Abstract
Plant-growth-promoting fungi (PGPF) play a central role in promoting sustainable agriculture by improving plant growth and resilience. The aim of this literature review is to survey the impacts of Trichoderma spp. and Penicillium spp. on various agricultural and horticultural plants. The information provided in this manuscript was obtained from randomized control experiments, review articles, and analytical studies and observations gathered from numerous literature sources such as Scopus, Google Scholar, PubMed, and Science Direct. The keywords used were the common and Latin names of various agricultural and horticultural species, fungal endophytes, plant-growth-promoting fungi, Trichoderma, Penicillium, microbial biostimulants, and biotic and abiotic stresses. Endophytic fungi refer to fungi that live in plant tissues throughout part of or the entire life cycle by starting a mutually beneficial symbiotic relationship with its host without any negative effects. They are also capable of producing compounds and a variety of bioactive components such as terpenoids, steroids, flavonoids, alkaloids, and phenolic components. Penicillium is extensively known for its production of secondary metabolites, its impact as a bioinoculant to help with crop productivity, and its effectiveness in sustainable crop production. The plant-growth-promotion effects of Trichoderma spp. are related to better absorption of mineral nutrients, enhanced morphological growth, better reproductive potential and yield, and better induction of disease resistance. Both Penicillium spp. and Trichoderma spp. are effective, affordable, safe, and eco-friendly biocontrol agents for various plant species, and they can be considered economically important microorganisms for both agricultural and horticultural sciences. The present review article aims to present the most up-to-date results and findings regarding the practical applications of two important types of PGPF, namely Penicillium spp., and Trichoderma spp., in agricultural and horticultural species, considering the mechanisms of actions of these species of fungi.
1. Introduction
Plant-growth-promoting microorganisms (PGPM) include fungi, bacteria, and other microorganisms that improve the performance of plants as well as increase their nutrient absorption under both abiotic and biotic stress conditions, with notable potential advantages for the growth and development of seedlings [1,2,3]. Plant-growth-promoting fungi (PGPF) are a group of non-pathogenic soil-borne filamentous fungi that exert positive effects on plants; these can enhance plant growth both indirectly and directly by producing phytohormones, fixing nitrogen, and inducing systemic resistance [4,5,6]. Trichoderma and Penicillium are the most effectual organisms among the numerous phosphate-solubilizing fungi that can dissolve insoluble phosphate and improve its uptake by plant roots, which are the main reasons for the plant’s appropriate growth and development [7,8,9]. In many research studies, it has been reported that a combination of biologically active compounds of plant or microbial origin, or their synthesized analogues, together with fungicides is a promising and sustainable approach to controlling crop diseases [10]. Fungi show significant metabolic properties because of their sophisticated genomic network, and they have notable importance because of their different roles in our world, such as in their applications in agriculture, industry, and medicine. Filamentous fungi of the genera Trichoderma have been widely studied because they can interact with and colonize plant roots via different processes that can increase plant growth through phytohormone synthesis, nutrient absorption, tolerance to abiotic stress, and the induction of systemic resistance as well as by acting as biological control factors. Their applications and their wonderful roles in environmentally friendly agricultural practices for different crops such as lettuce, soybean, wheat, corn, tomato, beans, etc., have been mentioned in previous research studies [11,12,13,14].
Some species of Penicillium produce different biologically active compounds [15,16,17], some of them with an extensive range of fungicidal action [18,19] and plant growth-stimulating activity [20]. Different species of Penicillium can significantly improve the dry and fresh weight of shoots as well as enhance the chlorophyll content [21]; for example, P. pinophilum can form arbuscular mycorrhizae, which can enhance the nitrogen content, photosynthesis rate, P content, and plant dry weight of strawberry plants [22]. The free-living soil fungi Trichoderma spp. are potential biological control agents of plant-parasitic nematodes [23], and they can control a wide range of economically notable plant pathogenic fungi, nematodes, bacteria, and viruses [24,25]. Trichoderma is the asexual stage of the filamentous Hypocrea genus belonging to the Ascomycota fungi division [26], and it is considered the most frequently isolated soil microorganism [27]. As PGPF, Penicillium spp. are known to grown in extreme conditions such as areas with high salt concentrations, and their application has increased in recent years due to their noticeable ability to increase salt-stress tolerance in different plant species as well as increase the levels of chemical components and improve the antioxidative system through the production of organic acids. Penicillium species can inhibit pathogens in the soil, and they are also highly effective in fixing potassium, dissolving phosphorus, and solubilizing soil-bound phosphate. In fact, Penicillium species can increase the absorption of phosphorus, improve soil conditions, help plants defend against diseases, and improve seed protection in plants facing pathogens. There has not been enough research on the effects of Penicillium fungi on plants, and more research is needed to study the effects of Penicillium fungi on soil phosphorus cycling and phosphorus uptake in plants as well as their importance in increasing plant growth. P. citrinum has been found to be a common endophytic fungus in cereal plants and has been isolated from various environmental conditions, ranging from permafrost sediments to agricultural fields and forest soils [28].
Trichoderma species such as T. harzianum, T. viride, T. atrovitide, T. hamatum, T. virens, and T. longibrachiatum are common in root and soil ecosystems, where they can establish root colonization, enhance crop productivity, growth and development, increase the use and uptake of nutrients, and improve resistance to abiotic stresses [29,30,31]. Chinnaperumal et al. [32] reported that Trichoderma viride can be considered an important inhibitor of the development of Helicoverpa armigera, with no negative impacts on earthworms. Panchalingam et al. [33] showed the positive effects of the combined use of a Streptomycetes consortium and Trichoderma as a potential biocontrol agent against the brown root rot pathogen Pyrrhoderma noxium (Corner) L.W. Zhou and Y.C. Dai in soil. The volatile organic compounds (VOCs) emitted by Trichoderma spp. have various effects against plant pathogenic fungi such as Sclerotinia nivalis, Cylindrocarpon destructans, Botrytis cinerea, Alternaria panax, Stagonosporopsis cucurbitacearum, Penicillium oxalicum, Ganoderma sp., Fusarium oxysporum, Sclerotium rolfsii, and Sclerotinia sclerotiorum [34,35]. Trichoderma is highly known as a biostimulant because of its main effects on plants, such as stimulating higher nutrient uptake efficiency, improving the rate of photosynthesis and metabolism, and as a potential biocontrol agent. Its different types of products can increase productivity and plant growth, and it can be applied as a good biocontrol agent that does not negatively influence other microorganisms in the soil. Trichoderma-based products have been identified as appropriate biological control agents for different plant pathogens, and they increase resistance to biotic stresses; thus, its different species have gained importance as microbial plant biostimulants in both agricultural and horticultural sciences. They can also mitigate the detrimental effects of abiotic stresses, improve nutritional quality and yield, and enhance plant growth. As important endophytic fungi, Trichoderma spp. can interact with other strains of the microbial community in the rhizosphere, which shows the importance of evaluating the ecological effects of different soil management and microbial-based biostimulants on the soil ecosystem.
Endophytes are known as microorganisms that can be found inside plant tissue without triggering any adverse impacts; rather, they reveal a positive influence on crop yield and plant growth, showing a high capacity for decreasing the need for fertilizer and stimulating plant biochemical components by providing osmoregulation, antioxidative defenses, and affecting nutrient uptake effectiveness during various biotic and abiotic stresses [36,37,38,39,40]. Different endophytic bacteria and fungi, including Bacillus, Actinomycetes, Trichoderma, Pseudomonas, and Epicoccum, were reported to elicit plant disease tolerance in cacao, potato, chili, cotton, and tomato [41]. It is also very important to choose suitable fungal strains, as some strains of the genera Penicillium are pathogens of crops and animals; P. allii is a pathogen of garlic, Penicillium spp. is a pathogen of pear, apple, and citrus fruit [42,43], and P. glabrum is a pathogen of garlic and onion [44].
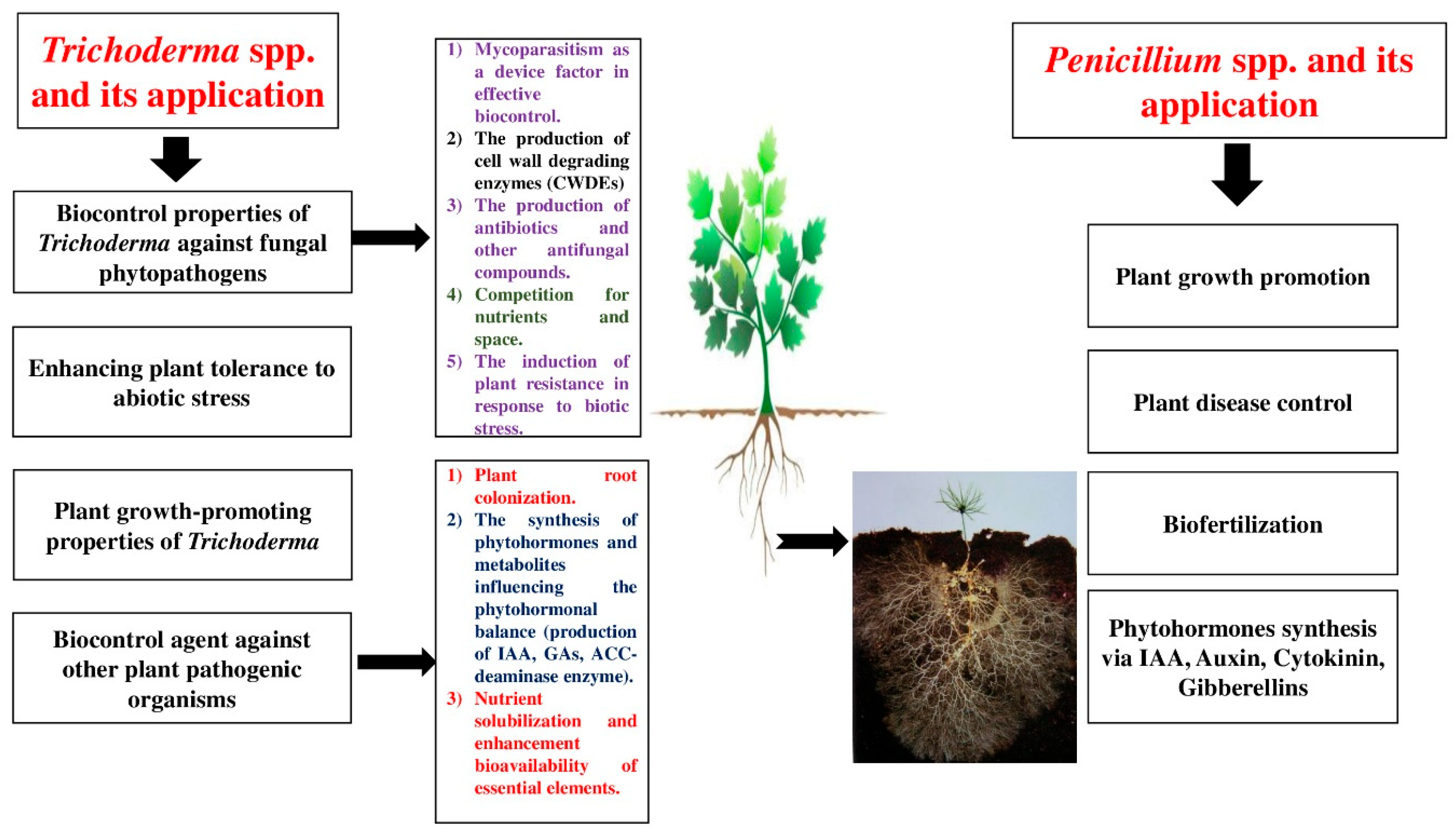
Plant–fungal interactions can be categorized as mutualistic, commensalistic, and pathogenic [45]. The most important mechanisms of PGPF involve the production of plant growth regulators such as abscisic acid, ethylene, gibberellins, cytokinins, and auxins; the production of organic acids and siderophores such as iron, zinc, potassium, phosphorus, and nitrogen; an increase in water uptake; the production of hydrolytic enzymes such as cellulases, pectinases, laccases, and xylanases; reductions in the amount of ethylene; relief from various abiotic stresses in harsh environments; and the induction of plant defense mechanisms against pathogens [46,47]. Trichoderma spp. and Penicillium spp. can be further studied to improve their ability as effective biocontrol agents; moreover, they show various antagonistic mechanisms against plant pathogens such as mycoparasitism, antibiosis, the promotion of plant growth, and competition for nutrients and space. They can be also considered important candidates for application in green technologies because of their great biostimulatory and biofertilization potential. The aim of this literature review is to survey the impacts of Trichoderma spp. and Penicillium spp. on various agricultural and horticultural plants, with the information provided in this manuscript obtained from randomized control experiments, review articles, and analytical studies and observations gathered from numerous literature sources such as Scopus, Google Scholar, PubMed, and Science Direct. The keywords used were the common and Latin names of various agricultural and horticultural species, fungal endophytes, plant-growth-promoting fungi, Trichoderma, Penicillium, microbial biostimulants, and biotic and abiotic stresses. The benefits and advantages of applications of both Penicillium spp. and Trichoderma spp. are presented in Figure 1.
Figure 1.
Studying the advantages of Penicillium spp. and Trichoderma spp.
2. Penicillium spp. as Plant-Growth-Promoting Fungi with Biocontrol Properties
Beneficial Penicillium spp. are used in the production of antibiotics, with the most famous advantage being the production of penicillin by P. chrysogenum, while some species produce griseofulvin, which is an antifungal drug. Other species are used in food and beverage production; in bioremediation, as some Penicillium species can break down different contaminants and pollutants in water and soil; and some species produce enzymes used in different industrial processes. They are also used to promote plant growth and stress tolerance, as certain species can help plants tolerate environmental stresses like drought, salinity, and heavy metals, and they can also increase the nutrient availability and promote root development in various plants. It is also reported that some species such as P. roqueforti and P. camemberti are important for cheese production. When using Penicillium for specific applications, it is essential to select the appropriate strain due to variations in their capabilities and properties.
Pencillium spp. are known to grow in extreme environments such as those with high salt concentrations, and some strains and species have been reported as PGPF with the ability to increase salt tolerance in different plant species [48,49,50,51,52]. Penicillium spp. colonize plant roots via several mechanisms, including endophytism, rhizosphere association, and mycorrhizal-like interactions [53,54,55]. They are often involved in a range of complex interactions with plants and use various strategies and develop distinct ways to mediate improvements in seed vigor, seed germination, plant growth, flowering, and productivity of host plants.
Root colonization by Penicillium spp. or its cell-free filtrate elicited an induced systemic resistance against infection by Pseudomonas syringae pv. Tomato DC3000 (Pst), leading to restricted pathogen growth and disease development [56]. Hossain et al. [56] showed that signal transduction leading to a GP16-2-mediated induced systemic resistance (ISR) requires responsiveness to JA and ET in an NPR1-dependent manner, while the cell-free filtrate (CF)-mediated ISR shows that salicylic acid (SA)-, JA-, ET-, and NPR1-dependent signaling is dispensable (at least individually). Moreover, root colonization by GP16-2 is not connected with a direct effect on the expression of known defense-related genes; however, it potentiates the activation of JA/ET-inducible basic chitinase (ChitB), which only becomes apparent after infection by Pst. But CF-mediated ISR is partly associated with the direct activation of marker genes responsive to both SA and JA/ET signaling pathways and partly associated with priming, leading to the activation of JA/ET-inducible ChitB and hevein-like protein (Hel) genes.
Species of Penicillium are ubiquitous fungi because of their ability to grow over a wide range of environments and conditions and their undemanding nutritional requirements; moreover, the genus is one of the largest groups of fungi. Its important species are P. chrysogenum, P. citreonigrum, P. citrinum, P. digitatum, and P. janthinellum [57,58,59]. Endophytic Penicillium species have different applications in (1) biotechnology—such as in the production of enzymes (such as lipase, inulinase, amylase, protease, cellulase, xylanase, β-glucosidase, etc. [60]), biotransformation [61,62], and the synthesis of nanoparticles like silver nanoparticles [63,64]; (2) agriculture—such as in phytoremediation, biocontrol, and insecticidal activities [65,66,67]; for example P. chrysogenum QEN-24S displayed potent activity against the pathogen Alternaria brassicae, which infects important crops such as oil seed rape, cabbage, and broccoli [67], while penicisteroids A and B, two new polyoxygenated steroids obtained from the culture extract of P. chrysogenum QEN-24S, revealed potent antifungal and cytotoxic activity in preliminary bioassays [65,66]; and (3) drug discovery—such as in immuno-suppressive [67], antifibrotic [68,69], neuroprotective [70], antidiabetic [70], anti-obesity [70,71,72], anti-inflammatory [22], antioxidative [22], anticancer, antiparasitic [73], antiviral [73,74], and antimicrobial [74] applications.
The species are also well-known for their unique potency in removing pollutants like heavy metals, such as mercury, lead, chromium, arsenic, and cadmium, from various ecosystems [75,76,77,78,79]. Sonderegger et al. [79] found that small, cysteine-rich, and cationic antifungal proteins (Aps) from filamentous ascomycetes, such as NFAP from Neosartorya fischeri and PAF from P. chrysogenum, are promising candidates for novel drug development. Garcia-Estrada et al. [80] showed that penicillin biosynthesis by P. chrysogenum is one of the best-characterized biological process from molecular, genetic, biochemical, and subcellular points of view, and omics studies have been conducted on this filamentous fungus over the last decade, which have contributed to gathering a deep knowledge about the molecular mechanisms underlying the improved productivity in industrial strains.
It has been reported that a halotolerant phenylacetate-degrading fungus P. chrysogenum CLONA2 strain can produce non-aromatic natural penicillin rather than benzylpenicillin, and it can be an appropriate option for aromatic compounds remediation in high-salinity regions [74]. Penicillium CLONA2, isolated from a salt mine at Algarve (Portugal), was identified as a variant of P. chrysogenum using ITS-5,85 rDNA and the D1/D2 domain of 28S rDNA sequences. Due to the ability of P. chrysogenum CLONA2 to degrade aromatic compounds, this strain can be considered an important organism for aromatic compound remediation in high-salinity environments. Garcia-Rico et al. [81] found that the heterotrimeric Gα protein, Pga1, of P. chrysogenum controls conidiation, vegetative growth, and secondary metabolite production. The secondary metabolites of P. chrysogenum are chrysogine, penicillins, sorrentanone, secalonic acids, and PR-toxins [82,83,84]. Guijarro et al. [85] reported that P. frequentans (Pf909) could decrease brown rot caused by Monilinia spp. in stone fruit, and that it could survive and establish actively in a broad range of climatic conditions. Arunthirumeni et al. [86] concluded that Penicillium spp. can produce secondary components that are effectual for the control of Spodoptera litura and Culex quinquefasciatus larvae. The combined application of a commercial azoxystrobin-based fungicide and the P. chrysogenum F-24-28 strain (DMP) induced to prolonged growth inhibition of F. culmorum, F. graminearum, and F. oxysporum at fungicide concentrations, which suggests that this approach can be used to control crop diseases [87]. Sikandar et al. [88] reported that P. chrysogenum Snef1216 has the potential to be used against Meloidogyne incognita, the main root-knot nematode, which is one of the most dangerous nematodes due to its high reproduction rate and extensive host range. P. citrinum has been found to be a common positive endophytic fungus of cereal plants like soybean and wheat [89]. Nguyen et al. [90] reported that P. citrinum can be used for the biological control of Plutella xylostella and Spodoptera litura in some crops. The non-volatile components produced by P. simplicissimum CEF-818, which is an endophyte from Gossypium hirsutum, were shown to strongly suppress the growth of the plant pathogen Verticillium dahliae isolate Vd080 [91,92,93]. It was shown that P. commune MC-9L could act as a biological agent against Sclerotinia sp. in fumigation assays [94,95]. It was also found that P. crustosum and P. chrysogenum isolated from Teucrium polium produced ammonia and IAA and showed a high phosphate solubilization capacity [96].
P. janthinellum can be used for the biological control of phytophthora root rot in azalea [97], while P. citrinum can noticeably enhance chemical metabolite production and yield [98]. Dry mycelium of P. chrysogenum is important in controlling fungal diseases in cotton [99], and it is also effective in increasing the germination index, germination rate, and seed germination rate in cucumber [100]. Moreover, P. chrysogenum can improve the flowering activity of jujube [101] and improve the yield and yield components of maize [102] and pearl millet [103]. Dry mycelium of P. chrysogenum can increase the resistance of seedlings against the root-knot nematode Meloidogyne javanica in tomato [104], and P. janthinellum LK5 can increase the resistance of plants against salinity stress [105] and increase metal phytoextraction while promoting crop physiological homeostasis [106]. The impacts of various species of Penicillium spp. on the yield and yield components of various plants are shown in Table 1.

Table 1.
The effects of different species of Penicillium spp. on the yield and yield components of plants.
3. Trichoderma spp. as Plant Growth-Promoting Fungi with Biocontrol Properties
The species of fungi belonging to Trichoderma genus are endophytic saprophytes, which can easily colonize the host root surface of plants, induce resistance, promote plant health as a biocontrol agent, and increase plant growth [125]. Trichoderma is highly known as a biostimulant because of its main impacts on plants, such as stimulating a higher nutrient uptake efficiency, improving the rate of metabolism and photosynthesis, and as a potential biocontrol agent. Its different types of products can increase productivity and the plant growth, and it can be applied as a good biocontrol agent without negatively influencing other microorganisms in the soil. The genus Trichoderma possesses mycoparasitic potential against pathogenic fungi [126]. It also has high potential for degrading pollutants [127,128]. At first, Trichoderma colonizes between living plant cells, resembling the early stages in an attack by soil-borne pathogens, and to start colonization, conidial germ tubes or hyphae growing toward and near the root in the rhizosphere almost adhere to the root epidermis [129,130].
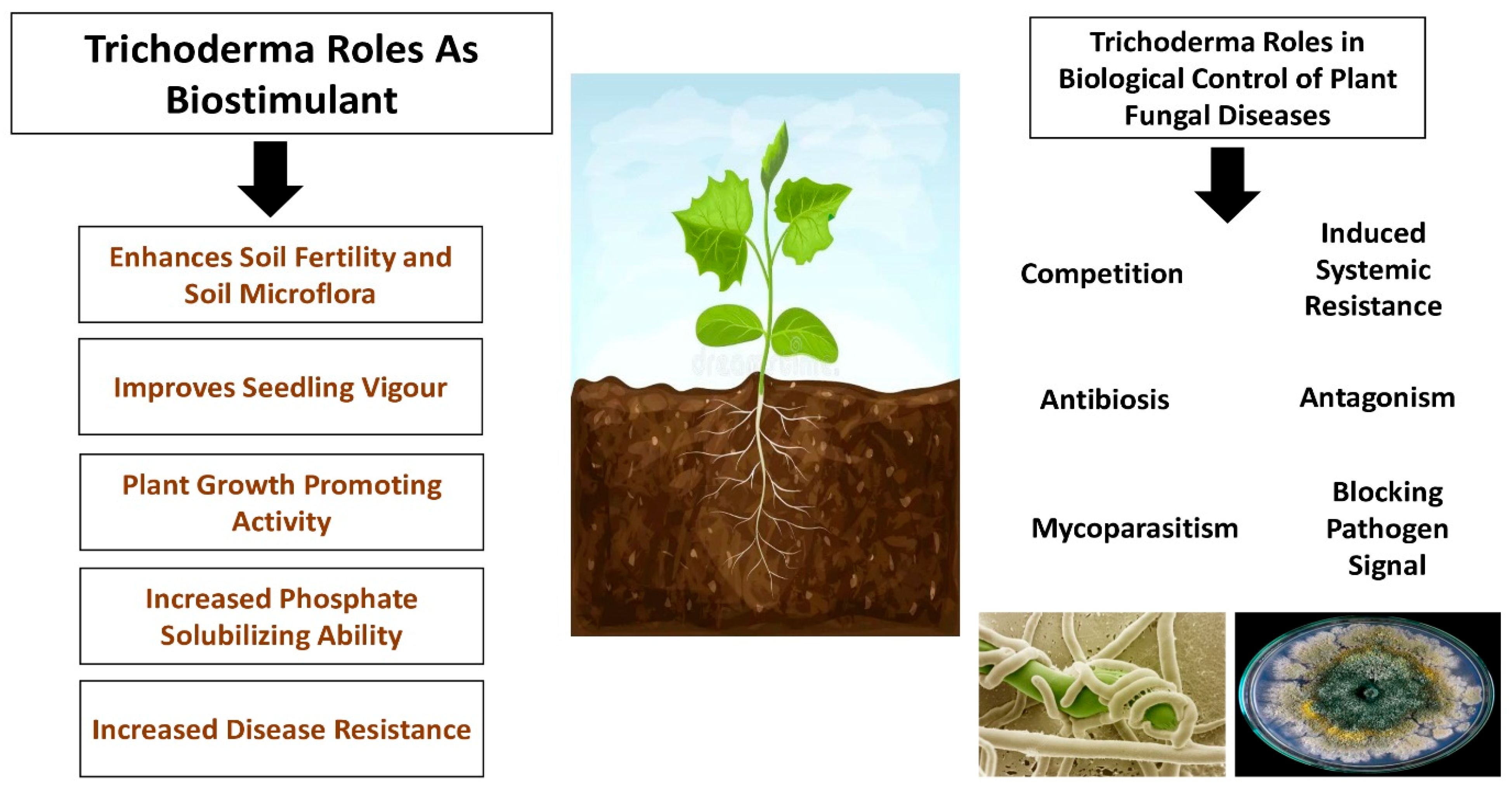
However, Pfordt et al. [131] reported that T. afroharzianum has been found as a pathogen causing ear rot disease on maize in Italy, France, and Germany, leading to massive infections on maize cobs, which has been confirmed even in other studies [132,133]. In 2020, T. afroharzianum was reported for the first time in Europe as an ear rot disease in maize [132]. The T. afroharzianum ear rot was characterized by a massive production of gray–green spores in the interkernel regions and on the outer surface of the husks, causing a significant reduction in the dry matter content and the premature germination of kernels [132,133]. The most important activities of Trichoderma spp. are shown in Figure 2.
Figure 2.
Plant biostimulatory actions of Trichoderma spp.
4. Journey of Trichoderma spp. Species
In the year 1794, the name Trichoderma was introduced, and in 1865, the sexual stage of T. viride (Hypocrea rufa) was reported. In 1932, the first evidence that T. lignorum (Hypocrea virens) has mycoparasitic and biocontrol abilities was presented, and in 1934, the first anti-microbial compound from Trichoderma spp., namely gliotoxin, was discovered. In 1957, the discovery of the effect of light on T. viride was reported. In general, when in darkness, Trichoderma grows indefinitely as mycelium, and a brief pulse of light applied to the actively growing zone of the mycelium leads to the formation of dark green mature conidia, forming a ring at what was the edge of the colony when light was applied; the first event induced by light is a fast, first-order, photochemical reaction that does not need the presence of molecular oxygen and is independent of temperature [134]. In 1972, the demonstration of biocontrol activity of T. harzianum against Sclerotium rolfsii in field conditions was reported, and in 1983, the cloning of the first Trichoderma species, which was T. reesei, was performed. In 1986, the report on the expression of growth promotion in the root was published, while in 1987, the successful transformation of T. reesei was reported, and in 1989, the first registration of a commercial formulation was reported. Evidence of the cloning of lectin-coated fibers by Trichoderma species was presented in 1992, and in 1998 and 1999, the identification of factors that induced the genes for mycoparasitism and the demonstration of Trichoderma internal colonization of plant roots was accomplished. In 2011, a genome comparison of three species of Trichoderma was conducted [135], and in 2022, five new Trichoderma species were reported [136,137,138,139].
5. Trichoderma spp. and Prospects for Application in Agriculture, Horticulture, and Organic Farming
Some scientific publications have shown that Trichoderma species can also significantly influence plant phytohormone networking and production by the secretion of enzymes that can change plant ethylene levels [140].
T. viride and T. longibrachiatum have shown effective larvicide impacts against dengue mosquitoes such as Aedes albopictus and Aedes aegypti [141]. T. asperellum also possesses both plant-growth-promoting and biocontrol activities [142]. It has also been reported that the combined application of T. koningii and T. virens with extracts of Chlorella vulgaris are significantly effective against severe disease-late wilt (LWD) of maize incited by Cephalosporium maydis Samra under both field and greenhouse conditions [143]. Previous studies confirmed the IAA production potential of Trichoderma species, including T. harzianum, T. asperellum, T. longibrachiatum, T. pinnatum, T. virens, T. asperelloides, T. guizhouense, T. atrobrunneum, T. simmonsii, and T. paratroviride [144]. It has been reported that one of the major and effective ways to control phytopathogenic microorganisms and to reduce the adverse impacts of heavy metals on plants is via the application of T. harzianum [145]. The presence of alfalfa seedlings with Trichoderma increased the available nutrient (K, P, and N) content in the soil and the alfalfa biomass [146], as Trichoderma acts as a nutrient mobilizer, improving the yield, yield components, and quality traits of the crops [147]. Trichoderma spp. have been found to degrade chlorpyrifos, benzimidazole fungicide, 2,2-dichlorovinyl dimethyl phosphate, and penthiopyrad [148]. T. hamatum can be considered for its plant-growth promotion [149] and various biocontrol activities, and it also possesses different beneficial activities such as antioxidative activity [150], antimicrobial properties [151], herbicidal activity [152], and insecticidal activity [153,154].
Trichoderma spp. has shown biocontrol activity by producing certain hydrolytic and antibiotic enzymes, such as β-1,3-glucanase and chitinase, which facilitate cell-wall degeneration and, ultimately, cause the death of pathogenic microorganisms [155]. Transcriptional parameters such as MYBs, WRKYs, and MYCs have shown important functions in priming as they act as regulatory nodes in the transcriptional network of systemic defense after stress recognition, and when it comes to long-lasting priming, Trichoderma spp. may have roles in the plants’ epigenetic regulation via DNA (hypo)methylation, histone replacement and modification, RNA-directed DNA methylation (RdDM), and DNA (hypo)methylation, and the inheritance of epigenetic markers can improve growth promotion and enhance the resistance of plants [156,157]. The most effectual biocontrol characteristics are pertain to T. harzianum, T. virens, T. pseudokoningii, T. koningii, T. asperellum, T. longibrachiatum, T. viride, and T. polysporum, which have a considerable effect on the development of plant diseases caused by F. culmorum, F. oxysporum, Verticillium dahliae, Gaeumannomyces graminis var. tritici, Pythium aphanidermatum, Sclerotium rolfsii, and R. solani in both field and greenhouse conditions [158].
Some researchers have reported that since many Trichoderma species are fungal parasitoids and symbiotic, they need to produce secondary metabolites and degradation enzymes to obtain nutrients from the host; thus, they have been developed as biocontrol factors for plant diseases [159]. T. hamatum has shown antibacterial effects on Xanthomonas campestris pv. armoraciae, and Xanthomonas euvesicatoria [160,161], Bacillus subtilis, Staphylococcus aureus, Pseudomonas aeruginosa, Serratia, and Acidovorax avenae [162], and Ralstonia solanecearum [160,163]. T. hamatum has also shown antifungal activity against Rhizoctonia solani in radish [164], Sclerotinia sclerotiorum in lettuce [164], Magnaporthe oryzae in Arabidopsis thaliana [165,166,167,168], Sclerotinia asari in Asarum rhizosphere [169], F. proliferatum, F. solani, and F. oxysporum in Aconitum carmichaelii Debx [170], Lasiodiplodia theobromae in Macadamia integrifolia [170], and Sclerotinia sclerotiorum in Arabidopsis thaliana [171]. Different species of Trichoderma, like. T. asperellum, T. atroviride, T. harzianum, T. viride, T. citrinoviride, T. koningii, and T. hamatum, can produce 6-pentyl-alpha-pyrone (6PP), which is a lactone with a coconut-like aroma that has special potency for increasing root hair development and root branching [172,173] and for improving plant health and growth [174]. The occurrence of Trichoderma spp. species in different environments is shown in Table 2.

Table 2.
Occurrence of Trichoderma spp. in different environments.
Compounds synthesized by Trichoderma spp. that are involved in plant interactions are IAA in T. virens, GA3 in Trichoderma spp., ABA in T. virens and T. atroviride, ethylene in T. atroviride, jasmonic acid (JA) in T. asperellum, and salicylic acid (SA) in T. atroviride [185]. A novel type II hydrophobin secreted by the biocontrol strain MK1 of T. longibrachiatum was characterized and isolated, and the corresponding gene (Hytlo1) was shown to have multiple functions in the Trichoderma–plant pathogen three-way interactions, while the purified protein showed direct antifungal and plant-growth promotion (PGP) activities as well as microbe-associated molecular patterns [185]. In this experiment, leaf infiltration with hydrophobin systemically enhanced the resistance to pathogens and activated defense-related responses involving phytoalexin, oxylipin, superoxide dismutase, reactive oxygen species, and pathogenesis-related formation or activity; moreover, hydrophobin stimulated root formation and growth, and a targeted knock-out of Hytlo1 significantly decreased both the antagonistic and PGP effects of the wild-type strain [185]. Polyketides in T. virens and Trichoderma sp. SCSIO41004; terpenes in T. virens, T. harzianum P1-4, T. citrinoviride, and T. harzianum R5; VOCs in T. atroviride and T. arundinaceum; and hydrophobin in T. virens, T. atroviride, and T. asperellum have different functions, such as in plant-growth promotion, and facilitate the plant–microbe interactions in the rhizosphere [186,187]. Claudia et al. [186] showed that the product of the TvCyt2 gene from T. virens encoded a new protein homologous to cytochrome p450, which is down-regulated at the beginning of the Trichoderma–Arabidopsis interaction, and Arabidopsis plants co-cultivated with the OETvCyt2 strains showed a stronger induction of systemic acquired resistance than plants co-cultivated with the WT strain, as well as increases in biomass and fitness, which show that the TvCyt2 gene is involved in secondary metabolite biosynthesis, and this can increase the antagonistic activity toward phytopathogenic fungi and the capacity to promote plant growth [186].
Some of the species of Trichoderma are active in regulating different genes in plants, such as T. asperelloides, which can up-regulate the MDAR gene in Arabidopsis and cucumber, which can increase osmo-protection and oxidative stress [188]; T. atroviride and T. virens, which can up-regulate AtERD14 in Arabidopsis and mitigate cold stress effects; T. parareesei, which can up-regulate PYL4, ERF1, ACCO1, and NCED3 in rapeseed, thus improving tolerance to salinity and drought; T. longibrachiatum in wheat, which can up-regulate CAT, POD, and SOD and increase the resistant of plants to salinity [189]; T. harzianum in tomato, which can improve the tolerance of seedlings to cold via P5CS and TAS14 [190]; T. harzianum, which regulates GST1 and Lox in potato, which can improve plant resistance to diseases [191]; and T. asperellum, which can up-regulate PdPapARF1 in poplar, which can promote growth and defense responses [192].
Some parameters that may encourage the market for Trichoderma-based biofungicides are that they should be broad-spectrum in action, show consistent field performance, have an extensive lifespan, be a cost-effective product, have easy accessibility and improved delivery systems, and there should be social awareness of their benefits among farmers [193]. Trichoderma spp. as potential biocontrol agents (BCAs) can be used for the effective management of different soil, foliar, and post-harvest plant pathogens, and they have gained more attention in recent years.
Some of the identified genes from Trichoderma species have different roles during the biocontrol interaction with phytopathogens, such as Tvsp1 from T. virens, which can protect cotton seedlings against R. solani [194]; Tag3 from T. asperellum, which is responsible for glucanase production for cell-wall degradation [195]; TgaA and TgaB from T. virens, which has shown biocontrol effectiveness for the management of R. solani and Sclerotium rolfsii [196]; ThPG1 from T. harzianum, which is needed for beneficial interactions between T. harzianum and the host [197]; ThPRT2 from T. harzianum, which has shown mycoparasitism activity against Botrytis cinerea [198]; tri5 of T. brevicompactum IBT40841, which has antifungal activity and has been used in the production of trichodermin against fungi causing infections in the human body [199], TvGST of T. virens, which provides enhanced tolerance against cadmium stress [200]; TrCCD1 of T. reesei, which can facilitate pigment production and hyphal growth [201]; egl1 of T. longibrachiatum, which shows antagonistic activity against Pythium ultimum [202]; qid74 of T. harzianum, which has roles in plant biofertilization and the root architecture [203]; tac1 of T. virens IMI 304061, which shows mycoparasitism against S. rolfsii and R. solani [204]; TrCCD1 of T. reesei, which can promote conidia formation and elongation of fungal hyphae; XI 1 of Trichoderma strain Y, which is helpful in hemicellulose breakdown [205]; tvhydii1 of T. reesei, which is important in mycoparasitism and plant–fungus interactions [206], gpr1 of T. atroviride, which is needed for the stability of cell walls and hyphal growth [207]; ipa-1 of T. virens, which has a role in antibiosis against R. solani [208]; TasXyn24.2 and TasXyn29.4 of T. asperellum, which can induce resistance and increase growth in seedlings [208], and agl1 of T. atroviride, which can be used for the biological control of plant pathogens. It has been reported that the application of grains (200 g + sugar (1%) + T. harzianum) showed 12.96% effectiveness in the management of chili wilt disease [209], and the application of vermicompost fortified with Trichoderma induced a reduction of 10.01% in the incidence of wilt in chili [209]. A mixture of ground grain + sugar solution (1%) + T. harzianum and the combined application of decomposed cow dung + a Trichoderma formulation has positive impacts on maize seeds [210], while the application of partially crushed grain + sugar (1%) solution + distilled water and the combined application of beech, fir, and chestnut + conidial suspensions of T. atroviride + distilled water + soy flour could increase the yield [211].
Guzman-Guzman et al. [212] reported that the biocontrol mechanisms of Trichoderma spp. against potential pathogens include antibiosis, parasitism, secondary metabolite production, plant defense system induction, and competition. Different Trichoderma species used in agriculture have different biocontrol characteristics; for example, T. atroviride, T. viride, T. longibrachiatum, T. virens, T. asperellum, and T. harzianum have demonstated parasitism, competition, plant defense induction, priming, secondary metabolite production, and antibiosis [213,214]. Chan et al. [215] reported that the T. harzianum strain CE92 can be used as a potential biocontrol agent for pathogenic wood rot fungal species such as Rigidoporus microporus, Phellinus noxius, and Fulvifomes siamensis. De Oliveira et al. [216] reported that T. harzianum is recommended for the biological control of the root-lesion nematode Pratylenchus brachyurus, which is an important nematode in sugarcane, sunflower, potato, millet, cotton, corn, and soybean, while Wu et al. [217] found that octahydronaphthalene derivatives from an endophytic fungus Trichoderma sp. can be used for managing Botrytis cinerea. Tamandegani et al. [218] also found that T. asperellum (Iran 3062C), which was isolated from potato fields in Hamedan, Iran, showed a strong ability to induce systemic resistance against cucumber mosaic virus (CMV) that involved the jasmonic acid (JA)/ethylene (ET)/salicylic acid (SA) signaling pathways. Biocontrol mechanisms applied by the Trichoderma genus against fungal pathogens include the use of plant-resistance elicitors, the stimulation of antioxidative enzymes, and phytoalexin production by the plants via Trichoderma and its metabolites, as well as mechanisms like mycoparasitism, utilization of the activity of proteases, glucanases, and chitinases, competition for space and nutrients, and the production of antifungal and antibiotic components such as polyketides, peptaibols, anthraquinones, pyrones, and terpenoids.
T. virens 6PS-2 was effective in controlling apple replant disease and in improving the growth and fruit quality of apple [219], while T. virens could improve defense pathways [220] and enhance plant growth promotion in Arabidopsis when applied as an important biocontrol agent [220]. T. harzianum could increase the tolerance of avocado seedlings to P. cinnamomic [221], and it can be used as biological control agent to control post-harvest pathogens such as Diaporthe sp. and Phomopsis perseae in avocado plants [222]. T. harzianum also can significantly control chickpea Fusarium wilt [223,224]. It is also effective in the control of root-knot nematode disease in cowpea [225]. Gupta et al. [226] reported that T. harzianum can induce a higher root canopy and a better root biomass and final yield in finger millet. Its application was also effective against dry root rot in mung bean [227,228]. Pandey et al. [229] found that T. harzianum could enhance the activity of antioxidative enzymes and decrease lipid peroxidation during drought stress in rice plants. The application of an organic additive with T. harzianum could reduce the levels of Sclerotium rolfsii, which is the main pathogen for potato [230]. Limdolthamand et al. [231] could decrease the infection of northern corn leaf blight in sweet corn and improve the growth of sweet corn plants. T. asperellum could improve the resistance of plants against Colletotrichum graminicola by increasing lignification in plants and by enhancing the activity of antioxidative enzymes [232], and it was effective in controlling Fusarium wilt in stevia plants [233]. Changes in the yield and yield components of various horticultural and agricultural plants following the application of different species of Trichoderma spp. are presented in Table 3.

Table 3.
The effects of different species of Trichoderma spp. on the yield and yield components of some agricultural and horticultural plants.
6. Conclusions
Endophytic fungi are very important in agricultural production because of their synergistic association with plants and their strong ability to stimulate plant growth because of their synthesis of phytohormones in response to biotic and abiotic stressors; these make them unique options as biofertilizers and biostimulants as well as as biocontrol factors against different types of diseases. Different species of Penicillium can significantly improve the fresh and dry weight of shoots, as well as stimulate the chlorophyll content. They can also improve the photosynthesis rate and the P and N content. P. janthinellum can be used to control phytophthora root rot in azalea, while P. citrinum YW322 can be applied to control ginseng root rot caused by Fusarium, and P. citrinum BTF08 can be applied against the pathogenic F. oxysporum s. sp. cubense race 4 in banana. P. chrysogenum can be used to control fungal diseases in cotton, and it is also appropriate for controlling root-knot nematodes in cucumber. P. citrinum can enhance the yield and chemical metabolites in choy sum, while P. chrysogenum strain 34-P can increase the fresh and dry biomass of maize, and P. chrysogenum (PenC-JSB9) is suitable for increasing the root and shoot length of pearl millet. Species of Trichoderma spp. are known as growth enhancers, stimulators of resistance in plants, biopesticides, and biofertilizers. T. harzianum is effective against dry root rot in mung bean, and T. asperelloides PSU-P1 can enhance the defense response against stem blight diseases in muskmelon. T. viride Tv-1511 can boost the concentrations of menthone, menthol, and pulegone in peppermint plants, while T. asperellum IIPRTH-31 and T. afroharzianum IIPRTH-33 can be used to control Fusarium wilt in pigeon pea plants. Among the different species, T. viride, T. longibrachiatum, T. atroviride, T. hamatum, and T. koningii have been reported to show nematicidal characteristics. Trichoderma spp. produces various secondary compounds with numerous beneficial impacts, such as xylanases, epipoly-thiodioxopiperazines, pyrones, peptaibols, volatile terpenes, nonvolatile terpenes, polyketides, siderophores, and cerato-plantanins. The most important impacts of Trichoderma inoculation are the destruction of pathogenic organisms and plant growth promotion. Their strains are considered to be among the most useful fungi in agriculture, horticulture, industrial enzyme production, and bioremediation. In conclusion, the application of Penicillium spp. and Trichoderma spp. is a promising method and an environmentally friendly practice for improving the growth and final yield of plants, and novel practices involving such biostimulants should be integrated in new farming systems. Areas that should be considered in future research include studying their mechanisms of action, assessing their long-term effects while considering sustainability and the effects of climate change, how to integrate both Trichoderma spp. and Penicillium spp. into sustainable agriculture, and investigating the effects of their combined application.
Author Contributions
W.S., writing—original draft preparation; M.H.S., writing—original draft preparation and editing; L.G., writing—original draft preparation. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Key R&D Program of China (Grant no. 2024YFA0918200). This work was also supported by the Scientific Research Project of Kweichow Moutai Liquor Co., Ltd. (MTGF2023050).
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare that this study received funding from Kweichow Moutai Liquor Co., Ltd. The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication.
References
- Shahrajabian, M.H.; Chaski, C.; Polyzos, N.; Tzortzakis, N.; Petropoulos, S.A. Sustainable agriculture systems in vegetable production using chitin and chitosan as plant biostimulants. Biomolecules 2021, 11, 819. [Google Scholar] [CrossRef] [PubMed]
- Shahrajabian, M.H.; Chaski, C.; Polyzos, N.; Petropoulos, S.A. Biostimulants application: A low input cropping management tool for sustainable farming of vegetables. Biomolecules 2021, 11, 698. [Google Scholar] [CrossRef] [PubMed]
- Shahrajabian, M.H.; Sun, W.; Cheng, Q. Using bacteria and fungi as plant biostimulants for sustainable agricultural production systems. Recent Pat. Biotechnol. 2022, 17, 206–244. [Google Scholar] [CrossRef]
- Sun, W.; Shahrajabian, M.H. The effectiveness of rhizobium bacteria on soil fertility and sustainable crop production under cover and catch crops management and green manuring. Not. Bot. Horti Agrobot. Cluj Napoca 2022, 50, 12560. [Google Scholar] [CrossRef]
- Chabbi, N.; Chafiki, S.; Telmoudi, M.; Labbassi, S.; Bouharroud, R.; Tahiri, A.; Mentag, R.; El-Amri, M.; Bendiab, K.; Hsissou, D.; et al. Plant growth promoting rhizobacteria improve seeds germination and growth of Argania spinosa. Plants 2024, 13, 2025. [Google Scholar] [CrossRef] [PubMed]
- Raish, S.M.; Sourani, O.M.; Abu-Elsaoud, A.M. Plant growth-promoting microorganisms as biocontrol agents: Mechanisms, challenges, and future prospects. Appl. Microbiol. 2025, 5, 44. [Google Scholar] [CrossRef]
- Embacher, J.; Seehauser, M.; Kappacher, C.; Stuppner, S.; Zeilinger, S.; Kirchmair, M.; Neuhauser, S. Serpula lacrymans reacts with a general, unspecialized chemical response during interaction with mycoparasitic Trichoderma spp. and bacteria. Fungal Ecol. 2023, 63, 101230. [Google Scholar] [CrossRef]
- Shahrajabian, M.H.; Cheng, Q.; Sun, W. The effects of amino acids, phenols and protein hydrolysates as biostimulants on sustainable crop production and alleviated stress. Recent Pat. Biotechnol. 2022, 16, 319–328. [Google Scholar] [CrossRef]
- Shahrajabian, M.H.; Petropoulos, S.A.; Sun, W. Survey of the influences of microbial biostimulants on horticultural crops: Case studies and successful paradigms. Horticulturae 2023, 9, 193. [Google Scholar] [CrossRef]
- Kim, K.; Lee, Y.; Ha, A.; Kim, J.-I.; Park, A.R.; Yu, N.H.; Son, H.; Choi, G.J.; Park, H.W.; Lee, C.W.; et al. Chemosensitization of Fusarium graminearum to chemical fungicides using cyclic lipopeptides produced by Bacillus amyloliquefaciens strain JCK-12. Front. Plant Sci. 2017, 8, 2010. [Google Scholar] [CrossRef]
- De Souza, R.R.; Moraes, M.P.; Paraginski, J.A.; Moreira, T.F.; Bittencourt, K.C.; Toebe, M. Effects of Trichoderma asperellum on germination indexes and seedling parameters of lettuce cultivars. Curr. Microbiol. 2022, 79, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.; Thakur, P.; Rathore, U.S.; Kumar, S.; Mishra, R.K.; Amaresan, N.; Pandey, S.; Mishra, M. Plant beneficial effects of Trichoderma spp. suppressin Fusarium wilt and enhancing growth in tomato. Vegetos 2022, 35, 188–195. [Google Scholar] [CrossRef]
- Sun, W.; Shahrajabian, M.H. The application of arbuscular mycorhhizal fungi as microbial biostimulant, sustainable approaches in modern agriculture. Plants 2023, 12, 3101. [Google Scholar] [CrossRef]
- Sun, W.; Shahrajabian, M.H.; Petrpoulos, S.A.; Shahrajabian, N. Developing sustainable agriculture systems in medicinal and aromatic plant production by using chitosan and chitin-based biostimulants. Plants 2023, 12, 2469. [Google Scholar] [CrossRef]
- Ma, H.-G.; Liu, Q.; Zhu, G.-L.; Liu, H.-S.; Zhu, W.-M. Marine natural products sourced from marine-derived Penicillium fungi. J. Asian Nat. Prod. Res. 2016, 18, 92–115. [Google Scholar] [CrossRef]
- Nobre, C.; Nascimento, A.K.C.D.; Silva, S.P.; Coelho, E.; Coimbra, M.A.; Cavalcanti, M.T.H.; Teixeira, J.A.; Porto, A.L.F. Process development for the production of prebiotic fructo-oligosaccarides by Penicillium citreonigrum. Bioresour. Technol. 2019, 282, 464–474. [Google Scholar] [CrossRef]
- Huang, J.-N.; Zou, Q.; Chen, J.; Xu, S.-H.; Luo, D.; Zhang, F.-G.; Lu, Y.-Y. Phenols and diketopiperazines isolated from Antarctic-derived fungi, Penicillium citreonigrum SP-6. Phytchem. Lett. 2018, 27, 114–118. [Google Scholar] [CrossRef]
- Huber, A.; Galgoczy, L.; Varadi, G.; Holzknecht, J.; Kakar, A.; Malanovic, N.; Leber, R.; Kochf, J.; Kellerf, M.A.; Battag, G.; et al. Two small, cysteine-rich and cationic antifungal proteins from Penicillium chrysogenum: A comparative study of PAF and PAFB. Biochim. Biophys. Acta-Biomembr. 2020, 1862, 183246. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Liu, X.; Zhao, H.; Ni, Y.; Lian, Q.; Qian, H.; He, B.; Liu, H.; Ma, Q. Biological control of Fusarium wilt of sesame by Penicillium bilaiae 47M-1. Biol. Control 2021, 158, 104601. [Google Scholar] [CrossRef]
- De Cal, A.; Sztejnberg, A.; Sabuquillo, P.; Melgarejo, P. Management Fusarium wilt on melon and watermelon by Penicillium oxalicum. Biol. Control 2009, 51, 480–486. [Google Scholar] [CrossRef]
- Waqas, M.; Khan, A.L.; Kamran, M.; Hamayun, M.; Kang, S.M.; Kim, Y.H.; Lee, I.J. Endophytic fungi produce gibberellins and indoleacetic acid and promotes host-plant growth during stress. Molecules 2012, 17, 10754–10773. [Google Scholar] [CrossRef]
- Fan, Y.; Luan, Y.; An, L.; Yu, K. Arbuscular mycorrhizae formed by Penicillium pinophilum improve the growth nutrient uptake, and photosynthesis of strawberry with two inoculum-types. Biotechnol. Lett. 2008, 30, 1489–1494. [Google Scholar] [CrossRef] [PubMed]
- Maccari, G.; Deodato, D.; Fiorucci, D.; Orofino, F.; Truglio, G.I.; Pasero, C.; Martini, R.; Luca, F.D.; Docquier, J.-D.; Botta, M. Design and synthesis of a novel inhibitor of T. viride chitinase through an in silico target fishing protocol. Bioorg. Med. Chem. Lett. 2017, 27, 3332–3336. [Google Scholar] [CrossRef] [PubMed]
- Jia, M.; Chen, J.; Liu, X.; Xie, M.; Nie, S.; Chen, Y.; Xie, J.; Yu, Q. Structural characteristics and functional properties of soluble dietary fiber from defatted rice bran obtained through Trichoderma viride fermentation. Food Hydrocoll. 2019, 94, 468–474. [Google Scholar] [CrossRef]
- Moya, P.; Barrera, V.; Cipollone, J.; Bedoya, C.; Kohan, L.; Tolerdo, A.; Sisterna, M. New isolates of Trichoderma spp. as biocontrol and plant growth-promoting agents in the pathosystem Pyrenophora teres-barley in Argentina. Biol. Control 2020, 141, 104152. [Google Scholar] [CrossRef]
- Tomah, A.A.; Zhang, Z.; Alamer, I.S.A.; Khattak, A.A.; Ahmed, T.; Hu, M.; Wang, D.; Xu, L.; Li, B.; Wang, Y. The potential of Trichoderma-mediated nanotechnology application in sustainable development scopes. Nanomaterials 2023, 13, 2475. [Google Scholar] [CrossRef]
- Druzhinia, I.S.; Seidl-Seiboth, V.; Herrera-Estrella, A.; Horwitz, B.A.; Kenerley, C.M.; Monte, E.; Mukherjee, P.K.; Zeilinger, S.; Grigoriev, I.V.; Kubicek, C.P. Trichoderma: The genomics of opportunistic success. Nat. Rev. Microbiol. 2011, 9, 749–759. [Google Scholar] [CrossRef]
- Wakiyama, M.; Tanaka, H.; Yoshihara, K.; Hayashi, S.; Ohta, K. Purification and properties of family-10 endo-1, 4-β-Xylanase from Penicillium citrinum and structural organization of encoding gene. J. Biosci. Bioeng. 2008, 105, 367–374. [Google Scholar] [CrossRef]
- Lipsa, R.; Tudorachi, N.; Darie-Nita, R.N.; Oprica, L.; Vasile, C.; Chiriac, A. Biodegradation of poly(lactic acid) and some of its based systems with Trichoderma viride. Int. J. Biol. Macromol. 2016, 88, 515–526. [Google Scholar] [CrossRef]
- Peil, S.; Beckers, S.J.; Fischer, J.; Wurm, F.R. Biodegradable, lignin-based encapsulation enables delivery of Trichoderma reesei with programmed enzymatic release against grapevine trunk diseases. Mat. Today Bio 2020, 7, 100061. [Google Scholar] [CrossRef]
- Sun, F.-S.; Yu, G.-H.; Ning, J.-Y.; Zhu, X.-D.; Goodman, B.A.; Wu, J. Biological removal of cadmiuum from biogas residues during vermicomposting, and the effect of earthworm hydrolysates on Trichoderma guizhouense sporulation. Biosour. Technol. 2020, 312, 123635. [Google Scholar] [CrossRef] [PubMed]
- Chinnapermal, K.; Govindasamy, B.; Paramasivam, D.; Dilipkumar, A.; Dhayalan, A.; Vadivel, A.; Sengodan, K.; Pachiappan, P. Bio-pesticidal effects of Trichoderma viride formulated titanium dioxide nanoparticle and their physiological and biochemical changes on Helicoverpa armigera (Hub.). Pest Biochem. Physiol. 2018, 149, 26–36. [Google Scholar] [CrossRef]
- Panchalingam, H.; Ashfield-Crook, N.; Naik, V.; Frenken, R.; Foster, K.; Tomlin, R.; Shapcott, A.; Kurtboke, D.I. Testing the biocontrol ability of a Trichoderma-streptomycetes consortium against Pyrrhoderma noxium (Corner) L.W. Zhou and Y.C. Dai in soil. J. Fungi 2023, 9, 67. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Pollastri, S.; Ruocco, M.; Monti, M.M.; Loreto, F. Volatile organic compounds in the interaction between plants and beneficial microorganisms. J. Plant Interact. 2022, 17, 840–852. [Google Scholar] [CrossRef]
- Tyskiewicz, R.; Nowak, A.; Ozimek, E.; Jaroszuk-Scisel, J. Trichoderma: The current status of its application in agriculture for the biocontrol of fungal phytopathogens and stimulation of plant growth. Int. J. Mol. Sci. 2022, 23, 2329. [Google Scholar] [CrossRef]
- Arnold, A.E.; Mejia, L.C.; Kyllo, D.; Rojas, E.; Maynard, Z.; Robbins, N.; Herre, E.A. Fungal endophytes limit pathogen damage in a tropical tree. Proc. Natl. Acad. Sci. USA 2003, 100, 15649–15654. [Google Scholar] [CrossRef]
- Redman, R.S.; Dunigan, D.D.; Rodriguez, R.J. Fungal symbiosis: From mutualism to parasitism, who controls the outcome, host or invader? New Phytol. 2001, 151, 705–716. [Google Scholar] [CrossRef]
- Redman, R.S.; Sheehan, K.B.; Stout, R.G.; Rodriguez, R.J.; Henson, J.M. Thermotolerance conferred to plant host and fungal endophyte during mutualistic symbiosis. Science 2002, 298, 1581. [Google Scholar] [CrossRef]
- Waller, F.; Achatz, B.; Baltruschat, H.; Fodor, J.; Becker, K.; Fischer, M.; Heier, T.; Huckelhoven, R.; Neumann, C.; Von Wettstein, D.; et al. The endophytic fungus Piriformis indica reprograms barley to salt-stress tolerance, disease resistance, and higher yield. Proc. Natl. Acad. Sci. USA 2005, 102, 13386–13391. [Google Scholar] [CrossRef]
- Marquez, L.M.; Redman, R.S.; Rodriguez, R.J.; Roossinck, M.J. A virus in a fungus in a plant-three way symbioses required for thermal tolerance. Science 2007, 315, 513–515. [Google Scholar] [CrossRef]
- Allaga, H.; Zhumakayev, A.; Buchner, R.; Kocsube, S.; Szucz, A.; Vagvolgyi, C.; Kredics, L.; Hatvani, L. Members of the Trichoderma harzianum species complex with mushroom pathogenic potential. Agronomy 2021, 11, 2434. [Google Scholar] [CrossRef]
- Louw, J.P.; Korsten, L. Pathogenicity and host susceptibility of Penicillium spp. on citrus. Plant Dis. 2015, 99, 21–30. [Google Scholar] [CrossRef]
- Salinas, M.C.; Cavagnaro, P.F. In vivo and in vitro screening for resistance against Penicillium allii in garlic accessions. Eur. J. Plant Pathol. 2020, 156, 173–187. [Google Scholar] [CrossRef]
- Dugan, F.M.; Lupien, S.L.; Vahling-Armstrong, C.; Chastagner, G.A.; Schroeder, B.K. Host range of Penicillium species causing blue mold of bulbs crops in Washington state and Idaho. Crop Prot. 2017, 96, 265–272. [Google Scholar] [CrossRef]
- Zhu, G.; Wang, X.; Chen, T.; Wang, S.; Chen, X.; Song, Z.; Shi, X.; Laborda, P. First report of Aspergillus flavus causing fruit rot on kiwifruit in China. Plant Dis. 2022, 106, 1990. [Google Scholar] [CrossRef]
- Poveda, J.; Eugui, D.; Abril-Urias, P.; Velasco, P. Endophytic fungi as direct plant growth promoters for sustainable agricultural production. Symbiosis 2021, 85, 1–19. [Google Scholar] [CrossRef]
- Mathis, K.A.; Bronstein, J.L. Our current understanding of commensalism. Annu. Rev. Ecol. Syst. 2020, 14, 167–189. [Google Scholar] [CrossRef]
- Duan, B.; Gao, Z.; Reymick, O.O.; Ouyang, Q.; Chen, Y.; Long, C.; Yang, B.; Tao, N. Cinnamaldehyde promotes the defense response in postharvest citrus fruit inoculated with Penicillium digitatum and Geotrichum citri-aurantii. Pest Biochem. Physiol. 2021, 179, 104976. [Google Scholar] [CrossRef]
- Liu, K.; Wang, L.; Jiang, B.; An, J.; Nian, B.; Wang, D.; Chen, L.; Ma, Y.; Wang, X.; Fan, J.; et al. Effect of inoculation with Penicillium chrysogenum on chemical components and fungal communities in fermentation of Pu-erh tea. Food Res. Int. 2021, 150, 110748. [Google Scholar] [CrossRef]
- Fierro, F.; Vaca, I.; Castillo, N.I.; Garcia-Rico, R.O.; Chavez, R. Penicillium chrysogenum, a vintage model with a cutting-edge profile in biotechnology. Microorganisms 2022, 10, 573. [Google Scholar] [CrossRef]
- Qi, B.; Jia, F.; Luo, Y.; Ding, N.; Li, S.; Shi, F.; Hai, Y.; Wang, L.; Zhu, Z.-X.; Liu, X.; et al. Two new diterpenoids from penicillium chrysogenum MT-12, and endophytic fungus isolated from Huperzia serrata. Nat. Prod. Res. 2022, 36, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Newaz, A.W.; Yong, K.; Yi, W.; Wu, B.; Zhang, Z. Antimicrobial metabolites from the Indonesian mangrove sediment-derived fungus Penicillium chrysogenum sp. ZZ1151. Nat. Prod. Res. 2023, 37, 1702–1708. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhang, J.; Liu, Y.; Feng, W. Biodegradation of hydrocarbons by Purpureocillium lilacinum and Penicillium chrysogenum from heavy oil sludge and their potential for bioremediation of contaminated soil. Int. Biodeter. Biodegrade 2023, 178, 105566. [Google Scholar] [CrossRef]
- Leitao, A.L. Potential of Penicillium species in the bioremediation field. Int. J. Environ. Res. Public Health 2009, 6, 1393–1417. [Google Scholar] [CrossRef]
- Visagie, C.M.; Houbraken, J.; Frisvad, J.C.; Hong, S.-B.; Klaassen, C.H.W.; Perrone, G.; Seifert, K.A.; Varga, J.; Yaguchi, T.; Samson, R.A. Identification and nomenclature of the genus Penicillium. Stud. Mycol. 2014, 78, 343–371. [Google Scholar] [CrossRef]
- Hossain, M.M.; Sultana, F.; Kubota, M.; Hyakumachi, M. Differential inducible defense mechanisms against bacterial speck pathogen in Arabidopsis thaliana by plant-growth-promoting-fungus Penicillium sp. GP16-2 and its cell free filtrate. Plant Soil 2008, 304, 227–239. [Google Scholar] [CrossRef]
- Ali, S.; Khan, A.L.; Ali, L.; Rizvi, T.S.; Khan, S.A.; Hussain, J.; Hamayun, M.; Al-Harrasi, A. Enzyme inhibitory metabolites from endophytic Penicillium citrinum isolated from Boswellia sacra. Arch. Microbiol. 2017, 199, 691–700. [Google Scholar] [CrossRef]
- Ali, T.; Inagaki, M.; Chai, H.; Wieboldt, T.; Rapplye, C.; Rakotondraibe, L.H. Halogenated compounds from directed fermentation of Penicillium concentricum, an endophytic fungus of the Liverwort Trichocolea tomentella. J. Nat. Prod. 2017, 80, 1397–1403. [Google Scholar] [CrossRef] [PubMed]
- Attia, E.Z.; Khalifa, B.A.; Shaban, G.M.; Abdelraheem, W.M.; Mustafa, M.; Abdelmohsen, U.R.; El-Katatny, M.H. Discovering the chemical profile antimicrobial and antibiofilm potentials of the endophytic fungus Penicillium chrysogenum isolated from Artemisia judaica L. assisted with docking studies. S. Afr. J. Bot. 2022, 151, 218–227. [Google Scholar] [CrossRef]
- Fu, S.F.; Wei, J.Y.; Chen, H.W.; Liu, Y.Y.; Lu, H.Y.; Chou, J.Y. Indole-3-acetic acid: A widespread physiological code in interactions of fungi with other organisms. Plant Signal Behav. 2015, 10, e1048052. [Google Scholar] [CrossRef]
- Shi, Y.; Xie, H.; Cao, L.; Zhang, R.; Xu, Z.; Wang, Z.; Deng, Z. Effects of Cd- and Pb-resistant endophytic fungi on growth and phytoextraction of Brassica napus in metal-contaminated soils. Environ. Sci. Pollut. Res. Int. 2017, 24, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Mehmannavaz, M.; Nickavar, B. Biotransformation of testosterone by the filamentous fungus Penicillium pinophilum. Arch. Microbiol. 2022, 204, 570. [Google Scholar] [CrossRef]
- Tian, H.; Ma, Y.J.; Li, W.Y.; Wang, J.W. Efficient degradation of triclosan by an endophytic fungus Penicillium oxalicum B4. Environ. Sci. Pollut. Res. Int. 2018, 25, 8963–8975. [Google Scholar] [CrossRef]
- Yassin, M.T.; Mostafa, A.A.-F.; Al-Askar, A.A.; Sayed, S.R.M.; Rady, A.M. Antagonistic activity of Trichoderma harzianum and Trichoderma viride strains against some fusarial pathogens causing stalk rot disease of maize, in vitro. J. King Saud. Univ. Sci. 2021, 33, 101363. [Google Scholar] [CrossRef]
- Gao, S.S.; Li, X.M.; Du, F.Y.; Li, C.S.; Proksch, P.; Wang, B.G. Secondary metabolites from a marine-derived endophytic fungus Penicillium chrysogenum QEN-24S. Mar. Drugs 2010, 9, 59–70. [Google Scholar] [CrossRef]
- Gao, S.S.; Li, X.M.; Li, C.S.; Proksch, P.; Wang, B.G. Penicisteroids A and B, antifungal and cytotoxic polyoxygenated steroids from the marine alga-derived endophytic fungus Penicillium chrysogenum QEN-24S. Bioorg. Med. Chem. Lett. 2011, 21, 2894–2897. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.S.; Li, C.M.; Zhang, Y.; Li, C.S.; Wang, B.G. Conidiogenones H and I, two new diterpenes of Cyclopiane class from a marine-derived endophytic fungus Penicillium chrysogenum QEN-24S. Chem. Biodivers. 2011, 8, 1748–1753. [Google Scholar] [CrossRef] [PubMed]
- Baitharu, I.; Jain, V.; Deep, S.N.; Shroff, S.; Sahu, J.K.; Naik, P.K.; Ilavazhagan, G. Withanolide A prevents neurodegeneration by modulating hippocampal glutathione biosynthesis during hypoxia. PLoS ONE 2014, 9, e105311. [Google Scholar] [CrossRef]
- Wang, W.; Zhai, Y.; Cao, K.; Tan, H.; Zhang, R. Endophytic bacterial and fungal microbiota in sprouts, roots and stems of rice (Oryza sativa L.). Microbiol. Res. 2016, 188–189, 1–8. [Google Scholar] [CrossRef]
- Wang, W.G.; Li, A.; Yan, B.C.; Niu, S.B.; Tang, J.W.; Li, X.N.; Du, X.; Challis, G.L.; Che, Y.; Sun, H.D.; et al. LC-MS-guided isolation of Penicilfuranone A: A new antifibrotic furancarboxylic acid from the plant endophytic fungus Penicillium sp. sh18. J. Nat. Prod. 2016, 79, 149–155. [Google Scholar] [CrossRef]
- Colovic, M.B.; Krstic, D.Z.; Lazarevic-Pasti, T.D.; Bondzic, A.M.; Vasic, V.M. Acetylcholinesterase inhibitors: Pharmacology and toxicology. Curr. Neuropharmacol. 2013, 11, 315–335. [Google Scholar] [CrossRef] [PubMed]
- Ateba, J.E.T.; Toghueo, R.M.K.; Awantu, A.F.; Mba’ning, B.M.; Gohlke, S.; Sahal, D.; Rodrigues-Filho, E.; Tsamo, E.; Boyom, F.F.; Sewald, N.; et al. Antiplasmodial properties and cytotoxicity of endophytic fungi from Symphonia globulifera (Clusiaceae). J. Fungi 2018, 4, E70. [Google Scholar] [CrossRef] [PubMed]
- Toghueo, R.M.K.; Boyom, F.F. Endophytic Penicillium species and their agricultural, biotechnological, and pharmaceutical applications. 3 Biotech 2020, 10, 1–35. [Google Scholar] [CrossRef]
- Leitao, A.L.; Garcia-Estrada, C.; Ullan, R.V.; Guedes, S.F.; Martin-Jimenez, P.; Mendes, B.; Martin, J.F. Penicillium chrysogenum var. halophenolicum, a new halotolerant strain with potential in the remediation of aromatic compounds in high salt environments. Microbiol. Res. 2012, 167, 79–89. [Google Scholar] [CrossRef]
- Ferreira-Guedes, S.; Leitao, A.L. Simultaneous removal of dihydroxybenzenes and toxicity reduction by Penicillium chrysogenum var. halophenolicum under saline conditions. Ecotoxicol. Environ. Saf. 2018, 150, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Francis, F.; Jaber, K.; Colinet, F.; Portetelle, D.; Haubruge, E. Purification of a new fungal mannose-specific lectin from Penicillium chrysogenum and its aphicidal properties. Fungal Biol. 2011, 115, 1093–1099. [Google Scholar] [CrossRef]
- Garcia-Estrada, C.; Martin, J.F.; Cueto, L.; Barreiro, C. Omics approaches applied to Penicillium chrysogenum and Penicillin production: Revealing the secrets of improved productivity. Genes 2020, 11, 712. [Google Scholar] [CrossRef]
- Martin, J.F. Insight into the genome of diverse Penicillium chrysogenum strains: Specific genes, cluster duplications and DNA fragment translocatins. Int. J. Mol. Sci. 2020, 21, 3936. [Google Scholar] [CrossRef] [PubMed]
- Sonderegger, C.; Galgoczy, L.; Garrigues, S.; Fizil, A.; Borics, A.; Manzanares, P.; Hegedus, N.; Huber, A.; Marcos, J.F.; Batta, G.; et al. A Pencillium chrysogenum-based expression system for the production of mall, cysteine-rich antifungal proteins for structural and functional analyses. Microb. Cell Fact. 2016, 15, 192. [Google Scholar] [CrossRef]
- Estrada-Rivera, M.; Hernandez-Onate, M.A.; Dautt-Castro, M.; Gallardo-Negrete, J.D.J.; Rebolledo-Prudencio, O.G.; Uresti-Rivera, E.E.; Arenas-Huertero, C.; Herrera-Estrella, A.; Casas-Flores, S. IPA-1 a putative chromatin remodeler/helicase-related protein of Trichodermis virens plays important roles in antibiosis against Rhizoctonia solani and induction of Arabidopsis systemic disease resistance. Mol. Plant Microbe Interact. 2020, 33, 808–824. [Google Scholar] [CrossRef]
- Garcia-Rico, R.O.; Martin, J.F.; Fierro, F. Heterotrimeric Ga protein Pga1 from Penicillium chrysogenum triggers germination in response to carbon sources and affects negatively resistance to different stress conditions. Fungal Gen. Biol. 2011, 48, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Akaniro, I.R.; Chibuikde, I.V.; Onwujekwe, E.C.; Gbadamosti, F.A.; Enyi, D.O.; Onwe, O.N. Penicillium species as chassis for biomanufacturing and environmental sustainability in the modern era: Progress, challenges, and future perspective. Fungal. Biol. Rev. 2023, 46, 100326. [Google Scholar] [CrossRef]
- Meng, L.; Sun, P.; Tang, H.; Li, L.; Draeger, S.; Schulz, B.; Krohn, K.; Hussain, H.; Zhang, W.; Yi, Y. Endophytic fungus Penicillium chrysogenum, a new source of hypocrellins. Biochem. Syst. Ecol. 2011, 39, 163–165. [Google Scholar] [CrossRef]
- Qin, J.; Teng, J.; Li, Z.; Xia, N.; Wei, B.; Huang, L. Expression of citrinin biosynthesis gene in Liupao tea and effect of Penicillium citrinum on tea quality. Fungal Gene Biol. 2022, 163, 103742. [Google Scholar] [CrossRef]
- Guijarro, B.; Larena, I.; Melgarejo, P.; Cal, A.D. Adaptive conditions and safety of the application of Penicillium frequentans as a biocontrol agent on stone fruit. Int. J. Food Microbiol. 2017, 254, 25–35. [Google Scholar] [CrossRef]
- Arunthirumeni, M.; Vinitha, G.; Shivakumar, M.S. Antifeedant and larvicidal activity of bioactive compounds isolated from entomopathogenic fungi Penicillium sp. for the control of agricultural and medically important insect pest (Spodoptera litura and Culex quinquefasciatus). Parasitol. Int. 2023, 92, 102688. [Google Scholar] [CrossRef]
- Karpova, N.V.; Yaderets, V.V.; Glagoleva, E.V.; Petrova, K.S.; Ovchinnikov, A.I.; Dzhavakhiya, V.V. Antifungal activity of the dry biomass of Penicillium chrysogenum F-24-28 and Is application in combination with azoxystrobin for efficient crop protection. Agriculture 2021, 11, 935. [Google Scholar] [CrossRef]
- Sikandar, A.; Zhang, M.; Wang, Y.; Zhu, X.; Liu, X.; Fan, H.; Xuan, Y.; Chen, L.; Duan, Y. In vitro evaluation of Penicillium chrysogenum Snef1216 against Meloidogyne incognita (root-knot nematode). Sci. Rep. 2020, 10, 8342. [Google Scholar] [CrossRef]
- Khan, S.A.; Hamayun, M.; Yoon, H.; Kim, H.-Y.; Suh, S.-J.; Hwang, S.-K.; Kim, J.-M.; Lee, I.-J.; Choo, Y.-S.; Yoon, U.-H.; et al. Plant growth promotion and Penicillium citrinum. BMC Microbiol. 2008, 8, 231. [Google Scholar] [CrossRef]
- Nguyen, H.C.; Lin, K.-H.; Nguyen, T.P.; Le, H.S.; Ngo, K.N.; Pham, D.C.; Tran, T.N.; Su, C.-H.; Barrow, C.J. Isolation and cultivation of Penicillium citrinum for biological control of Spodoptera litura and Plutella xylostella. Fermentation 2023, 9, 438. [Google Scholar] [CrossRef]
- Babu, J.V.; Popay, A.J.; Miles, C.O.; Wilkins, A.L.; di Menna, M.E.; Finch, S.C. Identification and structure elucidation of Janthitrems A and D from Penicillium janthinellum and determination of tremorgenic and anti-insect activity of Janthitrems A and B. J. Agric. Food Chem. 2018, 66, 13116–13125. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yang, X.; Lin, Y.; Yuan, J.; Lu, Y.; Zhu, X.; Li, J.; Li, M.; Lin, Y.; He, J.; et al. Meroterpenes and azaphilones from marine mangrove endophytic fungus Penicillium 303#. Fitoterapa 2014, 97, 241–246. [Google Scholar] [CrossRef]
- Li, X.D.; Miao, F.P.; Liang, X.R.; Ji, N.Y. Meroterpenes from an algicolous strain of Penicillium echinulatum. Magn. Reson. Chem. 2014, 52, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Katoch, M.; Pull, S. Endophytic fungi associated with Monarda citriodora, an aromatic and medicinal plant and their biocontrol potential. Pharm. Biol. 2017, 55, 1528–1535. [Google Scholar] [CrossRef]
- Li, Z.F.; Wang, L.F.; Feng, Z.L.; Zhao, L.H.; Shi, Y.Q.; Zhu, H.Q. Diversity of endophytic fungi from different Verticillium-wilt-resistant Gossypium hirsutum and evaluation of antifungal activity against Verticillium dahliae in vitro. J. Microbiol. Biotechnol. 2014, 24, 1149–1161. [Google Scholar] [CrossRef]
- Hassan, S.E. Plant growth-promoting activities for bacterial and fungal endophytes isolated from medicinal plant of Teucrium polium L. J. Adv. Res. 2017, 8, 687–695. [Google Scholar] [CrossRef]
- Ownley, B.H.; Benson, D.M. Evaluation of Penicillium janthinellum as a biological control of phytophthora root rot of Azalea. J. Amer. Soc. Hort. Sci. 1992, 117, 407–410. [Google Scholar] [CrossRef]
- Gu, K.; Chen, C.-Y.; Selvaraj, P.; Pavagadhi, S.; Yeap, Y.T.; Swarup, S.; Zheng, W.; Naqvi, N.I. Penicillium citrinum provides transkingdom growth benefits in Choy Sum (Brassica rapa var. parachinensis). J. Fungi 2023, 9, 420. [Google Scholar] [CrossRef]
- Dong, H.; Zhang, X.; Choen, Y.; Zhou, Y.; Li, W.; Li, Z. Dry mycelium of Penicillium chrysogenum protects cotton plants against wilt diseases and increases yield under field conditions. Crop Prot. 2006, 25, 324–330. [Google Scholar] [CrossRef]
- Sikandar, A.; Zhang, M.Y.; Zhu, X.F.; Wang, Y.Y.; Ahmed, M.; Iqbal, M.F.; Javeed, A.; Xuan, Y.H.; Fan, H.Y.; Liu, X.Y.; et al. Efficacy of Penicillium chrysogenum strain SNEF1216 against root-knot nematodes (Meloidogyne incognita) in cucumber (Cucumis sativus L.) under greenhouse conditions. Appl. Ecol. Environ. Res. 2019, 17, 12451–12464. [Google Scholar] [CrossRef]
- Li, S.-Y.; Yang, X.-Q.; Chen, J.-X.; Wu, Y.-M.; Yang, Y.-B.; Ding, Z.-T. The induced cryptic metabolites and antifungal activities from culture of Penicillium chrysogenum by supplementing with host Ziziphus jujuba extract. Phytochemistry 2022, 203, 113391. [Google Scholar] [CrossRef] [PubMed]
- Galeano, R.M.S.; Silva, S.M.; Yonekawa, M.K.A.; Guimaraes, N.C.D.A.; Giannesi, G.C.; Masui, D.C.; Correa, B.O.; Brasil, M.D.S.; Zanoelo, F.F. Penicillium chrysogenum strain 34-P promotes plant growth and improves initial development of maize under saline conditions. Rhizosphere 2023, 26, 100710. [Google Scholar] [CrossRef]
- Murali, M.; Sudisha, J.; Amruthesh, K.N.; Ito, S.-I.; Shetty, H.S. Rhizosphere fungus Penicillium chrysogenum promotes growth and induces defence-related genes and downy mildew disease resistance in pearl millet. Plant Biol. 2013, 15, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Gotlieb, D.; Oka, Y.; Ben-Daniel, B.-H.; Cohen, Y. Dry mycelium of Penicillium chrysogenum protects cucumber and tomato plants against root-knot nematode Meloidogyne javanica. Phytoparasitica 2003, 31, 217–225. [Google Scholar] [CrossRef]
- Khan, A.L.; Waqas, M.; Khan, A.R.; Hussain, J.; Kang, S.-M.; Gilani, S.A.; Hamayun, M.; Shin, J.-H.; Kamran, M.; Al-Harrasi, A.; et al. Fungal endophyte Penicillium janthinellum LK5 improves growth of ABA-deficient tomato under salinity. World J. Microbiol. Biotechnol. 2013, 29, 2133–2144. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.L.; Waqas, M.; Hussain, J.; Al-Harrasi, A.; Hamayun, M.; Lee, I.-J. Phytohormones enabled endophytic fungal symbiosis improve aluminum phytoextraction in tolerant Solanum lycopersicum: A examples of Penicillium janthinellum LK5 and comparison with exogenous GA3. J. Hazard. Mat. 2015, 295, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Ting, A.S.Y.; Mah, S.W.; Tee, C.S. Evaluating the feasibility of induced host resistance by endophytic isolate Penicillium citrinum BTF08 as a control mechanism for Fusarium wilt in banana plantlets. Biol. Control 2012, 61, 155–159. [Google Scholar] [CrossRef]
- Chen, S.; Dong, H.; Fan, Y.; Li, W.; Cohen, Y. Dry mycelium of Penicillium chrysogenum induces expression of pathogenesis-related protein genes and resistance against wilt diseases in Bt transgenic cotton. Biol. Control 2006, 39, 460–464. [Google Scholar] [CrossRef]
- Dong, H.; Li, W.; Zhang, D.; Tang, W. Differential expression of induced resistance by an aqueous extract of killed Penicillium chrysogenum against Verticillium wilt of cotton. Crop Prot. 2003, 22, 129–134. [Google Scholar] [CrossRef]
- Wang, H.; Mo, S.; Xia, Q.; Zhao, Z.; Chen, X.; Shen, X.; Yin, C.; Mao, Z. The interaction of the pathogen Fusarium proliferatum with Trichoderma asperellum characterized by transcriptome changes in apple rootstock roots. Physiol. Mol. Plant Pathol. 2022, 121, 101894. [Google Scholar] [CrossRef]
- Li, X.; Leng, J.; Yu, L.; Bai, H.; Li, X.; Wisniewski, M.; Liu, J.; Sui, Y. Efficacy of the biocontrol agent Trichoderma hamatum against Lasiodiplodia theobromae on macadamia. Front. Microbiol. 2022, 13, 994422. [Google Scholar] [CrossRef] [PubMed]
- Ibiang, S.R.; Usami, T.; Sakamoto, K.; Ibiang, Y.B. Lettuce tolerance to verticillium wilt after inoculation with Penicillium pinophilum and Rhizophagus intraradices. Physiol. Mol. Plant Pathol. 2023, 128, 102171. [Google Scholar] [CrossRef]
- Dong, H.; Cohen, Y. Extracts of killed Penicillium chrysogenum induce resistance against Fusarium wilt of melon. Phytoparasitica 2001, 29, 421–430. [Google Scholar] [CrossRef]
- Shen, F.; Wang, G.; Liu, X.; Zhu, S. Exogenous inoculation of endophyte Penicillium sp. alleviated pineapple internal browning during storage. Heliyon 2023, 9, e16258. [Google Scholar] [CrossRef]
- Maity, A.; Pal, R.K.; Chandra, R.; Singh, N.V. Penicillium pinophilum—A novel microorganism for nutrient management in pomegranate (Punica granatum L.). Sci. Hortic. 2014, 169, 111–117. [Google Scholar] [CrossRef]
- Rosa, C.A.R.; Keller, K.M.; Oliveira, A.A.; Almeida, T.X.; Keller, L.A.M.; Marassi, A.C.; Kruger, C.D.; Deveza, M.V.; Monteiro, B.S.; Nunes, L.M.T.; et al. Production of citreoviridin by Penicillium citreonigrum strains associated with rice consumption and beriberi cases in the Maranhao state, Brazil. Food Additt. Contam. Part A Chem. Anal. Control Expo. Risk Assess 2010, 27, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.C.; Duan, Y.M.; Wang, X.X.; Wang, J.G.; Xu, X.Y.; Chen, S.L.; Yin, P.; Zhu, S.; Li, X.J.; Chen, S.Y. The effect of dry mycelium of Penicillium chrysogenum on the growth of flue-cured tobacco on floating system and resistance against tobacco mosaic under field conditions. J. Yunnan Agric. Univ. 2013, 28, 169–174. [Google Scholar]
- Zhong, Y.; Li, Y.; Huang, K.; Chen, Z.-Z.; Fu, J.; Liu, C.-M.; Chen, S.-Y.; Wang, J.-G. Crude peptides extracted from dry mycelium of Penicillium chrysogenu serve as a micro-associated molecular pattern to induce systemic resistance against tobacco mosaic virus in tobacco. Physiol. Mol. Plant Pathol. 2021, 115, 101677. [Google Scholar] [CrossRef]
- Zhong, Y.; Li, Y.; Chen, Z.; Fu, J.; Li, X.; Zhang, B.; Chen, S.; Wang, J. Treatment of Penicillium chrysogenum extracts (PDMP) restricts the spread of Tobacco mosaic virus by priming callose deposition in Nicotiana benthamiana. Physiol. Mol. Plant Pathol. 2021, 113, 101569. [Google Scholar] [CrossRef]
- Zhong, Y.; Pen, J.-J.; Chen, Z.-Z.; Xie, H.; Luo, D.; Dai, J.-R.; Yan, F.; Wang, J.-G.; Dong, H.-Z.; Chen, S.-Y. Dry mycelium of Penicillium chrysogenum activates defense responses and restricts the spread of Tobacco Mosaic Virus in tobacco. Physiol. Mol. Plant Pathol. 2015, 92, 28–37. [Google Scholar] [CrossRef]
- Wang, J.; Chen, S. Dry mycelium of Penicillium chrysogenum protect flue-cured tobacco against brown spot and wildfire disease. New Biotechnol. 2012, 29, S190. [Google Scholar] [CrossRef]
- Li, Y.; Jiao, M.; Li, Y.; Zhong, Y.; Li, X.; Chen, Z.; Chen, S.; Wang, J. Penicillium chrysogenum polypeptide extract protects tobacco plants from tobacco mosaic virus infection through modulation of ABA biosynthesis and callose priming. J. Experim. Bot. 2021, 72, 3526–3539. [Google Scholar] [CrossRef]
- Fu, J.; Zhang, S.; Wu, J.; Chen, Y.; Zhong, Y.; Zhou, Y.; Wang, J.; Chen, S. Structural characterization of a polysaccharide from dry mycelium of Penicillium chrysogenum that induces resistance to Tobacco mosaic virus in tobacco plants. Int. J. Biol. Macromol. 2020, 156, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Saxena, S. Penicillium citrinum, a drought-tolerant endophytic fungus isolated from wheat (Triticum aestivum L.) leaves with plant growth-promoting abilities. Curr. Microbiol. 2023, 80, 184. [Google Scholar] [CrossRef]
- Narwade, J.D.; Odaneth, A.A.; Lele, S.S. Solid-state fermentation in an earthen vessel: Trichoderma viride spore-based biopesticide production using corn cobs. Fungal Biol. 2023, 127, 1146–1156. [Google Scholar] [CrossRef] [PubMed]
- Juric, S.; Dermic, E.; Topolovec-Pintaris, S.; Bedek, M.; Vincekovic, M. Physicochemical properties and release characteristics of caldium alginate microspheres loaded with Trichoderma viride spores. J. Integr. Agric. 2019, 18, 2534–2548. [Google Scholar] [CrossRef]
- Nabilah, B.; Purnomo, A.S.; Prasetyoko, D.; Rohmah, A.A. Methylene blue biodecolotization and biodegradation by immobilized mixed cultures of Trichoderma viride and Ralstonia pickettii into SA-PVA-Bentionite matrix. Arab J. Chem. 2023, 16, 104940. [Google Scholar] [CrossRef]
- Xu, S.; Luo, Y.; Han, Z.; Zhang, T.; Sun, L.; Zheng, G.; Wang, K.; Cheng, Z. Diatomite-Trichoderma viride composite microspheres for selective removal of anionic dyes and copper ions. J. Water Process Eng. 2023, 55, 104235. [Google Scholar] [CrossRef]
- Alonso-Ramirez, A.; Poveda, J.; Martin, H.; Hermosa, R.; Monte, E.; Nicolas, C. Salicylic acid prevents Trichoderma harzianum from entering the vascular system of roots. Mol. Plant Pathol. 2014, 15, 823–831. [Google Scholar] [CrossRef]
- Taylor, J.T.; Harting, R.; Shalaby, S.; Kenerley, C.M.; Braus, G.H.; Horwitz, B.A. Adhesion as a focus in Trichoderma-root interactions. J. Fungi 2022, 8, 372. [Google Scholar] [CrossRef]
- Pfordt, A.; Gaumann, P.; von Tiedemann, A. Pathogenicity of Trichoderma afroharzianum in cereal crops. Pathogen 2023, 12, 936. [Google Scholar] [CrossRef] [PubMed]
- Pfordt, A.; Schiwek, S.; Karlovsky, P.; von Tiedemann, A. Trichoderma afroharzianum ear rot- A new disease on maize in Europe. Front. Agron. 2020, 2, 547758. [Google Scholar] [CrossRef]
- Sanna, M.; Pugliese, M.; Gullino, M.L.; Mezzalama, M. First report of Trichoderma afroharzianum causing seed rot on maize in Italy. Plant Diseases 2022, 106, 1982. [Google Scholar] [CrossRef]
- Schmoll, M.; Esquivel-Naranjo, E.U.; Herrera-Estrella, A. Trichoderma in the light of day-Physiology and development. Fungal Genet. Biol. 2010, 47, 909–916. [Google Scholar] [CrossRef]
- Kubicek, C.P.; Herrera-Estrella, A.; Seidl-Seiboth, V.; Martinez, D.A.; Druzhini, I.S.; Thon, M.; Zeilinger, S.; Casas-Flores, S.; Horwitz, B.A.; Mukherjee, P.K. Comparative genome sequence analysis underscores mycoparasitism as the ancestral life style of Trichoderma. Genome Biol. 2011, 12, R40. [Google Scholar] [CrossRef]
- Schuster, A.; Bruno, K.S.; Collett, J.R.; Baker, S.E.; Seiboth, B.; Kubicek, C.P.; Schmoll, M. A versatile toolkit for high throughput functional genomics with Trichoderma reesei. Biotechnol. Biofuels 2012, 5, 1. [Google Scholar] [CrossRef]
- Chammem, H.; Nesler, A.; Pertot, I. Wood pellets as carriers of conidia of Trichoderma atroviride SC1 for soil application. Fungal Biol. 2021, 125, 989–998. [Google Scholar] [CrossRef]
- Zhang, G.-Z.; Yang, H.-T.; Zhang, X.-J.; Zhou, F.-Y.; Wu, X.-Q.; Xie, X.-Y.; Zhao, X.-Y.; Zhou, H.-Z. Five new species of Trichoderma from moist soils in China. Mycokeys 2022, 87, 133. [Google Scholar] [CrossRef]
- Woo, S.L.; Hermosa, R.; Lorito, M.; Monte, E. Trichoderma: A multipurpose, plant-beneficial microorganism for eco-sustainable agriculture. Nat. Rev. Microbiol. 2023, 21, 312–326. [Google Scholar] [CrossRef]
- Verma, H.; Kumar, D.; Kumar, V.; Kumari, M.; Singh, S.K.; Sharma, V.K.; Droby, S.; Santoyo, G.; White, J.F.; Kumar, A. The potential application of endophytes in management of stress from drought and salinity in crop plants. Microorganisms 2021, 9, 1729. [Google Scholar] [CrossRef]
- Perera, D.S.; Tharaka, W.G.H.; Amarasinghe, D.; Wickramarachchi, S.R. Extracellular extracts of antagonistic fungi, Trichoderma longibrachiatum and Trichoderma viride, as larvicides against dengue vectors, Aedes aegypti and Aedes albopictus. Acta Trop 2023, 238, 106747. [Google Scholar] [CrossRef] [PubMed]
- Moussa, Z.; Alanazi, Y.F.; Khateb, A.M.; Eldadamony, N.M.; Ismail, M.M.; Saber, W.I.; Darwish, D.B.E. Domiciliation of Trichoderma asperellum suppresses Globiosporangium ultimum and promotes pea growth, ultrastructure, and metabolic features. Microorganisms 2023, 11, 198. [Google Scholar] [CrossRef] [PubMed]
- Fayzalla, E.; Sadik, E.; Elwakil, M.; Gomah, A. Soil solarization for controlling Cephalosporium maydis, the cause of late wilt disease of maize in Egypt. Egypt J. Phytopathol. 1994, 22, 171–178. [Google Scholar]
- Legein, M.; Smets, W.; Vandenheuvel, D.; Eilers, T.; Muyshondt, B.; Prinsen, E.; Samson, R.; Lebeer, S. Modes of action of microbial biocontrol in the phyllosphere. Front. Microbiol. 2020, 11, 1619. [Google Scholar] [CrossRef]
- Alghuthaymi, M.A.; Abd-Elsalam, K.A.; AboDalam, H.M.; Ahmed, F.K.; Ravichandran, M.; Kalia, A.; Rai, M. Trichoderma: An eco-friendly source of nanomaterials for sustainable agroecosystems. J. Fungi 2022, 8, 367. [Google Scholar] [CrossRef]
- Zhang, F.; Xu, X.; Huo, Y.; Xiao, Y. Trichoderma-inoculation and mowing synergistically altered soil available nutrients, rhizosphere chemical compounds and soil microbial community, potentially driving alfalfa growth. Front. Microbiol. 2019, 9, 3241. [Google Scholar] [CrossRef]
- Molla, A.H.; Manjurul Haque, M.; Amdadul Haque, M.; Ilias, G.N.M. Trichoderma-enriched biofertilizer enhances production and nutritional quality of tomato (Lycopersicon esculentum Mill.) and minimizes NPK fertilizer use. Agric. Res. 2012, 1, 265–272. [Google Scholar] [CrossRef]
- Sun, J.; Yuan, X.; Li, Y.; Wang, X.; Chen, J. The pathway of 2,2-dichlorovinyl dimethyl phosphate (DDVP) degradation by Trichoderma atroviride strain T23 and characterization of a paraoxonase-like enzyme. Appl. Microbiol. Biotechnol. 2019, 103, 8947–8962. [Google Scholar] [CrossRef] [PubMed]
- Lodi, R.S.; Peng, C.; Dong, X.; Deng, P.; Peng, L. Trichoderma hamatum and its benefits. J. Fungi 2023, 9, 994. [Google Scholar] [CrossRef]
- Velasco, P.; Rodriguez, V.M.; Soengas, P.; Pveda, J. Content and antioxidant potential of different leafy Brassica vegetables. Plants 2021, 10, 2449. [Google Scholar] [CrossRef]
- Krause, M.S.; De Ceuster, T.J.J.; Tiquia, S.M.; Michel, F.C.; Madden, L.V.; Hoitink, H.A.J. Isolation and characterization of rhizobacteria from composts that suppress the severity of bacterial leaf spot of radish. Phytopathology 2003, 93, 1292–1300. [Google Scholar] [CrossRef] [PubMed]
- Daba, A.; Berecha, G.; Tadesse, M.; Belay, A. Evaluation of the herbicidal potential of some fungal species against Bidens Pilosa, the coffee farming wees. Saudi. J. Biol. Sci. 2021, 28, 6408–6416. [Google Scholar] [CrossRef] [PubMed]
- Wen, C.; Xiong, H.; Wen, J.; Wen, X.; Wang, C. Trichoderma species attract Coptotermes formosanus and antagonize termite pathogen Metarhizium anisopliae. Front. Microbiol. 2020, 11, 00653. [Google Scholar] [CrossRef] [PubMed]
- Lana, M.; Simon, O.; Velasco, P.; Rodriguez, V.M.; Caballero, P.; Poveda, J. First study on the root endophytic fungus Trichoderma hamatum as an entomopathogen: Development of a fungal bioinsecticide against cotton leafworm (Spodoptera littoralis). Microbiol. Res. 2023, 270, 127334. [Google Scholar] [CrossRef]
- Rai, S.; Kashyap, P.L.; Kumar, S.; Srivastava, A.K.; Ramteke, P.W. Identification, characterization and phylogenetic analysis of antifungal Trichoderma from tomato rhizosphere. Springerplus 2016, 5, 1939. [Google Scholar] [CrossRef]
- Sood, M.; Kapoor, D.; Kumar, V.; Sheteiwy, M.S.; Ramakrishnan, M.; Landi, M.; Araniti, F.; Sharma, A. Trichoderma: The secrets of a multitalented biocontrol agent. Plants 2020, 9, 762. [Google Scholar] [CrossRef]
- Moran-Diez, E.; Hermosa, R.; Ambrosino, P.; Cardoza, R.E.; Gutierrez, S.; Lorito, M.; Monte, E. ThPG1 endopolygalacturonase is required for the Trichoderma harzianum-plant beneficial interaction. Mol. Plant Microbe Interact. 2009, 22, 1021–1031. [Google Scholar] [CrossRef]
- Jaroszuk-Scisel, J.; Tyskiewicz, R.; Nowak, A.; Ozimek, E.; Majewska, M.; Hanaka, A.; Tyskiewicz, K.; Pawlik, A.; Janusz, G. Phytohormones (auxin, gibberellin) and ACC deaminase in vitro synthesized by the mycoparasitic Trichoderma DEMTkZ3A0 strain and changes in the level of auxin and plant resistance markers in wheat seedlings inoculated with this strain conidia. Int. J. Mol. Sci. 2019, 20, 4923. [Google Scholar] [CrossRef]
- An, X.-Y.; Cheng, G.-H.; Gao, H.-X.; Li, X.-F.; Yang, Y.; Li, D.; Li, Y. Phylogenetic analysis of Trichoderma species associated with green mold disease on mushrooms and two new pathogens on Ganoderma sichuanense. J. Fungi 2022, 8, 704. [Google Scholar] [CrossRef]
- Krause, M.S.; Madden, L.V.; Hoitink, H.A.J. Effect of potting mix microbial carrying capacity on biological control of Rhizoctonia damping-off of radish and Rhizoctonia crown root rot of poinsettia. Phytopathology 2001, 91, 1116–1123. [Google Scholar] [CrossRef]
- Alfano, G.; Lewis Ivey, M.L.; Cakir, C.; Bos, J.I.B.; Miller, S.A.; Madden, L.V.; Kamoun, S.; Hoitink, H.A.J. Systemic modulation of gene expression in tomato by Trichoderma hamatum 382. Phytopathology 2007, 97, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Baazeem, A.; Almanea, A.; Manikandan, P.; Alorabi, M.; Vijayaraghavan, P.; Abdel-Hadi, A. In vitro antibacterial, antifungal, nematocidal and growth promoting activities of Trichoderma hamatum fb10 and its secondary metabolites. J. Fungi 2021, 7, 331. [Google Scholar] [CrossRef] [PubMed]
- Wamani, A.O.; Muthomi, J.W.; Mutitu, E.; Waceke, W.J. Efficacy of microbial antagonists in the management of bacterial wilt of field-grown tomato. J. Nat. Pestic. Res. 2023, 6, 100051. [Google Scholar] [CrossRef]
- Shaw, S.; Le Cocq, K.; Paszkiewicz, K.; Moore, K.; Winsbury, R.; De Torres Zabala, M.; Studholme, D.J.; Salmon, D.; Thornton, C.R.; Grant, M.R. Transcriptional reprogramming underpins enhanced plant growth promotion by the biocontrol fungus Trichoderma hamatum gd12 during antagonistic interactions with Sclerotinia sclerotiorum in soil. Mol. Plant Pathol. 2016, 17, 1425–1441. [Google Scholar] [CrossRef]
- Studholme, D.J.; Harris, B.; Le Cocq, K.; Winsbury, R.; Perera, V.; Ryder, L.; Ward, J.L.; Beale, M.H.; Thornton, C.R.; Grant, M. Investigating the beneficial traits of Trichoderma hamatum GD12 for sustainable agriculture-insights from genomics. Front. Plant Sci. 2013, 4, 00258. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Shahrajabian, M.H. Survey on nitrogenase evolution by considering the importance of nitrogenase, its structure, and mechanism of nitrogenase. Not. Bot. Horti Agrobot. Cluj-Napoca 2024, 52, 13157. [Google Scholar] [CrossRef]
- Sun, W.; Shahrajabian, M.H.; Kuang, Y.; Wang, N. Amino acids biostimulants and protein hydrolysates in agricultural sciences. Plants 2024, 13, 210. [Google Scholar] [CrossRef]
- Sun, W.; Shahrajabian, M.H.; Soleymani, A. The roles of plant-growth-promoting rhizobacteria (PGPR)-based biostimulants for agricultural production systems. Plants 2024, 13, 613. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Wang, Y.; Suo, M.; Wu, H.; Zhao, M.; Yang, H. Inoculation with Penicillium citrinum aids ginseng in resisting Fusarium oxysporum by regulating the root and rhizosphere microbial communities. Rhizosphere 2022, 22, 100535. [Google Scholar] [CrossRef]
- Liu, R.; Chen, M.; Gao, J.; Luo, M.; Wang, G. Identification of antagonistic fungi and their antifungal activities against aconite root rot pathogens. Plant Signal Behav. 2023, 18, 2211852. [Google Scholar] [CrossRef]
- Poveda, J.; Rodriguez, V.M.; Abilleira, R.; Velasco, P. Trichoderma hamatum can act as inter-plant communicator of foliar pathogen infections by colonizing the roots of nearby plants: A new inter-plant wired communication. Plant Sci. 2023, 330, 111664. [Google Scholar] [CrossRef] [PubMed]
- Kottb, M.; Gigolashvili, T.; GroBkinsky, D.K.; Piechulla, B. Trichoderma volatiles effecting Arabidopsis: From inhibition to protection against phytopathogenic fungi. Front. Microbiol. 2015, 6, 995. [Google Scholar] [CrossRef]
- Garnica-Vergara, A.; Barrera-Ortiz, S.; Munoz-Parra, E.; Raya-Gonzalez, J.; Mendez-Bravo, A.; Macias-Rodriguez, L.; Ruiz-Herrera, L.F.; Lopez-Bucio, J. The volatile 6-pentyl-2H-pyran-2-one from Trichoderma atroviride regulates Arabidopsis thaliana root morphogenesis via auxin signaling and ETHYLENE INSENSITIVE 2 functioning. New Phytol. 2016, 209, 1496–1512. [Google Scholar] [CrossRef]
- Boukaew, S.; Kumla, J.; Prasertsan, P.; Cheirsilp, B.; Petlamul, W.; Srinuanpan, S. In vitro and in situ antifungal properties of a Trichoderma asperelloides SKRU-01 against aflatoxigenic aspergillus species. Food Control. 2023, 14, 110025. [Google Scholar] [CrossRef]
- Haouhach, S.; Karkachi, N.; Oguiba, B.; Sisaoui, A.; Chamorro, I.; Kihal, M.; Monte, E. Three new reports of Trichoderna in Algeria: T. atrobrunneum (South), T. longibrachiatum (South), and T. afroharzianum (Northwest). Microorganisms 2020, 8, 1455. [Google Scholar] [CrossRef]
- Ma, J.; Tsegaye, E.; Li, M.; Wu, B.; Jiang, X. Biodiversity of Trichoderma from grassland and forest ecosystems in Northern Xinjiang, China. 3 Biotech 2020, 10, 362. [Google Scholar] [CrossRef]
- Saravanakumar, K.; Fan, L.; Fu, K.; Yu, C.; Wang, M. Cellulase from Trichoderma harzianum interacts with roots and triggers induced systemic resistance to foliar disease in maize. Sci. Rep. 2016, 6, 35543. [Google Scholar] [CrossRef] [PubMed]
- Inglis, P.W.; Mello, S.C.M.; Martins, I.; Silva, J.B.T.; Macedo, K.; Sifuentes, D.N.; Valadares-Inglis, M.C. Trichoderma from Brazilian garlic and onion crop soils and description of two new species: Trichoderma azevedoi and Trichoderma peberdyi. PLoS ONE 2020, 15, e0228485. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, J.L.; Chen, J.; Mao, L.J.; Feng, X.X.; Zhang, C.L.; Lin, F.C. Trichoderma biodiversity of agricultural fields in east China reveals a gradient distribution of species. PLoS ONE 2016, 11, e0160613. [Google Scholar] [CrossRef]
- Cummings, N.; Ambrose, A.; Braithwaite, M.; Bissett, J.; Roslan, H.A.; Abdullah, J.; Stewart, A.; Agbayani, F.V.; Steyaert, J.; Hill, R.A. Diversity of root-endophytic Trichoderma from Malaysian Borneo. Mycol. Progr. 2016, 15, 45–50. [Google Scholar] [CrossRef]
- Swain, H.; Adak, T.; Mkherjee, A.K.; Sarangi, S.; Samal, P.; Khandual, A.; Jena, R.; Bhattacharyya, P.; Naik, S.K.; Mehetre, S.T.; et al. Seed biopriming with Trichoderma stains isolated from tree bark improves plant growth, antioxidative defense system in rice and enhance straw degradation capacity. Front. Microbiol. 2021, 12, 633881. [Google Scholar] [CrossRef] [PubMed]
- Blaszczyk, L.; Strakowska, J.; Chelkowski, J.; Gabka-Buszek, A.; Kaczmarek, J. Trichoderma species occurring on wood with decay symptoms in mountains forests in Central Europe: Genetic and enzymatic characterization. J. Appl. Genet. 2016, 57, 397–407. [Google Scholar] [CrossRef]
- Bohacz, J.; Kornillowicz-Kowalska, T. Modification of post-industrial lignin by fungal strains of the genus Trichoderma isolated from different composting stages. J. Environ. Manag. 2020, 266, 110573. [Google Scholar] [CrossRef] [PubMed]
- Kubiak, A.; Wolna-Maruwka, A.; Pilarska, A.A.; Niewiadomska, A.; Piotrowska-Cyplik, A. Fungi of the Trichoderma genus: Future perspectives of benefits in sustainable agriculture. Appl. Sci. 2023, 13, 6434. [Google Scholar] [CrossRef]
- Ruocco, M.; Lanzuise, S.; Lombardi, N.; Woo, S.L.; Vinale, F.; Marra, R.; Varlese, R.; Manganiello, G.; Pascale, A.; Scala, V.; et al. Multiple roles and effects of a novel Trichoderma hydrophobin. Mol. Plant-Microbe Interact. 2015, 28, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Valdespino, C.A.; Porras-Troncoso, M.D.; Corrales-Escobosa, A.R.; Wrobel, K.; Martinez-Hernandez, P.; Olmedo-Monfil, V. Functional characterization of TvVyt2, a member of the p450 monoocygenases from Trichoderma virens relevant during the association with plants and mycoparasitism. Mol. Plant-Microbe Interact. 2018, 31, 289–298. [Google Scholar] [CrossRef]
- Rimkus, A.; Namina, A.; Dzierkale, M.T.; Grigs, O.; Senkovs, M.; Larsson, S. Impact of growth conditions on the viability of Trichoderma asperellum during storage. Microorganisms 2023, 11, 1084. [Google Scholar] [CrossRef] [PubMed]
- Brotman, Y.; Briff, E.; Viterbo, A.; Chet, I. Role of swollenin, an expansin-like protein from Trichoderma, in plant root colonization. Plant Physiol. 2008, 147, 779–789. [Google Scholar] [CrossRef]
- Zhang, S.; Gan, Y.; Xu, B. Application of plant-growth-promoting fungi Trichoderma longibrachiatum T6 enhances tolerance of wheat to salt stress through improvement of antioxidative defense system and gene expression. Front. Plant Sci. 2016, 7, 1405. [Google Scholar] [CrossRef]
- Ghorbanpour, A.; Salimi, A.; Ghanbary, M.A.T.; Pirdashti, H.; Dehestani, A. The effect of Trichoderma harzianum in mitigating low temperature stress in tomato (Solanum lycopersicum L.) plants. Sci. Hortic. 2018, 230, 134–141. [Google Scholar] [CrossRef]
- Gallou, A.; Cranenbrouk, S.; Declerck, S. Trichoderma harzianum elicits defence response genes in roots of potato plantlets challenged by Rhizoctonia solani. Eur. J. Plant Pathol. 2008, 124, 219–230. [Google Scholar] [CrossRef]
- Wang, Y.; Hou, X.; Deng, J.; Yao, Z.; Lyu, M.; Zhang, R. Auxin response factor 1 acts as a positive regulator in the response of poplar to Trichoderma asperellum inoculation in over-expressing plants. Plants 2020, 9, 272. [Google Scholar] [CrossRef]
- Kumar, V.; Koul, B.; Taak, P.; Yadav, D.; Song, M. Journey of Trichoderma from pilot scale to mass production: A review. Agriculture 2023, 13, 2022. [Google Scholar] [CrossRef]
- Abbas, A.; Mubeen, M.; Zheng, H.; Sohail, M.A.; Shakeel, Q.; Solanki, M.K.; Iftikhar, Y.; Sharma, S.; Kashyap, B.K.; Hussain, S.; et al. Trichoderma spp. Genes involved in the biocontrol activity against Rhizoctonia solani. Front. Microbiol. 2022, 13, 884469. [Google Scholar] [CrossRef]
- Marcello, C.M.; Steindorff, A.S.; da Silva, S.P.; do Nascimento Silva, R.; Bataus, L.A.M.; Ulhoa, C.J. Expression analysis of the exo-B-1, 3-glucanase from the mycoparasitic fungus Trichoderma asperellum. Microbiol. Res. 2010, 165, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Latha, J.; Hadar, R.; Horwitz, B.A. Role of two G-protein alpha subunits, TgaA and TgaB, in the antagonism of plant pathogens by Trichoderma virens. Appl. Environ. Microbiol. 2004, 70, 542–549. [Google Scholar] [CrossRef]
- Moran-Diez, M.E.; Martinez de Alba, A.E.; Rubio, M.B.; Hermosa, R.; Monte, E. Trichoderma and the plant heritage priming responses. J. Fungi 2021, 7, 318. [Google Scholar] [CrossRef]
- Vizcaino, J.; Cardoza, R.; Hauser, M.; Hermosa, R.; Rey, M.; Llobell, A.; Becker, J.; Gutierrez, S.; Monte, E. ThPTR2, a di/tri-peptide transporter gene from Trichoderma harzianum. Fungal Genet. Biol. 2006, 43, 234–246. [Google Scholar] [CrossRef]
- Tijerino, A.; Cardoza, R.E.; Moraga, J.; Malmierca, M.G.; Vicente, F.; Aleu, J.; Collado, I.G.; Gutierrez, S.; Monte, E.; Hermosa, R. Overexpression of the trichodiene synthase gene trip5 increases trichodermin production and antimicrobial activity in Trichoderma brevicompactum. Fungal Genet. Biol. 2011, 48, 285–296. [Google Scholar] [CrossRef]
- Dixit, P.; Mukherjee, P.K.; Ramachandran, V.; Eapen, S. Glutathone transferase from Trichoderma virens enhances cadmium tolerance without enhancing its accumulation in transgenic Nicotiana tabacum. PLoS ONE 2011, 6, e16360. [Google Scholar] [CrossRef]
- Zhong, Y.H.; Wang, T.H.; Wang, X.L.; Zhang, G.T.; Yu, H.N. Identification and characterization of a novel gene, TrCCD1, and its possible function in hyphal growth and conidiospore development of Trichoderma reesei. Fungal Genet. Biol. 2009, 46, 255–263. [Google Scholar] [CrossRef]
- Migheli, Q. Biodiversity of the Genus Trichoderma and Identification of Marker Genes Involved in the Antagonism Between Trichoderma spp. and Plant Pathogenic Fungi. Ph.D. Thesis, University of Sassari, Sassari, Italy, 2008. [Google Scholar]
- Samolski, I.; Rincon, A.M.; Pinzon, L.M.; Viterbo, A.; Monte, E. The qid74 gene from Trichoderma harzianum has a role in root architecture and plant biofertilization. Microbiology 2012, 158, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, M.; Mukherjee, P.K.; Kale, S.P. cAMP signaling is involved in growth, germination, mycoparasitism and secondary metabolism in Trichoderma virens. Microbiology 2007, 153, 1734–1742. [Google Scholar] [CrossRef] [PubMed]
- Min, S.Y.; Kim, B.-G.; Lee, C.; Hur, H.-G.; Ahn, J.-H. Purification, characterization, and cDNA cloning of xylanase from fungus Trichoderma strain SY. J. Microbiol. Biotechnol. 2002, 12, 89–894. [Google Scholar]
- Guzman-Guzman, P.; Aleman-Duarte, M.I.; Delaye, L.; Herrera-Estrella, A.; Olmedo-Monfil, V. Identification of effector-like proteins in Trichoderma spp. and role of a hydrophobin in the plant-fungus interaction and mycoparasitism. BMC Genet. 2017, 18, 16. [Google Scholar] [CrossRef]
- Atanasova, L.; Gruber, S.; Lichius, A.; Radebner, T.; Abendstein, L.; Munsterkotter, M.; Stralis-Pavese, N.; Labaj, P.P.; Kreil, D.P.; Zeilinger, S. The Gpr1-regulated Sur7 family protein Sfp2 is required for hyphal growth and cell wall stability in the mycoparasite Trichoderma atroviride. Sci. Rep. 2018, 8, 12064. [Google Scholar] [CrossRef]
- Guo, R.; Ji, S.; Wang, Z.; Zhang, H.; Wang, Y.; Liu, Z. Trichoderma asperellum xylanases promote growth and induce resistance in poplar. Microbiol. Res. 2021, 248, 126767. [Google Scholar] [CrossRef]
- Nabi, S.U.; Raja, W.H.; Sharma, A.; Malik, G. Evaluation of different substrates for development of Trichoderma harzianum based stock cultures and their utilization in management of chilli wilt disease. Chem. Sci. Rev. Lett. 2017, 6, 2229–2235. [Google Scholar]
- Mohiddin, F.; Bashir, I.; Padder, S.A.; Hamid, B. Evaluation of different substrates for mass mltiplication of Trichoderma species. J. Pharmacogn. Phytochem. 2017, 6, 563–569. [Google Scholar]
- Andrzejak, R.; Janowska, B. Trichoderma spp. improves flowering, quality, and nutritional status of ornamental plants. Int. J. Mol. Sci. 2022, 23, 15662. [Google Scholar] [CrossRef]
- Guzman-Guzman, P.; Kumar, A.; de los Santos-Villalobos, S.; Parra-Cota, F.I.; Orozco-Mosqueda, M.D.C.; Fadiji, A.E.; Hyder, S.; Babalola, O.O.; Santoyo, G. Trichoderma species: Our best fungal allies in the biocontrol of plant diseases—A review. Plants 2023, 12, 432. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Cao, J.; Zhu, H.; Chen, C.; Lai, Y.; Zhang, Y. Trichodermatides A-D, four new polyketides from Trichoderma sp. XM-3. Fitoterapia 2023, 169, 105584. [Google Scholar] [CrossRef]
- Kassam, R.; Kranti, K.V.V.S.; Yadav, J.; Chatterjee, M.; Chawla, G.; Kundu, A.; Hada, A.; Thokala, P.D.; Shukla, L.; Mishra, J.; et al. Exploration of rhizsophere-dwelling nematophagous Trichoderma spp. using novel bait technique with root-knot nematode Meloidogyne incognita. Biol. Control 2023, 186, 105327. [Google Scholar] [CrossRef]
- Chan, M.E.; Tan, J.Y.; Lee, Y.Y.; Lee, D.; Fong, Y.K.; Mutwil, M.; Wong, J.Y.; Hong, Y. Locally isolated Trichoderma harzianum species have broad spectrum biocontrol activities against the wood rot fungal species through both volatile inhibition and mycoparasitism. J. Fungi 2023, 9, 675. [Google Scholar] [CrossRef]
- De Oliveira, C.M.; Oshiquiri, L.H.; Almeida, N.O.; Steindorf, A.S.; Rocha, M.R.D.; Georg, R.C.; Ulhoa, C.J. Trichoderma harzianum transcriptome in response to the nematode Pratylenchus branchyurus. Biol. Control 2023, 183, 105245. [Google Scholar] [CrossRef]
- Wu, J.-L.; Yu, Y.-H.; Yao, H.-Z.; Zhao, X.; Yuan, T.; Huang, Y.-H. Trichodermic acids from an endophytic Trichoderma sp. and their antifungal activity against the phytopathogen Botrytis cinerea. Phytochem. Lett. 2023, 56, 24–29. [Google Scholar] [CrossRef]
- Tamandegani, P.R.; Sharifnabi, B.; Massah, A.; Zahravi, M. Induced reprogramming of oxidative stress responses in cucumber by Trichoderma asperellum (Iran 3062C) enhances defense against cucumber mosaic virus. Biol. Control 2021, 164, 104779. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Z.; Lu, B.; Quan, X.; Zhao, G.; Zhang, Z.; Liu, W.; Tian, Y. Antagonistic potential of Trichoderma as a biocontrol agent Sclerotinia asari. Front. Microbiol. 2022, 13, 997050. [Google Scholar] [CrossRef]
- Bansal, R.; Sahoo, S.A.; Barvkar, V.T.; Srivastava, A.K.; Mukherjee, P.K. Trichoderma virens exerts herbicidal effect on Arabidopsis thaliana via modulation of amino acid metabolism. Plant Sci. 2023, 332, 111702. [Google Scholar] [CrossRef]
- McLeod, A.; Labuschagne, N.; Kotze, J. Evaluation of Trichoderma for biological control of avocado root rot in bark medium artificially infested with Phytophthora cinnamomi. S. Afr. Avocado Grow. Assoc. Yearb. 1995, 18, 32–37. [Google Scholar]
- Lopez-Lopez, M.E.; Del-Toro-Sanchez, C.L.; Gutierrez-Lomeli, M.; Ochoa-Ascencio, S.; Aguilar-Lopez, J.A.; Robles-Garcia, M.A.; Plascencia-Jatomea, M.; Bernal-Mercado, M.A.; Martinez-Cruz, O.; Avila-Novoa, M.G.; et al. Isolation and characterization of Trichoderma spp. for antagonistic activity against Avocado (Persea americana Mill.) fruit pathogens. Horticulturae 2022, 8, 714. [Google Scholar] [CrossRef]
- Meki, S.; Ahmed, S.; Sakhuja, P.K. Control of chickpea wilt (Fusarium oxysporum f.s.p. ciceris) using Trichoderma spp. in Ehtiopia. Arch. Phytopathol. Plant Prot. 2011, 44, 432–440. [Google Scholar] [CrossRef]
- Rawat, J.; Sanwal, P.; Saxena, J.; Prasad, R. Exploring the biochar as a suitable carrier for a bioinoculant Aspergillus niger K7 and its consequence on Eleusine coracana in field studies. J. Agric. Food Res. 2023, 14, 100825. [Google Scholar] [CrossRef]
- Olabiyi, T.I.; Ojo, O.J.; Adewuyi, B.O.; Moral, M.T. Impact assessment of need compost and Trichoderma harzianum solution in the control of root knot nematode disease on cowpea. Cogent Food Agric. 2016, 2, 1207843. [Google Scholar] [CrossRef]
- Gupta, R.; Singh, M.; Khan, B.R. Photosynthetic electron transport rate and root dynamics of finger millet in response to Trichoderma harzianum. Plant Signal Behav. 2022, 17, 2146373. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, A.; Ashraf, S.; Musheer, N. The antagonistic effect of locally isolated Trichoderma spp. against dry root rot of mungbean. Arch. Phytopathol. Plant Prot. 2021, 54, 1204–1210. [Google Scholar] [CrossRef]
- Barbosa, J.Z.; Hungria, M.; Prior, S.A.; Moura, M.C.; Poggere, G.; Motta, A.C.V. Improving yield and health of legume crops via co-inoculation with rhizobia and Trichoderma: A global meta-analysis. Appl. Soil Ecol. 2022, 176, 104493. [Google Scholar] [CrossRef]
- Pandey, V.; Ansari, M.W.; Tula, S.; Yadav, S.; Sahoo, R.K.; Shukla, N.; Bains, G.; Badal, S.; Chandra, S.; Gaur, A.K.; et al. Does-dependent response of Trichoderma harzianum in improving drought tolerance in rice genotypes. Plants 2016, 243, 1251–1264. [Google Scholar] [CrossRef]
- Chowdhury, M.R.; Ahmed, S.F.; Khalid, B.; Bony, Z.F.; Asha, J.F.; Bhuiyan, M.K.A. Biocontrol efficiency of microencapsulated Trichoderma harzianum coupled with organic additives against potato stem rot caused by Sclerotium rolfsii. Plant Stress 2023, 9, 100181. [Google Scholar] [CrossRef]
- Limdolthamand, S.; Songkumarn, P.; Suwannarat, S.; Jantasorn, A.; Dethoup, T. Bioconrol efficacy of endophytic Trichoderma spp. in fresh and dry powder formulations in controlling northern corn leaf blight in sweet corn. Biol. Control 2023, 181, 105217. [Google Scholar] [CrossRef]
- Manzar, N.; Singh, Y.; Kashyap, A.S.; Sahu, P.K.; Rajawat, M.V.S.; Bhowmik, A.; Sharma, P.K.; Saxena, A.K. Biocontrol potential of native Trichoderma spp. against anthracnose of great millet (Sorghum bicolour L.) from Tarai and hill regions of India. Biol. Control 2021, 152, 104474. [Google Scholar] [CrossRef]
- Diaz-Gutierrez, C.; Arroyave, C.; Llugany, M.; Pschenrieder, C.; Martos, S.; Pelaez, C. Trichoderma asperellum as a preventive and curative agent to control Fusarium wilt in Stevia rebaudiana. Biol. Control 2021, 155, 104537. [Google Scholar] [CrossRef]
- Wang, H.; Tang, W.; Mao, Y.; Ma, S.; Chen, X.; Shen, X.; Yin, C.; Mao, Z. Isolation of Trichoderma virens 6PS-2 and its effects on Fusarium prolideratum f. sp. Malus domestica MR5 related to apple replant disease (ARD) in China. Hortic. Plants J. 2020, 10, 1291–1308. [Google Scholar] [CrossRef]
- Kishmoto, K.; Matsui, K.; Ozawa, R.; Takabayashi, J. Volatile 1-Octen-3-Ol induced a defensive response in Arabidopsis thaliana. J. Gen. Plant Pathol. 2007, 73, 35–37. [Google Scholar] [CrossRef]
- Contretas-Cornejo, H.A.; Macias-Rodriguez, L.; Herrera-Estrella, A.; Lopez-Bucio, J. The 4-phosphopantetheinyl transferase of Trichoderma virens plays a role in plant protection against Botrytis cinerea through volatine organic compounds emission. Plant Soil 2014, 379, 261–274. [Google Scholar] [CrossRef]
- Lopez, A.C.; Alvarenga, A.E.; Zapata, P.D.; Luna, M.F.; Villalba, L.L. Trichoderma spp. from misiones, Argentina: Effective fungi to promote plant growth of the regional crop Ilex paraguariensis St. Hil. Mycology 2019, 10, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Ruano-Rosa, D.; Arjona-Girona, I.; Lopez-Herrera, C.J.L. Integrated control of avocado white root rot combining low concentrations of fluazinam and Trichoderma spp. Crop Prot. 2018, 112, 363–370. [Google Scholar] [CrossRef]
- Muthukathan, G.; Mukherjee, P.; Salaskar, D.; Pachauri, S.; Tak, H.; Ganapathi, T.R.; Mukherjee, P.K. Secretome of Trichoderma virens induced by banana roots-identification of novel fungal proteins for enhancing plant defence. Physiol. Mol. Plant Pathol. 2020, 110, 101476. [Google Scholar] [CrossRef]
- Almeida, N.O.; Oliveira, C.M.D.; Ulhoa, C.J.; Cortes, M.V.D.C.B.; Junior, M.L.; Rocha, M.R.D. Trichoderma harzianum and Trichoderma asperellum are potential biocontrol agents of Meloidogyne javanica in banana cv. Grande Naine. Biol. Control 2022, 175, 105054. [Google Scholar] [CrossRef]
- Sano, L.; Oliveira, L.L.B.D.; Leao, M.D.M.; Santos, J.E.D.A.D.; Medeiros, S.C.D.; Schneider, F.; Sousa, A.B.O.D.; Taniguchi, C.A.K.; Muniz, C.R.; Grangeiro, T.B.; et al. Trichoderma longibrachiatum as a biostimulant of micropropagated banana seedlings under acclimatization. Plant Physiol. Biochem. 2022, 190, 184–192. [Google Scholar] [CrossRef]
- Moreira, F.M.; Cairo, P.A.R.; Borges, A.L.; Silva, L.D.D.; Haddad, F. Investigating the ideal mixture of soil and organic compound with Bacillus sp. and Trichoderma asperellum inoculations for optimal growth and nutrient content of banana seedlings. S. Afr. J. Bot. 2021, 137, 249–256. [Google Scholar] [CrossRef]
- Costa, A.C.D.; Miranda, R.F.D.; Costa, F.A.; Ulhoa, C.J. Potential of Trichoderma piluliferum as a biocontrol agent of Colletotrichum musae in banana fruits. Biocatal. Agric. Biotechnol. 2021, 34, 102028. [Google Scholar] [CrossRef]
- Mayo, S.; Gutierrez, S.; Malmierca, M.G.; Lorenzana, A.; Campelo, M.P.; Hermosa, R.; Casquero, P.A. Influence of Rhizoctonia solani and Trichoderma spp. in growth of bean (Phaseolus vulgaris L.) and in the induction of plant defense-related genes. Front. Plant Sci. 2015, 6, 685. [Google Scholar] [CrossRef] [PubMed]
- Eslahi, N.; Kowsari, M.; Motallebi, M.; Zamani, M.R.; Moghadasi, Z. Influence of recombinant Trichoderma strains on growth of bean (Phaseolus vulgaris L.) by increased root colonization and induction of root growth related genes. Sci. Hortic. 2020, 261, 108932. [Google Scholar] [CrossRef]
- Silva, F.D.A.; Vieira, V.D.O.; Silva, R.C.D.; Pinheiro, D.G.; Soares, M.A. Introduction of Trichoderma spp. biocontrol strains against Sclerotinia scleotiorum (Lib.) de Bary change soil microbial community composition in common bean (Phaseolus vulgaris L.) cultivation. Biol. Control 2021, 163, 104755. [Google Scholar] [CrossRef]
- Ghoneem, K.M.; Al-Askar, A.A.; Saber, W.-E.I.A. A simple formula of the endophytic Trichoderma viride, a case study for the management of Rhizoctonia solani on the common bean. Life 2023, 13, 1358. [Google Scholar] [CrossRef] [PubMed]
- Abdelmoteleb, A.; Gonzalez-Mendoza, D.; Zayed, O. Cell-free culture filtrate of Trichoderma longibrachiatum AD-1 as alternative approach to control Fusarium solani and induce defense response Phaseolus vulgaris L. plants. Rhizosphere 2023, 25, 100648. [Google Scholar] [CrossRef]
- Bedine, M.A.B.; Iacomi, B.; Tchameni, S.N.; Sameza, M.L.; Fekam, F.B. Harnessing the phosphate-solubilizing ability of Trichoderma strains to improve plant growth, phosphorus uptake and photosynthetic pigment contents in common bean (Phaseolus vulgaris). Biocatal. Agric. Biotechnol. 2022, 45, 102510. [Google Scholar] [CrossRef]
- Boat, M.A.B.; Sameza, M.L.; Iacomi, B.; Tchameni, S.N.; Boyom, F.F. Screening, identification and evaluation of Trichoderma spp. for biocontrol potential of common bean damping-off pathogens. Biocontrol Sci. Technol. 2020, 30, 228–242. [Google Scholar] [CrossRef]
- Silva, F.D.A.; Vierira, V.D.O.; Carrenho, R.; Rodrigues, V.B.; Junior, M.L.; Silva, G.F.D.; Soares, M.A. Influence of the biocontrol agents Trichoderma spp. on the structure and functionality of the edaphic microbial community in common bean cultivars (Phaseolus vulgaris L.) inoculated with Sclerotinia sclerotiorum (Lib.) de Bary. Appl. Soil Ecol. 2021, 168, 104190. [Google Scholar] [CrossRef]
- Umadevi, P.; Anandaraj, M.; Srivastav, V.; Benjamin, S. Trichoderma harzianum MTCC 5179 impacts the population and functional dynamics of microbial community in the rhzisophere of black pepper (Piper nigrum L.). Brazil J. Microbiol. 2018, 49, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, D.A.; Makandar, R. Delineating host responses induced by Trichoderma in castor through comparative transcriptome analysis. Rhizosphere 2023, 27, 100745. [Google Scholar] [CrossRef]
- Prasad, R.D.; Chandrika, K.S.V.P.; Godbole, V. A novel chitosan biopolymer based Trichoderma delivery system: Storage stability, persistence and bio efficacy against seed and soil borne diseases of oilseed crops. Microbiol. Res. 2020, 237, 126487. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.; Das, A.; Kaman, P.K.; Kushwaha, K.P.S.; Bisht, A.S.; Narzary, P.R.; Roy, B. Comparative evaluation of physiological and biochemical chages in black pepper plants infected by Colletotrichum siamense in response to Trichoderma viride and T. harzianum application. Physiol. Mol. Plant Pathol. 2023, 128, 102170. [Google Scholar] [CrossRef]
- Tchameni, S.N.; Ngonkeu, M.E.L.; Begoude, B.A.D.; Nana, L.W.; Fokom, R.; Owona, A.D.; Mbarga, J.B.; Tchana, T.; Tondje, P.R.; Etoa, F.X.; et al. Effect of Trichoderma asperellum and arbuscular mycorrhizal fungi on cacao growth and resistance against black pod disease. Crop Prot. 2011, 30, 1321–1327. [Google Scholar] [CrossRef]
- Mbarga, J.B.; Begoude, B.A.D.; Ambang, Z.; Meboma, M.; Kuate, J.; Ewbank, W.; Hoopen, G.M.T. Field testing an oil-based Trichoderma asperellum formulation for the biological control of cacao black pod disease caused by Phytophthora megakarya. Crop Prot. 2020, 132, 105134. [Google Scholar] [CrossRef]
- Mbargo, J.B.; Begoude, B.A.D.; Ambang, Z.; Meboma, M.; Kuate, J.; Schiffers, B.; Ewbank, W.; Dedieu, L.; Hoopen, G.M.T. A new oil-based formulation of Trichoderma asperellum for the biological control of cacao black pod disease caused by Phytophthora megakarya. Biol. Control 2014, 77, 15–22. [Google Scholar] [CrossRef]
- Motlagh, M.R.S.; Abolghasemi, M. The effect of Trichoderma spp. isolates on some morphological traits of canola inoculated with Sclerotinia sclerotiorum and evaluation of their efficacy in biological control of pathogen. J. Saudi Soc. Agric. Sci. 2022, 21, 217–231. [Google Scholar] [CrossRef]
- Da Silva, J.S.A.D.; Medeiros, E.V.D.; Costa, D.P.D.; Souza, C.A.F.D.; Oliveira, J.B.D.; Franca, R.F.D.; Souza-Motta, C.M.; Lima, J.R.D.S.; Hammecker, C. Biochar and Trichoderma aureoviride URM 5158 as alternatives for the management of cassava root rot. Appl. Soil Ecol. 2022, 172, 104353. [Google Scholar] [CrossRef]
- Pradham, D.A.; Bagagoni, P.; Slathia, S.; Prasad, R.D. Characterization of Trichoderma strains for novel species-specific markers by multiplex PCR and antagonistic property against Alternaria ricini in castor (Ricinus communis L.). Biocatal. Agric. Biotechnol. 2023, 54, 102945. [Google Scholar] [CrossRef]
- Pradhan, D.A.; Bagagoni, P.; Makandar, R. Assessing rhizosphere Trichoderma asperellum strains for root colonizing and antagonistic competencies against Fusarium wilt through molecular and biochemical responses in castor. Biol. Control 2023, 184, 105280. [Google Scholar] [CrossRef]
- Dubey, S.C.; Tripathi, A.; Bhavani, R.; Singh, B. Evaluation of seed dressing and soil application formulation of Trichoderma species for integrated management of dry root rot of chickpea. Biocontrol. Sci. Technol. 2011, 21, 93–100. [Google Scholar] [CrossRef]
- Begum, M.F.; Rahman, M.A.; Firoz Alam, M. Biological control of Alternaria fruit rot of chili by Trichoderma species under field conditions. Mycobiology 2010, 38, 113–117. [Google Scholar] [CrossRef]
- Ji, S.; Liu, Z.; Liu, B.; Wang, Y.; Wang, J. The effect of Trichoderma biofertilizer on the quality of flowering Chinese cabbage and the soil environment. Sci. Hortic. 2020, 262, 109069. [Google Scholar] [CrossRef]
- De Lima, F.B.; Felix, C.; Osorio, N.; Alves, A.; Vitorino, R.; Domingues, P.; da Silva Ribeiro, R.T.; Esteves, A.C. Trichoderma harzianum T1A constitutively secretes proteins involved in the biological control of Guignardia citricarpa. Biol. Control 2017, 106, 99–109. [Google Scholar] [CrossRef]
- Bae, H.; Sicher, R.C.; Kim, M.S.; Kim, S.H.; Strem, M.D.; Melnick, R.L.; Bailey, B.A. The beneficial endophyte Trichoderma hamatum isolate DIS 219b promotes growth and delays the onset of the drought response in Theobroma cacao. J. Exp. Bot. 2009, 60, 3279–3295. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; An, X.; Lv, Y.; Khan, R.A.A.; Xue, M.; Chen, J.; Liu, T. Trichoderma asperellum GD040 upregulates defense-related genes and reduces lesion size in Coffea canephora leaves inoculated with Colletotrichum cairnsense. Biol. Control 2023, 181, 105213. [Google Scholar] [CrossRef]
- Howell, C.R.; Hanson, L.E.; Stipanovic, R.D.; Puckhaber, L.S. Induction of terpenoid synthesis in cotton roots and control of Rhizoctonia solani by seed treatment with Trichoderma virens. Phytopathology 2000, 90, 248–252. [Google Scholar] [CrossRef]
- Howell, C.R. Cotton seedling preemergence damping-off incited by Rhizopus oryzae and Pythium spp. and its biological control with Trichoderma spp. Phytopathology 2002, 92, 177–180. [Google Scholar] [CrossRef]
- Howell, C.R. Understanding the mechanisms employed by Trichoderma virens to effect biological control of cotton diseases. Phytopathology 2006, 96, 178–180. [Google Scholar] [CrossRef]
- Larran, S.; Simon, M.R.; Santamarina, M.P.; Caselles, J.R.; Consolo, V.F.; Perello, A. Endophytic Trichoderma strains increase soya bean growth and promote charcoal rot control. J. Saudi Soc. Agric. Sci. 2023, 22, 395–406. [Google Scholar] [CrossRef]
- Nakkeeran, S.; Renukadevi, P.; Marimuthu, T. Antagonistic potentiality of Trichoderma viride and assessment of its efficacy for the management of cotton root rot. Arch. Phytopathil. Plant Prot. 2005, 38, 209–225. [Google Scholar] [CrossRef]
- Gajera, H.P.; Hirpara, D.G.; Savaliya, D.D.; Golakiya, B.A. Extracellular metabolomics of Trichoderma biocontroller for antifungal action to restrain Rhizoctonia solani Kuhn in cotton. Physiol. Mol. Plant Pathol. 2020, 112, 101547. [Google Scholar] [CrossRef]
- Oliveira, C.M.D.; Almeida, N.O.; Cortes, M.V.D.C.B.; Junior, M.L.; Rocha, M.R.D.; Ulhoa, C.J. Biological control of Pratylenchus brachyurus with isolates of Trichoderma spp. on soybean. Biol. Control 2021, 152, 104425. [Google Scholar] [CrossRef]
- Szczech, M.; Nawrocka, J.; Felczynski, K.; Malolepsza, U.; Sobolewski, J.; Kowalska, B.; Maciorowski, R.; Jas, K.; Kancelista, A. Trichoderma atroviride TRS25 isolate reduces downy mildew and induces systemic defence responses in cucumber in field conditions. Sci. Hortic. 2017, 224, 17–26. [Google Scholar] [CrossRef]
- Nawrocka, J.; Malolepsza, U.; Szymczak, K.; Szczech, M. Involvement of metabolic components, volatile compounds, PR proteins and mechanical strengthening in multilayer protection of cucumber plants against Rhizoctonia solani activated by Trichoderma atroviride TRS25. Protoplasma 2018, 255, 359–373. [Google Scholar] [CrossRef]
- Yuan, M.; Huang, Y.; Ge, W.; Jia, Z.; Song, S.; Zhang, L.; Huang, Y. Involvement of jasmonic acid, ethylene and salicylic acid signaling pathways behind the systemic resistance induced by Trichoderma longibrachiatum H9 in cucumber. BMC Genom. 2019, 20, 144. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, Y. Effects of phosphate solubilization and phytohormone production of Trichoderma asperellum Q1 on promoting cucumber growth under salt stress. J. Integr. Agr. 2015, 14, 1588–1597. [Google Scholar] [CrossRef]
- Brotman, Y.; Landau, U.; Cuadros-Inostroza, A.; Takayuki, T.; Fernie, A.R.; Chet, I.; Viterbo, A.; Willmitzer, L. Trichoderma-plant root colonization: Escaping early plant defense responses and activation of the antioxidant machinery for saline stress tolerance. PLOS Pathog. 2013, 9, e1003221. [Google Scholar] [CrossRef]
- Wang, R.; Yu, X.; Yi, Y.; Norvienyeku, J.; Khan, R.A.A.; Zhang, M.; Ren, S.; Chen, J.; Liu, T. Biocontrol of cucumber Fuarium wilt by Trichoderma asperellum FJ035 dependent on antagonism and spatiotemporal competition with Fusarium oxysporum. Biol. Control 2023, 186, 105334. [Google Scholar] [CrossRef]
- Li, P.; Xie, D.; Chen, H.; Qiu, Y.; Zhang, X.; Zhang, S.; Wang, L.; Lin, H.; Li, X.; Liu, K. Secondary metabolites from marine derived fungus Penicillium chrysogenum Y19-1 with proangiogenic and antithrombotic activities. Biochem. System. Ecol. 2023, 107, 104625. [Google Scholar] [CrossRef]
- Ulrich, A.; Lerin, L.A.; Camargo, A.F.; Scapini, T.; Diering, N.L.; Bonafin, F.; Gasparetto, I.G.; Confortin, T.C.; Sansonovicz, P.F.; Fabaian, R.L.; et al. Alternative bioherbicide based on Trichoderma koningiopsis: Enzymatic characterization and its effect on cucumber plants and soil organism. Biocatal. Agric. Biotechnol. 2021, 36, 102127. [Google Scholar] [CrossRef]
- Intana, W.; Kumla, J.; Suwannarach, N.; Sunpapao, A. Biological control potential of a soil fungus Trichoderma asperellum K1-02 against Neoscytalidium dimidiatum causing stem canker of dragon fruit. Physiol. Mol. Plant Pathol. 2023, 128, 102151. [Google Scholar] [CrossRef]
- Admasu, W.; Sintayehu, A.; Gezahgne, A.; Terefework, Z. In vitro bioefficacy of Trichoderma species against two Botryosphaeriaceae fungi causing Eucalytpus stem canker disease in Ethiopia. J. Nat. Pest Res. 2023, 4, 100037. [Google Scholar] [CrossRef]
- Mbazia, A.; Youssef, N.O.B.; Kharrat, M. Tunisian isolates of Trichoderma spp. and Bacillus subtilis can control Botrytis fabae on faba bean. Biocontrol Sci. Technol. 2016, 26, 915–927. [Google Scholar] [CrossRef]
- Neme, A.; Leta, A.; Yones, A.M.; Tahir, M. Seedborne mycoflora of faba bean (Vicia fabae L.) and evaluation of plant extract and Trichoderma species against mycelium growth of selected fungi. Heliyon 2023, 9, e17291. [Google Scholar] [CrossRef]
- Gupta, V.P.; Sharma, D.D.; Mahasevaswamy, H.; Chandrashekar, D.S. Trichoderma pseudokoningii for hastening the decomposition of various sericultural wastes and impact of enriched composts on disease suppression in mulberry (Morus spp.). Arch. Phytopathol. Plant Prot. 2009, 7, 603–609. [Google Scholar] [CrossRef]
- Brito, F.D.S.; Costa, D.P.D.; Souza, C.A.F.D.; Almeida, D.T.D.R.G.F.D.; Leite, I.C.H.D.L.; Goncalves, E.P.; Medeiros, E.V.D. Selection and control efficacy of Trichoderma spp. against Fusarium solani and Lasiodiplodia theobromae causing root rot in forage cactus. Physiol. Mol. Plant Pathol. 2022, 122, 101900. [Google Scholar] [CrossRef]
- Mastan, A.; Rane, D.; Dastager, S.G.; Vivek Babu, C.S. Molecular insights of fungal endophyte co-inoculation with Trichoderma viride for the augmentation of forskolin biosynthesis in Coleus forskohlii. Phytochemistry 2021, 184, 112654. [Google Scholar] [CrossRef]
- Kupper, V.; Kortekamp, A.; Steiner, U. Combining Trichoderma koningiopsis and chitosan as a synergistic biocontrol and biostimulating complex to reduce copper rates for downy mildew control on grapevine. Biol. Control 2023, 185, 105293. [Google Scholar] [CrossRef]
- El-Mohamedy, R.; Ziedan, E.H.; Abdalla, A.M. Biological soil treatment with Trichoderma harzianum to control root rot disease grapevine (Vitis vinifera L.) in newly reclaimed lands in Bobaria province. Arch. Phytopathol. Plant Prot. 2010, 43, 73–87. [Google Scholar] [CrossRef]
- Csoto, A.; Kovacs, C.; Pal, K.; Nagy, A.; Peles, F.; Fekete, E.; Karaffa, L.; Kubicek, C.P.; Sandor, E. The biocontrol potential of endophytic Trichoderma fungi isolated from Hungarian grapevines, part II, grapevine stimulation. Pathogens 2023, 12, 2. [Google Scholar] [CrossRef]
- Song, H.; Asghari, M.; Zahedipour-Sheshglani, P.; Alizadeh, M.; Qian, S.; Diao, E. Modeling and optimizing the effects of Trichoderma on quality, decay extension rate and phytochemical compounds of Thompson seedless table grapes by the use of response surface methodology. Eur. J. Agron. 2023, 144, 126758. [Google Scholar] [CrossRef]
- Gajera, H.P.; Savaliya, D.D.; Patel, S.V.; Golakiya, B.A. Trichoderma viride induces pathogenesis related defense response against rot pathogen infection in groundnut (Arachis hypogaea L.). Infect Gene Evol. 2015, 34, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Gajera, H.P.; Katakpara, Z.A.; Golakiya, B.A. Antioxidant defense response induced by Trichoderma viride against Aspergillus niger Van Tieghem causing collar rot in groundnut (Arachis hypogaea L.). Microbiol. Pathogen. 2016, 91, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Ayyandurai, M.; Akila, R.; Manonmani, K.; Harish, S.; Mini, M.L.; Vellaikumar, S. Deciphering the mechanism of Trichoderma spp. consortia possessing volatile organic compounds and antifungal metabolites in the suppression of Sclerotium rolfsii in groundnut. Physiol. Mol. Plant Pathol. 2023, 125, 102005. [Google Scholar] [CrossRef]
- Hoa, P.T.B.; Tue, N.H.; Trang, H.T.Q.; Thu, H.A.; Nhung, L.N.H.; Luong, N.N.; Huy, N.X.; Tien, N.Q.D.; Loc, N.H. Enhancement of resistance against fungal pathogens in peanut (Arachis hypogaea L.) cultivar L14 by heterologous expression of gene encoding chitinase 42 kDa from Trichoderma asperellum SH16. S. Afr. J. Bot. 2023, 160, 673–681. [Google Scholar] [CrossRef]
- Boukaew, S.; Petlamul, W.; Srinuanpan, S.; Nooprom, K.; Zhang, Z. Heat stability of Trichoderma asperelloides SKRU-01 culture filtrates: Potential applications for controlling fungal spoilage and AFB1 production in peanuts. Int. J. Food Microbiol. 2024, 409, 110477. [Google Scholar] [CrossRef]
- Erazo, J.G.; Palacios, S.A.; Pastor, N.; Giordano, F.D.; Rovera, M.; Reynoso, M.M.; Venisse, J.S.; Torres, A.M. Biocontrol mechanisms of Trichoderma harzianum ITEM 3636 against peanut brown root rot caused by Fusarium solani RC 386. Biol. Control 2021, 164, 104774. [Google Scholar] [CrossRef]
- Yan, D.; Cai, N.; Nong, X.; Wang, G.; Wang, Q.; Ullah, H.; Tu, X.; Zhang, Z. Transcriptomic differences in response to Metarhizium anisopliae and Trichoderma harzianum uncovers major regulative genes and pathways for establishment of beneficial relationship in peanut. Biol. Control 2022, 172, 104964. [Google Scholar] [CrossRef]
- Song, X.; Jin, J.; Yin, H.; Wang, T.; Zong, H.; Wang, F.; Liu, J.; Huang, X.; Wang, B.; Chai, C.; et al. Trichoderma viride F7 improves peanut performance while remedying cadmium-contaminated soil with microplastics. Pedosphere 2023, 34, 1–20. [Google Scholar] [CrossRef]
- Sennoi, R.; Singkham, N.; Jogloy, S.; Boonlue, S.; Saksirirat, W.; Kesmala, T.; Patanothai, A. Biological control of southern stem rot caused by Sclerotium rolfsii using Trichoderma harzianum and arbuscular mycorrhizal fungi on Jerusalem artichoke (Helianthus tuberosus L.). Crop Prot. 2013, 54, 148–153. [Google Scholar] [CrossRef]
- Silva, L.R.D.; Valadares-Inglis, M.C.; Peixoto, G.H.S.; Luccas, B.E.G.D.; Muniz, P.H.P.C.; Magalhaes, D.M.; Moraes, M.C.B.; Mello, S.C.M.D. Volatile organic compounds emitted by Trichoderma azevedoi promote the growth of lettuce plants and delay the symptoms of white mold. Biol. Control 2021, 152, 104447. [Google Scholar] [CrossRef]
- Wonglom, P.; Ito, S.-I.; Supapao, A. Volatile organic compounds emitted from endophytic fungus Trichoderma asperellum T1 mediate antifungal activity, defense response and promote plant growth in lettuce (Lactuca sativa). Fungal Ecol. 2020, 43, 100867. [Google Scholar] [CrossRef]
- Baiyee, B.; Pornsuriya, C.; Ito, S.-I.; Sunpapao, A. Trichoderma spirale T76-1 displays biocontrol activity against leaf spot on lettuce (Lactuca sativa L.) caused by Corynespora cassiicola or Curvularia aeria. Biol. Control 2019, 129, 195–200. [Google Scholar] [CrossRef]
- Caporale, A.G.; Sommella, A.; Lorito, M.; Lombardi, N.; Azam, S.M.G.G.; Pigna, M.; Ruocco, M. Trichoderma spp. alleviate phytotoxicity in lettuce plants (Lactuca sativa L.) irrigated with arsenic-contaminated water. J. Plant Physiol. 2014, 171, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
- Santos-Villalobos, S.; Guzman-Ortiz, D.A.; Gomez-Lim, M.A.; Delano-Frier, J.P.; De-Folter, S.; Sanchez-Garcia, P.; Pena-Cabriales, J.J. Potential use of Trichoderma asperellum (Samuels, Liechfeldt et Nirenberg) T8a as a biological control agent against anthracnose in mango (Mangifera indica L.). Biol. Control 2013, 64, 37–44. [Google Scholar] [CrossRef]
- Zhan, X.; Khan, R.A.A.; Zhang, J.; Chen, J.; Yin, Y.; Tang, Z.; Wang, R.; Lu, B.; Liu, T. Control of postharvest stem-end rot on mango by antifungal metabolites of Trichoderma pinnatum LS2029-3. Sci. Hortic. 2023, 310, 111696. [Google Scholar] [CrossRef]
- Akladious, S.A.; Abbas, S.M. Application of Trichoderma harzianum T22 as a biofertilizer potential in maize growth. J. Plant Nutr. 2014, 37, 30–49. [Google Scholar] [CrossRef]
- Yassin, M.T.; Mostafa, A.A.-F.; Al-Askar, A.A. In vitro antagonistic activity of Trichoderma spp. against fungal pathogens causing black point disease of wheat. J. Taibah Univ. Sci. 2022, 16, 57–65. [Google Scholar] [CrossRef]
- Degani, O.; Dor, S. Trichoderma biological control to protect sensitive maize hybrids against Late wilt disease in the field. J. Fungi 2021, 7, 315. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Luo, X. Trichoderma koningiopsis controls Fusarium oxysporum causing damping-off in Pinus massoniana seedlings by regulating active oxygen metabolism osmotic potential, and the rhizosphere microbiome. Biol. Control 2020, 150, 104352. [Google Scholar] [CrossRef]
- Saravanakumar, K.; Wand, M.H. Isolation and molecular identification of Trichoderma species from wetland soil and their antagonistic activity against phytopathogens. Physiol. Mol. Plant Pathol. 2020, 109, 101458. [Google Scholar] [CrossRef]
- Karuppiah, V.; Li, Y.; Sun, J.; Vallikkannu, M.; Chen, J. Vel1 regulates the growth of Trichoderma atroviride during co-cultivation with Bacillus amyloliquefaciens and is essential for wheat root rot control. Biol. Control 2020, 151, 104374. [Google Scholar] [CrossRef]
- Galletti, S.; Paris, R.; Cianchetta, S. Selected isolates of Trichoderma gamsii induce different pathways of systemic resistance in maize upon Fuarium verticillioides challenge. Micriobiol. Res. 2020, 233, 126406. [Google Scholar] [CrossRef]
- Hasanloo, T.; Kowsari, M.; Naraghi, S.M.; Bagheri, O. Study of different Trichoderma strains on growth characteristics and silymarin accumulation of milk thistle plant. J. Plant Interact. 2010, 5, 45–49. [Google Scholar] [CrossRef]
- Hohmann, P.; Jones, E.E.; Hill, R.A.; Stewart, A. Understanding Trichoderma in the root system of Pinus radiata: Associations between rhizosphere colonisation and growth promotion for commercially growth seedlings. Fungal Biol. 2011, 115, 759–767. [Google Scholar] [CrossRef]
- Hohmann, P.; Jones, E.E.; Hill, R.A.; Stewart, A. Ecological studies of the bio-inoculant Trichoderma hamatum LU592 in the root system of Pinus radiata. FEMS Microbiol. Ecol. 2012, 80, 709–721. [Google Scholar] [CrossRef]
- Ruangwong, Q.-U.; Wonglom, P.; Phoka, N.; Suwannarach, N.; Lumyong, S.; Ito, S.-I.; Sunpapao, A. Biological control activity of Trichoderma asperelloides PSU-P1 against gummy stem blight in muskmelon (Cucumis melo). Physiol. Mol. Plant Pathol. 2021, 115, 101663. [Google Scholar] [CrossRef]
- Khare, A.; Singh, B.; Upadhyay, R. Biological control of Pythium aphanidermatum causing damping-off of mustard by mutants of Trichoderma viride 1433. J. Agric. Technol. 2010, 6, 231–243. [Google Scholar]
- Samlikamnoed, P.; Anothai, J.; Chairin, T. Defense-related enzyme production in oil palm seedlings against basal stem rot pathogen Ganoderma boninense and its biological control by Trichoderma asperellun. Physiol. Mol. Plant Pathol. 2023, 128, 102154. [Google Scholar] [CrossRef]
- Ben Amira, M.; Lopez, D.; Triki Mohamed, A.; Khouaja, A.; Chaar, H.; Fumanal, B.; Gousset-Dupont, A.; Bonhomme, L.; Label, P.; Goupil, P.; et al. Beneficial effect of Trichoderma harzianum strain Ths97 in biocontrolling Fusarium solani causal agent of root rot disease in olive trees. Biol. Control 2017, 110, 70–78. [Google Scholar] [CrossRef]
- Metwally, R.A.; Soliman, S.A.; Laref, A.A.H.A.; Abdelhameed, R.E. The individual and interactive role of arbuscular mycorrhizal fungi and Trichoderma viride on growth, protein content, amino acids fraction, and phosphatases enzyme activities of onion plant amended with fish waste. Ecotoxicol. Environ. Saf. 2021, 214, 112072. [Google Scholar] [CrossRef] [PubMed]
- Vargas, J.T.; Rodriguez-Monroy, M.; Meyer, M.L.; Montes-Belmont, R.; Sepulveda-Jimenez, G. Trichoderma asperellum ameliorates phytotoxic effects of copper in onion (Allium cepa L.). Environ. Exp. Bot. 2017, 136, 85–93. [Google Scholar] [CrossRef]
- Camacho-Luna, V.; Pizar-Quiroz, A.M.; Rodriguez-Hernandez, A.A.; Rodriguez-Monroy, M.; Sepulveda-Jimenez, G. Trichodera longibrachiatum, a biological control agent of Sclerotium cepivorum on onion plants under salt stress. Biol. Control 2023, 180, 105168. [Google Scholar] [CrossRef]
- Da Silva, L.R.; Rodrigues, L.L.D.B.; Zazaroni, A.B.; Castro, B.S.D.; Sifuentes, D.N.; Botelho, A.S.; Moraes, M.C.B.; Mello, S.C.M.D. Sclerotium rolfssi mycelial profile analysis by MALDI-TOF related to biological control, volatile organic compounds diversity and onion growth promotion, as influenced by Trichoderma spp. Biol. Control 2022, 172, 104970. [Google Scholar] [CrossRef]
- Bunbury-Blanchette, A.L.; Walker, A.K. Trichoderma species show biocontrol potential in dual culture and greenhouse bioassays against Fusarium basal rot of onion. Biol. Control 2019, 130, 127–135. [Google Scholar] [CrossRef]
- Arif, S.; Munis, M.F.H.; Liaquat, F.; Gulzar, S.; Haroon, U.; Zhao, L.; Zhang, Y. Trichoderma viride establishes biodefense against clubroot (Plasmodiophora brassicae) and fosters plant growth via colonizing root hairs in pak choi (Brassica campestris spp. chinensis). Biol. Control 2023, 183, 105265. [Google Scholar] [CrossRef]
- Gulzar, S.; Manzoor, M.A.; Liaquat, F.; Shah, I.H.; Rehman, A.; Hameed, M.K.; Arif, S.; Zhou, X.; Zhang, Y. Effects of melatonin and Trichoderma harzianum on pak choi yield, chlorophyll contents and antioxidant defense system under clubroot disease. S. Afr. J. Bot. 2023, 158, 292–300. [Google Scholar] [CrossRef]
- Nandini, B.; Hariprasad, P.; Niranjana, S.R.; Shetty, H.S.; Geetha, N.P. Elicitation of resistance in pearl millet by oligosaccharides of Trichoderma spp. against downy mildew disease. J. Plant Inter. 2013, 8, 45–55. [Google Scholar] [CrossRef]
- Nandini, B.; Geetha, N.; Prakash, H.S.; Hariparsad, P. Natural uptake of anti-oomycetes Trichoderma produced secondary metabolites from pearl millet seedlings- A new mechanism of biological control of downy mildew disease. Biol. Control 2021, 156, 104550. [Google Scholar] [CrossRef]
- Rochal, K.K.L.; Pierre, E.; Diane, Y.Y.; Sahu, K.P.; Vanessa, N.D.; Wankeu, K.; Herman, T.; Gilbert, G.T.P.; Sevilor, K.; Germain, K.; et al. Biological elicitor potential of endospheric Trichoderma and derived consortia against pepper (Capsicum annuum L.) leaf curl virus. Arch. Phytopathol. Plant Prot. 2021, 54, 1926–1952. [Google Scholar] [CrossRef]
- Eke, P.; Dinango, V.N.; Wakam, L.N.; Toghueo, R.M.K.; Kouokap, L.R.K.; Mabou, L.C.N.; Wankeu, T.H.K.; Ngomsi, P.; Boyom, F.F. Diagnosis and bioefficiancy of endospheric trichoderma strains of selected medicinal plant on pepper root rot and vascular wilt in Cameroon. Arch. Phytopathol. Plant Prot. 2020, 54, 1–15. [Google Scholar] [CrossRef]
- Demissie, S.; Megersa, G.; Meressa, B.H.; Muleta, D. Resistance levels of Ethiopian hot pepper (Capsicum spp.) varieties to a pathogenic Fusarium spp. and in vitro antagonistic effect of Trichoderma spp. Arch. Phytopathol. Plant Prot. 2021, 54, 647–663. [Google Scholar] [CrossRef]
- Pereira, T.D.S.; Macedo, A.G.; Silva, J.D.; Pinheiro, J.B.; Paula, A.M.D.; Biscaia, D.; Busato, J.G. Water-extractable fraction of vrmicomposts enriched with Trichoderma enhances the growth of bell pepper and tomato as well as their tolerance against Meloidogyne incognita. Sci. Hortic. 2020, 272, 109536. [Google Scholar] [CrossRef]
- Tomah, A.A.; Alamer, I.S.A.; Li, B.; Zhang, J.-Z. A new species of Trichoderma and gliotoxin role: A new observation in enhancing biocontrol potential of T. virens against Phytophthora capsici on chili pepper. Biol. Control 2020, 145, 104261. [Google Scholar] [CrossRef]
- Rokni, N.; Alizadeh, H.S.; Bazgir, E.; Darvishnia, M.; Mirzaei-Najafgholi, H. The tripartite consortium of Serendipita indica, Trichoderma simmonsii, and bell pepper (Capsicum annum). Biol. Control 2021, 158, 104608. [Google Scholar] [CrossRef]
- Guo, K.; Sui, Y.; Li, Z.; Huang, Y.; Zhang, H.; Wang, W. Colonization of Trichoderma viride Tv-1511 in perppermint (Mentha × piperita L.) roots promotes essential oil production by triggering ROS-mediated MAPK activation. Plant Physiol. Biochem. 2020, 151, 705–718. [Google Scholar] [CrossRef]
- Mishra, R.K.; Pandey, S.; Hazra, K.K.; Mishra, M.; Naik, S.J.S.; Bohra, A.; Parihar, A.K.; Rathore, U.S.; Naimuddin, K.K.; Singh, B.; et al. Biocontrol efficacy and induced defense mechanisms of indigenous Trichoderma strains against Fusarium wilt (F. udum (Butler) in pigeonpea. Physiol. Mol. Plant Pathol. 2023, 127, 102122. [Google Scholar] [CrossRef]
- Dehriya, K.; Shukla, A.; Ganaie, M.A.; Vyas, D. Individual and interactive role of Trichoderma and Mycorrhizae in controlling wilt disease and growth reduction in Cajanus cajan caused by Fusarium udum. Arch. Phytopathol. Plant Prot. 2015, 48, 50–61. [Google Scholar] [CrossRef]
- Kelley, W.D. Interactions of Phytophthora cinnamomi and Trichoderma spp. in relation to propagule production in soil cultures at 26 °C. Can. J. Microbiol. 1977, 23, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Mollah, M.M.I.; Hassan, N. Efficacy of Trichoderma harzianum, as a biological fungicide against fungal diseases of potato, late blight and early blight. J. Nat. Pest Res. 2023, 5, 100047. [Google Scholar] [CrossRef]
- Hassan, M.M.; Soliman, M.M.; Al-Otaibi, S.; El-Shehawi, A.M.; Taha, E.-K.A.; Sayed, S. The effectiveness of Xanthium strumamrium L. extract and Trichoderma spp. against pomegranate isolated. J. King Saud. Univ. Sci. 2022, 34, 102185. [Google Scholar] [CrossRef]
- Metz, N.; Hausladen, H. Trichoderma spp. as potential biological control agent against Alternaria solani in potato. Biol. Control 2022, 166, 104820. [Google Scholar] [CrossRef]
- Ommati, F.; Zaker, M. In vitro and greenhouse evaluations of Trichoderma isolates for biological control of potato wilt disease (Fusarium solani). Arch. Phytopathol. Plant Prot. 2012, 45, 1715–1723. [Google Scholar] [CrossRef]
- Sivakumar, D.; Wijeratnam, R.S.; Wijesundera, R.L.; Marikar, F.M.; Abeyesekere, M. Antagonistic effect of Trichoderna harzianm on postharvest pathogens of rambutan (Nephelium lappaceum). Phytoparasitica 2000, 28, 240–247. [Google Scholar] [CrossRef]
- Pandey, A.K.; Kumar, A.; Samota, M.K.; Tanti, A. Trichoderma reesei as an elicitor triggers defense responses in tea plant and delays gray blight symptoms. Pest Biochem. Physiol. 2022, 188, 105279. [Google Scholar] [CrossRef]
- Klaram, R.; Jantasorn, A.; Dethoup, T. Efficacy of marine antagonist, Trichoderma spp. as halo-tolerant biofungicide in controlling rice diseases and yield improvement. Biol. Control 2022, 172, 104985. [Google Scholar] [CrossRef]
- Chen, L.-H.; Zhang, J.; Shao, X.-H.; Wang, S.-S.; Miao, Q.-S.; Mao, X.-Y.; Zhai, Y.-M.; She, D.-L. Development and evaluation of Trichoderma asperellum preparation for control of sheath blight of rice (Oryza sativa L.). Biocontrol Sci. Technol. 2015, 25, 316–328. [Google Scholar] [CrossRef]
- Parizi, T.E.; Ansari, M.; Elaminejad, T. Evaluation of the potential of Trichoderma viride in the control of fungal pathogens of Roselle (Hibiscus sabdariffa L.) in vitro. Microbiol. Pathogen. 2012, 52, 201–205. [Google Scholar] [CrossRef]
- Elad, Y.; Chet, I.; Boyle, P.; Henis, Y. Parasitism of Trichoderma spp. on rhizoctonia solani and Sclerotium rolfsii-scanning electron microscopy and fluorescence microscopy. Phytopathology 1983, 73, 85–88. [Google Scholar] [CrossRef]
- Vecstaudza, D.; Grantina-levina, L.; Makarenkova, G.; Kasparinskis, R.; Selga, T.; Steinberga, V.; Stelmahere, S.; Steiner, C.; Muter, O. The impact of wood-derived biochar on the survival of Trichoderma spp. and growth of Secale cereale L. in sandy soil. Biocontrol Sci. Technol. 2018, 28, 341–358. [Google Scholar] [CrossRef]
- Arellano, A.D.V.; Dilva, G.M.D.; Guatimosim, E.; Dorneles, K.D.R.; Moreira, L.G.; Dallagnol, L.J. Seeds coated with Trichoderma atroviride and soil amended with silicon improve the resistance of Lolium multiflorum against Pyricularia oryzae. Biol. Control 2021, 154, 104499. [Google Scholar] [CrossRef]
- John, R.P.; Tyagi, R.D.; Prevost, D.; Brar, S.K.; Pouleur, S.; Surampalli, R.Y. Mycoparasitic Trichoderma viride as a biocontrol agent against Fusarium oxysporum f. sp. adzuki and Pythium arrhenomanes and as a growth promoter of soybean. Crop Prot. 2010, 29, 1452–1459. [Google Scholar] [CrossRef]
- Conte, E.D.; Magro, T.D.; Bem, L.C.D.; Dalmina, J.C.; Matte, J.A.; Schenkel, V.O.; Schwambach, J. Use of Trichoderma spp. in no-tillage system: Effect of soil and soybean crop. Biol. Control 2022, 171, 104941. [Google Scholar] [CrossRef]
- Ulrich, A.; Muller, C.; Gasparetto, I.G.; Bonafin, F.; Diering, N.L.; Camargo, A.F.; Reichert Junior, F.W.; Paudel, S.R.; Treichel, H.; Mossi, A.J. Bioherbicide effects of Trichoderma koningiopsis associated with commercial formulations of glyphosate in weeds and soybean plants. Crop Prot. 2023, 172, 106346. [Google Scholar] [CrossRef]
- Camatti, G.; Santos, F.M.D.; Rodrigues Junior, G.L.D.S.; Camargo, D.P.; Mandio, G.S.; Santos, J.R.P.; Silva, J.C.P.D. Bacillus- and Trichoderma-based products control the spiral nematode Helicotylenchus dihystera in soybean. Rhizosphere 2023, 27, 100717. [Google Scholar] [CrossRef]
- Gashash, E.A.; El-Sayed, S.A.; Kamel, W.M.; Sarhan, E.A.D. Evaluation of the potential of native Trichoderma spp. against root-rot diseases of soybean. Arch. Phytopathol. Plant Prot. 2023, 56, 1–14. [Google Scholar] [CrossRef]
- Sofy, M.; Mohamed, H.; Dawood, M.; Abu-Elsaoud, A.; Soliman, M. Integrated usage of Trichoderma harzianum and biochar to ameliorate salt stress on spinach plants. Arch. Agron. Soil Sci. 2022, 68, 2005–2026. [Google Scholar] [CrossRef]
- Naher, L.; Yusuf, U.K.; Ismail, A.; Hossain, K. Trichoderma spp.: A biocontrol agent for sustainable managment of plant diseases. Pak. J. Bot. 2014, 46, 1489–1493. [Google Scholar]
- Qiu, Y.-L.; Wang, N.; Zhang, J.-F.; Chen, Z.-H.; Yan, Y.-T.; Wu, F.; Li, H.-L. Application of indigenous honeybees in dispersing Trichoderma harzianum spores for control of the strawberry grey mould. Biocontrol Sci. Technol. 2021, 31, 418–429. [Google Scholar] [CrossRef]
- Kappel, L.; Kosa, N.; Gruber, S. The multilateral efficacy of chitosan and Trichoderma on sugar beet. J. Fungi 2022, 8, 137. [Google Scholar] [CrossRef] [PubMed]
- Paramasivan, M.; Chandrasekaran, A.; Mohan, S.; Muthukrishnan, N. Ecological management of tropical sugar beet (TSB) root rot (Sclerotium rolfsii (Sacc.) by rhizosphere Trichoderma species. Arch. Phytopathol. Plant Prot. 2014, 47, 1629–1644. [Google Scholar] [CrossRef]
- Upadhyay, J.P.; Mukhopadhyay, A.N. Biological control of Sclerotium rolfsii by Trichoderma harzianum in sugarbeet. Trop Pest Manag. 1986, 32, 215–220. [Google Scholar] [CrossRef]
- Kakvan, N.; Heydari, A.; Zamanizadeh, H.R.; Rezaee, S.; Naraghi, L. Development of new bioformulations using Trichoderma and Talaromyces fungal antagonists for biological control of sugar beet damping-off disease. Crop Prot. 2013, 53, 80–84. [Google Scholar] [CrossRef]
- Shukla, S.K.; Jaiswal, V.P.; Sharma, L.; Tiwari, R.; Pathak, A.D.; Gaur, A.; Awasthi, S.K.; Srivastava, A. Trash management and Trichoderma harzianum influencing photosynthesis, soil carbon sequestration, and growth and yield of sugarcane ratoon in subtropical India. Eur. J. Agron. 2022, 141, 126631. [Google Scholar] [CrossRef]
- Devi, S.S.; Sreenivasulu, Y.; Saritha, S.; Kumar, M.R.; Kumar, K.P.; Sudharak, P. Molecular diversity of native Trichoderma isolates against Fusarium oxysporum f. sp. lycopersici (Sacc.). A causal agent of Fusarium wilt in tomato (Lycopersicon esculentum Mill.). Arch. Phytopathol. Plant Prot. 2012, 45, 686–698. [Google Scholar] [CrossRef]
- Mushtaq, S.; Bareen, F.; Tayyeb, A.; Nazir, A. Autochthonous strains of trichoderma isolated from tannery solid waste improve phytoextraction potential of heavy metals by sunflower. Int. J. Phytoremed. 2023, 25, 1435–1454. [Google Scholar] [CrossRef]
- Arzanlou, M.; Khodaei, S.; Narmani, A.; Babai-Ahari, A.; Azar, A.M. Inhibitory effect of Trichoderma isolates on growth of Alternaria alternata, the causal agent of leaf spot disease on sunflower, under laboratory conditions. Arch. Phytopathol. Plant Prot. 2014, 47, 1592–1599. [Google Scholar] [CrossRef]
- Yadav, A.; Yadva, K.; Aggarwal, A. Impact of arbuscular mycorrhizal fungi with Trichoderma viride and Pseudomonas flurescens on growth, yield, and oil content in Helianthus annuus L. J. Essent Oil Bear Plants 2015, 18, 444–454. [Google Scholar] [CrossRef]
- Mathews, J.R.; Sivparsad, B.J.; Laing, M.D. Greenhouse evaluation of Trichoderma harzianum for the control of Sclerotinia wilt (Sclerotinia sclerotiorum) of sunflower. S. Afr. J. Plant Soil 2019, 36, 69–72. [Google Scholar] [CrossRef]
- Ozer, N.; Coskuntuna, A.; Sabudak, T. Trichoderma harzianum-induced defense in sunflower (Helianthus annuus L.) against Plasmopara halstedii with changes in metabolite profiling of roots. Biocontrol Sci. Technol. 2021, 31, 1403–1418. [Google Scholar] [CrossRef]
- Wang, X.; Xu, S.; Wu, S.; Feng, S.; Bai, Z.; Zhuang, G.; Zhuang, X. Effect of Trichoderma viride biofertilizer on ammonia volatilization from an alkaline soil in Northern China. J. Environ. Sci. 2018, 66, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Han, S.; Wang, G.; Liu, X.; Amombo, E.; Xie, Y.; Fu, J. The fungus Aspergillus aculeatus enhances salt-stress tolerance, metabolite accumulation, and improves forage quality in perennial ryegrass. Front. Microbiol. 2017, 8, 1664. [Google Scholar] [CrossRef] [PubMed]
- Andrzejak, R.; Janowska, B. Flowering, nutritioal status, and content of chloroplast pigments in leaves of Gladiolus hybridus L. Advances Red after application of Trichoderma spp. Sustainability 2022, 14, 4576. [Google Scholar] [CrossRef]
- Pandey, A.K.; Samota, M.K.; Tanti, A.J.; Babu, A. Trichoderma reesei induces defense-related biochemical markers associated with resistance to Fusarim dieback in tea crop. Biol. Control 2023, 180, 105200. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, C.; Gao, Z.-F.; Qiu, C.-W.; Shi, S.-H.; Chen, Z.-H.; Ali, M.A.; Wang, F.; Wu, F. Integrated physiological and omics analyses reveal the mechanism of beneficial fungi Trichoderma sp. alleviating cadmium toxicity in tobacco (Nicotiana tabacum L.). Ecotoxicol. Environ. Saf. 2023, 267, 115631. [Google Scholar] [CrossRef]
- Khalil, M.I.I.; Youssef, S.A.; Tartoura, K.A.; Eldesoky, A.A. Comparative evaluation of physiological and biochemical alteration in tomato plants infected by Alternaria alternata in response to Trichoderma viride and Chaetomium globosum application. Physiol. Mol. Plant Pathol. 2021, 115, 101671. [Google Scholar] [CrossRef]
- Olowe, O.M.; Nicola, L.; Asemoloye, M.D.; Akanmu, A.O.; Babalola, O.O. Trichoderma: Potential bio-resource for the management of tomato root rot diseases in Africa. Microbiol. Res. 2022, 257, 126978. [Google Scholar] [CrossRef]
- Devi, S.S.; Sreenivasulu, Y.; Rao, K.V.B. Protective role of Trichoderma logibrachiatum (WT2) on lead induced oxidative stress in Helianthus annus L. Indian J. Exp. Biol. 2017, 55, 235–241. [Google Scholar] [CrossRef]
- Singh, J.; Kumar, V.; Srivastava, S.; Kumar, A.; Singh, V.P. In vitro evaluation of Trichoderma species against Fusarium oxysporum f. sp. lycopersici causing tomato wilt. Plant Pathol. J. 2018, 17, 59–64. [Google Scholar] [CrossRef]
- Zehra, A.; Aamir, M.; Dubey, M.L.; Ansari, W.A.; Meena, M.; Swapnil, P.; Upadhyay, R.S.; Ali, M.A.; Al-Ghamdi, A.A.; Lee, J. Enhanced protection of tomato against Fusarium wilt through biopriming with Trichoderma harzianum. J. King Saud Univ. Sci. 2023, 35, 102466. [Google Scholar] [CrossRef]
- Al-Hazmi, A.S.; TariqJaveed, M. Effects of different inoculum densities of Trichoderma harzianum and Trichoderma viride against Meloidogyne javanica on tomato. Saudi J. Biol. Sci. 2016, 23, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Malolepsza, U.; Nawrocka, J.; Szczech, M. Trichoderma virens 106 inoculation stimulates defense enzyme activities and enhances phenolic levels in tomato plants leading to lowered Rhizoctonia solani infection. Biocontrol Sci. Technol. 2017, 27, 180–199. [Google Scholar] [CrossRef]
- Yan, Y.; Mao, Q.; Wang, Y.; Zhao, J.; Fun, Y.; Yang, Z.; Peng, X.; Zhang, M.; Bai, B.; Liu, A.; et al. Trichoderma harzianum induces resistance to root-knot nematodes by increasing secondary metabolite synthesis and defense-related enzyme activity in Solanum lycopersicum L. Biol. Control 2021, 158, 104609. [Google Scholar] [CrossRef]
- Islam, M.M.; Hossain, D.M.; Nonaka, M.; Harada, N. Biological control of tomato collar rot induced by Sclerotium rolfsii using Trichoderma species isolated in Bangladesh. Arch. Phytopathol. Plant Prot. 2017, 50, 109–116. [Google Scholar] [CrossRef]
- Rubio, M.B.; Dominguez, S.; Monte, E.; Hermosa, R. Comparative study of Trichoderma gene expression in interactions with tomato plants using high density oligonucleotide microarrays. Microbiology 2012, 158, 119–128. [Google Scholar] [CrossRef]
- Sani, M.N.H.; Hasan, M.; Uddain, J.; Subramaniam, S. Impact of application of Trichoderma and biochar on growth, productivity and nutritional quality of tomato under reduced N-P-K fertilization. Annal Agric. Sci. 2020, 65, 107–115. [Google Scholar] [CrossRef]
- Natsiopoulos, D.; Tziolias, A.; Lagogiannis, I.; Mantzoukas, S.; Eliopoulos, P.A. Growth-promoting and protective effect of Trichoderma atrobrunneum and T. simmonssi on tomato against soil-borne fungal pathogens. Crops 2022, 2, 202–217. [Google Scholar] [CrossRef]
- Tripathi, R.; Keswani, C.; Tewari, R. Trichoderma koningii enhances tolerance against thermal stress by regulating ROS metabolism in tomato (Solanum lycopersicum L.) plants. J. Plant Interact. 2021, 16, 116–126. [Google Scholar] [CrossRef]
- Mona, S.A.; Hashem, A.; Abd Allah, E.F.; Alqarawi, A.A.; Soliman, D.W.K.; Wirth, S.; Egamberdieva, D. Increased resistance of drought by Trichoderma harzianum fungal treatment correlates with increased secondary metabolites and proline content. J. Integr. Agric. 2017, 16, 1751–1757. [Google Scholar] [CrossRef]
- Shanmugaraj, C.; Kamil, D.; Kundu, A.; Singh, P.K.; Das, A.; Hussain, Z.; Gogoi, R.; Shashnak, P.R.; Gangaraj, R.; Chaithra, M. Exploring the potential biocontrol isolates of Trichoderma asperellum for management of collar rot disease in tomato. Horticulturae 2023, 9, 1116. [Google Scholar] [CrossRef]
- Sharma, A.; Salwan, R.; Sharma, V. Extracellular proteins of Trichoderma and their roles in plant health. S. Afr. J. Bot. 2022, 147, 359–369. [Google Scholar] [CrossRef]
- Vinale, F.; Sivasithamparam, K.; Ghisalberti, E.L.; Marra, R.; Barbetti, M.J.; Li, H.; Woo, S.L.; Lorito, M. A novel role for Trichoderma secondary metabolites in the interactions with plants. Physiol. Mol. Plant Pathol. 2008, 72, 80–86. [Google Scholar] [CrossRef]
- Carillo, P.; Woo, S.L.; Comite, E.; El-nakhel, C.; Rouphael, Y.; Fusco, G.M.; Borzacchiello, A.; Lanzuise, S.; Vinale, F. Application of Trichoderma harzianum, 6-Pentyl-a-Pyrone and plant biopolymer formulations modulate plant metabolism and fruit quality of plum tomatoes. Plants 2020, 9, 771. [Google Scholar] [CrossRef]
- De Palma, M.; Docimo, T.; Guida, G.; Salzano, M.; Albrizio, R.; Giorio, P.; Ruocco, M.; Tucci, M. Transcriptome modulation by the beneficial fungus Trichoderma longibrachiatum drives water stress response and recovery in tomato. Environ. Experim. Bot. 2021, 190, 104588. [Google Scholar] [CrossRef]
- Andrzejak, R.; Janowska, B.; Renska, B.; Kosiada, T. Effect of Trichoderma spp. and fertilization on the flowering of Begonia X tuberhybrida voss. Picotee sunburst. Agronomy 2021, 11, 1278. [Google Scholar] [CrossRef]
- Ghosh, S.K.; Pal, S.; Banerjee, S. Identification and pathogenicity Alternaria alternata causing leaf blight of Bacopa monnieri (L.) Wettst. and its biocontrol by Trichoderma species in agrifields—An ecofriendly approach. J. Appl. Res. Med. Aromat Plants 2022, 31, 100406. [Google Scholar] [CrossRef]
- Villa-Rodriguez, E.; Lugo-Enriquez, C.; Ferguson, S.; Parra-Cota, F.I.; Cira-Chavez, L.A.; Santos-Villalobos, S.D.I. Trichoderma harzianum sensu lato TSM39: A wheat microbiome fungus that mitigates spot blotch disease of wheat (Triticum turgidum L. subsp. durum) caused by Bipolaris sorokiniana. Biol. Control 2022, 175, 105055. [Google Scholar] [CrossRef]
- Javaid, A.; Sli, S. Herbicidal activity of culture filtrates of Trichoderma spp. against two problematic weeds of wheat. Nat. Prod. Res. 2011, 25, 730–740. [Google Scholar] [CrossRef]
- Makhlouf, K.E.; Boungab, K.; Mokrani, S. Synergistic effect of Pseudomonas azotoformans and Trichoderma gamsii in management of Fusarium crown rot of wheat. Arch. Phytopathol. Plant Prot. 2023, 56, 108–126. [Google Scholar] [CrossRef]
- Alvarez-Garcia, S.; Manga-Robles, A.; Encina, A.; Gutierrez, S.; Casquero, P.A. Novel culture chamber to evaluate in vitro plant-microbe volatile interactions; Effects of Trichoderma harzianum volatiles on wheat plantlets. Plant Sci. 2022, 320, 111286. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Dou, K.; Wang, S.; Wu, Q.; Ni, M.; Zhang, T.; Lu, Z.; Tang, J.; Chen, J. Elicitor hydrophobin Hyd1 interacts with ubiquilin1-like to induce maize systemic resistance. J. Integr. Plant Biol. 2020, 62, 509–526. [Google Scholar] [CrossRef] [PubMed]
- Karuppiah, V.; He, A.; Lu, Z.; Wang, X.; Li, Y.; Chen, J. Trichoderma asperellum GDFS1009-mediated maize resistance against Fusarium graminearum stalk rot and mycotoxin degradation. Biol. Control 2022, 174, 105026. [Google Scholar] [CrossRef]
- Abbey, J.A.; Percival, D.; Anku, K.; Schilder, A.; Asiedu, S. Managing Botrytis blossom bligh of wild blueberry through field sanitation, lime sulfur and Trichoderma application. Can. J. Plant Pathol. 2022, 44, 361–371. [Google Scholar] [CrossRef]
- Bello, F.; Montironi, I.D.; Medina, M.B.; Munitz, M.S.; Ferreira, F.V.; Williman, C.; Vazquez, D.; Cariddi, L.N.; Musumeci, M.A. Mycofumigation of postharvest blueberries with volatile compounds from Trichoderma atroviride IC-11 is a promising tool to control rots caused by Botrytis cinerea. Food Microbiol. 2022, 106, 104040. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).