Abstract
Coconut (Cocos nucifera L.) is an important agricultural commodity with substantial economic and nutritional value, widely used for various products, including coconut water. The sweetness is an important quality trait of coconut water, which is influenced by genetic and environmental factors. In this study, we utilized next-generation sequencing to identify genetic variations in the coconut genome associated with the sweetness of coconut water. Whole-genome resequencing of 49 coconut accessions, including diverse germplasm and an F2 population of 81 individuals, revealed ~27 M SNPs and ~1.5 M InDels. Sugar content measured by °Bx was highly variable across all accessions tested, with dwarf varieties generally sweeter. A comprehensive analysis of the sugar profiles revealed that sucrose was the major sugar contributing to sweetness. Allele mining of the 148 genes involved in sugar metabolism and transport and genotype–phenotype association tests revealed two significant SNPs in the hexose carrier protein (Cnu01G018720) and sucrose synthase (Cnu09G011120) genes associated with the higher sugar content in both the germplasm and F2 populations. This research provides valuable insights into the genetic basis of coconut sweetness and offers molecular markers for breeding programs aimed at improving coconut water quality. The identified variants can improve the selection process in breeding high-quality sweet coconut varieties and thus support the economic sustainability of coconut cultivation.
1. Introduction
Coconut (Cocos nucifera L.) is widely known for its many uses, especially as a food and energy crop. The fruit is the most valuable part of the coconut and provides a range of products such as coconut water, virgin coconut oil, copra and coconut milk [1]. Coconut cultivation covers more than 12 million hectares in 92 countries, underlining its global agricultural importance [2]. The versatility and economic importance of the fruit make it indispensable for both local consumption and international trade [3]. The world’s coconut populations are divided into two main groups: the Indo-Atlantic group and the Pacific group, which includes subgroups such as Southeast Asia, Micronesia, Polynesia, Melanesia and the Pacific coast of South and Central America [4,5]. Coconut varieties are divided into tall and dwarf tree varieties [6]. Tall coconuts, which cross-pollinate, have greater genetic diversity than dwarf coconuts, which self-pollinate and have less genetic diversity [5,7]. This genetic diversity is crucial for breeding programs and the development of new varieties with desirable traits. Dwarf varieties can be further subdivided based on the color of the nuts, e.g., green, yellow or brown [8]. Tall coconuts, which are more common worldwide, are valued primarily for their mature fruits, while dwarf coconuts are valued for the coconut water and tender flesh of their young fruits [5]. The liquid endosperm, which consists predominantly of glucose, sucrose and fructose, is richer in dwarf varieties, making them ideal for the production of coconut water [9]. Popular varieties for coconut water consumption are Green Dwarf, King Coconut, Aromatic Green Dwarf (Nam Hom) and Chowgat Orange Dwarf [9]. Coconut water contains sugars, minerals, vitamins, amino acids, enzymes, volatile aromatic compounds and other biochemical compounds [10]. Studies on the physicochemical properties of different coconut varieties have shown that dwarf coconuts, with their higher pH and lower titratable acidity, have a better flavor profile characterized by sweetness and acidity [10,11]. Sweetness is a crucial quality attribute that consumers value in coconut water. Improving the sweetness of coconuts is important for increasing their market value. The sweetness of coconut water is influenced by both the genetic makeup of the coconut and environmental factors such as soil type and climate [12]. In addition, it depends on the stage of maturity, with young coconuts generally producing sweeter water. Coconuts usually take 12 months to fully mature after pollination, with the sugar content in coconut water peaking after about 7 to 8 months before gradually decreasing [12]. Therefore, most coconut farms harvest the fruit at this optimum age to ensure the best quality of coconut water. Understanding the genetic and metabolic determinants of coconut sweetness has practical implications for breeding programs to improve this trait.
The sugar metabolism process is principally regulated by four important enzymes: neutral invertase, acid invertase, sucrose phosphate synthase and sucrose synthase [13,14]. Understanding the genetic basis of these metabolic pathways is crucial for the development of breeding strategies that can improve the sweetness of coconut water. Plants produce sucrose in photosynthetically active tissue through the process of photosynthesis and transport it into the phloem. Sucrose is then moved to sink organs, where it can be rapidly hydrolyzed into glucose and fructose [13,15]. Research on other crops has shown that manipulating the expression of genes involved in sugar metabolism can significantly alter the sugar content of fruits [16]. In tomato fruits, pathways involving invertase and sucrose synthase play an important role in sucrose metabolism [17]. In maize, sweetness is primarily controlled by the shrunken-2 (sh2) gene, which affects sucrose synthase, resulting in higher sugar accumulation in the kernels. The manipulation of these genes has helped to develop super sweet maize varieties that have a higher sugar content compared with normal maize [16]. Although research on the genes encoding the essential enzymes involved in sugar production has been conducted in a number of plants [14,16], no comparable study has been conducted on coconuts. In this study, we utilized the next-generation sequencing (NGS) technology of a collection of coconut differing in sweetness levels to characterize the genes involved in the sugar biosynthetic pathway in coconut and identify variants associated with sweetness. By analyzing these genes, we successfully identified natural variants in two candidate genes associated with the sweetness in coconut. The results of our study provide a fundamental understanding of genetic control over coconut sweetness and offer a pathway for genetic improvement of coconut cultivars through targeted breeding strategies.
2. Results
2.1. Whole-Genome Resequencing, Variant Calling and Population Study of Coconut Populations
The whole genome of 2 coconut populations, a germplasm of 49 coconut accessions and an F2 population of 81 individuals, was sequenced. The 49 coconut accessions represented a diversity of coconuts in Thailand, including 17 dwarf and 32 tall coconuts (Table 1). The F2 population was derived from a cross between a dwarf coconut and a tall coconut (Table S1). Whole-genome sequencing of the 130 coconut samples yielded a total of 42,044 million 150 bp paired-end reads with an average depth of 14.16× and an average genome coverage of 98.71%. After mapping with an in-house coconut reference genome, a set of 27,000,601 common SNPs was identified, along with 1,540,742 InDels (Table 1 and Table S1).

Table 1.
List of coconut accessions and details on genetic groups, sweetness (Brix) and sequencing and variant statistics.
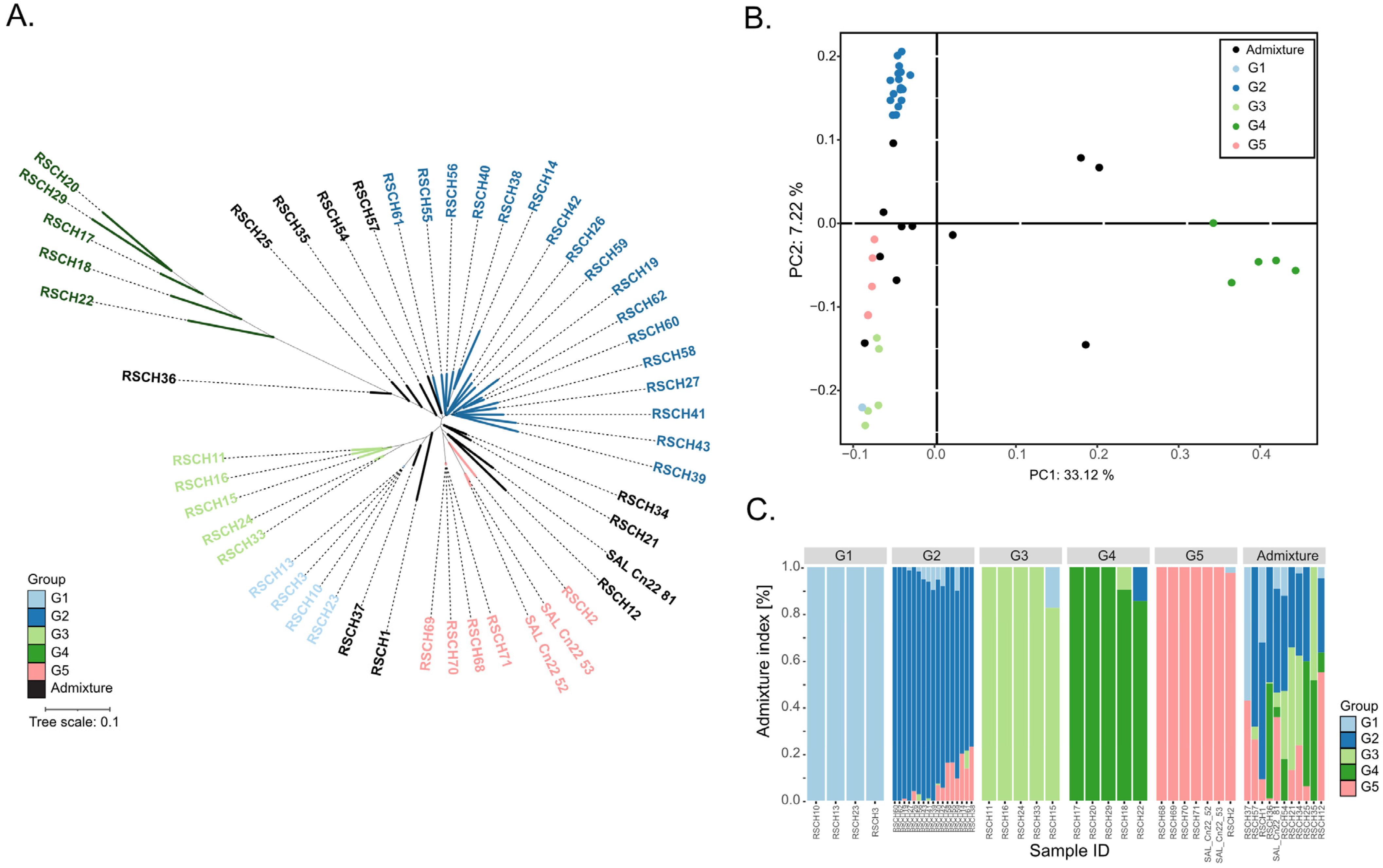
To investigate the population structure and genetic diversity of the 49 coconut accessions, we performed three analyses, namely phylogenetic tree analysis, principal coordinate analysis (PCoA) and STRUCTURE analysis. The genotype data used for these analyzes were 18,379,163 filtered SNPs of the 49 coconut accessions. The genetic relationships analyzed by UPGMA-based clustering and the population structure analyzed by PCoA and STRUCTURE revealed 5 major groups (G1–G5) among the 49 coconut accessions: G1, consisting of 4 dwarf accessions; G2, the largest group, consisting of 17 tall accessions; G3, consisting of 5 colored dwarf accessions; G4, consisting of 5 tall accessions with the highest distance matrix to the other groups; G5, consisting of 7 dwarf accessions (Figure 1; Table 1). Based on the results of the STRUCTURE analysis, 11 accessions were considered as admixtures. The population study results confirmed the diversity of coconut accessions used in this study.
Figure 1.
Population study of the 49 coconut accessions. (A) UPGMA phylogenetic tree. (B) Principal coordinate analysis (PCoA). (C) STRUCTURE analysis. Different genetic groups are highlighted by different colors.
2.2. Analysis of Sweetness and Sugar Profile
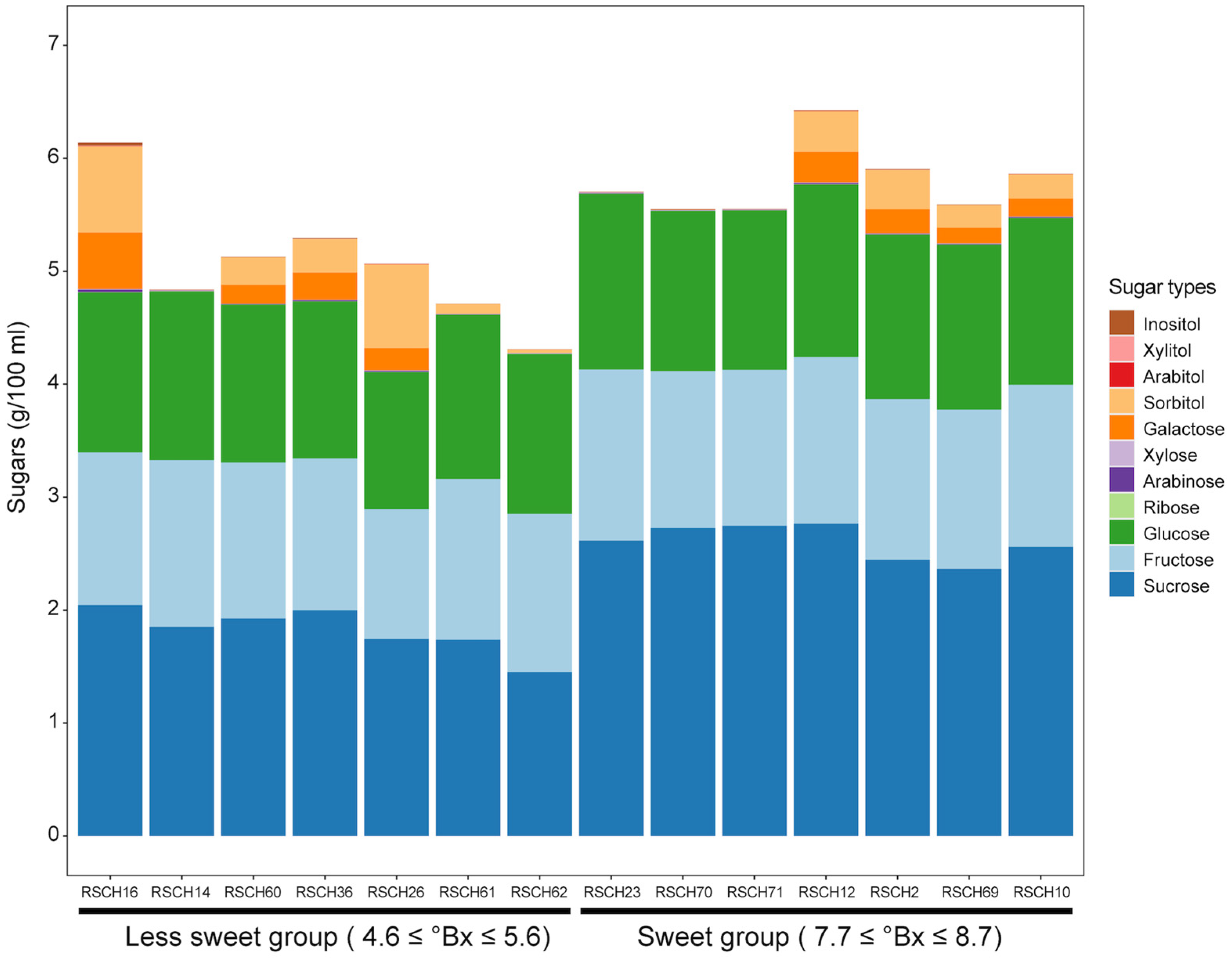
The sweetness of the coconut water was derived from the sugar content (°Bx) measured with a refractometer. The evaluated sugar content of the 49 coconut accessions ranged from 4.60 to 8.70 °Bx, with an average of 6.66 °Bx (Table 1). The sugar content of the dwarf varieties averaged 7.32 °Bx and that of the tall varieties 6.31 °Bx. The sweetest coconut water was found in the dwarf accession RSCH23 (8.7 °Bx) and the least sweet coconut water in the tall accession RSCH62 (4.6 °Bx) (Table 1). The evaluated sugar content of the 81 F2 individuals ranged from 5.40 to 7.60 °Bx, with an average of 6.46 °Bx. We then selected seven representatives of the coconut accessions with the highest sugar content values (namely the sweet group) of 7.7 ≤ °Bx ≤ 8.7 and seven accessions with the lowest sugar content values (namely the less-sweet group) of 4.7 ≤ °Bx ≤ 5.6 to analyze the sugar profile, focusing on disaccharides, i.e., sucrose; monosaccharides, i.e., fructose, glucose, ribose, arabinose, xylose and galactose; and sugar alcohols, i.e., sorbitol, arabitol, xylitol and inositol with HPAEC-PAD. Of these, the content of fructose, glucose and sucrose was higher in all the accessions studied (Figure 2). The type of sugar that was present in the greatest quantity in all accessions was sucrose. When comparing the two groups of coconut accessions, the average sucrose content in the coconut water of the accessions was significantly higher in the sweet group (2.60 ± 0.15 g/100 mL) than in the less-sweet group (1.82 ± 0.20 g/100 mL). For the other sugars and the sugar alcohol, there was no clear trend when comparing the two groups. This indicates that the accumulation of sucrose content is probably related to the higher sweetness of the coconut water.
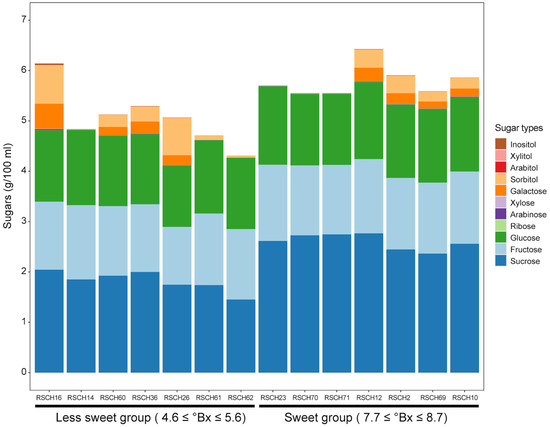
Figure 2.
The sugar content in coconut water from accessions with varying sweetness levels. A stacked bar graph illustrates the quantities of different sugars and sugar alcohols measured in coconut water extracted from 7-month-old fruits.
2.3. Identification of Genes Associated with Sweetness in Coconut
We used a gene mining approach to identify candidate genes associated with the sweetness of coconut water. A total of 148 genes involved in sugar metabolism and sugar transport were identified based on gene functional annotations in the coconut genome. These included genes encoding sucrose synthases and sugar transporters (Table S2). We then identified variants, i.e., SNPs and InDels, present in each gene. A total of 14,471 variants were identified with high confidence and compared among the 49 coconut accessions. Of these, the 871 variants were annotated with an effect on gene translation, i.e., missense variants causing amino acid changes and those causing stop-loss, stop-gain and frame shifts. Subsequently, these functional variants were selected to perform genotype–phenotype association tests with the sugar content in the °Bx scale determined in the 49 coconut accessions and an F2 population of 81 plants (Table S3). As a result, among the functional SNP and InDel variants, 57 SNPs within 35 genes were found to be significantly associated with sweetness in the 49 coconut accessions (p < 0.01; Table 2). After validation in the F2 population segregating for sugar content (°Bx), only SNPs 1_157345938 (A/G) and 9_119160271 (C/A) were found to be significantly associated with the trait (p < 0.05).

Table 2.
List of functional variants significantly associated with the sweetness in 49 coconut accessions.
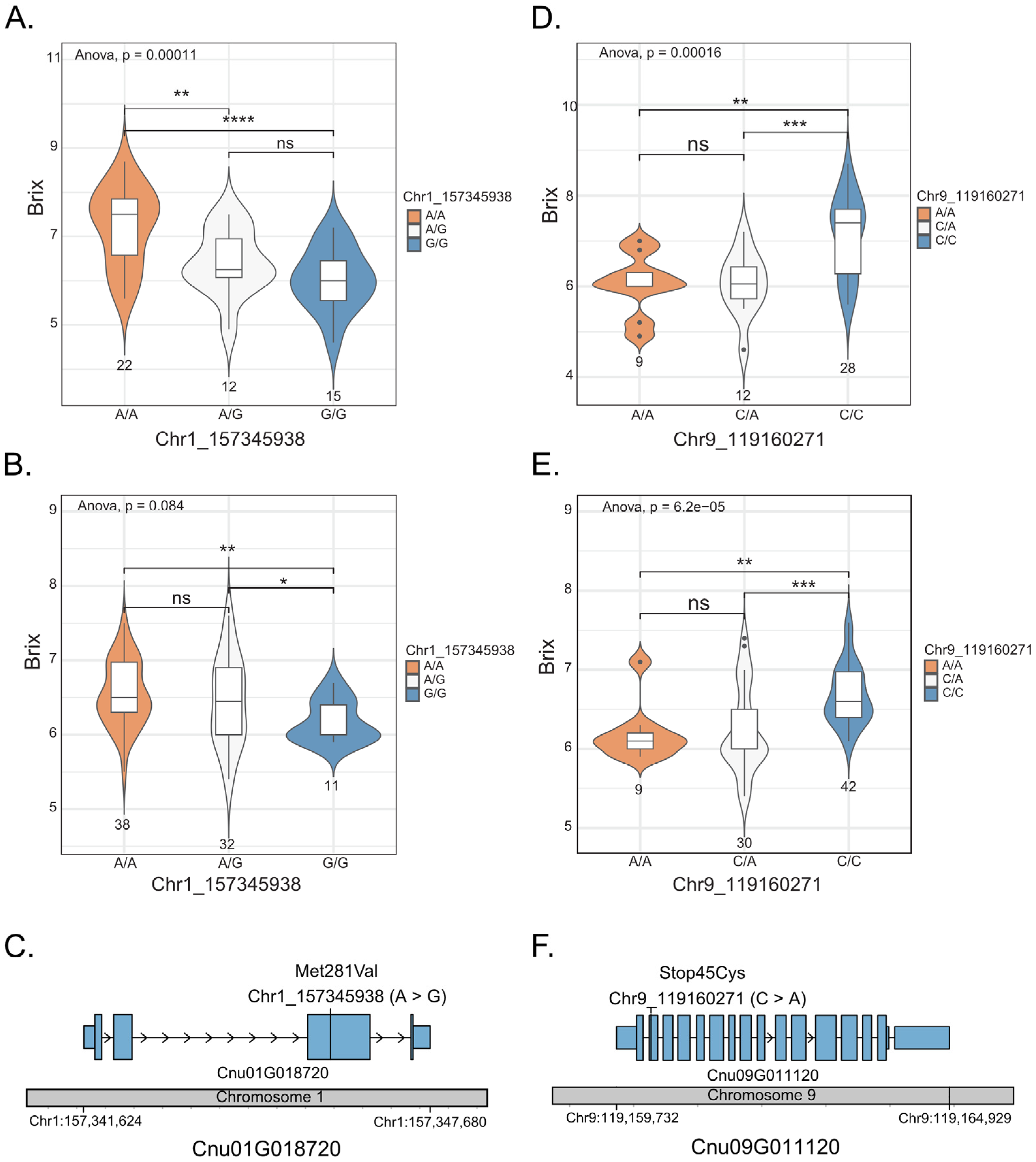
Among the 49 coconut accessions, the average °Bx value of the accessions with the homozygous genotype (A/A) at position 1_163346328 was 7.23 °Bx, which was significantly higher than that of the accessions with the homozygous genotype (GG) at the same position, which was 6.03 °Bx (Figure 3A). The average °Bx value of the accessions with the heterozygous genotype (A/G) was not significantly different from that of the accessions with the homozygous genotype (G/G) but it was significantly different from that of the accessions with the homozygous genotype (A/A). For SNP position 9_119160271, the average °Bx value of the accessions with the homozygous genotype (A/A; 7.11 °Bx) was also significantly higher than that of the accessions with the homozygous genotype (C/C; 6.04 °Bx). The average °Bx value of the accessions with the heterozygous genotype (A/C) was not significantly different from that of the accessions with the homozygous genotype (A/A) but it was significantly different from that of the accessions with the homozygous genotype (C/C; Figure 3D). A similar trend was confirmed in the F2 population (n = 81; Figure 3B,E). Based on these results, we considered the two SNPs as the candidate SNPs associated with coconut sweetness. The SNP 1_157345938 (A/G) was identified in exon 3 of the gene Cnu01G018720 (hexose carrier protein), where the nucleotide change A > G should have an effect on an amino acid change Met281Val (Figure 3C). SNP 9:119160271 was identified in exon 2 of the gene Cnu09G011120 (sucrose synthase 7), in which the nucleotide change C > A had an effect on a stop codon gain (TGC > TGA; Figure 3F).
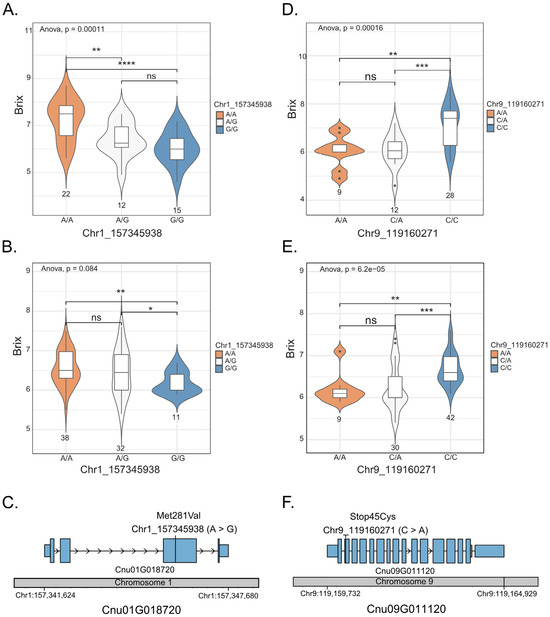
Figure 3.
Genotype–phenotype association on two genes, Cnu01G018720 and Cnu09G011120. The box plot/violin plots display the three different genotypes at SNP 1_157345938 on the gene Cnu01G018720, analyzed in 49 accessions (A) and 81 F2 individuals (B). The box plot/violin plots display the three different genotypes at SNP 9_119160271 on the gene Cnu09G011120, analyzed in 49 accessions (D) and 81 F2 individuals (E). The medians are indicated by solid horizontal lines in the box plots. The structure of Cnu01G018720 (C) and Cnu09G011120 (F) shows UTRs (small blue boxes), exons (large blue boxes) and introns (arrow lines). The grey bar represents the chromosome, with SNP positions in each gene indicated by vertical bars. * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001; ns (not significant).
3. Discussion
Coconut water is one of the best-known natural refreshing drinks. It is known for its health-promoting and energizing properties due to its sugars, dietary fiber, antioxidants, vitamins, minerals and phytohormones [10,18]. Over time, coconut water has gained popularity as a beverage that possesses both functional and nutritional properties, which has contributed to its economic growth. Improving the quality of coconut water will therefore increase its market value. The quality of coconut water is mainly related to its sweetness, which is mainly determined by its sugar content. The present study focused on identifying the genetic basis associated with the sweetness of coconut water so that the results can be used in a molecular breeding program to improve coconut for high quality coconut water. The coconut populations used in this study were a diverse germplasm of coconut in Thailand, which included 49 accessions of tall and dwarf types of Thai and international coconut cultivars and an F2 population of 81 individuals. The coconut water from the young fruits (7-month-old) of these coconut accessions and lines had different degrees of sweetness, as determined by the soluble solids’ content in degrees Brix (°Bx). The coconut water of most dwarf coconut accessions was sweeter than that of the coconut accessions of the tall group. This was confirmed by a recent study on the physicochemical properties and sensory acceptability of different coconut varieties, which found that the water of the dwarf coconut was more acceptable and palatable than the water of the tall coconut [11]. The comprehensive analysis of sugar profiles, including sugar alcohols, in coconut water provides insights into the broader metabolic pathways active in the different coconut genotypes. The results of the HPAEC-PAD analysis revealed the variations in the major sugar components such as sucrose, glucose and fructose, which collectively define the sweetness of coconut water. Our results show significant differences in sucrose concentration between the sweet and less-sweet coconut groups, suggesting that the different sweetness of each coconut group is due to their own sucrose metabolism. Higher sucrose concentrations in sweeter varieties indicate more efficient sucrose synthesis or a lower rate of sucrose degradation.
NGS technologies can be used to analyze diverse germplasm, leading to the discovery of candidate genes and trait-specific diagnostic markers. Rapid advances in NGS technologies have significantly reduced the cost and increased the speed of whole genome sequencing (WGS). This makes it possible to scan whole genomes or specific pathways to identify candidate genes in different accessions. For example, the application of NGS in tree breeding has led to the identification of candidate genes for traits such as fruit quality [19]. The application of WGS in this study provided a comprehensive overview of the genetic landscape of coconut cultivars. The groups of coconut accessions occurring in Thailand classified by the WGS-derived SNPs were similar to those identified by the GBS-derived SSRs in our previous study [6], except that the dwarf coconut cluster was further divided into three subgroups (G1, G3 and G5) in this study. This could be due to the high resolution of SNP markers used in this study compared with the much lower number of SSR markers used in the previous study. The genome-wide variants (SNPs and InDels) obtained from the WGS data were used for allele mining to identify candidate genes associated with sweetness in the 49 different coconut accessions in this study. Allele mining with NGS is a cost-effective method for identifying genetic variation in candidate genes and loci that are important for crop improvement. For instance, the allelic mining has been used to explore allelic diversity for nutrient-rich traits in rice [20]. The results of genotype-phenotype association analysis conducted on the 49 coconut accessions addressed two SNPs within a hexose carrier protein and a sucrose synthase gene. These SNPs were found to be strongly associated with variations in coconut sweetness, highlighting the potential of marker–trait association studies in linking genetic variants to the trait. Sucrose synthase plays a crucial role in sugar metabolism by catalyzing the reversible conversion of sucrose and uridine diphosphate (UDP) into UDP-glucose and fructose [21,22]. This enzyme plays a dual role in the synthesis and degradation of sucrose in response to metabolic demands, which is consistent with the observed differences in sugar content between different coconut genotypes [22]. This enzyme not only contributes to sucrose metabolism, but also provides UDP-glucose for the synthesis of cellulose in the cell wall, highlighting its role in structural and metabolic processes within the plant [23]. The significant SNP (C > A) in the sucrose synthase (Cnu09G011120) was annotated to possess the stop-gain effect that leads to the prematurely stop codon present in the exon 2, suggesting that this change may be critical for the increase in sucrose content of coconut water. Likewise, the hexose carrier protein (Cnu01G018720) may also affect the sugar metabolism in the coconut water. Hexose carrier proteins, also known as hexose transporters or sugar transporters, are integral to various physiological processes related to sugar metabolism [24]. The SNP (A > G) in this gene leads to the amino acid change (Met281Val) that may alter the activity of the enzyme and thereby directly modulate sucrose content. However, further studies are still needed to confirm the function of these two genes in sucrose metabolism in coconut. Nevertheless, the results from our study are crucial for the development of molecular markers that can be used in marker-assisted selection (MAS) to breed coconut varieties with improved sweetness that meet market demands and support the economic sustainability of coconut cultivation. In summary, this research provides a fundamental understanding of genetic control over coconut sweetness and offers a pathway for genetic improvement of coconut cultivars through targeted breeding strategies. The integration of genomic tools with traditional breeding approaches promises to improve the commercial viability and attractiveness of coconut products and support the sustainable development of the coconut industry.
4. Materials and Methods
4.1. Plant Materials
The coconut population used for this population study and allele mining consisted of 49 coconut accessions, including 32 accessions of tall coconut and 17 accessions of dwarf coconut. A total of 81 F2 plants from the cross of a tall coconut and an aromatic coconut were also used to validate the candidate SNPs. The 49 coconut accessions were grown at the KU-BEDO Coconut Biobank, Kasetsart University, Kamphaeng Saen Campus, Nakhon Pathom, Thailand. The F2 population was grown at the Trang Horticulture Research Center, Trang, Thailand. The plants were irrigated daily and fertilized every three months with chicken manure as organic fertilizer. Only one plant per accession was used to perform the experiment.
4.2. DNA Extraction, Whole-Genome Sequencing and Variant Calling
Young fresh coconut leaves were collected for DNA extraction. High-quality DNA from coconut leaf samples was extracted using the DNeasy Plant Mini Kit (Qiagen, Hilden, Germany). Whole genome sequencing (WGS) was performed using MGI-seq technology. The WGS process was performed by China National GeneBank (CNGB), Shenzhen, China. The sequence data obtained from whole genome sequencing of 49 coconut accessions and 81 F2 individuals were analyzed to identify variants, SNPs and InDels across the genome using the GATK software suite (4.6.0.0) [25]. The reference genome used for read mapping and variant calling was an unpublished reference genome of an aromatic dwarf coconut.
4.3. Population Structure Analysis
The genetic distance among the 49 coconut accessions was determined using Nei’s standard dissimilarity distance, based on 18,379,163 filtered SNPs, and a UPGMA phylogenetic tree was constructed with 500 bootstraps in MEGA X [26]. STRUCTURE analysis utilized a Bayesian model-based clustering algorithm in STRUCTURE version 2.3.4 [27], applying the admixture model with correlated allele frequencies. Each genetic cluster (K) value (K = 1–10) was analyzed in three independent runs, with a burn-in of 100,000 iterations followed by 100,000 Markov chain Monte Carlo (MCMC) repetitions. LnP(D) values were plotted to identify the ∆K plateau [28]. Genotypes were assigned to subpopulations if their probability of membership (Q value) was 0.70 or higher, while accessions with Q < 0.70 were considered genetically admixed. Principal coordinate analysis (PCoA) was conducted using DARwin 6.0.21 [29], and the final results were visualized with the ggplot2 R package [30].
4.4. Sweetness Evaluation and Sugar Profile Analysis
For the evaluation of sweetness, a total of five fruits (7-month-old) were collected from each coconut accession. The coconut water from each fruit was then collected and the total soluble sugar content was measured using a digital handheld reflectometer (MyBrix, Mettler-Toledo (Thailand) Co., Ltd., Bangkok, Thailand). The sweetness was expressed in degrees Brix (°Bx). Analysis of the different sugars from the 14 coconut accessions representing the sweet group and the less-sweet group was performed using high performance anion exchange chromatography with pulsed amperometric detector (HPAEC-PAD), model Dionex ICS-6000 DC and Chromeleon Console software version 7.2.10 (Thermo Fisher Scientific, Waltham, MA, USA). A CarboPac PA1 analytical column (4 × 250 mm) and a CarboPac PA1 guard column (4 × 50 mm) were used together with standard substances for quantification. Each accession was analyzed four times.
4.5. Allele Mining Genes in Sugar Metabolism and Transport and Genotype–Phenotype Association Analysis
The genes involved in sugar metabolism and sugar transport in coconuts were collected based on the gene functional annotations. The SNPs and InDels present in each of these genes were determined and compared among the 49 coconut accessions and 81 F2 individuals. The effects of SNPs and InDels in each gene were estimated using SnpEff [31]. A genotype–phenotype association analysis was performed on the 49 coconut accessions and 81 F2 individuals with a simple regression method using the lm() function in R (http://www.r-project.org, accessed on 1 June 2024). The genotype data used for the analysis were the SNPs and InDels in the genes and the phenotype data were the sugar content measured in degrees Brix (°Bx).
5. Conclusions
Our results provide valuable insights into the genetic control of coconut sweetness. The two significant SNPs identified in the sucrose synthase and hexose carrier genes could be used in breeding programs to develop better coconut varieties with improved sweet tastes, ultimately meeting consumer demands and supporting the economic viability of coconut breeding. This research not only contributes to our understanding of coconut biology but also has practical implications for the coconut industry, where there is a growing demand for high-quality sweet coconut water.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/plants13182548/s1, Table S1: The CDS sequence of Cnu09G011120 (sucrose synthase) shows a nucleotide change causing a premature stop codon; Table S2: List of genes involved in sucrose metabolism and sugar transport; Table S3: List of functional variants in the genes involved in sugar metabolism and transport tested in 49 coconut accessions.
Author Contributions
Conceptualization, S.A., S.W., V.R., Y.X. and R.X.; methodology, S.W. and A.P.; formal analysis, W.A., M.K., R.D. and N.P.; resources, T.K.; data curation, S.W.; writing—original draft preparation, M.K. and M.K.P.; writing—review and editing, S.W. and S.A.; supervision, S.A., S.W. and V.R.; funding acquisition, S.A. and Y.X. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Kasetsart University Research and Development Institute, KURDI, grant number FF (KU) 10.65; the Agriculture Research Development Agency (Public Organization), grant number PRP6705030710; the Scientific and Technological Cooperation Projects of Hainan Province (No. ZDYF2020215); Kasetsart University through the Graduate School Fellowship Program.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The SNP dataset presented in this study can be found in Zenodo repository (https://doi.org/10.5281/zenodo.13352204). The raw sequencing data generated in this study have been deposited with links to BioProject accession number PRJNA1153526 in the NCBI BioProject database (https://www.ncbi.nlm.nih.gov/bioproject/ (accessed on 1 June 2024)).
Acknowledgments
We would like to thank Chumphon Horticulture Research Center and Trang Horticulture Research Center for coconut plant materials used in this study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Muralidharan, K.; Jayashree, A. Value addition, product diversification and by-product utilization in coconut. Indian Coconut J. 2011, 7, 4–10. [Google Scholar]
- Lédo, A.d.S.; Passos, E.E.M.; Fontes, H.R.; Ferreira, J.M.S.; Talamini, V.; Vendrame, W.A. Advances in Coconut palm propagation. Rev. Bras. Frutic. 2019, 41, e-159. [Google Scholar] [CrossRef]
- Ignacio, I.-F.; Miguel, T.-S. Research opportunities on the coconut (Cocos nucifera L.) using new technologies. S. Afr. J. Bot. 2021, 141, 414–420. [Google Scholar] [CrossRef]
- Gunn, B.F.; Baudouin, L.; Olsen, K.M. Independent origins of cultivated coconut (Cocos nucifera L.) in the old world tropics. PLoS ONE 2011, 6, e21143. [Google Scholar] [CrossRef]
- Perera, L.; Perera, S.A.; Bandaranayake, C.K.; Harries, H.C. Coconut. In Oil Crops; Springer: New York, NY, USA, 2010; pp. 369–396. [Google Scholar]
- Riangwong, K.; Wanchana, S.; Aesomnuk, W.; Saensuk, C.; Nubankoh, P.; Ruanjaichon, V.; Kraithong, T.; Toojinda, T.; Vanavichit, A.; Arikit, S. Mining and validation of novel genotyping-by-sequencing (GBS)-based simple sequence repeats (SSRs) and their application for the estimation of the genetic diversity and population structure of coconuts (Cocos nucifera L.) in Thailand. Hortic. Res. 2020, 7, 156. [Google Scholar] [CrossRef]
- Kalaipandian, S.; Mu, Z.; Kong, E.Y.Y.; Biddle, J.; Cave, R.; Bazrafshan, A.; Wijayabandara, K.; Beveridge, F.C.; Nguyen, Q.; Adkins, S.W. Cloning Coconut via Somatic Embryogenesis: A Review of the Current Status and Future Prospects. Plants 2021, 10, 2050. [Google Scholar] [CrossRef]
- Dumhai, R.; Wanchana, S.; Saensuk, C.; Choowongkomon, K.; Mahatheeranont, S.; Kraithong, T.; Toojinda, T.; Vanavichit, A.; Arikit, S. Discovery of a novel CnAMADH2 allele associated with higher levels of 2-acetyl-1-pyrroline (2AP) in yellow dwarf coconut (Cocos nucifera L.). Sci. Hortic. 2019, 243, 490–497. [Google Scholar] [CrossRef]
- Burns, D.T.; Johnston, E.L.; Walker, M.J. Authenticity and the potability of coconut water-a critical review. J. AOAC Int. 2020, 103, 800–806. [Google Scholar] [CrossRef]
- Yong, J.W.H.; Ge, L.; Ng, Y.F.; Tan, S.N. The chemical composition and biological properties of coconut (Cocos nucifera L.) water. Molecules 2009, 14, 5144–5164. [Google Scholar] [CrossRef]
- Ab Mutalib, S.R.; Jailani, F. Physicochemical properties and sensory acceptability of different varieties of coconut water and flesh. Sci. Res. J. 2022, 19, 75. [Google Scholar] [CrossRef]
- Assa, R.R.; Prades, A.; Konan, A.G.; Nemlin, J.; Konan, J.-L. Sensory evaluation and sugars contents of coconut (Cocos nucifera L.) water during nuts ripening. Afr. J. Food Sci. 2013, 7, 186–192. [Google Scholar]
- Li, P.; Wang, L.; Liu, H.; Yuan, M. Impaired SWEET-mediated sugar transportation impacts starch metabolism in developing rice seeds. Crop J. 2022, 10, 98–108. [Google Scholar] [CrossRef]
- Wu, Y.; Lee, S.-K.; Yoo, Y.; Wei, J.; Kwon, S.-Y.; Lee, S.-W.; Jeon, J.-S.; An, G. Rice transcription factor osdof11 modulates sugar transport by promoting expression of sucrose transporter and SWEET genes. Mol. Plant 2018, 11, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Rolland, F.; Moore, B.; Sheen, J. Sugar sensing and signaling in plants. Plant Cell 2002, 14, S185–S205. [Google Scholar] [CrossRef]
- Shangguan, L.; Song, C.; Leng, X.; Kayesh, E.; Sun, X.; Fang, J. Mining and comparison of the genes encoding the key enzymes involved in sugar biosynthesis in apple, grape, and sweet orange. Sci. Hortic. 2014, 165, 311–318. [Google Scholar] [CrossRef]
- Nguyen-Quoc, B.; Foyer, C.H. A role for “futile cycles” involving invertase and sucrose synthase in sucrose metabolism of tomato fruit. J. Exp. Bot. 2001, 52, 881–889. [Google Scholar] [CrossRef]
- Aba, R.P.M.; Garcia, P.Y.Q.; Juan, J.K.B.; Linsangan, A.T. Influence of food safety knowledge, attitudes, and practices (KAP) of vendors in the City of Manila on microbiological quality of ready-to-drink coconut water. Food Humanit. 2023, 1, 119–127. [Google Scholar] [CrossRef]
- Badenes, M.L.; Fernández I Martí, A.; Ríos, G.; Rubio-Cabetas, M.J. Application of genomic technologies to the breeding of trees. Front. Genet. 2016, 7, 198. [Google Scholar] [CrossRef]
- Lokeshkumar, B.M.; Katral, A.; Sunitha, N.C.; Sah, R.P.; Krishnamurthy, S.L.; Molla, K.A.; Anilkumar, C. Allele mining in rice. In Allele Mining for Genomic Designing of Cereal Crops; CRC Press: Boca Raton, FL, USA, 2024; pp. 39–59. ISBN 9781003385004. [Google Scholar]
- Huang, Y.-C.; Hsiang, E.-C.; Yang, C.-C.; Wang, A.-Y. New insight into the catalytic properties of rice sucrose synthase. Plant Mol. Biol. 2016, 90, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Stein, O.; Granot, D. An overview of sucrose synthases in plants. Front. Plant Sci. 2019, 10, 95. [Google Scholar] [CrossRef] [PubMed]
- Coleman, H.D.; Yan, J.; Mansfield, S.D. Sucrose synthase affects carbon partitioning to increase cellulose production and altered cell wall ultrastructure. Proc. Natl. Acad. Sci. USA 2009, 106, 13118–13123. [Google Scholar] [CrossRef]
- Geiger, D. Plant glucose transporter structure and function. Pflügers Arch. 2020, 472, 1111–1128. [Google Scholar] [CrossRef]
- Van der Auwera, G.A.; Carneiro, M.O.; Hartl, C.; Poplin, R.; Del Angel, G.; Levy-Moonshine, A.; Jordan, T.; Shakir, K.; Roazen, D.; Thibault, J.; et al. From FastQ data to high confidence variant calls: The Genome Analysis Toolkit best practices pipeline. Curr. Protoc. Bioinform. 2013, 11, 11.10.1–11.10.33. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef]
- Earl, D.A.; vonHoldt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Perrier, X. DARwin Software. 2006. Available online: http://darwin.cirad.fr (accessed on 1 June 2024).
- Villanueva, R.A.M.; Chen, Z.J. ggplot2: Elegant Graphics for Data Analysis (2nd ed.). Meas. Interdiscip. Res. Perspect. 2019, 17, 160–167. [Google Scholar] [CrossRef]
- Cingolani, P.; Platts, A.; Wang, L.L.; Coon, M.; Nguyen, T.; Wang, L.; Land, S.J.; Lu, X.; Ruden, D.M. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 2012, 6, 80–92. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).