Romanian Viscum album L.—Untargeted Low-Molecular Metabolomic Approach to Engineered Viscum–AuNPs Carrier Assembly
Abstract
1. Introduction
2. Results and Discussion
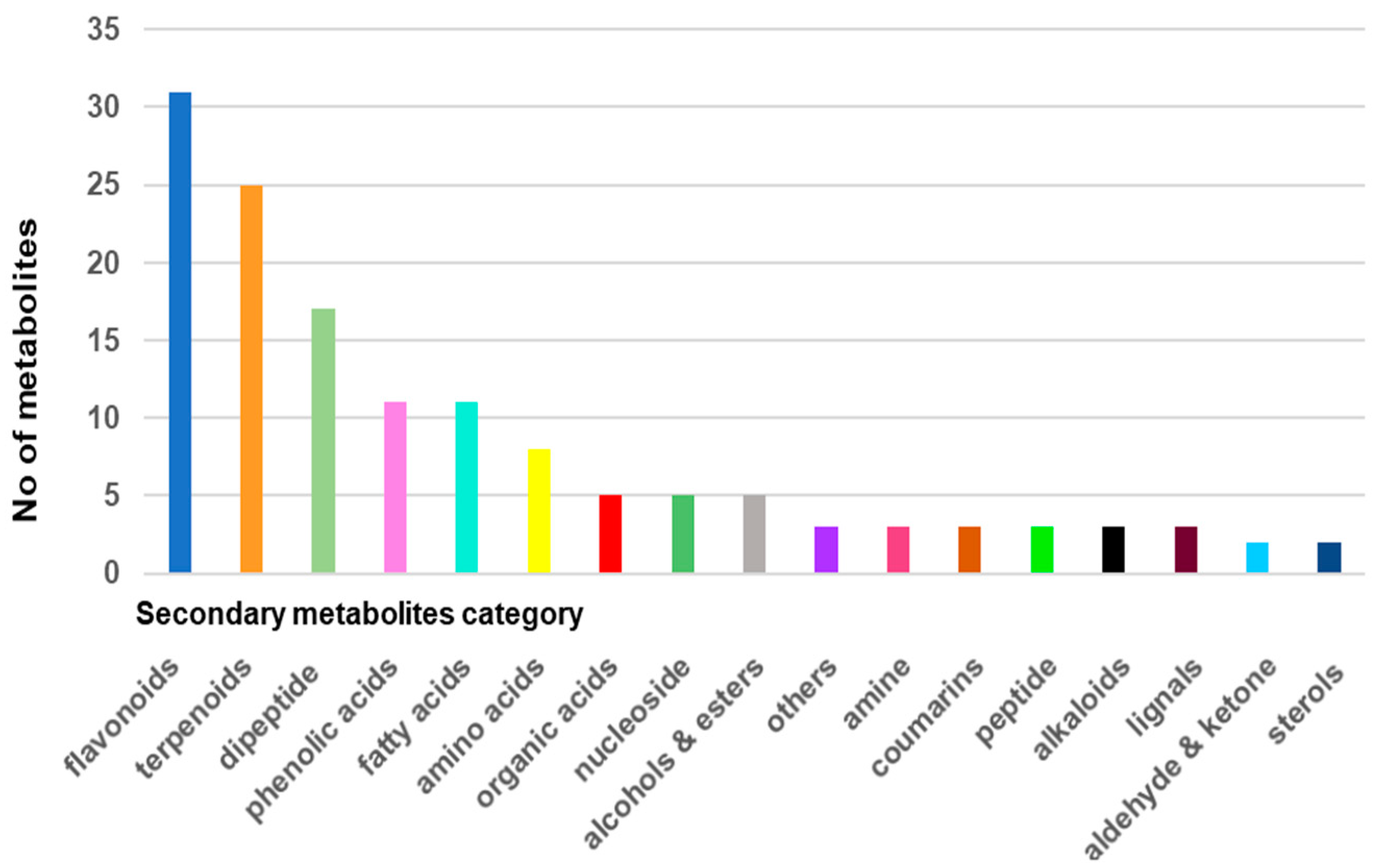
2.1. Screening and Classification of the Differential Metabolites
2.2. Engineered Viscum–AuNPs Carrier Assembly
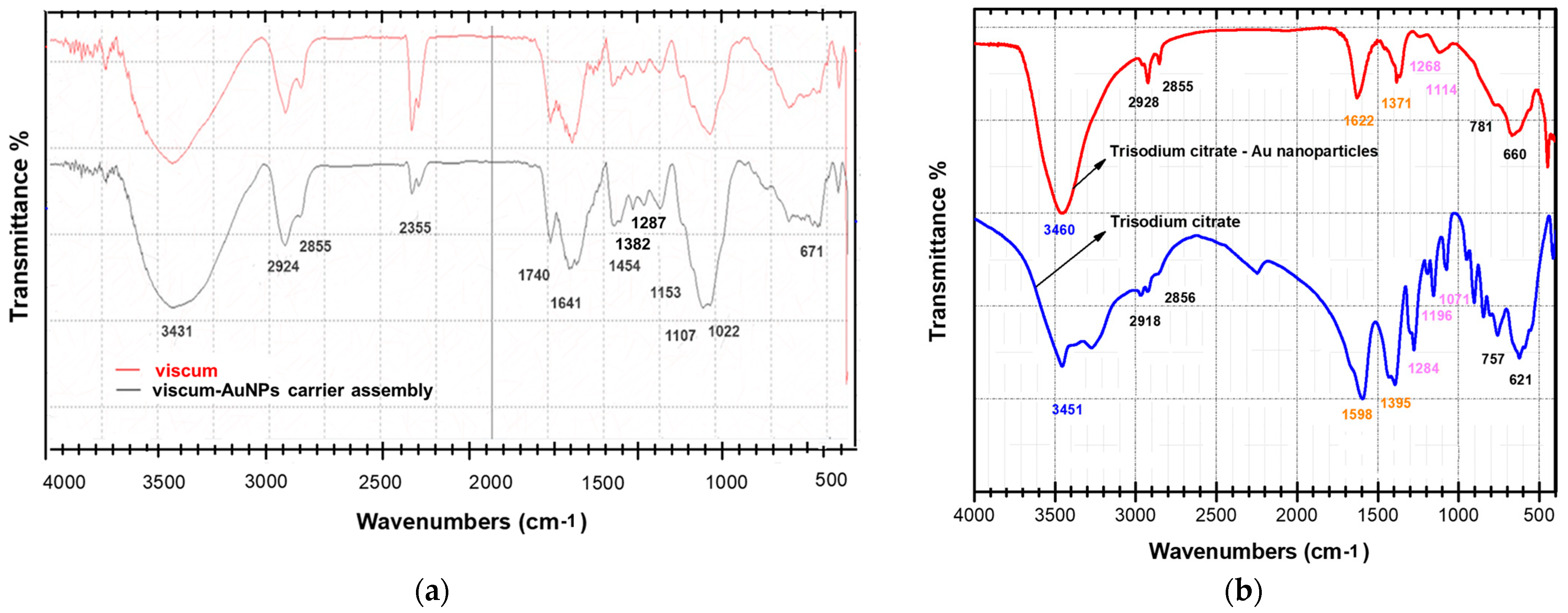
2.3. FT-IR Spectroscopy
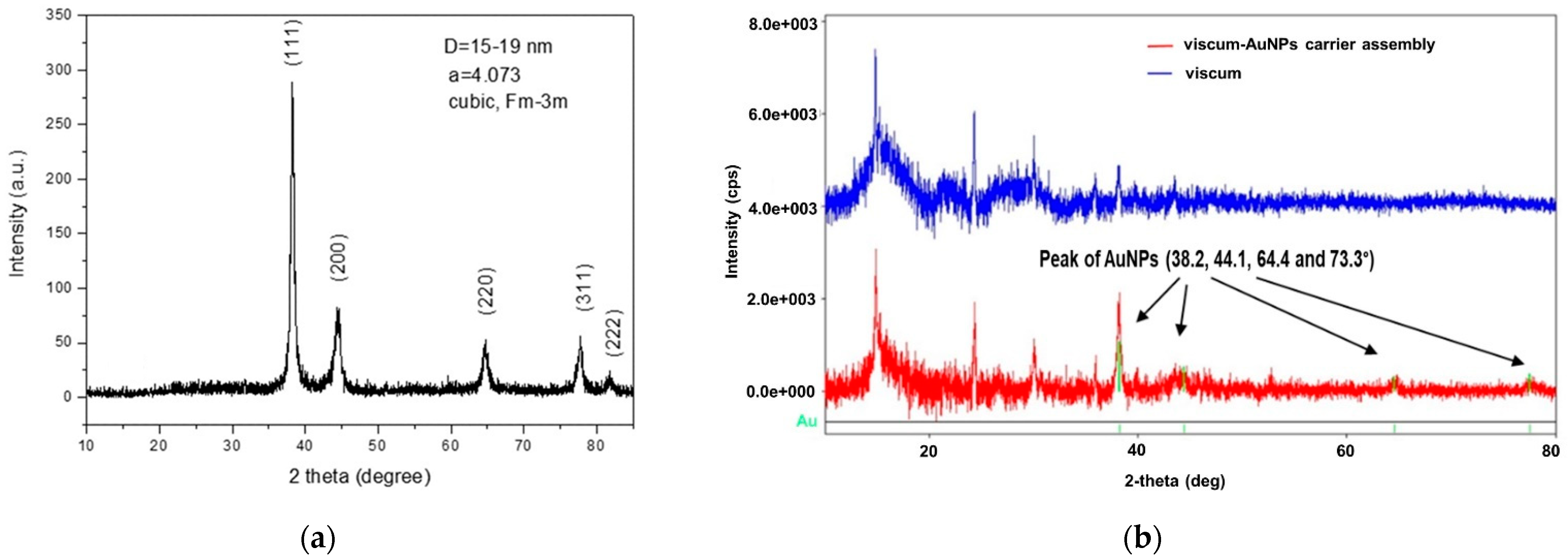
2.4. X-ray Diffraction Spectroscopy
2.5. Scanning Electron Microscopy (SEM)
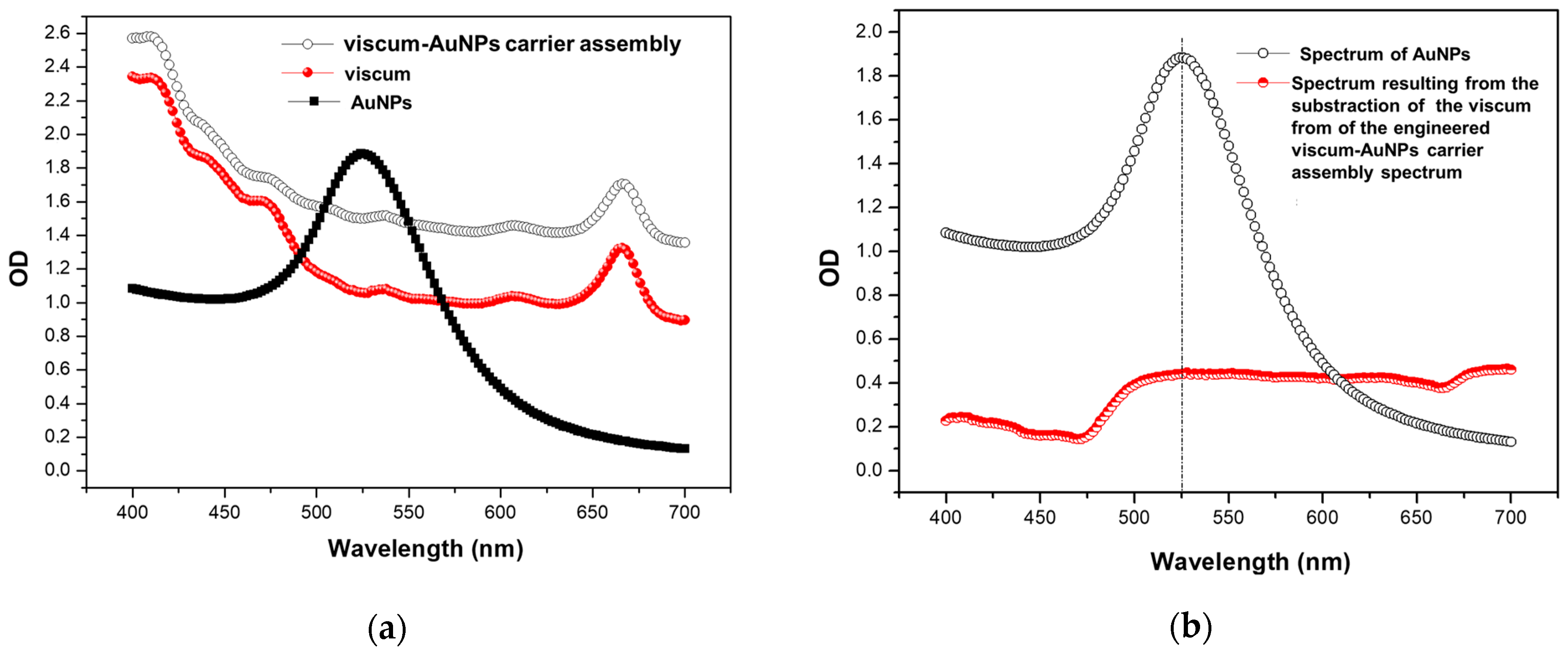
2.6. UV–VIS Spectroscopy
3. Materials and Methods
3.1. Carrier Assembly Components Preparation
AuNPs Synthesis
3.2. Plant Sample Preparation
3.3. Plant Preparation for Chemical Screening
3.4. Mass Spectrometry
3.5. Phyto-Engineered AuNPs Carrier Assembly Preparation
3.6. Characterisation of the Engineered Viscum–AuNPs Carrier Assembly
3.6.1. UV–Vis Analysis
3.6.2. Fourier Transform Infrared (FTIR) Spectroscopy
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barlow, B. Mistletoe in Folk Legend and Medicine. 2008. Available online: https://www.anbg.gov.au/mistletoe/folk-legend.html (accessed on 2 June 2022).
- Frazer, J.G. The Golden Bough: A Study in Magic and Religion; Palgrave Macmillan: London, UK, 1990. [Google Scholar]
- Gill, L.S.; Hawksworth, F.G. The Mistletoes: A Literature Review; U.S. Dept. Agr. Tech. Bul. 1242. U.S.; Government Printing Office: Washington, DC, USA, 1961. Available online: https://handle.nal.usda.gov/10113/CAT87201185 (accessed on 2 June 2022).
- Ogunmefun, O.T.; Olatunji, B.P.; Adarabioyo, M.I. Ethnomedicinal Survey on the Uses of Mistletoe in South-Western Nigeria. Eur. J. Med. Plants 2015, 8, 224–230. [Google Scholar] [CrossRef]
- Segneanu, A.; Velciov, S.M.; Olariu, S.; Cziple, F.; Damian, D.; Grozescu, I. Bioactive molecules profile from natural compounds. In Amino Acid-New Insights and Roles in Plant and Animal; Asao, T., Asaduzzaman, M., Eds.; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar]
- Szurpnicka, A.; Kowalczuk, A.; Szterk, A. Biological activity of mistletoe: In vitro and in vivo studies and mechanisms of action. Arch. Pharm. Res. 2020, 43, 593–629. [Google Scholar] [CrossRef]
- Kleszken, E.; Timar, A.V.; Memete, A.R.; Miere (Groza), F.; Vicas, S.I. On overview of bioactive compounds, biological and pharmacological effects of mistletoe (Viscum album L.). Pharmacophore 2022, 13, 10–26. [Google Scholar] [CrossRef]
- Freuding, M.; Keinki, C.; Micke, O.; Buentzel, J.; Huebner, J. Mistletoe in oncological treatment: A systematic review. J. Cancer Res. Clin. Oncol. 2019, 145, 695–707. [Google Scholar] [CrossRef] [PubMed]
- Mazalovska, M.; Calvin Kouokam, J. Plant-derived lectins as potential cancer therapeutics and diagnostic tools. BioMed Res. Int. 2020, 2020, 1–13. [Google Scholar] [CrossRef]
- Pietrzak, W.; Nowak, R. Impact of harvest conditions and host tree species on chemical composition and antioxidant activity of extracts from Viscum album L. Molecules 2021, 26, 3741. [Google Scholar] [CrossRef]
- Jäger, T.; Holandino, C.; Melo, M.N.D.O.; Peñaloza, E.M.C.; Oliveira, A.P.; Garrett, R.; Glauser, G.; Grazi, M.; Ramm, H.; Urech, K.; et al. Metabolomics by UHPLC-Q-TOF Reveals Host Tree-Dependent Phytochemical Variation in Viscum album L. Plants 2021, 10, 1726. [Google Scholar] [CrossRef]
- Majeed, M.; Rehman, R.U. Phytochemistry, Pharmacology, and Toxicity of an Epiphytic Medicinal Shrub Viscum album L. (White Berry Mistletoe). In Medicinal and Aromatic Plants; Aftab, T., Hakeem, K.R., Eds.; Springer: Cham, Switzerland, 2021; pp. 287–301. [Google Scholar]
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The traditional medicine and modern medicine from natural products. Molecules 2016, 21, 559. [Google Scholar] [CrossRef]
- Loef, M.; Walach, H. Quality of life in cancer patients treated with mistletoe: A systematic review and meta-analysis. BMC Complement Med. Ther. 2020, 20, 227. [Google Scholar] [CrossRef]
- Oei, S.L.; Thronicke, A.; Schad, F. Mistletoe and immunomodulation: Insights and implications for anticancer therapies. Evid.-Based Complementary Altern. Med. 2019, 2019, 1–6. [Google Scholar] [CrossRef]
- Marvibaigi, M.; Supriyanto, E.; Amini, N.; Abdul Majid, F.A.; Jaganathan, S.K. Preclinical and clinical effects of mistletoe against breast cancer. Biomed Res. Int. 2014, 2014, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Urech, K.; Schaller, G.; Jäggy, C. Viscotoxins, mistletoe lectins and their isoforms in mistletoe (Viscum album L.) extracts Iscador. Arzneimittelforschung 2011, 56, 428–434. [Google Scholar] [CrossRef]
- Vang Petersen, P. Warrior art, religion and symbolism. In The Spoils of Victory. The North in the Shadow of the Roman Empire; Jørgensen, L., Storgaard, B., Thomsen, L.G., Eds.; National Museet: Copenhagen, Denmark, 2003; pp. 286–293. [Google Scholar]
- Available online: https://finds.org.uk/staffshoardsymposium/papers/charlottebehr (accessed on 2 June 2022).
- Huaizhi, Z.; Yuantao, N. China’s ancient gold drugs. Gold Bull. 2001, 34, 24–29. [Google Scholar] [CrossRef]
- Patil-Bhole, T.; Wele, A.; Gudi, R.; Thakur, K.; Nadkarni, S.; Panmand, R.; Kale, B. Nanostructured gold in ancient Ayurvedic calcined drug ‘swarnabhasma’. J. Ayurveda Integr. Med. 2021, 12, 640–648. [Google Scholar] [CrossRef]
- Sharma, R.; Prajapati, P.K. Nanotechnology in medicine: Leads from Ayurveda. J. Pharma Bioallied Sci. Jan.-Mar. 2016, 8, 80–81. [Google Scholar] [CrossRef] [PubMed]
- Parimalam, S.S.; Badilescu, S.; Bhat, R.; Packirisamy, M. A narrative review of scientific validation of gold- and silver-based Indian medicines and their future scope. Longhua Chin. Med. 2020, 3, 10. [Google Scholar] [CrossRef]
- Metwaly, A.M.; Ghoneim, M.M.; Eissa, I.H.; Elsehemy, I.A.; Mostafa, A.E.; Hegazy, M.M.; Afifi, W.M.; Dou, D. Traditional ancient Egyptian medicine: A review. Saudi J. Biol. Sci. 2021, 28, 5823–5832. [Google Scholar] [CrossRef]
- Forshaw, R.J. The practice of dentistry in ancient Egypt. Br. Dent. J. 2009, 206, 481–486. [Google Scholar] [CrossRef]
- Console, R. Pharmaceutical use of gold from antiquity to the seventeenth century. Geol. Soc. Lond. Spec. Publ. 2013, 375, 171–191. [Google Scholar] [CrossRef]
- Pricker, S.P. Medical uses of gold compounds: Past, present and future. Gold Bull. 1996, 29, 53–60. [Google Scholar] [CrossRef]
- Balfourier, A.; Kolosnjaj-Tabi, J.; Luciani, N.; Carn, F.; Gazeau, F. Gold-based therapy: From past to present. Proc. Natl. Acad. Sci. USA 2020, 117, 22639–22648. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.H.; Su, C.H.; Wu, Y.J.; Lin, C.A.J.; Lee, C.H.; Shen, J.L.; Yeh, H.I. Application of gold in biomedicine: Past, present and future. Int. J. Gerontol. 2012, 6, 1–4. [Google Scholar] [CrossRef]
- Medici, S.; Peana, M.F.; Zoroddu, M.A. Noble metals in pharmaceuticals: Applications and limitations. In Biomedical Applications of Metals; Rai, M., Ingle, A., Medici, S., Eds.; Springer: Cham, Switzerland, 2018; pp. 3–48. [Google Scholar]
- Ott, I. On the medicinal chemistry of gold complexes as anticancer drugs. Coord. Chem. Rev. 2009, 253, 1670–1681. [Google Scholar] [CrossRef]
- Goddard, Z.R.; Marín, M.J.; Russell, D.A.; Searcey, M. Active targeting of gold nanoparticles as cancer therapeutics. Chem. Soc. Rev. 2020, 49, 8774–8789. [Google Scholar] [CrossRef] [PubMed]
- Yafout, M.; Ousaid, A.; Khayati, Y.; El Otmani, I.S. Gold nanoparticles as a drug delivery system for standard chemotherapeutics: A new lead for targeted pharmacological cancer treatments. Sci. Afr. 2021, 11, e00685. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, D.; Zhang, C.; Liu, H.; Hao, M.; Kan, S.; Liu, D.; Liu, W. The Applications of gold nanoparticles in the diagnosis and treatment of gastrointestinal cancer. Front. Oncol. 2022, 11, 819329. [Google Scholar] [CrossRef]
- Vlad, D.C.; Popescu, R.; Dumitrascu, V.; Cimporescu, A.; Vlad, C.S.; Vágvölgyi, C.; Krisch, J.; Dehelean, C.; Horhat, F.G. Phytocomponents identification in mistletoe (Viscum album) young leaves and branches, by GC-MS and antiproliferative effect on HEPG2 And MCF7 cell lines. Farmacia 2016, 64, 82–87. [Google Scholar]
- Paun, G.; Rotinberg, P.; Mihai, C.; Neagu, E.; Radu, G.L. Cytostatic activity of Viscum album L. extract processed by microfiltration and ultrafiltration. Rom. Biotechnol. Lett. 2011, 16, 6000–6007. [Google Scholar]
- Vicas, S.I.; Socaciu, C. The biological activity of European mistletoe (Viscum album) extracts and their pharmaceutical impact. Bull. USAMV-CN 2007, 63, 217–222. [Google Scholar]
- Sevastre, B.; Olah, N.K.; Hanganu, D.; Sárpataki, O.; Taulescu, M.; Mănălăchioae, R.; Marcus, I.; Cătoi, C. Viscum album L. alcoholic extract enhance the effect of doxorubicin in ehrlich carcinoma tumor cells. Rom. Biotechnol. Lett. 2012, 17, 6976–6981. [Google Scholar]
- Stan, R.L.; Hangan, A.C.; Dican, L.; Sevastre, B.; Hanganu, D.; Catoi, C.; Sarpataki, O.; Ionescu, C.M. Comparative study concerning mistletoe viscotoxins antitumor activity. Acta Biol. Hung. 2013, 64, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Vicaş, S.I.; Rugina, D.; Leopold, L.; Pintea, A.; Socaciu, C. HPLC Fingerprint of bioactive compounds and antioxidant activities of Viscum album from different host trees. Not. Bot Hort Agrobot Cluj 2011, 39, 48–57. [Google Scholar] [CrossRef]
- Yarnell, E. Synergy in Herbal Medicines: Part. J. Restor. Med. 2015, 1, 460–473. [Google Scholar] [CrossRef]
- Zhao, Q.; Luan, X.; Zheng, M.; Tian, X.H.; Zhao, J.; Zhang, W.D.; Ma, B.L. Synergistic mechanisms of constituents in herbal extracts during intestinal absorption: Focus on natural occurring nanoparticles. Pharmaceutics 2020, 12, 128. [Google Scholar] [CrossRef]
- Hussein, R.A.; El-Anssary, A.A. Plants secondary metabolites: The key drivers of the pharmacological actions of medicinal plants. In Herbal Medicine; Philip, F., Builders, Eds.; IntechOpen: Rijeka, Croatia, 2018. [Google Scholar]
- Chen, G.; Wang, S.; Huang, X.; Hong, J.; Du, L.; Zhang, L.; Ye, L. Environmental factors affecting growth and development of Banlangen (Radix Isatidis) in China. Afr. J. Plant. Sci. 2015, 9, 421–426. [Google Scholar]
- Pang, Z.; Chen, J.; Wang, T.; Gao, C.; Li, Z.; Guo, L.; Xu, J.; Cheng, Y. Linking plant secondary metabolites and plant microbiomes: A Review. Front. Plant. Sci. 2021, 12, 621276. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, Y.L.; Yang, Y.P.; Li, X.L. Chemical constituents of Viscum album var. meridianum. Biochem. Syst. Ecol. 2011, 39, 849–852. [Google Scholar] [CrossRef]
- Singh, B.N.; Saha, C.; Galun, D.; Dalip, K.U.; Bayry, J.; Kaveri, S.V. European Viscum album: A potent phytotherapeutic agent with multifarious phytochemicals, pharmacological properties and clinical evidence. RSC Adv. R. Soc. Chem. 2016, 6, 23837–23857. [Google Scholar] [CrossRef]
- Urech, K.; Baumgartner, S. Chemical Constituents of Viscum Album L. Implications for the Pharmaceutical Preparation of Mistletoe in Mistletoe: From Mythology to Evidence-Based Medicine; Zanker, K.S., Kaveri, S.V., Eds.; Transl Res Biomed; Karger: Basel, Switzerland, 2015; Volume 4, pp. 11–23. [Google Scholar]
- Song, C.; Wei, X.-Y.; Qiu, Z.-D.; Gong, L.; Chen, Z.-Y.; Ma, Y.; Shen, Y.; Zhao, Y.-J.; Wang, W.-H.; Lai, C.-J.-S.; et al. Exploring the resources of the genus Viscum for potential therapeutic applications. J. Ethnopharmacol. 2021, 277, 114233. [Google Scholar] [CrossRef]
- Patel, B.P.; Singh, P.K. Viscum articulatum Burm. F.: A review on its phytochemistry, pharmacology and traditional uses. J. Pharm Pharm. 2018, 70, 159–177. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, D.; Zhang, Q.; Qin, D.; Jiang, X.; Li, H.; Fang, l.; Cao, J.; Wu, H. Volatile components and nutritional qualities of Viscum articulatum Burm.f. parasitic on ancient tea trees. Food Sci. Nutr. 2019, 7, 3017–3029. [Google Scholar] [CrossRef] [PubMed]
- Peñaloza, E.; Holandino, C.; Scherr, C.; de Araujo, P.I.P.; Borges, R.M.; Urech, K.; Baumgartner, S.; Garrett, R. Comprehensive metabolome analysis of fermented aqueous extracts of Viscum album L. by Liquid chromatography-high resolution tandem mass spectrometry. Molecules 2020, 25, 4006. [Google Scholar] [CrossRef] [PubMed]
- Stefanucci, A.; Zengin, G.; Llorent-Martinez, E.J.; Dimmito, M.P.; Della Valle, A.; Pieretti, S.; Gunes, A.K.; Sinan, K.I.; Mollica, A. Viscum album L. homogenizer-assisted and ultrasound-assisted extracts as potential sources of bioactive compounds. J. Food Biochem. 2020, 44, e13377. [Google Scholar] [CrossRef] [PubMed]
- Holandino, C.; Melo, M.N.; Oliveira, A.P.; da Costa Batista, J.V.; Capella, M.A.M.; Garrett, R.; Grazi, M.; Ramm, H.; Torre, C.D.; Schaller, G.; et al. Phytochemical analysis and in vitro anti-proliferative activity of Viscum album ethanolic extracts. BMC Complement. Med. 2020, 20, 215. [Google Scholar] [CrossRef]
- Chandra, K. Bio Active Compounds Isolated from Mistletoe (Scurulla Oortiana (Korth) Danser Parasiting Tea Plant Camellia sinenis L.). Master’s Thesis, University of Adelaide, Adelaide, Australia, 1996. [Google Scholar]
- Fukunaga, T.; Kajikawa, I.; Nashiya, K.; Watanabe, Y.; Suzuki, N.; Takeya, K.; Itokawa, H. Studies on the constituents of the European mistletoe, Viscum album L.II. Chem. Pharm. Bull. 1988, 36, 1185–1189. [Google Scholar] [CrossRef][Green Version]
- Maher, S.; Fayyaz, S.; Naheed, N.; Dar, Z. The Isolation and screening of the bioactive compound of Viscum album against Meloidogyne incognita. Pak. J. Nematol. 2021, 39, 46–51. [Google Scholar] [CrossRef]
- Oluwaseun, A.A.; Ganiyu, O. Antioxidant properties of methanolic extracts of mistletoes (Viscum album) from cocoa and cashew trees in Nigeria. Afr. J. Biotechnol. 2008, 7, 3138–3142. [Google Scholar]
- Park, B.J.; Matsuta, T.; Samejima, H.; Park, C.H. In Chemical constituents of mistletoe (Viscum album L. var. coloratum Ohwi) Sung, J.; Lee, B.D.; Onjo, M. IOSR J. Pharm. Biol. Sci. 2017, 12, 19–23. [Google Scholar]
- Das, K.; Gezici, S. Secondary plant metabolites, their separation and identification, and role in human disease prevention. Ann. Phytomedicine Int. J. 2018, 7, 13–24. [Google Scholar] [CrossRef]
- Kim, S.-H.; Roszik, J.; Grimm, E.A.; Ekmekcioglu, S. Impact of L-arginine metabolism on immune response and anticancer immunotherapy. Front. Oncol. 2018, 8, 67. [Google Scholar] [CrossRef]
- Chiangjong, W.; Chutipongtanate, S.; Hongeng, S. Anticancer peptide: Physicochemical property, functional aspect and trend in clinical application (Review). Int. J. Oncol. 2020, 57, 678–696. [Google Scholar] [CrossRef] [PubMed]
- Lieu, E.L.; Nguyen, T.; Rhyne, S.; Kim, J. Amino acids in cancer. Exp. Mol. Med. 2020, 52, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Albaugh, V.L.; Pinzon-Guzman, C.; Barbul, A. Arginine metabolism and cancer. Surg. Oncol. March 2017, 115, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Dhama, K.; Karthik, K.; Khandia, R.; Munjal, A.; Tiwari, R.; Rana, R.; Khurana, S.K.; Ullah, S.; Khan, R.U.; Alagawany, M.; et al. Medicinal and therapeutic potential of herbs and plant metabolites/Extracts countering viral pathogens-Current knowledge and future prospects. Curr. Drug Metab. 2018, 19, 236–263. [Google Scholar] [CrossRef]
- Cox-Georgian, D.; Ramadoss, N.; Dona, C.; Basu, C. Therapeutic and medicinal uses of terpenes. In Medicinal Plants; Joshee, N., Dhekney, S., Parajuli, P., Eds.; Springer: Cham, Switzerland, 2019; pp. 333–359. [Google Scholar]
- Fongang Fotsing, Y.S.; Bankeu Kezetas, J.J. Terpenoids as important bioactive constituents of essential oils, In Essential oils—Bioactive Compounds, New Perspectives and Application; Santana de Oliveira, M., Almeida da Costa, W., Gomes Silva, S., Eds.; Intechopen: Rijeka, Croatia, 2020; ISBN 978–1-83962–698–2. [Google Scholar]
- Jahangeer, M.; Fatima, R.; Ashiq, M.; Basharat, A.; Qamar, S.A.; Bilal, M.; Iqbal, H.M. Therapeutic and biomedical potentialities of terpenoids–A Review. J. Pure Appl. Microbiol. 2021, 15, 471–483. [Google Scholar] [CrossRef]
- João Matos, M.; Santana, L.; Uriarte, E.; Abreu, O.A.; Molina, E.; Guardado Yordi, E. Coumarins—An important class of phytochemicals in phytochemicals-Isolation. In Characterisation and Role in Human Health; Venket Rao, A., Rao, L.G., Eds.; IntechOpen: Rijeka, Croatia, 2015. [Google Scholar]
- Küpeli Akkol, E.; Genç, Y.; Karpuz, B.; Sobarzo-Sánchez, E.; Capasso, R. Coumarins and coumarin-related compounds in pharmacotherapy of cancer. Cancers 2020, 12, 1959. [Google Scholar] [CrossRef]
- Kozłowska, A.; Szostak-Węgierek, D. Flavonoids–food sources, health benefits, and mechanisms involved. In Bioactive Molecules in Food; Reference Series in Phytochemistry; Mérillon, J.M., Ramawat, K., Eds.; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Kozłowska, A.; Szostak-Wegierek, D. Flavonoids-food sources and health benefits. Rocz. Panstw. Zakl. Hig. 2014, 65, 79–85. [Google Scholar]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Minatel, I.O.; Vanz Borges, C.; Ferreira, M.I.; Gomez, H.A.G.; Chen, C.Y.O.; Pace Pereira Lima, G. Phenolic compounds: Functional properties, impact of processing and bioavailability. In Phenolic Compounds-Biological Activity; Soto-Hernandez, M., Palma-Tenango, M., del Rosario Garcia-Mateos, M., Eds.; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar]
- Salehi, B.; Quispe, C.; Sharifi-Rad, J.; Cruz-Martins, N.; Nigam, M.; Mishra, A.P.; Konovalov, D.A.; Orobinskaya, V.; Abu-Reidah, I.M.; Zam, W.; et al. Phytosterols: From preclinical evidence to potential clinical applications. Front. Pharmacol. 2021, 11, 599959. [Google Scholar] [CrossRef]
- Nagy, K.; Tiuca, I.D. Importance of fatty acids in physiopathology of human body. In Fatty Acids; Catala, A., Ed.; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar]
- Kilcoyne, M.; Joshi, L. Carbohydrates in therapeutics. Cardiovasc. Hematol. Agents Med. Chem. 2007, 5, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Rajput, A.; Bhatia, A.; Kumar, A.; Kaur, H.; Sharma, P.; Kaur, P.; Singh, S.; Attri, S.; Buttar, H.; et al. Plant-based polysaccharides and their health Functions. Funct. Foods Health Dis. 2021, 11, 179–200. [Google Scholar]
- Hu, X.; Zhang, Y.; Ding, T.; Liu, J.; Zhao, H. Multifunctional gold nanoparticles: A novel nanomaterial for various medical applications and biological activities. Front. Bioeng. Biotechnol. 2020, 8, 990. [Google Scholar] [CrossRef] [PubMed]
- Yeh, Y.C.; Creran, B.; Rotello, V.M. Gold nanoparticles: Preparation, properties, and applications in bionanotechnology. Nanoscale 2012, 4, 1871–1880. [Google Scholar] [CrossRef] [PubMed]
- Siddique, S.; Chow, J.C.L. Gold nanoparticles for drug delivery and cancer therapy. Appl. Sci. 2020, 10, 3824. [Google Scholar] [CrossRef]
- Vega-Vásquez, P.; Mosier, N.S.; Irudayaraj, J. Nanoscale drug delivery systems: From Medicine to Agriculture. Front. Bioeng. Biotechnol. 2020, 8, 79. [Google Scholar] [CrossRef]
- Pala, R.; Anju, V.T.; Dyavaiah, M.; Busi, S.; Nauli, S.M. Nanoparticle-mediated drug delivery for the treatment of cardiovascular diseases. Int. J. Nanomed. 2020, 15, 3741–3769. [Google Scholar] [CrossRef]
- Boomi, P.; Ganesan, R.; Prabu Poorani, G.; Jegatheeswaran, S.; Balakumar, C.; Gurumallesh Prabu, H.; Anand, K.; Marimuthu Prabhu, N.; Jeyakanthan, J.; Saravanan, M. Phyto-engineered gold nanoparticles (AuNPs) with potential antibacterial, antioxidant, and wound healing activities under in vitro and in vivo conditions. Int. J. Nanomed. 2020, 15, 7553–7568. [Google Scholar] [CrossRef]
- Heneczkowski, M.; Kopacz, M.; Nowak, D.; Kuźniar, A. Infrared spectrum analysis of some flavonoids. Acta Pol. Pharm. Nov.-Dec. 2001, 58, 415–420. [Google Scholar]
- Segneanu, A.E.; Marin, C.N.; Ghirlea, I.O.; Feier, C.V.I.; Muntean, C.; Grozescu, I. Artemisia annua Growing Wild in Romania-A metabolite profile approach to target a drug delivery system based on magnetite nanoparticles. Plants 2021, 10, 2245. [Google Scholar] [CrossRef]
- Zhang, Y.C.; Deng, J.; Lin, X.L.; Li, Y.M.; Sheng, H.X.; Xia, B.H.; Lin, L.M. Use of ATR-FTIR spectroscopy and chemometrics for the variation of active components in different harvesting periods of Lonicera japonica. Int. J. Anal. Chem. 2022, 2022, 8850914. [Google Scholar] [CrossRef] [PubMed]
- Mabasa, X.E.; Mathomu, L.M.; Madala, N.E.; Musie, E.M.; Sigidi, M.T. Molecular spectroscopic (FTIR and UV-Vis) and hyphenated chromatographic (UHPLC-qTOF-MS) analysis and in vitro bioactivities of the Momordica balsamina leaf extract. Biochem. Res. Int. 2021, 2021, 2854217. [Google Scholar] [CrossRef] [PubMed]
- Scarsini, M.; Thurotte, A.; Veidl, B.; Amiard, F.; Niepceron, F.; Badawi, M.; Lagarde, F.; Schoefs, B.; Marchand, J. Metabolite quantification by Fourier Transform Infrared Spectroscopy in diatoms: Proof of concept on Phaeodactylum tricornutum. Front. Plant. Sci. 2021, 12, 756421. [Google Scholar] [CrossRef]
- Topală, C.M.; Tătarua, L.D.; Ducu, C. ATR-FTIR spectra fingerprinting of medicinal herbs extracts prepared using microwave extraction. Arab. J. Med. Aromat. Plants AJMAP 2017, 3, 1–9. [Google Scholar]
- Seuvre, A.M.; Mathlouthi, M. FT-IR. spectra of oligo- and poly-nucleotides. Carbohydr. Res. 1987, 169, 83–103. [Google Scholar] [CrossRef]
- Umashankar, T.; Govindappa, M.; Ramachandra, Y.L.; Rai, P.; Channabasava, S. Isolation and characterization of coumarin isolated from endophyte, Alternaria species-1 of Crotalaria pallida and its apoptotic action on HeLa cancer cell line. Metabolomics 2015, 5, 158. [Google Scholar]
- Salem, M.; Marzouk, M.; El-Kazak, A. Synthesis and characterization of some new coumarins with in vitro antitumor and antioxidant activity and high protective effects against DNA damage. Molecules 2016, 21, 249. [Google Scholar] [CrossRef]
- Baranska, M.; Schulz, H. Chapter 4 Determination of alkaloids through Infrared and Raman Spectroscopy. Alkaloids Chem. Biol. 2009, 67, 217–255. [Google Scholar]
- Pang, M.; Jiang, S.; Cao, L.; Pan, L. Novel synthesis of steryl esters from phytosterols and amino acid. J. Agric. Food Chem. 2011, 59, 10732–10736. [Google Scholar] [CrossRef]
- Thottoli, A.K.; Unni, A.K.A. Effect of trisodium citrate concentration on the particle growth of ZnS nanoparticles. J. Nanostructure Chem. 2013, 3, 56. [Google Scholar] [CrossRef]
- Tao, C. Antimicrobial activity and toxicity of gold nanoparticles: Research progress, challenges and prospects. Lett Appl Microbiol. 2018, 67, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Hitaishi, V.P.; Mazurenko, I.; Vengasseril Murali, A.; de Poulpiquet, A.; Coustillier, G.; Delaporte, P.; Lojou, E. Nanosecond laser–fabricated monolayer of gold nanoparticles on ITO for bioelectrocatalysis. Front. Chem. 2020, 8, 431. [Google Scholar] [CrossRef] [PubMed]
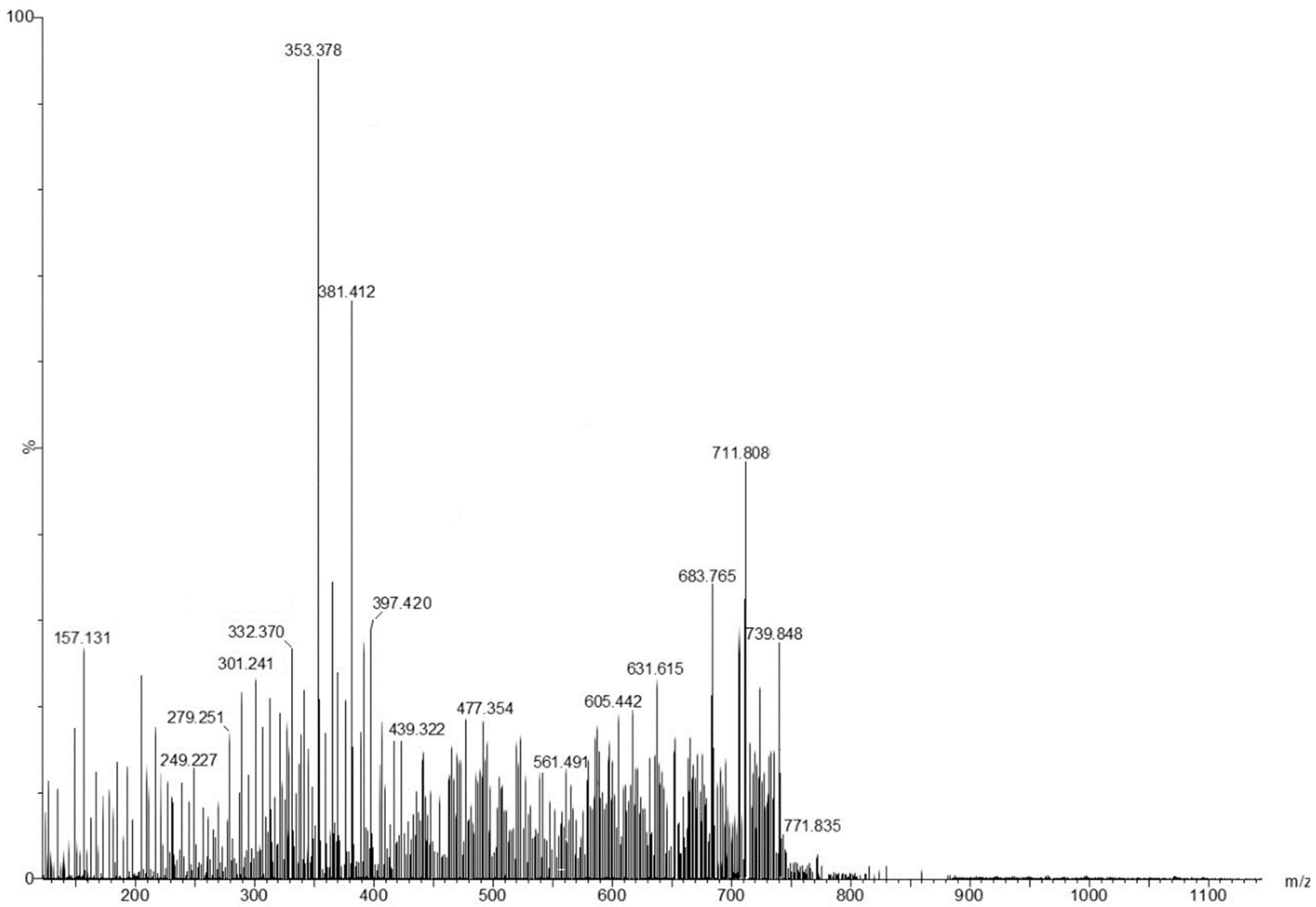



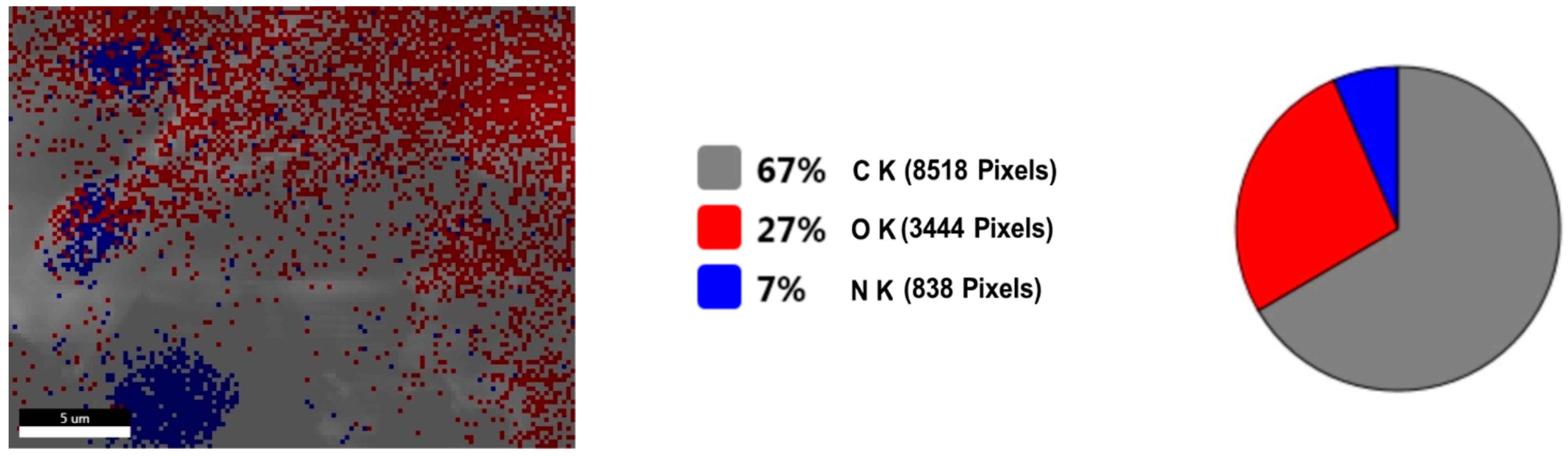

| Compound No. | m/z Detected | Theoretic m/z | Formula | Tentative of Identification | Category | Ref. |
|---|---|---|---|---|---|---|
| 1 | 104.16 | 104.17 | C5H14NO+ | choline | amine | [48] |
| 2 | 111.15 | 111.15 | C5H9N3 | histamine | amine | [48] |
| 3 | 121.14 | 121.16 | C3H7NO2S | cysteine | amino acids | [9] |
| 4 | 122.15 | 122.16 | C8H10O | 2-phenylethanol | alcohol | [50] |
| 5 | 128.22 | 128.21 | C8H16O | 1-octene-3-ol | alcohol | [51] |
| 6 | 131.16 | 131.17 | C6H13NO2 | leucine | amino acids | [52] |
| 7 | 136.22 | 136.23 | C10H16 | sabinene | terpenoids | [51] |
| 8 | 138.11 | 138.12 | C7H6O3 | salicylic acid | phenolic acid | [10,40] |
| 9 | 144.20 | 144.21 | C8H16O2 | octanoic acid | fatty acids | [51] |
| 10 | 146.13 | 146.14 | C9H6O2 | coumarin | coumarins | [49] |
| 11 | 146.22 | 146.21 | C7H16NO2+ | acetylcholine | amine | [48] |
| 12 | 147.12 | 147.13 | C5H9NO4 | glutamic acid | amino acids | [11,52] |
| 13 | 148.15 | 148.16 | C9H8O2 | cinamic acid | phenolic acid | [40] |
| 14 | 149.22 | 149.21 | C5H11NO2S | methionine | amino acids | [52] |
| 15 | 150.21 | 150.22 | C10H14O | safranal | terpenoids | [51] |
| 16 | 152.14 | 152.15 | C8H8O3 | vanillin | aldehydes, phenols | [50] |
| 17 | 152.21 | 152.23 | C10H16O | citral | terpenoids | [51] |
| 18 | 154.11 | 154.12 | C7H6O4 | gentisic acid | phenolic acid | [10,40] |
| 19 | 154.26 | 154.25 | C10H18O | geraniol | terpenoids | [51] |
| 20 | 155.14 | 155.15 | C6H9N3O2 | histidine | amino acids | [52] |
| 21 | 156.21 | 156.22 | C9H16O2 | nonanolide | lactone | [51] |
| 22 | 162.17 | 162.18 | C10H10O2 | cinnamic acid methyl ester | ester | [50] |
| 23 | 164.14 | 164.16 | C9H8O3 | p-coumaric acid | phenolic acid | [40,49] |
| 24 | 166.18 | 166.17 | C9H10O3 | tropic acid | organic acid | [52] |
| 25 | 170.11 | 170.12 | C7H6O5 | gallic acid | phenolic acid | [52] |
| 26 | 174.10 | 174.11 | C6H6O6 | aconic acid | miscellaneous | [52] |
| 27 | 174.19 | 174.2 | C6H14N4O2 | arginine | amino acids | [11,48,52] |
| 28 | 176.11 | 176.12 | C6H8O6 | ascorbic acid | organic acid | [40,49] |
| 29 | 176.16 | 176.17 | C10H8O3 | 6-hydroxy-4-methylcoumarin | coumarins | [52] |
| 30 | 180.15 | 180.16 | C9H8O4 | caffeic acid | phenolic acid | [40,52,53] |
| 31 | 181.18 | 181.19 | C9H11NO3 | tyrosine | amino acids | [11,52] |
| 32 | 182.16 | 182.17 | C9H10O4 | veratric acid | phenolic acid | [10] |
| 34 | 186.11 | 186.12 | C8H14N2O3 | prolyl-alanine | dipeptide | [52] |
| 35 | 188.21 | 188.22 | C8H16N2O3 | leucyl-glycine | dipeptide | [52] |
| 36 | 192.11 | 192.12 | C6H8O7 | citric acid | organic acid | [52] |
| 37 | 193.16 | 192.17 | C7H12O6 | quinic acid | organic acid | [49,53,54] |
| 38 | 192.29 | 192.3 | C13H20O | ionone | ketone | [51] |
| 39 | 194.17 | 194.18 | C10H10O4 | ferulic acid | phenolic acid | [10,40] |
| 40 | 194.30 | 194.31 | C13H22O | geranylacetone | terpenoids | [51] |
| 41 | 196.23 | 196.24 | C11H16O3 | loliolide | terpenoid | [52] |
| 42 | 196.27 | 196.29 | C12H20O2 | nerol acetate | terpenoids | [51] |
| 43 | 198.16 | 198.17 | C9H10O5 | syringic acid | phenolic acid | [40,53] |
| 44 | 202.23 | 202.25 | C9H18N2O3 | leucyl-alanine | dipeptide | [52] |
| 45 | 204.21 | 204.22 | C11H12N2O2 | tryptophan | amino acids | [52] |
| 46 | 204.34 | 204.35 | C15H24 | caryophyllene | terpenoids | [51] |
| 47 | 204.36 | 206.37 | C15H26 | cadinene | terpenoids | [51,52] |
| 48 | 208.19 | 208.21 | C8H16O6 | viscumitol | saccharides | [49] |
| 49 | 216.27 | 216.28 | C10H20N2O3 | valylvaline | dipeptide | [52] |
| 50 | 222.35 | 222.37 | C15H26O | cadinol | terpenoids | [51,52] |
| 51 | 224.19 | 224.21 | C11H12O5 | sinapic acid | phenolic acid | [40,53] |
| 52 | 224.29 | 224.3 | C13H20O3 | vomifoliol | terpenoids | [52] |
| 53 | 226.21 | 226.23 | C11H14O5 | genipin | terpenoids | [52] |
| 54 | 228.28 | 228.29 | C11H20N2O3 | prolyl-leucine | dipeptide | [52] |
| 55 | 229.22 | 229.23 | C9H15N3O4 | asparaginyl-proline | dipeptide | [52] |
| 56 | 230.29 | 230.3 | C15H18O2 | dehydrocostuslactone | terpenoids | [52] |
| 57 | 234.19 | 234.2 | C12H10O5 | 7-methoxycoumarin-4-acetic acid | coumarins | [52] |
| 58 | 234.37 | 234.38 | C15H26N2 | sparteine | alkaloid | [55] |
| 59 | 236.26 | 236.27 | C12H16N2O3 | phenylalanylalanine | dipeptide | [52] |
| 60 | 236.34 | 236.35 | C15H24O2 | curcumol | terpenoids | [52] |
| 61 | 240.49 | 240.5 | C17H36 | heptadecane | hydrocarbons, lipids | [51] |
| 62 | 242.21 | 242.23 | C10H14N2O5 | thymidine | nucleoside | [40,49] |
| 63 | 242.39 | 242.4 | C15H30O2 | pentadecanoic acid | fatty acids | [51] |
| 64 | 243.21 | 243.22 | C9H13N3O5 | cytidine | nucleoside | [52] |
| 65 | 244.19 | 244.2 | C9H12N2O6 | uridine | nucleoside | [52] |
| 66 | 244.23 | 244.24 | C10H16N2O5 | prolylglutamic acid | dipeptide | [52] |
| 67 | 244.31 | 244.33 | C12H24N2O3 | isoleucyl-isoleucine | dipeptide | [11,52] |
| 68 | 248.35 | 248.36 | C15H24N2O | lupanine | alkaloid | [55] |
| 69 | 250.26 | 250.27 | C8H14N2O5S | gamma-glutamylcysteine | dipeptide | [49] |
| 70 | 250.37 | 250.38 | C15H26N2O | retamine | alkaloid | [53] |
| 71 | 256.41 | 256.42 | C16H32O2 | palmitic acid | fatty acids | [52] |
| 72 | 262.29 | 262.3 | C14H18N2O3 | prolylphenylalanine | dipeptide | [52] |
| 74 | 264.31 | 264.32 | C14H20N2O3 | phenylalanylvaline | dipeptide | [52] |
| 74 | 267.23 | 267.24 | C10H13N5O4 | adenosine | nucleoside | [40,52] |
| 75 | 272.24 | 272.25 | C15H12O5 | naringenin | flavonoids | [40,54,55] |
| 76 | 278.29 | 278.3 | C14H18N2O4 | tyrosyl-L-proline | dipeptide | [55] |
| 77 | 278.39 | 278.4 | C18H30O2 | linolenic acid | fatty acids | [46] |
| 78 | 278.33 | 278.35 | C15H22N2O3 | leucyl-phenylalanine | dipeptide | [52] |
| 79 | 280.39 | 280.4 | C18H32O2 | linoleic acid | fatty acids | [52] |
| 80 | 283.21 | 283.24 | C10H13N5O5 | guanosine | nucleoside | [52] |
| 81 | 284.49 | 284.5 | C18H36O2 | stearic acid | fatty acids | [52] |
| 82 | 286.23 | 286.24 | C15H10O6 | luteolin | flavonoids | [52] |
| 83 | 288.26 | 288.25 | C15H12O6 | eriodictyol | flavonoids | [49] |
| 84 | 292.39 | 292.4 | C18H28O3 | 9-OxoOTrE | fatty acids | [52] |
| 85 | 292.34 | 294.35 | C15H22N2O4 | tyrosylleucine | dipeptide | [52] |
| 86 | 296.49 | 296.5 | C20H40O | phytol | terpenoids | [51] |
| 87 | 298.39 | 298.4 | C19H22O3 | acerogenin G | flavonoids | [46] |
| 88 | 300.25 | 300.26 | C16H12O6 | rhamnocitrin | flavonoids | [49] |
| 89 | 302.22 | 302.23 | C15H10O7 | quercetin | flavonoids | [40,53,54] |
| 90 | 302.27 | 302.28 | C16H14O6 | homoeriodictyol | flavonoids | [46] |
| 91 | 300.39 | 300.4 | C19H24O3 | centrolobol | flavonoids | [46] |
| 92 | 307.31 | 307.33 | C10H17N3O6S | glutathione | peptides | [48,49,52] |
| 93 | 308.49 | 308.5 | C20H36O2 | terpineol | terpenoids | [51] |
| 94 | 312.39 | 312.4 | C18H20N2O3 | phenylalanylphenylalanine | dipeptide | [52] |
| 95 | 312.49 | 312.5 | C20H40O2 | arachidic acid | fatty acids | [52] |
| 96 | 314.27 | 314.29 | C17H14O6 | ermanin | flavonoids | [52] |
| 97 | 314.49 | 314.5 | C18H34O4 | 12,13-DiHOME | fatty acids | [52] |
| 98 | 316.25 | 316.26 | C16H12O7 | rhamnetin | flavonoids | [52] |
| 99 | 317.39 | 317.4 | C17H23N3O3 | leucyl-tryptophan | dipeptide | [52] |
| 100 | 328.39 | 328.4 | C19H20O5 | hirsutanone | diarylheptanoids | [49,52] |
| 101 | 330.27 | 330.29 | C17H14O7 | rhamnazin | flavonoids | [49] |
| 102 | 341.39 | 341.4 | C17H31N3O4 | Ile-Pro-Ile | peptides | [52] |
| 103 | 342.33 | 342.34 | C16H22O8 | coniferin | glucoside | [56] |
| 104 | 344.39 | 344.3 | C18H16O7 | santin | flavonoids | [52] |
| 105 | 354.29 | 354.31 | C16H18O9 | chlorogenic acid | phenolic acid | [40,46,49,54] |
| 106 | 360.29 | 360.3 | C18H16O8 | rosmarinic acid | phenolic acid | [10,40] |
| 107 | 368.59 | 368.6 | C24H48O2 | lignoceric acid | fatty acids | [52] |
| 108 | 372.39 | 372.4 | C17H24O9 | syringin | flavonoids | [11,52] |
| 109 | 386.39 | 386.4 | C19H30O8 | roseoside | terpenoids | [52] |
| 110 | 388.39 | 388.4 | C21H24O7 | medioresinol | lignal | [52] |
| 111 | 399.69 | 399.7 | C26H52O2 | cerotic acid | fatty acids | [52] |
| 112 | 406.39 | 406.4 | C20H22O9 | viscutin-3 | flavonoids | [49] |
| 113 | 412.69 | 412.7 | C30H52 | lupane | terpenoids | [49] |
| 114 | 414.69 | 414.7 | C29H50O | β-sitosterol | sterols | [49,57] |
| 115 | 418.39 | 418.4 | C22H26O8 | syringaresinol | lignal | [49] |
| 116 | 426.69 | 426.7 | C30H50O | lupeol | terpenoids | [46,49] |
| 117 | 434.29 | 434.3 | C20H18O11 | avicularin | flavonoids | [55] |
| 118 | 434.39 | 434.4 | C21H22O10 | naringenin-7-O-glucoside | flavonoids | [54] |
| 119 | 442.39 | 442.4 | C20H26O11 | visartiside D | flavonoids | [54] |
| 120 | 442.69 | 442.7 | C30H50O2 | betulin | terpenoids | [46] |
| 121 | 448.39 | 448.4 | C21H20O11 | quercitrin | flavonoids | [49] |
| 122 | 450.39 | 450.4 | C21H22O11 | eriodictyol-7-O-glucoside | flavonoids | [50] |
| 123 | 456.69 | 456.7 | C30H48O3 | betulinic acid | terpenoids | [46,57] |
| 124 | 464.39 | 464.4 | C21H20O12 | hyperoside | flavonoids | [51] |
| 125 | 468.79 | 468.8 | C32H52O2 | β-amyrin acetate | terpenoids | [50] |
| 126 | 476.41 | 476.43 | C23H24O11 | flavoyadorinin B | flavonoids | [49] |
| 127 | 478.39 | 478.4 | C22H22O12 | isorhamnetin-3-O-rutinoside | flavonoids | [52,53,56] |
| 128 | 492.39 | 492.4 | C23H24O12 | flavoyadorinin A | flavonoids | [49] |
| 129 | 526.49 | 526.5 | C27H26O11 | viscutin-1 | flavonoids | [49] |
| 130 | 532.49 | 532.5 | C27H32O11 | visartiside E | flavonoids | [50] |
| 131 | 536.89 | 536.9 | C40H56 | carotene | miscellaneous | [48] |
| 132 | 565.79 | 565.8 | C30H55N5O5 | viscumamide | peptides | [58,59] |
| 133 | 568.49 | 568.5 | C29H28O12 | viscutin-2 | flavonoids | [49] |
| 134 | 576.79 | 576.8 | C35H60O6 | daucosterol | sterols | [52] |
| 135 | 596.49 | 596.5 | C27H32O15 | viscumneosides I | flavonoids | [49] |
| 136 | 608.49 | 608.5 | C28H32O15 | homoflavoyadorinin B | flavonoids | [52] |
| 137 | 636.59 | 636.6 | C29H32O16 | viscumneoside IV | flavonoids | [49] |
| 138 | 728.59 | 728.6 | C32H40O19 | viscumneoside V | flavonoids | [49] |
| 139 | 768.69 | 768.7 | C34H40O20 | viscumneoside VII | flavonoids | [49] |
| 140 | 784.96 | 784.97 | C41H68O14 | astragaloside IV | terpenoid | [49] |
| Chemical Class | Metabolite Name |
|---|---|
| Flavonoids | naringenin |
| acerogenin G | |
| luteolin | |
| eriodictyol | |
| rhamnocitrin | |
| centrolobol | |
| quercetin | |
| homoeriodictyol | |
| ermanin | |
| rhamnetin | |
| rhamnazin | |
| santin | |
| syringin | |
| viscutin-3 | |
| avicularin | |
| naringenin-7-O-glucoside | |
| visartiside D | |
| quercitrin | |
| eriodictyol-7-O-glucoside | |
| hyperoside | |
| flavoyadorinin B | |
| isorhamnetin-3-O-rutinoside | |
| flavoyadorinin A | |
| viscutin-1 | |
| visartiside E | |
| viscutin-2 | |
| viscumneosides I | |
| homoflavoyadorinin B | |
| viscumneoside IV | |
| viscumneoside V | |
| Amino acids and peptides | cysteine |
| leucine | |
| glutamic acid | |
| methionine | |
| histidine | |
| arginine | |
| tyrosine | |
| prolyl-alanine | |
| leucyl-glycine | |
| leucyl-alanine | |
| tryptophan | |
| valylvaline | |
| prolyl-leucine | |
| asparaginyl-proline | |
| phenylalanylalanine | |
| prolylglutamic acid | |
| isoleucyl-isoleucine | |
| gamma-glutamylcysteine | |
| prolylphenylalanine | |
| phenylalanylvaline | |
| tyrosyl-L-proline | |
| leucyl-phenylalanine | |
| tyrosylleucine | |
| glutathione | |
| phenylalanylphenylalanine | |
| leucyl-tryptophan | |
| Ile-Pro-Ile | |
| viscumamide | |
| Terpenoids | sabinene |
| safranal | |
| citral | |
| geraniol | |
| geranylacetone | |
| loliolide | |
| nerol acetate | |
| caryophyllene | |
| cadinene | |
| cadinol | |
| vomifoliol | |
| genipin | |
| dehydrocostuslactone | |
| curcumol | |
| phytol | |
| acerogenin G | |
| terpineol | |
| hirsutanone | |
| roseoside | |
| lupane | |
| lupeol | |
| betulin | |
| betulinic acid | |
| β-amyrin acetate | |
| astragaloside IV | |
| Phenolic acids | salicilyc acid |
| cinamic acid | |
| gentisic acid | |
| p-coumaric acid | |
| gallic acid | |
| caffeic acid | |
| veratric acid | |
| ferulic acid | |
| syringic acid | |
| chlorogenic acid | |
| rosmarinic acid | |
| Fatty acids | octanoic acid |
| pentadecanoic acid | |
| palmitic acid | |
| linolenic acid | |
| linoleic acid | |
| stearic acid | |
| 9-OxoOTrE | |
| arachidic acid | |
| 12,13-DiHOME | |
| lignoceric acid | |
| cerotic acid | |
| Organic acids | tropic acid |
| aconic acid | |
| ascorbic acid | |
| citric acid | |
| quinic acid | |
| Nucleosides | thymidine |
| cytidine | |
| uridine | |
| adenosine | |
| guanosine | |
| Alcohols and esters | 2-phenylethanol |
| 1-octene-3-ol | |
| cinnamic acid methyl ester | |
| viscumitol | |
| sinapic acid | |
| Amines | choline |
| histamine | |
| acetylcholine | |
| Coumarins | coumarin |
| 6-hydroxy-4-methylcoumarin | |
| 7-methoxycoumarin-4-acetic acid | |
| Alkaloids | sparteine |
| lupanine | |
| retamine | |
| Lignals | pinoresinol |
| medioresinol | |
| syringaresinol | |
| Sterols | β-sitosterol |
| daucosterol | |
| Aldehydes and ketones | vanillin |
| ionone | |
| Miscellaneous | nonanolide |
| heptadecane | |
| carotene | |
| coniferin |
| Metabolites | Wavenumber (cm–1) | Ref. |
|---|---|---|
| Amino acids, peptides, thionins | 3400; 3330–3130; 2530–2760; 2080–2140; 1724–1754 1687, 1675, 1663, 1654, 1644, 1632, 1621, 1611, 1610–1660, 1500–1600; | [5] |
| Flavonoids | 1734, 1703, 1634, 1627, 1580, 1522, 1460, 1440, 1410, 1367, 1315, 1255, 630 and 575 | [86,87,88] |
| Terpenoids | 2938.7, 1740, 1651, 810 | [87,89] |
| Phenolic acids | 1800–1650, 1734, 1720, 1627, 1522, 1440, 1410, 1420–1300, 1367, 1315, 1255, 1170–1100 | [88] |
| Fatty acids | 3020–3010, 2924–2915, 2855–2847, 2800–2900, 1746, 1710, 1250, 720 | [90,91] |
| Organic acids | 1255, 1378, 1440, 1410, 1376 | [88] |
| Nucleosides | 968, 1120, 1175, 1330, 1420, 1480, 1725, 3270, 3600 | [92] |
| Coumarins | 600–900, 1200–1000, 1028, 1254, 1450, 1608, 1627–1715, 1740, 1760, 2207–2210, 2963, 3362, 3381, 3399, 3434, | [93,94] |
| Alkaloids | 630–650, 925, 1200,1285, 1330, 1531, 1548, 1600–1650, 1653, 1658–1567, 1600, 1660, 1700, 1710, 3000, 3377, 3380, 3332, 3384.84 | [95] |
| Sterols | 740.5, 1062.5, 1192, 1383, 1465.6. 1737.5, 2937 | [96] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Segneanu, A.-E.; Marin, C.N.; Herea, D.D.; Stanusoiu, I.; Muntean, C.; Grozescu, I. Romanian Viscum album L.—Untargeted Low-Molecular Metabolomic Approach to Engineered Viscum–AuNPs Carrier Assembly. Plants 2022, 11, 1820. https://doi.org/10.3390/plants11141820
Segneanu A-E, Marin CN, Herea DD, Stanusoiu I, Muntean C, Grozescu I. Romanian Viscum album L.—Untargeted Low-Molecular Metabolomic Approach to Engineered Viscum–AuNPs Carrier Assembly. Plants. 2022; 11(14):1820. https://doi.org/10.3390/plants11141820
Chicago/Turabian StyleSegneanu, Adina-Elena, Catalin Nicolae Marin, Dumitru Daniel Herea, Ionut Stanusoiu, Cornelia Muntean, and Ioan Grozescu. 2022. "Romanian Viscum album L.—Untargeted Low-Molecular Metabolomic Approach to Engineered Viscum–AuNPs Carrier Assembly" Plants 11, no. 14: 1820. https://doi.org/10.3390/plants11141820
APA StyleSegneanu, A.-E., Marin, C. N., Herea, D. D., Stanusoiu, I., Muntean, C., & Grozescu, I. (2022). Romanian Viscum album L.—Untargeted Low-Molecular Metabolomic Approach to Engineered Viscum–AuNPs Carrier Assembly. Plants, 11(14), 1820. https://doi.org/10.3390/plants11141820








