Biological and Transcriptomic Characterization of Pre-Haustorial Resistance to Sunflower Broomrape (Orobanche cumana W.) in Sunflowers (Helianthus annuus)
Abstract
1. Introduction
2. Results
2.1. Effect of Grafting on the Source of the Resistance
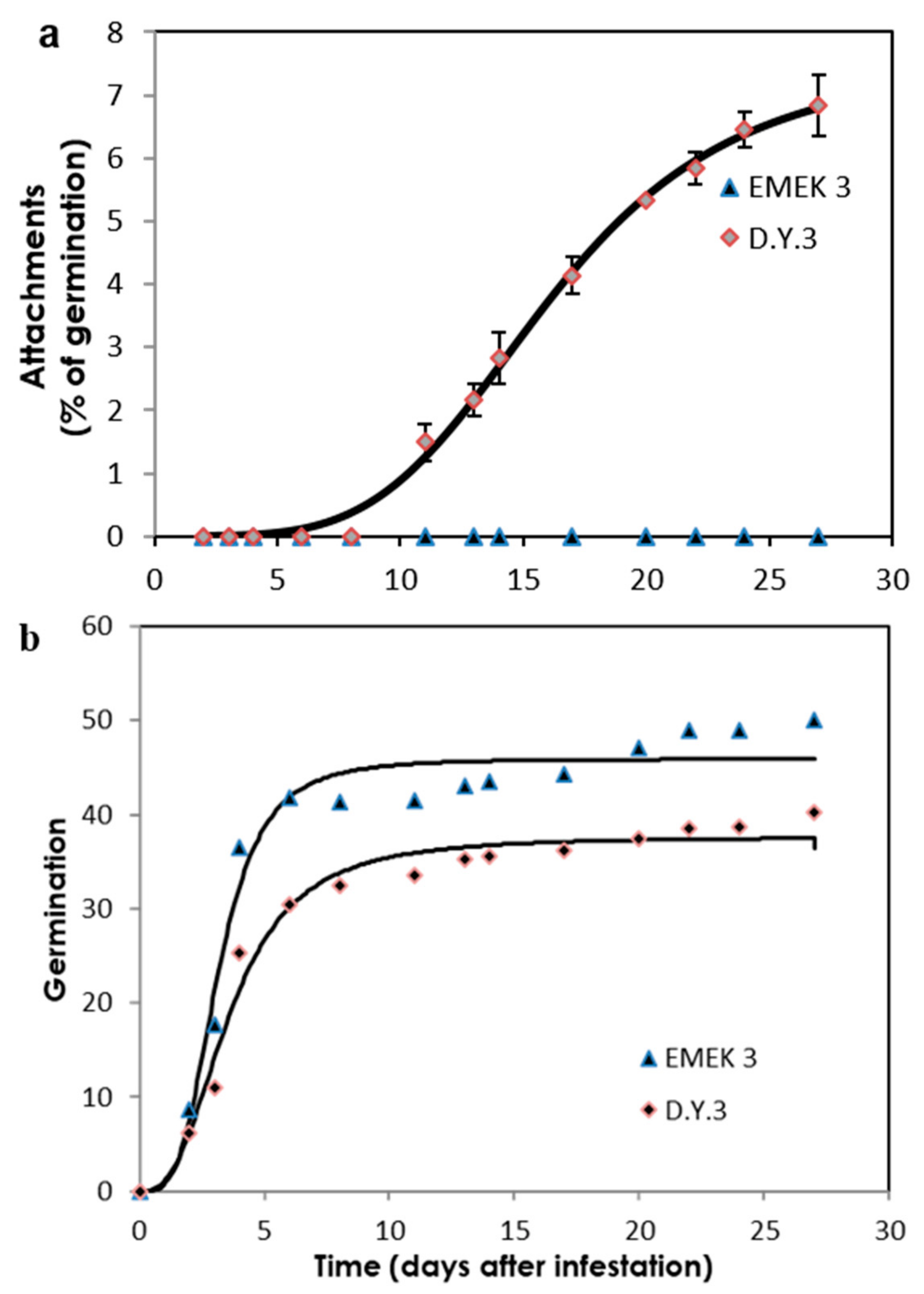
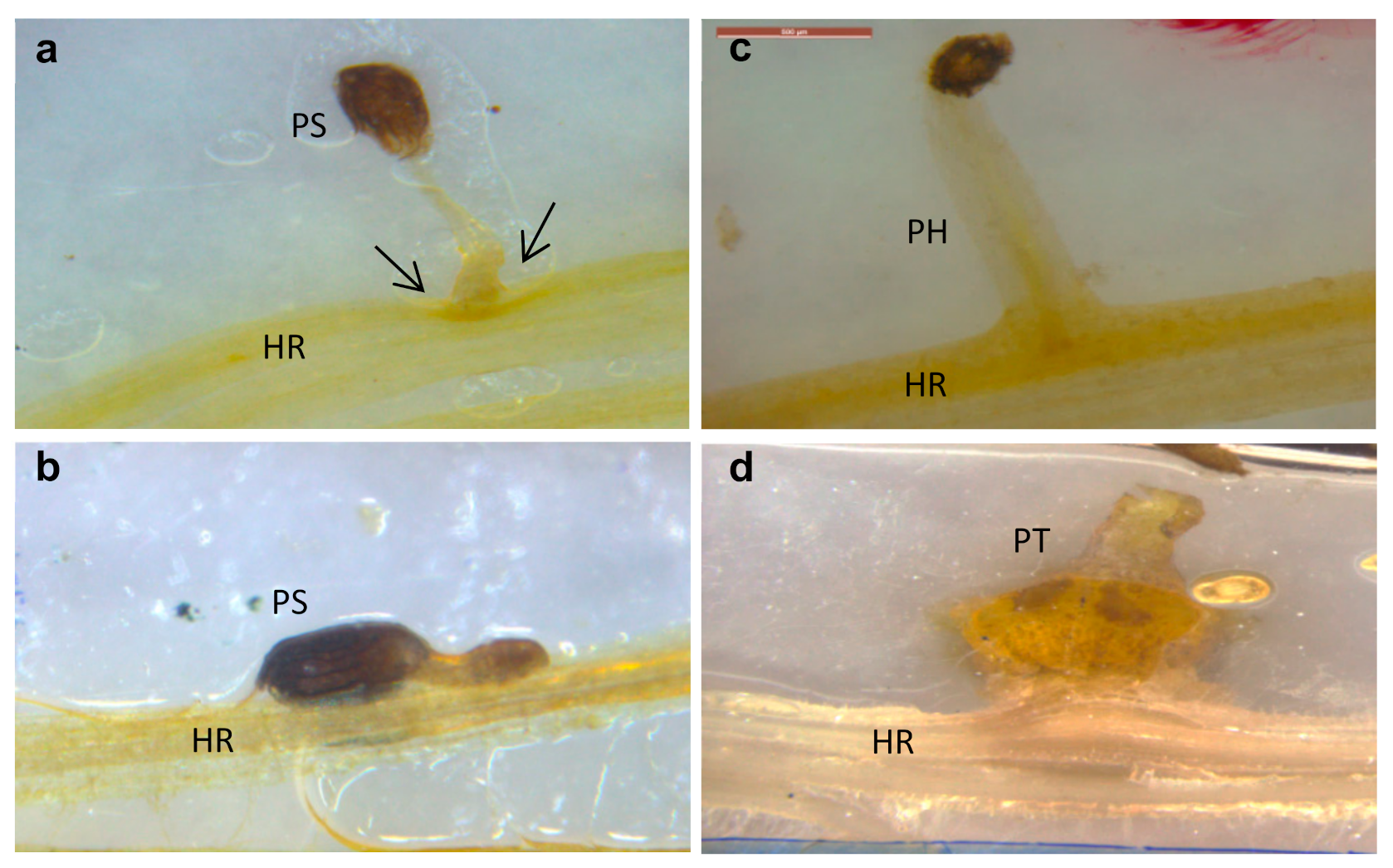
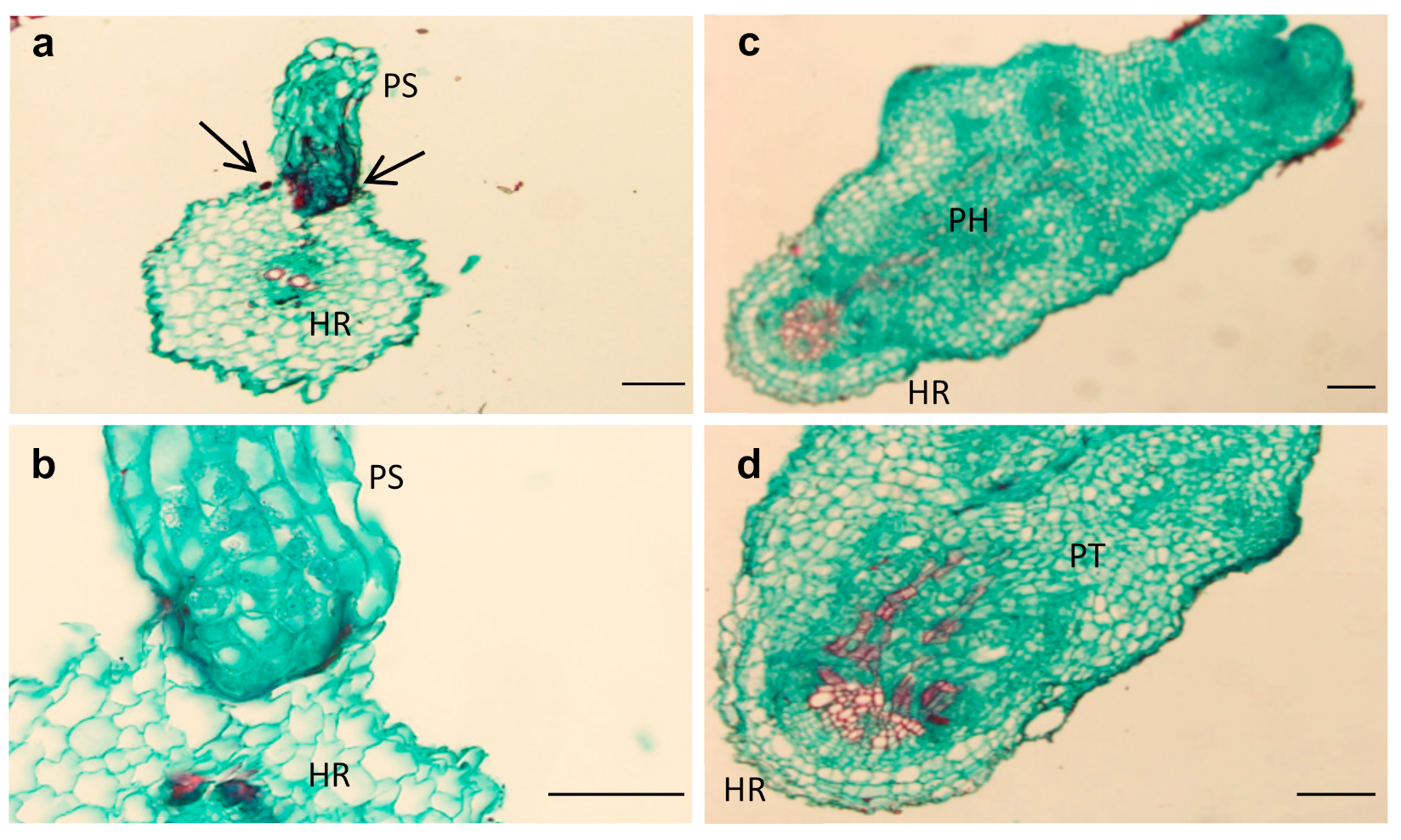
2.2. Sunflower–O. cumana Incompatibility
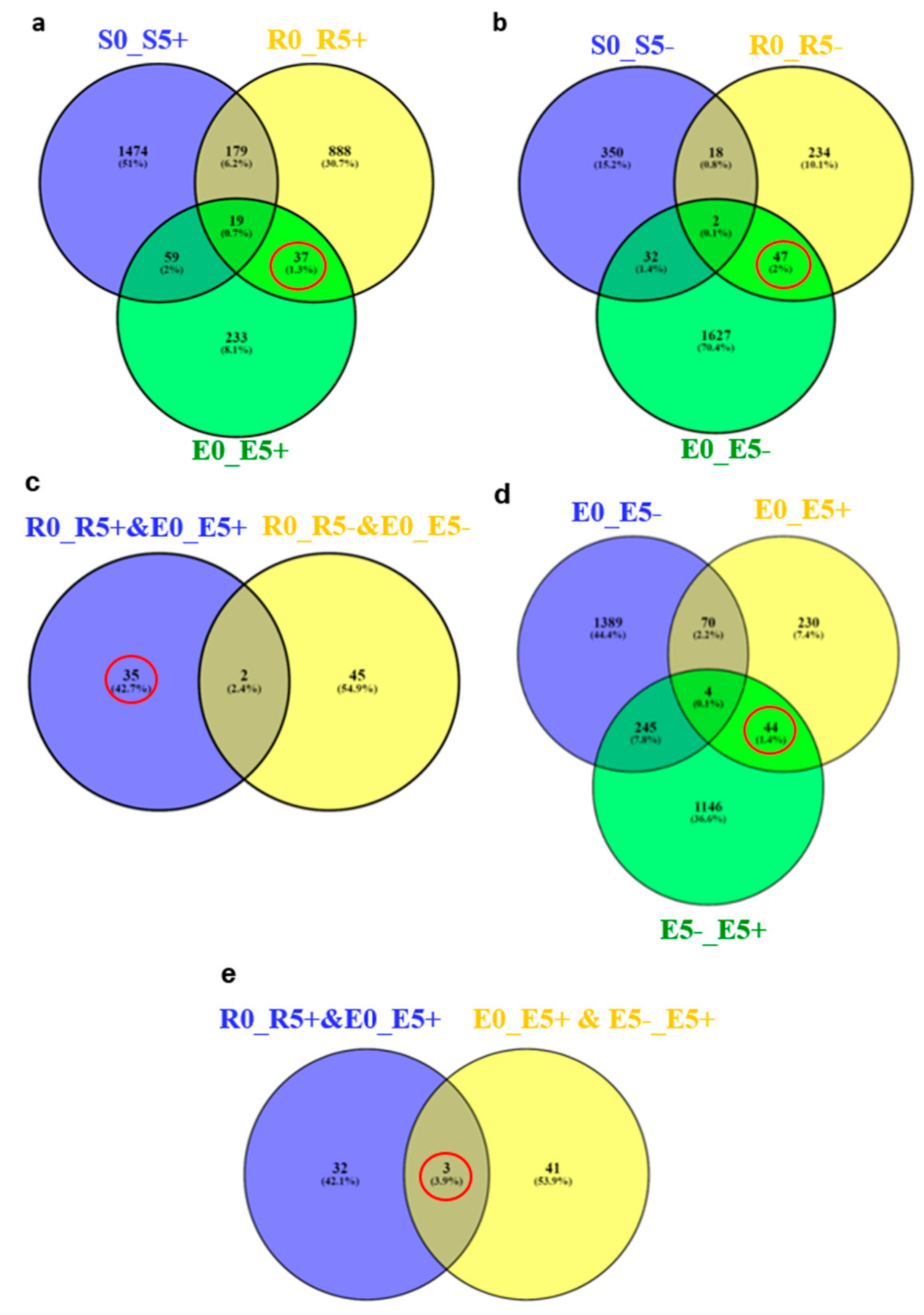
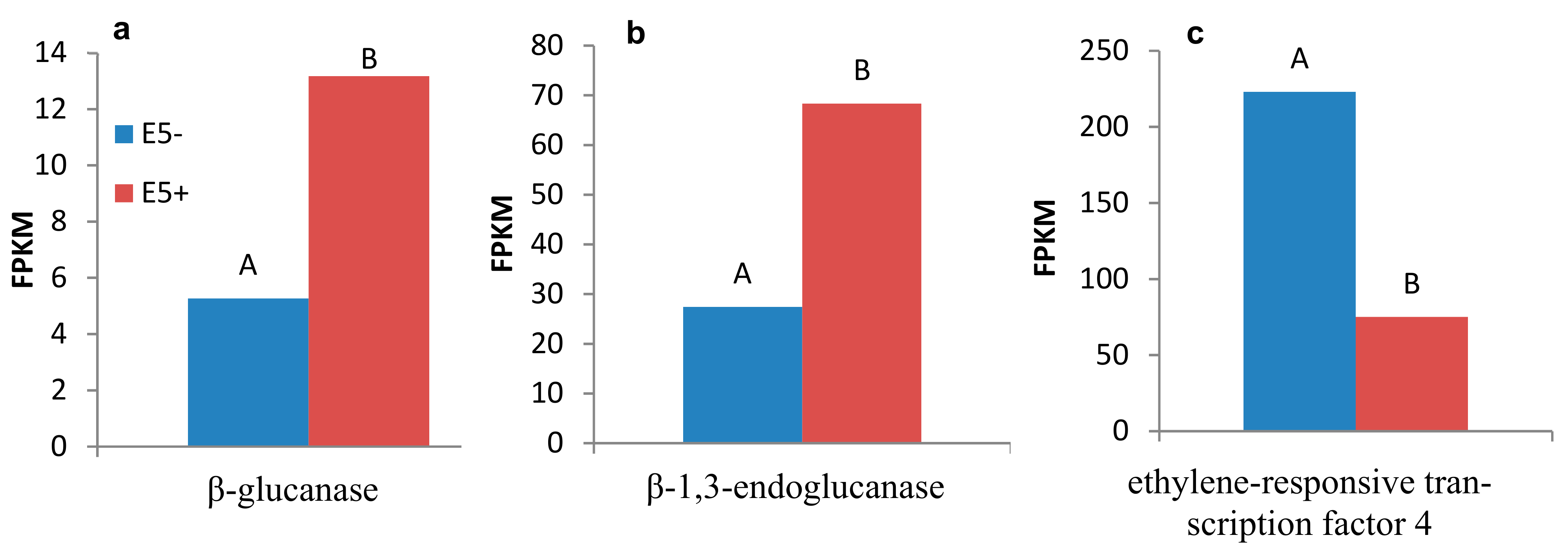
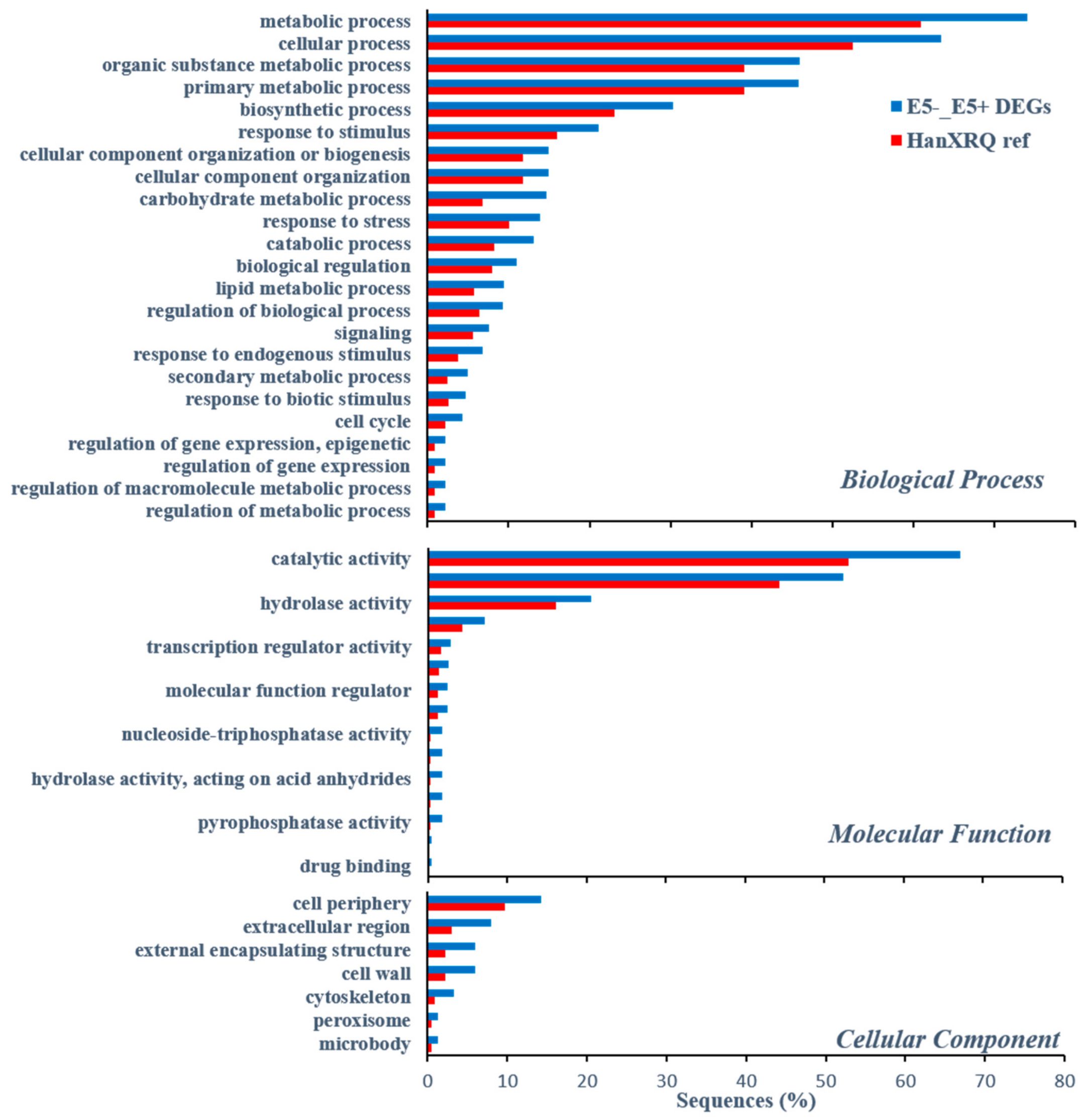
2.3. Identification of Candidate Resistance Genes, Using RNA-Sequencing
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Plant Materials and Growth Conditions
5.2. Preconditioning of O. cumana Seeds
5.3. Cultivation in Polyethylene Bag
5.4. Histological Analysis
5.5. Grafting Experiments
5.6. Statistical Analysis
5.7. Bulk Construction and RNA Extraction
5.8. RNA Sequencing and Mapping
5.9. RNA-Seq Data Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Parker, C. The parasitic weeds of the Orobanchaceae. In Parasitic Orobanchaceae; Joel, D.M., Gressel, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 313–344. [Google Scholar]
- Eizenberg, H.; Hershenhorn, J.; Ephrath, J.H.; Kanampiu, F. Chemical control. In Parasitic Orobanchaceae; Joel, D.M., Gressel, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 415–432. [Google Scholar]
- Eizenberg, H.; Hershenhorn, J.; Ephrath, J.E. Factors affecting the efficacy of Orobanche cumana chemical control in sunflower. Weed Res. 2009, 49, 308–315. [Google Scholar] [CrossRef]
- De Luque, A.P.; Moreno, M.; Rubiales, D. Host plant resistance against broomrapes (Orobanche spp.): Defence reactions and mechanisms of resistance. Ann. Appl. Biol. 2008, 152, 131–141. [Google Scholar] [CrossRef]
- Xie, X.; Yoneyama, K.; Yoneyama, K. The Strigolactone Story. Annu. Rev. Phytopathol. 2010, 48, 93–117. [Google Scholar] [CrossRef] [PubMed]
- Yoneyama, K.; Ruyter-Spira, C.; Bouwmeester, H. Induction of germination. In Parasitic Orobanchaceae: Parasitic Mechanisms and Control Strategies; Joel, D.M., Gressel, J., Musselman, L.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 167–194. ISBN 978-3-642-38146-1. [Google Scholar]
- Joel, D.M.; Bar, H. The seed and the seedling. In Parasitic Orobanchaceae: Parasitic Mechanisms and Control Strategies; Joel, D.M., Gressel, J., Musselman, L.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 147–165. ISBN 978-3-642-38146-1. [Google Scholar]
- Joel, D.M.; Losner-Goshen, D. The attachment organ of the parasitic angiosperms Orobanche cumana and O. aegyptiaca and its development. Can. J. Bot. 1994, 72, 564–574. [Google Scholar] [CrossRef]
- Letousey, P.; De Zélicourt, A.; Dos Santos, C.V.; Thoiron, S.; Monteau, F.; Simier, P.; Thalouarn, P.; Delavault, P. Molecular analysis of resistance mechanisms to Orobanche cumana in sunflower. Plant Pathol. 2007, 56, 536–546. [Google Scholar] [CrossRef]
- Echevarría-Zomeño, S.; de Luque, A.P.; Jorrín, J.; Maldonado, A.M. Pre-haustorial resistance to broomrape (Orobanche cumana) in sunflower (Helianthus annuus): Cytochemical studies. J. Exp. Bot. 2006, 57, 4189–4200. [Google Scholar] [CrossRef]
- De Luque, A.P.; González-Verdejo, C.I.; Lozano-Baena, M.-D.; Dita, M.A.; Cubero, J.I.; González-Melendi, P.; Risueño, M.C.; Rubiales, D. Protein cross-linking, peroxidase and β-1,3-endoglucanase involved in resistance of pea against Orobanche crenata. J. Exp. Bot. 2006, 57, 1461–1469. [Google Scholar] [CrossRef]
- De Luque, A.P.; Rubiales, D.; Cubero, J.I.; Press, M.C.; Scholes, J.; Yoneyama, K.; Takeuchi, Y.; Plakhine, D.; Joel, D.M. Interaction between Orobanche crenata and its Host Legumes: Unsuccessful Haustorial Penetration and Necrosis of the Developing Parasite. Ann. Bot. 2005, 95, 935–942. [Google Scholar] [CrossRef]
- Lozano-Baena, M.-D.; Prats, E.; Moreno, M.T.; Rubiales, D.; de Luque, A.P. Medicago truncatula as a Model for Nonhost Resistance in Legume-Parasitic Plant Interactions. Plant Physiol. 2007, 145, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Eizenberg, H.; Hershenhorn, J.; Plakhine, D.; Kleifeld, Y.; Shtienberg, D.; Rubin, B. Effect of temperature on susceptibility of sunflower varieties to sunflower broomrape (Orobanche cumana) and Egyptian broomrape (Orobanche aegyptiaca). Weed Sci. 2003, 51, 279–286. [Google Scholar] [CrossRef]
- Boller, T.; He, S.Y. Innate immunity in plants: An arms race between pattern recognition receptors in plants and effectors in microbial pathogens. Science 2009, 324, 742–744. [Google Scholar] [CrossRef]
- Molinero-Ruiz, L.; Delavault, P.; Pérez-Vich, B.; Pacureanu-Joita, M.; Bulos, M.; Altieri, E.; Domínguez, J. History of the race structure of Orobanche cumana and the breeding of sunflower for resistance to this parasitic weed: A review. Span. J. Agric. Res. 2015, 13, e10R01-01. [Google Scholar] [CrossRef]
- Pérez-Vich, B.; Akhtouch, B.; Knapp, S.J.; León, A.J.; Velasco, L.; Fernández-Martínez, J.M.; Berry, S.T. Quantitative trait loci for broomrape (Orobanche cumana Wallr.) resistance in sunflower. Theor. Appl. Genet. 2004, 109, 92–102. [Google Scholar] [CrossRef]
- Román, B.; Torres, A.M.; Rubiales, D.; Cubero, J.I.; Šatović, Z. Mapping of quantitative trait loci controlling broomrape (Orobanche crenata Forsk.) resistance in faba bean (Vicia faba L.). Genome 2002, 45, 1057–1063. [Google Scholar] [CrossRef]
- Vranceanu, A.V.; Tudor, V.A.; Stoenescu, F.M.; Pirvu, N. Virulence groups of Orobanche cumana Wallr. differential hosts and resistance sources and genes in sunflower. In Proceedings of the 9th International Sunflower Conference, Torremolinos, Spain, 8–13 June 1980; pp. 74–82. [Google Scholar]
- Flor, H.H. Current Status of the Gene-for-Gene Concept. Annu. Rev. Phytopathol. 1971, 9, 275–296. [Google Scholar] [CrossRef]
- Akhtouch, B.; Munoz-Ruz, J.; Melero-Vara, J.; Fernandez-Martinez, J.; Dominguez, J. Inheritance of resistance to race F of broomrape in sunflower lines of different origins. Plant Breed. 2002, 121, 266–268. [Google Scholar] [CrossRef]
- Fernandez-Martinez, J.; Perez-Vich, B.; Akhtouch, B.; Velasco, L.; Munoz-Ruz, J.; Melero-Vara, J.; Dominguez, J. Registration of Four Sunflower Germplasms Resistant to Race F of Broomrape. Crop Sci. 2004, 44, 1033–1034. [Google Scholar] [CrossRef]
- Perez-Vich, B.; Aktouch, B.; Mateos, A.; Velasco, L.; Jan, C.; Fernández, J.; Dominguez, J.; Fernández-Martínez, J. Dominance relationships for genes conferring resistance to broomrape (Orobanche cumana wallr.) in sunflower. Helia 2004, 27, 183–192. [Google Scholar] [CrossRef]
- Akhtouch, B.; del Moral, L.; Leon, A.; Velasco, L.; Martínez, J.M.F.; Pérez-Vich, B. Genetic study of recessive broomrape resistance in sunflower. Euphytica 2016, 209, 419–428. [Google Scholar] [CrossRef]
- Lu, Y.H.; Gagne, G.; Grezes-Besset, B.; Blanchard, P. Integration of a molecular linkage group containing the broomrape resistance gene Or5 into an RFLP map in sunflower. Genome 1999, 42, 453–456. [Google Scholar] [CrossRef]
- Lu, Y.H.; Melero-Vara, J.M.; Garcia-Tejada, J.A.; Blanchard, P. Development of SCAR markers linked to the gene Or5 conferring resistance to broomrape (Orobanche cumana Wallr.) in sunflower. Theor. Appl. Genet. 2000, 100, 625–632. [Google Scholar] [CrossRef]
- Tang, S.; Heesacker, A.; Kishore, V.K.; Fernandez, A.; Sadik, E.S.; Cole, G.; Knapp, S.J. Genetic Mapping of the Or5 Gene for Resistance to Orobanche Race E in Sunflower. Crop Sci. 2003, 43, 1021–1028. [Google Scholar] [CrossRef]
- Louarn, J.; Boniface, M.-C.; Pouilly, N.; Velasco, L.; Pérez-Vich, B.; Vincourt, P.; Muños, S. Sunflower Resistance to Broomrape (Orobanche cumana) Is Controlled by Specific QTLs for Different Parasitism Stages. Front. Plant Sci. 2016, 7, 590. [Google Scholar] [CrossRef]
- Imerovski, I.; Dedić, B.; Cvejic, S.; Miladinović, D.; Jocić, S.; Owens, G.L.; Tubić, N.K.; Rieseberg, L.H. BSA-seq mapping reveals major QTL for broomrape resistance in four sunflower lines. Mol. Breed. 2019, 39, 41. [Google Scholar] [CrossRef]
- Duriez, P.; Vautrin, S.; Auriac, M.C.; Bazerque, J.; Boniface, M.C.; Callot, C.; Carrère, S.; Cauet, S.; Chabaud, M.; Gentou, F.; et al. A receptor-like kinase enhances sunflower resistance to Orobanche cumana. Nat. Plants 2019, 5, 1211–1215. [Google Scholar] [CrossRef] [PubMed]
- Goldwasser, Y.; Hershenhorn, J.; Plakhine, D.; Kleifeld, Y.; Rubin, B. Biochemical factors involved in vetch resistance to Orobanche aegyptiaca. Physiol. Mol. Plant Pathol. 1999, 54, 87–96. [Google Scholar] [CrossRef]
- Goldwasser, Y.; Plakhine, D.; Kleifeld, Y.; Zamski, E.; Rubin, B. The Differential Susceptibility of Vetch (Vicia spp.) to Orobanche aegyptiaca: Anatomical Studies. Ann. Bot. 2000, 85, 257–262. [Google Scholar] [CrossRef]
- Yang, C.; Xu, L.; Zhang, N.; Islam, F.; Song, W.; Hu, L.; Liu, D.; Xie, X.; Zhou, W. iTRAQ-based proteomics of sunflower cultivars differing in resistance to parasitic weed Orobanche cumana. Proteomics 2017, 17, 1700009. [Google Scholar] [CrossRef]
- Antonova, T.S.; TerBorg, S.J. The role of peroxidase in the resistance of sunflower against Oronache cumana in Russia. Weed Res. 1996, 36, 113–121. [Google Scholar] [CrossRef]
- Labrousse, P.; Arnaud, M.C.; Serieys, H.; Bervillé, A.; Thalouarn, P. Several Mechanisms are Involved in Resistance of Helianthus to Orobanche cumana Wallr. Ann. Bot. 2001, 88, 859–868. [Google Scholar] [CrossRef]
- Dor, E.; Alperin, B.; Wininger, S.; Ben-Dor, B.; Somvanshi, V.S.; Koltai, H.; Kapulnik, Y.; Hershenhorn, J. Characterization of a novel tomato mutant resistant to the weedy parasites Orobanche and Phelipanche spp. Euphytica 2009, 171, 371–380. [Google Scholar] [CrossRef]
- Oliveros, J.C. VENNY. An Interactive Tool for Comparing Lists with Venn Diagrams. 2007. Available online: https://bioinfogp.cnb.csic.es/tools/venny/index.html (accessed on 8 February 2017).
- Simmons, C.R. The physiology and molecular biology of plant 1, 3-β-D-glucanases and 1, 3; 1, 4-β-D-glucanases. Crit. Rev. Plant Sci. 1994, 13, 325–387. [Google Scholar]
- Durrant, W.; Dong, X. Systemic acquired resistance. Annu. Rev. Phytopathol. 2004, 42, 185–209. [Google Scholar] [CrossRef] [PubMed]
- Saboki, E.; Usha, K.; Singh, B. Pathogenesis related (PR) proteins in plant defense mechanism. In Science against Microbial Pathogens: Communicating Current Research and Technological Advances; Mendez-Vilas, A., Ed.; Formatex: Badajoz, Spain, 2011; pp. 1043–1047. [Google Scholar]
- Van Loon, L. Induced resistance in plants and the role of pathogenesis-related proteins. Eur. J. Plant Pathol. 1997, 103, 753–765. [Google Scholar] [CrossRef]
- Van Loon, L.; Van Strien, E. The families of pathogenesis-related proteins, their activities, and comparative analysis of PR-1 type proteins. Physiol. Mol. Plant Pathol. 1999, 55, 85–97. [Google Scholar] [CrossRef]
- Castillejo, M.; Amiour, N.; Dumas-Gaudot, E.; Rubiales, D.; Jorrín, J.V. A proteomic approach to studying plant response to crenate broomrape (Orobanche crenata) in pea (Pisum sativum). Phytochemistry 2004, 65, 1817–1828. [Google Scholar] [CrossRef]
- Anderson, J.; Badruzsaufari, E.; Schenk, P.; Manners, J.M.; Desmond, O.J.; Ehlert, C.; MacLean, D.J.; Ebert, P.; Kazan, K. Antagonistic Interaction between Abscisic Acid and Jasmonate-Ethylene Signaling Pathways Modulates Defense Gene Expression and Disease Resistance in Arabidopsis. Plant Cell 2004, 16, 3460–3479. [Google Scholar] [CrossRef]
- Singh, K.B.; Foley, R.C.; Oñate-Sánchez, L. Transcription factors in plant defense and stress responses. Curr. Opin. Plant Biol. 2002, 5, 430–436. [Google Scholar] [CrossRef]
- Kazan, K. Negative regulation of defence and stress genes by EAR-motif-containing repressors. Trends Plant Sci. 2006, 11, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, S.Y.; Ohta, M.; Usui, A.; Shinshi, H.; Ohme-Takagi, M. Arabidopsis ethylene-responsive element binding factors act as transcriptional activators or repressors of GCC box–mediated gene expression. Plant Cell 2000, 12, 393–404. [Google Scholar] [PubMed]
- McGrath, K.; Dombrecht, B.; Manners, J.M.; Schenk, P.; Edgar, C.I.; Maclean, D.J.; Scheible, W.-R.; Udvardi, M.; Kazan, K. Repressor- and Activator-Type Ethylene Response Factors Functioning in Jasmonate Signaling and Disease Resistance Identified via a Genome-Wide Screen of Arabidopsis Transcription Factor Gene Expression. Plant Physiol. 2005, 139, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, P.; Liu, Y.; Wang, P. Identification of candidate gene for resistance to broomrape (Orobanche cumana) in sunflower by BSA-seq. Oil Crop Sci. 2020, 5. [Google Scholar] [CrossRef]
- Parker, C.; Dixon, N. The use of polyethylene bags in the culture and study of Striga spp. and other organisms on crop roots. Ann. Appl. Biol. 1983, 103, 485–488. [Google Scholar] [CrossRef]
- Eizenberg, H.; Plakhine, D.; Hershenhorn, J.; Kleifeld, Y.; Rubin, B. Resistance to broomrape (Orobanche spp.) in sunflower (Helianthus annuus L.) is temperature dependent. J. Exp. Bot. 2003, 54, 1305–1311. [Google Scholar] [CrossRef][Green Version]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 32. [Google Scholar]
- Ruzin, S.E. Plant Microtechnique and Microscopy; Oxford University Press: New York, NY, USA, 1999; Volume 198. [Google Scholar]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 29 December 2016).
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Badouin, H.; Gouzy, J.; Grassa, C.; Murat, F.; Staton, S.; Cottret, L.; Lelandais-Brière, C.; Owens, G.L.; Carrère, S.; Mayjonade, B.; et al. The sunflower genome provides insights into oil metabolism, flowering and Asterid evolution. Nature 2017, 546, 148–152. [Google Scholar] [CrossRef]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2012, 29, 15–21. [Google Scholar] [CrossRef]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Götz, S.; Garcia-Gomez, J.M.; Terol, J.; Williams, T.; Nagaraj, S.H.; Nueda, M.J.; Robles, M.; Talón, M.; Dopazo, J.; Conesa, A. High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res. 2008, 36, 3420–3435. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sisou, D.; Tadmor, Y.; Plakhine, D.; Ziadna, H.; Hübner, S.; Eizenberg, H. Biological and Transcriptomic Characterization of Pre-Haustorial Resistance to Sunflower Broomrape (Orobanche cumana W.) in Sunflowers (Helianthus annuus). Plants 2021, 10, 1810. https://doi.org/10.3390/plants10091810
Sisou D, Tadmor Y, Plakhine D, Ziadna H, Hübner S, Eizenberg H. Biological and Transcriptomic Characterization of Pre-Haustorial Resistance to Sunflower Broomrape (Orobanche cumana W.) in Sunflowers (Helianthus annuus). Plants. 2021; 10(9):1810. https://doi.org/10.3390/plants10091810
Chicago/Turabian StyleSisou, Dana, Yaakov Tadmor, Dina Plakhine, Hammam Ziadna, Sariel Hübner, and Hanan Eizenberg. 2021. "Biological and Transcriptomic Characterization of Pre-Haustorial Resistance to Sunflower Broomrape (Orobanche cumana W.) in Sunflowers (Helianthus annuus)" Plants 10, no. 9: 1810. https://doi.org/10.3390/plants10091810
APA StyleSisou, D., Tadmor, Y., Plakhine, D., Ziadna, H., Hübner, S., & Eizenberg, H. (2021). Biological and Transcriptomic Characterization of Pre-Haustorial Resistance to Sunflower Broomrape (Orobanche cumana W.) in Sunflowers (Helianthus annuus). Plants, 10(9), 1810. https://doi.org/10.3390/plants10091810





