Abstract
Recent advances in developing and screening candidate pharmacotherapies for psychiatric disorders have depended on rodent models. Eating disorders are a set of psychiatric disorders that have traditionally relied on behavioral therapies for effective long-term treatment. However, the clinical use of Lisdexamfatamine for binge eating disorder (BED) has furthered the notion of using pharmacotherapies for treating binge eating pathologies. While there are several binge eating rodent models, there is not a consensus on how to define pharmacological effectiveness within these models. Our purpose is to provide an overview of the potential pharmacotherapies or compounds tested in established rodent models of binge eating behavior. These findings will help provide guidance for determining pharmacological effectiveness for potential novel or repurposed pharmacotherapies.
1. Introduction
Advancements in uncovering novel therapeutics for eating disorders have been limited in the past several decades. Treatments for eating disorders have historically depended on cognitive behavioral and psychotherapies [1,2,3]. While these therapies remain effective for specific eating disorders and coping with individualized factors, the long-term effectiveness of these therapies is dependent on the availability of trained clinicians, patient commitment, and other clinically managed outcomes [4,5,6]. In recent years, there has been an interest in developing pharmacotherapies as adjuvant to improve behavioral and psychotherapy outcomes [7,8]. In the US, there are only two FDA-approved pharmacotherapies options for the treatment of eating disorders. Both medications are “repurposed” and were originally approved for other psychiatric illnesses [9]. Fluoxetine (FLX) is available for the clinical management of bulimia nervosa (BN), whereas lisdexamfetamine (LDX) is available for binge eating disorder (BED). To date, there is no pharmacotherapy approved for use in patients with anorexia nervosa (AN). Notably, BN and BED are two separate psychiatric disorders with binge eating as a core pathology but differ in their clinical diagnostic criteria and management [10].
FLX was FDA-approved in 1987 for the treatment of major depressive disorder and then approved for acute management and maintenance of BN in 1994 [11,12]. Efficacy of FLX was determined in two pivotal randomized placebo-controlled multi-center studies [13,14]. In an 8-week study (n = 387, 100% were female; DSM III-R defined BN), FLX (60 mg/day) had a 20% placebo-subtracted reduction of binge eating and a 31% placebo-subtracted reduction of vomiting episodes per week. Discontinuation rates were 37.2% for placebo and 30.2% for FLX [13]. Similar reductions in bulimic behaviors were demonstrated in a 16-week study (n = 398, 96.2% were female; DSM III-R defined BN) [14]. FLX (60 mg/d) had a 32% placebo-subtracted reduction in binge eating and a 29% placebo-reduction reduction of vomiting episodes per week. Discontinuation rates were 52% for placebo and 40.5% for FLX [14]. Recent evidence suggest that FLX is still considered effective for the management of BN, but approximately 60% of BN patients do not achieve remission with FLX or available psychotherapies or their combination [15].
The other available pharmacotherapy is LDX, which was initially FDA-approved for the management of attention deficit hyperactivity disorder (ADHD) in 2007 for children (6–12 years old) and 2008 for adults. In 2015, LDX was FDA-approved for the maintenance of moderate to severe BED [16]. Several randomized placebo-controlled phase III studies were conducted to determine the efficacy of LDX for BED. Two 12-week studies (Study 1: n = 379, 86.5% were female; Study 2: n = 366, 85.2% were female; DSM-IV TR defined BED; NCT01718483 and NCT01718509) demonstrated LDX (50 or 70 mg/day) had a 26.3% placebo-subtracted reduction (study 1) and a 37.6% placebo-subtracted reduction (study 2) in binge eating days/per week [17]. Discontinuation rates were 18% and 25.9% for placebo and 17.7% and 26.5% for LDX for each study, respectively [17]. Efficacy of LDX was further established in a 26-week study (n = 418, 87.1% female; DSM-IV TR defined BED; NCT02009163), which demonstrated a reduced relapse risk for BED patients [18]. LDX-treated subjects had a 3.7% relapse rate compared with 32.1% relapse rate in placebo-treated subjects [18]. LDX is also likely to improve the high impulsivity reported in some BED populations [19]. Using a within-subjects design, it was demonstrated that 8-week LDX (50 mg/day or 70 mg/day) decreased binge eating frequency in BED subjects (n = 41, 97.6% female; DSM-V defined BED) [19]. Notably, binge episode frequency was also quantified as binge days per week rather than discrete binge episodes, since self-reporting of when binge episodes start and finish can be difficult to delineate or recall [17]. Additional medications initially approved to treat other psychiatric illnesses have been investigated in BED or BN populations [20,21,22,23,24] and are reviewed in detail elsewhere [25,26].
Concerns for the safety and tolerability of pharmacotherapies for BN and BED arise on several levels. The frequency and severity of treatment emergent adverse events represents a major contributor to the discontinuation rate in efficacy trials [14,27]. Pregnancy risks are major concerns for women of child-bearing age, which represents the predominant age range of women seeking therapeutic assistance for eating disorders. For instance, LDX and FLX has not been systematically tested for their effects on a developing fetus, but there is ample evidence in rodent and non-rodent species to suggest a pregnancy risk (FDA Pregnancy category C) [12,28]. LDX also has an abuse and dependency potential with a long-term risk to elevate cardiovascular endpoints [29]. For the most part, though, FLX and LDX are considered to be well-tolerated medications for the treatment of BN and BED, respectively.
Unlikely other classes of medications [30], to date, there is no guidance for industry on developing pharmacotherapies for the clinical management of binge eating. However, in the FDA application for LDX for moderate to severe BED, there was a stated assertion by the sponsor that a mean reduction of 0.5 or more binge days per week was “clinically meaningful” [31]. Additional endpoints include number of subjects with total remission of binge eating and/or vomiting, improvements in structured diagnostic assessments (i.e., Y-BOCS-BE, BES, EDI, CGI, BDI, etc.), and triglyceride levels in BED patients [13,17,28,31]. As such, the primary endpoint for determining efficacy of a pharmacotherapy for BED or BN is a sustained significant reduction in the frequency of binge (BN/BED) or vomiting (BN) episodes [13,20]. A reduction in binge frequency outcomes could also represent only one component in multi-stage optimization innervation [32]. Nonetheless, reduction in binge frequency represents a measurable behavioral endpoint for determining pharmacotherapeutic effectiveness.
Animal models, specifically rodent models, are used to advance the screening and development of potential pharmacotherapies for psychiatric disorders [33,34]. In addition, animal models can assist in the early identification of some safety and tolerability-related issues. No animal model, however, can fully encompass all the intricacies of a psychiatric disorder. To be relevant and effective, animal models should be developed to ask specific hypothesis-testable questions for particular features of the complex psychiatric disease under investigation [35]. Within the field of eating disorders, binge eating has several core components that can be recapitulated in rodents for assessing the effectiveness of potential pharmacotherapies. While the sense of ‘loss of control’ of binge eating is a pathological feature that is difficult to objectively assess in animals, two aspects of binge eating that can be recapitulated are excessive ingestion of calories in a short period of time and the episodic pattern of eating. In addition, some rodent binge eating models have incorporated a caloric restriction component to mimic restrictive eating or cycling dieting influencing binge eating behaviors [10]. Rats and mice lack an emetic response [36], so calorie restriction in animal models also serves as a method of assessing the influence of calorie restrictive conditions on eating without vomiting. The purpose of this systematic review is not to further debate on which experimentally controlled factors appropriately recapitulate the core features of a clinical eating disorder. The purpose of this systematic review is to provide an overview of the potential pharmacotherapies or compounds tested in established rodent models of binge eating behavior. This information can be used to develop hypothesis-testable questions on pharmacotherapeutic options, standardized approaches to assess candidate drugs, and to provide guidance on future experimental design approaches building on existing findings in binge eating rodent models.
2. Materials and Methods
This systematic review was performed using Pubmed (https://pubmed.ncbi.nlm.nih.gov/; accessed 31 December 2022) and APA PsycInfo (https://psycnet.apa.org/search) web-based databases (accessed 31 December 2022). The Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (PRISMA) flowchart was used in the literature selections [37]. The following terms were used: “binge eating”, “binge eating disorder”, “bulimia nervosa”, “animal models”, “rodents”, “rat”, “mouse”, “intermittent”, “intermittent access”, “intermittent palatable food”, “dietary binge eating”, “stress eating”, “stress overeating”, “stress-induced binge eating”, “binge-like eating”, “binge-type eating”, and “binge feeding”. The searches were performed to include all studies with a full publication date before 1 January 2023 (not epub date). Inclusion criteria were studies published as peer-reviewed articles, written in the English language, and definition of the feeding schedule as “binge” or “binge-like” or “binge-type”. Articles were further selected based on pharmacology endpoints to included compounds, agents, drugs, or medication interventions on feeding outcomes. Exclusion criteria were studies that used liquid (i.e., Ensure® or chocolate drink) or sweetened solutions as binge foods. Our rationale for excluding liquid binge food studies was that while these represent related ingestive behaviors and brain processes, fluid intake is also regulated by osmotic and volumetric factors. These factors are distinguishable from eating. Additional studies were excluded that had alcohol- and/or drugs of abuse-related outcomes. Studies were also excluded if the binge paradigm with regards to binge food composition, access, frequency, or duration were poorly defined.
3. Results
3.1. Study Charactersitics and Selection
As detailed in Figure 1, there were a total of 434 studies screened, and 67 studies were used for systematic review. Studies were further classified based on targeted systems, combinations, or comparisons between compounds. These studies are summarized in Table 1, Table 2, Table 3, Table 4 and Table 5. Given the differences in study design, approach, binge food, and major pharmacological outcomes, the summary of these studies is best represented in a table format.
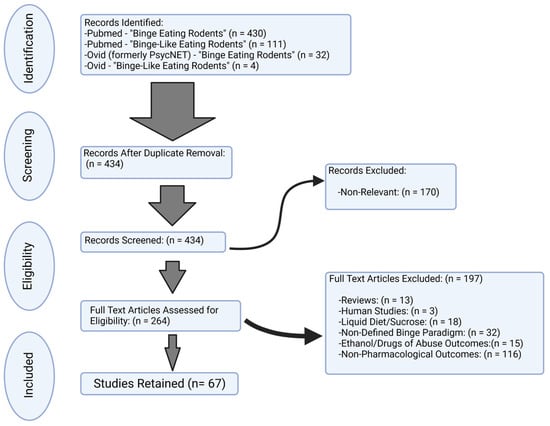
Figure 1.
Flow chart for inclusion/exclusion of peer-reviewed studies. In accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, a total of 67 studies were retained for the examination of pharmacological or compound assessment of binge-like eating behavior in rodents.
3.2. Biogenic Amines and Related Compounds
As described in Table 1, 16 studies [38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53] were identified that used compounds acting on the biogenic amine systems.

Table 1.
Biogenic amines and related compounds. Studies using adrenergic/noradrenergic, dopaminergic, serotonergic, GABA, and glutamatergic compounds.
Table 1.
Biogenic amines and related compounds. Studies using adrenergic/noradrenergic, dopaminergic, serotonergic, GABA, and glutamatergic compounds.
| Adrenergic/Noradrenergic | |||
| Study | Methods | Details | Major Findings |
| Hicks et al., 2020 [50] | Species: Rat Binge Food: Chocolate-flavored high-sucrose (50% kcal) AIN-76A-based pellets Access: 1 h Frequency: Daily Duration: >12 days Calorie Restriction: None | Male Wistar rats were training to nose poke for binge food or standard chow pellets on fixed ratio and progressive ratio (PR) testing. The dose-dependent effects of prazosin (0.5–2 mg/kg; IP) on binge food responding were assessed. |
|
| Bello et al., 2014 [43] | Species: Rat Binge Food: Vegetable shortening blended with 10% sucrose Access: 30 min Frequency: 2 non-consecutive days per week Duration: 6 weeks Calorie Restriction: Two groups of rats had a 24-h calorie restriction prior to the binge period twice a week (Restrict). | Female Sprague Dawley rats were exposed to the binge eating paradigm with or without intermittent calorie restriction. c-Fos immunoreactivity was measured in the medial prefrontal cortex (mPFC) and hypothalamic paraventricular nucleus (PVN) in response to binge food, with or without acute restraint stress (1 h). In another set of experiments, female rats were implanted with an osmotic minipump to provide a sustained release of guanfacine, alpha adrenergic 2A receptor agonist (0.5 mg/kg/day) or vehicle to determine the effects of binge food intake. |
|
| Bello et al., 2014 [44] | Species: Rat Binge Food: Vegetable shortening blended with 10% sucrose Access: 30 min Frequency: 2 non-consecutive days per week Duration: 10 weeks Calorie Restriction: Two groups had a 24 h calorie restriction prior to the binge period twice a week. (Restrict) | Male Sprague Dawley rats were exposed to the binge eating paradigm with or without intermittent calorie restriction. Noradrenergic involvement was assessed by feeding suppression to a selective norepinephrine reuptake inhibitor, nisoxetine (3 mg/kg; IP), dorsal noradrenergic bundle lesions with neurotoxin (DSP4), and single-unit electrophysiological recording of locus coeruleus (LC) neurons. |
|
| Dopaminergic | |||
| Study | Methods | Details | Major Findings |
| Presby et al., 2020 [51] | Species: Rat Binge Food: Ground Cadbury’s Dairy Milk Chocolate Access: 2 h Frequency: 12 non-consecutive days (1–3 days apart) Duration: 4 weeks Calorie Restriction: 85% of ad libitum feeding weights for 18 weeks. After that, 18 weeks of ad libitum feeding and normal weight before starting the binge feeding protocol. | Female Wistar rats were exposed to standard lab chow and divided into three groups, +chocolate, +standard chow, and +empty feeding dish. Effects of lisdexamfetamine (LDX; 0.185–1.5 mg/kg; IP) on operant responding and intake were measured. |
|
| Vickers et al., 2017 [47] | Species: Rat Binge Food: Ground chocolate (Cadbury’s Dairy Milk) Access: 2 h Frequency: Intermittent on Days 1, 2, 4, 6, 7, 9, 12, 14, 15, 18, 23, 25 and 28. Duration: >28 days Calorie Restriction: None | Female Wistar were trained on operant delay discounting lever pressing task (immediate vs. delayed reward) before exposure to the binge eating paradigm. Rats were dosed in a cross-over design with oral lisdexamfetamine (LDX; 0.3 mg/kg or 0.8 mg/kg) or vehicle after 2 weeks on the binge eating paradigm. |
|
| Heal et al., 2016 [46] | Species: Rat Binge Food: Ground chocolate (Cadbury’s Dairy Milk) Access: 2 h Frequency: Intermittent on Days 1, 2, 4, 6, 7, 9, 12, 14, 15, 18, 23, 25 and 28. Duration: >28 days Calorie Restriction: None | Female Wistar were exposed to the binge eating paradigm or no access to binge food (control) and trained on conditioned avoidance response task. Binge or control rats were dosed with lisdexamfetamine (LDX: 0.8 mg/kg) or vehicle to assess performance in novel food reward/punishment conflict paradigm. |
|
| Halpern et al., 2013 [42] | Species: Mouse Binge Food: High-fat diet (HFD; 60% fat, 20% carbohydrates, 20% protein) Access: 1 h Frequency: Daily Duration: 8 days (3 days; 5 treatment days) Calorie Restriction: None | Male C57BL/6J mice were implanted unilaterally into the left nucleus accumbens shell (NAS), core or dorsal striatum with a bipolar tungsten electrode. Mice were exposed to the binge eating paradigm and deep brain stimulation (DBS) with 150 µA on two non-consecutive days. Pharmacological blockage of DBS with D1R antagonist (SCH-23390, 0.075 mg/kg; IP) or D2 antagonist (raclopride; 3 mg/kg; IP) was also performed. |
|
| Serotonergic | |||
| Study | Methods | Details | Major Findings |
| Karth et al., 2022 [53] | Species: Mouse Binge Food: High-fat diet (39.7% Fat) Access: 24 h Frequency: Once per week Duration: >3 weeks Calorie Restriction: None | Male and female mice homozygous for a knockin (KI) of tryptophan hydroxylase 2 (Tph2; R439H) or littermate contols (WT). Tph2 KI mice have reduced (~60–80%) serotonin levels. FLX was administered acutely (30 mg/kg; or saline; IP) or chronically (150 mg/L or unadulterated drinking water) for 21 days. Serotonin-related gene expression was measured by qPCR from the raphe nucleus. |
|
| Blumenthal and Pratt, 2018 [48] | Species: Rat Binge Food: Vegetable shortening Access: 2 h Frequency: Daily Duration: ~22 days Calorie Restriction: None | Male Sprague Dawley rats were implanted with bilateral intracranial cannulas targeting the nucleus accumbens core. Rats received vehicle or DAMGO (mu-opioid receptor agonist; 0.025 μg) prior to the binge access. In addition, rats received fenfluramine (0, 0.6, or 3.0 mg/kg; IP) or lorcaserin (0, 0.3, or 1.0 mg/kg; IP). |
|
| Price et al., 2018 [49] | Species: Rat Binge Food: High-fat diet (45%) Access: 2 h Frequency: Once per week Duration: 5–9 weeks Calorie Restriction: None | Adult male Sprague Dawley rats were IP administered a novel 5HT-2cR agonist, WAY163909, 0.5, 1.0 or 2.0 mg/Kg) prior to binge access. |
|
| Chandler-Laney et al., 2007 [41] | Species: Rat Binge food: Double-Stuff Oreo cookies Access: 3 cycles of restriction–refeeding, each lasting 12 days. Refeeding consisted of presentation of cookies for a short period, followed by ad lib chow. Frequency: 12-day cycle: 5 days ad lib chow or restriction, 2 days ad lib chow and cookies, 4 days ad lib chow, 1 day stress/no stress. Duration: 6 cycles of 12 days of restriction/refeeding followed by a test day on day 13. Calorie Restriction: 66% of ad libitum standard chow. Stress: Acute foot shock. | Female Sprague Dawley rats were exposed to a binge paradigm with or without stress or restriction. The central effects of FLX (3 mg/kg;IP) on binge eating were asseseed. Levels of serotonin, dopamine, and metabolites were evaluated from key brain regions involved in feeding and reward. Serotonin, dopamine, norepinephrine, and their metabolites were measured using high-performance liquid chromatography (HPLC). |
|
| Placidi et al., 2004 [38] | Species: Rat Binge food: Nabisco Oreo cookies (4.7 kcal/g; 57%, 40%, and 3% of kcal from carbohydrates, fats, and protein, respectively. Access: 24 h Frequency: After every R-R/S cycle the animals had access to binge food and standard chow. Ad libitum access during test period. Duration: ~2 months Calorie Restriction: 4 days of 66% of control food intake followed by 6 days of free feeding (R + NS and R + S groups). | Female Sprague Dawley rats were exposed to binge paradigm with or without stress (i.e., acute foot shock; 0.6 mA). The role of serotonergic function in binge eating behaviors was investigated, specifically the influence of FLX (2 mg/kg; IP). |
|
| GABAergic | |||
| Study | Methods | Details | Major Findings |
| Wojnicki et al., 2006 [40] | Species: Rat Binge food: Crisco All-Vegetable shortening Access: 1 h Frequency: Daily (D) or intermittent (INT) 3 days/week. Duration: 4–6 months Calorie Restriction: None | Operant performance of 18 non-food deprived male Sprague Dawley rats was assessed under progressive ratio (PR) and concurrent PR-fixed ratio schedules of chow pellets and/or vegetable shortening reinforcement. Rats were then divided into 2 groups based on schedule of binge food access: High-Restriction binge group (1 h access 3 days/week) and Low-Restriction binge group (1 h access daily). The effects of (R)-baclofen (0.3, 0.6, 1.0 and 1.8 mg/kg) on operant performance were assessed. |
|
| Buda-Levin et al., 2005 [39] | Species: Rat Binge food: Vegetable shortening, chow + shortening mixture. Access: Vegetable shortening for 2 h on Monday, Wednesday, and Friday (MWF), continuous access to chow + shortening mixture, or standard chow alone. Frequency: Daily (D) or Intermittent (INT) on non-consecutive days, 3 days a week. Duration: ~4 months Calorie Restriction: None (30 min post-injection) | Male Sprague Dawley rats were divided into 3 groups: Binge (B), Fat-Matched (FM), and Chow (C) groups. Animals were exposed to binge food in INT or D. The effect of the GABA-B agonist, baclofen (0, 0.3, 0.6, 1.0, 1.8 mg/kg IP), was assessed for the reduction of binge food intake. |
|
| Glutamatergic | |||
| Study | Methods | Details | Major Findings |
| Oliveira et al., 2021 [52] | Species: Mouse Binge Food: High-fat diet: (45% fat) Access: Ad libitum, assesed 2.5 h after caloric restriction. Frequency: Daily Duration: 15 weeks Calorie Restriction: One period of 14 h food deprivation at week 12 prior to drug treatment. | The metabotropic glutamate receptor 5 (mGluR5) is known to modulate central reward pathways. The effect of the mGluR5 negative allosteric modulator VU0409106 (3, 7.5, or 15 mg/kg) or FLX (10 mg.kg) in regulating feeding and obesity parameters in diet-induced obese C57BL/6 mice was assessed. Food intake, body weight, inflammatory/hormonal levels, and behavioral tests were measured/performed. |
|
| Smith et al., 2015 [45] | Species: Rat Binge Food: Chocolate-flavored high-sucrose (50% kcal) AIN-76A-based pellets Access: 1 h Frequency: Daily Duration: >12 days Calorie Restriction: None | Male Wistar rats were trained on binge eating paradigm to operant respond (FR1) for binge food. Rats were administered an uncompetitive N-methyl-D-Aspartate antagonist, memantine (0, 1.25, 2.5, 5, and 10 mg/kg, IP), 30 min prior to the binge food or standard diet access. Another group of rats had cannula targeting the nucleus accumbens (NAcc) core or shell and received (0, 2.5, 10, and 20 µg/side) immediately before access. |
|
3.2.1. Noradrenergic/Adrenergic-Acting Compounds
Three compounds (prazosin, guanfacine, and nisoxetine) were used to target the central noradrenergic system in binge eating rodents [43,44,50]. Prazosin, an α-1 adrenergic receptor antagonist, increased the progressive ratio break point for binge food at all doses tested (0.5–2 mg/kg) in male rats [50]. Chronic guanfacine, an α2A-adrenergic receptor agonist (0.5 mg/kg/daily), increased binge eating in female rats exposed to limited binge food access without intermittent calorie restriction [43]. The selective norepinephrine reuptake inhibitor, nisoxetine (3 mg/kg), produced a feeding suppression in all groups, except the binge group with a history of intermittent calorie restriction [44].
3.2.2. Dopaminergic-Acting Compounds
Three compounds (LDX, SCH23390, and raclopride) were used to target the central dopaminergic system [42,46,47,51]. LDX at 1.5 mg/kg reduced operant responding for binge food in female rats [51]. At 0.8 mg/kg, LDX normalized binge eating-induced delay discounting and binge eating-induced avoidance behaviors in female rats [46,47]. In male mice with deep brain stimulation (DBS)-induced binge eating, raclopride (3 mg/kg), a dopamine D2 receptor antagonist, effectively reduced DBS-induced binge eating [42]. In contrast, SCH23390 (0.75 mg/kg), a dopamine D1 receptor antagonist, was not effective in reducing DBS-induced binge eating [42].
3.2.3. Serotonergic-Acting Compounds
Four compounds (FLX, fenfluramine, lorcaserin, and WAY163909) were used to target the central serotonergic system [38,41,48,49,53]. A selective serotonin reuptake inhibitor (SSRI), FLX was administered a single peripheral dose (30 mg/kg) or chronic dose (150 mg/L in drinking water for 21 days) to assess binge eating in mice with a knock-in of tryptophan hydroxylase 2 (Tph2) [53]. Peripherally administered FLX at 3 mg/kg reduced binge eating in female rats with a history of caloric restriction [41], whereas FLX at 2 mg/kg reduced binge eating in female rats exposed to acute foot shock stress [38]. Peripherally administered serotonergic drugs, such as fenfluramine (3 mg/kg), and lorcaserin (1 mg/kg), were used to assess intra-accumbens DAMGO-induced binge eating [48]. In addition, a novel 5HT2c receptor agonist, WAY163909, was effective at reducing binge eating at 1 mg/kg and at 2 mg/kg in male rats [49].
3.2.4. γ-Aminobutyric Acid (GABA)-Ergic Compounds
The GABAergic system was targeted in two studies using the GABAB agonist, baclofen, on binge eating in male rats [39,40]. Baclofen at 1 mg/kg reduced lever responding for binge food [40], whereas at 1.8 mg/kg baclofen reduced binge food intake under all access conditions [39].
3.2.5. Glutamatergic Compounds
The glutamatergic system was targeted with two compounds (memantine and VU0409106) [45,52]. VU0409106, a negative allosteric modulator of the metabotropic glutamate receptor 5 (mGluR5), reduced binge eating in mice at 7.5 or 15 mg/kg [52]. The uncompetitive N-methyl-D-Aspartate antagonist, memantine, (2.5, 5, or 10 mg/kg), administered peripherally and site-specifically into the nucleus accumbens shell (10 or 20 µg), reduced binge eating in male rats [45].
3.3. Opioidergic Compounds
As described in Table 2, there were a total of five studies [54,55,56,57,58] that targeted the opioid system. The opioid antagonists, naloxone (0.1–1 mg/kg) and naltrexone (1 or 3 mg/kg), showed reductions in binge eating in rats following peripheral administration [54,55,56]. The novel opioid antagonist, GSK1521498 (1 or 3 mg/kg), reduced binge eating in male and female rats [56]. Peripherally administered naloxone (10 or 30 mg/kg) or intra- paraventricular hypothalamic nucleus naloxone (10–100 nmol) reduced intra-accumbens DAMGO-induced binge eating [58]. Two studies demonstrated the binge promoting effects of butorphanol (8 mg/kg), an opioid agonist/antagonist, on binge eating in female rats [55,57].
3.4. Hormone Analogs
As described in Table 3, there were a total of 12 studies that targeted hormonal signaling pathways [59,60,61,62,63,64,65,66,67,68,69,70].
3.4.1. Estrogenic Compounds
The targets of sex steroids were examined in seven studies [59,60,61,62,63,64,65]. Several studies examined the effects of estradiol (E2) on binge eating behavior in ovariectomized (OVX) female mice. Results were mixed with E2 either reducing or not influencing binge eating behaviors [59,62,63,64,65]. E2 treatment combined with progesterone reduced binge food intake compared with the vehicle or progesterone alone [64,65]. However, the estrogen metabolite, 2-hydroxy-estradiol (2OH-E2), increased binge eating behavior in male and female rats [60,61].
3.4.2. Corticotropin-Releasing Factor (CRF) and Stress-Related Compounds
Compounds targeting stress-related hormonal and brain signaling were examined in two studies [68,69]. Specifically, CRF receptor antagonists, centrally administered D-Phe-CRF (12–41) or peripherally administered R121919 (20 mg/kg), reduced binge eating in rats with a history of calorie restriction and stress exposure [68,69]. Neither exogenous corticosterone nor metyrapone influenced binge eating [69].
3.4.3. Gut Peptide Analogs
Three studies used compounds that targeted glucagon-like peptide-1 (GLP-1) and ghrelin signaling [66,67,70]. Intra-accumbens administration of GLP-1 receptor agonist (exendin 4; ex4; 0.05 and 0.1 µg) and antagonist (exendin 9; ex9; 2.5 and 5 µg) reduced and increased, respectively, mu-opioid-stimulated binge eating [70]. Neither intra-accumbens ex4 nor ex9 alone influenced binge food intake [70]. Acute central ghrelin (1 or 2 µg) reduced binge food preference, whereas chronic central ghrelin (0.5 μg/h for 28 days) increased binge food intake in male rats [66]. Central ghrelin signaling was further delineated by using exogenous LEAP2, an antagonist for ghrelin receptor, and K-(D-1-Nal)-FwLL-NH2, a growth hormone secretagogue receptor (GHSR) inverse agonist, to reduce binge eating in male mice [67].
3.5. Additional Individual Compounds
As described in Table 4, there were a total of 17 studies that examined additional compounds individually that were insufficient in the number of studies to justify a single table or category [71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87].

Table 2.
Opioids and related compounds. Studies using opioidergic compounds and related targets.
Table 2.
Opioids and related compounds. Studies using opioidergic compounds and related targets.
| Opioidergic | |||
|---|---|---|---|
| Study | Methods | Details | Major Findings |
| Blasio et al., 2014 [54] | Species: Rat Binge Food: Chocolate-flavored high-sucrose (50% kcal) AIN-76A-based pellets Access: 1 h Frequency: Daily Duration: >12 days Calorie Restriction: None | Male Wistar rats were administered naloxone (0.03–0.3 mg/kg, SQ) or microinfused (5 µg or 25 µg/side) into the nucleus accumbens (NAcc) shell or medial frontal cortex (mPFC) to assess FR1 or breakpoint for binge food. Gene expression of opioid-related genes (POMC, PDyn and PEnk) was assessed in NAcc or mPFC. |
|
| Giuliano et al., 2012 [56] | Species: Rat Binge food: Chocolate-flavored pellets Access: 1 h chow access followed by 2 h food deprivation, 10 min access to chow feeder, and 10 min access to another chow feeder. Frequency: 15 consecutive days (preference) Duration: >2 months Calorie Restriction: 18 g/day lab chow for two weeks prior to the food-seeking experiment. | Male and female Lister Hooded rats were exposed to binge eating and food-seeking paradigms. The effects of the novel mu-opioid receptor antagonists, GSK1521498 (0.1, 1, and 3 mg/kg; SC) or Naltrexone (NTX, 0.1, 1, and 3 mg/kg; SC), on food seeking behavior (measured in rats trained to respond for reward under a second-order schedule of reinforcement) and binge-like eating were assessed. |
|
| Naleid et al., 2007 [58] | Species: Rat Binge food: Choice of fat diet (77% fat) or sucrose diet (69% carbohydrate) Access: 3 h Frequency: Daily (D) Duration: ~3 weeks per experiment following one-week post-surgery recovery time. Total ~2 months. Calorie Restriction: 80% of free-feeding intake for NTX no-choice experiment only. | Male Sprague Dawley rats were implanted bilaterally with guide cannula in the PVN. The effects of the mu-opioid agonist DAMGO (0, 0.025, 0.25, and 2.5 nm, injected unilaterally to stimulate feeding) and the general opioid antagonist NTX (0, 10, 30, and 100 nmol, injected bilaterally) to inhibit feeding in consumption of fat and sucrose diets were assessed. The effect of peripheral subcutaneous injections of NTX (SQ; 0, 0.03, 0.1, and 0.3 mg/kg) was also assessed. |
|
| Boggiano et al., 2005 [55] | Species: Rat Binge food: Oreo cookies Access: Binge eating was produced in the R + S group through 3 cycles of restriction-refeeding, each of which lasted 12 days. Refeeding consisted of presentation of cookies for a short period, followed by ad lib chow. Frequency: 12-day cycle: 5 days ad lib chow or restriction, 2 days ad lib chow and cookies, 4 days ad lib chow, 1 day stress/no stress. Duration: 3–17 cycles of 12 days of restriction/refeeding followed by a test day on day 13. | Female Sprague Dawley rats were exposed to the binge paradigm with or without stress or restriction. The groups were as follows: Restriction and Stress (R + S), Restriction but No Stress (R + NS), Stress but No history of Restriction (NR + S), and a group with No History of Restriction or Stress (NR + NS) The effects of naloxone (1 mg/kg) and butorphanol (8 mg/kg) on binge eating were assessed. |
|
| Hagan & Moss, 1991 [57] | Species: Rat Binge Food: Standard chow Access: 1–4 h Frequency: Daily Duration: >1 year Calorie Restriction: Limited access to 4 h per day for first deprivation (DEP) episode, 1-to-3 h for 2nd and 3rd episodes. | Female Sprague Dawley rats were deprived for 3 episodes down to 75% normal body weight (1st episode) or 80% normal body weight (2nd and 3rd episodes) by limiting access to standard chow to 4 h per day (1st episode) or 2–3 h per day (2nd and 3rd episodes), followed by recovery to normal weight. Control group had ad libitum access. Once recovered, food was removed 40–60 min before testing. Intake of chow was measured 1, 2, and 3 h after butorphanol (SQ; 8 mg/kg) or vehicle. |
|

Table 3.
Hormone analogs and related compounds. Studies that use estrogenic/androgenic, stress-related, or gut peptides compounds or their combination.
Table 3.
Hormone analogs and related compounds. Studies that use estrogenic/androgenic, stress-related, or gut peptides compounds or their combination.
| Estrogenic | |||
| Study | Methods | Details | Major Findings |
| Anversa et al., 2020 [59] | Species: Mouse Binge Food: Reese’s peanut butter drops and Nestlé chocolate drops. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Day 8 was the stress. Duration: 16 days, 2 cycles Calorie Restriction: 68% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Male and female C57BL/6J mice. A subset of female mice received ovariectomy (OVX), OVX + estrogen replacement, sham, or control (no surgery or stress). |
|
| Brutman et al., 2019 [62] | Species: Rat Binge Food: High-fat diet (40% fat) Access: 2 h Frequency: Every 3rd day (HFD-3D) or every day (HFD-ED) Duration: 45 days Calorie Restriction: None | Female Long Evans rats were exposed to HFD 3D, HFD-ED, or chow. Following the binge paradigm, rats were training for operant responding for sucrose pellets. A subset of rats underwent ovariectomy (OVX), or sham OVX, with or without estrogen (E2) treatment. |
|
| Di Bonaventura et al., 2017 [63] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were assessed by the vaginal cytology to determine ovarian phase. Additionally, rats underwent ovariectomy and received estrogen (E2) or oil (OIL) to determine the influence on binge eating. Brain region expression of phosphorylated extracellular signal-regulated kinases (pERK) were assessed. |
|
| Yu et al., 2011 [65] | Species: Rat Binge food: Vegetable shortening. Access: 1 h Frequency: Intermittent 3 non-consecutive days (INT) per week. Duration: 4 weeks Calorie Restriction: None | Female ovariectomized Sprague Dawley rats were assigned to 1/3 of 4-day cyclic hormone treatments: estradiol (E) benzoate, progesterone (P), both, or oil vehicle, followed by an INT binge feeding protocol. The individual roles of estradiol (E) and progesterone (P) in the control of food intake and body weight in ovariectomized rats were assessed. |
|
| Babbs et al., 2013 [60] | Species: Rat Binge Food: Vegetable shortening Access: 1 h in week 1, 40 min in week 2, and 20 min in weeks 3 and 4. Frequency: 3 times a week on non-consecutives days Duration: 4 weeks Calorie Restriction: None | Female Sprague Dawley rats were ovariectomized (OVX) and received 2-hydroxyestradiol (2-OHE2) or vehicle via osmotic pump. Binge eating behavior was also compared with sham OVX (Intact). Binge eating male Sprague Dawley rats were implanted with microdialysis cannula targeting the prefrontal cortex (PFC) to measure dopamine (DA) efflux-administered acute vehicle or 2-OHE2 (3 μg/kg; IP). |
|
| Babbs et al., 2011 [61] | Species: Rat Binge food: Vegetable shortening Access: 1 h Frequency: Daily (D) or Intermittent (INT) on non-consecutive days, 3 days a week. Duration: >5 weeks Calorie Restriction: None | Two cohorts (1 male and 1 female) of Sprague Dawley rats were exposed to a daily (D) or intermittent (INT) limited-access, binge eating paradigm for 5 weeks. Assessment of the estrogen system on binge eating behavior of 2OHE2 (1, 3, and 10 ug/kg; IP), vehicle, or 2-methoxyestradiol (2ME2; 3.0 µg/kg; IP) immediately prior to fat access following the 5-week binge-induction period. |
|
| Yu et al., 2008 [64] | Species: Rat Binge food: Vegetable shortening Access: 1 h Frequency: Daily (D; low restriction) or Intermittent (3 non-consecutive days; INT; high restriction) per week. Duration: 6 weeks Calorie Restriction: None | Ovariectomized female Sprague Dawley rats were exposed to a binge feeding paradigm of either chow only, INT, or D. The effect of ovarian hormones E (estradiol benzoate and P (progesterone), 2 ug/100 µL and 500 µg/100 µL sesame oil or oil vehicle, respectively, injected every 4 days) on binge feeding behavior was assessed. |
|
| Corticotropin-releasing factor (CRF)/Stress-Related Studies | |||
| Study | Methods | Details | Major Findings |
| Di Bonaventura et al., 2017 [69] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were exposed to the binge eating paradigm with CRF1 receptor antagonist R121919 (IP; 0,10. 20 mg/kg) or metyrapone (IP; 0, 50, 100 mg/kg). Non-stressed rats were treated with corticosterone (IP; 2.5 mg-15 mg/kg). In another set of rats, crhr1 mRNA was assessed in the BNST, CeA, BLA, and paraventricular nucleus. Lastly, binge eating was assesed following CeA or BLA injections of the CRF receptor antagonist D-Phe-CRF (12–41) (300 ng/side). |
|
| Di Bonaventura et al., 2014 [68] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were exposed to a binge eating paradigm with or without stress or restriction. The systemic effects of the CRF-1 receptor antagonist R121919 (10 or 20 mg/kg, SQ) were assessed. Intraventricular injections (ICV) or intra-BNST injection of nonselective CRF receptor antagonist D-Phe-CRF (12–41) (ICV dose 100–1000 ng; BNST dose; 10–50 ng/side) were also determined. Regional C-Fos immunoreactivity (IR) was assessed. |
|
| Gut peptides | |||
| Study | Methods | Details | Major Findings |
| Pierce-Messick and Pratt, 2020 [70] | Species: Rat Binge Food: High-fat, high-sucrose diet Access: 2 h Frequency: Daily (D) Duration: ~6 weeks Calorie Restriction: None | Male Sprague Dawley rats were implanted with bilateral cannula above the nucleus accumbens core. The effects of nucleus accumbens injections of the GLP-1 receptor agonist exendin 4 (EX4) (0, 0.05, and 0.1 µg/side) of antagonist exendin 9 (EX9) (0, 2.5, and 5.0 µg/side) on binge food consumption with and without simultaneous mu-opioid receptor stimulation (via DAMGO; 0.25 ug/side) was assessed. |
|
| Cornejo et al., 2019 [67] | Species: Mouse Binge Food: High palatable pellets (21.1% fat, 22.5% carbohydrates) Access: 2 h Frequency: Daily Duration: 4 consecutive days Calorie Restriction: None | Male C57BL/6J mice were investigated for growth hormone secretagogue receptor (GHSR) activity. Ghrelin (300 pmol; ICV) and LEAP2 (liver-expressed antimicrobial peptide 2; 600 pmol/g SQ; 600 pmol ICV), [D-Lys3]-GHRP-6 (2 nmol ICV), JMV2959, (3 nmol ICV), and K-(D-1-Nal)-FwLL-NH2 (1 nmol; ICV) were used to investigated GHSR in the periphery and brain. |
|
| Bake et al., 2017 [66] | Species: Rat and Mouse Binge Food: High-fat diet (60% Fat) Access: 2 h Frequency: Daily Duration: >2 weeks Calorie Restriction: None | Male Sprague Dawley rats were implanted with intracerebroventricular (ICV) cannulae or intra-VTA cannulae and administered acute ghrelin (1 or 2 µg/mL) prior to the binge. In a subset of rats, chronic ICV ghrelin (0.5 μg/h for 28 days) with 10 days standard chow/18 days on binge paradigm was evaluated. Another set of ghrelin receptor knockout (GHS-R KO) mice were examined for their binge phenotype. |
|

Table 4.
Additional compounds. Studies using PACAP, orexin, and other novel targeting compounds.
Table 4.
Additional compounds. Studies using PACAP, orexin, and other novel targeting compounds.
| Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) | |||
| Study | Methods | Details | Major Findings |
| Le et al., 2021 [80] | Species: Mouse Binge Food: High-fat diet Access: 1 h Frequency: Daily Duration: 5 days Calorie Restriction: None | Male and female C57BL/6/J (WT) tyrosine hydroxylase (TH-CRE) and pituitary adenylate cyclase-activating polypeptide (PACAP:CRE) mice (on a C57BL6/J background) were used. Experiments were designed to examine the ventromedial nucleus (VMN) PACAP neurons projects to the ventral tegmental area (VTA) and binge eating. Female WT mice were ovariectomized (OVX) and received vehicle or SQ estrogen daily. Immediately before binge access, WT mice were injected with PACAP1–38 (30 pmol, 0.2 μL) or 0.9% saline vehicle (0.2 μL) into the VTA. Electrophysiological recordings from TH-CRE and PACAP-CRE were taken. |
|
| Hurley et al., 2016 [77] | Species: Rat Binge Food: Western diet (41% Fat) Access: 15 min (meal 2; M2) Frequency: Daily Duration: 2 weeks Calorie Restriction: Daily 2 h standard chow access (meal 1; M1), 30 min before M2. | Male Sprague Dawley rats were implanted intra-ventromedial hypothalamus (VMN) and intra-nucleus accumbens (NAc) cannula. Binge eating paradigm consisted of M2 as binge food or standard chow as control. Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP; 50 pmol/side) was injected in the either intra-VMN or intra-NAc to investigate the regional PACAP influence on binge-like eating. |
|
| Orexin | |||
| Study | Methods | Details | Major Findings |
| Schuler and Koch, 2022 [87] | Species: Rat Binge Food: Vegetable fat Access: 1 h Frequency: Daily (non-binge) or Every other day (binge) Duration: 6 weeks Calorie Restriction: None | Male Lister Hooded rats were trained and screened for impulsivity on a five-choice serial reaction time task. After exposure to the binge eating paradigm, rats were bilaterally implanted with guide cannula target the NAC shell. Rats received microinfusions of either vehicle, orexin 1 receptor anatagonist (SB-338467; 500 ng/0.3 μL), or cocaine-and amphetamine-regulated transcript antibodies (CARTaB; 1:500) immediately before acess to the binge food. Microinfusions were 2–3 days apart. |
|
| Valdivia et al., 2015 [85] | Species: Mouse Binge Food: High-fat diet (HFD: 22.5% carbohydrate, 22.8% proteins, 21.1% fat, 23.0% fibers, 5.6% minerals, and 5.0% humidity). Access: 2 h Frequency: Daily Duration: 4 days Calorie Restriction: None | C57BL/6J mice (WT; wild type) were exposed to the 1 or 4 day of binge food, ad libitum binge food, or standard chow (no binge food). Regional immunoreactivity (IR) for tyrosine hydroxylase (TH), orexin, or c-Fos was determined. In separate experiments the influence of orexin, by injection of orexin 1 selective antagonist SB-334867 (5 mg/Kg, IP), and ghrelin, by use of growth hormone secretagogue receptor (GHSR)-null mice, were determined. |
|
| Piccoli et al., 2012 [83] | Species: Rat Binge food: Paste mixture of Nutella (52%), food pellets (33%), and water (15%). Access: 2 h for days 5–6 of the 8-day cycle Frequency: Three 8-day cycles: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day. Calorie Restriction: 4 days per week at 66% restriction of ad libitum chow. Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were exposed to a binge eating paradigm of restriction (R) and stress (S). Effects of GSK1059865 (a selective OX1R antagonist) (10 and 30 mg/kg; IP), JNJ-10397049 (a selective OX2R antagonist) (1, 3 mg/kg; IP), and SB-649868 (a dual OX1R/OX2R receptor antagonist) (3, 10 mg/kg; gavage) on binge eating were assessed. |
|
| Sigma (σ) receptors | |||
| Study | Methods | Details | Major Findings |
| Cifani et al., 2020 [72] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were exposed to the binge paradigm or three control conditions: No calorie restriction, No Stress, or No calorie and No Stress. A potent novel sigma1 (σ1), receptor antagonist,1,3-dioxane derivative, 9-Benzyl-3-phenyl-1,5-dioxa-9-azaspiro[5.5]undecane, was evaluated in binge eating rats. |
|
| Del Bello et al., 2019 [74] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were dosed with a novel σ1 receptor antagonist (1, 3, and 7 mg/Kg). Compound was the racemic spipethiane analogue (2-(1-Benzylpiperidin-4-yl)thiochroman-4-one) (±)-1. |
|
| Other targeting compounds | |||
| Study | Methods | Details | Major Findings |
| Kania et al., 2020 [79] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 36 days; test day (day 37). Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were exposed to the binge paradigm or control conditions (No calorie restriction on days 1–4 of the 8-day cycle and No Stress). Relaxin-family peptide-3 receptor (RXFP3) antagonist R3(B1-22)R (1 ug/0.5 uL in ACSF, 0.5 uL bilateral) or vehiclewere injected into the hypothalamic paraventricular nucleus (PVN; magnocellular region) 15 min before access to the binge food on test day. |
|
| Romano et al., 2020 [84] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were exposed to the binge paradigm or three control conditions: No calorie restriction, No Stress, or No calorie and No Stress. Oleoylethanolamide (OEA) was dose-dependently administered (2.5, 5, and 10 mg/kg; IP) to suppress binge eating. OEA effects on binge eating rats were measured by neural activation, c-Fos activation, oxytocin receptor expression, and monoamine turnover. OEA effect on oxytocin on corticotropin-releasing factor (CRF) and oxytocin mRNA in hypothalamic and extrahypothalamic regions were also examined. |
|
| Feltmann et al., 2018 [75] | Species: Rat Binge Food: Chocolate-flavored sucrose pellets Access: 10 min Frequency: daily Duration: 48 days Calorie Restriction: 2 h prior to binge food. | Male Lister Hooded rats were exposed to a binge paradigm of negative anticipatory contrast (10 min chow/10 min binge food). After stable intake, the dose-dependent effects of a novel monoamine stabilizer OSU6162 (SQ; 5, 10, 15, and 30 mg/kg) were assessed. |
|
| Ferragud et al., 2017 [76] | Species: Rat Binge Food: Chocolate-flavored, high-sucrose (50% kcal) AIN-76A-based diet (5TUL: 66.7% carbohydrate, 12% fat) Access: 1 h Frequency: Daily Duration: >15 days Calorie Restriction: None | Male Wistar rats were administered a highly selective Trace Amine-Associated Receptor 1 (TAAR1) full-agonist RO5256390 (0, 1, 3, 10 mg/kg, IP) 30 min prior to binge food access. |
|
| Hurley et al., 2016 [78] | Species: Rat Binge Food: Western diet (41% Fat) Access: 30 min Frequency: Daily Duration: 2 weeks Calorie Restriction: None | Male Sprague Dawley rats were dosed with N-acetylcysteine (NAC) or saline by systemic (90 mg/kg IP) or intraventricular (10 μg ICV) administration. Binge food intake and saccharin (0.15%) conditioned taste aversion (CTA) were assessed. |
|
| Bharne et al., 2015 [71] | Species: Rat Binge Food: Hhigh-fat, sweet palatable diet (HFSPD; 40% fat as vegetable shortening, 40% protein, and 20% carbohydrate with sweetened milk) Access: 2 h Frequency: Every other day Duration: 4 weeks Calorie Restriction: None | Male Wistar rats were implanted with lateral ventricle ICV cannula and the effects of cocaine- and amphetamine-regulated transcript peptide (CART; 1, 1.5 and 2 µg/rat), CART antibody (1:500 and 1:250 dilution, 5 µL/rat), and the CB1 inverse agonist rimonabant (1, 3, and 10 mg/kg; IP) before binge food access. Morphometric analysis of CART immunoreactivity in feeding- and reward-related brain areas was investigated. |
|
| Freund et al., 2015 [86] | Species: Rat Binge Food: All-vegetable shortening. Access: 1 h Frequency: Intermittent on non-consecutive days, 3 days a week. Duration: 5 weeks Calorie Restriction: None | Male and female Sprague Dawley rats received the tricyclic antidepressant clomipramine (CMI, 15 mg/kg; IP; BID) or vehicle during postnatal day (P) 9–16. Spontaneous alternation task, elevated plus maze, and marble burying were performed at three developmental time points P25, P60, and P100. In a separate experiment, rats were observed for sucrose pellet consumption, which began on P60 and P100. In another experiment, rats were exposed to binge eating paradigm on P55. Rats were also classified as binge-prone based on the upper 25% of consumption of chow. |
|
| Di Bonaventura et al., 2013 [82] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were exposed to a binge paradigm with or without stress or restriction. The central effects of nociception/orphanin FQ (N/OFQ) on binge eating were determined by intracerebroventricular (ICV) injection of N/OFQ (0.5-1 nmol/rat) or regionally mRNA by in situ hybridization of NOP receptor and ppN/OFQ. |
|
| Di Bonaventura et al., 2019 [88] | Species: Rat Binge Food: Sweetened hazelnut cocoa spread (52%), ground standard rat chow (33%), and 15% water. Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day Calorie Restriction: 66% of ad libitum standard chow Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats received a single dose of a A2A adenosine receptor antagonist ANR 94 (1 mg/kg), agonist VT 7 (0.1 mg/kg, IP), combination (pretreatment ANR 94 followed by VT 7 15 min later, IP), or vehicle before binge food access. Binge eating intake effects werestudied on A2A adenosine receptor gene expression and DNA methylation in the amygdaloid complex. |
|
| Di Bonaventura et al., 2012 [81] | Species: Rat Binge food: Paste mixture of Nutella (52%), food pellets (33%), and water (15%). Access: 2 h for days 5–6 of the 8-day cycle Frequency Three 8-day cycles: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day. Calorie Restriction: 4 days per week at 66% restriction of ad libitum chow. Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female rats were exposed to a restricting(R)/refeeding binge eating paradigm with or without stress (S). The effect of the A2A adenosine receptor (AR) agonists, CGS 21680 and VT 7 (0.1 and 0.05 mg/kg IP), on intake of highly palatable food (HPF) on sated rats, and low-palatability food (LPF) intake in food-deprived rats was assessed. |
|
| Cifani et al., 2010 [73] | Species: Rat Binge food: Paste mixture of Nutella (52%), food pellets (33%), and water (15%). Access: 2 h for days 5–6 of the 8-day cycle Frequency: 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days, day 25 test day. Calorie Restriction: 4 days per week at 66% restriction of ad libitum chow. Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were exposed to a model of binge eating by three 8-day cycles of food restriction/refeeding (R) and stress (S). The effect of Rhodiola rosea dry extract (3% rosavin, 3.12% salidroside) in this model of binge eating was assessed. Additionally, the bioactive constituents of rosavin and salidroside were assessed. |
|
3.5.1. Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP)
Two studies investigated site-specific injections of PACAP or PACAP1–38 [77,80]. Intra-accumbens PACAP (50 pmol/side) reduced binge eating, whereas ventromedial hypothalamic injections did not influence binge food intake in male rats [77]. Intra-VTA PACAP1–38 (30 pmol/side) reduced binge eating in male mice, but not in female or OVX female mice [80].
3.5.2. Orexin Receptor-Targeting Compounds
Two studies investigated peripheral injections of selective orexin receptor-1 (OX1) or orexin receptor-2 (OX2) or combined (OX1/OX2) antagonists [83,85]. A selective OX1 antagonist, GSK1059865, (10 or 30 mg/kg), and a dual OX1/OX2 receptor antagonist, SB-649868 (3 mg/kg), reduced binge eating in rats with stress exposure [83]. A selective OX2 antagonist, JNJ-10397049 (1 or 3 mg/kg), was not effective in reducing binge food intake [83]. In contrast, the selective OX1 antagonist, SB-334867 (5 mg/kg), only reduced intake for the first binge eating episode, but was not effective at preventing the escalation in binge food intake over time in mice [85]. In one study examining the centrally administered OX1 antagonist, bilateral microinfusions of SB-334867 (500 ng/0.3 μL per side) into the nucleus accumbens shell were ineffective at reducing binge eating in male rats with high or low impulsivity [87].
3.5.3. Sigma (σ) Receptors
Two studies examined novel sigma receptor antagonists in female rats with a history of calorie restriction and stress exposure [72,74]. Both the racemic spipethiane analogue (2-(1-Benzylpiperidin-4-yl)thiochroman-4-one) (±)-1 (3 or 7 mg/kg) and 1,3-dioxane derivative, 9-Benzyl-3-phenyl-1,5-dioxa-9-azaspiro[5.5]undecane (3 or 7 mg/kg) were effective in reducing binge eating behaviors [72,74].
3.5.4. Other Targeting Compounds
Site-specific injections into the PVN of the relaxin-family peptide-3 receptor (RXFP3) antagonist, R3(B1-22)R (1 µg/side), reduced binge eating in female rats [79]. Peripheral administration of oleoylethanolamide (OEA; 10 mg/kg) reduced binge eating in female rats with stress exposure [84]. Peripheral injections of a novel monoamine stabilizer OSU6162 (15 or 30 mg/kg) reduced binge eating in male rats [75].
A selective trace amine-associated receptor 1 (TAAR1) full-agonist RO5256390 (3 or 10 mg/kg) administered peripherally reduced binge food intake in male rats [76]. Peripheral (90 mg/kg) or central administration of N-acetylcysteine (10 µg ICV) reduced binge eating without producing a conditioned taste aversion to the binge food or sodium saccharin solution [78]. Central injections of the cocaine- and amphetamine-regulated transcript peptide (CART; 1.5 or 2 µg; ICV) reduced binge eating, whereas the central injections of the CART antibody (1:500 or 1:250) did not influence binge food intake [71]. In the same study, peripheral injections of the inverse agonist of the cannabinoid receptor 1 (CB1), rimonabant (3 or 10 mg/kg), reduced binge intake [71].
Peripheral administration of the tricyclic antidepressant clomipramine twice daily (15 mg/kg) prior to weaning resulted in adult binge eating phenotype in male and female rats [86]. Examination of nociception/orphanin FQ (N/OFQ) on binge eating indicated a differential dose-dependent effect [82]. Central injection of N/OFQ peptide (0.5 nmol; ICV) decreased binge intake at 30 min in rats with a history of calorie restriction and stress, but higher doses (1.0 nmol ICV) increased binge eating in rats with a history of calorie restriction without stress [82].
Peripheral administration of two of the A2A adenosine receptor agonists, CGS 21680 (0.1 mg/kg) and VT 7 (0.1 mg/kg), reduced binge food and non-binge food intake [81]. VT7 (0.1 mg/kg) also increased A2A receptor gene expression in the amygdaloid complex, whereas the A2A antagonist ANR 94 (1 mg/kg) increased %DNA methylation of the A2A gene promoter [88]. ANR94 combined with VT 7 had higher %DNA methylation than VT 7 alone [88]. Peripheral administration of extracts of the botanical Rhodiola rosea (10 or 20 mg/kg) reduced binge eating in female rats exposed to a history of calorie restriction and stress [73]. Additional experiments indicated salidroside (312–936 µg/kg), a bioactive component of R. rosea, reduced binge eating [73].
3.6. Combinational or Direct Comparisons of Multiple Compounds Acting on Different Systems
As described in Table 5, there were 16 studies that were combinational or were designed to be direct comparisons between compounds [89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104].
3.6.1. Fluoxetine (FLX) Compared with Other Compounds
Six studies [89,90,91,92,94,98] compared FLX with other compounds. In all studies, FLX was administered peripherally (IP) at dose ranges of 2.5 to 10 mg/kg in rats and 3 to 30 mg/kg in mice. FLX (30 mg/kg) reduced binge eating, whereas peripherally administered psilocybin (3 mg/kg) was ineffective on reducing binge eating in male mice [90]. In male and female mice, the peripherally administered nociceptin receptor antagonist, SB612111(10 mg/kg), reduced binge food intake in rats with the intermittent access schedule, whereas FLX (30 mg/kg) had a feeding reduction not specific to the binge paradigm [89]. FLX (10 mg/kg), lorcaserin (6 mg/kg), and fenfluramine (3 mg/kg) reduced binge eating in wild-type (WT) mice, but all serotonin-acting compounds were ineffective in mice lacking the serotonin (5HT) 2C receptor (5HT 2C null) [94]. In another study, FLX was compared with topiramate and baclofen in binge eating male mice [89]. In this study, two doses of FLX (10 or 30 mg/kg), while only one dose of baclofen (3 mg/kg), were effective at reducing binge eating [89]. In female rats with a history calorie restriction and stress, FLX (3 mg/kg) and topiramate (60 mg/kg) reduced binge food intake, whereas sibutramine (1 or 3 mg/kg) reduced binge food intake in all groups [98]. In contrast, the benzodiazepine, midazolam (5 or 10 mg/kg), increased binge food intake, except in those rats with a history of calorie restriction and stress exposure [89]. In a study in male rats with either a fat-rich or carbohydrate-rich binge food, FLX (2.5–10 mg/kg) preferentially suppressed carbohydrate-rich binge food, whereas as naloxone (1 mg/kg) suppressed fat-rich binge food [91]. Naloxone (1 mg/kg) increased FLX-induced suppression of both types of binge foods [91].
3.6.2. Lisdexamfetamine (LDX) Compared with Other Compounds
Three studies [93,95,97] compared LDX with other compounds. In all studies, LDX was administered orally at dose ranges of 0.15–1.5 mg/kg in mice and 0.1–100 mg/kg rats. Acute dosing with sibutramine (1 or 3 mg/kg) reduced binge eating; however, binge eating was not reduced at the 0.15–1.5 mg/kg dose range of LDX in female mice [93]. In female rats, oral dosing of a nootropic agent, piracetam (200 mg/kg), reduced binge eating to a similar extent as LDX (100 mg/kg) [95]. In a study comparing several compounds in female rats, nalmefene (0.1–1.0 mg/kg), SB-334867 (3–30 mg/kg), R-baclofen (1–10 mg/kg), sibutramine (1–5 mg/kg), rolipram (1–10 mg/kg), amphetamine (0.5–1.0 mg/kg), and LDX (0.3–1.5 mg/kg) reduced binge food intake. Sibutramine (0.3–5 mg/kg), rolipram (1–10 mg/kg), and baclofen (10 mg/kg) also reduced standard chow intake [97]. Prazosin (0.3 and 1.0 mg/kg) and SCH-23390 (0.1 mg/kg) attenuated LDX suppression of binge food intake [97].

Table 5.
Combinational or direct comparisons of multiple compounds acting on different systems.
Table 5.
Combinational or direct comparisons of multiple compounds acting on different systems.
| Fluoxetine Compared with Other Compounds | |||
| Study | Methods | Details | Major Findings |
| Fadahunsi et al., 2022 [90] | Species: Mice Binge Food: High-fat diet (58% fat) Access: 48 h (initial binge)/24 h (subsequent binges). Binge asessed after 2.5 h. Control group had binge food ad libitum. Frequency: Once a week Duration: 3 weeks Calorie Restriction: None | Male C57BL/6J mice were exposed to the binge eating paradigm To determine the influence of the serotinergic psycheldelic, psilocybin, mice received an IP injection of vehicle (saline), FLX (30 mg/kg), or psilocybin (3 mg/kg) 30 min prior to a scheduled binge. |
|
| Hardaway et al., 2016 [92] | Species: Mice Binge Food: High-fat diet (60% Fat) Access: 1 h Frequency: Daily Duration: >14 days Calorie Restriction: None | Male and female C57BL/6J mice were exposed to the binge eating paradigm, continuous access, or no access to the binge food. Binge food intake was assessed following nociceptin receptor antagonist (SB 612111, 0.1–10 mg/kg; IP) or FLX (30 mg/kg; IP) administration. |
|
| Xu et al., 2016 [94] | Species: Mice Binge Food: High-fat diet (40% kcal from fat) Access: 24 h; binge intake measured 2.5 h Frequency: Once per week Duration: >7 weeks Calorie Restriction: None | Transgenic mice were compared with wild-type (WT) mice. All were on C57 Bl/6 background. 5-Hydroxytryptamine (5HT) 2C Receptors (2C-null) mice, 2C null in dopamine (DA) neurons (DA-2C-KO), or 2C expressed only in DA neurons (DA-2C-RE). Mice were also exposed to 2C agonist, lorcaserin (6 mg/kg; IP), and other 5HT agents (FLX; 10 mg/kg; IP or d-fenfluramine; 3 mg/kg; IP). |
|
| Czyzyk et al., 2010 [89] | Species: Mice Binge Food: High-energy diet (Teklad 95217) Access: 48 h (initial binge)/24 h (subsequent binges). Binge asessed after 2.5 and 24 h. Control group had binge food ad libitum. Frequency: Continuous or once per week (Intermittent). Duration: ~8 weeks Calorie Restriction: None | Male C57BL/6 mice were split into 3 groups: Chow, Continuous, or Intermittent. Pharmacological evaluation of binge-like eating behavior was performed using baclofen (0.3, 1, and 3 mg/kg), topiramate (10, 30, and 100 mg/kg), and FLX (3, 10, and 30 mg/kg). |
|
| Cifani et al., 2009 [98] | Species: Rat Binge food: Paste mixture of Nutella (52%), food pellets (33%), and water (15%). Access: 2 h for days 5–6 and 13–14 of the 8-day cycles (second cycle). Frequency: Three 8-day cycle: 1–4 days of calorie restriction, 5–8 days ad libitum feeding. Duration: 24 days (test day 25). Calorie Restriction: 4 days per week at 66% restriction of ad libitum chow. Stress: 15 min exposure to sight and smell of binge food without the ability to consume it. (Frustration Stress) | Female Sprague Dawley rats were split into 4 groups: nonrestricted and not exposed to stress (NR + NS), restricted and not exposed to stress (R + NS), nonrestricted and exposed to stress (NR + S), and restricted and exposed to stress (R + S). Animals were exposed to the binge eating paradigm. The predictive validity of the restriction/refeeding/frustration stress model of binge eating was evaluated. The effects of FLX (3, 10 mg/kg), sibutramine (1, 3 mg/kg), topiramate (30, 60 mg/kg), and midazolam (5, 10 mg/kg) with vehicle were assessed. |
|
| Hagan et al., 1997 [91] | Species: Rat Binge food: Kellogg’s Froot Loops cereal (4.0 Kcal/g; 88% carbohydrate, 3% fat, 6% protein) given as palatable carbohydrate-rich food. Mars almond M&Ms (5.48 kCal/g; 57% carbohydrate, 30% fat, 8& protein) given as palatable fat-rich food. Access: 2 h Frequency: Daily (D) after injections for 2 h for each of the two experiments. Duration: 2 experiments in isolation 3 months apart; ~5 months total. Calorie Restriction: Animals were food-deprived 24 h before testing to induce feeding. | Male Sprague Dawley rats given ad libitum access to food and water. During eating tests, only the cereal and chocolate were available (no rat chow) along with water. Animals were randomly assigned to be pretreated with either SQ 1 mg/kg NAL or vehicle followed by an IP injection of one of 4 doses of FLX (0, 2.5, 5, and 10 mg/kg) with acess to binge food. Intakes were measured after 30 min and 2 h. At a 3 month time point, rats were pretreated with one of 4 doses of naloxone (NAL; 0, 0.01, 0.1, and 1 mg/kg); 10 min later, they were IP injected with 2.5 mg/kg FLX of vehicle and presented 20 min later with binge food access. |
|
| Lisdexamfetamine compared with other compounds | |||
| Study | Methods | Details | Major Findings |
| Sachdeo et al., 2019 [93] | Species: Mice Binge Food: Vegetable shortening blended with 10% sucrose Access: 30 min Frequency: 2 non- consecutive days per week Duration: 6 weeks Calorie Restriction: Two groups of mice had a 24 h calorie restriction prior to the binge period twice a week. | Female mice (C57Bl/6J background) with variant of OPRM1 gene were exposed to binge eating paradigm to determine the polymorphisms’ (A112G) influence on binge-like eating. The interactions of polymorphisms, binge eating, and lisdexamfetamine (LDX: 0.15–1.5 mg/kg: PO) and sibutramine (SIB; 0.3–3 mg/kg;PO) were also examined. |
|
| Hussain & Krishnamurthy, 2018 [95] | Species: Rat Binge Food: Oreo cookies Access: 2 h Frequency: Alternate days Duration: 28 days Calorie Restriction: None Stress: Rats were single housed at time of binge (Isolation stress) | Female Wister albino rats were exposed to binge or binge + stress conditions. A nootropic agent, piracetam (200 mg/kg, oral) was compared with lisdexamfetamine (LDX; 100 mg/kg, oral) for binge eating suppression in the binge + stress conditions. |
|
| Vickers et al., 2015 [97] | Species: Rat Binge Food: Ground chocolate (Cadbury’s Dairy Milk) Access: 2 h Frequency: Intermittent on non-consecutive days, 3 days a week. Duration: 28 days Calorie Restriction: None | Female Wistar rats were exposed to a binge eating paradigm. The dose-dependent effects of several compounds were examined; these included opioid antagonist nalmefene (0.1–1.0 mg/kg, SQ), orexin antagonist SB-334867 (3.0–30 mg/kg, IP), antipsychotic olanzapine (0.3–3.0 mg/kg, IP), GABAB agonist baclofen (1.0–10 mg/kg, IP), selective phosphodiesterase-4 inhibitor antidepressant rolipram (1.0–10 mg/kg, IP), norepinephrine and serotonin reuptake inhibitor sibutramine (0.3–5 mg/kg, PO), d-amphetamine (0.1–1.0 mg/kg, PO), and prodrug lisdexamfetamine (LDX; 0.1–1.5 mg/kg.,PO). To further investigate the effects of LDX, noradrenergic: prazosin (0.3 or 1.0 mg/kg, IP) or RX821002 (0.1 or 0.3 mg/kg, IP) or dopamine: raclopride (0.1 or 0.5 mg/kg, IP) or SCH-23390 (0.1 or 0.3 mg/kg, IP) in combination with LDX (1.0 mg/kg, PO) were considered. |
|
| Additional compound comparisons or combinations | |||
| Study | Methods | Details | Major Findings |
| Price et al., 2018 [96] | Species: Rat Binge Food: High-fat diet (45%) Access: 2 h (after a single 7-day ad libitum access period) Frequency: Once per week Duration: 5–9 weeks Calorie Restriction: None | Adult male Sprague Dawley rats were SQ administered lorcaserin (5HT-2cR agonist; 0.25, 0.5, or 1.0 mg/Kg) or pimavanserin (5HT-2aR antagonist/inverse agonist; 0.3, 1.0, or 3.0 mg/kg). Binge eating intake or binge occurrence (intake over 2 h intake during day 6 binge food ad libitium period) was assessed. |
|
| Corwin et al., 2016 [100] | Species: Rat Binge Food: Vegetable Shortening Access: 1 h Frequency: Intermittent on non-consecutive days or daily Duration: 5 to 8 weeks Calorie Restriction: None | Male Sprague Dawley rats were assessed for glutamate, GABA, dopamine, and opioid-related genes by qPCR 20 min before/after binge eating in PFC and VTA. Another set of rats were implanted with ventromedial (PL) or dorsomedial (M2) PFC cannula and injected with combined GABA R agonists (muscimol/baclofen), D1 R agonist (SKF 81297), D1 R antagonist (SCH 23390), D2 R agonist (quinpirole), and D2 R antagonist (eticlopride) before a scheduled binge episode. |
|
| Cao et al., 2014 [99] | Species: Mice Binge Food: High-fat diet (HFD; 40% fat) Access: 48 h initial exposure; 2.5 h Frequency: Once a week Duration: >2 weeks Calorie Restriction: None | Female C57BL/6 mice received bilateral ovariectomies (OVX) followed by 17β-estradiol (0.5 μg/d for 60 days; OVXE) or the containing vehicle (OVXV). Mice with tamoxifen-inducible CRE-recombinase on the tryptophan hydroxylase 2 (TPH2) promoter were used to visualize the serotonin-containing cells or to investigate the role of estrogen receptor alpha (ERα) on 5HT neurons. In addition, the roles of glucagon-like peptide (GLP-1) (4 ug/kg; SQ) as well as GLP-1 + E2 (4 ug/kg; SQ) were examined. |
|
| Popik et al., 2010 [102] | Species: Rat Binge food: Lard Access: 2 h Frequency: Daily Duration: 6 weeks Calorie Restriction: None | Male Sprague Dawley rats were exposed to a daily limited-access binge eating paradigm for 6 weeks. After 3 weeks, animals were given either sibutramine, memantine, or MTEP for 12 days, and their effects on binge eating behavior were assessed. In a separate experiment, rats were given ad lib access to standard chow and water for 2 weeks, followed by assignment of 1 of the 3 drugs for 7 consecutive days. Following treatment, animals were given vehicle and food consumption was measured for 5 days. |
|
| Pankevich et al., 2010 [101] | Species: Mice Binge Food: High-fat diet Access: Ad libitum access for 1 week (experiment 1). 1 h daily for stress experiment. Frequency: Ad libitum or daily (D) Duration: ~2 months Calorie Restriction: 75% of average caloric intake. | Male C57BL/6J mice were exposed to a model of moderate caloric restriction (CR; 10–15% weight loss), and physiological and behavioral stress measures were assessed. Changes in leptin, orexigenic hormones, melanin-concentrating hormone (MCH), and orexin levels due to stress and caloric restriction were also assessed, in addition to the effect of the MCH receptor-1 antagonist GSK-856464 (30 mg/kg; IP) and the SSRI citalopram (CIT; 20 mg/kg; IP) on binge feeding behavior. Tail suspension test on day 19 of restriction, restraint stress, and HPA axis stress response on day 21 were also performed. Chronic variable stress (CVS) was studied in a separate experiment (restraint stress). |
|
| Wong et al. 2009 [104] | Species: Rat Binge food: Fat/sucrose mixture (FSM) (3.2%, 10%, or 32% sucrose in vegetable shortening). Access: 1 h Frequency: Daily (D) or Intermittent (INT), 3 days/week. Duration: ~6–8 months Calorie Restriction: None | Male Sprague Daley rats were grouped according to two schedules of access, D or INT, to an optional FSM. The effects of the opioid antagonist naltrexone (0.03, 0.1, 0.3, 1 mg/kg; IP), the dopamine-2-like (D2) antagonist raclopride (0.03, 0.1, 0.3 mg/kg; IP), and the GABAA agonist baclofen (0.6, 1, 1.8, 3.2 mg/kg; IP) were assessed. |
|
| Will et al., 2004 [103] | Species: Rat Binge Food: High-fat diet Access: 2 h daily (D) Frequency: Daily during training, then every other day for treatment. Duration: 4–6 weeks Calorie Restriction: None | Adult male Sprague Dawley rats were implanted with bilateral guide cannulas into the nucleus accumbens core and one of the following four structures: Basolateral amygdala, central nucleus of the amygdala, posterior mediobasal amygdala, or the posterior lateral striatum. GABAA agonist, muscimol (20 ng/0.25 uL/side bilaterally), or saline were infused into the selected sites followed immediately by accumbens DAMGO (0.25 mg/0.5 uL/side bilaterally) or saline. |
|
3.6.3. Additional Compound Comparisons or Combinations
Seven studies [96,99,100,101,102,103,104] compared or examined the combinational effects with additional compound or agents. In female rats, two serotonergic compounds peripherally administered, lorcaserin (5HT 2c R agonist) or pimavanserin (5HT 2a antagonist/inverse agonist), were examined alone or combination in a binge paradigm to assess binge intake and binge episode occurrence (>binge food intake during intermittent vs. continuous access) [96]. Lorcaserin alone at 1.0 mg/kg and primavanserin alone at 1 or 3 mg/kg reduced binge intake, whereas the combination of lorcaserin (1 mg/kg) + primavanserin (0.3 mg/kg) reduced binge intake, binge occurrence, and weight gain [96]. Regional differences in the prefrontal cortex (PFC) of male rats to dopamine agonist/antagonists (SKF 81297, SCH 23390, quinpirole, or eticlopride) or GABA receptor-combined agonists (muscimol/baclofen) were assessed in male rats [100]. Intra-PFC (either pre-limbic or dorsomedial region) of the combined GABA agonists and dopamine (D2) receptor antagonist eticloprde (0.3 µg) into the intra-PFC dorsomedial region increased binge eating behavior [100]. In female OVX mice, combinations of GLP-4 and E2 reduced binge food intake; this effect was abolished in mice lacking estrogen receptor alpha (ER-α) on 5 HT neurons [99]. Peripheral administration of memantine (5 mg/kg) reduced binge food intake to a similar level as sibutramine (7.5 mg/kg) in male rats; however, memantine feeding suppression persisted post-treatment [102]. Peripheral administration of the MCH receptor-1 antagonist, GSK-856464 (30 mg/kg), suppressed binge intake compared with peripherally administered citalopram (20 mg/kg) in mice with a history of caloric restriction [101]. In male rats with intermittent access to a binge food of a fat mixture with different levels of sucrose sweetening (3.2–32%), peripheral administration of naltrexone (0.03–1 mg/kg), baclofen (0.6–3.2 mg/kg), and raclopride (0.03–0.3 mg/kg) were assessed [104]. All naltrexone doses tested reduced the fat/sugar mixtures (3.3–32%), whereas all baclofen doses reduced only 3.2% and 10% sugar/fat mixtures. Raclopride at 0.3 mg/kg reduced 3.2% and 10% sugar/fat mixtures; in contrast, at 0.1 mg/kg, raclopride increased 3.2% sugar/fat binge food intake [104]. In male rats, intra-accumbens DAMGO binge eating was blocked by microinjections of muscimol (20 ng/bilaterally) in either the basolateral or central nucleus of the amygdala [103].
4. Discussion
The purpose of this systematic review was to provide an overview of the existing literature on candidate pharmacotherapies or drug targets in rodent models of binge eating behaviors. Based on exclusionary/inclusionary criteria, we identified 67 peer-reviewed studies that used novel compounds or agents to influence binge eating behaviors in rats and mice. Not all studies were designed to test candidate pharmacotherapies. However, there were some similarities in the experimental design among the studies. For all studies, binge food was a highly palatable (e.g., sugar, fat, or combination) optional food item or diet. Each study employed an intermittent or limited access schedule of the binge food. In this sense, binge eating was related to the overconsumption of the highly palatable food, rather than overconsumption of standard (non-binge) diet. Binge eating behaviors in rodents are dependent on increasing the eating rate and amount of binge food consumed in a defined episode. As such, the episodic reduction in the amount of binge food is the major criterion used to determine the effectiveness of the potential pharmacotherapeutic agent. Using the amount of binge food consumed presents an inherent challenge when interpreting the findings from binge eating animal studies to potential therapeutic strategies for BED or BN, since clinical binge eating studies use a reduction in the number of binge days or binge episode frequency as one of the primary efficacy endpoints [22,105]. One way to address this challenge in rodent studies is to incorporate another dimension to binge eating measurements. For instance, binge occurrence (i.e., episodes of increase intake above baseline) or binge propensity (i.e., above the quartile of intake over episodes) are two measurements that could be incorporated into some binge eating paradigms to determine effectiveness of treatment [86,96,106]. Nonetheless, the measurements of binge amount, binge occurrence, and binge propensity do not fully encompass the interoceptive drive initiating binge eating bouts in persons with eating disorders.
Because FLX and LDX are FDA-approved medications for BN or BED [9,25], the incorporation of these medications in rodent binge eating studies assessing candidate pharmacotherapies should be given consideration. Of course, having a FLX or LDX treatment arm incorporated as a comparator is not feasible for all study designs, but would add relevance to findings. Indeed, 15 of the 67 studies included in this systematic review had used FLX or LDX. These studies represent a strong foundation for future studies of drug comparisons using rodent models.
5. Conclusions
Binge eating rodent models have provided valuable preclinical assessments for the screening and development of candidate pharmacotherapies. As with any animal model for a psychiatric condition, there are limitations in the data interpretation and extrapolation of the findings. Two ways to reduce the limitations and advance the development of future pharmacotherapies are to incorporate more binge eating dimension measurements, such as binge proneness and binge occurrence, and to use existing comparator treatments, such as LDX and FLX, in the study design.
Author Contributions
Conceptualization N.T.B.; methodology, N.T.B. and G.B.; writing—original draft preparation, G.B., J.D.C., S.E.F., L.H. and L.L.S.; writing—review and editing, N.T.B.; funding acquisition, N.T.B. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Office of the Assistant Secretary of Defense for Health Affairs through the Peer-Reviewed Medical Research Program under W81XWH-19-1-0016. Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Brambilla, F.; Draisci, A.; Peirone, A.; Brunetta, M. Combined cognitive-behavioral, psychopharmacological and nutritional therapy in eating disorders. 2. Anorexia nervosa--binge-eating/purging type. Neuropsychobiology 1995, 32, 64–67. [Google Scholar] [CrossRef]
- Murphy, R.; Straebler, S.; Cooper, Z.; Fairburn, C.G. Cognitive behavioral therapy for eating disorders. Psychiatr. Clin. North Am. 2010, 33, 611–627. [Google Scholar] [CrossRef]
- Wilson, G.T.; Fairburn, C.G. Cognitive treatments for eating disorders. J. Consult Clin. Psychol. 1993, 61, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Gorla, K.; Mathews, M. Pharmacological treatment of eating disorders. Psychiatry 2005, 2, 43–48. [Google Scholar]
- Sivyer, K.; Allen, E.; Cooper, Z.; Bailey-Straebler, S.; O’Connor, M.E.; Fairburn, C.G.; Murphy, R. Mediators of change in cognitive behavior therapy and interpersonal psychotherapy for eating disorders: A secondary analysis of a transdiagnostic randomized controlled trial. Int. J. Eat. Disord. 2020, 53, 1928–1940. [Google Scholar] [CrossRef]
- Troscianko, E.T.; Leon, M. Treating Eating: A Dynamical Systems Model of Eating Disorders. Front. Psychol. 2020, 11, 1801. [Google Scholar] [CrossRef]
- Mitchell, J.E.; Peterson, C.B.; Myers, T.; Wonderlich, S. Combining pharmacotherapy and psychotherapy in the treatment of patients with eating disorders. Psychiatr. Clin. N. Am. 2001, 24, 315–323. [Google Scholar] [CrossRef]
- Grilo, C.M.; Reas, D.L.; Mitchell, J.E. Combining Pharmacological and Psychological Treatments for Binge Eating Disorder: Current Status, Limitations, and Future Directions. Curr. Psychiatry Rep. 2016, 18, 55. [Google Scholar] [CrossRef] [PubMed]
- Bello, N.T.; Yeomans, B.L. Safety of pharmacotherapy options for bulimia nervosa and binge eating disorder. Expert Opin. Drug Saf. 2018, 17, 17–23. [Google Scholar] [CrossRef]
- Association, A.P. Diagnostic and Statistical Manual of Mental Disorders: DSM-V, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013; p. xxvii. 991p. [Google Scholar]
- Romano, S.J.; Halmi, K.A.; Sarkar, N.P.; Koke, S.C.; Lee, J.S. A placebo-controlled study of fluoxetine in continued treatment of bulimia nervosa after successful acute fluoxetine treatment. Am. J. Psychiatry 2002, 159, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Eli Lilly and Company PROZAC (fluoxetine hydrochloride) for oral use. Package Insert. 2017, pp. 1–25. Available online: https://pi.lilly.com/us/prozac.pdf (accessed on 31 December 2022).
- Fluoxetine Bulimia Nervosa Collaborative Study Group. Fluoxetine in the Treatment of Bulimia Nervosa. A Multicenter, Placebo-Controlled, Double-Blind Trial. Arch. Gen. Psychiatry 1992, 49, 139–147. [Google Scholar] [CrossRef]
- Goldstein, D.J.; Wilson, M.G.; Thompson, V.L.; Potvin, J.H.; Rampey, A.H., Jr. Long-term fluoxetine treatment of bulimia nervosa. Fluoxetine Bulimia Nervosa Research Group. Br. J. Psychiatry J. Ment. Sci. 1995, 166, 660–666. [Google Scholar] [CrossRef]
- Hagan, K.E.; Walsh, B.T. State of the Art: The Therapeutic Approaches to Bulimia Nervosa. Clin. Ther. 2021, 43, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Guerdjikova, A.I.; Mori, N.; Casuto, L.S.; McElroy, S.L. Novel pharmacologic treatment in acute binge eating disorder—Role of lisdexamfetamine. Neuropsychiatr. Dis. Treat 2016, 12, 833–841. [Google Scholar] [CrossRef] [PubMed]
- McElroy, S.L.; Hudson, J.; Ferreira-Cornwell, M.C.; Radewonuk, J.; Whitaker, T.; Gasior, M. Lisdexamfetamine Dimesylate for Adults with Moderate to Severe Binge Eating Disorder: Results of Two Pivotal Phase 3 Randomized Controlled Trials. Neuropsychopharmacology 2016, 41, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.I.; McElroy, S.L.; Ferreira-Cornwell, M.C.; Radewonuk, J.; Gasior, M. Efficacy of Lisdexamfetamine in Adults With Moderate to Severe Binge-Eating Disorder: A Randomized Clinical Trial. JAMA Psychiatry 2017, 74, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, K.R.; Aparicio, L.; Braund, T.A.; Yang, J.; Harvie, G.; Harris, A.; Hay, P.J.; Touyz, S.; Kohn, M.R. Impulsivity and Its Relationship With Lisdexamfetamine Dimesylate Treatment in Binge Eating Disorder. Front. Psychol. 2021, 12, 716010. [Google Scholar] [CrossRef]
- McElroy, S.L.; Guerdjikova, A.; Kotwal, R.; Welge, J.A.; Nelson, E.B.; Lake, K.A.; Keck, P.E., Jr.; Hudson, J.I. Atomoxetine in the treatment of binge-eating disorder: A randomized placebo-controlled trial. J. Clin. Psychiatry 2007, 68, 390–398. [Google Scholar] [CrossRef]
- Quilty, L.C.; Allen, T.A.; Davis, C.; Knyahnytska, Y.; Kaplan, A.S. A randomized comparison of long acting methylphenidate and cognitive behavioral therapy in the treatment of binge eating disorder. Psychiatry Res. 2019, 273, 467–474. [Google Scholar] [CrossRef]
- McElroy, S.L.; Hudson, J.I.; Grilo, C.M.; Guerdjikova, A.I.; Deng, L.; Koblan, K.S.; Goldman, R.; Navia, B.; Hopkins, S.; Loebel, A. Efficacy and Safety of Dasotraline in Adults With Binge-Eating Disorder: A Randomized, Placebo-Controlled, Flexible-Dose Clinical Trial. J. Clin. Psychiatry 2020, 81, 19m13068. [Google Scholar] [CrossRef]
- McElroy, S.L.; Shapira, N.A.; Arnold, L.M.; Keck, P.E.; Rosenthal, N.R.; Wu, S.C.; Capece, J.A.; Fazzio, L.; Hudson, J.I. Topiramate in the long-term treatment of binge-eating disorder associated with obesity. J. Clin. Psychiatry 2004, 65, 1463–1469. [Google Scholar] [CrossRef] [PubMed]
- Milano, W.; Petrella, C.; Sabatino, C.; Capasso, A. Treatment of bulimia nervosa with sertraline: A randomized controlled trial. Adv. Ther. 2004, 21, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Heal, D.J.; Gosden, J. What pharmacological interventions are effective in binge-eating disorder? Insights from a critical evaluation of the evidence from clinical trials. Int. J. Int. J. Obes. 2022, 46, 677–695. [Google Scholar] [CrossRef]
- Shapiro, J.R.; Berkman, N.D.; Brownley, K.A.; Sedway, J.A.; Lohr, K.N.; Bulik, C.M. Bulimia nervosa treatment: A systematic review of randomized controlled trials. Int. J. Eat. Disord. 2007, 40, 321–336. [Google Scholar] [CrossRef] [PubMed]
- Wilfley, D.E.; Crow, S.J.; Hudson, J.I.; Mitchell, J.E.; Berkowitz, R.I.; Blakesley, V.; Walsh, B.T.; Sibutramine Binge Eating Disorder Research, G. Efficacy of sibutramine for the treatment of binge eating disorder: A randomized multicenter placebo-controlled double-blind study. Am. J. Psychiatry 2008, 165, 51–58. [Google Scholar] [CrossRef]
- Shire US Inc. VYVANSE® (Lisdexamfetamine Dimesylate) Chewable Tablets, for Oral Use, CII. Initial U.S. Approval: 2007. 2015. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/021977s036s037lbledt.pdf (accessed on 31 December 2022).
- Coghill, D.R.; Caballero, B.; Sorooshian, S.; Civil, R. A systematic review of the safety of lisdexamfetamine dimesylate. CNS Drugs 2014, 28, 497–511. [Google Scholar] [CrossRef] [PubMed]
- Colman, E. Food and Drug Administration’s Obesity Drug Guidance Document: A short history. Circulation 2012, 125, 2156–2164. [Google Scholar] [CrossRef]
- Food and Drug Administration, Center for Drug Evaluation and Research.Application Number: 21-977/S037. Vyvanse for Moderate to Severe Binge Eating Disorder. (BED). Center for Drug Evaluation and Research. FDA 2015. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2015/021977Orig1s037.pdf (accessed on 1 December 2022).
- Collins, L.M. Optimization of Behavioral, Biobehavioral, and Biomedical Interventions The Multiphase Optimization Strategy (MOST); Springer: New York, NY, USA, 2018; pp. 67–113. [Google Scholar]
- Kenny, P.J.; Hoyer, D.; Koob, G.F. Animal Models of Addiction and Neuropsychiatric Disorders and Their Role in Drug Discovery: Honoring the Legacy of Athina Markou. Biol. Psychiatry 2018, 83, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Nestler, E.J.; Hyman, S.E. Animal models of neuropsychiatric disorders. Nat. Neurosci. 2010, 13, 1161–1169. [Google Scholar] [CrossRef]
- National Institute of Mental Health. Notice of NIMH’s Considerations Regarding the Use of Animal Neurobehavioral Approaches in Basic and Pre-Clinical Studies. 2019; NOT-MH-19-053. Available online: https://grants.nih.gov/grants/guide/notice-files/NOT-MH-19-053.html (accessed on 1 December 2022).
- Horn, C.C.; Kimball, B.A.; Wang, H.; Kaus, J.; Dienel, S.; Nagy, A.; Gathright, G.R.; Yates, B.J.; Andrews, P.L. Why can’t rodents vomit? A comparative behavioral, anatomical, and physiological study. PLoS ONE 2013, 8, e60537. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Placidi, R.J.; Chandler, P.C.; Oswald, K.D.; Maldonado, C.; Wauford, P.K.; Boggiano, M.M. Stress and hunger alter the anorectic efficacy of fluoxetine in binge-eating rats with a history of caloric restriction. Int. J. Eat. Disord. 2004, 36, 328–341. [Google Scholar] [CrossRef] [PubMed]
- Buda-Levin, A.; Wojnicki, F.H.; Corwin, R.L. Baclofen reduces fat intake under binge-type conditions. Physiol. Behav. 2005, 86, 176–184. [Google Scholar] [CrossRef]
- Wojnicki, F.H.; Roberts, D.C.; Corwin, R.L. Effects of baclofen on operant performance for food pellets and vegetable shortening after a history of binge-type behavior in non-food deprived rats. Pharm. Behav. Biochem. Behav. 2006, 84, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Chandler-Laney, P.C.; Castaneda, E.; Viana, J.B.; Oswald, K.D.; Maldonado, C.R.; Boggiano, M.M. A history of human-like dieting alters serotonergic control of feeding and neurochemical balance in a rat model of binge-eating. Int. J. Eat. Disord. 2007, 40, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Halpern, C.H.; Tekriwal, A.; Santollo, J.; Keating, J.G.; Wolf, J.A.; Daniels, D.; Bale, T.L. Amelioration of binge eating by nucleus accumbens shell deep brain stimulation in mice involves D2 receptor modulation. J. Neurosci. 2013, 33, 7122–7129. [Google Scholar] [CrossRef]
- Bello, N.T.; Walters, A.L.; Verpeut, J.L.; Caverly, J. Dietary-induced binge eating increases prefrontal cortex neural activation to restraint stress and increases binge food consumption following chronic guanfacine. Pharm. Behav. Biochem. Behav. 2014, 125, 21–28. [Google Scholar] [CrossRef]
- Bello, N.T.; Yeh, C.Y.; Verpeut, J.L.; Walters, A.L. Binge-like eating attenuates nisoxetine feeding suppression, stress activation, and brain norepinephrine activity. PLoS ONE 2014, 9, e93610. [Google Scholar] [CrossRef]
- Smith, K.L.; Rao, R.R.; Velazquez-Sanchez, C.; Valenza, M.; Giuliano, C.; Everitt, B.J.; Sabino, V.; Cottone, P. The uncompetitive N-methyl-D-aspartate antagonist memantine reduces binge-like eating, food-seeking behavior, and compulsive eating: Role of the nucleus accumbens shell. Neuropsychopharmacology 2015, 40, 1163–1171. [Google Scholar] [CrossRef]
- Heal, D.J.; Goddard, S.; Brammer, R.J.; Hutson, P.H.; Vickers, S.P. Lisdexamfetamine reduces the compulsive and perseverative behaviour of binge-eating rats in a novel food reward/punished responding conflict model. J. Psychopharmacol. 2016, 30, 662–675. [Google Scholar] [CrossRef]
- Vickers, S.P.; Goddard, S.; Brammer, R.J.; Hutson, P.H.; Heal, D.J. Investigation of impulsivity in binge-eating rats in a delay-discounting task and its prevention by the d-amphetamine prodrug, lisdexamfetamine. J. Psychopharmacol. 2017, 31, 784–797. [Google Scholar] [CrossRef]
- Blumenthal, S.A.; Pratt, W.E. d-Fenfluramine and lorcaserin inhibit the binge-like feeding induced by mu-opioid receptor stimulation of the nucleus accumbens in the rat. Neurosci. Lett. 2018, 687, 43–48. [Google Scholar] [CrossRef]
- Price, A.E.; Anastasio, N.C.; Stutz, S.J.; Hommel, J.D.; Cunningham, K.A. Serotonin 5-HT2C Receptor Activation Suppresses Binge Intake and the Reinforcing and Motivational Properties of High-Fat Food. Front. Pharmacol. 2018, 9, 821. [Google Scholar] [CrossRef]
- Hicks, C.; Sabino, V.; Cottone, P. The Alpha-1 Adrenergic Receptor Antagonist Prazosin Reduces Binge-Like Eating in Rats. Nutrients 2020, 12, 1569. [Google Scholar] [CrossRef] [PubMed]
- Presby, R.E.; Rotolo, R.A.; Yang, J.H.; Correa, M.; Salamone, J.D. Lisdexamfetamine suppresses instrumental and consummatory behaviors supported by foods with varying degrees of palatability: Exploration of a binge-like eating model. Pharm. Behav. Biochem. Behav. 2020, 189, 172851. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, T.P.D.; Goncalves, B.D.C.; Oliveira, B.S.; de Oliveira, A.C.P.; Reis, H.J.; Ferreira, C.N.; Aguiar, D.C.; de Miranda, A.S.; Ribeiro, F.M.; Vieira, E.M.L.; et al. Negative Modulation of the Metabotropic Glutamate Receptor Type 5 as a Potential Therapeutic Strategy in Obesity and Binge-Like Eating Behavior. Front. Neurosci. 2021, 15, 631311. [Google Scholar] [CrossRef] [PubMed]
- Karth, M.D.; Baugher, B.J.; Pellechia, S.A.; Huq, S.N.; Warner, A.K.; Karth, M.M.; Sachs, B.D. Brain serotonin deficiency and fluoxetine lead to sex-specific effects on binge-like food consumption in mice. Psychopharmacology 2022, 239, 2975–2984. [Google Scholar] [CrossRef]
- Blasio, A.; Steardo, L.; Sabino, V.; Cottone, P. Opioid system in the medial prefrontal cortex mediates binge-like eating. Addict Biol. 2014, 19, 652–662. [Google Scholar] [CrossRef]
- Boggiano, M.M.; Chandler, P.C.; Viana, J.B.; Oswald, K.D.; Maldonado, C.R.; Wauford, P.K. Combined dieting and stress evoke exaggerated responses to opioids in binge-eating rats. Behav. Neurosci. 2005, 119, 1207–1214. [Google Scholar] [CrossRef]
- Giuliano, C.; Robbins, T.W.; Nathan, P.J.; Bullmore, E.T.; Everitt, B.J. Inhibition of opioid transmission at the mu-opioid receptor prevents both food seeking and binge-like eating. Neuropsychopharmacology 2012, 37, 2643–2652. [Google Scholar] [CrossRef]
- Hagan, M.M.; Moss, D.E. An animal model of bulimia nervosa: Opioid sensitivity to fasting episodes. Pharm. Behav. Biochem. Behav. 1991, 39, 421–422. [Google Scholar] [CrossRef] [PubMed]
- Naleid, A.M.; Grace, M.K.; Chimukangara, M.; Billington, C.J.; Levine, A.S. Paraventricular opioids alter intake of high-fat but not high-sucrose diet depending on diet preference in a binge model of feeding. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R99–R105. [Google Scholar] [CrossRef]
- Anversa, R.G.; Campbell, E.J.; Ch’ng, S.S.; Gogos, A.; Lawrence, A.J.; Brown, R.M. A model of emotional stress-induced binge eating in female mice with no history of food restriction. Genes Brain Behav. 2020, 19, e12613. [Google Scholar] [CrossRef]
- Babbs, R.K.; Unger, E.L.; Corwin, R.L. 2-Hydroxyestradiol enhances binge onset in female rats and reduces prefrontal cortical dopamine in male rats. Behav. Horm. Behav. 2013, 63, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Babbs, R.K.; Wojnicki, F.H.; Corwin, R.L. Effect of 2-hydroxyestradiol on binge intake in rats. Physiol. Behav. 2011, 103, 508–512. [Google Scholar] [CrossRef]
- Brutman, J.N.; Sirohi, S.; Davis, J.F. Examining the Impact of Estrogen on Binge Feeding, Food-Motivated Behavior, and Body Weight in Female Rats. Obesity 2019, 27, 1617–1626. [Google Scholar] [CrossRef]
- Di Bonaventura, M.V.M.; Lutz, T.A.; Romano, A.; Pucci, M.; Geary, N.; Asarian, L.; Cifani, C. Estrogenic suppression of binge-like eating elicited by cyclic food restriction and frustrative-nonreward stress in female rats. Int. J. Eat. Disord. 2017, 50, 624–635. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Geary, N.; Corwin, R.L. Ovarian hormones inhibit fat intake under binge-type conditions in ovariectomized rats. Physiol. Behav. 2008, 95, 501–507. [Google Scholar] [CrossRef]
- Yu, Z.; Geary, N.; Corwin, R.L. Individual effects of estradiol and progesterone on food intake and body weight in ovariectomized binge rats. Physiol. Behav. 2011, 104, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Bake, T.; Hellgren, K.T.; Dickson, S.L. Acute ghrelin changes food preference from a high-fat diet to chow during binge-like eating in rodents. J. Neuroendocrinol. 2017, 29, 12463. [Google Scholar] [CrossRef]
- Cornejo, M.P.; Castrogiovanni, D.; Schioth, H.B.; Reynaldo, M.; Marie, J.; Fehrentz, J.A.; Perello, M. Growth hormone secretagogue receptor signalling affects high-fat intake independently of plasma levels of ghrelin and LEAP2, in a 4-day binge eating model. J. Neuroendocrinol. 2019, 31, e12785. [Google Scholar] [CrossRef] [PubMed]
- Micioni Di Bonaventura, M.V.; Ciccocioppo, R.; Romano, A.; Bossert, J.M.; Rice, K.C.; Ubaldi, M.; St Laurent, R.; Gaetani, S.; Massi, M.; Shaham, Y.; et al. Role of bed nucleus of the stria terminalis corticotrophin-releasing factor receptors in frustration stress-induced binge-like palatable food consumption in female rats with a history of food restriction. J. Neurosci. 2014, 34, 11316–11324. [Google Scholar] [CrossRef] [PubMed]
- Micioni Di Bonaventura, M.V.; Ubaldi, M.; Giusepponi, M.E.; Rice, K.C.; Massi, M.; Ciccocioppo, R.; Cifani, C. Hypothalamic CRF1 receptor mechanisms are not sufficient to account for binge-like palatable food consumption in female rats. Int. J. Eat. Disord. 2017, 50, 1194–1204. [Google Scholar] [CrossRef]
- Pierce-Messick, Z.; Pratt, W.E. Glucagon-like peptide-1 receptors modulate the binge-like feeding induced by micro-opioid receptor stimulation of the nucleus accumbens in the rat. Neuroreport 2020, 31, 1283–1288. [Google Scholar] [CrossRef]
- Bharne, A.P.; Borkar, C.D.; Subhedar, N.K.; Kokare, D.M. Differential expression of CART in feeding and reward circuits in binge eating rat model. Behav. Brain Res. 2015, 291, 219–231. [Google Scholar] [CrossRef]
- Cifani, C.; Micioni Di Bonaventura, E.; Botticelli, L.; Del Bello, F.; Giorgioni, G.; Pavletic, P.; Piergentili, A.; Quaglia, W.; Bonifazi, A.; Schepmann, D.; et al. Novel Highly Potent and Selective Sigma1 Receptor Antagonists Effectively Block the Binge Eating Episode in Female Rats. ACS Chem. Neurosci. 2020, 11, 3107–3116. [Google Scholar] [CrossRef]
- Cifani, C.; Micioni Di Bonaventura, M.V.; Vitale, G.; Ruggieri, V.; Ciccocioppo, R.; Massi, M. Effect of salidroside, active principle of Rhodiola rosea extract, on binge eating. Physiol. Behav. 2010, 101, 555–562. [Google Scholar] [CrossRef]
- Del Bello, F.; Micioni Di Bonaventura, M.V.; Bonifazi, A.; Wunsch, B.; Schepmann, D.; Giancola, J.B.; Micioni Di Bonaventura, E.; Vistoli, G.; Giorgioni, G.; Quaglia, W.; et al. Investigation of the Role of Chirality in the Interaction with sigma Receptors and Effect on Binge Eating Episode of a Potent sigma1 Antagonist Analogue of Spipethiane. ACS Chem. Neurosci. 2019, 10, 3391–3397. [Google Scholar] [CrossRef] [PubMed]
- Feltmann, K.; Giuliano, C.; Everitt, B.J.; Steensland, P.; Alsio, J. The Effects of the Monoamine Stabilizer (-)-OSU6162 on Binge-Like Eating and Cue-Controlled Food-Seeking Behavior in Rats. Neuropsychopharmacology 2018, 43, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Ferragud, A.; Howell, A.D.; Moore, C.F.; Ta, T.L.; Hoener, M.C.; Sabino, V.; Cottone, P. The Trace Amine-Associated Receptor 1 Agonist RO5256390 Blocks Compulsive, Binge-like Eating in Rats. Neuropsychopharmacology 2017, 42, 1458–1470. [Google Scholar] [CrossRef] [PubMed]
- Hurley, M.M.; Maunze, B.; Block, M.E.; Frenkel, M.M.; Reilly, M.J.; Kim, E.; Chen, Y.; Li, Y.; Baker, D.A.; Liu, Q.S.; et al. Pituitary Adenylate-Cyclase Activating Polypeptide Regulates Hunger- and Palatability-Induced Binge Eating. Front. Neurosci. 2016, 10, 383. [Google Scholar] [CrossRef] [PubMed]
- Hurley, M.M.; Resch, J.M.; Maunze, B.; Frenkel, M.M.; Baker, D.A.; Choi, S. N-acetylcysteine decreases binge eating in a rodent model. Int. J. Obes. 2016, 40, 1183–1186. [Google Scholar] [CrossRef] [PubMed]
- Kania, A.; Szlaga, A.; Sambak, P.; Gugula, A.; Blasiak, E.; Micioni Di Bonaventura, M.V.; Hossain, M.A.; Cifani, C.; Hess, G.; Gundlach, A.L.; et al. RLN3/RXFP3 Signaling in the PVN Inhibits Magnocellular Neurons via M-like Current Activation and Contributes to Binge Eating Behavior. J. Neurosci. 2020, 40, 5362–5375. [Google Scholar] [CrossRef]
- Le, N.; Hernandez, J.; Gastelum, C.; Perez, L.; Vahrson, I.; Sayers, S.; Wagner, E.J. Pituitary Adenylate Cyclase Activating Polypeptide Inhibits A10 Dopamine Neurons and Suppresses the Binge-like Consumption of Palatable Food. Neuroscience 2021, 478, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Micioni Di Bonaventura, M.V.; Cifani, C.; Lambertucci, C.; Volpini, R.; Cristalli, G.; Massi, M. A2A adenosine receptor agonists reduce both high-palatability and low-palatability food intake in female rats. Behav. Pharmacol. 2012, 23, 567–574. [Google Scholar] [CrossRef]
- Micioni Di Bonaventura, M.V.; Ubaldi, M.; Liberati, S.; Ciccocioppo, R.; Massi, M.; Cifani, C. Caloric restriction increases the sensitivity to the hyperphagic effect of nociceptin/orphanin FQ limiting its ability to reduce binge eating in female rats. Psychopharmacology 2013, 228, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, L.; Micioni Di Bonaventura, M.V.; Cifani, C.; Costantini, V.J.; Massagrande, M.; Montanari, D.; Martinelli, P.; Antolini, M.; Ciccocioppo, R.; Massi, M.; et al. Role of orexin-1 receptor mechanisms on compulsive food consumption in a model of binge eating in female rats. Neuropsychopharmacology 2012, 37, 1999–2011. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Micioni Di Bonaventura, M.V.; Gallelli, C.A.; Koczwara, J.B.; Smeets, D.; Giusepponi, M.E.; De Ceglia, M.; Friuli, M.; Micioni Di Bonaventura, E.; Scuderi, C.; et al. Oleoylethanolamide decreases frustration stress-induced binge-like eating in female rats: A novel potential treatment for binge eating disorder. Neuropsychopharmacology 2020, 45, 1931–1941. [Google Scholar] [CrossRef]
- Valdivia, S.; Cornejo, M.P.; Reynaldo, M.; De Francesco, P.N.; Perello, M. Escalation in high fat intake in a binge eating model differentially engages dopamine neurons of the ventral tegmental area and requires ghrelin signaling. Psychoneuroendocrinology 2015, 60, 206–216. [Google Scholar] [CrossRef]
- Freund, N.; Thompson, B.S.; Norman, K.J.; Einhorn, P.; Andersen, S.L. Developmental emergence of an obsessive-compulsive phenotype and binge behavior in rats. Psychopharmacology 2015, 232, 3173–3181. [Google Scholar] [CrossRef]
- Schuller, J.; Koch, M. Investigating a role of orexin and ‘cocaine- and amphetamine-regulated transcript’ in the nucleus accumbens shell in binge eating of male rats and the relationship with impulsivity. Physiol. Behav. 2022, 257, 114000. [Google Scholar] [CrossRef]
- Micioni Di Bonaventura, M.V.; Pucci, M.; Giusepponi, M.E.; Romano, A.; Lambertucci, C.; Volpini, R.; Micioni Di Bonaventura, E.; Gaetani, S.; Maccarrone, M.; D’Addario, C.; et al. Regulation of adenosine A(2A) receptor gene expression in a model of binge eating in the amygdaloid complex of female rats. J. Psychopharmacol. 2019, 33, 1550–1561. [Google Scholar] [CrossRef]
- Czyzyk, T.A.; Sahr, A.E.; Statnick, M.A. A model of binge-like eating behavior in mice that does not require food deprivation or stress. Obes. (Silver Spring) 2010, 18, 1710–1717. [Google Scholar] [CrossRef]
- Fadahunsi, N.; Lund, J.; Breum, A.W.; Mathiesen, C.V.; Larsen, I.B.; Knudsen, G.M.; Klein, A.B.; Clemmensen, C. Acute and long-term effects of psilocybin on energy balance and feeding behavior in mice. Transl. Psychiatry 2022, 12, 330. [Google Scholar] [CrossRef]
- Hagan, M.M.; Holguin, F.D.; Cabello, C.E.; Hanscom, D.R.; Moss, D.E. Combined naloxone and fluoxetine on deprivation-induced binge eating of palatable foods in rats. Pharm. Behav. Biochem. Behav. 1997, 58, 1103–1107. [Google Scholar] [CrossRef]
- Hardaway, J.A.; Jensen, J.; Kim, M.; Mazzone, C.M.; Sugam, J.A.; Diberto, J.F.; Lowery-Gionta, E.G.; Hwa, L.S.; Pleil, K.E.; Bulik, C.M.; et al. Nociceptin receptor antagonist SB 612111 decreases high fat diet binge eating. Behav. Brain Res. 2016, 307, 25–34. [Google Scholar] [CrossRef]
- Sachdeo, B.L.Y.; Yu, L.; Giunta, G.M.; Bello, N.T. Binge-Like Eating Is Not Influenced by the Murine Model of OPRM1 A118G Polymorphism. Front. Psychol. 2019, 10, 246. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; He, Y.; Cao, X.; Valencia-Torres, L.; Yan, X.; Saito, K.; Wang, C.; Yang, Y.; Hinton, A., Jr.; Zhu, L.; et al. Activation of Serotonin 2C Receptors in Dopamine Neurons Inhibits Binge-like Eating in Mice. Biol. Psychiatry 2017, 81, 737–747. [Google Scholar] [CrossRef]
- Hussain, Y.; Krishnamurthy, S. Piracetam attenuates binge eating disorder related symptoms in rats. Pharm. Behav. Biochem. Behav. 2018, 169, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Price, A.E.; Brehm, V.D.; Hommel, J.D.; Anastasio, N.C.; Cunningham, K.A. Pimavanserin and Lorcaserin Attenuate Measures of Binge Eating in Male Sprague-Dawley Rats. Front. Pharm. 2018, 9, 1424. [Google Scholar] [CrossRef] [PubMed]
- Vickers, S.P.; Hackett, D.; Murray, F.; Hutson, P.H.; Heal, D.J. Effects of lisdexamfetamine in a rat model of binge-eating. J. Psychopharmacol. 2015, 29, 1290–1307. [Google Scholar] [CrossRef]
- Cifani, C.; Polidori, C.; Melotto, S.; Ciccocioppo, R.; Massi, M. A preclinical model of binge eating elicited by yo-yo dieting and stressful exposure to food: Effect of sibutramine, fluoxetine, topiramate, and midazolam. Psychopharmacology 2009, 204, 113–125. [Google Scholar] [CrossRef]
- Cao, X.; Xu, P.; Oyola, M.G.; Xia, Y.; Yan, X.; Saito, K.; Zou, F.; Wang, C.; Yang, Y.; Hinton, A., Jr.; et al. Estrogens stimulate serotonin neurons to inhibit binge-like eating in mice. J. Clin. Investig. 2014, 124, 4351–4362. [Google Scholar] [CrossRef]
- Corwin, R.L.; Wojnicki, F.H.; Zimmer, D.J.; Babbs, R.K.; McGrath, L.E.; Olivos, D.R.; Mietlicki-Baase, E.G.; Hayes, M.R. Binge-type eating disrupts dopaminergic and GABAergic signaling in the prefrontal cortex and ventral tegmental area. Obesity 2016, 24, 2118–2125. [Google Scholar] [CrossRef]
- Pankevich, D.E.; Teegarden, S.L.; Hedin, A.D.; Jensen, C.L.; Bale, T.L. Caloric restriction experience reprograms stress and orexigenic pathways and promotes binge eating. J. Neurosci. 2010, 30, 16399–16407. [Google Scholar] [CrossRef] [PubMed]
- Popik, P.; Kos, T.; Zhang, Y.; Bisaga, A. Memantine reduces consumption of highly palatable food in a rat model of binge eating. Amino. Acids 2011, 40, 477–485. [Google Scholar] [CrossRef]
- Will, M.J.; Franzblau, E.B.; Kelley, A.E. The amygdala is critical for opioid-mediated binge eating of fat. Neuroreport 2004, 15, 1857–1860. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.J.; Wojnicki, F.H.; Corwin, R.L. Baclofen, raclopride, and naltrexone differentially affect intake of fat/sucrose mixtures under limited access conditions. Pharm. Biochem. Behav. 2009, 92, 528–536. [Google Scholar] [CrossRef] [PubMed]
- McElroy, S.L.; Hudson, J.I.; Mitchell, J.E.; Wilfley, D.; Ferreira-Cornwell, M.C.; Gao, J.; Wang, J.; Whitaker, T.; Jonas, J.; Gasior, M. Efficacy and safety of lisdexamfetamine for treatment of adults with moderate to severe binge-eating disorder: A randomized clinical trial. JAMA Psychiatry 2015, 72, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Boggiano, M.M.; Artiga, A.I.; Pritchett, C.E.; Chandler-Laney, P.C.; Smith, M.L.; Eldridge, A.J. High intake of palatable food predicts binge-eating independent of susceptibility to obesity: An animal model of lean vs obese binge-eating and obesity with and without binge-eating. Int. J. Obes. 2007, 31, 1357–1367. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).