Benzimidazole Containing Acetamide Derivatives Attenuate Neuroinflammation and Oxidative Stress in Ethanol-Induced Neurodegeneration
Abstract
1. Introduction
2. Material and Methods
2.1. Chemicals
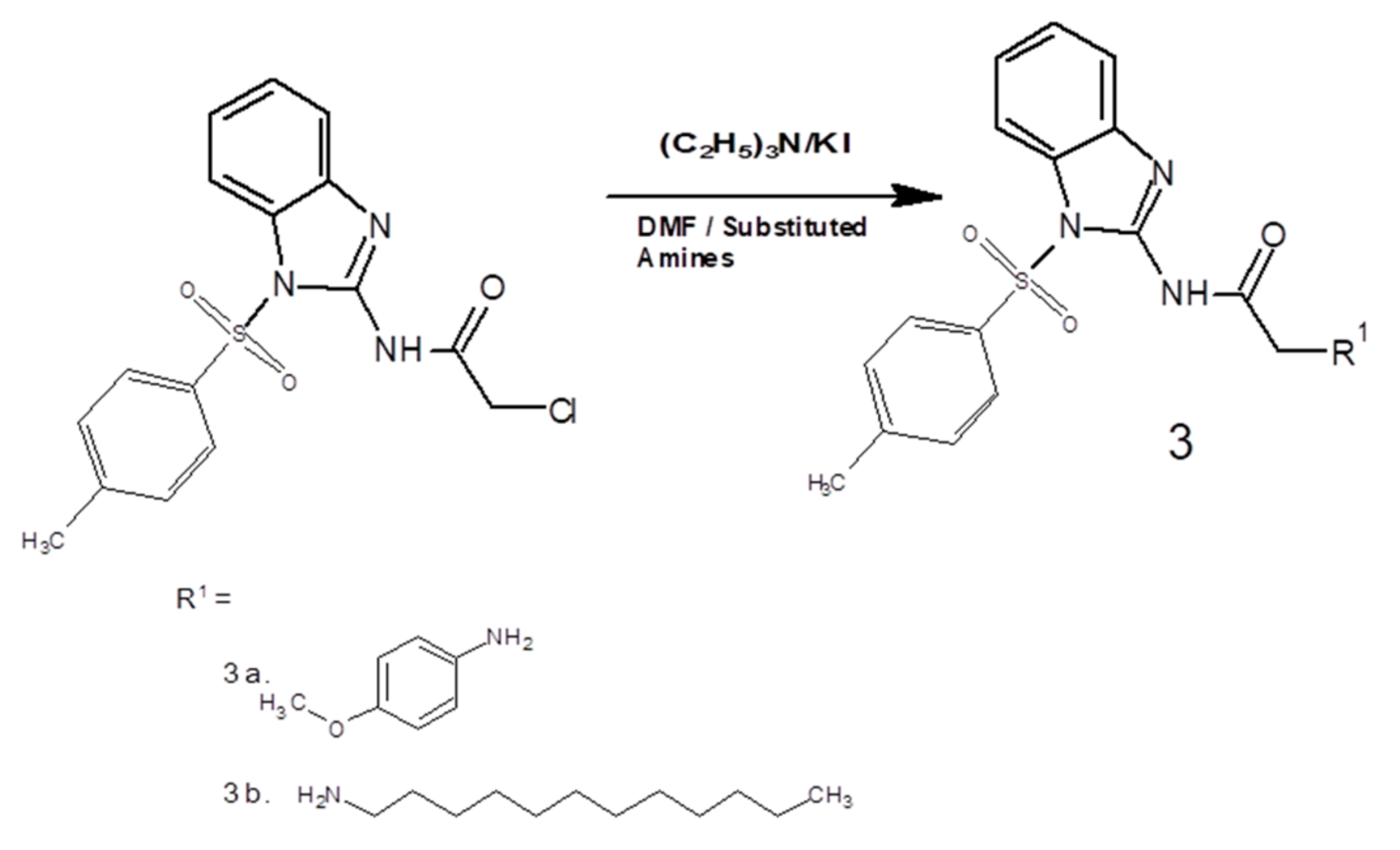
2.2. General Procedure for the Synthesis of Benzimidazole Acetamide Derivatives (3a and 3b)
2.3. Molecular Docking
2.4. Animals and Drug Treatment
2.5. Behavioral Tests
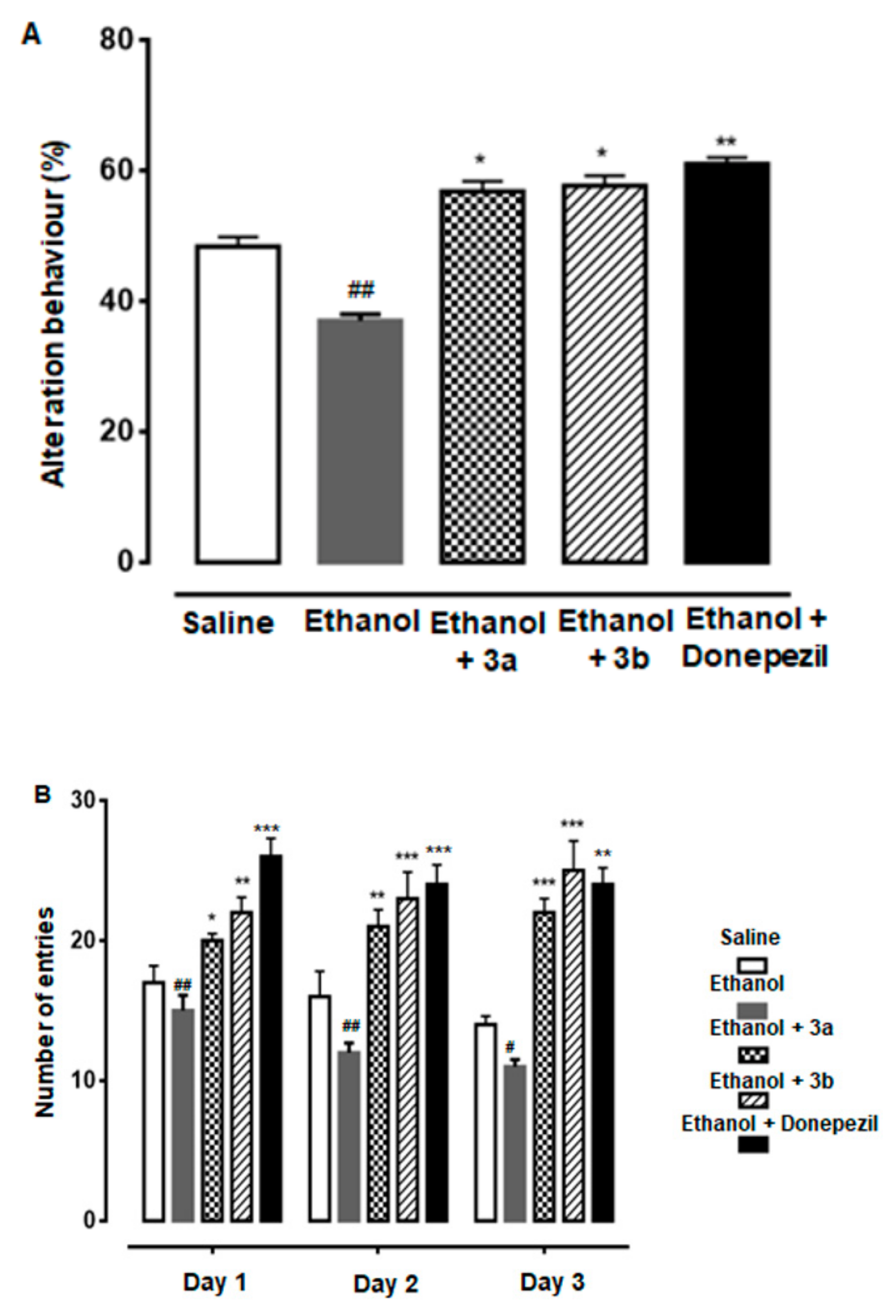
2.5.1. Y-Maze Test
2.5.2. Morris Water Maze (MWM) Test
2.6. Enzyme-Linked Immunosorbent Assays (ELISAs)
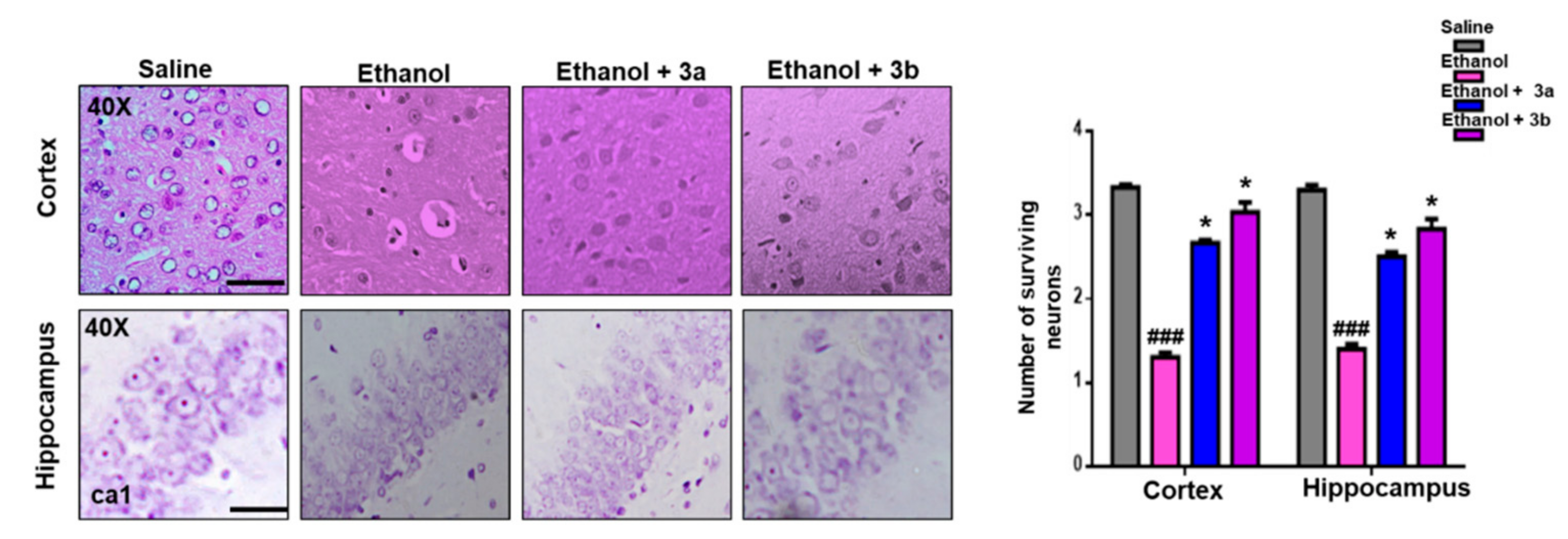
2.7. Morphological Analysis
2.7.1. Hematoxylin and Eosin (H&E) Staining
2.7.2. Immunohistochemical Evaluation
2.8. Oxidative Enzymes Exploration
2.8.1. GSH and GSH S-Transferase (GST) Assay
2.8.2. Lipid Peroxidation Assay
2.8.3. Nitric Oxide Assay
2.9. Statistical Analysis
3. Results
3.1. Spectral Analysis of (3a) [2-(4-methoxyanilino)-N-[1-(4-methylbenzene-1-sulfonyl)-1H-benzimidazol-2-yl] acetamide]
3.2. Spectral Analysis of (3b) [2-(Dodecylamino)-N-[1-(4-methylbenzene-1-sulfonyl)-1H-benzimidazol-2-yl] acetamide]
3.3. Docking Evaluation
3.4. Effect of Compound 3a and 3b on Alternation Behavior
3.5. Effect of Compound 3a and 3b on Escape Latency
3.6. Effect on Ethanol-Induced Neurodegeneration
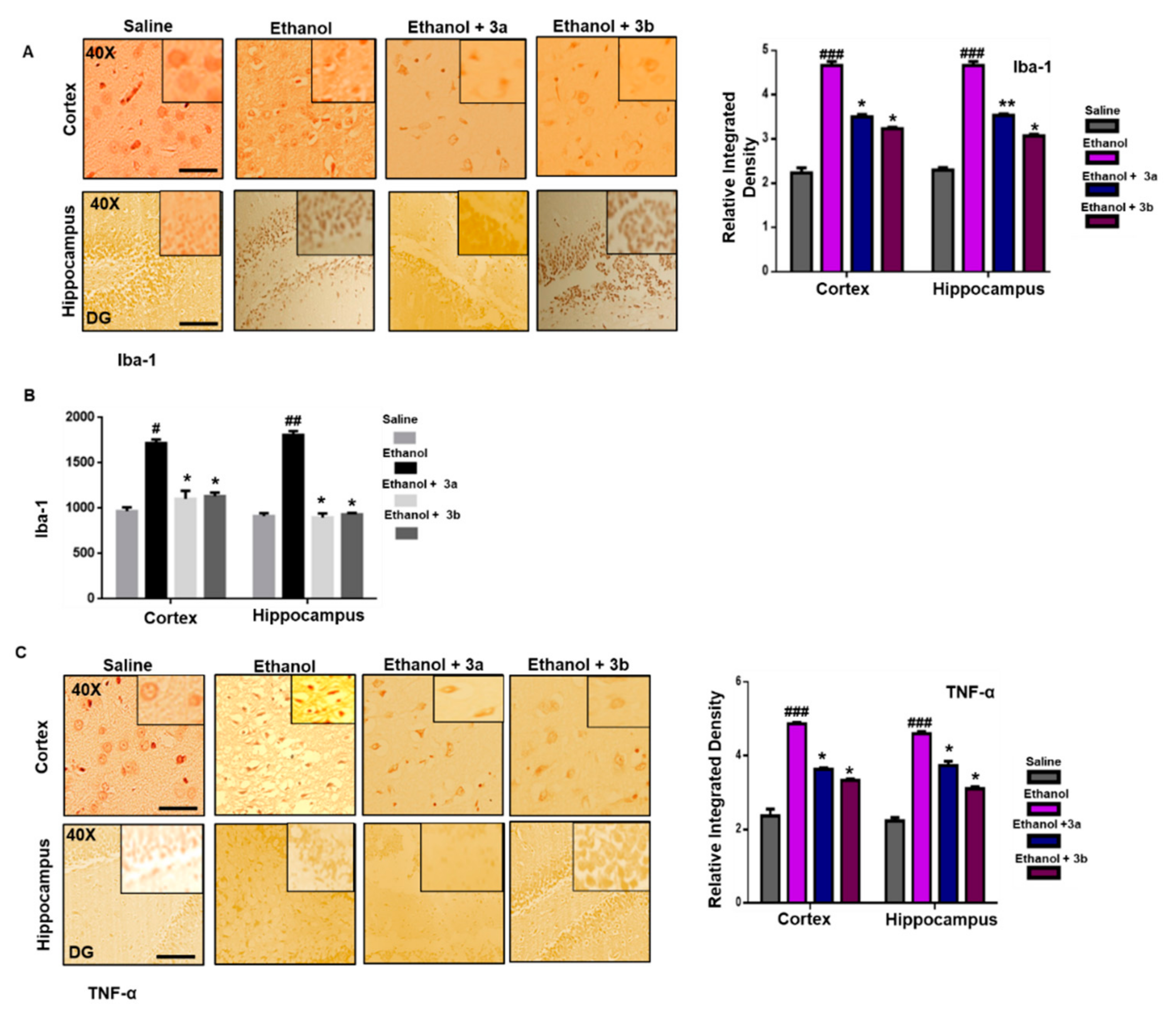
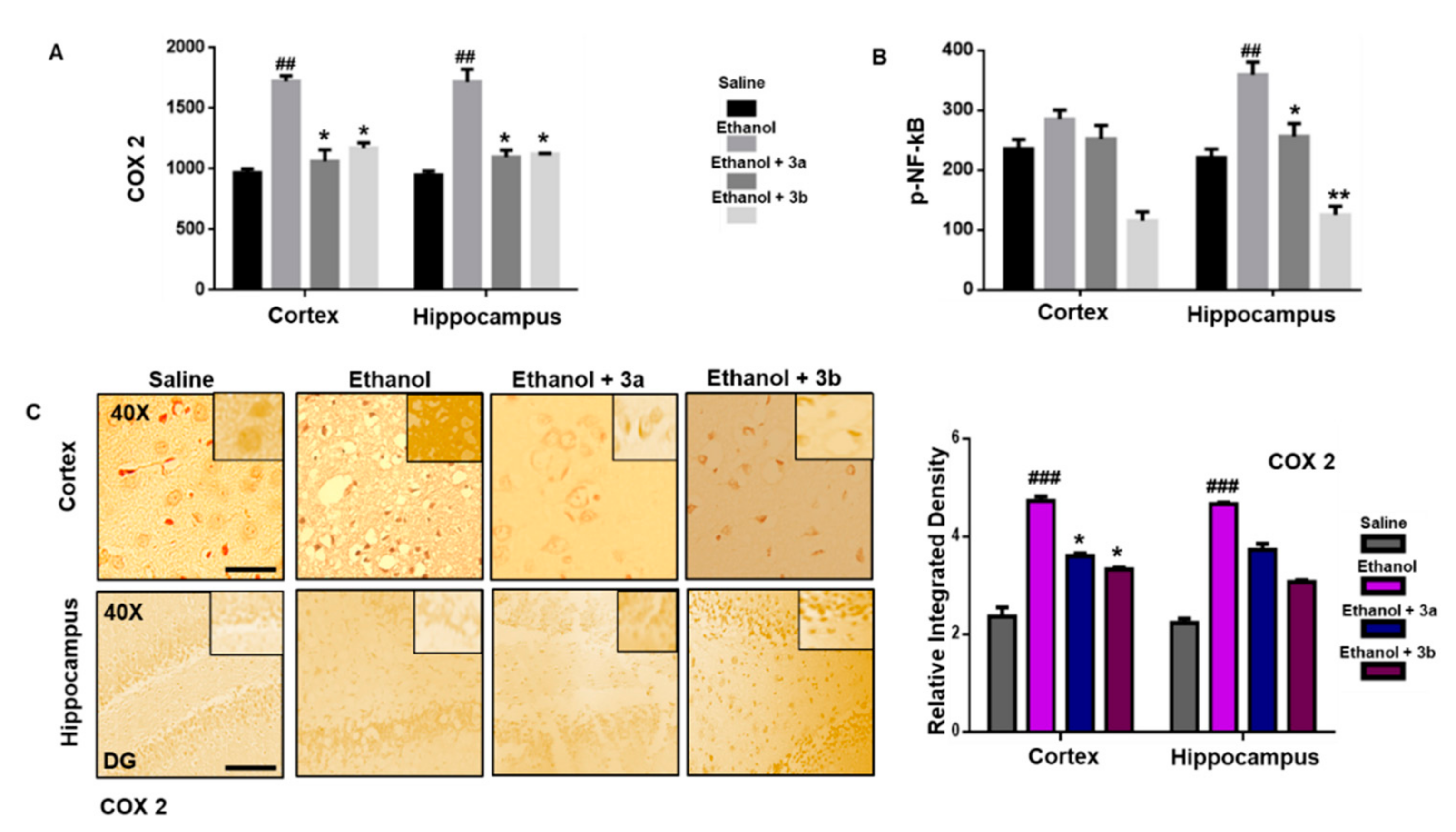
3.7. Pre-Treatment Dosage Regimen Downregulated Neuroinflammation
3.8. Pre-Treatment Dosage Regimen Attenuated the Ethanol-Induced Inflammatory Mediators
3.9. Effect on Oxidative Enzyme Variations
3.10. Effect on Ethanol-Induced Lipid Peroxidation (LPO)
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Castegna, A.; Aksenov, M.; Aksenova, M.; Thongboonkerd, V.; Klein, J.B.; Pierce, W.M.; Booze, R.; Markesbery, W.R.; Butterfield, D.A. Proteomic identification of oxidatively modified proteins in Alzheimer’s disease brain. Part I: creatine kinase BB, glutamine synthase, and ubiquitin carboxy-terminal hydrolase L-1. Free Radic. Biol. Med. 2002, 33, 562–571. [Google Scholar] [CrossRef]
- Olanow, C.W. A radical hypothesis for neurodegeneration. Trends Neurosci. 1993, 16, 439–444. [Google Scholar] [CrossRef]
- Shah, F.A.; Kury, L.A.; Li, T.; Zeb, A.; Koh, P.O.; Liu, F.; Zhou, Q.; Hussain, I.; Khan, A.U.; Jiang, Y.; et al. Polydatin Attenuates Neuronal Loss via Reducing Neuroinflammation and Oxidative Stress in Rat MCAO Models. Front. Pharmacol. 2019, 10, 663. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.A.; Liu, G.; Al Kury, L.T.; Zeb, A.; Koh, P.-O.; Abbas, M.; Li, T.; Yang, X.; Liu, F.; Jiang, Y.; et al. Melatonin Protects MCAO-Induced Neuronal Loss via NR2A Mediated Prosurvival Pathways. Front. Pharmacol. 2019, 10, 297. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tocchetti, C.G.; Krieg, T.; Moens, A.L. Oxidative and nitrosative stress in the maintenance of myocardial function. Free Radic. Biol. Med. 2012, 53, 1531–1540. [Google Scholar] [CrossRef]
- Al Kury, L.T.; Zeb, A.; Abidin, Z.U.; Irshad, N.; Malik, I.; Alvi, A.M.; Khalil, A.A.K.; Ahmad, S.; Faheem, M.; Khan, A.U.; et al. Neuroprotective effects of melatonin and celecoxib against ethanol-induced neurodegeneration: a computational and pharmacological approach. Drug Des. Dev. 2019, 13, 2715–2727. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.A.; Park, D.J.; Koh, P.O. Identification of Proteins Differentially Expressed by Quercetin Treatment in a Middle Cerebral Artery Occlusion Model: A Proteomics Approach. Neurochem. Res. 2018, 43, 1608–1623. [Google Scholar] [CrossRef]
- Spranger, M.; Fontana, A. Activation of Microglia: A Dangerous Interlude in Immune Function in the Brain. Neuroscientist 1996, 2, 293–299. [Google Scholar] [CrossRef]
- Vilhardt, F. Microglia: Phagocyte and glia cell. Int. J. Biochem. Cell Biol. 2005, 37, 17–21. [Google Scholar] [CrossRef]
- Gebicke-Haerter, P.J. Microarrays and expression profiling in microglia research and in inflammatory brain disorders. J. Neurosci. Res. 2005, 81, 327–341. [Google Scholar] [CrossRef]
- Byrnes, K.R.; Garay, J.; Di Giovanni, S.; De Biase, A.; Knoblach, S.M.; Hoffman, E.P.; Movsesyan, V.; Faden, A.I. Expression of two temporally distinct microglia-related gene clusters after spinal cord injury. Glia 2006, 53, 420–433. [Google Scholar] [CrossRef] [PubMed]
- Lenzlinger, P.M.; Hans, V.H.; Joller-Jemelka, H.I.; Trentz, O.; Morganti-Kossmann, M.C.; Kossmann, T. Markers for cell-mediated immune response are elevated in cerebrospinal fluid and serum after severe traumatic brain injury in humans. J. Neurotrauma 2001, 18, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Ali, T.; Rehman, S.U.; Shah, F.A.; Kim, M.O. Acute dose of melatonin via Nrf2 dependently prevents acute ethanol-induced neurotoxicity in the developing rodent brain. J. Neuroinflamm. 2018, 15, 119. [Google Scholar] [CrossRef] [PubMed]
- Aloisi, F. Immune function of microglia. Glia 2001, 36, 165–179. [Google Scholar] [CrossRef]
- Reddy, V.D.; Padmavathi, P.; Kavitha, G.; Saradamma, B.; Varadacharyulu, N. Alcohol-induced oxidative/nitrosative stress alters brain mitochondrial membrane properties. Mol. Cell. Biochem. 2013, 375, 39–47. [Google Scholar] [CrossRef]
- Akira, S.; Takeda, K.; Kaisho, T. Toll-like receptors: Critical proteins linking innate and acquired immunity. Nat. Immunol. 2001, 2, 675–680. [Google Scholar] [CrossRef]
- Amor, S.; Puentes, F.; Baker, D.; van der Valk, P. Inflammation in neurodegenerative diseases. Immunology 2010, 129, 154–169. [Google Scholar] [CrossRef]
- Martinon, F. Dangerous liaisons: Mitochondrial DNA meets the NLRP3 inflammasome. Immunity 2012, 36, 313–315. [Google Scholar] [CrossRef]
- Lampron, A.; ElAli, A.; Rivest, S. Innate immunity in the CNS: Redefining the relationship between the CNS and Its environment. Neuron 2013, 78, 214–232. [Google Scholar] [CrossRef]
- O’Neill, L.A.; Bowie, A.G. The family of five: TIR-domain-containing adaptors in Toll-like receptor signalling. Nat. Rev. Immunol. 2007, 7, 353–364. [Google Scholar] [CrossRef]
- Takeda, K.; Kaisho, T.; Akira, S. Toll-like receptors. Ann. Rev. Immun. 2003, 21, 335–376. [Google Scholar] [CrossRef] [PubMed]
- Harrison, T.S.; Keating, G.M. Zolpidem. CNS Drugs 2005, 19, 65–89. [Google Scholar] [CrossRef] [PubMed]
- Margiotta, N.; Ostuni, R.; Ranaldo, R.; Denora, N.; Laquintana, V.; Trapani, G.; Liso, G.; Natile, G. Synthesis and characterization of a platinum(II) complex tethered to a ligand of the peripheral benzodiazepine receptor. J. Med. Chem. 2007, 50, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.-Z.; Li, S.-Q.; Liao, W.-L.; Xie, Z.-G.; Wang, M.-S.; Cao, Y.; Zhang, J.; Xu, Z.-G. Efficient method for the synthesis of fused benzimidazole–imidazoles via deprotection and cyclization reactions. Tetrahedron 2015, 71, 8424–8427. [Google Scholar] [CrossRef]
- Evans, B.E.; Rittle, K.E.; Bock, M.G.; DiPardo, R.M.; Freidinger, R.M.; Whitter, W.L.; Lundell, G.F.; Veber, D.F.; Anderson, P.S.; Chang, R.S.; et al. Methods for drug discovery: Development of potent, selective, orally effective cholecystokinin antagonists. J. Med. Chem. 1988, 31, 2235–2246. [Google Scholar] [CrossRef]
- Mader, M.; de Dios, A.; Shih, C.; Bonjouklian, R.; Li, T.; White, W.; de Uralde, B.L.; Sánchez-Martinez, C.; del Prado, M.; Jaramillo, C.; et al. Imidazolyl benzimidazoles and imidazo[4,5-b]pyridines as potent p38α MAP kinase inhibitors with excellent in vivo antiinflammatory properties. Bioorg. Med. Chem. Lett. 2008, 18, 179–183. [Google Scholar] [CrossRef]
- Zawawi, N.K.N.A.; Taha, M.; Ahmat, N.; Wadood, A.; Ismail, N.H.; Rahim, F.; Azam, S.S.; Abdullah, N. Benzimidazole derivatives as new α-glucosidase inhibitors and in silico studies. Bioorg. Chem. 2016, 64, 29–36. [Google Scholar] [CrossRef]
- El-Feky, S.A.H.; Abd El-Samii, Z.K.; Osman, N.A.; Lashine, J.; Kamel, M.A.; Thabet, H.K. Synthesis, molecular docking and anti-inflammatory screening of novel quinoline incorporated pyrazole derivatives using the Pfitzinger reaction II. Bioorg. Chem. 2015, 58, 104–116. [Google Scholar] [CrossRef]
- Gaba, M.; Gaba, P.; Uppal, D.; Dhingra, N.; Bahia, M.S.; Silakari, O.; Mohan, C. Benzimidazole derivatives: Search for GI-friendly anti-inflammatory analgesic agents. Acta Pharm. Sin. 2015, 5, 337–342. [Google Scholar] [CrossRef]
- Zhu, J.; Wu, C.-F.; Li, X.; Wu, G.-S.; Xie, S.; Hu, Q.-N.; Deng, Z.; Zhu, M.X.; Luo, H.-R.; Hong, X. Synthesis, biological evaluation and molecular modeling of substituted 2-aminobenzimidazoles as novel inhibitors of acetylcholinesterase and butyrylcholinesterase. Bioorg. Med. Chem. 2013, 21, 4218–4224. [Google Scholar] [CrossRef]
- Hay, L.A.; Koenig, T.M.; Ginah, F.O.; Copp, J.D.; Mitchell, D. Palladium-Catalyzed Hydroarylation of Propiolamides. A Regio- and Stereocontrolled Method for Preparing 3,3-Diarylacrylamides. J. Org. Chem. 1998, 63, 5050–5058. [Google Scholar] [CrossRef]
- Ashraf, Z.; Alamgeer, A.; Rasool, R.; Hassan, M.; Ahsan, H.; Afzal, S.; Afzal, K.; Cho, H.; Kim, S.J. Synthesis, Bioevaluation and Molecular Dynamic Simulation Studies of Dexibuprofen-Antioxidant Mutual Prodrugs. Int. J. Mol. Sci. 2016, 17, 2151. [Google Scholar] [CrossRef] [PubMed]
- Abbas, Q.; Ashraf, Z.; Hassan, M.; Nadeem, H.; Latif, M.; Afzal, S.; Seo, S.-Y. Development of highly potent melanogenesis inhibitor by in vitro, in vivo and computational studies. Drug. Des. Dev. 2017, 11, 2029–2046. [Google Scholar] [CrossRef] [PubMed]
- Volkamer, A.; Kuhn, D.; Grombacher, T.; Rippmann, F.; Rarey, M. Combining global and local measures for structure-based druggability predictions. J. Chem. Inf. Model. 2012, 52, 360–372. [Google Scholar] [CrossRef] [PubMed]
- O’Boyle, N.M.; Banck, M.; James, C.A.; Morley, C.; Vandermeersch, T.; Hutchison, G.R. Open Babel: An open chemical toolbox. J. Cheminform. 2011, 3, 33. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]
- Bashir, M.A.; Khan, A.U.; Badshah, H.; Rodrigues-Filho, E.; Din, Z.U.; Khan, A. Synthesis, characterization, molecular docking evaluation, antidepressant, and anti-Alzheimer effects of dibenzylidene ketone derivatives. Drug Dev. Res. 2019, 80, 595–605. [Google Scholar] [CrossRef]
- Khan, A.; Shal, B.; Naveed, M.; Shah, F.A.; Atiq, A.; Khan, N.U.; Kim, Y.S.; Khan, S. Matrine ameliorates anxiety and depression-like behaviour by targeting hyperammonemia-induced neuroinflammation and oxidative stress in CCl4 model of liver injury. Neurotoxicology 2019, 72, 38–50. [Google Scholar] [CrossRef]
- Khan, S.; Shehzad, O.; Chun, J.; Choi, R.J.; Park, S.; Islam, M.N.; Choi, J.S.; Kim, Y.S. Anti-hyperalgesic and anti-allodynic activities of capillarisin via suppression of inflammatory signaling in animal model. J. Ethnopharmacol. 2014, 152, 478–486. [Google Scholar] [CrossRef]
- Davies, C.A.; Loddick, S.A.; Toulmond, S.; Stroemer, R.P.; Hunt, J.; Rothwell, N.J. The progression and topographic distribution of interleukin-1beta expression after permanent middle cerebral artery occlusion in the rat. J. Cereb. Blood Flow Metab. 1999, 19, 87–98. [Google Scholar] [CrossRef]
- Nakajima, K.; Tohyama, Y.; Kohsaka, S.; Kurihara, T. Protein kinase C alpha requirement in the activation of p38 mitogen-activated protein kinase, which is linked to the induction of tumor necrosis factor alpha in lipopolysaccharide-stimulated microglia. Neurochem. Int. 2004, 44, 205–214. [Google Scholar] [CrossRef]
- Akiyama, H.; Barger, S.; Barnum, S.; Bradt, B.; Bauer, J.; Cole, G.M.; Cooper, N.R.; Eikelenboom, P.; Emmerling, M.; Fiebich, B.L.; et al. Inflammation and Alzheimer’s disease. Neurobiol. Aging 2000, 21, 383–421. [Google Scholar] [CrossRef]
- Eikelenboom, P.; Bate, C.; Van Gool, W.A.; Hoozemans, J.J.; Rozemuller, J.M.; Veerhuis, R.; Williams, A. Neuroinflammation in Alzheimer’s disease and prion disease. Glia 2002, 40, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Gage, F.H. Adult hippocampal neurogenesis and its role in Alzheimer’s disease. Mol. Neurodegener. 2011, 6, 85. [Google Scholar] [CrossRef] [PubMed]
- Pascual, M.; Blanco, A.M.; Cauli, O.; Minarro, J.; Guerri, C. Intermittent ethanol exposure induces inflammatory brain damage and causes long-term behavioural alterations in adolescent rats. Eur. J. Neurosci. 2007, 25, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Vetreno, R.P.; Lawrimore, C.J.; Rowsey, P.J.; Crews, F.T. Persistent Adult Neuroimmune Activation and Loss of Hippocampal Neurogenesis Following Adolescent Ethanol Exposure: Blockade by Exercise and the Anti-inflammatory Drug Indomethacin. Front. Neurosci. 2018, 12, 200. [Google Scholar] [CrossRef]
- Ajani, O.O.; Aderohunmu, D.V.; Ikpo, C.O.; Adedapo, A.E.; Olanrewaju, I.O. Functionalized Benzimidazole Scaffolds: Privileged Heterocycle for Drug Design in Therapeutic Medicine. Arch. Pharm. 2016, 349, 475–506. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, W.; Khan, M.F.; Verma, G.; Shaquiquzzaman, M.; Rizvi, M.A.; Mehdi, S.H.; Akhter, M.; Alam, M.M. Therapeutic evolution of benzimidazole derivatives in the last quinquennial period. Eur. J. Med. Chem. 2017, 126, 705–753. [Google Scholar] [CrossRef] [PubMed]
- Bansal, Y.; Kaur, M.; Silakari, O. Benzimidazole–ibuprofen/mesalamine conjugates: Potential candidates for multifactorial diseases. Eur. J. Med. Chem. 2015, 89, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Bansal, Y.; Silakari, O. 2-Aminobenzimidazole conjugates of NSAIDS: Novel compounds with immunomodulatory, anti-inflammatory and antioxidant actions. Med. Chem. Res. 2015, 24, 1170–1179. [Google Scholar] [CrossRef]
- He, X.; Lakkaraju, S.K.; Hanscom, M.; Zhao, Z.; Wu, J.; Stoica, B.; MacKerell, A.D., Jr.; Faden, A.I.; Xue, F. Acyl-2-aminobenzimidazoles: A novel class of neuroprotective agents targeting mGluR5. Bioorg. Med. Chem. 2015, 23, 2211–2220. [Google Scholar] [CrossRef] [PubMed]
- Baudy, R.B.; Fletcher, H., 3rd; Yardley, J.P.; Zaleska, M.M.; Bramlett, D.R.; Tasse, R.P.; Kowal, D.M.; Katz, A.H.; Moyer, J.A.; Abou-Gharbia, M. Design, synthesis, SAR, and biological evaluation of highly potent benzimidazole-spaced phosphono-alpha-amino acid competitive NMDA antagonists of the AP-6 type. J. Med. Chem. 2001, 44, 1516–1529. [Google Scholar] [CrossRef] [PubMed]
- Yoon, Y.K.; Ali, M.A.; Wei, A.C.; Choon, T.S.; Khaw, K.Y.; Murugaiyah, V.; Osman, H.; Masand, V.H. Synthesis, characterization, and molecular docking analysis of novel benzimidazole derivatives as cholinesterase inhibitors. Bioorg. Chem. 2013, 49, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Boggu, P.; Venkateswararao, E.; Manickam, M.; Kwak, D.; Kim, Y.; Jung, S.H. Exploration of 2-benzylbenzimidazole scaffold as novel inhibitor of NF-kappaB. Bioorg. Med. Chem. 2016, 24, 1872–1878. [Google Scholar] [CrossRef]
- Arora, R.K.; Kaur, N.; Bansal, Y.; Bansal, G. Novel coumarin–benzimidazole derivatives as antioxidants and safer anti-inflammatory agents. Acta Pharm. Sin. 2014, 4, 368–375. [Google Scholar] [CrossRef]
- Tournier, C.; Hess, P.; Yang, D.D.; Xu, J.; Turner, T.K.; Nimnual, A.; Bar-Sagi, D.; Jones, S.N.; Flavell, R.A.; Davis, R.J. Requirement of JNK for stress-induced activation of the cytochrome c-mediated death pathway. Science 2000, 288, 870–874. [Google Scholar] [CrossRef]
- Liu, J.; Lin, A. Role of JNK activation in apoptosis: A double-edged sword. Cell Res. 2005, 15, 36–42. [Google Scholar] [CrossRef]
- Valles, S.L.; Blanco, A.M.; Pascual, M.; Guerri, C. Chronic ethanol treatment enhances inflammatory mediators and cell death in the brain and in astrocytes. Brain Pathol. 2004, 14, 365–371. [Google Scholar] [CrossRef]
- Alfonso-Loeches, S.; Pascual-Lucas, M.; Blanco, A.M.; Sanchez-Vera, I.; Guerri, C. Pivotal role of TLR4 receptors in alcohol-induced neuroinflammation and brain damage. J. Neurosci. 2010, 30, 8285–8295. [Google Scholar] [CrossRef]
- Tumiatti, V.; Minarini, A.; Bolognesi, M.L.; Milelli, A.; Rosini, M.; Melchiorre, C. Tacrine derivatives and Alzheimer’s disease. Curr. Med. Chem. 2010, 17, 1825–1838. [Google Scholar] [CrossRef]
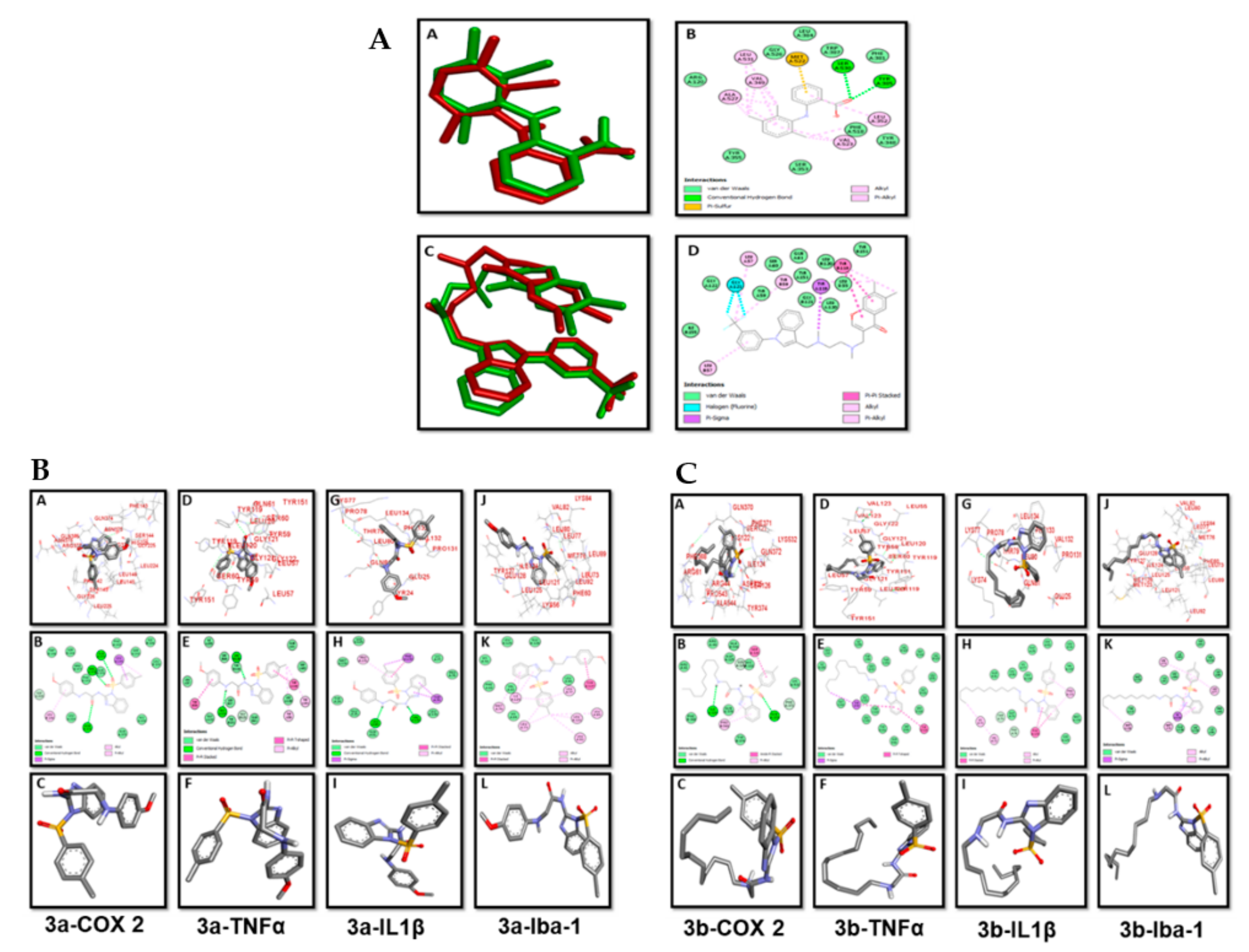
| Compounds | COX2 | TNF-α | IL-1β | Iba-1 |
|---|---|---|---|---|
| Binding Energies (kcal/mol) | ||||
| 3a | –8.9 | –8.4 | –7.6 | –7.3 |
| 3b | –7.4 | –7.4 | –6 | –7 |
| Co-crystal | –9.1 | –9.3 | - | - |
| Group | GSH (mg/1100 g Tissue) | GST (pmol) | i-NOS (pmol) | LPO |
|---|---|---|---|---|
| Saline | 54.88 ± 3.78 | 40.42 ± 1.30 | 21.02± 7.61 | 79.32 ±0.70 |
| Ethanol | 3.30 ± 0.97 ### | 2.78 ± 0.45 ### | 85 ± 6 ### | 224.54 ± 4.32 ### |
| Ethanol + 3a | 34.3 ± 2.4 ** | 28.05 ± 4.3 ** | 63.15 ± 3.8 * | 160.45 ± 8.9 * |
| Ethanol + 3b | 40.7 ± 4.1 ** | 35.8 ± 5.1 *** | 65.2 ± 4.56 * | 130.8 ± 2.82 ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imran, M.; Al Kury, L.T.; Nadeem, H.; Shah, F.A.; Abbas, M.; Naz, S.; Khan, A.-u.; Li, S. Benzimidazole Containing Acetamide Derivatives Attenuate Neuroinflammation and Oxidative Stress in Ethanol-Induced Neurodegeneration. Biomolecules 2020, 10, 108. https://doi.org/10.3390/biom10010108
Imran M, Al Kury LT, Nadeem H, Shah FA, Abbas M, Naz S, Khan A-u, Li S. Benzimidazole Containing Acetamide Derivatives Attenuate Neuroinflammation and Oxidative Stress in Ethanol-Induced Neurodegeneration. Biomolecules. 2020; 10(1):108. https://doi.org/10.3390/biom10010108
Chicago/Turabian StyleImran, Muhammad, Lina Tariq Al Kury, Humaira Nadeem, Fawad Ali Shah, Muzaffar Abbas, Shagufta Naz, Arif-ullah Khan, and Shupeng Li. 2020. "Benzimidazole Containing Acetamide Derivatives Attenuate Neuroinflammation and Oxidative Stress in Ethanol-Induced Neurodegeneration" Biomolecules 10, no. 1: 108. https://doi.org/10.3390/biom10010108
APA StyleImran, M., Al Kury, L. T., Nadeem, H., Shah, F. A., Abbas, M., Naz, S., Khan, A.-u., & Li, S. (2020). Benzimidazole Containing Acetamide Derivatives Attenuate Neuroinflammation and Oxidative Stress in Ethanol-Induced Neurodegeneration. Biomolecules, 10(1), 108. https://doi.org/10.3390/biom10010108





