Abstract
To assess the association of environmental chemical factors with osteopenia and/or bone fractures. All data were extracted from the National Health and Nutrition Survey (NHANES) 2017–2018 of American adults aged 20–59 years old; invalid data were excluded based on dual-energy X-ray absorptiometry. For the ultimate valid data set, multivariate logistic regression models were applied to evaluate the association of environmental chemical factors with osteopenia and bone fractures. The valid dataset was obtained from 2640 individuals, who completed a questionnaire of demographic characteristics. Urinary manganese and monomethylarsonic acid were positively associated with osteopenia in American adults, but not bone fracture. However, several environmental factors (e.g., arsenous acid, arsenocholine, dimethylarsinic acid, and 2-thioxothiazolidine-4-carboxylic acid) did not affect bone mineral density, but were significantly associated with bone fracture. Multiple environmental chemical factors significantly affect bone mass or fracture risk. However, the risk of environmental chemical factors on fractures is independent of osteopenia in US Adults. The influence of environmental chemical factors on bone quality should be considered and monitored.
1. Introduction
Osteoporosis is a chronic skeletal disorder, characterized by compromised bone mineral density (BMD) and bone strength, impaired bone microstructure, and increased fracture risk at several sites (e.g., hip, wrist, spine, and other skeletal sites). BMD is the commonly used parameter for bone health evaluation; the total body and lumbar spine are preferred sites for BMD assessment [1]. Bone mass increases significantly during childhood and adolescence, and reaches its peak value at approximately 20 years [2]. Reduced BMD could result in osteoporosis [3]. Numerous risk factors contribute to BMD reduction and bone fracture, including nutritional, genetic, and metabolic factors, low physical activity, drugs, and cigarette smoking [4,5,6]. Additionally, environmental factors play an important role in bone health.
It has been confirmed by some evidence that bone health can be affected by heavy metals. In a retrospective study of itai–itai disease cases, Inaba et al. [7] found that high-dose cadmium exposure induced generalized osteomalacia and osteoporosis, resulting in multiple bone fractures. Increased osteoporosis-related bone fracture risk has also been observed after high selenium level exposure [8]. However, heavy metals are not only detrimental to bone health. Existing studies indicated that zinc and mercury deficiency cause bone loss [9,10]. Campbell et al. [11] observed that higher lead exposure led to higher BMD in children. Furthermore, dietary manganese intake is positively correlated with lumbar BMD. Lower copper levels were associated with lower BMD, and long-term exposure to manganese and copper increased the risk of osteoporosis [12,13,14]. As well as heavy metals, polycyclic aromatic hydrocarbons (PAHs) are toxic to bone health; they are characterized by disrupted bone homeostasis [15], leading to the inflammation of various joints with synovitis, progressive bone deterioration, and bone metabolic diseases [16,17,18].
Most previous studies have focused on the impact of a single environmental factor on BMD or fracture, and believed that reducing bone mineral density would lead to fracture to a certain degree. In this cross-sectional study, we analyzed the effect of different environmental chemical factors on BMD and bone fractures based on data from the 2017–2018 cycle of the National Health and Nutrition Survey (NHANES), and explored the association of multiple environmental chemical factors with BMD and bone fractures.
2. Materials and Methods
2.1. Data Sources and Study Design
The NHANES is a population-based, national, cross-sectional survey, initiated by the National Health Statistics Center of the US. The database combines information collected in interviews and physical examinations to collect health and nutritional status data of US adults and children from a representative sample of the noninstitutionalized civilian population; data are released every 2 years. In this study, we extracted demographic data from the 2017–2018 cycle of the NHANES, comprising data from 9254 individuals. We screened 3419 individuals in the 20–59 age range, of whom 2253 individuals underwent dual-energy X-ray absorptiometry (DXA) to determine total BMD. According to valid DXA data and self-reported information regarding fractures of the hip, wrist, and spine; in total, data were included for analysis of the associations of environmental chemical factors with BMD and bone fracture from 2640 individuals (aged 20–59 years) and 292 individuals (aged 50–59 years), respectively. For dual-energy x-ray absorptiometry, the inclusion criteria in the present study included the following: (1) participants aged 20–59 years; (2) whole body scan completed; (3) all regions were valid. The exclusion criteria were as follows: (1) participants with a positive urine pregnancy test and/or self-reported pregnancy; (2) a history of radiographic contrast material use in past 7 days; (3) weight over 450 pounds or height over 6′5″ (DXA table limitation). In this study, all data were recorded in the NHANES, which the Ethics Committee of the National Health Statistics Center approved.
2.2. Biomonitoring
Urine samples were collected from one-third of eligible subjects in the cohort, processed, stored at −30 °C, and shipped to the US National Center for Environmental Health for testing. The concentrations of a number of metals in urine specimens were determined by inductively coupled plasma–mass spectrometry with dynamic reaction cell technology (ICP-DRC-MS). Ultra-performance liquid chromatography coupled with electrospray tandem mass spectrometry was used to measure volatile organic compound (VOC) metabolites [19,20]. For speciated arsenic, high-performance liquid chromatography was used to separate the species and coupled to ICP-DRC-MS to detect arsenic concentration [21].
2.3. Bone Densitometry
The reference range for standardizing different bones is determined according to the International Committee for Standards in Bone Densitometry [22], and classified by gender, race, and ethnicity. Based on the T-score formula, a BMD value between 1 standard deviation (SD) and 2.5 SD below the reference mean indicates osteopenia, while a value over 2.5 SD indicates osteoporosis [23].
In the NHANES, the DXA examination provided representative BMD data for different bone tissue sites, including the whole body, trunk, pelvis, thoracic vertebrae, lumbar vertebrae, and limbs. The examinees in the 2017–2018 NHANES were between 8 and 59 years old. Herein, we selected the adult data from the participants aged 20–59 to analyze, and pregnant women were excluded. In the DXA, the whole body was scanned using a Hologic Discovery Model A densitometer (Hologic, Inc., Bedford, MA, USA), and information was collected using Hologic Apex version 3.2 software. All scans were analyzed using Hologic Apex version 4.0 software [24].
2.4. Statistical Analysis
All statistical analyses were conducted using R version 4.2.1 and SPSS version 26.0 for Windows (IBM); p < 0.05 was considered statistically significant. A multivariate logistic regression model was applied to evaluate correlations of different environmental chemical factors with BMD and fracture of the hip, wrist, and spine; categorical outcomes were reported through p value and the odds ratio with a 95% confidence interval (CI). The multivariate logistic regression model was adjusted to control for potential confounders by covariates, including age, gender, body mass index (BMI), race or ethnicity, education level, country of birth, the ratio of family income to poverty, and current smoking status.
3. Results
3.1. Demographic Characteristics of Participants with Osteopenia
In this study, 3419 individuals aged 20–59 years (mean, 40.24 years; 95% CI, 39.80–40.69 years) from the 2017–2018 NHANES completed a questionnaire on demographic characteristics. Using the DXA scans as a further selection criterion, the final valid data for analysis was from 2640 individuals. The specific selection process is shown in Figure 1.
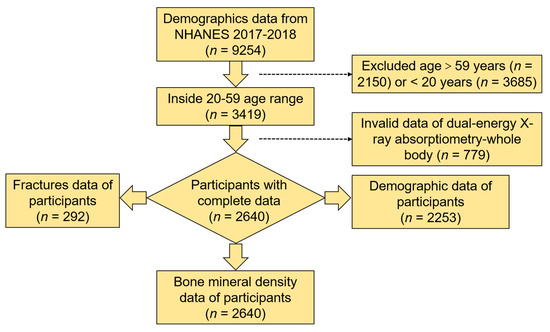
Figure 1.
Flow chart of the selection process from the 2017–2018 NHANES.
Using total BMD as a selection criterion, 2253 individuals (1073 (47.63%) men and 1180 (52.33%) women) with osteopenia were selected; the data analysis is given in Table 1. Americans who were female (p < 0.001), over 65 years old (p < 0.001), had a lower BMI (p < 0.001), were educated to high school graduation or equivalent or to less than 9th grade (p = 0.049), or who had been born abroad (p = 0.008) were more likely to have an increased risk of osteopenia. A recent study [25] demonstrated that fracture incidence varies with socioeconomic status, race, and ethnicity, but race, ethnicity (p = 0.801), family income (p = 0.175), and current smoking status (p = 0.089) have no significant effect on BMD in the US.

Table 1.
Characteristics of American adults with osteopenia in 2017–2018.
3.2. Associations between Different Environmental Chemical Factors and Osteopenia
Means, SDs, and reference ranges of BMD and cut-off values of osteopenia and osteoporosis corresponding to the World Health Organization diagnostic criteria at major anatomic sites for the reference groups were classified by sex, race, and ethnicity [26]. The specific reference values are given in Table 2.

Table 2.
Mean bone mineral density of osteopenia and osteoporosis aged 20–29 years in 2017–2018.
A multivariate logistic regression model was adjusted by covariates, demonstrating the association between environmental chemical factors and BMD at different skeletal sites (Table 3). For total body, lower levels exposure of urinary mercury (odds ratio (OR), 0.567; CI, 0.357–0.900; p = 0.016, 2-methylhippuric acid (OR, 0.425; CI, 0.232–0.778; p = 0.006), and N-acetyl-S-(2-hydroxypropyl)-L-cysteine (OR, 0.415; CI, 0.175–0.982; p = 0.045) were in statistically significant association with osteopenia. For the specific body sites, lower levels of urinary barium (OR, 0.130; CI, 0.017–0.983, p = 0.048) are obviously associated with left arm osteopenia; lower levels of N-acetyl-S-(n-propyl)-L-cysteine (OR, 0.502; CI, 0.298–0.844, p = 0.009) and N-acetyl-S-(phenyl-2-hydroxyethyl)-L-cysteine (OR, 0.552; CI, 0.324–0.939, p = 0.028) remained in statistically significant association with osteopenia in the right arm. Lower levels of urinary barium (OR, 0.088; CI, 0.009–0.864; p = 0.037), mercury (OR, 0.514; CI, 0.341–0.773; p = 0.001), 2-methylhippuric acid (OR, 0.487; CI, 0.275–0.863; p = 0.014), and N-acetyl-S-(2-hydroxypropyl)-L-cysteine (CYHA) (OR, 0.56; CI, 0.322–0.975; p = 0.04) remained in statistically significant association with left leg osteopenia. Lower levels of urinary barium (OR, 0.093; CI, 0.009–0.912; p = 0.041), mercury (OR, 0.529; CI, 0.350–0.798; p = 0.002), 2-methylhippuric acid (OR, 0.450; CI, 0.255–0.795; p = 0.006), and N-acetyl-S-(4-hydroxy-2-methyl-2-buten-1-yl)-L-cysteine (IPM3) (OR, 0.566; CI, 0.337–0.949, p = 0.031) were in statistically significant association with right leg osteopenia.

Table 3.
Associations with multiple environmental chemical factors and osteopenia in 2017–2018.
There is a strong association of high urinary manganese with osteopenia in the left arm (OR, 1.610; CI, 1.015–2.554; p = 0.043) or pelvis (OR, 1.587; CI, 1.001–2.516; p = 0.05) and monomethylarsonic acid (OR, 1.451; CI, 1.007–2.090; p = 0.046) with osteopenia of the lumbar spine. The results are given in Table 3.
3.3. Associations between Different Environmental Chemical Factors and Fracture
In this study, fracture data from the hip, wrist, and spine were combined into one dataset, owing to the small amount of data. Table 4 summarizes the associations of different environmental chemical factors with bone fractures at the hip, wrist, and spine in the multivariate models. Adjusted by covariants, high levels of arsenous acid (OR, 2.578, CI: 1.358–4.893; p = 0.004), arsenobetaine (OR, 2.978; CI, 1.052–8.427; p = 0.04), dimethylarsinic acid (OR, 2.087; CI, 1.06–4.112; p = 0.033), monomethylarsonic acid (OR, 2.276; CI, 1.225–4.226; p = 0.009), and 2-thioxothiazolidine-4-carboxylic acid (OR, 1.804; CI, 1.004–3.242; p = 0.048) were associated with an increased prevalence of osteoporotic fractures.

Table 4.
Associations with multiple environmental chemical factors and fractures in 2017–2018.
4. Discussion
Environmental chemical factors have different effects on bone development, and vary depending on the degree of exposure. In this cross-sectional study, we examined the association of environmental chemicals with BMD and bone fractures among a large sample of American adults from NHANES 2017–2018. The results in the present study demonstrate that urinary manganese, barium, mercury, dimethylarsinic acid, 2-methylhippuric acid, CYHA cysteine, N-acetyl-S-(2-hydroxypropyl)-L-cysteine, and IPM3 cysteine has a potential effect on osteopenia in American adults while not associated with fractures. Interestingly, the environmental chemicals (arsenous acid, arsenocholine, dimethylarsinic acid, monomethylarsonic acid, and 2-thioxothiazolidine-4-carboxylic acid) causing fracture are entirely different from the environmental chemicals that significantly reduce bone mass. The impact of environmental chemicals on fracture may not be caused by low bone mass. It is well known that osteoporosis is due to changes in bone microarchitecture and bone strength leading to fractures [27]. Bone strength is related to both BMD and bone microstructure [27]. However, only the assessment of BMD is available in clinical routine care today. Measuring bone turnover markers is the best non-invasive and easy non-time-consuming way to assess bone quality and the relationship between bone formation and bone resorption [28]. The latter increases physiologically with age, particularly in postmenopausal women, and may increase the risk of fractures.
Due to estrogen changes, fracture incidence in women over 50 is approximately twice that in men [29,30]; it is consistent with our findings. However, there is no information on the number of peri- and postmenopausal women in this database of 2017–2018 NHANES; it would be beneficial to investigate these data since the frequency of osteopenia in females was significantly higher than in males.
Previous data confirmed that environmental factors have an impact on BMD. Cadmium exposure inhibits osteoblast and osteoclast differentiation via the P2X7/PI3K/AKT signaling pathway, and causes osteoporosis by promoting osteoclast and osteoblast apoptosis [31]. In a steady state, bone contains about 80%–90% of the total body lead burden [32]. Cui et al. [33] showed that lead affected the lumbar BMD more than the femur, and that the impact on women was more robust. As mentioned above, the dietary intake of manganese is positively related to the lumbar spine, although one study found that blood manganese exposure was negatively related to BMD [34]. Based on these two results, our study found a strong positive correlation between urinary manganese exposure and osteopenia; this result is consistent with the study of Karamati et al. [12]. In addition, the zinc/copper ratio in serum and bone is positively correlated with systemic BMD and BMC in older men with osteoarthritis [35]. Serum selenium level was positively correlated with BMD in the forearm and heel [36]. However, dietary selenium intake as a function of BMD shows an inverted U-shaped trend, owing to the status of selenium in the body [37]. Blood mercury is positively associated with spine BMD and negatively associated with femoral BMD [38]. Our study confirms that urinary mercury is positively associated with the BMD of the legs and total body. Furthermore, to our knowledge, we did not find any research on the relationship between barium and BMD; however, we found that low-level urinary barium exposure in the left arm and legs may significantly increase the risk of osteopenia.
Nguyen et al. [39] indicated that PAHs could inhibit absorption of osteoclasts and synthesis of osteoblasts, and damage bone remodeling. Izawa et al. [40] suggested that benzopyrene accelerated the development of osteoclasts. High exposure to 3-methylcholanthrene [41] results in bone loss and decreases bone mechanical properties. Hsueh et al. [42] demonstrated that high total urinary arsenic levels significantly increased the risk of bone loss. Consistently, we found that high urinary levels of monomethylarsonic acid had a significant correlation with osteopenia, and that high levels of arsenous acid, arsenocholine, dimethylarsinic acid, and dimethylarsinic acid significantly increased the possibility of fragility fracture. No other environmental factors (including heavy metals) have a significant impact on fracture. Subsequently, our study verified a negative correlation between lower VOC exposure with osteopenia. Some factors, including 2-methylhippuric acid, N-acetyl-S-(2-hydroxypropyl)-L-cysteine, N-acetyl-S-(phenyl-2-hydroxyethyl)-L-cysteine, CYHA cysteine, and IPM3 cysteine, and 2-thioxothiazolidine-4-carboxylic acid, have a potential role in osteopenia and only 2-Thioxothiazolidine-4-carboxylic acid was found to increase the risk of fracture significantly.
This study has some limitations. First, the sample size is not large enough to obtain more results specific to each bone region, and due to the lack of fracture data for the arm and pelvis, we cannot determine whether manganese increases the risk of fracture, although manganese has a significant effect on BMD reduction. More data should be collected to investigate the effects of environmental chemical factors on different bone regions. Moreover, the data of the individuals chosen in this study, collected in 2017 and 2018, can only reflect a short-term influence of environmental chemical factors; the time span should be extended for long-term effects.
5. Conclusions
The results of this study demonstrate that environmental chemical factors have strong associations with osteopenia and/or fracture. However, several factors (especially arsenic compounds) cause bone fragility fracture, but no bone loss. We considered that environmental chemical-induced fracture is independent of BMD. Further studies should explore the exact mechanisms of these factors, which will be helpful in the prevention and treatment of bone diseases caused by environmental problems.
Author Contributions
Conceptualization, R.L. and B.W.; methodology, R.L., Y.A. and C.C.; formal analysis, R.L., J.Z. and C.L.; investigation, R.L., Y.A. and Z.Z.; resources, B.W. and S.C.; data curation, R.L. and X.L.; writing—original draft preparation, R.L., Y.A. and B.W.; writing—review and editing, B.W.; visualization, R.L.; supervision, R.L.; project administration, X.L.; funding acquisition, B.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Natural Science Foundation of Chongqing, China (CSTB2022NSCQ-MSX0110), the Science and Technology Research Program of Chongqing Municipal Education Commission (KJQN202000421), and the Program for Youth Innovation in Future Medicine, Chongqing Medical University (W0046).
Institutional Review Board Statement
The study was performed in accordance with the Declaration of Helsinki, and approved by the NCHS Ethics Review Board (ERB) (Protocol Code #2018-01, effective beginning October 26, 2017; continuation of Protocol Code #2011-17, effective through 26 October 2017).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study in NHANES.
Data Availability Statement
Data available in a publicly accessible repository. Publicly available datasets were analyzed in this study. This data can be found in the National Health and Nutrition Examination Survey, https://wwwn.cdc.gov/nchs/nhanes/Default.aspx (accessed on 30 January 2023).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Medina-Gomez, C.; Kemp, J.P.; Trajanoska, K.; Luan, J.; Chesi, A.; Ahluwalia, T.S.; Mook-Kanamori, D.O.; Ham, A.; Hartwig, F.P.; Evans, D.S.; et al. Life-Course Genome-wide Association Study Meta-analysis of Total Body BMD and Assessment of Age-Specific Effects. Am. J. Hum. Genet. 2018, 102, 88–102. [Google Scholar] [CrossRef]
- Xue, S.; Kemal, O.; Lu, M.; Lix, L.M.; Leslie, W.D.; Yang, S. Age at attainment of peak bone mineral density and its associated factors: The National Health and Nutrition Examination Survey 2005–2014. Bone 2020, 131, 115163. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, R.; Naldini, G.; Chiavarini, M. Dietary Patterns in Relation to Low Bone Mineral Density and Fracture Risk: A Systematic Review and Meta-Analysis. Adv. Nutr. 2019, 10, 219–236. [Google Scholar] [CrossRef] [PubMed]
- Compston, J.E.; McClung, M.R.; Leslie, W.D. Osteoporosis. Lancet 2019, 393, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Ströhle, A.; Hahn, A. Nutrition and bone health: What ist the evidence? Med. Mon. Fur Pharm. 2016, 39, 236–244, quiz 245. [Google Scholar]
- Trajanoska, K.; Rivadeneira, F. The genetic architecture of osteoporosis and fracture risk. Bone 2019, 126, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Inaba, T.; Kobayashi, E.; Suwazono, Y.; Uetani, M.; Oishi, M.; Nakagawa, H.; Nogawa, K. Estimation of cumulative cadmium intake causing Itai-itai disease. Toxicol. Lett. 2005, 159, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Galvez-Fernandez, M.; Grau-Perez, M.; Garcia-Barrera, T.; Ramirez-Acosta, S.; Gomez-Ariza, J.L.; Perez-Gomez, B.; Galan-Labaca, I.; Navas-Acien, A.; Redon, J.; Briongos-Figuero, L.S.; et al. Arsenic, cadmium, and selenium exposures and bone mineral density-related endpoints: The HORTEGA study. Free Radic. Biol. Med. 2021, 162, 392–400. [Google Scholar] [CrossRef]
- Ryz, N.R.; Weiler, H.A.; Taylor, C.G. Zinc deficiency reduces bone mineral density in the spine of young adult rats: A pilot study. Ann. Nutr. Metab. 2009, 54, 218–226. [Google Scholar] [CrossRef]
- Lim, H.S.; Lee, H.H.; Kim, T.H.; Lee, B.R. Relationship between Heavy Metal Exposure and Bone Mineral Density in Korean Adult. J. Bone Metab. 2016, 23, 223–231. [Google Scholar] [CrossRef]
- Campbell, J.R.; Rosier, R.N.; Novotny, L.; Puzas, J.E. The association between environmental lead exposure and bone density in children. Environ. Health Perspect. 2004, 112, 1200–1203. [Google Scholar] [CrossRef] [PubMed]
- Karamati, M.; Yousefian-Sanni, M.; Shariati-Bafghi, S.E.; Rashidkhani, B. Major nutrient patterns and bone mineral density among postmenopausal Iranian women. Calcif. Tissue Int. 2014, 94, 648–658. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Ge, X.; Liu, Z.; Huang, L.; Zhou, Y.; Liu, P.; Qin, L.; Lin, S.; Liu, C.; Hou, Q.; et al. Association between long-term occupational manganese exposure and bone quality among retired workers. Environ. Sci. Pollut. Res. Int. 2020, 27, 482–489. [Google Scholar] [CrossRef]
- Qu, X.; He, Z.; Qiao, H.; Zhai, Z.; Mao, Z.; Yu, Z.; Dai, K. Serum copper levels are associated with bone mineral density and total fracture. J. Orthop. Transl. 2018, 14, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Kao, T.W.; Wang, C.C.; Wu, C.J.; Zhou, Y.C.; Chen, W.L. Association between polycyclic aromatic hydrocarbons exposure and bone turnover in adults. Eur. J. Endocrinol. 2020, 182, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Meng, X.; Sun, Y.; Jia, C. Association between polycyclic aromatic hydrocarbons and osteoporosis: Data from NHANES, 2005-2014. Arch. Osteoporos. 2018, 13, 112. [Google Scholar] [CrossRef]
- Guo, J.; Huang, Y.; Bian, S.; Zhao, C.; Jin, Y.; Yu, D.; Wu, X.; Zhang, D.; Cao, W.; Jing, F.; et al. Associations of urinary polycyclic aromatic hydrocarbons with bone mass density and osteoporosis in U.S. adults, NHANES 2005-2010. Environ. Pollut. (Barking Essex 1987) 2018, 240, 209–218. [Google Scholar] [CrossRef]
- Li, J.; Li, X.; Xia, Y.; Fan, H.; Fan, D.; Xi, X.; Ye, Q.; Zhu, Y.; Xiao, C. Subgroup analysis of the relationship between polycyclic aromatic hydrocarbons and rheumatoid arthritis: Data from the National Health and Nutrition Examination Survey, 2003–2014. Sci. Total Environ. 2021, 775, 145841. [Google Scholar] [CrossRef]
- Alwis, K.U.; Blount, B.C.; Britt, A.S.; Patel, D.; Ashley, D.L. Simultaneous analysis of 28 urinary VOC metabolites using ultra high performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry (UPLC-ESI/MSMS). Anal. Chim. Acta 2012, 750, 152–160. [Google Scholar] [CrossRef]
- National Health and Nutrition Examination Survey. 2017–2018 Data Documentation, Codebook, and Frequencies: Volatile Organic Compound (VOC) Metabolites—Urine (UVOC_J). Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2017-2018/UVOC_J.htm (accessed on 30 January 2023).
- National Health and Nutrition Examination Survey. 2017–2018 Data Documentation, Codebook, and Frequencies: Arsenics—Speciated—Urine (UAS_J). Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2017-2018/UAS_J.htm (accessed on 30 January 2023).
- Looker, A.C.; Orwoll, E.S.; Johnston, C.C., Jr.; Lindsay, R.L.; Wahner, H.W.; Dunn, W.L.; Calvo, M.S.; Harris, T.B.; Heyse, S.P. Prevalence of low femoral bone density in older U.S. adults from NHANES III. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1997, 12, 1761–1768. [Google Scholar] [CrossRef]
- Cohen, L.L.; Berry, J.G.; Ma, N.S.; Cook, D.L.; Hedequist, D.J.; Karlin, L.I.; Emans, J.B.; Hresko, M.T.; Snyder, B.D.; Glotzbecker, M.P. Spinal Fusion in Pediatric Patients With Low Bone Density: Defining the Value of DXA. J. Pediatr. Orthop. 2022, 42, e713–e719. [Google Scholar] [CrossRef] [PubMed]
- National Health and Nutrition Examination Survey. 2017–2018 Data Documentation, Codebook, and Frequencies: Dual-Energy X-ray Absorptiometry—Whole Body (DXX_J). Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2017-2018/DXX_J.htm (accessed on 30 January 2023).
- Fuggle, N.R.; Curtis, E.M.; Ward, K.A.; Harvey, N.C.; Dennison, E.M.; Cooper, C. Fracture prediction, imaging and screening in osteoporosis. Nat. Rev. Endocrinol. 2019, 15, 535–547. [Google Scholar] [CrossRef]
- Samelson, E.J.; Broe, K.E.; Xu, H.; Yang, L.; Boyd, S.; Biver, E.; Szulc, P.; Adachi, J.; Amin, S.; Atkinson, E.; et al. Cortical and trabecular bone microarchitecture as an independent predictor of incident fracture risk in older women and men in the Bone Microarchitecture International Consortium (BoMIC): A prospective study. Lancet Diabetes Endocrinol. 2019, 7, 34–43. [Google Scholar] [CrossRef]
- Brandi, M.L. Microarchitecture, the key to bone quality. Rheumatology 2009, 48, iv3–iv8. [Google Scholar] [CrossRef]
- Eastell, R.; Szulc, P. Use of bone turnover markers in postmenopausal osteoporosis. Lancet Diabetes Endocrinol. 2017, 5, 908–923. [Google Scholar] [CrossRef] [PubMed]
- Clynes, M.A.; Harvey, N.C.; Curtis, E.M.; Fuggle, N.R.; Dennison, E.M.; Cooper, C. The epidemiology of osteoporosis. Br. Med. Bull. 2020, 133, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Management of Osteoporosis in Postmenopausal Women: The 2021 Position Statement of The North American Menopause Society’’ Editorial Panel. Management of osteoporosis in postmenopausal women: The 2021 position statement of The North American Menopause Society. Menopause 2021, 28, 973–997. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Ran, D.; Zhao, H.; Song, R.; Zou, H.; Gu, J.; Yuan, Y.; Bian, J.; Zhu, J.; Liu, Z. Cadmium exposure triggers osteoporosis in duck via P2X7/PI3K/AKT-mediated osteoblast and osteoclast differentiation. Sci. Total Environ. 2021, 750, 141638. [Google Scholar] [CrossRef]
- Baloh, R.W. Laboratory diagnosis of increased lead absorption. Arch. Environ. Health 1974, 28, 198–208. [Google Scholar] [CrossRef]
- Cui, A.; Xiao, P.; Hu, B.; Ma, Y.; Fan, Z.; Wang, H.; Zhou, F.; Zhuang, Y. Blood Lead Level Is Negatively Associated With Bone Mineral Density in U.S. Children and Adolescents Aged 8-19 Years. Front. Endocrinol. 2022, 13, 928752. [Google Scholar] [CrossRef]
- Liu, J.; Tang, Y.; Chen, Y.; Zhang, X.; Xia, Y.; Geng, B. Association between blood manganese and bone mineral density in US adolescents. Environ. Sci. Pollut. Res. Int. 2022. [Google Scholar] [CrossRef] [PubMed]
- Rył, A.; Miazgowski, T.; Szylińska, A.; Turoń-Skrzypińska, A.; Jurewicz, A.; Bohatyrewicz, A.; Rotter, I. Bone Health in Aging Men: Does Zinc and Cuprum Level Matter? Biomolecules 2021, 11, 237. [Google Scholar] [CrossRef] [PubMed]
- Qu, Z.; Yang, F.; Yan, Y.; Hong, J.; Wang, W.; Li, S.; Jiang, G.; Yan, S. Relationship between Serum Nutritional Factors and Bone Mineral Density: A Mendelian Randomization Study. J. Clin. Endocrinol. Metab. 2021, 106, e2434–e2443. [Google Scholar] [CrossRef] [PubMed]
- Xue, G.; Liu, R. Association between dietary selenium intake and bone mineral density in the US general population. Ann. Transl. Med. 2022, 10, 869. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Yi, Q.; Wang, S.; Xia, Y.; Geng, B. Normal concentration range of blood mercury and bone mineral density: A cross-sectional study of National Health and Nutrition Examination Survey (NHANES) 2005–2010. Environ. Sci. Pollut. Res. Int. 2022, 29, 7743–7757. [Google Scholar] [CrossRef]
- Nguyen, N.T.; Nakahama, T.; Nguyen, C.H.; Tran, T.T.; Le, V.S.; Chu, H.H.; Kishimoto, T. Aryl hydrocarbon receptor antagonism and its role in rheumatoid arthritis. J. Exp. Pharmacol. 2015, 7, 29–35. [Google Scholar] [CrossRef]
- Izawa, T.; Arakaki, R.; Mori, H.; Tsunematsu, T.; Kudo, Y.; Tanaka, E.; Ishimaru, N. The Nuclear Receptor AhR Controls Bone Homeostasis by Regulating Osteoclast Differentiation via the RANK/c-Fos Signaling Axis. J. Immunol. 2016, 197, 4639–4650. [Google Scholar] [CrossRef]
- Lee, L.L.; Lee, J.S.; Waldman, S.D.; Casper, R.F.; Grynpas, M.D. Polycyclic aromatic hydrocarbons present in cigarette smoke cause bone loss in an ovariectomized rat model. Bone 2002, 30, 917–923. [Google Scholar] [CrossRef]
- Hsueh, Y.M.; Huang, Y.L.; Chen, H.H.; Shiue, H.S.; Lin, Y.C.; Hsieh, R.L. Alcohol Consumption Moderated the Association Between Levels of High Blood Lead or Total Urinary Arsenic and Bone Loss. Front. Endocrinol. 2021, 12, 782174. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).