Abstract
Aesthetic medicine has progressed from the early 2000s fascination with bio-stimulation to the current dominance of hyaluronic acid (HA) fillers, prized for immediate, predictable, and reversible volumizing effects. Recently, demand for more natural results, stronger emphasis on skin quality, and increased post-pandemic self-scrutiny have renewed interest in regenerative strategies, sometimes called the “second wave of bio-stimulation.” This trend highlights the need for clearer terminology and a cautious, evidence-based reading of proposed biological mechanisms. This narrative review proposes a framework in which bio-regeneration denotes a hypothesized, controlled induction of physiological processes, fibroblast activation, collagen and elastin synthesis, extracellular matrix remodeling, and immune modulation, potentially producing sustained improvements in dermal structure and function beyond simple filling. Among emerging technologies, polyethylene glycol diglycidyl ether (PEGDE) cross-linking is reported to create a stable, flexible HA scaffold with homogeneous tissue integration, favorable rheology, thermal stability, and a reduced inflammatory profile, supporting safer multimodal use with energy-based devices. The framework is illustrated with PEGDE-crosslinked HA combined with low-concentration calcium hydroxyapatite (CaHA), exemplified by a PEGDE-HA filler containing CaHA microspheres plus glycine and L-proline. These formulations aim to deliver immediate correction via HA and delayed stimulatory effects possibly driven by gradual CaHA exposure and macrophage-associated signaling. Available clinical, imaging, and histological observations, including prospective ultrasound and biopsy assessments, suggest progressive dermal thickening and predominant type I collagen expression, without pathological inflammation or granuloma formation. Although evidence remains preliminary and largely non-comparative, findings are compatible with controlled remodeling and resolving inflammation; however, the underlying mechanism and any ‘regenerative’ versus ‘reparative’ classification require controlled comparative studies.
1. Background
At the beginning of the 2000s, the field of aesthetic medicine experienced what could be described as the so-called “peak of bio-stimulation.” At that time, physicians and researchers were particularly interested in treatments that were not only volumizing or filling but also able to trigger endogenous regenerative processes within the skin. Techniques using autologous materials, such as platelet-rich plasma (PRP), and early collagen stimulators like poly-L-lactic acid (PLLA) or calcium hydroxyapatite (CaHA), were perceived as innovative because they addressed skin quality, not just contour.
Later, this trend gradually declined and, for many years, was overshadowed by the rapid development and popularity of hyaluronic acid (HA) fillers. HA fillers quickly became the gold standard on facial rejuvenation because of their safety profile, reversibility, and immediate, predictable results. This practical advantage, combined with strong commercial promotion and patient demand for fast “lunchtime procedures”, led to a shift away from bio-stimulatory concepts and toward primarily volumetric correction.
Over the past decade, aesthetic ideals have progressively evolved toward a more natural, balanced appearance. This shift has been influenced by multiple factors, including changing media narratives, the public visibility of actors and public figures favoring subtle and natural-looking results over overfilled appearances, and the emergence of new generational concepts emphasizing skin quality, authenticity, and healthy aging.
The COVID-19 pandemic represented a significant phase that accelerated this ongoing transformation. Increased digital communication and self-observation further heightened patient awareness of skin quality and facial harmony, while clinicians simultaneously recognized the limitations of purely volumetric approaches. Although effective in contour correction, these techniques may not fully address the progressive decline in skin quality associated with aging.
Together, these factors rekindled interest in regenerative and bio-stimulatory strategies, including advanced CaHA formulations, hybrid HA-dermal fillers, and energy-based devices used alone or in combination with injectable therapies. This renewed focus on physiological tissue improvement is often described as the “second wave of bio-stimulation,” a term commonly used to denote a stronger scientific understanding of aging biology and a growing body of objective clinical evidence.
“In the early 2000s, bio-stimulation reached its peak, but then declined as fillers took over. With COVID, however, bio-stimulation made a comeback—this time in a different way, supported by stronger scientific understanding of the aging process and more evidence.”
The practical considerations and treatment suggestions presented in this section reflect the clinical experience of the co-authors, selected to ensure broad geographic representation and long-standing involvement in aesthetic medicine. The authors have long-standing clinical experience with the majority of currently available biostimulators and dermal filler technologies, including PEGDE-HA and CaHA-based systems. In addition, they regularly participate as faculty and/or speakers in national and international scientific meetings and congresses across Europe, Latin America, and Asia, contributing to professional education and exchange of evidence-based clinical practices. This background is intended to support the scientific credibility and clinical relevance of the statements provided, while acknowledging that recommendations should be interpreted within the limits of a narrative review.
2. Definition of Bio-Stimulation/Bio-Revitalization/Bio-Regeneration
One of the key challenges in the field of aesthetic medicine is the lack of a unified definition. Different authors have used “bio-stimulation,” “bio-revitalization”, or “bio-regeneration,” interchangeably, which creates confusion. Considering the diversity of definitions and interpretations, as well as the limited understanding of the underlying biological mechanisms of many commonly used products, it is necessary to first establish a working definition that can be applied consistently.
Bio-stimulation should be understood as the ability of a product or treatment to trigger the body’s own physiological processes that contribute to restoration of skin structure and function.
It implies activation of fibroblasts and stimulation of collagen and extracellular matrix production, rather than simply acting as a filler.
Bio-revitalization refers to the restoration of skin vitality by supplying essential substances (such as hyaluronic acid and cofactors) that improve hydration, elasticity, and cellular function.
Bio-regeneration is a broader term, which may include bio-stimulation but also covers immunomodulation, angiogenesis, and metabolic improvements.
To advance the field, it is essential to establish a common language. The current understanding of bio-regeneration and bio-stimulation is no longer what it was two decades ago. Today, it refers to a physiologically oriented, evidence-based process rather than a purely marketing concept.
Proposed conceptual definition:
“Bio-regeneration in aesthetic medicine refers to the controlled and progressive induction of physiological processes, including fibroblast activation, collagen and elastin synthesis, and dermal matrix remodeling, which result in improvement of skin quality, elasticity, and structural integrity. Bio-regeneration encompasses these mechanisms and extends to the modulation of cellular and immune responses that are thought to contribute to long-term tissue health.”
3. Why Bio-Regeneration?
Bio-regeneration in aesthetic medicine is a conceptual, biologically oriented approach that aims to restore skin structure, function, and vitality by targeting the extracellular matrix (ECM) and its cellular components [1]. Central to this process is the stimulation of fibroblasts, which produce collagen, elastin, and other ECM proteins essential for maintaining skin elasticity, firmness, and youthful appearance [2,3]. Physiological bio-regeneration goes beyond simple volumization, supporting vascularization, ECM remodeling, and metabolic balance, thereby promoting gradual and controlled improvements in skin function rather than merely achieving superficial aesthetic changes. By engaging biomechanical receptors on fibroblastic membranes, bio-regenerative approaches may influence cellular behaviour, supporting collagen production, myofibroblast differentiation, and extracellular matrix remodeling, thereby contributing to improved skin structure and resilience [4].
Within this framework, physiological bio-regeneration is positioned as a strategy that directly targets the cellular and extracellular changes underlying skin aging. The approach addresses both intrinsic and extrinsic aging mechanisms. Physiological skin aging is gradual, characterized by slow collagen loss, decreased fibroblast activity, reduced cell turnover, and diminished hydration, leading to mild progressive loss of elasticity and volume. Pathological aging, accelerated by chronic inflammation, oxidative stress, hormonal changes, and metabolic imbalances, results in excessive collagen degradation, impaired wound healing, structural collapse, and increased skin fragility [5,6,7,8]. Bio-regeneration therapies are intended to harness the body’s natural regenerative potential to counteract these processes, enhance skin elasticity, hydration, and circulation, and supporting long-term tissue homeostasis [9].
Injectable treatments, such as bio-stimulating hyaluronic acid, calcium hydroxyapatite, polycaprolactone (PCL), poly-D,L-lactic acid (PDLLA), or poly-L-lactic acid (PLLA), provide direct cellular activation and ECM remodeling [1,10]. Device-based technologies including fractional lasers, microneedling, microneedling radiofrequency (RF), high-intensity focused ultrasound (HIFU), and infrared (IR) light, offer controlled stimulation that complements injectables [11,12]. Biologically active compounds, such as biomimetic peptides, growth factors, and platelet-rich plasma (PRP), can further enhance regenerative responses and optimize outcomes. These interventions aim to reduce wrinkles, restore lost volume, improve skin texture, and prevent aging-related changes, all while supporting physiological repair mechanisms [13,14,15].
Personalization is a cornerstone of effective bio-regeneration. Every patient is unique, and aging is multifactorial, involving intrinsic factors (genetics, natural fibroblast slowdown), extrinsic factors (UV exposure, pollution, lifestyle), and metabolic or inflammatory influences (chronic inflammation, glycation, hormonal shifts) [7]. Understanding each patient’s skin, aging pattern, and specific needs allows clinicians to select the optimal combination of therapies and techniques, ensuring safe, effective, and long-lasting results. True physiological bio-regeneration works with the body, not against it, leveraging bio-compatible stimulators, synergistic multi-modal approaches, and respect for the skin’s healing cycles to achieve both structural and aesthetic improvements.
In the past, the dominant “anti-aging” approach was aimed at masking visible signs of aging. The focus was primarily on correction, using surgical procedures or line-filling hyaluronic acids, often resulting in unnatural or short-lived effects [16]. The paradigm was defined by a “fight against aging.” Today, and looking forward, the emphasis is on healthy aging and regenerative aesthetics. The goal is not simply to conceal, but to support skin biology and tissue homeostasis. Modern tools including PEGylated hyaluronic acid, calcium hydroxyapatite, bio-stimulators, and energy-based technologies may allow for physiological regeneration, natural rejuvenation, and long-term improvements in tissue quality. This represents a shift from symptom management to biological stimulation, from restoring shape to restoring biology.
4. PEGDE as a Cross-Linking Agent
Among the agents currently applied for cross-linking in soft tissue fillers are Butanediol Diglycidyl Ether (BDDE), 1,8-Diepoxyoctane (DEO), Divinyl Sulfone (DVS), and Polyethylene Glycol Diglycidyl Ether (PEGDE). Manufacturers employ these agents differently, varying the degree of cross-linking, the ratio of cross-linker to HA, and the final HA concentration. These technical differences are decisive for the viscoelastic properties of each gel, which directly shape the clinical effect and durability of the aesthetic outcome [13,14]. Alongside anatomical knowledge, appropriate technique, and patient selection, choosing the right filler formulation remains a cornerstone of safety and complication prevention.
One of the more recent developments in filler technology is the application of PEGDE as a cross-linker. Experimental data indicate that PEGDE may improve both the performance and the safety profile of HA-based fillers. As polymers, PEGDE and HA together create a three-dimensional, scaffold-like matrix, which allows the filler to integrate homogeneously with the surrounding tissue [15,17,18]. PEGDE-crosslinked fillers show favorable rheological features such as viscosity, cohesiveness, and adaptability, supporting both natural integration and predictable handling [19,20,21].
In a preclinical comparative study, PEGDE-crosslinked HA hydrogels exhibited higher storage modulus, reduced swelling, and greater resistance to hyaluronidase-mediated degradation than BDDE-crosslinked HA, alongside more favorable fibroblast spreading, supporting the notion that PEGDE cross-linking may confer enhanced physicochemical stability and tissue-compatible cell–matrix interactions [22].
Commercial introduction of PEGDE as a cross-linking platform has therefore opened new possibilities in aesthetic procedures. Unlike traditional cross-linking chemistries, PEG produces a more uniform and flexible network, which enhances the mechanical resistance and thermal stability of the resulting gels [23,24,25,26,27]. Its well-documented biocompatibility is another key advantage, associated with reduced local inflammatory signalling and low immunogenicity. This is of particular relevance in high-risk or delicate anatomical areas. Moreover, these properties make PEGylated fillers well-suited for combined use with energy-based technologies such as IR, laser, or RF within the same session, without compromising safety or tissue integrity [23,24,25,26,27]. Two features stand out in particular: their thermodynamic stability and their ability to limit local inflammatory activation.
The ability of a filler to withstand heat is especially relevant when treatments involve devices that deliver energy to the skin. The thermal resistance of an HA gel depends largely on its cross-linking system. Studies have shown that PEGDE-crosslinked fillers are more resilient to heat than BDDE-based products [27]. This becomes critical when combining HA fillers with infrared, RF, or HIFU, as thermal degradation of the filler matrix can mimic mechanical stress and trigger endogenous hyaluronidase activity. In parallel, tissue damage induced by heat can provoke inflammation, further accelerating enzymatic degradation [28]. In this context, PEGylated fillers offer a margin of safety by retaining structural stability and minimizing the risk of inflammatory breakdown during multimodal procedures.
Safety is another domain where PEGDE appears to provide a tangible advantage. Any dermal filler, regardless of composition, may elicit local recruitment of neutrophils, monocytes, and macrophages, with downstream fibroblast activation. The inflammatory profile is shaped by each component of the formulation: HA, water content, and the cross-linker itself. While HA is a natural polysaccharide, its degradation fragments may display pro-inflammatory activity. Water can also influence osmotic balance locally, contributing to irritation. The cross-linker therefore plays a key role in the overall immunological response. PEGDE, in contrast to more traditional agents, has shown immunomodulatory properties: it appears to blunt proinflammatory pathways and may even exert local anti-inflammatory effects [26,29].
In vitro research supports this, showing that PEGDE-HA fillers reduce immune cell recruitment, reactive oxygen species formation, and expression of cytokines such as TNF and IL-8 under both resting and stimulated conditions [26,30,31]. This suggests a lower risk of immune-mediated adverse reactions, including granuloma formation or cellulitis, and may even promote an anti-inflammatory immune phenotype [30,31].
These findings have been reinforced in vivo: PEGylated fillers demonstrate diminished antigen presentation, reflected by lower counts of CD4+ and CD8+ T cells, B cells, and macrophages in the surrounding tissue. Both retrospective and prospective studies have confirmed this reduced immune activation [25,26]. Long-term clinical experience likewise supports their high safety, with no reports of granulomas, foreign body reactions, or related complications over three years of follow-up [31].
5. PEGDE-HA + CaHA and the Possible Mechanism of Bio-Regeneration
PEGDE-crosslinked hyaluronic acid combined with calcium hydroxyapatite, glycine, and L-proline is intended to combine immediate volumizing effects with a delayed bio-stimulatory response, consistent with a conceptual dual-phase mechanism of action that integrates both immediate volumizing and long-term bio-stimulatory effects.
Upon injection, the PEGDE-crosslinked hyaluronic acid matrix provides immediate tissue hydration, subtle volume restoration, and contour enhancement [32]. Its rheological and viscoelastic properties generate a transient mechanical lifting and support effect within the dermal and subdermal layers, contributing to early aesthetic improvement [20,24]. At the same time, the low-concentration CaHA microspheres (10–12 µm) remain evenly dispersed within the PEGDE-HA matrix, potentially forming a three-dimensional scaffold that may help maintain local tissue tension and may contribute to mechanical stimulation of fibroblasts through controlled micro-strain [33].
As the PEGDE-crosslinked hyaluronic acid gradually degrades, CaHA particles become progressively exposed to the surrounding tissue [33]. This delayed exposure is hypothesized to contribute to a controlled remodeling response, potentially involving collagen deposition and signals associated with vascular changes. Interactions between macrophages and small-sized CaHA particles have been proposed to promote the release of mediators such as transforming growth factor-β1 (TGF-β1) and platelet-derived growth factor (PDGF), which may, in turn, support gradual fibroblast activation and collagen synthesis [34]. This mechanistic hypothesis is biologically plausible in light of animal data showing that HA–hydroxyapatite composite fillers increase collagen type I and elastic fiber synthesis alongside upregulation of TGF-β/Smad-associated signaling (e.g., EGFR, Smad2/3, procollagen, elastin and fibrillin expression) in a nude mouse model [35].
Although the precise in vivo mechanism remains under investigation, available preclinical evidence is consistent with a dual-phase model. The initial phase is characterized by immediate, predominantly mechanical correction, likely supported by the intrinsic water-binding behavior of HA hydrogels, consistent with the rapid clinical improvement observed directly after injection [36]. The secondary phase may be associated with biologically mediated tissue remodeling. Morphological and ultrastructural analyses of human skin several months after injection of a PEGylated filler revealed uniform tissue integration without encapsulation or inflammatory reactions, along with fibroblast changes suggestive of increased extracellular matrix production [19]. Supporting these findings, in vitro studies have shown that PEG-cross-linked HA containing low concentrations of CaHA significantly upregulates collagen synthesis in human fibroblasts, indicating a potential bioactive role of CaHA beyond volumization [37]. Nevertheless, in the absence of controlled comparator arms (e.g., HA alone, CaHA alone, or sham treatment), the relative contribution of PEGDE cross-linking, CaHA content, and procedural factors cannot be determined with certainty.
PEGDE-HA + CaHA has been reported to address both dermal and subcutaneous tissue concerns, particularly in the correction of mid-face volume deficits [36]. The effects of PEGDE-HA + CaHA may not be limited to collagen production, but extend to vascularization, immune modulation, and broader tissue physiology [26,38], reflecting its potential role in bio-regeneration. The formulation itself also plays an important role in supporting these outcomes, and includes the amino acids proline and glycine, which act as osmolytes to help control hydrogel swelling after implantation. Proline, an osmoprotectant, mitigates water stress and supports turgor balance, while glycine contributes to cell volume regulation and stabilization of the hydrogel structure, preventing uncontrolled swelling [39]. Beyond their physicochemical role, proline and glycine also enhance tissue hydration and softness, functioning as humectant and emollient agents. Together, these properties support the stability of the hydrogel, particularly with respect to its viscoelastic behaviour and thermodynamic balance [39].
6. Clinical Evidence
The in vitro and in vivo data described above provide strong biological rationale supporting the hypothesis that the combination of hyaluronic acid and calcium hydroxyapatite may promote sustained improvements in skin quality, consistent with a dermal remodeling process rather than solely transient volumetric effects. Clinical experience further suggests durable, long-lasting outcomes [23]. In addition, a recent systematic review reports that combined HA–CaHA formulations demonstrate greater biostimulatory potential than hyaluronic acid alone, although substantial heterogeneity across study designs and experimental models limits direct comparison [40].
High-frequency ultrasound (HF-USG) studies have consistently demonstrated measurable increases in dermal thickness following treatment with PEGDE-crosslinked hyaluronic acid-based fillers, both with and without calcium hydroxylapatite, accompanied by reduction of the subepidermal low-echogenic band (SLEB). As these parameters are widely accepted non-invasive markers of intrinsic and extrinsic skin aging, their modulation after treatment has been interpreted as reflecting improvements in dermal hydration, collagen organization, and overall skin quality [41].
Clinical imaging and long-term observational studies further indicate that PEGDE-crosslinked fillers integrate homogeneously within the tissue, without evidence of irregular distribution, nodular accumulation, or encapsulation, and are associated with structural changes consistent with collagen-related dermal remodeling [23,36,37]. The incorporation of CaHA into PEGDE-HA formulations has been associated with enhanced collagen-related responses, suggesting that CaHA contributes to the durability and bio-regenerative profile of these combinations beyond the rheological properties of hyaluronic acid alone [37,42].
Histological analyses from clinical settings provide convergent evidence supporting a controlled and transient tissue response following PEGDE-HA + CaHA administration. Early post-injection evaluations, performed approximately 21 days after treatment, demonstrate moderately increased fibroblast activity, early collagen deposition, and mild inflammatory infiltration; findings considered consistent with a physiological tissue response rather than pathological inflammation [25].
Longitudinal immunohistochemical assessments indicate that markers of immune cell infiltration, including CD8, CD34, and CD68, decrease progressively over time, supporting resolution of the early immune response without progression to chronic inflammation [26,38]. Similar histological and clinical safety profiles have been reported in both healthy individuals and patients with autoimmune conditions, including Hashimoto’s thyroiditis, treated with PEGDE-HA–based fillers, with follow-up periods extending up to 150 days and no evidence of delayed inflammatory reactions or granuloma formation [26]. Notably, PEGylated hydrogels have been shown to protect calcium hydroxyapatite particles from immediate phagocytosis, thereby enabling their gradual release over time [37,43,44]. This observation is clinically relevant for practitioners, as the persistence of foreign materials such as soft tissue fillers can, in some cases, be associated with adverse immune reactions, including granuloma formation.
Prospective Clinical and Histological Study
A previously unpublished, prospective clinical and histological study, conducted in an independent laboratory, evaluated the biological and clinical effects of PEGDE-crosslinked hyaluronic acid combined with calcium hydroxyapatite. Fourteen healthy volunteers received a single subdermal injection and were followed for nine months. The study design included serial high-frequency ultrasound imaging, skin biopsies with histological and immunohistochemical analyses, electron microscopy, and longitudinal laboratory assessments.
Sustained clinical improvement and high patient satisfaction were observed throughout the follow-up period. High-frequency ultrasound demonstrated a progressive, time-dependent increase in dermal thickness, with mean values rising by approximately 7.8% at 30 days, 22.2% at 4 months, and 30.6% at 9 months (Figure 1). These structural changes occurred in the context of homogeneous tissue integration and in the absence of pathological neovascularization or granuloma formation.
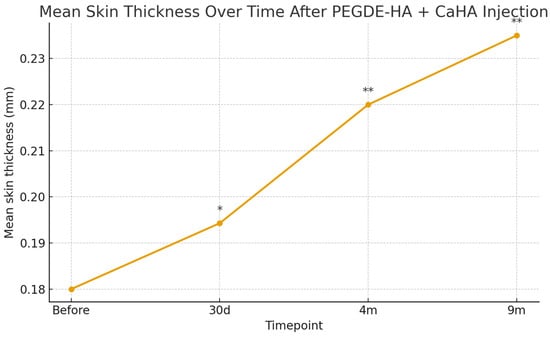
Figure 1.
Mean total skin thickness measured by high-frequency ultrasound at baseline and at 30 days, 4 months, and 9 months after injection of PEGDE-crosslinked HA with CaHA. A progressive, statistically significant increase in dermal thickness was observed over time (one-way ANOVA, p < 0.001), with the most pronounced improvement at 9 months; n = 14. Error bars indicate standard deviation; asterisks denote significant differences versus baseline (* p < 0.05; ** p < 0.01).
Histology and immunohistochemistry offered detailed mechanistic insights. IHC demonstrated a selective, time-dependent increase in type I collagen, with staining intensity becoming most pronounced at nine months (Table 1). In contrast, collagen types III, IV, and VII remained stable and unchanged throughout the study period. Type VII collagen, a key component of anchoring fibrils at the dermal–epidermal junction, ensures stable adhesion between the epidermis and dermis; its unchanged expression indicates that no injury-induced basement membrane repair was triggered [45,46]. Similarly, type IV collagen, essential for the structural integrity of basal membranes, showed consistent expression at all time points, confirming that no mechanical or inflammatory disruption occurred [47].

Table 1.
Semi-quantitative evaluation of type I collagen expression in skin biopsies.
The absence of detectable upregulation of type III collagen, commonly associated with early inflammatory or reparative wound-healing responses, suggests a remodeling pattern that does not prominently involve classical injury-driven repair cascades. However, this interpretation remains inferential and should not be construed as definitive evidence distinguishing regenerative from reparative mechanisms.
Electron microscopy corroborated these findings, revealing a predominance of mature type I collagen fibrils and activated fibroblasts in close proximity to hydroxyapatite particles, without foreign-body giant cells or macrophage-mediated reactions. Systemic laboratory parameters remained stable over time, with no signs of immune perturbation, and adverse events were limited to transient, mild injection-site reactions (Table 2).

Table 2.
Dynamics of cytokine and IgE levels.
Independent preclinical data provide an external line of support for the collagen I–dominant response observed in this human study. In a nude mouse model, crosslinked hyaluronic acid–hydroxyapatite composite fillers were associated with increased collagen type I expression and elastic fiber formation [35].
Taken together, these findings suggest that PEGDE-crosslinked HA combined with CaHA functions as a volumizing filler with additional biological activity that may support long-term dermal remodeling, while preserving basement membrane integrity and avoiding pronounced inflammatory responses. However, in the absence of controlled comparator arms (e.g., HA alone, CaHA alone, or sham-treated sites), causal attribution to PEGDE cross-linking, CaHA content, or their interaction cannot be established with certainty.
Several limitations should be acknowledged. The study was conducted in a small cohort (n = 14) without randomization or placebo controls, limiting statistical power and causal inference. Collagen expression was assessed using semi-quantitative immunohistochemistry, which may introduce inter-observer variability, and automated quantitative image analysis was not performed. Finally, longer-term follow-up is required to assess durability beyond nine months. Nonetheless, these data provide preliminary evidence of a consistent tissue response and support further investigation in larger, controlled studies with appropriate comparator groups and extended follow-up.
7. Positioning of PEGDE-HA + CaHA Among Other Options
The mechanism of action of currently marketed poly-L-lactic acid (PLLA), calcium hydroxyapatite CaHA or hyaluronic acid products carries important clinical implications, particularly with respect to injection technique, optimization of aesthetic outcomes, and minimization of adverse events [48]. When administered into the skin or subcutaneous tissue, these agents gradually increase tissue volume, correcting wrinkles and sagging [49,50]. Each treatment session stimulates collagen production, with the magnitude depending on both concentration and injected volume, which must be individually tailored. Repeated sessions progressively enhance extracellular matrix deposition, improving skin firmness and contour.
Unlike PLLA, CaHA provides immediate effects due to the carrier carboxymethyl cellulose gel [48]. The glycerine in the gel can induce transient edema lasting 24–72 h [49]. Thanks to its viscosity, density, and cohesiveness, CaHA allows effective tissue lifting and instant contouring. It is also suitable for supraperiosteal application and for restoring volume in areas of bone resorption [49,50].
Because the clinical effects of biostimulators may take weeks to manifest, it is crucial to allow sufficient time between sessions to permit the biological response. Additional treatments are typically scheduled at intervals of at least four weeks to avoid overcorrection [48]. Response time and degree of correction are influenced by patient-specific factors, including age, sex, skin quality, phototype, and lifestyle. Histological studies in animal models have shown that intradermal injections produce greater collagen formation than subdermal delivery; however, they are also associated with a higher incidence of palpable nodules and undulations, which can generally be managed conservatively with digital massage or saline/lidocaine infiltration [50,51].
From a practical standpoint, PLLA requires hydration several hours before injection, whereas CaHA can be administered directly or combined with lidocaine immediately prior to use. CaHA offers both immediate and long-lasting volumizing effects, though initial edema may occur due to the glycerine content. In contrast, PLLA’s immediate post-injection effect arises from the diluent volume and subsides within 24–48 h, with the true effect emerging gradually as neocollagenesis induces dermal thickening [52].
PEGDE-HA + CaHA represents a novel approach, being the first PEGDE-crosslinked hyaluronic acid filler to integrate CaHA within a single matrix. This dual mechanism differentiates it from conventional HA suspensions and standard CaHA-based biostimulators. The PEGDE-crosslinked HA component provides hydration, elasticity, and volumetric support, while also acting as a scaffold that ensures homogeneous dispersion of CaHA particles, thereby providing a more controlled stimulus for fibroblast activation and neocollagenesis.
Evidence increasingly indicates that particle morphology plays a decisive role in CaHA behaviour. Beyond particle size alone, factors such as shape, surface characteristics and aggregation significantly influence macrophage uptake, with smooth and spherical particles being less susceptible to phagocytosis than irregular ones [44]. Traditional CaHA fillers, typically containing particles in the range of 25–45 μm, are therefore understood to induce neocollagenesis primarily through mechanical fibroblast stimulation and the gradual degradation of relatively large particles. More recent perspectives expand this view. Turkevych proposed that, in addition to direct fibroblast activation, CaHA may also interact with adipocytes and fibroblasts within subcutaneous white adipose tissue (sWAT), potentially modulating local biomechanics and broadening its bio-regenerative impact beyond classical collagen induction [43].
Within this evolving framework, the PEGDE-crosslinked HA–CaHA matrix in this formulation, incorporating smaller CaHA particles of 8 to 12 μm uniformly dispersed within the dense HA network, may contribute to controlled particle exposure and moderated release kinetics. This architecture may limit immediate contact with phagocytic cells and reduce rapid clearance, allowing the particles to remain bioactive over an extended period and sustain a regenerative stimulus. The prolonged release profile and durable bio-stimulatory effect were demonstrated in the 2023 study by Zerbianti et al. [36].
CaHA concentration also appears to play a crucial role, as very low concentrations (1–3%) may influence the formation and secretion of exosomes from surrounding cells, contributing to long-term improvements in skin appearance [43,53]. Furthermore, the combination of hydroxyapatite and hyaluronic acid exerts a synergistic effect: HA enhances extracellular matrix hydration and facilitates uniform dispersion of CaHA particles within the tissue. This may promote consistent tissue integration and could explain the absence of local accumulations, granulomas, or fibrosis, supporting both the safety and efficacy profile of PEGDE-HA + CaHA.
Histological assessments of PEGDE-HA + CaHA confirmed the absence of inflammatory infiltrates, foreign-body giant cells, and macrophage-driven reactions. Systemic laboratory analyses further demonstrated stable cytokine profiles and preserved IFN-γ production, findings that argue against granulomatous or immunosuppressive responses [26,33]. Clinically, this mechanistic advantage translates into a more predictable safety profile, with a markedly reduced risk of nodules, fibrosis, and delayed adverse events.
8. Practical Guidance and Clinical Algorithm
Based on expert opinions, PEGDE-HA + CaHA is particularly recommended in:
8.1. General Aging & Skin Quality
- Patients with early to moderate signs of skin aging, where dermal quality improvement is a primary goal;
- Patients seeking improvement in firmness, elasticity, hydration, and skin texture, in addition to subtle contouring;
- Patients seeking preventive treatment to counteract accelerated aging.
8.2. Corrective & Reparative Indications
- Cases requiring mild volumization combined with bio-regeneration, especially in delicate facial zones;
- Patients with skin imperfections, including acne scars, stretch marks, and post-surgical scars.
8.3. Special Clinical Situations
- Patients with compromised tissue quality (after illness or oncological treatment), where regenerative support is valuable;
- Patients with autoimmune conditions (e.g., Hashimoto’s thyroiditis), provided that eligibility and safety are carefully assessed by the treating physician [26].
- Patients experiencing significant weight loss, including those treated with GLP-1 receptor agonists, where loss of subcutaneous tissue and skin quality deterioration require supportive regenerative therapy;
- Menopausal patients presenting with skin dullness, dryness, and reduced vitality due to hormonal changes, where bio-regeneration can help restore hydration and dermal balance.
8.4. Treatment Strategies
- Situations where multi-modal therapy is indicated, combining injectables with energy-based devices (infrared, fractional laser, RF) within the same session to enhance synergistic regenerative effects;
- Cases where gentle stimulation of fibroblasts is desired to support long-term tissue homeostasis without overcorrection.
- When repeated sessions are required for biostimulatory products, consensus recommendations commonly use multi-week intervals between sessions to allow the biological response to evolve, supporting a staged, lower-risk approach.
Other options, such as pure HA fillers or CaHA suspensions, may be considered when the therapeutic goal is limited to volumetric correction or to strong bio-stimulation without the need for hydration and elasticity. PEGDE-HA + CaHA thus fills a unique therapeutic niche by combining regenerative and corrective effects in a single product.
9. Conclusions
Physiological bio-regeneration represents an evolving paradigm in aesthetic medicine, shifting the focus from purely volumetric correction toward sustained improvement in skin structure, function, and quality through the controlled induction of endogenous processes. Within this conceptual framework, PEGDE-crosslinked hyaluronic acid combined with low-concentration calcium hydroxyapatite may represent a coherent technological and biological strategy, providing immediate structural support and hydration followed by a delayed, low-inflammatory stimulatory effect potentially associated with fibroblast activation and selective type I collagen enhancement. Available clinical, imaging, and histological observations, although preliminary and largely derived from small, non-comparative cohorts, consistently indicate homogeneous tissue integration, progressive increases in dermal thickness, and the absence of pathological inflammation or granuloma formation: findings that are compatible with a remodeling-oriented response rather than a classical injury-driven wound-healing process. Nevertheless, the methodological limitations of the current evidence base underscore the need for robust controlled trials with quantitative endpoints and longer follow-up to confirm underlying mechanisms, durability, and optimal clinical indications, ensuring that the bio-regenerative concept remains grounded in physiology and evidence rather than terminology alone.
Author Contributions
Conceptualization, M.C., R.F.d.C.I., F.M., I.R.K., R.A.S.V., D.P.F., S.Z. and D.P.; methodology, M.C. and F.M.; formal analysis, M.C. and F.M.; data curation, M.C. and F.M.; writing—original draft preparation, M.C.; writing—review & editing, M.C., R.F.d.C.I., F.M., I.R.K., R.A.S.V., D.P.F., S.Z. and D.P.; supervision, M.C. and F.M.; project administration, M.C. and F.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Local Ethics Committee of Clinic of Professor Yutskovskaya (date of approval: 29 February 2024).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest related to this work. The authors have no financial or personal relationships that could inappropriately influence or bias the content of this publication. M.C. is affiliated with Agora clinical educational center, Milan, Italy; R.F.d.C.I. with Fercasy Clinica Medicina Estética, Madrid, Spain; F.M. with Private Practice, Casa di Cura Villa Mafalda, Rome, Italy; I.R.K. with BK Aesthetic Medicine, Asturias, Spain; R.A.S.V. with Private Practice “Dr. Sandoval Skin Care Clinic”, Bogotá, Colombia; D.P.F. with Clínica Forjaz, Lisboa, Portugal; S.Z. with Clinica Doux, Sao Paulo, Brazil; and D.P. with The Demis Clinic, Bangkok, Thailand. The institutions had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Barbosa, A.D.P. Regeneration in Aesthetic Medicine: Mechanisms, Evidence, and Clinical Boundaries. J. Cosmet. Dermatol. 2026, 25, e70669. [Google Scholar] [CrossRef]
- Zorina, A.; Zorin, V.; Isaev, A.; Kudlay, D.; Vasileva, M.; Kopnin, P. Dermal Fibroblasts as the Main Target for Skin Anti-Age Correction Using a Combination of Regenerative Medicine Methods. Curr. Issues Mol. Biol. 2023, 45, 3829–3847. [Google Scholar] [CrossRef]
- Boraldi, F.; Lofaro, F.D.; Bonacorsi, S.; Mazzilli, A.; Garcia-Fernandez, M.; Quaglino, D. The Role of Fibroblasts in Skin Homeostasis and Repair. Biomedicines 2024, 12, 1586. [Google Scholar] [CrossRef]
- Plikus, M.V.; Wang, X.; Sinha, S.; Forte, E.; Thompson, S.M.; Herzog, E.L.; Driskell, R.R.; Rosenthal, N.; Biernaskie, J.; Horsley, V. Fibroblasts: Origins, definitions, and functions in health and disease. Cell 2021, 184, 3852–3872. [Google Scholar] [CrossRef]
- Dorosz, A.; Skoczeń, A.; Kulesza, M.; Wawrzynów, W.; Jakubowska, M.M.; Kruk, A.; Rutecka, N.; Miłoś, M.; Kuśnierz-Gibała, A.; Kaczmarek, B. The Impact of Environmental Factors on Skin and Tissue Ageing: Mechanisms, Effects, and Preventive Strategies. J. Educ. Health Sport 2025, 79, 58282. [Google Scholar] [CrossRef]
- Chambers, E.S.; Vukmanovic-Stejic, M. Skin barrier immunity and ageing. Immunology 2020, 160, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, M.; Khan, A.; Gupta, M. Skin Ageing: Pathophysiology and Current Market Treatment Approaches. Curr. Aging Sci. 2020, 13, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Challa, V.; Prajapati, S.K.; Gangani, S.; Yadav, D.; Lekkala, L.; Jain, S.; Yadav, H. Microbiome–Aging–Wrinkles Axis of Skin: Molecular Insights and Microbial Interventions. Int. J. Mol. Sci. 2025, 26, 10022. [Google Scholar] [CrossRef]
- Dutra Alves, N.S.; Reigado, G.R.; Santos, M.; Caldeira, I.D.S.; Hernandes, H.D.S.; Freitas-Marchi, B.L.; Zhivov, E.; Chambergo, F.S.; Nunes, V.A. Advances in regenerative medicine-based approaches for skin regeneration and rejuvenation. Front. Bioeng. Biotechnol. 2025, 13, 1527854. [Google Scholar] [CrossRef]
- Lee, K.W.A.; Kim, H.; Song, J.K.; Sydorchuk, O.; Fai, W.K.; Rosellini, I.; Kim, H.; Lau, K.H.; Gold, M.H.; Yi, K. Injectable Biostimulator in Adipose Tissue: An Update and Literature Review. Sci. Pharm. 2025, 93, 62. [Google Scholar] [CrossRef]
- Gold, M.; Biesman, B.; Cohen, J.L.; Goldberg, D.J.; Guenin, S.; Schlesinger, T.; Taher, Z. Real-World Experience Using a Multi-Modality System using Intense Pulsed Light, Radiofrequency Microneedling, High-Intensity Focused Ultrasound, or Thermal Radiofrequency, 808, HIFU for Skin Rejuvenation Treatment. J. Cosmet. Dermatol. 2024, 23, 1–11. [Google Scholar] [CrossRef]
- Jia, X.; Feng, Y. Energy-Based Skin Rejuvenation: A Review of Mechanisms and Thermal Effects. J. Cosmet. Dermatol. 2025, 24, e16657. [Google Scholar] [CrossRef]
- Khunmanee, S.; Jeong, Y.; Park, H. Crosslinking method of hyaluronic-based hydrogel for biomedical applications. J. Tissue Eng. 2017, 8, 2041731417726464. [Google Scholar] [CrossRef]
- Pierre, S.; Liew, S.; Bernardin, A. Basics of Dermal Filler Rheology. Dermatol. Surg. 2015, 41, S120–S126. [Google Scholar] [CrossRef]
- Leach, J.B.; Schmidt, C.E. Characterization of protein release from photocrosslinkable hyaluronic acid-polyethylene glycol hydrogel tissue engineering scaffolds. Biomaterials 2005, 26, 125–135. [Google Scholar] [CrossRef]
- Peng, C.X.; Xv, W.; Ao, Y.-J. A Review: Causes, Consequences, and Management Strategies of Facial Overfilling. Clin. Cosmet. Investig. Dermatol. 2025, 18, 1857–1864. [Google Scholar] [CrossRef] [PubMed]
- Aurand, E.R. The Characterization of Hyaluronic Acid and Polyethylene Glycol Hydrogels for Neural Tissue Engineering. Doctoral Dissertation, University of Colorado Denver, Denver, CO, USA, 2014. [Google Scholar]
- Hassan, W.; Dong, Y.; Wang, W. Encapsulation and 3D culture of human adipose-derived stem cells in an in-situ crosslinked hybrid hydrogel composed of PEG-based hyperbranched copolymer and hyaluronic acid. Stem Cell Res. Ther. 2013, 4, 32. [Google Scholar] [CrossRef] [PubMed]
- Zerbinati, N.; D’Este, E.; Farina, A.; Rauso, R.; Cherubino, M.; Calligaro, A. Morphological evidences following pegylated filler treatment in human skin. J. Biol. Regul. Homeost. Agents 2017, 31, 79–85. [Google Scholar]
- Zerbinati, N.; Sommatis, S.; Maccario, C.; Capillo, M.C.; Grimaldi, G.; Alonci, G.; Protasoni, M.; Rauso, R.; Mocchi, R. Toward Physicochemical and Rheological Characterization of Different Injectable Hyaluronic Acid Dermal Fillers Cross-Linked with Polyethylene Glycol Diglycidyl Ether. Polymers 2021, 13, 948. [Google Scholar] [CrossRef] [PubMed]
- Zerbinati, N.; Esposito, C.; Cipolla, G.; Calligaro, A.; Monticelli, D.; Martina, V.; Golubovic, M.; Binic, I.; Sigova, J.; Gallo, A.L.; et al. Chemical and mechanical characterization of hyaluronic acid hydrogel cross-linked with polyethylen glycol and its use in dermatology. Dermatol. Ther. 2020, 33, e13747. [Google Scholar] [CrossRef]
- Ho-Yong, L.; Seol-Ha, J.; Jae-Uk, B.; Ju-Ha, S.; Hyoun-Ee, K. Hyaluronic acid hydrogels cross-linked by polyethylene glycol diglycidyl ether (PEGDE) for long-lasting dermal filler applications. In Proceedings of the 10th World Biomaterials Congress, Montréal, QC, Canada, 17–22 May 2016; Frontiers: Lausanne, Switzerland, 2016; Volume 4. [Google Scholar] [CrossRef]
- Rauso, R.; Nicoletti, G.F.; Bove, P.; Rauso, G.M.; Fragola, R.; Lo Giudice, G.; Zerbinati, N. Clinical Experience with PEGylated Hyaluronic Acid Fillers: A 3-year Retrospective Study. Open Access Maced. J. Med. Sci. 2021, 9, 1168–1173. [Google Scholar] [CrossRef]
- Zerbinati, N.; Capillo, M.C.; Sommatis, S.; Maccario, C.; Alonci, G.; Rauso, R.; Galadari, H.; Guida, S.; Mocchi, R. Rheological Investigation as Tool to Assess Physicochemical Stability of a Hyaluronic Acid Dermal Filler Cross-Linked with Polyethylene Glycol Diglycidyl Ether and Containing Calcium Hydroxyapatite, Glycine and L-Proline. Gels 2022, 8, 264. [Google Scholar] [CrossRef]
- Kubik, P.; Jankau, J.; Rauso, R.; Galadari, H.; Protasoni, M.; Gruszczyński, W.; Grzanka, D.; Smolińska, M.; Antosik, P.; Piesiaków, M.-L.; et al. HA PEGylated Filler in Association with an Infrared Energy Device for the Treatment of Facial Skin Aging: 150 Day Follow-Up Data Report. Pharmaceuticals 2022, 15, 1355. [Google Scholar] [CrossRef]
- Kubik, P.; Gallo, D.; Tanda, M.L.; Jankau, J.; Rauso, R.; Gruszczyński, W.; Pawłowska, A.; Chrapczyński, P.; Malinowski, M.; Grzanka, D.; et al. Evaluation of the Safety of Neauvia Stimulate Injectable Product in Patients with Autoimmune Thyroid Diseases Based on Histopathological Examinations and Retrospective Analysis of Medical Records. Gels 2023, 9, 440. [Google Scholar] [CrossRef]
- Kubik, P.; Gruszczyński, W. Heat Influence on Different Hyaluronic Acid Fillers. J. Appl. Cosmetol. 2023, 41, 20–27. [Google Scholar] [CrossRef]
- Mayer, R.L. Hyaluronidase and inflammation of the skin. Ann. N. Y. Acad. Sci. 1950, 52, 1041–1045. [Google Scholar] [CrossRef]
- Decates, T.; Kadouch, J.; Velthuis, P.; Rustemeyer, T. Immediate nor Delayed Type Hypersensitivity Plays a Role in Late Inflammatory Reactions After Hyaluronic Acid Filler Injections. Clin. Cosmet. Investig. Dermatol. 2021, 14, 581–589. [Google Scholar] [CrossRef]
- Marino, F.; Cosentino, M.; Legnaro, M.; Luini, A.; Sigova, J.; Mocchi, R.; Lotti, T.; Zerbinati, N. Immune profile of hyaluronic acid hydrogel polyethylene glycol crosslinked: An in vitro evaluation in human polymorphonuclear leukocytes. Dermatol. Ther. 2020, 33, e13388. [Google Scholar] [CrossRef]
- Jeong, C.H.; Kim, D.H.; Yune, J.H.; Kwon, H.C.; Shin, D.-M.; Sohn, H.; Lee, K.H.; Choi, B.; Kim, E.S.; Kang, J.H.; et al. In vitro toxicity assessment of crosslinking agents used in hyaluronic acid dermal filler. Toxicol. Vitr. 2021, 70, 105034. [Google Scholar] [CrossRef] [PubMed]
- Zerbinati, N.; Carugno, A.; Guida, S.; Mocchi, R.; Sommatis, S.; Cipolla, G.; Rauso, R.; Galadari, H.; Fratton, Z.; Errichetti, E.; et al. Assessment of Safety and Tissue Integration of PEGDE-Based Hyaluronic Acid Filler for Severe Nasolabial Folds: A Prospective Observational Study with Biophysical and Ultrasound Evaluation. Cosmetics 2025, 12, 275. [Google Scholar] [CrossRef]
- Zerbinati, N.; Lotti, T.; Monticelli, D.; Rauso, R.; González-Isaza, P.; D’Este, E.; Calligaro, A.; Sommatis, S.; Maccario, C.; Mocchi, R.; et al. In Vitro Evaluation of the Biosafety of Hyaluronic Acid PEG Cross-Linked with Micromolecules of Calcium Hydroxyapatite in Low Concentration. Open Access Maced. J. Med. Sci. 2018, 6, 15–19. [Google Scholar] [CrossRef]
- Yutskovskaya, Y.A.; Kogan, E.A. Improved Neocollagenesis and Skin Mechanical Properties After Injection of Diluted Calcium Hydroxylapatite in the Neck and Décolletage:A Pilot Study. J. Drugs Dermatol. JDD 2017, 16, 68–74. [Google Scholar]
- Fan, Y.; Choi, T.-H.; Chung, J.-H.; Jeon, Y.-K.; Kim, S. Hyaluronic acid-cross-linked filler stimulates collagen type 1 and elastic fiber synthesis in skin through the TGF-β/Smad signaling pathway in a nude mouse model. J. Plast. Reconstr. Aesthet. Surg. 2019, 72, 1355–1362. [Google Scholar] [CrossRef]
- Zerbinati, N.; D’Este, E.; De Silvestri, A.; Zullino, M.; Rabbiosi, G.; Guida, S.; Kubik, P.; Stabile, G.; Mocchi, R.; Barlusconi, C.; et al. Efficacy of Pegylated Hyaluronic Acid Filler Enriched with Calcium Hydroxyapatite: A 24-Week Post-Market, Observational, Prospective, Open-Label, Single-Center Study. J. Funct. Biomater. 2023, 14, 345. [Google Scholar] [CrossRef] [PubMed]
- Zerbinati, N.; Rauso, R.; Gonzalez, P.; Cherubino, M.; D’Este, E.; Calligaro, A.; Sommatis, S.; Maccario, C.; Mocchi, R. In vitro evaluation of collagen production on human fibroblasts treated with hyaluronic acid peg cross-linked with micromolecules of calcium hydroxyapatite in low concentration. J. Biol. Regul. Homeost. Agents 2017, 31, 87–90. [Google Scholar]
- Kubik, P.; Gruszczyński, W. Safety of PEGylated Hyaluronic Acid Filler for the Treatment of Facial Skin Aging: Case Report. Clin. Case Rep. Int. 2024, 8, 1679. [Google Scholar]
- Martina, V.; Gallo, A.; Tarantino, E.; Esposito, C.; Zerbinati, U.; Mocchi, R.; Monticelli, D.; Lotti, T.; Tirant, M.; Thuong, N.V.; et al. Viscoelastic properties and thermodynamic balance improvement of a hyaluronic acid hydrogel enriched with proline and glycyne. J. Biol. Regul. Homeost. Agents 2019, 33, 1955–1959. [Google Scholar] [PubMed]
- Meçani, R.; Amiri, M.; Kadouch, J.; Sajic, D.; Lin, F.; Cheung, J.; Barrera, D.; Haroon, O.; Sil-Zavaleta, S.; Chao, Y.; et al. Combined and Hybrid Treatments of Hyaluronic Acid (HA) and Calcium Hydroxylapatite (CaHA): A Systematic Review of Mechanisms of Action, Aesthetic Effectiveness, Satisfaction, and Safety Profile. Aesth. Plast. Surg. 2025, 49, 5292–5313. [Google Scholar] [CrossRef]
- Tafur, M.G. A Comparative Case Study on Echographic Behaviour of Selected Soft Tissue Fillers. J. Appl. Cosmetol. 2025, 43, 16–24. [Google Scholar] [CrossRef]
- Zerbinati, N. Pegylated hyaluronic acid filler enriched with calcium hydroxyapatite treatment of human skin: Collagen renewal demonstrated through morphometric computerized analysis. J. Biol. Regul. Homeost. Agents 2019, 33, 1967–1971. [Google Scholar] [CrossRef]
- Turkevych, A.; Turkevych, D. Influence of calcium hydroxylapatite on the soft tissues—A critical viewpoint. J. Appl. Cosmetol. 2022, 40, 19–27. [Google Scholar]
- Lebre, F.; Sridharan, R.; Sawkins, M.J.; Kelly, D.J.; O’Brien, F.J.; Lavelle, E.C. The shape and size of hydroxyapatite particles dictate inflammatory responses following implantation. Sci. Rep. 2017, 7, 2922. [Google Scholar] [CrossRef] [PubMed]
- Conradt, G.; Hausser, I.; Nyström, A. Epidermal or Dermal Collagen VII Is Sufficient for Skin Integrity: Insights to Anchoring Fibril Homeostasis. J. Investig. Dermatol. 2024, 144, 1301–1310.e7. [Google Scholar] [CrossRef]
- Burgeson, R.E. Type VII collagen, anchoring fibrils, and epidermolysis bullosa. J. Investig. Dermatol. 1993, 101, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Pöschl, E.; Schlötzer-Schrehardt, U.; Brachvogel, B.; Saito, K.; Ninomiya, Y.; Mayer, U. Collagen IV is essential for basement membrane stability but dispensable for initiation of its assembly during early development. Development 2004, 131, 1619–1628. [Google Scholar] [CrossRef] [PubMed]
- Lacombe, V. Sculptra: A Stimulatory Filler. Facial Plast. Surg. 2009, 25, 095–099. [Google Scholar] [CrossRef]
- Bauer, U.; Graivier, M.H. Optimizing injectable poly-L-lactic acid administration for soft tissue augmentation: The rationale for three treatment sessions. Can. J. Plast. Surg. 2011, 19, e22–e27. [Google Scholar] [CrossRef]
- Hevia, O. A Retrospective Review of Calcium Hydroxylapatite for Correction of Volume Loss in the Infraorbital Region. Dermatol. Surg. 2009, 35, 1487. [Google Scholar] [CrossRef]
- Breithaupt, A.; Fitzgerald, R. Collagen Stimulators. Facial Plast. Surg. Clin. N. Am. 2015, 23, 459–469. [Google Scholar] [CrossRef]
- Cunha, M.G.D.; Engracia, M.; Souza, L.G.D.; Machado Filho, C.D. Bioestimuladores e seus mecanismos de ação. Surg. Cosmet. Dermatol. 2020, 12, 109–117. [Google Scholar] [CrossRef]
- De, A.; Chakraborty, D.; Agarwal, I.; Sarda, A. Present and Future Use of Exosomes in Dermatology. Indian. J. Dermatol. 2024, 69, 461–470. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.