Abstract
Collagen is an essential ingredient in dietary supplements for its anti-ageing benefits, and l-cystine-based supplementation has garnered interest for its ability to improve skin condition. Our study aimed to evaluate the effects of oral supplementation combining l-cystine and fish collagen peptides at two different dosages on mature (55–65 years) and young (18–30 years) skin types. Two randomised, three-arm, double-blind, placebo-controlled trials were conducted. A total of 198 Asian women were allocated into equal groups (5.5 or 11 g of active supplement or placebo daily for 12 weeks). In the Young panel, global aesthetic improvement, cutaneous pigmentation homogeneity, texture, redness, acne scars, and UV sensitivity were investigated. In the Mature panel, complexion radiance, crow’s feet, cutaneous thickness, moisturising, pigmentation homogeneity, texture, and skin colour were investigated. In mature women, the most notable improvements after 12 weeks were hydration, cutaneous thickness, and wrinkles. In younger women, improvements in cutaneous texture, reduced skin redness, and increased UV photoprotection were the most notable effects. Oral administration of the dietary supplement containing l-cystine and collagen peptides was perfectly well tolerated. In addition to the already known benefits for mature skin, this supplementation could also benefit younger people with skin imperfections.
1. Introduction
Collagens constitute a large family of proteins with a simple fundamental repeated Gly–X–Y amino acid sequence, which is the defining signature of these molecules. X and Y are frequently proline (Pro) and hydroxyproline (Hyp) residues, respectively. The sequence of these base units forms a polypeptide chain wound in an α chain. Several chains can be twisted around each other into a triple helix to form a rigid structure. Twenty-nine types of collagens have been identified thus far. They differ according to the composition of their α chains and their organisation as homo- or hetero-trimers. Type I collagen, synthesised by fibroblasts, is the most common in the dermis. The main function of collagens is to provide structural support to the extracellular matrix (ECM) of connective tissues such as skin, bone joints, and tendons [,,]. To overcome the low water solubility of native collagen proteins, which limits their potential utilisation in liquid applications, a process of controlled hydrolysis that leads to low-molecular-weight, smaller peptides has been proposed. As a result, collagen hydrolysates are now commonly used by the cosmetic industry, owing to their skin benefits and favourable biocompatibility and immunogenicity profiles []. In particular, oral supplementation with collagen peptides has proven to be very useful for skin anti-ageing treatment. Because ageing has many physiological and functional consequences that originate at the molecular level, type I collagen with its very long half-life (approximately 15 years) undergoes progressive modifications over time. Therefore, supplying the body with peptides that promote collagen synthesis may be a logical solution.
l-cystine is the amino acid dimer formed when a pair of l-cysteine molecules are joined by a disulfide bond through oxidation. l-cystine is found in most high-protein foods such as eggs, meat, dairy products, and whole grains. In humans, this molecule is particularly abundant in skin, hair, and nails. At the cellular level, it is the main source of l-cysteine, which is usually the limiting amino acid for the synthesis of the powerful antioxidant glutathione. l-cystine is highly available as a dietary supplement, is quickly absorbed, and provides enough cysteine to the cells for glutathione production. Interestingly, the mechanisms controlling the eumelanin/pheomelanin ratio are dependent on l-cysteine, glutathione, and tyrosinase-related protein [,,]. In addition, since reactive oxygen species are known to activate melanogenesis, antioxidants exhibit skin lightening activity through their quenching effects []. Recently, novel interventions based on cystine- or cysteine-based oral supplementation have been garnering interest for their ability to improve skin, hair, and nail conditions [,,].
In recent years, functional foods that are claimed to have skin benefits have greatly increased. In Asia, hydrolysed collagen is one of the foremost health ingredients. Apart from its effects on hair, bone, and joint health, collagen is primarily recognised as being useful in anti-ageing skin treatment. Since having a smooth, light, and homogeneous complexion is highly desired in most Asian countries by women and men of all ages, we theorised that combining collagen hydrolysates with l-cystine in one dietary supplement could provide a two-in-one solution for both young individuals with skin imperfections, who wish to obtain a better skin quality or reduce skin roughness, and for older individuals who desire to fight signs of skin ageing.
To date, no randomised placebo-controlled study has evaluated the response to the combination of collagen hydrolysates and l-cystine. We therefore conducted two clinical trials aiming at evaluating the effect on skin condition of such a combination, called Naticol®-CySkin®, which was taken by the oral route at two different dosages. The first clinical study was conducted on mature women whose skin presented signs of ageing, such as wrinkles and a dull complexion. The second clinical trial was conducted on younger women whose skin presented moderate acne. Collagen hydrolysates, registered under the Naticol® brand name, were obtained from the native collagen present in fish skins by denaturation and specific enzymatic cleavage, while l-cystine was obtained industrially by the hydrolysis of natural keratin from poultry feathers. In this paper, we report the results from these two trials, subsequently referred to as the “Mature skin study” and “Young skin study”. In the trial with younger women, acne-prone skin was used as a model of imperfections.
2. Materials and Methods
2.1. Study Design and Ethical Considerations
These two randomised, double-blind, placebo-controlled, three-arm, parallel-group studies were conducted in compliance with the ethical principles of the Declaration of Helsinki. The Good Clinical Practices and Personal Data Protection Act B.E. 2562 (2019) of Thailand was observed. Approvals of the protocols, CRFs, and informed consent documents were granted by the Human Research Ethics Committee of Thammasat University (Faculty of Medicine). The 268/2021 and 255/2021 certificates of approval were granted to the Mature skin study (project MTU-EC-IM-5-180/64) and the Young skin study (project MTU-EC-IM-5-179/64), respectively. Because Thailand currently has no legal or regulatory requirement for clinical trial registration in a public registry, the two trials were not registered in the Thai Clinical Trials Registry. All participants gave their signed informed consent, as well as authorised the use of their photographs for publication, prior to the commencement of any study-related procedures. Both studies had a duration of 12 weeks and included 3 visits each (Figure 1).
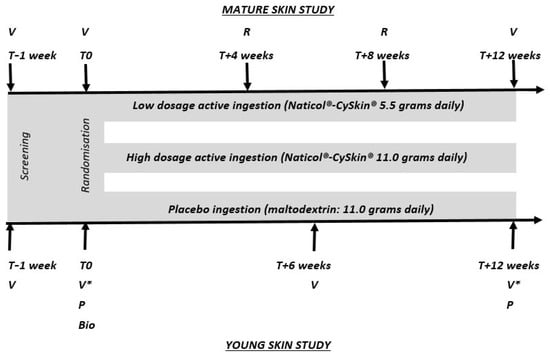
Figure 1.
Flowcharts of the two studies evaluating the in vivo effects of the Naticol-CySkin supplement administered orally at dosages of 5.5 or 11 g per day in young skin presenting imperfections and in mature skin panels. V: visit at the testing centre; R: reminder calls to the participants; P: pregnancy test; Bio: bioavailability study on 5 participants; *: including an additional visit at T + 24 h for the determination of the Minimum Erythema Dose.
2.2. Subjects
The two studies included healthy Asian females having skin phototypes III and IV, of any skin type; not having ingested oral supplements for at least one month and willing to abstain from oral supplements (excepted the tested one) during the study period; and agreeing to not modify their nutrition habits during the trial period. These women were eligible for the Mature skin study if they were 55–65 years old, had wrinkles and/or fine crow’s feet with a grade ≥ 2 according to the Skin Ageing Atlas, Vol 2. Asian type [], and presented a dull complexion. To be included in the Young skin study, the women had to be 18–30 years of age with mixed acne types, i.e., inflammatory and retentional acne lesions, acne scars and cutaneous redness, and rough skin. A negative urine pregnancy test was required to be eligible for the Young skin study.
The main exclusion criteria, common to both studies, were a diagnosed or highly probable allergy to fish and/or seafood; liver diseases such as cirrhosis and hepatitis; eating disorders; excessive alcohol consumption; having followed a particular diet in the 2 months before the beginning of the study (hypocaloric, gluten-free, hyperproteic); or a body mass index (BMI) higher than 27.0 kg.m−2.
For the Mature skin study, additional exclusion criteria were any hormonal treatments, aesthetic and/or surgical procedures, utilisation of cosmetic products, or ingestion of dietary supplements that could affect the natural ageing or the colour of the skin. For the Young skin study, additional exclusion criteria were any hormonal treatments, physical (phototherapy, laser, etc.) or chemical (peeling) interventions, or local benzoyl-peroxide-based, local retinoid-based, or oral (retinoid, cimetidine, zinc gluconate, antibiotics, etc.) acne treatments. Women taking medication with a photosensitivity potential or having a history of abnormal response to the sun and nursing women were also excluded from the study.
At the baseline visit, all participants received oral and written instructions to follow a list of restrictions and proscriptions dealing with topical or systemic medicinal, cosmetic, or aesthetic treatments that could interfere with the trial assessments. They were also instructed to avoid excessive sun exposure and artificial UV light during the entire 12-week study period. A total of 198 women were recruited and equally allocated to the Mature skin and Young skin studies.
2.3. Randomisation and Interventions
For the two studies, the investigational product was a nutritional supplement (Naticol®-CySkin®), manufactured by Kovic Kate International (Thailand) (Bangkok, Thailand). It contained l-cystine manufactured by BCF Life Sciences (Pleucadeuc, France) and fish collagen peptides (2 kDa) manufactured by Weishardt International (Graulhet, France). Two different dosages were under investigation, i.e., 5.5 or 11 g of Naticol®-CySkin®. In each case, the composition was 90% collagen–10% l-cystine, without excipient. The complete amino acid composition of the Naticol®-CySkin® formulation is presented in Figure 2. Another product was a placebo containing only excipients (11 g of maltodextrin). All products were presented in sachets to be taken orally once daily, 20 min before a meal in the morning, for a total duration of 12 weeks. The investigational products were presented to the participants and investigators in blinded packaging to ensure the double-blind nature of the trials. Each participant received oral and written instructions for the proper preparation and intake of the investigated oral supplements.
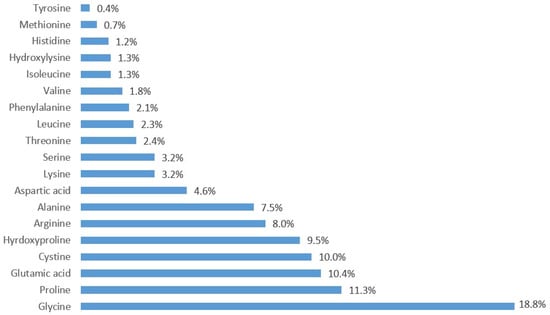
Figure 2.
Aminogram of the Naticol®-CySkin® formulation (in %).
In both studies, the participants were randomly assigned in a ratio of 1:1:1 to receive either 5.5 g Naticol®-CySkin®, or 11 g Naticol®-CySkin®, or the placebo products. At each visit to the test centre, sachets were counted to ensure that the appropriate amounts of test products were being used. The participants’ compliance with product utilisation was also monitored through daily logs.
2.4. Study Procedures
In both studies, all measurements and evaluations were performed after a systematic 20-min period of acclimatisation and rest in a controlled environment (temperature: 22 ± 2 °C, hygrometry: 50 ± 10%). The experimental procedures were performed at the same time at all appointments to avoid bias due to the natural circadian variations. The participants were either in the supine position or seated with their forearms on a table, depending on whether the face or the forearm was the site under investigation. All measurements and evaluations were performed by the same blinded technicians over the course of the study. An overview of the trial designs is shown in Figure 1. To further avoid any bias, the measurement sites (left or right forearm, left or right half-face) were selected randomly using proprietary software; to ensure consistent site placement across visits, each location was individually mapped before the initial time point T0. These maps were then used for accurate repositioning during subsequent visits.
2.5. Assessment Methods
2.5.1. Assessments in the Mature Skin Study
The effects of the investigated products upon crow’s feet were evaluated by grading their severity according to the reference scale for women described in the Skin Ageing Atlas dedicated to Asian skin []. For this clinical scoring, participants were seated between two symmetrically installed daylight lamps, their head and shoulders covered by a black cap and gown. Grading was performed by one trained judge, the same one for the entire study duration. The judge could give intermediate scores (.33 and .66) when the crow’s feet severity was between two consecutive marks.
Hydration of the outer layers of the skin was evaluated by the electrical capacitance method with a Corneometer CM 825 (Courage + Khazaka electronic, Köln, Germany). Capacitance, expressed in arbitrary units (a.u.), was measured in triplicate on the cheekbone and then averaged.
The thickness of the skin (i.e., epidermis + dermis) was determined for each participant on the temple and one inner forearm—sites considered to be photoexposed and photoprotected, respectively—using high-resolution ultrasonic images obtained with a 20 MHz ultrasound scanner (DermaLab® Combo, Cortex Technology, Aalborg, Denmark). The depth examined for such emission frequency is approximately 3 mm; axial and lateral image resolutions are 60 and 200 μm, respectively.
To measure skin colour, a Spectrophotometer CM700d (Konica Minolta, Tokyo, Japan) was used, calibrated before each use according to the manufacturer’s recommendations by means of a CM-A177 white ceramic plate. Triplicate measurements of L*a*b* values from the CIELAB colour space were performed on normal skin (i.e., on a non-hypo- or hyperpigmented area) on the left or right cheek. Average values were then calculated. The so-called individual typological angle (ITA°), where ITA° is equal to Arctg[(L* − 50)/b*].(180/π), was calculated, as it gives the best description of a lightening effect []. For the assessment of the whitening/lightening effect, the lighter the skin, the higher the L* and ITA° values.
The primary objective of the Mature skin study was to assess and compare the in vivo effects of the three tested formulas upon ageing signs (crow’s feet, cutaneous atrophy, and skin dryness). The other investigated variables were secondary objectives.
2.5.2. Assessments in the Young Skin Study
A dermatological evaluation was performed at the end of the study using whole-face diffused light photographs to assess the overall beauty/quality of the skin. Firstly, photographs taken at T0, T + 6 weeks, and T + 12 weeks for each given participant were presented simultaneously, side-by-side, to a dermatologist, who was aware of the time the photographs were taken but unaware of the tested treatment. Efficacy was evaluated after 6 and 12 weeks using the Global Aesthetic Improvement Scale (GAIS) (grades: 0–4). Secondly, a side-by-side forced blind comparison of the same whole face Tn vs. T0 photographs was conducted. The principle of the assessment was as follows: pairs of pictures of a given participant were blindly presented to the dermatologist, who tried to arrange the photographs in order as “before–after treatment”. If the order was correct, then a score of +1 was allotted to the evaluation. If incorrect, a score of 0 was given. A completely random arrangement in order would merit an average score of 0.5, meaning that no visible improvement was noticeable. Therefore, the mean values obtained after these evaluations were compared to a 0.5 theoretical mean. To confirm the validity of the effect, the observed mean score must be significantly greater than the theoretical mean of 0.5, and the observed confidence interval (CI) must not include the value 0.5.
In order to assess the anti-inflammatory and healing potentials of the test products, images of the cheeks presenting acne lesions and scars were acquired with an Antera 3D® camera (Miravex Limited, Dublin, Ireland). A map of the haemoglobin distribution was created for all images acquired at T0, T + 6, and T + 12 weeks, allowing the subsequent determination of the average haemoglobin concentration per unit area. A decrease in the average haemoglobin value was expected to confirm a diminution in the redness of skin with imperfections such as acne. Antera 3D® images were also used to monitor the changes in the skin roughness over the study period. The average roughness Ra, the root mean squared roughness Rq, and the elevation span Rt were the investigated variables.
The anti-inflammatory effect was evaluated using a UV-induced model of moderate inflammatory reaction. This model corresponds to the first step of the in vivo determination of the sun protection factor (SPF) of sunscreen products according to the ISO 24444 norm []. This evaluation was performed by Solar Test Asia Co., Ltd. (Bangkok, Thailand), a company specialising in the realisation of in vivo solar tests. Briefly, the principle of this evaluation consisted of determining the minimal erythema dose (MED) at baseline and at the end of the treatment period. If the MEDend of test is higher than the MEDbaseline, the skin has a reduced sensitivity to UV injury and an improved inflammatory response. The solar simulator used as a UV source was a Xenon Multiport light, model 601-300W (Solar Light Company, LLC, Glenside, PA, USA), which emits a continuous spectrum from 290 to 400 nm. The spectrum of the lamp was modified using UG 11 and WG 320 filters to comply with the emission spectrum defined by the ISO 24444 standard. The UV erythemal fluxes (mW/cm2.eff) were measured using a PMA2100 Radiometer (Solar Light Company, LLC, Glenside, PA, USA). Six increasing UV doses were administered to the test area on the back (on six different subsites), with a 25% geometric progression. Skin responses were assessed visually by a trained and validated technician to determine the MED 16 to 24 h after UV exposure. This visual assessment was made with the participant lying prone, under a daylight-type lamp with an illumination of at least 500 lux.
Health check-ups were performed during the pre-inclusion visit, before the first ingestion of the test products, after 6 weeks, and at the end of the treatment period. In addition to a general physical examination, routine haematology, blood chemistry, and a urine analysis were performed. The ultra-sensitive C-reactive protein (hs-CRP) inflammatory status marker was determined as well. These safety assessments were conducted by Bangkok Medical Laboratory (Bangkok, Thailand).
Because collagen peptides are the main ingredient of the oral supplementation comprising l-cystine and fish collagen peptides (>90.09%) and have a higher molecular weight compared to cystine, which is in a free amino acid form, we wanted to ensure that Naticol® was bioavailable after ingestion. A bioavailability study was carried out on 5 participants from the Young skin panel at the end of the T0 visit. This study was independently conducted at Bangkok Medical Laboratory (Thailand) in order to maintain the double-blind nature of the main trial. After 12 h of fasting, the volunteers ingested one dose of Naticol®-CySkin® at 11 g. Venous blood was collected from the cubital vein before and at 30, 60, 90, and 180 min post-ingestion. The samples were hydrolysed overnight with chlorohydric acid to release free amino acids. Plasma levels of hydroxyproline were then determined.
The primary outcomes of the Young skin study were the overall beauty/quality of the skin assessed according to the Global Aesthetic Improvement Scale and the side-by-side forced blind comparisons of Tn vs. T0 photographs. The other investigated variables were secondary objectives.
2.5.3. Assessments Common to the Mature Skin and Young Skin Studies
A photography-based technique was employed to investigate the changes in skin pigmentation homogeneity and texture roughness. This technique consisted of obtaining high-resolution photographs of one randomly chosen half-face for each participant. To ensure consistent and reproducible lighting and subject positions, a VISIA-CR Imaging System (Canfield Scientific Europe, Utrecht, the Netherlands) was used. Two photographs were taken: one under cross-polarised light and a second one under diffuse light. To analyse skin texture and pigmentary imperfections, a dedicated script was developed, using the ImageJ2 software and the open-source platform Fiji [,]. In both cases, the first step was removing the background from the image. A contrast segmentation step was then carried out to detect the target objects (roughness for texture and pigmentary disorders). A final binarisation step allowed us to obtain mappings characterising either the texture or the colour homogeneity of the skin (Figure 3). From these texture and pigmentation maps, two variables were computed: the surface area of the detected objects characterising roughness density and the surface area of the detected objects characterising pigmentation surface intensity, linked to pigmentation homogeneity. Both variables were expressed as pixels2.
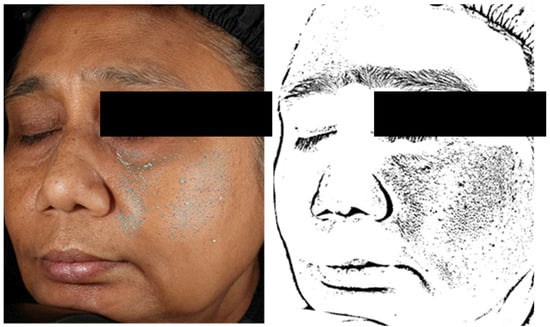
Figure 3.
Illustration of the detection of rough areas and obtention of a texture map from photographs taken with a VISIA-CR Imaging System (Canfield Scientific Europe, Utrecht, The Netherlands).
Adverse events reported by the participants themselves or observed by the investigators were collected during the study period. The type of sign, intensity, duration, and evolution were used to derive a conclusion regarding the possible causality between the observed sign and the treatment under investigation.
2.6. Data Analysis and Statistics
All statistical analyses were performed using SigmaStat 3.5 (Systat Software Inc., San Jose, CA, USA) and Past 1.37 (Paleontological Association) software. Quantitative variables were described as mean values with their respective standard deviations. Qualitative variables were reported as numbers and percentages. Verification of the normality of the distributions was performed using the Shapiro–Wilk test, with a threshold set at 1%. Depending on the normality of the data, Student’s t-test or a non-parametric Wilcoxon test was used. Differences among the three groups were tested using ANOVA after verification of the normality and homogeneity of variances using the Shapiro–Wilk test at 1% and the Levene test at 5%, respectively. Dunnett and Tukey post hoc tests were performed to determine which pairs of groups presented significant differences. The threshold of significance was set at p < 0.05. Tolerance analysis was performed on those participants who received at least one treatment dose, whereas the analysis of efficacy included all participants who completed the study without any major protocol deviation (Figure 4).
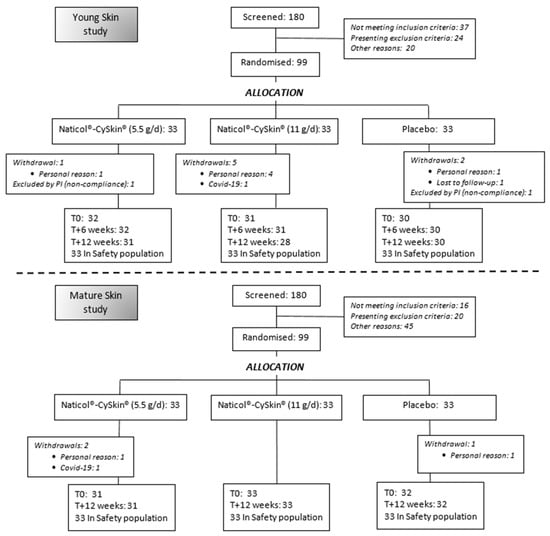
Figure 4.
Consolidated Standards of Reporting Trials (CONSORT) flowchart diagram of participants in the Mature skin and Young skin studies (PI: Principal Investigator).
Sample sizes were calculated for both studies based on their respective primary objectives, as is the usual practice in clinical research, with an α level of significance of 5% and a power of 80%. For the Mature skin study, the minimum number of participants was 30 per group, whereas a sample size of 21 participants per group was determined for the Young skin study. We took a conservative approach to account for possible dropouts and to ensure similarly sized studies: the sample size was adjusted to 33 participants per group for both trials, aiming to have at least 30 assessable participants per group at the final visit.
3. Results
3.1. Baseline Demographic Characteristics
From the 360 screened participants, 198 participants were enrolled in the Mature skin and Young skin studies (99 subjects in each trial). A total of 96 participants completed the Mature skin study, and 89 completed the Young skin study. Eleven withdrawals (nine due to personal reasons and two due to the occurrence of COVID-19) were recorded, and two subjects were excluded by the principal investigator because of insufficient compliance with the ingestion of the investigated product (Figure 4). The demographics of the participants are presented in Table 1.

Table 1.
Baseline demographics and skin characteristics of the study samples.
3.2. Compliance of the Subjects
In the Mature skin study, participants in the 5.5 g Naticol®-CySkin®, 11 g Naticol®-CySkin®, and placebo groups presented the following average consumption after 12 weeks of intervention: 84 ± 3, 85 ± 2, and 85 ± 2 sachets, respectively. At the end of the 12-week period, participants in the Young skin study used an average of 83 ± 1, 83 ± 2, and 83 ± 3 sachets of Naticol®-CySkin® (5.5 g/d), Naticol®-CySkin® (11 g/d), and placebo, respectively. All of these consumption levels were not only very close to the expected number of sachets for full compliance but also highly consistent with each other. As indicated in Section 3.1, two participants from the Young skin study were found to have complied poorly with the usage instructions for the test products and were excluded from the analysis by the principal investigator (see Figure 4). With the exception of these two participants, we concluded that the compliance of those who completed the studies was very satisfactory with regard to the use of the investigated products.
3.3. Moisturising Effects in Mature Participants
Compared to the placebo, only the higher dosage of Naticol®-CySkin® (11 g per day) significantly improved the hydration of the outer layers of the skin (Table 2). Indeed, a significant increase in the mean capacitance, measured by corneometry, was found from 62.7 ± 12.8 a.u. at baseline up to 65.8 ± 10.3 a.u. after 12 weeks of daily intake (i.e., +4.9%; p < 0.01). In contrast, the lower 5.5 g dosage of Naticol®-CySkin® did not significantly modify the moisturising of the skin: 64.5 ± 12.1 a.u. at T0; 64.8 ± 10.3 a.u. at T + 12 weeks (i.e., +0.5%), whereas a very small and non-significant −0.6% decrease in the mean capacitance of the skin was observed for the placebo group (from 63.3 ± 9.5 a.u. down to 62.9 ± 8.3 a.u.) during the same period. At baseline, no significant difference in the mean capacitance values from all groups was found (p = 0.813). The statistical comparison of the skin hydration variations of the three groups showed that the moisturising effect induced by the daily use of 11 g of Naticol®-CySkin® was significantly superior to the skin hydration variation observed with the placebo (p < 0.05).

Table 2.
Summary of the changes from baseline for the investigated variables in the Mature skin study for the three intervention groups.
3.4. Anti-Wrinkle Effects in Mature Participants
Data from the clinical evaluation of the crow’s feet area are summarised in Table 2. Significant reduction in the appearance of the crow’s feet area, indicating the severity of wrinkles, compared with baseline was observed following daily intakes of Naticol®-CySkin® (5.5 g/d) and Naticol®-CySkin® (11 g/d) (by −3.9% and −3.5%, respectively; p < 1.10−4 for both groups) for 12 weeks. A significant decrease in the mean score of the crow’s feet severity was also observed in the placebo group (−3.1%; p < 1.10−3). Although the two investigated dosages of Naticol®-CySkin® slightly outperformed the placebo, the difference between the three groups was not statistically significant (p = 0.810).
3.5. Thickening of the Skin in Mature Participants
The 12-week intervention with the two dosages of Naticol®-CySkin® resulted in global and significant increases in cutaneous thickness, a marker of anti-ageing effects, for both investigated sites, whereas participants treated with the placebo did not exhibit significant variations during the study period (Table 2). On the photoexposed temple, the average increase in skin thickness for the participants in the 5.5 g/d Naticol®-CySkin® group was 65.6 ± 104.9 μm compared to baseline (i.e., +5.6%; p < 0.01). On the photoprotected inner forearm skin, the observed increase was 38.2 ± 60.7 μm (+3.9%; p < 0.01). With the higher 11 g/d dose of Naticol®-CySkin®, the average thickening observed compared to baseline was more significant: 111.0 ± 100.8 μm for the temple (i.e., +9.6%; p < 1.10−6) and 76.2 ± 50.2 μm for the inner forearm (i.e., +8.3%; p < 1.10−8). For the placebo group, no significant effect on the average thickness of the temple and forearm skin was found after 12 weeks. The statistical comparison of the three tested treatments evidenced that Naticol®-CySkin® at 5.5 and 11 g/d both induced significantly superior thickening of the temple skin than the placebo (p < 0.05). With regard to the forearm skin, the thickening effect of the higher 11 g/d dose of Naticol®-CySkin® was significantly superior to the effects observed with either 5.5 g/d Naticol®-CySkin® or the placebo (p < 0.01).
3.6. Lightening of the Skin in Mature Participants
Colour measurements of the facial skin by spectrophotometry showed a general lightening between T0 and T + 12 weeks for all groups (Table 2). The 5.5 g/d dose of Naticol®-CySkin® was the only investigated treatment for which statistical significance was reached, as demonstrated by the marked increases in the mean luminance L* (+0.8%; p < 0.05) and in the mean individual typological angle (ITA°) (+3.7%; p < 0.05) compared to baseline. Tendencies toward a lightening of the skin were observed after 12 weeks of treatment with Naticol®-CySkin® (11 g/d) and with the placebo as well, since the observed increases in ITA° after 12 weeks did not reach the level of statistical significance. It should be noted that a significant increase in the mean L* was observed for the placebo group (+0.5%; p < 0.05). The statistical comparison of the three groups did not evidence significant differences between the mean variations of L* (p = 0.698) or ITA° (p = 0.602) of the facial skin after 12 weeks of study.
3.7. Effects upon Skin Roughness and Pigmentation Imperfections in Mature Participants
The computer-assisted image analysis of the digital photographs taken at T0 and T + 12 weeks showed a significant improvement in both the texture and the homogeneity of the pigmentation of the facial skin after 12 weeks in the 5.5 g/d Naticol®-CySkin® group only (Table 2). Indeed, when compared to baseline, a significant decrease in the mean total surface of detected roughness was observed (−2.7%; p < 0.05). During the same period, the mean total pigmented area decreased by −3.7% (p < 0.05). Surprisingly, no significant improvements in the texture or pigmentation homogeneity were observed after using the higher dose of the active compound, whereas a significant improvement in the pigmentation homogeneity was observed in the placebo group (−3.5%; p < 0.05). When the mean variations of the three groups were compared, no significant differences were found for the total pigmented area variable (p = 0.158) and the total surface of detected roughness (p = 0.569).
3.8. Safety and Tolerability in Young Participants
All the results from the general physical examination and laboratory investigations performed before the first ingestion of the test products, and after 6 and 12 weeks, are summarised in Table 3.

Table 3.
Summary of the results of physical examinations, haematology, blood chemistry, and urine analysis performed over a 12-week period for the three intervention groups in the Young skin study.
Haematology, clinical chemistry (fasting blood sugar, lipid profile, uric acid), hs-CRP, electrolyte (calcium), liver, and kidney functions were normal at baseline and remained in the normal biological ranges during the entire study period for the three groups of participants. Although marginally significant changes were observed for some variables compared to baseline (see Table 3 for details), all the investigated variables remained within the normal healthy clinical reference ranges for Thai adult women and were deemed not clinically significant. No adverse events related to the tested formulations were observed or reported in the Young skin and Mature skin studies. Therefore, after 6 and 12 weeks of once daily oral administration, the tested Naticol®-CySkin® (5.5 g/d), placebo, and Naticol®-CySkin® (11 g/d) were very well tolerated by the participants.
3.9. Bioavailability
All participants who took Naticol®-CySkin® presented increased systemic levels of free hydroxyproline after ingestion, confirming Naticol® availability (Figure 5). A plasma concentration peak was observed 90 min after ingestion for all participants. Additionally, at all times, free hydroxyproline concentrations exhibited disparities between individuals, thereby highlighting the inter-individual variations in collagen bioavailability after intake. Despite the concentration variability among the participants, the kinetics were comparable for all of them, following a typical bell curve.
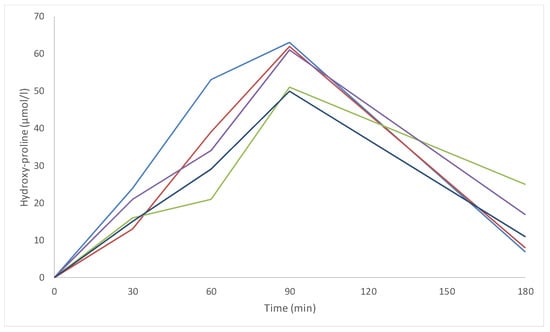
Figure 5.
Individual baseline corrected concentration–time curves of the free hydroxyproline (Hyp) plasmatic concentration after intake of the study product (11 g). Different colours indicate different study participants.
3.10. Global Aesthetic Improvement in Young Participants
The detailed results from the side-by-side forced blind comparisons of T0 and Tn photographs performed by the dermatologist are presented in Table 4. Since an average score superior or equal to the random score of 0.5 and its corresponding calculated confidence interval not including the 0.5 random score were the two necessary conditions to confirm an improvement in the skin appearance on the photographs, we can conclude that Naticol®-CySkin® (11 g/d) significantly improved the facial skin of the participants after 6 weeks (mean score of 0.77 ± 0.43; CI = 0.62–0.92). At the end of the study period, the improvement did not reach the level of statistical significance. It should be noted that a significant improvement was also found at T + 12 weeks for the placebo group (0.70 ± 0.47; CI = 0.53–0.87). A tendency toward an improvement in the facial skin was observed after 12 weeks of Naticol®-CySkin® (5.5 g/d). The blinded dermatologist assessed that a majority of the participants presented improvements in the GAIS scores. Results were very similar between the three groups, and the improvements were observed after 6 weeks, then plateaued. At 6 and 12 weeks, the proportion of participants who greatly improved their GAIS scores was higher for those who used Naticol®-CySkin® (11 g/d) compared to the other groups, although in a marginal manner only.

Table 4.
Summary of the side-by-side forced blind comparisons and blind assessment of the Global Aesthetic Improvement Scale by a dermatologist after 6 and 12 weeks of oral administration of Naticol®-CySkin® (5.5 g/d), Naticol®-CySkin® (11 g/d), or placebo.
3.11. Effects upon Skin Roughness and Pigmentation Imperfections in Young Participants
From the computer-assisted image analysis of the digital photographs taken at T0 and T + 12 weeks, a significant improvement in the homogeneity of the pigmentation of the facial skin was observed for the 5.5 and 11 g/d Naticol®-CySkin® groups (Table 5). This improvement progressed during the study period for the 11 g/d group (−8.2% after 6 weeks; p < 0.01; −10.5% after 12 weeks; p < 0.01). At the final visit, 71% of the panellists exhibited a more homogeneous facial pigmentation after taking 11 g/d Naticol®-CySkin®. Improvement in the homogeneity of the facial pigmentation was also found in the 5.5 g/d group, but only after 12 weeks of intervention (−6.6%; p < 0.05). Significant improvements in the pigmentation homogeneity were also observed in the placebo group: −6.0% after 6 weeks (p < 0.05) and −11.0% after 12 weeks (p < 0.01). When the mean variations of the three groups were compared, no significant differences were found for the total pigmented area variable, whatever the time of examination.

Table 5.
Summary of the changes from baseline for the investigated variables in the Young skin study for the three intervention groups.
With regard to the analysis of skin texture, only Naticol®-CySkin® (5.5 g/d) induced a significant decrease in the mean total surface of detected roughness (−7.8%; p < 0.05) after 12 weeks of utilisation, meaning that the facial skin was smoother than at the baseline visit (at 6 weeks, the improvement was not significant). A progressive improvement in the cutaneous texture, i.e., a tendency to a smoothing of the skin, although not significant, was observed after use of the 11 g/d dosage, whereas no effect was observed in the placebo group. The inter-group comparison did not evidence a superiority of one intervention over the others regarding the total surface of detected roughness at either 6 or 12 weeks.
Roughness of the facial skin was also investigated using the Antera 3D® imaging system and by determining the standard skin surface variables (Table 5). All the roughness variables (Ra, Rq, and Rt) were significantly improved after 6 and 12 weeks for all treatments under investigation, implying that better skin smoothness and a less coarse texture were achieved. Comparisons of the variations from baseline between groups showed significant differences for Ra (p = 0.013 and p = 0.017 at weeks 6 and 12, respectively) and for Rq (p = 0.019 and p = 0.027 at weeks 6 and 12, respectively). These differences favoured 5.5 g/d Naticol®-CySkin®, which allowed achieving the greatest reductions for the Ra, Rq, and Rt after 6 and 12 weeks of administration compared to baseline (Table 5).
3.12. Effect upon Skin Redness and Anti-Inflammatory Effect in Young Participants
Redness of the facial skin was also investigated, on areas where inflammation caused by acne lesions was present, by using the Antera 3D® imaging system. The average haemoglobin (Hb) level and the variation in Hb level were significantly improved after 6 and 12 weeks for the three investigated treatments, indicating that reduced redness and a better uniformity of the vascular component of the skin pigmentation were achieved (Table 5). An inter-group comparison of the mean variations in the two investigated variables evidenced a significant difference with regard to redness uniformity after 6 weeks (p < 0.01).
With regard to the UV-induced erythema model, the two tested dosages of Naticol®-CySkin® exhibited anti-inflammatory effects after 12 weeks of daily intake (Figure 6). Indeed, significant increases in the MED were observed with the 5.5 g/d (+6.14 ± 8.63 mJ.cm2.eff or +13.8%; p < 1 × 10−3) and the 11 g/d dosages (+6.62 ± 9.39 mJ.cm2.eff, i.e., +15.4%; p < 1 × 10−3). These increases were observed in 71.0% and 71.4% of the participants, respectively. A tendency toward a higher resistance to UV-induced erythema was also observed in the placebo group, for which the +4.03 ± 11.14 mJ.cm2.eff increase in MED was close to statistical significance. However, when the inter-group mean variations in the MED compared to baseline were compared, no significant difference was observed (p = 0.558).
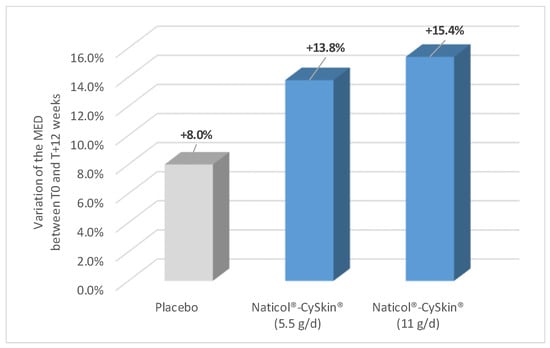
Figure 6.
Evolution of the Minimal Erythema Dose (MED) compared to baseline (expressed as % of variation) for the three intervention groups.
4. Discussion
To address the increasing need for better management of resources, the extraction of molecules of interest from by-products is worth the effort, from both ethical and economic perspectives. The cosmetic and nutraceutical industries can benefit from sourcing active ingredients such as collagen extracted from by-products. In our studies, we evaluated an oral supplement containing fish collagen hydrolysates. Because the amino acid content of these collagen peptides was poor in l-cystine, the oral supplement was complemented with l-cystine obtained industrially by the hydrolysis of natural keratin from poultry feathers. Moreover, if collagen is known to be effective on fibroblasts at the dermis level, l-cystine is recognised as active on keratinocytes at the epidermis level, which led us to hypothesise the possible complementary effects of the two ingredients on the two skin layers.
Collagen is a major constituent of the extracellular matrix of the dermis. The positive effects of collagen hydrolysates prepared from bovine, porcine, and fish sources on the moisturising, barrier function, anti-ageing, antioxidant, and biomechanical properties have been demonstrated for topical and oral formulations. A comprehensive review of their in vivo properties and mechanisms of action was published recently []. On the other hand, three in vitro studies (unpublished results) have proven that l-cystine improves keratinocyte proliferation and wound healing and has a depigmenting effect in reconstructed Asian skin when it is combined with reduced glutathione (GSH). We published an earlier study finding that l-cystine used as a food supplement produced hair and nail benefits (when associated with a natural keratin hydrolysate) and was able to lighten the skin and attenuate facial dark spots when associated with GSH in humans [,]. To date, most clinical studies aiming at evaluating collagen-based oral formulations have assessed anti-ageing effects. In the present work, the first study with 55–65-year-old women (“Mature skin study”) aimed at confirming whether the combination of collagen hydrolysates and l-Cys could alleviate signs of skin ageing. The second study, with 18–30-year-old women, focused more on the ability of the same combination to improve skin health and beauty. In these two trials, two different daily doses were investigated (5 g Naticol/0.5 g l-cystine and 10 g Naticol/1 g l-cystine).
Our studies confirmed several points. Firstly, the association of l-cystine with collagen peptides of fish origin was shown to be well tolerated at the two investigated dosages (5.5 and 11 g daily for 12 weeks). Indeed, no adverse events related to the investigated products were reported or observed in the two clinical studies. Haematology, clinical chemistry, and liver and kidney function examinations performed on the women participating in the Young skin study evidenced no clinically significant changes. Previous clinical trials had shown that human consumption of hydrolysed collagen-based dietary supplements was deemed safe []. l-cystine is safely used as a nutrient added to different foods. Therefore, it was unlikely that its association with collagen hydrolysates could induce any tolerability problems.
Several results from the Mature skin study are consistent with previously demonstrated beneficial properties of collagen-based oral supplements. In particular, Naticol®-CySkin® (11 g per day) significantly improved the hydration of the outer layers of the skin compared to baseline and to placebo. The moisturising effect of the 5.5 g/d dosage was not statistically significant. Improving skin moisture or enhancing the ability of the skin to retain moisture is important since it is known to contribute to the alleviation of skin photoageing [,]. The effect upon the dermis, as shown by the very significant increases in the cutaneous thickness measured on the photoexposed and photoprotected sites, was also a confirmation. Indeed, oral collagen supplementation or collagen-peptide-based oral nutritional supplements have previously been found to increase the dermal thickness and to reduce skin wrinkles [,,,,]. However, in our Mature skin study, the anti-wrinkle effect of Naticol®-CySkin® could not be validated. Although the two investigated dosages of the active oral supplement were slightly more effective than the placebo, the lack of a statistically significant difference between the three arms of the study prevented a conclusion about the anti-wrinkle effect. A recent systematic review and meta-analysis of randomised controlled trials by Myunk et al. found insufficient clinical evidence to support the use of collagen supplements for preventing or treating skin aging, also noting a possible bias as studies funded by pharmaceutical companies more often reported significant effects []. These findings highlight the need for more high-quality, independent clinical studies to establish the true efficacy of collagen-based supplements and eliminate potential conflicts of interest.
In the Young skin study, the most interesting results were those showing benefits for skin imperfections such as roughness using the 5.5 g/d dosage of Naticol®-CySkin®. In many cases, the 5.5 g/d dosage of Naticol®-CySkin® achieved superior results to the 11 g/d administration. This was particularly the case regarding improvements in the texture/smoothness and redness of the skin in the younger population. This raises the question of an optimal dose of the active ingredients, 11 g being over the optimum. This question probably needs further study.
Interestingly, the two dosages of Naticol®-CySkin® significantly improved the skin’s reaction to UV exposure, as indicated by the increases in the MED of the panellists after 12 weeks of treatment. While the observed increases in MED within the active groups did not reach statistical significance compared to the placebo group, the magnitude of these differences was appreciable and may have achieved significance with a larger sample size. This anti-inflammatory or photoprotective effect has rarely been found for oral supplement products so far and generally for formulations containing actives other than collagen or l-cystine [,]. Zague et al., who evaluated the effects of collagen hydrolysate supplementation on diverse skin ageing markers, found significant reductions in inflammation markers [].
With regard to the mechanisms of action, collagen hydrolysates are low-molecular-weight molecules that are more easily absorbed and used by tissues, including skin, compared to native collagen. After ingestion, collagen hydrolysates are further cleaved by enzymes during gastrointestinal passage. This gives rise to elevated levels of free amino acids and bioactive peptides in the blood circulation [,]. Hydrolysed collagen has been shown to block the increase in MMPs and collagen breakdown by reducing skin oxidation levels and to stimulate the TGF-Beta/Smad signalling pathway, thus restoring collagen synthesis and reducing skin inflammation. Taking hydrolysed collagen supplements can reduce UV-induced skin ageing and stimulate procollagen synthesis. Clinically, this results in improved skin moisturising and elasticity, reduced wrinkles and thickening of photoaged skin, and protection against UV radiation, as demonstrated by several clinical studies []. Cystine also plays a significant protective role because it is involved in the biosynthesis of glutathione, a potent antioxidant that helps protect the skin from oxidative damage. Because reactive oxygen species (ROS) are known to activate melanogenesis, the antioxidant properties of l-Cys and glutathione have a quenching effect on ROS, which explains the skin lightening effect and improvement in pigmentation []. Cystine is also involved in collagen synthesis and cross-linking, two essential processes for maintaining dermis structure and skin biomechanical properties. Cystine and cysteine form a redox pair, with cystine being the stable, natural form found in food. Upon ingestion, cystine can be partially reduced to cysteine in the small intestine due to its reductive environment and the action of enzymes and glutathione. Both cystine and cysteine are absorbed by the gut via specific transporters. Cysteine is the dominant intra-cellular form (80%) while cystine predominates in the plasma (also ~80%). In essence, cystine serves as a reservoir for cysteine, with their balance and conversion depending on the environment within the body.
Generalising the observed results to males or non-Asian populations is questionable and is a limitation of these trials. Nonetheless, prior studies conducted in Caucasian populations—including those with Middle Eastern and East Asian origins—indicate that generalisation may be possible [,,,].
5. Conclusions
Unlike topical products, oral supplements can have a systemic effect, supporting the rationale of the popular “beauty from within” concept. Numerous clinical studies have demonstrated the various cosmetic benefits of either oral collagen or oral cystine, including enhanced cutaneous moisturising, reduction of ageing signs such as wrinkles or dermal atrophy, regulation of skin pigmentation, and anti-inflammatory effects, to name the most frequent claims. Our two 12-week studies with young and mature Asian women, wherein l-cystine was combined with collagen hydrolysate, confirmed that this combination is a safe option when administered orally and is likely to mitigate some cutaneous signs of ageing, improve skin hydration and texture, and exhibit some anti-inflammatory activity, which can help improve the overall appearance of facial skin. Interestingly, the benefits do not only affect mature persons; younger women with skin imperfections may also benefit from daily usage of an oral combination of l-cystine and collagen peptides. However, our findings must be confirmed by further studies, since several observed effects did not differ significantly from the placebo. Until now, collagen-based supplements, with or without add-ons, have mostly been intended for mature consumers. Therefore, a novel finding of our work is that an oral supplementation combining l-cystine and fish collagen peptides can also be beneficial for young adults with skin imperfections such as moderate acne and roughness.
Author Contributions
Conceptualisation, C.B.-B. and J.D.; methodology, F.P., K.C., C.B.-B. and J.D.; validation, C.B.-B., J.-P.S., M.R. and K.C.; formal analysis, F.P. and K.C.; investigation, K.C. and K.S.; writing—original draft preparation, F.P.; writing—review and editing, M.R., J.-P.S., C.B.-B. and J.D.; supervision, F.P.; project administration, K.C.; funding acquisition, C.B.-B. and J.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research was sponsored by Weishardt International (Graulhet, France) and BCF Life Sciences (Pleucadeuc, France).
Institutional Review Board Statement
This study followed the Good Clinical Practices and Personal Data Protection Act B.E. 2562 (2019) of Thailand. Approvals of the protocols, CRFs, and informed consent documents were granted by the Human Research Ethics Committee of Thammasat University (Faculty of Medicine), with certificates of approval 268/2021 (25/10/2021) and 255/2021 (15/10/2021) granted to the Mature skin study (project MTU-EC-IM-5-180/64), and the Young skin study (project MTU-EC-IM-5-179/64), respectively.
Informed Consent Statement
Written informed consent was obtained from all subjects involved in these clinical trials, including the permission to use their photographs for publication.
Data Availability Statement
Data supporting the findings of the two clinical studies presented in this article are available from the corresponding author upon reasonable request.
Acknowledgments
We are grateful to the participants in these two clinical trials.
Conflicts of Interest
C.B.-B., J.-P.S., M.R. and J.D. were employees of the study’s sponsors at the time of the performance of the trials. The other authors declare no conflicts of interest. The funders had no role in the design of the studies, the collection, analyses, and interpretation of data, or in the writing of the manuscript.
References
- Salo, A.M.; Myllyharju, J. Prolyl and lysyl hydroxylases in collagen synthesis. Exp. Dermatol. 2021, 30, 38–49. [Google Scholar] [CrossRef]
- León-López, A.; Morales-Peñaloza, A.; Martínez-Juárez, V.M.; Vargas-Torres, A.; Zeugolis, D.I.; Águirre-Alvarez, G. Hydrolyzed collagen—Sources and applications. Molecules 2019, 24, 4031. [Google Scholar] [CrossRef]
- Shenoy, M.; Abdul, N.S.; Qamar, Z.; Al Bahri, B.M.; Al Ghalayini, K.Z.K.; Kakti, A. Collagen structure, synthesis, and its applications: A systematic review. Cureus 2022, 14, e24856. [Google Scholar] [CrossRef] [PubMed]
- Avila Rodriguez, M.I.; Rodríguez Barroso, L.G.; Sánchez, M.L. Collagen: A review on its sources and potential cosmetic applications. J. Cosmet. Dermatol. 2018, 17, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Land, E.J.; Ito, S.; Wakamatsu, K.; Riley, P.A. Rate constants for the first two chemical steps of eumelanogenesis. Pigment. Cell Res. 2003, 16, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Ren, W.; Yang, G.; Duan, J.; Huang, X.; Fang, R.; Li, C.; Li, T.; Yin, Y.; Hou, Y.; et al. L-cysteine metabolism and its nutritional implication. Mol. Food Res. 2016, 60, 134–146. [Google Scholar] [CrossRef]
- Del Marmol, V.; Ito, S.; Bouchard, B.; Libert, A.; Wakamatsu, K.; Ghanem, G.; Solano, F. Cysteine deprivation promotes eumelanogenesis in human melanoma cells. J. Investig. Dermatol. 1996, 107, 698–702. [Google Scholar] [CrossRef]
- Simon, J.D.; Peles, D.; Wakamatsu, K.; Ito, S. Current challenges in understanding melanogenesis: Bridging chemistry, biological control, morphology, and function. Pigment. Cell Melanoma Res. 2009, 22, 563–579. [Google Scholar] [CrossRef]
- McPherson, R.A.; Hardy, G. Clinical and nutritional benefits of cysteine-enriched protein supplements. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 562–568. [Google Scholar] [CrossRef]
- Duperray, J.; Sergheraert, R.; Chalothorn, K.; Tachalerdmanee, P.; Perin, F. The effects of the oral supplementation of L-cystine associated with reduced L-Glutathione-GSH on human skin pigmentation: A randomized, double-blind, benchmark- and placebo-controlled clinical trial. J. Cosmet. Dermatol. 2022, 21, 802–813. [Google Scholar] [CrossRef]
- Nobile, V.; Duperray, J.; Cestone, E.; Sergheraert, S.; Tursi, F. Efficacy and safety of L-cystine associated or not to a natural keratin (Kera-Diet®) hydrolysate on hair and nails: Randomized, placebo-controlled, clinical trial on healthy females. J. Cosmo. Trichol. 2019, 5, 142–150. [Google Scholar]
- Bazin, R.; Flament, F. Skin Ageing Atlas. Volume 2, Asian Type; Editions Med’Com: Paris, France, 2010; pp. 40–41. [Google Scholar]
- Petit, L.; Pierard, G.E. Skin-lightening products revisited. Int. J. Cosmet. Sci. 2003, 25, 169–181. [Google Scholar] [CrossRef] [PubMed]
- ISO 24444:2019; Cosmetics—Sun Protection Test Methods—In Vivo Determination of the Sun Protection Factor (SPF). American National Standards Institute (ANSI): New York, NY, USA, 2019.
- Schindelin, J.; Argand-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji—An open source platform for biological image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Rueden, C.T.; Schindelin, J.; Hiner, M.C.; DeZonia, B.E.; Walter, A.E.; Arena, E.T.; Eliceiri, K.W. ImageJ2: ImageJ for the next generation of scientific image data. BMC Bioinform. 2017, 18, 529. [Google Scholar] [CrossRef]
- Aguirre-Cruz, G.; León-López, A.; Cruz-Gómez, V.; Jiménez-Alvarado, R.; Águirre-Alvarez, G. Collagen hydrolysates for skin protection: Oral administration and topical formulation. Antioxidants 2020, 9, 181. [Google Scholar] [CrossRef]
- Geng, R.; Kang, S.G.; Huang, K.; Tong, T. Boosting the photoaged skin: The potential role of dietary components. Nutrients 2021, 13, 1691. [Google Scholar] [CrossRef]
- Asserin, J.; Lati, E.; Shioya, T.; Prawitt, J. The effect of oral collagen peptide supplementation on skin moisture and the dermal collagen network: Evidence from an ex vivo model and randomized, placebo-controlled clinical trials. J. Cosmet. Dermatol. 2015, 14, 291–301. [Google Scholar] [CrossRef]
- Addor, F.A.S.; Vieira, J.C.; Melo, C.S.A. Improvement of dermal parameters in aged skin after oral use of a nutrient supplement. Clin. Cosmet. Investig. Dermatol. 2018, 30, 195–201. [Google Scholar] [CrossRef]
- Evans, M.; Lewis, A.D.; Zakaria, N.; Pelipyagina, T.; Guthrie, N. A randomized, triple-blind, placebo-controlled, parallel study to evaluate the efficacy of a freshwater marine collagen on skin wrinkles and elasticity. J. Cosmet. Dermatol. 2021, 20, 825–834. [Google Scholar] [CrossRef]
- Proksch, E.; Schunck, M.; Zague, V.; Segger, D.; Degwert, J.; Oesser, S. Oral intake of specific bioactive collagen peptides reduces skin wrinkles and increases dermal matrix synthesis. Skin Pharmacol. Physiol. 2014, 27, 113–119. [Google Scholar] [CrossRef]
- Laing, S.; Bielfeldt, S.; Ehrenberg, C.; Wilhelm, K.P. A dermonutrient containing special collagen peptides improves skin structure and function: A randomized, placebo-controlled, triple-blind trial using confocal laser scanning microscopy on the cosmetic effects and tolerance of a drinkable collagen supplement. J. Med. Food 2020, 23, 147–152. [Google Scholar] [PubMed]
- Myung, S.K.; Park, Y. Effects of collagen supplements on skin aging: A systematic review and meta-analysis of randomized controlled trials. Am. J. Med. 2025, 138, 1264–1277. [Google Scholar] [CrossRef] [PubMed]
- Nobile, V.; Burioli, A.; Yu, S.; Zhifeng, S.; Cestone, E.; Insolia, V.; Zaccaria, V.; Malfa, G.A. Photoprotective and antiaging effects of a standardized red orange (Citrus sinensis (L.) Osbeck) extract in Asian and Caucasian subjects: A randomized, double-blind, controlled study. Nutrients 2022, 14, 2241. [Google Scholar] [CrossRef] [PubMed]
- Morse, N.L.; Reid, A.J.; St-Onge, M. An open-label clinical trial assessing the efficacy and safety of Bend skincare anti-ageing formula on minimal erythema dose in skin. Photodermatol. Photoimmunol. Photomed. 2018, 34, 152–161. [Google Scholar] [CrossRef]
- Zague, V.; de Freitas, V.; da Costa Rosa, M.; de Castro, G.A.; Jaeger, R.G.; Machado-Santelli, G.M. Collagen hydrolysate intake increases skin collagen expression and suppresses matrix metalloproteinase 2 activity. J. Med. Food 2011, 14, 618–624. [Google Scholar] [CrossRef]
- Virgilio, N.; Schön, C.; Mödinger, Y.; van der Steen, B.; Vlemickx, S.; van Holthoon, F.L.; Kleinnijenhuis, A.J.; Silva, C.I.F.; Prawitt, J. Absorption of bioactive peptides following collagen hydrolysate intake: A randomized, double-blind crossover study in healthy individuals. Front. Nutr. 2024, 11, 1416643. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Q.; Qian, J.; Liang, Q.; Wang, Z.; Xu, J.; He, S.; Ma, H. Bioavailability and bioavailable forms of collagen after oral administration to rats. J. Agric. Food Chem. 2015, 63, 3752–3756. [Google Scholar] [CrossRef]
- Liu, H.; Dong, J.; Du, R.; Gao, Y.; Zhao, P. Collagen study advances for photoageing skin. Photodermatol. Photoimmunol. Photomed. 2024, 40, e12931. [Google Scholar] [CrossRef]
- Samadi, A.; Movaffaghi, M.; Kazemi, F.; Yazdanparast, T.; Nasrollahi, S.A.; Firooz, A. Tolerability and efficacy assessment of an oral collagen supplement for the improvement of biophysical and ultrasonic parameters of skin in Middle Eastern consumers. J. Cosmet. Dermatol. 2023, 22, 2252–2258. [Google Scholar] [CrossRef]
- Vleminckx, S.; Virgilio, N.; Asserin, J.; Prawitt, J.; Silva, C.I.F. Influence of collagen peptide supplementation on visible signs of skin and nail health and –aging in an East Asian population: A double-blind, randomized, placebo-controlled trial. J. Cosmet. Dermatol. 2023, 23, 3645–3653. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).