Genetic Control of Photosynthesis in Sugarcane During Successive Ratoon Cycles
Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Tested Sugarcane Genotypes and Field Experimental Design
2.2. Field Data Collection
2.3. Sugarcane Quality Assessment
2.4. Statistical Analysis
3. Results
3.1. Joint Variance Analysis of Sugarcane Photosynthetic Traits
3.2. Principal Component Analysis of Sugarcane Photosynthetic Traits
3.3. Cluster Analysis and Discriminant Analysis of Sugarcane Photosynthetic Traits
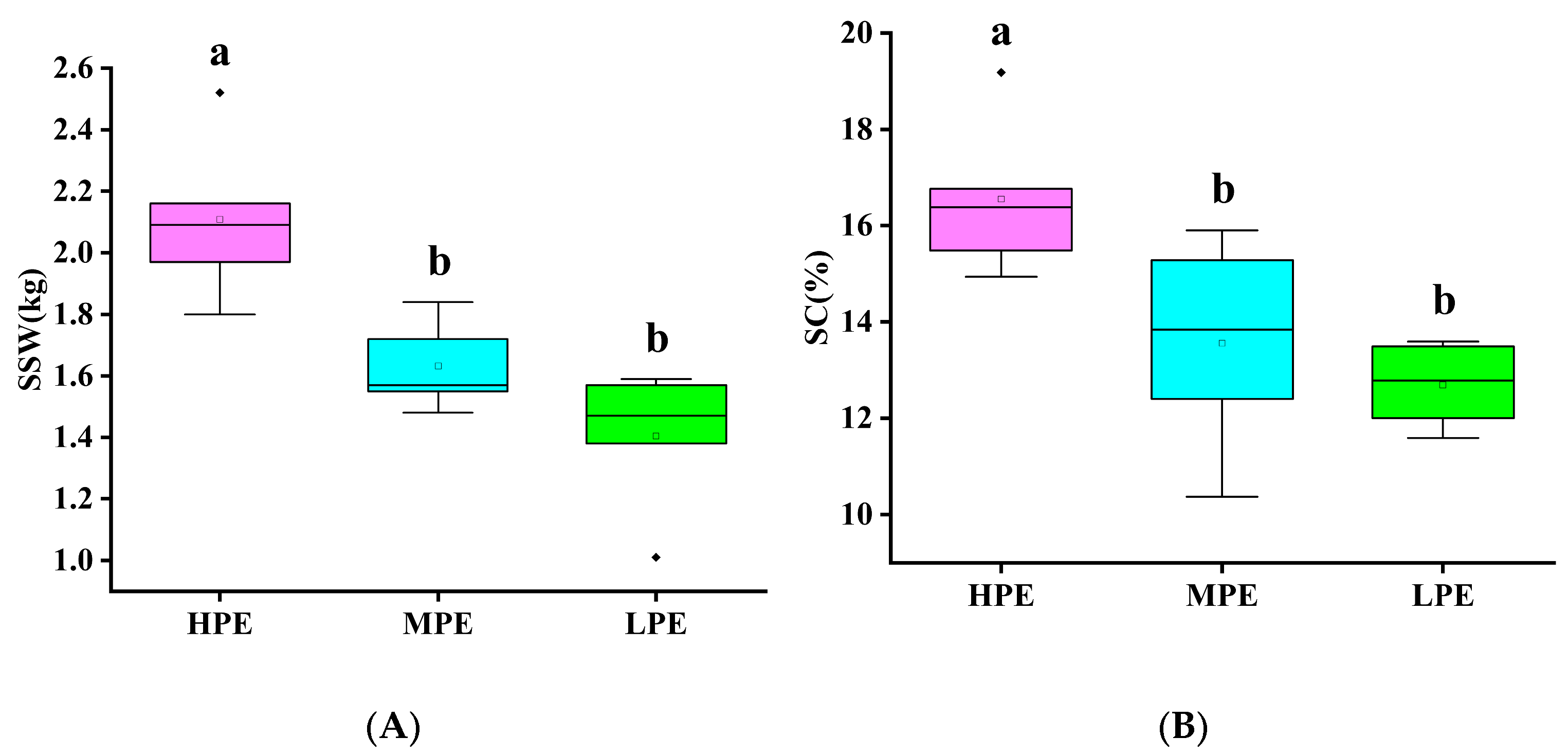
3.4. Quality Analysis of Genotypes for Photosynthetic Efficiency in Sugarcane
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Macrelli, S.; Mogensen, J.; Zacchi, G. Techno-economic evaluation of 2nd generation bioethanol production from sugarcane bagasse and leaves integrated with the sugar-based ethanol process. Biotechnol. Biofuels 2012, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Ngammuangtueng, P.; Nilsalab, P.; Chomwong, Y.; Wongruang, P.; Jakrawatana, N.; Sandhu, S.; Gheewala, S.H. Water-energy-food nexus of local bioeconomy hub and future climate change impact implication. J. Clean. Prod. 2023, 339, 136543. [Google Scholar] [CrossRef]
- Hiloidhari, M.; Haran, S.; Banerjee, R.; Rao, A.B. Life cycle energy-carbon-water footprints of sugar, ethanol and electricity from sugarcane. Bioresour. Technol. 2021, 330, 125012. [Google Scholar] [CrossRef]
- Leanasawat, N.; Kosittrakun, M.; Lontom, W.; Patcharin, S. Physiological and Agronomic Traits of Certain Sugarcane Genotypes Grown under Field Conditions as Influenced by Early Drought Stress. Agronomy 2021, 11, 2319. [Google Scholar] [CrossRef]
- Long, S.P.; Zhu, X.; Naidu, S.L.; Ort, D.R. Can improvement in photosynthesis increase crop yields? Plant Cell Environ. 2006, 29, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.G.; Long, S.P.; Ort, D.R. Improving photosynthetic efficiency for greater yield. Annu. Rev. Plant Biol. 2010, 61, 235–261. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Elsheery, N.I.; Sunoj, V.S.J.; Wen, Y.; Zhu, J.J.; Muralidharan, G.; Cao, K. Foliar application of nanoparticles mitigates the chilling effect on photosynthesis and photoprotection in sugarcane. Plant Physiol. Biochem. 2020, 149, 50–60. [Google Scholar] [CrossRef]
- Fernandez-Calleja, M.; Monteagudo, A.; Casas, A.M.; Boutin, C.; Pin, P.A.; Morales, F.; Igartua, E. Rapid On-Site Phenotyping via Field Fluorimeter Detects Differences in Photosynthetic Performance in a Hybrid-Parent Barley Germplasm Set. Sensors 2020, 20, 1486. [Google Scholar] [CrossRef]
- Ogawa, T.; Sonoike, K. Screening of mutants using chlorophyll fluorescence. J. Plant Res. 2021, 134, 653–664. [Google Scholar] [CrossRef]
- Hegde, V.; Sunoj, V.S.J.; Pradhan, A.; Mohanty, D.; Rathod, T.; Rane, J. Chlorophyll fluorescence transients as indicators for rapid detection of desiccation in sugarcane (Saccharum officinarum L.). Plant Physiol. Rep. 2025, 30, 673–681. [Google Scholar] [CrossRef]
- Qi, Z.; Xu, C.; Tang, R.; Zhang, Q.; Sun, W.; Guan, C.; Wang, Y.; Zhang, M.; Ding, J.; Zhang, Y.; et al. Response of Photosynthesis and Chlorophyll Fluorescence to Nitrogen Changes in Rice with Different Nitrogen Use Efficiencies. Plants 2025, 14, 1465. [Google Scholar] [CrossRef] [PubMed]
- Klughammer, C.; Schreiber, U. Complementary PS II quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the Saturation Pulse method. PAM Appl. Notes 2008, 1, 27–35. [Google Scholar]
- Peršić, V.; Ament, A.; Antunović Dunić, J.; Drezner, G.; Cesar, V. PEG-induced physiological drought for screening winter wheat genotypes sensitivity-integrated biochemical and chlorophyll a fluorescence analysis. Front. Plant Sci. 2022, 13, 987702. [Google Scholar] [CrossRef]
- Lazarevic, B.; Satovic, Z.; Nimac, A.; Vidak, M.; Gunjaca, J.; Politeo, O.; Carovic-Stanko, K. Application of Phenotyping Methods in Detection of Drought and Salinity Stress in Basil (Ocimum basilicum L.). Front. Plant Sci. 2021, 12, 629441. [Google Scholar] [CrossRef]
- Pfundel, E.E. Simultaneously measuring pulse-amplitude-modulated (PAM) chlorophyll fluorescence of leaves at wavelengths shorter and longer than 700 nm. Photosynth. Res. 2021, 147, 345–358. [Google Scholar] [CrossRef] [PubMed]
- De Souza, A.P.; Gaspar, M.; Da Silva, E.A.; Ulian, E.C.; Waclawovsky, A.J.; Nishiyama, M.Y.; Dos Santos, R.V.; Teixeira, M.M.; Souza, G.M.; Buckeridge, M.S. Elevated CO2 increases photosynthesis, biomass and productivity, and modifies gene expression in sugarcane. Plant Cell Environ. 2008, 31, 1116–1127. [Google Scholar] [CrossRef]
- Sales, C.R.G.; Wang, Y.; Evers, J.B.; Kromdijk, J. Improving C4 photosynthesis to increase productivity under optimal and suboptimal conditions. J. Exp. Bot. 2021, 72, 5942–5960. [Google Scholar] [CrossRef]
- Sommer, S.G.; Han, E.; Li, X.; Rosenqvist, E.; Liu, F. The Chlorophyll Fluorescence Parameter Fv/Fm Correlates with Loss of Grain Yield after Severe Drought in Three Wheat Genotypes Grown at Two CO2 Concentrations. Plants 2023, 12, 436. [Google Scholar] [CrossRef]
- Wang, M.; Li, X.; Shen, Y.; Adnan, M.Y.; Mao, L.; Lu, P.; Hu, Q.; Jiang, F.; Khan, M.T.; Deng, Z.; et al. A systematic high-throughput phenotyping assay for sugarcane stalk quality characterization by near-infrared spectroscopy. Plant Methods 2020, 17, 76. [Google Scholar] [CrossRef]
- Li, C.; Jackson, P.A.; Lu, X.; Xu, C.; Cai, Q.; Basnayake, J.W.M.; Lakshmanan, P.; Ghannoum, O.; Fan, Y. Genotypic variation in transpiration efficiency due to differences in photosynthetic capacity among sugarcane-related clones. J. Exp. Bot. 2017, 68, 2377–2385. [Google Scholar] [CrossRef]
- Singh, R.K.P.; Sudhir Pratap, S.; Singh, S.B. Correlation and path analysis in sugarcane ratoon. Sugar Tech 2005, 7, 176–178. [Google Scholar] [CrossRef]
- Wei, Y.; Yuzhi, X.; Khan, A.; Jiang, C.; Li, H.; Wu, Y.; Zhang, C.; Wang, M.; Chen, J.; Zeng, L. Analysis of Photosynthetic Characteristics and Screening High Light-Efficiency Germplasm in Sugarcane. Plants 2024, 13, 587. [Google Scholar] [CrossRef]
- Baker, N.R.; Rosenqvist, E. Applications of chlorophyll fluorescence can improve crop production strategies: An examination of future possibilities. J. Exp. Bot. 2004, 55, 1607–1621. [Google Scholar] [CrossRef]
- Wang, R.; Peng, W.; Hu, L.; Wu, W. Study on screening of canola varieties with high light use efficiency and evaluation of selecting indices. OCL 2020, 27, 43. [Google Scholar] [CrossRef]
- Latzel, V.; Fischer, M.; Groot, M.; Gutzat, R.; Lampei, C.; Ouborg, J.; Parepa, M.; Schmid, K.; Vergeer, P.; Zhang, Y.; et al. Parental environmental effects are common and strong, but unpredictable, in Arabidopsis thaliana. New Phytol. 2023, 237, 1014–1023. [Google Scholar] [CrossRef]
- Luo, T.; Zhou, Z.; Deng, Y.; Fan, Y.; Qiu, L.; Chen, R.; Yan, H.; Zhou, H.; Lakshmanan, P.; Wu, J.; et al. Transcriptome and metabolome analyses reveal new insights into chlorophyll, photosynthesis, metal ion and phenylpropanoids related pathways during sugarcane ratoon chlorosis. BMC Plant Biol. 2022, 22, 222. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.B.; Mahenderakar, M.D.; Jugran, A.K.; Singh, R.K.; Srivastava, R.K. Assessing genetic diversity and population structure of sugarcane cultivars, progenitor species and genera using microsatellite (SSR) markers. Gene 2020, 753, 144800. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.N.; Teferi, T.J.; Ambaw, A.M.; Gabi, M.T.; Koua, P.; Léon, J.; Ballvora, A. New drought-adaptive loci underlying candidate genes on wheat chromosome 4B with improved photosynthesis and yield responses. Physiol. Plant. 2021, 173, 2166–2180. [Google Scholar] [CrossRef]
- Tsujimoto, K.; Hikosaka, K. Estimating leaf photosynthesis of C3 plants grown under different environments from pigment index, photochemical reflectance index, and chlorophyll fluorescence. Photosynth. Res. 2021, 148, 33–46. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, L.; Wang, W.; Zhang, Z.; Du, H.; Qu, Z.; Li, X.; Xiang, H. Differences in Codon Usage Bias between Photosynthesis-Related Genes and Genetic System-Related Genes of Chloroplast Genomes in Cultivated and Wild Solanum Species. Int. J. Mol. Sci. 2018, 19, 3142. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Xiong, Z.; Zhang, Z.; Wei, Y.; Xiong, D.; Wang, F.; Huang, J. Exploration of chlorophyll fluorescence characteristics gene regulatory in rice (Oryza sativa L.): A genome-wide association study. Front. Plant Sci. 2023, 14, 1234866. [Google Scholar] [CrossRef] [PubMed]
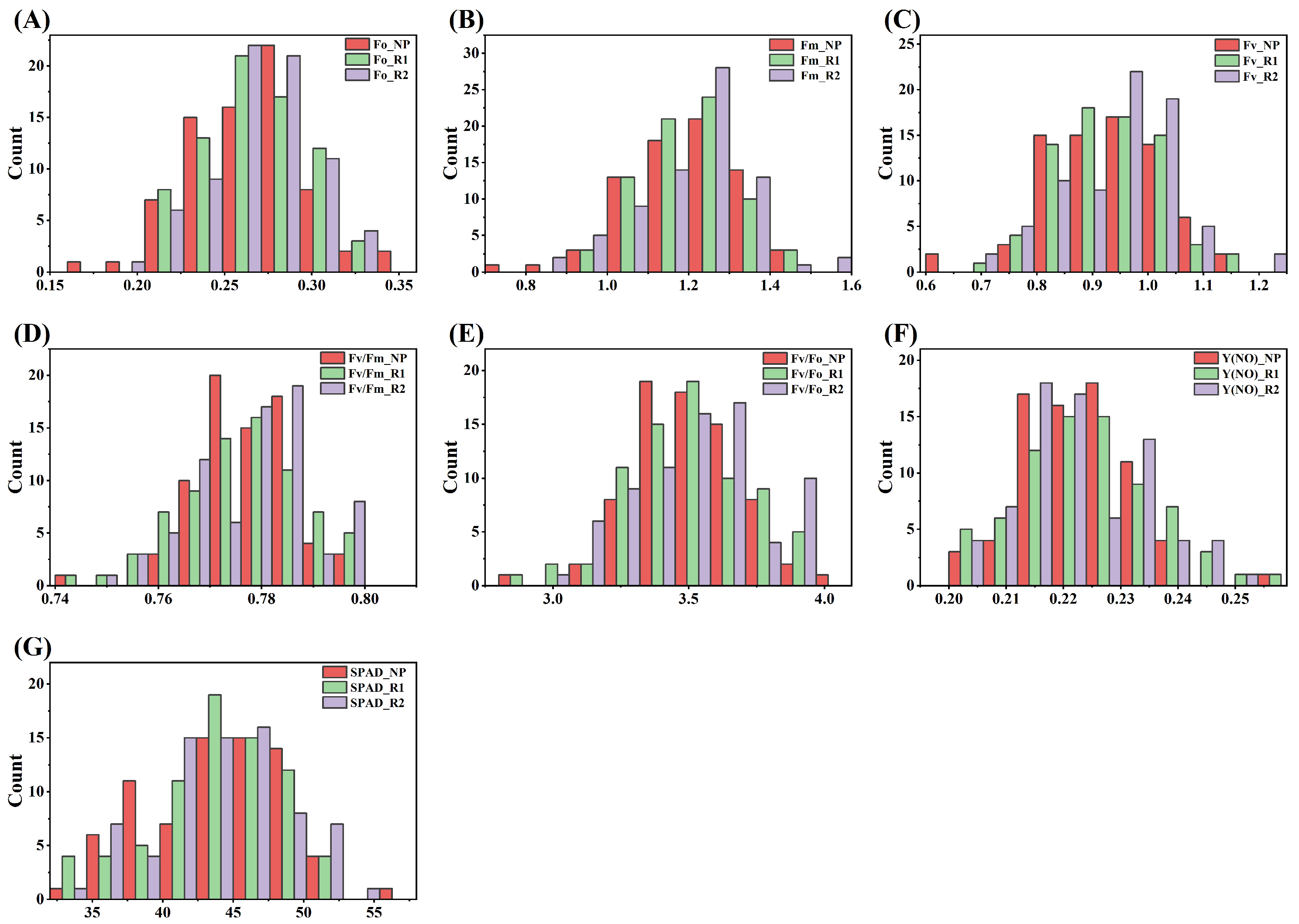
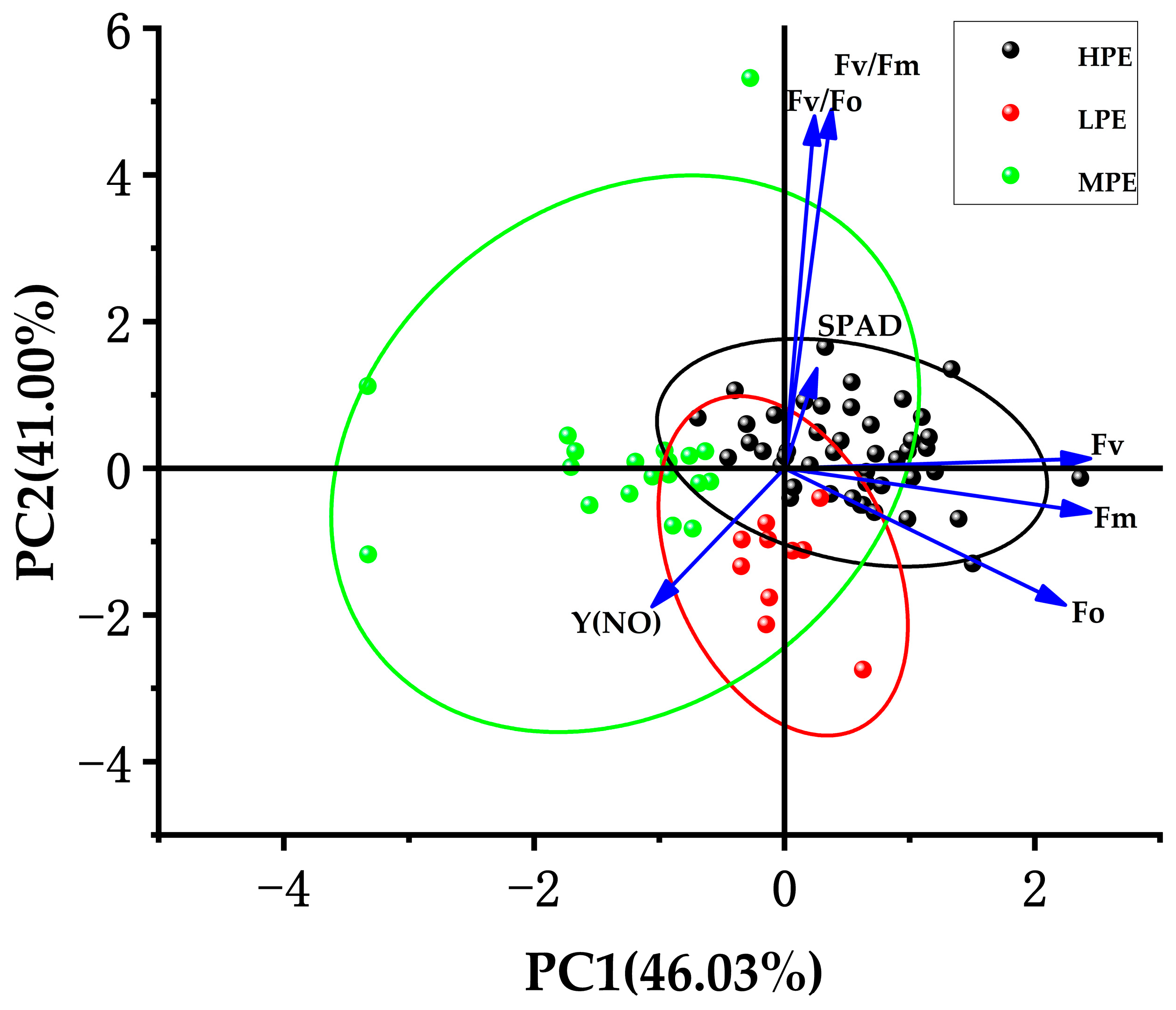
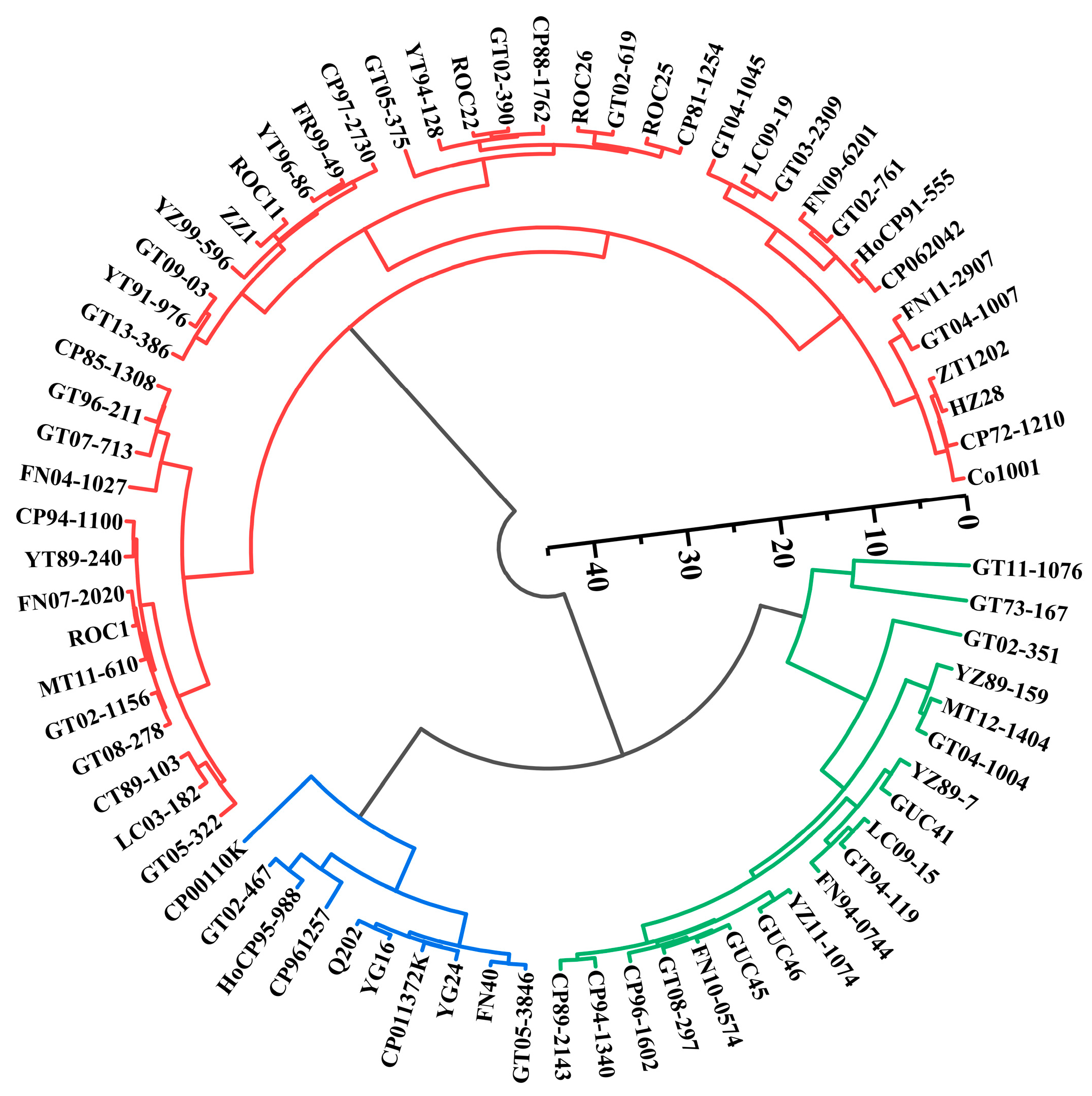

| Source of Variation | Df | SPAD | Fo | Fm | Fv | Fv/Fm | Fv/Fo | Y(NO) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sum Sq | SS% | Sum Sq | SS% | Sum Sq | SS% | Sum Sq | SS% | Sum Sq | SS% | Sum Sq | SS% | Sum Sq | SS% | ||
| Line | 73 | 172.3 *** | 61.50 | 0.006851 *** | 46.87 | 0.11749 *** | 43.88 | 0.07181 *** | 40.74 | 0.0006805 *** | 23.73 | 0.3618 *** | 21.54 | 0.0006805 *** | 23.73 |
| Rep | 2 | 11.7 | 0.11 | 0.000564 | 0.10 | 0.03095 | 0.32 | 0.02345 | 0.37 | 0.0002672 | 0.25 | 0.1667 | 0.27 | 0.0002672 | 0.25 |
| Year | 2 | 411.3 *** | 4.02 | 0.00139 | 0.26 | 0.00996 | 0.10 | 0.00391 | 0.06 | 0.0000822 | 0.08 | 0.0826 | 0.14 | 0.0000822 | 0.08 |
| Line:Rep | 146 | 18.2 *** | 12.98 | 0.00158 *** | 21.61 | 0.0281 *** | 20.99 | 0.01878 *** | 21.31 | 0.0003598 ** | 25.10 | 0.2372 *** | 28.25 | 0.0003598 ** | 25.10 |
| Residuals | 442 | 9.9 | 21.39 | 0.000752 | 31.16 | 0.01535 | 34.71 | 0.01097 | 37.67 | 0.0002408 | 50.84 | 0.1382 | 49.81 | 0.0002408 | 50.84 |
| h2B (%) | 86.00 | 81.82 | 81.39 | 80.57 | 72.27 | 70.30 | 72.27 | ||||||||
| NO. | Genotype | SPAD | Y(NO) | Fo | Fm | Fv | Fv/Fm | Fv/Fo | Clustering |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Co1001 | 40.43 | 0.22 | 0.27 | 1.22 | 0.95 | 0.78 | 3.48 | HPE |
| 2 | CP062042 | 47.23 | 0.22 | 0.27 | 1.15 | 0.90 | 0.78 | 3.66 | HPE |
| 3 | CP81-1254 | 45.57 | 0.21 | 0.28 | 1.36 | 1.08 | 0.79 | 3.85 | HPE |
| 4 | CP85-1308 | 47.13 | 0.23 | 0.33 | 1.41 | 1.08 | 0.77 | 3.27 | HPE |
| 5 | CP88-1762 | 46.43 | 0.21 | 0.30 | 1.35 | 1.05 | 0.79 | 3.74 | HPE |
| 6 | CP94-1100 | 48.63 | 0.24 | 0.29 | 1.30 | 1.02 | 0.78 | 3.60 | HPE |
| 7 | CP97-2730 | 46.27 | 0.23 | 0.27 | 1.26 | 0.99 | 0.79 | 3.77 | HPE |
| 8 | CT89-103 | 49.70 | 0.25 | 0.27 | 1.22 | 0.95 | 0.78 | 3.58 | HPE |
| 9 | FN04-1027 | 49.10 | 0.22 | 0.33 | 1.52 | 1.19 | 0.78 | 3.65 | HPE |
| 10 | FN07-2020 | 46.80 | 0.23 | 0.29 | 1.30 | 1.00 | 0.77 | 3.44 | HPE |
| 11 | FR99-49 | 43.60 | 0.21 | 0.27 | 1.27 | 1.00 | 0.79 | 3.67 | HPE |
| 12 | GT02-390 | 44.70 | 0.21 | 0.29 | 1.35 | 1.06 | 0.79 | 3.71 | HPE |
| 13 | GT02-619 | 49.23 | 0.21 | 0.29 | 1.35 | 1.06 | 0.79 | 3.71 | HPE |
| 14 | GT02-761 | 48.37 | 0.22 | 0.25 | 1.18 | 0.93 | 0.79 | 3.72 | HPE |
| 15 | GT05-322 | 56.77 | 0.22 | 0.29 | 1.29 | 1.00 | 0.78 | 3.57 | HPE |
| 16 | GT05-375 | 49.53 | 0.15 | 0.30 | 1.34 | 1.05 | 0.80 | 4.00 | HPE |
| 17 | GT09-03 | 44.07 | 0.20 | 0.27 | 1.28 | 1.01 | 0.80 | 4.02 | HPE |
| 18 | GT13-386 | 47.30 | 0.20 | 0.26 | 1.23 | 0.99 | 0.80 | 4.14 | HPE |
| 19 | GT96-211 | 50.07 | 0.23 | 0.31 | 1.33 | 1.03 | 0.77 | 3.39 | HPE |
| 20 | HoCP91-555 | 46.07 | 0.21 | 0.26 | 1.19 | 0.93 | 0.78 | 3.61 | HPE |
| 21 | LC03-182 | 49.33 | 0.23 | 0.28 | 1.22 | 0.94 | 0.77 | 3.48 | HPE |
| 22 | ROC1 | 49.33 | 0.23 | 0.29 | 1.30 | 1.00 | 0.77 | 3.42 | HPE |
| 23 | ROC11 | 45.03 | 0.23 | 0.29 | 1.29 | 1.03 | 0.79 | 3.87 | HPE |
| 24 | ROC22 | 43.80 | 0.21 | 0.29 | 1.34 | 1.05 | 0.79 | 3.67 | HPE |
| 25 | ROC25 | 47.97 | 0.20 | 0.27 | 1.34 | 1.07 | 0.80 | 3.90 | HPE |
| 26 | ROC26 | 50.27 | 0.21 | 0.29 | 1.36 | 1.06 | 0.79 | 3.73 | HPE |
| 27 | YT89-240 | 47.93 | 0.22 | 0.30 | 1.35 | 1.05 | 0.78 | 3.56 | HPE |
| 28 | YT91-976 | 43.33 | 0.21 | 0.26 | 1.25 | 0.99 | 0.79 | 3.94 | HPE |
| 29 | YT96-86 | 45.70 | 0.21 | 0.27 | 1.27 | 1.00 | 0.79 | 3.71 | HPE |
| 30 | YZ99-596 | 51.47 | 0.23 | 0.27 | 1.23 | 0.96 | 0.79 | 3.86 | HPE |
| 31 | ZZ1 | 46.60 | 0.22 | 0.27 | 1.27 | 1.01 | 0.80 | 3.89 | HPE |
| 32 | CP72-1210 | 39.57 | 0.23 | 0.27 | 1.26 | 0.99 | 0.78 | 3.70 | HPE |
| 33 | FN09-6201 | 49.50 | 0.23 | 0.24 | 1.12 | 0.88 | 0.79 | 3.71 | HPE |
| 34 | GT02-1156 | 47.00 | 0.22 | 0.28 | 1.26 | 0.98 | 0.78 | 3.47 | HPE |
| 35 | GT03-2309 | 42.93 | 0.20 | 0.24 | 1.16 | 0.92 | 0.80 | 3.90 | HPE |
| 36 | GT04-1007 | 41.13 | 0.22 | 0.29 | 1.29 | 1.01 | 0.78 | 3.52 | HPE |
| 37 | GT04-1045 | 36.47 | 0.22 | 0.25 | 1.17 | 0.92 | 0.78 | 3.74 | HPE |
| 38 | GT07-713 | 42.83 | 0.22 | 0.31 | 1.41 | 1.09 | 0.78 | 3.47 | HPE |
| 39 | GT08-278 | 46.30 | 0.22 | 0.29 | 1.30 | 1.02 | 0.78 | 3.54 | HPE |
| 40 | HZ28 | 44.50 | 0.22 | 0.26 | 1.22 | 0.96 | 0.78 | 3.65 | HPE |
| 41 | LC09-19 | 41.47 | 0.21 | 0.25 | 1.21 | 0.96 | 0.79 | 3.87 | HPE |
| 42 | MT11-610 | 50.13 | 0.22 | 0.29 | 1.31 | 1.02 | 0.78 | 3.50 | HPE |
| 43 | YT94-128 | 42.07 | 0.22 | 0.30 | 1.39 | 1.09 | 0.79 | 3.68 | HPE |
| 44 | ZT1202 | 41.67 | 0.21 | 0.26 | 1.22 | 0.96 | 0.78 | 3.65 | HPE |
| 45 | FN11-2907 | 37.47 | 0.22 | 0.29 | 1.32 | 1.03 | 0.78 | 3.51 | HPE |
| 46 | CP89-2143 | 48.10 | 0.22 | 0.26 | 1.11 | 0.85 | 0.78 | 3.61 | MPE |
| 47 | CP94-1340 | 43.93 | 0.22 | 0.26 | 1.06 | 0.81 | 0.78 | 3.63 | MPE |
| 48 | GT04-1004 | 51.87 | 0.23 | 0.23 | 0.97 | 0.75 | 0.77 | 3.40 | MPE |
| 49 | GT73-167 | 44.17 | 0.21 | 0.23 | 1.09 | 0.92 | 0.84 | 6.09 | MPE |
| 50 | MT12-1404 | 48.20 | 0.22 | 0.21 | 0.98 | 0.76 | 0.78 | 3.56 | MPE |
| 51 | CP96-1602 | 46.73 | 0.22 | 0.23 | 1.05 | 0.82 | 0.78 | 3.51 | MPE |
| 52 | FN94-0744 | 44.97 | 0.23 | 0.23 | 1.01 | 0.77 | 0.77 | 3.33 | MPE |
| 53 | GT08-297 | 46.37 | 0.22 | 0.24 | 1.11 | 0.87 | 0.78 | 3.59 | MPE |
| 54 | GT11-1076 | 45.73 | 0.56 | 0.21 | 0.92 | 0.71 | 0.78 | 3.47 | MPE |
| 55 | GT94-119 | 50.20 | 0.23 | 0.24 | 1.07 | 0.83 | 0.77 | 3.42 | MPE |
| 56 | GUC41 | 48.03 | 0.24 | 0.26 | 1.09 | 0.84 | 0.77 | 3.26 | MPE |
| 57 | GUC46 | 46.37 | 0.23 | 0.25 | 1.14 | 0.88 | 0.78 | 3.48 | MPE |
| 58 | LC09-15 | 48.50 | 0.23 | 0.24 | 1.05 | 0.81 | 0.77 | 3.38 | MPE |
| 59 | YZ11-1074 | 45.47 | 0.22 | 0.25 | 1.12 | 0.87 | 0.78 | 3.47 | MPE |
| 60 | YZ89-159 | 41.77 | 0.22 | 0.22 | 0.99 | 0.78 | 0.78 | 3.57 | MPE |
| 61 | YZ89-7 | 41.73 | 0.23 | 0.26 | 1.12 | 0.86 | 0.77 | 3.32 | MPE |
| 62 | FN10-0574 | 44.60 | 0.22 | 0.24 | 1.09 | 0.85 | 0.78 | 3.54 | MPE |
| 63 | GT02-351 | 43.07 | 0.21 | 0.14 | 0.79 | 0.64 | 0.79 | 3.72 | MPE |
| 64 | GUC45 | 44.03 | 0.22 | 0.23 | 1.11 | 0.86 | 0.78 | 3.48 | MPE |
| 65 | CP00110K | 40.03 | 0.22 | 0.28 | 1.26 | 0.98 | 0.78 | 3.52 | LPE |
| 66 | GT02-467 | 35.70 | 0.26 | 0.29 | 1.22 | 0.93 | 0.76 | 3.20 | LPE |
| 67 | Q202 | 41.67 | 0.24 | 0.28 | 1.20 | 0.92 | 0.77 | 3.33 | LPE |
| 68 | YG16 | 43.27 | 0.23 | 0.27 | 1.20 | 0.92 | 0.77 | 3.36 | LPE |
| 69 | CP011372K | 41.20 | 0.24 | 0.28 | 1.17 | 0.90 | 0.76 | 3.21 | LPE |
| 70 | CP961257 | 42.87 | 0.26 | 0.32 | 1.34 | 0.99 | 0.74 | 2.86 | LPE |
| 71 | FN40 | 43.23 | 0.23 | 0.28 | 1.24 | 0.96 | 0.77 | 3.26 | LPE |
| 72 | GT05-3846 | 44.97 | 0.24 | 0.29 | 1.23 | 0.94 | 0.76 | 3.25 | LPE |
| 73 | HoCP95-988 | 34.23 | 0.29 | 0.30 | 1.23 | 0.94 | 0.76 | 3.13 | LPE |
| 74 | YG24 | 38.20 | 0.23 | 0.27 | 1.18 | 0.91 | 0.77 | 3.32 | LPE |
| Grade of Photosynthetic Efficiency | High Photosynthetic Efficiency (HPE) | Moderate Photosynthetic Efficiency (MPE) | Low Photosynthetic Efficiency (LPE) |
|---|---|---|---|
| Tested genotypes | Co1001, CP062042, CP81-1254, CP85-1308, CP88-1762, CP94-1100, CP97-2730, CT89-103, FN04-1027, FN07-2020, FR99-49, GT02-390, GT02-619, GT02-761, GT05-322, GT05-375, GT09-03, GT13-386, GT96-211, HoCP91-555, LC03-182, ROC1, ROC11, ROC22, ROC25, ROC26, YT89-240, YT91-976, YT96-86, YZ99-596, ZZ1, CP72-1210, FN09-6201, GT02-1156, GT03-2309, GT04-1007, GT04-1045, GT07-713, GT08-278, HZ28, LC09-19, MT11-610, YT94-128, ZT1202, FN11-2907 | CP89-2143, CP94-1340, GT04-1004, GT73-167, MT12-1404, CP96-1602, FN94-0744, GT08-297, GT11-1076, GT94-119, GUC41, GUC46, LC09-15, YZ11-1074, YZ89-159, YZ89-7, FN10-0574, GT02-351, GUC45 | CP00110K, GT02-467, Q202, YG16, CP011372K, CP961257, FN40, GT05-3846, HoCP95-988, YG24 |
| Fm | 1.28 ± 0.08 A | 1.05 ± 0.09 B | 1.23 ± 0.05 A |
| Fo | 0.28 ± 0.02 A | 0.23 ± 0.03 B | 0.29 ± 0.01 A |
| Fv | 1.01 ± 0.06 A | 0.81 ± 0.07 C | 0.94 ± 0.03 B |
| Fv/Fm | 0.79 ± 0.01 A | 0.78 ± 0.02 A | 0.76 ± 0.01 A |
| Fv/Fo | 3.68 ± 0.18 A | 3.62 ± 0.61 A | 3.25 ± 0.17 B |
| Y(NO) | 0.22 ± 0.01 A | 0.24 ± 0.08 A | 0.24 ± 0.02 A |
| SPAD | 46.02 ± 3.89 A | 45.99 ± 2.71 A | 40.54 ± 3.50 B |
| Total | 45 | 19 | 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhang, C.; Wei, Y.; Xu, Y.; Khan, A.; Jiang, C.; Li, H.; Chen, J.; Wu, Y.; Yang, Z.; Chen, J.; et al. Genetic Control of Photosynthesis in Sugarcane During Successive Ratoon Cycles. Biology 2026, 15, 75. https://doi.org/10.3390/biology15010075
Zhang C, Wei Y, Xu Y, Khan A, Jiang C, Li H, Chen J, Wu Y, Yang Z, Chen J, et al. Genetic Control of Photosynthesis in Sugarcane During Successive Ratoon Cycles. Biology. 2026; 15(1):75. https://doi.org/10.3390/biology15010075
Chicago/Turabian StyleZhang, Chi, Yibin Wei, Yuzhi Xu, Abdullah Khan, Chunxiu Jiang, Huojian Li, Jun Chen, Yuling Wu, Zuli Yang, Jiafu Chen, and et al. 2026. "Genetic Control of Photosynthesis in Sugarcane During Successive Ratoon Cycles" Biology 15, no. 1: 75. https://doi.org/10.3390/biology15010075
APA StyleZhang, C., Wei, Y., Xu, Y., Khan, A., Jiang, C., Li, H., Chen, J., Wu, Y., Yang, Z., Chen, J., Liang, F., Xu, J., Zhang, M., & Bao, Y. (2026). Genetic Control of Photosynthesis in Sugarcane During Successive Ratoon Cycles. Biology, 15(1), 75. https://doi.org/10.3390/biology15010075









