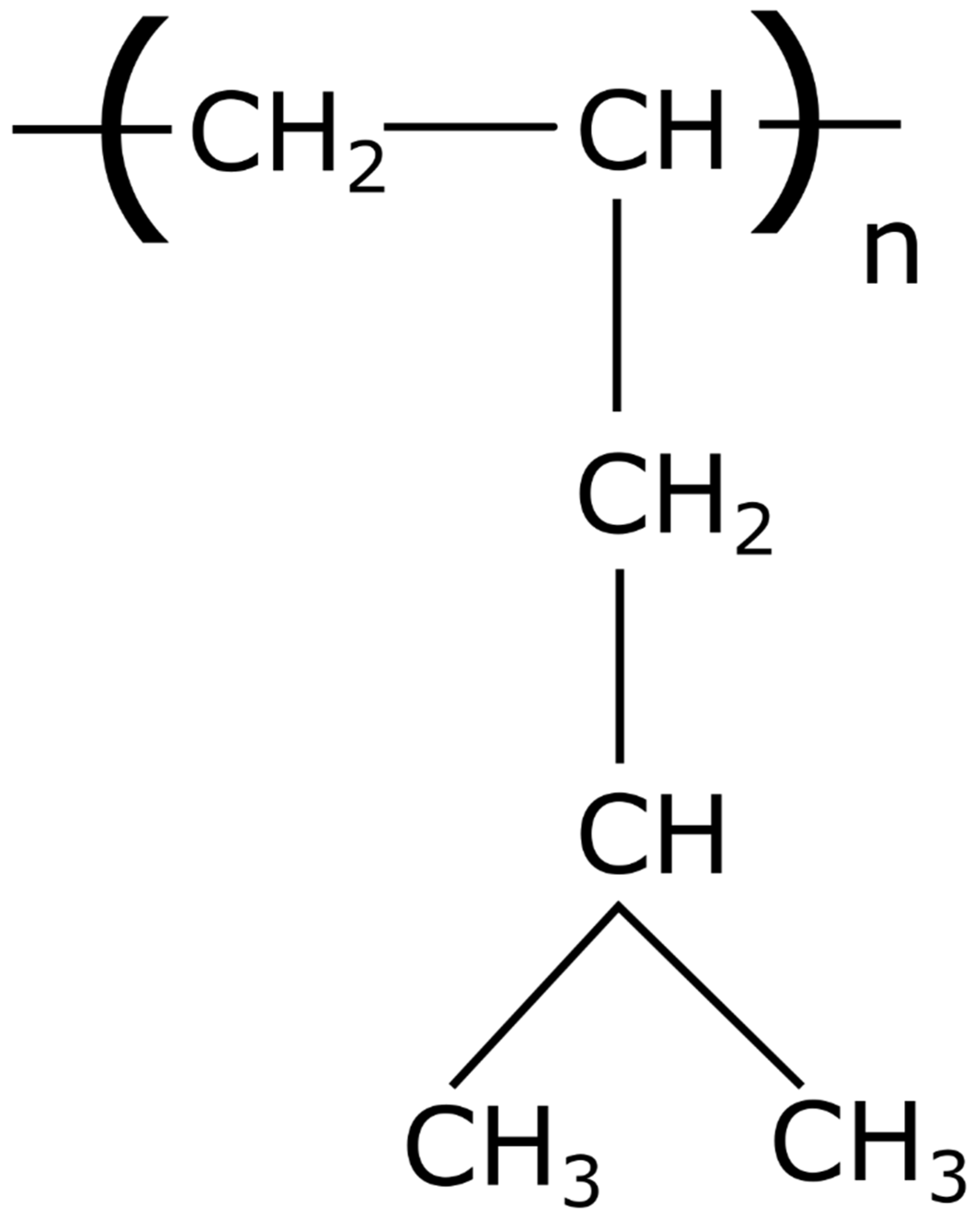
Preparation of Hollow Fiber Membranes Based On Poly(4-methyl-1-pentene) for Gas Separation
Abstract
:1. Introduction
2. Materials and Methods
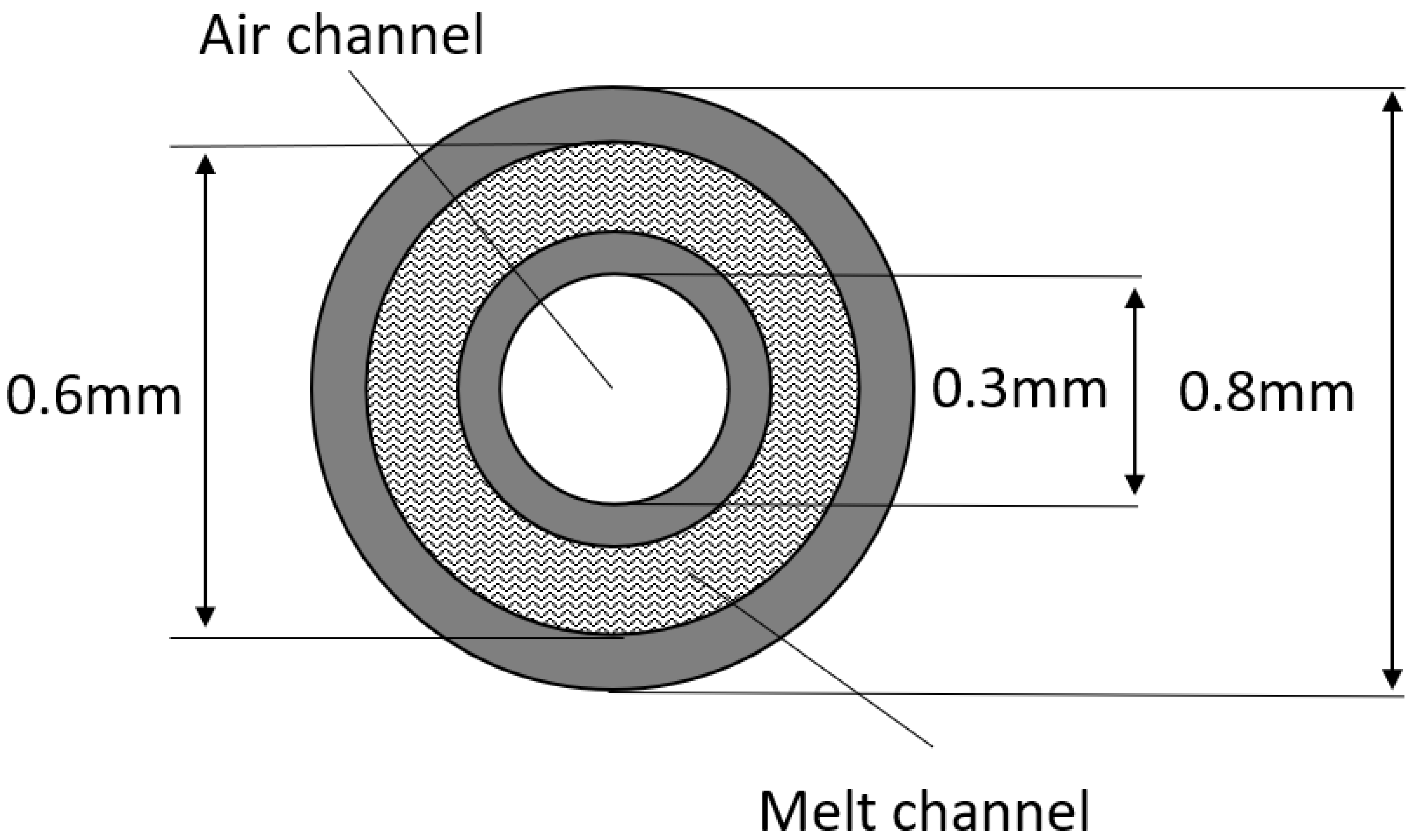
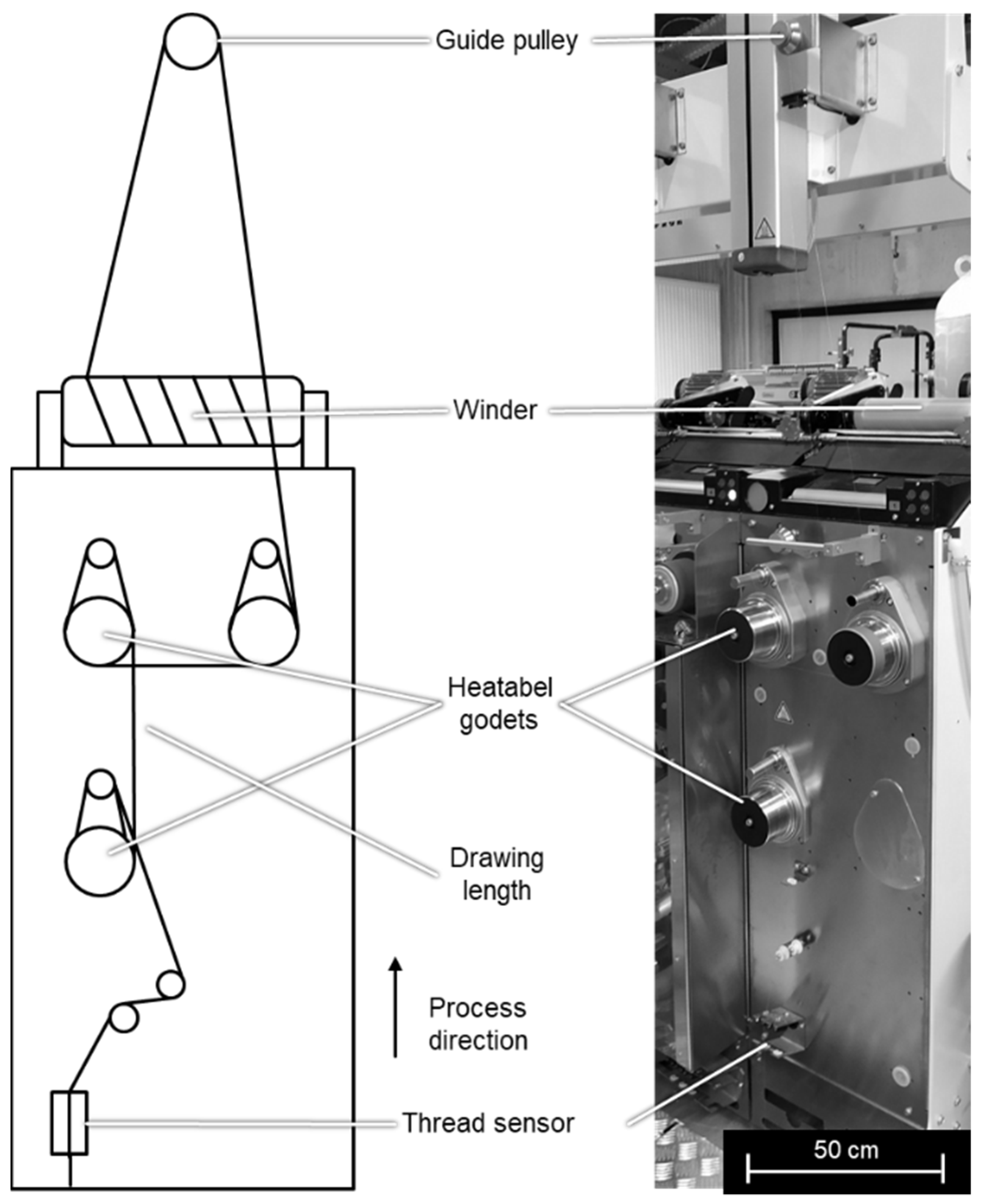
2.1. Fiber Preparation Method
2.2. Gas Permeability Measurement Method
3. Results and Discussion
3.1. Effects of Post-Drawing on the Cross Section Geometry
3.2. Influence of Production Conditions on Gas Transport and Release Properties
3.3. Investigation of the Peculiarities of Permeability of Mixtures in Membrane Modules with PMP HFs
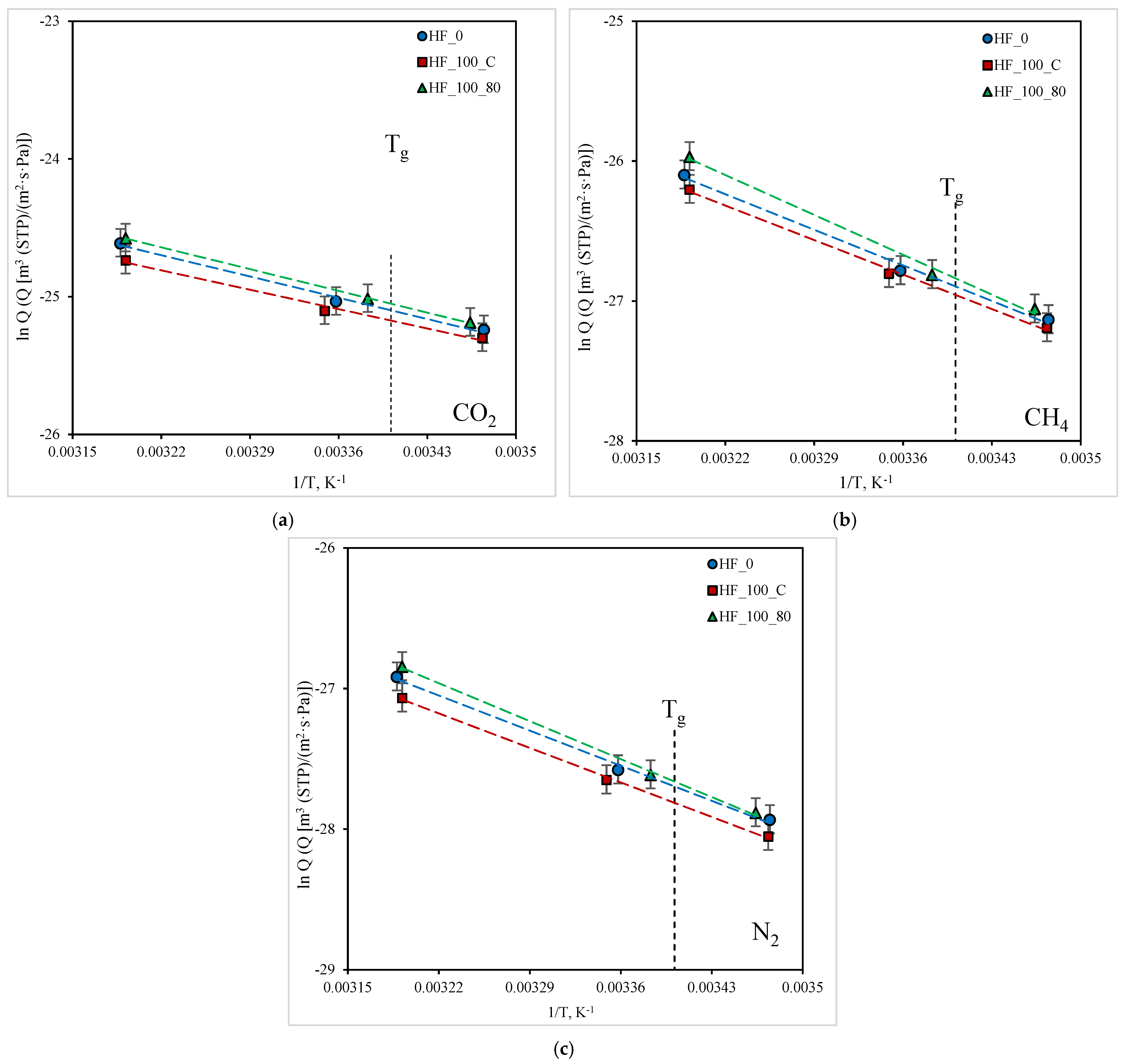
3.4. Effect of Temperature on the Gas Transport Properties of PMP HFs
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baker, R.W. Membrane Technology and Application, 2nd ed.; John Wiley & Sons, Ltd: San Francisco, CA, USA, 2012; p. 574. [Google Scholar]
- Buller, K.-U. Heat and Heat-Resistant Polymers; Chemistry: Moscow, Russia, 1984; p. 1056. [Google Scholar]
- He, T.; Porter, R.S. Characterization of uniaxially drawn poly(4-methyl-pentene-1). Polymer 1987, 28, 1321–1325. [Google Scholar] [CrossRef]
- Chen, X.Y.; Vinh-Thang, H.; Ramirez, A.A.; Rodrigue, D.; Kaliaguine, S. Membrane gas separation technologies for biogas upgrading. RSC Adv. 2015, 5, 24399–24448. [Google Scholar] [CrossRef]
- Kostrov, J.A.; Mostrova, G.B.; Ignatienko, T.I.; Arbashnikov, A.J.; Khutorski, B.I. Graviton hollow gas-separating fibre. Fibre Chem. 1987, 18, 479–481. [Google Scholar] [CrossRef]
- Abedini, R.; Omidkhah, M.; Dorosti, F. Hydrogen separation and purification with poly(4-methyl-1-pentene)/MIL 53 mixed matrix membrane based on reverse selectivity. Int. J. Hydrogen Energy 2014, 39, 7897–7909. [Google Scholar] [CrossRef]
- Nematollahi, M.H.; Dehaghani, A.H.S. CO2/CH4 separation with poly(4-methyl-1-pentyne) (TPX) based mixed matrix membrane filled with Al2O3 nanoparticles. Korean J. Chem. Eng. 2016, 33, 657–665. [Google Scholar] [CrossRef]
- Wolinska-Grabczyk, A.; Jankowski, A.; Sekuła, R.; Kruczek, B. Separation of SF6 from binary mixtures with N2 using commercial poly(4-methyl-1-pentene) films. Sep. Sci. Technol. 2011, 46, 1231–1240. [Google Scholar] [CrossRef]
- Mitsui Chemical, Inc. Available online: https://us.mitsuichemicals.com/index.htm (accessed on 22 May 2021).
- Pelzer, M.; Vad, T.; Becker, A.; Gries, T.; Markova, S.; Teplyakov, V. Melt spinning and characterization of hollow fibers from poly(4-methyl-1-pentene). J. Appl. Polym. Sci. 2021, 138, 49630. [Google Scholar] [CrossRef]
- Mohr, J.M.; Paul, D.R.; Mslna, T.E.; Lagow, R.J. Surface fluorination of composite membranes. Part I. Transport properties. J. Membr. Sci. 1991, 55, 131–148. [Google Scholar] [CrossRef]
- Puleo, A.C.; Paul, D.R.; Wong, P.K. Gas sorption and transport in semicrystalline poly(4-methyl-1-pentene). Polymer 1989, 30, 1357–1366. [Google Scholar] [CrossRef]
- Markova, S.Y.; Beckman, I.N.; Teplyakov, V.V. Diffusion of C1-C3 Alkanes in Semicrystalline Poly(4-Methyl-1-Pentene) as a Two-Phase Polymeric System. Int. J. Mem. Sci. Tech. 2017, 4, 28–36. [Google Scholar]
- Sridhar, S.; Veerapur, R.S.; Patil, M.B.; Gudasi, K.B.; Aminabhavi, T.M. Matrimid polyimide membranes for the separation of carbon dioxide from methane. J. Appl. Polym. Sci. 2007, 106, 1585–1594. [Google Scholar] [CrossRef]
- Donohue, M.D.; Minhas, B.S.; Lee, S.Y. Permeation behavior of carbon dioxidemethane mixtures in cellulose acetate membranes. J. Membr. Sci. 1989, 42, 197–214. [Google Scholar] [CrossRef]
- Yeom, C.K.; Lee, S.H.; Lee, J.M. Study of transport of pure and mixed CO2/N2 gases through polymeric membranes. J. Appl. Polym. Sci. 2000, 78, 179–189. [Google Scholar] [CrossRef]
- Markova, S.Y.; Pelzer, M.; Shalygin, M.G.; Vad, T.; Gries, T.; Teplyakov, V.V. Gas separating hollow fibres from Poly(4-methyl-1-pentene): A new development. Sep. Purif. Technol. 2022, 278, 119534. [Google Scholar] [CrossRef]




| Volumetric Polymer Feed (cm3/Rotation) | Extruder Rotation Speed (Rotations/min) | Spin Head Temperature (°C) | Specific Mass Throughput (g mm−2 h−1) | Winding Speed (m/min) |
|---|---|---|---|---|
| 0.16 | 13.13 | 280 | 103 | 25 |
| HF Sample | Process Conditions | Internal Diameter (µm) | External Diameter (µm) | Wall Thickness (µm) |
|---|---|---|---|---|
| Graviton HFs | 40 | 80 | 20 | |
| HF_0 | see Table 1 | 111 | 187 | 38 |
| HF_100_C | continuously drawn by 100% between unheated (20 °C) godets | 93 | 173 | 40 |
| HF_100_80 | continuously drawn by 100% at 80 °C godet temperature | 88 | 141 | 27 |
| Sample | XC | Tg, °C | Tm, °C | ΔHf, (J g−1) |
|---|---|---|---|---|
| Graviton HF | 0.54 | 30 | 235 | 33.5 |
| HF_0 | 0.32 | 21 | 227 | 20.0 |
| HF_100_C | 0.32 | 22 | 227 | 19.6 |
| HF_100_80 | 0.36 | 23 | 227 | 22.1 |
| Gas | CO2 | CH4 | N2 | Selectivity | Ref. | |
|---|---|---|---|---|---|---|
| Sample | Permeability Coefficient, Barrer * | CO2/CH4 | CO2/N2 | |||
| PMP film | 108 | 20.1 | 8.50 | 5.4 | 12.7 | [11] |
| PMP (Q) (Xc = 0.51; Tg = 30 °C) | 108 | 19.8 | 9.26 | 5.5 | 11.7 | [12] |
| PMP (S) (Xc = 0.61; Tg = 27 °C) | 85.0 | 14.0 | 6.74 | 6.1 | 12.6 | |
| PMP (A) (Xc = 0.76; Tg = 22 °C) | 74.0 | 13.0 | 5.93 | 5.7 | 12.5 | |
| PMP (H) (Xc = 0.68; Tg = 37 °C) | 82.0 | 14.2 | 6.68 | 5.8 | 12.3 | |
| PMP (2.5X) (Xc = 0.38; Tg = 40 °C) | 86.0 | 14.6 | 6.55 | 5.9 | 13.1 | |
| PMP (10X) (Xc = 0.20; Tg = 53 °C) | 68.0 | 10.5 | 5.02 | 6.5 | 13.5 | |
| PMP film (Xc = 0.69; Tg = 29.7 °C) | - | 14.0 | - | - | - | [13] |
| PMP Graviton HFs | 42.3 | 7.3 | 4.5 | 5.8 | 9.4 | This work |
| PMP HF_0 | 68.4 | 12 | 5.37 | 5.7 | 12.7 | This work |
| PMP HF_100_C | 67.1 | 12.2 | 5.26 | 5.5 | 12.8 | This work |
| PMP HF_100_80 | 48.7 | 8.1 | 3.62 | 6.0 | 13.5 | This work |
| CO2/CH4 Mixture Composition, % vol. | 100/0 | 90/10 | 27/73 | 10/90 | 0/100 |
|---|---|---|---|---|---|
| Q(CO2)·1012, m3 (STP)/(m2·s·Pa) | 24.1 | 24.6 | 21.8 | 25.2 | – |
| Q(CH4)·1012, m3 (STP)/(m2·s·Pa) | – | 3.9 | 4.1 | 3.9 | 3.8 |
| CO2/N2 Mixture Composition, % vol. | 100/0 | 27/73 | 15/85 | 12/88 | 0/100 |
|---|---|---|---|---|---|
| Q(CO2)·1012, m3 (STP)/(m2·s·Pa) | 24.1 | 21.6 | 22.8 | 23.5 | – |
| Q(N2)·1012, m3 (STP)/(m2·s·Pa) | – | 1.6 | 1.7 | 1.8 | 1.6 |
| Composition of the Mixture, % vol. | 90/10 | 27/73 | 15/85 | 12/88 | 10/90 | αid |
|---|---|---|---|---|---|---|
| Mixture Selectivity (α) | ||||||
| CO2/CH4 | 6.3 | 5.3 | – | – | 6.4 | 6.4 |
| CO2/N2 | – | 13.3 | 13.5 | 13.3 | – | 15.2 |
| Gas | EP, kJ/mol | ||
|---|---|---|---|
| HF_0 | HF_100_C | HF_100_80 | |
| CO2 | 16.7 | 18.4 | 18.8 |
| CH4 | 29.3 | 30.2 | 33.9 |
| N2 | 29.2 | 29.6 | 32.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dukhov, A.V.; Pelzer, M.; Markova, S.Y.; Syrtsova, D.A.; Shalygin, M.G.; Gries, T.; Teplyakov, V.V. Preparation of Hollow Fiber Membranes Based On Poly(4-methyl-1-pentene) for Gas Separation. Fibers 2022, 10, 1. https://doi.org/10.3390/fib10010001
Dukhov AV, Pelzer M, Markova SY, Syrtsova DA, Shalygin MG, Gries T, Teplyakov VV. Preparation of Hollow Fiber Membranes Based On Poly(4-methyl-1-pentene) for Gas Separation. Fibers. 2022; 10(1):1. https://doi.org/10.3390/fib10010001
Chicago/Turabian StyleDukhov, Anton V., Martin Pelzer, Svetlana Yu. Markova, Daria A. Syrtsova, Maxim G. Shalygin, Thomas Gries, and Vladimir V. Teplyakov. 2022. "Preparation of Hollow Fiber Membranes Based On Poly(4-methyl-1-pentene) for Gas Separation" Fibers 10, no. 1: 1. https://doi.org/10.3390/fib10010001
APA StyleDukhov, A. V., Pelzer, M., Markova, S. Y., Syrtsova, D. A., Shalygin, M. G., Gries, T., & Teplyakov, V. V. (2022). Preparation of Hollow Fiber Membranes Based On Poly(4-methyl-1-pentene) for Gas Separation. Fibers, 10(1), 1. https://doi.org/10.3390/fib10010001







