Highlights
What are the main findings?
- CaCO3 nanopowder (CCNP) was chelated using citric acid (CA) and tartaric acid (TA).
- The chelation reaction produced wood coatings with improved thermal properties.
What are the implications of the main findings?
- The coatings promoted char formation and inorganic residues under O2.
- TA-based coating delayed combustion by limiting the surface temperature to 200 °C.
Abstract
This study presents a sustainable strategy for improving the thermal properties of pine wood through the application of calcium carbonate nanopowder (CCNP) chelated with polycarboxylic acids (citric acid (CA) and tartaric acid (TA)) as coatings. The chelation reaction was confirmed by the detection of carbon dioxide (CO2) gas. CCNP was characterized using microscopy and particle size analysis. The formation of crystalline calcium citrate and calcium tartrate was verified using FTIR and Raman spectroscopies, and XRD analysis. Wood treatment was conducted using different volumetric ratios of CA and TA. The CA-TA-treated (coated) wood blocks achieved the highest mass gain after treatment of around 89%, while the pure TA treatment exhibited enhanced leaching resistance, maintaining around 69% mass gain after leaching test. TGA conducted under oxidative (air) conditions showed that the coatings promoted char formation and produced inorganic residues from 6.4% to 7.8%, with the control resulting in negligible residual mass. Flame retardancy tests showed that the chelated coatings effectively delayed combustion and inhibited heat transfer, with the TA treatment showing improved flame retardancy performance by limiting the surface temperature to ~200 °C after 60 s of exposure, as compared to >550 °C for the control.
1. Introduction
The increasing demand for environmentally friendly and sustainable construction materials featuring outstanding fire safety and durability standards has given rise to interest in using wood and lignocellulosic biocomposites due to their carbon sequestration abilities, aesthetic versatility, and high strength-to-weight ratios [1,2,3]. The inherent flammability of wood, in addition to its susceptibility to thermal degradation and dimensional instability caused by variations in moisture levels, presents an important challenge to its widespread adoption in high-rise and high-risk construction applications [4,5]. Historically, the preservation and fire resistance of wood have mainly relied on halogenated chemicals, boron salts, phosphorus-containing moieties, and heavy metal-based preservatives [6,7,8]. Although effective, these traditional methods are under increasing regulations due to their persistence in the environment, effects on human health, and the production of harmful volatiles during combustion [9]. Therefore, it is imperative to satisfy the urgent need for developing wood modification strategies involving non-toxic, renewable, and bio-based materials that provide enhanced thermal properties and flame retardancy.
Polycarboxylic acids are organic compounds with at least two carboxylic acid (−COOH) groups, such as citric acid (CA, tricarboxylic acid) and tartaric acid (TA, dicarboxylic acid). The production of CA has evolved from conventional petroleum-derived synthesis to fermentation using microorganisms, therefore positioning it as one of the most important biotechnological products globally. A recent study has shown that fermentation using Aspergillus niger PD-66 can be employed for the bioconversion of carbohydrate substrates into CA [10]. Citrus processing produces around 146 million tons of waste every year, generating a considerable renewable feedstock for CA production [11]. In addition to citrus waste, additional substrates have also been studied, including orange and pineapple waste and glycerol waste from biodiesel production [10,12]. A recent study showed that a closed-loop production process utilizing engineered marine cyanobacterium (Synechococcus sp. PCC 7002) for the direct photosynthetic conversion of CO2 to CA was feasible [13]. This advancement achieves carbon neutrality through the use of atmospheric CO2 rather than relying on biomass feedstocks, indicating an advancement towards the production of more sustainable specialty chemicals and other value-added products [14,15,16]. CA is utilized in the food, pharmaceutical, chemical, and metallurgical industries due to its non-toxic properties and its chelating and sequestering abilities [17]. CA has also been used as a crosslinker to enhance the stability and fire retardancy of wood through modifications with phosphorylated sucrose stearate [18]. Tartaric acid (TA) has also been studied recently due to its ability to effectively act as a chelating agent to sequester metal ions, particularly in wastewater treatment and environmental remediation [19,20]. TA exhibits a relatively restricted distribution in higher plants, accumulating at high concentrations mostly within the Vitaceae family which includes grapes [21]. TA is categorized as a “specialized primary metabolite” derived from the degradation of ascorbic acid via carbohydrate metabolic pathways without any identified primary metabolic function. Although small amounts can be observed in other fruits (such as apples [22] and mangoes [23]), the grape berry acts as the main natural source of TA. The wine industry produces significant amounts of byproducts containing TA, such as calcium tartrate deposits (wine stone), grape pomace, fermentation yeast residues, and vinasse (byproduct of distillation). A study involving the wine industry of Moldova has shown extensive technological approaches for the extraction of TA from waste materials with minimal expenses. The procedure involves the extraction and purification of calcium tartrate deposits and subsequent conversion into TA via acidification or decalcification processes [24]. A corresponding diethyl ester with reactive hydroxyl groups produced from TA has also been used as a cross-linker to react with phosphorus-containing compounds, serving as a monomer for the synthesis of different flame-retardant (FR) polymers and oligomers through step-growth transesterification [25]. Calcium carbonate (CaCO3) is a common compound found in nature and is widely used in different applications, such as construction, paper, paints, health supplements, and most importantly, as an antacid [26]. Calcium is an important part of CaCO3 and an essential component in the mineralization of bones and shells in some microorganisms and animals [27,28,29,30]. CaCO3 has also been extensively studied due to its excellent thermal properties and potential as bio-based fire retardant [31,32,33]. In terms of sustainability, eggshell waste (consisting of over 90% CaCO3), constitutes the most readily available renewable source of CaCO3 with around 95% of this waste currently disposed of in landfills worldwide [34,35].
In this study, CA and TA were utilized as polycarboxylic acids in chelating calcium from CaCO3 and as potential cross-linkers to bind Ca2+ to wood. The study also focuses on the effects of the resulting calcium citrate and calcium tartrate-based cross-linked chelate coating on the thermal properties and fire retardancy of wood (loblolly pine). The specific objectives of this study were (1) to prepare coating materials through CA and TA chelation of Ca2+ in the form of calcium carbonate nanopowder (CCNP); (2) to characterize the chemical properties of the starting materials and the coatings produced through Laser diffraction-based particle size analysis (PSA), Scanning Electron Microscopy (SEM), Attenuated Total Reflectance-Fourier Transform Infrared Spectroscopy (ATR-FTIR), Raman Spectroscopy, X-ray Diffraction (XRD), Gas Chromatography-Thermal Conductivity Detector (GC-TCD); (3) to characterize the thermal properties of the starting materials and the coatings produced through Thermogravimetric Analysis (TGA); (4) to treat pine wood samples using vacuum impregnation; and lastly (5) to characterize the thermal properties and fire retardancy of the coated (treated) pine wood samples through TGA and a modified UL 94 flammability test standard method, respectively.
2. Materials and Methods
2.1. Materials
Citric acid (C6H8O7, anhydrous, ACS 99.9%), and tartaric acid (C4H6O6, anhydrous, ≥99%) were acquired from J.T. Baker Chemical Co. (Phillipsburg, NJ, USA). CCNP (CaCO3, 98%) was purchased from US Research Nanomaterials, Inc. (Houston, TX, USA). The chemical reagents were used as received without additional purification. Deionized water (18.2 MΩ·cm resistivity at 22.5 °C) was used for all experiments. Loblolly pine free of defects was initially divided into sticks and subsequently into end-matched blocks measuring approximately 18 mm × 18 mm × 5 mm (tangential (T) × radial (R) × longitudinal (L)). Prior to wood treatment, the pine wood blocks were subjected to oven drying at 105 °C for 24 h.
2.2. Chelation
CCNP was chelated and converted to calcium citrate (Ca-citrate) and calcium tartrate (Ca-tartrate) in the presence of CA (Equation (1)) and TA (Equation (2)), respectively.
3CaCO3(s) + 2C6H8O7(aq)
→ Ca3(C6H5O7)2(s) + 3H2O(l)
+ 3CO2(g) ↑
CaCO3(s) + C4H6O6(aq)
→ CaC4H4O6(s) + H2O(l) + CO2(g)
↑
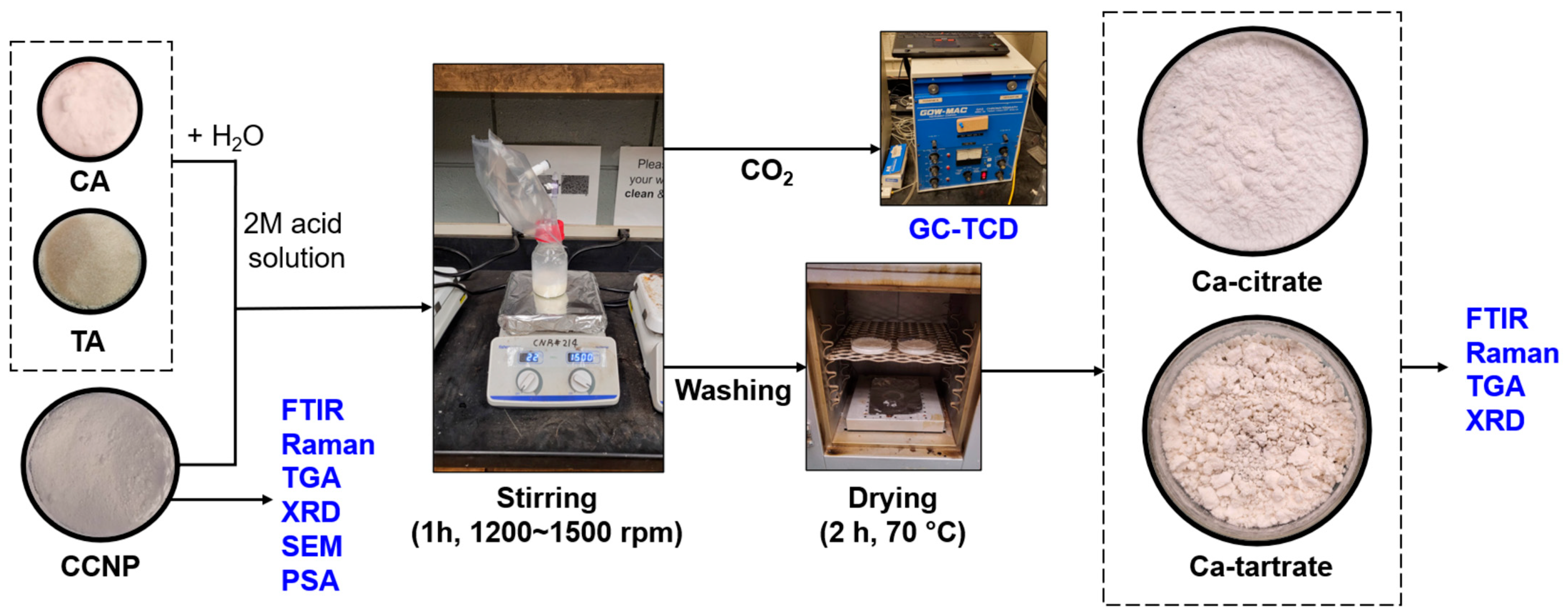
The preparation of both Ca-citrate and Ca-tartrate was carried out by weighing 5 g of CCNP and placing it in a 250 mL culture bottle with a cap that was connected to a Tedlar gas bag. Then, 40 mL of either 2M CA or 2M TA was slowly added to each bottle containing CCNP [36]. Due to the difference in viscosity between the solutions reacted with CA and TA, the mixture for Ca-citrate was stirred at 1200 rpm and Ca-tartrate at 1500 rpm, both for 1 h to ensure that no additional CO2 generation was observed, signifying a complete chelation reaction. The formed products were washed three times with 100 mL of water to remove unreacted ions and excess acids and then oven-dried at 70 °C for 2 h. The products were stored in air-tight gas bags prior to further analysis. Figure 1 shows the process diagram for chelating CCNP with CA or TA to produce the coating materials.
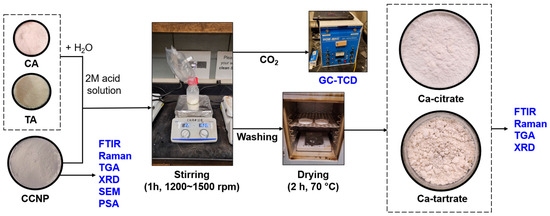
Figure 1.
Process diagram for CCNP chelation using CA or TA.
2.3. Particle Size Analysis (PSA)
The particle size distribution of CCNP was determined using a Bettersizer 2600 particle size analyzer equipped with a BT-802 wet dispersion module (Costa Mesa, CA, USA). For sample preparation, roughly 1.0 g of sample was dissolved in 45 mL of water inside a centrifuge tube. This stock solution was diluted further by taking at least 1 mL from the stock solution and transferring to another centrifuge tube and adding 45 mL of water-ethanol solution (1:1 v/v) and ultrasonicated (15 W) for 10 min. Drops of the resulting suspension was added into the analyzer reservoir while being stirred at 3000 rpm until 5%–15% obscuration was reached. Each sample went through an automated background measurement and data acquisition with 64 scans per run. The particle size distribution was determined using Mie scattering theory, that uses a refractive index of 1.596 for CCNP and 1.333 for water. The analysis was conducted in triplicate, with a repeatability of ≤0.5% for D50 values.
2.4. Scanning Electron Microscopy (SEM)
SEM was employed to investigate the microstructural surface morphology of CCNP with a Zeiss Supra 35VP (Oberkochen, Germany), equipped with a fixed emission gun and a secondary electron (SE2) backscatter detector. Prior to SEM analysis, dried powdered CCNP samples were produced and coated with approximately 10 nm of gold. The analysis was conducted with a 30 µm aperture size, a 10 mm working distance, and a 10 kV accelerating voltage in low vacuum mode under nitrogen pressure.
2.5. Attenuated Total Reflectance-Fourier Transform Infrared Spectroscopy (ATR-FTIR)
The chemical composition shift resulting from the chelation of Ca2+ from CCNP by CA and TA (coating materials) was confirmed using ATR-FTIR spectroscopy. The FTIR analysis utilized a Thermo Fisher Scientific Nicolet iS5 FTIR spectrometer (Madison, WI, USA) equipped with a ZnSe ATR crystal. Samples were placed on the ATR crystal and subjected to 64 successive scans at a resolution of 4 cm−1. The obtained spectral data was averaged and underwent baseline correction and normalization using Thermo Fisher Scientific OMNIC v9.8 software.
2.6. Raman Spectroscopy
The CCNP and coating material samples were also analyzed using a Thermo Fisher Scientific DXR3 Flex Raman spectrometer equipped with a 5.0 mW laser at 532 nm and an aperture size of 50 µm (Madison, WI, USA). Samples were placed on the sample mount and initially focused by using the built-in optical microscope (µView LMPlanFI 20X/0.40). The laser was initialized and the sample was subjected to 32 successive scans within a Raman shift range of 50–3500 cm−1 at a resolution of 5 cm−1. The obtained spectral data were baseline corrected and normalized using Thermo Fisher Scientific OMNIC v9.8 software.
2.7. X-Ray Diffraction (XRD)
XRD analysis was conducted to determine the crystallinity and phase composition of CCNP and coating material samples using a Siemens D5000 diffractometer (Munich, Germany) under Cu Kα radiation (λ = 0.15 nm) with a step size of 0.02° and an angular range scan (2θ range) of 2–90°.
2.8. Gas Chromatography-Thermal Conductivity Detector (GC-TCD)
The gas produced was captured in a Tedlar bags and analyzed using a GOW-MAC Series 350 gas chromatograph (Bethlehem, PA, USA) equipped with thermal conductivity detector (GC-TCD) to verify both the CA and TA chelation of CCNP through CO2 generation. Gas samples were injected into the sampling port to a Porapak N packed column (2.0 mm ID × 1.8 m) at room temperature with helium as carrier gas. The test was performed in triplicate.
2.9. Wood Coating Treatment
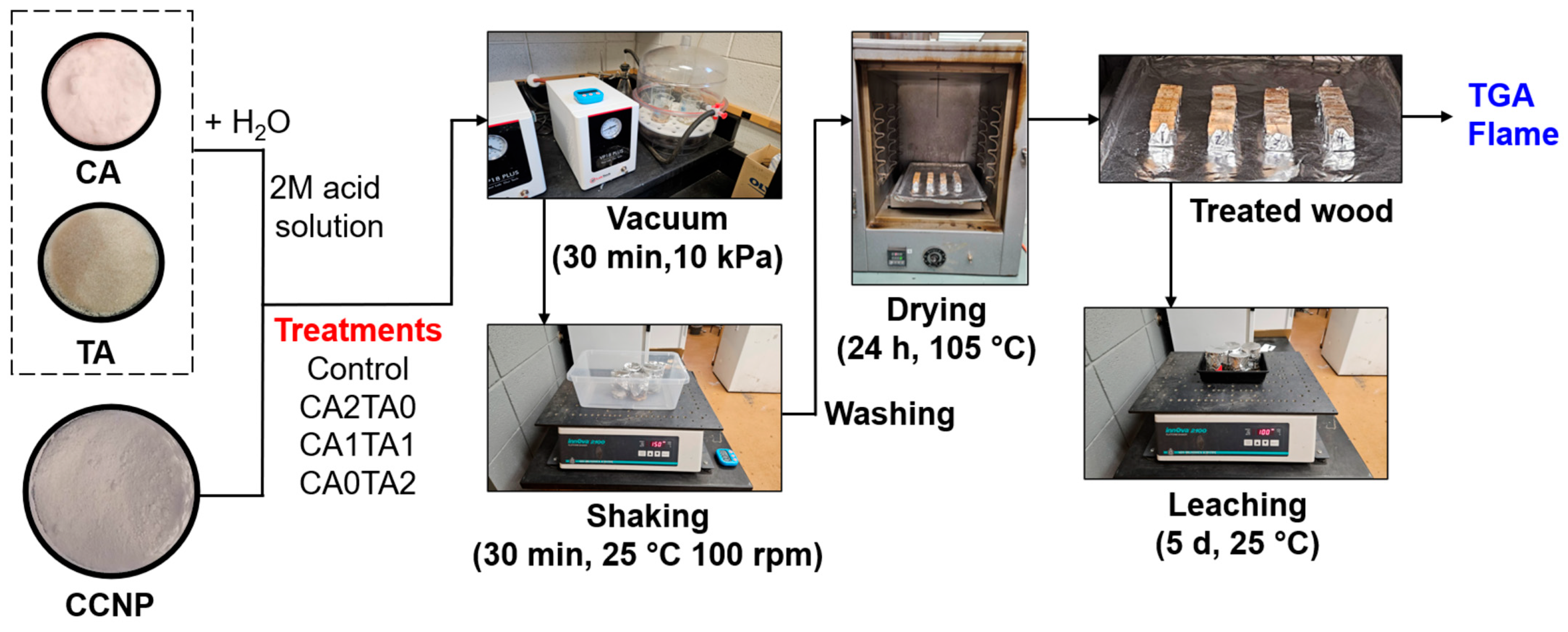
Wood treatment was carried out by placing 10 pine wood blocks in a 250 mL beaker for each treatment. The treatment solutions used followed different volumetric ratios for CA and TA and fixed CCNP amount: (1) control (40 mL water); (2) CA2TA0 (40 mL 2M CA and 5 g CCNP); (3) CA1TA1 (20 mL 2M CA + 20 mL 2M TA and 5 g CCNP); and (4) CA0TA2 (40 mL 2M TA and 5 g CCNP). Each treatment solution was immediately added to a corresponding beaker containing the wood blocks and vacuum treated at 10 kPa for 30 min. Afterwards, the beakers were covered and placed on a New Brunswick Scientific Innova 2100 platform shaker (Edison, NJ, USA) preset to 100 rpm for another 30 min. The ambient temperature was maintained between around 25 °C during the treatment. The control and treated wood blocks were washed with water and then oven-dried at 105 °C for at least 24 h or until a stable mass was attained. The mass gain of the pine wood blocks after coating treatment was calculated using Equation (3).
where mb and ma are the mass of the pine wood blocks oven-dried at 105 °C before and after the coating treatment, respectively. Figure 2 shows the process diagram for wood coating treatment using chelated CCNP.

Figure 2.
Process diagram for wood coating treatment of wood pine blocks.
2.10. Leaching Test
Leaching test was performed on control, CA2TA0-treated, CA1TA1-treated, and CA0TA2-treated wood blocks in accordance with a modified AWPA E11-16 standard method [37]. The wood blocks from each treatment were soaked in water at a wood-to-water volume ratio of 1:15. The beakers containing the wood blocks and water were sealed and fixed on a New Brunswick Scientific Innova 2100 platform shaker (Edison, NJ, USA) preset to 100 rpm for continuous agitation. The ambient temperature was sustained around 25 °C during the test. The leaching test involved replacing the water once every 24 h for 5 d. After the leaching period, the wood blocks were oven-dried at 105 °C for at least 24 h or until a stable mass was attained. The leaching test results were incorporated into the mass gain results after treatment to obtain the final mass of the pine wood blocks. The leaching test was performed in triplicate.
2.11. Thermogravimetric Analysis (TGA)
The thermal properties of the control, CA2TA0-treated, CA1TA1-treated, and CA0TA2-treated wood blocks were assessed using a PerkinElmer TGA 7 (Waltham, MA, USA) instrument. The wood blocks were ball-milled to produce fine powdered samples, wherein 4–5 mg were placed into platinum pans. For each run, the sample was heated from 30 °C to 800 °C at a rate of 20 °C/min under air or nitrogen with a flow rate of 20 mL/min. The collection and analysis of data was performed with Pyris v13.3 software (PerkinElmer). TGA was also utilized for analyzing CCNP, starting materials (CA and TA), and coating materials (Ca-citrate and Ca-tartrate) under the same conditions except that the temperature programming was set at 30–900 °C. TGA was conducted in triplicate.
2.12. Flame Retardancy Test
The flame retardancy of the control, CA2TA0-treated, CA1TA1-treated, and CA0TA2-treated wood blocks was evaluated using a modified UL 94 flammability standard test method, outlined in the literature [38,39]. Each wood block sample was subjected to the flame of an alcohol burner maintained at around 900 °C (monitored by a thermocouple) and ignited for 20 s, after which the flame source was withdrawn. The surface temperature variations in the ignited wood block were recorded with a Testo 868S thermal imaging camera (Titisee-Neustadt, Germany), positioned, and stabilized 0.45 m from the sample. The combustion process was recorded, with surface temperature measured at 10-s intervals. The recording ended with the extinction of the flame in order to record the time of flame-out. The obtained temperature data was utilized to generate a temperature profile for each experimental run. The test was performed in five replicates.
3. Results and Discussion
3.1. Structural Characterization of CCNP
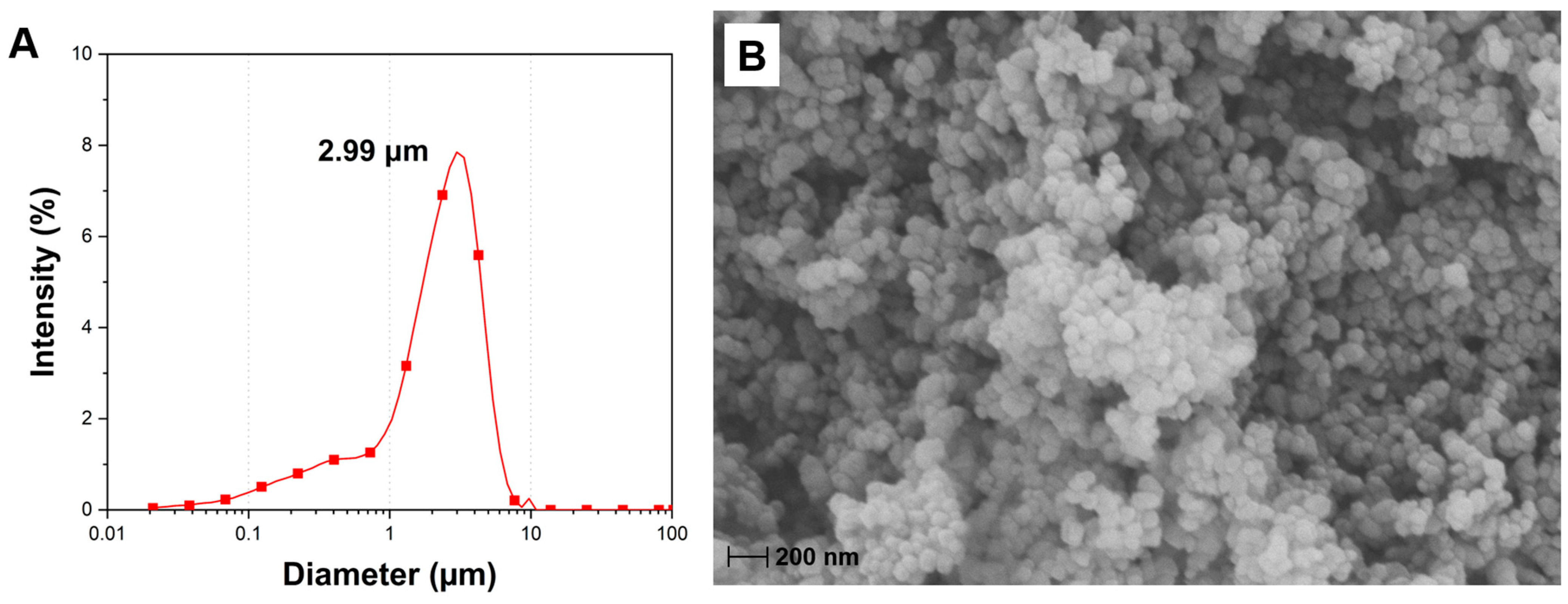
The particle size distribution of CCNP is shown in Figure 3A. The analysis showed particle size ranging from 21 nm to 9.7 μm, a prominent peak at 2.99 μm, and a volume weighted mean (D [4,3]) of 2.3 μm. SEM analysis was performed to visualize the microstructural and surface morphology of CCNP and to verify the particle size results obtained from PSA (Figure 3B). The SEM micrograph shows that the CCNP consists of near-spherical nanoparticles. The individual particles, visually approximated to be in the nanoscale range of 20–100 nm were aggregated into larger and dense clusters. This agglomeration leads to surface roughness due to the compact arrangement of the constituent nanoparticles and results from strong Van der Waals forces and hydrogen bonding interactions typical of carbonate particles, particularly in the presence of ambient moisture [40,41]. Comparing the SEM micrograph with the PSA result suggests a discrepancy. SEM verifies the presence of particles within the nanometer range, although PSA indicates a mean diameter in the micrometer range. The finding implies that laser diffraction-based PSA measured the hydrodynamic diameter of the stable aggregates instead of the individual nanoparticles [42]. Nanoscale CaCO3 particles can be advantageous for subsequent reactions due to higher surface area and availability of superficial functional groups, as compared to larger CaCO3 particles. A research study reported a specific surface area of 6.99 m2/g for CaCO3 nanopowder and 4.26 m2/g for CaCO3 micropowder [43]. Another study comparing the efficiencies of both CaCO3/CMC (carboxymethyl chitosan) microspheres and nanospheres reported a higher cumulative release (%) over time for the nanospheres compared to the microspheres, owing to the higher encapsulation efficiency and drug loading ability of the nanoparticles due to its improved surface area [44].
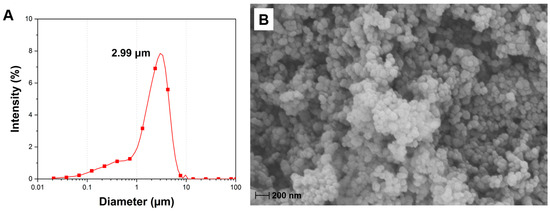
Figure 3.
(A) Particle size analysis (D [4,3] = 2.3 μm) and (B) SEM micrograph of CCNP (25,000× magnification).
3.2. FTIR and Raman Spectroscopy
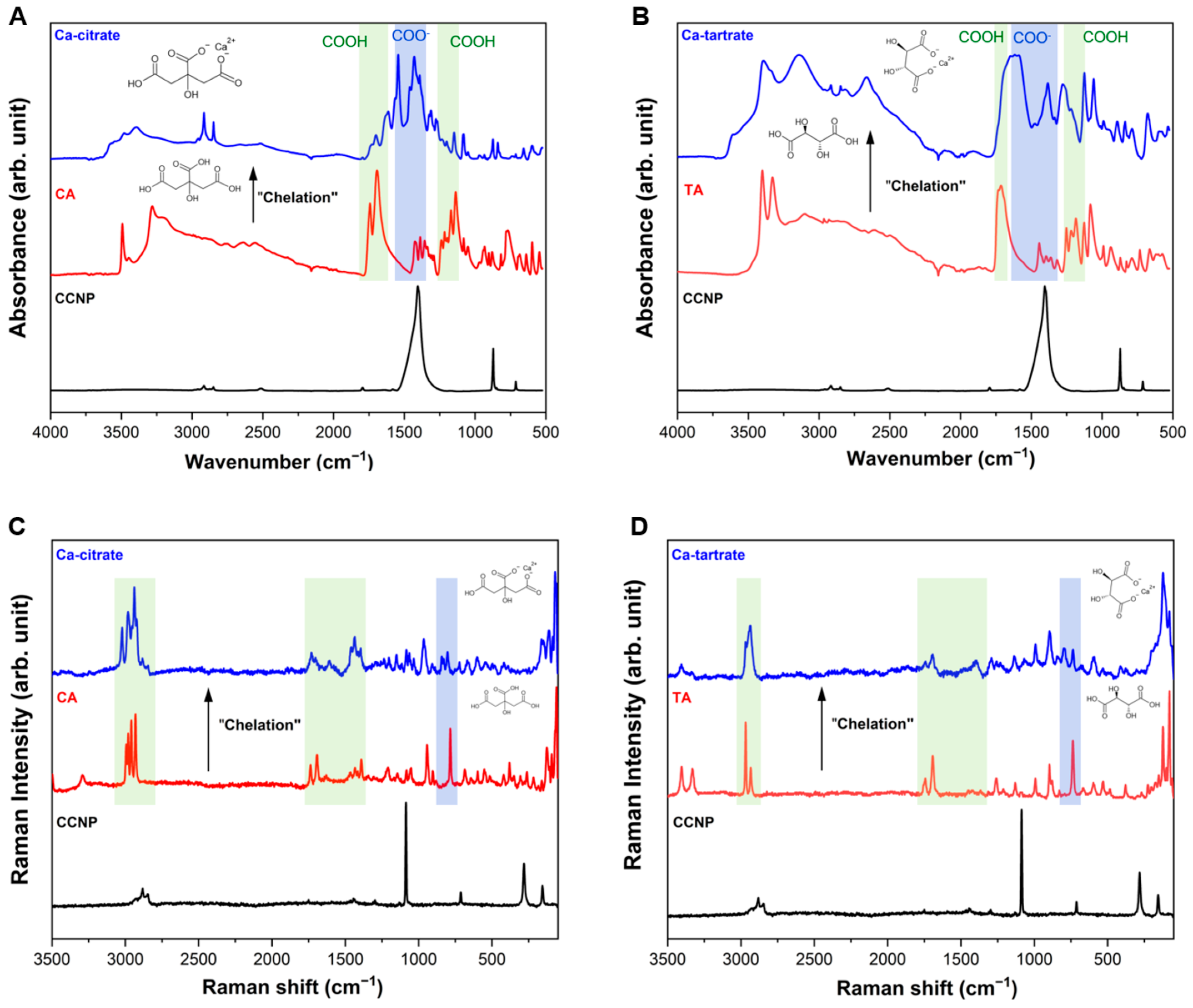
The successful chelation of Ca2+ from CCNP using CA and TA to form Ca-citrate and Ca-tartrate, respectively, was confirmed via FTIR and Raman spectroscopies. The FTIR spectra provided evidence of the transformation of the carboxylic acid (−COOH) groups into carboxylate ligands and subsequently forming a metal-ion chelate [45]. Figure 4A shows the FTIR spectra and chelation reaction CCNP with CA. The spectrum of CA (red) exhibited a characteristic and intense absorption band at approximately 1650–1780 cm−1 (C=O) and a symmetric stretching vibration and around 1135–1250 cm−1 due to O−H bend in COOH (shaded green) [46,47]. Meanwhile for Ca-citrate (blue), the same peaks and bands disappeared due to the deprotonation to COO− and subsequent chelation to Ca2+, which corresponds to the appearance of a new band with two distinct peaks (1460–1580 cm−1) resulting from the strong coupling effect of the antisymmetric stretching vibration characterized by high infrared activity (shaded blue) [46]. A similar pattern was observed in the formation of Ca-tartrate (Figure 4B). The TA spectrum (red) shows a carbonyl stretch at around 1650–1760 cm−1 and absorption bands in the 1130–1260 cm−1 range, corresponding to protonated O−H in COOH (shaded green). The two broad bands observed in Ca-tartrate (blue) at around 1350–1640 cm−1 indicate complex coordination involving deprotonation (COO−) and bidentate binding typical of calcium-carboxylate structures (shaded blue) [48]. For CCNP (black in both Figure 4A,B), the FTIR spectral result displays the characteristic sharp vibrational modes of calcite, most notably the carbonate asymmetric stretch which is obscured in the product spectra indicating high conversion. Distinct absorption bands appeared at around 1450 cm−1 corresponding to C−O stretching in the carbonate ion, while 870 cm−1 is associated with C−O bending vibrations [49].
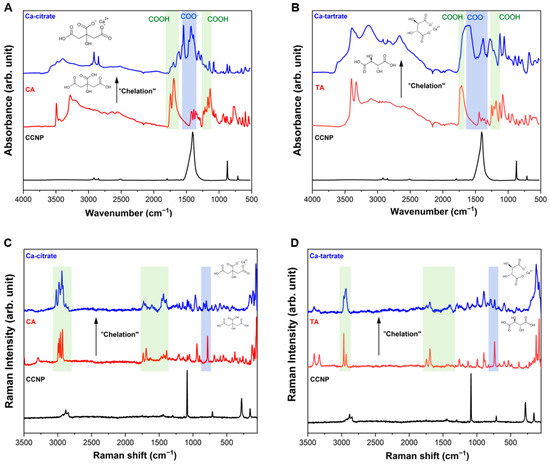
Figure 4.
FTIR spectra of (A) CCNP chelation using CA forming Ca-citrate and (B) CCNP chelation using TA forming Ca-tartrate. Raman spectra of (C) CCNP chelation using CA forming Ca-citrate and (D) CCNP chelation using TA forming Ca-tartrate.
Raman spectroscopy was employed to validate the FTIR results and to investigate the changes in structure, polarity, and crystallinity of the chelate products. The Raman spectra for both Ca-citrate (Figure 4C) and Ca-tartrate (Figure 4D) confirms the presence of the carboxylate moiety in the chelate products (shaded green), exhibiting an increase in band intensity between 1370 and 1470 cm−1 and an accompanying reduction between 1670 and 1790 cm−1. The shift in the C−C and C−O stretching modes within the fingerprint region, relative to the free acids, indicates crystal lattice reorganization following calcium coordination [50]. Coordination-induced shifts in polarization often eliminate the carboxylate-associated bands (Eg symmetry COO bending mode, shaded blue) at around 700–750 cm−1 region, and can increase the lattice or coordination properties at lower frequency compared to the free acids [51,52]. In both figures, the spectra of the chelate products (blue) differ from their corresponding starting materials (red) in the shaded green carboxylate (COO−) regions (1350–1777 cm−1 and 2875–3060 cm−1), indicating transformation into new coordination bonds. The changes observed using Raman spectroscopy were consistent with the interpretation obtained from FTIR spectroscopy, showing the conversion of COOH in free acids into Ca-coordinated carboxylate groups. For reference, CCNP (black) exhibits a pronounced symmetric stretching bands of the carbonate ion at 1086 cm−1 and 710 cm−1, corresponding to the v1 and v4 modes of calcite, respectively [53]. Minor bands were also observed at around 280 cm−1 and 140 cm−1, which corresponds to Ca2+ [54].
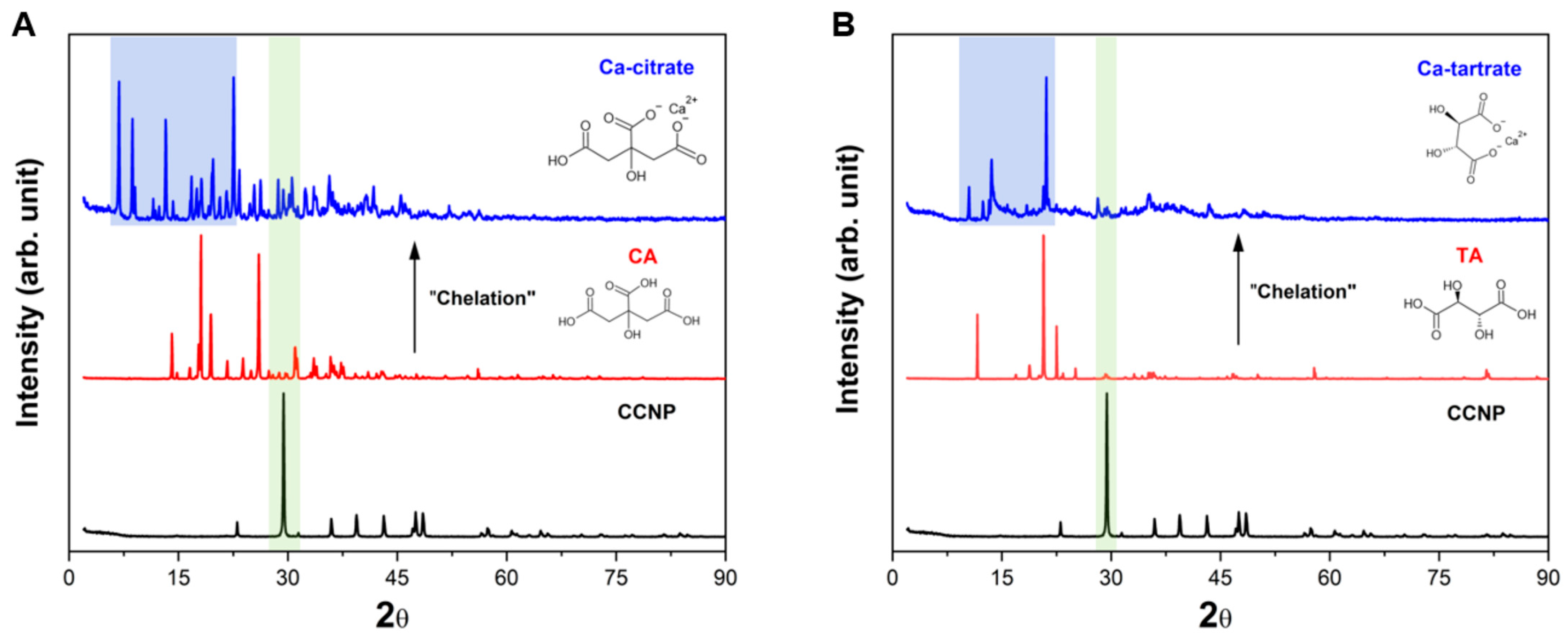
3.3. X-Ray Diffraction (XRD)
The crystalline properties and phase identification of CCNP and the resulting chelation products with CA and TA were analyzed by XRD. The XRD pattern for the CCNP precursor (black, Figure 5A,B) exhibited distinct sharp peaks, indicating a high degree of crystallinity. The major peak observed at 29.4° was characteristic of the (104) plane of calcite (calcium carbonate). Additional peaks were also observed at around 23.0°, 36.0°, 39.4°, 43.2°, 47.5°, and 48.5° corresponding to the (102), (110), (113), (202), (108), and (116) hkl planes of the rhombohedral calcite phase, respectively [55]. No distinct peaks for vaterite or aragonite polymorphs were detected, confirming that the CCNP precursor was calcite [56], verifying the results from FTIR and Raman spectroscopy.
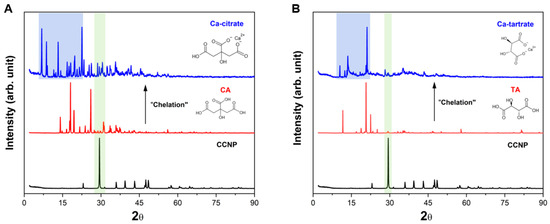
Figure 5.
XRD patterns of (A) CCNP chelation using CA forming Ca-citrate and (B) CCNP chelation using TA forming Ca-tartrate.
Figure 5A shows the transformation of CCNP chelated with CA into calcium citrate. The characteristic calcite peak of CCNP (29.4°) disappeared (shaded green) following chelation with CA, indicating the conversion of carbonate and subsequent release as carbon dioxide (Equation (1)). The Ca-citrate product (blue) exhibits a new crystalline phase (triclinic) characterized by strong diffraction peaks in the low-angle region at 2θ < 20°. Significant peaks in this region have been identified at 2θ = 6.8°, 8.5°, 11.8°, 13.2°, and 19.5° (shaded blue), including a distinct peak at 22.5°, in agreement with the formation of calcium citrate tetrahydrate [36,57]. Figure 5B shows the chelation of CCNP with TA. Similar to the chelation with CA, the reaction with TA leads to the disappearance of the calcite (104) peak at 29.4° (shaded green), confirming the conversion of carbonate into carbon dioxide (Equation (2)). The resulting Ca-tartrate XRD pattern (blue) shows a distinctive pattern of diffraction peaks, in contrast with both the CCNP (black) and the TA (red). Significant peaks were observed at 2θ = 10.5°, 13.6°, 17.0°, and 21.1° (shaded blue). These findings agree with the literature results regarding calcium tartrate tetrahydrate, which generally exhibits an orthorhombic crystal structure [58]. The sharp intensity of the peaks indicates that the chelation process produced highly crystalline Ca-tartrate. The hydration of both Ca-citrate and Ca-tartrate products was due to the incorporation of water into the crystal lattice during the chelation reaction.
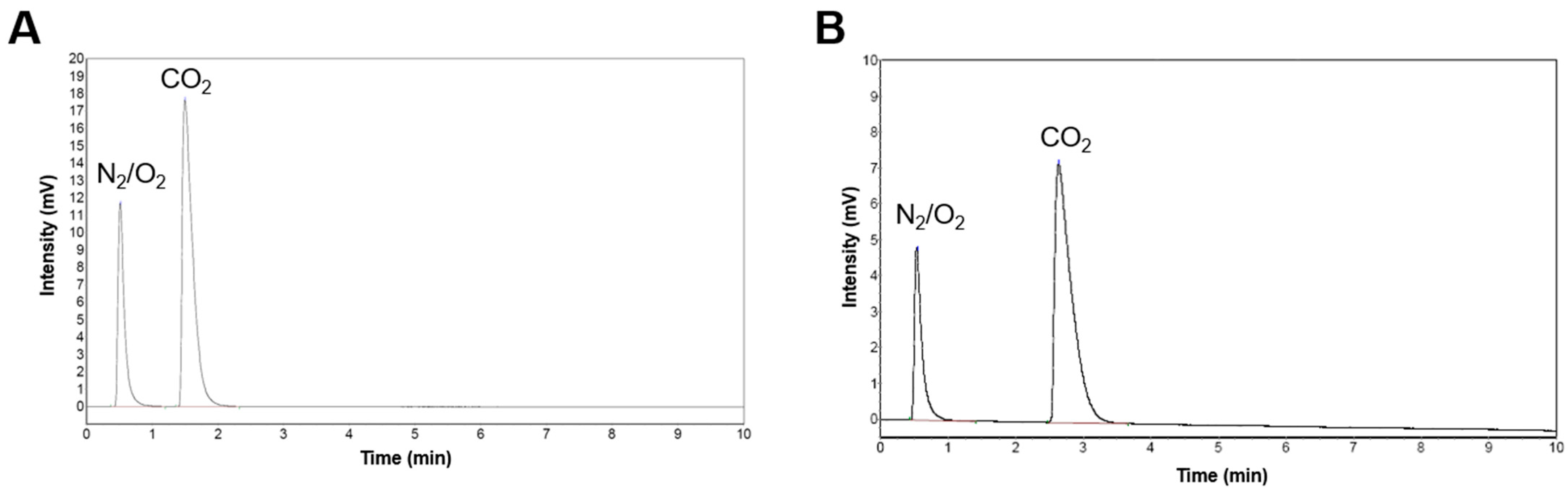
3.4. Gas Chromatography-Thermal Conductivity Detector (GC-TCD)
To further confirm the chelation reaction, the gas produced during the process was collected and analyzed by GC-TCD. The results show the formation of CO2 as the gaseous byproduct in both reactions involving CA and TA. In both chromatograms, the second peak in both chromatograms (Peak 2) was identified as CO2. The chelation of CCNP using CA (Figure 6A) produced CO2 and was eluted at a retention time of 1.5 min and exhibiting a relative abundance of 71% (Area %). Meanwhile, for the chelation of CCNP using TA (Figure 6B), the CO2 peak eluted at Rt of 2.6 min, which constitutes 77% of the total peak area. The difference in Rt between the two chelation reactions shows slight variations in chromatographic conditions, such as operating temperatures (oven temperature) or carrier gas flow rates, on different days as evidenced by the corresponding small displacement of the air (N2/O2) peak. This follows the chelation reaction in which a H+ from the carboxyl groups of CA or TA protonate the carbonate groups of CCNP, forming a weak and unstable carbonic acid, promoting the formation and subsequent release of CO2 in aqueous conditions. The detection of CO2 verifies the conversion of carbonate through chelation, demonstrating the reactivity of both CA and TA with CCNP.
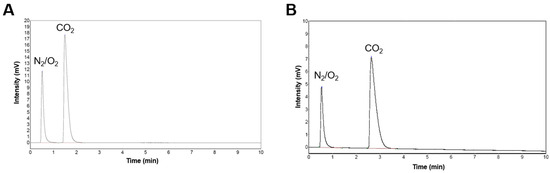
Figure 6.
Gas chromatograms of (A) the gas product of CCNP chelation using CA and (B) using TA.
3.5. Wood Treatment
3.5.1. Coating Formation and Mass Gain
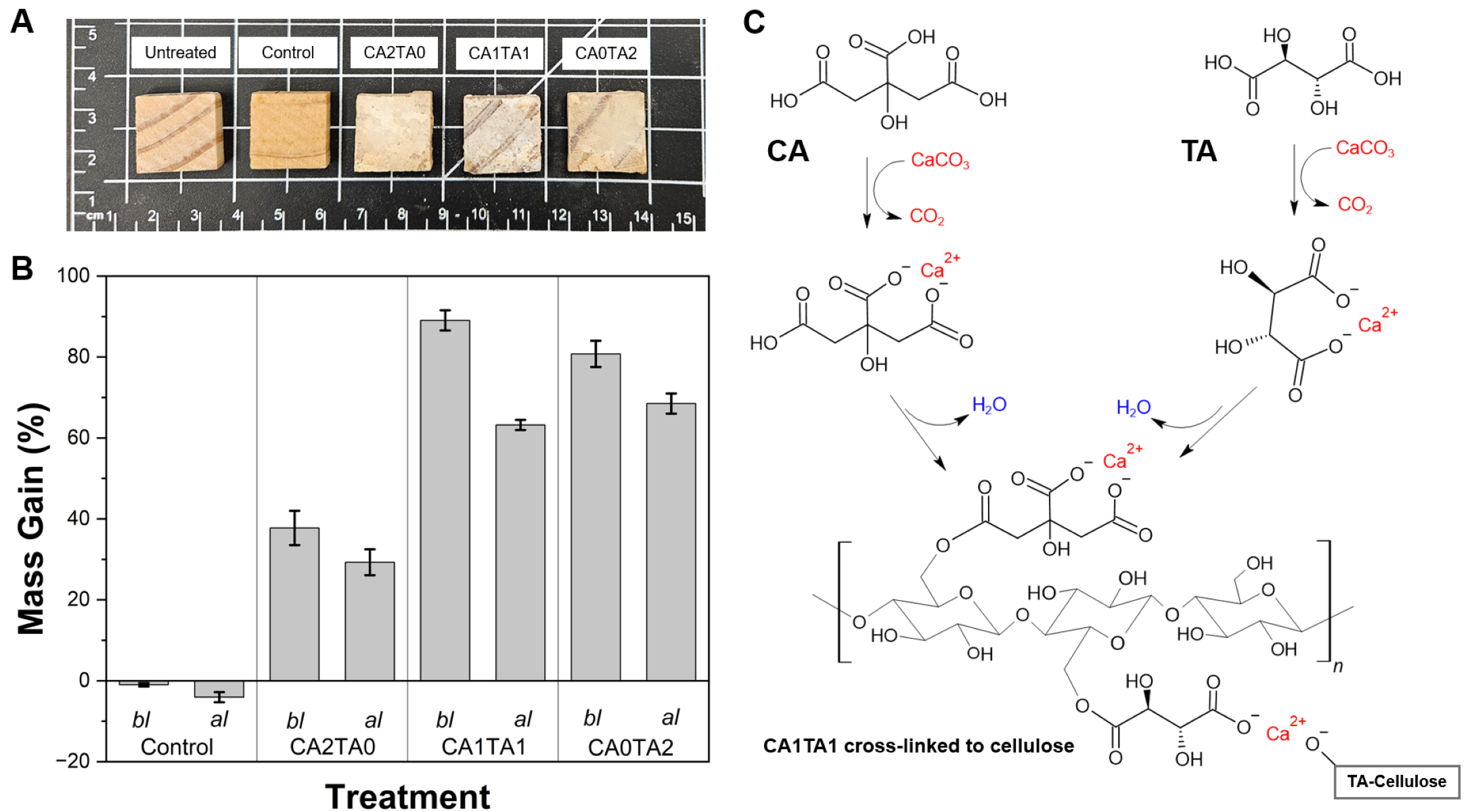
The macroscopic morphology of the pine wood blocks after vacuum-impregnation and chelation reaction of CCNP with the polycarboxylic acids is shown in Figure 7A. Besides facilitating the penetration of the Ca2+ and the carboxylate ions in solution into the wood structure, vacuum treatment also served as a means to push the equilibrium reaction forward by continuously removing CO2 during the chelation process. The untreated and control (water-treated) samples exhibited the light brown coloration of natural pine (visible grain patterns and an absence of surface deposits). On the other hand, the wood blocks subjected to different chelation solutions (CA2TA0, CA1TA1, and CA0TA2) exhibited color and surface changes. A dark white precipitate was deposited on the surface of the treated (coated) wood, due to the formation of insoluble calcium-organic complexes (Ca-citrate and Ca-tartrate). This observation refers to the macroscopic accumulation of the chelated product. Because the calcium-organic complexes have lower solubility than the precursors, they precipitate out of the solution onto the wood surface and within the surface voids during the treatment and drying phases. The coating formation is driven by a two-step mechanism. First, the vacuum treatment facilitates the penetration of soluble ions and carboxylate ligands into the porous wood structure. Second, as the chelation reaction proceeds and the solvent evaporates during drying, insoluble calcium-organic complexes precipitate and deposit, subsequent esterification of the ligands with the hydroxyl groups of the cellulosic chains of wood occurs, ultimately forming cross-linked structures. The coating opacity increased with the addition of TA, demonstrated by CA1TA1 and CA0TA2 addition, exhibiting a denser surface coating compared to the CA2TA0 (pure CA treatment). The mass gain (%), as seen in Figure 7B (bl), was evaluated to quantify the efficiency of the treatments. The control samples showed a slightly negative mass change (around −1.0%) due to the leaching of water-soluble wood extractives during the vacuum and stirring process. However, all chelated treatments produced significant mass increases, confirming the successful incorporation and retention of the mineral–organic complexes on the surface and within the wood matrix after vacuum treatment. The nature of the organic acid affected the degree of mass increase. The CA2TA0 treatment resulted in a mass increase of around 38%. The addition of TA led to enhancements in mass gain. This was evidenced in the mixed acid system (CA1TA1) achieving the highest initial mass increase of approximately 89%, versus the pure TA system (CA0TA2) with around 81%. This indicates that the Ca-tartrate and mixed-chelate complexes undergo deposition more readily on the surface and within the lignocellulosic structure than the pure Ca-citrate complexes under the given reaction conditions (2M concentration, ambient temperature). Figure 7C highlights the proposed chelation of Ca2+ by both CA and TA, subsequent esterification, and the cross-linking reaction schematic with the lignocellulosic hydroxyl groups in wood after the dehydration step. Take note of the ability of the tartrate ligand to bind with Ca2+ bound to another tartrate ligand–cellulose chain (TA-Cellulose) which produces a highly cross-linked coating product. This explains the higher mass gain observed for Ca-tartrate-containing treatments.
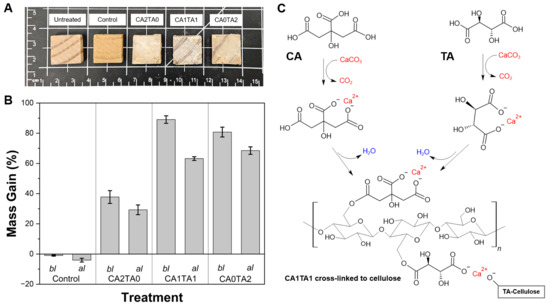
Figure 7.
(A) Macroscopic morphology of the pine wood blocks after chelation. (B) Mass gain (%) of the control and coated pine wood blocks (n = 10) after chelation and before leaching (bl) and the corresponding mass gain after leaching (al). There is a significant statistical difference between the control and treated wood blocks before and after leaching test, as evidenced by ANOVA (p < 0.05). (C) Proposed cross-linking reaction schematic of Ca2+ chelation by both CA and TA forming a hypothetical CA1TA1 product after the esterification and dehydration steps.
3.5.2. Leaching
A modified 5-day leaching test was performed to evaluate the leachability of the coatings for potential outdoor or wet applications, following a modified AWPA E11-16 standard. The retention of mass gain following leaching is depicted in Figure 7B (al). The control samples exhibited increased mass loss following the overall leaching period, decreasing to around −4%, corresponding to further solubilization of extractives in the aqueous medium. For the coated samples, a reduction in mass gain was observed for all groups, with a significant amount of the coating remaining within the wood. The CA2TA0 samples showed a mass gain of around 29%. Although the CA1TA1 treatment exhibited the highest initial deposition, it had a significant reduction during leaching (from approximately 89% to around 63%). On the other hand, the CA0TA2 treatment showed improved stability. Despite starting with a lower mass gain than CA1TA1, the CA0TA2 samples reached the highest mass gain for all treatments after leaching at approximately 69%. This indicates that the Ca-tartrate complexes produced in the CA0TA2 treatment exhibited reduced water solubility when compared to the Ca-citrate or mixed-acid complexes. Therefore, while the synergistic combination of CA and TA (CA1TA1) improves initial mass gain, the addition of just TA (CA0TA2) seemed to be the most effective for producing leach-resistant coatings on pine wood. This is due to the relative hydrophilicity of Ca-citrate compared to Ca-tartrate [46] and the ability of TA to act as a compatibilizer, reacting with the hydroxyl groups of the cellulose chain by esterification/transesterification reactions, making it less susceptible to leaching [59].
3.6. Thermal Degradation
3.6.1. Coating Materials
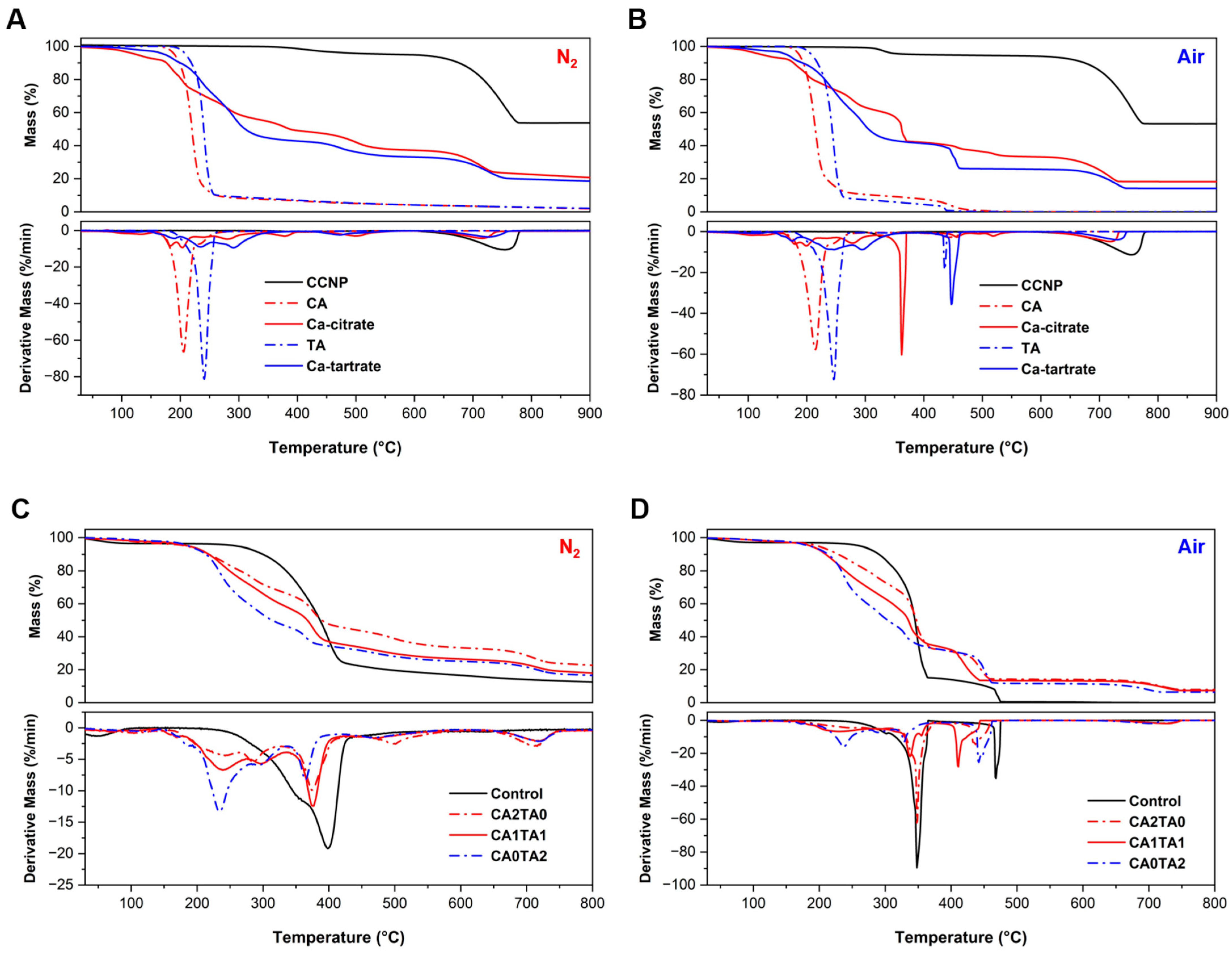
TGA and derivative thermogravimetry (DTG) were utilized to quantify the thermal stability and decomposition process of the starting materials (CCNP, CA, and TA) and the chelated coating products (Ca-citrate and Ca-tartrate) in both pyrolysis (N2) and oxidative (air) conditions, as shown in Figure 8A and Figure 8B, respectively. In both conditions, CCNP (black) showed significant thermal stability up to around 600 °C. Between 680 °C and 750 °C, a single and notable mass loss step was observed, corresponding to the decarbonation of calcite [60,61]. The stoichiometric yield of calcium oxide from calcium carbonate (56% by mass) is in accordance with the residual mass obtained in TGA at around 52%–54%. On the other hand, at lower temperatures, the CA and TA (dotted lines) degraded rapidly, leaving minimal residual mass (<2%) and confirming complete volatilization or combustion. Compared to its precursors, the Ca-citrate product (red) exhibited a multi-step decomposition profile. First, the release of absorbed water from calcium citrate resulted in an initial mass loss starting around 180 °C. Then after dehydration, the product gradually degraded in the N2 atmosphere, likely as a result of the citrate ligand breaking down into intermediate species such aconitate or itaconate derivatives [62,63]. However, there was a significant secondary mass loss under air at a peak temperature of around 360 °C. The oxidative combustion of the organic ligand can be observed through a major secondary peak in the DTG curve, which did not occur in the inert N2 atmosphere. After the degradation of the organic components of the chelated products, the inorganic calcium components remained (CaCO3 or CaO) with a final residual mass range of 18%–20% [64,65].
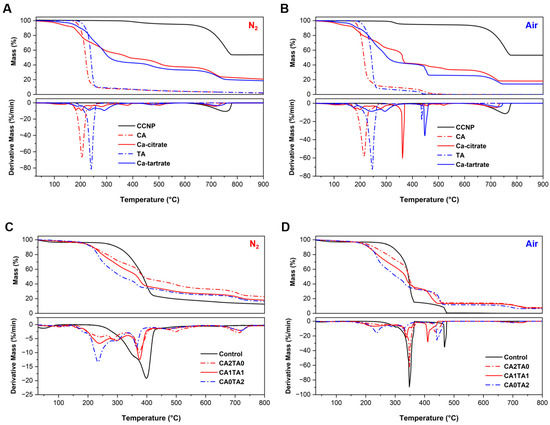
Figure 8.
(A) TGA and DTG thermograms of the coating materials under pyrolysis conditions (N2). (B) TGA and DTG thermograms of the coating materials under oxidative conditions (air). (C) TGA and DTG thermograms of the control and coated pine wood under pyrolysis conditions (N2). (D) TGA and DTG thermograms of the control and coated pine wood under oxidative conditions (air).
Compared to Ca-citrate (red), the Ca-tartrate product (blue) showed higher thermal stability in the initial stages. The multi-step thermal degradation of Ca-tartrate started at 202 °C in N2 and was noticeably greater at 445 °C in air. Sequential decomposition stages following a minor mass loss stage due to dehydration and subsequent ligand degradation were observed around 200–260 °C. The Ca-tartrate profile in the air atmosphere exhibits a sharp and delayed decomposition step that occurs at 448 °C. This step (similarly observed with TA at 434 °C) indicates the formation of a thermally stable intermediate, such as calcium oxalate, which is known to degrade into calcium carbonate at this temperature range (400–500 °C) [66]. The tartrate ligand combusts more readily in the oxidative atmosphere, producing oxalate species before being converted to oxide [67]. The more efficient combustion of carbonaceous residues was confirmed by TGA, showing that the final residual mass for Ca-tartrate in air was lower (14.7%) compared to that of N2 (18.7%). The presence of oxygen (air) shifted the thermal degradation pathways from pyrolysis to combustion when comparing the two conditions. Sharper DTG peaks and distinct secondary decomposition stages (364 °C for Ca-citrate and 448 °C for Ca-tartrate) were observed. These stages correspond to the rapid oxidation of organic intermediates and char that are persistent under N2 conditions. Table 1 and Table 2 summarize the thermogravimetric parameters under pyrolysis (N2) and oxidative (air) conditions for the coating materials, respectively.

Table 1.
Thermogravimetric parameters under pyrolysis (N2) conditions for the coating materials.

Table 2.
Thermogravimetric parameters under oxidative (air) conditions for the coating materials.
3.6.2. Coated Wood
TGA and DTG were also utilized to quantify thermal stability and decomposition of control and treated (coated) wood samples in N2 (Figure 8C) and air (Figure 8D). The control pine wood showed a typical three-stage decomposition profile under an inert atmosphere. First is the initial dehydration step below 150 °C, followed by a major degradation step between 250 °C and 450 °C (Tpeak at 398 °C) attributed to the depolymerization of cellulose and hemicellulose, and a much stable degradation step over a wide temperature range representing the pyrolysis of lignin [33,68,69,70,71,72]. The thermal degradation pathway was significantly altered by the incorporation of the chelated CCNP coatings. In comparison to the control, the coated samples (CA2TA0, CA1TA1, and CA0TA2) exhibited decreased degradation onset temperatures (Tonset). The catalytic effect of organic acids and calcium species on the dehydration and depolymerization of hemicellulose causes this decreased initial thermal stability [73]. Additionally, distinct secondary peaks not observed from the control were present in the DTG thermograms for the coated samples. First, prominent shoulder peaks were observed under the cellulose decomposition temperature in the low-temperature range, corresponding to rapid cellulose/hemicellulose degradation (200–300 °C), immediately followed by the thermal degradation of the citrate and tartrate ligands to form intermediate products (350–400 °C) [64,74,75,76]. On the other hand, decomposition was observed for all coated samples in the 680–720 °C range (high-temperature region), which is a characteristic of calcium carbonate decarbonation, as previously discussed [60]. Prior to their subsequent degradation into calcium oxide, the calcium species in the wood structure (chelates) transform into stable calcium carbonate intermediates under elevated temperatures [77]. From 12.5% for the control to 16.6%–22.7% for the coated samples, the residual mass at 800 °C increased significantly. This increase is a result of both the improved char formation promoted by the coating treatments and the resulting inorganic calcium oxide (CaO) formation.
Meanwhile, the thermo-oxidative degradation profiles under air (combustion) provide valuable information on the final inorganic content. The oxidation of cellulose/hemicellulose components (Tpeak around 345 °C) and the combustion of the residual char (Tpeak > 465 °C) were the two distinct stages of combustion observed in the control sample. The control exhibited complete combustion of the organic wood components, yielding negligible residual mass. On the other hand, the coated samples showed multi-stage combustion processes and significantly higher residual masses. The treatments affected the char oxidation process and accelerated the initial combustion stage. The decarboxylation of citrate and tartrate ligands within the coatings to produce CO2 was promoted in the presence of O2 (under air), as evidenced by the Tpeak observed around 350 °C, since no similar decomposition reaction occurred under N2 atmosphere [78,79]. The final residual masses from TGA in air and the initial mass gain (bl) shown in Figure 7B can be quantitatively correlated. The residual masses in the coated samples originate entirely from the inorganic calcium component of the coating (CaO), while for the control pine wood, no residue was observed under air at 800 °C. First, with an initial mass gain of 89%, CA1TA1 yielded the highest mass gain. The same CA1TA1-treated wood sample produced a residual mass of 7.4% of the total coated wood block mass under air in TGA. Next, CA0TA2 produced a TGA residual mass of 6.4% and an initial mass increase after treatment of 81%. Lastly, CA2TA0 produced the highest TGA residual mass of 7.8% while having the lowest initial mass gain after treatment of about 38%. Although the successful incorporation of the CCNP-derived coatings was confirmed by the significant inorganic residues observed in all coated samples, the lack of a direct linear relationship between initial mass gain after treatment (%) and TGA residual mass (%) points to variations in the chemical properties of the chelated coating products. Compared to the tartrate-containing coated samples (CA1TA1 and CA0TA2), which include more organic components which contributed to initial mass increase but mostly degraded during combustion, the CA2TA0 coating exhibits a higher mineral-to-organic ratio, following the initial mass gain after treatment and the TGA residual mass. Table 3 and Table 4 summarize the thermogravimetric parameters under pyrolysis (N2) and oxidative (air) conditions for the coated wood samples, respectively.

Table 3.
Thermogravimetric parameters under pyrolysis (N2) conditions for the wood samples.

Table 4.
Thermogravimetric parameters under oxidative (air) conditions for the wood samples.
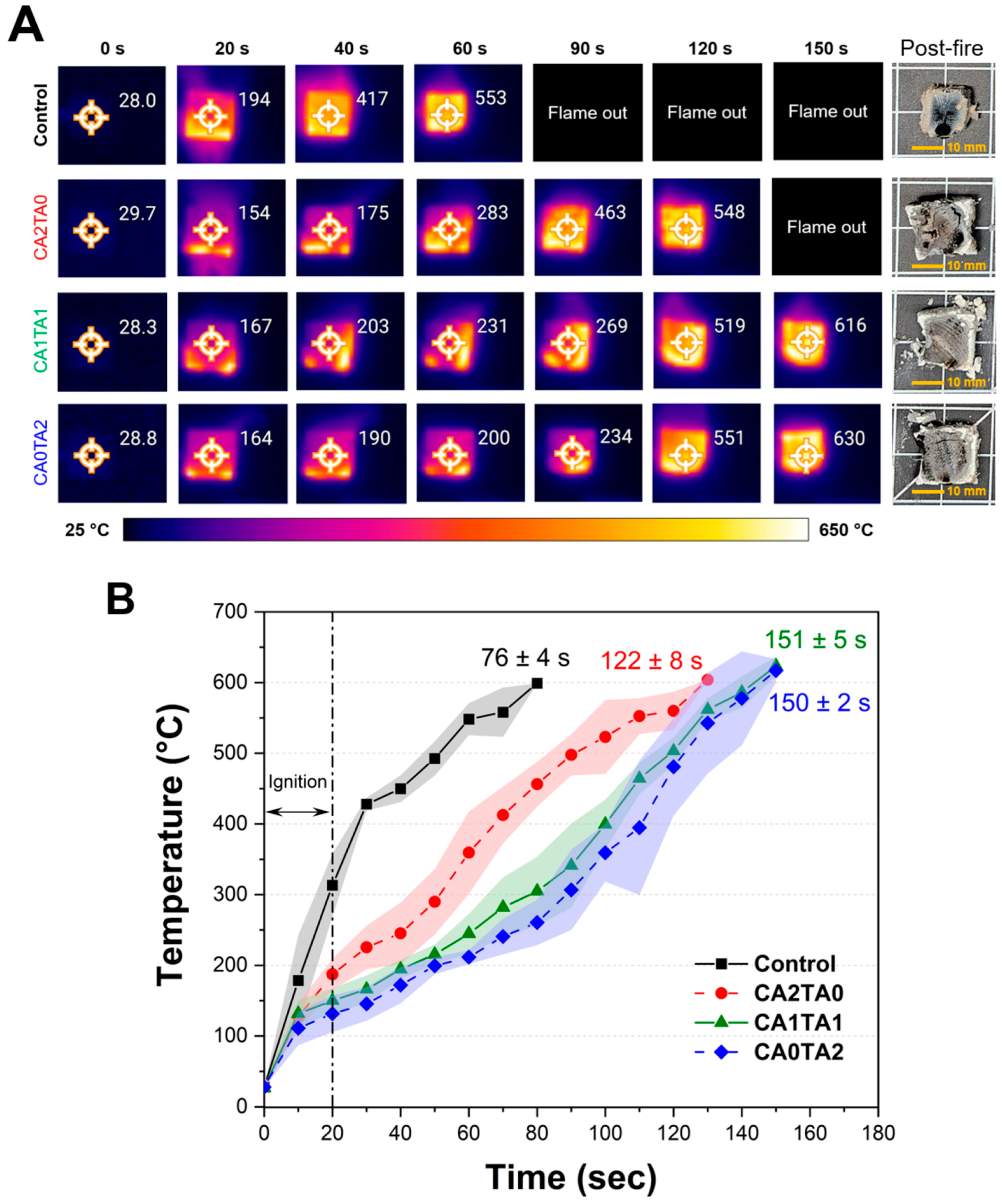
3.7. Flame Retardancy
A modified UL 94 flammability test was used to evaluate the flame retardancy of the pine wood blocks treated (coated) with chelated CCNP. Figure 9A shows the sequential infrared thermal images of representative wood samples featuring the surface temperatures during combustion, along with the post-fire macroscopic photographs of the residual wood samples after combustion. The corresponding temperature profile of the wood block samples is shown in Figure 9B. The control sample immediately caught fire when exposed to the flame source. At around 40 s, which was 20 s after the removal of the flame source, the surface temperature had surged to 417 °C, increasing to 553 °C by 60 s. The wood block has undergone complete combustion in 76 ± 4 s due to the rapid flame spread and substantial heat release, leaving only a small amount of residue. The result corresponds to the TGA results under air, where the control sample exhibited rapid mass loss and negligible residual mass, verifying the complete oxidation of lignocellulosic material without char stabilization. On the other hand, the treated samples showed reduced heat propagation and delayed ignition. At 60 s, the temperature of the CA2TA0 (citrate-only) sample increased gradually (reaching 283 °C), which was almost half of the control at the same interval. However, the flame continued until 122 ± 8 s. The incorporation of TA significantly improved flame resistance. Both CA1TA1- (mixed acid) and CA0TA2 (tartrate-only)-treated samples maintained lower surface temperatures during the initial stages of combustion. At 60 s, the surface temperatures were confined to 231 °C and 200 °C, respectively. As noted in the figure, the “flame out” periods were considerably extended to 151 ± 5 s for CA1TA1 and 150 ± 2 s for CA0TA2. The post-fire images for all coated samples show a more preserved dimensional stability of the wood blocks, now composed of carbonaceous char and inorganic residues. This corresponds to the significant residual masses (6.4%–7.8%) observed in the air-atmosphere TGA, suggesting that the chelated coatings promote char formation [80]. In contrast to the sharp increase in temperature of the control, Figure 9B shows an apparent slower temperature increase for the coated samples between 20 and 80 s. This flame retardation effect can be attributed to the multi-step degradation of the coating components observed in the TGA and DTG thermograms. The endothermic dehydration of the chelated salts and the subsequent decarbonation of CaCO3 around 680–720 °C served as a heat sink by absorbing thermal energy and diluting combustible gases with non-flammable ones [81]. Furthermore, the production of persistent CaO forms a thermal barrier that protects the underlying wood from heat flux and oxygen transport, prolonging the time to structural disintegration. The conversion of CCNP into tartrate-based coatings (CA0TA2) provides optimal thermal insulation. This treatment delays the temperature increase while slowing thermal decomposition in accordance with both the thermal imaging and temperature profile results, leading to a more preserved structure post-fire compared to the control or citrate-only treatments. The reported thermal property improvements, which include limiting the surface temperatures to around 200 °C and delaying the combustion of the bulk material, were due to the high loading achieved, particularly for CA1TA1 and CA0TA2 (>80%) after the coating treatment. For real-world structural applications, this trade-off between weight and fire safety would need to be evaluated based on specific building codes and construction policies.
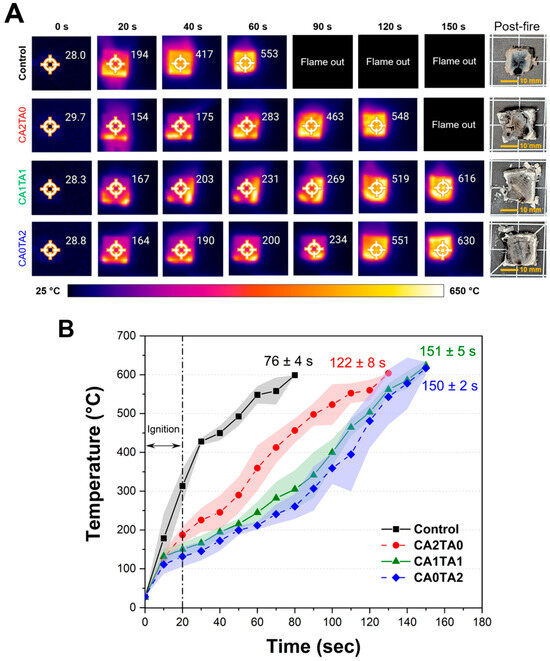
Figure 9.
(A) Thermal images of representative control and coated pine wood blocks, alongside the post-fire images of the wood blocks after combustion. (B) Temperature profile of the control and coated pine wood blocks (n = 5). The values (mean ± sd) indicated in the figure correspond to the flame-out time.
4. Related Studies and Recommendations
Recent studies focusing on metal ion coordination (chelation/complexation) with organic acids have proven effective in improving the thermal properties of materials. These strategies minimize the effects of thermal degradation, flammability, and heat transfer through the formation of stable metal–organic complexes. A good example is the improvement of polyvinyl chloride (PVC), which is intrinsically unstable at processing temperatures, emitting hydrogen chloride that accelerates subsequent thermal degradation. A related study utilized calcium and zinc stearates (metal carboxylates) as heat stabilizers. Their strategy is based on a synergistic mechanism, wherein zinc carboxylates rapidly substitute unstable chlorine atoms on the polymer chain, while calcium carboxylates neutralize the liberated hydrogen chloride and restore the zinc active sites to prevent “zinc burning” [82]. In related studies, metal chelation has also been found to improve fire resistance through two primary pathways. First is bio-based charring, wherein upon combustion, the metal complexes catalyze the dehydration of cellulose/wood, subsequently forming a dense and thermally insulating carbonaceous char layer that blocks heat transfer and suppresses smoke. An example of this involves phytic acid, a natural organophosphate, chelating Zn ions to form metal phytates to improve the flame retardancy of Chinese fir wood [83]. The second pathway involves barrier effects, where metal–organic frameworks (MOFs) formed by chelating metal cross-linking are incorporated into polymers that decompose endothermically. The material releases non-flammable gases upon combustion, diluting the fuel supply and leaving behind a porous metal-oxide/carbon barrier that protects the underlying material from fire [84]. Organic acids, like stearic acid, are excellent phase change materials (PCM) due to their high latent heat. However, the materials suffer from liquid leakage during melting. A recent study focusing on “shape-stabilization” by chelation into the porous MOF resulted in confinement and interactions between the acid and metals to prevent thermal leakage [85]. Table 5 summarizes the recent studies employing organic acids to bind metal ions to improve the thermal properties of materials.

Table 5.
Studies highlighting the use of metal ion coordination with organic acids for improving the thermal properties of materials.
The evaluation of mechanical properties, such as bending strength, modulus of elasticity, and surface hardness, is important for the overall material property assessment and for future work, but the primary aim of this study was to establish the preparation of coatings, material characterization, and evaluation of the thermal property improvements of the polycarboxylic acid/CCNP coatings on wood. Thus, the wood block dimensions had been intentionally selected to accommodate the constraints of the TGA instrument and the modified UL 94 flammability test, rather than the much larger dimensions required for traditional mechanical testing. Regarding adhesion, although a pull-off test was not conducted, the modified leaching test involving continuous agitation provided a rigorous alternative to test the stability and resistance to leaching in aqueous conditions of the coatings. In addition, even though cone calorimetry is an effective method for obtaining heat release results, the use of the modified UL 94 method coupled with thermal imaging afforded important findings regarding the inhibition of heat transfer. The decrease in surface temperature (200 °C compared to 553 °C) demonstrated that coating the material hindered flame propagation. Lastly, while SEM/EDS could provide and verify microstructural morphology, the presence and effectiveness of the coating were established through macroscopic imaging, mass gain after treatment, and improved thermal properties of the bulk material after coating. This study suggests that the employed thermal characterization techniques, together with comprehensive FTIR, Raman, and XRD analyses, sufficiently confirm the deposition and effectiveness of the coatings.
5. Conclusions
The focus of the current study was the development of sustainable wood coatings with improved thermal properties by chelating calcium carbonate nanopowder (CCNP) with citric acid (CA) and tartaric acid (TA). Characterization tests using FTIR, Raman spectroscopy, and XRD analysis confirmed the conversion of CCNP, CA, and TA into crystalline calcium citrate and calcium tartrate. An important correlation was observed between the treatment (coating) efficiency and leachability. The mixed-acid treatment (CA1TA1) achieved the highest mass gain of around 89%. However, the pure TA treatment (CA0TA2) exhibited more resilience to leaching, maintaining around 69% of its mass as compared to only 29% for the citrate-based coating (CA2TA0). These results indicate that tartrate ligands produce a relatively more stable and less soluble complex within the wood structure. Thermogravimetric analysis showed that the coatings act as effective heat sinks through their ability to undergo multi-step degradation (dehydration, decarbonation, and decarboxylation) and promote char formation, leading to significant inorganic residues in oxidative (air) conditions. The process was verified using flammability tests, in which the CA0TA2-treated samples exhibited optimal flame retardancy. The coating delayed combustion and limited the surface temperatures to around 200 °C (compared to the >550 °C for control) while slowing down the thermal decomposition of the wood during burning and leading to a more intact structure post-fire. This bio-based and sustainable strategy provides a potential non-toxic alternative to conventional chemical-based fire retardants.
Author Contributions
Conceptualization, J.V.T.; methodology, J.V.T.; validation, J.V.T. and A.G.M.; writing—original draft preparation, J.V.T.; writing—review and editing, J.V.T. and A.G.M.; visualization, J.V.T.; supervision, A.G.M.; project administration, A.G.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the University of Idaho P3R1 matching grant.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All relevant data are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| al | After Leaching |
| ATR-FTIR | Attenuated Total Reflectance-Fourier Transform Infrared (Spectroscopy) |
| AWPA | American Wood Protection Association |
| bl | Before Leaching |
| CA | Citric Acid |
| CCNP | Calcium Carbonate Nanopowder |
| DI | Deionized (Water) |
| DTG | Derivative Thermogravimetry |
| EDTA | Ethylenediaminetetraacetic Acid |
| GC-TCD | Gas Chromatography-Thermal Conductivity Detector |
| GLDA | L-Glutamic acid-N,N-Diacetic Acid |
| HEDTA | N-(Hydroxyethyl)-Ethylenediaminetriacetic Acid |
| MOF | Metal–Organic Frameworks |
| NTA | Nitrilotriacetic Acid |
| PCM | Phase Change Materials |
| PSA | Particle Size Analysis |
| PVC | Polyvinyl Chloride |
| Rt | Retention Time |
| SEM | Scanning Electron Microscopy |
| TA | Tartaric Acid |
| TGA | Thermogravimetric Analysis |
| Tonset | Onset Temperature |
| TPeak | Peak Temperature |
| UL 94 | Underwriters Laboratories 94 (Burning Test) |
| XRD | X-Ray Diffraction |
References
- Tongco, J.V. Effects of Low Concentration Coupling Agent Addition on the Physicomechanical Behavior of Wood Fiber/HDPE Composite. Mediterr. J. Chem. 2025, 15, 187–195. [Google Scholar] [CrossRef]
- Tongco, J.V. Effects of Treatment Methods on Surface Morphology and Performance of Modified Natural Fiber Biopolymers and Biocomposites—A Comprehensive Review. J. Polym. Res. 2025, 32, 443. [Google Scholar] [CrossRef]
- Akpan, E.I.; Wetzel, B.; Friedrich, K. Eco-Friendly and Sustainable Processing of Wood-Based Materials. Green Chem. 2021, 23, 2198–2232. [Google Scholar] [CrossRef]
- Teacă, C.-A.; Roşu, D.; Mustaţă, F.; Rusu, T.; Roşu, L.; Roşca, I.; Varganici, C.-D. Natural Bio-Based Products for Wood Coating and Protection against Degradation: A Review. Bioresources 2019, 14, 4873–4901. [Google Scholar] [CrossRef]
- Kolya, H.; Kang, C.-W. Eco-Friendly Polymer Nanocomposite Coatings for Next-Generation Fire Retardants for Building Materials. Polymers 2024, 16, 2045. [Google Scholar] [CrossRef] [PubMed]
- Vani, C.N.; Prajwal, S.; Sundararaj, R.; Dhamodaran, T.K. Chemical preservatives in wood protection. In Science of Wood Degradation and Its Protection; Springer: Singapore, 2022; pp. 559–587. ISBN 978-981-16-8797-6. [Google Scholar]
- Barbero-López, A.; Akkanen, J.; Lappalainen, R.; Peräniemi, S.; Haapala, A. Bio-Based Wood Preservatives: Their Efficiency, Leaching and Ecotoxicity Compared to a Commercial Wood Preservative. Sci. Total Environ. 2021, 753, 142013. [Google Scholar] [CrossRef]
- Schiopu, N.; Tiruta-Barna, L. Wood preservatives. In Toxicity of Building Materials; Woodhead Publishing: Cambridge, UK, 2012; pp. 138–165. ISBN 9780857091222. [Google Scholar]
- Dietenberger, M. Update for Combustion Properties of Wood Components. Fire Mater. 2002, 26, 255–267. [Google Scholar] [CrossRef]
- Książek, E.E.; Janczar-Smuga, M.; Pietkiewicz, J.J.; Walaszczyk, E. Optimization of Medium Constituents for the Production of Citric Acid from Waste Glycerol Using the Central Composite Rotatable Design of Experiments. Molecules 2023, 28, 3268. [Google Scholar] [CrossRef]
- Chen, Y.; Barzee, T.J.; Zhang, R.; Pan, Z. Citrus. In Integrated Processing Technologies for Food and Agricultural By-Products; Pan, Z., Zhang, R., Zicari, S., Eds.; Academic Press: Amsterdam, The Netherlands, 2019; pp. 217–242. ISBN 9780128141397. [Google Scholar]
- Kuforiji, O.O.; Kuboye, A.O.; Odunfa, S.A. Orange and Pineapple Wastes as Potential Substrates for Citric Acid Production. Int. J. Plant Biol. 2010, 1, 19–21. [Google Scholar] [CrossRef]
- Zhang, L.; Bryan, S.J.; Selão, T.T. Sustainable Citric Acid Production from CO2 in an Engineered Cyanobacterium. Front. Microbiol. 2022, 13, 973244. [Google Scholar] [CrossRef]
- Mruthunjaya, K.; Manjula, S.N.; Anand, A.; Kenchegowda, M.; Sharma, H. Valorization of citrus waste for the synthesis of value added products. In Valorization of Citrus Food Waste; Springer: Cham, Switzerland, 2025; pp. 179–213. ISBN 978-3-031-77999-2. [Google Scholar]
- Pratiksha; Adhikary, M. Industrial production of citrus by-products and its processing techniques. In Citrus Fruits and Juice: Processing and Quality Profiling; Springer: Singapore, 2024; pp. 391–417. ISBN 978-981-99-8699-6. [Google Scholar]
- Mahato, N.; Sharma, K.; Sinha, M.; Baral, E.R.; Koteswararao, R.; Dhyani, A.; Hwan Cho, M.; Cho, S. Bio-Sorbents, Industrially Important Chemicals and Novel Materials from Citrus Processing Waste as a Sustainable and Renewable Bioresource: A Review. J. Adv. Res. 2020, 23, 61–82. [Google Scholar] [CrossRef]
- Show, P.L.; Oladele, K.O.; Siew, Q.Y.; Aziz Zakry, F.A.; Lan, J.C.W.; Ling, T.C. Overview of Citric Acid Production from Aspergillus niger. Front. Life Sci. 2015, 8, 271–283. [Google Scholar] [CrossRef]
- Dong, Y.; Xue, Q.; Fu, Z.; Yan, Y.; Lu, Y.; Liu, Y.; Li, J. Enhancing Wood Stability and Fire Retardancy through Citric Acid and Phosphorylated Sucrose Stearate Cross-Linking Modification. Constr. Build. Mater. 2023, 393, 131946. [Google Scholar] [CrossRef]
- Saffari, V.R.; Saffari, M. Effects of EDTA, Citric Acid, and Tartaric Acid Application on Growth, Phytoremediation Potential, and Antioxidant Response of Calendula officinalis L. in a Cadmium-Spiked Calcareous Soil. Int. J. Phytoremediat. 2020, 22, 1204–1214. [Google Scholar] [CrossRef]
- Tang, S.; Wang, Z.; Yuan, D.; Zhang, C.; Rao, Y.; Wang, Z.; Yin, K. Ferrous Ion-Tartaric Acid Chelation Promoted Calcium Peroxide Fenton-like Reactions for Simulated Organic Wastewater Treatment. J. Clean. Prod. 2020, 268, 122253. [Google Scholar] [CrossRef]
- Burbidge, C.A.; Ford, C.M.; Melino, V.J.; Wong, D.C.J.; Jia, Y.; Jenkins, C.L.D.; Soole, K.L.; Castellarin, S.D.; Darriet, P.; Rienth, M.; et al. Biosynthesis and Cellular Functions of Tartaric Acid in Grapevines. Front. Plant Sci. 2021, 12, 643024. [Google Scholar] [CrossRef]
- Celik, F.; Gundogdu, M.; Ercisli, S.; Kaki, B.; Berk, S.; Ilhan, G.; Sagbas, H.I. Variation in Organic Acid, Sugar and Phenolic Compounds in Fruits of Historical Apple Cultivars. Not. Bot. Horti Agrobot. Cluj. Napoca. 2018, 46, 622–629. [Google Scholar] [CrossRef]
- Dai, T.; Wan, J.; Li, J.; Luo, G.; Li, L.; Wu, H.; Xu, W.; Zheng, B.; Wang, S.; Luo, C.; et al. Comprehensive Evaluation of Fruit Sugar and Acid Quality of Mango Germplasm Based on Principal Component and Cluster Analysis. J. Fruit Sci. 2022, 39, 2253–2263. [Google Scholar] [CrossRef]
- Reşitca, V. Possibility and Necessity of Tartaric Acid Production in the Republic of Moldova. J. Eng. Sci. 2022, 29, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Howell, B.A.; Carter, K.E.; Dangalle, H. Flame Retardants Based on Tartaric Acid: A Renewable by-Product of the Wine Industry. ACS Symp. Ser. 2011, 1063, 133–152. [Google Scholar] [CrossRef]
- Dhami, N.K.; Reddy, M.S.; Mukherjee, M.S. Biomineralization of Calcium Carbonates and Their Engineered Applications: A Review. Front. Microbiol. 2013, 4, 314. [Google Scholar] [CrossRef]
- Hammes, F.; Verstraete, W. Key Roles of PH and Calcium Metabolism in Microbial Carbonate Precipitation. Rev. Environ. Sci. Biotechnol. 2002, 1, 3–7. [Google Scholar] [CrossRef]
- Ghosh, T.; Bhaduri, S.; Montemagno, C.; Kumar, A. Sporosarcina Pasteurii Can Form Nanoscale Calcium Carbonate Crystals on Cell Surface. PLoS ONE 2019, 14, e0210339. [Google Scholar] [CrossRef]
- Beniash, E.; Aizenberg, J.; Addadi, L.; Weiner, S. Amorphous Calcium Carbonate Transforms into Calcite during Sea Urchin Larval Spicule Growth. Proc. R. Soc. B Biol. Sci. 1997, 264, 461. [Google Scholar] [CrossRef]
- Anbu, P.; Kang, C.H.; Shin, Y.J.; So, J.S. Formations of Calcium Carbonate Minerals by Bacteria and Its Multiple Applications. Springerplus 2016, 5, 250. [Google Scholar] [CrossRef]
- Abdolmohammadi, S.; Siyamak, S.; Ibrahim, N.A.; Wan Yunus, W.M.Z.; Ab Rahman, M.Z.; Azizi, S.; Fatehi, A. Enhancement of Mechanical and Thermal Properties of Polycaprolactone/Chitosan Blend by Calcium Carbonate Nanoparticles. Int. J. Mol. Sci. 2012, 13, 4508–4522. [Google Scholar] [CrossRef] [PubMed]
- Merk, V.; Chanana, M.; Gaan, S.; Burgert, I. Mineralization of Wood by Calcium Carbonate Insertion for Improved Flame Retardancy. Holzforschung 2016, 70, 867–876. [Google Scholar] [CrossRef]
- Tongco, J.V.; McDonald, A.G. Bioinspired Improvement of Lignocellulosic Bio-Based Materials Against Fire and Fungi—A Comprehensive Review. Bioresour. Bioprod. 2026, 2, 3. [Google Scholar] [CrossRef]
- Elkordy, M.M.; El-Senousi, N.A.; Zein, H.; Mohamed, T.A. Valorization of Eggshell Waste into Calcium and Collagen-Based Nutritional Supplements. Asian Food Sci. J. 2025, 24, 47–60. [Google Scholar] [CrossRef]
- Adaikalam, K.; Teli, A.M.; Marimuthu, K.P.; Ramesh, S.; Lee, H.; Kim, H.S.; Kim, H.S. Energy Storage Application of CaO/Graphite Nanocomposite Powder Obtained from Waste Eggshells and Used Lithium-Ion Batteries as a Sustainable Development Approach. Nanomaterials 2024, 14, 1129. [Google Scholar] [CrossRef]
- Chanwetprasat, P.; Seangarun, C.; Seesanong, S.; Boonchom, B.; Laohavisuti, N.; Boonmee, W.; Rungrojchaipon, P. Effect of Citric Acid Concentration on the Transformation of Aragonite CaCO3 to Calcium Citrate Using Cockle Shells as a Green Calcium Source. Materials 2025, 18, 2003. [Google Scholar] [CrossRef]
- AWPA E11-16; American Wood Protection Association Standard Method for Accelerated Evaluation of Preservative Leaching. American Wood Protection Association: Birmingham, AL, USA, 2016.
- UL-94; Standard for Tests for Flammability of Plastic Materials for Parts in Devices and Appliances. American National Standards Institute—Underwriters’ Laboratories: Northbrook, IL, USA, 1996.
- Spieß, B.; Metzsch-Zilligen, E.; Pfaendner, R. Mechanistic Evaluation of Flame Retardants during UL94 Standard Testing via IR-Camera. Polym. Test. 2021, 103, 107320. [Google Scholar] [CrossRef]
- Leukel, S.; Mondeshki, M.; Tremel, W. Hydrogen Bonding in Amorphous Alkaline Earth Carbonates. Inorg. Chem. 2018, 57, 11289–11298. [Google Scholar] [CrossRef] [PubMed]
- Schuitemaker, A.; Aufort, J.; Koziara, K.B.; Demichelis, R.; Raiteri, P.; Gale, J.D. Simulating the Binding of Key Organic Functional Groups to Aqueous Calcium Carbonate Species. Phys. Chem. Chem. Phys. 2021, 23, 27253–27265. [Google Scholar] [CrossRef]
- Fatehah, M.O.; Aziz, H.A.; Stoll, S. Nanoparticle Properties, Behavior, Fate in Aquatic Systems and Characterization Methods. J. Colloid Sci. Biotechnol. 2014, 3, 1–30. [Google Scholar] [CrossRef]
- Paik, S.-Y.-R.; Ryu, J.; Ko, S. Comparison of Physicochemical Properties of Calcium Carbonate Nano- and Micro-Powders. Food Eng. Prog. 2012, 16, 134–138. [Google Scholar] [CrossRef]
- Wang, J.; Chen, J.S.; Zong, J.Y.; Zhao, D.; Li, F.; Zhuo, R.X.; Cheng, S.X. Calcium Carbonate/Carboxymethyl Chitosan Hybrid Microspheres and Nanospheres for Drug Delivery. J. Phys. Chem. C 2010, 114, 18940–18945. [Google Scholar] [CrossRef]
- Miller, M.E.; McKinnon, L.P.; Walker, E.B. Quantitative Measurement of Metal Chelation by Fourier Transform Infrared Spectroscopy. Anal. Chem. Res. 2015, 6, 32–35. [Google Scholar] [CrossRef]
- Li, J.; Liu, Y.; Gao, Y.; Zhong, L.; Zou, Q.; Lai, X. Preparation and Properties of Calcium Citrate Nanosheets for Bone Graft Substitute. Bioengineered 2016, 7, 376–381. [Google Scholar] [CrossRef]
- Tsioptsias, C.; Panagiotou, A.; Mitlianga, P. Thermal Behavior and Infrared Absorbance Bands of Citric Acid. Appl. Sci. 2024, 14, 8406. [Google Scholar] [CrossRef]
- Turki, T.; Othmani, M.; Bac, C.G.; Rachdi, F.; Bouzouita, K. Surface Modification of Zinc-Containing Hydroxyapatite by Tartaric Acid. Appl. Surf. Sci. 2013, 284, 66–71. [Google Scholar] [CrossRef]
- He, L.; Bao, G.; Jin, X.; Zhang, R.; Qin, D. Eco-Friendly in-Situ Mineralization of Bamboo for Flame Retardancy. Ind. Crops Prod. 2023, 197, 116644. [Google Scholar] [CrossRef]
- Mitterdorfer, C.; Bernard, J.; Klauser, F.; Winkel, K.; Kohl, I.; Liedl, K.R.; Grothe, H.; Mayer, E.; Loerting, T. Local Structural Order in Carbonic Acid Polymorphs: Raman and FT-IR Spectroscopy. J. Raman Spectrosc. 2012, 43, 108–115. [Google Scholar] [CrossRef]
- Alves, J.F.; Edwards, H.G.M.; Korsakov, A.; de Oliveira, L.F.C. Revisiting the Raman Spectra of Carbonate Minerals. Minerals 2023, 13, 1358. [Google Scholar] [CrossRef]
- Roy, S.; Chamberlin, B.; Matzger, A.J. Polymorph Discrimination Using Low Wavenumber Raman Spectroscopy. Org. Process Res. Dev. 2013, 17, 976–980. [Google Scholar] [CrossRef]
- Harris, J.; Mey, I.; Hajir, M.; Mondeshki, M.; Wolf, S.E. Pseudomorphic Transformation of Amorphous Calcium Carbonate Films Follows Spherulitic Growth Mechanisms and Can Give Rise to Crystal Lattice Tilting. CrystEngComm 2015, 17, 6831–6837. [Google Scholar] [CrossRef]
- Rudolph, W.W.; Irmer, G. Hydration of the Calcium(II) Ion in an Aqueous Solution of Common Anions (ClO4−, Cl−, Br−, and NO3−). Dalton Trans. 2013, 42, 3919–3935. [Google Scholar] [CrossRef]
- Zhou, G.T.; Yu, J.C.; Wang, X.C.; Zhang, L.Z. Sonochemical Synthesis of Aragonite-Type Calcium Carbonate with Different Morphologies. New J. Chem. 2004, 28, 1027–1031. [Google Scholar] [CrossRef]
- Boudaira, B.; Harabi, A.; Bouzerara, F.; Zenikheri, F.; Foughali, L.; Guechi, A. Preparation and Characterization of Membrane Supports for Microfiltration and Ultrafiltration Using Kaolin (DD2) and CaCO3. Desalination Water Treat. 2016, 57, 5258–5265. [Google Scholar] [CrossRef]
- Rimsueb, N.; Cherdchom, S.; Aksornkitti, V.; Khotavivattana, T.; Sereemaspun, A.; Rojanathanes, R. Feeding Cells with a Novel “Trojan” Carrier: Citrate Nanoparticles. ACS Omega 2020, 5, 7418–7423. [Google Scholar] [CrossRef] [PubMed]
- Shajan, X.S.; Mahadevan, C. On the Growth of Calcium Tartrate Tetrahydrate Single Crystals. Bull. Mater. Sci. 2004, 27, 327–331. [Google Scholar] [CrossRef]
- Olivato, J.B.; Müller, C.M.O.; Carvalho, G.M.; Yamashita, F.; Grossmann, M.V.E. Physical and Structural Characterisation of Starch/Polyester Blends with Tartaric Acid. Mater. Sci. Eng. C 2014, 39, 35–39. [Google Scholar] [CrossRef]
- Galan, I.; Glasser, F.P.; Andrade, C. Calcium Carbonate Decomposition. J. Therm. Anal. Calorim. 2013, 111, 1197–1202. [Google Scholar] [CrossRef]
- Palandri, A.; Gilot, P.; Prado, G. A Kinetic Study of the Decarbonation of CaCO3. J. Anal. Appl. Pyrolysis 1993, 27, 119–130. [Google Scholar] [CrossRef]
- Rajendran, M.; Rao, M.S. Formation of BaTiO3 from Citrate Precursor. J. Solid State Chem. 1994, 113, 239–247. [Google Scholar] [CrossRef]
- Van Werde, K.; Mondelaers, D.; Vanhoyland, G.; Nelis, D.; Van Bael, M.K.; Mullens, J.; Van Poucke, L.C.; Van Der Veken, B.; Desseyn, H.O. Thermal Decomposition of the Ammonium Zinc Acetate Citrate Precursor for Aqueous Chemical Solution Deposition of ZnO. J. Mater. Sci. 2002, 37, 81–88. [Google Scholar] [CrossRef]
- Mansour, S.A.A. Thermal Decomposition of Calcium Citrate Tetrahydrate. Thermochim. Acta 1994, 233, 243–256. [Google Scholar] [CrossRef]
- Li, X.-G.; Lv, Y.; Ma, B.-G.; Wang, W.-Q.; Jian, S.-W. Decomposition Kinetic Characteristics of Calcium Carbonate Containing Organic Acids by TGA. Arab. J. Chem. 2017, 10, S2534–S2538. [Google Scholar] [CrossRef]
- Hourlier, D. Thermal Decomposition of Calcium Oxalate: Beyond Appearances. J. Therm. Anal. Calorim. 2019, 136, 2221–2229. [Google Scholar] [CrossRef]
- Shajan, X.S.; Mahadevan, C. FT-IR Spectroscopic and Thermal Studies on Pure and Impurity Added Calcium Tartrate Tetrahydrate Crystals. Cryst. Res. Technol. 2005, 40, 598–602. [Google Scholar] [CrossRef]
- Poletto, M.; Zattera, A.J.; Forte, M.M.C.; Santana, R.M.C. Thermal Decomposition of Wood: Influence of Wood Components and Cellulose Crystallite Size. Bioresour. Technol. 2012, 109, 148–153. [Google Scholar] [CrossRef]
- Slopiecka, K.; Bartocci, P.; Fantozzi, F. Thermogravimetric Analysis and Kinetic Study of Poplar Wood Pyrolysis. Appl. Energy 2012, 97, 491–497. [Google Scholar] [CrossRef]
- Tongco, J.V.; Cai, L. Durability and thermal properties of biomineralized southern yellow pine (SYP) via Bacterium sporosarcina Pasteurii. In Proceedings of the the 120th Annual Meeting of the American Wood Protection Association, Portland, ME, USA, 1 May 2024; American Wood Protection Association: Portland, ME, USA, 2024; pp. 1–8. [Google Scholar]
- del Carmen Recio-Ruiz, M.; Ruiz-Rosas, R.; García-Mateos, F.J.; Valero-Romero, M.J.; Rosas, J.M.; Rodríguez-Mirasol, J.; Cordero, T. An Integrated Approach to the Valorization of Pyrolysis Products from Lignocellulosic Residues and By-Products. Biomass Bioenergy 2025, 196, 107676. [Google Scholar] [CrossRef]
- Sebio-Puñal, T.; Naya, S.; López-Beceiro, J.; Tarrío-Saavedra, J.; Artiaga, R. Thermogravimetric Analysis of Wood, Holocellulose, and Lignin from Five Wood Species. J. Therm. Anal. Calorim. 2012, 109, 1163–1167. [Google Scholar] [CrossRef]
- Patwardhan, P.R.; Satrio, J.A.; Brown, R.C.; Shanks, B.H. Influence of Inorganic Salts on the Primary Pyrolysis Products of Cellulose. Bioresour. Technol. 2010, 101, 4646–4655. [Google Scholar] [CrossRef] [PubMed]
- Vallan, L.; Imahori, H. Citric Acid-Based Carbon Dots and Their Application in Energy Conversion. ACS Appl. Electron. Mater. 2022, 4, 4231–4257. [Google Scholar] [CrossRef]
- Wyrzykowski, D.; Hebanowska, E.; Nowak-Wiczk, G.; Makowski, M.; Chmurzyński, L. Thermal Behaviour of Citric Acid and Isomeric Aconitic Acids. J. Therm. Anal. Calorim. 2010, 104, 731–735. [Google Scholar] [CrossRef]
- Chattaway, F.D.; Ray, F.E. II.—The Decomposition of Tartaric Acid by Heat. J. Chem. Soc. Trans. 1921, 119, 34–37. [Google Scholar] [CrossRef]
- Rodriguez-Navarro, C.; Ruiz-Agudo, E.; Luque, A.; Rodriguez-Navarro, A.B.; Ortega-Huertas, M. Thermal Decomposition of Calcite: Mechanisms of Formation and Textural Evolution of CaO Nanocrystals. Am. Mineral. 2009, 94, 578–593. [Google Scholar] [CrossRef]
- Verduyckt, J.; De Vos, D.E. Highly Selective One-Step Dehydration, Decarboxylation and Hydrogenation of Citric Acid to Methylsuccinic Acid. Chem. Sci. 2017, 8, 2616–2620. [Google Scholar] [CrossRef]
- Czégény, Z.; Bozi, J.; Sebestyén, Z.; Blazsó, M.; Jakab, E.; Barta-Rajnai, E.; Forster, M.; Nicol, J.; McAdam, K.G.; Liu, C. Thermal Behaviour of Selected Flavour Ingredients and Additives under Simulated Cigarette Combustion and Tobacco Heating Conditions. J. Anal. Appl. Pyrolysis 2016, 121, 190–204. [Google Scholar] [CrossRef]
- Pondelak, A.; Škapin, A.S.; Knez, N.; Knez, F.; Pazlar, T. Improving the Flame Retardancy of Wood Using an Eco-Friendly Mineralisation Process. Green Chem. 2021, 23, 1130–1135. [Google Scholar] [CrossRef]
- Hernandez, V.; Romero, R.; Arias, S.; Contreras, D. A Novel Method for Calcium Carbonate Deposition in Wood That Increases Carbon Dioxide Concentration and Fire Resistance. Coatings 2022, 12, 72. [Google Scholar] [CrossRef]
- Putrawan, I.D.G.A.; Indarto, A.; Octavia, Y. Thermal Stabilization of Polyvinyl Chloride by Calcium and Zinc Carboxylates Derived from Byproduct of Palm Oil Refining. Heliyon 2022, 8, e10079. [Google Scholar] [CrossRef]
- Fan, S.; Gao, X.; Yang, X.; Li, X. Infusing Phytate-Based Biomass Flame Retardants into the Cellulose Lumens of Chinese Fir Wood Attains Superior Flame Retardant Efficacy. Int. J. Biol. Macromol. 2024, 258, 128975. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Zhang, Z.; Wang, Z.; Lin, Y.; Pan, Y.T.; Yao, K. Emerging Roles of Metal–Organic Frameworks as Flame Retardants: Recent Advances and Future Perspectives in Thermoplastic Polymers. Materials 2025, 19, 150. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Li, M. Stearic Acid-Modified MOF-Based Composite Phase Change Materials for Solar-Thermal Energy Conversion and Storage. Sol. Energy 2023, 262, 111843. [Google Scholar] [CrossRef]
- Sokhanvarian, K.; Nasr-El-Din, H.A.; Wang, G.; De-Wolf, C.A. Thermal Stability of Various Chelates That Are Used in the Oilfield and Potential Damage Due to Their Decomposition Products. In Proceedings of the SPE International Production and Operations Conference & Exhibition, Doha, Qatar, 14 May 2012; Volume 2, pp. 1263–1283. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.