Carbon-Related Defects as a Source for the Enhancement of Yellow Luminescence of Unintentionally Doped GaN
Abstract
1. Introduction
2. Materials and Methods
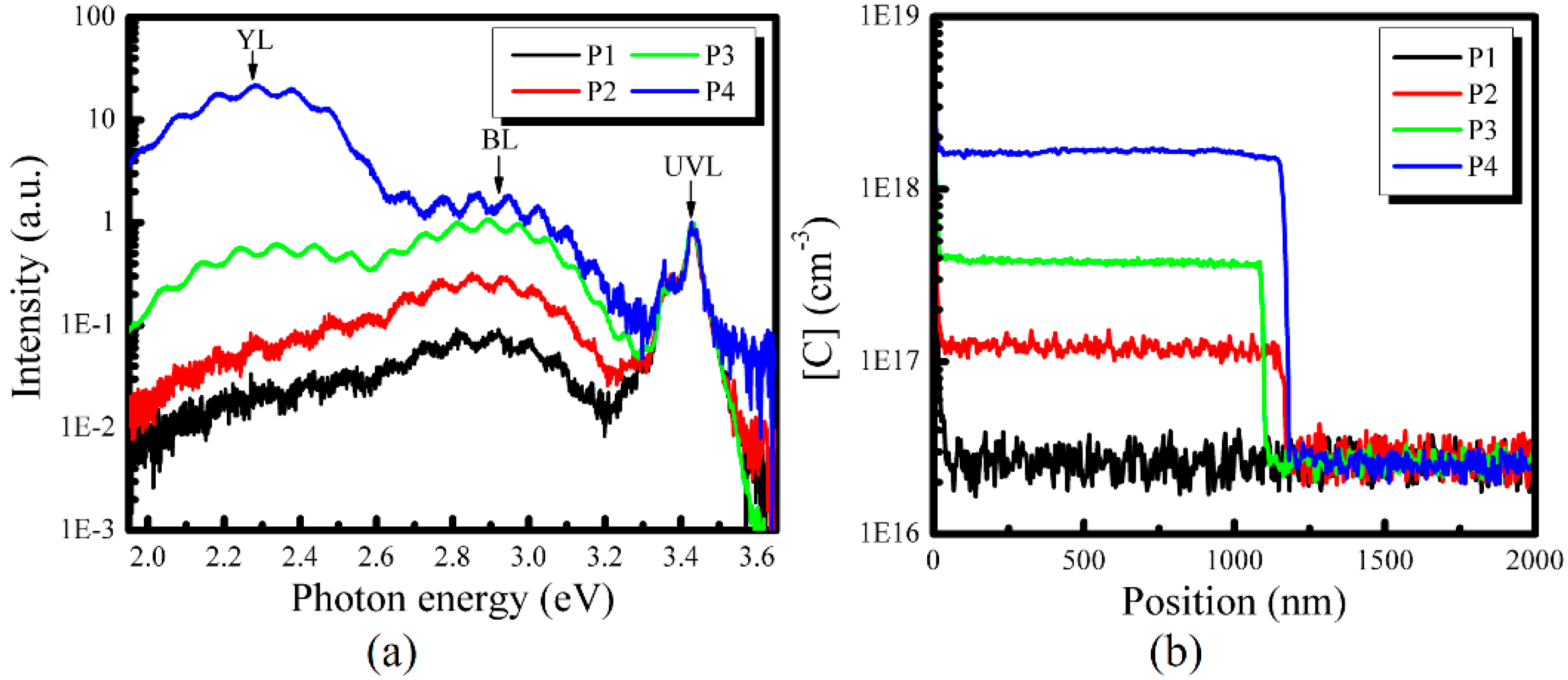
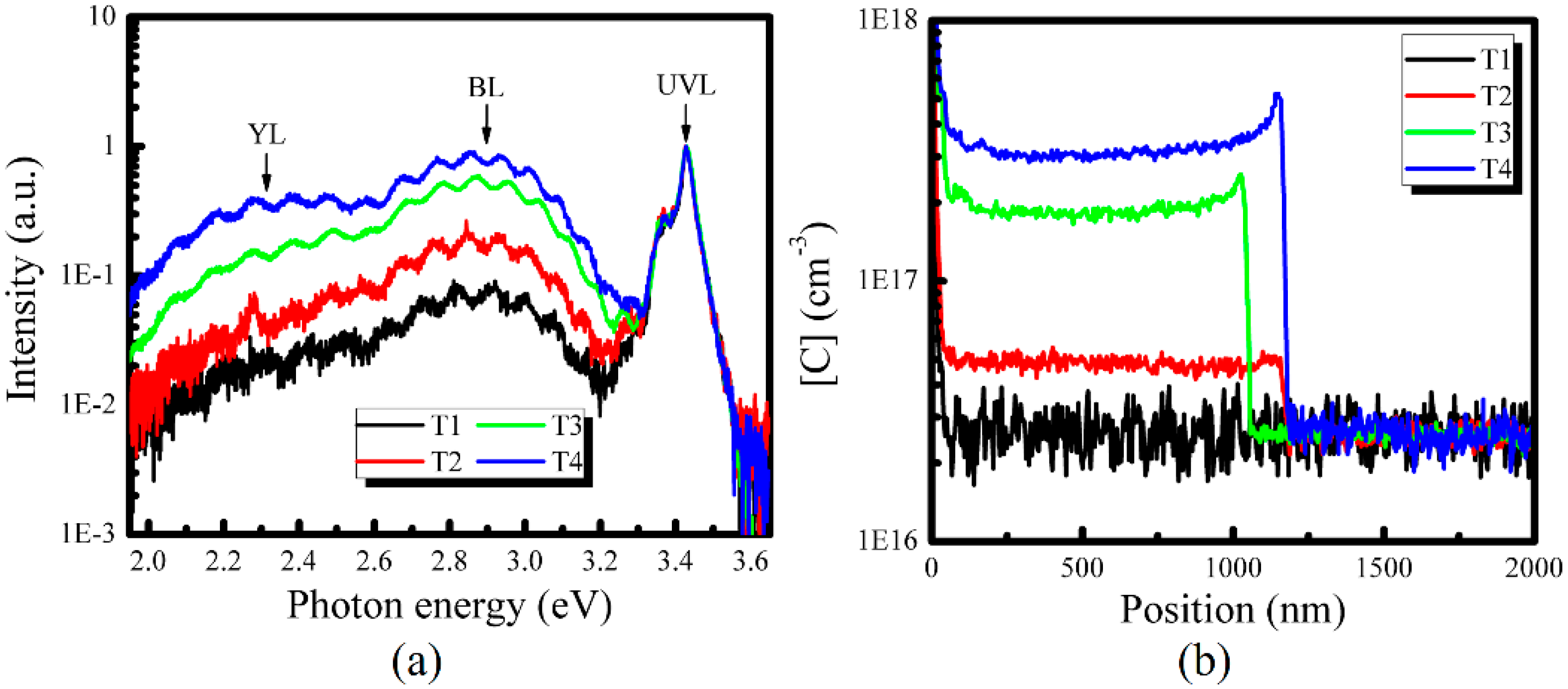
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Amano, H. Development of GaN-based blue LEDs and metalorganic vapor phase epitaxy of GaN and related materials. Prog. Cryst. Growth CH 2016, 62, 126–135. [Google Scholar] [CrossRef]
- Sun, Q.; Yan, W.; Feng, M.X.; Li, Z.C.; Feng, B.; Zhao, H.M.; Yang, H. GaN-on-Si blue/white LEDs: Epitaxy, chip, and package. J. Semicond. 2016, 37, 044006. [Google Scholar] [CrossRef]
- Murayama, M.; Nakayama, Y.; Yamazaki, K.; Hoshina, Y.; Watanabe, H.; Fuutagawa, N.; Kawanishi, H.; Uemura, T.; Narui, H. Watt-Class Green (530 nm) and Blue (465 nm) Laser Diodes. Phys. Status Solidi A 2018, 215, 1700513. [Google Scholar] [CrossRef]
- Jiang, L.R.; Liu, J.P.; Tian, A.Q.; Cheng, Y.; Li, Z.C.; Zhang, L.Q.; Zhang, S.M.; Li, D.Y.; Ikeda, M.; Yang, H. GaN-based green laser diodes. J. Semicond. 2016, 37, 111001. [Google Scholar] [CrossRef]
- Zhao, D.G.; Yang, J.; Liu, Z.S.; Chen, P.; Zhu, J.J.; Jiang, D.S.; Shi, Y.S.; Wang, H.; Duan, L.H.; Zhang, L.Q.; et al. Fabrication of room temperature continuous-wave operation GaN-based ultraviolet laser diodes. J. Semicond. 2017, 38, 051001. [Google Scholar] [CrossRef]
- Wu, H.L.; Wu, W.C.; Zhang, H.X.; Chen, Y.D.; Wu, Z.S.; Wang, G.; Jiang, H. All AlGaN epitaxial structure solar-blind avalanche photodiodes with high efficiency and high gain. App. Phys. Exp. 2016, 9, 052103. [Google Scholar] [CrossRef]
- Kuech, T.F. III-V compound semiconductors: Growth and structures. Prog. Cryst. Growth CH 2016, 62, 352–370. [Google Scholar] [CrossRef]
- Flack, T.J.; Pushpakaran, B.N.; Bayne, S.B. GaN Technology for Power Electronic Applications: A Review. J. Electron. Mater. 2016, 45, 2673–2682. [Google Scholar] [CrossRef]
- Calleja, E.; Sanchez, F.J.; Basak, D.; Sanchez-Garcia, M.A.; Munoz, E.; Izpura, I.; Calle, F.; Tijero, J.M.G.; Sanchez-Rojas, J.L.; Beaumont, B.; et al. Yellow luminescence and related deep states in undoped GaN. Phys. Rev. B 1997, 55, 4689–4694. [Google Scholar] [CrossRef]
- Julkarnain, M.; Kamata, N.; Fukuda, T.; Arakawa, Y. Yellow luminescence band in undoped GaN revealed by two-wavelength excited photoluminescence. Opt. Mater. 2016, 60, 481–486. [Google Scholar] [CrossRef]
- Julkarnain, M.; Fukuda, T.; Kamata, N.; Arakawa, Y. A direct evidence of allocating yellow luminescence band in undoped GaN by two-wavelength excited photoluminescence. Appl. Phys. Lett. 2015, 107, 212102. [Google Scholar] [CrossRef]
- Neugebauer, J.; Van de Walle, C.G. Gallium vacancies and the yellow luminescence in GaN. Appl. Phys. Lett. 1996, 69, 503–505. [Google Scholar] [CrossRef]
- Xu, H.Y.; Hu, X.B.; Xu, X.G.; Shen, Y.; Qu, S.; Wang, C.X.; Li, S.Q. Gallium vacancies related yellow luminescence in N-face GaN epitaxial film. Appl. Surf. Sci. 2012, 258, 6451–6454. [Google Scholar] [CrossRef]
- Armitage, R.; Hong, W.; Yang, Q.; Feick, H.; Gebauer, J.; Weber, E.R.; Hautakangas, S.; Saarinen, K. Contributions from gallium vacancies and carbon-related defects to the “yellow luminescence” in GaN. Appl. Phys. Lett. 2003, 82, 3457–3459. [Google Scholar] [CrossRef]
- Lyons, J.L.; Janotti, A.; Van de Walle, C.G. Carbon impurities and the yellow luminescence in GaN. Appl. Phys. Lett. 2010, 97, 152108. [Google Scholar] [CrossRef]
- Reshchikov, M.A.; Demchenko, D.O.; Usikov, A.; Helava, H.; Makarov, Y. Carbon defects as sources of the green and yellow luminescence bands in undoped GaN. Phys. Rev. B 2014, 90, 235203. [Google Scholar] [CrossRef]
- Ogino, T.; Aoki, M. Mechanism of Yellow Luminescence in GaN. Jpn. J. Appl. Phys. 1980, 19, 2395–2405. [Google Scholar] [CrossRef]
- Zhao, D.G.; Jiang, D.S.; Zhu, J.J.; Liu, Z.S.; Wang, H.; Zhang, S.M.; Wang, Y.T.; Yang, H. Role of edge dislocation and Si impurity in linking the blue luminescence and yellow luminescence in n-type GaN films. Appl. Phys. Lett. 2009, 95, 041901. [Google Scholar] [CrossRef]
- Reshchikov, M.A.; Ghimire, P.; Demchenko, D.O. Magnesium acceptor in gallium nitride. I. Photoluminescence from Mg-doped GaN. Phys. Rev. B 2018, 97, 205204. [Google Scholar] [CrossRef]
- Armitage, R.; Yang, Q.; Weber, E.R. Analysis of the carbon-related “blue” luminescence in GaN. J. Appl. Phys. 2005, 97, 073524. [Google Scholar] [CrossRef]
- Mäkelä, J.; Tuominen, M.; Nieminen, T.; Yasir, M.; Kuzmin, M.; Dahl, J.; Punkkinen, M.; Laukkanen, P.; Kokko, K.; Osiecki, J.; et al. Comparison of Chemical, Electronic, and Optical Properties of Mg-Doped AlGaN. J. Chem. Phys. C 2016, 120, 28591–28597. [Google Scholar] [CrossRef]
- Li, S.T.; Jiang, F.Y.; Han, G.F.; Wang, L.; Xiong, C.B.; Peng, X.X.; Mo, H.L. Relationship between structure characteristic and blue luminescence in unintentional doped GaN layers. Mater. Sci. Eng. B 2005, 122, 72–75. [Google Scholar] [CrossRef]
- Reshchikov, M.A.; Korotkov, R.Y. Analysis of the temperature and excitation intensity dependencies of photoluminescence in undoped GaN films. Phys. Rev. B 2001, 64, 115205. [Google Scholar] [CrossRef]
- Yang, H.C.; Lin, T.Y.; Chen, Y.F. Nature of the 2.8-eV photoluminescence band in Si-doped GaN. Phys. Rev. B 2000, 62, 12593–12596. [Google Scholar] [CrossRef]
- Demchenko, D.O.; Diallo, I.C.; Reshchikov, M.A. Hydrogen-carbon complexes and the blue luminescence band in GaN. J. Appl. Phys. 2016, 119, 10. [Google Scholar] [CrossRef]





| Sample | Pressure (Torr) | Temperature (°C) | NH3 (L/min) | [C] (cm−3) |
|---|---|---|---|---|
| P1/T1 | 200 | 1110 | 3 | 2.6 × 1016 |
| P2 | 100 | 1110 | 3 | 1.2 × 1017 |
| P3 | 75 | 1110 | 3 | 3.7 × 1017 |
| P4 | 50 | 1110 | 3 | 1.6 × 1018 |
| T2 | 200 | 1050 | 3 | 4.9 × 1016 |
| T3 | 200 | 1020 | 3 | 1.8 × 1017 |
| T4/F2 | 200 | 1000 | 3 | 3.1 × 1017 |
| F1 | 200 | 1000 | 6 | 8.0 × 1016 |
| F3 | 200 | 1000 | 2 | 1.0 × 1018 |
| F4 | 200 | 1000 | 1 | 3.1 × 1018 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, F.; Zhao, D.; Jiang, D.; Liu, Z.; Zhu, J.; Chen, P.; Yang, J.; Liu, S.; Xing, Y.; Zhang, L.; et al. Carbon-Related Defects as a Source for the Enhancement of Yellow Luminescence of Unintentionally Doped GaN. Nanomaterials 2018, 8, 744. https://doi.org/10.3390/nano8090744
Liang F, Zhao D, Jiang D, Liu Z, Zhu J, Chen P, Yang J, Liu S, Xing Y, Zhang L, et al. Carbon-Related Defects as a Source for the Enhancement of Yellow Luminescence of Unintentionally Doped GaN. Nanomaterials. 2018; 8(9):744. https://doi.org/10.3390/nano8090744
Chicago/Turabian StyleLiang, Feng, Degang Zhao, Desheng Jiang, Zongshun Liu, Jianjun Zhu, Ping Chen, Jing Yang, Shuangtao Liu, Yao Xing, Liqun Zhang, and et al. 2018. "Carbon-Related Defects as a Source for the Enhancement of Yellow Luminescence of Unintentionally Doped GaN" Nanomaterials 8, no. 9: 744. https://doi.org/10.3390/nano8090744
APA StyleLiang, F., Zhao, D., Jiang, D., Liu, Z., Zhu, J., Chen, P., Yang, J., Liu, S., Xing, Y., Zhang, L., Li, M., Zhang, Y., & Du, G. (2018). Carbon-Related Defects as a Source for the Enhancement of Yellow Luminescence of Unintentionally Doped GaN. Nanomaterials, 8(9), 744. https://doi.org/10.3390/nano8090744





