Abstract
This study aimed to investigate the seasonal variation of zooplankton diversity and abundance in the Nokoué Lagoon in southern Benin. Through extensive sampling, a total of 109 zooplanktonic taxa were identified and quantified. The average zooplankton abundance was found to be 60 individuals per liter, with copepods and rotifers being the most dominant groups, comprising 68.1% and 29.1% of the total abundance, respectively. The key factor identified as driving the structure of the zooplanktonic assemblages was salinity, which showed significant seasonal variation. The results revealed that during the high water period, when the lagoon was filled with fresh water, rotifers were dominant, zooplanktonic diversity was highest, and abundances were quite high. Conversely, during the low water period, when the lagoon was characterized by brackish water, diversity was minimal, and abundance decreased slightly. The study also found that some areas of the lagoon showed high abundances independent of salinity levels, suggesting that other factors such as riverine inputs or the presence of acadjas (home-made brush parks used as fish traps) may also have notable effects on the zooplankton community. Overall, the findings of this study provide valuable insights into the functioning of one of the most biologically productive lagoons in West Africa.
1. Introduction
Located in southeastern Benin, the Nokoué Lagoon has been recognized by the Ramsar Convention as a wetland of international importance (Ramsar Site no. 1018). This lagoon represents the largest continental water body in Benin and one of the most biologically productive in West Africa in terms of annual fish catch yields [1]. It contributes to 70% of the national fisheries production [2] and is home to the largest lacustrine villages in West Africa populated by 50,000 people including 12,000 fishermen. Although the Nokoué Lagoon forms a vast natural space that sustains a rich ecosystem, it is surrounded by numerous urbanizations that total more than 1.5 million inhabitants and exert a strong anthropic pressure [3]. Despite its importance for the socio-economic development of Benin and its vulnerability to urban development and global changes, Nokoué Lagoon has been relatively poorly studied. Therefore, it is essential to better characterize and understand the functioning of its ecosystem in order to implement future sustainable management plans.
One of the particularities of Nokoué Lagoon is the strong seasonal variation of its salinity, which varies on average from less than 1 during the heavy rainy season in northern Benin (September–October) and progressively increases during the dry season (December–April) to reach a mean value of ~25 in April [4,5]. These strong seasonal changes in salinity have been shown to structure and strongly impact certain trophic levels of the Nokoué Lagoon ecosystem, such as macroinvertebrates, mangrove oysters, or ichthyofauna [6,7,8]. However, zooplankton is a key organism in the aquatic food chain as it serves as an intermediary species that allows energy transfer between phytoplankton and higher trophic levels such as zooplanktivorous fish. As we know that salinity can control the response of phytoplankton to nutrients and significantly alter zooplankton dynamics in different lagoon ecosystems (e.g., [9,10,11,12]), we therefore hypothesize that strong seasonal variations in salinity in the Nokoué Lagoon generate important changes in zooplankton diversity and abundance.
Although salinity is the parameter that exhibits the greatest seasonal variation and is presumed to be the major environmental parameter impacting the Nokoué ecosystem, other physico-chemical parameters (temperature, depth, pH, turbidity, dissolved oxygen concentration, chlorophyll) also exhibit significant seasonal variations (e.g., [3]). Given their short life cycles, zooplankton organisms can respond rapidly and sensitively to many physical, chemical, and biological changes in aquatic ecosystems [13,14,15]. Zooplankton are therefore highly sensitive to environmental changes, and changes in zooplankton community composition or abundance are often considered indicative of environmental disturbances in coastal lagoons (e.g., [16,17,18,19]). The second hypothesis of this study is therefore that the zooplankton community of Nokoué Lagoon may respond to other physico-chemical parameters than salinity and that the presence and dominance of certain zooplanktonic species could reflect particular environmental conditions.
Furthermore, given that relatively strong spatial gradients in salinity and other physicochemical parameters may exist in the lagoon [3,4,5], we believe that the zooplankton community may respond to these environmental gradients and show spatial differences across the lagoon. In particular, the zooplanktonic community near the river mouths (impacted by freshwater flows) might probably differ from that near the connection between Nokoué Lagoon and the Atlantic Ocean (impacted by saltwater flows).
Despite the importance of zooplankton in the trophic chain and for maintaining the high biological productivity of Nokoué Lagoon, knowledge on the zooplankton of this lagoon is very limited. Indeed, to the best of our knowledge, the only study that has explored the zooplankton community of Nokoué and its relationship with environmental parameters is [20]. In particular, these authors showed that (i) total zooplankton species richness was 31 taxa, (ii) zooplanktonic abundance was relatively low and dominated by copepods, and (iii) the zooplanktonic community responded mainly to nitrate and ammonium concentrations. Unfortunately, as also mentioned by these authors, the sampling was only conducted during a 4-month period (June–September 2015) in the rainy season (low salinity) and did not allow them to investigate the seasonal variation over a full hydrological cycle. Consequently, our study, based on bimonthly sampling carried out during a complete year, aims to fill this gap, and has the following objectives:
- To inventory the zooplanktonic fauna of Nokoué Lagoon,
- To describe the spatio-temporal variations of zooplankton on a seasonal scale, in terms of diversity and abundance,
- To determine the main physicochemical drivers of the zooplankton community and to verify if salinity is the determining parameter.
2. Materials and Methods
2.1. Study Area and Annual Hydrological Cycle
Nokoué Lagoon is a shallow lagoon (~1.3 m depth on average during the dry season) that extends from 2°20′ E to 2°35′ E in longitude and 6°20′ N to 6°30′ N in latitude. This lagoon is bounded to the west by the Abomey-Calavi plateau, to the east by the Porto-Novo Lagoon, to the north by the deltaic floodplain of the Ouémé and Sô Rivers, and to the south by the city of Cotonou (Figure 1). It extends approximately over 20 km from west to east and a maximum of 11 km from south to north, covering an area of ~150 km2 at low water period [3,21,22]. The Cotonou Channel, which is 4 km long and 300 m wide, connects Nokoué Lagoon to the Atlantic Ocean and thus allows freshwater and saltwater exchanges between these two environments (Figure 1) [5]. Nokoué Lagoon also communicates in its western part with the small Djonou River (a few meters wide and a few kilometers long) and in the east with the Porto-Novo lagoon via the Totchè canal, but they have little effect on the dynamics of Nokoué Lagoon [21,22,23,24].
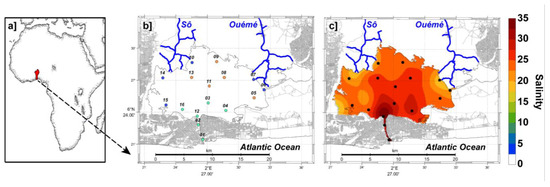
Figure 1.
Location of Nokoué Lagoon (Benin) and hydrographic stations carried out between November 2019 and September 2020. (a) Benin location in West-Africa. (b) Mean salinity (SAL) during high water period in October (SAL ~ 0) and (c) during low water period in April (SAL ~ 25). Station numbers are shown in b]; green dots correspond to stations influenced by the Cotonou Canal and the Atlantic Ocean, blue dots by rivers, and red dots are intermediate stations more representative of the central zone of the lagoon.
The annual hydrological regime of Nokoué Lagoon can be characterized by three main periods [3,23,25]. First, during the heavy rainy season in northern Benin (September–November), the lagoon is filled of freshwater and its water-level strongly increases [5,23]. During this period, the highest species richness of ichthyofauna is observed and the lagoon is populated by freshwater fish species [8]. Second, during the dry season (December–April), the lagoon reaches its low water level and is filled with brackish water with an average salinity of ~25 at the end of the dry season [5,23]. During this period, the species richness of the main taxonomic families of the ichthyofauna decreases and the lagoon is depopulated of some freshwater species not tolerant to these high salinity levels [8]. In contrast, some marine fish species enter the lagoon during this season. Third, during the main rainy season in southern Benin (May–July), the lagoon level rises slightly, and its salinity gradually decreases to an average value of less than 10 [5,23].
2.2. Station Location and Sampling Strategy
This study relies on the analysis of six bimonthly campaigns conducted on Nokoué Lagoon and the Cotonou Channel between November 2019 and September 2020. Each of the field campaigns took place during two consecutive days around the 15th of the corresponding month. They consisted of 16 physicochemical and biological sampling stations (13 in Nokoué Lagoon and 3 in the Cotonou Channel) distributed rather homogeneously in the study area in order to monitor various environmental and biological conditions (Figure 1a). Sampling stations were performed during daytime between ~8 am to ~5 pm. A total of 96 samples were thus taken over the 6 bimonthly campaigns.
2.3. Environmental Parameters
For each of the 96 samples (6 campaigns of 16 stations), the following environmental parameters were acquired at the surface (within the top 20 cm) using both a Conductivity-Temperature-Depth (CTD) probe (Valeport-CTD+) and a multi-parameter probe (WTW-3630IDS): temperature (TEMP, in °C), salinity (SAL), turbidity (TURB, in FTU), dissolved oxygen (DO, in mg L−1). As the water-level of the Nokoué Lagoon strongly varies both spatially and temporally [23], the water-depth (DEPTH, in m) at each station was measured using a Garmin GPSmap 421S echosounder.
Water samples were collected at the surface using a 10 L bucket for measurements of total (organic and inorganic) suspended solids (TSS, in mg L−1), particulate organic matter (POM, in mg L−1), and Chlorophyll-a (CHL-a, in mg m−3). For each water sample, 500 mL were immediately filtered onboard using Whatman GF/F glass fiber filters with a 0.7 µm pore size. The CHL-a concentrations were determined by spectrophotometer after extraction from the filters using 90% acetone [26,27]. For TSS and POM, between 100 and 500 mL (depending on the water turbidity) of water samples were filtered onto both reweighed GF/F filters and Nucleopore membrane filters (0.4 µm pore size). After filtration, filters were dried at 55 °C for 24 h and reweighed to determine TSS concentrations. The GF/F filters were then combusted at 550 °C for 2 h and reweighed to estimate ash weight, POM, and percentage of organic matter (%POM). Based on a statistical analysis, we determined that TSS concentration and turbidity were highly correlated (r2 = 0.97, n = 742, p < 0.05) and collinear (variance inflation factor, VIF = 14.96). We therefore only retained turbidity and discarded TSS from subsequent analyses. Similarly, as organic and inorganic fractions are complementary, we only retained %POM.
2.4. Zooplankton Collection and Sample Analysis
Zooplankton community composition was determined from 96 samples collected at the 16 stations using a plankton net having a mouth opening diameter of 40 cm and a mesh size of 50 µm. A mechanical flowmeter (General Oceanics-2030R6) was placed on the opening of the net to estimate the volume of water filtered. At each site, the net was rinsed with water from the station, and then towed horizontally at a depth of ~50 cm for 30 to 45 s, which allows an average filtration of ~3–4 m3 of water. The collected filtrate was then transferred to a pillbox and preserved in 70 mL of 70% ethanol.
The identification and enumeration of zooplankton was done using a digital microscope (Optika B-290TB) and a panel of taxonomic keys [28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47]. For each station, 1 mL of sample was diluted 5 times with distilled water. Then, 1 mL of this subsample (corresponding to a replicate) was observed under the microscope. The identification and enumeration of zooplankton was carried out on at least 5 independent replicas per station, until a species richness plateau was reached, i.e., when no new zooplankton species were observed in three successive replicas. The zooplankton composition and relative abundance for each station was then determined from all pooled replicas. For each sample, counts of zooplankton taxa were converted to abundance (ind. L−1) taking the number of investigated replicas, the subsample ratio, and the total volume of the sample.
In the present study, copepod nauplii could not be diagnosed at the species level and were thus pooled into one single taxon. Some species could not be formally identified (Table 1) but they were distinguished between them taking into account several morphological characteristics. Each differentiated morphotype was assigned to a distinct (unnamed) species. Differentiation of indeterminate copepod species was performed based on (i) the number of antenna segments, (ii) the cephalosome shape, (iii) the number and appearance of metasoma segments, (iv) the number and size of urosoma segments, and (v) the number and size of setae on each furca. As for the undetermined species of rotifers, they could be distinguished by (i) the body shape and (ii) the appearance and position of certain organs (see references quoted above for taxonomic keys).

Table 1.
List of zooplankton taxa, their relative abundance (in %) and frequency of occurrence (in %). The inventoried taxa were sampled in Nokoué Lagoon during 6 bimonthly surveys carried out between November 2019 and September 2020. The last column indicates the codes of the most frequent (Focc > 0.25) and abundant (relative abundance higher than 0.5%) taxa, used to study their relationship with environmental variables.
The zooplanktonic specific diversity (H’) was estimated by the Shannon index [48,49]:
where i is a specific species, S is the total number of species (or species richness), ni is the number of individuals for species i, and N is the total number of individuals considering all species.
Species evenness was determined by the Pielou’s equitability index (J) [50,51,52]:
This index, which represents the distribution of individuals over species, varies between 0 when a single species dominates, and 1 if all species have an identical abundance.
In addition, the frequency of occurrence (Focc) of each species in all 96 samples was computed (Table 1). A species is considered as frequent if Focc ≥ 0.5, occasional if 0.25 ≤ Focc < 0.5, infrequent, accidental if 0.05 ≤ Focc < 0.25, and rare if Focc < 0.05.
2.5. Data Interpolation and Statistics
For each field campaign, all the results (species richness, abundance, H’, J) were spatially interpolated onto a regular grid of ~100 m × 100 m resolution, using an objective interpolation scheme implemented in MATLAB® [53,54,55].
To test for general significant spatial or temporal variability in the environmental parameters (TEMP, SAL, DEPTH, TURB, %POM, DO, CHL-a) and zooplankton groups, several statistical tests were used. The non-parametric Wilcoxon–Mann–Whitney test (WMW-test) was used to test the significance of variations between the minimum and maximum mean values observed between 2 stations or between 2 campaigns. Similarly, the non-parametric Kruskal–Wallis test (H-test) was applied to test the significance of overall variations observed between the 6 campaigns or between the 16 stations.
In order to test for the effects of the seasons (bi-monthly surveys) and geographic locations (and their interactions) on the environmental variables and zooplankton data, we also used two-way analyses of variance (ANOVA). Two-way ANOVA were performed on Box-Cox transformed data (to approach the hypotheses of normality, independence and homogeneity), and the data were grouped into 3 sub-regions (Figure 1b): the first group includes stations under the direct influence of the Cotonou Channel (green dots in Figure 1b); the second group includes stations under the influence of rivers (blue dots in Figure 1b), while the third group includes stations more representative of the lagoon environment (orange dots in Figure 1b).
General relationships between the diversity and abundance of major zooplankton groups or taxa and environmental variables were investigated using redundancy analyses (RDA) [56]. To do so, we considered only zooplankton groups representing at least 3% of the total diversity and abundance. A Monte Carlo permutation test was used to check the significance (p < 0.05) of the relationships between the zooplankton and each environmental variable, using 10,000 randomizations. Environmental variables were ranked according to their quantitative importance through manual selection based on the Monte Carlo permutation test. Environmental variables that do not significantly (p < 0.05) increase the explained variance were removed from RDA analyses. Prior to RDA analyses, zooplankton abundances were Hellinger-transformed to down-weight the influence of rare species having low counts and many zeros [57]. Note that RDA analyses were used since detrended canonical correspondence analysis (DCCA) revealed maximum gradient lengths of the response data lower than 2 [58].
We also related abundance and species richness of the main zooplankton groups and taxa to individual environmental variables using generalized additive models (GAMs) [59]. The GAMs allow for nonlinearity in the relationships between the predictor variable (environmental parameter) and the response variable (zooplankton abundance or diversity). Low-rank thin plate splines were applied, and the smoothing parameters were determined through restricted maximum likelihood (REML). The best models, associated with the lowest values of Akaike’s information criterion (AIC) and the highest explained deviances, included all 7 environmental variables and 5 degrees of freedom for the smoothing curve functions. In these models, response variables were linked to the additive predictors using log-link functions and Tweedie family distributions. Explained deviances of the GAMs and p-values were examined to retain and describe only those environmental variables that significantly (p < 0.05) explained zooplankton variations. The GAMs were carried out using untransformed abundance data.
All statistical analyses, the RDA, and the GAMs were performed using the free R Statistical Software (version 4.0.2; R Foundation for Statistical Computing, Vienna, Austria) and CANOCO 4.5 software [60]. In particular, we used the R package mgcv [61,62].
3. Results
3.1. Seasonal Variations of Environmental Parameters
Figure 2 shows the seasonal variations of the physicochemical parameters in Nokoué Lagoon from November 2019 to September 2020. H-tests revealed that all physicochemical factors exhibited significant (p < 0.05) bimonthly differences and some of these variables (SAL, DEPTH, DO) also showed significant mean differences between the overall stations (Table 2).
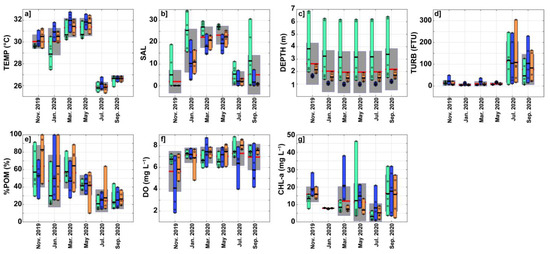
Figure 2.
Seasonal variation of environmental parameters for the six bimonthly surveys carried out between November 2019 and September 2020. (a) Temperature (TEMP, in °C). (b) Salinity (SAL). (c) Station depth (DEPTH, in m). (d) Turbidity (TURB, in FTU). (e) Fraction of particulate organic matter in total suspended material (%POM, in %). (f) Dissolved oxygen concentration (DO, in mg L−1). (g) Chlorophyll-a concentration (CHL-a, in mg L−1). On each subplot, the gray bars show the mean value (red horizontal line) +/− one standard deviation for all 16 stations, while the green, blue, and orange vertical bars show the range of data in each sub-region defined in Figure 1b. These colored bars thus extend from the minimum to the maximum value observed in each sub-region, and the black horizontal lines show the mean values of the data indicated by the black circles.

Table 2.
Results of statistical analyses on environmental and zooplankton variables. Wilcoxon–Mann–Whitney test (WMW-test) was used to test the variations between the minimum (Min.) and maximum (Max.) mean values observed between two campaigns (spatial average) or between two stations (temporal average). Kruskal–Wallis test (H-test) was applied to test the significance of overall variations observed between the six campaigns (temporal analysis, df = 5) or between the 16 stations (spatial analysis, df = 15). Significance levels (S.L.): * p < 0.05, ** p < 0.01, *** p < 0.001, n.s. not significant.
A significant water temperature decrease was observed between maximum values of 30–31 °C from November 2019 to May 2020, and minimum values of 26–27 °C in July and September 2020 during the rainy and flood season (Figure 2a and Table 2). The lagoon is rather spatially homogeneous in temperature, with no significant difference in mean temperature between the stations (TEMP = 28.4–30.1 °C on average, see Table 2). However, the two-way ANOVA analysis revealed significant spatial and temporal variations (without interaction) in the three sub-regions considered (Table 3 and Figure 2a): temperatures tended to be cooler in the Cotonou Channel close to the Atlantic Ocean (green bars in Figure 1a) and warmer in the lagoon and towards the river mouths (orange and blue bars in Figure 2a), more particularly during dry season in January-May.

Table 3.
Significant F-values (p < 0.05) derived from two-way ANOVA on the influence of seasons (bi-monthly campaigns), sub-regions and interaction between seasons and sub-regions on environmental and zooplankton variables. Significance levels: * p < 0.05, ** p < 0.01, *** p < 0.001, n.s. not significant.
Mean salinity values strongly increased from November (SAL ~ 0) to May (SAL > 23), before decreasing to less than 5 in July and September (Figure 2b). The two salinity outliers of November 2019 (SAL of 10–20) and September 2020 (SAL of 20–30) were observed in the Cotonou Channel during flood tide. Indeed, during September–November, strong river discharges were observed and seawater can only marginally penetrate the Cotonou Channel under the effect of the tide (e.g., [5,23]). During most of the bimonthly surveys, the minimum salinity values were observed near the river mouths while the maximum values were observed in the Cotonou Channel sub-region (see also [5]). Both the nonparametric tests and the two-way ANOVA revealed significant spatial and temporal salinity variations (Table 2 and Table 3).
Figure 2c shows that November 2019 corresponded to the end of the high water period whereas May 2020 corresponded to the end of the low water period. Statistical tests suggested that seasonal and spatial DEPTH variations were significant (Table 2 and Table 3). Bathymetry is significantly deeper in the Cotonou Channel (DEPTH > 6 m for Stations 1 and 2) than in the lagoon or close to the river mouths (Figure 2c). This large spatial variability partly masks the non-negligible water-level decrease of 0.60–0.70 m observed between high and low water seasons (see also [23]).
Turbidity was relatively low (<15 FTU) from November 2019 to May 2020, and strongly increased to reach mean values of ~100 FTU at the beginning of the rainy season in July 2020 (Figure 2d and Table 2). The TURB showed significant temporal and spatial variability in the three sub-regions, with higher values close to the river mouths (Figure 2d and Table 3). A closer inspection of the turbidity data revealed the presence of a turbid plume that extends to the south of the Ouémé (east of the lagoon), particularly during the rainy season. Figure 2e showed that %POM, which represents the organic fraction of TSS, was maximum in November 2019 (%POM ~ 60%) and decreased to a minimum in July-September 2020 (%POM ~ 25%). Although statistical analyses did not reveal significant spatial variations in %POM (p > 0.05; Table 2), the %POM was generally slightly higher in the lagoon environment, than close to the river mouth or in the Cotonou Channel (Figure 2e). Similarly, the %POM was on average significantly (WKW-test, p < 0.05) lower at Station 7 (%POM ~ 24%) than at Station 13 (%POM ~ 64%) (Table 2). High TURB values (Figure 2d) were associated with low %POM values (Figure 2e), suggesting that river plumes observed at the beginning of the wet season transport mineral sediments towards the lagoon, thus decreasing the proportion of organic matter.
Mean DO concentration varied from a minimum of less than 6 mg L−1 in November 2019 (Figure 2f), during a flood period when the lagoon was filled of freshwater. In contrast, higher DO concentrations (DO > 7 mg L−1) were observed during low water season between January and July of 2020. Both spatial and temporal variations were significant (p < 0.05, Table 2 and Table 3). During high water period, the water near the river mouths tended to be much less oxygenated than in the rest of the lagoon (Figure 2f).
The CHL-a showed significant temporal variability (p < 0.05 Table 2 and Table 3) with stronger values (>15 mg m−3) during high-water periods in November 2019 and September 2020, and lower values (<10 mg m−3) in low-water season between January and July 2020 (Figure 2g). Significantly higher CHL-a concentrations were also generally observed near the river mouths (Figure 2g and Table 3). In contrast to the other months, CHL-a in January 2020 was very homogeneous throughout the lagoon and had relatively low values (5–10 mg m−3).
3.2. General Distribution of the Zooplankton Community
A total of 109 zooplanktonic species were inventoried, including 81 taxa of rotifers, 20 of copepods (without considering nauplii), five of cladocerans and three organisms that belonged to three other zooplankton groups (Eumalacostraca, Mollusca, and Ostracoda) (Table 1 and Figure 3). Among all the organisms identified, one rotifer taxon (Brachionus plicatilis), four copepod taxa (cyclopoid sp. 1, Ectocyclops sp., calanoid sp. 1, and Microsetella sp.), copepod nauplii and unidentified mollusks were considered frequent species, with Focc ranging from 51% to ~98% (Table 1). The other species identified were either occasional (15 taxa), accidental (46 taxa) or rare (42 taxa) species (Table 1).
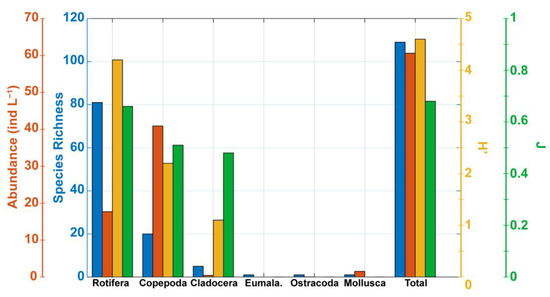
Figure 3.
Overall species richness (blue), mean abundance (red), Shannon species diversity index (H’, yellow) and Piélou equitability index (J, green) for each of the zooplankton groups (Rotifera, Copepoda, Cladocera, Eumalacostraca, Ostracoda, Mollusca) as well as for the entire zooplankton community (Total).
Considering the six field campaigns, the average abundance of zooplankton organisms was ~60 ind L−1. This abundance was dominated by copepods (68.1% of the individuals) and rotifers (29.1%) (Figure 3). The average relative abundance of Mollusca was weak (2.5%), whereas cladocerans, Eumalacostraca, and Ostracoda were negligible (Figure 3). For the rest of the manuscript, we focused mainly on the two main groups of zooplankton, namely copepods and rotifers, which represented 92.7% of the total species richness and 97.2% of the zooplankton abundance.
The Shannon diversity index showed an overall value of 4.6 (Figure 3). This index, computed for each group, indicated that rotifers were the most diverse group (H’ = 4.2) followed by copepods (H’ = 2.2) (Figure 3). On average, the relative abundance of each species was rather homogeneous within the copepods, rotifers and cladocerans (J ~ 0.5–0.7) (Figure 3).
On average over the six surveys, the number of zooplanktonic species observed varied non-significantly (p > 0.05) between 13 and 18 species depending on the stations (Table 2). Similarly, no significant variation in species richness was observed between the three sub-regions considered (Table 3). Despite a significant decrease in mean zooplankton abundance from a maximum of ~190 ind L−1 in the northwest of the Lagoon to a minimum of ~20 ind L−1 in the Cotonou Channel (p < 0.05), no significant spatial difference (p > 0.05) was observed when considering the abundance distributions at each of the 16 stations or between the three sub-regions (Table 2). This may be due to the presence of multiple intermediate values that reduced noise and increased the statistical power of the analysis, thereby masking the dissimilarity that existed between the two extreme abundance distributions observed between the Channel and northwest of the Lagoon.
On average, copepods were the dominant group, representing 50–80% of the total abundance in the lagoon and reaching almost 90% at the connection of the Cotonou Channel with the Atlantic Ocean (Station 1) (Table 2). In contrast, the relative abundance of rotifers was relatively low (10–50%) over the whole lagoon (Table 2). Spatial variations in relative fractions of copepods or rotifers were not significant (p > 0.05; Table 2 and Table 3).
The averaged Shannon species diversity index values varied from 1.7 to 2.7 but these weak spatial variations were not significant (p > 0.05; Table 2). Piélou’s equitability index values broadly followed the H’ distribution, suggesting that where specific diversity was higher in the lagoon, abundance was more evenly distributed among the different zooplankton species. The Piélou’s equitability index values significantly varied between 0.4 and 0.7 (p < 0.01; Table 2).
3.3. Seasonal Variation of the Zooplankton Community
Zooplankton species richness peaked during the flood period in November 2019, with 30–40 distinct zooplanktonic taxa at each station (Figure 4a). During the low water period, from January to May 2020, the species richness decreased sharply down to 10–15 taxa at each station. During the rainy period (July 2020) and the beginning of the high water period (September 2020), species richness gradually increased and reached 20–30 species per station in September 2020. These seasonal variations in species richness were highly significant (p < 0.001) unlike spatial variations (Table 2 and Table 3).
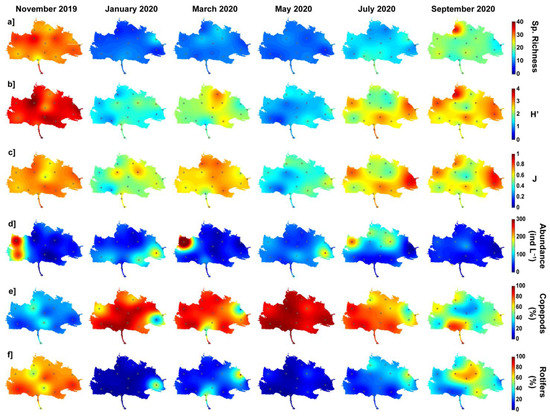
Figure 4.
Spatial distribution of bimonthly variation of different zooplankton parameters. (a) Species richness (1st row), (b) Shannon species diversity index (H’, 2nd row), (c) Piélou equitability index (J, 3rd row), (d) Total abundance (in ind L−1, 4th row), (e) Copepod relative abundance (in %, 5th row), and (f) Rotifer relative abundance (in %, 6th row).
Spatio-temporal variation in zooplankton abundance was more complex (Figure 4d). Throughout the year, abundance was relatively low in the lagoon (typically 15–40 ind L−1) except in specific locations, usually to the east or west, where values could locally increase to 200–400 ind L−1. The observed spatio-temporal variations, considering all stations/campaigns or the three sub-regions, are not statistically significant (Table 2 and Table 3). However, abundance distributions differed significantly (p < 0.05) between Station 2 located in the Cotonou Channel (mean abundance of 20 ind L−1) and Station 14 located in the northwestern part of the lagoon (>190 ind L−1) (Table 2).
The relative proportion of copepods varied inversely with abundance (Figure 4e). In November 2019 and September 2020, the percentage of copepods was relatively low in most of the lagoon, with relative abundance values below 60% and 40% in September and November, respectively. During this period, the zooplanktonic assemblage was therefore dominated in abundance by rotifers (Figure 4f). However, during the low water period, from January to May 2020, the proportion of copepods significantly increased (p < 0.001; Table 2 and Table 3) and exceeded 80% in most areas of the lagoon (Figure 4e). In contrast to temporal variations, spatial variations were not significant except for the proportion of rotifers which showed, on average, significantly (p < 0.05) lower values in the Cotonou Channel (Station 1~11%) than near the Ouémé River (Station 7~51%) (Table 2).
The combination of species richness and abundance led to diversity index values that varied similarly to species richness (Figure 4b). The Piélou equitability index values varied similarly, suggesting that abundance was relatively well distributed among species when diversity was high, while some species dominated when abundance was low (Figure 4c). The observed bi-monthly variations of both H’ and J were significant (p < 0.001; Table 2 and Table 3).
In general, spatial variations in zooplankton were not statistically significant, either between all stations or between the three sub-regions more influenced by rivers or the ocean (Table 2 and Table 3). However, we showed that the zooplankton community of the Nokoué Lagoon tended to significantly vary on a seasonal scale, and that it was likely to be strongly impacted by environmental parameters that also showed significant seasonal variability (Figure 2 and Table 2 and Table 3). Therefore, we analyzed more thoroughly the relationships between physicochemical factors and zooplankton composition.
3.4. Relationship between Rotifer and Copepod Diversity and Environmental Factors
Zooplankton diversity of the two most abundant groups (rotifers and copepods) was combined with environmental variables to identify the main drivers of the general zooplankton community structure using a RDA (Figure 5a). Four environmental variables significantly constrained (p < 0.05) the overall variance of the zooplankton diversity, explaining 54.8% of the total variability. The environmental variable with the greatest explanatory power for zooplankton species richness was salinity (F = 59.6, p < 0.001) which explained 38.8% of the total variability, followed by dissolved oxygen (F = 15.5, p < 0.001) which explained 8.8%. Turbidity (F = 7.9, p < 0.01) and station depth (F = 5.5, p < 0.01) were of secondary importance, explaining 4.4% and 2.8% of the total variance, respectively. The first RDA axis was mainly scored by salinity (r = −0.64) followed by oxygen (r = −0.43), whereas axis 2 was mainly scored by water-depth (r = 0.26) followed by the three other parameters (r = ±0.12–0.15) (Figure 5a). The RDA analysis revealed that higher copepod diversity was primarily associated with higher salinities (Figure 5a). In contrast, higher rotifer species richness was related to weaker SAL and DO values.
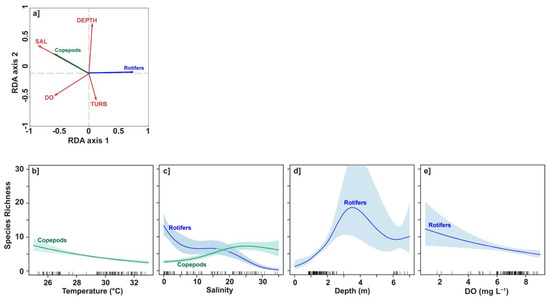
Figure 5.
Redundancy analyses (RDA) and generalized additive models (GAMs) relating diversity of copepods and rotifers to environmental variables. (a) RDA diagram of species richness and environmental parameters. (b–e) GAMs of species richness versus temperature (TEMP), salinity (SAL), depth (DEPTH) and dissolved oxygen (DO). Only significant (p < 0.05) relationships are shown for copepods and rotifers. Shaded areas indicate the 95% confidence intervals.
To further investigate the impact of key environmental factors on the diversity of each of the dominant groups (rotifers and copepods), the GAMs were fitted on the response of rotifer and copepod species richness to environmental parameters. Considering all seven environmental variables, the GAMs had the highest explained deviance (76% for rotifers and 57.8% for copepods) and adjusted R2 (0.70 for rotifers and 0.53 for copepods). The response plots were shown in Figure 5b–e, only for variables with a significant effect (p < 0.05).
The most significant parameter for copepod diversity was SAL followed by TEMP (p < 0.001). Copepod diversity decreased almost linearly with temperature, slightly varying from 7–8 species for TEMP < 26 °C to 3–4 species for TEMP > 32 °C (Figure 5b). In contrast copepod species richness increased with salinity, from ~3–4 species in freshwater to 6–7 species for SAL > 20 (Figure 5c). Copepod species richness was not significantly (p > 0.05) influenced by other environmental variables and was therefore not shown in Figure 5d,e.
In a similar way, the most significant parameter for rotifer diversity was SAL (p < 0.001) followed by DEPTH (p < 0.001) and DO (p < 0.01). The other environmental parameters do not significantly (p > 0.05) influence rotifer species richness. The evolution of rotifer species richness as a function of salinity showed opposite variations to that of copepods, with a much higher diversity in low-salinity water, and only very few species persisted when SAL > 20 (Figure 5c). The diversity of rotifers tended to be higher at the deepest (Figure 5d) or least oxygenated (Figure 5e) stations. Note, however, the large errors for depths between 3–6 m (Figure 5d) or DO values less than 5 mg L−1, due to the lack of data in these ranges.
3.5. Relationship between Rotifer and Copepod Abundance and Environmental Factors
We investigated the main drivers of the general zooplankton abundance, applying a RDA to the abundance of the two main zooplankton groups (i.e., rotifers and copepods) combined with environmental variables (Figure 6a). The RDA used three variables that significantly constrained (p < 0.05) the overall variance of the zooplankton abundance, explaining 47.8% of the total variability: the environmental variable with the greatest explanatory power for zooplankton abundance was salinity (F = 55.9, p < 0.001) which explained 37.3% of the total variability. Water temperature (F = 9.6, p < 0.01) and station depth (F = 8.1, p < 0.01) were of secondary importance, explaining 5.5% and 5% of the total variance, respectively. The first RDA axis was mainly scored by salinity (r = 0.62) followed by temperature (r = 0.19), whereas axis 2 was mainly scored by temperature (r = −0.29) followed by water depth and salinity (r = ±0.07) (Figure 6a). The RDA analysis revealed that higher abundances of copepods and lower abundances of rotifers were mainly associated with higher salinities.
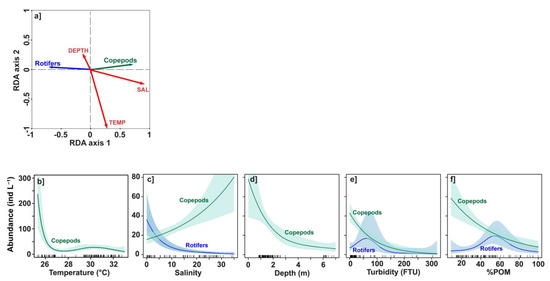
Figure 6.
Redundancy analyses (RDA) and generalized additive models (GAMs) relating abundance of copepods and rotifers to environmental variables. (a) RDA diagram of abundances and environmental parameters. (b–f) GAMs of abundances versus temperature (TEMP), salinity (SAL), depth (DEPTH), turbidity (TURB) and dissolved oxygen (DO). Only significant (p < 0.05) relationships are shown for copepods and rotifers. Note the change in scale between subplots (b) and (c–e). Shaded areas indicate the 95% confidence intervals.
Nonlinear relationships between the abundances of each of the dominant zooplankton groups (rotifers and copepods) and environmental variables were also explored through GAMs (Figure 6b–f). The GAMs including the seven environmental variables have explained deviances of 50.6% for rotifers and 55.2% for copepods, and adjusted R2 of 0.14 for rotifers and 0.41 for copepods. As previously, response plots were presented in Figure 6b–e, only for variables with a significant effect (p < 0.05).
The copepod abundance variability is significantly related to five environmental parameters (p < 0.001; Figure 6b–f). Copepod abundance strongly decreased for temperature between 25.5 °C and 26 °C, and then remained around 15–25 ind L−1 for TEMP > 26 °C (Figure 6b). Note, however, that the high abundance values for TEMP < 26 °C were highly uncertain because of the lack of data in these temperature ranges. Copepod abundance increased progressively from ~20 ind L−1 in freshwater to more than 60 ind L−1 for SAL > 30 (Figure 6c). For the other environmental variables (DEPTH, TURB, %POM), the abundance of copepods was found to be higher the lower the depth of the stations, the turbidity of the water and the fraction of organic matter (Figure 6d–f).
The rotifer abundance variability was significantly related to SAL (p < 0.001) followed by %POM (p < 0.01) and TURB (p < 0.05). Rotifers were more abundant for lower salinities and even dominant (>20 ind L−1) for SAL < 3. Rotifer abundance rapidly decreased with salinity and was less than 10 ind L−1 for SAL > 10 (Figure 6c). Variations in rotifer abundance as a function of TURB and %POM were more complex and showed a bell shape with relatively high values (~20 ind L−1) for intermediate values of turbidity (~50 FTU) and organic matter (~50%) (Figure 6e,f). However, associated errors were also important for these fitted GAMs. In contrast to copepods, station depths and water temperature did not have a significant impact (p > 0.05) on variations in rotifer abundance.
3.6. Relationship between Abundance of the Most Frequent Taxa and Environmental Factors
In this section, we first examined the general relationship between the abundances of the most frequent (Focc > 25%) and most abundant (>0.5%) zooplanktonic taxa and environmental parameters, using an RDA. Therefore, 16 taxa were considered, including five copepod taxa (including Nauplius), 10 rotifer taxa and Mollusca sp. The list of these taxa and their frequency of occurrence were presented in Table 1 and Figure 7. The RDA used four variables that significantly constrained (p < 0.05) the overall variance of the species-specific abundances, explaining 23% of the total variability (Figure 7a). The environmental variable with the greatest explanatory power was again salinity (F = 12.0, p < 0.001), which explained 11.3% of the total variability. The DO concentration (F = 5.1, p < 0.01), TEMP (F = 4.2, p < 0.01) and CHL-a (F = 3.9, p < 0.01) concentration, were of secondary importance, explaining 4.7%, 3.6%, and 3.4% of the variance, respectively. The first RDA axis was mainly scored by SAL (r = 0.67) followed by CHL-a (r = −0.40), whereas axis 2 was mainly scored by DO (r = 0.54) and TEMP (r = −0.40) (Figure 6a). Higher abundances of the most frequent copepod taxa were in general associated with higher salinities, whereas the abundance of the most frequent rotifer taxa likely increased with low salinity (or high CHL-a) values.
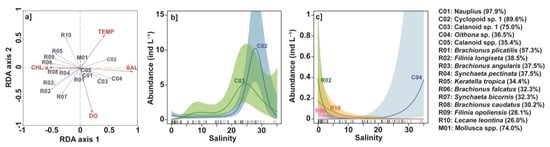
Figure 7.
Redundancy analyses (RDA) and generalized additive models (GAMs) relating abundance of the most frequent zooplankton taxa to environmental variables. (a) RDA diagram of taxa abundances and environmental parameters. (b,c) GAMs of taxa abundances versus salinity (SAL). Only the three most significant (p < 0.05) relationships are shown for copepods and rotifers. Note the change in scale between subplots (b) and (c). Taxa were coded according to the legend shown on the right (see also Table 1). Shaded areas indicate the 95% confidence intervals.
To further investigate the impact of environmental variables on the most frequently observed zooplankton taxa, we used GAMs to relate the seven environmental variables to these different taxa. Depending on the taxon considered, the explained deviance varied between a minimum value of 33.4% for Calanoid spp. (coded C05) and a maximum value of 94.5% for Brachionus caudatus (coded R08) (Table 4). The taxa with the strongest relationship with environmental variables (highest explained deviance) belonged to the rotifer group (e.g., Brachionus caudatus, Brachionus angularis, Filinia opoliensis, Brachionus falcatus, Lecane leontina) (Table 4). The most important variable was SAL, followed by CHL-a, as also noted on the RDA (Table 4 and Figure 7a).

Table 4.
Each row summarizes the GAM for that particular taxa: percent deviance explained (%Deviance) and significance of each environmental variable. Black cells represent p-values < 0.001, dark gray cells represent p-values < 0.01, light gray cells represent p-values < 0.05, and white cells represent non-significant p-values.
Salinity was significantly related (p < 0.05) to the abundance of 13 of the 16 taxa most frequently and abundantly observed in Nokoué Lagoon (Table 4), and very significantly related (p < 0.001) to nine of them. We were therefore studying more specifically the relationship between zooplankton taxa and salinity. However, for the sake of clarity and conciseness, we presented in Figure 7b,c only the three taxa of copepods (Cyclopoid sp. 1, Calanoid sp. 1, Oithona sp.) and rotifers (Filinia longiseta, Lecane leontina, Brachionus falcatus) for which the partial deviances explained by salinity were the most important. Concerning the copepods, abundances of Cyclopoid sp. 1 (coded C02) and Calanoid sp. 1 (coded C03) showed almost unimodal distributions, with maximum abundances for salinities between 20 and 30 (Figure 7b). The abundance of Oithona sp. (coded C04) also increased for salinities above 20, but its abundance is an order of magnitude lower than that of the other two copepod taxa (Figure 7c). Concerning the rotifers, abundances of Filinia longiseta (coded R02) Brachionus falcatus (coded R06) strongly increased for SAL < 5, whereas the abundance of Lecane leontina (coded R10) started to increase for SAL < 15 (Figure 7c). In very low-salinity waters, Filinia longiseta dominated the two other copepod taxa. Although not shown, the abundance of other rotifer taxa showed similar relationships with salinity, with the exception of Brachionus plicatilis (coded R01) which showed a roughly unimodal distribution with a marked maximum for a SAL ~ 8. This planktonic species is known to be a euryhaline rotifer that tolerates a wide range of salinities.
4. Discussion
4.1. Zooplankton Composition and Diversity
This study enabled us to identify 109 distinct zooplanktonic taxa in the Nokoué Lagoon, including 81 taxa of rotifers, 20 of copepods, five of cladocerans and three other organisms. Representing ~75% of the species richness, rotifers were thus the most diversified group, as also observed in other regional tropical lagoons, from Ivory Coast to Nigeria [63,64,65,66]. The total species richness of 109 zooplanktonic taxa recorded in our study far exceeded that previously reported in Nokoué Lagoon (31 taxa) [20]. This difference could be explained by the sampling that covered here the whole hydrological cycle and a wide range of salinity, while in the previous study zooplankton were analyzed between June and September when salinity was low [20]. In addition, the methodology implemented in our study (see Section 2.1.) was different from that used previously (mesh size, volumes filtered, location of sampling stations, number of replicates, etc.). Note that taxa not identified to specific levels (see Table 1) have been conscientiously distinguished from each other on the basis of clear morphological features (see Section 2.3). As a consequence, the taxonomic list obtained here (Table 1), and the resulting greater diversity cannot be attributed to misclassification and erroneous separation of individuals belonging to the same species.
Relationships between zooplankton diversity and environmental variables, as investigated through RDA and GAMs, showed that salinity was the key parameter for species richness, explaining a large part of the total variance. Consequently, seasonal salinity variations in Nokoué Lagoon, which strongly varied from 0 in the flood period to ~24 in low-water period (Figure 2b; see also [5]), were associated with strong changes in the structure and diversity of zooplankton assemblages. During low-salinity (flood) periods, a higher diversity of rotifers, and thus zooplankton, was observed (Figure 4, Figure 5 and Figure 6), consistently with other regional studies e.g., [65,67]. During floods, a relatively high freshwater inflow (up to 1200 m3 s−1) from the Sô and Ouémé rivers was observed [23,68]. These river discharges could transport into the lagoon the numerous zooplankton species (~100 zooplankton taxa, including ~90 rotifers) observed in the rivers [69]. Furthermore, the high dominance of rotifers during the flood period in November 2019 (Figure 4f) was associated with relatively low DO values (Figure 2f), as also suggested by the GAMs (Figure 5e). These low DO values are likely related to the degradation of a significant amount of POM in the lagoon (Figure 2e) that can originate, during the flood season, from several sources, such as the Ouémé deltaic plain, urban water drainage, or acadjas (i.e., traditionally made bamboo parks used by local fishermen to grow fish) [19,69]. The degradation of this POM, which consumes oxygen, was generally the main source of nutrients for rotifers [10,63,70,71] which may have increased rotifer diversity during the period of very low salinity and low DO concentrations during flooding. The composition and diversity of rotifers can also reflect a certain level of eutrophication in Nokoué Lagoon. Indeed, rotifers are among the only zooplanktonic organisms that are resistant to high organic matter enrichment and dissolved oxygen depletion [72,73]. The high diversity of Brachionidae and the presence of Brachionus falcatus (Figure 7), could be an indicator of eutrophication in Nokoué Lagoon [74,75], as well as the species Cephalodella gibba which was often associated with high POM concentrations. In the future, additional measurements of eutrophication, especially nitrogen and phosphorus compounds, would be needed to better interpret zooplankton structure and their use as a bioindicator of environmental conditions in Nokoué Lagoon.
During the low water period, Nokoué Lagoon was very salty, and we observed a strong decrease in the diversity of rotifers associated with an increase in the diversity of copepods (Figure 4 and Figure 5), once again corroborating other regional studies [11,66]. Salinity tends to cause stressful ecological conditions and decreases biodiversity in lagoon ecosystems, particularly rotifers and cladocerans [12]. Rotifers are typically dulcaquicolous organisms (1488 taxa out of 1570 described in this phylum [76]). Only halotolerant rotifers could withstand the haline stress observed in Nokoué Lagoon, such as Synchaeta bicornis, or more particularly Brachionus plicatilis as observed in Figure 7 [77,78,79]. These species could tolerate wide variations in salinity [79,80,81,82].
In Nokoué Lagoon, total species richness decreased progressively with increasing salinity. These results may seem to contradict the well-accepted Remane’s (1934) theory [83], according to which taxonomic diversity would reach a minimum, called the Artenminimum, for salinities of 5 to 8 [83,84,85]. However, this concept of Artenminimum was mainly based on observations of the diversity of benthic invertebrates in the Baltic Sea, and not of the diversity of pelagic organisms such as zooplankton. Although Remane’s species-minimum model has been widely used to explain biodiversity changes along haline gradients, it has also been shown to be inadequate to explain phyto- and zooplanktonic diversities in estuarine habitats (e.g., [86,87,88,89]). Indeed, in these regions, and as observed in the Nokoué Lagoon, planktonic organisms often did not show minimal diversity at salinities intermediate between marine and freshwater (e.g., [89,90,91,92,93,94,95]).
4.2. Zooplankton Abundance
Zooplankton abundance was, on average, ~60 ind L−1 across the Nokoué Lagoon, but locally as high as 100–300 ind L−1 (Figure 3 and Figure 4). These values were of the same order of magnitude as those obtained in other regional lagoons: 100–500 ind L−1 in Fresco Lagoon, 120–160 ind L−1 in Aby-Tendo-Ehy lagoons, and 50–250 ind L−1 in Ebrié Lagoon [65,66,67]. However, they were slightly lower than the average abundances of 100–400 ind L−1 obtained previously in Nokoué Lagoon [20]. This difference may be explained by inter-annual variations as well as by the use of a net with much finer mesh by these authors (30 μm instead of 50 μm), which was likely to collect smaller zooplankton organisms, thus increasing zooplankton abundance.
One of our hypotheses was that the Nokoué Lagoon could show significant differences between the Cotonou Channel located near the Atlantic Ocean and the areas near the river mouths. The separation of the stations into three distinct groups showed indeed that some environmental variables, and in particular salinity, had significant variations (p < 0.05) between the three sub-regions (Figure 2 and Table 3) but that this was not the case for zooplankton diversity and abundance (Table 3). However, significant differences on zooplankton may appear between stations with extreme values (see MWM-test in Table 2). In particular, local increase in zooplankton abundance was noted at some stations located at the west and east of Nokoué Lagoon (Figure 4). First, this could be related to the freshwater inflows from the So and Ouémé rivers, which resulted in local desalination processes in these particular areas of the lagoon [5]. These events may be favorable to freshwater zooplankton communities, especially rotifers as well as nauplii, which could maintain high abundances in less saline environments (Figure 6 and Figure 7). Second, the relatively high zooplankton abundance in the western part of Nokoué Lagoon could also be related to the increased presence of brush park fisheries (acadjas), used in these areas for trapping and artisanal fishing e.g., [96,97]. Indeed, these artificial parks lead to the local development and increase of biological productivity due to the contribution of nutrients through the decomposition of organic matter from woody materials [1,96,98]. Moreover, acadjas parks, which are made up of more than 15 branches per square meter and extend over several hectares [96,97], tend to modify local hydrodynamics by decreasing the intensity of winds and currents [99]. This reduced vertical mixing and turbulence may also enhance the development of zooplankton (e.g., [100]). Thus, higher nutrient concentration and lower turbulence in these zones could explain the higher local zooplankton abundance. Accordingly, it has already been shown that phytoplankton and zooplankton abundances could be four times higher, but that their diversity was lower, in the acadjas than in the surrounding areas [99,101]. However, our current dataset did not allow us to highlight a possible link between the proximity of the acadjas to our stations (all carried out outside of these brush parks) and the structure of the zooplankton community. For instance, we did not observe any increased presence of species with benthic tendencies (e.g., Lecanidae, Lepadellidae, Mytilinidae, Notommatidae, Chydoridae, Macrothricidae) or bacterivores (e.g., Brachionidae) at these particular stations. More specific studies on the impact of acadjas on the structure of planktonic communities will therefore be necessary to reach any definitive conclusions.
The RDA and GAMs analyses revealed a strong relationship between salinity and zooplankton abundance. Indeed, salinity was the primary driver of zooplankton abundance, explaining 37% of the overall variance in zooplankton taxa abundance, and 23% of the variance in the most frequent taxa. Our results also showed that zooplankton abundance was higher during the dry season, associated with high salinities (low-water period), than in the wet season characterized by low salinity (flood period). These results are similar to those noted in Ivorian lagoons of Aby-Tendo-Ehy, Fresco and Ebrié [65,66,67]. The high zooplanktonic abundance during the low-water period was related to the dominance of copepods and in particular their larvae (nauplii) which represented ~60% of the relative zooplanktonic abundance. During this period, the dominance of brackish water conditioned the proliferation of these halotolerant taxa of copepods from the coastal ocean. The rotifers were then in the minority.
Copepods in the Nokoué Lagoon included both freshwater and marine species, which may have contrasting responses to environmental parameters. However, due to limitations in our dataset, we were unable to differentiate between unidentified Calanoida and Cyclopoida species in terms of their affinity for freshwater or marine environments. Nevertheless, many of these unidentified copepod species were observed throughout the year and were able to tolerate highly contrasting salinity gradients. Based on our analysis, Cyclopoid sp. 1 and Calanoid sp. 1 were likely to correspond to marine species due to their higher abundance in higher salinity conditions (Figure 7). Identified copepod species were divided into freshwater and marine groups based on their known affinity for salinity. However, the low frequencies of occurrence and relative abundances of these identified species (Table 1) limited the ability to draw robust conclusions about the relationship between salinity and copepod diversity. Further research, with a longer monitoring period and a more comprehensive zooplankton dataset, is needed to fully understand the contrasting relationships that freshwater and marine copepod species may have with environmental parameters in the Nokoué Lagoon. It would also be interesting to investigate the percentage of freshwater and marine species present in the copepod community and how this changes in response to environmental conditions, which could provide valuable insights into the ecological dynamics of copepods in the lagoon.
Finally, on an interannual scale, between June and September 2015, 90% of the zooplanktonic abundance was copepods and 10% was rotifers [20]. Between July and September 2020, our study showed a reduced proportion of copepods (~70%) and an increase proportion of rotifers (30%). This interannual variability could be explained by the difference in salinity between the two periods. In June-September 2015, the average salinity was of 6.2 [20], while it was only ~3 in July-September 2020 (Figure 2). Based on the highlighted relationships between salinity and zooplankton species distribution (Figure 6 and Figure 7), it was therefore consistent to observe a lower abundance of copepods between July and September 2020. Although interannual variations in salinity probably explain the changes in copepod and rotifer distributions between 2015 and 2020, a temporal shift in zooplanktonic successions during these 2 years cannot be ruled out either, which cannot be assessed with our bimonthly sampling approach.
5. Conclusions
Based on the analysis of biological data from six bimonthly campaigns, we identified 109 distinct zooplankton taxa in Nokoué Lagoon. Average zooplanktonic abundance was ~60 ind L−1 but increased locally to 100–300 ind L−1. This abundance was largely dominated by copepods and rotifers, which represented, on average, 68.1% and 29.1% of organisms, respectively.
Environmental parameters showed significant seasonal variations, especially in terms of the salinity. Indeed, Nokoué Lagoon was filled with fresh water during the flood period in November 2019 and progressively salinized to reach average salinity values of ~22–25 during the low water period in March-May 2020. We showed, through redundancy analyses, that salinity was the key parameter that structured the zooplanktonic ecosystem, both for taxon diversity and abundance. Therefore, on a seasonal scale, a strong shift was observed in the zooplanktonic community. Indeed, during the flood period, abundance was quite high and zooplanktonic diversity was maximal. During this short period, rotifers were dominant and about 30 zooplanktonic species could be observed in each sampling station. In contrast, during the low water period, the diversity became minimal (less than 10 species in each station), and the abundance slightly decreased. However, some zooplankton hotspots were observed in the west and east of the lagoon, likely independent of salinity. These localized areas of high abundance may have been under the influence of other processes, such as the Sô and Ouémé river inputs and/or the presence of acadjas brush parks whose effects on zooplankton could not be determined with our dataset. More specific studies as well as the continuation of regular and long-term sampling will help us to understand further such aspects of zooplanktonic structuring and its fine-scale spatio-temporal variations.
The results obtained in this study provided valuable information on the seasonal variations of zooplankton in Nokoué Lagoon, which is one of the most biologically productive in West Africa. Future studies should focus on the simultaneous analysis of the different compartments of the trophic chain (phytoplankton, zooplankton, fish) and their responses to environmental variables. Such an approach will complete our study and lead to a comprehensive view of the functioning of this rich ecosystem that provides 70% of the Beninese fishery resource.
Author Contributions
Contributed to conception and design: A.C., H.H.A. and G.D. Contributed to acquisition of data: F.T.O., T.T.A., F.U.D.-S., V.O.O. and M.B.D. Contributed to zooplankton identification: F.T.O., T.T.A., F.U.D.-S. and F.A. Contributed to analysis and interpretation of data: F.T.O., A.C. and F.A. Contributed to statistical analyses, article writing and figure design: A.C. and F.T.O. Contributed to the proofreading and correction of the article: all authors. All authors have read and agreed to the published version of the manuscript.
Funding
Field campaigns and instrumentation were supported by IRD. This work is a contribution to the « JEAI SAFUME » project funded by IRD. V. OKPEITCHA was funded by OmiDelta project of the Embassy of the Netherlands in Benin, through a scholarship grant of the National Institute of Water (INE). F. OUINSOU and T. AVOCEGAN received funding from IRD.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
Special thanks to the members and crew participating to the bimonthly surveys, and in particular A. ASSOGBA, A. B. TIGO, M. BENOIST, and J. AZANKPO. Collaboration of Team 2/ODA-INE is also acknowledged.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lalèyè, P.; Villanueva, M.; Entsua-Mensah, C.M.; Moreau, J. A review of the aquatic living resources in Gulf of Guinea lagoons with particular emphasis on fisheries management. J. Afrotropical Zool. 2007, 10, 123–136. [Google Scholar]
- Gnohossou, P. La Faune Benthique d’une Lagune Ouest Africaine (le Lac Nokoue au Benin), Diversite, Abondance, Variations Temporelles et Spatiales, Place dans la Chaine Trophique. PhD Thesis, Institut National Polytechnique de Toulouse, Toulouse, France, 2006; p. 169. [Google Scholar]
- Djihouessi, M.B.; Aina, M.P. A review of hydrodynamics and water quality of Lake Nokoué: Current state of knowledge and prospects for further research. Reg. Stud. Mar. Sci. 2018, 18, 57–67. [Google Scholar] [CrossRef]
- Mama, D.; Aina, M.; Alassane, A.; Boukari, O.; Chouti, W.; Deluchat, V.; Bowen, J.; Afouda, A.; Baudu, M. Caractérisation physico-chimique et évaluation du risque d’eutrophisation du lac Nokoué (Bénin). Int. J. Biol. Chem. Sci. 2011, 5, 20–76. [Google Scholar] [CrossRef]
- Okpeitcha, V.; Chaigneau, A.; Morel, Y.; Stieglitz, T.; Pomalegni, Y.; Sohou, Z.; Mama, D. Seasonal and interannual variability of salinity in a large West-African lagoon (Nokoué Lagoon, Benin). Estuar. Coast. Shelf Sci. 2021, 264, 107689. [Google Scholar] [CrossRef]
- Odountan, O.; de Bisthoven, L.J.; Koudenoukpo, C.; Abou, Y. Spatio-temporal variation of environmental variables and aquatic macroinvertebrate assemblages in Lake Nokoué, a RAMSAR site of Benin. Afr. J. Aquat. Sci. 2019, 44, 219–231. [Google Scholar] [CrossRef]
- Agadjihouede, H.; Akele, D.G.; Gougbedji, A.U.M.; Laleye, P.A. Exploitation de l’huître des mangroves Crassostrea Gasar (Adanson, 1757) dans le lac Nokoué au Bénin. Eur. Sci. J. 2017, 13, 352. [Google Scholar] [CrossRef]
- Lalèyè, P.; Niyonkuru, C.; Moreau, J.; Teugels, G. Spatial and seasonal distribution of the ichthyofauna of Lake Nokoué, Bénin, west Africa. Afr. J. Aquat. Sci. 2003, 28, 151–161. [Google Scholar] [CrossRef]
- Marcarelli, A.M.; Wurtsbaugh, W.A.; Griset, O. Salinity controls phytoplankton response to nutrient enrichment in the Great Salt Lake, Utah, USA. Can. J. Fish. Aquat. Sci. 2006, 63, 2236–2248. [Google Scholar] [CrossRef]
- Zakaria, H.Y.; Radwan, A.A.; Said, M.A. Influence of salinity variations on zooplankton community in El-Mex Bay, Alexandria, Egypt. Egypt. J. Aquat. Res. 2017, 33, 52–67. [Google Scholar]
- Nkwoji, J.A.; Onyema, I.C.; Igbo, J.K. Wet season spatial occurrence of phytoplankton and zooplankton in Lagos Lagoon, Nigeria. Sci. World J. 2010, 5, 7–14. [Google Scholar] [CrossRef]
- Paturej, E.; Gutkowska, A. The effect of salinity levels on the structure of zooplankton communities. Arch. Biol. Sci. 2015, 67, 483–492. [Google Scholar] [CrossRef]
- Beaugrand, G. The North Sea regime shift: Evidence, causes, mechanisms and consequences. Prog. Oceanogr. 2004, 60, 245–262. [Google Scholar] [CrossRef]
- Bonnet, D.; Frid, C. Seven copepod species considered as indicators of water-mass influence and changes: Results from a Northumberland coastal station. ICES J. Mar. Sci. 2004, 61, 485–491. [Google Scholar] [CrossRef]
- Giamali, C.; Kontakiotis, G.; Antonarakou, A.; Koskeridou, E. Ecological Constraints of Plankton Bio-Indicators for Water Column Stratification and Productivity: A Case Study of the Holocene North Aegean Sedimentary Record. J. Mar. Sci. Eng. 2021, 9, 1249. [Google Scholar] [CrossRef]
- Bērziņš, B.; Pejler, B. Rotifer occurrence in relation to pH. Hydrobiologia 1987, 147, 107–116. [Google Scholar] [CrossRef]
- Kuczynski, D. The rotifer fauna of Argentine Patagonia as a potential limnological indicator. Hydrobiologia 1987, 150, 3–10. [Google Scholar] [CrossRef]
- Branco, C.W.; de Assis Esteves, F.; Kozlowsky-Suzuki, B. The zooplankton and other limnological features of a humic coastal lagoon (Lagoa Comprida, Mace, RJ) in Brazil. Hydrobiologia 2000, 437, 71–81. [Google Scholar] [CrossRef]
- Branco, C.W.; Kozlowsky-Suzuki, B.; Esteves, F.A. Environmental changes and zooplankton temporal and spatial variation in a disturbed Brazilian coastal lagoon. Braz. J. Biol. 2007, 67, 251–262. [Google Scholar] [CrossRef]
- Adandedjan, D.; Makponse, E.; Hinvi, L.C.; Laleye, P. Données préliminaires sur la diversité du zooplancton du lac Nokoué (Sud-Bénin). J. Appl. Biosci. 2017, 115, 11476–11489. [Google Scholar] [CrossRef]
- Le Barbé, L.; Alé, G.; Millet, B.; Texier, H.; Borel, Y.; Gualde, R. Les Ressources en Eaux Superficielles de la République du Bénin; Monographies hydrologiques; ORSTOM: Paris, France, 1993; Volume 11, 540p. [Google Scholar]
- Mama, D.; Chouti, W.; Alassane, A.; Changotade, O.; Alapini, F.; Boukari, M. Etude dynamique des apports en éléments majeurs et nutritifs des eaux de la lagune de Porto-Novo (Sud Bénin). Int. J. Biol. Chem. Sci. 2011, 5, 1278–1293. [Google Scholar] [CrossRef]
- Chaigneau, A.; Okpeitcha, V.O.; Morel, Y.; Stieglitz, T.; Assogba, A.; Benoist, M.; Allamel, P.; Honfo, J.; Awoulbang Sakpak, T.D.; Rétif, F.; et al. From seasonal flood pulse to seiche: Multi-frequency water-level fluctuations in a large shallow tropical lagoon (Nokoué Lagoon, Benin). Estuar. Coast. Shelf Sci. 2022, 267, 107767. [Google Scholar] [CrossRef]
- Texier, H.; Colleuil, B.; Profizi, J.-P.; Dossou, C. Lake Nokoue, lagoonal environment of South-Benin costal margin: Bathymetry, sedimentary facies, salinites, molluscus and vegetation. Bull. Inst. Géol. Bassin D’aquitaine 1980, 28, 115–142. [Google Scholar]
- Texier, H.; Dossou, C.; Colleuil, B. Étude de l’environnement lagunaire du domaine margino-littoral sud-béninois. Étude hydrologique préliminaire du lac Nokoué. Bull. L’institut Géologie Du Bassin D’aquitaine 1979, 25, 149–166. [Google Scholar]
- Wetzel, R.A.; Likens, G.E. Limnological Analyses; Springer: New York, NY, USA, 1991; Volume 391, pp. 15–166. [Google Scholar]
- Strickland, J.D.H.; Parsons, T.R. A Practical Handbook of Seawater Analysis; Fisheries Research Board of Canada: Ottawa, ON, Canada, 1972. [Google Scholar]
- Ahlström, E.H. A revision of the Rotatorian genera Brachionus an Platyias with descriptions new species and two new varieties. Bull. Am. Mus. Nat. Hist. 1940, 77, 148–184. [Google Scholar]
- Durand, J.R.; Leveque, C. Flore et Faune Aquatiques de l’Afrique Sahélo-Soudanienne: Tome 1; ORSTOM: Paris, France, 1980. [Google Scholar]
- Dussart, B.H. Les Copépodes. In Flore et Faune Aquatiques de l’Afrique Sahélo Soudanienne: Tome 1; ORSTOM: Paris, France, 1980; pp. 333–356. [Google Scholar]
- Conway, D.V.P. Marine zooplankton of southern Britain. Part 1: Radiolaria, Heliozoa, Foraminifera, Ciliophora, Cnidaria, Ctenophora, Platyhelminthes, Nemertea, Rotifera and Mollusca. Mar. Biol. Assoc. U. K. 2012, 25, 138. [Google Scholar]
- Conway, D.V.P. Marine zooplankton of southern Britain. Part 2: Arachnida, Pycnogonida, Cladocera, Facetotecta, Cirripedia and Copepoda. Mar. Biol. Assoc. U. K. 2012, 26, 163. [Google Scholar]
- Conway, D.V.P. Marine zooplankton of southern Britain. Part 3: Ostracoda, Stomatopoda, Nebaliacea, Mysida, Amphipoda, Isopoda, Cumacea, Euphausiacea, Decapoda, Annelida, Tardigrada, Nematoda, Phoronida, Bryozoa, Entoprocta, Brachiopoda, Echinodermata, Chaetognatha, Hemichordata and Chordata. Mar. Biol. Assoc. U. K. 2015, 27, 271. [Google Scholar]
- Conway, D.V.P.; White, R.G.; Hugues-Dit-Ciles, J.; Gallienne, C.P.; Robins, D.B. Guide to the coastal and surface zooplankton of the south-western Indian Ocean. Mar. Biol. Assoc. U. K. 2003, 15, 354. [Google Scholar]
- Carling, K.; Ater, I.; Pellam, M.; Bouchard, A.; Mihuc, T. A Guide to the Zooplankton of Lake Champlain; Plattsburgh State University of New York: Plattsburgh, NY, USA, 2004; Volume 1, pp. 38–66. [Google Scholar]
- Fontaneto, D.; De Smet, W.H.; Melone, G. Identification key to the genera of marine rotifers worldwide. Meiofauna Mar. 2008, 16, 75–99. [Google Scholar]
- Yamani, F.Y.; Skryabin, V.; Gubanova, A.; Khvorov, S.; Prusova, I. Marine Zooplankton: Practical Guide for the Northwestern Arabian Gulf; Kuwait Institute for Scientific Research: Kuwait City, Kuwait, 2011; p. 12. [Google Scholar]
- Haney, J.F.; Aliberti, M.A.; Allan, E.; Allard, S.; Bauers, D.J.; Beagen, W.S.; Bradtr, R.; Carlson, B.; Carlson, S.C.; Doan, U.M.; et al. An-Image-Based Key to the Zooplankton of North America; Version 5.0; University of New Hampshire Center for Freshwater Biology: Durham, NH, USA, 2013. [Google Scholar]
- LaMay, M.; Hayes-Pontius, E.; Ater, I.M.; Mihuc, T.B. A revised key to the zooplankton of Lake Champlain. Sci. Discipulorum 2013, 6, 141. [Google Scholar]
- Ezz, S.M.A.; Aziz, A.N.E.; Abou Zaid, M.M.; El Raey, M.; Abo-Taleb, H.A. Environmental assessment of El-Mex Bay, Southeastern Mediterranean by using Rotifera as a plankton bio-indicator. Egypt. J. Aquat. Res. 2014, 40, 43–57. [Google Scholar]
- Swadling, K.; Slotwinski, A.; Davies, C.; Beard, J.; McKinnon, A.D.; Coman, F.; Murphy, N.; Tonks, M.; Rochester, W.; Conway, D.V.P.; et al. Australian Marine Zooplankton: A Taxonomic Guide and Atlas. In Image Key-Zooplankton; The University of Tasmania: Hobart, Australia, 2013; Available online: https://www.imas.utas.edu.au/zooplankton/image-key (accessed on 1 February 2023).
- Glime, J.M. Arthropods: Crustacea—Copepoda and Cladocera. Chapter 10–1. Bryophyt. Ecol. 2017, 2, 10–120. [Google Scholar]
- Glime, J.M. Invertebrates: Rotifer Taxa—Monogononta Chapter 4–7 a-b-c. Bryophyt. Ecol. 2017, 2, 7–37. [Google Scholar]
- Santhanam, P.; Perumal, P.; Begum, A. A Method of Collection, Preservation and Identification of Marine Zooplankton. In Basic and Applied Zooplankton Biology; Springer: Berlin/Heidelberg, Germany, 2019; pp. 1–44. [Google Scholar]
- Wilke, T.; Ahlrichs, W.H.; Bininda-Emonds, O.R.P. A weighted taxonomic matrix key for species of the rotifer genus Synchaeta (Rotifera, Monogononta, Synchaetidae). ZooKeys 2019, 871, 40. [Google Scholar] [CrossRef] [PubMed]
- Available online: http://rotifera.hausdernatur.at/ (accessed on 1 February 2023).
- Available online: https://www.shetlandlochs.com/species/eukaryota/animalia/rotifera/eurotatoria/ploima/asplanchnidae/asplanchna (accessed on 1 February 2023).
- Shannon, C.E.; Weaver, W.W. The Mathematical Theory of Communications; University of Illinois Press: Urbana, IL, USA, 1963; 117p. [Google Scholar]
- Peet, R.K. The measurement of species diversity. Annu. Rev. Ecol. Syst. 1974, 5, 285–307. [Google Scholar] [CrossRef]
- Pielou, E.C. The measurement of diversity in different types of biological collections. J. Theor. Biol. 1966, 13, 131–144. [Google Scholar] [CrossRef]
- Magurran, A.E. Ecological Diversity and Its Measurement; Princeton University Press: Princeton, NJ, USA, 1988. [Google Scholar]
- Grall, J.; Coïc, N. Synthèse des méthodes d’évaluation de la qualité du benthos en milieu côtier. Rapp. IFREMER 2005, 91, 2711826. [Google Scholar]
- Bretherton, F.; Davis, R.; Fandry, C. A technique for objective analysis and design of oceanographic experiments applied to MODE-73. Deep-Sea Res. Oceanogr. Abstr. 1976, 23, 559–582. [Google Scholar] [CrossRef]
- McIntosh, P. Oceanographic data interpolation: Objective analysis and splines. J. Geophys. Res. 1990, 95, 13529–13541. [Google Scholar] [CrossRef]
- Wong, A.P.S.; Johnson, G.C.; Owens, W.B. Delayed-mode calibration of autonomous CTD profiling float salinity data by y–S climatology. J. Atmos. Ocean. Technol. 2003, 20, 308–318. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology; Elsevier: Amsterdam, The Netherland, 1998; 853p. [Google Scholar]
- Legendre, P.; Gallagher, E.D. Ecologically meaningful transformations for ordination of species data. Oecologia 2001, 129, 271–280. [Google Scholar] [CrossRef]
- Ter Braak, C.J.; Prentice, I.C. A theory of gradient analysis. Adv. Ecol. Res. 1988, 18, 271–317. [Google Scholar]
- Hastie, T.J. Generalized Additive Models. In Statistical Models in S.; Routledge: New-York, USA, 2017; pp. 249–307. [Google Scholar]
- Ter Braak, C.J.; Smilauer, P. CANOCO Reference Manual and Canodraw for Windows User’s Guide: Softare for Canonical Community Ordination (Version 4.5); Microcomputer Power: Ithaca, NY, USA, 2002. [Google Scholar]
- Wood, S. Generalized Additive Models: An Introduction with R.; Chapman & Hall/CRC: Boca Raton, FL, USA, 2006; 422p. [Google Scholar]
- Wood, S. mgcv: Mixed GAM Computation Vehicle with GCV/AIC/REML Smoothness Estimation; University of Bath: Bath, UK, 2012. [Google Scholar]
- Badsi, H.; Ali, H.O.; Loudiki, M.; Hafa, M.E.; Chakli, R.; Aamiri, A. Ecological factors affecting the distribution of zooplankton community in the Massa Lagoon (Southern Morocco). Afr. J. Environ. Sci. Technol. 2010, 4, 751–762. [Google Scholar]
- Okogwu, I.O. Seasonal variations of species composition and abundance of zooplankton in Ehoma Lake, a floodplain lake in Nigeria. Rev. Biol. Trop. 2010, 58, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Appiah, Y.S.; Étilé, R.N.; Kouamé, K.A.; Paul, E. Zooplankton diversity and its relationships with environmental variables in a West African tropical coastal lagoon (Ebrié lagoon, Côte d’Ivoire, West Africa). J. Biodivers. Environ. Sci. 2018, 13, 1–16. [Google Scholar]
- Étilé, R.N.; Aka, M.N.; Blahoua, G.K.; Kouamélan, P.E. Zooplankton diversity and distribution in a Fresco Lagoon (West Africa, Côte d’Ivoire). Int. Res. J. Environ. Sci. 2018, 7, 9–20. [Google Scholar]
- Monney, I.A.; Etile, R.N.; Ouattara, I.N.; Kone, T. Seasonal distribution of zooplankton in the Aby-Tendo-Ehy lagoons system (Côte d’Ivoire, West Africa). Int. J. Biol. Chem. Sci. 2016, 9, 23–62. [Google Scholar] [CrossRef]
- Morel, Y.; Chaigneau, A.; Okpeitcha, V.O.; Stieglitz, T.; Assogba, A.; Duhaut, T.; Rétif, F.; Peugeot, C.; Sohou, Z. Terrestrial or oceanic forcing? Water level variations in coastal lagoons constrained by river inflow and ocean tides. Adv. Water Resour. 2022, 169, 104309. [Google Scholar] [CrossRef]
- Houssou, A.; Montchowui, E.; Bonou, C. Composition and structure of zooplankton community in ouémé river basin, republic of Benin. J. Entomol. Zool. Stud. 2017, 10, 336–344. [Google Scholar]
- Adjahouinou, D.C.; Liady, N.D.; Fiogbe, E.D. Diversité phytoplanctonique et niveau de pollution des eaux du collecteur de Dantokpa (Cotonou-Bénin). Int. J. Biol. Chem. Sci. 2012, 6, 1938–1949. [Google Scholar] [CrossRef]
- Sukumaran, P.K.; Das, A.K. Distribution and abundance of rrotifera in relation to the water quality of selected tropical reservoirs. Indian J. Fish. 2004, 51, 295–301. [Google Scholar]
- Özbay, H.; Altindağ, A. Zooplankton abundance in the River Kars, Northeast Turkey: Impact of environmental variables. Afr. J. Biotechnol. 2009, 8, 5814–5818. [Google Scholar]
- Onana, F.M.; Zébazé Togouet, S.H.; Nyamsi, T.; Domche, T.; Ngassam, P. Distribution spatio-temporelle du zooplancton en relation avec les facteurs abiotiques dans un hydrosystème urbain: Le ruisseau de Kondi, Cameroun. J. Appl. Biosci. 2014, 82, 7326–7338. [Google Scholar] [CrossRef]
- Zannatul, F.; Muktadir, A.K.M. A Review: Potentiality of Zooplankton as Bioindicator. Am. J. Appl. Sci. 2009, 10, 1815–1819. [Google Scholar]
- Koudenoukpo, C.Z.; Chikou, A.; Zebaze, S.H.T.; Mvondo, N.; Hazoume, R.U.; Houndonougbo, P.K.; Laleye, P.A. Zooplanctons et Macroinvertébrés aquatiques: Vers un assemblage de bioindicateurs pour un meilleur monitoring des écosystèmes aquatiques en région tropicale. Int. J. Innov. Appl. Stud. 2017, 20, 276. [Google Scholar]
- Segers, H. Global diversity of rotifère (Phylum Rotifera) in freshwater. Hydrobiologia 2008, 595, 49–59. [Google Scholar] [CrossRef]
- Sarma, S.S.S.; Nandini, S.; Morales-Ventura, J.; Delgado-Martínez, I.; González-Valverde, L. Effects of NaCl salinity on the population dynamics of freshwater zooplankton (rotifers and cladocerans). Aquat. Ecol. 2006, 40, 349–360. [Google Scholar] [CrossRef]
- Epp, R.W.; Winston, P.W. Osmotic regulation in the brackish-water rotifer Brachionus plicatilis (MULLER). J. Exp. Biol. 1977, 68, 151–156. [Google Scholar] [CrossRef]
- Lowe, C.D.; Kemp, S.J.; Díaz-Avalos, C.; Montagnes, D.J.S. How does salinity tolerance influence the distributions of Brachionus plicatilis sibling species? Mar. Biol. 2007, 150, 377–386. [Google Scholar] [CrossRef]
- Walker, K.F. A synopsis of ecological information on the saline lake rotifer Brachionus plicatilis Muller 1786. Hydrobiologia 1981, 81, 159–167. [Google Scholar] [CrossRef]
- Miracle, R.M.; Serra, M.; Oltra, R.; Vicente, E. Differencial distribution of Brachionus species in the coastal lagoons. Verh. Internat. Verein. Limnol. 1988, 25, 2006–2015. [Google Scholar]
- Arcifa, M.S.; Castilho, M.S.M.; Carmouze, J.-P. Composition et évolution du zooplancton dans une lagune tropicale (Brésil) au cours d’une période marquée par une mortalité de poissons. Hydrobiol. Trop. 1994, 27, 251–263. [Google Scholar]
- Remane, A. Die Brackwasserfauna. Verhandlungen der Deutschen Zoologischen Gesellschaft. Greifswald 1934, 36, 34–74. [Google Scholar]
- Khlebovich, V.V.; Abramova, E.N. Some problems of crustacean taxonomy related to the phenomenon of horohalinicum. Hydrobiologia 2000, 417, 109–113. [Google Scholar] [CrossRef]
- Telesh, I.V.; Schubert, H.; Skarlato, S.O. Life in the salinity gradient: Discovering mechanisms behind a new biodiversity pattern. Estuar. Coast. Shelf Sci. 2013, 135, 317–327. [Google Scholar] [CrossRef]
- Boesch, D.F.; Diaz, R.J.; Virnstein, R.W. Effets de la tempête tropicale Agnes sur les communautés macrobenthiques à fond meuble des estuaires James et York et de la partie inférieure de la baie de Chesapeake. Chesap. Sci. 1976, 17, 246–259. [Google Scholar] [CrossRef]
- Attrill, M.J. Un modèle linéaire testable pour les tendances de la diversité dans les estuaires. J. D’écologie Anim. 2002, 71, 262–269. [Google Scholar] [CrossRef]
- Telesh, I.V.; Schubert, H.; Skarlato, S.O. Revisiting Remane’s concept: Evidence for high plankton diversity and a protistan species maximum in the horohalinicum of the Baltic Sea. Mar. Ecol. Prog. Ser. 2011, 421, 1–11. [Google Scholar] [CrossRef]
- Whitfield, A.K.; Elliott, M.; Basset, A.; Blaber, S.J.M.; West, R.J. Paradigms in estuarine ecology—A review of the Remane diagram with a suggested revised model for estuaries. Estuar. Coast. Shelf Sci. 2012, 97, 78–90. [Google Scholar] [CrossRef]
- Laprise, R.; Dodson, J.J. Environmental variability as a factor controlling spatial patterns in distribution and species diversity of zooplankton in the St. Lawrence Estuary. Mar. Ecol.-Prog. Ser. 1994, 107, 67. [Google Scholar] [CrossRef]
- Crump, B.C.; Armbrust, E.V.; Baross, J.A. Phylogenetic analysis of particle-attached and free-living bacterial communities in the Columbia River, its estuary, and the adjacent coastal ocean. Appl. Environ. Microbiol. 1999, 65, 3192–3204. [Google Scholar] [CrossRef]
- Dolan, J.R.; Gallegos, C.L. Estuarine diversity of tintinnids (planktonic ciliates). J. Plankton Res. 2001, 23, 1009–1027. [Google Scholar] [CrossRef]
- Hewson, I.; Fuhrman, J.A. Richness and diversity of bacterioplankton species along an estuarine gradient in Moreton Bay, Australia. Appl. Environ. Microbiol. 2004, 70, 3425–3433. [Google Scholar] [CrossRef]
- Telesh, I.V. Plankton of the Baltic estuarine ecosystems with emphasis on Neva Estuary: A review of present knowledge and research perspectives. Mar. Pollut. Bull. 2004, 49, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Duggan, S.; McKinnon, A.D.; Carleton, J.H. Zooplankton in an Australian tropical estuary. Estuaries Coasts 2008, 31, 455–467. [Google Scholar] [CrossRef]
- Welcomme, R.L. An evaluation of acadja method of fishing as practiced in the coastal lagoons of Dahomey (West africa). J. Fish Bid. 1972, 4, 39–55. [Google Scholar] [CrossRef]
- Chaffra, A.S.; Agbon, A.C.; Tchibozo, E.A.M. Cartographie par télédétection des Acadjas, une technique de pêche illicite sur le lac Nokoué au Bénin. Sci. Tech. 2020, 5, 11–29. [Google Scholar]
- Welcomme, R.L.; Kapetsky, I.C. Acadjas: The Brush Park Fisheries of Benin, West Africa. ICLARM Newsl. 1981, 4, 3–4. [Google Scholar]
- Guiral, D.; Gourbault, N.; Helleouet, M.-N. Étude sédimentologique et méiobenthos d’un écosystème lagunaire modifié par un récif artificiel à vocation aquacole: L’acadja. Oceanol. Acta 1995, 18, 543–555. [Google Scholar]
- Visser, A.W.; Stips, A. Turbulence and zooplankton production: Insights from PROVESS. J. Sea Res. 2002, 47, 317–329. [Google Scholar] [CrossRef]
- Guiral, D.; Arfi, R.; Da, K.P.; Konan-Brou, A.A. Communautés, biomasses et productions algales au sein d’un récif artificiel (acaja) en milieu lagunaire tropical. Rev. Hydrobiol. Trop. 1993, 26, 219–228. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).