Abstract
This study evaluated the effects of biostimulant treatments on the ionic composition and yield of ‘Formosa’ papaya (Carica papaya L.) subjected to varying water replacement levels. The research utilized a randomized complete block design in a split-plot scheme comprising three irrigation depths (100%, 75%, and 50% ETc) and four biological treatments: control, Trichoderma harzianum, Ascophyllum nodosum, and Bacillus aryabhattai. Contrary to initial expectations, water restriction was associated with increased yield and leaf concentrations of zinc (Zn) and nitrogen (N), challenging the hypothesis that water restriction limits production. Quantitative results showed that T. harzianum under 75% ETc attained 93.29 kg plant−1, whereas the control at 50% ETc recorded 19.14 g kg−1 of N. Under 50% ETc, B. aryabhattai increased the bacterial population to 10.46 log10 CFU g−1 soil compared to the control. The T. harzianum-based biostimulant reduced leaf sodium (Na) under 75% ETc and maintained the nutrient accumulation order K > N > Ca > Fe > Mn > Zn > Cu > Na. Conversely, B. aryabhattai and A. nodosum improved yield under 100% ETc through N accumulation. This study confirms that microbial and seaweed-based biostimulants mitigate water stress through rhizosphere modification and nutritional homeostasis, offering a practical strategy for sustainable fruit production in semi-arid regions.
1. Introduction
Papaya (Carica papaya L.) is a fruit crop of global economic importance, with Brazil as the world’s second-largest producer. The crop plays a strategic role in the socioeconomic development of semi-arid regions in the Brazilian Northeast, where agricultural activities face constraints imposed by prolonged droughts [1,2]. Although production is concentrated in irrigated areas of states such as Bahia and Espírito Santo, recurrent water scarcity has forced the adoption of management strategies based on deficit irrigation levels, challenging orchard sustainability [3,4].
Water deficit impairs plant physiology by inhibiting root growth and limiting mass flow and diffusion—the primary pathways for nutrient uptake. Under water restriction, plants close their stomata to prevent dehydration, which reduces the xylem transport of essential elements such as calcium (Ca) and boron (B) [5]. This blockage triggers the remobilization of mobile nutrients from mature to young leaves, resulting in metabolic expenditure and nutritional imbalances that compromise both flowering and fruiting [6,7]. Investigating the ionic composition under varying water replacement levels is essential because the shift in mineral profiles reflects the plant’s capacity to maintain metabolic stability and osmotic adjustment under reduced water availability [4,8].
As a mitigating strategy, soil-applied biostimulants act through rhizosphere modification and metabolic regulation [8]. The fungus Trichoderma harzianum, in addition to solubilizing soil nutrients via acidification, modulates the hormonal balance of auxins and abscisic acid (ABA) [9]. Furthermore, it induces the expression of resistance genes, such as Heat Shock Proteins (HSP70), priming the cellular machinery to withstand oxidative stress [10]. In a distinct mechanism, the plant growth-promoting rhizobacterium (PGPR) Bacillus aryabhattai promotes tissue longevity. Beyond forming protective biofilms on the roots, it synthesizes the enzyme 1-aminocyclopropane-1-carboxylate (ACC) deaminase, which degrades the ethylene precursor in the plant [11]. This process results in the reduction of this hormone, thereby delaying leaf senescence and chlorophyll degradation, maintaining photosynthetic activity for longer periods under drought conditions [12]. Similarly, the seaweed extract of Ascophyllum nodosum functions as a metabolic supplement. Rich in mannitol, betaines, and polysaccharides, it provides compatible solutes that adjust the intracellular osmotic potential (osmotic adjustment) [13]. This process preserves turgidity and membrane integrity against dehydration while also stimulating cell division through cytokinin-like activity [14].
The efficacy of these biological agents has been reported in annual crops such as maize, common bean, and wheat [13,15]. Despite these advances, a scientific gap exists regarding the interactive effects of biostimulants and deficit irrigation on long-cycle tropical fruit trees. Specifically, the mechanisms by which biological inputs modulate the mineral profile to sustain yield under progressive water stress remain insufficiently understood. This study advances current knowledge by determining how specific microbial strains and seaweed extracts influence nutrient absorption and translocation patterns, establishing a link between biological rhizosphere modification and mineral homeostasis. In this context, the hypothesis is that the soil application of these biostimulants promotes the uptake and translocation of nutrients through rhizosphere modifications and hormonal signaling, thereby maintaining ionic homeostasis. Thus, the objective of this study was to evaluate the effects of T. harzianum, B. aryabhattai, and A. nodosum on the mineral nutrition and yield of ‘Formosa’ papaya under different irrigation depths.
2. Materials and Methods
2.1. Study Site and Climate Conditions
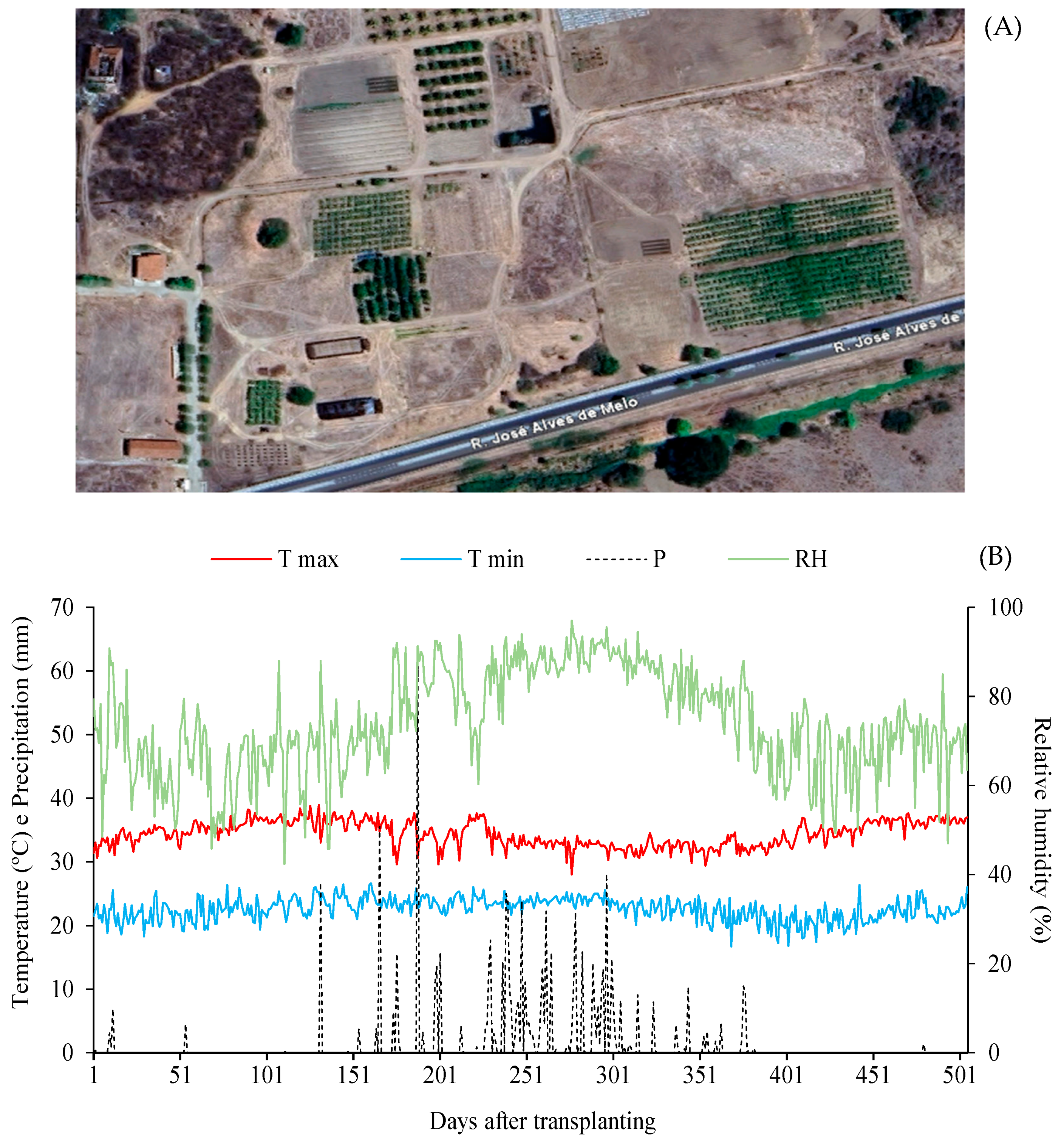
The study was conducted between 28 June 2023, and 12 November 2024, totaling 505 days of experimental monitoring. The geographic location (6°48′51.7″ S, 37°56′13.8″ W; average altitude 190 m) of the experimental site in São Domingos, Paraíba, Brazil, is shown in Figure 1A. Climatic conditions were compared with historical regional data, characterized by a mean annual temperature of 26.7 °C and rainfall of 800 mm year−1 [16]. Daily climatic data, including maximum and minimum temperatures, mean relative humidity, and rainfall, obtained from the São Gonçalo Meteorological Station (Sousa, PB), are shown in Figure 1B.
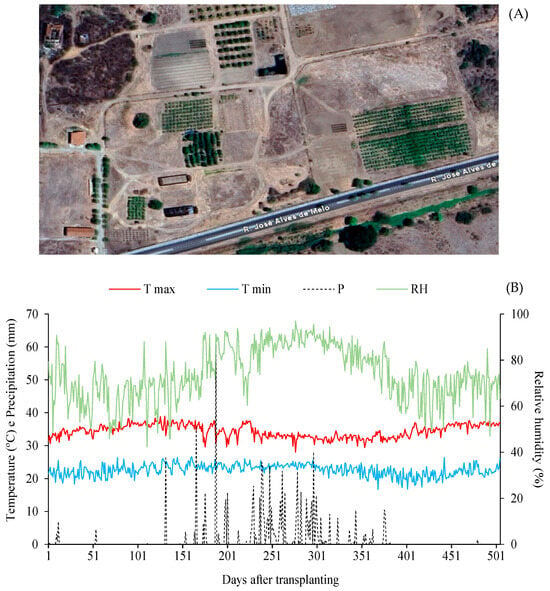
Figure 1.
(A) Satellite map and geographic location of the experimental area at the ‘Rolando Enrique Rivas Castellón’ Experimental Farm, São Domingos, Paraíba, Brazil (6°48′51.7″ S, 37°56′13.8″ W; 190 m altitude); (B) Climatic data of maximum (Tmax) and minimum (Tmin) temperature, precipitation (P), and mean relative humidity (RH) recorded during the experimental period from 29 June 2023, to 12 November 2024.
2.2. Experimental Design and Treatments
The experiment was conducted using a randomized complete block design (RCBD) in a split-plot scheme with four replications. The main plots comprised three irrigation depths (L): 100%, 75%, and 50% of crop evapotranspiration (ETc). The subplots consisted of four biostimulant treatments: (B1) Control (no biostimulant); (B2) Trichoderma harzianum; (B3) Ascophyllum nodosum; and (B4) Bacillus aryabhattai. Each subplot contained three plants. Border plants were positioned at the edges of the experimental area (at a spacing of 2.0 m between plants and 3.5 m between rows) to mitigate external influences.
2.3. Biostimulant Sources and Application
The study evaluated three distinct biostimulant treatments compared to a control group: (i) a fungus-based biostimulant consisting of the Trichoderma harzianum strain; (ii) a bacterial-based biostimulant utilizing the Bacillus aryabhattai strain; and (iii) an algal-based extract derived from the seaweed Ascophyllum nodosum. These agents were applied individually to assess their specific interactions with the irrigation regimes. The biostimulant sources were Trichodermil SC 1306® (T. harzianum Rifai, strain ESALQ-1306, minimum 2.0 × 109 viable conidia mL−1), Aryacompost® (B. aryabhattai, 4 × 108 CFU g−1), and Alga 95® (A. nodosum extract, 95% concentration). The biostimulants were applied to the soil around the root system (15 cm from the stem) following the manufacturers’ recommendations: Alga 95® (0.75 kg ha−1), Aryacompost® (0.5 kg ha−1), and Trichodermil SC 1306® (1.0 L ha−1). Applications began five days after transplanting and were repeated every 50 days, totaling four applications. Irrigation depths were established following the methodology of Melo et al. [17].
2.4. Plant Material, Soil Preparation, and Characterization
Seedlings of the ‘Papaya Sunrise’ (Formosa group) cultivar—characterized by high vigor, yield, and fruits weighing 700–1500 g [18]—were produced in a greenhouse (50% shade) using JKS® trays (162 cells, 50 mL). The substrate consisted of a 2:1 ratio (v/v) of local soil and well-cured cattle manure. Seedlings were irrigated daily with water (ECw = 0.3 dS m−1).
Soil preparation involved harrowing and leveling. Soil samples for physical and chemical characterization were collected at a depth of 0–0.30 m in June 2023, prior to the installation of the experimental setup. Analyses were performed according to the standard protocols described by Teixeira et al. [19]: the pipette method was used for granulometric analysis (sand, silt, and clay); pH was determined in water (1:2.5); phosphorus (P) and potassium (K) were extracted using Mehlich-1 solution; exchangeable calcium (Ca2+), magnesium (Mg2+), and aluminum (Al3+) were extracted with 1 mol L−1 KCl; and organic matter (OM) was determined via wet oxidation with potassium dichromate
The soil physical properties were: sand (73.51 dag kg−1), silt (20.14 dag kg−1), clay (6.35 dag kg−1), exchangeable sodium percentage (ESP) of 0.79%, and moisture at field capacity (33.42 kPa) and permanent wilting point (1519.5 kPa) of 15.78 and 6.41 dag kg−1, respectively. Chemical properties were: pH (H2O 1:2.5): 7.19; Organic Matter: 1.40 g kg−1; P: 5.95 mg kg−1; K+: 0.49 cmolc kg−1; Na+: 0.07 cmolc kg−1; Ca2+: 4.70 cmolc kg−1; Mg2+: 3.63 cmolc kg−1; Al3+: 0; H+: 0; ECse: 0.58 dS m−1; and SARse: 1.40 (mmol L−1)0.5.
2.5. Soil Microbiological and Respirometric Analysis
Soil samples (0–0.30 m) were collected before the experiment and during the fruiting phase, stored at −4 °C, and equilibrated at room temperature for eight hours before analysis. Total densities of bacteria, fungi, and actinomycetes were determined according to Hungria and Araújo [20] using serial dilutions (10−1 to 10−10) in saline solution. Culture media included Potato Dextrose Agar (PDA), Nutrient Agar (NA), and PDA + starch for fungi, bacteria, and actinomycetes, respectively. Bacterial counts were expressed as Log10 Colony Forming Units (CFU) per gram of soil, while fungi and actinomycetes were estimated by the Most Probable Number (MPN) using the McCrady table (Log10 MPN).
Soil microbial respiration (CO2 evolution) was measured following Mendonça and Matos [21] by capturing C- CO2 in a 0.5 M NaOH solution within a hermetically sealed environment. After 48 h of incubation, the solution was titrated with HCl using BaCl2 (0.05 M) to calculate C- CO2 (mg) according to Equation (1):
where: B = HCl volume for blank (mL); V = HCl volume for sample (mL); M = HCl molarity; 6 = constant for carbon mass; V1 = total NaOH volume (mL); and V2 = titrated NaOH volume (mL).
Regarding soil microbial activity, the B1 (Control) treatment recorded 9.12 log10 CFU g−1 soil for bacteria, 5.38 log MPN for fungi, 5.05 log MPN for actinomycetes, and 6.15 mg for C-CO2. Under the 50% ETc irrigation depth, the treatments B1, B2, B3, and B4 presented bacterial counts of 9.52, 10.46, 9.95, and 10.20 log10 CFU g−1 soil; fungal populations of 5.38, 5.38, 5.38, and 5.05 log MPN; actinomycetes levels of 5.38, 5.72, 6.05, and 5.05 log MPN; and C-CO2 values of 9.82, 11.72, 12.75, and 12.45 mg, respectively.
At the 75% ETc depth, the values for B1, B2, B3, and B4 were: 9.07, 10.52, 10.57, and 9.88 for bacteria (log10 CFU g−1 soil); 5.17, 5.17, 5.17, and 5.17 for fungi (log MPN); 5.47, 5.17, 5.17, and 5.17 for actinomycetes (log MPN); and 12.16, 11.87, 13.77, and 36.04 mg for C-CO2. Finally, for the 100% ETc treatments, B1, B2, B3, and B4 recorded bacterial counts of 9.81, 9.62, 9.69, and 9.83 log10 CFU g−1 soil; fungal levels of 5.38, 5.05, 5.05, and 5.05 log MPN; actinomycetes counts of 5.05, 6.05, 5.05, and 5.05 log MPN; and C-CO2 values of 16.12, 10.84, 10.26, and 12.45 mg, respectively.
2.6. Field Management, Fertilization, and Irrigation
Plants were grown in furrows (0.4 × 1.0 × 30 m) spaced 3.5 m apart. Foundation fertilization included 8.72 kg ha−1 of phosphorus (P), applied as single superphosphate. Top-dressing fertilization with nitrogen (N), P, and potassium (K) followed Embrapa recommendations [22]. The nutrient sources utilized were urea, potassium sulfate, and monoammonium phosphate (MAP). In the first year, total applications reached 260 kg ha−1 of N, 17.44 kg ha−1 of P, and 74.7 kg ha−1 of K. During the second year, doses consisted of 240 kg ha−1 of N, 21.8 kg ha−1 of P, and 78.85 kg ha−1 of K. Fertilizers were applied via fertigation every 15 days.
Foliar micronutrient applications were performed every 30 days using the Dripsol micro® compound, with the following composition: Mg = 1.1%; B = 0.85%; Cu (Cu-EDTA) = 0.5%; Fe (Fe-EDTA) = 3.4%; Mn (Mn-EDTA) = 3.2%; Mo = 0.05%; and Zn = 4.2%. The product contained 70% chelating agent (EDTA) and was applied at a concentration of 0.5 g L−1, according to the manufacturer’s recommendations.
Transplanting was performed after the technical preparation of the soil and furrows. The seedlings, produced in trays with 162 cells (50 mL each), were manually transplanted into individual holes created within the furrows using a 32 mm diameter pipe. This procedure ensured a precise fit for the substrate plug, facilitating root establishment and immediate connection with the soil solution.
The irrigation water was sourced from an artesian well located at the Experimental Farm, presenting the following chemical characteristics: pH = 7.22; electrical conductivity (ECw) = 1.032 dS m−1; Ca2+ = 1.24 mmolc L−1; Mg2+ = 4.66 mmolc L−1; Na+ = 7.56 mmolc L−1; K+ = 0.03 mmolc L−1; CO32− = 0 mmolc L−1; HCO3− = 3.55 mmolc L−1; Cl− = 5.70 mmolc L−1; presence of SO42−; and a Sodium Adsorption Ratio (SAR) = 4.40 (mmol L−1)0.5.
The plants were irrigated daily in the morning. Irrigation depths were estimated based on crop evapotranspiration (ETc), with the 100% ETc depth calculated according to Equation (2), as described by Bernardo et al. [23]. The 75% and 50% ETc levels were estimated relative to the 100% ETc value:
where: ETc: crop evapotranspiration (mm day−1); ETo: Penman–Monteith reference evapotranspiration (mm day−1); Kc: crop coefficient (dimensionless).
The reference evapotranspiration (ETo) was determined daily using the Penman–Monteith method, based on climatic data from the São Gonçalo Meteorological Station in Sousa—PB. The crop coefficients (Kc) corresponded to the phenological stages of the crop: Kc values of 0.64, 1.16, and 1.19 were adopted for the vegetative (46–95 DAT), flowering (96–201 DAT), and fruiting (after 202 DAT) stages, respectively, following Montenegro et al. [24].
The control of the water volume for each calculated irrigation depth was performed daily at a fixed time, based on the emitter flow rate and the time required to reach the target ETc proportions. Climatic variables were monitored daily; when precipitation occurred, the rainfall volume was measured and deducted from the irrigation depths for each treatment. Irrigation was suspended when precipitation exceeded the irrigation requirement and was resumed based on soil moisture monitoring via Time Domain Reflectometry – TDR (Milano, Roma).
Soil moisture content was monitored using TDR, which emits high-frequency electromagnetic pulses through a sensor connected to a coaxial cable. The reflections recorded over time are read as distance and related to the dielectric constant [25]. An access tube was installed for each treatment at a depth of 1.0 m and a distance of 0.15 m from the plant, distributed throughout the experiment. The TDR was previously calibrated in the cultivation area. Readings were taken weekly at 08:00 AM, and the data collected at depths with the highest root volume (between 10 and 40 cm) were averaged. These data served to monitor soil moisture relative to the water volume applied in each irrigation treatment corresponding to ETc.
2.7. Plant Sexing and Cultural Practices
To ensure a productive stand of hermaphrodite plants, three seedlings were transplanted at each planting site using the method described in Section 2.6. Sexing was performed at 96 days after transplanting (DAT) upon the identification of the first flower buds. At this stage, thinning was conducted to remove surplus plants, leaving only the most vigorous hermaphrodite individual at each site. Cultural practices included the removal of lateral shoots, weed control, and staking with wooden supports to prevent lodging.
2.8. Foliar Nutrient and Yield Analysis
The evaluation of leaf nutrient concentrations (expressed in g kg−1 for macronutrients and mg kg−1 for micronutrients) was conducted at 205 DAT. For each experimental plot, one leaf (the 4th from the apex of the canopy) was sampled from each of the three plants [26], totaling three leaves per treatment replication to form a composite sample. Regarding the digestion procedures, N was determined via the Kjeldahl method after sulfuric acid digestion, and S was analyzed via nitric–perchloric digestion [27]. All other elements (P, K, Ca, Mg, Na, Fe, Zn, Mn, Cu) were determined after microwave-assisted digestion using concentrated nitric acid (HNO3) [27].
Fruit yield was determined cumulatively by weighing all fruits harvested from 246 to 505 DAT. The results are expressed as fresh fruit mass (kg per plant), representing the marketable yield as fruits met the commercial weight standards for the ‘Formosa’ group. No moisture standardization was applied to the fruits, as yield is conventionally reported as fresh weight for this crop.
2.9. Statistical Analysis
The experimental data, totaling 48 observations (n = 48, derived from 12 treatments with 4 replications each), were subjected to analysis of variance (ANOVA) using the F-test (p ≤ 0.05). Statistical assumptions of normality and homogeneity of variances were previously verified using the Shapiro–Wilk and Bartlett tests, respectively. In cases of significant interaction or main effects, means were compared using Tukey’s test (p ≤ 0.05). The univariate analyses (ANOVA and means comparison) were performed using SISVAR software, version 5.6, developed by Daniel Furtado Ferreira at the Federal University of Lavras (UFLA), Brazil [28]. To explore the relationships between irrigation depths and biostimulants, multivariate analysis through Principal Component Analysis (PCA) and Pearson correlation (p ≤ 0.05) were conducted. These multivariate procedures were executed using R software, version 4.3.2, developed by the R Core Team at the R Foundation for Statistical Computing (Vienna, Austria) [29].
3. Results
3.1. Leaf Macronutrient Contents
The interaction between irrigation depths and biostimulants significantly influenced leaf nitrogen (N) and potassium (K) contents (Table 1). Biostimulants significantly affected calcium (Ca) levels in papaya plants, while no significant effects were observed for phosphorus (P), magnesium (Mg), or sulfur (S).

Table 1.
Summary of the analysis of variance (ANOVA) for leaf contents of nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), and sulfur (S) of ‘Formosa’ papaya grown under different irrigation depths (ID) and biostimulant application (Bio) at 205 days after transplanting (DAT).
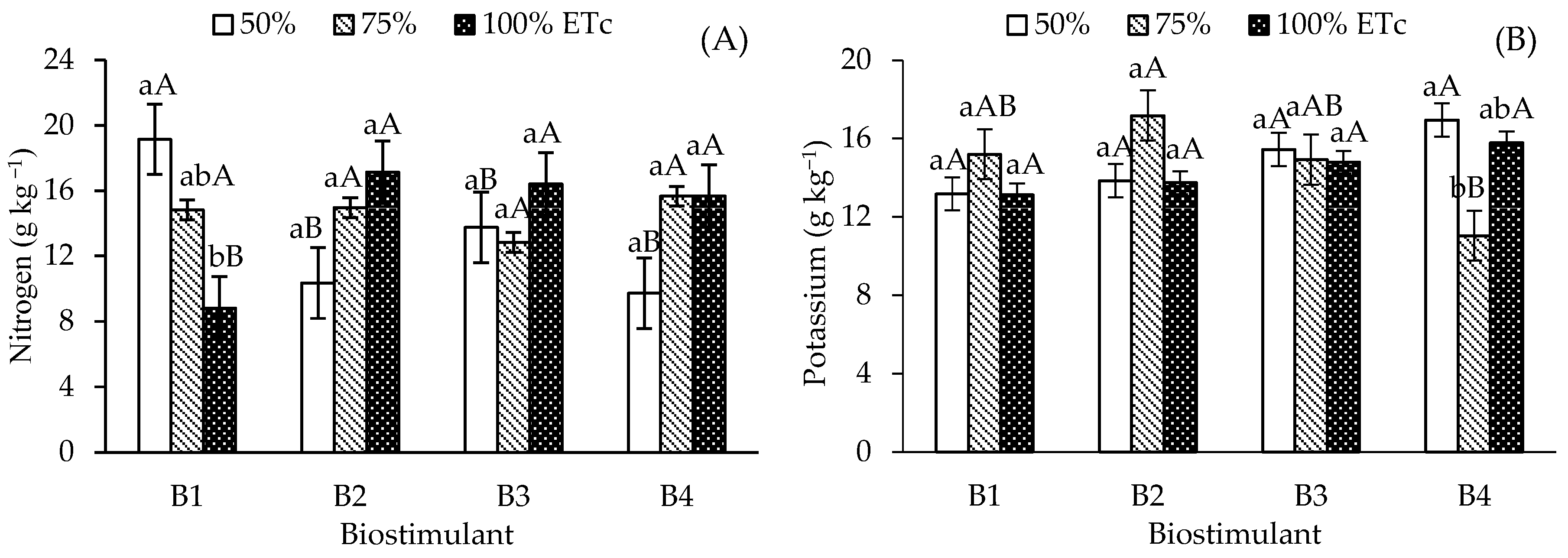
Nitrogen accumulation in papaya leaves was higher in control plants (without biostimulants) under the 50% ETc irrigation depth, differing from those under 100% ETc, but remaining similar to levels observed at 75% ETc (Figure 2A). In contrast, plants treated with B. aryabhattai, T. harzianum, and A. nodosum showed no significant differences in N content across the various irrigation depths. Regarding the effect of biostimulants under 50% ETc irrigation, N content in the control treatment (19.14 g kg−1) was significantly higher than in plants treated with B. aryabhattai, T. harzianum, and A. nodosum. No significant differences were found among biostimulants at the 75% ETc depth. However, under 100% ETc, the application of all three biostimulants resulted in similar N values, which were significantly higher than those of the control.
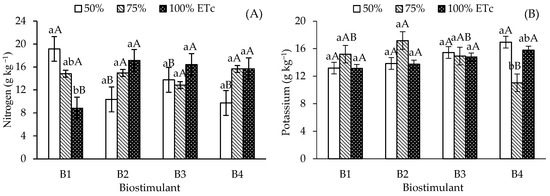
Figure 2.
Leaf contents of nitrogen—N (A) and potassium—K (B) of ‘Formosa’ papaya as a function of the interaction between irrigation depths and biostimulant application at 205 days after transplanting (DAT). B1—control; B2—Bacillus aryabhattai; B3—Trichoderma harzianum; B4—Ascophyllum nodosum. Bars followed by the same lowercase letter indicate no significant difference between irrigation depths within the same biostimulant treatment, while the same uppercase letters indicate no significant difference among biostimulants within the same irrigation depth, according to Tukey’s test (p ≤ 0.05).
Foliar potassium (K) content (Figure 2B) showed no significant differences across irrigation depths for the control, B. aryabhattai, and T. harzianum treatments. The application of A. nodosum increased leaf K levels in plants irrigated at 50% ETc (16.95 g kg-1), compared to 11.04 g kg-1 at 75% ETc. Under full irrigation (100% ETc), no significant differences occurred among biostimulant treatments, with a mean value of 15.78 g kg-1. At the 75% ETc irrigation depth, the use of B. aryabhattai and A. nodosum resulted in significant differences in K content; B. aryabhattai achieved the highest mean (17.17 g kg-1), although it did not differ significantly from the control and T. harzianum under the same conditions.
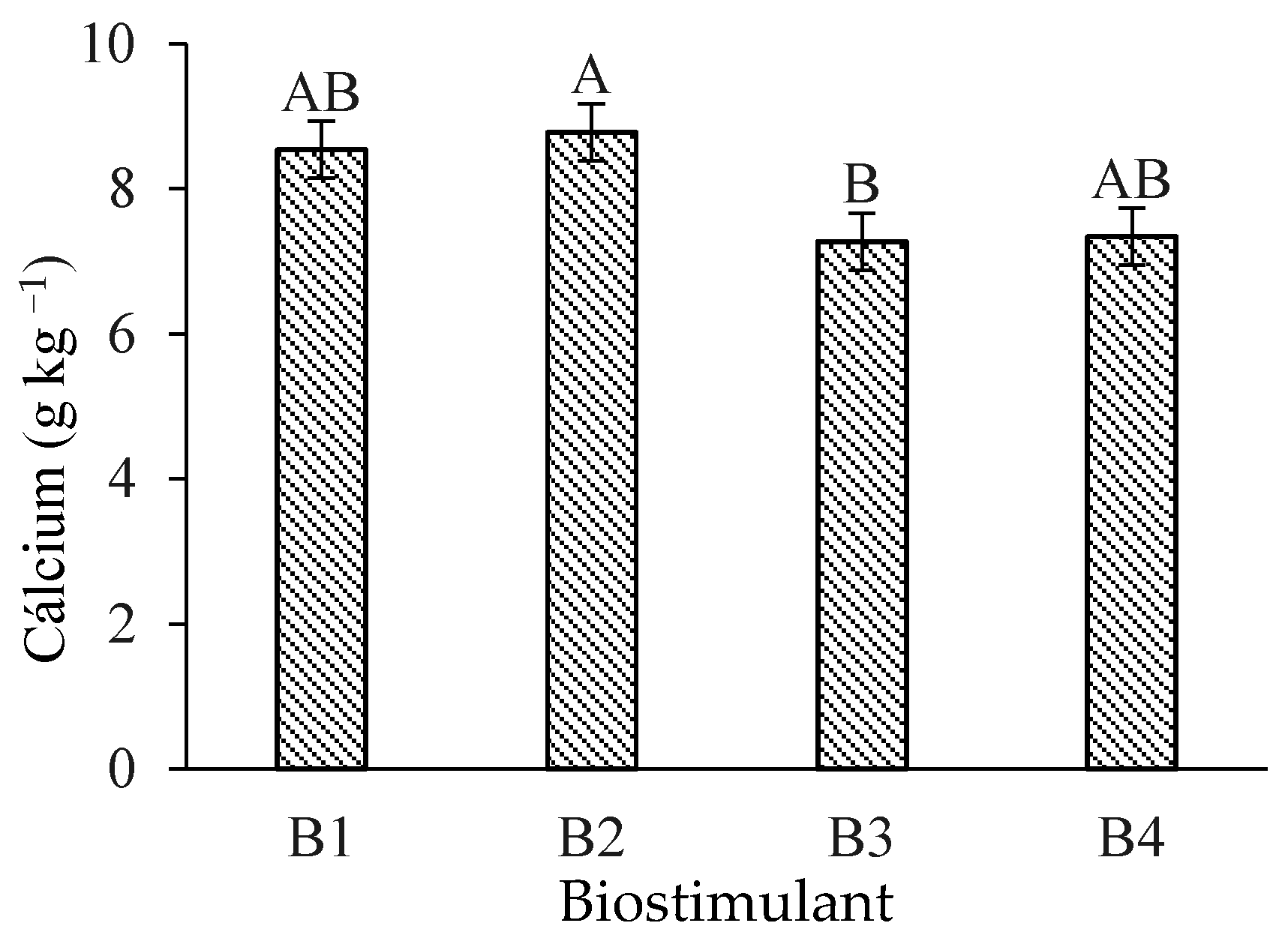
Leaf calcium (Ca) content in ‘Formosa’ papaya was significantly higher under B. aryabhattai application compared to T. harzianum (Figure 3). However, no significant differences in Ca levels occurred when comparing the control treatment with B. aryabhattai and A. nodosum.
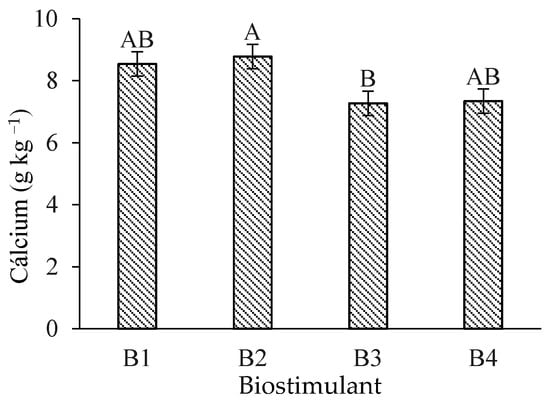
Figure 3.
Leaf calcium (Ca) contents of ‘Formosa’ papaya as a function of biostimulant application at 205 days after transplanting (DAT). B1—control; B2—Bacillus aryabhattai; B3—Trichoderma harzianum; B4—Ascophyllum nodosum. Bars followed by the same uppercase letter indicate no significant difference between biostimulants according to Tukey’s test (p ≤ 0.05).
3.2. Leaf Micronutrient Contents and Fruit Yield
The interaction between irrigation depths and biostimulants had a significant effect on leaf sodium (Na), manganese (Mn), zinc (Zn), iron (Fe), and copper (Cu) contents, as well as on fruit yield (Table 2). No significant effects were observed for chloride (Cl) levels.

Table 2.
Summary of the analysis of variance (ANOVA) for leaf contents of sodium (Na), chloride (Cl), manganese (Mn), zinc (Zn), iron (Fe), and copper (Cu) at 205 days after transplanting (DAT), and yield per plant (PRODF) at 505 DAT of ‘Formosa’ papaya grown under different irrigation depths (ID) and biostimulant application (Bio).
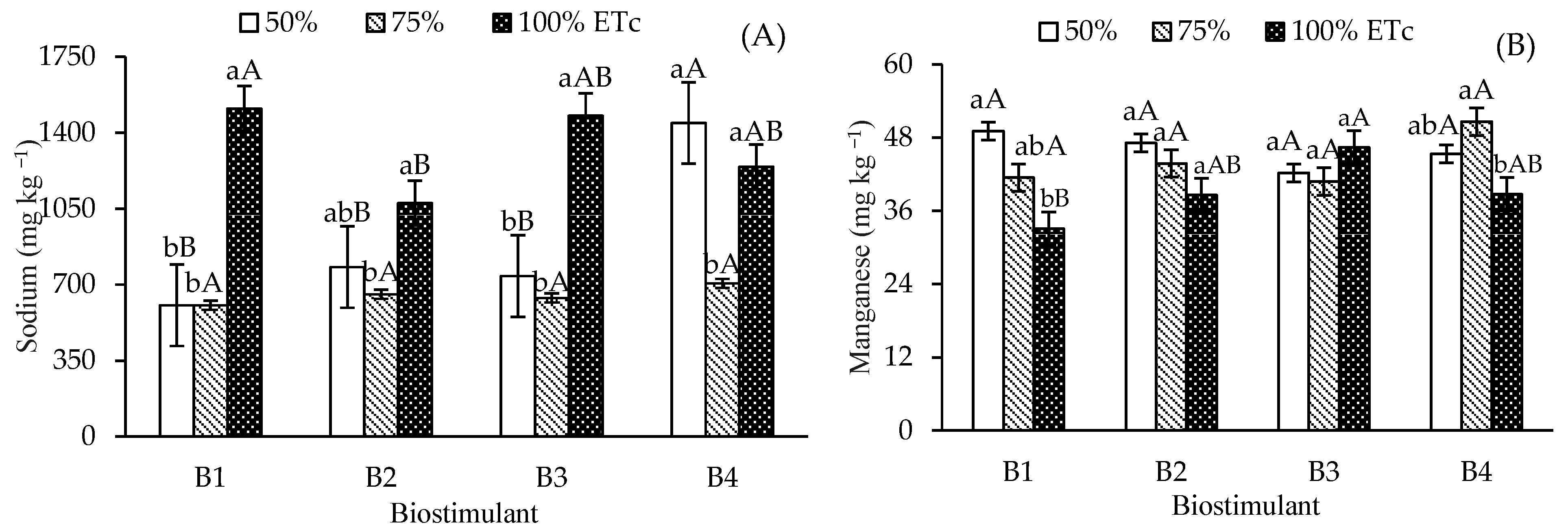
Leaf sodium contents were highest in control plants and those treated with T. harzianum under 100% ETc irrigation, compared to 50% and 75% ETc (Figure 4A). B. aryabhattai also increased Na levels at 100% ETc compared to 75% ETc, though it remained statistically similar to the 50% ETc depth. For A. nodosum, irrigation at 50% and 100% ETc promoted higher Na contents than at 75% ETc. At the 50% ETc depth, Na reduction occurred in the control, B. aryabhattai, and T. harzianum treatments; these values were significantly lower than those observed with A. nodosum.
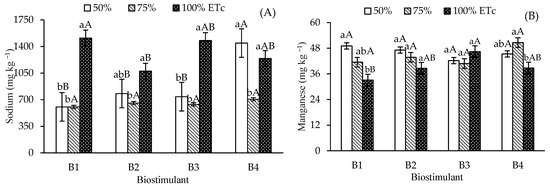
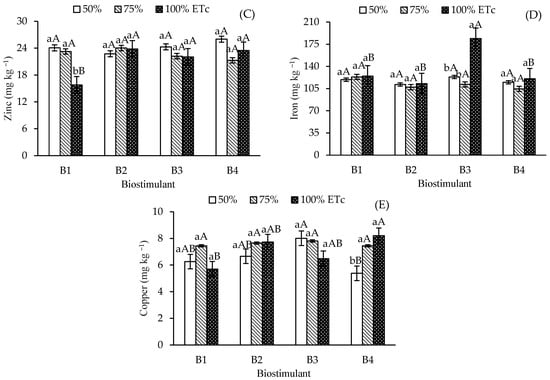
Figure 4.
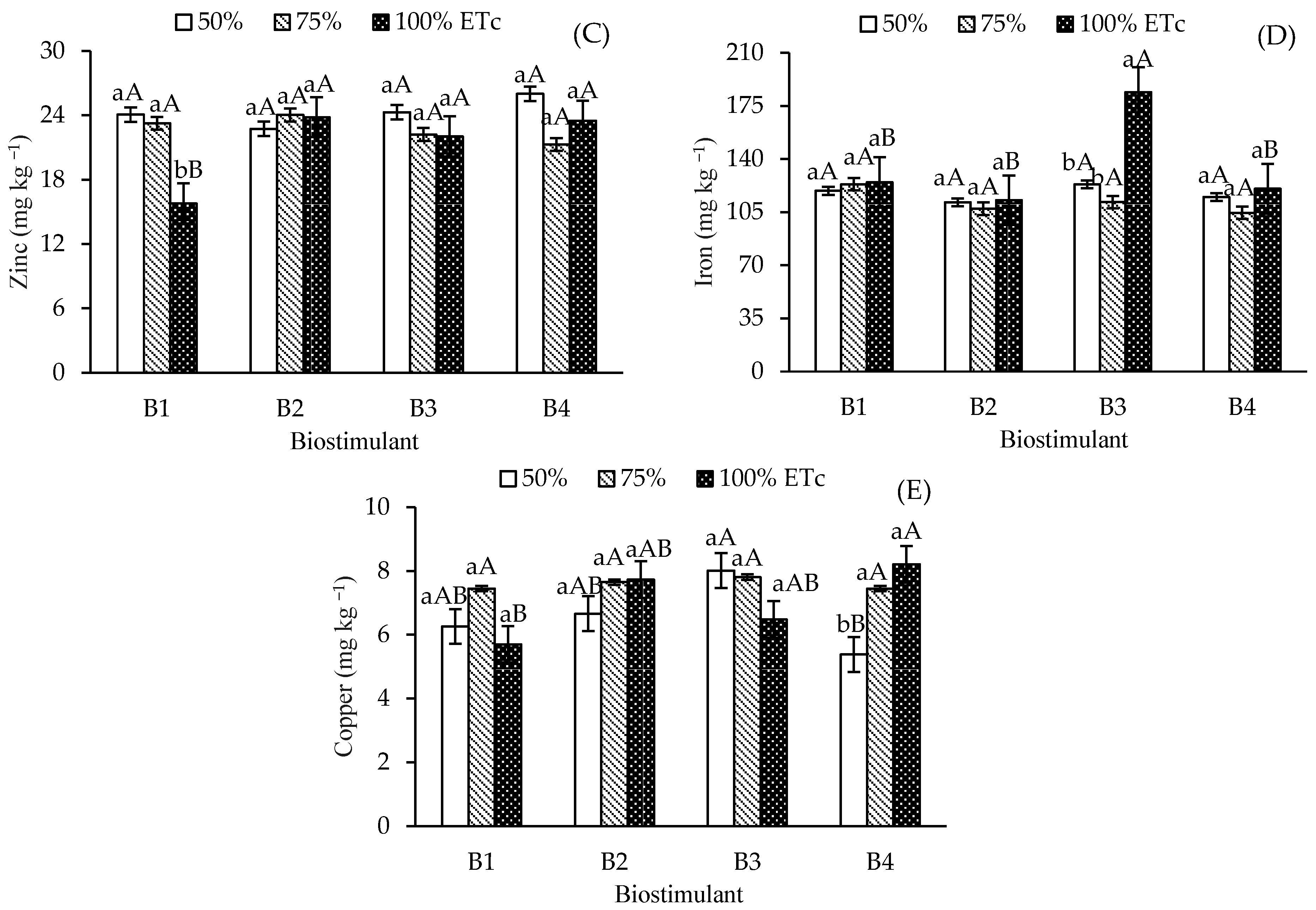
Leaf contents of sodium—Na (A), manganese—Mn (B), zinc—Zn (C), iron—Fe (D), and copper—Cu (E) of ‘Formosa’ papaya as a function of the interaction between irrigation depths and biostimulant application at 205 days after transplanting (DAT). B1—control; B2—Bacillus aryabhattai; B3—Trichoderma harzianum; B4—Ascophyllum nodosum. Bars followed by the same lowercase letter indicate no significant difference between irrigation depths within the same biostimulant treatment, while the same uppercase letters indicate no significant difference among biostimulants within the same irrigation depth, according to Tukey’s test (p ≤ 0.05).
Leaf manganese (Mn) content in control plants at 50% ETc was 49.08 mg kg−1, which differed significantly from the 33.08 mg kg−1 observed at 100% ETc (Figure 4B). No significant Mn variations occurred across irrigation depths for B. aryabhattai and T. harzianum. However, A. nodosum at 75%ETc increased Mn content (50.6 mg kg−1) compared to 100% ETc. Under 100% ETc, T. harzianum showed significant differences from the control, attaining Mn levels similar to B. aryabhattai and A. nodosum.
Foliar zinc (Zn) content was higher in control plants irrigated at 50% and 75% ETc than in those under full irrigation (100% ETc) (Figure 4C). In contrast, irrigation depths did not affect Zn levels in plants treated with biostimulants. Under full irrigation, biostimulant-treated plants showed significantly higher Zn levels than the control. Iron (Fe) levels (Figure 4D) were higher in T. harzianum-treated plants under 100% ETc (184.01 mg kg-1) compared to other irrigation depths and biostimulants. Copper (Cu) content (Figure 4E) increased in A. nodosum-treated plants at 75% and 100% ETc compared to 50% ETc.
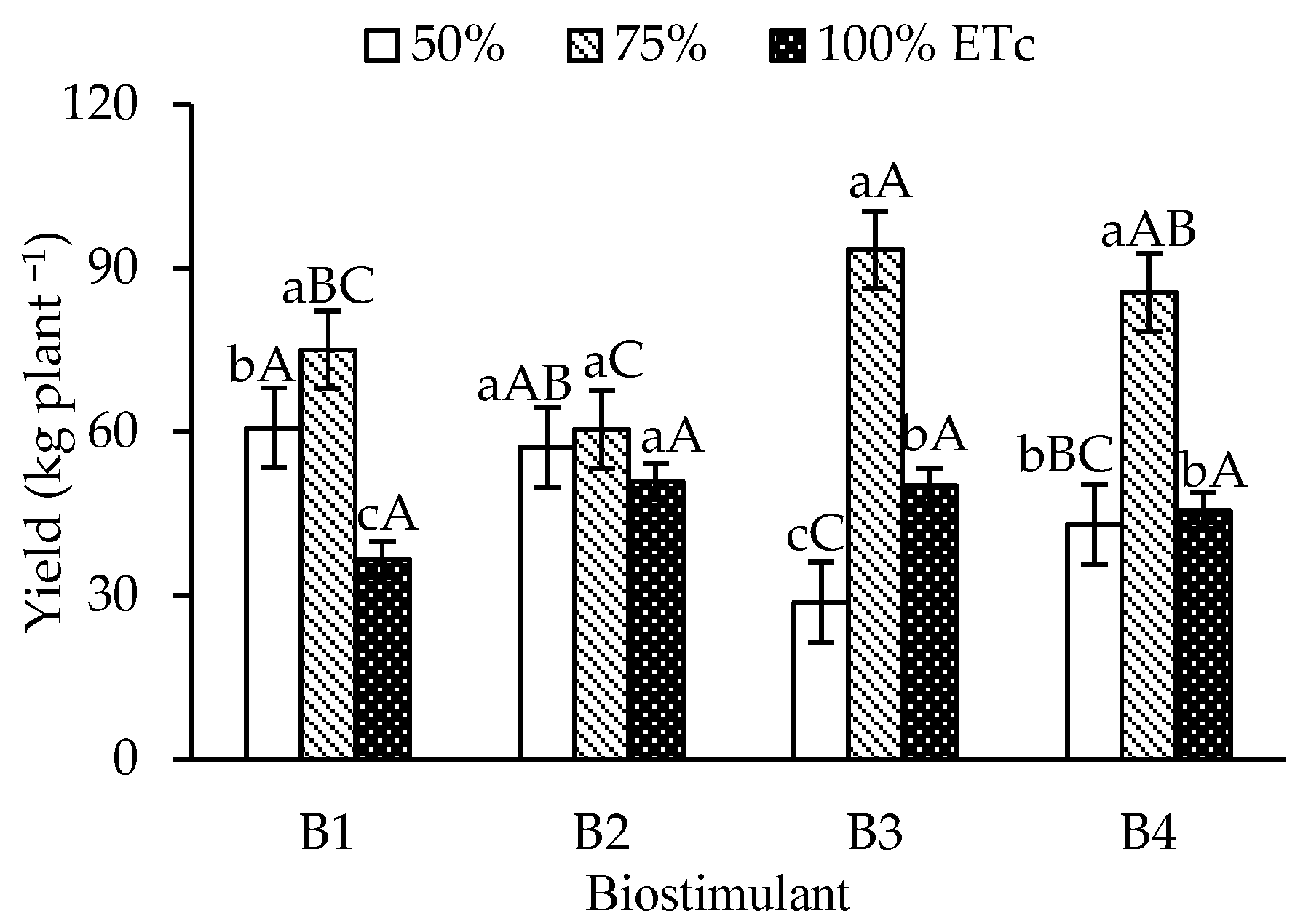
The yield of ‘Formosa’ papaya (Figure 5) increased under 75% ETc irrigation in the control, T. harzianum, and A. nodosum treatments compared to 50% and 100% ETc. For B. aryabhattai, no significant yield differences were observed between irrigation depths. Under 75% ETc, T. harzianum attained the highest yield (93.29 kg plant-1), which was significantly higher than that of the control and B. aryabhattai, but similar to that of A. nodosum (85.51 kg plant-1).
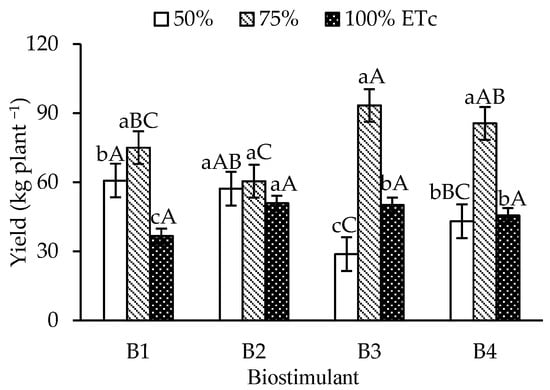
Figure 5.
Yield per plant of ‘Formosa’ papaya as a function of the interaction between irrigation depths and biostimulant application at 505 days after transplanting (DAT). B1—control; B2—Bacillus aryabhattai; B3—Trichoderma harzianum; B4—Ascophyllum nodosum. Bars followed by the same lowercase letter indicate no significant difference between irrigation depths within the same biostimulant treatment, while the same uppercase letters indicate no significant difference among biostimulants within the same irrigation depth, according to Tukey’s test (p ≤ 0.05).
3.3. Multivariate Analysis and Correlations
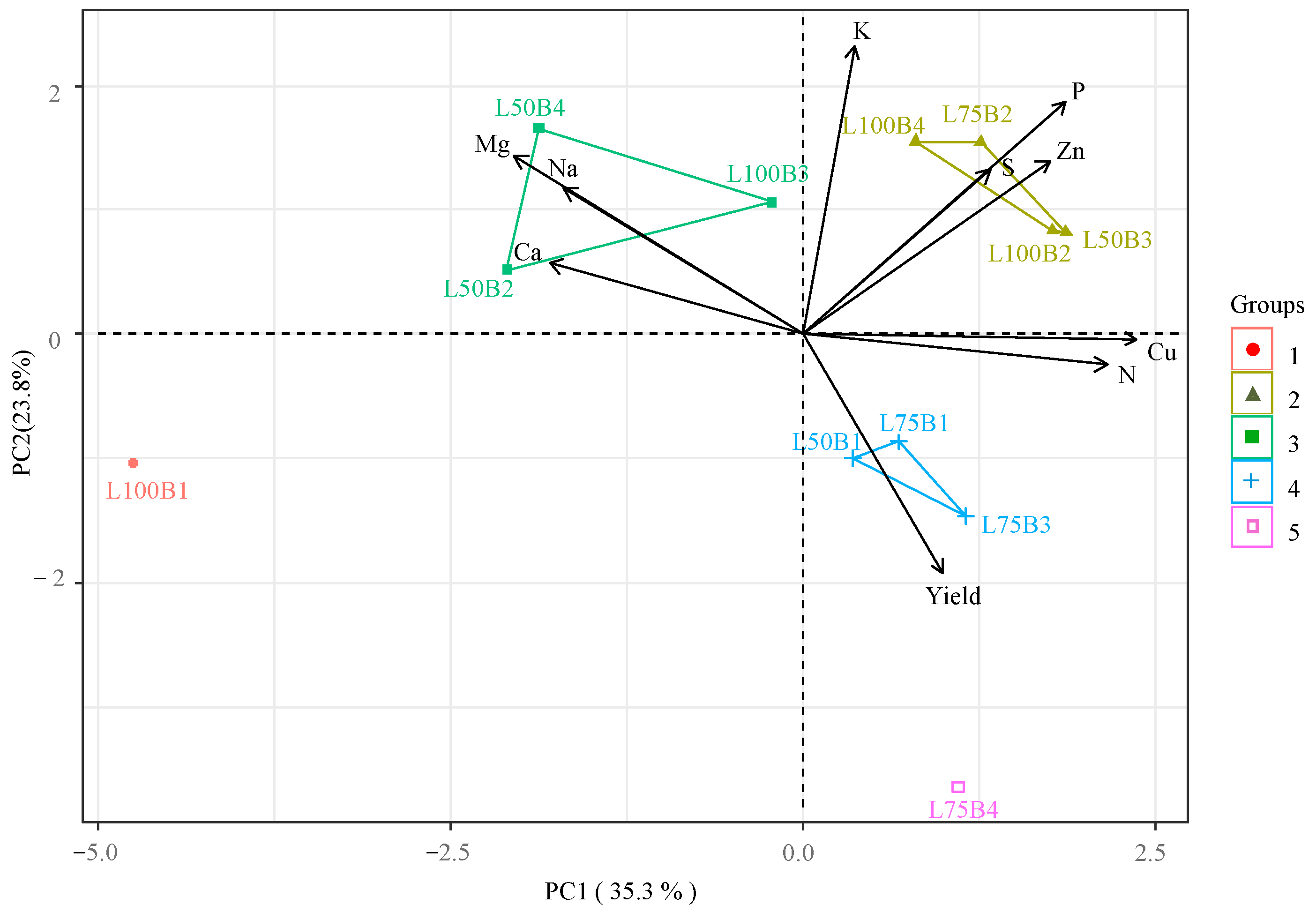
Principal Component Analysis (PCA) explained 59.11% of the total variance for the interaction between irrigation depths and biostimulants, with PC1 and PC2 accounting for 35.33% and 23.78%, respectively (Figure 6). Nitrogen (N), phosphorus (P), zinc (Zn), and copper (Cu) contents were positively correlated with each other, primarily influenced by Group 2 treatments (L75B, L100A, L100T, L100B, and L50T). Group 2 also contributed to higher K levels in PC2. These elements exhibited opposite effects to Ca, Na, and Mg, likely influenced by Group 1 (L100C) and Group 3 (L50A and L50B). Treatment L75A (Group 4) occupied an isolated position, showing the highest contribution to the papaya yield parameter, which was inversely proportional to leaf K contents.
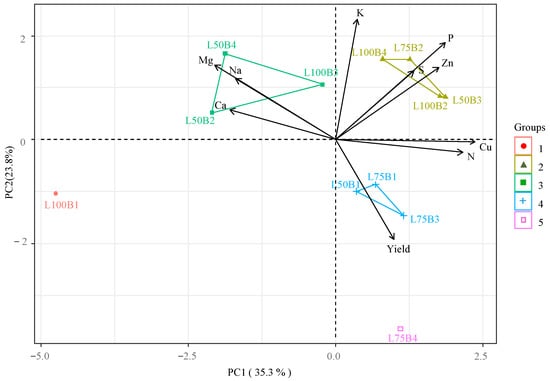
Figure 6.
Two-dimensional projection of principal component scores for the factors irrigation depth and biostimulants, and variables analyzed in the first two principal components (PC1 and PC2), for ‘Formosa’ papaya plants at 205 days after transplanting (DAT). L100% = 100% crop evapotranspiration (ETc) irrigation depth; L75% = 75% ETc irrigation depth; L50% = 50% ETc irrigation depth; B1—control (no biostimulants); B2—Bacillus aryabhattai; B3—Trichoderma harzianum; B4—Ascophyllum nodosum. Nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), sulfur (S), sodium (Na), zinc (Zn), and copper (Cu). ● — group 1 (L100B1); ▲— group 2 (L100B2, L100B4, L75B2 e L50B3); ■— group 3 (L100B3, L50B2 e L50B4); + — group 4 (L75B1, L75B3 e L50B1); and □ — group 5 (L75B4).
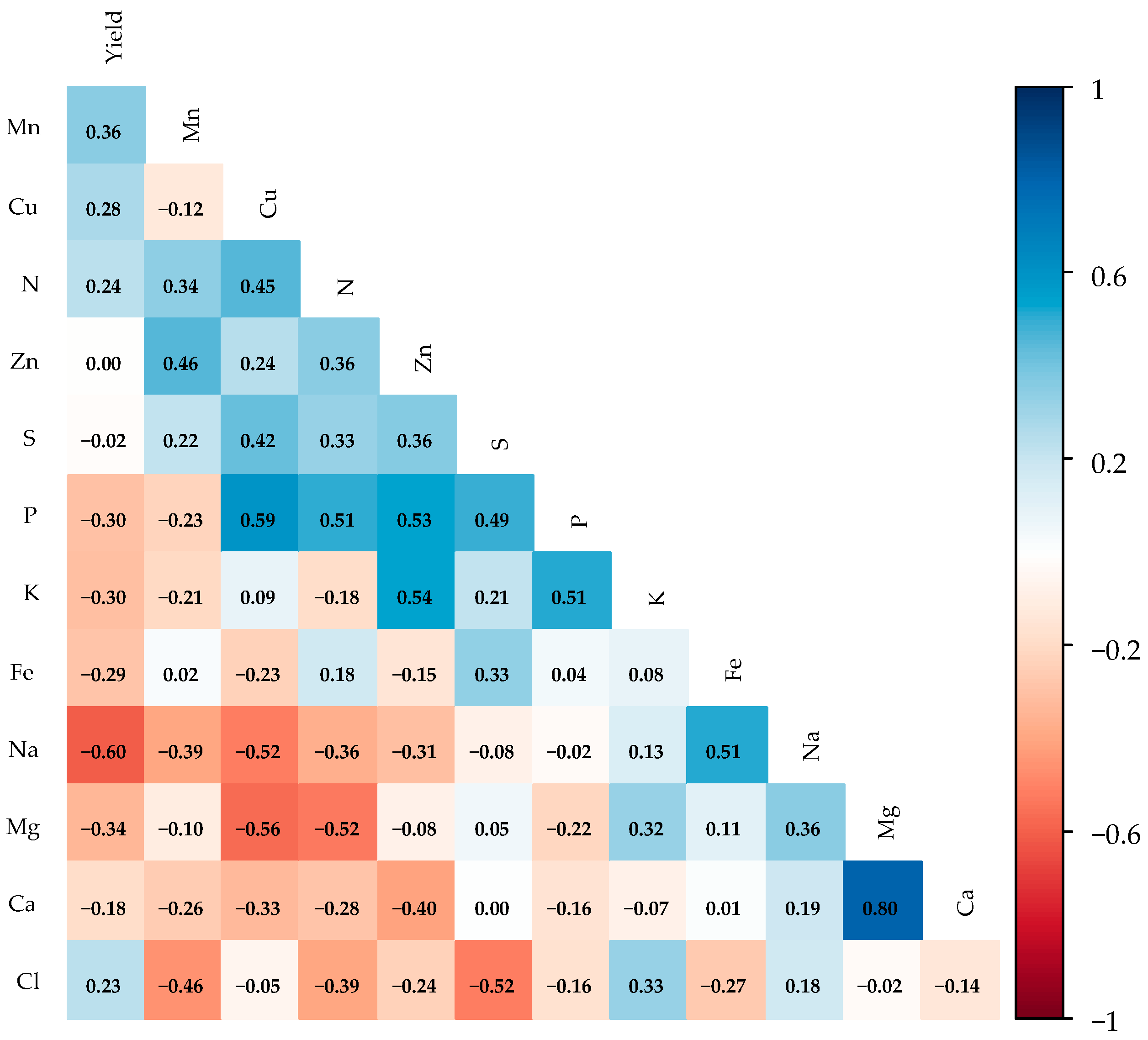
The Pearson correlation matrix (Figure 7) revealed that papaya yield shows weak correlations with the leaf nutritional status of the plants, with a notable negative correlation between yield and Na (r = −0.60). However, a strong positive correlation was found between magnesium and calcium (r = 0.80). Moderate positive correlations were observed between Zn and K (r = 0.54), and between P and K (r = 0.51), Zn (r = 0.53), and N (r = 0.51). Moderate negative correlations were identified between Cl and S (r = −0.52), Mg and N (r = −0.52), Mg and Cu (r = −0.56), and Na and Cu (r = −0.52).
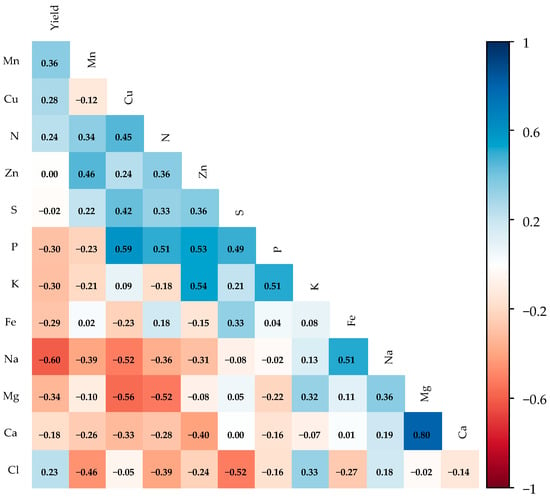
Figure 7.
Pearson correlation matrices for leaf macro- and micronutrient variables during the fruiting stage of ‘Formosa’ papaya under different irrigation depths and biostimulant applications at 205 days after transplanting (DAT) (n = 48). Values above 0.50 are significant (p ≤ 0.05) according to Pearson’s correlation test. Nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), sulfur (S), sodium (Na), chloride (Cl), manganese (Mn), zinc (Zn), iron (Fe), and copper (Cu).
4. Discussion
The results of this study contrast with the nutritional declines typically expected under extreme water deficit, demonstrating that leaf nutrient accumulation was maintained or increased, with yield gains surpassing the control group. This physiological response is directly linked to the specific edaphoclimatic context of the experiment. Although conducted in a semi-arid region, the recorded mild temperatures and a relative humidity of 70.52% likely acted as a buffer, mitigating atmospheric demand and supporting stomatal conductance even under reduced irrigation depths [4,16].
Soil physical characteristics and water quality were also decisive factors. In sandy-textured soil with low Cation Exchange Capacity (CEC), excessive water in the 100% ETc treatment likely accelerated the leaching of mobile bases, including K+ and NO3− [30]. Additionally, the continuous salt input from the irrigation water established a saline environment in the root zone under full irrigation, as evidenced by the increase in leaf Na and its negative correlation with yield [17,31]. Conversely, under water deficit, the alternation between drying cycles and precipitation may have triggered programmed acclimation processes, inducing adaptive responses without reaching metabolic thresholds that compromise flowering [6].
The increase in leaf Nitrogen (N) and Zinc (Zn) under water restriction indicates improved metabolic efficiency. N is a constituent of osmoregulatory compounds, such as proline, which are essential for osmotic homeostasis and photosynthetic pigment integrity [7,32]. Simultaneously, Zn acts as an essential enzymatic cofactor for tryptophan synthesis and antioxidant protection via Superoxide Dismutase (SOD) [33]. The stability of these elements prevented the physiological destabilization observed during the sensitive fruiting stage [34].
The application of biostimulants resulted in positive responses by overcoming the leaching observed under full irrigation. The presence of these microorganisms in the rhizosphere likely creates a “biological reservoir”, reducing nutrient loss through percolation in sandy soils [35]. Furthermore, these compounds facilitate antioxidant defense signaling, contributing to the management of Reactive Oxygen Species (ROS) in the leaves [36].
Regarding microbiological activity, the substantial increase in the bacterial population under Bacillus aryabhattai (10.46 log10 CFU g−1 soil) confirms its capacity to colonize the rhizosphere and produce exopolysaccharides, forming a moisture-retaining biofilm [15]. This density enhances the activity of the ACC deaminase enzyme, which degrades the ethylene precursor and delays stress-induced leaf senescence [11]. This mechanism explains the maintenance of leaf N and the exclusion of Na, even when final yield is limited by severe restriction [12].
In the Trichoderma harzianum treatment, the marked increase in the actinomycetes population at 50% ETc suggests a synergism where the fungus stimulates the native microbiota to mineralize organic matter [8,37]. The yield gain at 75% ETc with T. harzianum is linked to root growth stimulation and rhizosphere acidification via organic acid release, which solubilizes low-mobility elements [10]. This justifies the observed increases in Fe and Mn levels [9,38]. However, yield loss at the 50% ETc depth may be associated with the metabolic cost of maintaining an extensive root system under severe carbon restriction [39].
Unlike living microorganisms, Ascophyllum nodosum acts as an immediate chemical signaler. Rich in betaines and mannitol, it improves soil aggregation and nutrient chelation, explaining the increase in Zn and Cu at 100% ETc [13,14]. However, the supply of labile carbon from the extract may have caused temporary nitrogen immobilization due to competitive demand between the plant and the stimulated microbiota, explaining the reduced leaf N and yield at the 50% ETc depth [40,41].
In general, while leaf nutrient levels are lower than some classical recommendations, they were not limiting to production [26,42]. The yield per plant surpassed values reported in recent regional studies [1,2], suggesting that the interaction between mild tropical conditions and biostimulant activity maximized fruit set and filling. This evidence highlights that while biostimulants offer effective mitigation, their efficiency is strictly modulated by stress severity and interaction with soil microbiota [6]. Finally, the nutrient accumulation order (K > N > Ca > Fe > Mn > Zn > Cu > Na) remained consistent with crop requirements in most treatments, confirming the maintenance of nutritional homeostasis [43].
5. Conclusions
The results indicate that water restriction at 75% ETc is associated with increased yield and foliar concentrations of zinc (Zn) and nitrogen (N). Application of Trichoderma harzianum potentiates the effects of moderate water restriction (75% ETc), correlating with reduced leaf sodium (Na) levels and stable yield while preserving the nutrient accumulation order (K > N > Ca > Fe > Mn > Zn > Cu > Na). Conversely, Bacillus aryabhattai and Ascophyllum nodosum demonstrate potential to enhance yield under 100% ETc by promoting N accumulation. Severe water restriction (50% ETc) is linked to an altered nutrient accumulation order (N > K > Ca > Fe > Mn > Zn > Cu > Na). These findings demonstrate that specific biostimulants mitigate water stress through rhizosphere modification and nutritional homeostasis. Nevertheless, given the environmental variability and the study’s focus on a single cycle, these responses reflect the specific experimental conditions. Future investigations across multiple cycles and diverse locations are recommended to further clarify the underlying physiological mechanisms.
Author Contributions
Conceptualization, M.V.D. and G.S.d.L.; methodology, M.V.D., R.T.d.F., G.S.d.L., H.R.G. and L.A.d.A.S.; software, M.V.D. and R.T.d.F.; validation, M.V.D., G.S.d.L., H.R.G. and L.A.d.A.S.; formal analysis, M.V.D., R.T.d.F., G.S.d.L., H.R.G. and L.A.d.A.S.; investigation, M.V.D., J.d.S.d.S., A.P.N.F., J.S.N., I.A.R., C.N.d.L., G.d.S.d.S., L.F.S.S., L.d.A.S. and S.S.d.S.; resources, G.S.d.L., H.R.G. and R.G.N.; data curation, M.V.D., R.T.d.F., G.S.d.L. and L.A.d.A.S.; writing—original draft preparation, M.V.D., R.T.d.F., G.S.d.L., H.R.G. and L.A.d.A.S.; writing—review and editing, M.V.D., G.S.d.L., H.R.G. and R.G.N.; visualization, M.V.D. and R.T.d.F.; supervision, G.S.d.L., H.R.G. and L.A.d.A.S.; project administration, M.V.D. and G.S.d.L.; funding acquisition, G.S.d.L. and H.R.G. All authors have read and agreed to the published version of the manuscript.
Funding
INCT in Sustainable Agriculture in the Tropical Semi-Arid Region-INCTAgriS (CNPq/Funcap/CAPES), processes 406570/2022-1 (CNPq) and Process INCT-35960-62747.65.95/51 (Funcap).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
To the National Council for Scientific and Technological Development (CNPq) and the Coordination for the Improvement of Higher Education Personnel (CAPES) for the support in carrying out this research, and to the postgraduate programs in Agricultural Engineering and Tropical Horticulture, in addition to INCTAgriS.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Gondim, J.E.F.; Portela, J.C.; Rocha Mendes, K.; Silva, E.F.; Oliveira Gomes, D.V.; Dias, M.C.C.; Antunes, L.F. Land uses and climatic seasonality modulate biological attributes in areas under family farming management in the Caatinga, semi-arid region of Brazil. Environ. Monit. Assess. 2025, 197, 1158. [Google Scholar] [CrossRef] [PubMed]
- Galeano, E.A.V.; Ventura, J.A.; Martins, D.d.S. Socioeconomic characterization of employment in the papaya production chain in Espírito Santo. Int. J. Prof. Bus. Rev. 2024, 9, e04678. [Google Scholar] [CrossRef]
- IBGE—Instituto Brasileiro de Geografia e Estatística. Produção de Mamão. Available online: https://www.ibge.gov.br/explica/producao-agropecuaria/mamao/br (accessed on 12 November 2025).
- Rezende, L.F.; Alves, L.; Barbosa, A.A.; Sales, A.T.; Pedra, G.U.; Menezes, R.S.C.; Arcoverde, G.F.; Ometto, J.P. Greening and water use efficiency during a period of high frequency of droughts in the Brazilian semi-arid. Front. Water 2023, 5, e1295286. [Google Scholar] [CrossRef]
- Krishankumar, S.; Hunter, J.J.; Alyafei, M.; Hamed, F.; Subramaniam, S.; Ramlal, A.; Amiri, K.M. Physiological, biochemical and elemental responses of grafted grapevines under drought stress: Insights into tolerance mechanisms. BMC Plant Biol. 2025, 25, 385. [Google Scholar] [CrossRef]
- Mahouachi, J.; Marrero-Díaz, E. Plant growth and fruit nutrient changes in Carica papaya L. Genotypes subjected to regulated deficit irrigation. Life 2022, 12, e1831. [Google Scholar] [CrossRef]
- Bang, T.C.; Husted, S.; Laursen, K.H.; Persson, D.P.; Schjoerring, J.K. The molecular–physiological functions of mineral macronutrients and their consequences for deficiency symptoms in plants. New Phytol. 2021, 225, 2446–2469. [Google Scholar] [CrossRef]
- Zhang, L.; Jin, Q.; Guan, Y.; Liu, Z.; Pan, X.; Zhang, Y.; Zhang, Y.; Wang, Q. Trichoderma spp. promotes ginseng biomass by influencing the soil microbial community. Front. Microbiol. 2024, 15, 1283492. [Google Scholar] [CrossRef] [PubMed]
- Akbari, S.I.; Prismantoro, D.; Permadi, N.; Rossiana, N.; Miranti, M.; Mispan, M.S.; Mohamed, Z.; Doni, F. Bioprospecting the roles of Trichoderma in alleviating plants’ drought tolerance: Principles, mechanisms of action, and prospects. Microbiol. Res. 2024, 283, e127665. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Chen, S.; Lv, P.; Li, Y.; Li, J.; Jiang, F.; Wu, Z.; Shen, Q.; Zhou, R. Positive Role of Trichoderma harzianum in Increasing Plant Tolerance to Abiotic Stresses: A Review. Antioxidants 2025, 14, 807. [Google Scholar] [CrossRef]
- Abuhena, M.; Zhakypbek, Y.; Uteyev, Y.; Aben, A.; Kamarkhan, Z.; Allakhverdiev, S.I.; Sadvakassova, A.K.; Al Rashid, J.; Karim, M.D.; Kuanysh, T.T.; et al. An overview of Bacillus species in agriculture for growth promotion, biocontrol and dry tolerance. ES Food Agrofor. 2024, 18, 1321. [Google Scholar] [CrossRef]
- Etesami, H.; Jeong, B.R.; Glick, B.R. Potential use of Bacillus spp. as an effective biostimulant against abiotic stresses in crops—A review. Curr. Res. Biotechnol. 2023, 5, e100128. [Google Scholar] [CrossRef]
- Ahmed, M.; Ullah, H.; Himanshu, S.K.; García-Caparrós, P.; Tisarum, R.; Cha-um, S.; Datta, A. Ascophyllum nodosum seaweed extract and potassium alleviate drought damage in tomato by improving plant water relations, photosynthetic performance, and stomatal function. J. Appl. Phycol. 2024, 36, 2255–2268. [Google Scholar] [CrossRef]
- Kumari, S.; Sehrawat, K.D.; Phogat, D.; Sehrawat, A.R.; Chaudhary, R.; Sushkova, S.N.; Voloshina, M.S.; Rajput, V.D.; Shmaraeva, A.N.; Minkina, T.M.; et al. Ascophyllum nodosum (L.) Le Jolis, a pivotal biostimulant toward sustainable agriculture: A comprehensive review. Agriculture 2023, 13, 1179. [Google Scholar] [CrossRef]
- Sousa, H.C.; de Sousa, G.G.; Viana, T.V.d.A.; Pereira, A.P.d.A.; Lessa, C.I.N.; de Souza, M.V.P.; Guilherme, J.M.d.S.; Goes, G.F.; Alves, F.G.d.S.; Gomes, S.P.; et al. Bacillus aryabhattai mitigates the effects of salt and water stress on the agronomic performance of maize under an agroecological system. Agriculture 2023, 13, e1150. [Google Scholar] [CrossRef]
- Álvares, C.A.; Stape, J.L.; Sentelhas, P.C.; Gonçalves, J.D.M.; Sparovek, G. Köppen’s climate classification map for Brazil. Meteorol. Z 2013, 22, 711–728. [Google Scholar] [CrossRef] [PubMed]
- de Melo, A.S.; Sousa, M.S.d.S.; Dutra, A.F.; Brito, M.E.B.; da Silva, J.M.; Viégas, P.R.A.; Maia Júnior, S.d.O.; Dutra, W.F. Physiological and productive performance of papaya plants irrigated in a semiarid environment. Comun. Sci. 2021, 12, e3718. [Google Scholar]
- ISLA—Sementes. 158—Mamão Formosa. Available online: https://www.isla.com.br/mamao-formosa-sunrise-env-linha-super-und-158-61 (accessed on 4 December 2024).
- Teixeira, P.C.; Donagemma, G.K.; Fontana, A.; Teixeira, W.G. Manual de Métodos de Análise de Solo; Embrapa: Rio de Janeiro, Brasil, 2017; p. 573p. [Google Scholar]
- Hungria, M.; Araujo, R.S. Manual de Métodos Empregados em Estudos de Microbiologia Agrícola; Embrapa Serviço de Produção e Informação: Brasília, Brasil, 1994; p. 542p. [Google Scholar]
- Mendonça, E.S.; Matos, E.S. Matéria Orgânica do Solo: Métodos de Análises; Universidade Federal de Viçosa: Viçosa, Brasil, 2005; p. 107p. [Google Scholar]
- EMBRAPA—Empresa Brasileira de Pesquisa Agropecuária. A Cultura do Mamão, 3rd ed.; Coleção Plantar; Informação Tecnológica: Brasília, Brasil, 2009; p. 119p. [Google Scholar]
- Bernardo, S.; Soares, A.A.; Mantovani, E.C. Manual de Irrigação, 9th ed.; Editora da Universidade Federal de Viçosa: Viçosa, Brasil, 2019; p. 545p. [Google Scholar]
- Montenegro, A.A.T.; Bezerra, F.M.L.; Lima, R.N. Evapotranspiração e coeficientes de cultura do mamoeiro para a região litorânea do Ceará. Eng. Agric. 2024, 24, 464–472. [Google Scholar] [CrossRef]
- Topp, G.C.; Davis, J.L.; Annan, A.P. Electromagnetic determination of soil water content: Measurement in coaxial transmission lines. Water Resour. Res. 1980, 16, 574–582. [Google Scholar] [CrossRef]
- Marinho, C.S.; Monnerat, P.H.; de Carvalho, A.J.C.; Marins, S.L.D.; Vieira, A. Análise química do pecíolo e limbo foliar como indicador do estado nutricional dos mamoeiros ‘Solo’ e ‘Formosa’. Sci. Agric. 2002, 59, 373–381. [Google Scholar] [CrossRef]
- da Silva, F.C. Manual de Análises Químicas de Solos, Plantas e Fertilizantes, 2nd ed.; rev. ampl.; Embrapa Informação Tecnológica: Brasília, Brasil, 2009; 627p, Available online: http://www.infoteca.cnptia.embrapa.br/infoteca/handle/doc/330496 (accessed on 9 March 2026).
- Ferreira, D.F. Sisvar: A computer analysis system to fixed effects split-plot type designs. Rev. Bras. Biom 2019, 37, 529–535. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2024. [Google Scholar]
- Abramova, T.V.; Buchkina, N.P. Short-term effect of heavy precipitation on nutrient leaching from arable sandy loam soil amended with fertiliser and biochar. Zemdirbyste 2022, 109, 3–10. [Google Scholar] [CrossRef]
- Yamashita, A.; Hata, T. Sustainability of irrigation fruit farming in terms of water supply-demand situation: Case study of the middle basin of São Francisco River, Northeast Brazil. Geogr. Rev. Jpn. Ser. B 2021, 94, 1–17. [Google Scholar] [CrossRef]
- Sulaman, S.; Nadeem, M.; Shabaan, M.; Orman, S.; Anwar-ul-Haq, M.; Zulfigar, U. Exogenous application of nitrogen (N) and potassium (K) improves drought tolerance in plants: A review. J. Soil Sci. Plant Nutr. 2025, 25, 4850–4865. [Google Scholar] [CrossRef]
- Macwan, A.H.; Shelat, H.N.S.; Jhala, Y.K.; Shah, S.N. Utilization of zinc solubilizing bacteria for better growth and development of summer groundnut (Arachis hypogaea L.). Pharma Innov. J. 2022, 11, 1027–1035. [Google Scholar]
- Xu, E.; Liu, Y.; Gu, D.; Zhan, X.; Li, J.; Zhou, K.; Zhang, P.; Zou, Y. Molecular mechanisms of plant responses to copper: From deficiency to excess. Int. J. Mol. Sci. 2024, 25, e6993. [Google Scholar] [CrossRef] [PubMed]
- Sanjuán, J.; Nápoles, M.C.; Pérez-Mendoza, D.; Lorite, M.J.; Rodríguez-Navarro, D.N. Microbials for agriculture: Why do they call them biostimulants when they mean probiotics? Microorganisms 2023, 11, e153. [Google Scholar] [CrossRef]
- Ahluwalia, O.; Singh, P.C.; Bhatia, R. A review on drought stress in plants: Implications, mitigation and the role of plant growth promoting rhizobacteria. Resour. Environ. Sustain. 2021, 5, e100032. [Google Scholar] [CrossRef]
- Khan, M.T.; Supronienė, S.; Žvirdauskienė, R.; Aleinikovienė, J. Climate, soil, and microbes: Interactions shaping organic matter decomposition in croplands. Agronomy 2025, 15, 1928. [Google Scholar] [CrossRef]
- Sorahinobar, M.; Yusefieh, N.; Rezayian, M.; Shahbazi, M. Multifaceted role of Trichoderma harzianum isolates in mitigating drought stress and promoting adaptive responses in barley cultivars. Sci. Rep. 2025, 15, e26552. [Google Scholar] [CrossRef]
- Vukelić, I.D.; Prokić, L.T.; Racić, G.M.; Pešić, M.B.; Bojović, M.M.; Sierka, E.M.; Kalaji, H.M.; Panković, D.M. Effects of Trichoderma harzianum on photosynthetic characteristics and fruit quality of tomato plants. Int. J. Mol. Sci. 2021, 22, e6961. [Google Scholar] [CrossRef]
- Dell’Aversana, E.; Cirillo, V.; Oosten, M.J.V.; Stasio, E.D.; Saiano, K.; Woodrow, P.; Ciarmiello, L.F.; Maggio, A.; Carillo, P. Ascophyllum nodosum based extracts counteract salinity stress in tomato by remodeling leaf nitrogen metabolism. Plants 2021, 10, e1044. [Google Scholar] [CrossRef] [PubMed]
- Frioni, T.; VanderWeide, J.; Palliotti, A.; Tombesi, S.; Poni, S.; Sabbatini, P. Foliar vs. soil application of Ascophyllum nodosum extracts to improve grapevine water stress tolerance. Sci. Hortic. 2021, 277, e109807. [Google Scholar] [CrossRef]
- Santos, E.M.; Cavalcante, Í.H.L.; Silva Júnior, G.B.; Albano, F.G.; Lima, F.N.; Sousa, A.M.; Cavalcante, L.F. Estado nutricional do mamoeiro Formosa (cv. Caliman 01) em função de adubação com NK e espaçamento de plantio. Comun. Sci. 2014, 5, 229–240. [Google Scholar]
- da Costa, A.N.; da Costa, A.d.F.S.; Ferreguetti, G.A. Manejo da fertilidade do solo e da nutrição do mamoeiro. Inf. Agropecu. 2013, 34, 38–47. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.