Abstract
Background/Objectives: Cardiac surgery, particularly procedures performed with cardiopulmonary bypass (CPB), carries a high risk of neurological complications, including postoperative delirium (POD), which affects 16–73% of patients and increases the likelihood of long-term cognitive impairment. Brain-derived neurotrophic factor (BDNF), a neurotrophin involved in neuronal function, synaptic plasticity, and inflammatory regulation processes, including its Val66Met polymorphism, has been implicated as a potential predictor of POD. This study aimed to evaluate the relationship between perioperative plasma BDNF levels, the BDNF Val66Met polymorphism, and the incidence of POD in patients undergoing elective cardiac surgery with CPB. Methods: This prospective observational single-center study enrolled 287 adults scheduled for elective isolated coronary artery bypass grafting (CABG) with CPB, of whom 107 met all inclusion criteria for final analysis. Exclusion criteria included urgent surgery and pre-existing cognitive or psychiatric disorders. Preoperative evaluation included cognitive testing (MoCA), laboratory and biochemical analysis, and genotyping for BDNF Val66Met. Postoperatively, patients were assessed for POD using the CAM-ICU scale for the first three consecutive days. Cognitive function (using MoCA) and other neurological complications were evaluated during hospitalization, at 30-day and 12-month follow-up. Associations between biomarkers, genetic factors, and clinical outcomes were analyzed. Results: POD occurred in 19.6% of patients who were older, had higher EuroSCORE II, greater coronary disease burden, more frequent prior stroke and chronic kidney disease, and lower neutrophil counts. POD was significantly associated with prolonged hospital stay, need for continuous renal replacement therapy, and reoperation. The BDNF Val66Met polymorphism was present in 31.8% of patients but was not associated with POD, although carriers exhibited higher plasma BDNF concentrations across all time points. Conclusions: Perioperative plasma BDNF concentrations and the BDNF Val66Met polymorphism were not independently associated with the occurrence of POD in elective CABG patients. However, POD was significantly linked to prolonged hospitalization and reoperations. Neurological complications remain an important challenge in cardiac surgery, emphasizing the need for further research and early identification strategies to improve postoperative outcomes.
1. Introduction
Each year, more than two million cardiac surgical procedures are performed worldwide, with an increasing proportion of patients being elderly individuals with comorbidities such as hypertension and diabetes mellitus. This patient population is at heightened risk of postoperative complications, including neurological sequelae [1]. According to the American College of Cardiology (ACC) and the American Heart Association (AHA) classification, neurological complications following cardiac surgery are categorized as type I neuronal injuries (e.g., stroke, transient ischemic attacks) and type II injuries, which encompass postoperative delirium and postoperative cognitive dysfunction (POCD) [2].
Delirium is an acute disturbance of consciousness and attention, accompanied by alterations in cognitive processes such as memory, orientation, and perception [3]. It typically develops within hours to days and is characterized by a fluctuating course of symptoms throughout the day. Delirium is classified according to its etiology—for example, alcohol-related delirium or delirium secondary to critical illness, with sedative-associated, hypoxic, and septic delirium and unclassified phenotypes [4]. In surgical patients, the condition is referred to as postoperative delirium (POD), although diagnostic criteria remain identical to those used for delirium of other causes [3,5]. Management is clearly defined in the updated POD guidelines of the European Society of Anaesthesiology and Intensive Care (ESAIC) [6].
Three clinical subtypes of delirium are recognized: hyperactive, hypoactive, and mixed. This classification is based primarily on psychomotor features [3,7,8,9]. Hyperactive delirium is characterized by increased psychomotor agitation, restlessness, aggression, and frequent hallucinations or delusions [10,11]. In contrast, hypoactive delirium is marked by social withdrawal, apathy, excessive somnolence, and reduced responsiveness to external stimuli. Due to its subtle clinical presentation and absence of overt agitation, hypoactive delirium frequently remains underdiagnosed, contributing to poorer outcomes and increased risk of long-term health consequences [7,8,9].
Despite these differences, all delirium subtypes share certain core clinical features. The most prominent are disturbances of attention and awareness, manifesting as difficulty focusing, inability to follow instructions, and disorientation regarding time, place, or even personal identity [8,12]. Sleep–wake cycle disruptions are also common, with nocturnal insomnia and excessive daytime somnolence impairing patient functioning and presenting further clinical challenges [13,14]. Persistent delirium, lasting for weeks or months, is not uncommon, and approximately 20% of patients still exhibit symptoms at six months post-onset [15].
Accurate recognition of delirium is essential for timely interventions that reduce stress, improve comfort, and shorten its duration [3,16,17]. Education of healthcare providers in recognizing delirium subtypes enables earlier management and helps prevent adverse outcomes [7,9,16,18,19,20].
Brain-derived neurotrophic factor (BDNF) is a neurotrophin mainly expressed in the central nervous system, which regulates neuronal survival, differentiation, and synaptic plasticity [21,22]. It is critical for neurogenesis, neuronal regeneration, and cellular homeostasis, while also modulating brain plasticity in response to environmental stimuli. Beyond neurodevelopment, BDNF has been linked to immunological processes and cognitive regulation [22,23,24].
Cardiac surgery with cardiopulmonary bypass induces significant physiological and inflammatory stress that may alter circulating and cerebral BDNF concentration. Reduced BDNF concentrations have been associated with impaired neuronal recovery and increased susceptibility to neurocognitive disorders, including delirium [5,25,26].
A common genetic variant of the BDNF gene, Val66Met, results from a valine-to-methionine substitution at codon 66, affecting protein processing and secretion. Epidemiological studies suggest that the Met allele occurs in the global population with a frequency ranging from approximately 20% to as much as 50% in selected ethnic groups, although specific values depend on geographic and ethnic origin. In the European population, the frequency of this mutation (heterozygous or homozygous) is estimated at approximately 15–30%, making it the most common mutation in this gene [27,28]. This polymorphism has been implicated in psychiatric and neurodegenerative conditions, and carriers of the Met allele may be more vulnerable to memory disturbances and neuroinflammatory responses under systemic stress [22,28,29,30,31,32]. These findings suggest that BDNF Val66Met may represent a genetic determinant of susceptibility to postoperative delirium [32,33].
Aim of the Study
The aim of this study is to investigate the impact of circulating BDNF concentrations and the BDNF Val66Met polymorphism on the occurrence of postoperative delirium in patients undergoing cardiac surgery with cardiopulmonary bypass.
2. Materials and Methods
A prospective observational clinical study was conducted between September 2020 and June 2024 at a tertiary university hospital of the Pomeranian Medical University in Szczecin.
2.1. Ethical Considerations
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board, i.e., Bioethics Committee of the Pomeranian Medical University in Szczecin, approval no. KB-0012/55/19 issued on 25 February 2019. All eligible patients who agreed to participate received detailed information about the study from a member of the research team and signed written informed consent before enrolment and prior to the initiation of study procedures. To ensure data confidentiality, all analyses were performed on coded (anonymized) data.
2.2. Patient Enrollment
Patients enrolled in this prospective observational study were hospitalized in the Department of Cardiac Surgery in the University Hospital No. 2 of the Pomeranian Medical University. Under a strict study protocol, we enrolled only patients undergoing elective isolated coronary artery bypass grafting (CABG) with the use of cardiopulmonary bypass to ensure a homogeneous study population. Postoperatively, patients were admitted to the post-anaesthesia care unit (PACU) and later returned to the ward at the Department of Cardiac Surgery.
Each enrolled patient received complete information regarding the study from a research team member and was given the opportunity to ask questions and obtain clarifications, in accordance with ICH-GCP standards. Only those patients who signed informed consent for participation, follow-up visits, and both direct and indirect contact during follow-up were included in the study.
2.3. Inclusion Criteria
According to the study protocol, patients were eligible for enrolment if they met the following criteria:
- Male or female, aged 18–90 years.
- Scheduled for elective CABG surgery with the use of cardiopulmonary bypass.
- Provided written informed consent in accordance with ICH-GCP and national regulations.
2.4. Exclusion Criteria
Patients were excluded from the study if any of the following criteria applied:
- Emergency surgical procedures.
- Patients initially qualified for CABG, in whom the surgical approach was modified intraoperatively.
- History of cognitive impairment.
- History of psychiatric disorders (including depression, dementia, psychosis, or schizophrenia).
The study included preoperative, perioperative, and postoperative assessments of each participant.
2.5. Demographic Data and Baseline Assessment
All data were collected through patient interviews and/or review of medical records. Information was recorded using study-specific case report forms (CRFs). In addition to demographic characteristics, attention was given to comorbidities and chronic medication use. Preoperatively, cognitive function was assessed using the Montreal Cognitive Assessment (MoCA, © Z. Nasreddine, MoCA, Montreal, QC, Canada). Baseline data also included medical history, physical examination, and standard anthropometric measurements. The preoperative evaluation comprised routine laboratory tests, including complete blood count, inflammatory markers, renal function parameters, glycated haemoglobin, and arterial blood gas analysis.
2.6. Perioperative Period
All patients underwent elective cardiac surgery using cardiopulmonary bypass under general anesthesia with endotracheal intubation, following a standardised institutional anesthesia protocol. Induction of general anesthesia was achieved using propofol (Fresenius Kabi, Bad Homburg, Germany) and fentanyl (Polfa Warszawa S.A., Warsaw, Poland), followed by neuromuscular blockade with pancuronium (Jelfa S.A., Jelenia Góra, Poland). Endotracheal intubation was performed, and anesthesia was subsequently maintained with inhaled sevoflurane (AbbVie Inc., North Chicago, IL, USA).
Intraoperative analgesia was provided with fentanyl (Polfa Warszawa S.A., Warsaw, Poland). Prior to surgery, patients provided additional consent for genetic testing. During surgery, blood samples were collected at three different time points for BDNF concentration analysis.
2.7. Postoperative Period
The postoperative period was monitored in accordance with the standard institutional protocol for patient management and surveillance. In the immediate postoperative phase, patients were assessed for central nervous system dysfunction. Agitation was evaluated using the Richmond Agitation-Sedation Scale (RASS), while delirium was screened with the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU), performed twice daily—between 6:00–8:00 a.m. and 4:00–6:00 p.m. for the first 3 postoperative days. A structured neurological examination was also performed to detect focal neurological deficits, transient ischemic attack (TIA), stroke, or motor weakness.
Between 48 and 72 h after surgery, the MoCA was repeated to assess changes in cognitive performance. For the first three postoperative days, patients were assessed twice daily with the CAM-ICU to confirm or exclude delirium, alongside evaluation of pain intensity using the Numeric Rating Scale (NRS). The management for POD and pain followed a standardised institutional protocol for all patients undergoing cardiac surgery and included both diagnostic procedures and non-pharmacological interventions, as well as pharmacological treatment (dexmedetomidine, haloperidol) if needed. These assessments were repeated one month after surgery, coinciding with the routine postoperative follow-up visit.
2.8. Long-Term Follow-Up
All patients were followed for 12 months postoperatively. Cognitive function was reassessed using the MoCA, and persistent postoperative pain was evaluated. During the one-year follow-up, the incidence of neurological complications was recorded, including ischemic stroke, reversible ischemic neurologic deficit (RIND), transient ischemic attack (TIA), postoperative delirium (POD), ICU delirium (severe delirium), seizures, postoperative depression, persistent postoperative pain, and postoperative cognitive dysfunction (POCD).
2.9. Genetic Analysis
Blood samples collected for genetic analysis were subjected to DNA isolation using Genomic Mini AX Blood 300 Spin kit (A&A Biotechnology, Gdańsk, Poland) and amplification using Real-Time PCR System (Thermo Fisher Scientific, Waltham, MA, USA). The genetic assessment included evaluation of the frequency of the BDNF Val66Met polymorphism (G196A, rs6265, located at 11p13), with classification of patients according to homozygous and heterozygous variants, using PCR Mix Plus Green kit (A&A Biotechnology, Gdańsk, Poland). A standardized genetic test was used only for this single gene.
2.10. Biochemical Analysis
Serum BDNF concentrations were measured using the Human BDNF ELISA Kit (Sigma-Aldrich, St. Louis, MO, USA) according to the manufacturer’s instructions. Absorbance was measured using the EnVision 2104 (PerkinElmer, Waltham, MA, USA), and results were expressed in pg/mL and corrected for haematocrit.
2.11. Statistical Analysis
The study population was stratified into two subgroups based on the presence or absence of delirium (DEL+ and DEL−). Epidemiological and clinical data, leukocyte indices, and biochemical and genetic results were analyzed in relation to the following outcomes: ICU mortality, in-hospital mortality, duration of mechanical ventilation, incidence of neurological complications (including stroke, transient ischemic attack, postoperative seizures, severe delirium, frequency and severity of cognitive impairment, and severity of postoperative pain).
All statistical analyses were performed using Statistica 13 software (StatSoft, Inc., Tulsa, OK, USA). Median and interquartile ranges (upper and lower quartiles) were calculated for continuous variables, whereas nominal and categorical data were presented as percentages. Since the distribution of continuous variables deviated from normality, non-parametric tests (Mann–Whitney U test) were applied.
The chi-square test was used for nominal data, while within-group comparisons were performed using the Wilcoxon test. Logistic regression analysis was employed to assess associations between variables. A p-value < 0.05 was considered statistically significant.
- Graphical presentation
Figures depicting plasma BDNF concentrations were generated using median and interquartile range (IQR, Q1–Q3) values derived directly from the summary data in corresponding tables. Box-and-whisker plots display the interquartile range as the box, the median as a horizontal line, and whiskers limited to the IQR. Distinct colors were applied to differentiate study groups for clarity and consistency.
3. Results
3.1. Study Population
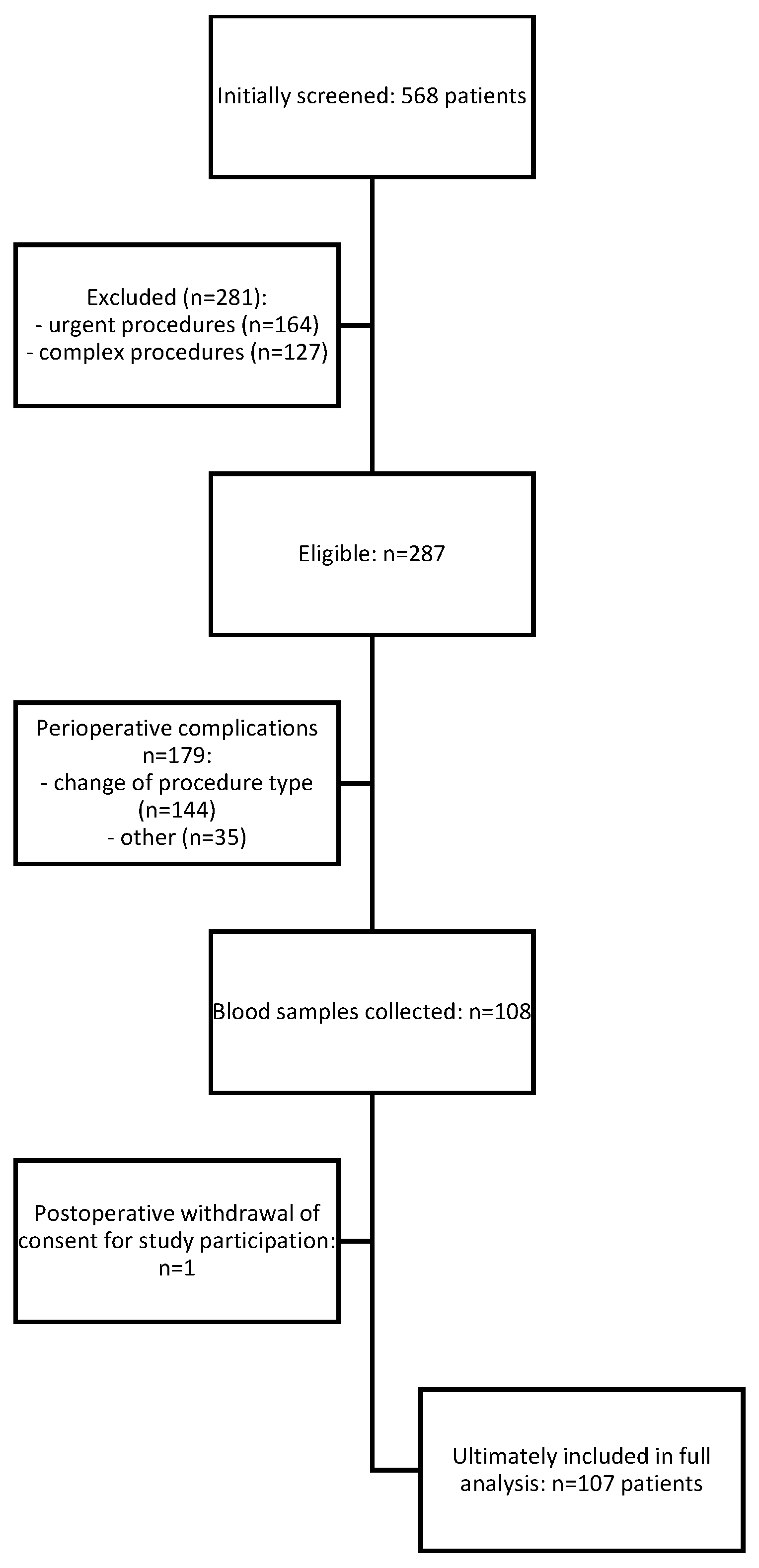
Of the 568 patients initially screened, 281 were excluded due to not meeting the inclusion criteria (urgent or complex surgical procedures). Among the 287 patients eligible for intraoperative enrolment, 179 were excluded intraoperatively: in 144 cases due to modification of the planned surgical procedure, and in 35 cases for perioperative reasons determined by the study team (including failure to collect blood samples, haemolysis, or incomplete neurocognitive testing within protocol-defined time windows).
Of the remaining 108 patients, one withdrew consent during follow-up. Thus, 107 patients were included in the final analysis (Figure 1). The primary stratification criterion was the presence or absence of postoperative delirium (POD) during hospitalization.
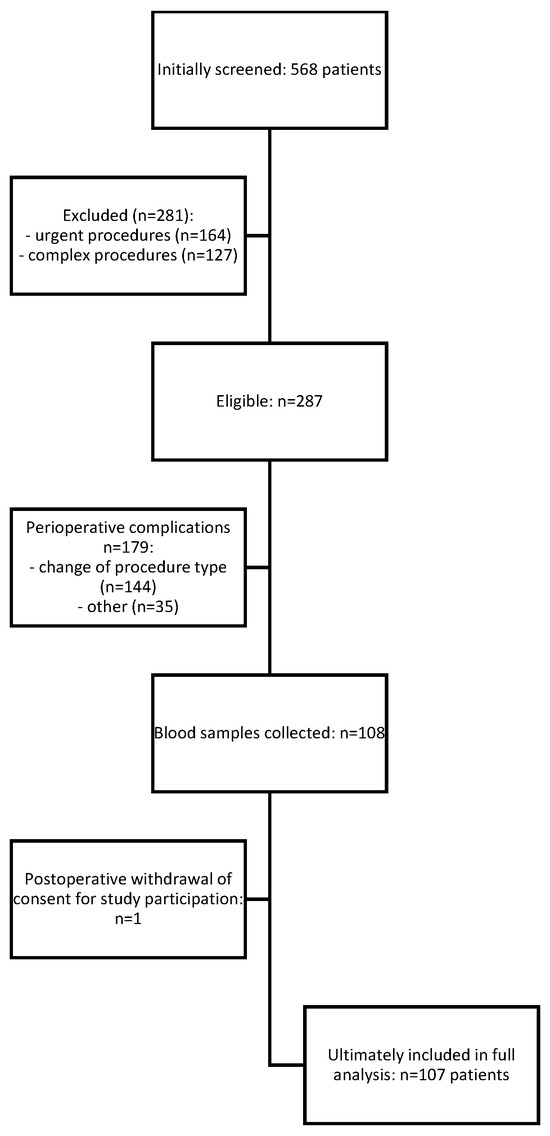
Figure 1.
Study protocol—STROBE diagram of the study.
Baseline characteristics of the study cohort are summarized in Table 1 and Table 2. The mean age was 67 years, and most patients were overweight. Median left ventricular ejection fraction was 45%.

Table 1.
Baseline characteristics of the study cohort (n = 107).

Table 2.
Comorbidities and medications of the study cohort (n = 107).
The majority were men (82.6%), and the most prevalent comorbidities were hypertension, hypercholesterolemia, and prior myocardial infarction.
3.2. Preoperative Characteristics
Patients were divided into those who experienced POD (DEL+) and who did not experience POD (DEL−) groups. Demographic and comorbidity data are shown in Table 3 and Table 4. The proportion of men and women did not differ significantly between groups. Hypertension, hypercholesterolemia, and prior myocardial infarction were the most common comorbidities overall.

Table 3.
Baseline characteristics of the study population stratified by subgroups.

Table 4.
Baseline characteristics of the study population–qualitative data.
There were no significant demographic differences between groups except for age, which was higher in the DEL+ group (69 vs. 67 years, p = 0.031). Patients with POD exhibited more advanced coronary artery disease, as measured by the CCS (Canadian Cardiovascular Society) angina grading scale (p = 0.006), and had higher predicted operative risk according to EuroSCORE II (median 3.54 vs. 2.25, p = 0.006). Prior stroke and chronic kidney disease were also significantly associated with POD.
Notably, POD was more frequent in patients receiving low-molecular-weight heparin before surgery, although not as bridging therapy during anticoagulant switching. No associations were found between POD and other preoperative medications.
There were no significant group differences in BMI, frailty score, baseline left ventricular ejection fraction, or ASA physical status. Similarly, the prevalence of other comorbidities (including ischemic heart disease, chronic heart failure, atrial fibrillation, diabetes, peripheral vascular disease, thyroid disease, COPD, cancer, or autoimmune disorders) did not differ between groups.
Preoperative laboratory testing revealed significantly lower baseline neutrophil counts in the DEL+ group (p = 0.032). Other baseline laboratory values were comparable between groups (Table 5).

Table 5.
Preoperative laboratory testing.
3.3. Intraoperative Findings
Intraoperative data are presented in Table 6 and Table 7. The pre-, intra-, and post-bypass plasma concentrations of brain-derived neurotrophic factor (BDNF) were lower in the DEL+ group, though differences did not reach statistical significance. Duration of anesthesia, cardiopulmonary bypass (CPB) time, total surgical time, and the type of cardioplegia used were not associated with POD incidence. Similarly, transfusion of blood products in the perioperative or early postoperative period did not influence POD risk.

Table 6.
Intraoperative data.

Table 7.
Postoperative data.
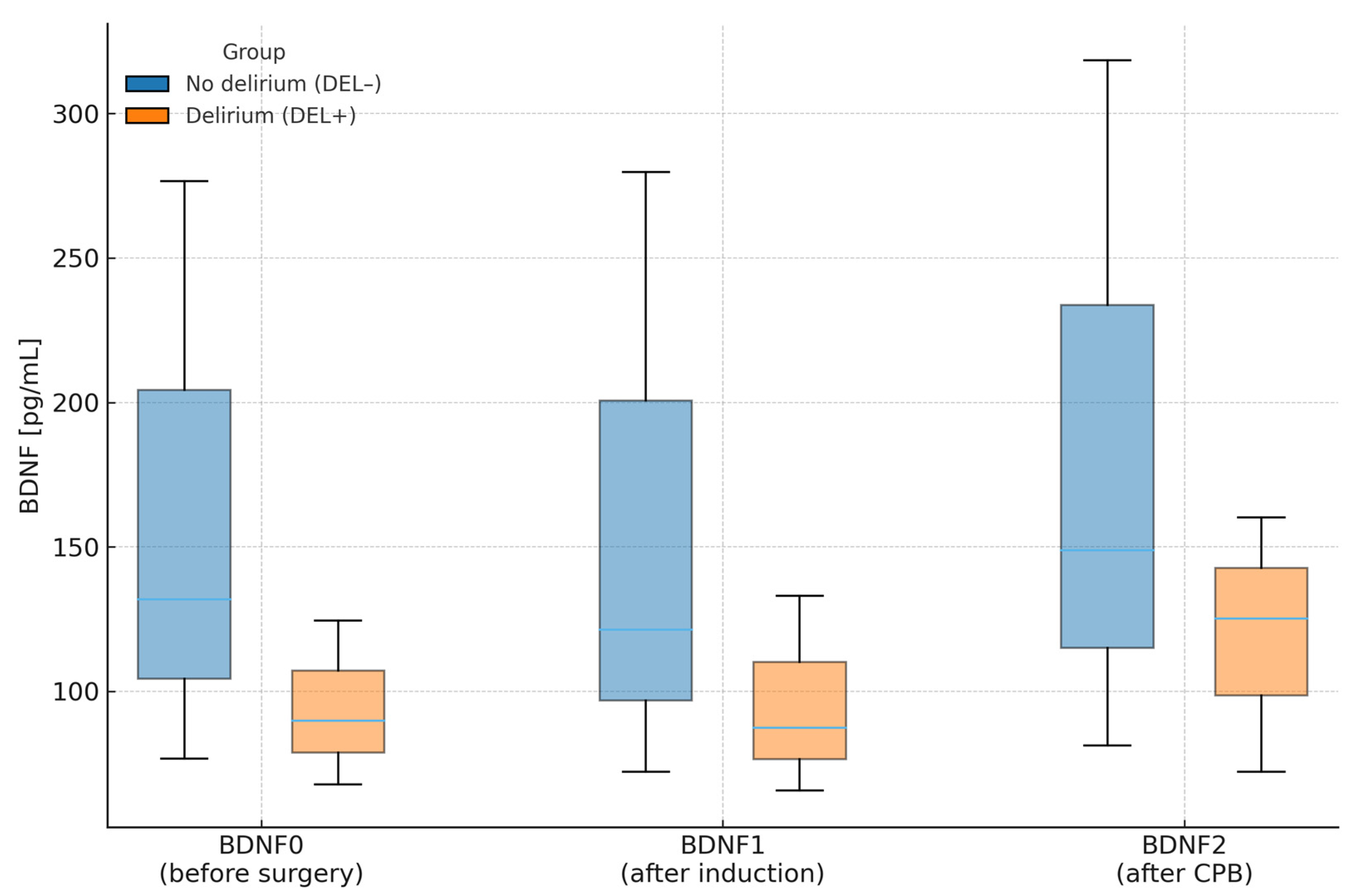
Figure 2 illustrates the median and IQR BDNF concentrations across perioperative time points in patients with (DEL+) and without (DEL−) delirium, corresponding directly to data presented in Table 6. Consistent with those results, patients who developed delirium tended to have lower perioperative BDNF levels, though these differences were not statistically significant. Perioperative trends within each group followed similar patterns without meaningful temporal variation.
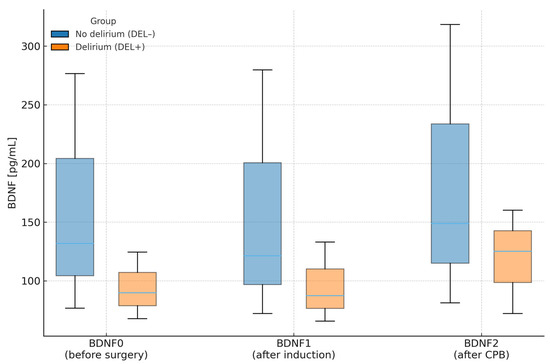
Figure 2.
Plasma BDNF concentrations (median and interquartile range) at three perioperative time points in patients with (DEL+) and without (DEL−) postoperative delirium. Legend: Boxes show IQR (Q1–Q3); horizontal line = median. CPB—cardiopulmonary bypass.
3.4. Postoperative Outcomes
As shown in Table 8, postoperative acute kidney injury requiring renal replacement therapy was strongly associated with POD (p < 0.001), as was the need for reoperation (p = 0.036). POD was also associated with prolonged length of stay (median 8 vs. 6 days, p = 0.012). In contrast, POD did not significantly affect postoperative mortality.

Table 8.
Cognitive function assessment.
3.5. Cognitive Assessment
Cognitive function was assessed perioperatively using the Richmond Agitation-Sedation Scale (RASS) and the Montreal Cognitive Assessment (MoCA). For statistical analysis, MoCA scores were categorized into four groups: 0 (no impairment), 1 (mild impairment), 2 (moderate impairment), and 3 (severe impairment). Postoperative testing (48–72 h) revealed a strong correlation between POD occurrence and impaired MoCA performance, consistent with the cognitive deficits inherent to delirium (Table 8).
3.6. BDNF Val66Met Polymorphism
Genotyping for the BDNF Val66Met polymorphism was performed in all 107 patients (Table 9). The variant was present in 34 patients (31.8%), including 31 heterozygotes and 3 homozygotes. The distribution of the polymorphism did not differ significantly between groups. Interestingly, the variant appeared more frequently in patients without POD (34.9% vs. 19.1%), though this trend did not reach statistical significance (p = 0.126).

Table 9.
Genotyping for the BDNF Val66Met polymorphism.
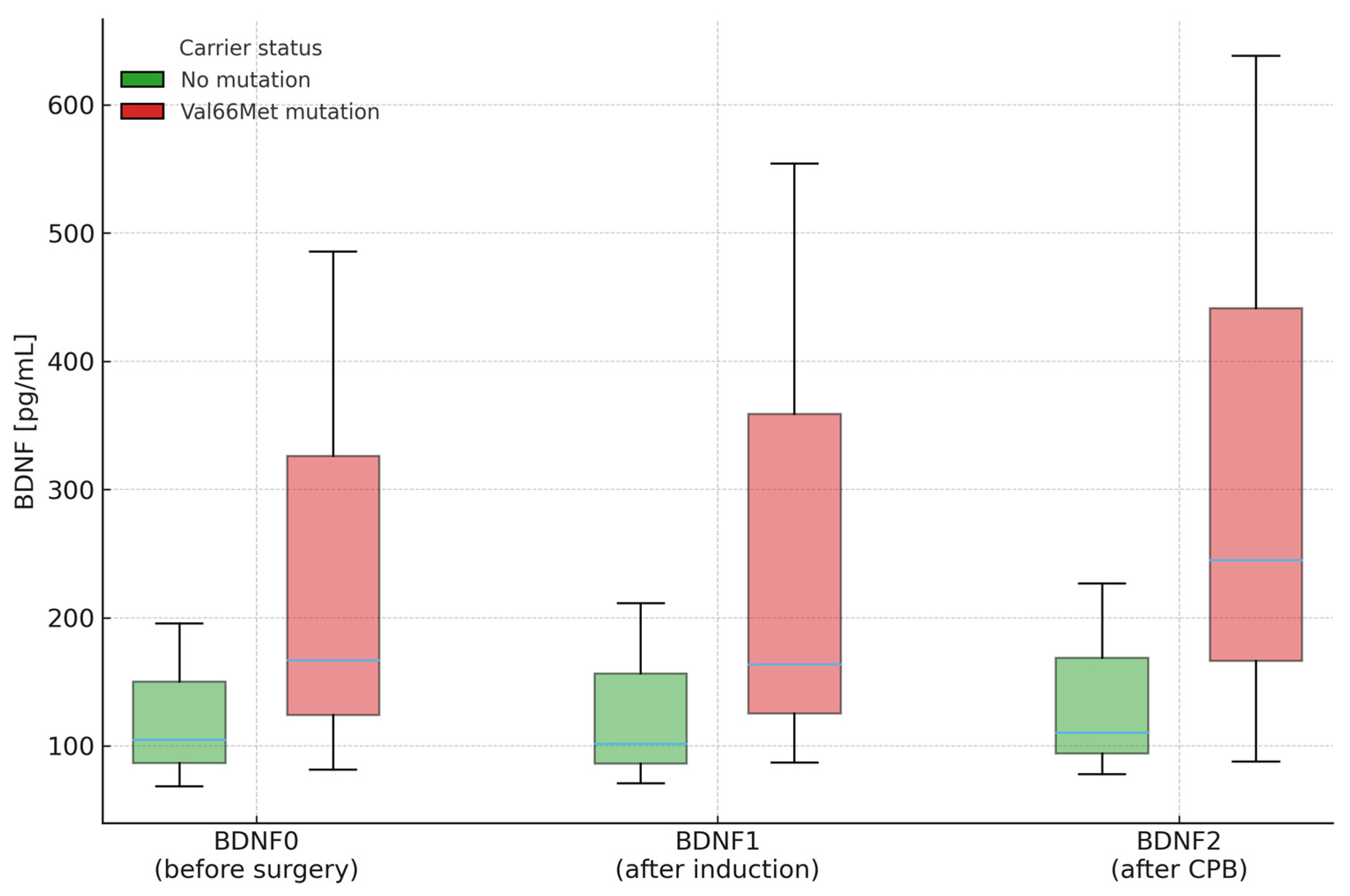
Across all perioperative time points, carriers of Val66Met exhibited higher plasma BDNF concentrations than non-carriers (BDNF0 = 166.48 vs. 104.73 pg/mL, p = 0.003; BDNF1 = 163.68 vs. 101.56 pg/mL, p = 0.024; BDNF2 = 244.89 vs. 110.24 pg/mL, p = 0.010), although within-patient changes (ΔBDNF) did not differ significantly between groups. Logistic-regression analysis confirmed a significant association between the polymorphism and absolute BDNF concentrations at early perioperative time points. Figure 3 depicts these medians and interquartile ranges exactly as reported in Table 9.
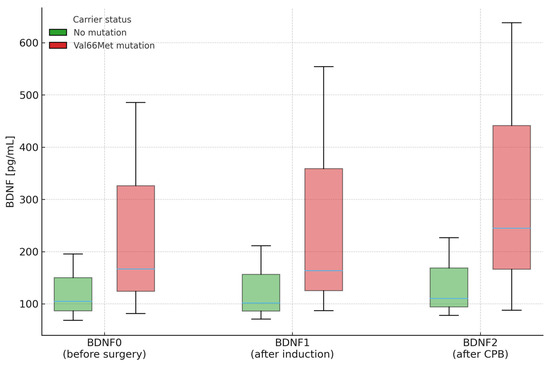
Figure 3.
Plasma BDNF concentrations (median and interquartile range) at three perioperative time points stratified by Val66Met carrier status. Legend: Labels as above. Boxes show IQR (Q1–Q3); horizontal line = median. CPB—cardiopulmonary bypass.
3.7. Logistic Regression of Perioperative Risk Factors for POD
Multivariable logistic regression was used to assess the association between perioperative factors and POD (Table 10). POD was independently associated with prolonged hospital stay (OR = 1.204, p = 0.009), reoperation (OR = 6.510, p = 0.021), and renal replacement therapy (OR = 26.563, p = 0.004), as visible in Table 10. Preoperative C-reactive protein (CRP) concentration was also an independent predictor of POD (OR = 1.187, p = 0.039).

Table 10.
Multivariable logistic regression.
4. Discussion
This study examined the relationship between perioperative BDNF concentrations, the Val66Met polymorphism, and postoperative delirium (POD) incidence in patients undergoing isolated coronary artery bypass grafting (CABG). Our key findings demonstrate that while neither BDNF levels nor the Val66Met polymorphism independently predicted POD, delirium occurred in 19.6% of patients and was strongly associated with adverse postoperative outcomes, including prolonged hospitalization, reoperations, and renal complications. These results contribute to the evolving understanding of perioperative neurocognitive disorders and their clinical implications.
The study cohort represented a typical population undergoing CABG in our institution, with demographic and clinical characteristics consistent with other reports of cardiac surgical patients [19,34,35]. The mean age was 67 years, and the majority were men. The prototypical participant was overweight (BMI 29.4), hypertensive, hypercholesterolemic, and had a history of myocardial infarction. These preoperative comorbidities reflect the well-established continuum of cardiovascular risk factors observed in cardiac surgical populations worldwide [12,36,37,38]. Interestingly, only 14% of patients had a documented history of peripheral vascular disease, and despite a high prevalence of smoking, the proportion with a diagnosis of chronic obstructive pulmonary disease (COPD) was low (4.7%). This may reflect underdiagnosis rather than true absence of disease.
Santos et al. studied a Brazilian cohort and reported a somewhat different risk factor profile compared with our population. In their study, hypertension (>70%) and diabetes were the most prevalent comorbidities, while smoking was less common (~45%), and overweight status was present in <10% of patients. Beta-blockers, nitrates, and diuretics were most frequently used, whereas statin use was not reported [39]. Given the study’s date (2004), both therapeutic strategies and socioeconomic conditions have changed substantially, which may account for these differences. Similarly, Yokoyama et al., analysing Japanese cardiac surgery patients, reported hypertension (45%) and diabetes (40%) as leading comorbidities [40].
In our cohort, no patients experienced stroke or transient ischemic attack (TIA) during hospitalization, while POD occurred in 21 patients (19.6%). The reported incidence of neurological complications after cardiac surgery varies across studies. Raffa et al., in a retrospective analysis of 2121 patients undergoing various cardiac procedures, found major stroke in 1.7% and TIA in 2.5% [41]. Roach et al. (1996) reported a 2.8% incidence of perioperative myocardial infarction and 2.7% incidence of cognitive impairment following elective CABG [42]. Bruggemans et al., in a prospective study of 101 CABG patients, demonstrated memory impairment in 64% at discharge, with nearly complete resolution by 3 months [43].
POD remains a serious and independent complication of postoperative care in cardiac surgery [44]. Patients who developed POD were, on average, nearly 4 years older (70.1 vs. 66.3 years). This age effect is well recognized, with large-scale studies consistently identifying advanced age as a robust risk factor for POD [15,45]. Advanced age is not only a determinant of delirium after surgery, but also across general hospitalized and critically ill populations, with prevalence reported to exceed 70% in some ICU cohorts [46,47]. Standardized diagnostic tools such as CAM-ICU or ICDSC are therefore recommended, and their routine use is part of our institutional practice [8]. The most recent ESAIC guidelines recommend incorporating age, ASA physical status, comorbidities, and cognitive screening tools such as MMSE or MoCA into POD risk assessment [6].
In our study, a high proportion of patients reported regular or occasional alcohol consumption, and only 28% were nonsmokers. Moreover, POD incidence was associated with severity of coronary artery disease (measured by CCS classification), history of stroke, and chronic kidney disease. These findings align with prior studies [48,49]. Gosselt et al. demonstrated associations between POD, age, and renal dysfunction, though unlike our findings and prior work from our center, they did not observe a significant impact of coronary artery disease severity [49,50,51].
4.1. Preoperative Laboratory Parameters
Patients who developed delirium exhibited lower preoperative neutrophil counts, accompanied by a trend toward lower leukocyte and haemoglobin levels and higher creatinine and CRP. Although none of these parameters exceeded laboratory reference ranges or reached consistent statistical significance, the directionality is noteworthy. Previous studies have identified inflammatory and hematologic derangements as potential contributors to POD. Santos et al. reported higher preoperative urea and creatinine levels in delirium patients, while Rudolph et al. described similar associations, albeit without statistical robustness [38,39]. Our findings suggest that subtle immune or hematologic alterations may lower physiological reserve, thereby predisposing patients to postoperative cognitive vulnerability.
4.2. Anticoagulant Therapy
A noteworthy observation was the association between low-molecular-weight heparin (LMWH) use and increased POD incidence. This represents a novel finding in cardiac surgery literature, as previous studies have not systematically examined this relationship. While we interpret this primarily as confounding by indication—patients receiving LMWH typically have greater comorbidity burden—the association warrants further investigation. Interestingly, evidence from non-surgical populations suggests LMWH may have neuroprotective properties, highlighting the need for cardiac surgery-specific research into anticoagulant strategies and their neurocognitive implications [39].
4.3. Cardiopulmonary Bypass and Operative Factors
Contrary to expectations, intraoperative variables—including duration of anesthesia, total operative time, and CPB duration—did not significantly differ between delirium and non-delirium patients. Although patients with POD had slightly longer CPB times, the difference was not statistically significant. These results are consistent with those of Rudolph et al., who observed trends toward prolonged CPB and anesthetic exposure in delirium patients but failed to demonstrate independent predictive value [38]. However, the biological plausibility of CPB-related neuroinflammation and microembolization as contributors to POD remains valid and deserves continued mechanistic investigation.
The conducted study showed that patients who developed postoperative delirium received more units of packed red blood cells transfusions than patients without delirium. The result did not achieve statistical significance. This is consistent with data reported in the literature. Gosselt et al. demonstrated that transfusion of blood products is one of the most significant risk factors for delirium after procedures using cardiopulmonary bypass [50].
4.4. Postoperative Complications
Delirium was strongly associated with adverse postoperative trajectories. Patients with POD required reoperation and renal replacement therapy more frequently and experienced prolonged hospitalization. Importantly, AKI and the need for dialysis emerged as robust correlates of delirium, consistent with prior reports linking renal dysfunction to acute cerebral complications. Siew et al. demonstrated that AKI independently predicts delirium and coma in critically ill patients, while Kotfis et al. reported a tenfold increased risk of POD following CABG in patients with AKI [51,52]. In contrast to some multicenter series, we did not observe increased mortality among delirium patients, possibly reflecting sample size limitations or the effectiveness of early detection and management strategies within our ICU setting.
4.5. Dynamics of BDNF Levels
BDNF is increasingly recognized as a critical mediator of synaptic plasticity, neuroinflammation, and neuronal survival [53]. In our study, pre-, intra-, and post-bypass plasma concentrations of brain-derived neurotrophic factor (BDNF) were lower in patients who developed postoperative delirium (DEL+) compared with those who did not (DEL−), although these differences did not reach statistical significance. This observation is in line with previous studies showing that reduced circulating BDNF concentrations are associated with neurodegenerative and neuropsychiatric conditions, including Alzheimer’s disease, depression, and schizophrenia, all characterized by impaired synaptic plasticity and neuronal resilience [31,53,54]. These findings also align with Grandi et al., who reported that reductions in BDNF preceded delirium onset [55]. However, we did not observe dynamic fluctuations in BDNF trajectories that could serve as predictive biomarkers. While BDNF remains biologically plausible as a marker of neurocognitive resilience, its clinical utility as a standalone predictor of POD remains uncertain.
It is conceivable that lower perioperative BDNF concentrations may reflect a pre-existing vulnerability of neuronal networks and a reduced capacity for neuroplastic recovery after major surgical stress. The observed trend toward lower BDNF levels in the DEL+ group, even in the absence of statistical significance, may therefore suggest a pathophysiological link between decreased neurotrophic support and the development of delirium. However, as demonstrated by the lack of significant differences in anesthesia duration, CPB time, total surgical time, and transfusion rates, intraoperative factors alone are unlikely to explain these findings. Future studies with larger cohorts and serial assessments of BDNF kinetics are warranted to clarify whether perioperative BDNF decline represents a contributing mechanism or merely an epiphenomenon in delirium pathogenesis.
4.6. Val66Met Polymorphism
The Val66Met polymorphism, which affects activity-dependent BDNF secretion, has been implicated in susceptibility to neurodegenerative and psychiatric disorders [27,32,56]. In our population, the mutation frequency was 31.8%, consistent with prior studies in Poland and comparable to frequencies reported in other Caucasian cohorts [28]. Unexpectedly, carriers of the Val66Met variant exhibited significantly higher BDNF plasma levels at all time points compared to non-carriers, contradicting prior reports that link the polymorphism to impaired BDNF release and reduced synaptic plasticity [27,28,32]. Importantly, the presence of the polymorphism was not associated with delirium incidence or with changes in BDNF kinetics, suggesting that its influence may be context-dependent or modulated by additional genetic and environmental factors.
4.7. Limitations
This study has several limitations. The most significant is the relatively small sample size, largely attributable to pandemic-related restrictions on elective CABG procedures and stringent inclusion criteria aimed at ensuring a homogeneous cohort. While these restrictions enhance internal validity, they limit generalizability. Furthermore, intraoperative exclusions due to surgical scope changes further reduced the analytic population. Despite these constraints, the study provides valuable insights by focusing exclusively on isolated CABG, in contrast to most previous research that combined CABG with valve procedures, thereby diluting procedure-specific associations. The relatively small sample size limited the statistical power of the regression analysis and the ability to adjust for multiple confounders. As a result, the regression findings should be interpreted cautiously and viewed as exploratory, generating hypotheses for future, larger-scale studies designed to validate these associations.
4.8. Implications and Future Directions
Our findings reinforce the multifactorial nature of postoperative delirium, with preoperative frailty markers, renal complications, and neurotrophic regulation all playing potential roles. The unexpected association of the Val66Met polymorphism, common genetic variant of the BDNF gene, with elevated BDNF levels warrants replication in larger cohorts and may suggest compensatory mechanisms in response to surgical stress. The Val66Met polymorphism has been implicated in psychiatric and neurodegenerative conditions. Carriers of the Met allele may be more vulnerable to memory disturbances and neuroinflammatory responses, especially under surgical stress; therefore, our findings suggest that BDNF Val66Met may represent a genetic determinant of susceptibility to postoperative delirium.
Future studies should integrate multimodal biomarkers—including inflammatory mediators, neuroimaging, and genetic profiling—to improve risk stratification and guide preventive strategies. Ultimately, a precision medicine approach will be essential to mitigate the burden of delirium in vulnerable cardiac surgery patients.
5. Conclusions
This prospective single-center study of patients undergoing isolated coronary artery bypass grafting (CABG) did not demonstrate a significant association between perioperative changes in BDNF concentrations or presence of the BDNF Val66Met gene mutation and the incidence of postoperative delirium. The occurrence of postoperative delirium was, however, significantly associated with a prolonged length of hospital stay, although it did not increase the risk of mortality. Neurological complications in the postoperative period, including postoperative delirium, remain an important adverse outcome following cardiac surgery and warrant further investigation. Early identification and management of these disturbances may meaningfully influence patient outcomes and functional recovery after surgery.
Author Contributions
Conceptualization, K.L. and K.K.; Methodology, K.L., A.S., B.D., E.C.-H., E.O.-J. and K.K.; Validation, E.Z., J.P., B.D. and K.K.; Resources, K.L., E.C.-H., E.O.-J., E.Z., J.P. and K.K.; Writing—original draft preparation, K.L., A.S., E.C.-H., E.O.-J., E.Z., J.P., B.D. and K.K.; Writing—review and editing, K.L., A.S., E.C.-H., E.O.-J., E.Z., J.P., B.D. and K.K. Supervision, J.P., B.D. and K.K.; Project administration, K.L. and K.K.; Visualization, K.L. and A.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was approved by the Bioethics Committee of the Pomeranian Medical University in Szczecin (approval no. KB-0012/55/19) on 25 February 2019, and was carried out in accordance with the most recent revision of the Declaration of Helsinki, the International Council for Harmonisation Good Clinical Practice Guidelines (ICH-GCP E6, R2), and the current Istanbul Declaration.
Informed Consent Statement
All eligible patients who agreed to participate received detailed information about the study from a member of the research team and signed written informed consent before enrolment and prior to the initiation of study procedures. To ensure data confidentiality, all analyses were performed on coded (anonymized) data.
Data Availability Statement
Data supporting the findings of this study can be obtained from the corresponding author upon reasonable request by researchers.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Natarajan, A.; Samadian, S.; Clark, S. Coronary Artery Bypass Surgery in Elderly People. Postgrad. Med. J. 2007, 83, 154–158. [Google Scholar] [CrossRef]
- Eagle, K.A.; Guyton, R.A.; Davidoff, R.; Ewy, G.A.; Fonger, J.; Gardner, T.J.; Gott, J.P.; Herrmann, H.C.; Marlow, R.A.; Nugent, W.; et al. ACC/AHA Guidelines for Coronary Artery Bypass Graft Surgery: Executive Summary and Recommendations: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1991 Guidelines for Coronary Artery Bypass Graft Surgery). Circulation 1999, 100, 1464–1480. [Google Scholar] [CrossRef]
- Neufeld, K.J.; Thomas, C. Delirium: Definition, Epidemiology, and Diagnosis. J. Clin. Neurophysiol. 2013, 30, 438–442. [Google Scholar] [CrossRef]
- Girard, T.D.; Thompson, J.L.; Pandharipande, P.P.; Brummel, N.E.; Jackson, J.C.; Patel, M.B.; Hughes, C.G.; Chandrasekhar, R.; Pun, B.T.; Boehm, L.M.; et al. Clinical Phenotypes of Delirium during Critical Illness and Severity of Subsequent Long-Term Cognitive Impairment: A Prospective Cohort Study. Lancet Respir. Med. 2018, 6, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.Z.; Liu, C.X.; Zhou, L.G.; Yang, Y.; Wang, Y. Postoperative Delirium, Neuroinflammation, and Influencing Factors of Postoperative Delirium: A Review. Medicine 2023, 102, E32991. [Google Scholar] [CrossRef] [PubMed]
- Aldecoa, C.; Bettelli, G.; Bilotta, F.; Sanders, R.D.; Aceto, P.; Audisio, R.; Cherubini, A.; Cunningham, C.; Dabrowski, W.; Forookhi, A.; et al. Update of the European Society of Anaesthesiology and Intensive Care Medicine Evidence-Based and Consensus-Based Guideline on Postoperative Delirium in Adult Patients. Eur. J. Anaesthesiol. 2024, 41, 81–108. [Google Scholar] [CrossRef] [PubMed]
- Tavares-Pereira, J.; Pereira Novaes, L.; Luz, M.; Brandao Barreto, B.; Gusmao-Flores, D. Identifying and Treating Hypoactive Delirium: Back to Square One? Intensive Care Med. 2025, 51, 201–202. [Google Scholar] [CrossRef]
- Stollings, J.L.; Kotfis, K.; Chanques, G.; Pun, B.T.; Pandharipande, P.P.; Ely, E.W. Delirium in Critical Illness: Clinical Manifestations, Outcomes, and Management. Intensive Care Med. 2021, 47, 1089–1103. [Google Scholar] [CrossRef]
- Kotfis, K.; Mesa, P.; Ely, E.W. How to End Quiet Suffering in the Intensive Care Unit? Identifying and Treating Hypoactive Delirium. Intensive Care Med. 2024, 50, 1695–1698. [Google Scholar] [CrossRef]
- Zakhary, T.; Ahmed, I.; Luttfi, I.; Montasser, M. Quetiapine Versus Haloperidol in the Management of Hyperactive Delirium: Randomized Controlled Trial. Neurocrit. Care 2024, 41, 550–557. [Google Scholar] [CrossRef]
- Ramírez Echeverría, M.d.L.; Schoo, C.; Paul, M. Delirium; StatPearls: Treasure Island, FL, USA, 2025. [Google Scholar]
- Marcantonio, E.R. Delirium in Hospitalized Older Adults. N. Engl. J. Med. 2017, 377, 1456–1466. [Google Scholar] [CrossRef]
- Kang, J.; Cho, Y.S.; Lee, M.; Yun, S.; Jeong, Y.J.; Won, Y.H.; Hong, J.; Kim, S. Effects of Nonpharmacological Interventions on Sleep Improvement and Delirium Prevention in Critically Ill Patients: A Systematic Review and Meta-Analysis. Aust. Crit. Care 2023, 36, 640–649. [Google Scholar] [CrossRef]
- Kuhlmann, J.; Alhammadi, E.; Mevissen, A.; Möllmann, H. Delirium and Sleep Disturbances—A Narrative Review. Z. Gerontol. Geriatr. 2023, 56, 539–544. [Google Scholar] [CrossRef]
- Cole, M.G.; Ciampi, A.; Belzile, E.; Zhong, L. Persistent Delirium in Older Hospital Patients: A Systematic Review of Frequency and Prognosis. Age Ageing 2009, 38, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Marra, A.; Kotfis, K.; Hosie, A.; MacLullich, A.M.J.; Pandharipande, P.P.; Ely, E.W.; Pun, B.T. Delirium Monitoring: Yes or No? That Is The Question. Am. J. Crit. Care 2019, 28, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Marra, A.; Pandharipande, P.P.; Patel, M.B. Intensive Care Unit Delirium and Intensive Care Unit-Related Posttraumatic Stress Disorder. Surg. Clin. N. Am. 2017, 97, 1215–1235. [Google Scholar] [CrossRef]
- Kotfis, K.; Marra, A.; Ely, E.W. ICU Delirium—A Diagnostic and Therapeutic Challenge in the Intensive Care Unit. Anaesthesiol. Intensive Ther. 2018, 50, 160–167. [Google Scholar] [CrossRef]
- Szylińska, A.; Rotter, I.; Listewnik, M.; Lechowicz, K.; Brykczyński, M.; Dzidek, S.; Żukowski, M.; Kotfis, K. Postoperative Delirium in Patients with Chronic Obstructive Pulmonary Disease after Coronary Artery Bypass Grafting. Medicina 2020, 56, 342. [Google Scholar] [CrossRef]
- Girard, T.D.; Jackson, J.C.; Pandharipande, P.P.; Pun, B.T.; Thompson, J.L.; Shintani, A.K.; Gordon, S.M.; Canonico, A.E.; Dittus, R.S.; Bernard, G.R.; et al. Delirium as a Predictor of Long-Term Cognitive Impairment in Survivors of Critical Illness. Crit. Care Med. 2010, 38, 1513–1520. [Google Scholar] [CrossRef]
- Brigadski, T.; Leßmann, V. The Physiology of Regulated BDNF Release. Cell Tissue Res. 2020, 382, 15–45. [Google Scholar] [CrossRef] [PubMed]
- Numakawa, T.; Suzuki, S.; Kumamaru, E.; Adachi, N.; Richards, M.; Kunugi, H. BDNF Function and Intracellular Signaling in Neurons. Histol. Histopathol. 2010, 25, 237–258. [Google Scholar] [CrossRef]
- Milyutina, Y.P.; Arutjunyan, A.V.; Korenevsky, A.V.; Selkov, S.A.; Kogan, I.Y. Neurotrophins: Are They Involved in Immune Tolerance in Pregnancy? Am. J. Reprod. Immunol. 2023, 89, e13694. [Google Scholar] [CrossRef] [PubMed]
- Chow, R.; Wessels, J.M.; Foster, W.G. Brain-Derived Neurotrophic Factor (BDNF) Expression and Function in the Mammalian Reproductive Tract. Hum. Reprod. Update 2020, 26, 545–564. [Google Scholar] [CrossRef] [PubMed]
- Travica, N.; Aslam, H.; O’Neil, A.; Lane, M.M.; Berk, M.; Gamage, E.; Walder, K.; Liu, Z.S.J.; Segasby, T.; Marx, W. Brain Derived Neurotrophic Factor in Perioperative Neurocognitive Disorders: Current Evidence and Future Directions. Neurobiol. Learn. Mem. 2022, 193, 107656. [Google Scholar] [CrossRef]
- Miniksar, Ö.H.; Çiçekçioğlu, F.; Kılıç, M.; Honca, M.; Miniksar, D.Y.; Gocmen, A.Y.; Kaçmaz, O.; Öz, H. Decreased Brain-Derived Neurotrophic Factor Levels May Predict Early Perioperative Neurocognitive Disorder in Patients Undergoing Coronary Artery Bypass Surgery: A Prospective Observational Pilot Study. J. Clin. Anesth. 2021, 71, 110235. [Google Scholar] [CrossRef]
- Finan, J.D.; Udani, S.V.; Patel, V.; Bailes, J.E. The Influence of the Val66Met Polymorphism of Brain-Derived Neurotrophic Factor on Neurological Function after Traumatic Brain Injury. J. Alzheimers Dis. 2018, 65, 1055. [Google Scholar] [CrossRef]
- Suchanek, R.; Owczarek, A.; Paul-Samojedny, M.; Kowalczyk, M.; Kucia, K.; Kowalski, J. BDNF Val66met Polymorphism Is Associated with Age at Onset and Intensity of Symptoms of Paranoid Schizophrenia in a Polish Population. J. Neuropsychiatry Clin. Neurosci. 2013, 25, 88–94. [Google Scholar] [CrossRef]
- Notaras, M.; van den Buuse, M. Brain-Derived Neurotrophic Factor (BDNF): Novel Insights into Regulation and Genetic Variation. Neuroscientist 2019, 25, 434–454. [Google Scholar] [CrossRef] [PubMed]
- Hempstead, B.L. Brain-Derived Neurotrophic Factor: Three Ligands, Many Actions. Trans. Am. Clin. Climatol. Assoc. 2015, 126, 9–19. [Google Scholar]
- Notaras, M.; van den Buuse, M. Neurobiology of BDNF in Fear Memory, Sensitivity to Stress, and Stress-Related Disorders. Mol. Psychiatry 2020, 25, 2251–2274. [Google Scholar] [CrossRef]
- Notaras, M.; Hill, R.; Van Den Buuse, M. The BDNF Gene Val66Met Polymorphism as a Modifier of Psychiatric Disorder Susceptibility: Progress and Controversy. Mol. Psychiatry 2015, 20, 916–930. [Google Scholar] [CrossRef]
- Lima Giacobbo, B.; Doorduin, J.; Klein, H.C.; Dierckx, R.A.J.O.; Bromberg, E.; de Vries, E.F.J. Brain-Derived Neurotrophic Factor in Brain Disorders: Focus on Neuroinflammation. Mol. Neurobiol. 2019, 56, 3295–3312. [Google Scholar] [CrossRef]
- Kotfis, K.; Szylińska, A.; Listewnik, M.; Lechowicz, K.; Kosiorowska, M.; Drożdżal, S.; Brykczyński, M.; Rotter, I.; Żukowski, M. Balancing Intubation Time with Postoperative Risk in Cardiac Surgery Patients—A Retrospective Cohort Analysis. Ther. Clin. Risk Manag. 2018, 14, 2203–2212. [Google Scholar] [CrossRef]
- Lechowicz, K.; Szylińska, A.; Listewnik, M.; Drożdżal, S.; Tomska, N.; Rotter, I.; Kotfis, K. Cardiac Delirium Index for Predicting the Occurrence of Postoperative Delirium in Adult Patients After Coronary Artery Bypass Grafting. Clin. Interv. Aging 2021, 16, 487. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.-H.; Chen, Y.-C.; Chu, C.-C.; Wang, J.-J.; Liao, K.-M. Postoperative Complications After Coronary Artery Bypass Grafting in Patients With Chronic Obstructive Pulmonary Disease. Medicine 2016, 95, e2926. [Google Scholar] [CrossRef] [PubMed]
- Shaefi, S.; Mittel, A.; Loberman, D.; Ramakrishna, H. Off-Pump Versus On-Pump Coronary Artery Bypass Grafting—A Systematic Review and Analysis of Clinical Outcomes. J. Cardiothorac. Vasc. Anesth. 2019, 33, 232–244. [Google Scholar] [CrossRef]
- Rudolph, J.L.; Jones, R.N.; Levkoff, S.E.; Rockett, C.; Inouye, S.K.; Sellke, F.W.; Khuri, S.F.; Lipsitz, L.A.; Ramlawi, B.; Levitsky, S.; et al. Derivation and Validation of a Preoperative Prediction Rule for Delirium after Cardiac Surgery. Circulation 2008, 119, 229. [Google Scholar] [CrossRef] [PubMed]
- Santana Santos, F.; Tadeu Velasco, I.; Fráguas, R. Risk Factors for Delirium in the Elderly after Coronary Artery Bypass Graft Surgery. Int. Psychogeriatr. 2004, 16, 175–193. [Google Scholar] [CrossRef]
- Yokoyama, C.; Yoshitnai, K.; Ogata, S.; Fukushima, S.; Matsuda, H. Effect of Postoperative Delirium after Cardiovascular Surgery on 5-Year Mortality. JA Clin. Rep. 2023, 9, 66. [Google Scholar] [CrossRef]
- Raffa, G.M.; Agnello, F.; Occhipinti, G.; Miraglia, R.; Lo Re, V.; Marrone, G.; Tuzzolino, F.; Arcadipane, A.; Pilato, M.; Luca, A. Neurological Complications after Cardiac Surgery: A Retrospective Case-Control Study of Risk Factors and Outcome. J. Cardiothorac. Surg. 2019, 14, 23. [Google Scholar] [CrossRef]
- Roach, G.W.; Kanchuger, M.; Mangano, C.M.; Newman, M.; Nussmeier, N.; Wolman, R.; Aggarwal, A.; Marschall, K.; Graham, S.H.; Ley, C.; et al. Adverse Cerebral Outcomes after Coronary Bypass Surgery. N. Engl. J. Med. 1996, 335, 1857–1864. [Google Scholar] [CrossRef]
- Bruggemans, E.F. Cognitive Dysfunction after Cardiac Surgery: Pathophysiological Mechanisms and Preventive Strategies. Neth. Heart J. 2013, 21, 70–73. [Google Scholar] [CrossRef] [PubMed]
- Mevorach, L.; Forookhi, A.; Farcomeni, A.; Romagnoli, S.; Bilotta, F. Perioperative Risk Factors Associated with Increased Incidence of Postoperative Delirium: Systematic Review, Meta-Analysis, and Grading of Recommendations Assessment, Development, and Evaluation System Report of Clinical Literature. Br. J. Anaesth. 2023, 130, e254–e262. [Google Scholar] [CrossRef]
- Burkhart, C.S.; Dell-Kuster, S.; Gamberini, M.; Moeckli, A.; Grapow, M.; Filipovic, M.; Seeberger, M.D.; Monsch, A.U.; Strebel, S.P.; Steiner, L.A. Modifiable and Nonmodifiable Risk Factors for Postoperative Delirium after Cardiac Surgery with Cardiopulmonary Bypass. J. Cardiothorac. Vasc. Anesth. 2010, 24, 555–559. [Google Scholar] [CrossRef]
- McPherson, J.A.; Wagner, C.E.; Boehm, L.M.; Hall, J.D.; Johnson, D.C.; Miller, L.R.; Burns, K.M.; Thompson, J.L.; Shintani, A.K.; Ely, E.W.; et al. Delirium in the Cardiovascular ICU: Exploring Modifiable Risk Factors. Crit. Care Med. 2013, 41, 405–413. [Google Scholar] [CrossRef]
- Gottesman, R.F.; Grega, M.A.; Bailey, M.M.; Pham, L.D.; Zeger, S.L.; Baumgartner, W.A.; Selnes, O.A.; McKhann, G.M. Delirium after Coronary Artery Bypass Graft Surgery and Late Mortality. Ann. Neurol. 2010, 67, 338–344. [Google Scholar] [CrossRef]
- Zhong, X.; Lin, J.-Y.; Li, L.; Barrett, A.M.; Poeran, J.; Mazumdar, M. Derivation and Validation of a Novel Comorbidity-Based Delirium Risk Index to Predict Postoperative Delirium Using National Administrative Healthcare Database. Health Serv. Res. 2021, 56, 154–165. [Google Scholar] [CrossRef]
- Kotfis, K.; Szylińska, A.; Listewnik, M.; Strzelbicka, M.; Brykczyński, M.; Rotter, I.; Żukowski, M. Early Delirium after Cardiac Surgery: An Analysis of Incidence and Risk Factors in Elderly (≥65 Years) and Very Elderly (≥80 Years) Patients. Clin. Interv. Aging 2018, 13, 1061–1070. [Google Scholar] [CrossRef]
- Gosselt, A.N.; Slooter, A.J.; Boere, P.R.; Zaal, I.J. Risk Factors for Delirium after On-Pump Cardiac Surgery: A Systematic Review. Crit. Care 2015, 19, 346. [Google Scholar] [CrossRef] [PubMed]
- Kotfis, K.; Ślozowska, J.; Listewnik, M.; Szylińska, A.; Rotter, I. The Impact of Acute Kidney Injury in the Perioperative Period on the Incidence of Postoperative Delirium in Patients Undergoing Coronary Artery Bypass Grafting—Observational Cohort Study. Int. J. Environ. Res. Public Health 2020, 17, 1440. [Google Scholar] [CrossRef] [PubMed]
- Siew, E.D.; Fissell, W.H.; Tripp, C.M.; Blume, J.D.; Wilson, M.D.; Clark, A.J.; Vincz, A.J.; Ely, E.W.; Pandharipande, P.P.; Girard, T.D. Acute Kidney Injury as a Risk Factor for Delirium and Coma during Critical Illness. Am. J. Respir. Crit. Care Med. 2017, 195, 1597–1607. [Google Scholar] [CrossRef]
- Hovens, I.B.; van Leeuwen, B.L.; Mariani, M.A.; Kraneveld, A.D.; Schoemaker, R.G. Postoperative Cognitive Dysfunction and Neuroinflammation; Cardiac Surgery and Abdominal Surgery Are Not the Same. Brain Behav. Immun. 2016, 54, 178–193. [Google Scholar] [CrossRef] [PubMed]
- Ritter, C.; Miranda, A.S.; Giombelli, V.R.; Tomasi, C.D.; Comim, C.M.; Teixeira, A.L.; Quevedo, J.; Dal-Pizzol, F. Brain-Derived Neurotrophic Factor Plasma Levels Are Associated with Mortality in Critically Ill Patients Even in the Absence of Brain Injury. Crit. Care 2012, 16, R234. [Google Scholar] [CrossRef] [PubMed]
- Grandi, C.; Tomasi, C.D.; Fernandes, K.; Stertz, L.; Kapczinski, F.; Quevedo, J.; Dal-Pizzol, F.; Ritter, C. Brain-Derived Neurotrophic Factor and Neuron-Specific Enolase, but Not S100β, Levels Are Associated to the Occurrence of Delirium in Intensive Care Unit Patients. J. Crit. Care 2011, 26, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Gliwińska, A.; Czubilińska-Łada, J.; Więckiewicz, G.; Świętochowska, E.; Badeński, A.; Dworak, M.; Szczepańska, M. The Role of Brain-Derived Neurotrophic Factor (BDNF) in Diagnosis and Treatment of Epilepsy, Depression, Schizophrenia, Anorexia Nervosa and Alzheimer’s Disease as Highly Drug-Resistant Diseases: A Narrative Review. Brain Sci. 2023, 13, 163. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).