Systematic Item Content and Overlap Analysis of Self-Reported Multiple Sleep Disorder Screening Questionnaires in Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of Self-Reported Multiple Sleep Disorder Screening Questionnaires
2.2. Extraction and Selection of Items
2.3. Extraction of Symptoms from Items
2.3.1. First Step: Symptom Extraction from Items within Each Questionnaire
- 1.
- From a methodological point of view, we used a double-blind method based on a panel of medical examiners outside the sleep community to increase reliability. Therefore, in addition to sleep experts (AR, RL, and JAM), this step was also carried out by medical students (LV, JLP, and IZ). This double-blind approach was used to develop large-scale questionnaire studies in psychiatric epidemiology, which led in particular to the constitution of the DSM-III, such as the US Mental Hygiene Movement, the Midtown Manhattan Study, and the Stirling County studies [43,44,45]. In those studies, the opinion of examiners outside the (sleep medicine) community was considered particularly important to avoid as much as possible value-laden choices and the influence of theoretical backgrounds on the fitting of the items of self-reported questionnaires with diagnostic criteria. Examiners’ opinions were then cross-referenced with those of the sleep experts.
- 2.
- From a content analysis point of view, we differentiated three kinds of symptoms within the items: compound symptoms, specific symptoms, and idiosyncratic symptoms, according to the terminology used in previous content overlap analysis studies [23,24,25,26,27,28,29,30].
- i.
- A compound symptom was extracted from an item referring simultaneously to at least two distinct symptoms. For instance, in the SDS-CL-25, item 6, “I am tired, fatigued, or sleepy during the day,” refers to both symptoms “Fatigue” and “Daytime sleepiness.”
- ii.
- A specific symptom was extracted from an item that refers specifically and solely to it. For instance, item 10 of the SDS-CL-25 “I snore” refers only to the “Snoring” symptom. When the same symptom was extracted from items referring to it in a specific way and in a compound way, we considered this symptom specific.
- iii.
- A symptom was considered idiosyncratic if it was extracted from an item that featured only once in a single questionnaire among all the questionnaires analyzed. For instance, the symptom “Hot/cool sensation” is present only in the PSQI in items 5f and 5g “Feel too cold”/“Feel too hot.”
2.3.2. Second Step: Harmonization between Questionnaires
2.3.3. Third Step: Aggregation of Data in a File
2.4. Statistical Analysis
2.4.1. Analysis of the Number and Frequency of Sleep Symptoms
2.4.2. Analysis of Content Overlap
2.4.3. Data Visualization of the Reproducibility of Content Overlap Results
2.4.4. Availability and Reproducibility of Results
3. Results
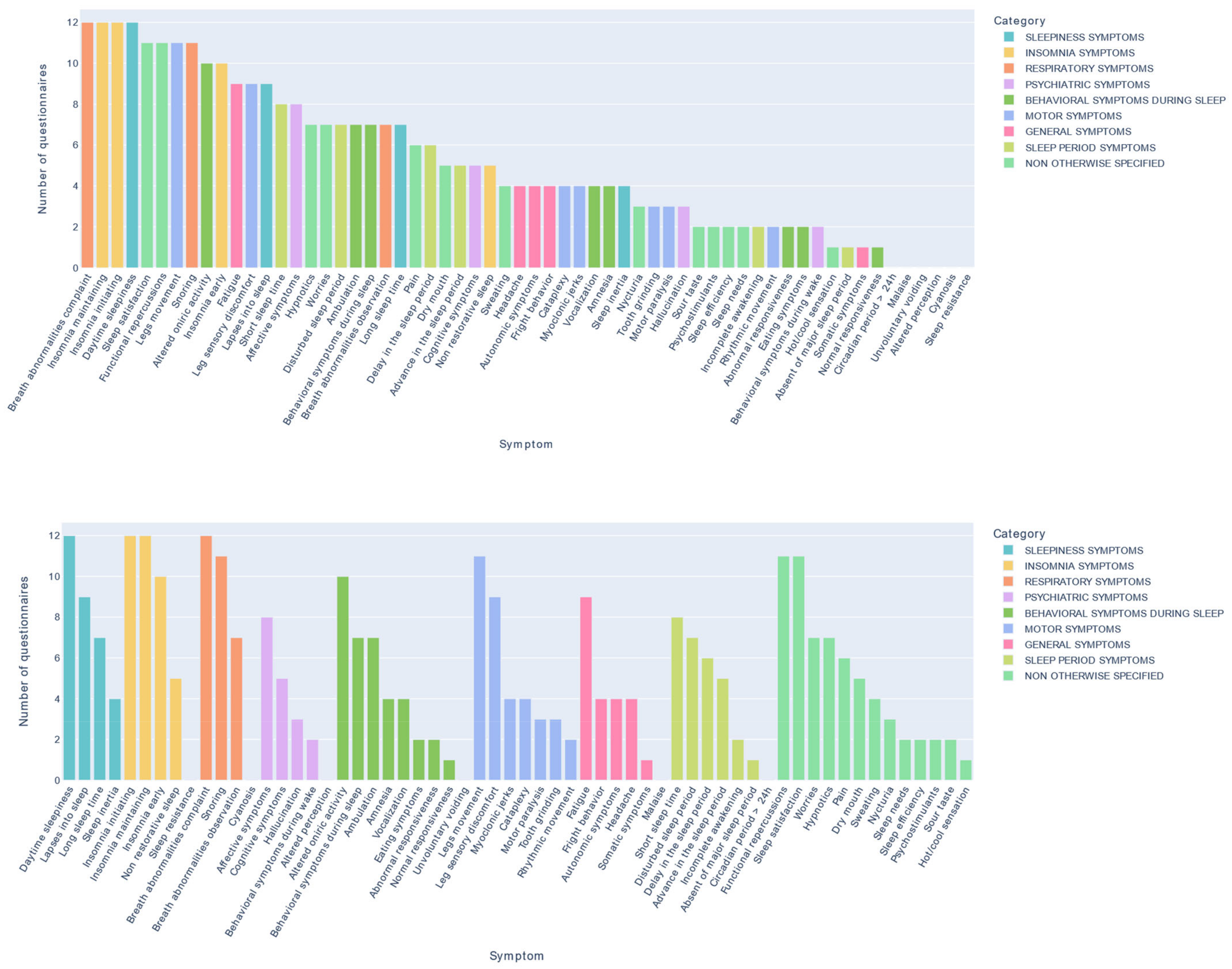
3.1. Number and Frequency of Sleep Symptoms
3.2. Analysis and Data Visualization of Content Overlap
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Academy of Sleep Medicine. International Classification of Sleep Medicine, 3rd ed.; American Academy of Sleep Medicine: Darien, IL, USA, 2014. [Google Scholar]
- Gauld, C.; Lopez, R.; Morin, C.; Geoffroy, P.A.; Maquet, J.; Desvergnes, P.; McGonigal, A.; Dauvilliers, Y.; Philip, P.; Dumas, G.; et al. Symptom network analysis of the sleep disorders diagnostic criteria based on the clinical text of the ICSD-3. J. Sleep Res. 2022, 31, e13435. [Google Scholar] [CrossRef] [PubMed]
- Gauld, C.; Lopez, R.; Morin, C.M.; Maquet, J.; McGonigal, A.; Geoffroy, P.A.; Fakra, E.; Philip, P.; Dumas, G.; Micoulaud-Franchi, J.A. Why do sleep disorders belong to mental disorder classifications? A network analysis of the “Sleep-Wake Disorders” section of the DSM-5. J. Psychiatr. Res. 2021, 142, 153–159. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorder, 5th ed.; (DSM-5); American Psychiatric Association: Washington, DC, USA, 2014. [Google Scholar]
- Gauld, C.; Lopez, R.; Geoffroy, P.A.; Morin, C.M.; Guichard, K.; Giroux, E.; Dauvilliers, Y.; Dumas, G.; Philip, P.; Micoulaud-Franchi, J.A. A systematic analysis of ICSD-3 diagnostic criteria and proposal for further structured iteration. Sleep Med. Rev. 2021, 58, 101439. [Google Scholar] [CrossRef]
- Edinger, J.D.; Morin, C. Sleep disorders classification and diagnosis. In The Oxford Handbook of Sleep and Sleep Disorder; Morin, C., Espie, C.A., Eds.; Oxford Library of Psychology, Oxford University Press: Oxford, UK, 2012; pp. 361–382. [Google Scholar]
- Pevernagie, D.A.; Gnidovec-Strazisar, B.; Grote, L.; Heinzer, R.; McNicholas, W.T.; Penzel, T.; Randerath, W.; Schiza, S.; Verbraecken, J.; Arnardottir, E.S. On the rise and fall of the apnea-hypopnea index: A historical review and critical appraisal. J. Sleep Res. 2020, 29, e13066. [Google Scholar] [CrossRef]
- Arnardottir, E.S.; Islind, A.S.; Oskarsdottir, M.; Olafsdottir, K.A.; August, E.; Jonasdottir, L.; Hrubos-Strom, H.; Saavedra, J.M.; Grote, L.; Hedner, J.; et al. The Sleep Revolution project: The concept and objectives. J. Sleep Res. 2022, 31, e13630. [Google Scholar] [CrossRef]
- McNicholas, W.T.; Pevernagie, D. Obstructive sleep apnea: Transition from pathophysiology to an integrative disease model. J. Sleep Res. 2022, 31, e13616. [Google Scholar] [CrossRef] [PubMed]
- Randerath, W.; Bassetti, C.L.; Bonsignore, M.R.; Farre, R.; Ferini-Strambi, L.; Grote, L.; Hedner, J.; Kohler, M.; Martinez-Garcia, M.A.; Mihaicuta, S.; et al. Challenges and perspectives in obstructive sleep apnoea: Report by an ad hoc working group of the Sleep Disordered Breathing Group of the European Respiratory Society and the European Sleep Research Society. Eur. Respir. J. 2018, 52, 1702616. [Google Scholar] [CrossRef]
- Buysse, D.J.; Young, T.; Edinger, J.D.; Carroll, J.; Kotagal, S. Clinicians’ use of the International Classification of Sleep Disorders: Results of a national survey. Sleep 2003, 26, 48–51. [Google Scholar]
- Edinger, J.D.; Bonnet, M.H.; Bootzin, R.R.; Doghramji, K.; Dorsey, C.M.; Espie, C.A.; Jamieson, A.O.; McCall, W.V.; Morin, C.M.; Stepanski, E.J.; et al. Derivation of research diagnostic criteria for insomnia: Report of an American Academy of Sleep Medicine Work Group. Sleep 2004, 27, 1567–1596. [Google Scholar] [CrossRef]
- Riemann, D.; Baglioni, C.; Bassetti, C.; Bjorvatn, B.; Dolenc Groselj, L.; Ellis, J.G.; Espie, C.A.; Garcia-Borreguero, D.; Gjerstad, M.; Goncalves, M.; et al. European guideline for the diagnosis and treatment of insomnia. J. Sleep Res. 2017, 26, 675–700. [Google Scholar] [CrossRef]
- American Academy of Sleep Medicine. Sleep-related breathing disorders in adults: Recommendations for syndrome definition and measurement techniques in clinical research. The Report of an American Academy of Sleep Medicine Task Force. Sleep 1999, 22, 667–689. [Google Scholar] [CrossRef]
- Allen, R.P.; Picchietti, D.; Hening, W.A.; Trenkwalder, C.; Walters, A.S.; Montplaisi, J.; Restless Legs Syndrome, D.; Epidemiology workshop at the National Institutes of Health; International Restless Legs Syndrome Study Group. Restless legs syndrome: Diagnostic criteria, special considerations, and epidemiology. A report from the restless legs syndrome diagnosis and epidemiology workshop at the National Institutes of Health. Sleep Med. 2003, 4, 101–119. [Google Scholar] [CrossRef]
- Lammers, G.J.; Bassetti, C.L.A.; Dolenc-Groselj, L.; Jennum, P.J.; Kallweit, U.; Khatami, R.; Lecendreux, M.; Manconi, M.; Mayer, G.; Partinen, M.; et al. Diagnosis of central disorders of hypersomnolence: A reappraisal by European experts. Sleep Med. Rev. 2020, 52, 101306. [Google Scholar] [CrossRef] [PubMed]
- Castelnovo, A.; Lopez, R.; Proserpio, P.; Nobili, L.; Dauvilliers, Y. NREM sleep parasomnias as disorders of sleep-state dissociation. Nat. Rev. Neurol. 2018, 14, 470–481. [Google Scholar] [CrossRef] [PubMed]
- Schenck, C.H.; Montplaisir, J.Y.; Frauscher, B.; Hogl, B.; Gagnon, J.F.; Postuma, R.; Sonka, K.; Jennum, P.; Partinen, M.; Arnulf, I.; et al. Rapid eye movement sleep behavior disorder: Devising controlled active treatment studies for symptomatic and neuroprotective therapy—A consensus statement from the International Rapid Eye Movement Sleep Behavior Disorder Study Group. Sleep Med. 2013, 14, 795–806. [Google Scholar] [CrossRef]
- Lobbezoo, F.; Ahlberg, J.; Raphael, K.G.; Wetselaar, P.; Glaros, A.G.; Kato, T.; Santiago, V.; Winocur, E.; De Laat, A.; De Leeuw, R.; et al. International consensus on the assessment of bruxism: Report of a work in progress. J. Oral Rehabil. 2018, 45, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Gauld, C.; Lopez, R.; Philip, P.; Taillard, J.; Morin, C.M.; Geoffroy, P.A.; Micoulaud-Franchi, J.A. A Systematic Review of Sleep-Wake Disorder Diagnostic Criteria Reliability Studies. Biomedicines 2022, 10, 1616. [Google Scholar] [CrossRef]
- Pevernagie, D.; Bauters, F.A.; Hertegonne, K. The Role of Patient-Reported Outcomes in Sleep Measurements. Sleep Med. Clin. 2021, 16, 595–606. [Google Scholar] [CrossRef]
- Klingman, K.J.; Jungquist, C.R.; Perlis, M.L. Questionnaires that screen for multiple sleep disorders. Sleep Med. Rev. 2017, 32, 37–44. [Google Scholar] [CrossRef]
- Fried, E.I. The 52 symptoms of major depression: Lack of content overlap among seven common depression scales. J. Affect. Disord. 2017, 208, 191–197. [Google Scholar] [CrossRef]
- Charvet, C.; Boutron, I.; Morvan, Y.; Le Berre, C.; Touboul, S.; Gaillard, R.; Fried, E.; Chevance, A. How to measure mental pain: A systematic review assessing measures of mental pain. Evid. Based Ment. Health 2022, 25, e4. [Google Scholar] [CrossRef] [PubMed]
- Chrobak, A.A.; Siwek, M.; Dudek, D.; Rybakowski, J.K. Content overlap analysis of 64 (hypo)mania symptoms among seven common rating scales. Int. J. Methods Psychiatr. Res. 2018, 27, e1737. [Google Scholar] [CrossRef] [PubMed]
- Fried, E.I.; Flake, J.K.; Robinaugh, D.J. Revisiting the theoretical and methodological foundations of depression measurement. Nat. Rev. Psychol. 2022, 1, 358–368. [Google Scholar] [CrossRef]
- Visontay, R.; Sunderland, M.; Grisham, J.; Slade, T. Content overlap between youth OCD scales: Heterogeneity among symptoms probed and implications. J. Obs.-Compuls. Relat. Disord. 2019, 21, 6–12. [Google Scholar] [CrossRef]
- Wall, A.; Lee, E. What do Anxiety Scales Really Measure? An Item Content Analysis of Self-Report Measures of Anxiety. J. Psychopathol. Behav. Assess. 2022, 44, 1148–1157. [Google Scholar] [CrossRef]
- Karstoft, K.I.; Armour, C. What we talk about when we talk about trauma: Content overlap and heterogeneity in the assessment of trauma exposure. J. Trauma. Stress 2022. online ahead of print. [Google Scholar] [CrossRef]
- Chrobak, A.A.; Krupa, A.; Dudek, D.; Siwek, M. How soft are neurological soft signs? Content overlap analysis of 71 symptoms among seven most commonly used neurological soft signs scales. J. Psychiatr. Res. 2021, 138, 404–412. [Google Scholar] [CrossRef]
- Borsboom, D.; Cramer, A.O.; Schmittmann, V.D.; Epskamp, S.; Waldorp, L.J. The small world of psychopathology. PLoS ONE 2011, 6, e27407. [Google Scholar] [CrossRef]
- Arroll, B.; Fernando, A., 3rd; Falloon, K.; Warman, G.; Goodyear-Smith, F. Development, validation (diagnostic accuracy) and audit of the Auckland Sleep Questionnaire: A new tool for diagnosing causes of sleep disorders in primary care. J. Prim. Health Care 2011, 3, 107–113. [Google Scholar] [CrossRef]
- Bailes, S.; Baltzan, M.; Rizzo, D.; Fichten, C.S.; Amsel, R.; Libman, E. A diagnostic symptom profile for sleep disorder in primary care patients. J. Psychosom. Res. 2008, 64, 427–433. [Google Scholar] [CrossRef]
- Douglass, A.B.; Bornstein, R.; Nino-Murcia, G.; Keenan, S.; Miles, L.; Zarcone, V.P., Jr.; Guilleminault, C.; Dement, W.C. The Sleep Disorders Questionnaire. I: Creation and multivariate structure of SDQ. Sleep 1994, 17, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Kerkhof, G.A.; Geuke, M.E.; Brouwer, A.; Rijsman, R.M.; Schimsheimer, R.J.; Van Kasteel, V. Holland Sleep Disorders Questionnaire: A new sleep disorders questionnaire based on the International Classification of Sleep Disorders-2. J. Sleep Res. 2013, 22, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Koffel, E.; Watson, D. Development and initial validation of the Iowa Sleep Disturbances Inventory. Assessment 2010, 17, 423–439. [Google Scholar] [CrossRef] [PubMed]
- Roth, T.; Zammit, G.; Kushida, C.; Doghramji, K.; Mathias, S.D.; Wong, J.M.; Buysse, D.J. A new questionnaire to detect sleep disorders. Sleep Med. 2002, 3, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Spoormaker, V.I.; Verbeek, I.; van den Bout, J.; Klip, E.C. Initial validation of the SLEEP-50 questionnaire. Behav. Sleep Med. 2005, 3, 227–246. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Bobes, J.; González, M.P.; Vallejo, J.; Sáiz, J.; Gibert, J.; Ayuso, J.L.; Rico, F. Oviedo Sleep Questionnaire (OSQ): A new semistructured interview for sleep disorders. Eur. Neuropsychopharmacol. 1998, 8, S162. [Google Scholar] [CrossRef]
- Partinen, M.; Gislason, T. Basic Nordic Sleep Questionnaire (BNSQ): A quantitated measure of subjective sleep complaints. J. Sleep Res. 1995, 4, 150–155. [Google Scholar] [CrossRef]
- Klingman, K.J.; Jungquist, C.R.; Perlis, M.L. Introducing the sleep disorders symptom checklist-25: A primary care friendly and comprehensive screener for sleep disorders. Sleep Med. Res. 2017, 8, 17–25. [Google Scholar] [CrossRef]
- Srole, L.; Langner, T.S.; Michael, S.T.; Opler, M.K.; Rennie, T.A.C. Mental Health in the Metropolis: The Midtown Manhattan Study; McGraw-Hill: New York, NY, USA, 1962. [Google Scholar]
- Coleman, J. Social Capital in the Creation of Human Capital. Am. J. Sociol. 1988, 94, S95–S120. [Google Scholar] [CrossRef]
- March, D.; Oppenheimer, G.M. Social disorder and diagnostic order: The US Mental Hygiene Movement, the Midtown Manhattan study and the development of psychiatric epidemiology in the 20th century. Int. J. Epidemiol. 2014, 43 (Suppl. S1), i29–i42. [Google Scholar] [CrossRef] [PubMed]
- Fried, E.I.; Nesse, R.M. Depression is not a consistent syndrome: An investigation of unique symptom patterns in the STAR*D study. J. Affect. Disord. 2015, 172, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Evans, J. Straightforward Statistics for the Behavioral Sciences; Brooks/Cole: Pacific Grove, CA, USA, 1996. [Google Scholar]
- Python. Package Plotly. Available online: https://plotly.com/python/ (accessed on 22 December 2022).
- Association of Sleep Disorders Centers; Association for the Psychophysiological Study of Sleep. Diagnostic classification of sleep and arousal disorders. Sleep 1979, 2, 1–154. [Google Scholar]
- Lavie, P. Who was the first to use the term Pickwickian in connection with sleepy patients? History of sleep apnoea syndrome. Sleep Med. Rev. 2008, 12, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Mollayeva, T.; Thurairajah, P.; Burton, K.; Mollayeva, S.; Shapiro, C.M.; Colantonio, A. The Pittsburgh sleep quality index as a screening tool for sleep dysfunction in clinical and non-clinical samples: A systematic review and meta-analysis. Sleep Med. Rev. 2016, 25, 52–73. [Google Scholar] [CrossRef] [PubMed]

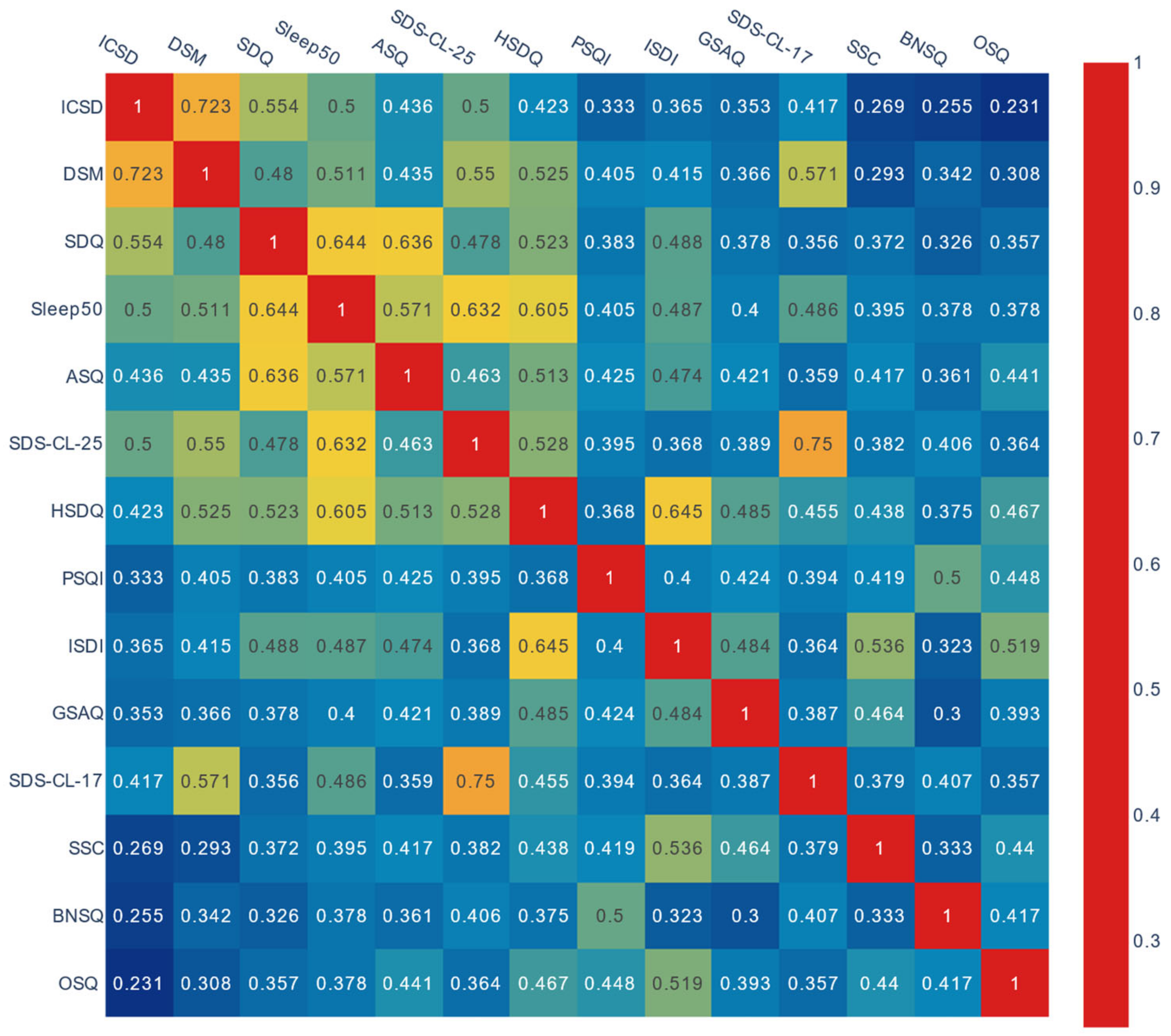
| Average Jaccard Index | Items | Specific Symptoms | Compound Symptoms | Total Number of Symptoms | |
|---|---|---|---|---|---|
| SDQ | 0.449 | 175 | 32 | 8 | 40 |
| Sleep50 | 0.498 | 50 | 31 | 3 | 34 |
| ASQ | 0.462 | 84 | 23 | 9 | 32 |
| SDS-CL-25 | 0.469 | 25 | 20 | 8 | 28 |
| HSDQ | 0.491 | 32 | 15 | 12 | 27 |
| PSQI | 0.415 | 24 | 13 | 12 | 25 |
| ISDI | 0.462 | 86 | 16 | 8 | 23 |
| GSAQ | 0.411 | 11 | 4 | 18 | 22 |
| SDS-CL-17 | 0.427 | 17 | 13 | 8 | 21 |
| SSC | 0.416 | 22 | 17 | 2 | 19 |
| BNSQ | 0.375 | 22 | 11 | 6 | 17 |
| OSQ | 0.416 | 19 | 12 | 5 | 17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gauld, C.; Martin, V.P.; Richaud, A.; Baillieul, S.; Vicente, L.; Perromat, J.-L.; Zreik, I.; Taillard, J.; Geoffroy, P.A.; Lopez, R.; et al. Systematic Item Content and Overlap Analysis of Self-Reported Multiple Sleep Disorder Screening Questionnaires in Adults. J. Clin. Med. 2023, 12, 852. https://doi.org/10.3390/jcm12030852
Gauld C, Martin VP, Richaud A, Baillieul S, Vicente L, Perromat J-L, Zreik I, Taillard J, Geoffroy PA, Lopez R, et al. Systematic Item Content and Overlap Analysis of Self-Reported Multiple Sleep Disorder Screening Questionnaires in Adults. Journal of Clinical Medicine. 2023; 12(3):852. https://doi.org/10.3390/jcm12030852
Chicago/Turabian StyleGauld, Christophe, Vincent P. Martin, Alexandre Richaud, Sébastien Baillieul, Lucie Vicente, Jean-Lorrain Perromat, Issa Zreik, Jacques Taillard, Pierre Alexis Geoffroy, Régis Lopez, and et al. 2023. "Systematic Item Content and Overlap Analysis of Self-Reported Multiple Sleep Disorder Screening Questionnaires in Adults" Journal of Clinical Medicine 12, no. 3: 852. https://doi.org/10.3390/jcm12030852
APA StyleGauld, C., Martin, V. P., Richaud, A., Baillieul, S., Vicente, L., Perromat, J.-L., Zreik, I., Taillard, J., Geoffroy, P. A., Lopez, R., & Micoulaud-Franchi, J.-A. (2023). Systematic Item Content and Overlap Analysis of Self-Reported Multiple Sleep Disorder Screening Questionnaires in Adults. Journal of Clinical Medicine, 12(3), 852. https://doi.org/10.3390/jcm12030852









