Controversial Aspects of Diagnostics and Therapy of Arthritis of the Temporomandibular Joint in Rheumatoid and Juvenile Idiopathic Arthritis—An Analysis of Evidence- and Consensus-Based Recommendations Based on an Interdisciplinary Guideline Project
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Project
2.2. Systematic Literature Search
2.3. Assessment of Evidence
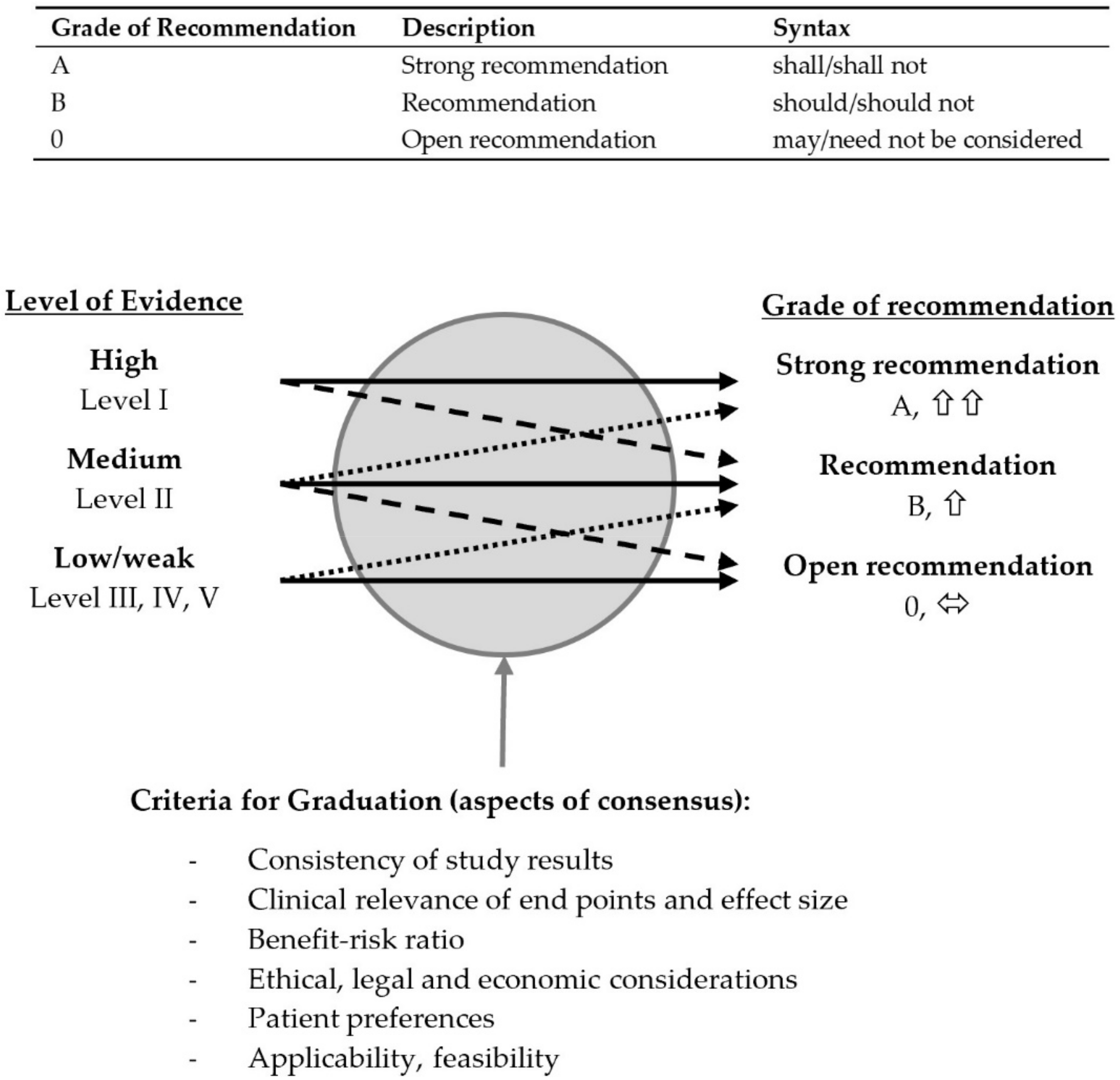
2.4. Wording of Recommendation and Structured Consensus Procedure
2.5. Statistics
3. Results
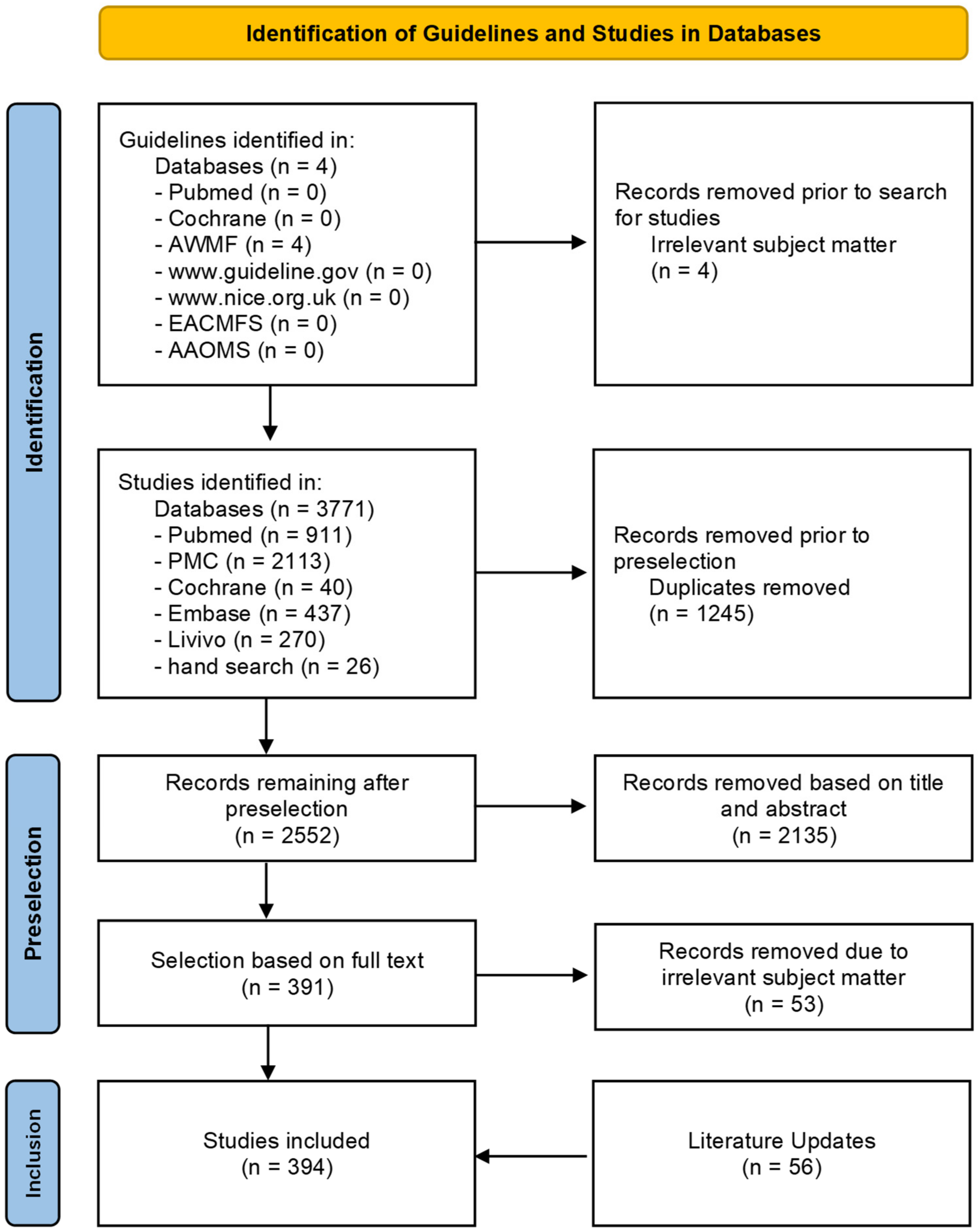
3.1. Systematic Literature Search
3.2. Consensus Process
3.2.1. OMFS Consensus Phase (K1) (Initial Draft Version Consensus)
3.2.2. Interdisciplinary Consensus Phase K2
3.2.3. Statistical Analysis of the Consensus Process
| Proportion of Abstentions | Consensus | ||
|---|---|---|---|
| Consensus OMFS (K1) | Mean | 0.1027 | 0.8562 |
| Standard deviation | 0.23345 | 0.33050 | |
| Interdisciplinary Consensus (K2) | Mean | 0.1288 | 0.9282 |
| Standard deviation | 0.16198 | 0.21870 | |
| Mann–Whitney test—exact signature (2-tailed) | <0.001 | 0.227 | |
| No Consensus | Consensus | Strong Consensus | |
|---|---|---|---|
| Consensus OMFS (K1) | 9.7% | 3.2% | 87.1% |
| Interdisciplinary Consensus (K2) | 5.2% | 3.9% | 89.2% |
| Total | 7.2% | 3.5% | 89.1% |
| Mann–Whitney test—exact signature (2-tailed) | 0.489 | ||
| Round 1 | Round 2 | |
|---|---|---|
| Consensus OMFS (K1) | 88.5% | 11.5% |
| Interdisciplinary Consensus (K2) | 92.4% | 7.6% |
| Chi-squared test—Fisher’s exact test—exact signature (2-tailed) | 0.559 | |
| Proportion of Abstentions | Consensus | ||
|---|---|---|---|
| JIA | Mean | 0.0903 | 0.9358 |
| Standard deviation | 0.14482 | 0.15857 | |
| RA | Mean | 0.1010 | 0.9343 |
| Standard deviation | 0.14722 | 0.14943 | |
| Mann–Whitney test—exact signature (2-tailed) | 0.684 | 0.735 | |
| No Consensus | Consensus | Strong Consensus | |
|---|---|---|---|
| JIA | 4.2% | 4.2% | 91.7% |
| RA | 0% | 27.3% | 72.7% |
| Total | 2.9% | 11.4% | 85.7% |
| Mann–Whitney test—exact signature (2-tailed) | 0.297 | ||
| Round 1 | Round 2 | |
|---|---|---|
| JIA | 90.2% | 9.8% |
| RA | 86.4% | 13.6% |
| Chi-squared test—Fisher’s exact test—exact signature (2-tailed) | 0.687 | |
3.2.4. Identification of Controversial Areas in the Consensus Process
- Criterion 1: Simple consensus not achieved (agreement ≤ 75%) in at least one round
- Criterion 2: Modification of text required to achieve a higher level of consensus
- -
- from approval by majority (≤75%) to simple consensus (76–95%) or from
- -
- simple consensus to strong consensus (>95%)
- Criterion 3: Proportion of abstentions >25%
- Criterion 4: Persistent dissenting vote despite modification
- Diagnostics: Orthopantomography (OPG) for initial imaging, cone-beam computed tomography (CBCT) compared to CT, sonography for detection of active arthritis, examination of synovial fluids and histopathological surveying using Krenn scores and bone scintigraphy, biopsy of components of the masticatory muscles, electromyography and instrumental recording of movements as supplementary diagnostic methods.
- Therapy: Intraarticular application of corticosteroids (IACI), the use of costochondral grafts (CCG) in JIA, the NSAR concept by Nørholt and colleagues in distraction osteogenesis and LeFort I osteotomy.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Petty, R.E.; Southwood, T.R.; Manners, P.; Baum, J.; Glass, D.N.; Goldenberg, J.; He, X.; Maldonado-Cocco, J.; Orozco-Alcala, J.; Prieur, A.-M.; et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: Second revision, Edmonton, 2001. J. Rheumatol. 2004, 31, 390–392. [Google Scholar]
- O’Connor, R.C.; Fawthrop, F.; Salha, R.; Sidebottom, A.J. Management of the temporomandibular joint in inflammatory arthritis: Involvement of surgical procedures. Eur. J. Rheumatol. 2017, 4, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Reich, R.H.; Lindern, J.J. Funktionelle Kiefergelenkschirurgie. In Mund-Kiefer-Gesichtschirurgie; 4. vollst. überarb. Aufl.; Horch, H.H., Ed.; Urban & Fischer bei Elsevier: München, Germany, 2007; pp. 189–191. ISBN 978-3-437-31383-7. [Google Scholar]
- Stoustrup, P.; Resnick, C.M.; Pedersen, T.K.; Abramowicz, S.; Michelotti, A.; Küseler, A.; Verna, C.; Kellenberger, C.J.; Berit Nordal, E.; Caserta, G.; et al. Standardizing Terminology and Assessment for Orofacial Conditions in Juvenile Idiopathic Arthritis: International, Multidisciplinary Consensus-based Recommendations. J. Rheumatol. 2019, 46, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Küseler, A.; Pedersen, T.K.; Gelineck, J.; Herlin, T. A 2 year followup study of enhanced magnetic resonance imaging and clinical examination of the temporomandibular joint in children with juvenile idiopathic arthritis. J. Rheumatol. 2005, 32, 162–169. [Google Scholar] [PubMed]
- Zwir, L.M.L.F.; Terreri, M.T.R.A.; Sousa, S.A.; Fernandes, A.R.C.; Guimarães, A.S.; Hilário, M.O.E. Are temporomandibular joint signs and symptoms associated with magnetic resonance imaging findings in juvenile idiopathic arthritis patients? A longitudinal study. Clin. Rheumatol. 2015, 34, 2057–2063. [Google Scholar] [CrossRef]
- Pantoja, L.L.Q.; de Toledo, I.P.; Pupo, Y.M.; Porporatti, A.L.; de Luca Canto, G.; Zwir, L.F.; Guerra, E.N.S. Prevalence of degenerative joint disease of the temporomandibular joint: A systematic review. Clin. Oral Investig. 2018, 23, 2475–2488. [Google Scholar] [CrossRef]
- Arabshahi, B.; Cron, R.Q. Temporomandibular joint arthritis in juvenile idiopathic arthritis: The forgotten joint. Curr. Opin. Rheumatol. 2006, 18, 490–495. [Google Scholar] [CrossRef]
- Weiss, P.F.; Arabshahi, B.; Johnson, A.; Bilaniuk, L.T.; Zarnow, D.; Cahill, A.M.; Feudtner, C.; Cron, R.Q. High prevalence of temporomandibular joint arthritis at disease onset in children with juvenile idiopathic arthritis, as detected by magnetic resonance imaging but not by ultrasound. Arthritis Rheum. 2008, 58, 1189–1196. [Google Scholar] [CrossRef]
- Tzaribachev, N.; Weber, D.; Horger, M. Juvenile idiopathische Arthritis: Der leise Zerstörer kindlicher Kiefergelenke. Z. Rheumatol. 2010, 69, 124–129. [Google Scholar] [CrossRef]
- Stoustrup, P.; Twilt, M.; Spiegel, L.; Kristensen, K.D.; Koos, B.; Pedersen, T.K.; Küseler, A.; Cron, R.Q.; Abramowicz, S.; Verna, C.; et al. Clinical Orofacial Examination in Juvenile Idiopathic Arthritis: International Consensus-based Recommendations for Monitoring Patients in Clinical Practice and Research Studies. J. Rheumatol. 2017, 44, 326–333. [Google Scholar] [CrossRef]
- Stoustrup, P.; Herlin, T.; Spiegel, L.; Rahimi, H.; Koos, B.; Pedersen, T.K.; Twilt, M. Standardizing the clinical orofacial examination in Juvenile idiopathic arthritis: An interdisciplinary, consensus-based, short screening protocol. J. Rheumatol. 2019, 47, 1397–1404. [Google Scholar] [CrossRef] [PubMed]
- Tolend, M.A.; Twilt, M.; Cron, R.Q.; Tzaribachev, N.; Guleria, S.; von Kalle, T.; Koos, B.; Miller, E.; Stimec, J.; Vaid, Y.; et al. Toward Establishing a Standardized Magnetic Resonance Imaging Scoring System for Temporomandibular Joints in Juvenile Idiopathic Arthritis. Arthritis Care Res. 2018, 70, 758–767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, E.; Inarejos Clemente, E.J.; Tzaribachev, N.; Guleria, S.; Tolend, M.; Meyers, A.B.; von Kalle, T.; Stimec, J.; Koos, B.; Appenzeller, S.; et al. Imaging of temporomandibular joint abnormalities in juvenile idiopathic arthritis with a focus on developing a magnetic resonance imaging protocol. Pediatr. Radiol. 2018, 48, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Resnick, C.M.; Frid, P.; Norholt, S.E.; Stoustrup, P.; Peacock, Z.S.; Kaban, L.B.; Pedersen, T.K.; Abramowicz, S. An Algorithm for Management of Dentofacial Deformity Resulting from Juvenile Idiopathic Arthritis: Results of a Multinational Consensus Conference. J. Oral Maxillofac. Surg. 2019, 77, 1152.e1–1152.e33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Samson, D.; Schoelles, K.M. Developing the Topic and Structuring Systematic Reviews of Medical Tests: Utility of PICOTS, Analytic Frameworks, Decision Trees, and Other Frameworks.: Chapter 2 of Methods Guide for Medical Test Reviews (AHRQ Publication No. 12-EHC017). Available online: https://effectivehealthcare.ahrq.gov/sites/default/files/pdf/methods-guidance-tests-topics_methods.pdf (accessed on 17 March 2022).
- Phillips, B.; Ball, C.; Sackett, D.; Badenoch, D.; Straus, S.; Haynes, B.; Dawes, M.; Howick, J. Oxford Centre for Evidence-Based Medicine: Levels of Evidence (March 2009). Available online: https://www.cebm.ox.ac.uk/resources/levels-of-evidence/oxford-centre-for-evidence-based-medicine-levels-of-evidence-march-2009 (accessed on 6 November 2021).
- Muche-Borowski, C.; Selbmann, H.K.; Nothacker, M.; Müller, W.; Kopp, I. Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (AWMF)-Ständige Kommission Leitlinien. AWMF-Regelwerk “Leitlinien”. 1. Auflage 2012. Available online: http://www.awmf.org/leitlinien/awmf-regelwerk.html (accessed on 6 November 2021).
- Pawlaczyk-Kamienska, T.; Pawlaczyk-Wróblewska, E.; Borysewicz-Lewicka, M. Early diagnosis of temporomandibular joint arthritis in children with juvenile idiopathic arthritis. A systematic review. Eur. J. Paediatr. Dent. 2020, 21, 219–226. [Google Scholar] [CrossRef]
- Bag, A.K.; Gaddikeri, S.; Singhal, A.; Hardin, S.; Tran, B.D.; Medina, J.A.; Curé, J.K. Imaging of the temporomandibular joint: An update. World J. Radiol. 2014, 6, 567–582. [Google Scholar] [CrossRef]
- Küseler, A.; Pedersen, T.K.; Herlin, T.; Gelineck, J. Contrast enhanced magnetic resonance imaging as a method to diagnose early inflammatory changes in the temporomandibular joint in children with juvenile chronic arthritis. J. Rheumatol. 1998, 25, 1406–1412. [Google Scholar]
- Pedersen, T.K.; Küseler, A.; Gelineck, J.; Herlin, T. A Prospective Study of Magnetic Resonance and Radiographic Imaging in Relation to Symptoms and Clinical Findings of the Temporomandibular Joint in Children with Juvenile Idiopathic Arthritis. J. Rheumatol. 2008, 35, 1668–1675. [Google Scholar]
- Hechler, B.L.; Phero, J.A.; van Mater, H.; Matthews, N.S. Ultrasound versus magnetic resonance imaging of the temporomandibular joint in juvenile idiopathic arthritis: A systematic review. Int. J. Oral Maxillofac. Surg. 2018, 47, 83–89. [Google Scholar] [CrossRef]
- Resnick, C.M.; Vakilian, P.M.; Breen, M.; Zurakowski, D.; Caruso, P.; Henderson, L.; Nigrovic, P.A.; Kaban, L.B.; Peacock, Z.S. Quantifying Temporomandibular Joint Synovitis in Children with Juvenile Idiopathic Arthritis. Arthritis Care Res. 2016, 68, 1795–1802. [Google Scholar] [CrossRef] [PubMed]
- Larheim, T.A.; Abrahamsson, A.-K.; Kristensen, M.; Arvidsson, L.Z. Temporomandibular Joint Diagnostics Using CBCT. Available online: https://www.birpublications.org/doi/pdf/10.1259/dmfr.20140235 (accessed on 24 May 2018).
- Zain-Alabdeen, E.H.; Alsadhan, R.I. A comparative study of accuracy of detection of surface osseous changes in the temporomandibular joint using multidetector CT and cone beam CT. Dentomaxillofac. Radiol. 2012, 41, 185–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barghan, S.; Tetradis, S.; Mallya, S. Application of cone beam computed tomography for assessment of the temporomandibular joints. Aust. Dent. J. 2012, 57, 109–118. [Google Scholar] [CrossRef]
- Zhang, Z.-L.; Shi, X.-Q.; Ma, X.; Li, G. Detection Accuracy of Condylar Defects in Cone Beam CT Images Scanned with Different Resolutions and Units. Available online: https://www.birpublications.org/doi/pdf/10.1259/dmfr.20130414 (accessed on 26 May 2018).
- Okano, T.; Harata, Y.; Sugihara, Y.; Sakaino, R.; Tsuchida, R.; Iwai, K.; Seki, K.; Araki, K. Absorbed and effective doses from cone beam volumetric imaging for implant planning. Dentomaxillofac. Radiol. 2009, 38, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Kadesjö, N.; Benchimol, D.; Falahat, B.; Näsström, K.; Shi, X.-Q. Evaluation of the Effective Dose of Cone Beam CT and Multislice CT for Temporomandibular Joint Examinations at Optimized Exposure Levels. Available online: https://www.birpublications.org/doi/pdf/10.1259/dmfr.20150041 (accessed on 24 May 2018).
- Wiesener, T. GOÄ-Ratgeber: Digitale Volumentomographie (DVT). Dtsch. Aerzteblatt Int. 2014, 111, A-415. [Google Scholar]
- Kalaykova, S.I.; Klitsie, A.T.; Visscher, C.M.; Naeije, M.; Lobbezoo, F. A Retrospective Study on Possible Predictive Factors for Long-term Temporomandibular Joint Degeneration and Impaired Mobility in Juvenile Arthritis Patients. J. Oral Facial Pain Headache 2017, 31, 165–171. [Google Scholar] [CrossRef] [Green Version]
- Cedströmer, A.-L.; Andlin-Sobocki, A.; Berntson, L.; Hedenberg-Magnusson, B.; Dahlström, L. Temporomandibular Signs, Symptoms, Joint Alterations and Disease Activity in Juvenile Idiopathic Arthritis—An Observational Study. Available online: https://ped-rheum.biomedcentral.com/articles/10.1186/1546-0096-11-37 (accessed on 24 May 2018).
- Poveda-Roda, R.; Bagan, J.; Carbonell, E.; Margaix, M. Diagnostic validity (sensitivity and specificity) of panoramic X-rays in osteoarthrosis of the temporomandibular joint. Cranio 2015, 33, 189–194. [Google Scholar] [CrossRef]
- Im, Y.-G.; Lee, J.-S.; Park, J.-I.; Lim, H.-S.; Kim, B.-G.; Kim, J.-H. Diagnostic accuracy and reliability of panoramic temporomandibular joint (TMJ) radiography to detect bony lesions in patients with TMJ osteoarthritis. J. Dent. Sci. 2018, 13, 396–404. [Google Scholar] [CrossRef]
- Hussain, A.M.; Packota, G.; Major, P.W.; Flores-Mir, C. Role of different imaging modalities in assessment of temporomandibular joint erosions and osteophytes: A systematic review. Dentomaxillofac. Radiol. 2008, 37, 63–71. [Google Scholar] [CrossRef]
- Mupparapu, M.; Oak, S.; Chang, Y.-C.; Alavi, A. Conventional and functional imaging in the evaluation of temporomandibular joint rheumatoid arthritis: A systematic review. Quintessence Int. 2019, 50, 742–753. [Google Scholar] [CrossRef]
- Alstergren, P.; Appelgren, A.; Appelgren, B.; Kopp, S.; Lundeberg, T.; Theodorsson, E. Determination of temporomandibular joint fluid concentrations using vitamin B12 as an internal standard. Eur. J. Oral Sci. 1995, 103, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Alstergren, P.; Kopp, S.; Theodorsson, E. Synovial fluid sampling from the temporomandibular joint: Sample quality criteria and levels of interleukin-1 ß and serotonin. Acta Odontol. Scand. 1999, 57, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, K.D.; Stoustrup, P.; Alstergren, P.; Küseler, A.; Herlin, T.; Pedersen, T.K. Signs and symptoms after temporomandibular joint washing and cannula placement assessed by cone beam computerized tomography. Acta Odontol. Scand. 2015, 73, 454–460. [Google Scholar] [CrossRef]
- Alstergren, P.; Appelgren, A.; Appelgren, B.; Kopp, S.; Nordahl, S.; Theodorsson, E. Measurement of joint aspirate dilution by a spectrophotometer capillary tube system. Scand. J. Clin. Lab. Investig. 1996, 56, 415–420. [Google Scholar] [CrossRef]
- Alstergren, P.; Pigg, M.; Kopp, S. Clinical diagnosis of temporomandibular joint arthritis. J. Oral Rehabil. 2018, 45, 269–281. [Google Scholar] [CrossRef]
- Krenn, V.; Morawietz, L.; Burmester, G.-R.; Kinne, R.W.; Mueller-Ladner, U.; Muller, B.; Haupl, T. Synovitis score: Discrimination between chronic low-grade and high-grade synovitis. Histopathology 2006, 49, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Najm, A.; Le Goff, B.; Venet, G.; Garraud, T.; Amiaud, J.; Biha, N.; Charrier, C.; Touchais, S.; Crenn, V.; Blanchard, F.; et al. IMSYC immunologic synovitis score: A new score for synovial membrane characterization in inflammatory and non-inflammatory arthritis. Jt. Bone Spine 2019, 86, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Krenn, V.; Perino, G.; Rüther, W.; Krenn, V.T.; Huber, M.; Hügle, T.; Najm, A.; Müller, S.; Boettner, F.; Pessler, F.; et al. 15 years of the histopathological synovitis score, further development and review: A diagnostic score for rheumatology and orthopaedics. Pathol. Res. Pract. 2017, 213, 874–881. [Google Scholar] [CrossRef]
- Kopp, S.; Akerman, S.; Nilner, M. Short-term effects of intra-articular sodium hyaluronate, glucocorticoid, and saline injections on rheumatoid arthritis of the temporomandibular joint. J. Craniomandib. Disord. 1991, 5, 231–238. [Google Scholar]
- Alstergren, P.; Appelgren, A.; Appelgren, B.; Kopp, S.; Lundeberg, T.; Theodorsson, E. The effect on joint fluid concentration of neuropeptide Y by intraarticular injection of glucocorticoid in temporomandibular joint arthritis. Acta Odontol. Scand. 1996, 54, 1–7. [Google Scholar] [CrossRef]
- Antonarakis, G.S.; Blanc, A.; Courvoisier, D.S.; Scolozzi, P. Effect of intra-articular corticosteroid injections on pain and mouth opening in juvenile idiopathic arthritis with temporomandibular involvement: A systematic review and meta-analysis. J. Craniomaxillofac. Surg. 2020, 48, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Poswillo, D. Experimental investigation of the effects of intra-articular hydrocortisone and high condylectomy on the mandibular condyle. Oral Surg. Oral Med. Oral Pathol. 1970, 30, 161–173. [Google Scholar] [CrossRef]
- Aggarwal, S.; Kumar, A. A cortisone-wrecked and bony ankylosed temporomandibular joint. Plast. Reconstr. Surg. 1989, 83, 1084–1085. [Google Scholar] [CrossRef]
- Haddad, I.K. Temporomandibular joint osteoarthrosis. Histopathological study of the effects of intra-articular injection of triamcinolone acetonide. Saudi Med. J. 2000, 21, 675–679. [Google Scholar] [PubMed]
- Wernecke, C.; Braun, H.J.; Dragoo, J.L. The Effect of Intra-Articular Corticosteroids on Articular Cartilage: A Systematic Review. Available online: http://journals.sagepub.com/doi/pdf/10.1177/2325967115581163 (accessed on 26 May 2018).
- Fouda, A.A. Association between Intra-Articular Corticosteroid Injection and Temporomandibular Joint Structure Changes. Int. Arch. Oral Maxillofac. Surg. 2018, 2, 1–6. [Google Scholar] [CrossRef]
- Lochbühler, N.; Saurenmann, R.K.; Müller, L.; Kellenberger, C.J. Magnetic Resonance Imaging Assessment of Temporomandibular Joint Involvement and Mandibular Growth Following Corticosteroid Injection in Juvenile Idiopathic Arthritis. J. Rheumatol. 2015, 42, 1514–1522. [Google Scholar] [CrossRef] [PubMed]
- Ringold, S.; Thapa, M.; Shaw, E.A.; Wallace, C.A. Heterotopic ossification of the temporomandibular joint in juvenile idiopathic arthritis. J. Rheumatol. 2011, 38, 1423–1428. [Google Scholar] [CrossRef]
- Stoll, M.L.; Amin, D.; Powell, K.K.; Poholek, C.H.; Strait, R.H.; Aban, I.; Beukelman, T.; Young, D.W.; Cron, R.Q.; Waite, P.D. Risk Factors for Intraarticular Heterotopic Bone Formation in the Temporomandibular Joint in Juvenile Idiopathic Arthritis. Available online: http://www.jrheum.org/content/45/9/1301 (accessed on 1 November 2018).
- Silbermann, M.; Toister, Z.; Lewinson, D. Corticosteroid-induced enhanced mineralization in neonatal condylar cartilage. Clin. Orthop. Relat. Res. 1977, 129, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Silbermann, M.; Maor, G. Mandibular growth retardation in corticosteroid-treated juvenile mice. Anat. Rec. 1979, 194, 355–367. [Google Scholar] [CrossRef]
- Silbermann, M.; Weiss, A.; Raz, E. Retardative effects of a corticosteroid hormone upon chondrocyte growth in the mandibular condyle of neonatal mice. J. Craniofac. Genet. Dev. Biol. 1981, 1, 109–122. [Google Scholar]
- Resnick, C.M.; Pedersen, T.K.; Abramowicz, S.; Twilt, M.; Stoustrup, P.B. Time to Reconsider Management of the Temporomandibular Joint in Juvenile Idiopathic Arthritis. J. Oral Maxillofac. Surg. 2018, 76, 1145–1146. [Google Scholar] [CrossRef]
- Holwegner, C.; Reinhardt, A.L.; Schmid, M.J.; Marx, D.B.; Reinhardt, R.A. Impact of local steroid or statin treatment of experimental temporomandibular joint arthritis on bone growth in young rats. Am. J. Orthod. Dentofacial Orthop. 2015, 147, 80–88. [Google Scholar] [CrossRef] [PubMed]
- George, M.D.; Owen, C.M.; Reinhardt, A.L.; Giannini, P.J.; Marx, D.B.; Reinhardt, R.A. Effect of simvastatin injections on temporomandibular joint inflammation in growing rats. J. Oral Maxillofac. Surg. 2013, 71, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Zotti, F.; Albanese, M.; Rodella, L.F.; Nocini, P.F. Platelet-Rich Plasma in Treatment of Temporomandibular Joint Dysfunctions: Narrative Review. Int. J. Mol. Sci. 2019, 20, 277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naujokat, H.; Sengebusch, A.; Loger, K.; Möller, B.; Açil, Y.; Wiltfang, J. Therapy of antigen-induced arthritis of the temporomandibular joint via platelet-rich plasma injections in domestic pigs. J. Craniomaxillofac. Surg. 2021, 49, 726–731. [Google Scholar] [CrossRef]
- Sidebottom, A.J.; Gruber, E. One-year prospective outcome analysis and complications following total replacement of the temporomandibular joint with the TMJ Concepts system. Br. J. Oral Maxillofac. Surg. 2013, 51, 620–624. [Google Scholar] [CrossRef]
- Wolford, L.M.; Cottrell, D.A.; Henry, C. Sternoclavicular grafts for temporomandibular joint reconstruction. J. Oral Maxillofac. Surg. 1994, 52, 119–128. [Google Scholar] [CrossRef]
- Mehra, P.; Wolford, L.M.; Baran, S.; Cassano, D.S. Single-stage comprehensive surgical treatment of the rheumatoid arthritis temporomandibular joint patient. J. Oral Maxillofac. Surg. 2009, 67, 1859–1872. [Google Scholar] [CrossRef] [PubMed]
- Neff, A.; Ahlers, O.; Eger, T.; Feurer, I.; Giannakopoulos, N.; Hell, B.; Kleinheinz, J.; Knaup, I.; Kolk, A.; Ottl, P.; et al. S3-Leitlinie: Totaler Alloplastischer Kiefergelenkersatz: AWMF-Register-Nr. 007-106 (Langversion). Available online: https://www.awmf.org/uploads/tx_szleitlinien/007-106l_S3_Totaler_alloplastischer_Kiefergelenkersatz_2020-04.pdf (accessed on 6 November 2021).
- Felix, V.B.; Cabral, D.R.G.; Almeida, A.B.W.; de Soares, E.D.; de Moraes Fernandes, K.J. Ankylosis of the Temporomandibular Joint and Reconstruction with a Costochondral Graft in a Patient with Juvenile Idiopathic Arthritis. J. Craniofac. Surg. 2017, 28, 203–206. [Google Scholar] [CrossRef]
- Stringer, D.E.; Gilbert, D.H.; Herford, A.S.; Boyne, P.J. A method of treating the patient with postpubescent juvenile rheumatoid arthritis. J. Oral Maxillofac. Surg. 2007, 65, 1998–2004. [Google Scholar] [CrossRef]
- Svensson, B.; Feldmann, G.; Rindler, A. Early surgical-orthodontic treatment of mandibular hypoplasia in juvenile chronic arthritis. J. Craniomaxillofac. Surg. 1993, 21, 67–75. [Google Scholar] [CrossRef]
- Svensson, B.; Adell, R. Costochondral grafts to replace mandibular condyles in juvenile chronic arthritis patients: Long-term effects on facial growth. J. Craniomaxillofac. Surg. 1998, 26, 275–285. [Google Scholar] [CrossRef]
- Balaji, S.M.; Balaji, P. Overgrowth of costochondral graft in temporomandibular joint ankylosis reconstruction: A retrospective study. Indian J. Dent. Res. 2017, 28, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Peltomäki, T.; Vähätalo, K.; Rönning, O. The effect of a unilateral costochondral graft on the growth of the marmoset mandible. J. Oral Maxillofac. Surg. 2002, 60, 1307–1314. [Google Scholar] [CrossRef]
- Peltomäki, T. Growth of a costochondral graft in the rat temporomandibular joint. J. Oral Maxillofac. Surg. 1992, 50, 851–858. [Google Scholar] [CrossRef]
- Peltomäki, T.; Rönning, O. Interrelationship between size and tissue-separating potential of costochondral transplants. Eur. J. Orthod. 1991, 13, 459–465. [Google Scholar] [CrossRef]
- Peltomäki, T. Histologic structure of human costochondral junction. Plast. Reconstr. Surg. 1994, 94, 585–588. [Google Scholar] [CrossRef]
- Mercuri, L.G.; Swift, J.Q. Considerations for the use of alloplastic temporomandibular joint replacement in the growing patient. J. Oral Maxillofac. Surg. 2009, 67, 1979–1990. [Google Scholar] [CrossRef]
- Cascone, P.; Basile, E.; Angeletti, D.; Vellone, V.; Ramieri, V. TMJ replacement utilizing patient-fitted TMJ TJR devices in a re-ankylosis child. J. Craniomaxillofac. Surg. 2016, 44, 493–499. [Google Scholar] [CrossRef]
- Chanchareonsook, N.; Tideman, H.; Lee, S.; Hollister, S.J.; Flanagan, C.; Jansen, J.A. Mandibular reconstruction with a bioactive-coated cementless Ti6Al4V modular endoprosthesis in Macaca fascicularis. Int. J. Oral Maxillofac. Surg. 2014, 43, 758–768. [Google Scholar] [CrossRef]
- Turpin, D.L. Juvenile rheumatoid arthritis: A 14-year posttreatment evaluation. Angle Orthod. 1989, 59, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Leshem, D.; Tompson, B.; Britto, J.A.; Forrest, C.R.; Phillips, J.H. Orthognathic surgery in juvenile rheumatoid arthritis patients. Plast. Reconstr. Surg. 2006, 117, 1941–1946. [Google Scholar] [CrossRef] [PubMed]
- Nothacker, M.; Blödt, S.; Muche-Borowski, C.; Selbmann, H.K.; Müller, W.; Erstling, S.; Kopp, I. Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (AWMF)-Ständige Kommission Leitlinien. AWMF-Regelwerk “Leitlinien”. 2. Auflage 2020. Available online: http://www.awmf.org/leitlinien/awmf-regelwerk.html (accessed on 27 November 2021).
- Kopp, I.B.; Selbmann, H.-K.; Koller, M. Konsensusfindung in evidenzbasierten Leitlinien–Vom Mythos zur rationalen Strategie. Z. Ärztliche Fortbild. Qual. Im Gesundheitswesen Ger. J. Qual. Health Care 2007, 101, 89–95. [Google Scholar] [CrossRef] [PubMed]
| LoE | Study Type | |
|---|---|---|
| I | a | Meta analysis/systematic review of studies with LoE Ib |
| b | “randomized controlled clinical trial” (RCT) | |
| II | a | Meta analysis/systematic review of studies with LoE IIb |
| b | “controlled clinical trial” (CCT)/comparative prospective cohort study (with control group) | |
| III | a | Meta analysis/systematic review of studies with LoE IIIb |
| b | Retrospective cohort study/case-control study | |
| IV | Non-controlled observational study > 1 patient (e.g., case series), studies other than in vivo studies of human subjects (e.g., animal experiment, cadaver study), consensus paper | |
| V | Case study, non-systematic secondary literature, expert opinion |
| Symbol | Criteria |
|---|---|
| ++ | High quality, overwhelming majority of criteria fulfilled (>75%), no risk or low risk of bias |
| + | Acceptable quality, majority of criteria fulfilled (50–75%), medium risk of bias |
| − | Low quality, majority of criteria not fulfilled (<50%), considerable risk of bias |
| 0 | Unacceptable, study rejected due to insufficient quality |
| Symbol | Criteria |
|---|---|
| k++ | High clinical relevance, overwhelming majority of criteria fulfilled (>75%) |
| k+ | Acceptable clinical relevance, majority of criteria fulfilled (50–75%) |
| k− | Low clinical relevance, majority of criteria not fulfilled (<50%) |
| k0 | Study without clinical relevance, study removed |
| Agreement | AWMF Definition |
|---|---|
| >95% | Strong consensus |
| 76–95% | Consensus |
| 50–75% | Approval by majority |
| <50% | No consensus |
| Item (Final Version) | LoE | Type | Criteria | Comment/Discussion |
|---|---|---|---|---|
| 1. In cases of suspected chronic-rheumatic TMJ arthritis, OPG provides a cost-efficient, low-risk and widely available method for initial medical imaging for detection of advanced bony involvement of the temporomandibular joint. | IIb | St | Criterion 2: K2.1 Consensus (77%), 2 comments → Adaptation of text, Statement added K2.2 Strong consensus (100%) | Insufficient detection ability for subtler bony pathologies was pointed out, and therefore suitability as initial screening tool was questioned. |
| 2. CBCT may be used for evaluation of bony structures of the temporomandibular joint in chronic rheumatic TMJ arthritis as potential dosage-efficient alternative to CT. Radiation exposure will largely depend on choice of device and examination protocol. | IV | R (0) | Criterion 2: K2.1 Consensus (85%), 3 comments, 2/13 dissenting votes → Adaptation of text K2.2 Strong consensus (100%) | Perceived higher dosage efficiency and cost efficiency of CBCT compared to CT questioned. |
| 3. Sonography is currently not considered a suitable means for diagnosis and monitoring of progress of TMJ arthritis with underlying chronic rheumatic disorder due to lack of standardization and limited availability of studies. | IIIb | St | Criterion 2: K2.1 Consensus (91%), abstentions 2/13 (15%), 2 Comments → Adaptation of text, K2.2 Strong consensus (100%), abstentions 2/13 (15%) | Discussion on the role of sonography as a well-established method for the assessment of joint alterations due to arthritis in general vs. its limitations for anatomical reasons, and as not well established, for the TMJ. |
| 4. The superior sensitivity of bone scintigraphy provides for early detection of bone remodelling processes—however, at the price of specificity. For diagnosis and monitoring of progress of chronic rheumatic arthritis of the temporomandibular joint, it is indeed a third-line choice diagnostic device. Its use should be avoided in children and adolescents due to the radiation exposure involved. | V | St | Criteria 1,2 and 3: K1.1 No consensus (50%), 4/6 abstentions (67%), 1 comment → Adaptation of text, K1.2 Strong consensus (100%) | Particular emphasis on the insufficient specificity of the method. |
| 5. Synovialis analysis by means of the Krenn Score may, in individual cases, be considered for the purpose of further assessment and differential diagnosis, independent of an intervention otherwise indicated. | IIIb | R (0) | Criteria 2 and 3: K2.1 Consensus (88%), 1 dissenting vote, abstentions 4/13 (31%), 1 comment → Adaptation of text, expert consensus added K2.2 Strong consensus (100%), abstentions 4/13 (31%) | Discussion on the degree of invasiveness of the procedure, as it is only insufficiently established in oral and maxillofacial surgery. |
| 6. Biopsy of components of the masticatory muscles as additional examination method is not considered a useful approach in the context mentioned above. | V | St | Criteria 1 and 3: K1.1 No consensus (33%), abstentions 3/6 (50%) K2.2 No consensus (100%) 2, abstentions 9/13 (69%) | |
| 7. If clinical indicators point to structural damage in the absence of pain (“silent arthritis of the temporomandibular joint”) and in case of borderline MRI-diagnostic findings, extraction and examination of synovial fluid from the temporomandibular joint may be considered in individual cases in patients >17 years of age. | IIIb | R (0) | Criteria 1, 2 and 3: K2.1 No consensus (66%), abstentions 3/6 (50%), 1 comment → Adaptation of text, K2.2 Strong consensus (100%) | Discussion on the degree of invasiveness of the procedure, as it is only insufficiently established in oral and maxillofacial surgery. |
| 8. Electromyography provides a possible additional diagnostic option. | V | St | Criteria 1 and 3: K1.1 No consensus (0%), abstentions 4/6 (66%) K2.2 No consensus (0%), abstentions 8/13 (62%) | |
| 9. Instrumental recording of the movements of the mandible provides a possible additional diagnostic option. | IIb | St | Criteria 1 and 3: K1.1 No consensus (0%), abstentions 5/6 (83%) K2.2 No consensus (29%), abstentions 6/13 (46%) | |
| 10. Due to the risk of severe complications (chondrotoxicity), no recommendation could be made by the guideline group in favour of IACI in cases where the temporomandibular joint exclusively is affected, or if it is intended as an additional measure during medication therapy, or for bridging during transition between medications in adults. | V | St | Criteria 1, 2 and 3: K1.1 No consensus (50%), abstentions 2/6 (33%), 1 comment → Adaptation of text, 2 statements added K2.1 Consensus (88%), abstentions 5/13 (39%) → Adaptation of text K2.2 Strong consensus (100%), abstentions 5/13 (39%) | Call for limitation to one-time application and avoidance of continuous therapy |
| 11. Due to the risk of severe complications (disturbance of mandibular growth, heterotopic ossification), no recommendation could be made by the guideline group in favour of IACI, if it is intended as an additional measure during medication therapy, or for bridging during transition between medications in JIA. | IV | St | Criteria 1, 2 and 3: K1.1 No consensus (50%), abstentions 2/6 (33%), 1 comment → Adaptation of text, 2 statements and 1 recommendation added K2.1 Consensus (90%), abstentions 3/13 (23%) → Adaptation of text K2.2 Strong consensus (100%), abstentions 3/13 (23%) | Call for limitation to one-time application and avoidance of continuous therapy |
| 12. In cases of loss of function of the temporomandibular joint in adolescents with JIA, otherwise refractory to therapy (e.g., ankylosis), despite sometimes grave side effects, autologous reconstruction of the TMJ may be performed by means of a costochondral graft. | IV | R (0) | Criterion 4: K1.1 Consensus (83%), 1 dissenting vote, 1 comment→ 1 statement added K1.2 Consensus (83%), 1 dissenting vote | Method criticized as no longer state-of-the-art in view of side effects |
| 13. The approach of Nørholt and colleagues, i.e., application of NSAIDs one hour prior to distractor activation and use of an occlusal splint to shift the load from the temporomandibular joint to the teeth, has been approved. | IV | St | Criteria 1 and 3: K1.1 No consensus (75%), abstentions 2/6 (33%) K2.2 No consensus (100%) 2, abstentions 7/13 (54%) | |
| 14. For skeletal deformities in the context of TMJ involvement in JIA, or as a result of JIA, Le Fort I osteotomy for correction of occlusion and repair of an anterior open bite after the end of the growth phase, is a possible option in select cases, provided the underlying disease is inactive/well controlled or adequately managed, as otherwise there is a risk of recurrence. Furthermore, factors such as sufficient posterior airway space (PAS) and the basic dentofacial aspects of orthognathic surgery need to be considered. | IV | St | Criterion 2: K1.1 Consensus (83%), 1 comment → Adaptation of text K1.2 Strong consensus (100%) | Call for limitation to individual cases and added basic prerequisites for the intervention |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmidt, C.; Reich, R.; Koos, B.; Ertel, T.; Ahlers, M.O.; Arbogast, M.; Feurer, I.; Habermann-Krebs, M.; Hilgenfeld, T.; Hirsch, C.; et al. Controversial Aspects of Diagnostics and Therapy of Arthritis of the Temporomandibular Joint in Rheumatoid and Juvenile Idiopathic Arthritis—An Analysis of Evidence- and Consensus-Based Recommendations Based on an Interdisciplinary Guideline Project. J. Clin. Med. 2022, 11, 1761. https://doi.org/10.3390/jcm11071761
Schmidt C, Reich R, Koos B, Ertel T, Ahlers MO, Arbogast M, Feurer I, Habermann-Krebs M, Hilgenfeld T, Hirsch C, et al. Controversial Aspects of Diagnostics and Therapy of Arthritis of the Temporomandibular Joint in Rheumatoid and Juvenile Idiopathic Arthritis—An Analysis of Evidence- and Consensus-Based Recommendations Based on an Interdisciplinary Guideline Project. Journal of Clinical Medicine. 2022; 11(7):1761. https://doi.org/10.3390/jcm11071761
Chicago/Turabian StyleSchmidt, Christopher, Rudolf Reich, Bernd Koos, Taila Ertel, Marcus Oliver Ahlers, Martin Arbogast, Ima Feurer, Mario Habermann-Krebs, Tim Hilgenfeld, Christian Hirsch, and et al. 2022. "Controversial Aspects of Diagnostics and Therapy of Arthritis of the Temporomandibular Joint in Rheumatoid and Juvenile Idiopathic Arthritis—An Analysis of Evidence- and Consensus-Based Recommendations Based on an Interdisciplinary Guideline Project" Journal of Clinical Medicine 11, no. 7: 1761. https://doi.org/10.3390/jcm11071761
APA StyleSchmidt, C., Reich, R., Koos, B., Ertel, T., Ahlers, M. O., Arbogast, M., Feurer, I., Habermann-Krebs, M., Hilgenfeld, T., Hirsch, C., Hügle, B., von Kalle, T., Kleinheinz, J., Kolk, A., Ottl, P., Pautke, C., Riechmann, M., Schön, A., Skroch, L., ... Neff, A. (2022). Controversial Aspects of Diagnostics and Therapy of Arthritis of the Temporomandibular Joint in Rheumatoid and Juvenile Idiopathic Arthritis—An Analysis of Evidence- and Consensus-Based Recommendations Based on an Interdisciplinary Guideline Project. Journal of Clinical Medicine, 11(7), 1761. https://doi.org/10.3390/jcm11071761







