The Use of Non-Invasive Continuous Blood Pressure Measuring (ClearSight®) during Central Neuraxial Anaesthesia for Caesarean Section—A Retrospective Validation Study
Abstract: Background
1. Introduction
2. Materials and Methods
Statistical Analysis
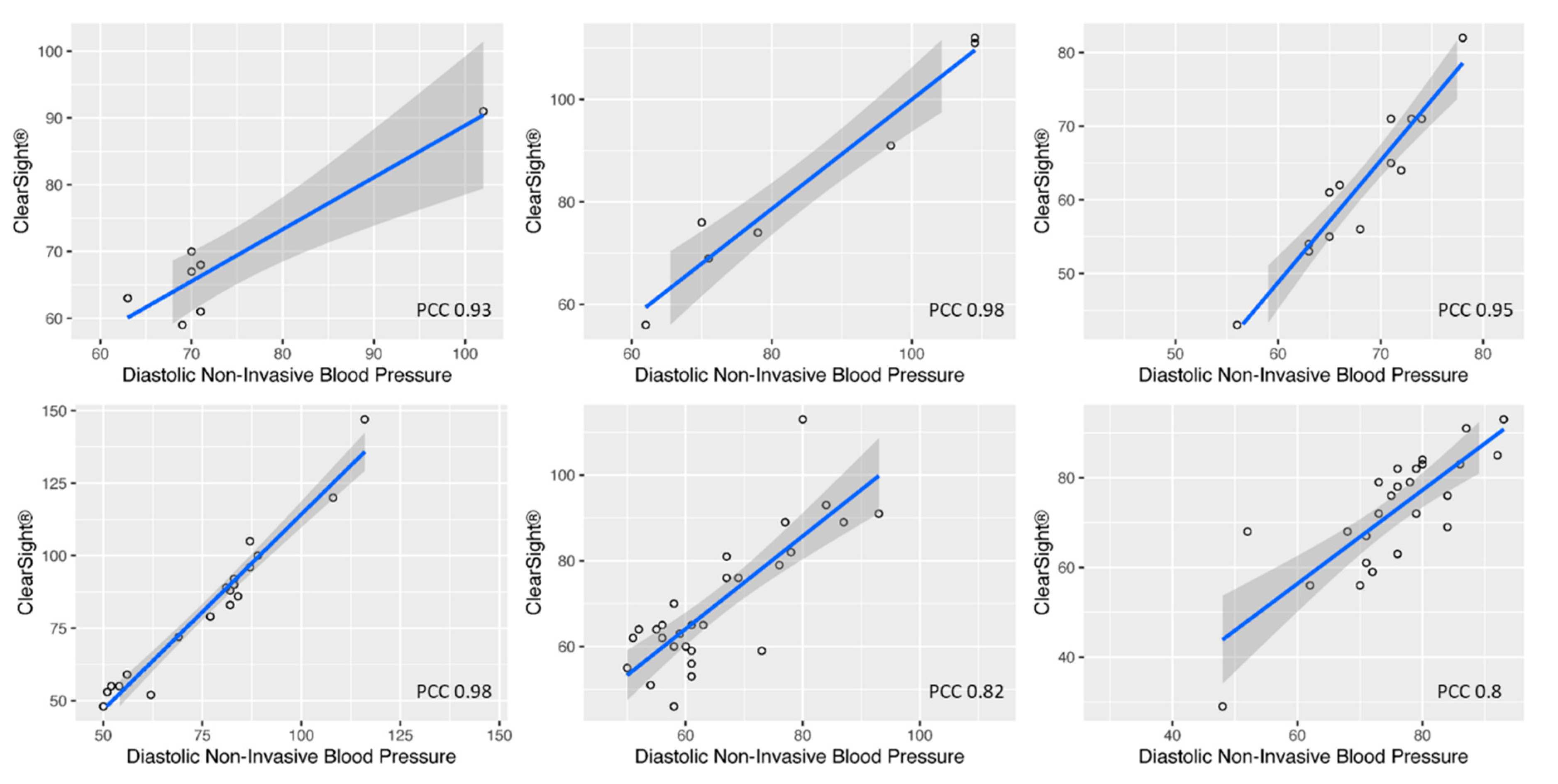
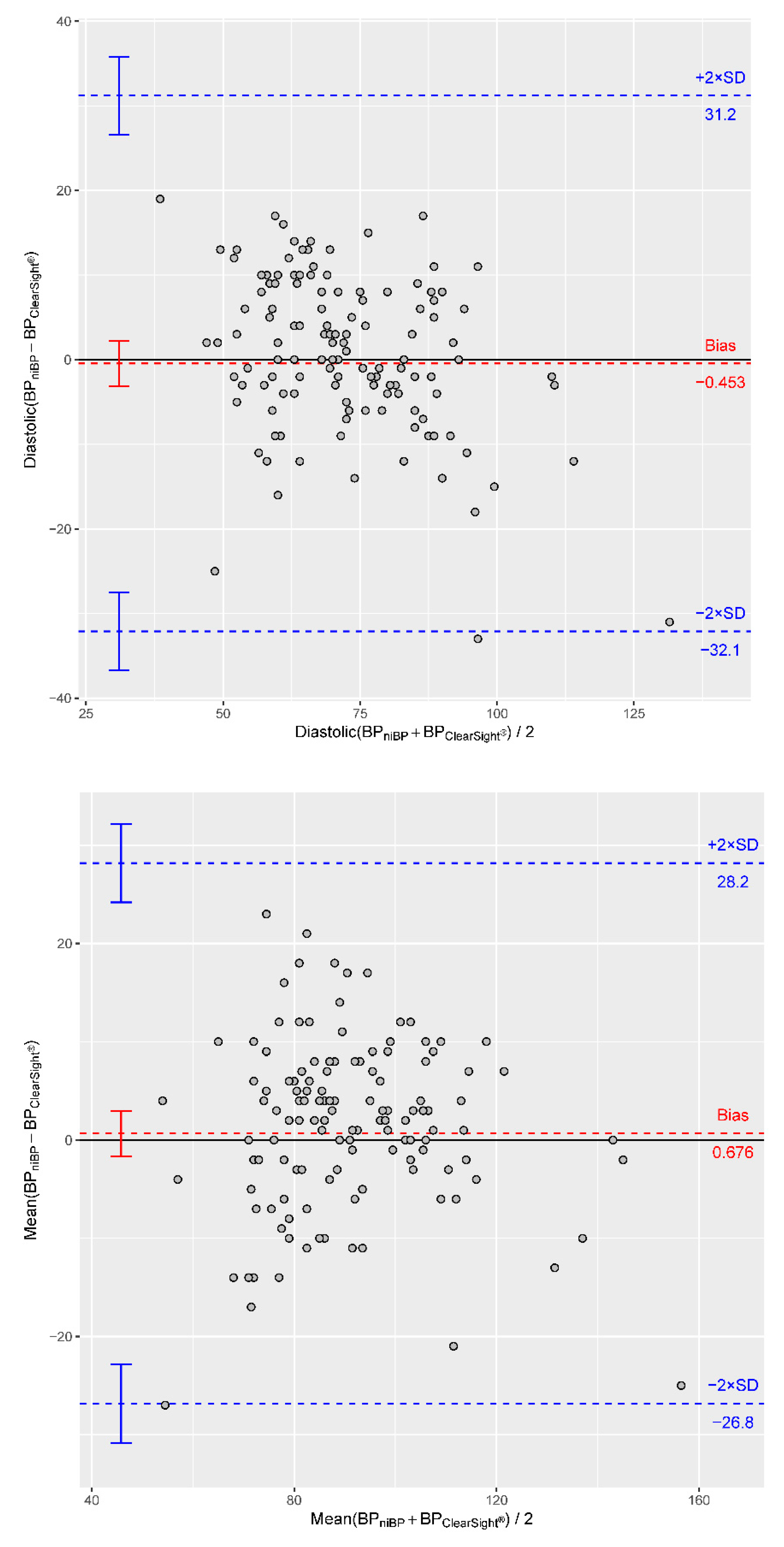
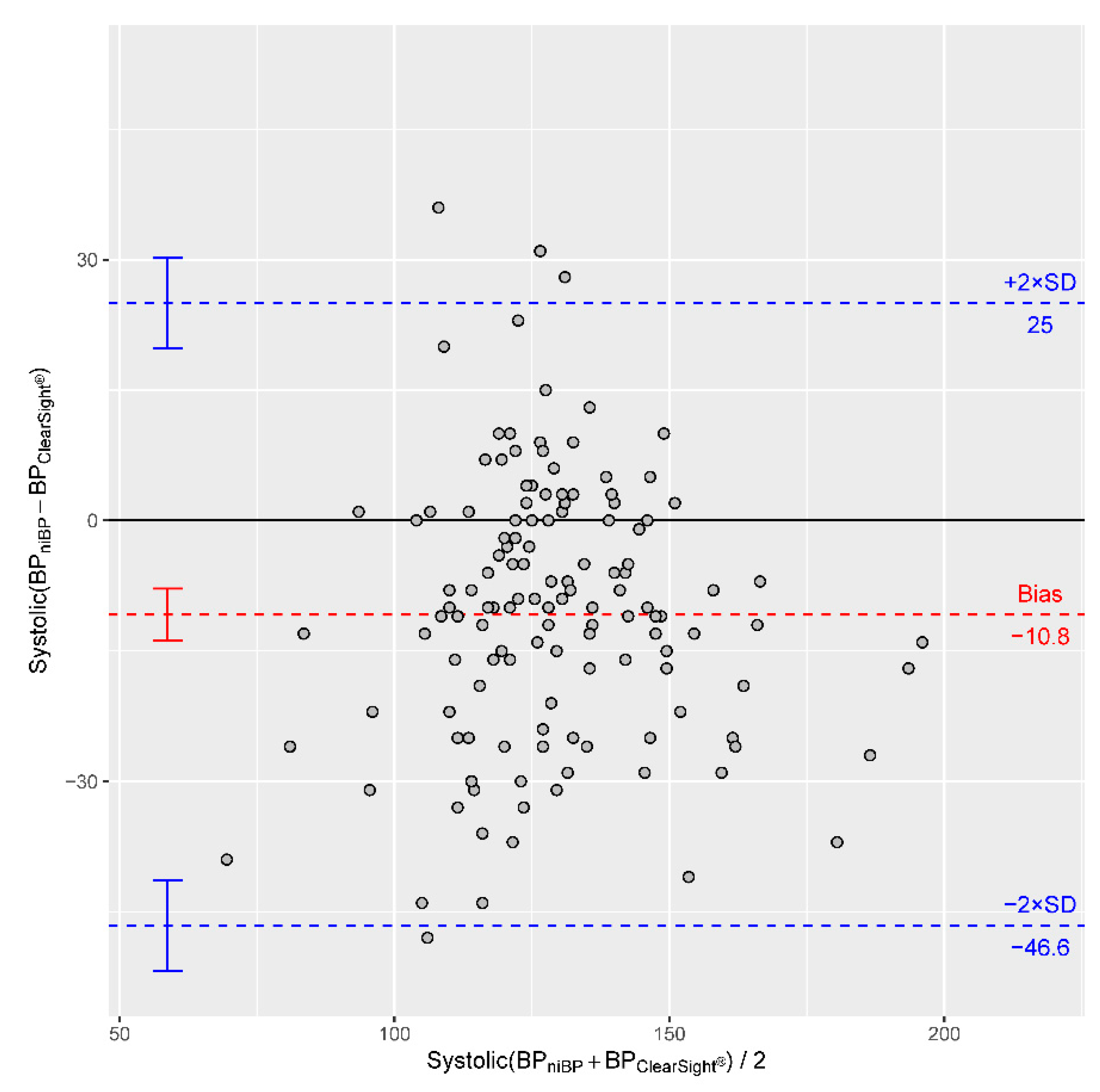
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AAMI | Association for the Advancement of Medical Instrumentation |
| ASA | American Society of Anesthesiologists |
| BMI | Body-Mass Index |
| CI | Confidence Interval |
| CSE | Combined Spinal Epidural |
| CSF | Cerebrospinal Fluid |
| G | Gauge |
| HPI | Hypotension Prediction Index |
| Hz | Hertz |
| ICU | Intensive Care Unit |
| IQR | Interquartile Range |
| L | Lumbal |
| MAP | Mean Arterial Pressure |
| NIBP | Non-Invasive Blood Pressure |
| PCC | Pearson Correlation Coefficient |
| PDMS | Patient Data Management System |
| RRdia | Diastolic Blood Pressure |
| RRmean | Mean Blood Pressure |
| RRsys | Systolic Blood Pressure |
| SD | Standard Deviation |
| SSA | Single-Shot Spinal Anaesthesia |
| TH | Thoracic |
References
- Klöhr, S.; Roth, R.; Hofmann, T.; Rossaint, R.; Heesen, M. Definitions of hypotension after spinal anaesthesia for caesarean section: Literature search and application to parturients. Acta Anaesthesiol. Scand. 2010, 54, 909–921. [Google Scholar] [CrossRef] [PubMed]
- Kranke, P.; Geldner, G.; Kienbaum, P.; Gerbershagen, H.J.; Chappell, D.; Wallenborn, J.; Huljic, S.; Koch, T.; Keller, T.; Weber, S.; et al. Treatment of spinal anaesthesia-induced hypotension with cafedrine/theodrenaline versus ephedrine during caesarean section: Results from HYPOTENS, a national, multicentre, prospective, noninterventional study. Eur. J. Anaesthesiol. 2021, 38, 1067–1076. [Google Scholar] [CrossRef]
- Kranke, P. Die Geburtshilfliche Anästhesie; Springer: Berlin/Heidelberg, Germany, 2018; pp. 79–99. [Google Scholar]
- Langesæter, E.; Gibbs, M.; Dyer, R.A. The role of cardiac output monitoring in obstetric anesthesia. Curr. Opin. Anaesthesiol. 2015, 28, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Langesæter, E.; A Dyer, R. Maternal haemodynamic changes during spinal anaesthesia for caesarean section. Curr. Opin. Anaesthesiol. 2011, 24, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Chooi, C.; Cox, J.J.; Lumb, R.S.; Middleton, P.; Chemali, M.; Emmett, R.S.; Simmons, S.W.; Cyna, A.M. Techniques for preventing hypotension during spinal anaesthesia for caesarean section. Cochrane Database Syst. Rev. 2017, 8, CD002251. [Google Scholar] [CrossRef] [PubMed]
- Massoth, C.; Chappell, D.; Kranke, P.; Wenk, M. Supine hypotensive syndrome of pregnancy: A review of current knowledge. Eur. J. Anaesthesiol. 2021, 39, 236–243. [Google Scholar] [CrossRef]
- Lewis, P.S.; on behalf of the British and Irish Hypertension Society’s Blood Pressure Measurement Working Party. Oscillometric measurement of blood pressure: A simplified explanation. A technical note on behalf of the British and Irish Hypertension Society. J. Hum. Hypertens. 2019, 33, 349–351. [Google Scholar] [CrossRef]
- Ramsey, M. Noninvasive automatic determination of mean arterial pressure. Med Biol. Eng. Comput. 1979, 17, 11–18. [Google Scholar] [CrossRef] [PubMed]
- David, W.; Lin, H.; Andrew, L. Invasive and Concomitant Noninvasive Intraoperative blood pressure monitoring. Anesthesiology 2011, 115, 5. [Google Scholar]
- Bur, A.; Hirschl, M.M.; Herkner, H.; Oschatz, E.; Kofler, J.; Woisetschläger, C.; Laggner, A.N. Accuracy of oscillometric blood pressure measurement according to the relation between cuff size and upper-arm circumference in critically ill patients. Crit. Care Med. 2000, 28, 371–376. [Google Scholar] [CrossRef]
- Maheshwari, K.; Khanna, S.; Bajracharya, G.R.; Makarova, N.; Riter, Q.; Raza, S.; Cywinski, J.B.; Argalious, M.; Kurz, A.; Sessler, D.I. A Randomized Trial of Continuous Noninvasive Blood Pressure Monitoring During Noncardiac Surgery. Anesthesia Analg. 2018, 127, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Jokinen, J.; Adametz, V.; Kredel, M.; Muellenbach, R.M.; Hönig, A.; Wöckel, A.; Dietl, J.; Roewer, N.; Kranke, P. [Spinal anesthesia in high-volume, low-concentration technique for Caesarean sections: Retrospective analysis]. Spinalanästhesie zur Sectio caesarea in “High-volume-low-concentration”-Technik. Der Anaesthesist 2014, 64, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Rogge, D.E.; Nicklas, J.Y.; Schön, G.; Grothe, O.; Haas, S.A.; Reuter, D.A.; Saugel, B. Continuous Noninvasive Arterial Pressure Monitoring in Obese Patients During Bariatric Surgery: An Evaluation of the Vascular Unloading Technique (Clearsight system). Anesthesia Analg. 2019, 128, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Bland, M.; Altman, D. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 327, 307–310. [Google Scholar] [CrossRef]
- Alexander, R. A note on averaging correlations. Bull. Psychon. Soc. 1990, 28, 335–336. [Google Scholar] [CrossRef] [Green Version]
- Wesseling, K.H.; Settels, J.J.; De Wit, B. The Measurement of Continuous Finger Arterial Pressure Noninvasively in Stationary Subjects. In Biological and Psychological Factors in Cardiovascular Disease; Schmidt, T.H., Dembroski, T.M., Blümchen, G., Eds.; Springer: Berlin/Heidelberg, Germany, 1986; pp. 355–375. [Google Scholar] [CrossRef]
- Penáz, J.; Voigt, A.; Teichmann, W. Contribution to the continuous indirect blood pressure measurement. Z Gesamte Inn. Med. 1976, 31, 1030–1033. [Google Scholar]
- Wesseling, K.H.; Jansen, J.R.; Settels, J.J.; Schreuder, J.J. Computation of aortic flow from pressure in humans using a nonlinear, three-element model. J. Appl. Physiol. 1993, 74, 2566–2573. [Google Scholar] [CrossRef]
- Tanioku, T.; Yoshida, A.; Aratani, Y.; Fujii, K.; Kawamata, T. Validation of noninvasive continuous arterial pressure measurement by ClearSight System™ during induction of anesthesia for cardiovascular surgery. BMC Anesthesiol. 2020, 20, 1–7. [Google Scholar] [CrossRef]
- Schumann, R.; Meidert, A.S.; Bonney, I.; Koutentis, C.; Wesselink, W.; Kouz, K.; Saugel, B. Intraoperative Blood Pressure Monitoring in Obese Patients. Anesthesiology 2020, 134, 179–188. [Google Scholar] [CrossRef]
- Juri, T.; Suehiro, K.; Kimura, A.; Mukai, A.; Tanaka, K.; Yamada, T.; Mori, T.; Nishikawa, K. Impact of non-invasive continuous blood pressure monitoring on maternal hypotension during cesarean delivery: A randomized-controlled study. J. Anesthesia 2018, 32, 822–830. [Google Scholar] [CrossRef]
- Frassanito, L.; Giuri, P.P.; Vassalli, F.; Piersanti, A.; Longo, A.; Zanfini, B.A.; Catarci, S.; Fagotti, A.; Scambia, G.; Draisci, G. Hypotension Prediction Index with non-invasive continuous arterial pressure waveforms (ClearSight): Clinical performance in Gynaecologic Oncologic Surgery. Int. J. Clin. Monit. Comput. 2021, 1–8. [Google Scholar] [CrossRef]
- Frassanito, L.; Sonnino, C.; Piersanti, A.; Zanfini, B.A.; Catarci, S.; Giuri, P.P.; Scorzoni, M.; Gonnella, G.L.; Antonelli, M.; Draisci, G. Performance of the Hypotension Prediction Index With Noninvasive Arterial Pressure Waveforms in Awake Cesarean Delivery Patients Under Spinal Anesthesia. Anesthesia Analg. 2022, 134, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Chung, J.; Bae, J.; Cho, Y.J.; Nam, K.; Jeon, Y. Continuous Non-Invasive Arterial Pressure Monitoring (ClearSight System) and Ankle Blood Pressure Measurements as Alternatives to Conventional Arm Blood Pressure. J. Clin. Med. 2020, 9, 3615. [Google Scholar] [CrossRef] [PubMed]
- Noto, A.; Sanfilippo, F.; De Salvo, G.; Crimi, C.; Benedetto, F.; Watson, X.; Cecconi, M.; David, A. Noninvasive continuous arterial pressure monitoring with Clearsight during awake carotid endarterectomy. Eur. J. Anaesthesiol. 2019, 36, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Sakai, Y.; Yasuo, T.M.; Oyama, T.; Murakami, C.; Kakuta, N.; Tanaka, K. Noninvasive continuous blood pressure monitoring by the ClearSight system during robot-assisted laparoscopic radical prostatectomy. J. Med Investig. 2018, 65, 69–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.-W.; Lee, S.; Kim, H.; Kim, Y.-J.; Kim, M.; Choi, J.-H. Accuracy of noninvasive continuous arterial pressure monitoring using ClearSight during one-lung ventilation. Medicine 2021, 100, e25152. [Google Scholar] [CrossRef]
- Yokose, M.; Mihara, T.; Takaya, M.; Yamamoto, T.; Saigusa, Y.; Takaki, S.; Goto, T. The perfusion index measured by the pulse oximeter affects the agreement between ClearSight and the arterial catheter-based blood pressures: A prospective observational study. PLoS ONE 2019, 14, e0219511. [Google Scholar] [CrossRef] [PubMed]
- Duclos, G.; Hili, A.; Resseguier, N.; Kelway, C.; Haddam, M.; Bourgoin, A.; Carcopino, X.; Zieleskiewicz, L.; Leone, M. Clearsight™ use for haemodynamic monitoring during the third trimester of pregnancy—A validation study. Int. J. Obstet. Anesthesia 2018, 36, 85–95. [Google Scholar] [CrossRef]
- Yang, S.-H.; Lin, Y.-S.; Lee, C.-N.; Cheng, Y.-J.; Chen, Y.-H.; Chiu, H.-C.; Wu, C.-Y. Implications of Continuous Noninvasive Finger Cuff Arterial Pressure Device Use during Cesarean Delivery for Goal-Directed Fluid Therapy Preload Optimization: A Randomized Controlled Trial. BioMed. Res. Int. 2021, 2021, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Eley, V.A.; Roberts, L.; Rickards, L.; Pelecanos, A.; Blackie, A.; Zhang, C.; Christensen, R.; Barrett, H.L. Arm and finger measurements in the third trimester: Implications for blood pressure measurement. Pregnancy Hypertens. Int. J. Women’s Cardiovasc. Health 2018, 14, 105–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sola, J.; Delgado-Gonzalo, R. The Handbook of Cuffless Blood Pressure Monitoring; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Langwieser, N.; Prechtl, L.; Meidert, A.S.; Hapfelmeier, A.; Bradaric, C.; Ibrahim, T.; Laugwitz, K.-L.; Schmid, R.M.; Wagner, J.Y.; Saugel, B. Radial artery applanation tonometry for continuous noninvasive arterial blood pressure monitoring in the cardiac intensive care unit. Clin. Res. Cardiol. 2015, 104, 518–524. [Google Scholar] [CrossRef]
- Ahn, J.H.; Song, J.; Choi, I.; Youn, J.; Cho, J.W. Validation of Blood Pressure Measurement Using a Smartwatch in Patients With Parkinson’s Disease. Front. Neurol. 2021, 12, 650929. [Google Scholar] [CrossRef] [PubMed]

| Pat.01 | Pat.02 | Pat.03 | Pat.04 | Pat.12 | Pat.25 | Pat.26 | Pat.28 | Pat.29 | Pat.30 | Pat.31 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Datapoints | 13 | 19 | 25 | 30 | 6 | 6 | 7 | 6 | 10 | 7 | 10 |
| Median (IQR) | |
|---|---|
| ASA | 2 (0) |
| Age [years] | 36 (6.5) |
| Height [cm] | 169 (9) |
| Weight [kg] | 92 (33.3) |
| BMI [kg/m2] | 34.21 (17.24) |
| Bias | CI of Bias | −2 SD with CI | +2 SD with CI | |
|---|---|---|---|---|
| RRsys | −10.82 | −13.8 to −7.8 | −46.6 (−51.8 to −41.4) | 25.0 (19.8 to 30.2) |
| RRmean | 0.68 | −1.6 to 3.0 | −26.8 (−30.8 to −22.8) | 28.2 (24.2 to 32.2) |
| RRdia | −0.45 | −3.1 to 2.2 | −32.1 (−36.7 to −27.5) | 31.2 (26.6 to 35.8) |
| Author | Year | Cohort | Study Size | RRsys [mmHg] | RRmean [mmHg] | RRdia [mmHg] | Data Points |
|---|---|---|---|---|---|---|---|
| Lee et al. [25] | 2020 | Breast cancer surgery | 10 | −6.0 (−24.3 to 12.3) | −3.8 (−19.7 to 12.1) | −1.2 (−16.1 to 13.7) | 245 |
| Noto et al. [26] | 2019 | Awake carotid endarterectomy | 30 | −3 (−22.1 to 16) | −6.8 (−20.1 to 6.3) | −9 (−19.7 to 1.5) | 2672 |
| Rogge et al. [14] | 2019 | Obese patients | 35 | 6.8 (−14.4 to 27.9) | 1.1 (−13.5 to 15.6) | 0.8 (−12.9 to 14.4) | 97,623 |
| Sakai et al. [27] | 2018 | Robot-assisted laparoscopic radical prostatectomy | 10 | −2.99 (−34.4 to 28.06) | −9.26 (−32.0 to 13.50) | −12.03 (−33.3 to 9.2) | 210 |
| Sang-Wook et al. [28] | 2021 | One-lung ventilation | 26 | −5.18 (−37.81 to 27.45) 1 | 1.05 (−18.85 to 20.95) 1 | 5.16 (−12.4 to 22.7) 1 | 8408 |
| Schumann et al. [21] | 2021 | Obese patients | 90 | −7 (−35 to 20) | −1 (−23 to 21) | 0 (−22.0 to 22.0) | 538 |
| Tanioku et al. [20] | 2020 | Cardiovascular surgery | 18 | 13.2 (−21.2 to 47.4) | −3.9 (−19.2 to 11.4) | −9.1 (−23.4 to 5.2) | 3068 |
| Yokose et al. [29] | 2019 | Major abdominal surgery | 30 | 0.7 (−24.6 to 26.0) | 7.9 (−7.6 to 21.3) | 10.1 (−3.8 to 24.0) | 6312 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helmer, P.; Helf, D.; Sammeth, M.; Winkler, B.; Hottenrott, S.; Meybohm, P.; Kranke, P. The Use of Non-Invasive Continuous Blood Pressure Measuring (ClearSight®) during Central Neuraxial Anaesthesia for Caesarean Section—A Retrospective Validation Study. J. Clin. Med. 2022, 11, 4498. https://doi.org/10.3390/jcm11154498
Helmer P, Helf D, Sammeth M, Winkler B, Hottenrott S, Meybohm P, Kranke P. The Use of Non-Invasive Continuous Blood Pressure Measuring (ClearSight®) during Central Neuraxial Anaesthesia for Caesarean Section—A Retrospective Validation Study. Journal of Clinical Medicine. 2022; 11(15):4498. https://doi.org/10.3390/jcm11154498
Chicago/Turabian StyleHelmer, Philipp, Daniel Helf, Michael Sammeth, Bernd Winkler, Sebastian Hottenrott, Patrick Meybohm, and Peter Kranke. 2022. "The Use of Non-Invasive Continuous Blood Pressure Measuring (ClearSight®) during Central Neuraxial Anaesthesia for Caesarean Section—A Retrospective Validation Study" Journal of Clinical Medicine 11, no. 15: 4498. https://doi.org/10.3390/jcm11154498
APA StyleHelmer, P., Helf, D., Sammeth, M., Winkler, B., Hottenrott, S., Meybohm, P., & Kranke, P. (2022). The Use of Non-Invasive Continuous Blood Pressure Measuring (ClearSight®) during Central Neuraxial Anaesthesia for Caesarean Section—A Retrospective Validation Study. Journal of Clinical Medicine, 11(15), 4498. https://doi.org/10.3390/jcm11154498






