Clinical Significance of Velamentous Cord Insertion Prenatally Diagnosed in Twin Pregnancy
Abstract
1. Introduction
2. Material and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Raisanen, S.; Georgiadis, L.; Harju, M.; Keski-Nisula, L.; Heinonen, S. Risk factors and adverse pregnancy outcomes among births affected by velamentous umbilical cord insertion: A retrospective population-based register study. Eur. J. Obstet. Gynecol. Reprod Biol. 2012, 165, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Buchanan-Hughes, A.; Bobrowska, A.; Visintin, C.; Attilakos, G.; Marshall, J. Velamentous cord insertion: Results from a rapid review of incidence, risk factors, adverse outcomes and screening. Syst. Rev. 2020, 9, 147. [Google Scholar] [CrossRef] [PubMed]
- Cambiaso, O.; Zhao, D.P.; Abasolo, J.I.; Aiello, H.A.; Oepkes, D.; Lopriore, E.; Otano, L. Discordance of cord insertions as a predictor of discordant fetal growth in monochorionic twins. Placenta 2016, 47, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Ebbing, C.; Kiserud, T.; Johnsen, S.L.; Albrechtsen, S.; Rasmussen, S. Prevalence, risk factors and outcomes of velamentous and marginal cord insertions: A population-based study of 634, 741 pregnancies. PLoS ONE 2013, 8, e70380. [Google Scholar] [CrossRef]
- Eddleman, K.A.; Lockwood, C.J.; Berkowitz, G.S.; Lapinski, R.H.; Berkowitz, R.L. Clinical significance and sonographic diagnosis of velamentous umbilical cord insertion. Am. J. Perinatol. 1992, 9, 123–126. [Google Scholar] [CrossRef]
- Kent, E.M.; Breathnach, F.M.; Gillan, J.E.; McAuliffe, F.M.; Geary, M.P.; Daly, S.; Higgins, J.R.; Dornan, J.; Morrison, J.J.; Burke, G.; et al. Placental cord insertion and birth weight discordance in twin pregnancies: Results of the national prospective ESPRiT Study. Am. J. Obstet. Gynecol. 2011, 205, 376.e1–376.e7. [Google Scholar] [CrossRef]
- Hanley, M.L.; Ananth, C.V.; Shen-Schwarz, S.; Smulian, J.C.; Lai, Y.L.; Vintzileos, A.M. Placental cord insertion and birth weight discordancy in twin gestations. Obstet. Gynecol. 2002, 99, 477–482. [Google Scholar] [CrossRef]
- Lopriore, E.; Sueters, M.; Middeldorp, J.M.; Oepkes, D.; Walther, F.J.; Vandenbussche, F.P. Velamentous cord insertion and unequal placental territories in monochorionic twins with and without twin-to-twin-transfusion syndrome. Am. J. Obstet. Gynecol. 2007, 196, 159.e1–159.e5. [Google Scholar] [CrossRef]
- Yonetani, N.; Ishii, K.; Kawamura, H.; Mabuchi, A.; Hayashi, S.; Mitsuda, N. Significance of Velamentous cord insertion for twin-twin transfusion syndrome. Fetal. Diagn. Ther. 2015, 38, 276–281. [Google Scholar] [CrossRef]
- Ebbing, C.; Kiserud, T.; Johnsen, S.L.; Albrechtsen, S.; Rasmussen, S. Third stage of labor risks in velamentous and marginal cord insertion: A population-based study. Acta Obstet. Gynecol. Scand. 2015, 94, 878–883. [Google Scholar] [CrossRef]
- Ebbing, C.; Johnsen, S.L.; Albrechtsen, S.; Sunde, I.D.; Vekseth, C.; Rasmussen, S. Velamentous or marginal cord insertion and the risk of spontaneous preterm birth, prelabor rupture of the membranes, and anomalous cord length: A population-based study. Acta Obstet. Gynecol. Scand. 2017, 96, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda, W. Velamentous insertion of the umbilical cord: A first-trimester sonographic screening study. J. Ultrasound Med. 2006, 25, 963–968. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.J.O. The management of multiple pregnancy. Early Hum. Dev. 2006, 82, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Jauniaux, E.; Ayres-de-Campos, D.; Langhoff-Roos, J.; Fox, K.A.; Collins, S. FIGO classification for the clinical diagnosis of placenta accreta spectrum disorders. Int. J. Gynecol. Obstet. 2019, 146, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.; Beune, I.; Hecher, K.; Wynia, K.; Ganzevoort, W.; Reed, K.; Lewi, L.; Oepkes, D.; Gratacos, E.; Thilaganathan, B.; et al. Consensus definition and essential reporting parameters of selective fetal growth restriction in twin pregnancy: A Delphi procedure. Ultrasound Obstet. Gynecol. 2019, 53, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Gratacos, E.; Lewis, L.; Munoz, B.; Acosta-Rojas, R.; Hernandez-Andrade, E.; Martinez, J.M.; Carreras, E.; Deprest, J. A classification system for selective intrauterine growth restriction in monochorionic pregnancies according to umbilical artery Doppler flow in the smaller twin. Ultrasound Obstet. Gynecol. 2007, 30, 28–34. [Google Scholar] [CrossRef]
- Quintero, R.A.; Morales, W.J.; Allen, M.H.; Bornick, P.W.; Johnson, P.K.; Kruger, M. Staging of twin-twin transfusion syndrome. J Perinatol. 1999, 19, 550–555. [Google Scholar] [CrossRef]
- Yerlikaya, G.; Pils, S.; Springer, S.; Chalubinski, K.; Ott, J. Velamentous cord insertion as a risk factor for obstetric outcome: A retrospective casecontrol study. Arch. Gynecol. Obstet. 2016, 293, 975–981. [Google Scholar] [CrossRef]
- Heinonen, S.; Ryynanen, M.; Kirkinen, P.; Saarikoski, S. Perinatal diagnostic evaluation of velamentous umbilical cord insertion: Clinical, Doppler, and ultrasonic findings. Obstet. Gynecol. 1996, 87, 112–117. [Google Scholar] [CrossRef]
- Ananth, C.V.; Savitz, D.A.; Williams, M.A. Placental abruption and its association with hypertension and prolonged rupture of membranes: A methodologic review and meta-analysis. Obstet. Gynecol. 1996, 88, 309–318. [Google Scholar] [CrossRef]
- De Paepe, M.E.; Shapiro, S.; Young, L.; Luks, F.I. Placental characteristics of selective birth weight discordance in diamniotic-monochorionic twin gestations. Placenta 2010, 31, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Hack, K.E.A.; Nikkels, P.G.J.; Koopman-Esseboom, C.; Derks, J.B.; Elias, S.G.; van Gemert, M.J.C.; Visser, G.H.A. Placental characteristics of monochorionic diamniotic twin pregnancies in relation to perinatal outcome. Placenta 2008, 29, 976–981. [Google Scholar] [CrossRef] [PubMed]
- Costa-Castro, T.; Zhao, D.P.; Lipa, M.; Haak, M.C.; Oepkes, D.; Severo, M.; Montenegro, N.; Matias, A.; Lopriore, E. Velamentous cord insertion in dichorionic and monochorionic twin pregnancies—Does it make a difference? Placenta 2016, 42, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Kalafat, E.; Thilaganathan, A.; Papageorghiou, A.; Bhide, A.; Khalil, A. Significance of placental cord insertion site in twin pregnancy. Ultrasound Obstet. Gynecol. 2018, 52, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, E.A.; Javid, N.; Duncombe, G.; Li, Z.; Safi, N.; Cincotta, R.; Homer, C.S.E.; Halliday, L.; Oyelese, Y. Vasa previa diagnosis, clinical practice, and outcomes in Australia. Obstet. Gynecol. 2017, 130, 591–598. [Google Scholar] [CrossRef]
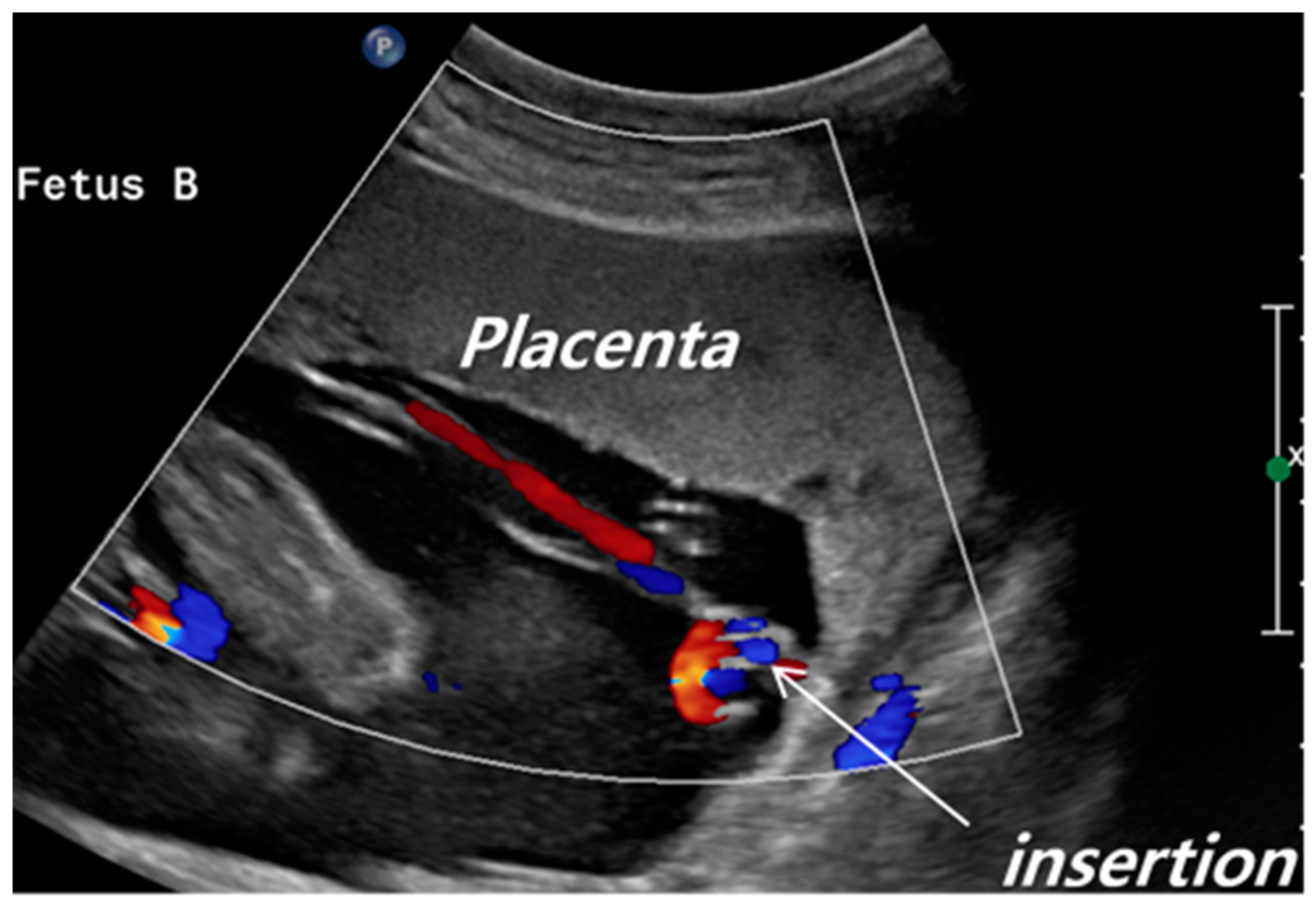
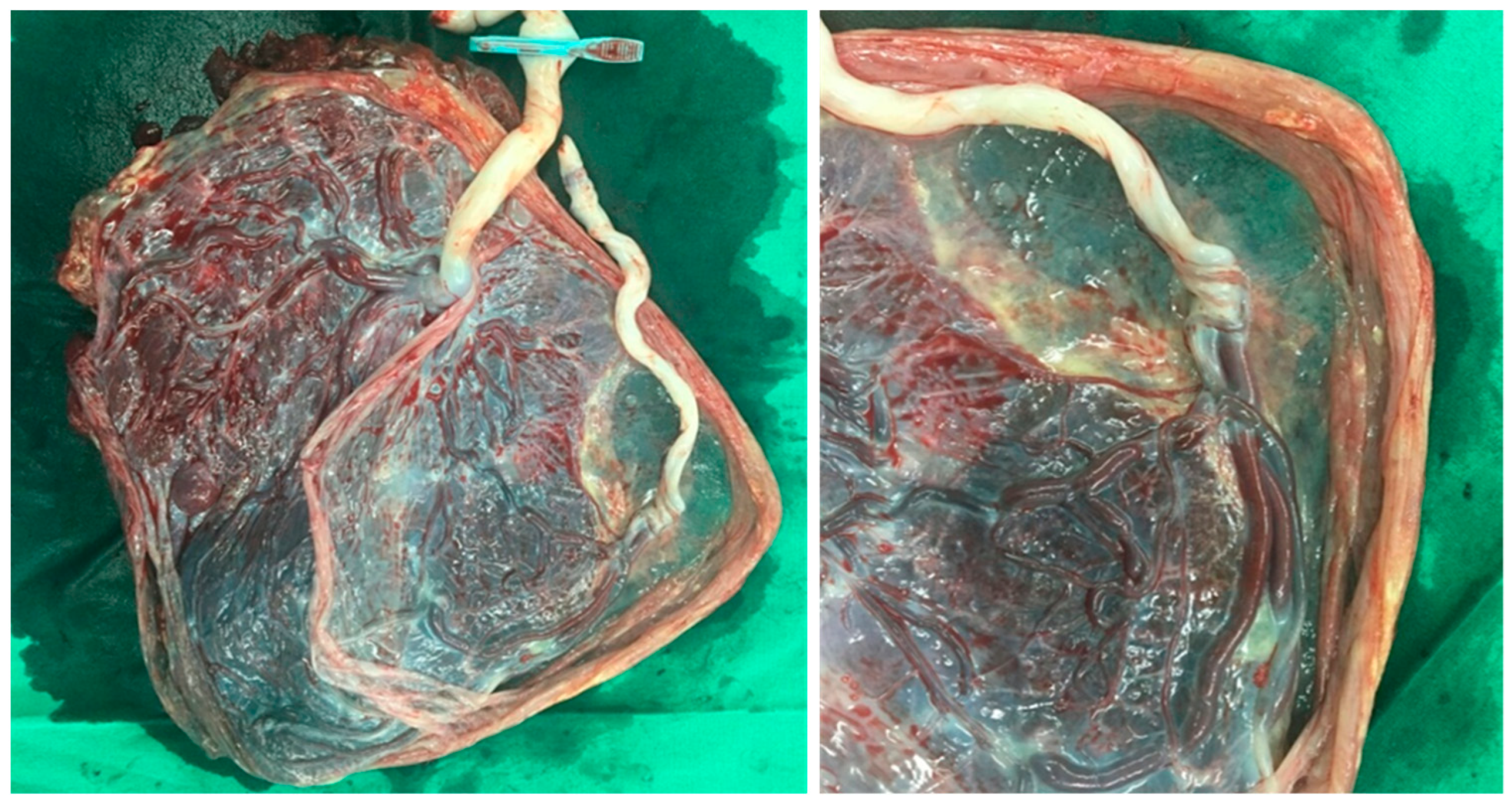
| Velamentous | Non-Velamentous | p | |
|---|---|---|---|
| (n = 58) | (n = 883) | ||
| Maternal characteristics | |||
| Maternal age at delivery (years) | 34.7 ± 3.3 | 35.3 ± 2.7 | 0.128 |
| Pregnancy conceived via ART | 48 (82.8%) | 733 (83.0%) | 0.969 |
| GA at delivery (weeks) | 36.0 ± 2.1 | 36.5 ± 1.4 | 0.077 |
| Gravidity | 1.7 ± 1.2 | 1.7 ± 0.9 | 0.948 |
| Nulliparous | 46 (79.3%) | 768 (87.0%) | 0.202 |
| MCDA | 12 (20.7%) | 141 (16.0%) | 0.633 |
| DCDA | 46 (79.3%) | 742 (84.0%) | 0.633 |
| Pregnancy outcomes | |||
| Preeclampsia | 5 (8.6%) | 115 (13.0%) | 0.395 |
| GDM | 4 (6.9%) | 79 (8.9%) | 0.643 |
| PPROM | 10 (17.2%) | 115 (13.0%) | 0.466 |
| Placenta previa | 3 (5.2%) | 26 (2.9%) | 0.491 |
| Vasa previa | 4 (6.9%) | 0 | 0.008 |
| PAS | 3 (5.2%) | 0 | 0.022 |
| Postpartum hemorrhage | 28 (48.3%) | 353 (40.0%) | 0.311 |
| PTB < 37 weeks | 26 (44.8%) | 331 (37.5%) | 0.264 |
| PTB < 34 weeks | 6 (10.3%) | 60 (6.8%) | 0.305 |
| Birth-weight discordance ≥ 20% | 12 (20.7 %) | 164 (18.6%) | 0.689 |
| Birth-weight discordance ≥ 25% | 6 (10.3%) | 47 (5.3%) | 0.108 |
| sFGR | 10 (17.2%) | 156 (17.7%) | 0.934 |
| Neonatal outcomes | n = 116 fetuses | n = 1766 fetuses | |
| Birth weight (g) | 2418 ± 499 | 2377 ± 371 | 0.408 |
| 1-min Apgar Score | 7.3 ± 0.86 | 7.4 ± 0.76 | 0.293 |
| 5-min Apgar Score | 8.4 ± 0.65 | 8.4 ± 0.67 | 0.661 |
| NICU admission | 51 (44.0%) | 653 (37%) | 0.222 |
| MC (n = 12) | DC (n = 46) | p | |
|---|---|---|---|
| Maternal characteristics | |||
| Maternal age at delivery (years) | 33.8 ± 3.0 | 35.0 ± 3.3 | 0.325 |
| Pregnancy conceived via ART | 2 (16.7%) | 46 (100%) | 0 |
| GA at delivery (weeks) | 35.0 ± 2.5 | 36.2 ± 1.9 | 0.078 |
| Pregnancy outcomes | |||
| Preeclampsia | 1 (8.3%) | 4 (8.7%) | 0.968 |
| GDM | 0 | 4(8.7%) | 0.29 |
| PPROM | 2 (16.7%) | 8 (17.4%) | 0.953 |
| Placenta previa | 0 | 3 (6.5%) | 0.364 |
| Vasa previa | 1 (8.3%) | 3 (6.5%) | 0.825 |
| PAS | 0 | 3 (6.5%) | 0.364 |
| Postpartum hemorrhage | 6 (50.0%) | 22 (47.8%) | 0.893 |
| PTB < 37 weeks | 8 (66.7%) | 18 (39.1%) | 0.088 |
| PTB < 34 weeks | 2 (16.7%) | 4 (8.7%) | 0.419 |
| Birth-weight discordance ≥ 20% | 4 (33.3%) | 8 (17.4%) | 0.549 |
| Birth-weight discordance ≥ 25% | 3 (25.0%) | 3 (6.5%) | 0.061 |
| sFGR | 3 (25.0%) | 7 (15.2%) | 0.424 |
| Neonatal outcomes | n = 24 fetuses | n = 92 fetuses | |
| Birth weight (g) | 2186 ± 485 | 2479 ± 487 | 0.01 |
| 1-min Apgar Score | 6.8 ± 0.9 | 7.4 ± 0.8 | 0.002 |
| 5-min Apgar Score | 7.9 ± 0.6 | 8.5 ± 0.6 | 0 |
| NICU admission | 14 (58.3%) | 37 (40.2%) | 0.111 |
| Velamentous | Non-velamentous | p | |
|---|---|---|---|
| (n = 46) | (n = 742) | ||
| Maternal characteristics | |||
| Age at delivery (years) | 34.9 ± 3.3 | 35.2 ± 3.1 | 0.106 |
| Pregnancy conceived via ART | 46 (100%) | 626 (95.1%) | 0.133 |
| GA at delivery (weeks) | 36.2 ± 1.9 | 36.6 ± 1.4 | 0.196 |
| Nulliparous, n (%) | 36 (78.3%) | 603 (91.6%) | 0.195 |
| Pregnancy outcomes | |||
| Preeclampsia | 4 (8.7%) | 70 (10.6%) | 0.714 |
| GDM | 4 (8.7%) | 63 (9.6%) | 0.876 |
| PPROM | 8 (17.4%) | 94 (14.3%) | 0.693 |
| Placenta previa | 3 (6.5%) | 24 (3.6%) | 0.443 |
| Vasa previa | 3 (6.5%) | 0 | 0.018 |
| PAS | 3 (6.5%) | 0 | 0.018 |
| Postpartum hemorrhage | 22 (47.8%) | 274 (41.6%) | 0.499 |
| PTB < 37weeks | 18 (39.1%) | 247 (33.3%) | 0.416 |
| PTB < 34weeks | 4 (8.7%) | 46 (6.2%) | 0.5 |
| Birth-weight discordance ≥ 20% | 8 (17.4%) | 141 (21.4%) | 0.582 |
| Birth-weight discordance ≥ 25% | 3 (6.5%) | 39 (5.9%) | 0.897 |
| sFGR | 7 (15.2%) | 132 (17.8%) | 0.657 |
| Neonatal outcomes | n = 92 fetuses | n = 1484 fetuses | |
| Birth weight (g) | 2479 ± 487 | 2408 ± 357 | 0.184 |
| 1-min Apgar Score | 7.4 ± 0.8 | 7.4 ± 0.7 | 0.914 |
| 5-min Apgar Score | 8.6 ± 0.6 | 8.5 ± 0.6 | 0.658 |
| NICU admission | 37 (40.2%) | 485 (32.7%) | 0.228 |
| Velamentous (n = 12) | Non-Velamentous (n = 141) | p | |
|---|---|---|---|
| Maternal characteristics | |||
| Age at delivery (years) | 33.8 ± 3.0 | 34.5 ± 3.1 | 0.576 |
| Pregnancy conceived via ART | 2 (16.7%) | 23 (16.3%) | 0.887 |
| GA at delivery (weeks) | 35.0 ± 2.5 | 35.8 ± 1.8 | 0.323 |
| Nulliparous, n (%) | 8 (66.7%) | 78 (55.3%) | 0.820 |
| Pregnancy outcomes | |||
| Preeclampsia | 1 (8.3%) | 11 (7.8%) | 0.254 |
| GDM | 0 | 8 (5.7%) | 0.378 |
| PPROM | 2 (16.7%) | 8 (5.7%) | 0.378 |
| Placenta previa | 1 (8.3%) | 0 | 0.240 |
| Vasa previa | 1 (8.3%) | 0 | 0.240 |
| PAS | 0 | 0 | - |
| Postpartum hemorrhage | 6 (50.0%) | 39 (27.7%) | 0.315 |
| PTB < 37weeks | 8 (66.7%) | 84 (59.6%) | 0.630 |
| PTB < 34weeks | 2 (16.7%) | 14 (9.9%) | 0.464 |
| Birth-weight discordance ≥ 20% | 4 (33.3%) | 23 (16.3%) | 0.378 |
| Birth-weight discordance ≥ 25% | 3 (25.0%) | 8 (5.7%) | 0.161 |
| sFGR | 3 (25.0%) | 24 (17.0%) | 0.486 |
| TTTS | 0 | 7 (5.0%) | 0.400 |
| Neonatal outcomes | n = 24 fetuses | n = 282 fetuses | |
| Birth weight (g) | 2186 ± 485 | 2214 ± 413 | 0.816 |
| 1-min Apgar Score | 6.8 ± 0.9 | 7.2 ± 0.9 | 0.123 |
| 5-min Apgar Score | 7.9 ± 0.6 | 8.1 ± 0.8 | 0.283 |
| NICU admission | 14 (58.3%) | 148 (52.5%) | 0.938 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.-M.; Lee, S.; Park, M.-K.; Han, Y.J.; Kim, M.Y.; Boo, H.Y.; Chung, J.H. Clinical Significance of Velamentous Cord Insertion Prenatally Diagnosed in Twin Pregnancy. J. Clin. Med. 2021, 10, 572. https://doi.org/10.3390/jcm10040572
Lee H-M, Lee S, Park M-K, Han YJ, Kim MY, Boo HY, Chung JH. Clinical Significance of Velamentous Cord Insertion Prenatally Diagnosed in Twin Pregnancy. Journal of Clinical Medicine. 2021; 10(4):572. https://doi.org/10.3390/jcm10040572
Chicago/Turabian StyleLee, Hyun-Mi, SiWon Lee, Min-Kyung Park, You Jung Han, Moon Young Kim, Hye Yeon Boo, and Jin Hoon Chung. 2021. "Clinical Significance of Velamentous Cord Insertion Prenatally Diagnosed in Twin Pregnancy" Journal of Clinical Medicine 10, no. 4: 572. https://doi.org/10.3390/jcm10040572
APA StyleLee, H.-M., Lee, S., Park, M.-K., Han, Y. J., Kim, M. Y., Boo, H. Y., & Chung, J. H. (2021). Clinical Significance of Velamentous Cord Insertion Prenatally Diagnosed in Twin Pregnancy. Journal of Clinical Medicine, 10(4), 572. https://doi.org/10.3390/jcm10040572






