Sunflower Oilcake as a Potential Source for the Development of Edible Membranes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Film Development
2.3. SFOC Characterization
2.4. Film Characterization
2.4.1. Affinity to Water
2.4.2. Barrier Properties
2.4.3. Thickness, Density, Tensile Strength and Hardness
2.4.4. Optical Properties
2.4.5. FT-IR
2.4.6. Scanning Electron Microscopy (SEM)
2.4.7. Antimicrobial Analysis
2.5. Statistical Analysis
3. Results
3.1. Sunflower Oilcake Characterization
3.2. Water Affinity
3.3. Barrier Properties
3.3.1. Water Vapor Permeability (WVP) and Water Vapor Transmission Rate (WVTR)
3.3.2. Oxygen Permeability (OxyP)
3.3.3. Oil Permeability (OP)
3.4. Thickness, Density, Tensile Strength and Hardness
3.5. Optical Properties
3.5.1. Color Analysis
3.5.2. UV–VIS Spectra
3.6. FT-IR
3.7. Apparence and Morphologhy
3.8. Microbiological Stability
| Microorganism | Control, cfu | SFOC1, cfu | SFOC2, cfu | SFOC3, cfu | SFOC4, cfu | SFOC5, cfu | Limits |
|---|---|---|---|---|---|---|---|
| Total count | 1 | 2 | 1 | 4 | 4 | 15 | <50 cfu 1 <100 cfu 2 |
| Enterococcus | Absent | Absent | Absent | Absent | Absent | 1 | <150 cfu 4 |
| Coliforms | Absent | Absent | Absent | Absent | Absent | Absent | <10 cfu 1 Absent 2 <50 cfu 3 |
| Enterobacteriaceae | Absent | Absent | Absent | Absent | Absent | Absent | Absent1 |
| Yeast and molds | Absent | Absent | Absent | Absent | Absent | 1 | <100 cfu 1 Absent 2 <102 cfu 3 |
| Bacillus cereus | Absent | Absent | Absent | Absent | Absent | 1 | 102–103 5 |
| E. coli | Absent | Absent | Absent | Absent | Absent | Absent | Absent 1,2,3 |
| Salmonella | Absent | Absent | Absent | Absent | Absent | Absent | Absent 1,2,3 |
| Staphylococcus aureus | Absent | Absent | Absent | Absent | Absent | Absent | Absent 1,2 |
| Listeria | Absent | Absent | Absent | Absent | Absent | Absent | Absent 1,2 |
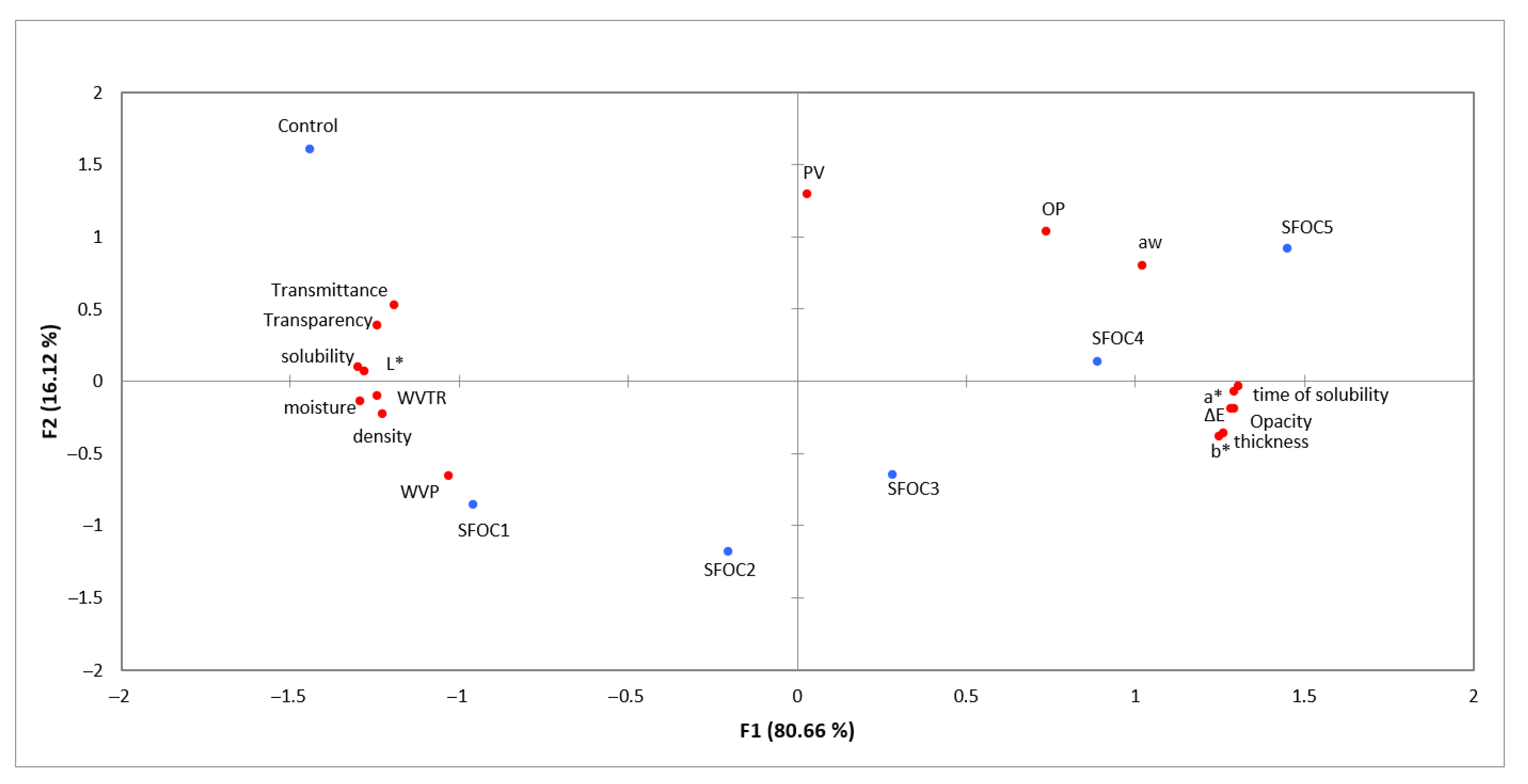
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Trajkovska Petkoska, A.; Daniloski, D.; D’Cunha, N.M.; Naumovski, N.; Broach, A.T. Edible packaging: Sustainable solutions and novel trends in food packaging. Food Res. Int. 2021, 140, 109981. [Google Scholar] [CrossRef] [PubMed]
- Thulasisingh, A.; Kumar, K.; Yamunadevi, B.; Poojitha, N.; SuhailMadharHanif, S.; Kannaiyan, S. Biodegradable packaging materials. Polym. Bull. 2021, 79, 4467–4496. [Google Scholar] [CrossRef]
- Afzal, A.; Khaliq, Z.; Ahmad, S.; Ahmad, F.; Noor, A.; Qadir, M.B. Development and characterization of biodegradable composite film. Environ. Technol. Innov. 2021, 23, 101664. [Google Scholar] [CrossRef]
- Sharma, N.; Bhardwaj, A.; Joshi, S.; Poonia, A. Scope, functions, and novelty of packaging edibles. In Edible Food Packaging; Springer: Berlin/Heidelberg, Germany, 2022; pp. 87–101. [Google Scholar] [CrossRef]
- Mohamed, S.A.A.; El-Sakhawy, M.; El-Sakhawy, M.A.M. Polysaccharides, Protein and Lipid-Based Natural Edible Films in Food Packaging: A Review. Carbohydr. Polym. 2020, 238, 116178. [Google Scholar] [CrossRef]
- Ajesh Kumar, V.; Hasan, M.; Mangaraj, S.; Pravitha, M.; Verma, D.K.; Srivastav, P.P. Trends in Edible Packaging Films and its Prospective Future in Food: A Review. Appl. Food Res. 2022, 2, 100118. [Google Scholar] [CrossRef]
- Imre, B.; García, L.; Puglia, D.; Vilaplana, F. Reactive compatibilization of plant polysaccharides and biobased polymers: Review on current strategies, expectations and reality. Carbohydr. Polym. 2019, 209, 20–37. [Google Scholar] [CrossRef]
- Hassan, B.; Chatha, S.A.S.; Hussain, A.I.; Zia, K.M.; Akhtar, N. Recent advances on polysaccharides, lipids and protein based edible films and coatings: A review. Int. J. Biol. Macromol. 2018, 109, 1095–1107. [Google Scholar] [CrossRef]
- Khwaldia, K.; Arab-Tehrany, E.; Desobry, S. Biopolymer Coatings on Paper Packaging Materials. Compr. Rev. Food Sci. Food Saf. 2010, 9, 82–91. [Google Scholar] [CrossRef]
- Tavassoli-Kafrani, E.; Gamage, M.V.; Dumée, L.F.; Kong, L.; Zhao, S. Edible films and coatings for shelf life extension of mango: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 2432–2459. [Google Scholar] [CrossRef]
- Galus, S.; Kibar, E.A.A.; Gniewosz, M.; Kraśniewska, K. Novel materials in the preparation of edible films and coatings—A review. Coatings 2020, 10, 674. [Google Scholar] [CrossRef]
- Salgado, P.R.; Ortiz, C.M.; Musso, Y.S.; di Giorgio, L.; Mauri, A.N. Edible films and coatings containing bioactives. Curr. Opin. Food Sci. 2015, 5, 86–92. [Google Scholar] [CrossRef]
- Borrello, M.; Caracciolo, F.; Lombardi, A.; Pascucci, S.; Cembalo, L. Consumers’ perspective on circular economy strategy for reducing food waste. Sustainability 2017, 9, 141. [Google Scholar] [CrossRef]
- Singh, R.; Langyan, S.; Sangwan, S.; Rohtagi, B.; Khandelwal, A. Protein for Human Consumption from Oilseed Cakes: A Review. Front. Sustain. Food Syst. 2022, 6, 101. [Google Scholar] [CrossRef]
- Arrutia, F.; Binner, E.; Williams, P.; Waldron, K.W. Oilseeds beyond oil: Press cakes and meals supplying global protein requirements. Trends Food Sci. Technol. 2020, 100, 88–102. [Google Scholar] [CrossRef]
- Popović, S.; Hromiš, N.; Šuput, D.; Bulut, S.; Romanić, R.; Lazić, V. Valorization of By-Products from the Production of Pressed Edible Oils to Produce Biopolymer Films, 1st ed.; Academic Press: London, UK, 2020; ISBN 9780128181881. [Google Scholar]
- Adeleke, B.S.; Babalola, O.O. Oilseed crop sunflower (Helianthus annuus) as a source of food: Nutritional and health benefits. Food Sci. Nutr. 2020, 8, 4666–4684. [Google Scholar] [CrossRef] [PubMed]
- Ancuţa, P.; Sonia, A. Oil press-cakes and meals valorization through circular economy approaches: A review. Appl. Sci. 2020, 10, 7432. [Google Scholar] [CrossRef]
- Gupta, A.; Sharma, R. Oilseed as potential functional food Ingredient. In Trends & Prospects in Food Technology, Processing and Preservation; Today and Tomorrow’s Printers and Publishers: New Delhi, India, 2019; pp. 191–215. [Google Scholar]
- Otles, S.; Despoudi, S.; Bucatariu, C.; Kartal, C. Valorization, and Sustainability in the Food Industry; Elsevier Inc.: Amsterdam, The Netherlands, 2015; ISBN 978-0-12-800351-0. [Google Scholar]
- Baiano, A. Recovery of biomolecules from food wastes—A review. Molecules 2014, 19, 14821–14842. [Google Scholar] [CrossRef]
- Hamed, I.; Jakobsen, A.N. Sustainable edible packaging systems based on active compounds from food processing byproducts: A review. Compr. Rev. Food Sci. Food Saf. 2022, 21, 198–226. [Google Scholar] [CrossRef]
- Esposito, B.; Sessa, M.R.; Sica, D.; Malandrino, O. Towards circular economy in the agri-food sector. A systematic literature review. Sustainability 2020, 12, 7401. [Google Scholar] [CrossRef]
- Šuput, D.; Lazić, V.; Popović, S.; Hromiš, N.; Bulut, S.; Pezo, L.; Banićević, J. Effect of process parameters on biopolymer films based on sunflower oil cake. J. Process. Energy Agric. 2018, 22, 125–128. [Google Scholar] [CrossRef]
- Šuput, D.; Popović, S.; Hromiš, N.; Bulut, S.; Lazić, V. Biopolymer films properties change affected by essential oils addition. J. Process. Energy Agric. 2019, 23, 61–65. [Google Scholar] [CrossRef]
- Popović, S.; Lazić, V.; Popović, L.; Vaštag, Ž.; Peričin, D. Effect of the addition of pumpkin oil cake to gelatin to produce biodegradable composite films. Int. J. Food Sci. Technol. 2010, 45, 1184–1190. [Google Scholar] [CrossRef]
- Popović, S.; Peričin, D.; Vaštag, Ž.; Popović, L.; Lazić, V. Evaluation of edible film-forming ability of pumpkin oil cake; Effect of pH and temperature. Food Hydrocoll. 2011, 25, 470–476. [Google Scholar] [CrossRef]
- Mirpoor, S.F.; Giosafatto, C.V.L.; di Girolamo, R.; Famiglietti, M. Hemp (Cannabis sativa) seed oilcake as a promising by-product for developing protein-based films: Effect of transglutaminase-induced crosslinking. Food Packag. Shelf Life 2022, 31, 100779. [Google Scholar] [CrossRef]
- Jang, S.A.; Lim, G.O.; Song, K. Bin Preparation and Mechanical Properties of Edible Rapeseed Protein Films. J. Food Sci. 2011, 76, 218–223. [Google Scholar] [CrossRef]
- Jang, S.A.; Shin, Y.J.; Song, K. Bin Effect of rapeseed protein-gelatin film containing grapefruit seed extract on “Maehyang” strawberry quality. Int. J. Food Sci. Technol. 2011, 46, 620–625. [Google Scholar] [CrossRef]
- Puscaselu, R.G.; Anchidin-Norocel, L.; Petraru, A.; Ursachi, F. Strategies and Challenges for Successful Implementation of Green Economy Concept: Edible Materials for Meat Products Packaging. Foods 2021, 10, 3035. [Google Scholar] [CrossRef]
- Mihalcea, A.; Amariei, S. Study on Contamination with Some Mycotoxins in Maize and Maize-Derived Foods. Appl. Sci. 2022, 12, 2579. [Google Scholar] [CrossRef]
- Pérez-Mateos, M.; Montero, P.; Gómez-Guillén, M.C. Formulation and stability of biodegradable films made from cod gelatin and sunflower oil blends. Food Hydrocoll. 2009, 23, 53–61. [Google Scholar] [CrossRef]
- Gheorghita Puscaselu, R.; Amariei, S.; Norocel, L.; Gutt, G. New edible packaging material with function in shelf life extension: Applications for the meat and cheese industries. Foods 2020, 9, 562. [Google Scholar] [CrossRef]
- Kanmani, P.; Rhim, J.W. Antimicrobial and physical-mechanical properties of agar-based films incorporated with grapefruit seed extract. Carbohydr. Polym. 2014, 102, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Salgado, P.R.; Fernández, G.B.; Drago, S.R.; Mauri, A.N. Addition of bovine plasma hydrolysates improves the antioxidant properties of soybean and sunflower protein-based films. Food Hydrocoll. 2011, 25, 1433–1440. [Google Scholar] [CrossRef]
- Luo, Y.; Liu, H.; Yang, S.; Zeng, J.; Wu, Z. Sodium alginate-based green packaging films functionalized by guava leaf extracts and their bioactivities. Materials 2019, 12, 2923. [Google Scholar] [CrossRef] [PubMed]
- Fahrullah, F.; Eka Radiati, L.; Purwadi; Rosyidi, D. The physical characteristics of whey based edible film added with konjac. Curr. Res. Nutr. Food Sci. 2020, 8, 333–339. [Google Scholar] [CrossRef]
- Greenspan, L. Humidity Fixed Points of Binary Saturated Aqueous Solutions. J. Res. Natl. Bur. Stand. Sect. A Phys. Chem. 1977, 81, 89–96. [Google Scholar] [CrossRef]
- Chambi, H.N.M.; Grosso, C.R.F. Mechanical and water vapor permeability properties of biodegradables films based on methylcellulose, glucomannan, pectin and gelatin. Ciênc. E Tecnol. Aliment. 2011, 31, 739–746. [Google Scholar] [CrossRef]
- Tural, S.; Turhan, S. Properties of Edible Films Made from Anchovy By-Product Proteins and Determination of Optimum Protein and Glycerol Concentration by the TOPSIS Method. J. Aquat. Food Prod. Technol. 2017, 26, 640–654. [Google Scholar] [CrossRef]
- Cao, L.; Ge, T.; Meng, F.; Xu, S.; Li, J.; Wang, L. An edible oil packaging film with improved barrier properties and heat sealability from cassia gum incorporating carboxylated cellulose nano crystal whisker. Food Hydrocoll. 2020, 98, 105251. [Google Scholar] [CrossRef]
- Cruz-Diaz, K.; Cobos, Á.; Fernández-Valle, M.E.; Díaz, O.; Cambero, M.I. Characterization of edible films from whey proteins treated with heat, ultrasounds and/or transglutaminase. Application in cheese slices packaging. Food Packag. Shelf Life 2019, 22, 100397. [Google Scholar] [CrossRef]
- Sandoval, D.C.G.; Sosa, B.L.; Martínez-Ávila, G.C.G.; Fuentes, H.R.; Abarca, V.H.A.; Rojas, R. Formulation and characterization of edible films based on organic mucilage from Mexican Opuntia ficus-indica. Coatings 2019, 9, 506. [Google Scholar] [CrossRef]
- Rhim, J.W.; Wu, Y.; Weller, C.L.; Schnepf, M. Physical characteristics of a composite film of soy protein isolate and propyleneglycol alginate. J. Food Sci. 1999, 64, 149–152. [Google Scholar] [CrossRef]
- Jancikova, S.; Dordevic, D.; Nejezchlebova, M.; Sedlacek, P.; Jakub, T.; Bohuslava, T. Edible films from carrageenan/orange essential oil/trehalose—Structure, optical properties, and antimicrobial activity. Polymers 2021, 13, 332. [Google Scholar] [CrossRef]
- Kassab, Z.; El Achaby, M.; Tamraoui, Y.; Sehaqui, H.; Bouhfid, R.; Qaiss, A.E.K. Sunflower oil cake-derived cellulose nanocrystals: Extraction, physico-chemical characteristics and potential application. Int. J. Biol. Macromol. 2019, 136, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Petraru, A.; Ursachi, F.; Amariei, S. Nutritional characteristics assessment of sunflower seeds, oil and cake. Perspective of using sunflower oilcakes as a functional ingredient. Plants 2021, 10, 2487. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Khuntia, A.; Mitra, J. Thermodynamic and mathematical background of water activity and its significance from microbiological point of view for food products. In Trends & Prospects in Food Technology, Processing and Perservation; Today & Tomorrow’s Printers and Publishers: New Dehli, India, 2018; pp. 1–12. [Google Scholar]
- Nur Alim, B.; Dekker, M.; Fogliano, V.; Heising, J. Development of a moisture-activated antimicrobial film containing ground mustard seeds and its application on meat in active packaging system. Food Packag. Shelf Life 2021, 30, 100753. [Google Scholar] [CrossRef]
- Mathlouthi, M. Water content, water activity, water structure and the stability of foodstuffs. Food Control 2001, 12, 409–417. [Google Scholar] [CrossRef]
- Kandasamy, S.; Yoo, J.; Yun, J.; Kang, H.; Seol, K.; Kim, H. Application of Whey Protein-Based Edible Films and Coatings in Food Industries: An Updated Overview. Coatings 2021, 11, 1056. [Google Scholar] [CrossRef]
- Drozłowska, E. Valorization of flaxseed oil cake residual from cold-press oil production as a material for preparation of spray-dried functional powders for foodDrozłowska, Emilia Łopusiewiapplications as emulsion stabilizers. Biomolecules 2020, 10, 153. [Google Scholar] [CrossRef]
- Mikus, M.; Galus, S.; Ciurzy, A. Development and Characterization of Novel Composite Films Based on Soy Protein Isolate and Oilseed Flours. Molecules 2021, 26, 3738. [Google Scholar] [CrossRef]
- Vartiainen, J.; Vähä-Nissi, M.; Harlin, A. Biopolymer Films and Coatings in Packaging Applications—A Review of Recent Developments. Mater. Sci. Appl. 2014, 5, 708–718. [Google Scholar] [CrossRef]
- Jost, V.; Kobsik, K.; Schmid, M.; Noller, K. Influence of plasticiser on the barrier, mechanical and grease resistance properties of afile. Carbohydr. Polym. 2014, 110, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhang, B.; Qin, Y.; Li, F.; Yang, S.; Lu, P.; Wang, L.; Fan, J. Preparation and characterization of antifungal coating films composed of sodium alginate and cyclolipopeptides produced by Bacillus subtilis. Int. J. Biol. Macromol. 2020, 143, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Nowicki, M.; Richter, A.; Wolf, B.; Kaczmarek, H. Nanoscale mechanical properties of polymers irradiated by UV. Polymer 2003, 44, 6599–6606. [Google Scholar] [CrossRef]
- Sabo, B.; Bečica, T.; Keleš, N.; Kovačević, D.; Kovačević, D. The impact of packaging transparency on product attractiveness. J. Graph. Eng. Des. 2017, 8, 5–9. [Google Scholar] [CrossRef]
- Amariei, S.; Ursachi, F.; Petraru, A. Development of New Biodegradable Agar-Alginate Membranes for Food Packaging. Membranes 2022, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Riyanta, A.B.; Riyanto, S.; Lukitaningsih, E.; Rohman, A. The employment of Fourier Transform Infrared Spectroscopy (FTIR) and chemometrics for analysis of candlenut oil in binary mixture with grape seed oil. Food Res. 2020, 4, 184–190. [Google Scholar] [CrossRef]
- Matwijczuk, A.; Oniszczuk, T.; Matwijczuk, A.; Chruściel, E.; Kocira, A.; Niemczynowicz, A.; Wójtowicz, A.; Combrzy´nski, M.; Wiącek, D. Use of FTIR Spectroscopy and Chemometrics with Respect to Storage Conditions of Moldavian Dragonhead Oil. Sustainability 2019, 11, 6414. [Google Scholar] [CrossRef]
- Barth, A. Infrared spectroscopy of proteins. Biochim. Biophys. Acta BBA Bioenerg. 2007, 1767, 1073–1101. [Google Scholar] [CrossRef]
- Parodi, E. Extraction of proteins and residual oil from flax (Linum usitatissimum), camelina (Camelina sativa), and sunflower (Helianthus annuus) oilseed press cakes. Biomass Convers. Biorefinery 2021, 1–12. [Google Scholar] [CrossRef]
- Lun, L.W.; Anas, A.; Gunny, N.; Kasim, F.H. Fourier Transform Infrared Spectroscopy (FTIR) analysis of Paddy Straw Pulp treated using Deep Eutectic Solvent. AIP Conf. Proc. 2017, 1835, 020049. [Google Scholar] [CrossRef]
- Soofi, M.; Alizadeh, A.; Hamishehkar, H.; Almasi, H.; Roufegarinejad, L. Preparation of nanobiocomposite film based on lemon waste containing cellulose nanofiber and savory essential oil: A new biodegradable active packaging system. Int. J. Biol. Macromol. 2021, 169, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Chawla, R.; Sivakumar, S.; Kaur, H.; Mishra, S.K. Effect of starch based edible antimicrobial films and modified atmospheric packaging (MAP) on extended life of composite sweetmeat. Carbohydr. Polym. Technol. Appl. 2021, 2, 100055. [Google Scholar] [CrossRef]
- Venkatesan, J.; Bhatnagar, I.; Manivasagan, P.; Kang, K.H.; Kim, S.K. Alginate composites for bone tissue engineering: A review. Int. J. Biol. Macromol. 2015, 72, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Leid, J.G.; Willson, C.J.; Shirtliff, M.E.; Hassett, D.J.; Parsek, M.R.; Jeffers, A.K. The Exopolysaccharide Alginate Protects Pseudomonas aeruginosa Biofilm Bacteria from IFN-γ-Mediated Macrophage Killing. J. Immunol. 2005, 175, 7512–7518. [Google Scholar] [CrossRef]
- Julien, C.-K.; Edith, A.A.; Thomas, A.D.; Mireille, D. Microbiological quality of raw vegetables and ready to eat products sold in Abidjan (Cte dIvoire) markets. Afr. J. Microbiol. Res. 2017, 11, 204–210. [Google Scholar] [CrossRef]
- Yu, P.; Yu, S.; Wang, J.; Guo, H.; Zhang, Y.; Liao, X.; Zhang, J.; Wu, S.; Gu, Q.; Xue, L.; et al. Bacillus cereus isolated from vegetables in China: Incidence, genetic diversity, virulence genes, and antimicrobial resistance. Front. Microbiol. 2019, 10, 948. [Google Scholar] [CrossRef]


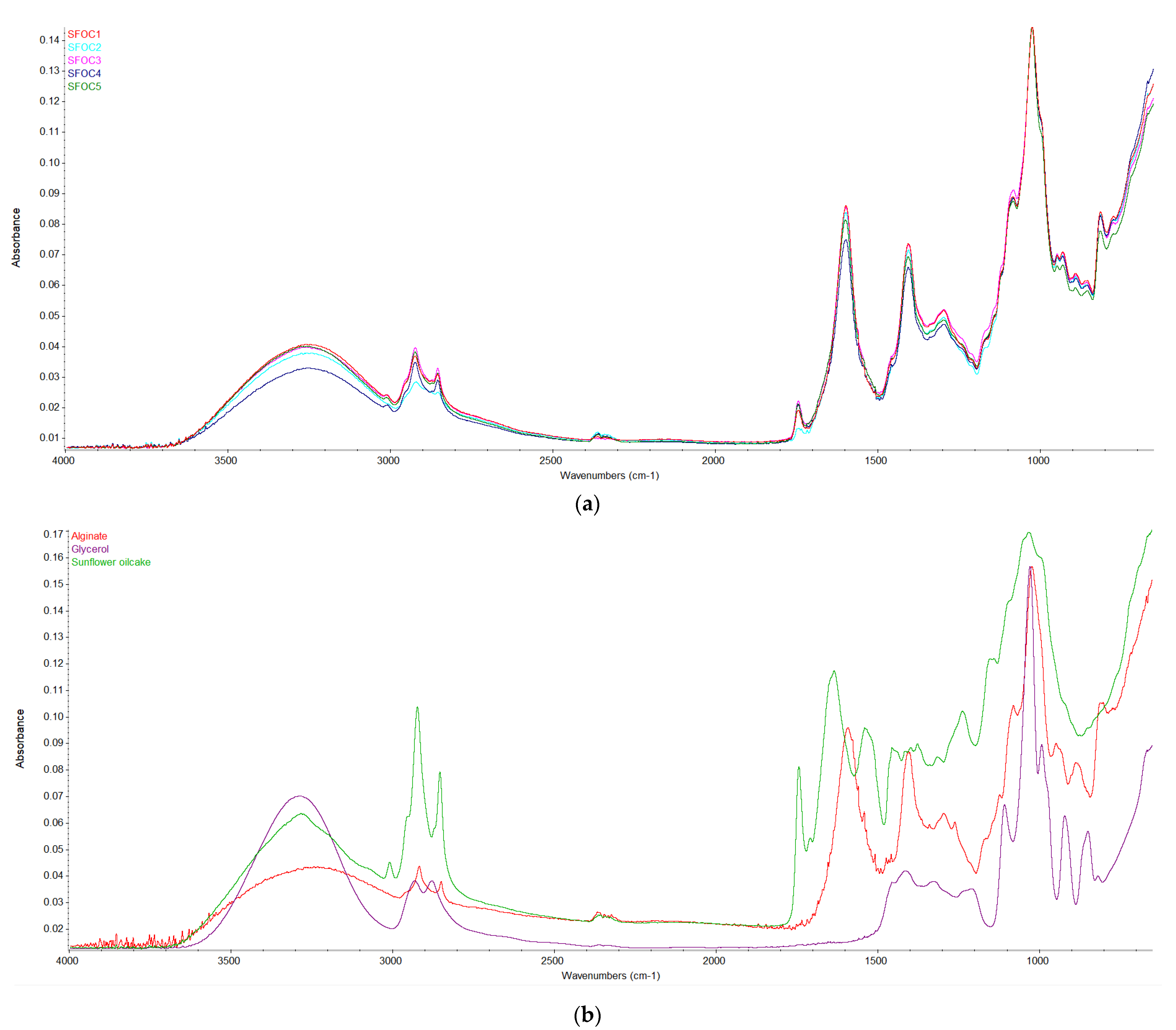

| Sample | Sodium Alginate, g | Glycerol, g | Sunflower Oilcake, g | Water, mL |
|---|---|---|---|---|
| Control | 1.00 | 0.50 | 0 | 100 |
| SFOC1 | 1.00 | 0.50 | 0.1 | 100 |
| SFOC2 | 1.00 | 0.50 | 0.2 | 100 |
| SFOC3 | 1.00 | 0.50 | 0.3 | 100 |
| SFOC4 | 1.00 | 0.50 | 0.4 | 100 |
| SFOC5 | 1.00 | 0.50 | 0.5 | 100 |
| Sample | Limit of Detection, µg/Kg | Limit of Quantification, µg/Kg | Results, µg/Kg | Maximum Limit *, µg/Kg |
|---|---|---|---|---|
| Zearalenone | 10 | 15 | 35.22 ± 3.96 | 2000 |
| Ochratoxin A | 0.5 | 1.5 | 8.38 ± 1.36 | 50 |
| Aflatoxin B1 | 0.3 | 0.7 | <LOQ | 10 |
| Deoxynivalenol | 0.011 | 0.042 | <LOD | 0.9 |
| Sample | aw | Moisture Content, % | Solubility, % | Time of Solubility, min |
|---|---|---|---|---|
| Control | 0.32 ± 0.01 c | 19.07 ± 0.78 a | 96.32 ± 0.11 a | 1.14 ± 0.08 a |
| SFOC1 | 0.27 ± 0.01 d | 18.73 ± 0.23 a | 95.13 ± 0.39 a | 1.39 ± 0.11 b |
| SFOC2 | 0.29 ± 0.01 d | 17.28 ± 0.80 b | 89.25 ± 0.69 a,b | 2.30 ± 0.09 c |
| SFOC3 | 0.32 ± 0.01 c | 15.34 ± 0.76 c | 86.11 ± 0.84 a,b | 2.42 ± 0.08 c |
| SFOC4 | 0.35 ± 0.01 b | 14.35 ± 0.60 d | 83.68 ± 4.71 b | 3.04 ± 0.04 d |
| SFOC5 | 0.40 ± 0.03 a | 13.07 ± 0.42 e | 82.79 ± 2.87 b | 3.19 ± 0.16 e |
| Sample | WVP, g × mm/KPa × h × m2 | WVTR, g/h | PV, meq O2/kg | OP, g × mm × m−2 × day−1 |
|---|---|---|---|---|
| Control | 1.66 × 10−4 ± 5.00 × 10−7 c | 13.55 ± 0.05 c | 5.50 ± 0.24 a | 0.027 ± 0.001 d |
| SFOC1 | 1.98 × 10−4 ± 1.02 × 10−5 d | 13.78 ± 0.74 c | 2.24 ± 0.14 d | 0.017 ± 0.000 d |
| SFOC2 | 1.95 × 10−4 ± 2.45 × 10−6 d | 11.56 ± 0.18 b | 2.24 ± 0.14 d | 0.018 ± 0.001 d |
| SFOC3 | 1.37 × 10−4 ± 1.00 × 10−6 b | 7.94 ± 0.05 a | 2.33 ± 0.00 d | 0.019 ± 0.001 c |
| SFOC4 | 1.30 × 10−4 ± 1.00 × 10−6 b | 7.34 ± 0.22 a | 3.50 ± 0.24 c | 0.030 ± 0.001 b |
| SFOC5 | 1.13 × 10−4 ± 1.30 × 10−6 a | 6.95 ± 0.93 a | 4.83 ± 0.24 b | 0.034 ± 0.000 a |
| Sample | Thickness, Mm | Density, g/cm3 | TS, MPa | Hardness, N |
|---|---|---|---|---|
| Control | 0.029 ± 0.01 a | 1.53 ± 0.03 a | 27.11 ± 1.97 a | 25.11 ± 0.19 a |
| SFOC1 | 0.034 ± 0.002 b | 1.45 ± 0.03 a,b | 22.15 ± 1.36 b | 22.26 ± 0.74 b |
| SFOC2 | 0.040 ± 0.002 c | 1.39 ± 0.01 b | 18.24 ± 0.22 c | 21.89 ± 0.26 c |
| SFOC3 | 0.041 ± 0.003 c | 1.38 ± 0.02 b | 15.73 ± 0.28 d | 18.39 ± 1.69 d |
| SFOC4 | 0.043 ± 0.003 c,d | 1.25 ± 0.04 c | 12.17 ± 0.39 e | 15.70 ± 0.50 e |
| SFOC5 | 0.044 ± 0.003 d | 1.04 ± 0.01 d | 8.66 ± 0.77 f | 12.52 ± 0.50 f |
| Sample | L* | a* | b* | ΔE | Opacity, UA/mm | Transparency, %/mm | Transmittance, % |
|---|---|---|---|---|---|---|---|
| Control | 92.97 ± 0.33 e | −5.53 ± 0.03 a | 10.31 ± 0.31 a | 1.74 ± 0.02 d | 6.50 ± 0.09 f | 65.90 ± 0.01 a | 81.52 ± 0.07 a |
| SFOC1 | 87.04 ± 2.01 d | −4.66 ± 0.37 b | 15.61 ± 1.31 b | 9.72 ± 0.55 c | 12.29 ± 0.03 e | 48.50 ± 0.02 b | 44.55 ± 0.06 b |
| SFOC2 | 83.74 ± 1.68 c | −4.11 ± 0.36 b,c | 18.95 ± 1.56 c | 14.47 ± 0.99 b | 17.30 ± 0.03 d | 34.50 ± 0.02 c | 23.98 ± 0.05 d |
| SFOC3 | 81.01 ± 1.84 c | −3.53 ± 0.34 c | 19.79 ± 1.42 cd | 17.10 ± 0.63 b | 17.95 ± 0.02 c | 33.92 ± 0.06 c | 24.54 ± 0.06 c |
| SFOC4 | 76.78 ± 3.25 b | −2.71 ± 0.74 d | 22.00 ± 2.44 d,e | 21.88 ± 0.07 a | 20.65 ± 0.02 b | 29.19 ± 1.25 d | 13.77 ± 0.50 e |
| SFOC5 | 73.10 ± 0.73 a | −2.10 ± 0.17 d | 23.46 ± 0.17 e | 25.77 ± 0.70 a | 26.06 ± 0.01 a | 20.90 ± 0.17 e | 8.38 ± 0.02 f |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petraru, A.; Amariei, S. Sunflower Oilcake as a Potential Source for the Development of Edible Membranes. Membranes 2022, 12, 789. https://doi.org/10.3390/membranes12080789
Petraru A, Amariei S. Sunflower Oilcake as a Potential Source for the Development of Edible Membranes. Membranes. 2022; 12(8):789. https://doi.org/10.3390/membranes12080789
Chicago/Turabian StylePetraru, Ancuţa, and Sonia Amariei. 2022. "Sunflower Oilcake as a Potential Source for the Development of Edible Membranes" Membranes 12, no. 8: 789. https://doi.org/10.3390/membranes12080789
APA StylePetraru, A., & Amariei, S. (2022). Sunflower Oilcake as a Potential Source for the Development of Edible Membranes. Membranes, 12(8), 789. https://doi.org/10.3390/membranes12080789






