Tyrosine Kinase Inhibitors Do Not Promote a Decrease in SARS-CoV-2 Anti-Spike IgG after BNT162b2 Vaccination in Chronic Myeloid Leukemia: A Prospective Observational Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Assessment of Serological Response
2.3. Presence or Absence of COVID-19
2.4. Statistical Analysis
3. Results
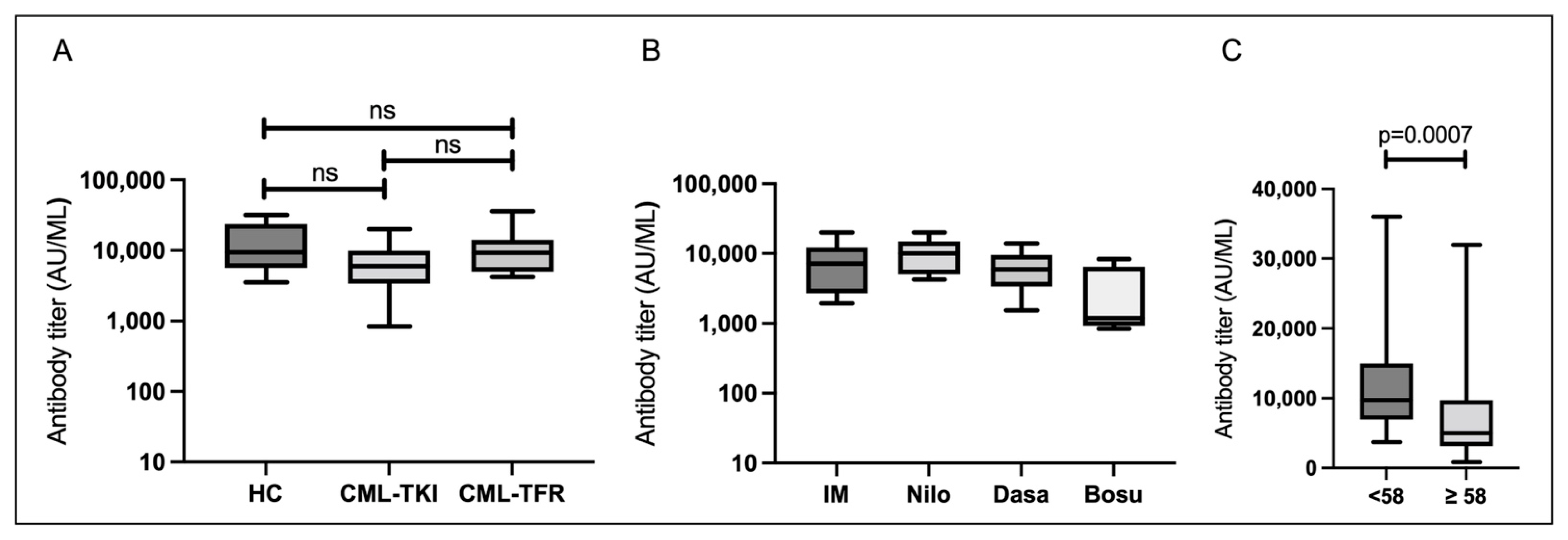
3.1. Characteristics of Participants
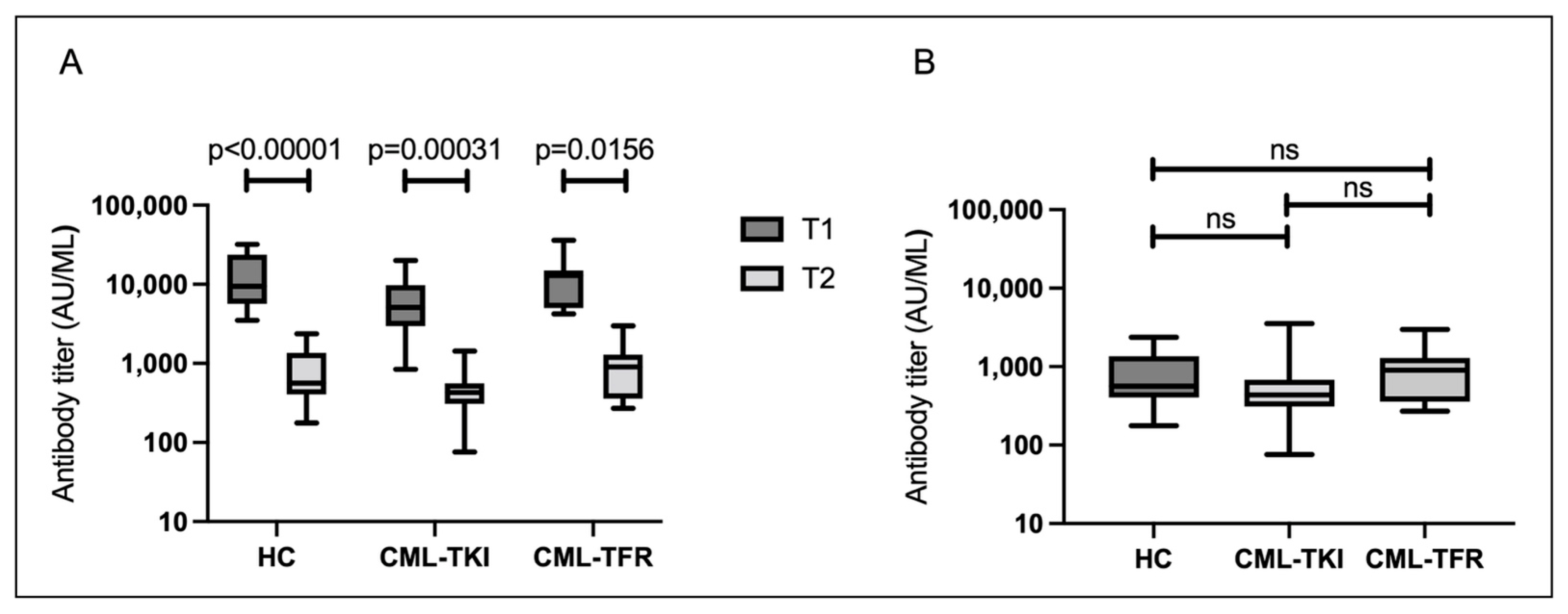
3.2. Serological Responses 1–5 Weeks after the Second Vaccination (T1)
3.3. Serological Responses Approximately 6 Months after the Second Vaccination (T2)
3.4. Molecular Response of CML at T2 in CML-TKI and CML-TFR Groups
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cohen, C.; Kleynhans, J.; von Gottberg, A.; McMorrow, M.L.; Wolter, N.; Bhiman, J.N.; Moyes, J.; du Plessis, M.; Carrim, M.; Buys, A.; et al. SARS-CoV-2 incidence, transmission, and reinfection in a rural and an urban setting: Results of the PHIRST-C cohort study, South Africa, 2020–2021. Lancet Infect. Dis. 2022, 22, 821–834. [Google Scholar] [CrossRef]
- Vijenthira, A.; Gong, I.Y.; Fox, T.A.; Booth, S.; Cook, G.; Fattizzo, B.; Martín-Moro, F.; Razanamahery, J.; Riches, J.C.; Zwicker, J.; et al. Outcomes of patients with hematologic malignancies and COVID-19: A systematic review and meta-analysis of 3377 patients. Blood 2020, 136, 2881–2892. [Google Scholar] [CrossRef] [PubMed]
- Passamonti, F.; Cattaneo, C.; Arcaini, L.; Bruna, R.; Cavo, M.; Merli, F.; Angelucci, E.; Krampera, M.; Cairoli, R.; Della Porta, M.G.; et al. Clinical characteristics and risk factors associated with COVID-19 severity in patients with haematological malignancies in Italy: A retrospective, multicentre, cohort study. Lancet Haematol. 2020, 7, e737–e745. [Google Scholar] [CrossRef]
- Lauring, A.S.; Tenforde, M.W.; Chappell, J.D.; Gaglani, M.; Ginde, A.A.; McNeal, T.; Ghamande, S.; Douin, D.J.; Talbot, H.K.; Casey, J.D.; et al. Clinical severity of, and effectiveness of mRNA vaccines against, covid-19 from omicron, delta, and alpha SARS-CoV-2 variants in the United States: Prospective observational study. Bmj 2022, 376, e069761. [Google Scholar] [CrossRef] [PubMed]
- Dooling, K.; Gargano, J.W.; Moulia, D.; Wallace, M.; Rosenblum, H.G.; Blain, A.E.; Hadler, S.C.; Plumb, I.D.; Moline, H.; Gerstein, J.; et al. Use of Pfizer-BioNTech COVID-19 vaccine in persons aged ≥16 years: Recommendations of the Advisory Committee on Immunization Practices—United States, September 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1344–1348. [Google Scholar] [CrossRef] [PubMed]
- Peeling, R.W.; Olliaro, P.L.; Boeras, D.I.; Fongwen, N. Scaling up COVID-19 rapid antigen tests: Promises and challenges. Lancet Infect. Dis. 2021, 21, e290–e295. [Google Scholar] [CrossRef]
- Liu, R.; He, L.; Hu, Y.; Luo, Z.; Zhang, J. A serological aptamer-assisted proximity ligation assay for COVID-19 diagnosis and seeking neutralizing aptamers. Chem. Sci. 2020, 11, 12157–12164. [Google Scholar] [CrossRef]
- Narasimhan, M.; Mahimainathan, L.; Araj, E.; Clark, A.E.; Markantonis, J.; Green, A.; Xu, J.; SoRelle, J.A.; Alexis, C.; Fankhauser, K.; et al. Clinical evaluation of the Abbott Alinity SARS-CoV-2 Spike-Specific Quantitative IgG and IgM Assays among infected, recovered, and vaccinated groups. J. Clin. Microbiol. 2021, 59, e0038821. [Google Scholar] [CrossRef]
- Hall, V.; Foulkes, S.; Insalata, F.; Kirwan, P.; Saei, A.; Atti, A.; Wellington, E.; Khawam, J.; Munro, K.; Cole, M.; et al. Protection against SARS-CoV-2 after Covid-19 vaccination and previous infection. N. Engl. J. Med. 2022, 386, 1207–1220. [Google Scholar] [CrossRef]
- Tut, G.; Lancaster, T.; Krutikov, M.; Sylla, P.; Bone, D.; Kaur, N.; Spalkova, E.; Bentley, C.; Amin, U.; Jadir, A.T.; et al. Profile of humoral and cellular immune responses to single doses of BNT162b2 or ChAdOx1 nCoV-19 vaccines in residents and staff within residential care homes (VIVALDI): An observational study. Lancet Healthy Longev. 2021, 2, e544–e553. [Google Scholar] [CrossRef]
- Feikin, D.R.; Higdon, M.M.; Abu-Raddad, L.J.; Andrews, N.; Araos, R.; Goldberg, Y.; Groome, M.J.; Huppert, A.; O’Brien, K.L.; Smith, P.G.; et al. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: Results of a systematic review and meta-regression. Lancet 2022, 399, 924–944. [Google Scholar] [CrossRef]
- Hughes, T.P.; Hochhaus, A.; Branford, S.; Müller, M.C.; Kaeda, J.S.; Foroni, L.; Druker, B.J.; Guilhot, F.; Larson, R.A.; O’Brien, S.G.; et al. Long-term prognostic significance of early molecular response to imatinib in newly diagnosed chronic myeloid leukemia: An analysis from the International Randomized Study of Interferon and STI571 (IRIS). Blood 2010, 116, 3758–3765. [Google Scholar] [CrossRef]
- Branford, S.; Yeung, D.T.; Ross, D.M.; Prime, J.A.; Field, C.R.; Altamura, H.K.; Yeoman, A.L.; Georgievski, J.; Jamison, B.A.; Phillis, S.; et al. Early molecular response and female sex strongly predict stable undetectable BCR-ABL1, the criteria for imatinib discontinuation in patients with CML. Blood 2013, 121, 3818–3824. [Google Scholar] [CrossRef]
- Ross, D.M.; Branford, S.; Seymour, J.F.; Schwarer, A.P.; Arthur, C.; Yeung, D.T.; Dang, P.; Goyne, J.M.; Slader, C.; Filshie, R.J.; et al. Safety and efficacy of imatinib cessation for CML patients with stable undetectable minimal residual disease: Results from the TWISTER study. Blood 2013, 122, 515–522. [Google Scholar] [CrossRef]
- Mahon, F.X.; Réa, D.; Guilhot, J.; Guilhot, F.; Huguet, F.; Nicolini, F.; Legros, L.; Charbonnier, A.; Guerci, A.; Varet, B.; et al. Discontinuation of imatinib in patients with chronic myeloid leukaemia who have maintained complete molecular remission for at least 2 years: The prospective, multicentre Stop Imatinib (STIM) trial. Lancet Oncol. 2010, 11, 1029–1035. [Google Scholar] [CrossRef]
- Saussele, S.; Richter, J.; Guilhot, J.; Gruber, F.X.; Hjorth-Hansen, H.; Almeida, A.; Janssen, J.; Mayer, J.; Koskenvesa, P.; Panayiotidis, P.; et al. Discontinuation of tyrosine kinase inhibitor therapy in chronic myeloid leukaemia (EURO-SKI): A prespecified interim analysis of a prospective, multicentre, non-randomised, trial. Lancet Oncol. 2018, 19, 747–757. [Google Scholar] [CrossRef]
- Takahashi, N.; Nishiwaki, K.; Nakaseko, C.; Aotsuka, N.; Sano, K.; Ohwada, C.; Kuroki, J.; Kimura, H.; Tokuhira, M.; Mitani, K.; et al. Treatment-free remission after two-year consolidation therapy with nilotinib in patients with chronic myeloid leukemia: STAT2 trial in Japan. Haematologica 2018, 103, 1835–1842. [Google Scholar] [CrossRef]
- Takahashi, N.; Tauchi, T.; Kitamura, K.; Miyamura, K.; Saburi, Y.; Hatta, Y.; Miyata, Y.; Kobayashi, S.; Usuki, K.; Matsumura, I.; et al. Deeper molecular response is a predictive factor for treatment-free remission after imatinib discontinuation in patients with chronic phase chronic myeloid leukemia: The JALSG-STIM213 study. Int. J. Hematol. 2018, 107, 185–193. [Google Scholar] [CrossRef]
- Kimura, S.; Imagawa, J.; Murai, K.; Hino, M.; Kitawaki, T.; Okada, M.; Tanaka, H.; Shindo, M.; Kumagai, T.; Ikezoe, T.; et al. Treatment-free remission after first-line dasatinib discontinuation in patients with chronic myeloid leukaemia (first-line DADI trial): A single-arm, multicentre, phase 2 trial. Lancet Haematol. 2020, 7, e218–e225. [Google Scholar] [CrossRef]
- Ohyashiki, K.; Katagiri, S.; Tauchi, T.; Ohyashiki, J.H.; Maeda, Y.; Matsumura, I.; Kyo, T. Increased natural killer cells and decreased CD3(+)CD8(+)CD62L(+) T cells in CML patients who sustained complete molecular remission after discontinuation of imatinib. Br. J. Haematol. 2012, 157, 254–256. [Google Scholar] [CrossRef]
- Hughes, A.; Yong, A.S.M. Immune effector recovery in chronic myeloid leukemia and treatment-free remission. Front. Immunol. 2017, 8, 469. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, Y.; Sugiyama, D.; Matsumura, I.; Minami, Y.; Miura, M.; Atsuta, Y.; Ohtake, S.; Kiyoi, H.; Miyazaki, Y.; Nishikawa, H.; et al. Regulatory T cell as a biomarker of treatment-free remission in patients with chronic myeloid leukemia. Cancers 2021, 13, 5904. [Google Scholar] [CrossRef] [PubMed]
- Claudiani, S.; Apperley, J.F.; Parker, E.L.; Marchesin, F.; Katsanovskaja, K.; Palanicawandar, R.; Innes, A.J.; Tedder, R.S.; McClure, M.O.; Milojkovic, D. Durable humoral responses after the second anti-SARS-CoV-2 vaccine dose in chronic myeloid leukaemia patients on tyrosine kinase inhibitors. Br. J. Haematol. 2022, 197, e1–e4. [Google Scholar] [CrossRef]
- Harrington, P.; Doores, K.J.; Radia, D.; O’Reilly, A.; Lam, H.P.J.; Seow, J.; Graham, C.; Lechmere, T.; McLornan, D.; Dillon, R.; et al. Single dose of BNT162b2 mRNA vaccine against severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) induces neutralising antibody and polyfunctional T-cell responses in patients with chronic myeloid leukaemia. Br. J. Haematol. 2021, 194, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Herzog Tzarfati, K.; Gutwein, O.; Apel, A.; Rahimi-Levene, N.; Sadovnik, M.; Harel, L.; Benveniste-Levkovitz, P.; Bar Chaim, A.; Koren-Michowitz, M. BNT162b2 COVID-19 vaccine is significantly less effective in patients with hematologic malignancies. Am. J. Hematol. 2021, 96, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Malard, F.; Gaugler, B.; Gozlan, J.; Bouquet, L.; Fofana, D.; Siblany, L.; Eshagh, D.; Adotevi, O.; Laheurte, C.; Ricard, L.; et al. Weak immunogenicity of SARS-CoV-2 vaccine in patients with hematologic malignancies. Blood Cancer J. 2021, 11, 142. [Google Scholar] [CrossRef] [PubMed]
- Maneikis, K.; Šablauskas, K.; Ringelevičiūtė, U.; Vaitekėnaitė, V.; Čekauskienė, R.; Kryžauskaitė, L.; Naumovas, D.; Banys, V.; Pečeliūnas, V.; Beinortas, T.; et al. Immunogenicity of the BNT162b2 COVID-19 mRNA vaccine and early clinical outcomes in patients with haematological malignancies in Lithuania: A national prospective cohort study. Lancet Haematol. 2021, 8, e583–e592. [Google Scholar] [CrossRef]
- Avivi, I.; Balaban, R.; Shragai, T.; Sheffer, G.; Morales, M.; Aharon, A.; Lowenton-Spier, N.; Trestman, S.; Perry, C.; Benyamini, N.; et al. Humoral response rate and predictors of response to BNT162b2 mRNA COVID19 vaccine in patients with multiple myeloma. Br. J. Haematol. 2021, 195, 186–193. [Google Scholar] [CrossRef]
- Mori, A.; Onozawa, M.; Tsukamoto, S.; Ishio, T.; Yokoyama, E.; Izumiyama, K.; Saito, M.; Muraki, H.; Morioka, M.; Teshima, T.; et al. Humoral response to mRNA-based COVID-19 vaccine in patients with myeloid malignancies. Br. J. Haematol. 2022, 197, 691–696. [Google Scholar] [CrossRef]
- Deininger, M.W.; Shah, N.P.; Altman, J.K.; Berman, E.; Bhatia, R.; Bhatnagar, B.; DeAngelo, D.J.; Gotlib, J.; Hobbs, G.; Maness, L.; et al. Chronic Myeloid Leukemia, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2020, 18, 1385–1415. [Google Scholar] [CrossRef]
- de Lavallade, H.; Khoder, A.; Hart, M.; Sarvaria, A.; Sekine, T.; Alsuliman, A.; Mielke, S.; Bazeos, A.; Stringaris, K.; Ali, S.; et al. Tyrosine kinase inhibitors impair B-cell immune responses in CML through off-target inhibition of kinases important for cell signaling. Blood 2013, 122, 227–238. [Google Scholar] [CrossRef]
- Rajala, H.L.M.; Missiry, M.E.; Ruusila, A.; Koskenvesa, P.; Brümmendorf, T.H.; Gjertsen, B.T.; Janssen, J.; Lotfi, K.; Markevärn, B.; Olsson-Strömberg, U.; et al. Tyrosine kinase inhibitor therapy-induced changes in humoral immunity in patients with chronic myeloid leukemia. J. Cancer Res. Clin. Oncol. 2017, 143, 1543–1554. [Google Scholar] [CrossRef]
- Totadri, S.; Thipparapu, S.; Aggarwal, R.; Sharma, M.; Naseem, S.; Jain, R.; Trehan, A.; Malhotra, P.; Varma, N.; Bansal, D. Imatinib-induced hypogammaglobulinemia in children and adolescents with chronic myeloid leukemia. Pediatr. Hematol. Oncol. 2020, 37, 539–544. [Google Scholar] [CrossRef]
- Bettoni da Cunha-Riehm, C.; Hildebrand, V.; Nathrath, M.; Metzler, M.; Suttorp, M. Vaccination with live attenuated vaccines in four children with chronic myeloid leukemia while on imatinib treatment. Front. Immunol. 2020, 11, 628. [Google Scholar] [CrossRef]
- de Lavallade, H.; Garland, P.; Sekine, T.; Hoschler, K.; Marin, D.; Stringaris, K.; Loucaides, E.; Howe, K.; Szydlo, R.; Kanfer, E.; et al. Repeated vaccination is required to optimize seroprotection against H1N1 in the immunocompromised host. Haematologica 2011, 96, 307–314. [Google Scholar] [CrossRef]
- Seggewiss, R.; Loré, K.; Greiner, E.; Magnusson, M.K.; Price, D.A.; Douek, D.C.; Dunbar, C.E.; Wiestner, A. Imatinib inhibits T-cell receptor-mediated T-cell proliferation and activation in a dose-dependent manner. Blood 2005, 105, 2473–2479. [Google Scholar] [CrossRef]
- Mumprecht, S.; Matter, M.; Pavelic, V.; Ochsenbein, A.F. Imatinib mesylate selectively impairs expansion of memory cytotoxic T cells without affecting the control of primary viral infections. Blood 2006, 108, 3406–3413. [Google Scholar] [CrossRef]
- English, E.; Cook, L.E.; Piec, I.; Dervisevic, S.; Fraser, W.D.; John, W.G. Performance of the Abbott SARS-CoV-2 IgG II quantitative antibody assay including the new variants of concern, VOC 202012/V1 (United Kingdom) and VOC 202012/V2 (South Africa), and first steps towards global harmonization of COVID-19 antibody methods. J. Clin. Microbiol. 2021, 59, e0028821. [Google Scholar] [CrossRef]
- Jung, K.; Shin, S.; Nam, M.; Hong, Y.J.; Roh, E.Y.; Park, K.U.; Song, E.Y. Performance evaluation of three automated quantitative immunoassays and their correlation with a surrogate virus neutralization test in coronavirus disease 19 patients and pre-pandemic controls. J. Clin. Lab. Anal. 2021, 35, e23921. [Google Scholar] [CrossRef]
- How, J.; Gallagher, K.M.E.; Liu, Y.; Katsis, K.; Elder, E.L.; Larson, R.C.; Leick, M.B.; Neuberg, D.; Maus, M.V.; Hobbs, G.S. Antibody and T-cell responses to SARS-CoV-2 vaccination in myeloproliferative neoplasm patients. Leukemia 2022, 36, 1176–1179. [Google Scholar] [CrossRef]
- Šušol, O.; Hájková, B.; Zelená, H.; Hájek, R. Third dose of COVID-19 vaccine restores immune response in patients with haematological malignancies after loss of protective antibody titres. Br. J. Haematol. 2022, 197, 302–305. [Google Scholar] [CrossRef] [PubMed]
| HC (n = 16) | CML-TKI (n = 32) | CML-TFR (n = 10) | |
|---|---|---|---|
| Age (years), median (range) | 45 (30–92) | 62 (29–81) | 61.5 (47–80) |
| Sex (male/female) | 8/8 | 20/12 | 6/4 |
| TKI (administration or discontinuation) | |||
| Imatinib | 6 | 7 | |
| Nilotinib | 9 | 2 | |
| Dasatinib | 12 | 1 | |
| Bosutinib | 5 | 0 | |
| TKI dose (mg), median (range) | |||
| Imatinib | 350 (100–600) | ||
| Nilotinib | 600 (150–600) | ||
| Dasatinib | 60 (20–100) | ||
| Bosutinib | 200 (100–400) | ||
| Duration of TFR (months), median (range) | 68.5 (31–130) | ||
| CML response according to ELN criteria | |||
| Non-MMR | 1 | 0 | |
| MMR | 3 | 0 | |
| MR4.0 | 11 | 1 | |
| MR4.5 | 2 | 2 | |
| Undetectable | 15 | 7 | |
| Duration from 2nd VC to T1 (days) | 27 (15–30) * | 17.5 (7–35) | 25.5 (8–31) |
| Duration from 2nd VC to T2 (days) | 204 (190–215) * | 174 (151–207) | 174 (164–183) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katagiri, S.; Akahane, D.; Otsuki, S.; Suto, A.; Yamada, A.; Suguro, T.; Asano, M.; Yoshizawa, S.; Tanaka, Y.; Furuya, N.; et al. Tyrosine Kinase Inhibitors Do Not Promote a Decrease in SARS-CoV-2 Anti-Spike IgG after BNT162b2 Vaccination in Chronic Myeloid Leukemia: A Prospective Observational Study. Vaccines 2022, 10, 1404. https://doi.org/10.3390/vaccines10091404
Katagiri S, Akahane D, Otsuki S, Suto A, Yamada A, Suguro T, Asano M, Yoshizawa S, Tanaka Y, Furuya N, et al. Tyrosine Kinase Inhibitors Do Not Promote a Decrease in SARS-CoV-2 Anti-Spike IgG after BNT162b2 Vaccination in Chronic Myeloid Leukemia: A Prospective Observational Study. Vaccines. 2022; 10(9):1404. https://doi.org/10.3390/vaccines10091404
Chicago/Turabian StyleKatagiri, Seiichiro, Daigo Akahane, Shunsuke Otsuki, Arisa Suto, Akiko Yamada, Tamiko Suguro, Michiyo Asano, Seiichiro Yoshizawa, Yuko Tanaka, Nahoko Furuya, and et al. 2022. "Tyrosine Kinase Inhibitors Do Not Promote a Decrease in SARS-CoV-2 Anti-Spike IgG after BNT162b2 Vaccination in Chronic Myeloid Leukemia: A Prospective Observational Study" Vaccines 10, no. 9: 1404. https://doi.org/10.3390/vaccines10091404
APA StyleKatagiri, S., Akahane, D., Otsuki, S., Suto, A., Yamada, A., Suguro, T., Asano, M., Yoshizawa, S., Tanaka, Y., Furuya, N., Fujimoto, H., Okabe, S., Gotoh, M., Ito, Y., & Gotoh, A. (2022). Tyrosine Kinase Inhibitors Do Not Promote a Decrease in SARS-CoV-2 Anti-Spike IgG after BNT162b2 Vaccination in Chronic Myeloid Leukemia: A Prospective Observational Study. Vaccines, 10(9), 1404. https://doi.org/10.3390/vaccines10091404






