Cytotoxic, Antimicrobial, Antioxidant Properties and Effects on Cell Migration of Phenolic Compounds of Selected Transylvanian Medicinal Plants
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Chemicals
2.2. Studied Plant Taxa and Plant Extraction
2.3. Analyses of Phenolic Compounds by HPLC with Diode-array Detector and Electrospray Ionization with MS
2.3.1. HPLC Conditions
2.3.2. MS Conditions
2.4. Determination of Minimum Inhibitory Concentration (MIC80) with Microdilution Method
2.5. Total Antioxidant Capacity (TAC) Assays
2.5.1. Oxygen Radical Absorbance Capacity (ORAC) Assay
2.5.2. Enhanced Chemiluminescence (ECL) Assay
2.5.3. 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Radical Scavenging Assay
2.5.4. Trolox Equivalent Antioxidant Capacity (TEAC) Assay
2.5.5. Calculation of Total Antioxidant Capacities (TAC)
2.6. Cell Cultures
2.7. Quantification of Intracellular ROS
2.8. Plate Reader Cytotoxicity Test
2.9. Flow Cytometric Cytotoxicity Test
2.10. In Vitro “Wound Healing” Assay
2.11. Statistical Analyses
3. Results
3.1. Qualitative Analysis of Phenolic Compounds in Plant Extracts with LC-DAD-ESI-MS/MS
3.2. Determination of Minimum Inhibitory Concentration (MIC80)
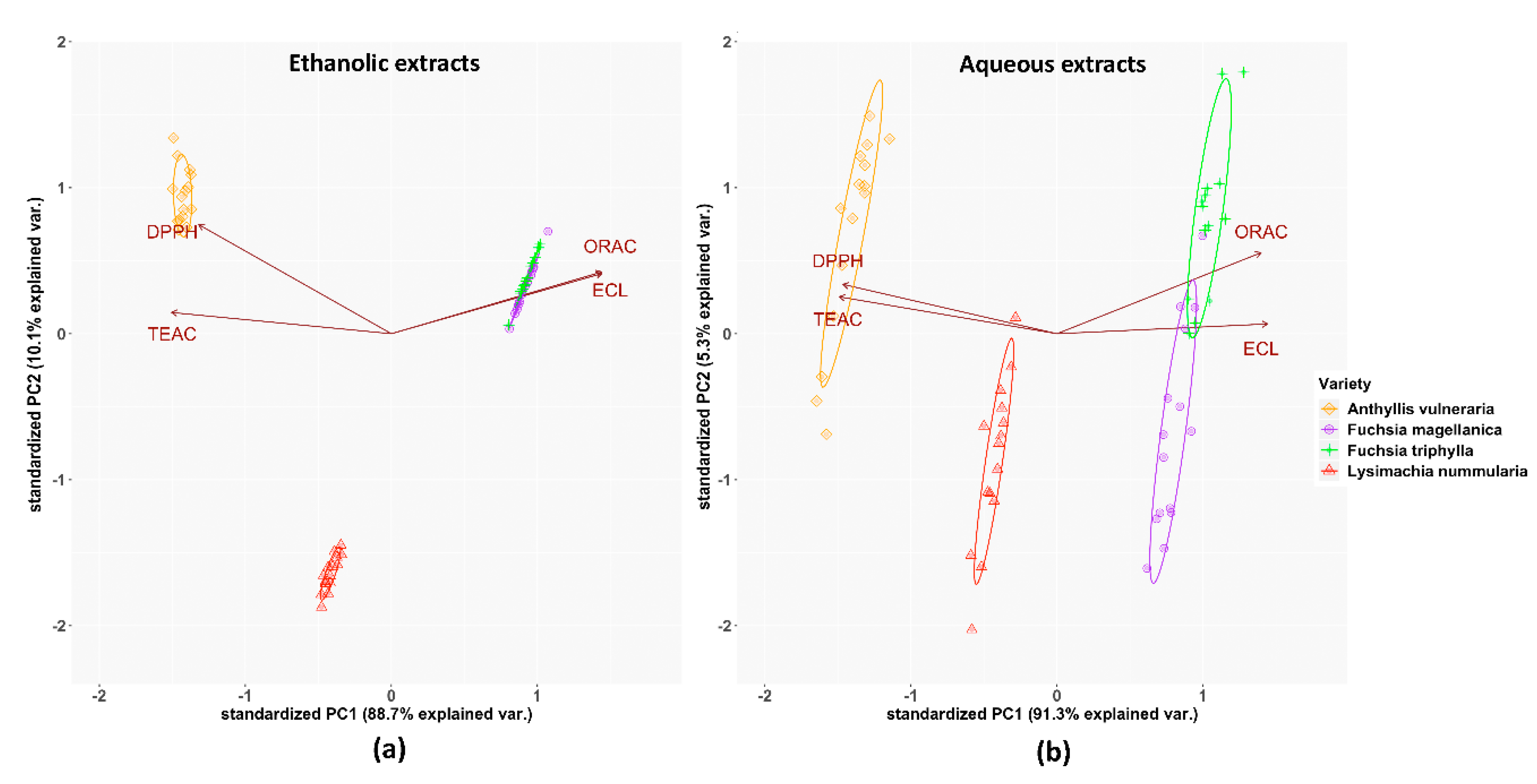
3.3. Total Antioxidant Capacity (TAC) Assays
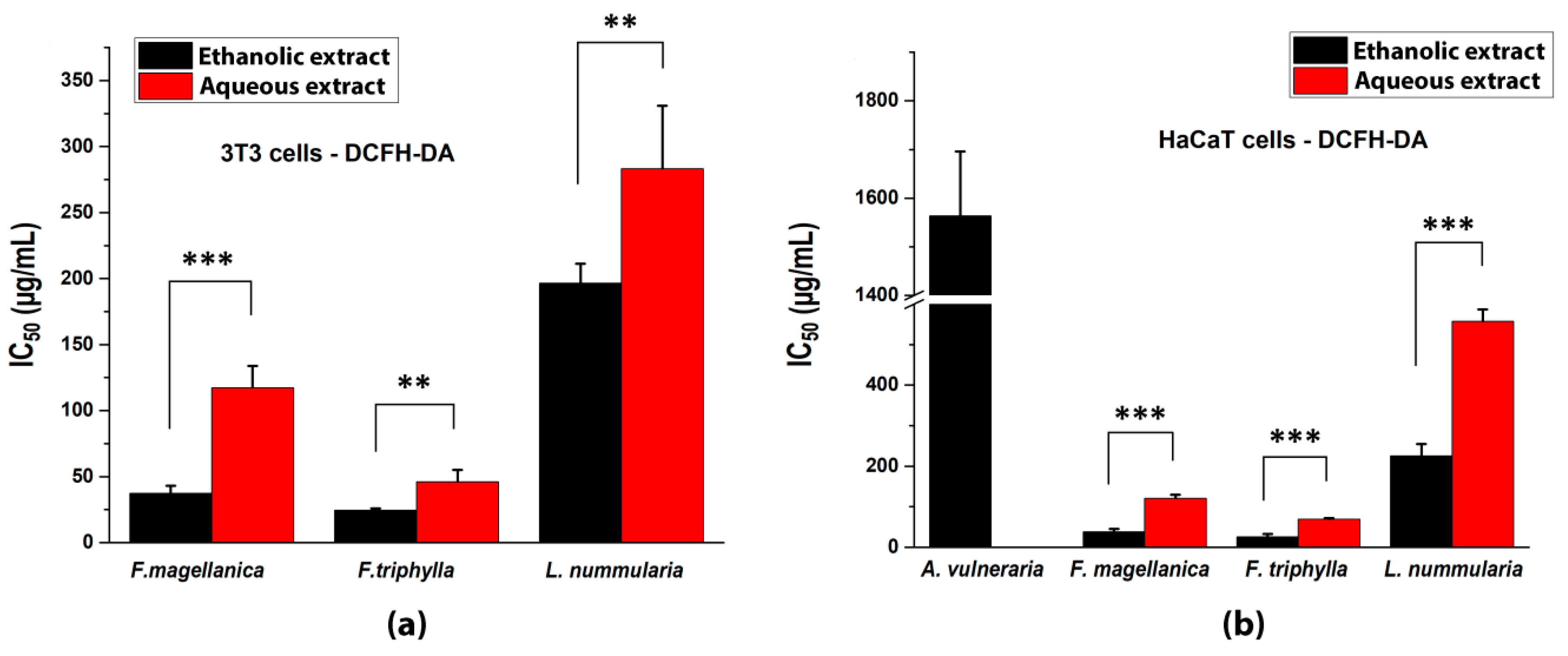
3.4. Inhibition of Intracellular ROS Production
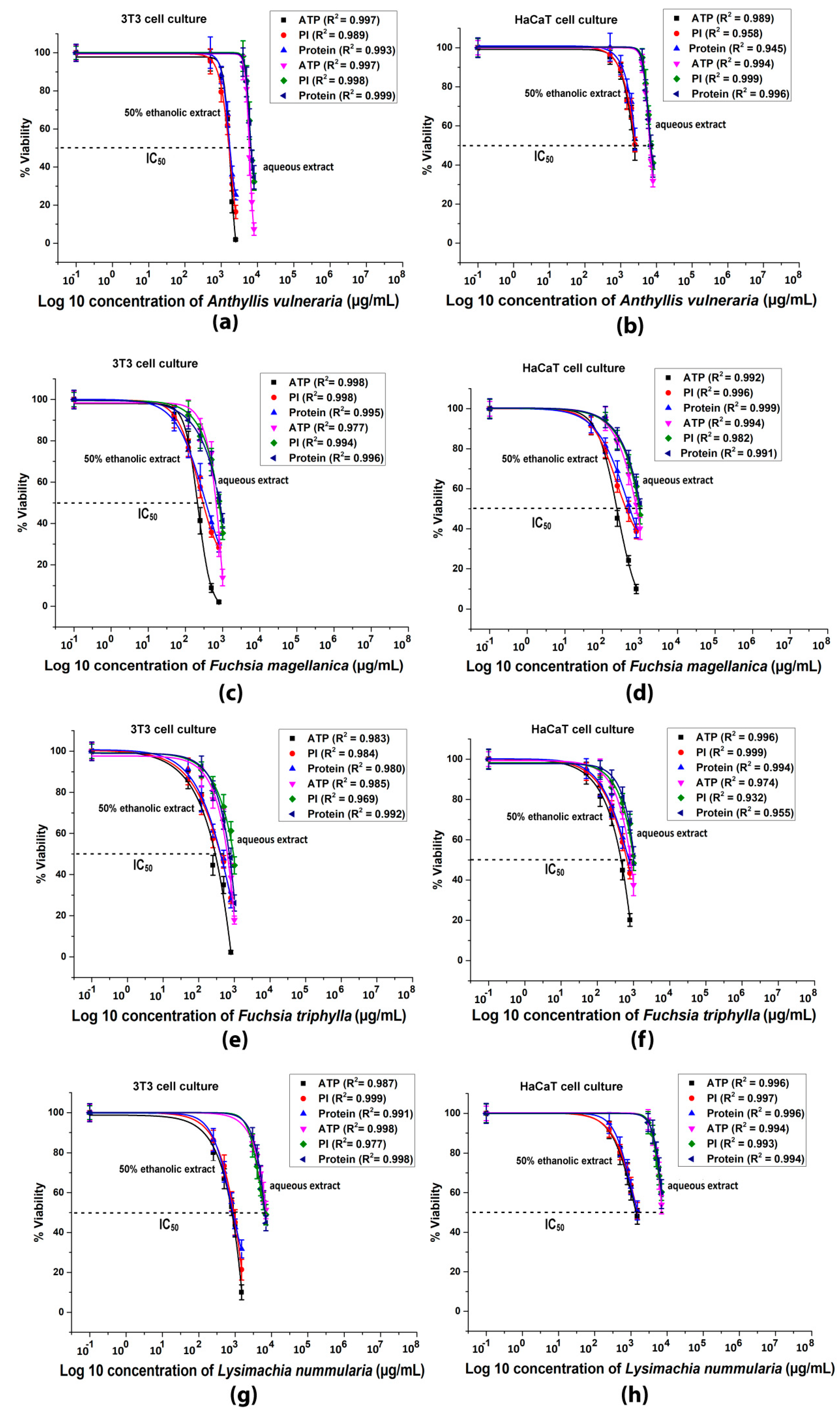
3.5. Plate Reader Cytotoxicity Tests
3.6. Flow Cytometric Cytotoxicity Test
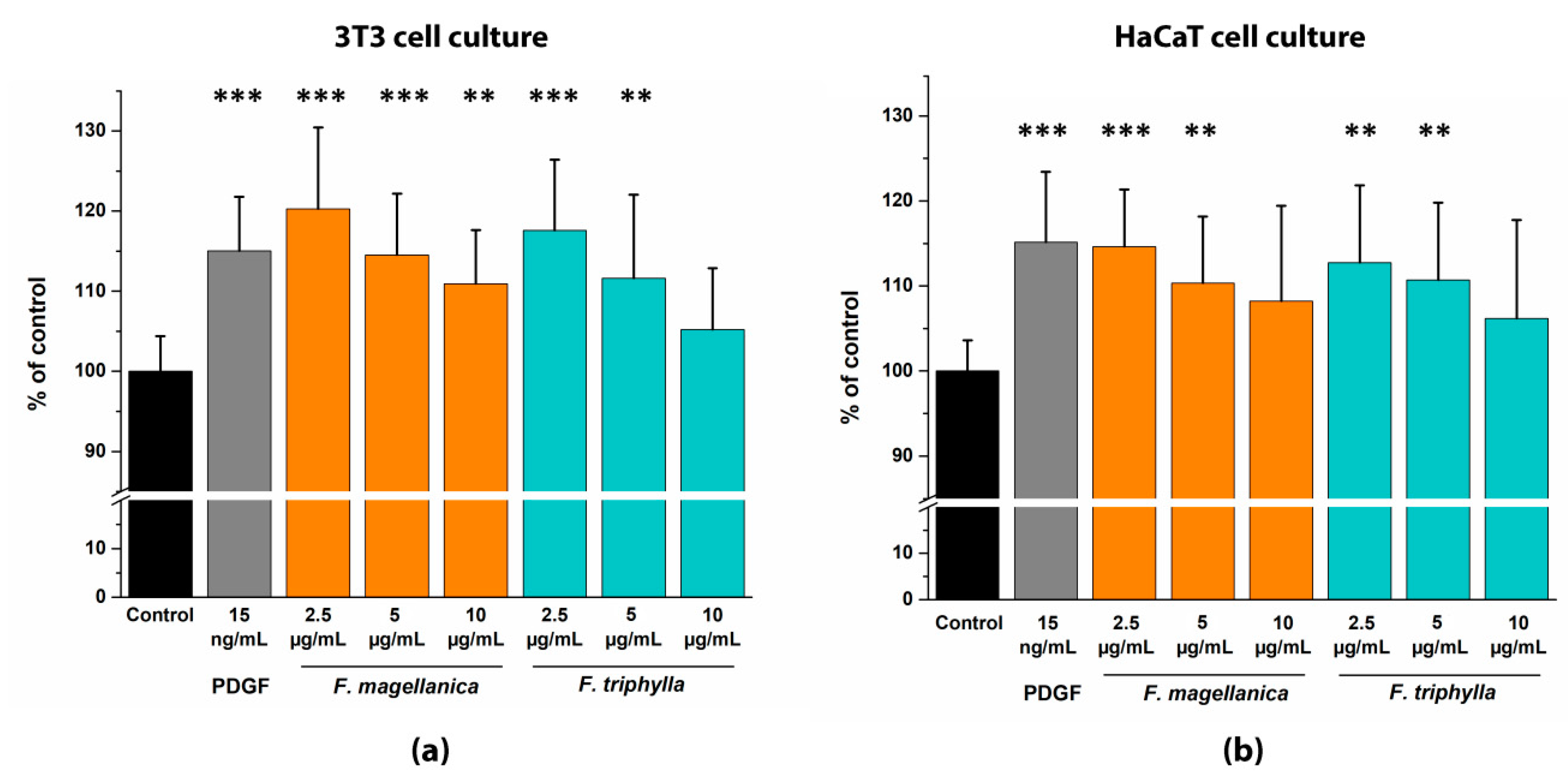
3.7. In vitro Migration Test
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Patay, E.B.; Sali, N.; Koszegi, T.; Csepregi, R.; Balazs, V.L.; Nemeth, T.S.; Nemeth, T.; Papp, N. Antioxidant potential, tannin and polyphenol contents of seed and pericarp of three Coffea species. Asian Pac. J. Trop. Med. 2016, 9, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Csepregi, K.; Neugart, S.; Schreiner, M.; Hideg, E. Comparative Evaluation of Total Antioxidant Capacities of Plant Polyphenols. Molecules 2016, 21, 208. [Google Scholar] [CrossRef] [PubMed]
- Maqsood, S.; Benjakul, S. Comparative studies of four different phenolic compounds on in vitro antioxidative activity and the preventive effect on lipid oxidation of fish oil emulsion and fish mince. Food Chem. 2010, 119, 123–132. [Google Scholar] [CrossRef]
- Tutin, T.; Heywood, V.; Burges, N.; Moore, D.; Valentine, D.; Walters, S.; Webb, D. Flora Europaea, 2nd ed.; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Szabó, L.G. Medicinal Plants; Schmidt und Co. Ltd. Melius Foundation: Baksa-Pecs, Hungary, 2005. (In Hungarian) [Google Scholar]
- Butura, V. Ethnobotanical Encyclopaedia of Romania; Editura Ştiinţifică şi Enciclopedică: Bucharest, Romania, 1979. (In Hungarian) [Google Scholar]
- Borza, A. Dictionary of Ethnobotany; Editura Academiei Republicii Socialiste Romania: Bucharest, Romania, 1968. (In Romanian) [Google Scholar]
- Makay, B. Handbook of Healing with Plants; Felső-Magyarországi Kiadó: Nyíregyháza, Hungary, 1994. (In Hungarian) [Google Scholar]
- Péntek, J.; Szabó, T.A. People and Plants. Ethnobotanical Knowledge of Rural People in Tara Călatei; Kriterion Publ. House: Bucharest, Romania, 1985. (In Hungarian) [Google Scholar]
- Gonnett, J.-F.; Jay, M. Les aglycones flavoniques d’Anthyllis vulneraria. Phytochemistry 1972, 11, 2313–2316. [Google Scholar] [CrossRef]
- Nartowska, J.; Wawer, I.; Strzelecka, H. Triterpenoid sapogenin from Anthyllis vulneraria L. Acta Pol. Pharm. 2001, 58, 289–291. [Google Scholar]
- Ghalem, M.; Merghache, S.; Ghalem, S.; Belarbi, M. Phenolic contents and in vitro antioxidant activity of some secondary metabolites of Anthyllis vulneraria L. from Algeria. Int. J. Med. Pharm. Sci. 2012, 2, 51–64. [Google Scholar]
- Halász, P. Plants in the Everyday Life and Traditions of Moldovan Hungarians; General Press: Budapest, Hungary, 2010. (In Hungarian) [Google Scholar]
- Papp, N.; Horváth, D. Traditionns and Ethnomedicine in Crăciunel; Homoródkarácsonyfalvi Közbirtokosság: Homoródkarácsonyfalva, Romania, 2016. (In Hungarian) [Google Scholar]
- Rácz, G.; Füzi, J. Medicinal Plants in Covasna County; Directorate of Agriculture and Food Industry: Sepsiszentgyörgy, Romania, 1973. (In Hungarian) [Google Scholar]
- Gub, J. Ethnobotanical Data along the Nagy-Homoród. In Plants in the Folklore; Zsigmond, G., Ed.; Magyar Köztársaság Kulturális Intézete: Bucharest, Romania, 2005. (In Hungarian) [Google Scholar]
- Papp, N. Traditions and Ethnomedicine in Lueat; Lövétei Közbirtokossága és Polgármestei Hivatal: Lövéte, Romania, 2018. (In Hungarian) [Google Scholar]
- Toth, A.; Toth, G.; Kery, A. Polyphenol composition and antioxidant capacity of three Lysimachia species. Nat. Prod. Commun. 2014, 9, 1473–1478. [Google Scholar] [CrossRef]
- Podolak, I.; Koczurkiewicz, P.; Galanty, A.; Michalik, M. Cytotoxic triterpene saponins from the underground parts of six Lysimachia L. species. Biochem. Syst. Ecol. 2013, 47, 116–120. [Google Scholar] [CrossRef]
- Boris, G. Ethnobotanical Use of Medicinal Plants in Lueta. Bachelor’s Thesis, University of Pécs, Pécs, Hungary, 2010. (In Hungarian). [Google Scholar]
- Crowden, R.K.; Wright, J.; Harborne, J.B. Anthocyanins of Fuchsia (Onagraceae). Phytochemistry 1977, 16, 400–402. [Google Scholar] [CrossRef]
- Ruiz, A.; Hermosín-Gutiérrez, I.; Vergara, C.; Baer, D.; Zapata, M.; Hitschfeld, A.; Obando, L.; Mardones, C. Anthocyanin profiles in south Patagonian wild berries by HPLC-DAD-ESI-MS/MS. Food Res. Int. 2013, 51, 706–713. [Google Scholar] [CrossRef]
- Williams, C.A.; Garnock-Jones, P.J. Leaf flavonoids and other phenolic glycosides and the taxonomy and phylogeny of Fuchsia sect. skinnera (Onagraceae). Phytochemistry 1986, 25, 2547–2549. [Google Scholar] [CrossRef]
- Ghosh, P.K.; Gaba, A. Phyto-extracts in wound healing. J. Pharm. Pharm. Sci. Publ. Can. Soc. Pharm. Sci. Soc. Can. Sci. Pharm. 2013, 16, 760–820. [Google Scholar] [CrossRef] [PubMed]
- Adetutu, A.; Morgan, W.A.; Corcoran, O. Antibacterial, antioxidant and fibroblast growth stimulation activity of crude extracts of Bridelia ferruginea leaf, a wound-healing plant of Nigeria. J. Ethnopharmacol. 2011, 133, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Süntar, I.; Akkol, E.K.; Nahar, L.; Sarker, S.D. Wound healing and antioxidant properties: Do they coexist in plants? Free Radic. Antioxid. 2012, 2, 1–7. [Google Scholar] [CrossRef]
- Behm, B.; Babilas, P.; Landthaler, M.; Schreml, S. Cytokines, chemokines and growth factors in wound healing. J. Eur. Acad. Dermatol. Venereol. JEADV 2012, 26, 812–820. [Google Scholar] [CrossRef]
- Csepregi, R.; Temesfoi, V.; Poor, M.; Faust, Z.; Koszegi, T. Green Fluorescent Protein-Based Viability Assay in a Multiparametric Configuration. Molecules 2018, 23, 1575. [Google Scholar] [CrossRef]
- Lee, J.H.; Cho, S.; Paik, H.D.; Choi, C.W.; Nam, K.T.; Hwang, S.G.; Kim, S.K. Investigation on antibacterial and antioxidant activities, phenolic and flavonoid contents of some thai edible plants as an alternative for antibiotics. Asian-Australas. J. Anim. 2014, 27, 1461–1468. [Google Scholar] [CrossRef]
- Das, S.; Gazdag, Z.; Szente, L.; Meggyes, M.; Horvath, G.; Lemli, B.; Kunsagi-Mate, S.; Kuzma, M.; Koszegi, T. Antioxidant and antimicrobial properties of randomly methylated beta cyclodextrin-captured essential oils. Food Chem. 2019, 278, 305–313. [Google Scholar] [CrossRef]
- Koszegi, T.; Sali, N.; Raknic, M.; Horvath-Szalai, Z.; Csepregi, R.; Koncic, M.Z.; Papp, N.; Poor, M. A novel luminol-based enhanced chemiluminescence antioxidant capacity microplate assay for use in different biological matrices. J. Pharmacol. Toxicol. Methods 2017, 88, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Bertoncelj, J.; Doberšek, U.; Jamnik, M.; Golob, T. Evaluation of the phenolic content, antioxidant activity and colour of Slovenian honey. Food Chem. 2007, 105, 822–828. [Google Scholar] [CrossRef]
- Beretta, G.; Granata, P.; Ferrero, M.; Orioli, M.; Facino, R.M. Standardization of antioxidant properties of honey by a combination of spectrophotometric/fluorimetric assays and chemometrics. Anal. Chim. Acta 2005, 533, 185–191. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Stratil, P.; Klejdus, B.; Kuban, V. Determination of phenolic compounds and their antioxidant activity in fruits and cereals. Talanta 2007, 71, 1741–1751. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.; Liu, D.; Yu, X.; Sun, H.; Li, Y. A Caco-2 cell-based quantitative antioxidant activity assay for antioxidants. Food Chem. 2015, 175, 601–608. [Google Scholar] [CrossRef]
- Balaiya, S.; Chalam, K.V. An In vitro Assay to Quantify Nitrosative Component of Oxidative Stress. J. Mol. Genet. Med. Int. J. Biomed. Res. 2014, 8. [Google Scholar] [CrossRef]
- Kalyanaraman, B.; Darley-Usmar, V.; Davies, K.J.; Dennery, P.A.; Forman, H.J.; Grisham, M.B.; Mann, G.E.; Moore, K.; Roberts, L.J., 2nd; Ischiropoulos, H. Measuring reactive oxygen and nitrogen species with fluorescent probes: Challenges and limitations. Free Radic. Biol. Med. 2012, 52, 1–6. [Google Scholar] [CrossRef]
- Clifford, M.N.; Johnston, K.L.; Knight, S.; Kuhnert, N. Hierarchical scheme for LC-MSn identification of chlorogenic acids. J. Agric. Food Chem. 2003, 51, 2900–2911. [Google Scholar] [CrossRef]
- Said, R.B.; Hamed, A.I.; Mahalel, U.A.; Al-Ayed, A.S.; Kowalczyk, M.; Moldoch, J.; Oleszek, W.; Stochmal, A. Tentative Characterization of Polyphenolic Compounds in the Male Flowers of Phoenix dactylifera by Liquid Chromatography Coupled with Mass Spectrometry and DFT. Int. J. Mol. Sci. 2017, 18, 512. [Google Scholar] [CrossRef]
- Alberti, A.; Beni, S.; Lacko, E.; Riba, P.; Al-Khrasani, M.; Kery, A. Characterization of phenolic compounds and antinociceptive activity of Sempervivum tectorum L. leaf juice. J. Pharm. Biomed. Anal. 2012, 70, 143–150. [Google Scholar] [CrossRef]
- Lorenz, P.; Conrad, J.; Bertrams, J.; Berger, M.; Duckstein, S.; Meyer, U.; Stintzing, F.C. Investigations into the Phenolic Constituents of Dog’s Mercury (Mercurialis perennis L.) by LC-MS/MS and GC-MS analyses. Phytochem. Anal. 2012, 23, 60–71. [Google Scholar] [CrossRef]
- Abad-García, B.; Berrueta, L.A.; Garmón-Lobato, S.; Gallo, B.; Vicente, F. A general analytical strategy for the characterization of phenolic compounds in fruit juices by high-performance liquid chromatography with diode-array detection coupled to electrospray ionization and triple quadrupole mass spectrometry. J. Chromatogr. A 2009, 1216, 5398–5415. [Google Scholar] [CrossRef] [PubMed]
- Rösch, D.; Krumbein, A.; Mügge, C.; Kroh, L.W. Structural Investigations of Flavonol Glycosides from Sea Buckthorn (Hippophae rhamnoides) Pomace by NMR Spectroscopy and HPLC-ESI-MS. J. Agric. Food Chem. 2004, 52, 4039–4046. [Google Scholar] [CrossRef] [PubMed]
- Simirgiotis, M.J.; Schmeda-Hirschmann, G. Direct identification of phenolic constituents in Boldo Folium (Peumus boldus Mol.) infusions by high-performance liquid chromatography with diode array detection and electrospray ionization tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 443–449. [Google Scholar] [CrossRef]
- Kang, J.; Price, W.E.; Ashton, J.; Tapsell, L.C.; Johnson, S. Identification and characterization of phenolic compounds in hydromethanolic extracts of sorghum wholegrains by LC-ESI-MS. Food Chem. 2016, 211, 215–226. [Google Scholar] [CrossRef]
- Chen, Y.; Yu, H.; Wu, H.; Pan, Y.; Wang, K.; Jin, Y.; Zhang, C. Characterization and Quantification by LC-MS/MS of the Chemical Components of the Heating Products of the Flavonoids Extract in Pollen Typhae for Transformation Rule Exploration. Molecules 2015, 20, 18352–18366. [Google Scholar] [CrossRef] [PubMed]
- Tóth, A.; Riethmüller, E.; Alberti, Á.; Végh, K.; Kéry, Á. Comparative Phytochemical Screening of Phenoloids in Lysimachia Species. Eur. Chem. Bull. 2012, 1, 27–30. [Google Scholar]
- Lin, L.Z.; Harnly, J.M. Identification of the phenolic components of chrysanthemum flower (Chrysanthemum morifolium Ramat). Food Chem. 2010, 120, 319–326. [Google Scholar] [CrossRef]
- de Brito, E.S.; de Araujo, M.C.; Lin, L.Z.; Harnly, J. Determination of the flavonoid components of cashew apple (Anacardium occidentale) by LC-DAD-ESI/MS. Food Chem. 2007, 105, 1112–1118. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, A.; Kumar, B. Identification and characterization of phenolics and terpenoids from ethanolic extracts of Phyllanthus species by HPLC-ESI-QTOF-MS/MS. J. Pharm. Anal. 2017, 7, 214–222. [Google Scholar] [CrossRef]
- Mena, P.; Cirlini, M.; Tassotti, M.; Herrlinger, K.A.; Dall’Asta, C.; Del Rio, D. Phytochemical Profiling of Flavonoids, Phenolic Acids, Terpenoids, and Volatile Fraction of a Rosemary (Rosmarinus officinalis L.) Extract. Molecules 2016, 21, 1576. [Google Scholar] [CrossRef]
- Bouaoudia-Madi, N.; Boulekbache-Makhlouf, L.; Madani, K.; Silva, A.M.S.; Dairi, S.; Oukhmanou-Bensidhoum, S.; Cardoso, S.M. Optimization of Ultrasound-Assisted Extraction of Polyphenols from Myrtus communis L. Pericarp. Antioxidants 2019, 8, 205. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, C.; Li, H.; Yu, T.; Tan, L. Simultaneous Determination of Formononetin, Calycosin and Rhamnocitrin from Astragalus Complanatus by UHPLC-MS-MS in Rat Plasma: Application to a Pharmacokinetic Study. J. Chromatogr. Sci. 2016, 54, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Prior, R.L. Identification and characterization of anthocyanins by high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry in common foods in the United States: Vegetables, nuts, and grains. J. Agric. Food Chem. 2005, 53, 3101–3113. [Google Scholar] [CrossRef] [PubMed]
- Mazzuca, P.; Ferranti, P.; Picariello, G.; Chianese, L.; Addeo, F. Mass spectrometry in the study of anthocyanins and their derivatives: Differentiation of Vitis vinifera and hybrid grapes by liquid chromatography/electrospray ionization mass spectrometry and tandem mass spectrometry. J. Mass Spectrom. JMS 2005, 40, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Pavelescu, L.A. On reactive oxygen species measurement in living systems. J. Med. Life 2015, 8, 38–42. [Google Scholar] [PubMed]
- Csepregi, R.; Bencsik, T.; Papp, N. Examination of secondary metabolites and antioxidant capacity of Anthyllis vulneraria, Fuchsia sp., Galium mollugo and Veronica beccabunga. Acta Biol. Hung. 2016, 67, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.; Wong, S.K.; Mohamed, I.N.; Mohamed, N.; Chin, K.Y.; Ima-Nirwana, S.; Shuid, A.N. Wound Healing Properties of Selected Natural Products. Int. J. Environ. Res. Public Health 2018, 15, 2360. [Google Scholar] [CrossRef]
- Harborne, J.B. Phytochemical Methods; Fakenham Press Limited: New York, NY, USA, 1973. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, J.J. Plant Phenolics: Extraction, Analysis and Their Antioxidant and Anticancer Properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Rahman, M.M.; Ichiyanagi, T.; Komiyama, T.; Hatano, Y.; Konishi, T. Superoxide radical- and peroxynitrite-scavenging activity of anthocyanins; structure-activity relationship and their synergism. Free Radic. Res. 2006, 40, 993–1002. [Google Scholar] [CrossRef]
- Ali, H.M.; Almagribi, W.; Al-Rashidi, M.N. Antiradical and reductant activities of anthocyanidins and anthocyanins, structure-activity relationship and synthesis. Food Chem. 2016, 194, 1275–1282. [Google Scholar] [CrossRef]
- Sun, X.H.; Zhou, T.T.; Wei, C.H.; Lan, W.Q.; Zhao, Y.; Pan, Y.J.; Wu, V.C.H. Antibacterial effect and mechanism of anthocyanin rich Chinese wild blueberry extract on various foodborne pathogens. Food Control 2018, 94, 155–161. [Google Scholar] [CrossRef]
- Van de Velde, F.; Esposito, D.; Grace, M.H.; Pirovani, M.E.; Lila, M.A. Anti-inflammatory and wound healing properties of polyphenolic extracts from strawberry and blackberry fruits. Food Res. Int. 2019, 121, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Nizamutdinova, I.T.; Kim, Y.M.; Chung, J.I.; Shin, S.C.; Jeong, Y.K.; Seo, H.G.; Lee, J.H.; Chang, K.C.; Kim, H.J. Anthocyanins from black soybean seed coats stimulate wound healing in fibroblasts and keratinocytes and prevent inflammation in endothelial cells. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2009, 47, 2806–2812. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Chen, J.; Xiao, A.; Liu, L. Antibacterial Activity of Polyphenols: Structure-Activity Relationship and Influence of Hyperglycemic Condition. Molecules 2017, 22, 1913. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Bhatnagar, P.; Mishra, S.; Kumar, P.; Shukla, Y.; Gupta, K.C. PLGA-encapsulated tea polyphenols enhance the chemotherapeutic efficacy of cisplatin against human cancer cells and mice bearing Ehrlich ascites carcinoma. Int. J. Nanomed. 2015, 10, 6789–6809. [Google Scholar] [CrossRef] [PubMed]
- Alsamri, H.; El Hasasna, H.; Al Dhaheri, Y.; Eid, A.H.; Attoub, S.; Iratni, R. Carnosol, a Natural Polyphenol, Inhibits Migration, Metastasis, and Tumor Growth of Breast Cancer via a ROS-Dependent Proteasome Degradation of STAT3. Front. Oncol. 2019, 9, 743. [Google Scholar] [CrossRef]
- LaFoya, B.; Munroe, J.A.; Albig, A.R. A comparison of resveratrol and other polyphenolic compounds on Notch activation and endothelial cell activity. PLoS ONE 2019, 14, e0210607. [Google Scholar] [CrossRef]
- Ye, J.C.; Hsiao, M.W.; Hsieh, C.H.; Wu, W.C.; Hung, Y.C.; Chang, W.C. Analysis of caffeic acid extraction from Ocimum gratissimum Linn. by high performance liquid chromatography and its effects on a cervical cancer cell line. J. Obstet. Gynecol. 2010, 49, 266–271. [Google Scholar] [CrossRef]
- Sanderson, J.T.; Clabault, H.; Patton, C.; Lassalle-Claux, G.; Jean-Francois, J.; Pare, A.F.; Hebert, M.J.; Surette, M.E.; Touaibia, M. Antiproliferative, antiandrogenic and cytotoxic effects of novel caffeic acid derivatives in LNCaP human androgen-dependent prostate cancer cells. Bioorg. Med. Chem. 2013, 21, 7182–7193. [Google Scholar] [CrossRef]
- Nasr Bouzaiene, N.; Kilani Jaziri, S.; Kovacic, H.; Chekir-Ghedira, L.; Ghedira, K.; Luis, J. The effects of caffeic, coumaric and ferulic acids on proliferation, superoxide production, adhesion and migration of human tumor cells in vitro. Eur. J. Pharmacol. 2015, 766, 99–105. [Google Scholar] [CrossRef]
- Kampa, M.; Alexaki, V.I.; Notas, G.; Nifli, A.P.; Nistikaki, A.; Hatzoglou, A.; Bakogeorgou, E.; Kouimtzoglou, E.; Blekas, G.; Boskou, D.; et al. Antiproliferative and apoptotic effects of selective phenolic acids on T47D human breast cancer cells: Potential mechanisms of action. Breast Cancer Res. BCR 2004, 6, R63–R74. [Google Scholar] [CrossRef] [PubMed]
- Brudzynski, K.; Carlone, R. Stage-dependent modulation of limb regeneration by caffeic acid phenethyl ester (CAPE)--immunocytochemical evidence of a CAPE-evoked delay in mesenchyme formation and limb regeneration. J. Exp. Zool. Part A Comp. Exp. Biol. 2004, 301, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Ly, C.; Ferrier, J.; Gaudet, J.; Yockell-Lelievre, J.; Arnason, J.T.; Gruslin, A.; Bainbridge, S. Vaccinium angustifolium (lowbush blueberry) leaf extract increases extravillous trophoblast cell migration and invasion in vitro. Phytother. Res. PTR 2018, 32, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Bueno, F.G.; Panizzon, G.P.; Mello, E.V.; Lechtenberg, M.; Petereit, F.; de Mello, J.C.; Hensel, A. Hydrolyzable tannins from hydroalcoholic extract from Poincianella pluviosa stem bark and its wound-healing properties: Phytochemical investigations and influence on in vitro cell physiology of human keratinocytes and dermal fibroblasts. Fitoterapia 2014, 99, 252–260. [Google Scholar] [CrossRef]
- Cornelli, U. Antioxidant use in nutraceuticals. Clin. Dermatol. 2009, 27, 175–194. [Google Scholar] [CrossRef]
- Han, J.H.; Lee, H.J.; Cho, M.R.; Chang, N.; Kim, Y.; Oh, S.Y.; Kang, M.H. Total antioxidant capacity of the Korean diet. Nutr. Res. Pract. 2014, 8, 183–191. [Google Scholar] [CrossRef]
- Milicevic, A. The relationship between antioxidant activity, first electrochemical oxidation potential, and spin population of flavonoid radicals. Arch. Ind. Hyg. Toxicol. 2019, 70, 134–139. [Google Scholar] [CrossRef]
- Apak, R.; Ozyurek, M.; Guclu, K.; Capanoglu, E. Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)-Based Assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef]
- Wolfe, K.L.; Liu, R.H. Cellular antioxidant activity (CAA) assay for assessing antioxidants, foods, and dietary supplements. J. Agric. Food Chem. 2007, 55, 8896–8907. [Google Scholar] [CrossRef]
- Vang Mouritzen, M.; Jenssen, H. Optimized Scratch Assay for In Vitro Testing of Cell Migration with an Automated Optical Camera. J. Vis. Exp. JoVE 2018. [Google Scholar] [CrossRef]
- Scheitza, S.; Bonifas, J.; Blomeke, B. Variable NAT1 enzyme activity in long-term cultured human HaCaT keratinocytes. J. Toxicol. Environ. Health Part A 2012, 75, 471–477. [Google Scholar] [CrossRef]
- Brooks, R.F.; Riddle, P.N. The 3T3 cell cycle at low proliferation rates. J. Cell Sci. 1988, 90 Pt 4, 601–612. [Google Scholar]
- Werner, S.; Krieg, T.; Smola, H. Keratinocyte-fibroblast interactions in wound healing. J. Investig. Dermatol. 2007, 127, 998–1008. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Maheshwari, A.; Chandra, A. Biomarkers for wound healing and their evaluation. J. Wound Care 2016, 25, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Lindley, L.E.; Stojadinovic, O.; Pastar, I.; Tomic-Canic, M. Biology and Biomarkers for Wound Healing. Plast. Reconstr. Surg. 2016, 138, 18S–28S. [Google Scholar] [CrossRef] [PubMed]



| No. | tR (min) | λmax (nm) | [M−H]− (m/z) | Fragment ions (m/z) | Tentative Characterization a | Presence and Relative Abundance (%) of Compounds in the Leaf Extracts b | Ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AvEc | AvWc | FmEc | FmWc | Ft Ec | FtWc | LnEc | LnWc | |||||||
| 1 | 2.0 | 315 | 343 | 179, 135 | Caffeic acid derivative | 0.1 | [39] | |||||||
| 2 | 2.2 | 306 | 533, 375, | 217, 173, 149 | Cinnamoylquinic acid derivative | 0.3 | [39] | |||||||
| 3 | 2.5 | 315 | 341 (683) | 179, 149, 135 | Caffeoyl-O-hexoside (dimer) | 0.2 | 0.3 | 0.2 | [40] | |||||
| 4 | 2.6 | 265, 312 | 337 | 267, 191, 163, 149, 135 | 5-p-Coumaroylquinic acid | 0.2 | 0.1 | 0.1 | [39] | |||||
| 5 | 3.2 | 288sh, 311 | 639, 353 | 191 | Caffeoylquinic acid derivative | 0.3 | 0.9 | [39] | ||||||
| 6 | 7.1 | 272 | 331 | 169, 125 | Galloyl hexoside | 0.2 | 0.2 | [41] | ||||||
| 7 | 9.5 | 311 | 355 | 191 | Coumaroyl glucarate isomer | 0.1 | 2.9 | [42] | ||||||
| 8 | 10.1 | 298sh, 320 | 371 | 209, 191, 179 | Caffeoylquinic acid derivative | 0.5 | [39] | |||||||
| 9 | 10.5 | 260 | 611 | 305 | n.i. | 2.0 | 1.8 | - | ||||||
| 10 | 10.5 | 298, 310 | 355 | 209, 191, 163 | Coumaroyl glucarate isomer | 0.5 | 1.3 | [42] | ||||||
| 11 | 10.6 | 298, 320 | 549 | 387, 369, 267, 249, 137 | Cinnamic acid derivative | 0.3 | [43] | |||||||
| 12 | 10.6 | 298 | 331 | 169, 125 | Galloyl hexoside | 2.6 | [41] | |||||||
| 13 | 10.9 | 300 | 301 | 168, 150, 125 | Galloyl pentoside | 0.2 | [41] | |||||||
| 14 | 11.9 | 282sh, 307 | 355 (711) | 271, 209, 191 | Coumaroyl glucarate isomer (dimer) | 3.4 | [42] | |||||||
| 15 | 12.6 | 283sh, 312 | 355 | 271, 209, 191 | Coumaroyl glucarate isomer | 1.9 | 5.0 | 6.9 | [42] | |||||
| 16 | 13.0 | 290, 328sh | 297, (595) | 179, 161, 135 | Caffeic acid derivative (dimer) | 2.0 | 2.4 | 4.0 | [39] | |||||
| 17 | 13.0 | 296 | 575 | 413, 351, 267, 249, 163, 113 | Coumaric acid derivative | 0.5 | [39] | |||||||
| 18 | 13.1 | 310 | 385 | 209, 191 | Feruloyl glucarate | 2.2 | 3.9 | [42] | ||||||
| 19 | 13.2 | 255, 350 | 787 | 625, 462, 301, 299 | Quercetin 3-O-hexosyl-hexosyl-7-O-hexoside | 1.0 | [44] | |||||||
| 20 | 13.3 | 283sh, 312 | 385 | 209, 191 | Feruloyl glucarate | 0.7 | 3.0 | 10.2 | [42] | |||||
| 21 | 13.5 | 265, 349 | 771 | 609, 462, 301, 299, 285, 284, 283, 179 | Quercetin 3-O-hexosyl-desoxyhexosyl-7-O-hexoside | 2.2 | 1.0 | [44] | ||||||
| 22 | 13.8 | 288sh, 312 | 385 | 271, 209, 191, 163, 146, 119 | Feruloyl glucarate | 3.0 | 2.3 | [42] | ||||||
| 23 | 13.9 | 265, 349 | 771 | 609, 446, 445, 285, 284, 283, 179 | Kaempferol 3-O-hexosyl-hexosyl-7-O-hexoside | 2.6 | 0.9 | [44] | ||||||
| 24 | 13.9 | 276 | 475, 453 | - | n.i. | 2.5 | - | |||||||
| 25 | 14.4 | 264, 351 | 755 | 593, 446, 284, 283 | Kaempferol 3-O-hexosyl-desoxyhexosyl-7-O-hexoside | 2.5 | 2.9 | [45] | ||||||
| 26 | 15.0 | 272 | 651 | 399, 325, 163 | n.i. | 1.9 | 2.9 | - | ||||||
| 27 | 15.2 | 256, 354 | 625 | 463, 462, 301, 299 | Quercetin 3-O-hexosyl-7-O-hexoside | 1.6 | 1.2 | [45] | ||||||
| 28 | 15.7 | 257, 352 | 595 | 462, 433, 301, 299, 271 | Quercetin 3-O-pentosyl-7-O-hexoside | 0.7 | 1.2 | [41] | ||||||
| 29 | 15.8 | 268 | 305 | 225, 147, 135 | n.i. | 0.8 | 0.5 | 0.9 | 1.3 | - | ||||
| 30 | 16.2 | 265, 347 | 609 | 447, 446, 285, 283 | Kaempferol 3-O-hexosyl-7-O-hexoside | 1.7 | 0.5 | [41] | ||||||
| 31 | 16.4 | 278 | 449 | 357, 275 | n.i. | 1.5 | 0.9 | - | ||||||
| 32 | 16.5 | 288sh, 321 | 575 | 443, 267, 249, 193, 175 | 1,3-O-Diferuloylglycerol pentoside | 0.7 | [46] | |||||||
| 33 | 16.6 | 254, 354 | 639 | 519, 477, 461, 315, 314, 313, 299, 151 | Isorhamnetin 3-O-hexosyl-7-O-hexoside | 4.6 | 1.4 | [47] | ||||||
| 34 | 17.2 | 268, 295sh | 693 | 477, 345, 327, 315, 300, 207, 183, 165 | n.i. | 1.8 | - | |||||||
| 35 | 17.4 | 252, 269sh, 332 | 443 | 267, 249, 193, 175, 149, 134, 113 | 1,3-O-Diferuloylglycerol | 5.1 | 4.4 | [46] | ||||||
| 36 | 17.5 | 252, 269sh, 352 | 963 | 801, 625 | Flavonoid | 0.6 | [43] | |||||||
| 37 | 18.0 | 260 | 197 | n.i. | 3.2 | - | ||||||||
| 38 | 18.0 | 268, 350 | 639 | 319, 301, 283, 239, 213, 203, 197, 157, 142, 130, 116, 109 | Flavonoid | 6.2 | 6.5 | [43] | ||||||
| 39 | 18.1 | 276 | 521 | 337, 191, 163 | Coumaroylquinic acid derivative | 1.2 | 0.7 | [39] | ||||||
| 40 | 18.2 | 266, 350 | 625 | 463, 300, 271, 255, 243, 179 | Quercetin 3-O-hexosyl-hexoside | 2.9 | 2.5 | [47] | ||||||
| 41 | 19.2 | 274 | 387 (775) | 169, 151, 124 | Gallic acid derivative (dimer) | 0.4 | 0.2 | [46] | ||||||
| 42 | 19.3 | 274 | 537 | 271, 211, 169, 151, 124 | Gallic acid derivative | 0.2 | 0.1 | [46] | ||||||
| 43 | 19.3 | 266, 357 | 625 | 479, 306 | Myricetin 3-O-desoxyhexosyl-hexoside | 2.3 | 1.5 | [48] | ||||||
| 44 | 19.3 | 266, 355 | 609 | 429, 285, 284, 255, 227 | Kaempferol 3-O-hexosyl-hexoside | 1.2 | 2.6 | [41] | ||||||
| 45 | 19.4 | 267, 357 | 479 | 316 | Myricetin 3-O-hexoside | 1.1 | 0.8 | - | ||||||
| 46 | 19.5 | 262, 355 | 615 | 463, 301, 300, 271, 169 | Quercetin galloyl hexoside | 0.4 | 0.4 | 1.8 | 1.0 | [47] | ||||
| 47 | 19.6 | 264, 352 | 639 | 459, 315, 314, 257 | Isorhamnetin 3-O-hexosyl-hexoside | 1.9 | [49] | |||||||
| 48 | 19.8 | 266, 331 | 593 | 429, 284, 255, 227 | Kaempferol 3-O-desoxyhexosyl-hexoside | 2.7 | 1.7 | [41] | ||||||
| 49 | 20.2 | 260, 354 | 463 | 317, 316 | Myricetin 3-O-desoxyhexoside | 22.0 | 17.2 | [50] | ||||||
| 50 | 20.3 | 255, 293sh, 359 | 615 | 301 | Quercetin galloyl hexoside | 0.1 | 0.1 | [47] | ||||||
| 51 | 20.5 | 259, 356 | 477 | 301, 283, 255, 179, 151, 121 | Quercetin glucuronide | 0.5 | 0.2 | 0.6 | 0.6 | [47] | ||||
| 52 | 20.6 | 257, 268sh, 356 | 433 | 301, 300 | Quercetin 3-O-pentoside | 0.1 | 0.1 | [47] | ||||||
| 53 | 20.7 | 257, 268sh, 356 | 615 | 301 | Quercetin galloyl hexoside | 0.9 | 0.5 | [51] | ||||||
| 54 | 20.7 | 260, 353 | 463 | 317, 316 | Myricetin 3-O-desoxyhexoside | 2.4 | [50] | |||||||
| 55 | 20.8 | 257, 355 | 463 | 301, 300, 271, 255, 243, 179, 163, 151 | Quercetin 3-O-hexoside | 3.6 | 4.9 | 0.1 | 0.1 | 0.6 | 0.5 | [41] | ||
| 56 | 20.9 | 266, 350 | 579 | 463, 315, 313 | Isorhamnetin-3-O-pentosyl-7-O-pentoside | 5.0 | 3.8 | [47] | ||||||
| 57 | 20.9 | 257, 355 | 609 | 463, 301, 300, 299 | Quercetin 3-O-desoxyhexosyl-7-O-hexoside | 0.1 | 0.1 | 0.2 | 0.5 | 0.8 | 1.8 | [48] | ||
| 58 | 21 | 257, 355 | 599 | 447, 313, 285, 169 | Kaempferol galloyl hexoside | 0.1 | 0.1 | [43] | ||||||
| 59 | 21.3 | 256, 356 | 433 | 300, 271, 255, 151 | Quercetin 3-O-pentoside | 2.4 | 2.0 | 0.2 | 0.1 | 0.4 | 0.4 | [47] | ||
| 60 | 21.5 | 254, 368 | 301 | 284, 245, 229, 201, 185, 145, 129, 117 | Ellagic acid | 0.3 | 0.1 | 1.1 | 0.6 | [51] | ||||
| 61 | 21.7 | 266, 350 | 447 | 284, 255, 227, 151 | Kaempferol 3-O-hexoside | 2.6 | 2.2 | 0.2 | 0.1 | [41] | ||||
| 62 | 21.7 | 266, 350 | 447 | 301, 300, 271, 255, 179, 151 | Quercetin 3-O-desoxyhexoside | 1.1 | 1.2 | [47] | ||||||
| 63 | 21.8 | 266, 332 | 705 | 437, 407, 325, 245, 231, 199, 163, 121 | n.i. | 2.4 | 1.8 | - | ||||||
| 64 | 21.8 | 266, 350 | 521 | 331, 271, 211, 169 | Galloyl hexoside derivative | 0.5 | 0.3 | [41] | ||||||
| 65 | 21.9 | 517 | 267, 249, 205, 161, 113 | n.i. | 2.0 | 1.2 | - | |||||||
| 66 | 22.0 | 266, 347 | 447 | 284, 255, 227 | Kaempferol 3-O-hexoside | 0.4 | 0.2 | [41] | ||||||
| 67 | 22.0 | 255, 350 | 477 | 315, 314, 285, 271, 257, 243 | Isorhamnetin 3-O-hexoside | 1.8 | 4.2 | [52] | ||||||
| 68 | 22.0 | 264, 340 | 477 | 331, 317 | Myricetin desoxyhexoside derivative | 2.8 | 3.4 | [53] | ||||||
| 69 | 22.0 | 264, 340 | 447 | 331, 317 | Myricetin derivative | 2.5 | 1.0 | [53] | ||||||
| 70 | 22.3 | 266, 340 | 639 | 477, 459, 315, 314, 267 | Isorhamnetin 3-O-hexosyl-hexoside | 1.1 | 2.6 | [52] | ||||||
| 71 | 22.3 | 266, 347 | 417 | 285, 284, 255, 227 | Kaempferol 3-O-pentoside | 0.3 | 0.2 | [41] | ||||||
| 72 | 22.4 | 267, 328 | 727 | 551, 491, 415, 267, 249 | Ferulic acid derivative | 2.0 | [54] | |||||||
| 73 | 22.4 | 623 | 431, 371, 345, 317, 301, 299 | n.i. | 0.8 | 0.5 | - | |||||||
| 74 | 22.5 | 267, 328 | 727 | 551, 415, 267, 183 | Ferulic acid derivative | 1.4 | [54] | |||||||
| 75 | 22.8 | 255, 266sh, 334 | 447 | 314, 285, 271, 257, 243 | Isorhamnetin 3-O-pentoside | 1.3 | [52] | |||||||
| 76 | 22.9 | 266, 351 | 599 | 447, 301, 300, 179, 151 | Quercetin 3-O-desoxyhexoside derivative | 0.1 | 0.1 | [47] | ||||||
| 77 | 23.4 | 266, 348 | 623 | 443, 299, 298, 283, 271 | Rhamnocitrin 3-O-dihexozid | 3.8 | 3.2 | [54] | ||||||
| 78 | 23.7 | 268, 339 | 609 | 315, 314, 193 | Isorhamnetin 3-O-pentosyl-hexoside | 0.6 | 1.5 | [52] | ||||||
| 79 | 23.8 | 268, 329 | 799 | 623, 485, 397, 299, 298 | n.i. | 2.1 | 0.7 | - | ||||||
| 80 | 24.8 | 477 | 301, 267, 249, 227, 209, 183, 165, 113 | Ellagic acid derivative | 0.6 | [51] | ||||||||
| 81 | 24.9 | 250, 370 | 531 | 301, 300 | Quercetin derivative | 0.1 | 0.5 | [47] | ||||||
| 82 | 24.9 | 266, 344 | 593 | 413, 299, 298, 283 | Rhamnocitrin 3-O-hexosyl-pentoside | 1.1 | 0.7 | [54] | ||||||
| No. | tR (min) | [M + H]+ (m/z) | Fragment Ions (m/z) | Tentative Characterization a | Presence and Relative Abundance (%) of Compounds in the Leaf Extracts b | Ref. | |||
|---|---|---|---|---|---|---|---|---|---|
| FmEc | FmWc | FtEc | FtWc | ||||||
| I | 2.0 | 767 | 453, 153 | Anthocyanin (n.i.) | 1.1 | - | |||
| II | 2.7 | 611 | 449, 287 | Cyanidin dihexoside | 3.0 | 18.3 | [55,56] | ||
| III | 2.9 | 783 | 303 | Anthocyanin (n.i.) | 5.9 | - | |||
| IV | 9.1 | 625 | 463, 301 | Peonidin dihexoside | 15.7 | [55,56] | |||
| V | 10.3 | 625 | 463, 301 | Peonidin dihexoside | 18.9 | 39.9 | 5.3 | 5.4 | [55,56] |
| VI | 10.7 | 487 | 325, 185 | Anthocyanidin hexoside | 4.2 | - | |||
| VII | 11.4 | 441 | 249 | Anthocyanin (n.i.) | 9.6 | - | |||
| VIII | 11.8 | 411 | 249 | Anthocyanin (n.i.) | 6.1 | 9.6 | - | ||
| MIC80 (µg/mL) | |||
|---|---|---|---|
| Test Bacteria | Treatment | Ethanolic Extracts | Aqueous Extracts |
| Escherichia coli | erythromycin | 30.56–42.88 | |
| A. vulneraria | N.D. | N.D. | |
| F. magellanica | N.D. | N.D. | |
| F. triphylla | N.D. | N.D. | |
| L. nummularia | N.D. | N.D. | |
| Pseudomonas aeruginosa | erythromycin | 69.91–82.76 | |
| A. vulneraria | N.D. | N.D. | |
| F. magellanica | 55.03–58.91 | N.D. | |
| F. triphylla | 49.03–58.87 | N.D. | |
| L. nummularia | N.D. | N.D. | |
| Staphylococcus aureus | erythromycin | 0.09–0.25 | |
| A. vulneraria | 715.90–835.92 | N.D. | |
| F. magellanica | 4.81–7.65 | 17.13–25.03 | |
| F. triphylla | 5.32–6.73 | 30.24–36.99 | |
| L. nummularia | 1063.75–1181.29 | N.D. | |
| Bacillus subtilis | erythromycin | 0.10–0.18 | |
| A. vulneraria | 288.02–313.01 | N.D. | |
| F. magellanica | 13.91–15.72 | 52.73–65.03 | |
| F. triphylla | 14.55–22.66 | 33.16–45.33 | |
| L. nummularia | 160.90–164.28 | N.D. | |
| Streptococcus pyogenes | erythromycin | 0.08–0.09 | |
| A. vulneraria | 220.06–236.30 | N.D. | |
| F. magellanica | 11.83–14.99 | 41.72–44.91 | |
| F. triphylla | 11.61–17.92 | 41.95–43.23 | |
| L. nummularia | 179.43–203.56 | N.D. | |
| 3T3 Cells | ||||
|---|---|---|---|---|
| Treatment Groups | Annexin V–7AAD Method | |||
| Live Cells (%) | Necrotic Cells (%) | Early Apoptotic Cells (%) | Late Apoptotic Cells (%) | |
| Control cells | 97.13 | 2.06 | 0.66 | 0.15 |
| 50 µg/mL A. vulneraria | 96.29 | 2.46 | 1.00 | 0.25 |
| 100 µg/mL A. vulneraria | 96.00 | 2.90 | 0.83 | 0.27 |
| 200 µg/mL A. vulneraria | 96.89 | 2.22 | 0.70 | 0.20 |
| 2.5 µg/mL F. magellanica | 96.95 | 2.03 | 0.75 | 0.26 |
| 5 µg/mL F. magellanica | 96.27 | 2.67 | 0.79 | 0.27 |
| 10 µg/mL F. magellanica | 96.62 | 2.25 | 0.78 | 0.34 |
| 2.5 µg/mL F. triphylla | 97.74 | 1.26 | 0.84 | 0.17 |
| 5 µg/mL F. triphylla | 98.15 | 1.01 | 0.71 | 0.13 |
| 10 µg/mL F. triphylla | 97.57 | 1.50 | 0.62 | 0.30 |
| 10 µg/mL L. nummularia | 95.63 | 2.98 | 1.05 | 0.33 |
| 25 µg/mL L. nummularia | 95.33 | 3.31 | 1.07 | 0.29 |
| 50 µg/mL L. nummularia | 95.48 | 2.81 | 1.34 | 0.38 |
| HaCaT Cells | ||||
|---|---|---|---|---|
| Treatment Groups | Annexin V–7AAD Method | |||
| Live Cells (%) | Necrotic Cells (%) | Early Apoptotic Cells (%) | Late Apoptotic Cells (%) | |
| Control cells | 99.04 | 1.31 | 0.49 | 0.47 |
| 50 µg/mL A. vulneraria | 96.84 | 1.56 | 0.51 | 1.09 |
| 100 µg/mL A. vulneraria | 95.29 | 3.36 | 0.51 | 0.83 |
| 200 µg/mL A. vulneraria | 96.06 | 2.60 | 0.35 | 0.99 |
| 2.5 µg/mL F. magellanica | 97.76 | 1.39 | 0.47 | 0.37 |
| 5 µg/mL F. magellanica | 97.08 | 2.19 | 0.38 | 0.34 |
| 10 µg/mL F. magellanica | 97.56 | 1.61 | 0.54 | 0.28 |
| 2.5 µg/mL F. triphylla | 97.98 | 1.06 | 0.54 | 0.42 |
| 5 µg/mL F. triphylla | 97.68 | 1.33 | 0.54 | 0.45 |
| 10 µg/mL F. triphylla | 98.04 | 0.91 | 0.63 | 0.42 |
| 10 µg/mL L. nummularia | 97.19 | 1.70 | 0.42 | 0.69 |
| 25 µg/mL L. nummularia | 96.89 | 1.86 | 0.41 | 0.84 |
| 50 µg/mL L. nummularia | 96.15 | 2.49 | 0.44 | 0.91 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Csepregi, R.; Temesfői, V.; Das, S.; Alberti, Á.; Tóth, C.A.; Herczeg, R.; Papp, N.; Kőszegi, T. Cytotoxic, Antimicrobial, Antioxidant Properties and Effects on Cell Migration of Phenolic Compounds of Selected Transylvanian Medicinal Plants. Antioxidants 2020, 9, 166. https://doi.org/10.3390/antiox9020166
Csepregi R, Temesfői V, Das S, Alberti Á, Tóth CA, Herczeg R, Papp N, Kőszegi T. Cytotoxic, Antimicrobial, Antioxidant Properties and Effects on Cell Migration of Phenolic Compounds of Selected Transylvanian Medicinal Plants. Antioxidants. 2020; 9(2):166. https://doi.org/10.3390/antiox9020166
Chicago/Turabian StyleCsepregi, Rita, Viktória Temesfői, Sourav Das, Ágnes Alberti, Csenge Anna Tóth, Róbert Herczeg, Nóra Papp, and Tamás Kőszegi. 2020. "Cytotoxic, Antimicrobial, Antioxidant Properties and Effects on Cell Migration of Phenolic Compounds of Selected Transylvanian Medicinal Plants" Antioxidants 9, no. 2: 166. https://doi.org/10.3390/antiox9020166
APA StyleCsepregi, R., Temesfői, V., Das, S., Alberti, Á., Tóth, C. A., Herczeg, R., Papp, N., & Kőszegi, T. (2020). Cytotoxic, Antimicrobial, Antioxidant Properties and Effects on Cell Migration of Phenolic Compounds of Selected Transylvanian Medicinal Plants. Antioxidants, 9(2), 166. https://doi.org/10.3390/antiox9020166






