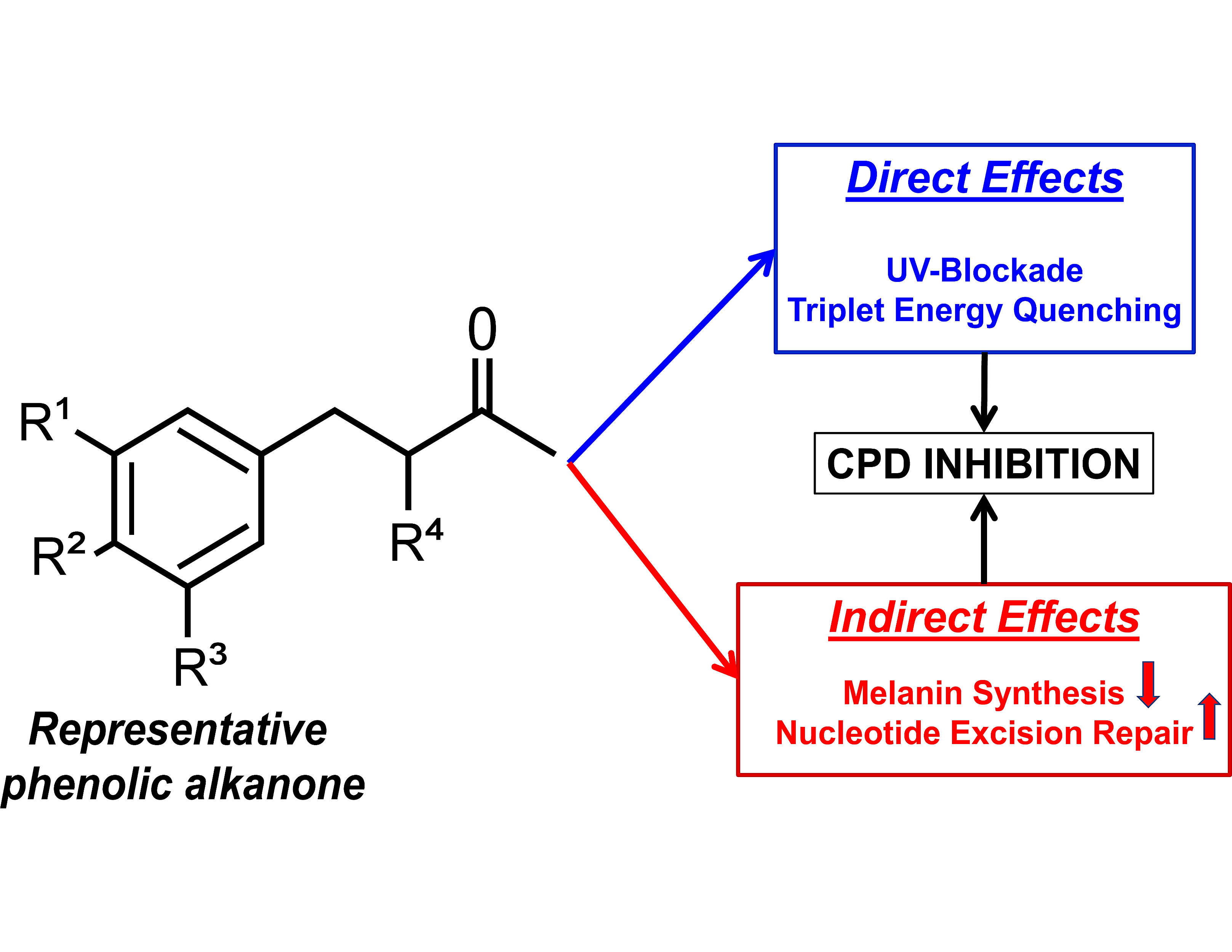
1. Introduction
In the USA, ~9000 people are diagnosed with skin cancers every day, with more than two deaths occurring every hour [
1,
2]. Sunburns and chronic sun exposure double the risk of skin cancers [
2], mostly through bulky and extremely mutagenic DNA photoproducts called cyclobutane pyrimidine dimers (CPDs). CPDs lead to cytosine to thymine (C to T) transition mutations, also known as UV signature mutations [
3,
4]. CPDs are generated in picoseconds in response to UV exposure (incident or iCPDs). The pigment melanin, produced by melanocytes, is known to act as a sunshield owing to its abnormally broad absorption spectrum and partial antioxidant nature [
5]. Two subtypes of melanoma, eumelanin and pheomelanin, have different capabilities with regard to protecting the skin. Eumelanin is dark/black in color and a potent sunshield and antioxidant [
6]. Pheomelanin is less photostable, and more oxidative compared to eumelanin, which is added upon by the fact its synthesis consumes cysteine, an integral part of the cellular antioxidant glutathione [
6,
7]. Contrary to its known photoprotective properties, Dr. Brash and his team discovered a novel pathway, called melanin chemiexcitation (MeCh) that implicates melanin in the generation of CPDs in complete absence of UV radiation [
8]. In MeCh, UV exposure induces nitric oxide synthase (NOS) and NADPH oxidase (NOX) activities to produce peroxynitrite (ONOO
−), which subsequently reacts with melanin to generate triplet-excited melanin-carbonyls (MCs). These MCs, which are as energetic as UV photons, can transfer their energy to di-pyrimidines, generating CPDs without UV exposure (delayed or dark CPDs, dCPDs). At any time-point post UV exposure, approximately 50% of CPDs are generated by MeCh [
8,
9] suggesting a potentially carcinogenic role for melanin. We also demonstrated that MeCh might be operative without any UV exposure, due only to endogenously hyperactive NOS in pigmented melanocytes [
8]. Pursuing this further, we identified genomic sites that are hypersensitive to iCPDs and dCPDs [
9]. These findings suggest a carcinogenic role for melanin. This also indicates that the total amount of CPDs generated by UV exposure has always been underestimated, and so is the mutational load in sunlight-induced skin cancers [
9].
Sunscreens significantly inhibit iCPD formation during sun exposure. Sunscreens form a continuous film that creates an effective interface between the sunlight and the skin. During sun exposure, sunscreen compounds are evenly dispersed within the film, and intercept and neutralize UV photons before they can reach and damage the underlying skin. Indeed, research has established that regular use of sunscreens does delay or reduce the incidences of melanoma and non-melanoma skin cancers [
10,
11]. Several mechanisms have been proposed for this [
12,
13]; however, the sunscreen-mediated inhibition of iCPD formation seems to be the best explanation. The fact that sunscreens reduce the total amount of CPDs significantly has been observed in both in situ and in vivo models [
14,
15,
16,
17,
18].
While effective during periods of sun exposure, it should be noted that sunscreen compounds can also act as a photosensitizers [
19] and can be cytotoxic [
20,
21,
22,
23]. Additionally, sunscreens are completely ineffective against dCPD formation once sun exposure ends. This is particularly important in the case of pigmented melanocytes, which have melanin as a raw material, in addition to nuclear and cytoplasmic NOS activity as an inducer of melanin chemiexcitation [
8]. Previously, we demonstrated that a plant-based phenolic alkanone called 3-(4-hydroxy, 3-methoxybenzyl) -pentane-2,4-dione (INCI: acetyl zingerone (AZ)) effectively blocked melanin chemiexcitation and inhibited dCPD generation [
24]. We postulated that AZ could intervene in the MeCh pathway, mainly by scavenging ONOO
− or by physically quenching the triplet energy of melanin-carbonyls via energy transfer [
25]. Using expressional microarrays on reconstituted human epidermis, we also reported that AZ increased the expression of NOTCH pathway genes such as
NOTCH1 and
MAML3 and decreased the expression of genes linked to ECM disassembly (
MMP3,
CTSV,
NOXO1) and reactive oxygen species metabolism (
PMAIP1,
ARG2). In in vitro assays, AZ also inhibited the activity
of MMP-1, MMP-3, and MMP-12 and consistently increased the expression of genes encoding collagens, proteoglycans, ECM regulators, and ECM glycoproteins, while concomitantly opposing the gene expression patterns associated with fibroblast senescence, keratinocyte differentiation, and IL-17A stimulation [
26].
In the current study, we further evaluated AZ as well as six of its chemical analogs for their effectiveness in blocking both iCPD and dCPD formation and investigated their basic structure–activity requirements for this property alongside mechanisms of action. We investigated their cellular toxicity, effects on DNA repair, and efficiency in inhibiting CPD generation in response to exposure to solar-simulated UV (ssUV). We identified MBPD (3-(4-Methoxy-benzyl)-pentane-2,4-dione) as one analog which effectively inhibited ~80% of the total CPD formation (iCPDs + dCPDs) in pigmented mouse and human melanocytes. Taken together, our results strongly support the use of AZ and MBPD as skincare additives in sunscreens as well as other topically applied products to bolster protection against CPD formation.
Figure 1 displays the general backbone of AZ’s chemical structure and summarizes the overall findings of this research.
2. Materials and Methods
2.1. Acetyl Zingerone and Its Analogs
The chemical synthesis of Acetyl Zingerone has been described previously by us [
24]. Briefly, vanillin (4-hydroxy-3-methoxybenzaldehyde) was condensed with acetylacetone to obtain 3-(4-hydroxy, 3-methoxybenzylidine) -pentane-2,4-dione, and the reaction intermediates were catalytically hydrogenated to obtain the target compound. The purity of AZ was estimated to be >99% as determined by HPLC. The structure was confirmed by
1HNMR,
13CNMR, and mass spectral analysis. Acetyl zingerone (trade name: Synoxyl
® AZ) is commercially available from Sytheon, Parsippany, New Jersey, USA. AZ is REACH-registered (EC# 820-605-0) and is safe for topical applications, as documented by the studies available for review on the ECHA website. The backbone structure is shown in
Figure 1. The functional groups on all the “Rs” and the chemical names with their respective abbreviations are listed below.
3-(4-Hydroxy-3-methoxybenzyl)-pentane-2,4-dione [Acetyl Zingerone (Synoxyl
® AZ)] R
1 = H, R
2 = OH, R
3 = OCH
3, R
4 = COCH
3 (
AZ). This is the parent compound which we have published previously for its capabilities in blocking delayed CPD generation [
17].
2-Vanillyl-acetoacetic acid ethyl ester, R1 = H, R2 = OH, R3 = OCH3, R4 = COOCH2CH3 (VAAE).
3-(3,4,5-trimethoxy-benzyl)-pentane-2,4-dione, R1 = R2 = R3 = OCH3, R4 = COCH3, (TMBPD).
3-(4-Methoxy-benzyl)-pentane-2,4-dione, R1 = R3 = H, R2 = OCH3, R4 = COCH3 (MBPD).
3-(3,4-Dimethoxy-benzyl)-pentane-2,4-dione [Acetyl Zingerone Methyl Ether (Synoxyl® MAZ)] R1 = H, R2 = R3 = OCH3, R4 = COCH3 (DMBPD).
4-(4-Hydroxy-3-methoxyphenyl)-butan-2-one [Zingerone]. Purchased from Sigma-Aldrich, Cat. W312420. R1 = H, R2 = OH, R3 = OCH3, R4 = H.
4-(4-Methoxyphenyl)-butan-2-one [Raspberry Ketone Methyl Ether RKME]. Purchased from Sigma-Aldrich, Cat. W267201. R1 = R3 = R4 = H, R2 = OCH3.
AZ and its analogs were dissolved in 200 mg/mL DMSO and diluted in cell culture medium at various concentrations for treating the cells in the culture. The respective molar concentrations are mentioned in the results and figure legends for each AZ analog.
2.2. Cell Types and Source
Various human and mouse skin cells were used for testing the efficacy of AZ and its analogues as sunscreens and inhibitors of MeCh. This included primary human skin fibroblasts (NBHF); primary human skin melanocytes (PHM); pigmented, C57BL/6 mouse melanocytes (Mel-a); and tyrosinase mutated, isogenic albino melanocytes (Mel-c). All were purchased from the specimen resource core of the Dermatology Department, Yale School of Medicine, New Haven, under a material transfer agreement (MTA). Mel-a and Mel-c, originally called Melan-a and Melan-c, are immortalized, C57BL/6 melanocytes generated by Dr. Bennett’s group. Mel-a cells were generated from embryonic melanoblasts of inbred C57BL/6 mice [
27]. Mel-c (albino melanocyte) cells were generated from the epidermis of C57BL/6 mice carrying a mutation in the albino locus which codes for tyrosinase [
28]. HaCaT, human keratinocyte cells, were purchased from AddexBio, San Diego, CA, USA (Cat. T0020001).
2.3. Cell Culture, Treatment of Cells with AZ and Its Analogs, and Cell Survival Assays
Mel-a and Mel-c melanocytes were cultured in a OptiMem base (Cat. 31985-070, Life Technologies, Carlsbad, CA, USA) supplemented with 7% horse serum (Cat. 100-508, Gemini Bio-Products, W. Sacramento, CA, USA), and 10 ng/mL (16.2 nM) TPA (12-O-tetradecanoylphorbol-13-acetate, Cat. P1585, Sigma, St. Louis, MO, USA) [
8]. NBHF and HaCaT cells were cultured in high glucose DMEM supplemented with 5% FBS (GeminiBio, Cat. 100-500) [
9]. Cell culture medium for every cell type was supplemented with 1× penicillin/streptomycin (diluted from 100×, which is 100 U/mL penicillin and 100 µg/mL streptomycin, Cat. 15140-122, Life Technologies, Carlsbad, CA, USA) throughout the experiment, and with Mycoplasma Removal Agent (Cat. BUF035, Bio-Rad Laboratories, Hercules, CA, USA) for the first 3 passages. Cells were regularly tested for Mycoplasma using PCR based methods (Cat. MP0025, Sigma). Cells were treated with various concentrations of AZ and its analogs in 96-well plates (Cat. 165305, ThermoFisher Scientific, Waltham, MA, USA) for 72 h. Cell survival was assessed using the oxidation of Resazurin to the fluorescent Resorufin (Alamar blue dye, Cat. BUF012A, Bio-Rad, Hercules, CA, USA) following manufacturer’s instructions and published protocols [
29]. In brief, Alamar blue was added to cells, they were incubated for 1–3 h, and survival was assessed in the form of fluorescence emitted by the live cells. Untreated or DMSO-treated cells were used as controls. Each experiment was repeated >3 times. Data analyses were performed using GraphPad Prism. Statistical significance and IC
50 values were estimated using 2-way ANOVA and other analyses methods on GraphPad Prism.
2.4. UV Exposure and Estimation of CPDs
For UV exposures, we used a “SOL-UV6 ultraviolet solar simulator” from Newport irradiators. It is equipped with a Xenon arc lamp where the output is varied using the integrated variable attenuator aperture, which provides the ability to vary the output from 10% to 100% of the maximum available solar constants. The output spectrum is flat from 320 nm to 400 nm and no values are below 300 nm. This is abbreviated as “ssUV” for “solar-simulated UV” throughout the text. Various cell types seeded at ~50% confluency, were treated with the AZ and analogs at concentrations mentioned in the data figures for 24–h. Cells at ~80–90% confluency (untreated controls), were washed twice with PBS and exposed to 18 kJ/m
2 of ssUV in minimal amount of PBS (ex. 2 mL PBS in a 60 mm cell culture dish) without any dish lid in place. According to our previous research, AZ and its analogs do not absorb UV wavelengths (
Supplementary Figure S1) and are more photostable compared to several well-known skincare product additives [
24]. The ssUV apparatus was monitored almost daily using UV meters for any emissions below 300 nm. The dose was decided based upon the 80–90% cell survival post UV exposure. After exposure, one time point was collected immediately to assess iCPDs, whereas more dishes were incubated in cell culture medium, inside the cell culture incubator, for various time points to be collected post UV to assess dCPDs. For dCPD induction, it is important that the cell culture incubator is not opened for at least 30 min (the first time point post UV) to avoid possible interference from the CO
2/O
2 ratio, which will be altered upon opening the incubator doors. Cells were collected by scraping and washed once with PBS, which was followed by DNA isolation using a Quick DNA miniprep Kit (Cat. D3024, Zymo Research, Irvine, CA, USA). CPDs were assessed using our previously established DNA ELISA protocols [
8]. In brief, ELISA was performed using mouse monoclonal Anti-CPD (Cat. CAC-NM-DND-001, Clone TDM-2, CosmoBio, Inc., Carlsbad, CA, USA). Flat-bottom, 96-well plates (Cat. 2801, Thermo Scientific, Waltham, MA, USA) were coated overnight with 50 µL of protamine sulphate (0.005–0.05%, Cat. P3369, Sigma, St. Lois, MO, USA) in distilled water. After washing twice with water, heat-denatured DNA was plated in triplicates from each sample, into the protamine sulphate coated plate wells (50–100 ng/well in 50 µL PBS) and incubated overnight at 37 °C. The plates were washed 5 times with 200 µL of PBST (PBS containing 0.05% Tween-20 Cat. P7949, Sigma), and blocked with 2–4% FBS in PBST (blocking buffer) for 1 h at room temperature (RT). The plates were incubated with 100 µL of a 1:1000 dilution of anti-CPD antibody in blocking buffer, and incubated for 30 min at 37 °C, which was followed by 5 washes in PBST. Next, the plates were incubated with 100 µL of a 1:2000 dilution of Biotin-F(ab’)2 fragment of anti-mouse IgG(H+L) (Cat. B-2763, Life Technologies) in blocking buffer for 30 min at 37 °C. Detection was conducted using 100 µL of a 1:10,000 dilution of Streptavidin HRP (Cat. 434323, Life Technologies) in 2% FBS for 30 min at 37 °C, which was followed by 5 washes. The plates were washed once with citrate phosphate buffer (24.3 mM citric acid monohydrate, J.T. Baker Cat. 0110-01- and 51.4-mM sodium phosphate dibasic, Sigma Cat. S7907, pH 5.0). The o-phenylene diamine dihydrochloride (Cat. P8287-50TAB, Sigma) was used as a HRP substrate at 400 µg/mL in citrate phosphate buffer for 5–10 min. A measure of 50 µL of 2M H
2SO
4 was used to stop the reaction as soon as the color developed (5–10 min incubation in substrate), and OD was measured at 490 nm using a Synergy-2 multimode microplate reader (BioTek, Winooski, VT, USA).
2.5. Modulation of Amount of Melanin in Melanocytes
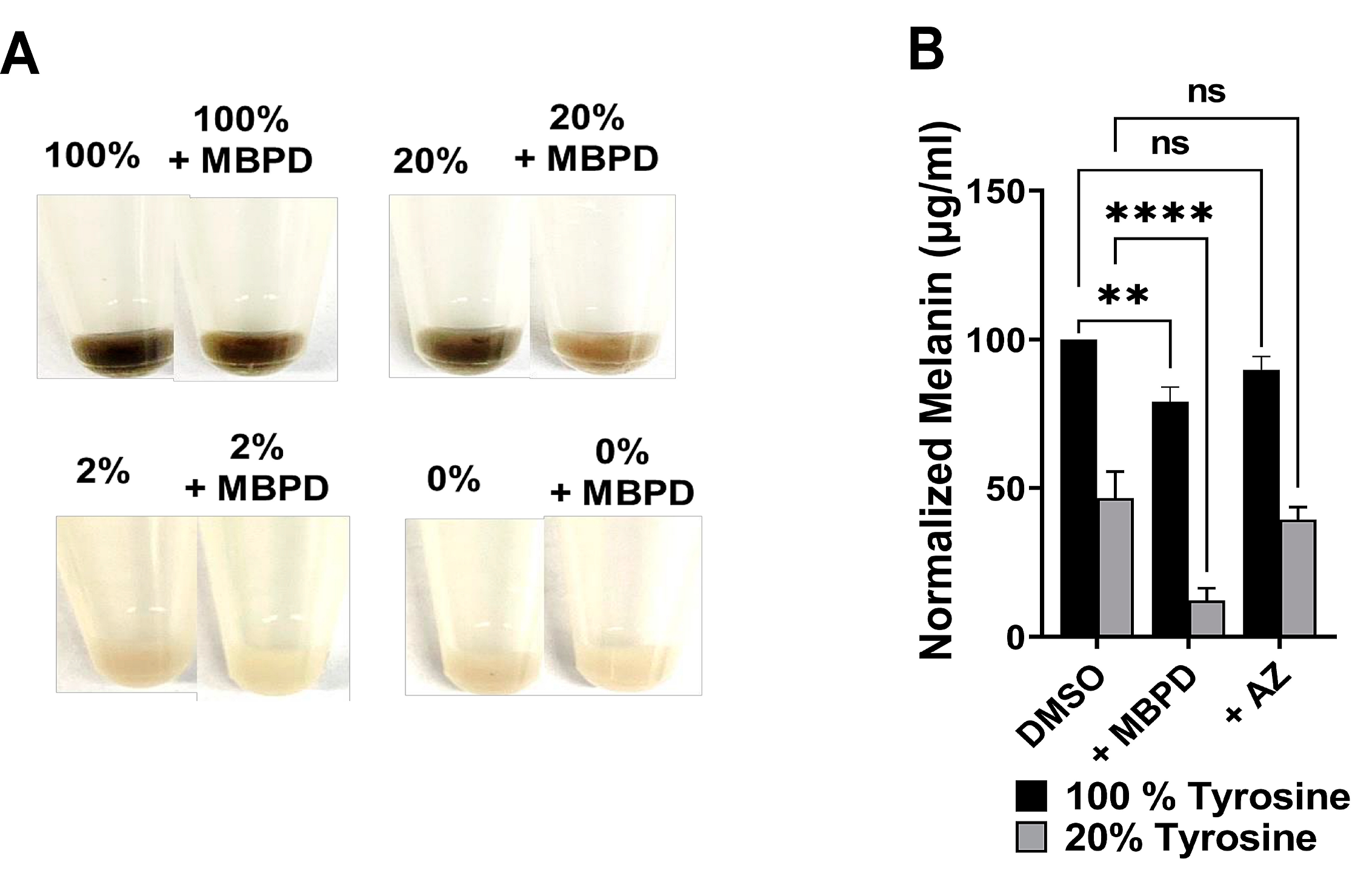
The amino acid tyrosine is essential for melanin synthesis, and it is added to the OptiMem medium at a proprietary concentration by ThermoFisher. Assuming this concentration to be “100% tyrosine”, we purchased the “custom-prepared, tyrosine-free” medium from ThermoFisher. This medium lacked tyrosine or its precursors, which include L-tyrosine, L-tyrosine-2Na-2H2O, L-tyrosine-Di-sodium salt, Hypoxanthine, and Sodium Hypoxanthine. Mel-a (pigmented mouse melanocyte) cells were cultured in regular cell culture medium containing 100% tyrosine, and in medium containing 20%, 4%, or 0% of the total tyrosine amount. This was achieved by diluting the regular OptiMem with the one containing no tyrosine (tyrosine-free, 0% tyrosine). Cells were cultured in these media for two weeks, and 200 µg/mL of MBPD or AZ was then added, and the mixture underwent a further culture for 2–3 weeks in the respective media. Since the cells cultured in 0%-tyrosine medium lost their pigment over time (Figure 5) and the cell pellets were almost white or opaque in color, we assumed that the minimal amount of tyrosine from the horse serum used in the cell culture medium was not sufficient for melanin synthesis. To estimate the amount of melanin, 3 × 106–5 × 106 cells were lysed in 1M sodium hydroxide (NaOH) by heating them at 100 °C for 1–2 h, which was followed by a 10-min spin at 14,000 rpm at room temperature to remove any cell debris. Absorbance of the supernatant was recorded using the supernatant in a 96-well plate in triplicates from each sample, at 460–490 nm. Synthetic melanin (Cat. M0418, Sigma) was dissolved in methanol and used as a positive control to generate standard curves to estimate the melanin amount in cell lysates. The number of cells in each pellet was used as a normalization factor for each experimental condition. In a complementary experiment, cells were first bleached out of melanin completely by culturing them in tyrosine-free medium, followed by 100/20/4/0% tyrosine (not shown) treatment. This seemed to be cytotoxic, comparatively more so in 100% tyrosine, probably owing to the toxicity of the sudden rush of melanin derivatives and the oxidative stress generated during melanin synthesis.
2.6. Quantitative PCR (qPCR)-Based Assessment of Expression of Nucleotide Excision Repair (NER) Pathway Genes
Various cell types were collected 24-h post incubation with 200 µg/mL AZ and MBPD, with and without ssUV exposure. RNA was isolated using a Quick-RNA isolation kit (Cat. R1050, Zymo Research). The cDNA was prepared using a High-Capacity cDNA Reverse Transcription Kit (Cat. 4368814, ThermoFisher Scientific). The gene expression of XPA, XPC, and MITF genes was assessed using the following primers. β-actin was used as an internal control for normalizations and data analyses. F1-R1 and F2-R2 represents two primer pairs which were used in the qPCR experiments.
XRCC1:
F1: TGGTGCTCAGTGGCTTCCAGAA, R1: TGGGAGTGTTGGCAAAGGCACA
F2: AATGGCGAGGACCCGTATGC, R2: CACGTAGCGGATGAGCCTCC
XPA:
F1: GAAGAACCCACGCCATTCACAG, R1: CTCGGTTTTCCTGCCTCACTTC
XPC:
F1: GGTATTGTCGTGGAGAAGCAGTC, R1: CACGGTTAGAGAAGCCTTTCACC
F2: ATTGCGTGCATACCTTGCAC, R2: TATCTCCTCAAACCCTGCTC
TYRP1:
F1: AGCCACAGGATGTCACTCAGTG, R1: GCAGGGTCATATTTTCCCGTGG
F2: CCAGAAAATTCTCACAGTCAGGAG, R2: CCATATCCAAGGCCCTGACA
MITF:
F1: GATCGACCTCTACAGCAACCAG, R1: GCTCTTGCTTCAGACTCTGTGG
F2: AGCAAGAGCATTGGCTAAAGA,R2: GCATGTCTGGATCATTTGACT
DCT:
F1: GCAAGATTGCCTGTCTCTCCAG, R1: CTTGAGAGTCCAGTGTTCCGTC
F2: TTGAGAGGAGAGGAAAGGGC, R2: CACGCCATCCAAGGTCATGC
2.7. Immunofluorescence for Nitrotyrosine
Posttranslational modification nitrotyrosine was used as a marker of nitric oxide synthase (NOS) activity. Melanocytes were cultured on collagen-coated, glass-bottom dishes (cat. P35G-1.5-14-C, MatTek, Ashland, MA, USA). After various treatments, cells were fixed with 4% formaldehyde for 10 min at RT, washed with PBS, and permeabilized with 0.5% Triton-X in PBS for 10 min. After blocking for non-specific antibody binding using 5% goat serum (Cat. 16210064, ThermoFisher Scientific) and 1% globulin free BSA (Cat. 001-000-161, Jackson ImmunoResearch, West Grove, PA, USA) in 0.1% Triton-x in PBS (blocking buffer) for 2 h at room temperature, cells were incubated with 1:1000 dilution of anti-nitrotyrosine antibody (Cat. A-21285, ThermoFisher Scientific) overnight at 4 °C. Detection was conducted using Alexa Fluor 488 Goat Anti-rabbit IgG (Cat. A-11008, Life Technologies). Imaging was performed using a ZEISS Axio Observer microscope. Approximately 5–10 images per condition, with ~70–100 cells per image, were quantitated for nitrotyrosine fluorescence using ImageJ software.
4. Discussion
UV exposure damages the DNA directly through CPDs and 6-4 photoproducts (6-4PP), and indirectly through oxidative DNA damage such as 8-Oxo-guanosine (8-oxoGua) [
35]. Both types of damage lead to single-strand DNA breaks during the process of DNA repair. CPDs are the most predominant DNA lesions generated by various UV wavelengths [
36] and are responsible for >80% of mutations, called “UV signature, C to T transition mutations” in sunlight induced melanoma [
3,
37]. Accordingly, effective blockade of CPD formation is the first line of defense against the damaging effects of UV exposure and can aid in the prevention of skin cancers. While sunscreens provide effective protection against erythema and iCPD formation, it is well recognized today that sunscreens can neither prevent the formation of sun-induced reactive oxygen species (ROS) within skin, nor protect against the formation of delayed CPDs (dCPDs) [
38,
39,
40]. These deficiencies in the protective capacity of sunscreens, however, can be shored up with the inclusion of ingredients that separately provide the ability within skin either to attenuate excess ROS or block dCPD formation.
In previous research, we documented the ability of AZ, with its unique chemical structure, to neutralize all the most reactive forms of ROS, including hydroxy- and peroxy- radicals, singlet oxygen, and peroxynitrite, with high efficiencies. We also observed significant attenuation of excess ROS in keratinocytes induced by UVA exposure, and the blockade of dCPD formation after AZ was added to melanocytes following exposure to UVA [
24,
25,
26]. The ability to block dCPD formation was furthermore attributed to AZ’s capacity to scavenge peroxynitrite, reduce high-energy dioxetane intermediates on melanin via H
٠ atom donation, or function as a triplet energy acceptor for triplet melanin carbonyls (MCs), all of which could reduce the extent of dCPD formation from the melanin chemiexcitation (MeCh) pathway.
In this research, we further evaluated AZ, along with six of its structural analogs, for their effectiveness in blocking iCPD formation during direct exposure to UV in several different cell types, and dCPD formation in pigmented melanocytes. Regarding AZ’s ability to block dCPD formation, two important structure–activity relationships were revealed in these results. First, the replacement of AZ’s phenolic group with a methoxy group (MBPD), which prevents the possibility of H
٠atom donation, did not impact the ability of MBPD to block dCPD formation (
Figure 4C), suggesting that AZ’s efficacy does not arise from its ability to reduce dioxetane intermediates. Secondly, replacing the pentane-2,4-dione group with a ketone group (RMKE) completely abrogated the ability of the molecule to prevent CPD formation (
Figure 4A), which suggests that maintaining the integrity of the pentane-2,4-dione group plays a central role in dCPD inhibition. Overall, these results support that the most likely explanations for AZ’s ability to inhibit the MeCh pathway stem from its ability to scavenge peroxynitrite or function as a triplet energy quencher for MCs. Interestingly, the energy quenching of triplet carbonyls by conventional antioxidant molecules has received considerable attention as a mechanism to prevent dCPD formation [
41]. It would thus be interesting to evaluate AZ and MBPD within the same model system that was used to evaluate these other antioxidants to ascertain their triplet quenching capabilities.
The ability of AZ and several of its analogs to block iCPD formation was also a surprising discovery. Interestingly, all the analogs (TMBPD, DMBPD, and MBPD) that retained the structural integrity of pentane-2,4-dione group except AZ reduced iCPD formation to some extent in NBHF cells (
Figure 3), with the highest efficacy displayed by MBPD. Indeed, a separate dose response study (
Figure 3C) highlighted that a dose of just 10 ug/mL of MBPD inhibited iCPD formation by ~30–40%. These results indicate that these analogs operate through an energy transfer mechanism from an excited state of DNA following UV absorption to the analogs, which subsequently dissipate the excess energy as heat. This notion is further reinforced by the fact that AZ and its analogs display negligible absorption across the UV spectrum (
Supplementary Figure S1), which rules out the possibility that they could be operating through a simple sunscreen-type effect. However, additional studies are required to elucidate the possibilities for energy transfer, including singlet–singlet as well as triplet–triplet energy transfers.
Since MBPD inhibited iCPDs and dCPDs maximally at considerably lower doses and inhibited melanin synthesis, we selected this analog for further analyses. MBPD inhibited both NOS activity (
Figure 7) and melanin synthesis (
Figure 5) to a higher extent than AZ, which had minimal effect on melanin synthesis. These results also indicate the MBPD-mediated inhibition of endogenous and UV-induced MeCh since NOS and melanin constitute two of the essential requirements for this pathway. This further suggests the existence of dCPDs in pigmented melanocytes even without UV exposure, due only to chronic, endogenous melanin chemiexcitation. The ~50% drop in iCPD formation at the 0H time point (
Figure 4C) potentially arises from MBDP’s ability to impede endogenous dCPD formation from the MeCh pathway during the 24-h incubation period by inhibiting melanin synthesis, upregulating nucleotide excision repair (NER), or scavenging ONOO
¯, with the effects of NER upregulation possibly being magnified since there are now fewer “starting CPDs” to be repaired. However, based upon our previous research [
8,
9], we also predict that the amount of pre-UV CPDs should not be as high as 50% of all the CPDs generated by UV immediately after UV exposure at time 0H. Accordingly, a more detailed investigation is needed to completely understand the blockade of iCPDs by MBPD. Skin is neuronal in origin, and UV exposure also causes endocrine effects through nitric oxide [
42,
43]. However, a correlation between nitric oxide-mediated endocrine responses and iCPDs and dCPDs is beyond the scope of this manuscript.
The upregulation of NER in both pigmented and albino melanocytes by MBPD, but only in pigmented cells by AZ, suggests a specific function and close association of AZ with the melanin synthesis pathway. It is known that DNA damage enhances melanin synthesis [
44]; however, its correlation with CPDs is completely unknown. Our preliminary qPCR data, which need further validation, suggest the upregulation of two melanin synthesis pathway genes,
Dct and
Tyrp1, in the first 24 h of incubation with AZ and MBPD both (unpublished, not shown). This needs detailed investigation since the melanin estimation (
Figure 5) was performed 3-6 weeks post incubation with MBPD and AZ, while the assessment of iCPDs and dCPDs, and
Dct and
Tyrp1 expression, were performed within 24–48 h post incubation. We believe that the expressional upregulation of
Dct and
Tyrp1 mRNA is short-lived and does not translate into the pigment melanin. In addition, melanin monomers are concentrated in perinuclear-coated vesicles during melanin synthesis [
45]. These monomers are lipophilic and potentially able to enter the nucleus, suggesting the presence of melanin in the cytoplasm and nucleus, which is potentially carcinogenic owing to NOS-induced chemiexcitation [
8]. Accordingly, we predict that MBPD inhibits processes associated with either tyrosine oxidation or the assembly of melanin monomers around the nucleus, which reflects lack of dCPDs within 48 h; whereas, visible downregulation of melanin synthesis occurs only when cells have undergone enough cell divisions to lose their remaining pigment. Further investigations are underway to reveal a correlation specifically in pigmented melanocytes between iCPD inhibition by MBPD and the melanin synthesis pathway. This also includes the investigation of iCPD inhibition by MBPD and AZ, and NER upregulation by MBPD in both the pigmented and isogenic albino melanocytes.
We are establishing cell culture-based methods to transfer the pigment melanin into keratinocytes. This includes “keratinocytes–melanocytes” co-culture, the incubation of keratinocytes with isolated melanosomes, or culturing cells in melanocytic-conditioned medium. This will assist in differentiating whether the iCPD inhibition by AZ and MBPD is associated with the melanin synthesis pathway, or if just the presence of the pigment melanin is sufficient for the downstream effect. Such functions will block MeCh and thus prevent CPD generation, leading to a significant reduction in the total amount of CPDs at any time point post UV exposure.
In conclusion, we propose that AZ and MBPD are extremely effective in protecting skin cells from the carcinogenic impacts of UV exposure by blocking the formation of CPDs. Based upon the experimental outcomes, we predict that that AZ and MBPD operate through two distinct mechanisms. Both are efficient ONOO− scavengers and antioxidants and upregulate NER. However, MBPD is mostly operative through the inhibition of MeCh by inhibiting NOS and melanin synthesis, while AZ is mostly operative through NER upregulation and possibly triplet energy quenching. Detailed studies are underway to further elucidate the mechanisms responsible for CPD inhibition by both AZ and MBPD and several other AZ analogs.
In this regard, our research presented here showcases that AZ and its analog MBPD constitute two highly efficient compounds that not only inhibit the formation of dCPDs but also iCPDs. Since various cell types tolerated milli-molar (mM) concentrations of AZ and MBPD, we propose that AZ and MBPD appear to be safe and highly efficient “next-generation additives” to bolster the protection properties of sunscreens, cosmetics, or other specialized clinical skincare applications.