Molecular Mechanisms and Therapeutic Potential of α- and β-Asarone in the Treatment of Neurological Disorders
Abstract
1. Introduction
2. Occurrence, Bioavailability, and Pharmacokinetics of α- and β-Asarone
2.1. Bioavailability and Pharmacokinetics of α- and β-Asarone
2.2. Toxicology of α- and β-Asarone: Preclinical Studies
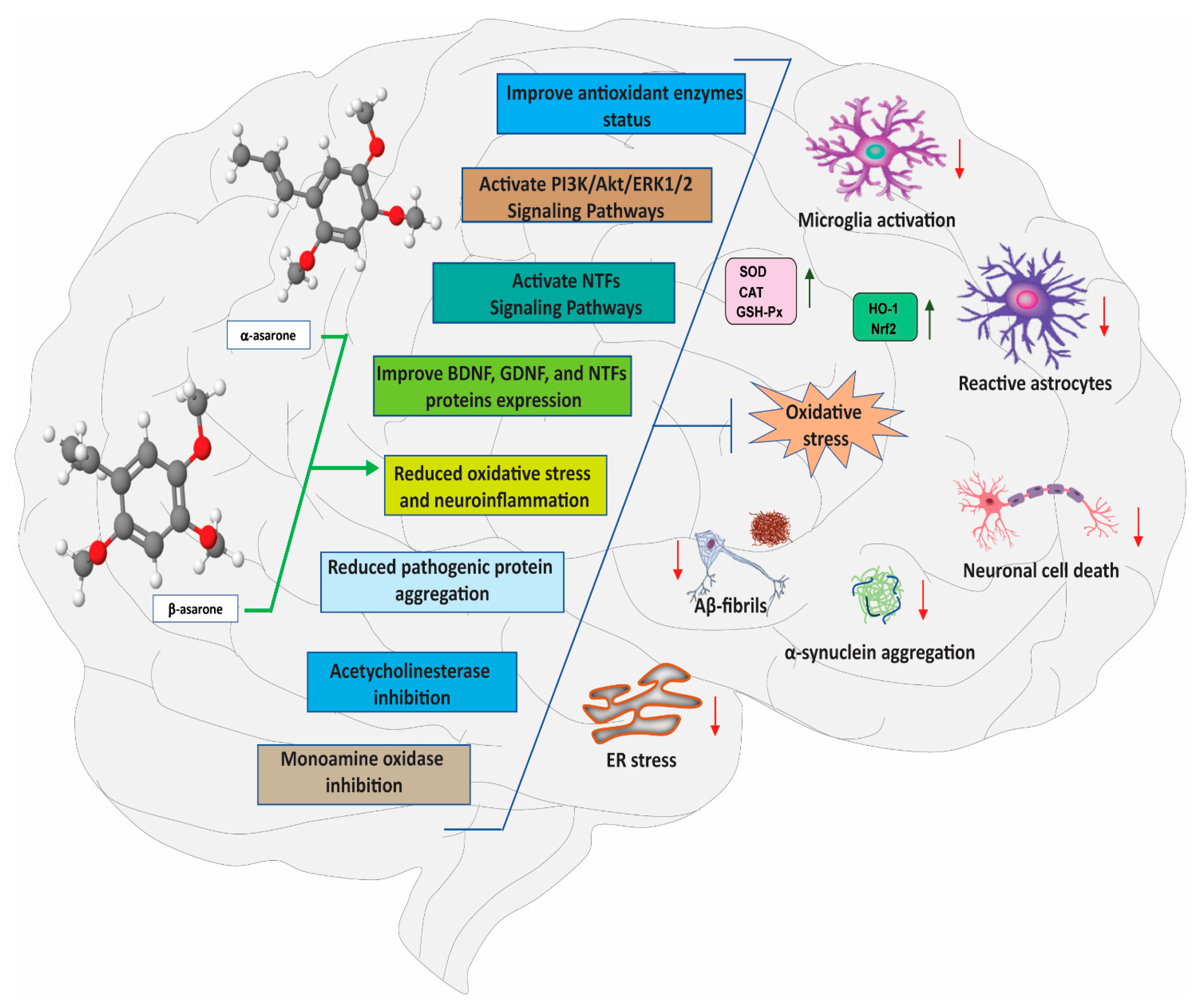
3. Neuroprotective Effects of α- and β-Asarone
3.1. Effects of α- and β-Asarone on Oxidative Stress
 ) and arrows (→) indicate inhibition and activation by α- and β-asarone, respectively. ERK, extracellular signal-regulated kinase; Akt, protein kinase B; KEAP1, Kelch-like ECH-associated protein 1; Nrf2, nuclear factor erythroid 2-related factor 2; HO-1, heme oxygenase-1; ARE, antioxidant response element; CREB, cAMP-response element-binding protein; Bcl2, B-cell lymphoma 2; NF-κB, nuclear factor-kappa B; IκB, inhibitory kappa B; ERK1/2, extracellular signal-regulated kinases 1/2; NTFs, neurotrophic factors.
) and arrows (→) indicate inhibition and activation by α- and β-asarone, respectively. ERK, extracellular signal-regulated kinase; Akt, protein kinase B; KEAP1, Kelch-like ECH-associated protein 1; Nrf2, nuclear factor erythroid 2-related factor 2; HO-1, heme oxygenase-1; ARE, antioxidant response element; CREB, cAMP-response element-binding protein; Bcl2, B-cell lymphoma 2; NF-κB, nuclear factor-kappa B; IκB, inhibitory kappa B; ERK1/2, extracellular signal-regulated kinases 1/2; NTFs, neurotrophic factors.
 ) and arrows (→) indicate inhibition and activation by α- and β-asarone, respectively. ERK, extracellular signal-regulated kinase; Akt, protein kinase B; KEAP1, Kelch-like ECH-associated protein 1; Nrf2, nuclear factor erythroid 2-related factor 2; HO-1, heme oxygenase-1; ARE, antioxidant response element; CREB, cAMP-response element-binding protein; Bcl2, B-cell lymphoma 2; NF-κB, nuclear factor-kappa B; IκB, inhibitory kappa B; ERK1/2, extracellular signal-regulated kinases 1/2; NTFs, neurotrophic factors.
) and arrows (→) indicate inhibition and activation by α- and β-asarone, respectively. ERK, extracellular signal-regulated kinase; Akt, protein kinase B; KEAP1, Kelch-like ECH-associated protein 1; Nrf2, nuclear factor erythroid 2-related factor 2; HO-1, heme oxygenase-1; ARE, antioxidant response element; CREB, cAMP-response element-binding protein; Bcl2, B-cell lymphoma 2; NF-κB, nuclear factor-kappa B; IκB, inhibitory kappa B; ERK1/2, extracellular signal-regulated kinases 1/2; NTFs, neurotrophic factors.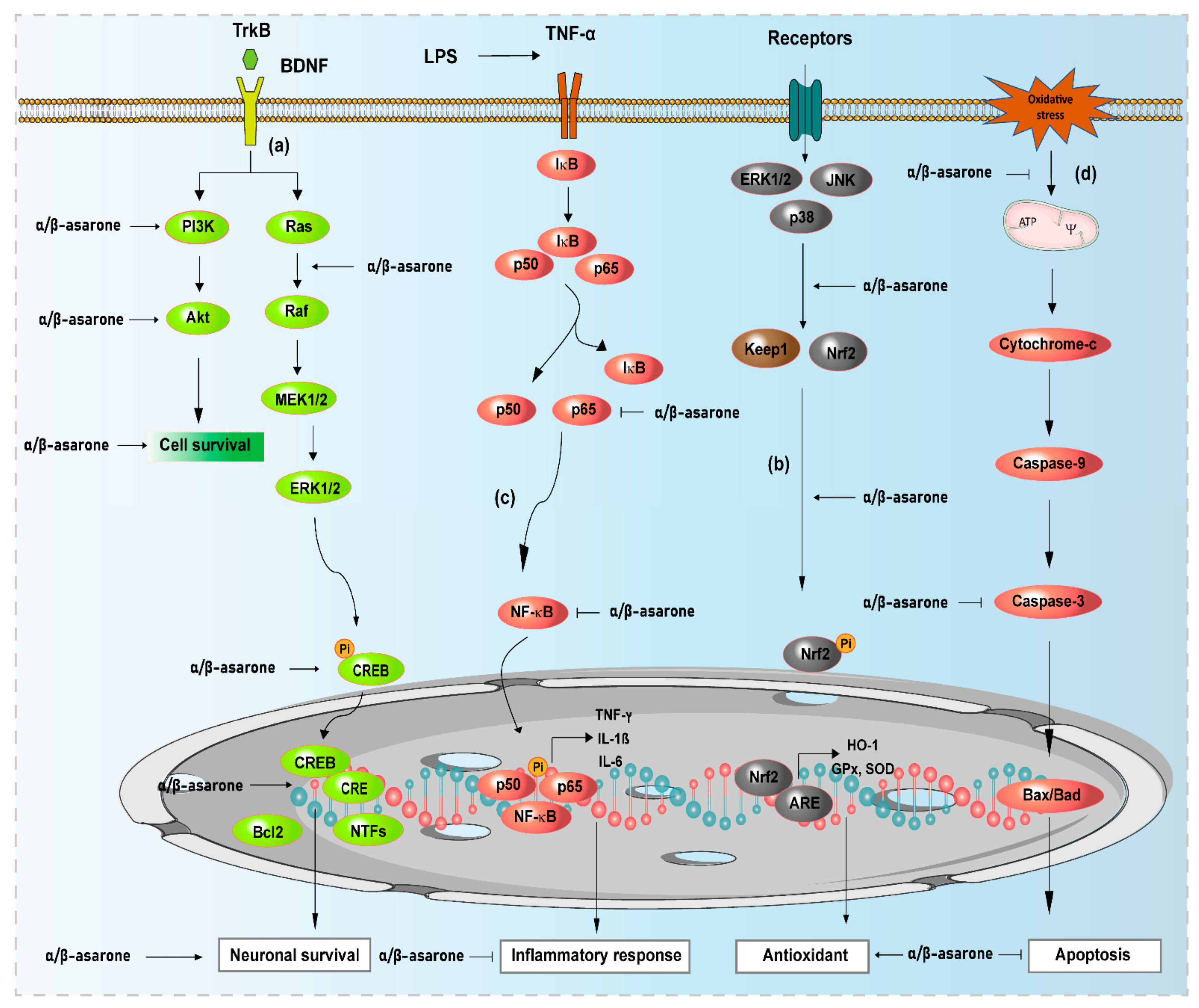
| In Vitro/In Vivo | Study Model | Main Mechanism | Dose and Route of Administration | Reference |
|---|---|---|---|---|
| α-asarone | ||||
| BV-2 | LPS/Parkinson’s disease (100 ng for 24 h) | ↓ Microglial activation ↓ Neuroinflammation ↓ NF-κB activation | α-asarone (10, 50 and 250 µM., for 24 h) | [25] |
| PC12 and cultured rat astrocytes | tBHP/Dementia (0–300 μM., for 3 h) | ↑ Neurotrophic factors ↑ Neurogenesis ↑ Akt activation ↑ Antioxidant response ↓ Oxidative stress ↓ PKA signalling ↓ Apoptosis | α- and β-asarone (15, 30 and 50 µM., 48 h) | [43,73,74] |
| Wistar rat | Noise stress/Stress model (100 dBA/4 h/d for 30 days) | ↓ Oxidative stress ↓ AChE activity ↓ HSP70mRNA expression | α-asarone (9 mg/kg−1, i.p., 30 days) | [75] |
| ICR mouse | Scopolamine hydrochloride/Alzheimer’s disease (2 mg/kg, i.p., for 2 days) | ↑ Motor performance ↓ Oxidative stress ↓ AChE activity | α-asarone (3, 10 and 30 mg/kg, p.o., 15 days) | [76] |
| C57BL/6 mice | Nicotine/Stress model (10–200 µg/mL for 40 days) | ↑ Motor performance ↑ Neurotrophic factors ↑ p-CREB protein expression ↓ Weight loss | α-asarone (5, 10 and 20 mg/kg, i.p., 8 days) | [77] |
| APP/PS1 transgenic mice | Submicron emulsion injection/Alzheimer’s disease | ↑ Motor performance ↑ Neuronal morphology ↑ Neuronal cell survival ↓ Neuroinflammation ↓ Aβ and tau aggregation ↓ Autophagosomes | α-asarone (30 and 60 mg/kg, i.p., 3 months) | [28] |
| Wistar rat | LPS/Neurotoxicity (30 µg/paw., for 6 h) | ↑ Motor performance ↑ Cognitive function ↑ Anti-inflammatory ↑ Anti-nociceptive action ↓ Neuroinflammation ↓ LPS toxicity | α-asarone (3, 10 and 30 mg/kg, p.o., for 7 h) | [78] |
| C57BL/6 mice | MPTP/Parkinson’s disease (18 mg/kg, i.p., four injections at 2 h intervals for one day) | ↑ Motor performance ↑ DA levels ↑ Cognitive function ↑ Anti-inflammatory ↑ TH-positive cells ↓ Neuroinflammation ↓ NF-κB activation | α-asarone (10 mg/kg, p.o., 15 days) | [25] |
| C57BL/6J mice | Ethanol/Dementia (Saline, i.p. + 2 g/kg ethanol, i.g., treatment duration not mentioned) | ↑ Motor performance ↑ Cognitive function ↓ NMDA receptors ↓ SYNI activity ↓ Glu levels | α-asarone (7.5, 15 and 30 mg/kg, i.p., treatment duration not mentioned) | [79] |
| Wistar rat | Submicron emulsion injection/Alzheimer’s disease | ↑ Motor performance ↑ Cognitive function ↑ Hippocampal neurons ↓ Aβ deposits | α-asarone (10 and 25 mg/kg, i.p., for 28 days) | [80] |
| β-asarone | ||||
| PC12 | H2O2/Neurotoxicity (400 µM for 24 h) | ↓ Oxidative stress ↓ ROS production ↑ Nrf2 and HO-1 activation | β-asarone (15, 30 and 60 µg/mL, for 24 h) | [71] |
| SH-SY5Y | Aβ/Alzheimer’s disease (SH-SY5Y, 20 μM for 24 h) | ↓ Oxidative stress ↓ ROS production ↓ Apoptosis ↑ ASK1 siRNA activity | β-asarone (10–100 µg/mL, for 24 h) | [81] |
| SH-SY5Y | Aβ25-35/Alzheimer’s disease (20 μM for 24 h) | ↓ Oxidative stress ↓ ROS production ↓ Neuroinflammation ↓ Apoptosis ↑ Autophagy efficiency ↑ Bcl2 protein expression | β-asarone (10, 50 and 100 µM, for 24 h) | [82] |
| Wistar rat | Middle cerebral artery occlusion (MCAO)/Ischemia | ↑ Motor performance ↓ Oxidative stress | β-asarone (10, 20 and 30 mg/kg, p.o., for 30 days) | [83] |
| Wistar rat PC12 cells | MCAO/Ischemia OGD/R for 24 h | ↑ Cell viability ↑ Motor performance ↓ Brain infarct volume ↓ Apoptosis ↓ Neuronal cell injury ↓ Neuroinflammation | α-asarone (10 and 20 mg/kg, i.v., for 24 h) α-asarone (12, 24, 48 μM for 24 h) | [84] |
| APP/PS1 transgenic mice | Alzheimer’s disease | ↓ Senile plaques ↓ Aβ40 and Aβ42 aggregation ↓ Autophagosomes ↑ p62 expression | β-asarone (10, 20 and 40 mg/kg, i.g., for 30 days) | [85] |
| Wistar rat | 6-OHDA/Parkinson’s disease (4 mg/mL, 6 µL in each rat for 30 days) | ↑ DA levels ↑ TH-positive cells ↑ HSP70 expression ↑ p62 expression ↑ Neuronal cell survival ↓ α-synuclein ↓ Macroautophagy ↓ Autophagosomes | β-asarone (15 mg/kg, i.g., for 30 days) | [29] |
| C57BL/6 mice | MK-801/Schizophrenia (0.1 mg/kg, i.p., for 7 days) | ↑ Motor performance ↑ Body weight ↑ Cognitive function ↑ Synaptophysin ↑ Postsynaptic density ↑ Cognitive function ↑ Anti-inflammatory ↓ Neuroinflammation ↓ Microglia activation | β-asarone (25 mg/kg, i.g., for 14 days) | [86] |
| Wistar rat | CUMS/Depression (CUMS for 21 days) | ↑ Motor performance ↑ Body weight ↑ Cognitive function ↑ Neurogenesis ↑ BrdU-positive cells ↑ ERK1/2 activation ↑ CREB activation | β-asarone (25 mg/kg, i.g., for 28 days) | [87] |
| PC12 | Aβ (1–42)/Alzheimer’s disease (20 μM for 24 h) | ↑ Cell viability ↑ Bcl2 protein expression ↓ Apoptosis ↓ JNK signalling | β-asarone (7.5, 15, and 30 μg/mL, for 24 h) | [88] |
| Wistar rat | 6-OHDA/Parkinson’s disease (4 µg/µL, 6 µL in each rat for 28 days) | ↑ Motor performance ↑ DA levels ↑ TH-positive cells ↑ p62 expression ↑ Bcl2 expression ↓ α-synuclein ↓ Apoptosis ↓ JNK signalling | β-asarone (10, 20, 40 and 75 mg/kg, i.g., for 28 days) | [89] |
| AβPP/PS1 double-transgenic mice | Alzheimer’s disease | ↑ Motor performance ↑ Cognitive function ↑ CREB activation ↑ Bcl2 expression ↑ CaMKII-α-positive cells ↓ Neuronal apoptosis | β-asarone (7 and 21 mg/kg, i.g., for 4 months) | [90] |
3.2. Effects of α- and β-Asarone on Neuroprotective Signaling Pathways
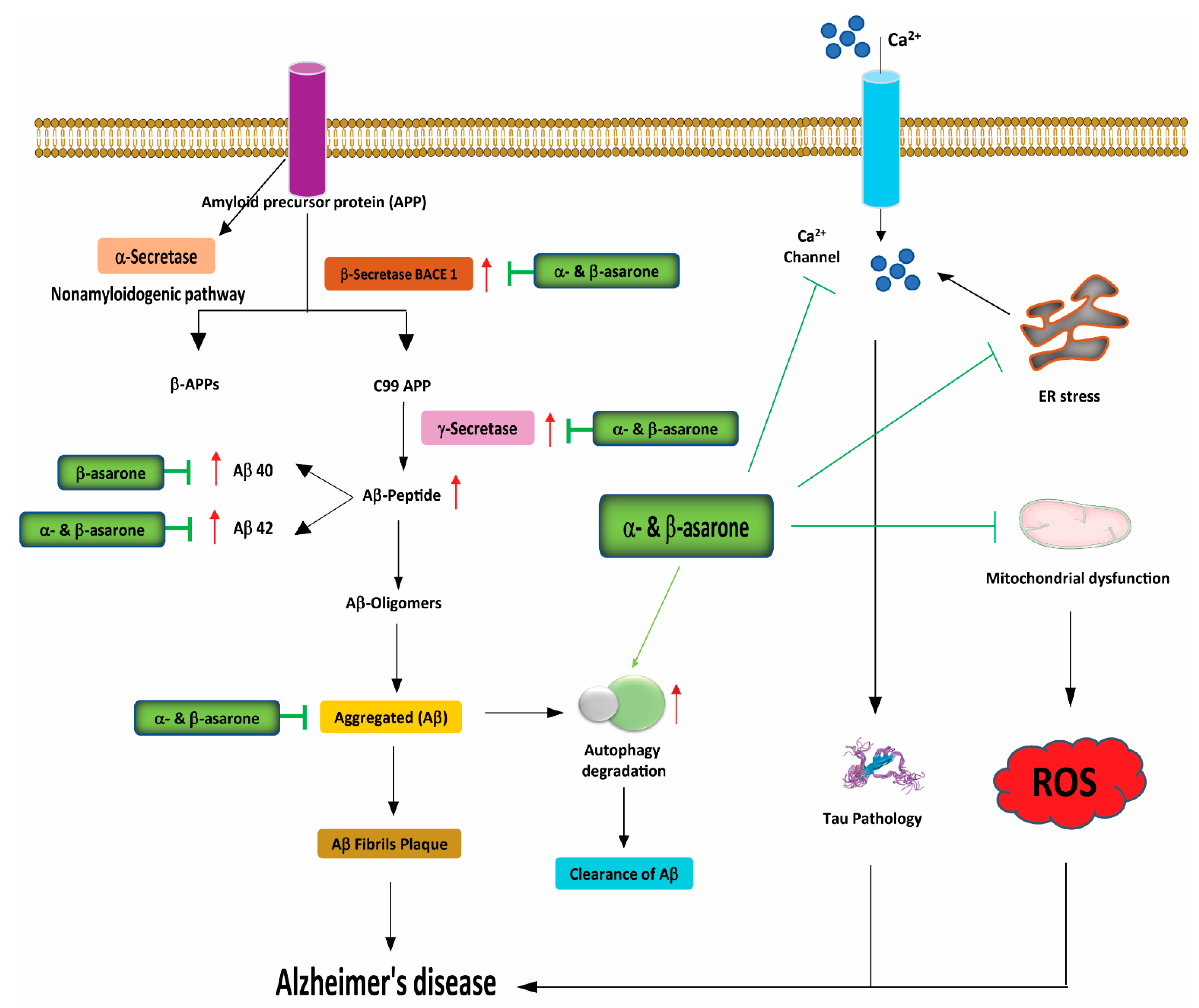
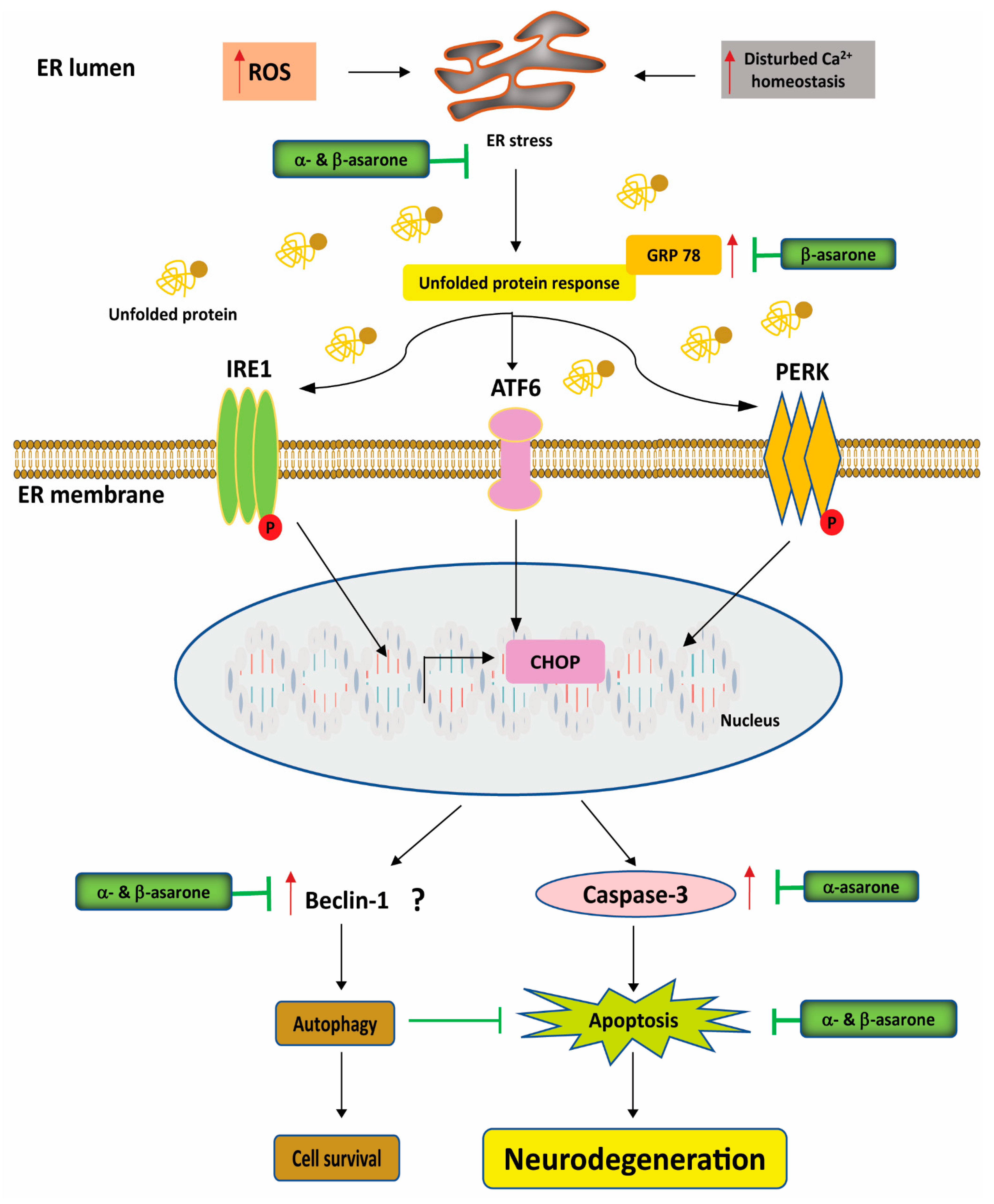
3.3. Effects of α- and β-Asarone on Proteostasis, ER Stress, and Autophagy
3.4. Effects of α- and β-Asarone on Neuroinflammation
3.5. Effects of α- and β-Asarone on Neurogenesis, Neurotransmitter Metabolism, and Neuronal Cell Death
4. Neuroprotective Effects of α- and β-Asarone on Other Neurological Disorders
5. Challenges to the Translation of the Neuroprotective Effects of α- and β-Asarone from Bench to Bedside
6. Conclusions and Future Prospects
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Neurological Disorders: Public Health Challenges. Available online: https://www.who.int/publications/i/item/9789241563369 (accessed on 5 October 2021).
- MacDonald, B.K.; Cockerell, O.C.; Sander, J.W.A.S.; Shorvon, S.D. The incidence and lifetime prevalence of neurological disorders in a prospective community-based study in the UK. Brain 2000, 123 Pt 4, 665–676. [Google Scholar] [CrossRef] [PubMed]
- Gorman, A.M. Neuronal cell death in neurodegenerative diseases: Recurring themes around protein handling. J. Cell. Mol. Med. 2008, 12, 2263–2280. [Google Scholar] [CrossRef] [PubMed]
- Iuchi, K.; Takai, T.; Hisatomi, H. Cell Death via Lipid Peroxidation and Protein Aggregation Diseases. Biology 2021, 10, 399. [Google Scholar] [CrossRef] [PubMed]
- Jha, S.K.; Jha, N.K.; Kumar, D.; Ambasta, R.K.; Kumar, P. Linking mitochondrial dysfunction, metabolic syndrome and stress signaling in Neurodegeneration. Biochim. Biophys. Acta BBA—Mol. Basis Dis. 2017, 1863, 1132–1146. [Google Scholar] [CrossRef] [PubMed]
- Jurcau, A. Insights into the Pathogenesis of Neurodegenerative Diseases: Focus on Mitochondrial Dysfunction and Oxidative Stress. Int. J. Mol. Sci. 2021, 22, 11847. [Google Scholar] [CrossRef] [PubMed]
- Deture, M.A.; Dickson, D.W. The neuropathological diagnosis of Alzheimer’s disease. Mol. Neurodegener. 2019, 14, 32. [Google Scholar] [CrossRef]
- Chen, G.F.; Xu, T.H.; Yan, Y.; Zhou, Y.R.; Jiang, Y.; Melcher, K.; Xu, H.E. Amyloid beta: Structure, biology and structure-based therapeutic development. Acta Pharmacol. Sin. 2017, 38, 1205–1235. [Google Scholar] [CrossRef]
- Sanabria-Castro, A.; Alvarado-Echeverría, I.; Monge-Bonilla, C. Molecular Pathogenesis of Alzheimer’s Disease: An Update. Ann. Neurosci. 2017, 24, 46. [Google Scholar] [CrossRef]
- Kouli, A.; Torsney, K.M.; Kuan, W.-L. Parkinson’s Disease: Etiology, Neuropathology, and Pathogenesis. In Parkinson’s Disease: Pathogenesis and Clinical Aspects; Codon Publications: Brisbane, Australia, 2018; pp. 3–26. [Google Scholar]
- Maiti, P.; Manna, J.; Dunbar, G.L.; Maiti, P.; Dunbar, G.L. Current understanding of the molecular mechanisms in Parkinson’s disease: Targets for potential treatments. Transl. Neurodegener. 2017, 6, 28. [Google Scholar] [CrossRef]
- Michel, P.P.; Hirsch, E.C.; Hunot, S. Understanding Dopaminergic Cell Death Pathways in Parkinson Disease. Neuron 2016, 90, 675–691. [Google Scholar] [CrossRef]
- Gómez-Benito, M.; Granado, N.; García-Sanz, P.; Michel, A.; Dumoulin, M.; Moratalla, R. Modeling Parkinson’s Disease With the Alpha-Synuclein Protein. Front. Pharmacol. 2020, 11, 356. [Google Scholar] [CrossRef] [PubMed]
- Musuka, T.D.; Wilton, S.B.; Traboulsi, M.; Hill, M.D. Diagnosis and management of acute ischemic stroke: Speed is critical. Can. Med. Assoc. J. 2015, 187, 887–893. [Google Scholar] [CrossRef] [PubMed]
- Numis, A.L.; Fox, C.K. Arterial ischemic stroke in children: Risk factors and etiologies. Curr. Neurol. Neurosci. Rep. 2014, 14, 422. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Li, P.; Guo, Q.; Shang, K.; Yan, D.; Du, S.; Lu, Y. Pathophysiology and Biomarkers in Acute Ischemic Stroke—A Review. Trop. J. Pharm. Res. 2014, 12, 1097–1105. [Google Scholar] [CrossRef]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. Guidelines for the Early Management of Patients with Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association. Stroke 2019, 50, E344–E418. [Google Scholar] [PubMed]
- England, M.J.; Liverman, C.T.; Schultz, A.M.; Strawbridge, L.M. Epilepsy across the spectrum: Promoting health and understanding. A summary of the Institute of Medicine report. Epilepsy Behav. 2012, 25, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Muhigwa, A.; Preux, P.M.; Gérard, D.; Marin, B.; Boumediène, F.; Ntamwira, C.; Tsai, C.H. Comorbidities of epilepsy in low and middle-income countries: Systematic review and meta-analysis. Sci. Rep. 2020, 10, 9015. [Google Scholar] [CrossRef] [PubMed]
- Pearson-Smith, J.N.; Patel, M. Metabolic Dysfunction and Oxidative Stress in Epilepsy. Int. J. Mol. Sci. 2017, 18, 2635. [Google Scholar] [CrossRef]
- Hussain, R.; Zubair, H.; Pursell, S.; Shahab, M. Neurodegenerative Diseases: Regenerative Mechanisms and Novel Therapeutic Approaches. Brain Sci. 2018, 8, 177. [Google Scholar] [CrossRef]
- Ghorbanpour, M.; Varma, A. Medicinal plants and environmental challenges. In Medicinal Plants and Environmental Challenges; Springer: Cham, Switzerland, 2017; pp. 1–413. [Google Scholar]
- Balakrishnan, R.; Azam, S.; Cho, D.Y.; Su-Kim, I.; Choi, D.K. Natural Phytochemicals as Novel Therapeutic Strategies to Prevent and Treat Parkinson’s Disease: Current Knowledge and Future Perspectives. Oxid. Med. Cell. Longev. 2021, 2021, 6680935. [Google Scholar] [CrossRef]
- Lim, H.W.; Kumar, H.; Kim, B.W.; More, S.V.; Kim, I.W.; Park, J.I.; Park, S.Y.; Kim, S.K.; Choi, D.K. β-Asarone (cis-2,4,5-trimethoxy-1-allyl phenyl), attenuates pro-inflammatory mediators by inhibiting NF-κB signaling and the JNK pathway in LPS activated BV-2 microglia cells. Food Chem. Toxicol. 2014, 72, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.W.; Koppula, S.; Kumar, H.; Park, J.Y.; Kim, I.W.; More, S.V.; Kim, I.S.; Han, S.D.; Kim, S.K.; Yoon, S.H.; et al. α-Asarone attenuates microglia-mediated neuroinflammation by inhibiting NF kappa B activation and mitigates MPTP-induced behavioral deficits in a mouse model of Parkinson’s disease. Neuropharmacology 2015, 97, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Ahn, S.M.; Pak, M.E.; Jung, D.H.; Lee, S.Y.; Shin, H.K.; Choi, B.T. Positive effects of α-asarone on transplanted neural progenitor cells in a murine model of ischemic stroke. Phytomedicine 2018, 51, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Chellian, R.; Pandy, V.; Mohamed, Z. Pharmacology and toxicology of α- and β-Asarone: A review of preclinical evidence. Phytomedicine 2017, 32, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Zhang, D.; Liu, Q.; Zhang, J.; Mu, K.; Gao, X.; Zhang, K.; Li, H.; Wang, Q.; Zheng, Y. Alpha-asarone Improves Cognitive Function of APP/PS1 Mice and Reducing Aβ 42, P-tau and Neuroinflammation, and Promoting Neuron Survival in the Hippocampus. Neuroscience 2021, 458, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Deng, M.; He, Y.; Lu, S.; Liu, S.; Fang, Y. β-asarone increases MEF2D and TH levels and reduces α-synuclein level in 6-OHDA-induced rats via regulating the HSP70/MAPK/MEF2D/Beclin-1 pathway: Chaperone-mediated autophagy activation, macroautophagy inhibition and HSP70 up-expression. Behav. Brain Res. 2016, 313, 370–379. [Google Scholar] [CrossRef]
- Han, T.; Han, P.; Peng, W.; Wang, X.R. Antidepressant-like effects of essential oil and asarone, a major essential oil component from the rhizome of Acorus tatarinowii. Pharm. Biol. 2013, 51, 589–594. [Google Scholar] [CrossRef]
- Pa, H.; Xu, Y.; Cai, Q.; Wu, M.; Din, M. Effects of β-Asarone on Ischemic Stroke in Middle Cerebral Artery Occlusion Rats by an Nrf2-Antioxidant Response Elements (ARE) Pathway-Dependent Mechanism. Med. Sci. Monit. 2021, 27, e931884. [Google Scholar]
- Rajput, S.B.; Tonge, M.B.; Karuppayil, S.M. An overview on traditional uses and pharmacological profile of Acorus calamus Linn. (Sweet flag) and other Acorus species. Phytomedicine 2014, 21, 268–276. [Google Scholar] [CrossRef]
- Chamorro, G.; Salazar, M.; Salazar, S.; Mendoza, T. Pharmacology and toxicology of Guatteria gaumeri and alpha-asarone. Rev. Investig. Clin. 1993, 45, 597–604. (In Spanish) [Google Scholar]
- Mukherjee, P.K.; Kumar, V.; Mal, M.; Houghton, P.J. Acorus calamus: Scientific Validation of Ayurvedic Tradition from Natural Resources. Pharm. Biol. 2007, 45, 651–666. [Google Scholar] [CrossRef]
- Sharma, V.; Singh, I.; Chaudhary, P. Acorus calamus (The Healing Plant): A review on its medicinal potential, micropropagation and conservation. Nat. Prod. Res. 2014, 28, 1454–1466. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Sharma, R.; Gautam, D.; Kuca, K.; Nepovimova, E.; Martins, N. Role of Vacha (Acorus calamus Linn.) in Neurological and Metabolic Disorders: Evidence from Ethnopharmacology, Phytochemistry, Pharmacology and Clinical Study. J. Clin. Med. 2020, 9, 1176. [Google Scholar] [CrossRef] [PubMed]
- Muthuraman, A.; Singh, N. Acute and sub-acute oral toxicity profile of Acorus calamus (Sweet flag) in rodents. Asian Pac. J. Trop. Biomed. 2012, 2, S1017–S1023. [Google Scholar] [CrossRef]
- Muthuraman, A.; Singh, N. Attenuating effect of Acorus calamus extract in chronic constriction injury induced neuropathic pain in rats: An evidence of anti-oxidative, anti-inflammatory, neuroprotective and calcium inhibitory effects. BMC Complement. Altern. Med. 2011, 11, 24. [Google Scholar] [CrossRef]
- Jain, N.; Jain, R.; Jain, A.; Jain, D.K.; Chandel, H.S. Evaluation of wound-healing activity of Acorus calamus Linn. Nat. Prod. Res. 2010, 24, 534–541. [Google Scholar] [CrossRef]
- Kim, H.; Han, T.H.; Lee, S.G. Anti-inflammatory activity of a water extract of Acorus calamus L. leaves on keratinocyte HaCaT cells. J. Ethnopharmacol. 2009, 122, 149–156. [Google Scholar] [CrossRef]
- Shu, H.; Zhang, S.; Lei, Q.; Zhou, J.; Ji, Y.; Luo, B.; Hong, L.; Li, F.; Liu, B.; Long, C. Ethnobotany of Acorus in China. Acta Soc. Bot. Pol. 2018, 87, 3585. [Google Scholar] [CrossRef]
- Sarjan, H.N.; Divyashree, S.; Yajurvedi, H.N. The protective effect of the Vacha rhizome extract on chronic stress-induced immunodeficiency in rat. Pharm. Biol. 2017, 55, 1358. [Google Scholar] [CrossRef]
- Lam, K.Y.C.; Wu, Q.Y.; Hu, W.H.; Yao, P.; Wang, H.Y.; Dong, T.T.X.; Tsim, K.W.K. Asarones from Acori tatarinowii Rhizoma stimulate expression and secretion of neurotrophic factors in cultured astrocytes. Neurosci. Lett. 2019, 707, 134308. [Google Scholar] [CrossRef]
- Das, B.K.; Choukimath, S.M.; Gadad, P.C. Asarone and metformin delays experimentally induced hepatocellular carcinoma in diabetic milieu. Life Sci. 2019, 230, 10–18. [Google Scholar] [CrossRef]
- Das, B.K.; Swamy, A.V.; Koti, B.C.; Gadad, P.C. Experimental evidence for use of Acorus calamus (asarone) for cancer chemoprevention. Heliyon 2019, 5, e01585. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Lee, J.Y.; Yun, B.S.; Hwang, B.K. Antifungal Activity of β-Asarone from Rhizomes of Acorus gramineus. J. Agric. Food Chem. 2004, 52, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Han, Y.; Luo, L.; Zhang, Q.; Ning, B.; Fang, Y. β-asarone induces cell apoptosis, inhibits cell proliferation and decreases migration and invasion of glioma cells. Biomed. Pharmacother. 2018, 106, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.B.; Fang, Y.Q. Pharmacokinetics of β-asarone in rats. Acta Pharm. Sin. 2004, 39, 836–838. [Google Scholar]
- Lu, J.; Fu, T.; Qian, Y.; Zhang, Q.; Zhu, H.; Pan, L.; Guo, L.; Zhang, M. Distribution of α-asarone in brain following three different routes of administration in rats. Eur. J. Pharm. Sci. 2014, 63, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.; Gong, T.; Sun, X.; Zhang, Z.; Zhang, Y. Alpha-Asarone incorporated in mixed micelles suitable for intravenous administration: Formulation, in-vivo distribution and anaphylaxis study. Pharmazie 2011, 66, 875–880. [Google Scholar]
- Liu, L.; Fang, Y.Q. Analysis of the distribution of β-asarone in rat hippocampus, brainstem, cortex and cerebellum with gas chromatography-mass spectrometry (GC-MS). J. Med. Plants Res. 2011, 5, 1728–1734. [Google Scholar]
- Meng, X.; Zhao, X.; Wang, S.; Jia, P.; Bai, Y.; Liao, S.; Zheng, X. Simultaneous Determination of Volatile Constituents from Acorus tatarinowii Schott in Rat Plasma by Gas Chromatography-Mass Spectrometry with Selective Ion Monitoring and Application in Pharmacokinetic Study. J. Anal. Methods Chem. 2013, 2013, 949830. [Google Scholar] [CrossRef]
- Qian, Y.Y.; Lu, J.; Zhang, L.H.; Shi, F.Y.; Fu, T.M.; Guo, L.W. Pharmacokinetic study on dry powder inhalation administration of α-asarone in rats. China J. Chin. Mater. Med. 2015, 40, 739–743. [Google Scholar]
- Ramalingam, P.; Ganesan, P.; Prabakaran, D.S.; Gupta, P.K.; Jonnalagadda, S.; Govindarajan, K.; Vishnu, R.; Sivalingam, K.; Sodha, S.; Choi, D.K.; et al. Lipid Nanoparticles Improve the Uptake of α-Asarone Into the Brain Parenchyma: Formulation, Characterization, In Vivo Pharmacokinetics, and Brain Delivery. AAPS PharmSciTech 2020, 21, 299. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Zhou, J.; Ju, F.; Zhu, H. Intranasal delivery of α-asarone to the brain with lactoferrin-modified mPEG-PLA nanoparticles prepared by premix membrane emulsification. Drug Deliv. Transl. Res. 2018, 8, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Guo, L.W.; Fu, T.M.; Zhu, G.L.; Dai, Z.N.; Zhan, G.J.; Chen, L.L. Pharmacokinetics of α-asarone after intranasal and intravenous administration with PLA-α-asarone nanoparticles. China J. Chin. Mater. Med. 2017, 42, 2366–2372. (In Chinese) [Google Scholar]
- Wu, J.Y.; Li, Y.J.; Yang, L.; Hu, Y.Y.; Hu, X.B.; Tang, T.T.; Wang, J.M.; Liu, X.Y.; Xiang, D.X. Borneol and a-asarone as adjuvant agents for improving blood-brain barrier permeability of puerarin and tetramethylpyrazine by activating adenosine receptors. Drug Deliv. 2018, 25, 1858–1864. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Kant, K.; Sharma, R.; Kumar, A. Evaluation of In Silico Anti-parkinson Potential of β-asarone. Cent. Nerv. Syst. Agents Med. Chem. 2018, 18, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.Q.; Shi, C.; Liu, L.; Fang, R.M. Pharmacokinetics of β-asarone in rabbit blood, hippocampus, cortex, brain stem, thalamus and cerebellum. Pharmazie 2012, 67, 120–123. [Google Scholar] [PubMed]
- Chen, Q.X.; Miao, J.K.; Li, C.; Li, X.W.; Wu, X.M.; Zhang, X.P. Anticonvulsant activity of acute and chronic treatment with a-asarone from Acorus gramineus in seizure models. Biol. Pharm. Bull. 2013, 36, 23–30. [Google Scholar] [CrossRef]
- Morales-Ramírez, P.; Madrigal-Bujaidar, E.; Mercader-Martínez, J.; Cassani, M.; González, G.; Chamorro-Cevallos, G.; Salazar-Jacobo, M. Sister-chromatid exchange induction produced by in vivo and in vitro exposure to alpha-asarone. Mutat. Res. 1992, 279, 269–273. [Google Scholar] [CrossRef]
- European Commision. Opinion of the Scientific Committee on Food on the Presence of β-Asarone in Flavourings and Other Food Ingredients with Flavouring Properties; European Commision: Brussels, Belgium, 2002; pp. 1–15. [Google Scholar]
- Liu, L.; Wang, J.; Shi, L.; Zhang, W.; Du, X.; Wang, Z.; Zhang, Y. β-Asarone induces senescence in colorectal cancer cells by inducing lamin B1 expression. Phytomedicine 2013, 20, 512–520. [Google Scholar] [CrossRef]
- Uebel, T.; Hermes, L.; Haupenthal, S.; Müller, L.; Esselen, M. α-Asarone, β-asarone, and γ-asarone: Current status of toxicological evaluation. J. Appl. Toxicol. 2021, 41, 1166–1179. [Google Scholar] [CrossRef]
- Manzanero, S.; Santro, T.; Arumugam, T.V. Neuronal oxidative stress in acute ischemic stroke: Sources and contribution to cell injury. Neurochem. Int. 2013, 62, 712–718. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.J.; Rogers, J.C.; Katshu, M.Z.U.H.; Liddle, P.F.; Upthegrove, R. Oxidative Stress and the Pathophysiology and Symptom Profile of Schizophrenia Spectrum Disorders. Front. Psychiatry 2021, 12, 703452. [Google Scholar] [CrossRef] [PubMed]
- Asha Devi, S.; Mali, A.L.; Rahee, M.A.; Belinda, E.D.S. Antioxidant properties of Alpha asarone. Asian J. Biochem. 2014, 9, 107–113. [Google Scholar] [CrossRef][Green Version]
- Parki, A.; Chaubey, P.; Prakash, O.; Kumar, R.; Pant, A.K. Seasonal Variation in Essential Oil Compositions and Antioxidant Properties of Acorus calamus L. Accessions. Medicines 2017, 4, 81. [Google Scholar] [CrossRef] [PubMed]
- Loying, R.; Gogoi, R.; Sarma, N.; Borah, A.; Munda, S.; Pandey, S.K.; Lal, M. Chemical Compositions, In-vitro Antioxidant, Anti-microbial, Anti-inflammatory and Cytotoxic Activities of Essential Oil of Acorus calamus L. Rhizome from North-East India. J. Essent. Oil Bear. Plants 2019, 22, 1299–1312. [Google Scholar] [CrossRef]
- Pages, N.; Maurois, P.; Delplanque, B.; Bac, P.; Stables, J.P.; Tamariz, J.; Chamorro, G.; Vamecq, J. Activities of α-asarone in various animal seizure models and in biochemical assays might be essentially accounted for by antioxidant properties. Neurosci. Res. 2010, 68, 337–344. [Google Scholar] [CrossRef]
- Hei, X.; Xie, M.; Xu, J.; Li, J.; Liu, T. β-Asarone Exerts Antioxidative Effects on H2O2-Stimulated PC12 Cells by Activating Nrf2/HO-1 Pathway. Neurochem. Res. 2020, 45, 1953–1961. [Google Scholar] [CrossRef]
- Meng, M.; Zhang, L.; AI, D.; Wu, H.; Peng, W. β-Asarone Ameliorates β-Amyloid–Induced Neurotoxicity in PC12 Cells by Activating P13K/Akt/Nrf2 Signaling Pathway. Front. Pharmacol. 2021, 12, 1079. [Google Scholar] [CrossRef]
- Lam, K.Y.C.; Yao, P.; Wang, H.; Duan, R.; Dong, T.T.X.; Tsim, K.W.K. Asarone from Acori tatarinowii Rhizome prevents oxidative stress-induced cell injury in cultured astrocytes: A signaling triggered by Akt activation. PLoS ONE 2017, 12, e0179077. [Google Scholar] [CrossRef]
- Lam, K.Y.C.; Chen, J.; Lam, C.T.W.; Wu, Q.; Yao, P.; Dong, T.T.X.; Lin, H.; Tsim, K.W.K. Asarone from Acori tatarinowii Rhizoma Potentiates the Nerve Growth Factor-Induced Neuronal Differentiation in Cultured PC12 Cells: A Signaling Mediated by Protein Kinase A. PLoS ONE 2016, 11, e0163337. [Google Scholar] [CrossRef]
- Sundaramahalingam, M.; Ramasundaram, S.; Rathinasamy, S.D.; Natarajan, R.P.; Somasundaram, T. Role of Acorus calamus and alpha-asarone on hippocampal dependent memory in noise stress exposed rats. Pak. J. Biol. Sci. 2013, 16, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Kim, B.W.; Song, S.Y.; Kim, J.S.; Kim, I.S.; Kwon, Y.S.; Koppula, S.; Choi, D.K. Cognitive enhancing effects of alpha asarone in amnesic mice by influencing cholinergic and antioxidant defense mechanisms. Biosci. Biotechnol. Biochem. 2012, 76, 1518–1522. [Google Scholar] [CrossRef] [PubMed]
- Chellian, R.; Pandy, V.; Mohamed, Z. Alpha-asarone attenuates depression-like behavior in nicotine-withdrawn mice: Evidence for the modulation of hippocampal pCREB levels during nicotine-withdrawal. Eur. J. Pharmacol. 2018, 818, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Saldanha, A.A.; Vieira, L.; de Oliveira, F.M.; Lopes, D.d.O.; Ribeiro, R.I.M.d.A.; Thomé, R.G.; dos Santos, H.B.; Silva, D.B.; Carollo, C.A.; de Siqueira, J.M.; et al. Anti-inflammatory and central and peripheral anti-nociceptive activities of α-asarone through the inhibition of TNF-α production, leukocyte recruitment and iNOS expression, and participation of the adenosinergic and opioidergic systems. Inflammopharmacology 2020, 28, 1039–1052. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Xu, F.; Zhang, Q.; Li, X.; Guo, M.; Zhang, Y.; Wang, Z.; Wang, J.; Zhao, J.; Tian, Y.; et al. Effect of α-asarone on ethanol-induced learning and memory impairment in mice and its underlying mechanism. Life Sci. 2019, 238, 116898. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gao, X.; Liu, Q.; Zeng, L.; Zhang, K.; Mu, K.; Zhang, D.; Zou, H.; Wu, N.; Ou, J.; et al. Alpha-asarone improves cognitive function of aged rats by alleviating neuronal excitotoxicity via GABA A receptors. Neuropharmacology 2020, 162, 107843. [Google Scholar] [CrossRef] [PubMed]
- Zou, D.J.; Wang, G.; Liu, J.C.; Dong, M.X.; Li, X.M.; Zhang, C.; Zhou, L.; Wang, R.; Niu, Y.C. Beta-asarone attenuates beta-amyloid-induced apoptosis through the inhibition of the activation of apoptosis signal-regulating kinase 1 in SH-SY5Y cells. Pharmazie 2011, 66, 44–51. [Google Scholar]
- Chang, W.; Teng, J. β-asarone prevents Aβ25-35-induced inflammatory responses and autophagy in SH-SY5Y cells: Down expression Beclin-1, LC3B and up expression Bcl-2. Int. J. Clin. Exp. Med. 2015, 8, 20658. [Google Scholar]
- Yang, Y.X.; Chen, Y.T.; Zhou, X.J.; Hong, C.L.; Li, C.Y.; Guo, J.Y. Beta-asarone, a major component of Acorus tatarinowii Schott, attenuates focal cerebral ischemia induced by middle cerebral artery occlusion in rats. BMC Complement. Altern. Med. 2013, 13, 236. [Google Scholar] [CrossRef]
- Zhang, K.; Liu, Q.; Luo, L.; Feng, X.; Hu, Q.; Fan, X.; Mao, S. Neuroprotective Effect of Alpha-asarone on the Rats Model of Cerebral Ischemia-Reperfusion Stroke via Ameliorating Glial Activation and Autophagy. Neuroscience 2021, 473, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Huang, L.; Zhong, X. β-asarone modulates Beclin-1, LC3 and p62 expression to attenuate Aβ40 and Aβ42 levels in APP/PS1 transgenic mice with Alzheimer’s disease. Mol. Med. Rep. 2020, 21, 2095–2102. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Xu, X.; Li, F.; Xie, G.; Zhang, T. Anti-inflammatory treatment with β-asarone improves impairments in social interaction and cognition in MK-801 treated mice. Brain Res. Bull. 2019, 150, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Gao, Z.; Rong, H.; Jin, M.; Zhang, X. β-asarone reverses chronic unpredictable mild stress-induced depression-like behavior and promotes hippocampal neurogenesis in rats. Molecules 2014, 19, 5634–5649. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Xing, G.; Dong, M.; Zhou, L.; Li, J.; Wang, G.; Zou, D.; Wang, R.; Liu, J.; Niu, Y. Beta-asarone protection against beta-amyloid-induced neurotoxicity in PC12 cells via JNK signaling and modulation of Bcl-2 family proteins. Eur. J. Pharmacol. 2010, 635, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Gui, X.H.; Huang, L.P.; Deng, M.Z.; Fang, R.M.; Ke, X.H.; He, Y.P.; Li, L.; Fang, Y.Q. Neuroprotective Effects of β-Asarone Against 6-Hydroxy Dopamine-Induced Parkinsonism via JNK/Bcl-2/Beclin-1 Pathway. Mol. Neurobiol. 2016, 53, 83–94. [Google Scholar] [CrossRef]
- Wei, G.; Chen, Y.B.; Chen, D.F.; Lai, X.P.; Liu, D.H.; Deng, R.D.; Zhou, J.H.; Zhang, S.X.; Li, Y.W.; Lii, H.; et al. β-Asarone inhibits neuronal apoptosis via the CaMKII/CREB/Bcl-2 signaling pathway in an in vitro model and AβPP/PS1 mice. J. Alzheimer’s Dis. 2013, 33, 863–880. [Google Scholar] [CrossRef]
- Saki, G.; Eidi, A.; Mortazavi, P.; Panahi, N.; Vahdati, A. Effect of β-asarone in normal and β-amyloid-induced Alzheimeric rats. Arch. Med. Sci. 2020, 16, 699–706. [Google Scholar] [CrossRef]
- Manikandan, S.; Devi, R.S. Antioxidant property of alpha-asarone against noise-stress-induced changes in different regions of rat brain. Pharmacol. Res. 2005, 52, 467–474. [Google Scholar] [CrossRef]
- Sandeep, D.; Nair, C.K.K. Radioprotection by α-asarone: Prevention of genotoxicity and hematopoietic injury in mammalian organism. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2011, 722, 62–68. [Google Scholar] [CrossRef]
- Wang, B.L.; Xuan, L.; Dai, S.J.; Ji, L.T.; Li, C.Y.; Yang, Y.X. Protective effect of β-asarone on AD rat model induced by intracerebroventricular injection of Aβ₁₋₄₂ combined 2-VO and its mechanism. China J. Chin. Mater. Med. 2017, 42, 4847–4854. (In Chinese) [Google Scholar]
- Sánchez-Alegría, K.; Flores-León, M.; Avila-Muñoz, E.; Rodríguez-Corona, N.; Arias, C. PI3K Signaling in Neurons: A Central Node for the Control of Multiple Functions. Int. J. Mol. Sci. 2018, 19, 3725. [Google Scholar] [CrossRef] [PubMed]
- Long, H.Z.; Cheng, Y.; Zhou, Z.W.; Luo, H.Y.; Wen, D.D.; Gao, L.C. PI3K/AKT Signal Pathway: A Target of Natural Products in the Prevention and Treatment of Alzheimer’s Disease and Parkinson’s Disease. Front. Pharmacol. 2021, 12, 648636. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Nan, G. The extracellular signal-regulated kinase 1/2 pathway in neurological diseases: A potential therapeutic target (Review). Int. J. Mol. Med. 2017, 39, 1338–1346. [Google Scholar] [CrossRef] [PubMed]
- Naoi, M.; Shamoto-Nagai, M.; Maruyama, W. Neuroprotection of multifunctional phytochemicals as novel therapeutic strategy for neurodegenerative disorders: Antiapoptotic and antiamyloidogenic activities by modulation of cellular signal pathways. Future Neurol. 2019, 14, FNL9. [Google Scholar] [CrossRef]
- Xue, Z.; Guo, Y.; Zhang, S.; Huang, L.; He, Y.; Fang, R.; Fang, Y. Beta-asarone attenuates amyloid beta-induced autophagy via Akt/mTOR pathway in PC12 cells. Eur. J. Pharmacol. 2014, 741, 195–204. [Google Scholar] [CrossRef]
- He, Y.; He, J.N.; Fu, J.; Bao, Y.T.; Li, C.Y.; Yang, Y.X. Protective effect of β-asarone on PC12 cells injury induced by Aβ₁₋₄₂ astrocytic activation. China J. Chin. Mater. Med. 2016, 41, 1282–1288. (In Chinese) [Google Scholar]
- Mao, J.; Huang, S.; Liu, S.; Feng, X.L.; Yu, M.; Liu, J.; Sun, Y.E.; Chen, G.; Yu, Y.; Zhao, J.; et al. A herbal medicine for Alzheimer’s disease and its active constituents promote neural progenitor proliferation. Aging Cell 2015, 14, 784–796. [Google Scholar] [CrossRef]
- Soto, C.; Pritzkow, S. Protein misfolding, aggregation, and conformational strains in neurodegenerative diseases. Nat. Neurosci. 2018, 21, 1332–1340. [Google Scholar] [CrossRef]
- Vasili, E.; Dominguez-Meijide, A.; Outeiro, T.F. Spreading of α-synuclein and tau: A systematic comparison of the mechanisms involved. Front. Mol. Neurosci. 2019, 12, 107. [Google Scholar] [CrossRef]
- Kumar, D.; Kumar, P. Aβ, Tau, and α-Synuclein aggregation and integrated role of PARK2 in the regulation and clearance of toxic peptides. Neuropeptides 2019, 78, 101971. [Google Scholar] [CrossRef]
- Ciechanover, A.; Kwon, Y.T. Degradation of misfolded proteins in neurodegenerative diseases: Therapeutic targets and strategies. Exp. Mol. Med. 2015, 47, e147. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.L.; Gandhi, S. Crucial role of protein oligomerization in the pathogenesis of Alzheimer’s and Parkinson’s diseases. FEBS J. 2018, 285, 3631–3644. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Lipton, S.A. Cell death: Protein misfolding and neurodegenerative diseases. Apoptosis 2009, 14, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, P.; Park, H.; Baumann, M.; Dunlop, J.; Frydman, J.; Kopito, R.; McCampbell, A.; Leblanc, G.; Venkateswaran, A.; Nurmi, A. Protein misfolding in neurodegenerative diseases: Implications and strategies. Transl. Neurodegener. 2017, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Pietrobono, D.; Giacomelli, C.; Trincavelli, M.L.; Daniele, S.; Martini, C. Inhibitors of protein aggregates as novel drugs in neurodegenerative diseases. Glob. Drugs Ther. 2017, 2, 1–5. [Google Scholar]
- Bieschke, J. Natural Compounds May Open New Routes to Treatment of Amyloid Diseases. Neurotherapeutics 2013, 10, 429–439. [Google Scholar] [CrossRef]
- Javed, H.; Nagoor Meeran, M.F.; Azimullah, S.; Adem, A.; Sadek, B.; Ojha, S.K. Plant Extracts and Phytochemicals Targeting α-Synuclein Aggregation in Parkinson’s Disease Models. Front. Pharmacol. 2019, 9, 1555. [Google Scholar] [CrossRef]
- Kabir, M.T.; Uddin, M.S.; Begum, M.M.; Thangapandiyan, S.; Rahman, M.S.; Aleya, L.; Mathew, B.; Ahmed, M.; Barreto, G.E.; Ashraf, G.M. Cholinesterase Inhibitors for Alzheimer’s Disease: Multitargeting Strategy Based on Anti-Alzheimer’s Drugs Repositioning. Curr. Pharm. Des. 2019, 25, 3519–3535. [Google Scholar] [CrossRef]
- Shin, J.W.; Cheong, Y.J.; Koo, Y.M.; Kim, S.; Noh, C.K.; Son, Y.H.; Kang, C.; Sohn, N.W. α-Asarone ameliorates memory deficit in lipopolysaccharide-treated mice via suppression of pro-inflammatory cytokines and microglial activation. Biomol. Ther. 2014, 22, 17–26. [Google Scholar] [CrossRef]
- Wang, N.; Wang, H.; Li, L.; Li, Y.; Zhang, R. β-Asarone Inhibits Amyloid-β by Promoting Autophagy in a Cell Model of Alzheimer’s Disease. Front. Pharmacol. 2020, 10, 1529. [Google Scholar] [CrossRef]
- Lee, J.E.; Kim, N.; Yeo, J.Y.; Seo, D.G.; Kim, S.; Lee, J.S.; Hwang, K.W.; Park, S.Y. Anti-Amyloidogenic Effects of Asarone Derivatives From Perilla frutescens Leaves against Beta-Amyloid Aggregation and Nitric Oxide Production. Molecules 2019, 24, 4297. [Google Scholar] [CrossRef] [PubMed]
- Risiglione, P.; Zinghirino, F.; Di Rosa, M.C.; Magrì, A.; Messina, A. Alpha-Synuclein and Mitochondrial Dysfunction in Parkinson’s Disease: The Emerging Role of VDAC. Biomolecules 2021, 11, 718. [Google Scholar] [CrossRef] [PubMed]
- Moloney, T.C.; Hyland, R.; O’Toole, D.; Paucard, A.; Kirik, D.; O’Doherty, A.; Gorman, A.M.; Dowd, E. Heat shock protein 70 reduces α-synuclein-induced predegenerative neuronal dystrophy in the α-synuclein viral gene transfer rat model of Parkinson’s disease. CNS Neurosci. Ther. 2014, 20, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Wolfer, D.P.; Lipp, H.P.; Büeler, H. Hsp70 gene transfer by adeno-associated virus inhibits MPTP-induced nigrostriatal degeneration in the mouse model of Parkinson disease. Mol. Ther. 2005, 1, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.S.; Wang, Z.H.; Zhang, J.L.; Duan, Y.L.; Li, G.F.; Zheng, D.L. Beta-asarone protects against MPTP-induced Parkinson’s disease via regulating long non-coding RNA MALAT1 and inhibiting α-synuclein protein expression. Biomed. Pharmacother. 2016, 83, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Lehman, N.L. The ubiquitin proteasome system in neuropathology. Acta Neuropathol. 2009, 118, 329–347. [Google Scholar] [CrossRef]
- Cook, C.; Stetler, C.; Petrucelli, L. Disruption of protein quality control in Parkinson’s disease. Cold Spring Harb. Perspect. Med. 2012, 2, a009423. [Google Scholar] [CrossRef]
- Sprenkle, N.T.; Sims, S.G.; Sánchez, C.L.; Meares, G.P. Endoplasmic reticulum stress and inflammation in the central nervous system. Mol. Neurodegener. 2017, 12, 42. [Google Scholar] [CrossRef]
- Sano, R.; Reed, J.C. ER stress-induced cell death mechanisms. Biochim. Biophys. Acta 2013, 1833, 3460–3470. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.V.; Bredesen, D.E. Misfolded proteins, endoplasmic reticulum stress and neurodegeneration. Curr. Opin. Cell Biol. 2004, 16, 653–662. [Google Scholar] [CrossRef]
- Høyer-Hansen, M.; Jäättelä, M. Connecting endoplasmic reticulum stress to autophagy by unfolded protein response and calcium. Cell Death Differ. 2007, 14, 1576–1582. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Niemeijer, M.; van de Water, B.; Beltman, J.B. ATF6 Is a Critical Determinant of CHOP Dynamics during the Unfolded Protein Response. iScience 2020, 23, 100860. [Google Scholar] [CrossRef] [PubMed]
- Ryu, E.J.; Harding, H.P.; Angelastro, J.M.; Vitolo, O.V.; Ron, D.; Greene, L.A. Endoplasmic reticulum stress and the unfolded protein response in cellular models of Parkinson’s disease. J. Neurosci. 2002, 22, 10690–10698. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Gan, M.; Ebrahim, A.S.; Lin, W.L.; Melrose, H.L.; Yen, S.H.C. ER stress response plays an important role in aggregation of α-synuclein. Mol. Neurodegener. 2010, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Holtz, W.A.; Turetzky, J.M.; Jong, Y.J.I.; O’Malley, K.L. Oxidative stress-triggered unfolded protein response is upstream of intrinsic cell death evoked by parkinsonian mimetics. J. Neurochem. 2006, 99, 54–69. [Google Scholar] [CrossRef]
- Ning, B.; Deng, M.; Zhang, Q.; Wang, N.; Fang, Y. β-Asarone Inhibits IRE1/XBP1 Endoplasmic Reticulum Stress Pathway in 6-OHDA-Induced Parkinsonian Rats. Neurochem. Res. 2016, 41, 2097–2101. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Endoplasmic Reticulum Stress and the Inflammatory Basis of Metabolic Disease. Cell 2010, 140, 900–917. [Google Scholar] [CrossRef]
- Ning, B.; Zhang, Q.; Wang, N.; Deng, M.; Fang, Y. β-Asarone Regulates ER Stress and Autophagy Via Inhibition of the PERK/CHOP/Bcl-2/Beclin-1 Pathway in 6-OHDA-Induced Parkinsonian Rats. Neurochem. Res. 2019, 44, 1159–1166. [Google Scholar] [CrossRef]
- Park, S.H.; Kang, M.K.; Choi, Y.J.; Kim, Y.H.; Antika, L.D.; Lim, S.S.; Kang, Y.H. Dietary compound α-asarone alleviates ER stress-mediated apoptosis in 7β-hydroxycholesterol-challenged macrophages. Mol. Nutr. Food Res. 2016, 60, 1033–1047. [Google Scholar] [CrossRef]
- Gui, Y.; Li, A.; Zhang, J.; Li, G.; Ruan, X.; Guo, Q.; Zou, W. α-Asarone alleviated chronic constriction injury–induced neuropathic pain through inhibition of spinal endoplasmic reticulum stress in an liver X receptor–dependent manner. Anesth. Analg. 2018, 127, 775–783. [Google Scholar] [CrossRef]
- Mikami, M.; Takuya, O.; Yoshino, Y.; Nakamura, S.; Ito, K.; Kojima, H.; Takahashi, T.; Iddamalgoda, A.; Inoue, S.; Shimazawa, M. Acorus calamus extract and its component α-asarone attenuate murine hippocampal neuronal cell death induced by l-glutamate and tunicamycin. Biosci. Biotechnol. Biochem. 2021, 85, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Kang, J.H.; Lee, S. Autophagy in Neurodegenerative Diseases: A Hunter for Aggregates. Int. J. Mol. Sci. 2020, 21, 3369. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Rahman, M.R.; Zaman, T.; Uddin, M.S.; Islam, R.; Abdel-Daim, M.M.; Rhim, H. Emerging Potential of Naturally Occurring Autophagy Modulators Against Neurodegeneration. Curr. Pharm. Des. 2020, 26, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, A.; Liu, M.; Zheng, Y.; Yan, W.; Pan, L.; Li, Y.; Ma, S.; Zhang, X.; Cao, M.; Wu, Z.; et al. Natural compounds modulate the autophagy with potential implication of stroke. Acta Pharm. Sin. B 2021, 11, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Kang, M.K.; Choi, Y.J.; Kim, Y.H.; Antika, L.D.; Kim, D.Y.; Lee, E.J.; Lim, S.S.; Kang, Y.H. α-Asarone blocks 7β-hydroxycholesterol-exposed macrophage injury through blocking elF2α phosphorylation and prompting beclin-1-dependent autophagy. Oncotarget 2017, 8, 7370–7383. [Google Scholar] [CrossRef][Green Version]
- Harry, G.J.; Kraft, A.D. Microglia in the developing brain: A potential target with lifetime effects. Neurotoxicology 2012, 33, 191–206. [Google Scholar] [CrossRef]
- Jeong, H.-K.; Ji, K.; Min, K.; Joe, E.-H. Brain inflammation and microglia: Facts and misconceptions. Exp. Neurobiol. 2013, 22, 59–67. [Google Scholar] [CrossRef]
- Lyman, M.; Lloyd, D.G.; Ji, X.; Vizcaychipi, M.P.; Ma, D. Neuroinflammation: The role and consequences. Neurosci. Res. 2014, 79, 1–12. [Google Scholar] [CrossRef]
- Liu, C.Y.; Wang, X.; Liu, C.; Zhang, H.L. Pharmacological Targeting of Microglial Activation: New Therapeutic Approach. Front. Cell. Neurosci. 2019, 13, 514. [Google Scholar] [CrossRef]
- Bachiller, S.; Jiménez-Ferrer, I.; Paulus, A.; Yang, Y.; Swanberg, M.; Deierborg, T.; Boza-Serrano, A. Microglia in Neurological Diseases: A Road Map to Brain-Disease Dependent-Inflammatory Response. Front. Cell. Neurosci. 2018, 12, 488. [Google Scholar] [CrossRef]
- Fatoba, O.; Itokazu, T.; Yamashita, T. Microglia as therapeutic target in central nervous system disorders. J. Pharmacol. Sci. 2020, 144, 102–118. [Google Scholar] [CrossRef] [PubMed]
- Moussa, C.; Hebron, M.; Huang, X.; Ahn, J.; Rissman, R.A.; Aisen, P.S.; Turner, R.S. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J. Neuroinflamm. 2017, 14, 61. [Google Scholar] [CrossRef] [PubMed]
- Kaur, N.; Chugh, H.; Sakharkar, M.K.; Dhawan, U.; Chidambaram, S.B.; Chandra, R. Neuroinflammation Mechanisms and Phytotherapeutic Intervention: A Systematic Review. ACS Chem. Neurosci. 2020, 11, 3707–3731. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.J.; Lai, X.; Xu, Y.; Miao, J.K.; Li, C.; Liu, J.Y.; Hua, Y.Y.; Ma, Q.; Chen, Q. α-Asarone Attenuates Cognitive Deficit in a Pilocarpine-Induced Status Epilepticus Rat Model via a Decrease in the Nuclear Factor-κB Activation and Reduction in Microglia Neuroinflammation. Front. Neurol. 2017, 8, 661. [Google Scholar] [CrossRef] [PubMed]
- Jo, M.J.; Kumar, H.; Joshi, H.P.; Choi, H.; Ko, W.K.; Kim, J.M.; Hwang, S.S.S.; Park, S.Y.; Sohn, S.; Bello, A.B.; et al. Oral Administration of α-Asarone Promotes Functional Recovery in Rats With Spinal Cord Injury. Front. Pharmacol. 2018, 9, 445. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Li, Y.; Mao, J.; Pei, G. Neurogenesis-promoting natural product α-asarone modulates morphological dynamics of activated microglia. Front. Cell. Neurosci. 2016, 10, 280. [Google Scholar] [CrossRef]
- Reichardt, L.F. Neurotrophin-regulated signalling pathways. Philos. Trans. R. Soc. B Biol. Sci. 2006, 361, 1545. [Google Scholar] [CrossRef]
- Zhang, H.; Ozbay, F.; Lappalainen, J.; Kranzler, H.R.; Van Dyck, C.H.; Charney, D.S.; Price, L.H.; Southwick, S.; Yang, B.Z.; Rasmussen, A.; et al. Brain derived neurotrophic factor (BDNF) gene variants and Alzheimer’s disease, affective disorders, posttraumatic stress disorder, schizophrenia, and substance dependence. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2006, 141, 387–393. [Google Scholar] [CrossRef]
- Kumar Gupta, V.; Sharma, B. Role of Phytochemicals in Neurotrophins Mediated Regulation of Alzheimer’s Disease. Int. J. Complement. Altern. Med. 2017, 7, 00231. [Google Scholar]
- Maswood, N.; Grondin, R.; Zhang, Z.; Stanford, J.A.; Surgener, S.P.; Gash, D.M.; Gerhardt, G.A. Effects of chronic intraputamenal infusion of glial cell line-derived neurotrophic factor (GDNF) in aged Rhesus monkeys. Neurobiol. Aging 2002, 23, 881–889. [Google Scholar] [CrossRef]
- Grondin, R.; Zhang, Z.; Yi, A.; Cass, W.A.; Maswood, N.; Andersen, A.H.; Elsberry, D.D.; Klein, M.C.; Gerhardt, G.A.; Gash, D.M. Chronic, controlled GDNF infusion promotes structural and functional recovery in advanced parkinsonian monkeys. Brain 2002, 125, 2191–2201. [Google Scholar] [CrossRef] [PubMed]
- Grondin, R.; Cass, W.A.; Zhang, Z.; Stanford, J.A.; Gash, D.M.; Gerhardt, G.A. Glial cell line-derived neurotrophic factor increases stimulus-evoked dopamine release and motor speed in aged rhesus monkeys. J. Neurosci. 2003, 23, 1974–1980. [Google Scholar] [CrossRef] [PubMed]
- Stahl, K.; Mylonakou, M.N.; Stahl, K.; Amiry-Moghaddam, M.; Torp, R. Cytoprotective effects of growth factors: BDNF more potent than GDNF in an organotypic culture model of Parkinson’s disease. Brain Res. 2011, 1378, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Takeda, M. Intrathecal infusion of brain-derived neurotrophic factor protects nigral dopaminergic neurons from degenerative changes in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced monkey parkinsonian model. Hokkaido J. Med. Sci. 1995, 70, 829–838. [Google Scholar] [PubMed]
- Cui, X.; Lin, Q.; Liang, Y. Plant-Derived Antioxidants Protect the Nervous System From Aging by Inhibiting Oxidative Stress. Front. Aging Neurosci. 2020, 12, 209. [Google Scholar] [CrossRef]
- Dong, H.; Cong, W.; Guo, X.; Wang, Y.; Tong, S.; Li, Q.; Li, C. β-asarone relieves chronic unpredictable mild stress induced depression by regulating the extracellular signal-regulated kinase signaling pathway. Exp. Ther. Med. 2019, 18, 3767–3774. [Google Scholar] [CrossRef]
- Lee, B.; Sur, B.; Cho, S.G.; Yeom, M.; Shim, I.; Lee, H.; Hahm, D.H. Effect of beta-asarone on impairment of spatial working memory and apoptosis in the hippocampus of rats exposed to chronic corticosterone administration. Biomol. Ther. 2015, 23, 571–581. [Google Scholar] [CrossRef]
- Naoi, M.; Maruyama, W.; Shamoto-Nagai, M. Type A monoamine oxidase and serotonin are coordinately involved in depressive disorders: From neurotransmitter imbalance to impaired neurogenesis. J. Neural Transm. 2018, 125, 53–66. [Google Scholar] [CrossRef]
- Finberg, J.P.M.; Rabey, J.M. Inhibitors of MAO-A and MAO-B in psychiatry and neurology. Front. Pharmacol. 2016, 7, 340. [Google Scholar] [CrossRef]
- Huang, L.; Deng, M.; Fang, Y.; Li, L. Dynamic changes of five neurotransmitters and their related enzymes in various rat tissues following β-asarone and levodopa co-administration. Exp. Ther. Med. 2015, 10, 1566–1572. [Google Scholar] [CrossRef]
- Huang, L.; Deng, M.; Zhang, S.; Fang, Y.; Li, L. Coadministration of β-asarone and levodopa increases dopamine in rat brain by accelerating transformation of levodopa: A different mechanism from Madopar. Clin. Exp. Pharmacol. Physiol. 2014, 41, 685–690. [Google Scholar] [PubMed]
- Huang, L.; Deng, M.; Zhang, S.; Lu, S.; Gui, X.; Fang, Y. β-asarone and levodopa coadministration increases striatal levels of dopamine and levodopa and improves behavioral competence in Parkinson’s rat by enhancing dopa decarboxylase activity. Biomed. Pharmacother. 2017, 94, 666–678. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.Q.; Xue, W.Z.; Zou, R.X.; Xu, Y.; Du, Y.; Wang, S.; Xu, L.; Chen, Y.Z.; Wang, H.L.; Chen, X.T. β-Asarone Rescues Pb-Induced Impairments of Spatial Memory and Synaptogenesis in Rats. PLoS ONE 2016, 11, e0167401. [Google Scholar] [CrossRef]
- Jiménez-Balado, J.; Eich, T.S. GABAergic dysfunction, neural network hyperactivity and memory impairments in human aging and Alzheimer’s disease. Semin. Cell Dev. Biol. 2021, 116, 146–159. [Google Scholar] [CrossRef] [PubMed]
- Jakaria, M.; Park, S.Y.; Haque, M.; Karthivashan, G.; Kim, I.S.; Ganesan, P.; Choi, D.K. Neurotoxic agent-induced injury in neurodegenerative disease model: Focus on involvement of glutamate receptors. Front. Mol. Neurosci. 2018, 11, 307. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.J. Mitochondrial and Cell Death Mechanisms in Neurodegenerative Diseases. Pharmaceuticals 2010, 3, 839–915. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, M.; Jiang, J. Mitochondrial dysfunction in neurodegenerative diseases and drug targets via apoptotic signaling. Mitochondrion 2019, 49, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.; Ho Kim, Y.; Kong, J.Y.; Ha Yang, C.; Gook Park, C. Protection of cultured rat cortical neurons from excitotoxicity by asarone, a major essential oil component in the rhizomes of Acorus gramineus. Life Sci. 2002, 71, 591–599. [Google Scholar] [CrossRef]
- Irie, Y.; Keung, W.M. Rhizoma Acori graminei and its active principles protect PC-12 cells from the toxic effect of amyloid-beta peptide. Brain Res. 2003, 963, 282–289. [Google Scholar] [CrossRef]
- Tatton, W.G.; Olanow, C.W. Apoptosis in neurodegenerative diseases: The role of mitochondria. Biochim. Biophys. Acta BBA—Bioenerg. 1999, 1410, 195–213. [Google Scholar] [CrossRef]
- KA, J. Cell death mechanisms in neurodegeneration. J. Cell. Mol. Med. 2001, 5, 1–17. [Google Scholar]
- Fan, J.; Dawson, T.M.; Dawson, V.L. Cell Death Mechanisms of Neurodegeneration. Adv. Neurobiol. 2017, 15, 403–425. [Google Scholar] [PubMed]
- Geng, Y.; Li, C.; Liu, J.; Xing, G.; Zhou, L.; Dong, M.; Li, X.; Niu, Y. Beta-asarone improves cognitive function by suppressing neuronal apoptosis in the beta-amyloid hippocampus injection rats. Biol. Pharm. Bull. 2010, 33, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; She, H.; Li, W.; Yang, Q.; Guo, S.; Mao, Z. Modulation of Neuronal Survival Factor MEF2 by Kinases in Parkinson’s Disease. Front. Physiol. 2012, 3, 171. [Google Scholar] [CrossRef] [PubMed]
- She, H.; Yang, Q.; Shepherd, K.; Smith, Y.; Miller, G.; Testa, C.; Mao, Z. Direct regulation of complex I by mitochondrial MEF2D is disrupted in a mouse model of Parkinson disease and in human patients. J. Clin. Investig. 2011, 121, 930–940. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.D.; Mount, M.P.; Shree, R.; Callaghan, S.; Slack, R.S.; Anisman, H.; Vincent, I.; Wang, X.; Mao, Z.; Park, D.S. Calpain-regulated p35/cdk5 plays a central role in dopaminergic neuron death through modulation of the transcription factor myocyte enhancer factor 2. J. Neurosci. 2006, 26, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.; Li, Z.; Yu, S.; Yuan, C.; Hong, W.; Wang, Z.; Cui, J.; Shi, T.; Fang, Y. Blood-Based Gene Expression Profiles Models for Classification of Subsyndromal Symptomatic Depression and Major Depressive Disorder. PLoS ONE 2012, 7, e31283. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Lin, W.; Li, J.; Pan, Y.; Wang, W. The depressive-like behaviors are correlated with decreased phosphorylation of mitogen-activated protein kinases in rat brain following chronic forced swim stress. Behav. Brain Res. 2006, 175, 233–240. [Google Scholar] [CrossRef]
- Vogt, A.; Tamewitz, A.; Skoko, J.; Sikorski, R.P.; Giuliano, K.A.; Lazo, J.S. The benzo[c]phenanthridine alkaloid, sanguinarine, is a selective, cell-active inhibitor of mitogen-activated protein kinase phosphatase-1. J. Biol. Chem. 2005, 280, 19078–19086. [Google Scholar] [CrossRef]
- Duric, V.; Banasr, M.; Licznerski, P.; Schmidt, H.D.; Stockmeier, C.A.; Simen, A.A.; Newton, S.S.; Duman, R.S. A negative regulator of MAP kinase causes depressive behavior. Nat. Med. 2010, 16, 1328–1332. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.R.; Wang, W.; Li, S.S.; Dong, H.Y.; Zhang, X.J. β-asarone from Acorus gramineus alleviates depression by modulating MKP-1. Genet. Mol. Res. 2015, 14, 4495–4504. [Google Scholar] [CrossRef] [PubMed]
- Chellian, R.; Pandy, V.; Mohamed, Z. Biphasic Effects of α-Asarone on Immobility in the Tail Suspension Test: Evidence for the Involvement of the Noradrenergic and Serotonergic Systems in Its Antidepressant-Like Activity. Front. Pharmacol. 2016, 7, 72. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Chen, S.W.; Xu, N.; Liu, X.H.; Zhang, H.; Wang, Y.Z.; Xu, X.D. Anxiolytic-like effect of α-asarone in mice. Phytother. Res. 2012, 26, 1476–1481. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Sur, B.; Yeom, M.; Shim, I.; Lee, H.; Hahm, D.H. Alpha-Asarone, a Major Component of Acorus gramineus, Attenuates Corticosterone-Induced Anxiety-Like Behaviours via Modulating TrkB Signaling Process. Korean J. Physiol. Pharmacol. 2014, 18, 191–200. [Google Scholar] [CrossRef]
- He, X.; Bai, Y.; Zeng, M.; Zhao, Z.; Zhang, Q.; Xu, N.; Qin, F.; Wei, X.; Zhao, M.; Wu, N.; et al. Anticonvulsant activities of α-asaronol ((E)-3′-hydroxyasarone), an active constituent derived from α-asarone. Pharmacol. Rep. 2018, 70, 69–74. [Google Scholar] [CrossRef]
- Ma, W.C.; Zhang, Q.; Li, H.; Chu, T.; Shi, H.Y.; Mao, S.J. Literature review and cause analysis of adverse reactions caused by asarone injection. Chin. J. Pharmacovigil. 2010, 7, 243–246. [Google Scholar]
- Yang, H.Y.; Deng, Y.P. Literature analysis of 122 adverse drug reactions induced by asarone injection. Chin. J. Ethnomed. Ethnopharm. 2012, 6, 36–39. [Google Scholar]
- Guan, Y.M.; Liu, J.; Zhang, J.L.; Chen, L.H.; Zhu, W.F.; Zang, Z.Z.; Jin, C.; Wu, L. Preparation and evaluation of four kinds of mixed essential oil liposomes in Jieyu Anshen Formula. China J. Chin. Mater. Med. 2019, 44, 1363–1370. (In Chinese) [Google Scholar]
- Lungare, S.; Hallam, K.; Badhan, R.K.S. Phytochemical-loaded mesoporous silica nanoparticles for nose-to-brain olfactory drug delivery. Int. J. Pharm. 2016, 513, 280–293. [Google Scholar] [CrossRef]
- FAO. Joint FAO/WHO Expert Committee on Food Additives (JECFA); FAO: Rome, Italy, 1987. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balakrishnan, R.; Cho, D.-Y.; Kim, I.-S.; Seol, S.-H.; Choi, D.-K. Molecular Mechanisms and Therapeutic Potential of α- and β-Asarone in the Treatment of Neurological Disorders. Antioxidants 2022, 11, 281. https://doi.org/10.3390/antiox11020281
Balakrishnan R, Cho D-Y, Kim I-S, Seol S-H, Choi D-K. Molecular Mechanisms and Therapeutic Potential of α- and β-Asarone in the Treatment of Neurological Disorders. Antioxidants. 2022; 11(2):281. https://doi.org/10.3390/antiox11020281
Chicago/Turabian StyleBalakrishnan, Rengasamy, Duk-Yeon Cho, In-Su Kim, Sang-Ho Seol, and Dong-Kug Choi. 2022. "Molecular Mechanisms and Therapeutic Potential of α- and β-Asarone in the Treatment of Neurological Disorders" Antioxidants 11, no. 2: 281. https://doi.org/10.3390/antiox11020281
APA StyleBalakrishnan, R., Cho, D.-Y., Kim, I.-S., Seol, S.-H., & Choi, D.-K. (2022). Molecular Mechanisms and Therapeutic Potential of α- and β-Asarone in the Treatment of Neurological Disorders. Antioxidants, 11(2), 281. https://doi.org/10.3390/antiox11020281








