Highlights
What are the main findings?
- Objective physical activity predicted faster reaction times in inhibitory control and fewer lapses in sustained attention, whereas sleep duration showed no significant effects.
- In the low-activity subgroup, higher daily steps were unexpectedly associated with slower inhibitory control, possibly reflecting confounding factors or differential physiological adaptation.
What are the implications of the main findings?
- Habitual physical activity should be prioritised in educational settings to enhance adolescent cognitive efficiency.
- Future research must assess circadian timing and sleep variability, rather than relying solely on total sleep duration.
Abstract
Background/Objectives: Adolescence is a critical period for executive function (EF) maturation. While sleep and physical activity (PA) are key lifestyle factors, their longitudinal impact on EF in ecologically valid settings is insufficiently characterised. This study examined the associations between objectively measured sleep duration, daily steps, and EF performance across one academic year (~9 months). Methods: A longitudinal study was conducted with 168 Spanish adolescents (13–16 years). Sleep duration and daily steps were monitored using Fitbit Charge 6 wearables for 7-day periods at baseline (M1; September 2024) and follow-up (M2; June 2025). EFs were assessed using three validated tasks: Stroop (inhibitory control), Psychomotor Vigilance Task-Brief (PVT-B; sustained attention), and Paced Auditory Serial Addition Test (PASAT; working memory). Linear Mixed Models (LMM) were employed to analyse the effects of the fixed factors (i.e., Group and Time), and their interactions. Results: PA, but not sleep duration, significantly predicted executive performance. The High_PA group demonstrated faster reaction times in inhibitory control (p = 0.007) and significantly fewer attentional lapses in sustained attention (p = 0.014). In contrast, sleep duration showed no significant main effects on EF domains (p > 0.05). Regression analyses confirmed that higher daily steps predicted faster reaction times in inhibitory control in the total sample (r = −0.173, p = 0.002), although an unexpected positive association was observed in the Low_PA group for inhibitory control, warranting cautious interpretation. Conclusions: These findings suggest that habitual PA is associated with better EF performance in adolescents, whereas sleep duration alone (without considering timing or variability) showed no significant associations with cognitive outcomes. Sensitivity analyses using clinically informed thresholds and continuous standardised predictors confirmed the robustness of these findings.
1. Introduction
Adolescence is a critical period for executive function (EF) maturation, during which the prefrontal cortex undergoes substantial refinement through synaptic pruning and myelination [1]. Sleep and physical activity have emerged as modifiable lifestyle factors that may shape neurocognitive trajectories, yet their longitudinal associations with EF remain insufficiently characterised in ecologically valid settings [2,3].
1.1. Sleep and Cognitive Function
Sleep is a fundamental pillar of cognitive homeostasis, supporting memory consolidation, synaptic plasticity, and metabolic restoration [4]. However, adolescents experience a biologically driven phase delay in circadian rhythms, resulting in a natural preference for later sleep and wake times [5,6]. Internationally, early school start times (typically 08:00–08:30) exacerbate this mismatch, with over 60% of adolescents failing to meet the recommended 8–10 h of sleep [7,8,9], potentially compromising cognitive readiness during morning hours.
Evidence suggests domain-specific vulnerability in sleep–cognition relationships. Meta-analytic findings indicate that sleep deficits significantly impair cognitive flexibility, while effects on inhibitory control and working memory remain inconsistent [10]. Acute sleep restriction primarily affects vigilance-related processing speed rather than selective attention [11].
A critical gap persists in the reliance on self-reported sleep measures, which show poor concordance with objective metrics [12] and in adolescent populations too [13]. Although consumer-grade wearables now enable continuous, objective monitoring in free-living conditions [14,15], few longitudinal studies have leveraged this technology to examine sleep–cognition associations across a full academic year.
1.2. Physical Activity and Executive Function
Beyond sleep, physical activity (PA) has emerged as another modifiable lifestyle factor consistently linked to enhanced cognitive performance in youth. International guidelines recommend that children and adolescents engage in an average of 60 min of moderate-to-vigorous physical activity daily [16], yet adherence rates remain suboptimal. This is concerning given that mental fatigue accumulation during sedentary school hours impairs both physical and cognitive performance [17,18].
Chronic PA—characterised by regular engagement over weeks or months (typically ≥4–6 weeks in intervention studies)—is hypothesised to confer stable neurobiological adaptations, including improved white matter integrity [19], increased cerebral blood flow, and upregulation of neurotrophic factors such as brain-derived neurotrophic factor (BDNF), a process driven by hemodynamic shear stress and metabolic demands during exertion [20,21]. These structural and functional changes are thought to enhance processing speed and executive control, particularly in tasks requiring sustained attention and inhibitory processes [22,23].
1.3. Current Study
Translation to real-world school settings remains challenging, as factors such as variability in engagement and competing curricular demands may attenuate laboratory-observed effects [24]. Moreover, PA and sleep are not independent: emerging evidence suggests that PA may function as a non-photic zeitgeber, strengthening circadian amplitude and improving sleep consolidation [25], thereby indirectly supporting cognitive performance through circadian entrainment. This underscores the need for longitudinal research in ecologically valid environments employing objective behavioural monitoring.
The present study therefore aimed to examine the longitudinal associations between objectively measured sleep duration, physical activity, and executive function performance in Spanish adolescents across one academic year under ecologically valid conditions. By doing so, this study sought to provide actionable evidence for educators and policymakers seeking to optimise adolescent learning environments. Crucially, we employed wearable technology (Fitbit Charge 6) to capture continuous behavioural data in free-living conditions, reflecting adolescents’ natural daily routines rather than controlled laboratory settings. We assessed three core executive function domains [26]: inhibitory control, sustained attention, and working memory. Given the domain-specific effects reported in prior meta-analyses [10] and the mechanistic plausibility of PA as a circadian modulator [25], we hypothesised that:
- Higher sleep duration would predict better performance on tasks requiring cognitive flexibility and vigilance, with weaker or null associations for inhibitory control.
- The sleep–EF relationship would be domain-specific, with stronger associations for vigilance-related outcomes than for inhibitory control or working memory.
- Higher physical activity levels would predict faster reaction times, greater accuracy and fewer attentional lapses across all executive function domains.
- The PA-EF relationship would be particularly pronounced in the high-activity subgroup, reflecting threshold effects consistent with neurobiological adaptation models.
2. Materials and Methods
2.1. Participants
A total of 168 Spanish adolescents (85 boys [weight: 65.84 ± 19.27 kg; height: 172.56 ± 8.24 cm] and 83 girls [weight: 57.16 ± 9.90 kg; height: 162.29 ± 6.60 cm]; Mage = 14.54 ± 0.73 years) from seven secondary schools in the region of Extremadura voluntarily participated in this longitudinal study. A priori power analysis (G*Power 3.1; [27]) indicated a minimum of 92 participants (f2 = 0.15, α = 0.05, power = 0.80); our sample (118–160 valid cases) exceeded this threshold. The inclusion criteria were: (a) enrolment in compulsory secondary education (ESO), (b) age between 13 and 16 years, and (c) willingness to wear a Fitbit device for 7-day monitoring periods at each assessment point (M1 and M2). Exclusion criteria included: (a) diagnosed sleep disorders, (b) neurological or psychiatric conditions affecting cognitive function, (c) use of medications known to affect sleep or cognition, and (d) inability to complete the cognitive assessment battery.
Complete cognitive data were available for 152–160 participants at baseline (M1) and 141–152 at follow-up (M2), depending on the measure. Fitbit-derived sleep data were available for 155 participants at M1 and 118 at M2, while physical activity data (daily steps) were available for 160 participants at M1 and 141 at M2.
The participants were informed about the study procedures, and written informed consent was obtained from parents or legal guardians, with verbal assent obtained from all participants. The study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki and was approved by the Bioethics Committee of the University of Extremadura (approval protocol number: 94//2024).
2.2. Outcomes and Instruments
2.2.1. Sleep and Physical Activity Monitoring
Sleep duration was objectively assessed using Fitbit Charge 6 devices (Fitbit Inc., San Francisco, CA, USA) worn on the non-dominant wrist for 7 consecutive days at each measurement point. Sleep duration (hours per night) was automatically detected by the device’s multi-sensor algorithm combining photoplethysmography (PPG) and accelerometery. Although direct validation for the specific Charge 6 model is limited, validation studies of previous generations of this device series have demonstrated sensitivity exceeding 95% for detecting sleep periods in adolescent populations [14,28].
For analytical purposes, participants were classified into sleep groups based on the median split of average sleep duration: High Sleep (≥7.26 h) and Low Sleep (<7.26 h).
PA was objectively assessed using the same Fitbit Charge 6 devices worn for 7 consecutive days at each measurement point. Daily step count was used as the primary measure of physical activity. For analysis purposes, participants were classified into physical activity groups based on the median split of average daily steps: High Physical Activity (High_PA; ≥9650 steps/day) and Low Physical Activity (Low_PA; <9650 steps/day). The median split was chosen in the absence of validated population-specific thresholds, ensuring balanced group sizes for subsequent mixed model analyses. Importantly, group assignment was based on the overall median across both time points, ensuring that each participant remained in the same group at M1 and M2, thereby avoiding the classification instability associated with time-point-specific median splits.
2.2.2. Executive Function Assessment
The Stroop Colour-Word Task was used to assess inhibitory control [29,30]. Participants were presented with colour words (e.g., “RED”) displayed in incongruent ink colours (e.g., blue) and were required to name the ink colour while ignoring the word meaning. This task requires the inhibition of the automatic response to read the word itself, thereby taxing executive control processes. Reaction time (ms) and accuracy (%) were recorded as outcome measures over a 1 min administration.
The Brief Psychomotor Vigilance Task (PVT-B) was employed to assess sustained attention and vigilance [31]. This 3 min task required participants to respond as quickly as possible when a visual stimulus appeared at random intervals (1–4 s). Mean reaction time (ms) and number of lapses (responses >500 ms) were recorded as outcome measures.
The Paced Auditory Serial Addition Test (PASAT) was used to assess working memory and information processing speed [32]. Participants heard single-digit numbers presented every 3 s and were required to add each number to the one immediately preceding it. This 3 min task places demands on working memory, attention, and arithmetic processing. Reaction time (ms) and accuracy (%) were recorded as outcome measures.
2.3. Procedures
A longitudinal observational design was carried out over a complete academic year (September 2024 to June 2025). Data were collected at two time points: baseline (M1, September 2024) and follow-up (M2, June 2025). At each time point, participants wore Fitbit Charge 6 devices for 7 consecutive days [33,34] and completed the cognitive assessment battery in a quiet classroom setting. Cognitive tests were administered using the SOMA NPT application (version 13.0; SOMA Analytics, Lucerne, Switzerland) on iPad devices in the same order across all participants and time points. For the duration of the study, participants were encouraged to maintain their usual daily habits.
2.4. Statistical Analysis
Data preprocessing and statistical analyses were performed using a modern data science framework in Python (version 3.12). The analytical pipeline utilised pandas library for data manipulation and the statsmodels and scipy packages for advanced longitudinal modelling, ensuring reproducibility and robustness in the handling of large dataset structures. Linear Mixed Models (LMM) were employed as the primary analytical procedure. LMMs are particularly robust for handling unbalanced longitudinal data and missing observations, providing a more flexible alternative to traditional repeated measures ANOVA [35].
The modelling strategy included both fixed and random effects. Specifically, Group (High vs. Low Physical Activity; or High vs. Low Sleep Duration) and Time (M1 vs. M2) were included as fixed factors, along with their interaction term (Group × Time). To account for the non-independence of repeated measures and the inherent inter-individual variability, Participant ID was included as a random intercept in all models. While lifestyle factors were also analysed as continuous predictors in supplementary regressions, the median-split grouping was maintained in the primary LMM to examine specific threshold effects and differential developmental trajectories between high- and low-adherence subgroups [36]. The median-split approach was retained to facilitate interpretability and to explore potential non-linear or threshold-like patterns in real-world behavioural adherence, while complementary continuous analyses were conducted to mitigate information loss. Prior to the main analysis, a variance component analysis confirmed that the Intraclass Correlation Coefficient (ICC) exceeded 10% for all dependent variables, corroborating the necessity of treating participants as a random effect [36]. Model comparison using AIC confirmed that models with fixed factors (Group, Time) and interactions provided optimal fit for most executive function outcomes.
Post hoc comparisons were conducted to obtain estimated marginal means (EMMeans) and to identify specific differences between groups at each time point. Results from LMM are presented as coefficients and standard error (Coef ± SE). Additionally, to further explore the continuous relationship between lifestyle habits and cognitive outcomes, linear regression analyses were performed. These results are reported as Pearson’s correlation coefficients (r) and unstandardised regression coefficients, where β0 represents the intercept and β1 indicates the slope of the regression line.
Statistical significance was set at p < 0.05. Finally, the magnitude of the observed differences was quantified using Cohen’s d effect sizes [37]. Following conventional criteria, effect sizes were interpreted as small (0.20–0.49), medium (0.50–0.79), and large (≥0.80).
To assess the robustness of findings to the choice of grouping criterion, supplementary sensitivity analyses were conducted using (a) clinically informed thresholds (≥10,000 steps/day for physical activity [38,39]; ≥8 h/night for sleep duration [9]) and (b) continuous standardised predictors (z-scores) entered directly into LMMs. Model comparison using AIC confirmed that models with categorical fixed factors provided optimal fit for most outcomes, supporting the median-split approach as the primary analysis.
3. Results
Table 1 shows the descriptive statistics and main effects analyses of the LMM for executive function outcomes by sleep duration group. Regarding lifestyle variables across the sample, sleep duration decreased from M1 (7.47 ± 0.83 h) to M2 (7.06 ± 0.82 h). The median split for sleep duration was 7.26 h/night.

Table 1.
LMM descriptive statistics and main effects analyses for executive function outcomes by sleep duration group.
Sleep duration groups did not show significant main effects on any executive function measure. The main effect of Group was non-significant for Stroop RT (F(1, 307) = 0.81, p = 0.369; R2m = 0.241, R2c = 0.589), PVT-B RT (F(1, 303) = 0.01, p = 0.916; R2m = 0.286, R2c = 0.689), PVT-B lapses (F(1, 300) = 1.13, p = 0.288; R2m = 0.243, R2c = 0.618), PASAT RT (F(1, 306) = 0.93, p = 0.336; R2m = 0.431, R2c = 0.804), and PASAT accuracy (F(1, 306) = 0.89, p = 0.344; R2m = 0.362, R2c = 0.743).
Significant main effects of Time were observed for Stroop RT (F(1, 307) = 10.66, p = 0.001, R2m = 0.240, R2c = 0.585), PASAT RT (F(1, 306) = 117.43, p < 0.001, R2m = 0.433, R2c = 0.807), and PASAT accuracy (F(1, 306) = 57.10, p < 0.001, R2m = 0.358, R2c = 0.745), primarily reflecting practice effects. No significant Group × Time interactions were found. Post-hoc comparisons for sleep duration groups are presented in Table 2.

Table 2.
Multiple comparisons (intra- and inter-group) for executive function outcomes by sleep duration group.
Table 2.
Multiple comparisons (intra- and inter-group) for executive function outcomes by sleep duration group.
| Variable | Type | Comparison | p | d | CI (95%) |
|---|---|---|---|---|---|
| Stroop RT (ms) | Inter-group (M1) | High_Sleep vs. Low_Sleep | 0.433 | 0.12 | [−0.19, 0.43] |
| Inter-group (M2) | High_Sleep vs. Low_Sleep | 0.701 | 0.06 | [−0.26, 0.38] | |
| Intra-group | M1 vs. M2 (High_Sleep) | <0.001 *** | −0.42 | [−0.75, −0.09] | |
| Intra-group | M1 vs. M2 (Low_Sleep) | 0.038 * | −0.28 | [−0.60, 0.05] | |
| Stroop Acc (%) | Inter-group (M1) | High_Sleep vs. Low_Sleep | 0.325 | −0.16 | [−0.47, 0.15] |
| Inter-group (M2) | High_Sleep vs. Low_Sleep | 0.709 | 0.06 | [−0.26, 0.38] | |
| Intra-group | M1 vs. M2 (High_Sleep) | 0.868 | −0.02 | [−0.34, 0.30] | |
| Intra-group | M1 vs. M2 (Low_Sleep) | 0.052 | −0.18 | [−0.51, 0.14] | |
| PVT-B RT (ms) | Inter-group (M1) | High_Sleep vs. Low_Sleep | 0.824 | −0.04 | [−0.35, 0.28] |
| Inter-group (M2) | High_Sleep vs. Low_Sleep | 0.938 | 0.01 | [−0.31, 0.33] | |
| Intra-group | M1 vs. M2 (High_Sleep) | 0.185 | −0.14 | [−0.47, 0.18] | |
| Intra-group | M1 vs. M2 (Low_Sleep) | 0.401 | −0.10 | [−0.43, 0.23] | |
| PVT-B Lapses (n) | Inter-group (M1) | High_Sleep vs. Low_Sleep | 0.311 | −0.16 | [−0.48, 0.15] |
| Inter-group (M2) | High_Sleep vs. Low_Sleep | 0.818 | 0.04 | [−0.28, 0.36] | |
| Intra-group | M1 vs. M2 (High_Sleep) | 0.144 | 0.16 | [−0.17, 0.49] | |
| Intra-group | M1 vs. M2 (Low_Sleep) | 0.748 | −0.04 | [−0.38, 0.29] | |
| PASAT RT (ms) | Inter-group (M1) | High_Sleep vs. Low_Sleep | 0.464 | −0.12 | [−0.43, 0.19] |
| Inter-group (M2) | High_Sleep vs. Low_Sleep | 0.109 | −0.26 | [−0.58, 0.06] | |
| Intra-group | M1 vs. M2 (High_Sleep) | <0.001 *** | −1.05 | [−1.42, −0.69] | |
| Intra-group | M1 vs. M2 (Low_Sleep) | <0.001 *** | −0.95 | [−1.31, −0.59] | |
| PASAT Acc (%) | Inter-group (M1) | High_Sleep vs. Low_Sleep | 0.476 | 0.11 | [−0.20, 0.42] |
| Inter-group (M2) | High_Sleep vs. Low_Sleep | 0.020 * | 0.38 | [0.06, 0.71] | |
| Intra-group | M1 vs. M2 (High_Sleep) | <0.001 *** | 0.96 | [0.60, 1.32] | |
| Intra-group | M1 vs. M2 (Low_Sleep) | <0.001 *** | 0.59 | [0.25, 0.93] |
Note. d = Cohen’s d; CI = confidence interval. Bold values indicate statistically significant effects (p < 0.05). * p < 0.05, *** p < 0.001.
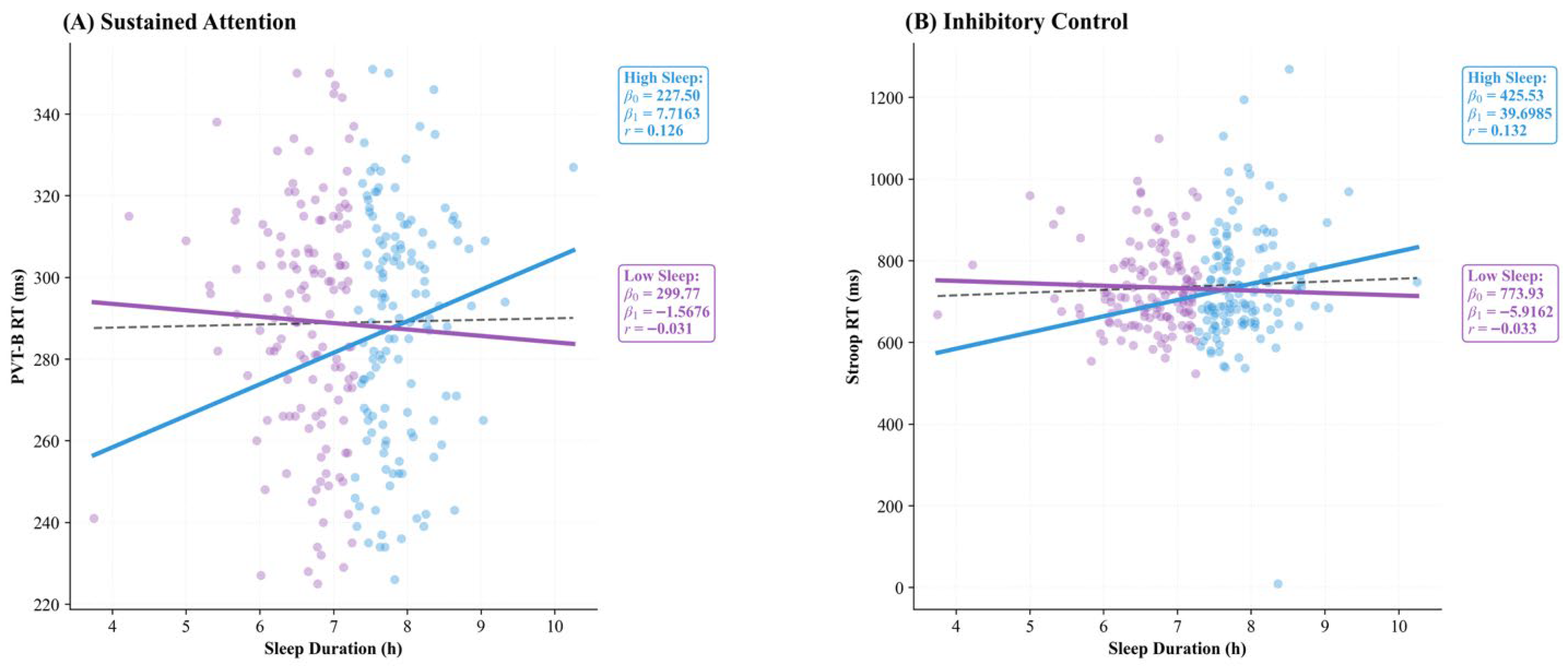
Linear regression analyses further corroborated the findings from the LMM, demonstrating an absence of a significant predictive relationship between sleep duration and cognitive performance (p > 0.05). As illustrated in Figure 1, the regression lines for both the total sample and the stratified groups (High_Sleep vs. Low_Sleep) remained remarkably flat for both sustained attention (Figure 1A; Total r = 0.011) and inhibitory control (Figure 1B; Total r = 0.046), indicating that sleep duration was not a primary driver of executive performance in this sample.
Figure 1.
Linear regression analysis between sleep duration and executive function. (A) Sustained attention (PVT-B RT) and (B) inhibitory control (Stroop RT) showed no significant associations with sleep duration in the total sample or subgroups (all p > 0.05). Dashed line = total sample; solid lines = group-specific regressions. High_Sleep ≥ 7.26 h/night; Low_Sleep < 7.26 h/night. Regression statistics (β0, β1, r) are displayed within each panel. Note: β0 = intercept; β1 = slope.
Given the absence of significant sleep duration effects on executive function performance, we next examined whether physical activity showed differential associations with EF outcomes. Table 3 shows the descriptive statistics and main effects analyses of the LMM for executive function outcomes by physical activity group. Regarding lifestyle variables across the sample daily steps decreased (from 10,927 ± 3450 to 8880 ± 3863 steps/day). The median split for physical activity groups was 9650 steps/day.

Table 3.
LMM descriptive statistics and main effects analyses for executive function outcomes by physical activity group.
A significant main effect of Group was found for Stroop RT (F(1, 307) = 7.40, p = 0.007), with the Low_PA group showing longer reaction times than the High_PA group. However, post hoc comparisons (Table 4) showed this effect was significant at M1 (p = 0.024) but not at M2 (p = 0.138), indicating the group difference was stronger at baseline. The model explained 25.3% of the variance through fixed effects (R2m = 0.253) and 59.5% when including random effects (R2c = 0.595). A significant main effect of Group was also found for PVT-B lapses (F(1, 300) = 6.05, p = 0.014), indicating that less active adolescents committed more attentional lapses. The model explained 24.7% of the variance through fixed effects (R2m = 0.247) and 60.8% when including random effects (R2c = 0.608).

Table 4.
Multiple comparisons (intra- and inter-group) for executive function outcomes by physical activity group.
For PASAT accuracy, a significant Group effect emerged (F(1, 306) = 4.08, p = 0.043), with the High_PA group showing higher accuracy. The model explained 36.4% of the variance through fixed effects (R2m = 0.364) and 74.4% when including random effects (R2c = 0.744). Significant main effects of Time were observed for PASAT RT (F(1, 306) = 114.41, p < 0.001; R2m = 0.432, R2c = 0.802) and PASAT accuracy (F(1, 306) = 42.38, p < 0.001), reflecting substantial practice effects. No significant Group × Time interactions were found, although substantial practice effects were observed for PASAT measures (p < 0.001).
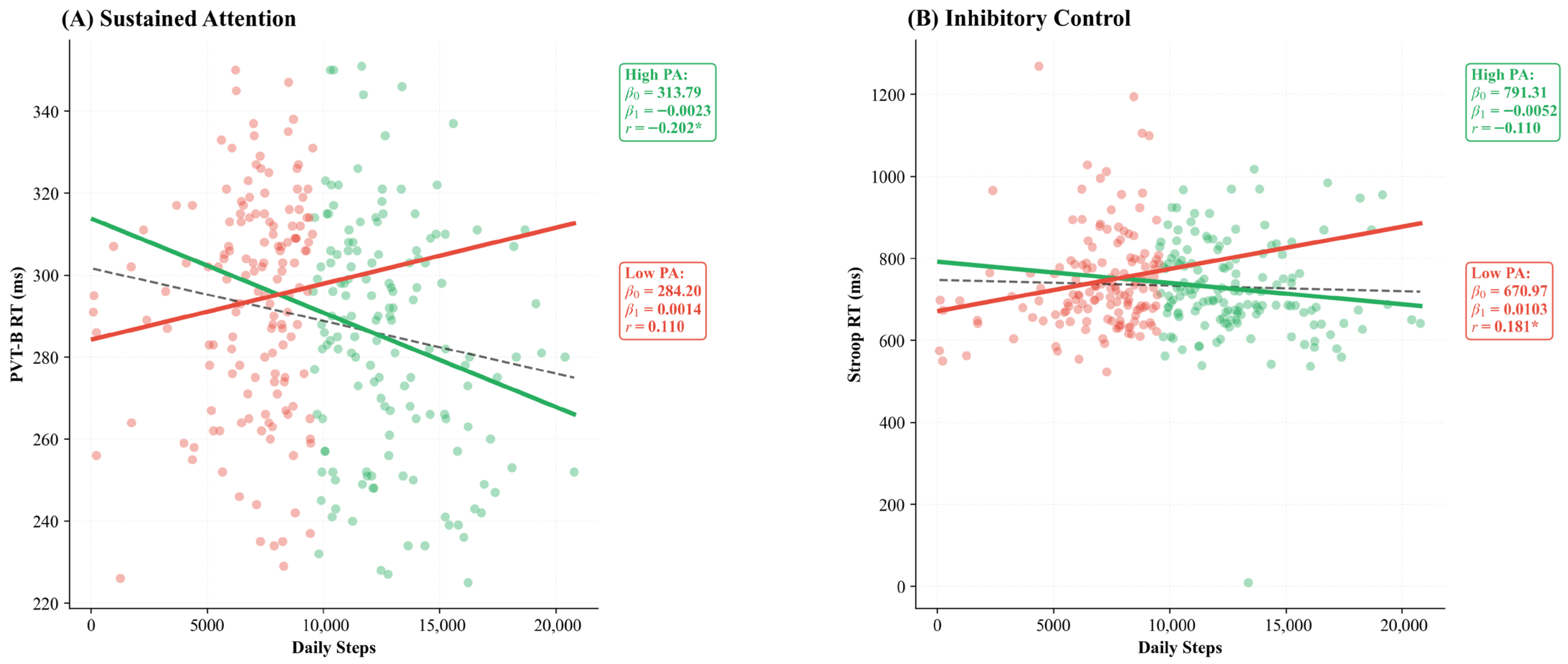
Beyond these longitudinal trends, linear regression analyses revealed that higher daily steps significantly predicted faster reaction times in the total sample (r = −0.173 **, p = 0.002, β1 = −0.0016). However, this benefit was specifically driven by the High_PA group (β1= −0.0023, p = 0.014), as shown in Figure 2A. Conversely, for inhibitory control (Stroop RT), a significant positive correlation emerged only within the Low_PA group (r = 0.181, p = 0.027, β1 = 0.0103), suggesting that adolescents with lower activity levels exhibit an unexpected increase in reaction time with higher daily steps (Figure 2B).
Figure 2.
Linear regression analysis between daily steps and executive function. (A) Higher daily steps predicted faster sustained attention (PVT-B RT) in the total sample (r = −0.173, p = 0.002), driven by the High_PA group (p = 0.014). (B) For inhibitory control (Stroop RT), a significant positive association emerged only in the Low_PA group (r = 0.181, p = 0.027). Dashed line = total sample; solid lines = group-specific regressions. High_PA ≥ 9650 steps/day; Low_PA < 9650 steps/day. Regression statistics (β0, β1, r) are displayed within each panel. Note: β0 = intercept; β1 = slope; * p < 0.05.
Sensitivity analyses confirmed the robustness of the primary findings. With clinically informed thresholds (≥10,000 steps/day; ≥8 h/night), the PA group effect on Stroop RT remained significant (p = 0.042). With continuous standardised predictors (z-scores), daily steps significantly predicted Stroop RT (β = −19.41, p = 0.037), PVT-B RT (β = −6.11, p = 0.007), and PVT-B Lapses (β = −3.92, p < 0.001), while sleep duration showed no significant associations (all p > 0.68). These converging results confirm that the PA–EF associations are not attributable to the choice of grouping criterion.
4. Discussion
The present longitudinal study examined the associations between objectively measured sleep duration, PA and EF performance in Spanish adolescents across one academic year. Our findings revealed a clear divergence: physical activity, but not sleep duration, significantly predicted executive performance, particularly in domains of inhibitory control and sustained attention. These results provide nuanced insights into the modifiable lifestyle factors that may support cognitive development during the critical window of adolescence. Importantly, these findings were observed under ecologically valid school conditions, where behavioural variability, competing academic demands, and imperfect adherence are the norm rather than the exception.
4.1. Sleep Duration and Executive Function: Interpreting Null Findings
Sleep duration, averaged across 7-day periods, did not predict executive function performance. Taken together, these findings suggest that total sleep duration, when averaged across several nights, may represent an insufficient proxy to capture the sleep-related processes most relevant to executive functioning during adolescence. This null finding aligns with the domain-specific vulnerability framework [10] which indicates that cognitive shifting is more sensitive to sleep loss than inhibitory control or working memory. Our null effects for Stroop and PASAT are consistent with this framework. Critically, these findings should not be interpreted as evidence that sleep is unimportant for cognition, but rather that duration alone may be an insufficient metric.
Several methodological factors may explain these null findings regarding sleep duration. First, our focus on mean sleep duration may have obscured the effects of night-to-night variability and circadian misalignment (e.g., social jetlag). Research indicates that high variability in sleep patterns and misalignment between biological and social rhythms impair executive function and processing speed independently of total sleep time [40,41,42]. Furthermore, experimental evidence suggests that circadian misalignment specifically degrades sustained attention and cognitive throughput, effects that are not captured by aggregate duration metrics [43]. Second, the sample’s restricted range (M1: 7.47 ± 0.83 h) represents mild insufficiency; populations with severe restriction (<6 h) show stronger cognitive deficits [44]. Third, ceiling effects in accuracy measures (>98% for Stroop) may have masked subtle impairments. Finally, unmeasured factors (time-since-awakening, caffeine use, screen time) may have modulated sleep–cognition relationships beyond duration alone [45,46].
4.2. Physical Activity as a Robust Predictor of Executive Performance
In stark contrast to sleep, physical activity emerged as a significant predictor of multiple executive function outcomes. The High_PA group demonstrated faster reaction times in inhibitory control (Stroop RT: p = 0.007) and fewer attentional lapses (PVT-B: p = 0.014), alongside higher working memory accuracy (PASAT: p = 0.043). These findings align with theoretical frameworks, such as the exercise-induced neuroplasticity model [47], proposing that chronic PA facilitates neurobiological adaptations and enhances neural efficiency [48]. Recent evidence published in this journal further corroborates that physical activity induces structural brain adaptations essential for executive development [49].
A central finding was the association between daily steps and reaction time, which showed a linear pattern in the total sample. In the total sample, higher daily activity significantly predicted faster reaction times in sustained attention tasks (r = −0.173, p = 0.002), a relationship primarily driven by the High_PA group (β1 = −0.0023, p = 0.014; Figure 2A). This pattern suggests that adolescents who habitually engage in higher levels of physical activity develop more efficient neural processing [50,51]. Furthermore, regular engagement in physical activity improves pacing strategies and effort regulation in youth, specifically through the maturation of metacognitive monitoring and planning [52]. Reaction time is a sensitive marker of neurodevelopmental integrity, indexing the efficiency of white matter maturation and myelination during the adolescent period [53,54]. The observed association corroborates meta-analytic evidence that PA interventions preferentially enhance processing speed and executive control in youth [55].
It is important to contextualise the practical significance of these associations. The correlation between daily steps and PVT-B reaction time (r = −0.173) corresponds to approximately 3% of shared variance, and Cohen’s d values for PA group comparisons ranged from small to moderate (d = 0.24–0.37). These modest effect sizes are consistent with the broader literature on lifestyle–cognition associations in youth, where small but consistent effects carry cumulative public health significance [23].
An important consideration is the potential for reverse causation: adolescents with better executive function may be more successful in maintaining regular physical activity through superior self-regulation and planning. This bidirectional relationship has been documented in adults [56] and is increasingly recognized in youth, where executive control facilitates the adherence to complex activity behaviours [57]. Our observational design cannot disentangle whether PA improves EF, EF facilitates PA adherence, or both processes operate reciprocally. Additionally, third-variable confounding (e.g., family support, socioeconomic resources) may explain observed associations independent of any causal pathway [58,59].
An unexpected finding was a positive correlation between daily steps and Stroop RT in the Low_PA group (r = 0.181, p = 0.027; Figure 2B), indicating that within the less active subgroup, higher step counts were associated with slower inhibitory control. This small effect should be interpreted with considerable caution, as the Low_PA group encompasses substantial heterogeneity (ranging from ~4000 to ~9650 steps/day), and confounding variables not included in the model, such as body mass index, screen time, or socioeconomic status, may account for this pattern. Several non-exclusive explanations remain plausible: (1) unaccustomed exertion in poorly conditioned individuals may temporarily compromise cognitive resources [60], (2) insufficient neurobiological scaffolding to benefit from irregular activity bursts [24], or (3) competing demands when combining increased activity with insufficient recovery [61], or (4) unmeasured third variables that covary with both moderate step counts and slower reaction times within this subgroup. Notably, the sensitivity analysis using the 10,000-step clinical threshold yielded a comparable pattern (r = 0.155, p = 0.048), suggesting that this association is not an artefact of the specific median-split criterion. Nonetheless, given the modest effect size (r2 ≈ 3%) and observational design, we refrain from attributing this pattern to any single causal mechanism. This pattern aligns with U-shaped models where moderate, habitual PA confers benefits, while both inactivity and erratic exertion may be suboptimal [62].
The absence of significant Group × Time interactions suggests that the PA-EF relationship remained stable across the academic year, despite the observed decline in average daily steps from M1 to M2 (10,927 to 8880 steps/day). This secular trend likely reflects increased academic demands [63] and reduced outdoor activity during winter months [64]. Notably, even within this context of declining activity, between-group differences persisted, underscoring the resilience of habitual PA effects on cognitive performance. It should be noted that daily step counts primarily reflect behavioural adherence to physical activity recommendations and may not fully capture qualitative aspects such as intensity distribution or contextual characteristics of movement. Importantly, this pattern should not be interpreted as a detrimental effect of physical activity per se, but rather as a potential mismatch between acute cognitive demands and the physiological adaptation levels of habitually low-active adolescents.
4.3. Strengths, Limitations and Future Directions
This study advances the field by integrating consumer-grade wearable technology with longitudinal cognitive assessment in an ecologically valid school setting. The Fitbit Charge 6 provided objective monitoring of sleep and physical activity at two assessment points across one academic year, circumventing the recall bias inherent in self-report measures. Recent large-scale analyses confirm significant discrepancies between youth-reported and Fitbit-derived sleep data, underscoring the necessity of objective monitoring for accurate cognitive profiling [13]. Validation studies of earlier Fitbit models (Charge 2, Charge 4) have demonstrated sensitivity exceeding 95% for sleep detection and strong concordance with research-grade actigraphy for step counting [14,15]. While the Charge 6 employs refined photoplethysmography (PPG) and accelerometery algorithms, direct validation against polysomnography remains limited. Nevertheless, for population-level research prioritising ecological validity over laboratory precision, these devices represent a viable translational tool [28].
Our use of Linear Mixed Models (LMM) to handle longitudinal data with missing observations represents a methodological strength. LMMs account for the non-independence of repeated measures and inter-individual variability, providing more robust estimates than traditional repeated-measures ANOVA [35]. The high conditional R2 values (ranging from 0.589 to 0.804) indicate that participant-level random effects captured substantial variance, corroborating the necessity of this analytical approach. Future iterations could extend these models to include time-varying covariates (e.g., daily stress, academic load) and explore non-linear trajectories using growth curve modelling.
Despite these methodological strengths, several limitations warrant consideration. First, the median-split approach to creating activity and sleep groups, while common in exploratory research, imposes artificial thresholds that may not reflect true functional categories. However, three lines of evidence support the robustness of our findings: (a) model comparison using AIC confirmed that categorical grouping provided optimal fit for most outcomes, (b) sensitivity analyses with clinically informed thresholds (≥10,000 steps/day; ≥8 h/night) replicated the primary results, and (c) continuous standardised predictors (z-scores) confirmed significant PA–EF associations independently of any grouping criterion. Future studies should employ person-cantered methods (e.g., latent profile analysis) to identify naturally occurring subgroups based on multivariate behavioural patterns.
Second, our reliance on sleep duration as the sole sleep metric neglects critical dimensions such as sleep timing (bedtime/wake time consistency), sleep efficiency (percentage of time in bed asleep), and sleep architecture (REM/NREM proportions). Fitbit devices capture these data, and future analyses should leverage them to test domain-specific hypotheses (e.g., REM sleep predicting cognitive flexibility; deep sleep predicting working memory consolidation). Notably, the sensitivity analysis with the 8 h clinical threshold revealed a significant effect on PASAT RT (p = 0.003) that was absent with the median split, suggesting that clinically meaningful sleep categories may have greater predictive value than sample-derived thresholds for specific cognitive domains.
Third, the observational design precludes causal inference. While our longitudinal approach strengthens temporal precedence claims, experimental manipulation (e.g., randomised PA interventions) is necessary to establish causality. Future research should also examine whether PA-EF relationships differ fundamentally between habitually active and sedentary youth. Ongoing trials in Spanish schools, such as the MOVESCHOOL study [65], will complement our findings by testing intervention efficacy. Additionally, substantial practice effects, particularly for the PASAT (Cohen’s d up to 1.07), may have obscured subtle lifestyle-related plasticity. Although the 9-month interval is long, task familiarity likely contributed to performance gains; yet accuracy at M2 (86%) remained below ceiling, indicating learning had not plateaued. Future studies should incorporate familiarisation sessions to attenuate this effect.
Fourth, we did not control potential mediators (e.g., BDNF levels, cortisol reactivity, screen time) or moderators (e.g., pubertal status, socioeconomic status, body composition) that could refine our understanding of PA-EF mechanisms. Multi-modal designs integrating biomarkers, neuroimaging, and ecological momentary assessment would provide mechanistic insights beyond behavioural correlations. Furthermore, previous research indicates that emotional intelligence and academic burnout significantly mediate the relationship between school performance and well-being [66,67], variables that could interact with the lifestyle factors assessed here.
Finally, the generalisability of our findings may be limited to Spanish adolescents in regions with similar school schedules and cultural norms around sleep and PA. Cross-cultural replications are essential, particularly in contexts with delayed school start times or different PA infrastructure.
4.4. Practical Applications
The robust PA-EF associations documented here carry direct implications for secondary education policy in Spain and beyond. Given that Spanish adolescents spend up to 78% of their school day sedentary [65,68], integrating movement-based interventions into the academic schedule may yield cognitive dividends without sacrificing instructional time. Evidence-based strategies include:
- Active breaks: Implementation of brief (5–10 min) bouts of moderate-intensity PA between lessons. Mechanistically, these bouts enhance cerebral blood flow and neural efficiency, facilitating the restoration of attentional resources depleted by prolonged cognitive effort [69]. Empirical evidence confirms that such breaks significantly improve students’ “on-task” behaviour and academic engagement [70,71], serving as an effective countermeasure against the mental load accumulation typically observed in school settings [17].
- Active commuting: Promoting walking or cycling to school as a daily PA opportunity that also serves as a circadian zeitgeber [72].
- Curriculum-integrated movement: Incorporating kinaesthetic learning activities (e.g., movement-based mnemonics, standing desks) into lesson plans. Recent meta-analyses indicate that integrating physical activity directly with academic content yields greater academic and behavioural benefits than non-integrated movement, likely through embodied cognition mechanisms [71,73].
While our data do not permit direct comparison of different PA intensities or frequencies, the consistent association between habitual daily step volume and cognitive performance, confirmed across median-split, clinical-threshold, and continuous-predictor analyses, aligns with emerging evidence that total daily movement volume, including light-to-moderate activity [74], may be a meaningful predictor of neurocognitive outcomes in youth [38]. Intervention programmes should therefore consider promoting consistent daily activity alongside intensity-focused targets, focusing on sustainable behaviour change rather than short-term performance gains [75,76].
5. Conclusions
This longitudinal study demonstrates that habitual physical activity is associated with better executive function performance in Spanish adolescents, particularly in inhibitory control and sustained attention. In contrast, sleep duration averaged across 7-day periods showed no significant associations with EF outcomes. These null findings likely reflect the complexity of sleep–cognition relationships, which extend beyond total duration to encompass sleep timing, night-to-night variability, and circadian alignment. Future research should expand beyond sleep duration to examine circadian alignment and sleep architecture, and randomised controlled trials are needed to establish whether the observed PA-EF associations reflect causal relationships that can be leveraged to enhance adolescent cognitive development. Taken together, these results suggest that under real-life school conditions, habitual physical activity emerges as a more robust and actionable correlate of executive functioning than sleep duration alone.
Author Contributions
Conceptualization, R.A.-M. and T.G.-C.; Methodology, R.A.-M.; Formal Analysis, R.A.-M.; Investigation, R.A.-M., A.R.-M., I.G.-P. and R.L.-M.; Data Curation, R.A.-M. and R.L.-M.; Writing—Original Draft Preparation, R.A.-M.; Writing—Review and Editing, A.R.-M., I.G.-P. and T.G.-C.; Visualisation, R.A.-M.; Supervision, A.R.-M., I.G.-P. and T.G.-C.; Project Administration, R.A.-M.; Funding Acquisition, R.A.-M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Spanish Ministry of Science and Innovation under the Knowledge Generation Projects 2022, grant number PID2022-140903NB-I00. A predoctoral FPI fellowship was awarded to R.A.M. by the Government of Spain (Ministry of Science and Innovation), grant number FPI-UEx/2023/5. This research has been co-financed 85% by the European Union, European Regional Development Fund, and the Government of Extremadura (Managing Authority: Ministry of Finance, GR24082).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of the University of Extremadura (protocol code 94/2024 and date of approval: 8 May 2024).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy and ethical restrictions involving minors.
Acknowledgments
During the preparation of this manuscript, the authors used ChatGPT (OpenAI, version GPT-4) to improve the clarity and readability of selected sentences. The authors reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| EF | Executive Function |
| PA | Physical Activity |
| BDNF | Brain-Derived Neurotrophic Factor |
| LMM | Linear Mixed Models |
| PVT-B | Psychomotor Vigilance Task-Brief |
| PASAT | Paced Auditory Serial Addition Test |
| RT | Reaction Time |
| PPG | Photoplethysmography |
| ICC | Intraclass Correlation Coefficient |
| SE | Standard Error |
References
- Diamond, A. Executive Functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef]
- Belluardo, G.; Meneo, D.; Cerolini, S.; Baglioni, C.; De Bartolo, P. Sleep, Physical Activity, and Executive Functions in Students: A Narrative Review. Clocks Sleep 2025, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Sewell, K.R.; Erickson, K.I.; Rainey-Smith, S.R.; Peiffer, J.J.; Sohrabi, H.R.; Brown, B.M. Relationships between Physical Activity, Sleep and Cognitive Function: A Narrative Review. Neurosci. Biobehav. Rev. 2021, 130, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Dewald, J.F.; Meijer, A.M.; Oort, F.J.; Kerkhof, G.A.; Bögels, S.M. The Influence of Sleep Quality, Sleep Duration and Sleepiness on School Performance in Children and Adolescents: A Meta-Analytic Review. Sleep Med. Rev. 2010, 14, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Crowley, S.J.; Acebo, C.; Carskadon, M.A. Sleep, Circadian Rhythms, and Delayed Phase in Adolescence. Sleep Med. 2007, 8, 602–612. [Google Scholar] [CrossRef]
- Crowley, S.J.; Wolfson, A.R.; Tarokh, L.; Carskadon, M.A. An Update on Adolescent Sleep: New Evidence Informing the Perfect Storm Model. J. Adolesc. 2018, 67, 55–65. [Google Scholar] [CrossRef]
- Galan-Lopez, P.; Domínguez, R.; Gísladóttir, T.; Sánchez-Oliver, A.J.; Pihu, M.; Ries, F.; Klonizakis, M. Sleep Quality and Duration in European Adolescents (The Adoleshealth Study): A Cross-Sectional, Quantitative Study. Children 2021, 8, 188. [Google Scholar] [CrossRef]
- Gradisar, M.; Gardner, G.; Dohnt, H. Recent Worldwide Sleep Patterns and Problems during Adolescence: A Review and Meta-Analysis of Age, Region, and Sleep. Sleep Med. 2011, 12, 110–118. [Google Scholar] [CrossRef]
- Paruthi, S.; Brooks, L.J.; D’Ambrosio, C.; Hall, W.A.; Kotagal, S.; Lloyd, R.M.; Malow, B.A.; Maski, K.; Nichols, C.; Quan, S.F.; et al. Consensus Statement of the American Academy of Sleep Medicine on the Recommended Amount of Sleep for Healthy Children: Methodology and Discussion. J. Clin. Sleep Med. 2016, 12, 1549–1561. [Google Scholar] [CrossRef]
- Molińska, M.; Złotogórska, A. Sleep Deficits and Executive Functions at Different Developmental Stages—Meta-Analysis. Pol. J. Aviat. Med. Bioeng. Psychol. 2017, 22, 27–38. [Google Scholar] [CrossRef]
- Cunningham, J.E.A.; Jones, S.A.H.; Eskes, G.A.; Rusak, B. Acute Sleep Restriction Has Differential Effects on Components of Attention. Front. Psychiatry 2018, 9, 499. [Google Scholar] [CrossRef] [PubMed]
- Prince, S.A.; Adamo, K.B.; Hamel, M.E.; Hardt, J.; Connor Gorber, S.; Tremblay, M. A Comparison of Direct versus Self-Report Measures for Assessing Physical Activity in Adults: A Systematic Review. Int. J. Behav. Nutr. Phys. Act. 2008, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Kiss, O.; Shaska, A.; Müller-Oehring, E.M.; Hasler, B.P.; Franzen, P.L.; FitzGerald, D.H.; Clark, D.B.; Baker, F.C. Assessment of Sleep Measures and Their Agreement: Youth-Reported, Caregiver-Reported, and Fitbit-Derived Data in a Large Early Adolescent Cohort. Sleep 2025, 48, zsaf065. [Google Scholar] [CrossRef] [PubMed]
- de Zambotti, M.; Goldstone, A.; Claudatos, S.; Colrain, I.M.; Baker, F.C. A Validation Study of Fitbit Charge 2TM Compared with Polysomnography in Adults. Chronobiol. Int. 2018, 35, 465–476. [Google Scholar] [CrossRef]
- Doheny, E.P.; Renerts, K.; Braun, A.; Werth, E.; Baumann, C.; Baumgartner, P.; Morgan-Jones, P.; Busse, M.; Lowery, M.M.; Jung, H.H. Assessment of Fitbit Charge 4 for Sleep Stage and Heart Rate Monitoring against Polysomnography and during Home Monitoring in Huntington’s Disease. J. Clin. Sleep Med. 2024, 20, 1163–1171. [Google Scholar] [CrossRef]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 Guidelines on Physical Activity and Sedentary Behaviour. Br. J. Sports Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef]
- Díaz-García, J.; González-Ponce, I.; Ponce-Bordón, J.C.; López-Gajardo, M.Á.; Ramírez-Bravo, I.; Rubio-Morales, A.; García-Calvo, T. Mental Load and Fatigue Assessment Instruments: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 419. [Google Scholar] [CrossRef]
- Van Cutsem, J.; Marcora, S.; De Pauw, K.; Bailey, S.; Meeusen, R.; Roelands, B. The Effects of Mental Fatigue on Physical Performance: A Systematic Review. Sports Med. 2017, 47, 1569–1588. [Google Scholar] [CrossRef]
- Chaddock-Heyman, L.; Erickson, K.I.; Kienzler, C.; Drollette, E.S.; Raine, L.B.; Kao, S.C.; Bensken, J.; Weisshappel, R.; Castelli, D.M.; Hillman, C.H.; et al. Physical Activity Increases White Matter Microstructure in Children. Front. Neurosci. 2018, 12, 950. [Google Scholar] [CrossRef]
- Cefis, M.; Chaney, R.; Wirtz, J.; Méloux, A.; Quirié, A.; Leger, C.; Prigent-Tessier, A.; Garnier, P. Molecular Mechanisms Underlying Physical Exercise-Induced Brain BDNF Overproduction. Front. Mol. Neurosci. 2023, 16, 1275924. [Google Scholar] [CrossRef]
- Edman, S.; Starck, J.; Corell, L.; Hangasjärvi, W.; von Finckenstein, A.; Reimeringer, M.; Reitzner, S.; Norrbom, J.; Moberg, M.; von Walden, F. Exercise-Induced Plasma Mature Brain-Derived Neurotrophic Factor Elevation in Children, Adolescents and Adults: Influence of Age, Maturity and Physical Activity. J. Physiol. 2025, 603, 2333–2347. [Google Scholar] [CrossRef] [PubMed]
- Baum, G.L.; Ciric, R.; Roalf, D.R.; Betzel, R.F.; Moore, T.M.; Shinohara, R.T.; Kahn, A.E.; Vandekar, S.N.; Rupert, P.E.; Quarmley, M.; et al. Modular Segregation of Structural Brain Networks Supports the Development of Executive Function in Youth. Curr. Biol. 2017, 27, 1561–1572.e8. [Google Scholar] [CrossRef] [PubMed]
- Hillman, C.H.; Erickson, K.I.; Kramer, A.F. Be Smart, Exercise Your Heart: Exercise Effects on Brain and Cognition. Nat. Rev. Neurosci. 2008, 9, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.; Tempest, G.D.; Hettinga, F.; Khudair, M.; McCullogh, N. The Impact of an Acute Bout of Physical Activity on Executive Function and Academic Achievement in Preadolescent Children: A Systematic Review and Meta-Analysis. Ment. Health Phys. Act. 2025, 28, 100685. [Google Scholar] [CrossRef]
- Youngstedt, S.D.; Elliott, J.A.; Kripke, D.F. Human Circadian Phase–Response Curves for Exercise. J. Physiol. 2019, 597, 2253–2268. [Google Scholar] [CrossRef]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A.; Wager, T.D. The Unity and Diversity of Executive Functions and Their Contributions to Complex “Frontal Lobe” Tasks: A Latent Variable Analysis. Cogn. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.G. Statistical Power Analyses Using G*Power 3.1:Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef]
- Menghini, L.; Yuksel, D.; Goldstone, A.; Baker, F.C.; de Zambotti, M. Performance of Fitbit Charge 3 against Polysomnography in Measuring Sleep in Adolescent Boys and Girls. Chronobiol. Int. 2021, 38, 1010–1022. [Google Scholar] [CrossRef]
- Scarpina, F.; Tagini, S. The Stroop Color and Word Test. Front. Psychol. 2017, 8, 557. [Google Scholar] [CrossRef]
- Stroop, J.R. Studies of Interference in Serial Verbal Reactions. J. Exp. Psychol. 1935, 18, 643–662. [Google Scholar] [CrossRef]
- Basner, M.; Mollicone, D.; Dinges, D.F. Validity and Sensitivity of a Brief Psychomotor Vigilance Test (PVT-B) to Total and Partial Sleep Deprivation. Acta Astronaut. 2011, 69, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Wiens, A.N.; Fuller, K.H.; Crossen, J.R. Paced Auditory Serial Addition Test: Adult Norms and Moderator Variables. J. Clin. Exp. Neuropsychol. 1997, 19, 473–483. [Google Scholar] [CrossRef]
- Meltzer, L.J.; Montgomery-Downs, H.E.; Insana, S.P.; Walsh, C.M. Use of Actigraphy for Assessment in Pediatric Sleep Research. Sleep Med. Rev. 2012, 16, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Acebo, C.; Sadeh, A.; Seifer, R.; Tzischinsky, O.; Wolfson, A.R.; Hafer, A.; Carskadon, M.A. Estimating Sleep Patterns with Activity Monitoring in Children and Adolescents: How Many Nights Are Necessary for Reliable Measures? Sleep 1999, 22, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Heck, R.H.; Thomas, S.L. An Introduction to Multilevel Modeling Techniques: MLM and SEM Approaches; Routledge: London, UK, 2020; Volume 3, ISBN 978-0-367-18242-7. [Google Scholar]
- West, B.T.; Welch, K.B.; Galecki, A.T. Linear Mixed Models: A Practical Guide Using Statistical Software, 3rd ed.; Taylor & Francis Group: Oxford, UK, 2022; ISBN 978-1-032-01932-1. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 1988; ISBN 0-8058-0283-5. [Google Scholar]
- Tudor-Locke, C.; Craig, C.L.; Beets, M.W.; Belton, S.; Cardon, G.M.; Duncan, S.; Hatano, Y.; Lubans, D.R.; Olds, T.S.; Raustorp, A.; et al. How Many Steps/Day Are Enough? For Children and Adolescents. Int. J. Behav. Nutr. Phys. Act. 2011, 8, 78. [Google Scholar] [CrossRef]
- Adams, M.A.; Johnson, W.D.; Tudor-Locke, C. Steps/Day Translation of the Moderate-to-Vigorous Physical Activity Guideline for Children and Adolescents. Int. J. Behav. Nutr. Phys. Act. 2013, 10, 49. [Google Scholar] [CrossRef]
- Kuula, L.; Pesonen, A.K.; Heinonen, K.; Kajantie, E.; Eriksson, J.G.; Andersson, S.; Lano, A.; Lahti, J.; Wolke, D.; Räikkönen, K. Naturally Occurring Circadian Rhythm and Sleep Duration Are Related to Executive Functions in Early Adulthood. J. Sleep Res. 2018, 27, 113–119. [Google Scholar] [CrossRef]
- Bei, B.; Wiley, J.F.; Trinder, J.; Manber, R. Beyond the Mean: A Systematic Review on the Correlates of Daily Intraindividual Variability of Sleep/Wake Patterns. Sleep Med. Rev. 2016, 28, 108–124. [Google Scholar] [CrossRef]
- Taillard, J.; Sagaspe, P.; Philip, P.; Bioulac, S. Sleep Timing, Chronotype and Social Jetlag: Impact on Cognitive Abilities and Psychiatric Disorders. Biochem. Pharmacol. 2021, 191, 114438. [Google Scholar] [CrossRef]
- Chellappa, S.L.; Morris, C.J.; Scheer, F.A.J.L. Daily Circadian Misalignment Impairs Human Cognitive Performance Task-Dependently. Sci. Rep. 2018, 8, 3041. [Google Scholar] [CrossRef]
- Lo, J.C.; Ong, J.L.; Leong, R.L.F.; Gooley, J.J.; Chee, M.W.L. Cognitive Performance, Sleepiness, and Mood in Partially Sleep Deprived Adolescents: The Need for Sleep Study. Sleep 2016, 39, 687–698. [Google Scholar] [CrossRef]
- Wallace, A.L.; Aguinaldo, L.; Thomas, M.L.; McCarthy, M.J.; Meruelo, A.D. Preliminary Findings on Caffeine Intake, Screen Time, Social Factors, and Psychological Well-Being: Their Impact on Chronotype and Sleep Health in Hispanic Adolescents. Sleep Adv. 2025, 6, zpaf019. [Google Scholar] [CrossRef] [PubMed]
- Calamaro, C.J.; Mason, T.B.A.; Ratcliffe, S.J. Adolescents Living the 24/7 Lifestyle: Effects of Caffeine and Technology on Sleep Duration and Daytime Functioning. Pediatrics 2009, 123, e1005–e1010. [Google Scholar] [CrossRef] [PubMed]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-Induced Neuroplasticity: A Mechanistic Model and Prospects for Promoting Plasticity. Neuroscientist 2019, 25, 65–85. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Smith, D.M. Neural Efficiency in Athletes: A Systematic Review. Front. Behav. Neurosci. 2021, 15, 698555. [Google Scholar] [CrossRef]
- Guzmán-Muñoz, E.; Concha-Cisternas, Y.; Jofré-Saldía, E.; Castillo-Paredes, A.; Molina-Márquez, I.; Yáñez-Sepúlveda, R. Physical Activity and Its Effects on Executive Functions and Brain Outcomes in Children: A Narrative Review. Brain Sci. 2025, 15, 1238. [Google Scholar] [CrossRef]
- Aly, M.; Kojima, H. Relationship of Regular Physical Activity with Neuroelectric Indices of Interference Processing in Young Adults. Psychophysiology 2020, 57, e13674. [Google Scholar] [CrossRef]
- Brooks, S.J.; Parks, S.M.; Stamoulis, C. Widespread Positive Direct and Indirect Effects of Regular Physical Activity on the Developing Functional Connectome in Early Adolescence. Cereb. Cortex 2021, 31, 4840–4852. [Google Scholar] [CrossRef]
- Menting, S.G.P.; Khudair, M.; Elferink-Gemser, M.T.; Hettinga, F.J. Unraveling the Role of (Meta-) Cognitive Functions in Pacing Behavior Development during Adolescence: Planning, Monitoring, and Adaptation. Med. Sci. Sports Exerc. 2023, 55, 1894–1904. [Google Scholar] [CrossRef]
- Tamnes, C.K.; Fjell, A.M.; Westlye, L.T.; Østby, Y.; Walhovd, K.B. Becoming Consistent: Developmental Reductions in Intraindividual Variability in Reaction Time Are Related to White Matter Integrity. J. Neurosci. 2012, 32, 972–982. [Google Scholar] [CrossRef]
- Scantlebury, N.; Cunningham, T.; Dockstader, C.; Laughlin, S.; Gaetz, W.; Rockel, C.; Dickson, J.; Mabbott, D. Relations between White Matter Maturation and Reaction Time in Childhood. J. Int. Neuropsychol. Soc. 2014, 20, 99–112. [Google Scholar] [CrossRef]
- Latino, F.; Tafuri, F. Physical Activity and Cognitive Functioning. Medicina 2024, 60, 216. [Google Scholar] [CrossRef] [PubMed]
- Daly, M.; McMinn, D.; Allan, J.L. A Bidirectional Relationship between Physical Activity and Executive Function in Older Adults. Front. Hum. Neurosci. 2015, 8, 1044. [Google Scholar] [CrossRef] [PubMed]
- Lubans, D.R.; Leahy, A.A.; Mavilidi, M.F.; Valkenborghs, S.R. Physical Activity, Fitness, and Executive Functions in Youth: Effects, Moderators, and Mechanisms. In Current Topics in Behavioral Neurosciences; Springer Science and Business Media Deutschland GmbH: Berlin/Heidelberg, Germany, 2022; Volume 53, pp. 103–130. [Google Scholar]
- McPherson, A.; Mackay, L.; Kunkel, J.; Duncan, S. Physical Activity, Cognition and Academic Performance: An Analysis of Mediating and Confounding Relationships in Primary School Children. BMC Public Health 2018, 18, 936. [Google Scholar] [CrossRef] [PubMed]
- Rakesh, D.; Lee, P.A.; Gaikwad, A.; McLaughlin, K.A. Annual Research Review: Associations of Socioeconomic Status with Cognitive Function, Language Ability, and Academic Achievement in Youth: A Systematic Review of Mechanisms and Protective Factors. J. Child Psychol. Psychiatry 2025, 66, 417–439. [Google Scholar] [CrossRef]
- Tomporowski, P.D. Effects of Acute Bouts of Exercise on Cognition. Acta Psychol. 2003, 112, 297–324. [Google Scholar] [CrossRef]
- You, Y.; Liu, J.; Wang, D.; Fu, Y.; Liu, R.; Ma, X. Cognitive Performance in Short Sleep Young Adults with Different Physical Activity Levels: A Cross-Sectional FNIRS Study. Brain Sci. 2023, 13, 171. [Google Scholar] [CrossRef]
- Lambourne, K.; Tomporowski, P. The Effect of Exercise-Induced Arousal on Cognitive Task Performance: A Meta-Regression Analysis. Brain Res. 2010, 1341, 12–24. [Google Scholar] [CrossRef]
- Mitáš, J.; Frömel, K.; Valach, P.; Suchomel, A.; Vorlíček, M.; Groffik, D. Secular Trends in the Achievement of Physical Activity Guidelines: Indicator of Sustainability of Healthy Lifestyle in Czech Adolescents. Sustainability 2020, 12, 5183. [Google Scholar] [CrossRef]
- Turrisi, T.B.; Bittel, K.M.; West, A.B.; Hojjatinia, S.; Hojjatinia, S.; Mama, S.K.; Lagoa, C.M.; Conroy, D.E. Seasons, Weather, and Device-Measured Movement Behaviors: A Scoping Review from 2006 to 2020. Int. J. Behav. Nutr. Phys. Act. 2021, 18, 24. [Google Scholar] [CrossRef]
- Bandera-Campos, F.J.; Grao-Cruces, A.; Camiletti-Moirón, D.; Martín-Acosta, F.; Muñoz-González, R.; González-Pérez, M.; Ruiz-Hermosa, A.; Vaquero-Solís, M.; Padilla-Moledo, C.; Sánchez-Oliva, D. Effectiveness of a Multicomponent Intervention to Promote Physical Activity during the School Day: Rationale and Methods of the MOVESCHOOL Study. Front. Public Health 2025, 13, 1565914. [Google Scholar] [CrossRef] [PubMed]
- Usán, P.U.; Bordás, C.S.; Mejías Abad, J.J. Relationship between Emotional Intelligence, Academic Burnout and School Performance in Adolescent Students. Rev. CES Psicol. 2020, 13, 125–139. [Google Scholar] [CrossRef]
- Liu, H.; Yao, M.; Li, J. Chinese Adolescents’ Achievement Goal Profiles and Their Relation to Academic Burnout, Learning Engagement, and Test Anxiety. Learn. Individ. Differ. 2020, 83–84, 101945. [Google Scholar] [CrossRef]
- Grao-Cruces, A.; Sánchez-Oliva, D.; Padilla-Moledo, C.; Izquierdo-Gómez, R.; Cabanas-Sánchez, V.; Castro-Piñero, J. Changes in the School and Non-School Sedentary Time in Youth: The UP&DOWN Longitudinal Study. J. Sports Sci. 2020, 38, 780–786. [Google Scholar] [CrossRef]
- Erickson, K.I.; Hillman, C.; Stillman, C.M.; Ballard, R.M.; Bloodgood, B.; Conroy, D.E.; Macko, R.; Marquez, D.X.; Petruzzello, S.J.; Powell, K.E. Physical Activity, Cognition, and Brain Outcomes: A Review of the 2018 Physical Activity Guidelines. Med. Sci. Sports Exerc. 2019, 51, 1242–1251. [Google Scholar] [CrossRef]
- Mahar, M.T.; Murphy, S.K.; Rowe, D.A.; Golden, J.; Shields, A.T.; Raedeke, T.D. Effects of a Classroom-Based Program on Physical Activity and on-Task Behavior. Med. Sci. Sports Exerc. 2006, 38, 2086–2094. [Google Scholar] [CrossRef]
- Mavilidi, M.F.; Pesce, C.; Benzing, V.; Schmidt, M.; Paas, F.; Okely, A.D.; Vazou, S. Meta-Analysis of Movement-Based Interventions to Aid Academic and Behavioral Outcomes: A Taxonomy of Relevance and Integration. Educ. Res. Rev. 2022, 37, 100478. [Google Scholar] [CrossRef]
- Ruiz-Hermosa, A.; Álvarez-Bueno, C.; Cavero-Redondo, I.; Martínez-Vizcaíno, V.; Redondo-Tébar, A.; Sánchez-López, M. Active Commuting to and from School, Cognitive Performance, and Academic Achievement in Children and Adolescents: A Systematic Review and Meta-Analysis of Observational Studies. Int. J. Environ. Res. Public Health 2019, 16, 1839. [Google Scholar] [CrossRef]
- Bedard, C.; St John, L.; Bremer, E.; Graham, J.D.; Cairney, J. A Systematic Review and Meta-Analysis on the Effects of Physically Active Classrooms on Educational and Enjoyment Outcomes in School Age Children. PLoS ONE 2018, 14, e021863. [Google Scholar] [CrossRef]
- Telford, D.M.; Meiring, R.M.; Gusso, S. Moving beyond Moderate-to-Vigorous Physical Activity: The Role of Light Physical Activity during Adolescence. Front. Sports Act. Living 2023, 5, 1282482. [Google Scholar] [CrossRef]
- van Sluijs, E.M.F.; Ekelund, U.; Crochemore-Silva, I.; Guthold, R.; Ha, A.; Lubans, D.; Oyeyemi, A.L.; Ding, D.; Katzmarzyk, P.T. Physical Activity Behaviours in Adolescence: Current Evidence and Opportunities for Intervention. Lancet 2021, 398, 429–442. [Google Scholar] [CrossRef]
- Cassar, S.; Salmon, J.; Timperio, A.; Naylor, P.J.; Van Nassau, F.; Contardo Ayala, A.M.; Koorts, H. Adoption, Implementation and Sustainability of School-Based Physical Activity and Sedentary Behaviour Interventions in Real-World Settings: A Systematic Review. Int. J. Behav. Nutr. Phys. Act. 2019, 16, 120. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.