Upper Limb Motor Planning in Individuals with Cerebral Palsy Aged between 3 and 21 Years Old: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Registration
2.2. Eligibility Criteria
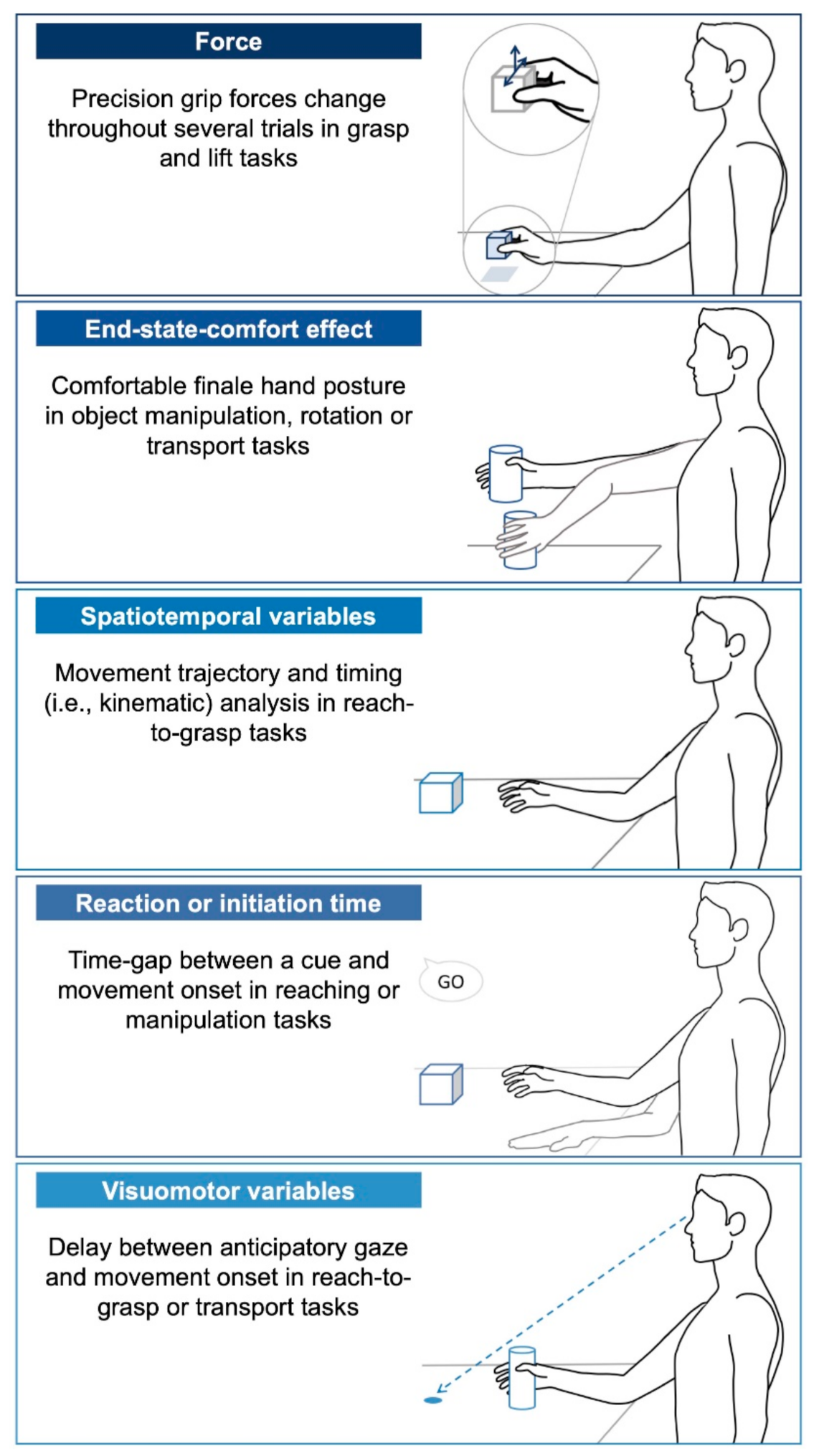
- Force: Grip (normal) and lift (tangential) forces scaling parameters are often measured through grasp and lift tasks where the participants must grasp an object between their thumb and finger and lift it from its support. The main motor planning variable is the development of change in force (force rate) exerted on the object prior to lifting necessary to lift it across trials and coordination of both forces (force ratios).
- End-state-comfort effect: This common motor planning variable is used to quantify how an individual anticipates the future state of their desired action [18]. Common research paradigms on end-state-comfort use an object manipulation task such as rotating a knob, rotating a bar or inserting a sword into a hole for younger children. This paradigm relies on the principle that an individual normally anticipates their final hand posture and thus, adapts their initial posture to minimize discomfort and allow flexibility for subsequent actions at the end of the task. Most often, the thumb points downward at the beginning of a desired action and it points upward when the rotation is completed, allowing the ability to supinate or pronate thereafter. “Comfort” experienced in the final posture is assumed to represent successful motor planning.
- Spatiotemporal variables: Movement trajectory is analyzed using kinematics, in particular during a reach-to-grasp task. The task can be divided into two segments: the reaching and grasping components [20]. To quantify motor planning in the reaching part (i.e., the movement towards the object without hand opening), the movement time required to complete the gesture or to attain a percentage of peak velocity is commonly measured in addition of bell-shaped velocity profiles and peak velocity scaled to distance. To quantify motor planning in the grasping part (i.e., the approach movement with hand opening), the time to reach maximum hand aperture is examined as well as peak velocity scaled to distance. These variables ultimately reflect the presence or absence of a motor planning deficit.
- Reaction time or movement initiation time: Both of these terms refer to the time-gap between a cue and movement onset to reflect motor planning. This variable is influenced by the complexity of the task, where longer reaction times are expected in more difficult or longer sequences of movement [50].
- Visuomotor variables: Temporal eye–hand coordination variables are often examined in reach-to-grasp tasks, in which an individual must move an object to a target [51,52]. The motor planning is assessed through the gaze that anticipates hand movement. Additionally, termed “movement onset asynchrony”, this variable quantifies the time delay between the first anticipatory gaze and hand movement initiation.
2.3. Data Sources
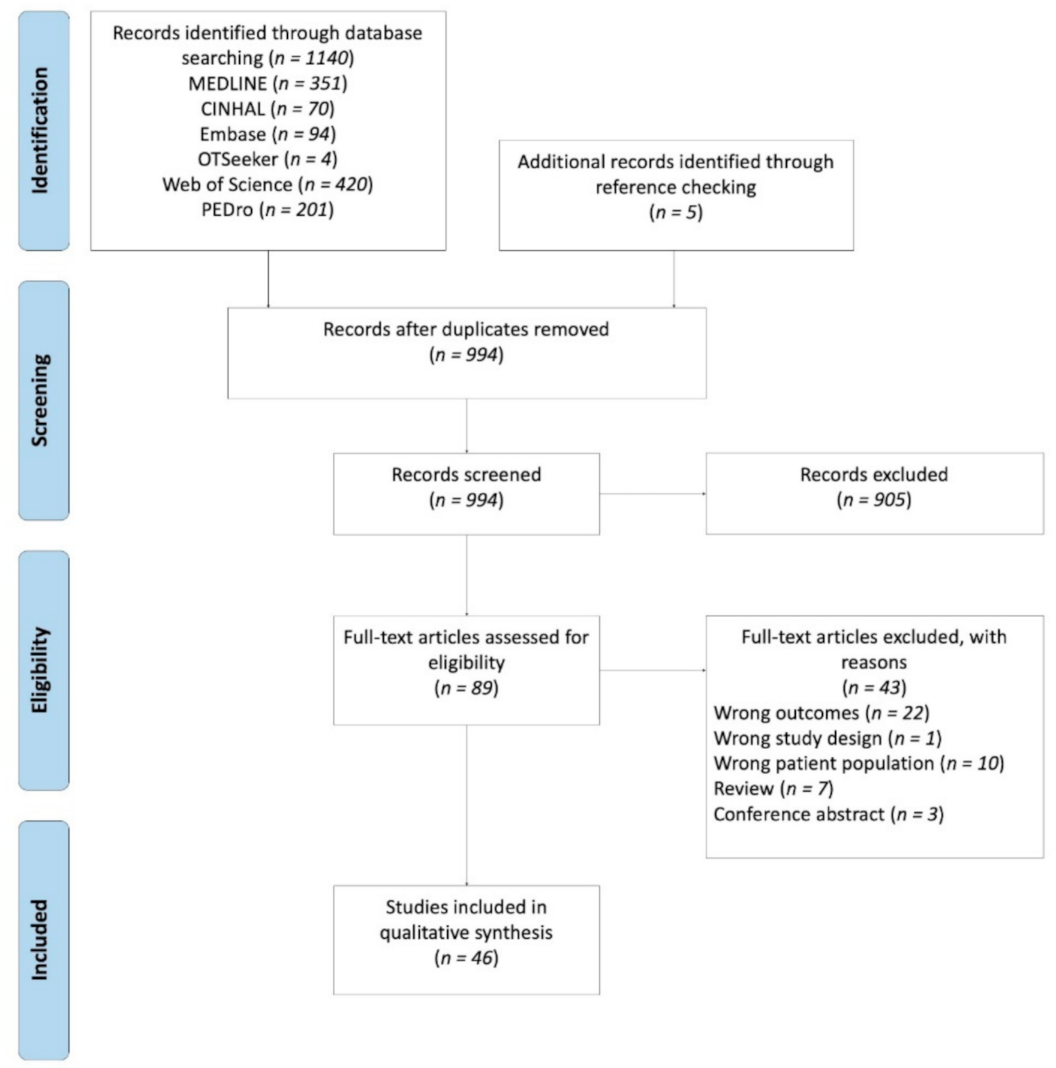
2.4. Study Selection
2.5. Data Extraction
2.6. Quality Assessment
2.7. Data Analysis
3. Results
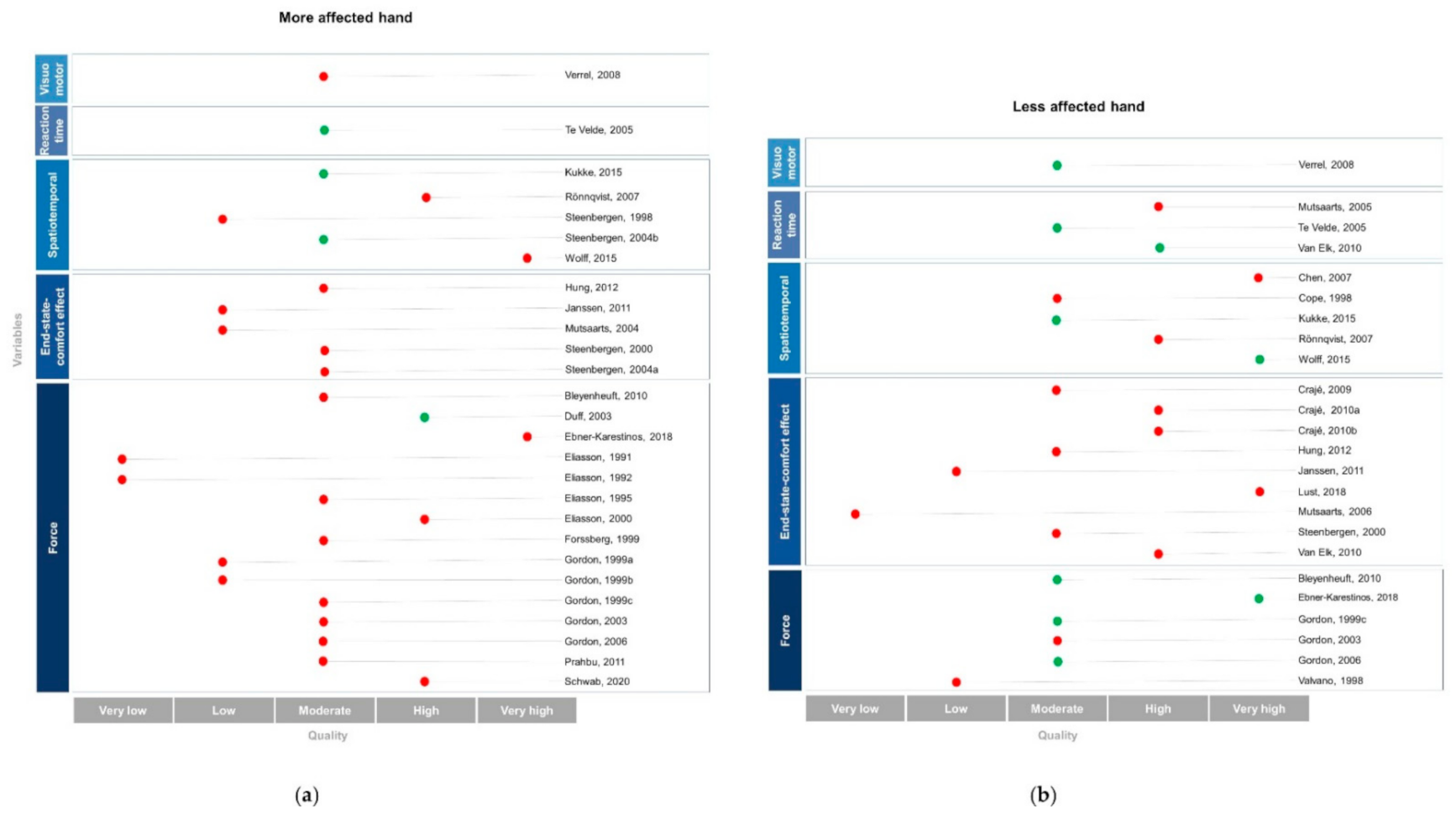
3.1. Quality Assessment
3.2. Participant’s Characteristic
3.3. Description of Results by Motor Planning Variable
3.3.1. Force
3.3.2. End-State-Comfort Effect
3.3.3. Spatiotemporal Variables
3.3.4. Reaction Time
3.3.5. Visuomotor Variables
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shevell, M.; Dagenais, L.; Oskoui, M. The epidemiology of cerebral palsy: New perspectives from a Canadian registry. Semin. Pediatr. Neurol. 2013, 20, 60–64. [Google Scholar] [CrossRef]
- Oskoui, M.; Coutinho, F.; Dykeman, J.; Jetté, N.; Pringsheim, T. An update on the prevalence of cerebral palsy: A systematic review and meta-analysis. Dev. Med. Child Neurol. 2013, 55, 509–519. [Google Scholar] [CrossRef]
- Boyle, C.A.; Boulet, S.; Schieve, L.A.; Cohen, R.A.; Blumberg, S.J.; Yeargin-Allsopp, M.; Visser, S.; Kogan, M.D. Trends in the prevalence of developmental disabilities in US children, 1997–2008. Pediatrics 2011, 127, 1034–1042. [Google Scholar] [CrossRef] [PubMed]
- Sellier, E.; Platt, M.J.; Andersen, G.L.; Krägeloh-Mann, I.; De La Cruz, J.; Cans, C.; Mejaski-Bosnjak, V. Decreasing prevalence in cerebral palsy, a multi-site European population-based study, 1980 to 2003. Dev. Med. Child Neurol. 2015, 58, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Krägeloh-Mann, I.; Horber, V. The role of magnetic resonance imaging in furthering understanding of the pathogenesis of cerebral palsy. Dev. Med. Child Neurol. 2007, 49, 948. [Google Scholar] [CrossRef]
- Robinson, M.N.; Peake, L.J.; Ditchfield, M.R.; Reid, S.M.; Lanigan, A.; Reddihough, D.S. Magnetic resonance imaging findings in a population-based cohort of children with cerebral palsy. Dev. Med. Child Neurol. 2009, 51, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Towsley, K.; Shevell, M.I.; Dagenais, L. Population-based study of neuroimaging findings in children with cerebral palsy. Eur. J. Paediatr. Neurol. 2011, 15, 29–35. [Google Scholar] [CrossRef]
- Ferrari, A. From movement to action: A new framework for cerebral palsy. Eur. J. Phys. Rehabil. Med. 2019, 55, 852–861. [Google Scholar] [CrossRef]
- James, S.; Ziviani, J.; Ware, R.S.; Boyd, R.N. Relationships between activities of daily living, upper limb function, and visual perception in children and adolescents with unilateral cerebral palsy. Dev. Med. Child Neurol. 2015, 57, 852–857. [Google Scholar] [CrossRef]
- Steenbergen, B.; Gordon, A.M. Activity limitation in hemiplegic cerebral palsy: Evidence for disorders in motor planning. Dev. Med. Child Neurol. 2006, 48, 780–783. [Google Scholar] [CrossRef]
- Johnson-Frey, S.H.; McCarty, M.E.; Keen, R. Reaching beyond spatial perception: Effects of intended furture actions on visually guided prehension. Vis. Cogn. 2004, 11, 371–399. [Google Scholar] [CrossRef]
- Wong, A.L.; Haith, A.M.; Krakauer, J.W. Motor planning. Neuroscientist 2015, 21, 385–398. [Google Scholar] [CrossRef]
- Kawato, M. Internal models for motor control and trajectory planning. Curr. Opin. Neurobiol. 1999, 9, 718–727. [Google Scholar] [CrossRef]
- Goodale, M.A. Transforming vision into action. Vision. Res. 2011, 51, 1567–1587. [Google Scholar] [CrossRef] [PubMed]
- Wolpert, D.M.; Flanagan, J.R. Motor prediction. Curr. Biol. 2001, 11, 729–732. [Google Scholar] [CrossRef]
- Forssberg, H.; Kinoshita, H.; Eliasson, A.C.; Johansson, R.S.; Westling, G.; Gordon, A.M. Development of human precision grip -II. Anticipatory control of isometric forces targeted for object’s weight. Exp. Brain Res. 1992, 90, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, A.C.; Forssberg, H.; Ikuta, K.; Apel, I.; Westling, G.; Johansson, R. Development of human precision grip-V. Anticipatory and triggered grip actions during sudden loading. Exp. Brain Res. 1995, 106, 425–433. [Google Scholar] [CrossRef]
- Rosenbaum, D.A.; Marchak, F.; Barnes, H.J.; Vaughan, J.; Slotta, J.D.; Jorgensen, M.J. Constraints for action selection: Overhand versus underhand grips. In Attention and Performance XIII, 1st ed.; Jeannerod, M., Ed.; Psychology Press: New York, NY, USA, 1990; pp. 321–342. [Google Scholar]
- Stöckel, T.; Hughes, C.M.L.; Schack, T. Representation of grasp postures and anticipatory motor planning in children. Psychol. Res. 2012, 76, 768–776. [Google Scholar] [CrossRef]
- Marteniuk, R.G.; Leavitt, J.L.; MacKenzie, C.L.; Athenes, S. Functional relationships between grasp and transport components in a prehension task. Hum. Mov. Sci. 1990, 9, 149–176. [Google Scholar] [CrossRef]
- Claxton, L.J.; Keen, R.; McCarty, M.E. Evidence of motor planning in infant reaching behavior. Psychol. Sci. 2003, 14, 354–356. [Google Scholar] [CrossRef]
- Hadders-Algra, M. Typical and atypical development of reaching and postural control in infancy. Dev. Med. Child Neurol. 2013, 55, 5–8. [Google Scholar] [CrossRef]
- Jeannerod, M. Visuomotor channels: Their integration in goal-directed prehension. Hum. Mov. Sci. 1999, 18, 201–218. [Google Scholar] [CrossRef]
- Braddick, O.; Atkinson, J. Visual control of manual actions: Brain mechanisms in typical development and developmental disorders. Dev. Med. Child Neurol. 2013, 55, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Johansson, R.S.; Ba, A. Eye-Hand Coordination in Object Manipulation. J. Neurosci. 2001, 21, 6917–6932. [Google Scholar] [CrossRef] [PubMed]
- Wiener, R.F.; Thurman, S.L.; Corbetta, D. Directing Gaze on a Scene Before Reaching for an Object Changes Over the First Year of Life. J. Mot. Learn. Dev. 2018, 6, 105–125. [Google Scholar] [CrossRef]
- Ferrel, C.; Bard, C.; Fleury, M. Coordination in childhood: Modifications of visuomotor representations in 6- to 11-year-old children. Exp. Brain Res. 2001, 138, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Bushnell, E.W.; Boudreau, J.P. Motor Development and the Mind: The Potential Role of Motor Abilities as a Determinant of Aspects of Perceptual Development. Child Dev. 1993, 64, 1005–1021. [Google Scholar] [CrossRef]
- Casey, B.J.; Galvan, A.; Hare, T.A. Changes in cerebral functional organization during cognitive development. Curr. Opin. Neurobiol. 2005, 15, 239–244. [Google Scholar] [CrossRef]
- Casey, B.J.; Tottenham, N.; Liston, C.; Durston, S. Imaging the developing brain: What have we learned about cognitive development? Trends Cogn. Sci. 2005, 9, 104–110. [Google Scholar] [CrossRef]
- Demsky, Y.; Carone, D.A.; Burns, W.J.; Sellers, A. Assessment of visual-motor coordination in 6-to 11-yr.-olds. Percept. Mot. Ski. 2000, 91, 311–321. [Google Scholar] [CrossRef]
- Pellizzer, G.; Hauert, C.A. Visuo-manual aiming movements in 6- to 10-year-old children: Evidence for an asymmetric and asynchronous development of information processes. Brain Cogn. 1996, 30, 175–193. [Google Scholar] [CrossRef][Green Version]
- Pennequin, V.; Sorel, O.; Fontaine, R. Motor planning between 4 and 7 years of age: Changes linked to executive functions. Brain Cogn. 2010, 74, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Serrien, D.J.; O’Regan, L. The development of motor planning strategies in children. Eur. J. Dev. Psychol. 2021, 18, 1–17. [Google Scholar] [CrossRef]
- Serrien, D.J.; Sovijärvi-Spapé, M.M.; Rana, G. Developmental changes in motor control: Insights from bimanual coordination. Dev. Psychol. 2014, 50, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Westerhausen, R.; Luders, E.; Specht, K.; Ofte, S.H.; Toga, A.W.; Thompson, P.M.; Helland, T.; Hugdahl, K. Structural and functional reorganization of the corpus callosum between the age of 6 and 8 years. Cereb. Cortex 2011, 21, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A. Executive functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed]
- Lebel, C.; Beaulieu, C. Longitudinal development of human brain wiring continues from childhood into adulthood. J. Neurosci. 2011, 31, 10937–10947. [Google Scholar] [CrossRef] [PubMed]
- Lebel, C.; Deoni, S. The development of brain white matter microstructure. Neuroimage 2018, 182, 207–218. [Google Scholar] [CrossRef]
- Lebel, C.; Walker, L.; Leemans, A.; Phillips, L.; Beaulieu, C. Microstructural maturation of the human brain from childhood to adulthood. Neuroimage 2008, 40, 1044–1055. [Google Scholar] [CrossRef]
- Bar-Haim, Y.; Bart, O. Motor function and social participation in kindergarten children. Soc. Dev. 2006, 15, 296–310. [Google Scholar] [CrossRef]
- Jovanovic, B.; Schwarzer, G. Learning to grasp efficiently: The development of motor planning and the role of observational learning. Vis. Res. 2011, 51, 945–954. [Google Scholar] [CrossRef]
- Wunsch, K.; Henning, A.; Aschersleben, G.; Weigelt, M. A Systematic Review of the End-State Comfort Effect in Normally Developing Children and in Children with Developmental Disorders. J. Mot. Learn. Dev. 2013, 1, 59–76. [Google Scholar] [CrossRef]
- Gordon, A.M.; Bleyenheuft, Y.; Steenbergen, B. Pathophysiology of impaired hand function in children with unilateral cerebral palsy. Dev. Med. Child Neurol. 2013, 55, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Steenbergen, B.; Jongbloed-Pereboom, M.; Spruijt, S.; Gordon, A.M. Impaired motor planning and motor imagery in children with unilateral spastic cerebral palsy: Challenges for the future of pediatric rehabilitation. Dev. Med. Child Neurol. 2013, 55, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Gordon, A.M. Impaired voluntary movement control and its rehabilitation in cerebral palsy. Adv. Exp. Med. Biol. 2016, 957, 291–311. [Google Scholar] [CrossRef]
- Steenbergen, B.; Verrel, J.; Gordon, A.M. Motor planning in congenital hemiplegia. Disabil. Rehabil. 2007, 29, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, D.A.; Van Heugten, C.M.; Caldwell, G.E. From cognition to biomechanics and back: The end-state comfort effect and the middle-is-faster effect. Acta Psychol. (Amst). 1996, 94, 59–85. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Estarli, M.; Barrera, E.S.A.; et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Systematic Reviews. 2016, 20, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Klapp, S.T. Reaction time analysis of two types of motor preparation for speech articulation: Action as a sequence of chunks. J. Mot. Behav. 2003, 35, 135–150. [Google Scholar] [CrossRef]
- Jeannerod, M. Specialized channels for cognitive responses. Cognition 1981, 10, 135–137. [Google Scholar] [CrossRef]
- Jeannerod, M. The timing of natural prehension movements. J. Mot. Behav. 1984, 16, 235–254. [Google Scholar] [CrossRef]
- Kmet, L.M.; Cook, L.S.; Lee, R.C. Standard Quality Assessment Criteria for Evaluating Primary Research Papers from a Variety of Fields; Alberta Heritage Foundation for Medical Research: Edmonton, AB, Canada, 2004. [Google Scholar]
- Balshem, H.; Helfand, M.; Sch, H.J.; Oxman, A.D.; Kunz, R.; Brozek, J.; Vist, G.E.; Falck-ytter, Y.; Meerpohl, J.; Norris, S.; et al. GRADE guidelines: 3. Rating the quality of evidence. J. Clin. Epidemiol. 2011, 64, 401–406. [Google Scholar] [CrossRef]
- Gwet, K.L. Handbook of Inter-Rater Reliability: The Definitive Guide to Measuring the Extent of Agreement among Raters, 3rd ed.; Advanced Analytics: Gaithersburg, MD, USA, 2014. [Google Scholar]
- Van Tulder, M.; Furlan, A.D.; Bombardier, C.; Bouter, L. Editorial Board of the Cochrane Collaboration Back Review Group Updated method guidelines for systematic reviews in the cochrane back review group. Spine 2003, 28, 1290–1299. [Google Scholar] [CrossRef]
- Haidich, A.B. Meta-analysis in medical research. Hippokratia 2010, 14 (Suppl. 1), 29–37. [Google Scholar] [PubMed]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; John Wiley & Sons: Oxford, UK, 2019. [Google Scholar]
- Fu, R.; Gartlehner, G.; Grant, M.; Shamliyan, T.; Sedrakyan, A.; Wilt, T.J.; Trikalinos, T.A. Conducting Quantitative Synthesis When Comparing Medical Interventions. Methods Guide for Effectiveness and Comparative Effectiveness Reviews; Agency for Healthcare Research Quality: Rockville, MD, USA, 2014.
- Lust, J.M.; Spruijt, S.; Wilson, P.H.; Steenbergen, B. Motor planning in children with cerebral palsy: A longitudinal perspective. J. Clin. Exp. Neuropsychol. 2018, 40, 559–566. [Google Scholar] [CrossRef]
- Krajenbrink, H.; Crichton, A.; Steenbergen, B.; Hoare, B. The development of anticipatory action planning in children with unilateral cerebral palsy. Res. Dev. Disabil. 2019, 85, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Van Mier, H. Movement Planning in Children with Motor Disorders: Diagnostic Implications of Pattern Complexity and Previewing in Copying. Dev. Neuropsychol. 1994, 10, 231–254. [Google Scholar] [CrossRef]
- Steenbergen, B.; Meulenbroek, R.G.J.; Rosenbaum, D.A. Constraints on grip selection in hemiparetic cerebral palsy: Effects of lesional side, end-point accuracy, and context. Cogn. Brain Res. 2004, 19, 145–159. [Google Scholar] [CrossRef] [PubMed]
- Valvano, J.; Newell, K.M. Practice of a precision isometric grip-force task by children with spastic cerebral palsy. Dev. Med. Child Neurol. 1998, 40, 464–473. [Google Scholar] [CrossRef]
- Mutalib, S.A.; Mace, M.; Burdet, E. Bimanual coordination during a physically coupled task in unilateral spastic cerebral palsy children. J. Neuroeng. Rehabil. 2019, 16, 1–12. [Google Scholar] [CrossRef]
- Islam, M.; Gordon, A.M.; Sköld, A.; Forssberg, H.; Eliasson, A.C. Grip force coordination during bimanual tasks in unilateral cerebral palsy. Dev. Med. Child Neurol. 2011, 53, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Smits-Engelsman, B.C.M.; Klingels, K.; Feys, H. Bimanual force coordination in children with spastic unilateral cerebral palsy. Res. Dev. Disabil. 2011, 32, 2011–2019. [Google Scholar] [CrossRef] [PubMed]
- Bleyenheuft, Y.; Thonnard, J.L. Predictive and reactive control of precision grip in children with congenital hemiplegia. Neurorehabil. Neural Repair 2010, 24, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, A.C.; Gordon, A.M.; Forssberg, H. Basic co-ordination of manipulative forces of children with cerebral palsy. Dev. Med. Child Neurol. 1991, 33, 661–670. [Google Scholar] [CrossRef]
- Prabhu, S.B.; Diermayr, G.; Gysin, P.; Gordon, A.M. Coordination of fingertip forces in object transport during gait in children with hemiplegic cerebral palsy. Dev. Med. Child Neurol. 2011, 53, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Ebner-Karestinos, D.; Flament, B.; Arnould, C.; Thonnard, J.L.; Bleyenheuft, Y. Precision grip control while walking down a step in children with unilateral cerebral palsy. PLoS ONE 2018, 13, e0191684. [Google Scholar] [CrossRef]
- Eliasson, A.C.; Gordon, A.M. Impaired force coordination during object release in children with hemiplegic cerebral palsy. Dev. Med. Child Neurol. 2000, 42, 228–234. [Google Scholar] [CrossRef]
- Schwab, S.M.; Grover, F.M.; Abney, D.H.; Silva, P.L.; Riley, M.A. Children and adolescents with cerebral palsy flexibly adapt grip control in response to variable task demands. Clin. Biomech. 2020, 80, 105149. [Google Scholar] [CrossRef]
- Forssberg, H.; Eliasson, A.; Redon-zouitenn, C.; Mercuri, E.; Dubowitz, L. Impaired grip-lift synergy in children with unilateral brain lesions. Brain 1999, 122, 1157–1168. [Google Scholar] [CrossRef]
- Gordon, A.M.; Lewis, S.R.; Eliasson, A.-C.; Duff, S.V. Object release under varying task constraints in children with hemiplegic cerebral palsy. Dev. Med. Child Neurol. 2003, 45, 240–248. [Google Scholar] [CrossRef]
- Gordon, A.M.; Charles, J.; Steenbergen, B. Fingertip force planning during grasp is disrupted by impaired sensorimotor integration in children with hemiplegic cerebral palsy. Pediatr. Res. 2006, 60, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, A.-C.; Gordon, A.M.; Forssberg, H. Impaired Anticipatory Control of Isometric Forces During Grasping by Children With Cerebral Palsy. Dev. Med. Child Neurol. 1992, 34, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Duff, S.V.; Gordon, A.M. Learning of grasp control in children with hemiplegic cerebral palsy. Dev. Med. Child Neurol. 2003, 45, 746–757. [Google Scholar] [CrossRef]
- Eliasson, A.-C.; Gordon, A.M.; Forssberg, H. Tactile Control of Isometric Fingertip Forces During Grasping in Children with Cerebral Palsy. Dev. Med. Child Neurol. 1995, 37, 72–84. [Google Scholar] [CrossRef]
- Gordon, A.M.; Duff, S.V. Fingertip forces during object manipulation in children with hemiplegic cerebral palsy. I: Anticipatory control. Dev. Med. Child Neurol. 1999, 41, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Gordon, A.M.; Charles, J.; Duff, S.V. Fingertip forces during object manipulation in children with hemiplegic cerebral palsy. II: Bilateral coordination. Dev. Med. Child Neurol. 1999, 41, 176–185. [Google Scholar] [CrossRef]
- Gordon, A.M.; Duff, S.V. Relation between clinical measures and fine manipulative control in children with hemiplegic cerebral palsy. Dev. Med. Child Neurol. 1999, 41, 586–591. [Google Scholar] [CrossRef]
- Eliasson, A.C.; Forssberg, H.; Hung, Y.-C.; Gordon, A.M. Development of Hand Function and Precision Grip Control in Individuals with Cerebral Palsy, A 13-Year Follow-up Study. Pediatrics 2006, 11, 1226–1236. [Google Scholar] [CrossRef]
- Janssen, L.; Steenbergen, B. Typical and atypical (cerebral palsy) development of unimanual and bimanual grasp planning. Res. Dev. Disabil. 2011, 32, 963–971. [Google Scholar] [CrossRef]
- Hung, Y.C.; Henderson, E.R.; Akbasheva, F.; Valte, L.; Ke, W.S.; Gordon, A.M. Planning and coordination of a reach-grasp-eat task in children with hemiplegia. Res. Dev. Disabil. 2012, 33, 1649–1657. [Google Scholar] [CrossRef]
- Steenbergen, B.; Hulstijn, W.; Dortmans, S. Constraints on grip selection in cerebral palsy: Minimising discomfort. Exp. Brain Res. 2000, 134, 385–397. [Google Scholar] [CrossRef]
- Mutsaarts, M.; Steenbergen, B.; Meulenbroek, R. A detailed analysis of the planning and execution of prehension movements by three adolescents with spastic hemiparesis due to cerebral palsy. Exp. Brain Res. 2004, 156, 293–304. [Google Scholar] [CrossRef]
- Crajé, C.; van der Kamp, J.; Steenbergen, B. Visual information for action planning in left and right congenital hemiparesis. Brain Res. 2009, 1261, 54–64. [Google Scholar] [CrossRef]
- Crajé, C.; van Elk, M.; Beeren, M.; van Schie, H.T.; Bekkering, H.; Steenbergen, B. Compromised motor planning and Motor Imagery in right Hemiparetic Cerebral Palsy. Res. Dev. Disabil. 2010, 31, 1313–1322. [Google Scholar] [CrossRef] [PubMed]
- Van Elk, M.; Crajé, C.; Beeren, M.E.G.V.; Steenbergen, B.; Van Schie, H.T.; Bekkering, H. Neural evidence for impaired action selection in right hemiparetic cerebral palsy. Brain Res. 2010, 1349, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Mutsaarts, M.; Steenbergen, B.; Bekkering, H. Anticipatory planning deficits and task context effects in hemiparetic cerebral palsy. Exp. Brain Res. 2006, 172, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Crajé, C.; Aarts, P.; Nijhuis-van der Sanden, M.; Steenbergen, B. Action planning in typically and atypically developing children (unilateral cerebral palsy). Res. Dev. Disabil. 2010, 31, 1039–1046. [Google Scholar] [CrossRef]
- Kirkpatrick, E.V.; Pearse, J.E.; Eyre, J.A.; Basu, A.P. Motor planning ability is not related to lesion side or functional manual ability in children with hemiplegic cerebral palsy. Exp. Brain Res. 2013, 231, 239–247. [Google Scholar] [CrossRef]
- Wolff, A.L.; Raghavan, P.; Kaminski, T.; Hillstrom, H.J.; Gordon, A.M. Differentiation of hand posture to object shape in children with unilateral spastic cerebral palsy. Res. Dev. Disabil. 2015, 45–46, 422–430. [Google Scholar] [CrossRef]
- Rönnqvist, L.; Rösblad, B. Kinematic analysis of unimanual reaching and grasping movements in children with hemiplegic cerebral palsy. Clin. Biomech. 2007, 22, 165–175. [Google Scholar] [CrossRef]
- Steenbergen, B.; Hulstijn, W.; Lemmens, I.H.L.; Meulenbroek, R.G.J. The timing of prehensile movements in subjects with cerebral palsy. Dev. Med. Child Neurol. 1998, 40, 108–114. [Google Scholar] [CrossRef]
- Steenbergen, B.; van der Kamp, J. Control of prehension in hemiparetic cerebral palsy: Similarities and differences between the ipsi- and contra-lesional sides of the body. Dev. Med. Child Neurol. 2004, 46, 325–332. [Google Scholar] [CrossRef]
- Kukke, S.N.; Curatalo, L.A.; De Campos, A.C.; Hallett, M.; Alter, K.E.; Damiano, D.L. Coordination of Reach-to-Grasp Kinematics in Individuals with Childhood-Onset Dystonia Due to Hemiplegic Cerebral Palsy. IEEE Trans. Neural Syst. Rehabil. Eng. 2016, 24, 582–590. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, T. Effect of task goals on the reaching patterns of children with cerebral palsy. J. Mot. Behav. 2007, 39, 317–325. [Google Scholar] [CrossRef]
- Trombly, C.A. Grasping in children with and without cerebral palsy a kinematic analysis. Scand. J. Occup. Ther. 1998, 5, 59–68. [Google Scholar] [CrossRef]
- Te Velde, A.F.; van der Kamp, J.; Becher, J.G.; van Bennekom, C.; Savelsbergh, G.J.P. Planning and control in a manual collision avoidance task by children with hemiparesis. Motor Control 2005, 9, 417–438. [Google Scholar] [CrossRef][Green Version]
- Mutsaarts, M.; Steenbergen, B.; Bekkering, H. Anticipatory planning of movement sequences in hemiparetic cerebral palsy. Motor Control 2005, 9, 439–458. [Google Scholar] [CrossRef]
- Surkar, S.M.; Hoffman, R.M.; Harbourne, R.; Kurz, M.J. Neural activation within the prefrontal cortices during the goal-directed motor actions of children with hemiplegic cerebral palsy. Neurophotonics 2018, 5, 1. [Google Scholar] [CrossRef]
- Verrel, J.; Bekkering, H.; Steenbergen, B. Eye-hand coordination during manual object transport with the affected and less affected hand in adolescents with hemiparetic cerebral palsy. Exp. Brain Res. 2008, 187, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Surkar, S.M.; Hoffman, R.M.; Davies, B.; Harbourne, R.; Kurz, M.J. Impaired anticipatory vision and visuomotor coordination affects action planning and execution in children with hemiplegic cerebral palsy. Res. Dev. Disabil. 2018, 80, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Poitras, I.; Martinie, O.; Robert, M.; Campeau-Lecours, A.; Mercier, C. Impact of somatosensory and visual deficits on upper limbs motor performance in individuals with cerebral palsy. Brain. Sci. 2021, 11, 744. [Google Scholar] [CrossRef]
- Mutsaarts, M.; Steenbergen, B.; Bekkering, H. Impaired motor imagery in right hemiparetic cerebral palsy. Neuropsychologia 2007, 45, 853–859. [Google Scholar] [CrossRef]
- Rigoldi, C.; Molteni, E.; Rozbaczylo, C.; Morante, M.; Albertini, G.; Bianchi, A.M.; Galli, M. Movement analysis and EEG recordings in children with hemiplegic cerebral palsy. Exp. Brain Res. 2012, 223, 517–524. [Google Scholar] [CrossRef]
- Payne, A.R.; Plimmer, B.; McDaid, A.; Davies, T.C. Eye movements show similar adaptations in temporal coordination to movement planning conditions in both people with and without cerebral palsy. Exp. Brain Res. 2017, 235, 1375–1385. [Google Scholar] [CrossRef] [PubMed]
- Blank, R.; Hermsdörfer, J. Basic motor capacity in relation to object manipulation and general manual ability in young children with spastic cerebral palsy. Neurosci. Lett. 2009, 450, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Bleyenheuft, Y.; Gordon, A.M. Precision grip control, sensory impairments and their interactions in children with hemiplegic cerebral palsy: A systematic review. Res. Dev. Disabil. 2013, 34, 3014–3028. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.J.; Wu, T.I.; Wu, W.L.; Su, F.C. Kinematical measure for spastic reaching in children with cerebral palsy. Clin. Biomech. 2005, 20, 381–388. [Google Scholar] [CrossRef]
- Gentilucci, M.; Toni, I.; Chieffi, S.; Pavesi, G. The role of proprioception in the control of prehension movements: A kinematic study in a peripherally deafferented patient and in normal subjects. Exp. Brain Res. 1994, 99, 483–500. [Google Scholar] [CrossRef] [PubMed]
- Lemon, R.N.; Johansson, R.S.; Westling, G. Corticospinal control during reach, grasp, and precision lift in man. J. Neurosci. 1995, 15, 6145–6156. [Google Scholar] [CrossRef]
- Wang, S.; Williams, J.; Wilmut, K. Constraints on Motor Planning Across the Life Span: Physical, Cognitive, and Motor Factors. Psychol. Aging 2019, 35, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Jongbloed-Pereboom, M.; Spruijt, S.; Nijhuis-Van Der Sanden, M.W.G.; Steenbergen, B. Measurement of Action Planning in Children, Adolescents, and Adults: A Comparison between 3 Tasks. Pediatr. Phys. Ther. 2016, 28, 33–39. [Google Scholar] [CrossRef]
- Adams, I.L.J.; Ferguson, G.D.; Lust, J.M.; Steenbergen, B.; Smits-Engelsman, B.C.M. Action planning and position sense in children with Developmental Coordination Disorder. Hum. Mov. Sci. 2016, 46, 196–208. [Google Scholar] [CrossRef]
- Bodimeade, H.L.; Whittingham, K.; Lloyd, O.; Boyd, R.N. Executive function in children and adolescents with unilateral cerebral palsy. Dev. Med. Child Neurol. 2013, 55, 926–933. [Google Scholar] [CrossRef]
- Bottcher, L.; Flachs, E.M.; Uldall, P. Attentional and executive impairments in children with spastic cerebral palsy. Dev. Med. Child Neurol. 2010, 52. [Google Scholar] [CrossRef]
- Guzzetta, A.; Mercuri, E.; Cioni, G. Visual disorders in children with brain lesions: 2. Visual impairment associated with cerebral palsy. Eur. J. Paediatr. Neurol. 2001, 5, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Stiers, P.; Vanneste, G.; Coene, S.; Vandenbussche, E. Visual-perceptual impairment in a random sample of children with cerebral palsy. Dev. Med. Child Neurol. 2002, 44, 370–382. [Google Scholar] [CrossRef] [PubMed]
- Menken, C.; Cermak, S.A.; Fisher, A. Evaluating the visual-perceptual skills of children with cerebral palsy. Am. J. Occup. Ther. Off. Publ. Am. Occup. Ther. Assoc. 1987, 41, 646–651. [Google Scholar] [CrossRef] [PubMed]
- Ickx, G.; Hatem, S.M.; Riquelme, I.; Friel, K.M.; Henne, C.; Araneda, R.; Gordon, A.M.; Bleyenheuft, Y. Impairments of visuospatial attention in children with unilateral spastic cerebral palsy. Neural Plast. 2018, 2018. [Google Scholar] [CrossRef]
- Hawe, R.L.; Kuczynski, A.M.; Kirton, A.; Dukelow, S.P. Assessment of bilateral motor skills and visuospatial attention in children with perinatal stroke using a robotic object hitting task. J. Neuroeng. Rehabil. 2020, 17, 1–12. [Google Scholar] [CrossRef]
- Ferrari, A.; Tersi, L.; Ferrari, A.; Sghedoni, A.; Chiari, L. Functional reaching discloses perceptive impairment in diplegic children with cerebral palsy. Gait Posture 2010, 32, 253–258. [Google Scholar] [CrossRef]
- Niermeyer, M.A.; Ziemnik, R.E.; Franchow, E.I.; Barron, C.A.; Suchy, Y. Greater naturally occurring expressive suppression is associated with poorer executive functioning and motor-sequence learning among older adults. J. Clin. Exp. Neuropsychol. 2019, 41, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Euler, M.J.; Niermeyer, M.A.; Suchy, Y. Neurocognitive and neurophysiological correlates of motor planning during familiar and novel contexts. Neuropsychology 2016, 30, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Stöckel, T.; Hughes, C.M.L. Effects of multiple planning constraints on the development of grasp posture planning in 6-to 10-year-old children. Dev. Psychol. 2015, 51, 1254–1261. [Google Scholar] [CrossRef]
- Himmelmann, K.; Horber, V.; Sellier, E.; De la Cruz, J.; Papavasiliou, A.; Krägeloh-Mann, I. Neuroimaging Patterns and Function in Cerebral Palsy—Application of an MRI Classification. Front. Neurol. 2021, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Mailleux, L.; Simon-Martinez, C.; Klingels, K.; Jaspers, E.; Desloovere, K.; Demaerel, P.; Fiori, S.; Guzzetta, A.; Ortibus, E.; Feys, H. Structural brain damage and upper limb kinematics in children with unilateral cerebral palsy. Front. Hum. Neurosci. 2017, 11. [Google Scholar] [CrossRef]
- Franki, I.; Mailleux, L.; Emsell, L.; Peedima, M.L.; Fehrenbach, A.; Feys, H.; Ortibus, E. The relationship between neuroimaging and motor outcome in children with cerebral palsy: A systematic review-Part A. Structural imaging. Res. Dev. Disabil. 2020, 100, 103606. [Google Scholar] [CrossRef] [PubMed]
- Mailleux, L.; Franki, I.; Emsell, L.; Peedima, M.L.; Fehrenbach, A.; Feys, H.; Ortibus, E. The relationship between neuroimaging and motor outcome in children with cerebral palsy: A systematic review—Part B diffusion imaging and tractography. Res. Dev. Disabil. 2020, 97, 103569. [Google Scholar] [CrossRef]
- Castro-Sierra, E.; Chico-Ponce de León, F.; Gordillo-Domínguez, L.F.; Raya-Gutiérrez, R. Surgery of tumors of the cerebellum and prefrontal cortex, and sensory memory and motor planning disturbances in children. Child’s Nerv. Syst. 2003, 19, 736–743. [Google Scholar] [CrossRef]
- Tartaglione, T.; Izzo, G.; Alexandre, A.; Botto, A.; Di Lella, G.M.; Gaudino, S.; Caldarelli, M.; Colosimo, C. MRI findings of olivary degeneration after surgery for posterior fossa tumours in children: Incidence, time course and correlation with tumour grading. Radiol. Medica 2015, 120, 474–482. [Google Scholar] [CrossRef]
- Bhoyroo, R.; Hands, B.; Wilmut, K.; Hyde, C.; Wigley, A. Investigating motor planning in children with DCD: Evidence from simple and complex grip-selection tasks. Hum. Mov. Sci. 2018, 61, 42–51. [Google Scholar] [CrossRef]
- Anat, D.; Miriam, R. Evidence for deficient motor planning in ADHD. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Hung, Y.C.; Robert, M.T.; Friel, K.M.; Gordon, A.M. Relationship Between Integrity of the Corpus Callosum and Bimanual Coordination in Children with Unilateral Spastic Cerebral Palsy. Front. Hum. Neurosci. 2019, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Robert, M.T.; Gutterman, J.; Ferre, C.L.; Chin, K.; Brandao, M.B.; Gordon, A.M.; Friel, K. Corpus Callosum Integrity Relates to Improvement of Upper-Extremity Function Following Intensive Rehabilitation in Children with Unilateral Spastic Cerebral Palsy. Neurorehabil. Neural Repair 2021, 35, 534–544. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinie, O.; Mercier, C.; Gordon, A.M.; Robert, M.T. Upper Limb Motor Planning in Individuals with Cerebral Palsy Aged between 3 and 21 Years Old: A Systematic Review. Brain Sci. 2021, 11, 920. https://doi.org/10.3390/brainsci11070920
Martinie O, Mercier C, Gordon AM, Robert MT. Upper Limb Motor Planning in Individuals with Cerebral Palsy Aged between 3 and 21 Years Old: A Systematic Review. Brain Sciences. 2021; 11(7):920. https://doi.org/10.3390/brainsci11070920
Chicago/Turabian StyleMartinie, Ophélie, Catherine Mercier, Andrew M. Gordon, and Maxime T. Robert. 2021. "Upper Limb Motor Planning in Individuals with Cerebral Palsy Aged between 3 and 21 Years Old: A Systematic Review" Brain Sciences 11, no. 7: 920. https://doi.org/10.3390/brainsci11070920
APA StyleMartinie, O., Mercier, C., Gordon, A. M., & Robert, M. T. (2021). Upper Limb Motor Planning in Individuals with Cerebral Palsy Aged between 3 and 21 Years Old: A Systematic Review. Brain Sciences, 11(7), 920. https://doi.org/10.3390/brainsci11070920






