In-Vitro Dynamic Bioaccessibility and Bioavailability as Key Parameters for Rational Engineering of Novel Curcumin Delivery Systems
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Methods
2.2.1. Preparation of Oleogel
2.2.2. Preparation of Oil Gelled-in-Water (Og/W) Simple Emulsion
2.2.3. Preparation of Water-in-Oil Gelled-in-Water (W1/Og/W2) Multiple Emulsion
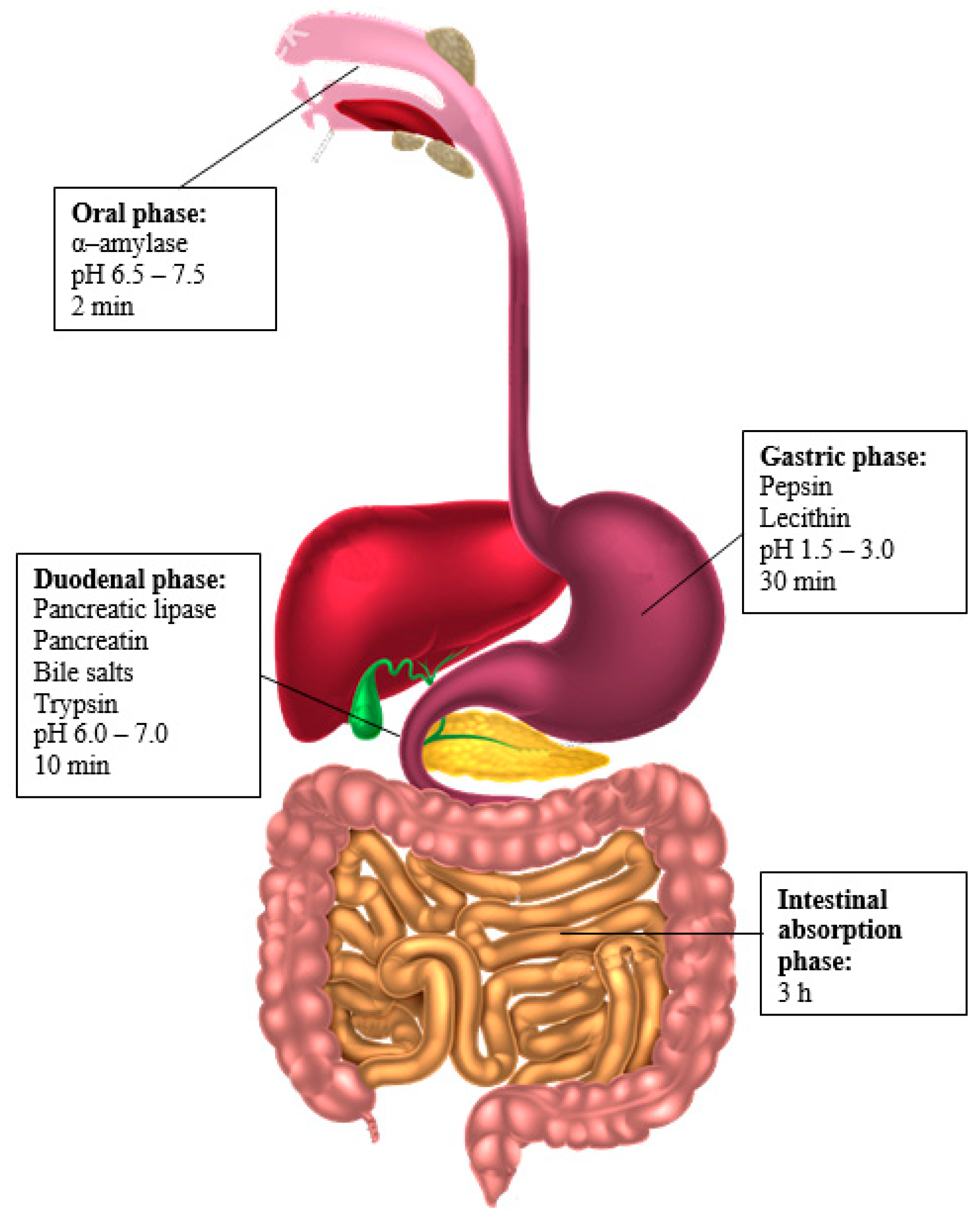
2.2.4. In Vitro Bioaccessibility and Bioavailability Assays
Equipment Overview
Essay Overview
- Simulation of the oral phase
- Simulation of the gastric phase
- Simulation of the duodenal phase
- Simulation of the intestinal absorption phase
Description of the Membrane Cleaning Protocol
- Physical cleaning.
- Chemical cleaning
- Washes or rinses.
2.2.5. Curcumin Concentration Measurements
2.2.6. Statistical Analysis
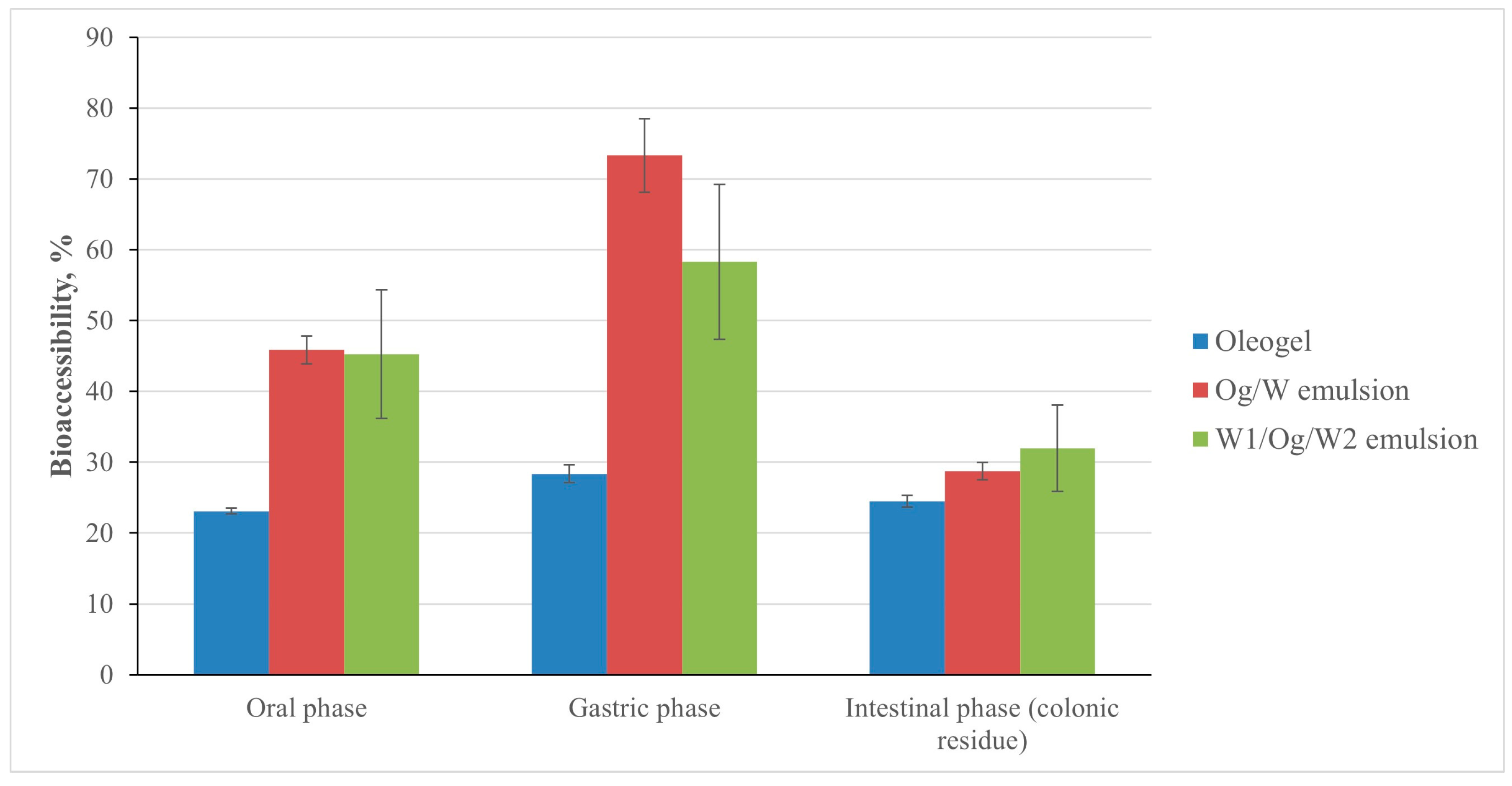
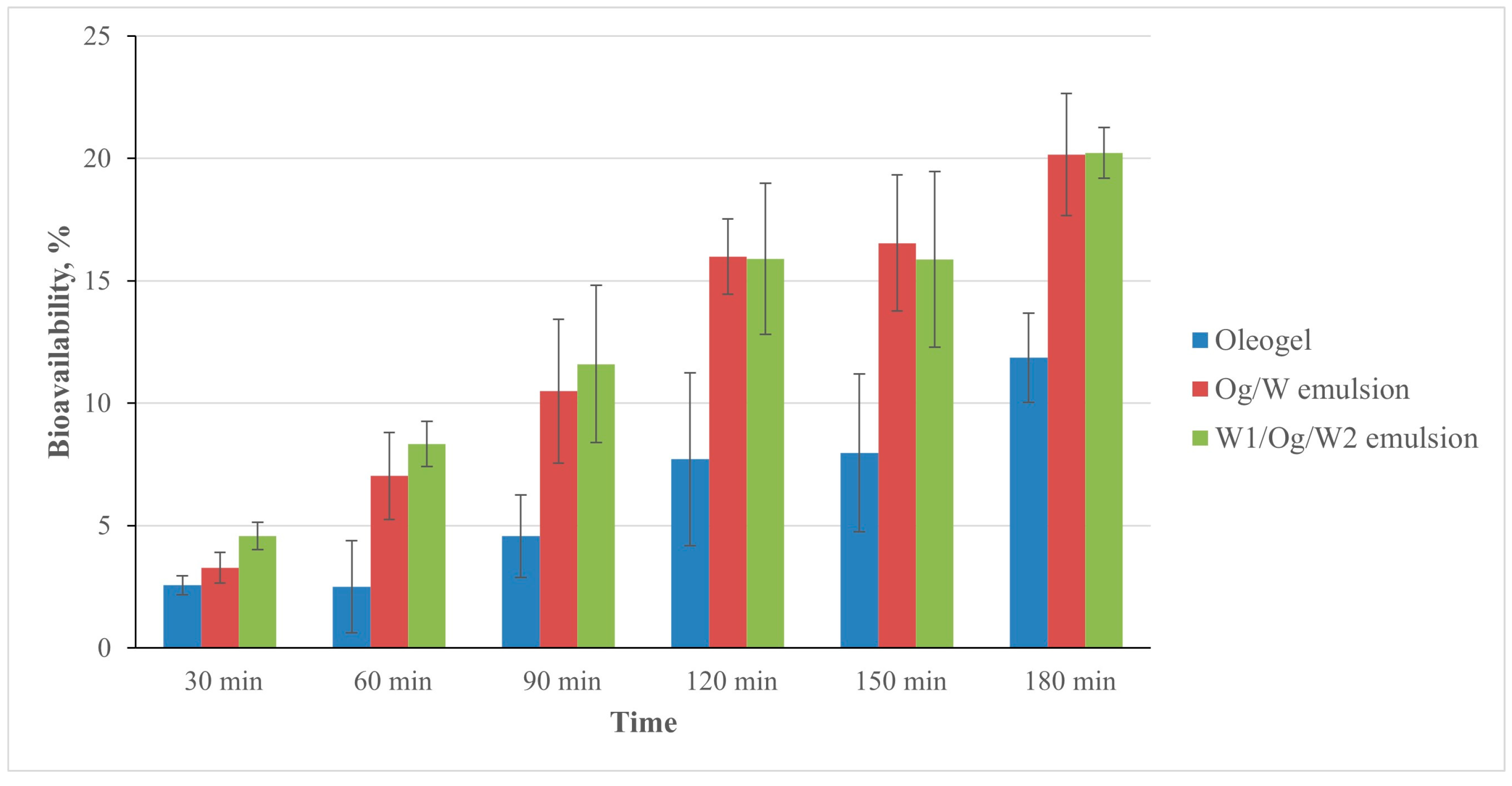
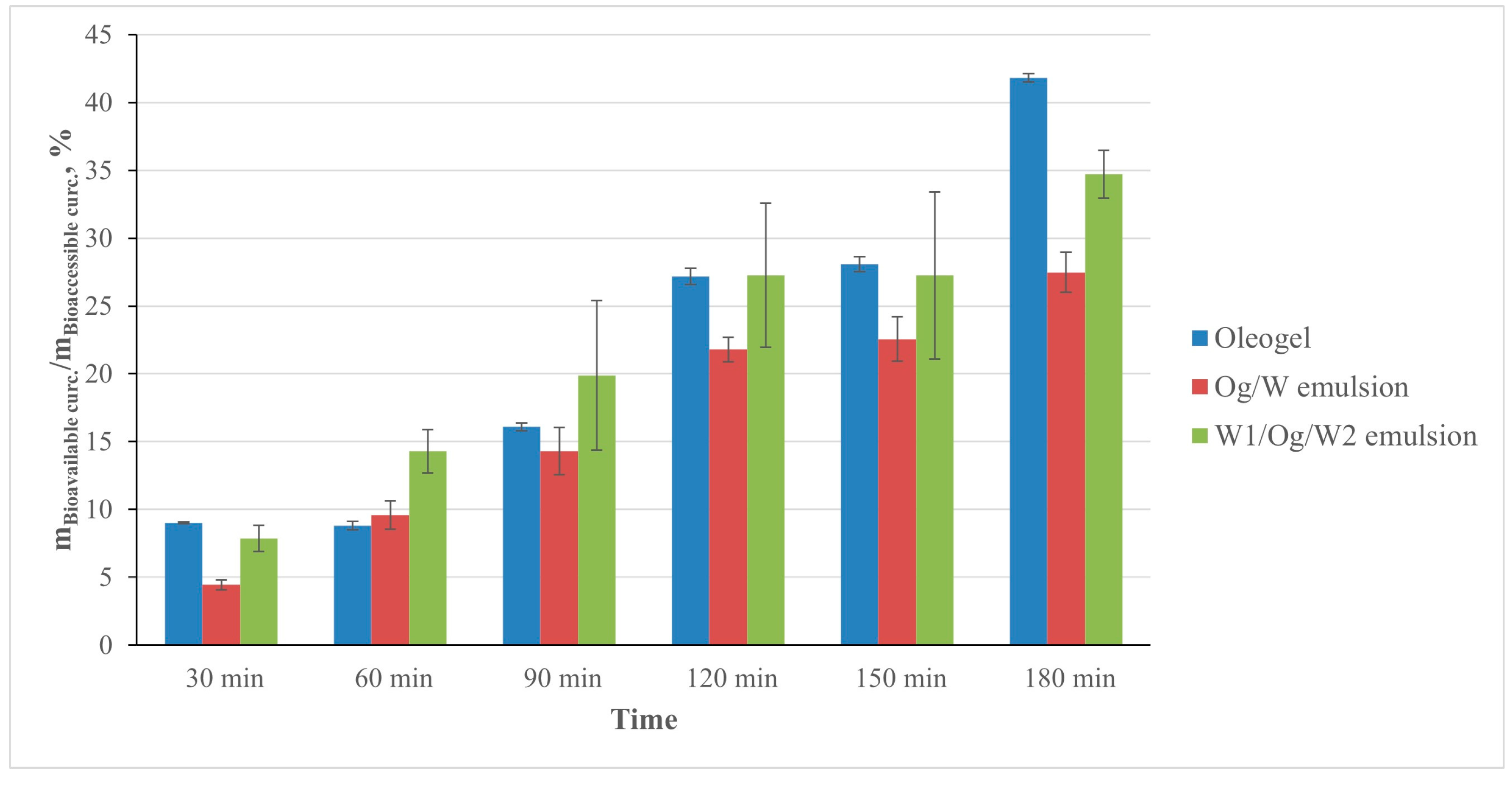
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Diplock, A.T.; Aggett, P.J.; Ashwell, M.; Bornet, F.; Fern, E.B.; Roberfroid, M.B. Scientific Concepts of Functional Foods in Europe. Consensus Document. Br. J. Nutr. 1999, 81, S1–S27. [Google Scholar] [CrossRef]
- Gil Hernández, Á.; Sánchez de Medina Contreras, F.; Ruiz López, M.; Camarero González, E.; Álvarez Hernández, J. Tratado de Nutrición, 1st ed.; Acción Médica: Madrid, Spain, 2005; ISBN 978-84-88336-40-8. [Google Scholar]
- Granato, D.; Barba, F.J.; Bursać Kovačević, D.; Lorenzo, J.M.; Cruz, A.G.; Putnik, P. Functional Foods: Product Development, Technological Trends, Efficacy Testing, and Safety. Annu. Rev. Food Sci. Technol. 2020, 11, 93–118. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of Curcumin: Problems and Promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Dulbecco, P.; Savarino, V. Therapeutic Potential of Curcumin in Digestive Diseases. World J. Gastroenterol. 2013, 19, 9256–9270. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.; Sanderson, I.R.; MacDonald, T.T. Curcumin as a Therapeutic Agent: The Evidence from In Vitro, Animal and Human Studies. Br. J. Nutr. 2010, 103, 1545–1557. [Google Scholar] [CrossRef]
- Grynkiewicz, G.; Ślifirski, P. Curcumin and Curcuminoids in Quest for Medicinal Status. Acta Biochim. Pol. 2012, 59, 201–212. [Google Scholar] [CrossRef]
- Maheshwari, R.K.; Singh, A.K.; Gaddipati, J.; Srimal, R.C. Multiple Biological Activities of Curcumin: A Short Review. Life Sci. 2006, 78, 2081–2087. [Google Scholar] [CrossRef]
- Ravindranath, V.; Chandrasekhara, N. Absorption and Tissue Distribution of Curcumin in Rats. Toxicology 1982, 22, 337. [Google Scholar] [CrossRef]
- Holder, G.M.; Plummer, J.L.; Ryan, A.J. The Metabolism and Excretion of Curcumin (1,7-Bis-(4-Hydroxy-3-Methoxyphenyl)-1,6-Heptadiene-3,5-Dione) in the Rat. Xenobiotica Fate Foreign Compd. Biol. Syst. 1978, 8, 761–768. [Google Scholar] [CrossRef]
- Prasad, S.; Tyagi, A.K.; Aggarwal, B.B. Recent Developments in Delivery, Bioavailability, Absorption and Metabolism of Curcumin: The Golden Pigment from Golden Spice. Cancer Res. Treat. Off. J. Korean Cancer Assoc. 2014, 46, 2–18. [Google Scholar] [CrossRef]
- Pan, M.-H.; Huang, T.-M.; Lin, J.-K. Biotransformation of Curcumin through Reduction and Glucuronidation in Mice. Drug Metab. Dispos. 1999, 27, 486–494. [Google Scholar] [CrossRef]
- Ireson, C.; Orr, S.; Jones, D.J.; Verschoyle, R.; Lim, C.K.; Luo, J.L.; Howells, L.; Plummer, S.; Jukes, R.; Williams, M.; et al. Characterization of Metabolites of the Chemopreventive Agent Curcumin in Human and Rat Hepatocytes and in the Rat in Vivo, and Evaluation of Their Ability to Inhibit Phorbol Ester-Induced Prostaglandin E2 Production. Cancer Res. 2001, 61, 1058–1064. [Google Scholar]
- Pal, A.; Sung, B.; Bhanu Prasad, B.A.; Schuber, P.T.; Prasad, S.; Aggarwal, B.B.; Bornmann, W.G. Curcumin Glucuronides: Assessing the Proliferative Activity Against Human Cell Lines. Bioorg. Med. Chem. 2014, 22, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.A.; McLelland, H.R.; Hill, K.A.; Ireson, C.R.; Euden, S.A.; Manson, M.M.; Pirmohamed, M.; Marnett, L.J.; Gescher, A.J.; Steward, W.P. Pharmacodynamic and Pharmacokinetic Study of Oral Curcuma Extract in Patients with Colorectal Cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2001, 7, 1894–1900. [Google Scholar]
- Wahlström, B.; Blennow, G. A Study on the Fate of Curcumin in the Rat. Acta Pharmacol. Toxicol. 1978, 43, 86–92. [Google Scholar] [CrossRef]
- Aggett, P.J. Population Reference Intakes and Micronutrient Bioavailability: A European Perspective. Am. J. Clin. Nutr. 2010, 91, 1433S–1437S. [Google Scholar] [CrossRef]
- Hurrell, R.; Egli, I. Iron Bioavailability and Dietary Reference Values. Am. J. Clin. Nutr. 2010, 91, 1461S–1467S. [Google Scholar] [CrossRef]
- Hur, S.J.; Lim, B.O.; Decker, E.A.; McClements, D.J. In Vitro Human Digestion Models for Food Applications. Food Chem. 2011, 125, 1–12. [Google Scholar] [CrossRef]
- Holst, B.; Williamson, G. Nutrients and Phytochemicals: From Bioavailability to Bioefficacy Beyond Antioxidants. Curr. Opin. Biotechnol. 2008, 19, 73–82. [Google Scholar] [CrossRef]
- Paixão Teixeira, J.L.; Lima Pallone, J.A.; Seiquer, I.; Morales-González, J.A.; Vellido-Pérez, J.A.; Martinez-Ferez, A. In Vitro Digestion Assays Using Dynamic Models for Essential Minerals in Brazilian Goat Cheeses. Food Anal. Methods 2022, 15, 2879–2889. [Google Scholar] [CrossRef]
- Rivas-Montoya, E.; Ochando-Pulido, J.M.; López-Romero, J.M.; Martinez-Ferez, A. Application of a novel gastrointestinal tract simulator system based on a membrane bioreactor (SimuGIT®) to study the stomach tolerance and effective delivery enhancement of nanoencapsulated macelignan. Chem. Eng. Sci. 2016, 140, 104–113. [Google Scholar] [CrossRef]
- Vellido Pérez, J.A. Diseño, Desarrollo y Optimización de Diferentes Sistemas de Liberación Modificada Para la Protección y Vehiculización de Ácidos Grasos Poliinsaturados Omega-3 y Curcumina. Ph.D. Thesis, University of Granada, Granada, Spain, 2021. [Google Scholar]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST Static In Vitro Simulation of Gastrointestinal Food Digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Vellido-Pérez, J.A.; Rodríguez-Remacho, C.; Rodríguez-Rodríguez, J.; Ochando-Pulido, J.M.; la Fuente, E.B.-D.; Martínez-Férez, A. Optimization of Oleogel Formulation for Curcumin Vehiculization and Lipid Oxidation Stability by Multi-Response Surface Methodology. Chem. Eng. Trans. 2019, 75, 427–432. [Google Scholar] [CrossRef]
- Vellido-Perez, J.A.; Ochando-Pulido, J.M.; Brito-de la Fuente, E.; Martinez-Ferez, A. Effect of Operating Parameters on the Physical and Chemical Stability of an Oil Gelled-in-Water Emulsified Curcumin Delivery System. J. Sci. Food Agric. 2021, 101, 6395–6406. [Google Scholar] [CrossRef]
- Vellido-Perez, J.A.; Ochando-Pulido, J.M.; Martinez-Ferez, A. Enhancing Physical and Chemical Stability of a Water-in-Oil Gelled-in-Water Multiple Emulsion Containing Omega-3 Polyunsaturated Fatty Acids and Curcumin by Draper-Lin Composite Design. LWT 2025, 215, 117265. [Google Scholar] [CrossRef]
- Godoy, V.; Martínez-Férez, A.; Martín-Lara, M.A.; Vellido-Pérez, J.A.; Calero, M.; Blázquez, G. Microplastics as Vectors of Chromium and Lead During Dynamic Simulation of the Human Gastrointestinal Tract. Sustainability 2020, 12, 4792. [Google Scholar] [CrossRef]
- Carrero, J.J.; Martín-Bautista, E.; Baró, L.; Fonollá, J.; Jiménez, J.; Boza, J.J.; López-Huertas, E. Efectos Cardiovasculares de Los Ácidos Grasos Omega-3 y Alternativas Para Incrementar Su Ingesta. Nutr. Hosp. 2005, 20, 63–69. [Google Scholar]
- Abad, P.; Arroyo-Manzanares, N.; Rivas-Montoya, E.; Ochando-Pulido, J.m.; Guillamon, E.; García-Campaña, A.M.; Martinez-Ferez, A. Effects of Different Vehiculization Strategies for the Allium Derivative Propyl Propane Thiosulfonate During Dynamic Simulation of the Pig Gastrointestinal Tract. Can. J. Anim. Sci. 2018, 99, 244–253. [Google Scholar] [CrossRef]
- Luo, N.; Ye, A.; Wolber, F.M.; Singh, H. Digestion behaviour of capsaicinoid-loaded emulsion gels and bioaccessibility of capsaicinoids: Effect of emulsifier type. Curr. Res. Food Sci. 2023, 6, 100473. [Google Scholar] [CrossRef]
- Eisinaite, V.; Duque Estrada, P.; Schroën, K.; Berton-Carabin, C.; Leskauskaite, D. Tayloring W/O/W emulsion composition for effective encapsulation: The role of PGPR in water transfer-induced swelling. Food Res. Int. 2018, 106, 722–728. [Google Scholar] [CrossRef]
- Lestari, M.L.A.D.; Indrayanto, G. Profiles of Drug Substances, Excipients and Related Methodology. Curcumin 2014, 39, 113–204. [Google Scholar] [CrossRef]
- Wang, Y.-J.; Pan, M.-H.; Cheng, A.-L.; Lin, L.-I.; Ho, Y.-S.; Hsieh, C.-Y.; Lin, J.-K. Stability of Curcumin in Buffer Solutions and Characterization of Its Degradation Products. J. Pharm. Biomed. Anal. 1997, 15, 1867–1876. [Google Scholar] [CrossRef]
- Tønnesen, H.H.; Karlsen, J. Studies on Curcumin and Curcuminoids—VI. Kinetics of Curcumin Degradation in Aqueous Solution. Z. Für Lebensm. Unters. Forsch. 1985, 180, 402–404. [Google Scholar] [CrossRef]
- Zhang, R.; McClements, D.J. Enhancing Nutraceutical Bioavailability by Controlling the Composition and Structure of Gastrointestinal Contents: Emulsion-Based Delivery and Excipient Systems. Food Struct. 2016, 10, 21–36. [Google Scholar] [CrossRef]
- Jagannathan, R.; Abraham, P.M.; Poddar, P. Temperature-Dependent Spectroscopic Evidences of Curcumin in Aqueous Medium: A Mechanistic Study of Its Solubility and Stability. J. Phys. Chem. B 2012, 116, 14533–14540. [Google Scholar] [CrossRef]
- Araiza-Calahorra, A.; Akhtar, M.; Sarkar, A. Recent Advances in Emulsion-Based Delivery Approaches for Curcumin: From Encapsulation to Bioaccessibility. Trends Food Sci. Technol. 2018, 71, 155–169. [Google Scholar] [CrossRef]
- Yao, M.; Xiao, H.; McClements, D.J. Delivery of Lipophilic Bioactives: Assembly, Disassembly, and Reassembly of Lipid Nanoparticles. Annu. Rev. Food Sci. Technol. 2014, 5, 53–81. [Google Scholar] [CrossRef]
- Jiao, J.; Rhodes, D.G.; Burgess, D.J. Multiple Emulsion Stability: Pressure Balance and Interfacial Film Strength. J. Colloid Interface Sci. 2002, 250, 444–450. [Google Scholar] [CrossRef]
- Pinheiro, A.C.; Lad, M.; Silva, H.D.; Coimbra, M.A.; Boland, M.; Vicente, A.A. Unravelling the Behaviour of Curcumin Nanoemulsions During In Vitro Digestion: Effect of the Surface Charge. Soft Matter 2013, 9, 3147–3154. [Google Scholar] [CrossRef]
- Lu, X.; Zhu, J.; Pana, Y.; Huang, Q. Assessment of dynamic bioaccessibility of curcumin encapsulated in milled starch particle stabilized Pickering emulsions using TNO’s gastrointestinal model. Food Funct. 2019, 10, 2583–2594. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vellido-Perez, J.A.; Martinez-Ferez, A. In-Vitro Dynamic Bioaccessibility and Bioavailability as Key Parameters for Rational Engineering of Novel Curcumin Delivery Systems. Appl. Sci. 2025, 15, 9417. https://doi.org/10.3390/app15179417
Vellido-Perez JA, Martinez-Ferez A. In-Vitro Dynamic Bioaccessibility and Bioavailability as Key Parameters for Rational Engineering of Novel Curcumin Delivery Systems. Applied Sciences. 2025; 15(17):9417. https://doi.org/10.3390/app15179417
Chicago/Turabian StyleVellido-Perez, Jose Antonio, and Antonio Martinez-Ferez. 2025. "In-Vitro Dynamic Bioaccessibility and Bioavailability as Key Parameters for Rational Engineering of Novel Curcumin Delivery Systems" Applied Sciences 15, no. 17: 9417. https://doi.org/10.3390/app15179417
APA StyleVellido-Perez, J. A., & Martinez-Ferez, A. (2025). In-Vitro Dynamic Bioaccessibility and Bioavailability as Key Parameters for Rational Engineering of Novel Curcumin Delivery Systems. Applied Sciences, 15(17), 9417. https://doi.org/10.3390/app15179417





