Antioxidant Activity of Panax ginseng to Regulate ROS in Various Chronic Diseases
Abstract
1. Introduction
2. Research Methodology
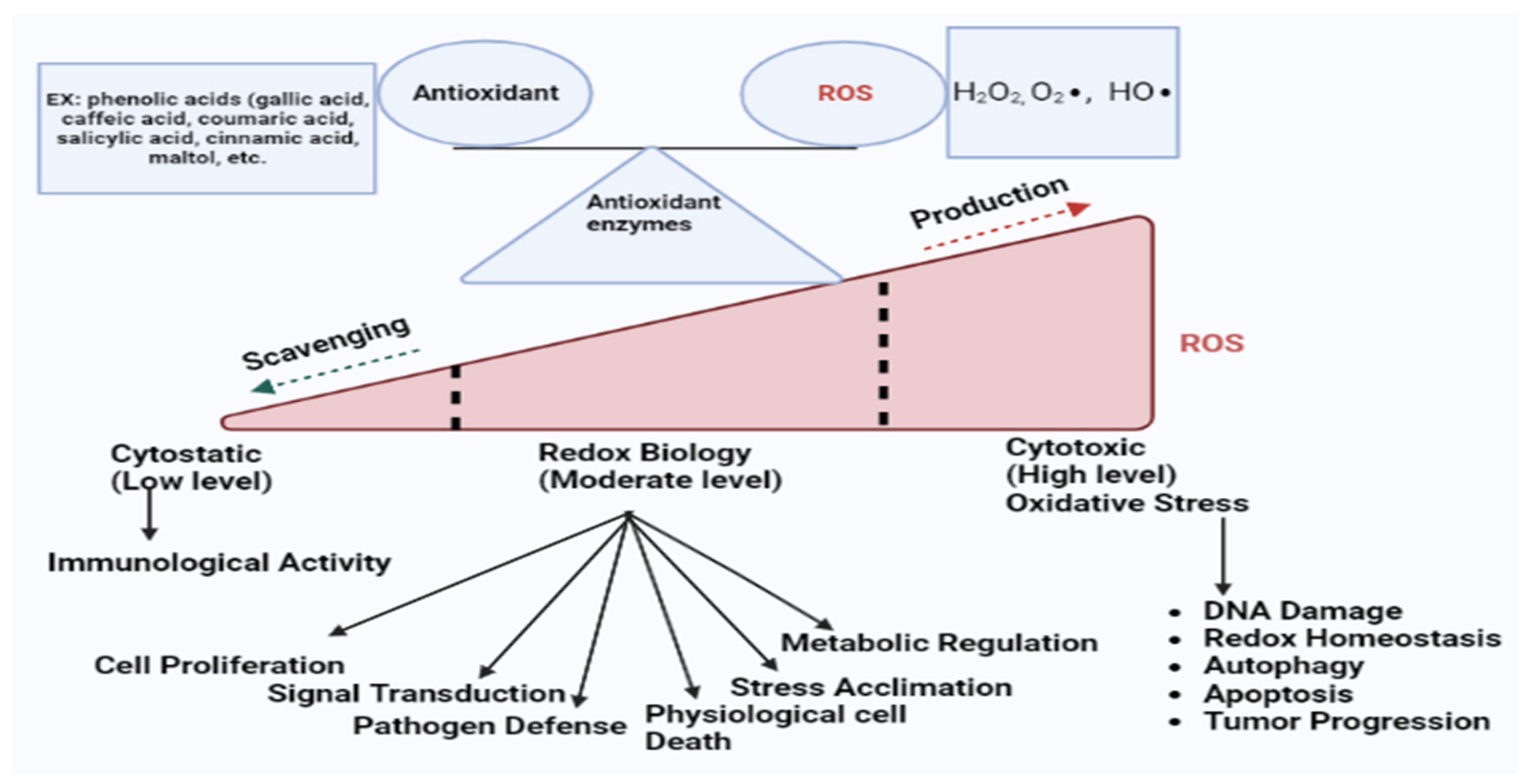
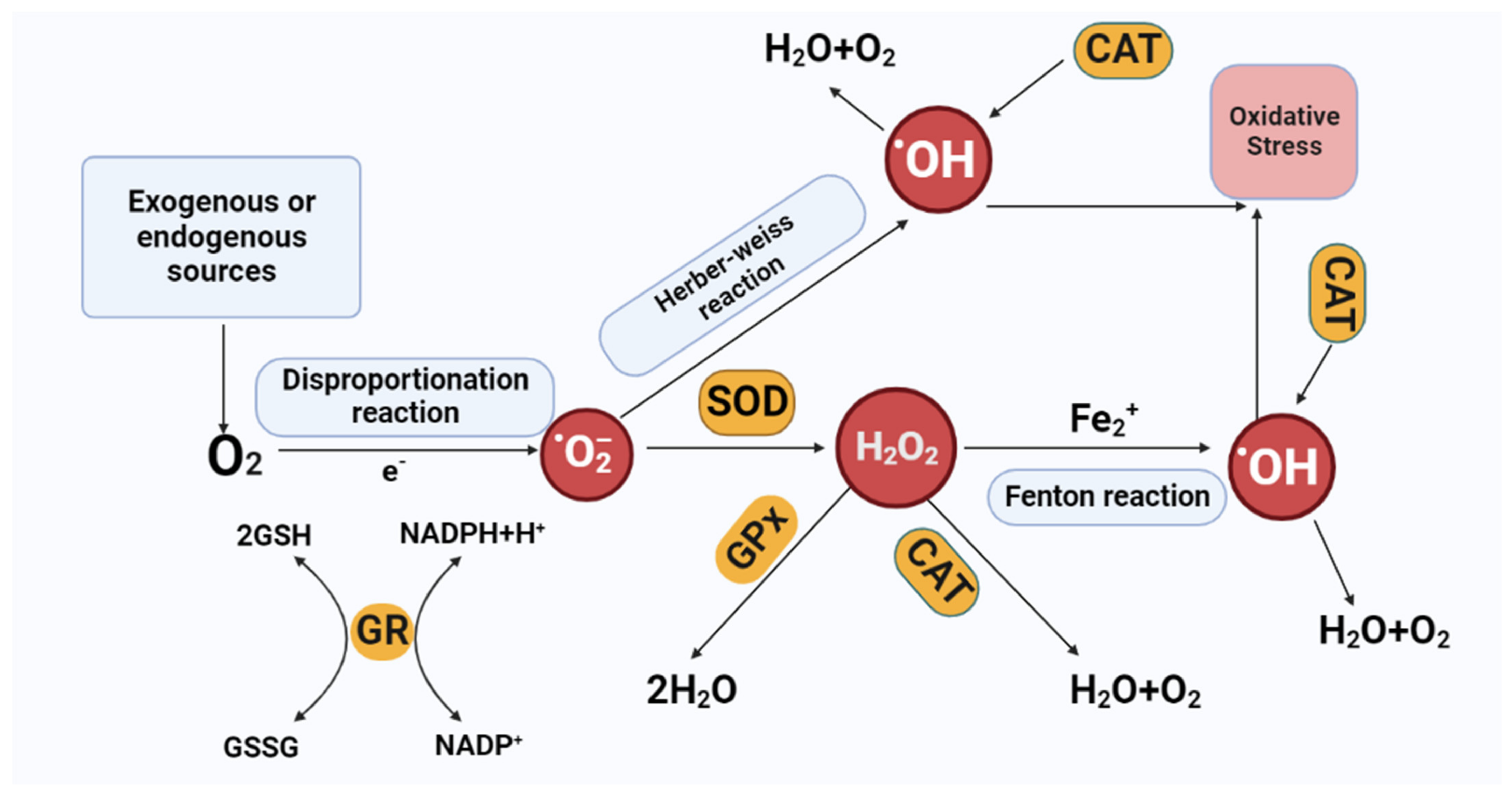
3. ROS, Oxidative Stress, and Antioxidants
4. Pathways Related to Oxidative Stress in Cells
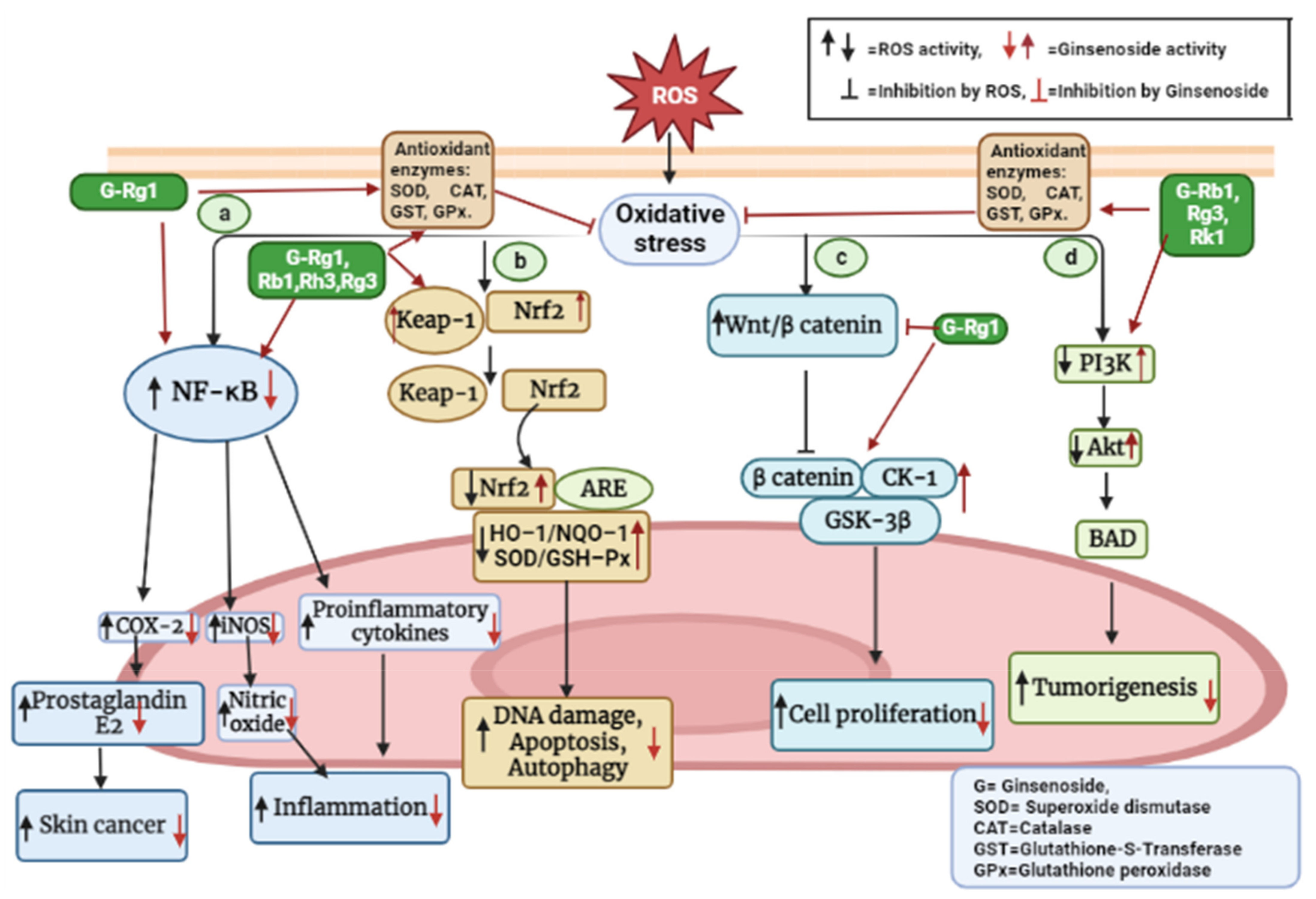
5. Roles of P. ginseng in the Oxidative-Stress-Related Signaling Pathways
6. P. ginseng in ROS-Mediated Diseases
6.1. Antioxidant Activities of P. ginseng in Sensor Impairment
6.1.1. Ototoxicity
6.1.2. Ocular Disease
6.2. Antioxidant Activities of P. ginseng in Neurogenerative Diseases
6.2.1. Parkinson’s Disease
6.2.2. Alzheimer’s Disease
6.2.3. Others
6.3. Antioxidant Activities of P. ginseng in Cardiovascular Diseases
6.4. Antioxidant Activities of P. ginseng in Cancer
6.5. Antioxidant Activities of P. ginseng in Other Diseases
7. Nano-Formulation Forms of Phenolic Compounds as a Solid Alternative in Current Treatment Methods
8. Discussion
9. Conclusion and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Angelova, N.; Kong, H.W.; Van Der Heijden, R.; Yang, S.Y.; Choi, Y.H.; Kim, H.K.; Wang, M.; Hankemeier, T.; Van Der Greef, J.; Xu, G. Recent methodology in the phytochemical analysis of ginseng. Phytochem. Anal. Int. J. Plant Chem. Biochem. Tech. 2008, 19, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.-S.; Park, H.-S.; Park, J.-W.; Li, S.-H.; Chun, Y.-S. Red ginseng and 20(S)-Rg3 control testosterone-induced prostate hy-perplasia by deregulating androgen receptor signaling. J. Nat. Med. 2012, 66, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Nocerino, E.; Amato, M.; Izzo, A.A. The aphrodisiac and adaptogenic properties of ginseng. Fitoterapia 2000, 71, S1–S5. [Google Scholar] [CrossRef]
- Metwaly, A.M.; Lianlian, Z.; Luqi, H.; Deqiang, D. Black ginseng and its saponins: Preparation, phytochemistry and pharmacological effects. Molecules 2019, 24, 1856. [Google Scholar] [CrossRef]
- Abbas, M.; Haq, M.; Anwar, F. Pharmaceutical and Medicinal Applications of Panax Ginseng and Ginsenosides and Their Theuropatic Role in Different. Nat. Volatiles Essent. Oils 2022, 9, 745–765. [Google Scholar]
- Kwak, G.-Y.; Ying, Z.; Awais, M.; Akter, R.; Xu, F.; Baik, S.; Jung, D.; Yang, D.C.; Wenying, Y. Discrimination of Panax ginseng from counterfeits using single nucleotide polymorphism: A focused review. Front. Plant Sci. 2022, 2116, 903306. [Google Scholar]
- Adil, M.; Jeong, B.R. In vitro cultivation of Panax ginseng CA Meyer. Ind. Crops Prod. 2018, 122, 239–251. [Google Scholar] [CrossRef]
- Chung, I.-M.; Lim, J.-J.; Ahn, M.-S.; Jeong, H.-N.; An, T.-J.; Kim, S.-H. Comparative phenolic compound profiles and antiox-idative activity of the fruit, leaves, and roots of Korean ginseng (Panax ginseng Meyer) according to cultivation years. J. Ginseng Res. 2016, 40, 68–75. [Google Scholar]
- Buettner, C.; Yeh, G.Y.; Phillips, R.S.; Mittleman, M.A.; Kaptchuk, T.J. Systematic review of the effects of ginseng on cardio-vascular risk factors. Ann. Pharmacother. 2006, 40, 83–95. [Google Scholar] [PubMed]
- Mao, Q.; Zhang, P.-H.; Wang, Q.; Li, S.-L. Ginsenoside F2 induces apoptosis in humor gastric carcinoma cells through reactive oxygen species-mitochondria pathway and modulation of ASK-1/JNK signaling cascade in vitro and in vivo. Phytomedicine 2014, 21, 515–522. [Google Scholar] [CrossRef]
- Li, L.; Wang, Y.; Guo, R.; Li, S.; Ni, J.; Gao, S.; Gao, X.; Mao, J.; Zhu, Y.; Wu, P.; et al. Ginsenoside Rg3-loaded, reactive oxygen species-responsive polymeric nanoparticles for alleviating myocardial ischemia-reperfusion injury. J. Control. Release 2020, 317, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.-S.; Song, J.H.; Choi, P.; Lee, J.H.; Kim, S.-Y.; Shin, K.-S.; Ham, J.; Kang, K.S. Stimulation of innate immune function by Panax ginseng after heat processing. J. Agric. Food Chem. 2018, 66, 4652–4659. [Google Scholar] [CrossRef]
- Zeng, D.; Wang, J.; Kong, P.; Chang, C.; Li, J.; Li, J. Ginsenoside Rg3 inhibits HIF-1α and VEGF expression in patient with acute leukemia via inhibiting the activation of PI3K/Akt and ERK1/2 pathways. Int. J. Clin. Exp. Pathol. 2014, 7, 2172–2178. [Google Scholar] [PubMed]
- Chung, K.-S.; Cho, S.-H.; Shin, J.-S.; Kim, D.-H.; Choi, J.-H.; Choi, S.Y.; Rhee, Y.K.; Hong, H.-D.; Lee, K.-T. Ginsenoside Rh2 induces cell cycle arrest and differentiation in human leukemia cells by upregulating TGF-β expression. Carcinogenesis 2012, 34, 331–340. [Google Scholar] [CrossRef]
- He, Y.; Hu, Z.; Li, A.; Zhu, Z.; Yang, N.; Ying, Z.; He, J.; Wang, C.; Yin, S.; Cheng, S. Recent advances in biotransformation of saponins. Molecules 2019, 24, 2365. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.-W.; Wang, H.-Y.; Zhang, H.; Wang, C.-Z.; Li, P.; Yuan, C.-S. Diagnostic ion filtering to characterize ginseng saponins by rapid liquid chromatography with time-of-flight mass spectrometry. J. Chromatogr. A 2012, 1230, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, A.; Kim, J.H.; Kim, J.-H.; Yi, Y.-S.; Cho, J.Y. Functional role of ginseng-derived compounds in cancer. J. Ginseng Res. 2018, 42, 248–254. [Google Scholar] [CrossRef]
- Jin, Y.; Huynh, D.T.N.; Nguyen, T.L.L.; Jeon, H.; Heo, K.-S. Therapeutic effects of ginsenosides on breast cancer growth and metastasis. Arch. Pharmacal Res. 2020, 43, 773–787. [Google Scholar] [CrossRef]
- Chae, S.; Kang, K.A.; Youn, U.; Park, J.S.; Hyun, J.W. A comparative study of the potential antioxidant activities of ginsenosides. J. Food Biochem. 2010, 34, 31–43. [Google Scholar] [CrossRef]
- He, B.; Chen, D.; Zhang, X.; Yang, R.; Yang, Y.; Chen, P.; Shen, Z. Oxidative Stress and Ginsenosides: An Update on the Molecular Mechanisms. Oxidative Med. Cell. Longev. 2022, 2022, 9299574. [Google Scholar] [CrossRef] [PubMed]
- Dröge, W. The plasma redox state and ageing. Ageing Res. Rev. 2002, 1, 257–278. [Google Scholar] [CrossRef]
- Trachootham, D.; Alexandre, J.; Huang, P. Targeting cancer cells by ROS-mediated mechanisms: A radical therapeutic ap-proach? Nat. Rev. Drug Discov. 2009, 8, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Bindu, M.; Annamalai, P. Combined Effect of Alcohol and Cigarette Smoke on Lipid Peroxidation and Antioxidant Status in Rats; CSIR: New Delhi, India, 2004; pp. 40–44. [Google Scholar]
- Chan, L.Y.; Kwok, H.H.; Chan, R.W.Y.; Peiris, M.J.S.; Mak, N.K.; Wong, R.N.S.; Chan, M.C.W.; Yue, P.Y.K. Dual functions of ginsenosides in protecting human endothelial cells against influenza H9N2-induced inflammation and apoptosis. J. Ethnopharmacol. 2011, 137, 1542–1546. [Google Scholar] [CrossRef]
- Khansari, N.; Shakiba, Y.; Mahmoudi, M. Chronic inflammation and oxidative stress as a major cause of age-related diseases and cancer. Recent Pat. Inflamm. Allergy Drug Discov. 2009, 3, 73–80. [Google Scholar] [CrossRef]
- Luo, Z.; Xu, X.; Sho, T.; Zhang, J.; Xu, W.; Yao, J.; Xu, J. ROS-induced autophagy regulates porcine trophectoderm cell apoptosis, proliferation, and differentiation. Am. J. Physiol. Cell Physiol. 2019, 316, C198–C209. [Google Scholar] [CrossRef] [PubMed]
- Devasagayam, T.P.; Steenken, S.; Obendorf, M.S.; Schulz, W.A.; Sies, H. Formation of 8-hydroxy (deoxy) guanosine and gen-eration of strand breaks at guanine residues in DNA by singlet oxygen. Biochemistry 1991, 30, 6283–6289. [Google Scholar] [CrossRef] [PubMed]
- Gutteridge, J.M.; Halliwell, B. Invited review free radicals in disease processes: A compilation of cause and consequence. Free. Radic. Res. Commun. 1993, 19, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Reactive oxygen species and the central nervous system. J. Neurochem. 1992, 59, 1609–1623. [Google Scholar] [CrossRef]
- Bhatia, V.; Tandon, R.K. Stress and the gastrointestinal tract. J. Gastroenterol. Hepatol. 2005, 20, 332–339. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 2015, 97, 55–74. [Google Scholar] [CrossRef]
- Alzoghaibi, M.A. Concepts of oxidative stress and antioxidant defense in Crohn’s disease. World J. Gastroenterol. WJG 2013, 19, 6540. [Google Scholar] [CrossRef]
- Pammi, S.S.S.; Suresh, B.; Giri, A. Antioxidant potential of medicinal plants. J. Crop Sci. Biotechnol. 2023, 26, 13–26. [Google Scholar] [CrossRef]
- Duarte, T.L.; Lunec, J. Review: When is an antioxidant not an antioxidant? A review of novel actions and reactions of vitamin C. Free Radic Res. 2005, 39, 671–686. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.C.; Zheng, G.J.; Li, A.M.; Richardson, B.J.; Lam, P.K. Relationships between tissue concentrations of polycyclic aromatic hydrocarbons and antioxidative responses of marine mussels, Perna viridis. Aquat. Toxicol. 2001, 52, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Biochemistry of oxidative stress. Biochem. Soc. Trans. 2007, 35, 1147–1150. [Google Scholar] [CrossRef]
- Kehrer, J.P.; Klotz, L.-O. Free radicals and related reactive species as mediators of tissue injury and disease: Implications for health. Crit. Rev. Toxicol. 2015, 45, 765–798. [Google Scholar] [CrossRef]
- Halliwell, B. Free radicals and antioxidants: Updating a personal view. Nutr. Rev. 2012, 70, 257–265. [Google Scholar] [CrossRef]
- Adwas, A.A.; Elsayed, A.; Azab, A.; Quwaydir, F. Oxidative stress and antioxidant mechanisms in human body. J. Appl. Bio-Technol. Bioeng. 2019, 6, 43–47. [Google Scholar]
- Ji, X.; Hou, C.; Shi, M.; Yan, Y.; Liu, Y. An insight into the research concerning Panax ginseng CA Meyer polysaccharides: A review. Food Rev. Int. 2022, 38, 1149–1165. [Google Scholar] [CrossRef]
- Azab, A.E.; Albasha, M.O.; Elsayed, A.S.I. Prevention of nephropathy by some natural sources of antioxidants. Yangtze Med. 2017, 1, 235. [Google Scholar] [CrossRef]
- Bassères, D.S.; Baldwin, A.S. Nuclear factor-κB and inhibitor of κB kinase pathways in oncogenic initiation and progression. Oncogene 2006, 25, 6817–6830. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.R.; Kim, D.H.; Kim, S.R.; An, H.J.; Lee, E.K.; Tanaka, T.; Kim, N.D.; Yokozawa, T.; Park, J.N.; Chung, H.Y. Anti-wrinkle effect of magnesium lithospermate B from Salvia miltiorrhiza BUNGE: Inhibition of MMPs via NF-κB signaling. PLoS ONE 2014, 9, e102689. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-K.; Kim, Y.; Na, K.-M.; Surh, Y.-J.; Kim, T.-Y. [6]-Gingerol prevents UVB-induced ROS production and COX-2 expression in vitro and in vivo. Free. Radic. Res. 2007, 41, 603–614. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Wen, X.; Hao, D.; Zhang, N.; He, G.; Jiang, X. NF-κB signaling in skin aging. Mech. Ageing Dev. 2019, 184, 111160. [Google Scholar] [CrossRef] [PubMed]
- Loboda, A.; Damulewicz, M.; Pyza, E.; Jozkowicz, A.; Dulak, J. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: An evolutionarily conserved mechanism. Cell. Mol. Life Sci. 2016, 73, 3221–3247. [Google Scholar] [CrossRef]
- Dai, X.; Yan, X.; Wintergerst, K.A.; Cai, L.; Keller, B.B.; Tan, Y. Nrf2: Redox and Metabolic Regulator of Stem Cell State and Function. Trends Mol. Med. 2020, 26, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Kensler, T.W.; Motohashi, H. The KEAP1-NRF2 system: A thiol-based sensor-effector apparatus for main-taining redox homeostasis. Physiol. Rev. 2018, 98, 1169–1203. [Google Scholar] [CrossRef]
- Katoh, M.; Katoh, M. WNT signaling pathway and stem cell signaling network. Clin. Cancer Res. 2007, 13, 4042–4045. [Google Scholar] [CrossRef]
- Kahn, M. Can we safely target the WNT pathway? Nat. Rev. Drug Discov. 2014, 13, 513–532. [Google Scholar] [CrossRef]
- Xie, Y.; Shi, X.; Sheng, K.; Han, G.; Li, W.; Zhao, Q.; Jiang, B.; Feng, J.; Li, J.; Gu, Y. PI3K/Akt signaling transduction pathway, erythropoiesis and glycolysis in hypoxia. Mol. Med. Rep. 2019, 19, 783–791. [Google Scholar] [CrossRef]
- Wen, Z.; Hou, W.; Wu, W.; Zhao, Y.; Dong, X.; Bai, X.; Peng, L.; Song, L. 6-O-galloylpaeoniflorin attenuates cerebral ischemia reperfusion-induced neuroinflammation and oxidative stress via PI3K/Akt/Nrf2 activation. Oxidative Med. Cell. Longev. 2018, 2018, 8678267. [Google Scholar] [CrossRef]
- Zu, G.; Guo, J.; Che, N.; Zhou, T.; Zhang, X. Protective effects of ginsenoside Rg1 on intestinal ischemia/reperfusion inju-ry-induced oxidative stress and apoptosis via activation of the Wnt/β-catenin pathway. Sci. Rep. 2016, 6, 38480. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Wang, X.; Tang, Y.; Huang, S.; Hu, C.-A.A.; Teng, Y. FGF19/FGFR4 signaling contributes to the resistance of hepato-cellular carcinoma to sorafenib. J. Exp. Clin. Cancer Res. 2017, 36, 8. [Google Scholar] [CrossRef]
- Zeng, X.; Li, J.; Li, Z. Ginsenoside Rd mitigates myocardial ischemia-reperfusion injury via Nrf2/HO-1 signaling pathway. Int. J. Clin. Exp. Med. 2015, 8, 14497. [Google Scholar]
- Dong, C.; Liu, P.; Wang, H.; Dong, M.; Li, G.; Li, Y. Ginsenoside Rb1 attenuates diabetic retinopathy in streptozotocin-induced diabetic rats. Acta Cirúrgica Bras. 2019, 34. [Google Scholar] [CrossRef]
- Liu, X.; Gu, X.; Yu, M.; Zi, Y.; Yu, H.; Wang, Y.; Xie, Y.; Xiang, L. Effects of ginsenoside Rb1 on oxidative stress injury in rat spinal cords by regulating the eNOS/Nrf2/HO-1 signaling pathway. Exp. Ther. Med. 2018, 16, 1079–1086. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yang, K.; Mao, R.; Zhong, D.; Xu, Z.; Xu, J.; Xiong, M. Ginsenoside Rg1 inhibits oxidative stress and inflammation in rats with spinal cord injury via Nrf2/HO-1 signaling pathway. Neuroreport 2021, 33, 81–89. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Y.; Zhang, M.; Gao, Y.; Yan, Z. Protective Effects of Ginsenosides (20R)-Rg3 on H2O2-Induced Myocardial Cell Injury by Activating Keap-1/Nrf2/HO-1 Signaling Pathway. Chem. Biodivers. 2021, 18, e2001007. [Google Scholar] [CrossRef]
- Wang, X.; Chen, L.; Wang, T.; Jiang, X.; Zhang, H.; Li, P.; Lv, B.; Gao, X. Ginsenoside Rg3 antagonizes adriamycin-induced cardiotoxicity by improving endothelial dysfunction from oxidative stress via upregulating the Nrf2-ARE pathway through the activation of akt. Phytomedicine 2015, 22, 875–884. [Google Scholar] [CrossRef]
- Song, W.; Wei, L.; Du, Y.; Wang, Y.; Jiang, S. Protective effect of ginsenoside metabolite compound K against diabetic nephropathy by inhibiting NLRP3 inflammasome activation and NF-κB/p38 signaling pathway in high-fat di-et/streptozotocin-induced diabetic mice. Int. Immunopharmacol. 2018, 63, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Ma, Q.; Fan, C.; Chen, X.; Zhang, H.; Tang, M. Ginsenosides Rb1 and Rg1 protect primary cultured astrocytes against oxygen-glucose deprivation/reoxygenation-induced injury via improving mitochondrial function. Int. J. Mol. Sci. 2019, 20, 6086. [Google Scholar] [CrossRef]
- Huang, W.-C.; Huang, T.-H.; Yeh, K.-W.; Chen, Y.-L.; Shen, S.-C.; Liou, C.-J. Ginsenoside Rg3 ameliorates allergic airway inflammation and oxidative stress in mice. J. Ginseng Res. 2021, 45, 654–664. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.-M.; Cao, Y.-L.; Dou, D.-Q. Protective Effect of Ginsenoside-Re against Cerebral Ischemia/Reperfusion Damage in Rats. Biol. Pharm. Bull. 2006, 29, 2502–2505. [Google Scholar] [CrossRef]
- Liu, L.; Vollmer, M.K.; Ahmad, A.S.; Fernandez, V.M.; Kim, H.; Doré, S. Pretreatment with Korean red ginseng or dimethyl fumarate attenuates reactive gliosis and confers sustained neuroprotection against cerebral hypoxic-ischemic damage by an Nrf2-dependent mechanism. Free Radic. Biol. Med. 2019, 131, 98–114. [Google Scholar] [CrossRef]
- Sohn, S.-H.; Kim, S.-K.; Kim, Y.-O.; Kim, H.-D.; Shin, Y.-S.; Yang, S.-O.; Kim, S.-Y.; Lee, S.-W. A comparison of antioxidant activity of Korean White and Red Ginsengs on H2O2-induced oxidative stress in HepG2 hepatoma cells. J. Ginseng Res. 2013, 37, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-H.; Jang, J.-H.; Chen, C.-Y.; Na, H.-K.; Surh, Y.-J. A formulated red ginseng extract rescues PC12 cells from PCB-induced oxidative cell death through Nrf2-mediated upregulation of heme oxygenase-1 and glutamate cysteine ligase. Toxicology 2010, 278, 131–139. [Google Scholar] [CrossRef]
- Sharma, J.; Goyal, P.K. Chemoprevention of chemical-induced skin cancer by Panax ginseng root extract. J. Ginseng Res. 2015, 39, 265–273. [Google Scholar] [CrossRef]
- Kim, H.-J.; Lee, S.-G.; Chae, I.-G.; Kim, M.-J.; Im, N.-K.; Yu, M.-H.; Lee, E.-J.; Lee, I.-S. Antioxidant effects of fermented red ginseng extracts in streptozotocin-induced diabetic rats. J. Ginseng Res. 2011, 35, 129. [Google Scholar] [CrossRef]
- Ramesh, T.; Kim, S.-W.; Hwang, S.-Y.; Sohn, S.-H.; Yoo, S.-K.; Kim, S.-K. Panax ginseng reduces oxidative stress and restores antioxidant capacity in aged rats. Nutr. Res. 2012, 32, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Lee, J. Antioxidant effects of Korean red ginseng extracts on the glutathione and lipid peroxidation in the liver of mouse treated with paraquat. Korean J. Biomed. Lab. Sci. 2000, 6, 45–53. [Google Scholar]
- Yokozawa, T.; Liu, Z.W.; Dong, E. A study of ginsenoside-Rd in a renal ischemia-reperfusion model. Nephron 1998, 78, 201–206. [Google Scholar] [CrossRef]
- Murphy, M.P.; Hartley, R.C. Mitochondria as a therapeutic target for common pathologies. Nat. Rev. Drug Discov. 2018, 17, 865–886. [Google Scholar] [CrossRef]
- Huang, Q.; Gao, S.; Zhao, D.; Li, X. Review of ginsenosides targeting mitochondrial function to treat multiple disorders: Current status and perspectives. J. Ginseng Res. 2021, 45, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Pak, J.H.; Kim, Y.; Yi, J.; Chung, J.W. Antioxidant therapy against oxidative damage of the inner ear: Protection and precon-ditioning. Antioxidants 2020, 9, 1076. [Google Scholar] [CrossRef] [PubMed]
- Choung, Y.H.; Kim, S.W.; Tian, C.; Min, J.Y.; Lee, H.K.; Park, S.N.; Lee, J.B.; Park, K. Korean red ginseng prevents gentamicin-induced hearing loss in rats. Laryngoscope 2011, 121, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, D.; Jiang, H.-Y.; Schacht, J.; Miller, J.M. Delayed production of free radicals following noise exposure. Brain Res. 2004, 1019, 201–209. [Google Scholar] [CrossRef]
- Hong, B.N.; Kim, S.Y.; Yi, T.H.; Kang, T.H. Post-exposure treatment with ginsenoside compound K ameliorates auditory functional injury associated with noise-induced hearing loss in mice. Neurosci. Lett. 2011, 487, 217–222. [Google Scholar] [CrossRef]
- Im, G.J.; Chang, J.W.; Choi, J.; Chae, S.W.; Ko, E.J.; Jung, H.H. Protective effect of Korean red ginseng extract on cisplatin ototoxicity in HEI-OC1 auditory cells. Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2010, 24, 614–621. [Google Scholar] [CrossRef]
- Moreau, K.L.; King, J.A. Protein misfolding and aggregation in cataract disease and prospects for prevention. Trends Mol. Med. 2012, 18, 273–282. [Google Scholar] [CrossRef]
- Truscott, R.J.W.; Friedrich, M.G. The etiology of human age-related cataract. Proteins don’t last forever. Biochim. Biophys. Acta BBA Gen. Subj. 2016, 1860, 192–198. [Google Scholar] [CrossRef]
- Park, C.; Cha, H.-J.; Song, K.-S.; Kim, H.-S.; Bang, E.; Lee, H.; Jin, C.-Y.; Kim, G.-Y.; Choi, Y.H. Nrf2-mediated activation of HO-1 is required in the blocking effect of compound K, a ginseng saponin metabolite, against oxidative stress damage in ARPE-19 human retinal pigment epithelial cells. J. Ginseng Res. 2022, in press. [Google Scholar] [CrossRef]
- Quigley, H.A.; Broman, A.T. The number of people with glaucoma worldwide in 2010 and 2020. Br. J. Ophthalmol. 2006, 90, 262–267. [Google Scholar] [CrossRef]
- Sim, R.H.; Sirasanagandla, S.R.; Das, S.; Teoh, S.L. Treatment of Glaucoma with Natural Products and Their Mechanism of Action: An Update. Nutrients 2022, 14, 534. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, J.; Huang, W.; Zeng, Z.; Yang, Y.; Zhu, B. Ginsenoside Rb1 protects rat retinal ganglion cells against hypoxia and oxidative stress. Mol. Med. Rep. 2013, 8, 1397–1403. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Yang, H.; Kim, J.Y.; Choi, W.; Seong, G.J.; Kim, C.Y.; Lee, J.M.; Bae, H.W. Effect of red ginseng on visual function and vision-related quality of life in patients with glaucoma. J. Ginseng Res. 2021, 45, 676–682. [Google Scholar] [CrossRef]
- Bae, H.W.; Kim, J.H.; Kim, S.; Kim, M.; Lee, N.; Hong, S.; Seong, G.J.; Kim, C.Y. Effect of Korean Red Ginseng supplementation on dry eye syndrome in glaucoma patients–A randomized, double-blind, placebo-controlled study. J. Ginseng Res. 2015, 39, 7–13. [Google Scholar] [CrossRef]
- Sayre, L.M.; Perry, G.; Smith, M.A. Oxidative Stress and Neurotoxicity. Chem. Res. Toxicol. 2008, 21, 172–188. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; O, W.; Li, W.; Jiang, Z.G.; Ghanbari, H.A. Oxidative stress and neurodegenerative disorders. Int. J. Mol. Sci. 2013, 14, 24438–24475. [Google Scholar] [CrossRef]
- Braak, H.; Del Tredici, K.; Rüb, U.; de Vos, R.A.; Jansen Steur, E.N.; Braak, E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol. Aging 2003, 24, 197–211. [Google Scholar] [CrossRef]
- Kang, M.H.; Ji, Y.-J.; Han, Y.M.; Jang, G.Y.; Kim, D.H.; Lee, J.H.; Kim, G.-S.; Choi, S.J.; Kim, H.D. Sasa borealis Ethanol Extract Protects PC12 Neuronal Cells against Oxidative Stress. Appl. Sci. 2022, 12, 6155. [Google Scholar] [CrossRef]
- Schapira, A.H. Molecular and clinical pathways to neuroprotection of dopaminergic drugs in Parkinson disease. Neurology 2009, 72, S44–S50. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Han, R.; Mak, S.; Han, Y. Protection against 1-methyl-4-phenylpyridinium ion (MPP+)-induced apoptosis by water extract of ginseng (Panax ginseng C.A. Meyer) in SH-SY5Y cells. J. Ethnopharmacol. 2011, 135, 34–42. [Google Scholar] [CrossRef]
- Chen, X.C.; Zhou, Y.C.; Chen, Y.; Zhu, Y.G.; Fang, F.; Chen, L.M. Ginsenoside Rg1 reduces MPTP-induced substantia nigra neuron loss by suppressing oxidative stress. Acta Pharmacol. Sin. 2005, 26, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Zhang, Y.X.; Zhang, Z.F. Effect of phosphorylated-ERK1/2 on inducible nitric oxide synthase expression in the sub-stantia nigra of mice with MPTP-induced Parkinson disease. Nan Fang Yi Ke Da Xue Xue Bao 2009, 29, 60–63. [Google Scholar] [PubMed]
- Chen, X.C.; Zhu, Y.G.; Zhu, L.A.; Huang, C.; Chen, Y.; Chen, L.M.; Fang, F.; Zhou, Y.C.; Zhao, C.H. Ginsenoside Rg1 attenuates dopamine-induced apoptosis in PC12 cells by suppressing oxidative stress. Eur. J. Pharmacol. 2003, 473, 1–7. [Google Scholar] [CrossRef]
- Xu, H.; Jiang, H.; Wang, J.; Xie, J. Rg1 protects the MPP+-treated MES23. 5 cells via attenuating DMT1 up-regulation and cellular iron uptake. Neuropharmacology 2010, 58, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.-B.; Liu, C.-Q.; Gao, X.; Zhang, W.-Q.; Wang, S.-W.; Cao, Y.-L. Possible mechanisms of the protection of ginsenoside Re against MPTP-induced apoptosis in substantia nigra neurons of Parkinson’s disease mouse model. J. Asian Nat. Prod. Res. 2005, 7, 215–224. [Google Scholar] [CrossRef]
- Selkoe, D.J.; Schenk, D. Alzheimer’s disease: Molecular understanding predicts amyloid-based therapeutics. Annu. Rev. Pharmacol. Toxicol. 2003, 43, 545. [Google Scholar] [CrossRef]
- Tanzi, R.E.; Bertram, L. The latest suspect. Nature 2008, 454, 707–708. [Google Scholar] [CrossRef]
- Bazan, N.G.; Palacios-Pelaez, R.; Lukiw, W.J. Hypoxia signaling to genes. Mol. Neurobiol. 2002, 26, 283–298. [Google Scholar] [CrossRef]
- Frank, B.; Gupta, S. A review of antioxidants and Alzheimer’s disease. Ann. Clin. Psychiatry 2005, 17, 269–286. [Google Scholar] [CrossRef] [PubMed]
- Finch, C.E.; Morgan, T.E. Systemic inflammation, infection, ApoE alleles, and Alzheimer disease: A position paper. Curr. Alzheimer Res. 2007, 4, 185–189. [Google Scholar] [CrossRef]
- Zhao, H.; Li, Q.; Zhang, Z.; Pei, X.; Wang, J.; Li, Y. Long-term ginsenoside consumption prevents memory loss in aged SAMP8 mice by decreasing oxidative stress and up-regulating the plasticity-related proteins in hippocampus. Brain Res. 2009, 1256, 111–122. [Google Scholar] [CrossRef]
- Qian, Y.-H.; Han, H.; Hu, X.-D.; Shi, L.-L. Protective effect of ginsenoside Rb1 on β-amyloid protein (1-42)-induced neuro-toxicity in cortical neurons. Neurol. Res. 2009, 31, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Wang, H.-T.; Li, C.-L.; Gao, X.-H.; Ding, J.-L.; Zhao, H.-H.; Lu, Y.-L. Ginsenoside Rb1 protects PC12 cells against β-amyloid-induced cell injury. Mol. Med. Rep. 2010, 3, 635–639. [Google Scholar] [PubMed]
- Liu, Q.; Kou, J.-P.; Yu, B.-Y. Ginsenoside Rg1 protects against hydrogen peroxide-induced cell death in PC12 cells via inhibiting NF-κB activation. Neurochem. Int. 2011, 58, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-H.; Du, G.-H. Ginsenoside Rg1 inhibits β-secretase activity in vitro and protects against Aβ-induced cytotoxicity in PC12 cells. J. Asian Nat. Prod. Res. 2009, 11, 604–612. [Google Scholar] [CrossRef] [PubMed]
- She, L.; Xiong, L.; Li, L.; Zhang, J.; Sun, J.; Wu, H.; Ren, J.; Wang, W.; Zhao, X.; Liang, G. Ginsenoside Rk3 ameliorates Aβ-induced neurotoxicity in APP/PS1 model mice via AMPK signaling pathway. Biomed. Pharmacother. 2023, 158, 114192. [Google Scholar] [CrossRef] [PubMed]
- Bouzroud, S.; El Maaiden, E.; Sobeh, M.; Merghoub, N.; Boukcim, H.; Kouisni, L.; El Kharrassi, Y. Biotechnological Approaches to Producing Natural Antioxidants: Anti-Ageing and Skin Longevity Prospects. Int. J. Mol. Sci. 2023, 24, 1397. [Google Scholar] [CrossRef] [PubMed]
- Hou, W.; Wang, Y.; Zheng, P.; Cui, R. Effects of ginseng on neurological disorders. Front. Cell. Neurosci. 2020, 14, 55. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liu, X.; Liu, J.; Cai, E.; Zhao, Y.; Li, H.; Zhang, L.; Li, P.; Gao, Y. Sesquiterpenoids from the root of Panax ginseng attenuates lipopolysaccharide-induced depressive-like behavior through the brain-derived neurotrophic fac-tor/tropomyosin-related kinase B and sirtuin type 1/nuclear factor-κB signaling pathways. J. Agric. Food Chem. 2018, 66, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Kelly, M.G.; Wierzbicki, E.L.; Escober-Nario, I.C.; Vollmer, M.K.; Doré, S. Nrf2 plays an essential role in long-term brain damage and neuroprotection of Korean red ginseng in a permanent cerebral ischemia model. Antioxidants 2019, 8, 273. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Zheng, L.; Lu, S.; Yang, Y. Neuroprotective effects of pretreatment of ginsenoside R b1 on severe cerebral ischemia-induced injuries in aged mice: Involvement of anti-oxidant signaling. Geriatr. Gerontol. Int. 2017, 17, 338–345. [Google Scholar] [CrossRef]
- Kim, J.; Shim, J.; Lee, S.; Cho, W.-H.; Hong, E.; Lee, J.H.; Han, J.-S.; Lee, H.J.; Lee, K.W. Rg3-enriched ginseng extract ameliorates scopolamine-induced learning deficits in mice. BMC Complement. Altern. Med. 2016, 16, 316. [Google Scholar] [CrossRef] [PubMed]
- Garbelli, R.; De Bock, F.; Medici, V.; Rousset, M.; Villani, F.; Boussadia, B.; Arango-Lievano, M.; Jeanneteau, F.; Daneman, R.; Bartolomei, F. PDGFRβ+ cells in human and experimental neuro-vascular dysplasia and seizures. Neuroscience 2015, 306, 18–27. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, Z.; Yu, S.; Zhao, C.; Xu, B.; Liu, R.; Xu, L.; Guo, Y. Neuroprotective Effect of Ginseng Fibrous Root Enzymatic Hydrolysate against Oxidative Stress. Molecules 2022, 27, 7824. [Google Scholar] [CrossRef]
- Kritharides, L.; Brown, A.; Brieger, D.; Ridell, T.; Zeitz, C.; Jeremy, R.; Tonkin, A.; Walsh, W.; White, H. Overview and de-terminants of cardiovascular disease in indigenous populations. Heart Lung Circ. 2010, 19, 337–343. [Google Scholar] [CrossRef]
- Pratt, C. Alternative prevention and treatment of cardiovascular disease, part 2. Prim. Care Clin. Off. Pract. 2010, 37, 339–366. [Google Scholar] [CrossRef] [PubMed]
- Ing, D.J.; Zang, J.; Dzau, V.J.; Webster, K.A.; Bishopric, N.H. Modulation of cytokine-induced cardiac myocyte apoptosis by nitric oxide, Bak, and Bcl-x. Circ. Res. 1999, 84, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Von Harsdorf, R.d.; Li, P.-F.; Dietz, R. Signaling pathways in reactive oxygen species–induced cardiomyocyte apoptosis. Circulation 1999, 99, 2934–2941. [Google Scholar] [CrossRef]
- Kanani, P.M.; Sinkey, C.A.; Browning, R.L.; Allaman, M.; Knapp, H.R.; Haynes, W.G. Role of oxidant stress in endothelial dysfunction produced by experimental hyperhomocyst (e) inemia in humans. Circulation 1999, 100, 1161–1168. [Google Scholar] [CrossRef]
- B Britten, M.; M Zeiher, A.; Schächinger, V. Clinical importance of coronary endothelial vasodilator dysfunction and thera-peutic options. J. Intern. Med. 1999, 245, 315–327. [Google Scholar] [CrossRef]
- Ferrari, R.; Agnoletti, L.; Comini, L.; Gaia, G.; Bachetti, T.; Cargnoni, A.; Ceconi, C.; Curello, S.; Visioli, O. Oxidative stress during myocardial ischaemia and heart failure. Eur. Heart J. 1998, 19, B2–B11. [Google Scholar] [CrossRef] [PubMed]
- Anversa, P.; Cheng, W.; Liu, Y.; Leri, A.; Redaelli, G.; Kajstura, J. Apoptosis and myocardial infarction. Basic Res. Cardiol. 1998, 93, s008–s012. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.H.; Ko, D.; Kim, J.-H. Cardioprotective potential of Korean Red Ginseng extract on isoproterenol-induced cardiac injury in rats. J. Ginseng Res. 2013, 37, 273. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Chai, H.; Lin, P.H.; Lumsden, A.B.; Yao, Q.; Chen, C. Ginsenoside Rb1 blocks homocysteine-induced endothelial dysfunction in porcine coronary arteries. J. Vasc. Surg. 2005, 41, 861–868. [Google Scholar] [CrossRef]
- Xie, J.-T.; Shao, Z.-H.; Vanden Hoek, T.L.; Chang, W.-T.; Li, J.; Mehendale, S.; Wang, C.-Z.; Hsu, C.-W.; Becker, L.B.; Yin, J.-J.; et al. Antioxidant effects of ginsenoside Re in cardiomyocytes. Eur. J. Pharmacol. 2006, 532, 201–207. [Google Scholar] [CrossRef]
- Yi, X.Q.; Li, T.; Wang, J.R.; Wong, V.K.W.; Luo, P.; Wong, I.Y.F.; Jiang, Z.H.; Liu, L.; Zhou, H. Total ginsenosides increase coronary perfusion flow in isolated rat hearts through activation of PI3K/Akt-eNOS signaling. Phytomedicine 2010, 17, 1006–1015. [Google Scholar] [CrossRef]
- Jeon, B.H.; Kim, C.S.; Park, K.S.; Lee, J.W.; Park, J.B.; Kim, K.-J.; Kim, S.H.; Chang, S.J.; Nam, K.Y. Effect of Korea red ginseng on the blood pressure in conscious hypertensive rats. Gen. Pharmacol. Vasc. Syst. 2000, 35, 135–141. [Google Scholar] [CrossRef]
- Qin, N.; Gong, Q.-h.; Wei, L.-w.; Wu, Q.; Huang, X.-n. Total Ginsenosides Inhibit the Right Ventricular Hypertrophy Induced by Monocrotaline in Rats. Biol. Pharm. Bull. 2008, 31, 1530–1535. [Google Scholar] [CrossRef]
- Hong, S.Y.; Kim, J.Y.; Ahn, H.Y.; Shin, J.-H.; Kwon, O. Panax ginseng Extract Rich in Ginsenoside Protopanaxatriol Attenuates Blood Pressure Elevation in Spontaneously Hypertensive Rats by Affecting the Akt-Dependent Phosphorylation of Endo-thelial Nitric Oxide Synthase. J. Agric. Food Chem. 2012, 60, 3086–3091. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Wu, L.; Li, C.R.; Wang, X.W.; Ma, Y.J.; Zhong, Z.y.; Zhao, H.B.; Cui, J.; Xun, S.F.; Huang, X.L. Ginsenoside Rg1 protects rat cardiomyocyte from hypoxia/reoxygenation oxidative injury via antioxidant and intracellular calcium homeostasis. J. Cell. Biochem. 2009, 108, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Tsutsumi, Y.M.; Tsutsumi, R.; Mawatari, K.; Nakaya, Y.; Kinoshita, M.; Tanaka, K.; Oshita, S. Compound K, a metabolite of ginsenosides, induces cardiac protection mediated nitric oxide via Akt/PI3K pathway. Life Sci. 2011, 88, 725–729. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hu, Z.; Sun, B.; Xu, J.; Jiang, J.; Luo, M. Ginsenoside Rg3 attenuates myocardial ischemia/reperfusion injury via Akt/endothelial nitric oxide synthase signaling and the B-cell lymphoma/B-cell lymphoma-associated X protein pathway. Mol. Med. Rep. 2015, 11, 4518–4524. [Google Scholar] [CrossRef]
- Qin, G.-W.; Lu, P.; Peng, L.; Jiang, W. Ginsenoside Rb1 inhibits cardiomyocyte autophagy via PI3K/Akt/mTOR signaling pathway and reduces myocardial ischemia/reperfusion injury. Am. J. Chin. Med. 2021, 49, 1913–1927. [Google Scholar] [CrossRef]
- Fan, Z.-X.; Yang, C.-J.; Li, Y.-H.; Yang, J.; Huang, C.-X. Ginsenoside Rh2 attenuates myocardial ischaemia-reperfusion injury by regulating the Nrf2/HO-1/NLRP3 signalling pathway. Exp. Ther. Med. 2023, 25, 35. [Google Scholar] [CrossRef]
- Sodrul, I.M.; Wang, C.; Chen, X.; Du, J.; Sun, H. Role of ginsenosides in reactive oxygen species-mediated anticancer therapy. Oncotarget 2018, 9, 2931. [Google Scholar] [CrossRef]
- Klaunig, J.E.; Xu, Y.; Isenberg, J.S.; Bachowski, S.; Kolaja, K.L.; Jiang, J.; Stevenson, D.E.; Walborg Jr, E.F. The role of oxidative stress in chemical carcinogenesis. Environ. Health Perspect. 1998, 106, 289–295. [Google Scholar]
- Cairns, R.A.; Harris, I.S.; Mak, T.W. Regulation of cancer cell metabolism. Nat. Rev. Cancer 2011, 11, 85–95. [Google Scholar] [CrossRef]
- Ichijo, H.; Nishida, E.; Irie, K.; Dijke, P.t.; Saitoh, M.; Moriguchi, T.; Takagi, M.; Matsumoto, K.; Miyazono, K.; Gotoh, Y. In-duction of apoptosis by ASK1, a mammalian MAPKKK that activates SAPK/JNK and p38 signaling pathways. Science 1997, 275, 90–94. [Google Scholar] [CrossRef]
- Moon, D.-O.; Kim, M.-O.; Choi, Y.H.; Hyun, J.W.; Chang, W.Y.; Kim, G.-Y. Butein induces G2/M phase arrest and apoptosis in human hepatoma cancer cells through ROS generation. Cancer Lett. 2010, 288, 204–213. [Google Scholar] [CrossRef]
- Gorrini, C.; Harris, I.S.; Mak, T.W. Modulation of oxidative stress as an anticancer strategy. Nat. Rev. Drug Discov. 2013, 12, 931–947. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z. Increased oxidative stress as a selective anticancer therapy. Oxidative Med. Cell. Longev. 2015, 2015, 294303. [Google Scholar] [CrossRef]
- De Giani, A.; Oldani, M.; Forcella, M.; Lasagni, M.; Fusi, P.; Di Gennaro, P. Synergistic Antioxidant Effect of Prebiotic Ginseng Berries Extract and Probiotic Strains on Healthy and Tumoral Colorectal Cell Lines. Int. J. Mol. Sci. 2023, 24, 373. [Google Scholar] [CrossRef] [PubMed]
- Nahar, J.; Boopathi, V.; Murugesan, M.; Rupa, E.J.; Yang, D.C.; Kang, S.C.; Mathiyalagan, R. Investigating the Anticancer Activity of G-Rh1 Using In Silico and In Vitro Studies (A549 Lung Cancer Cells). Molecules 2022, 27, 8311. [Google Scholar] [CrossRef]
- Al-Hussaniy, H.A.; Al-Kuraishy, H.M.; Abdulameer, A.-G.A. The Use of Panax Ginseng to Reduce the Cardiotoxicity of Doxorubicin and Study its Effect on Modulating Oxidative Stress, Inflammatory, and Apoptosis Pathways. Open Access Maced. J. Med. Sci. 2022, 10, 715–719. [Google Scholar] [CrossRef]
- Xia, T.; Wang, Y.N.; Zhou, C.X.; Wu, L.M.; Liu, Y.; Zeng, Q.H.; Zhang, X.L.; Yao, J.H.; Wang, M.; Fang, J.P. Ginsenoside Rh2 and Rg3 inhibit cell proliferation and induce apoptosis by increasing mitochondrial reactive oxygen species in human leukemia Jurkat cells. Mol. Med. Rep. 2017, 15, 3591–3598. [Google Scholar] [CrossRef]
- Xia, T.; Wang, J.; Wang, Y.; Wang, Y.; Cai, J.; Wang, M.; Chen, Q.; Song, J.; Yu, Z.; Huang, W. Inhibition of autophagy potentiates anticancer property of 20 (S)-ginsenoside Rh2 by promoting mitochondria-dependent apoptosis in human acute lympho-blastic leukaemia cells. Oncotarget 2016, 7, 27336. [Google Scholar] [CrossRef] [PubMed]
- Chae, S.; Kang, K.A.; Chang, W.Y.; Kim, M.J.; Lee, S.J.; Lee, Y.S.; Kim, H.S.; Kim, D.H.; Hyun, J.W. Effect of compound K, a metabolite of ginseng saponin, combined with γ-ray radiation in human lung cancer cells in vitro and in vivo. J. Ag-Ricultural Food Chem. 2009, 57, 5777–5782. [Google Scholar] [CrossRef]
- Zhu, C.; Liu, F.; Qian, W.; Zhang, T.; Li, F. Combined effect of sodium selenite and ginsenoside Rh2 on HCT116 human col-orectal carcinoma cells. Arch. Iran. Med. 2016, 19, 23. [Google Scholar]
- Wang, C.-Z.; Li, B.; Wen, X.-D.; Zhang, Z.; Yu, C.; Calway, T.D.; He, T.-C.; Du, W.; Yuan, C.-S. Paraptosis and NF-κB activation are associated with protopanaxadiol-induced cancer chemoprevention. BMC Complement. Altern. Med. 2013, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Meng, Y.; Sun, Q.; Zhang, Z.; Guo, X.; Sheng, X.; Tai, G.; Cheng, H.; Zhou, Y. Ginsenoside compound K sensitizes human colon cancer cells to TRAIL-induced apoptosis via autophagy-dependent and-independent DR5 upregulation. Cell Death Dis. 2016, 7, e2334. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Deng, Z.; Xiong, Z.; Zhang, B.; Yang, J.; Hu, J. A ROS-mediated lysosomal–mitochondrial pathway is induced by ginsenoside Rh2 in hepatoma HepG2 cells. Food Funct. 2015, 6, 3828–3837. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-J.; Yun, H.; Kim, D.-H.; Kang, I.; Choe, W.; Kim, S.-S.; Ha, J. AMP-activated protein kinase determines apoptotic sensitivity of cancer cells to ginsenoside-Rh2. J. Ginseng Res. 2014, 38, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-M.; Kim, S.-J.; Kim, J.-S.; Kang, H.-S. Reactive oxygen species mediated ginsenoside Rg3-and Rh2-induced apoptosis in hepatoma cells through mitochondrial signaling pathways. Food Chem. Toxicol. 2012, 50, 2736–2741. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.D.; Kang, K.A.; Zhang, R.; Lim, C.M.; Kim, H.S.; Kim, D.H.; Jeon, Y.J.; Lee, C.H.; Park, J.; Chang, W.Y. Ginseng saponin metabolite induces apoptosis in MCF-7 breast cancer cells through the modulation of AMP-activated protein kinase. Environ. Toxicol. Pharmacol. 2010, 30, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.-M.; Kim, D.-H.; Park, J.-H.; Na, H.-K.; Surh, Y.-J. Ginsenoside Rg3 induces apoptosis of human breast cancer (MDA-MB-231) cells. J. Cancer Prev. 2013, 18, 177. [Google Scholar] [CrossRef]
- Wang, D.; Markus, J.; Kim, Y.-J.; Wang, C.; Pérez, Z.E.J.; Ahn, S.; Aceituno, V.C.; Mathiyalagan, R.; Yang, D.C. Coalescence of functional gold and monodisperse silver nanoparticles mediated by black Panax ginseng Meyer root extract. Int. J. Nanomed. 2016, 11, 6621. [Google Scholar] [CrossRef]
- Ham, Y.-M.; Lim, J.-H.; Na, H.-K.; Choi, J.-S.; Park, B.-D.; Yim, H.; Lee, S.-K. Ginsenoside-Rh2-induced mitochondrial depo-larization and apoptosis are associated with reactive oxygen species-and Ca2+-mediated c-Jun NH2-terminal kinase 1 activation in HeLa cells. J. Pharmacol. Exp. Ther. 2006, 319, 1276–1285. [Google Scholar] [CrossRef]
- Wang, H.; Jiang, D.; Liu, J.; Ye, S.; Xiao, S.; Wang, W.; Sun, Z.; Xie, Y.; Wang, J. Compound K induces apoptosis of bladder cancer T24 cells via reactive oxygen species-mediated p38 MAPK pathway. Cancer Biother. Radiopharm. 2013, 28, 607–614. [Google Scholar] [CrossRef]
- Zheng, K.; Li, Y.; Wang, S.; Wang, X.; Liao, C.; Hu, X.; Fan, L.; Kang, Q.; Zeng, Y.; Wu, X. Inhibition of autophagosome-lysosome fusion by ginsenoside Ro via the ESR2-NCF1-ROS pathway sensitizes esophageal cancer cells to 5-fluorouracil-induced cell death via the CHEK1-mediated DNA damage checkpoint. Autophagy 2016, 12, 1593–1613. [Google Scholar] [CrossRef]
- Chen, X.-C.; Fang, F.; Zhu, Y.-G.; Chen, L.-M.; Zhou, Y.-C.; Chen, Y. Protective effect of ginsenoside Rg1 on MPP+-induced apoptosis in SHSY5Y cells. J. Neural Transm. 2003, 110, 835–845. [Google Scholar] [CrossRef]
- Choi, K.; Choi, C. Proapoptotic Ginsenosides Compound K and Rh2 Enhance Fas-induced Cell Death of Human Astrocytoma Cells Through Distinct Apoptotic Signaling Pathways. Cancer Res. Treat. 2009, 41, 36–44. [Google Scholar] [CrossRef]
- Sin, S.; Kim, S.Y.; Kim, S.S. Chronic treatment with ginsenoside Rg3 induces Akt-dependent senescence in human glioma cells. Int. J. Oncol. 2012, 41, 1669–1674. [Google Scholar] [CrossRef]
- Choi, Y.J.; Lee, H.J.; Kang, D.W.; Han, I.H.; Choi, B.K.; Cho, W.H. Ginsenoside Rg3 induces apoptosis in the U87MG human glioblastoma cell line through the MEK signaling pathway and reactive oxygen species. Oncol. Rep. 2013, 30, 1362–1370. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhao, J.; Wang, C.-Z.; Searle, J.; He, T.-C.; Yuan, C.-S.; Du, W. Ginsenoside Rh2 induces apoptosis and paraptosis-like cell death in colorectal cancer cells through activation of p53. Cancer Lett. 2011, 301, 185–192. [Google Scholar] [CrossRef]
- Lee, M.; Sorn, S.; Baek, S.; Jang, S.; Kim, S. Antioxidant and apoptotic effects of Korean white ginseng extracted with the same ratio of protopanaxadiol and protopanaxatriol saponins in human hepatoma HepG2 cells. Ann. N. Y. Acad. Sci. 2009, 1171, 217–227. [Google Scholar] [CrossRef]
- Ghafouri-Fard, S.; Balaei, N.; Shoorei, H.; Hasan, S.M.F.; Hussen, B.M.; Talebi, S.F.; Taheri, M.; Ayatollahi, S.A. The effects of Ginsenosides on PI3K/AKT signaling pathway. Mol. Biol. Rep. 2022, 49, 6701–6716. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.-H.; Nahar, J.; Mathiyalagan, R.; Rupa, E.J.; Ramadhania, Z.M.; Han, Y.; Yang, D.-C.; Kang, S.C. A Focused Review on Molecular Signalling Mechanisms of Ginsenosides Anti-Lung Cancer and Anti-inflammatory Activities. Anti Cancer Agents Med. Chem. Former. Curr. Med. Chem. Anti Cancer Agents 2023, 23, 3–14. [Google Scholar]
- Krata, N.; Zagożdżon, R.; Foroncewicz, B.; Mucha, K. Oxidative Stress in Kidney Diseases: The Cause or the Consequence? Arch. Immunol. Ther. Exp. 2018, 66, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Kong, Y.; Zhang, H. Oxidative stress, mitochondrial dysfunction, and aging. J. Signal Transduct. 2012, 2012, 646354. [Google Scholar] [CrossRef] [PubMed]
- Yokozawa, T.; Satoh, A.; Cho, E.J. Ginsenoside-Rd attenuates oxidative damage related to aging in senescence-accelerated mice. J. Pharm. Pharmacol. 2004, 56, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Oh, M.H.; Chung, H.Y.; Yong, H.S.; Kim, K.W.; Oura, H.; Yokozawa, T. Effects of ginsenoside Rb2 on the antioxidants in SAM-R/1 mice. Korean Biochem. J. 1992, 25, 492–497. [Google Scholar]
- Choi, W.; Kim, H.S.; Park, S.H.; Kim, D.; Hong, Y.D.; Kim, J.H.; Cho, J.Y. Syringaresinol derived from Panax ginseng berry attenuates oxidative stress-induced skin aging via autophagy. J. Ginseng Res. 2022, 46, 536–542. [Google Scholar] [CrossRef]
- Li, G.; Yang, L.; Feng, L.; Yang, J.; Li, Y.; An, J.; Li, D.; Xu, Y.; Gao, Y.; Li, J. Syringaresinol protects against type 1 diabetic cardiomyopathy by alleviating inflammation responses, cardiac fibrosis, and oxidative stress. Mol. Nutr. Food Res. 2020, 64, 2000231. [Google Scholar] [CrossRef]
- Jung, C.-H.; Seog, H.-M.; Choi, I.-W.; Choi, H.-D.; Cho, H.-Y. Effects of wild ginseng (Panax ginseng C.A. Meyer) leaves on lipid peroxidation levels and antioxidant enzyme activities in streptozotocin diabetic rats. J. Ethnopharmacol. 2005, 98, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Yokozawa, T.; Wu Liu, Z. The role of ginsenoside-Rd in cisplatin-induced acute renal failure. Ren. Fail. 2000, 22, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Liu, Z.; Wang, X.; Wang, C. Recent advances in panax ginseng CA Meyer as a herb for anti-fatigue: An effects and mechanisms review. Foods 2021, 10, 1030. [Google Scholar] [CrossRef] [PubMed]
- Han, S.Y.; Kim, J.; Kim, E.; Kim, S.H.; Seo, D.B.; Kim, J.-H.; Shin, S.S.; Cho, J.Y. AKT-targeted anti-inflammatory activity of Panax ginseng calyx ethanolic extract. J. Ginseng Res. 2018, 42, 496–503. [Google Scholar] [CrossRef]
- Ning, C.; Gao, X.; Wang, C.; Huo, X.; Liu, Z.; Sun, H.; Yang, X.; Sun, P.; Ma, X.; Meng, Q. Hepatoprotective effect of ginsenoside Rg1 from Panax ginseng on carbon tetrachloride-induced acute liver injury by activating Nrf2 signaling pathway in mice. Environ. Toxicol. 2018, 33, 1050–1060. [Google Scholar] [CrossRef]
- Ramadhania, Z.M.; Yang, D.U.; Moektiwardojo, M.; Han, Y.; Park, J.K.; Rupa, E.J.; Yang, D.C.; Lee, S.J.; Kang, S.C. Enhanced Anti-Skin Aging Effects of Fermented Black Ginseng (Panax ginseng CA Meyer) by Aspergillus niger KHNT-1. Appl. Sci. 2023, 13, 550. [Google Scholar] [CrossRef]
- Lakshmi, T.; Anitha, R.; Geetha, R. Panax ginseng–a universal panacea in the herbal medicine with diverse pharmacological spectrum–a review. Asian J. Pharm. Clin. Res. 2011, 4, 14–18. [Google Scholar]
- Patel, S.; Rauf, A. Adaptogenic herb ginseng (Panax) as medical food: Status quo and future prospects. Biomed. Pharma Cotherapy 2017, 85, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Shahrajabian, M.H.; Sun, W.; Cheng, Q. A review of ginseng species in different regions as a multipurpose herb in traditional Chinese medicine, modern herbology and pharmacological science. J. Med. Plants Res. 2019, 13, 213–226. [Google Scholar]
- Acero, N.; Ortega, T.; Villagrasa, V.; Leon, G.; Muñoz-Mingarro, D.; Castillo, E.; González-Rosende, M.E.; Borrás, S.; Rios, J.L.; Bosch-Morell, F. Phytotherapeutic alternatives for neurodegenerative dementias: Scientific review, discussion and therapeutic proposal. Phytother. Res. 2023. [Google Scholar] [CrossRef]
- Xu, X.-F.; Gao, Y.; Xu, S.-Y.; Liu, H.; Xue, X.; Zhang, Y.; Zhang, H.; Liu, M.-N.; Xiong, H.; Lin, R.-C. Remarkable impact of steam temperature on ginsenosides transformation from fresh ginseng to red ginseng. J. Ginseng Res. 2018, 42, 277–287. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhao, N.; Ye, X.; He, M.; Yang, Y.; Gao, H.; Zhang, X. Rapid discrimination between red and white ginseng based on unique mass-spectrometric features. J. Pharm. Biomed. Anal. 2019, 164, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Wang, P.; Li, T.; Huang, X.; Guo, W.; Yang, Y.; Yan, M.; Zhang, H.; Cai, D.; Jia, X. Self-assembled natural phytochemicals for synergistically antibacterial application from the enlightenment of traditional Chinese medicine combination. Acta Pharm. Sin. B 2020, 10, 1784–1795. [Google Scholar] [CrossRef]
- Watkins, R.; Wu, L.; Zhang, C.; Davis, R.M.; Xu, B. Natural product-based nanomedicine: Recent advances and issues. Interna-Tional J. Nanomed. 2015, 10, 6055. [Google Scholar]
- Qiao, L.; Han, M.; Gao, S.; Shao, X.; Wang, X.; Sun, L.; Fu, X.; Wei, Q. Research progress on nanotechnology for delivery of active ingredients from traditional Chinese medicines. J. Mater. Chem. B 2020, 8, 6333–6351. [Google Scholar] [CrossRef]
- Kong, W.; Wei, J.; Abidi, P.; Lin, M.; Inaba, S.; Li, C.; Wang, Y.; Wang, Z.; Si, S.; Pan, H. Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins. Nat. Med. 2004, 10, 1344–1351. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, X.; Shen, H.; He, Q.; Wu, Z.; Liao, W.; Yuan, M. Application of the nano-drug delivery system in treatment of cardiovascular diseases. Front. Bioeng. Biotechnol. 2020, 7, 489. [Google Scholar] [CrossRef]
- Yu, H.; Teng, L.; Meng, Q.; Li, Y.; Sun, X.; Lu, J.; Lee, R.J.; Teng, L. Development of liposomal Ginsenoside Rg3: Formulation optimization and evaluation of its anticancer effects. Int. J. Pharm. 2013, 450, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.; Huang, L.; Song, Y.; Liu, Z.; Liang, L.; Wang, L.; Wang, T. Preparation and pharmacological effects of minor ginsenoside nanoparticles: A review. Front. Pharmacol. 2022, 13, 974274. [Google Scholar] [CrossRef]
- Wang, X.; Zheng, W.; Shen, Q.; Wang, Y.; Tseng, Y.; Luo, Z.; Wang, X.; Shi, L.; Li, C.; Liu, J. Identification and construction of a novel biomimetic delivery system of paclitaxel and its targeting therapy for cancer. Signal Transduct. Target. Ther. 2021, 6, 33. [Google Scholar] [CrossRef]
- Alippilakkotte, S.; Sreejith, L. Pectin mediated synthesis of curcumin loaded poly(lactic acid) nanocapsules for cancer treat-ment. J. Drug Deliv. Sci. Technol. 2018, 48, 66–74. [Google Scholar] [CrossRef]
- Kumari, M.; Sharma, N.; Manchanda, R.; Gupta, N.; Syed, A.; Bahkali, A.H.; Nimesh, S. PGMD/curcumin nanoparticles for the treatment of breast cancer. Sci. Rep. 2021, 11, 3824. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.; Xie, J.; Zhang, X.; Cai, J.; Xia, S. Polysaccharide-based nanoparticles by chitosan and gum arabic polyelectrolyte complexation as carriers for curcumin. Food Hydrocoll. 2016, 57, 236–245. [Google Scholar] [CrossRef]
- Martínez-Ávila, M.; Guajardo-Flores, D.; Antunes-Ricardo, M. Nanoformulations applied to the delivery of phenolic com-pound. In Phytochemical Nanodelivery Systems as Potential Biopharmaceuticals; Elsevier: Amsterdam, The Netherlands, 2023; pp. 283–325. [Google Scholar]
- Yan, L.; Wang, R.; Wang, H.; Sheng, K.; Liu, C.; Qu, H.; Ma, A.; Zheng, L. Formulation and characterization of chitosan hy-drochloride and carboxymethyl chitosan encapsulated quercetin nanoparticles for controlled applications in foods system and simulated gastrointestinal condition. Food Hydrocoll. 2018, 84, 450–457. [Google Scholar] [CrossRef]
- Choudhary, A.; Kant, V.; Jangir, B.L.; Joshi, V.G. Quercetin loaded chitosan tripolyphosphate nanoparticles accelerated cu-taneous wound healing in Wistar rats. Eur. J. Pharmacol. 2020, 880, 173172. [Google Scholar] [CrossRef]
- Bhatt, P.; Fnu, G.; Bhatia, D.; Shahid, A.; Sutariya, V. Nanodelivery of Resveratrol-Loaded PLGA Nanoparticles for Age-Related Macular Degeneration. AAPS PharmSciTech 2020, 21, 291. [Google Scholar] [CrossRef]
- Saha, M.; Saha, D.R.; Ulhosna, T.; Sharker, S.M.; Shohag, M.H.; Islam, M.S.; Ray, S.K.; Rahman, G.M.S.; Reza, H.M. QbD based development of resveratrol-loaded mucoadhesive lecithin/chitosan nanoparticles for prolonged ocular drug delivery. J. Drug Deliv. Sci. Technol. 2021, 63, 102480. [Google Scholar] [CrossRef]
- Chmielowski, R.A.; Abdelhamid, D.S.; Faig, J.J.; Petersen, L.K.; Gardner, C.R.; Uhrich, K.E.; Joseph, L.B.; Moghe, P.V. Athero-inflammatory nanotherapeutics: Ferulic acid-based poly (anhydride-ester) nanoparticles attenuate foam cell for-mation by regulating macrophage lipogenesis and reactive oxygen species generation. Acta Biomater. 2017, 57, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.U.; Fatima, K.; Aisha, S.; Hamza, B.; Malik, F. Redox balance and autophagy regulation in cancer progression and their therapeutic perspective. Med. Oncol. 2022, 40, 12. [Google Scholar] [CrossRef]
- Chodari, L.; Dilsiz Aytemir, M.; Vahedi, P.; Alipour, M.; Vahed, S.Z.; Khatibi, S.M.H.; Ahmadian, E.; Ardalan, M.; Eftekhari, A. Targeting mitochondrial biogenesis with polyphenol compounds. Oxidative Med. Cell. Longev. 2021, 2021, 4946711. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Wang, X.; Liu, H.; Lv, C.; Lu, J. Structural characterization and antioxidant activity of oligosaccharides from Panax ginseng C. A. Meyer. Int. J. Biol. Macromol. 2020, 150, 737–745. [Google Scholar] [CrossRef]
- Paik, D.J.; Lee, C.H. Review of cases of patient risk associated with ginseng abuse and misuse. J. Ginseng Res. 2015, 39, 89–93. [Google Scholar] [CrossRef]
| Diseases | Ginseng/Ginsenosides | Action | Model | References |
|---|---|---|---|---|
| Intestinal I/R injury | Rg1 | ↓ROS, ↓apoptosis ↓ROS, ↓tunnel positive cell | In vitro In vivo | [52] |
| Adriamycin-induced cardiotoxicity | Rg3 | ↓ROS, ↓MDA, ↑SOD, ↑eNOS | In vitro | [60] |
| Inflammation in asthma | Rg3 | ↑Nrf2, ↑HO-1, ↑SOD, ↑GSH, ↓MDA | In vivo/Mice | [63] |
| Diabetic nephropathy | CK | ↓ROS, ↓IL-1β | In vivo/Mice | [61] |
| Myocardial cell injury | 20(S)-Rg3 and 20(R) | ↓ROS, ↓MDA ↑SOD, ↑GSH-Px, ↑CAT | In vitro | [59] |
| Myocardial ischemia-reperfusion | Rd | ↓CK, ↓LDH, ↑HO-1 | In vivo/Rats | [54] |
| Spinal cord injury | Rb1 | ↓MDA, ↑SOD, ↑CAT, ↑GSH | In vivo/Rats | [57] |
| Rat SCI model | Rg1 | ↓MDA, ↑SOD, ↑GSH | In vivo/Rats | [58] |
| Cerebral ischemia/reperfusion damage | Re | ↑SOD, ↑GSH-Px, ↓MDA, | In vivo/Rats | [64] |
| Mitochondrial oxidation | Rb1, Rg1 | ↑CAT, ↑complexes I-V, ↑ATP ↓ROS | In vivo/Mice | [62] |
| Cerebral hypoxic-ischemic damage | Red ginseng | ↑NQO1, ↑HO1, ↑SOD2, ↑Gpx1, ↓IL-1β, ↓iNOS | In vivo | [65] |
| Hepatic disease | Red ginseng | ↓MDA, ↓DCF, ↑GPx, ↑GR, ↑CAT, ↑SOD | In vitro | [66] |
| Pheochromocytoma | Red ginseng | ↓DCF, ↑GCLC, ↑SOD, ↑CAT ↑Nrf2, ↑HO-1 | In vitro | [67] |
| Skin cancer | Red ginseng | ↑GSH, ↑SOD, ↑CAT, ↑Vit C, ↓TBARS | In vivo/Mice | [68] |
| Diabetes | Red ginseng | ↑GSH, ↑SOD, ↑CAT, ↑GPx, ↑GR, ↓MDA | In vivo/Mice | [69] |
| Aging | Red ginseng | ↓MDA, ↑SOD, ↑CAT, ↑GPx, ↑GR, ↑GST | [70] | |
| Nerve disorder | Red ginseng | ↑GPx, ↓MDA, ↓ROS | In vivo/Mice | [71] |
| Kidney disease | Rd | ↑SOD, ↑CAT, ↑GPx, ↓MDA, ↓urea nitrogen, ↓creatinine | In vivo/Rats | [72] |
| Cancer Cell | Ginseng/Ginsenosides | Mechanisms | Effect | References |
|---|---|---|---|---|
| Leukemia Jurkat cells | Rh2 and Rg3 | ROS↑, MTP↓, caspase-3/9↑, Bax/Bcl-2↑, Cyt C↑ | Stimulated mitochondrial ROS that inhibited cell proliferation and induced apoptosis. | [149] |
| Acute Lymphoblastic leukemia cells | 20(S)-Ginsenoside Rh2 | ROS↑, MTP↓, Cyt C↑, caspase-3/9↑, LC3-I↑, LC3-II↑, Atg5↓, Beclin-1↑ | Generated mitochondrial ROS that inhibited autophagy and induced apoptosis. | [150] |
| Lung cancer NCI-H460 cell | Compound K | ROS↑, MMP↓, caspase-3↑ | Increased γ-ray-induced apoptosis by enhancing intracellular ROS production that reduced MMP and increased caspase-3. | [151] |
| Colorectal cancer HCT-116 | Rh2 | ROS↓, Bax/Bcl-2↑, capase-3↑, G1↓, S ↓ | Reduced the formation of ROS which activated autophagy and antiproliferative effects. | [152] |
| Colorectal cancer | Protopanaxadiol | ROS↑, NF-κB↑ | ROS production activated the NF-ĸB pathway that induced paraptosis. | [153] |
| Colon cancer HCT-116 cells | Compound K | ROS↑, Mcl-1↓, Bcl-2↓, survivin↓, XIAP↓, cFLIP↓, Bax↑, tBid↑, Cyt C↑, LC3-II↑, Atg7↑, JNK↑, ERK↓, p38↓, p53↑, DR5↑, CHOP↑ | Stimulated TRAIL-induced apoptosis by upregulating DR5 in both autophagy in a dependent and dependent manner. | [154] |
| Hepatoma HepG2 cells | Rh2 | Caspase-3/9↑, cytosol Cat B↓, leupeptin (Leu) ↑, MTP↓, Bid↑, tBid↑, Cyt C↓ | Mitochondrial apoptotic and ROS-accumulation- pathway-induced apoptosis. | [155] |
| Hepatoma HepG2 cells | Rh2 | PARP↑, ROS↑, p-p38↑, p-AMPK↑ | Activation of AMPK-mediated ROS generation that induced apoptosis. | [156] |
| Hepatoma Hep3B cells | Rg3 and Rh2 | ROS↑, caspase-3↑, Bcl-2↓, Bax↑, Cyt C↑, MTP↓ | Mitochondria-mediated apoptosis pathway and ROS generation caused apoptosis. | [157] |
| Breast cancer MCF-7 cells | Compound K | MTP↓, AMPK↑, COX-2↓, PGE2↓ | Modulation of the AMPK signaling pathway and ROS production induced apoptosis. | [158] |
| Breast cancer MDA-MB-231 cells | Rg3 | ROS↑, Bax/Bcl-2↓, MTP↓, caspase-3↑, PARP↑ | Apoptosis was caused by the activation of the mitochondrial death pathway. | [159] |
| Breast cancer MCF-7 cells | BG-AuNps and BG-AgNps | ROS↑ | ROS production caused oxidative cell damage and apoptosis. | [160] |
| Cervical carcinoma HeLa cells | Rh2 | ROS↑, MTP↓, Caspase-3↓, JNK1↑, SEK1↑, JNK2↑, c-Jun↑, Smac↑, Bax↑, Ca2+↑ | Initiated apoptosis by generating ROS and Ca2+ which activated SEK1 and JNK1. | [161] |
| Bladder cancer T24 cells | Compound K | Cyt C↑, Bax↑, Bcl- 2↓, p-p38MAPK↑, procaspase-3/9↑, p38↑, ROS↑, glutathione↓ | Induced apoptosis through ROS-mediated P38 MAPK pathway. | [162] |
| Gastric carcinoma SGC7901 cells | F2 | PARP↓, ASK-1↑, JNK↑, Bcl-2↓, Cyt C↑, Caspase-3/9↑ | Induced apoptosis via modulating the ASK-1/JNK signaling cascade and ROS– mitochondria pathway. | [10] |
| Esophageal cancer cells | Ro | ROS↑, CYBB/Nox2↑, LC3B-II↑, ATG7↑, ESR2↓, NCF1↑, SQSTM1/p62↑, CSTB↓ CSTD↓, p-CHEK1↑, EGFR↑, DDIT3↑, ATM↑, ATR↑, BRCA1↑, GFP-LC3B puncta↑, Lysosomal pH↑, autophagic vacuoles↑ | ERSR2-NCF1-ROS signaling pathway suppressed autophagy and decreased CHEK1 degradation which sensitized 5-fluorouracil inducing cell death. | [163] |
| Neuroblastoma SH-SY5Y cell | Rg1 | ROS↓, JNK↑, caspase-3↑ | Inhibited the generation of ROS and activated the JNK pathway to inhibit MPP+-induced apoptosis. | [164] |
| Astrocytoma CRT-MG cells | Compound K, ginsenoside Rh2 | ROS↑, caspase-3↑, Cyt C↑, p-p38↑, Fas↑, MIP↓ | Increased Fas-dependent apoptosis in ROS/caspase/ mitochondrial pathways. | [165] |
| Neuroblastoma SH-SY5Y cell | Ginseng water extract | ROS↑, Bcl-2↓, Bax↑, Cyt C↑, caspase-3↑ | Alleviated oxidative stress in the mitochondrial apoptotic pathway to prevent MPP+-induced apoptosis. | [166] |
| GliomaU87 cells | 20(S)-ginsenoside Rg3 | ROS↑, p21↑, p16↑, p53↑, Akt↑ | Enhanced ROS generation via Akt- and p53/p21-dependent pathways that induced senescence-like growth arrest. | [167] |
| Glioblastoma U87MG cells | Rg3 | Bcl-2↓, Bax↑, pro-caspase3↓, MEK1/2↑, ROS↑ | Induced apoptosis via ROS and MEK pathway. | [168] |
| Lung cancer A549 cells | Rh1 | ↑P53, ↑Bax, ↑caspase 3, ↑caspase 9, ↓Bcl | Intrinsic pathway. | [147] |
| Colorectal carcinoma SW-480 and HCT-116 | Rh2 | Bax↑, Bad ↑, ROS ↑, NF-ĸB↑, Bad↓, Bcl-xL↓, P53↓, Cytosolic vacuolization↓ | ROS/NF-ĸB signaling pathway/P53 and NF-ĸB signaling pathway. (Upregulation of ROS induced apoptosis by activating NF-ĸB and P53.) | [169] |
| Hepatoma HepG2 cells | Korean white ginseng extract | Cyt C↑, c-Jun↑, SAPK↑, MDA↓, Caspase-3↑, Iĸ-b↓, | Apoptosis induction through JNK/NF- ĸB/Cyt C apoptotic pathway and antioxidant activities. | [170] |
| Breast cancer MDA-MB-231 cells | Rg3 | NF-ĸB↓, Bcl-2↓, Akt ↓, IĸB↓, p-ERK↓ | NF-ĸB inhibition via Akt and ERK inactivation. | [171] |
| Lung cancer A549, H1299, and H358 cells | Rg3 | EMT↓, EGFR ↓, MAPK↓, NF-κB↓ | MAPK and NF-κB blocking to induce apoptosis. | [172] |
| Compounds | Nano-Formulations | Benefits | References |
|---|---|---|---|
| Ginsenoside Rg3 | Mixed micelles containing baicalin copolymer P123. G-Rg3 conjugated with nanoparticle. G-Rg3 with CNC nanocomposite. Liposomes of G-Rg3 | Increases drug absorption in cancer cells and boosts baicalin solubility. Inhibits the growth of hepatocellular carcinoma (HCC) and prevents lung metastasis. Enhances antioxidant activity. Antitumor activity is greater than free formulation. Increases uptake, sustains release, bioavailability, and enhances cytotoxicity in cancer cells. | [194,197,198] |
| Ginsenoside Rg5 | Liposomes of G-Rg5 | Inhibits tumor growth, tumor targeting. | [199] |
| Ginsenoside Rh2 | Micelles | Enhances water solubility and drug uptake. Elongates drug retention. | |
| Mesoporous silicas | Excellent biocompatibility to normal cell (Hacat cell). | ||
| Curcumin | PLA polymer, chitosan, gum arabic | Enhances cellular uptake, and antioxidant and anticancer activities. | [200,201] |
| Increases antioxidant activity. | [202] | ||
| Quercetin | PLA | Induces self-life. | [203] |
| Chitosan-CMC | Enhances stability, solubility, and antioxidant activity. | [204] | |
| Chitosan-pluronic F-127-STPP | Wound healing. | [205] | |
| Resveratrol | PLGA-PEG | Improves nonvascular effects. | [206] |
| Lecithin/chitosan | Promotes anti-inflammatory effects. | [207] | |
| Ferulic acid | Ferulic acid polymer nanoparticles | Reduces ROS activity. | [208] |
| Nanoparticles of poly ferulic acid | Improved colon cancer treatment. | [208] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morshed, M.N.; Ahn, J.C.; Mathiyalagan, R.; Rupa, E.J.; Akter, R.; Karim, M.R.; Jung, D.H.; Yang, D.U.; Yang, D.C.; Jung, S.K. Antioxidant Activity of Panax ginseng to Regulate ROS in Various Chronic Diseases. Appl. Sci. 2023, 13, 2893. https://doi.org/10.3390/app13052893
Morshed MN, Ahn JC, Mathiyalagan R, Rupa EJ, Akter R, Karim MR, Jung DH, Yang DU, Yang DC, Jung SK. Antioxidant Activity of Panax ginseng to Regulate ROS in Various Chronic Diseases. Applied Sciences. 2023; 13(5):2893. https://doi.org/10.3390/app13052893
Chicago/Turabian StyleMorshed, Md Niaj, Jong Chan Ahn, Ramya Mathiyalagan, Esrat Jahan Rupa, Reshmi Akter, Md. Rezaul Karim, Dae Hyo Jung, Dong Uk Yang, Deok Chun Yang, and Seok Kyu Jung. 2023. "Antioxidant Activity of Panax ginseng to Regulate ROS in Various Chronic Diseases" Applied Sciences 13, no. 5: 2893. https://doi.org/10.3390/app13052893
APA StyleMorshed, M. N., Ahn, J. C., Mathiyalagan, R., Rupa, E. J., Akter, R., Karim, M. R., Jung, D. H., Yang, D. U., Yang, D. C., & Jung, S. K. (2023). Antioxidant Activity of Panax ginseng to Regulate ROS in Various Chronic Diseases. Applied Sciences, 13(5), 2893. https://doi.org/10.3390/app13052893









