Study of the Ciprofloxacin Adsorption of Activated Carbon Prepared from Mangosteen Peel
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Fabrication Procedure of Activated Carbon Derived from Mangosteen Peel
2.1.2. Investigations on the Physical Properties and Surface Characteristics of Activated Carbon MP and ACMP
2.2. Antibiotic Ciprofloxacin
2.3. CIP Adsorption Experiments
2.4. Physical Parameters of the Activated Carbon
2.4.1. Moisture Determination
2.4.2. Ash Ratio Determination
2.4.3. Density Determination
2.5. Iodine Index of the Coal
2.6. Determination of the Isoelectric Point of the ACMP
3. Results and Discussion
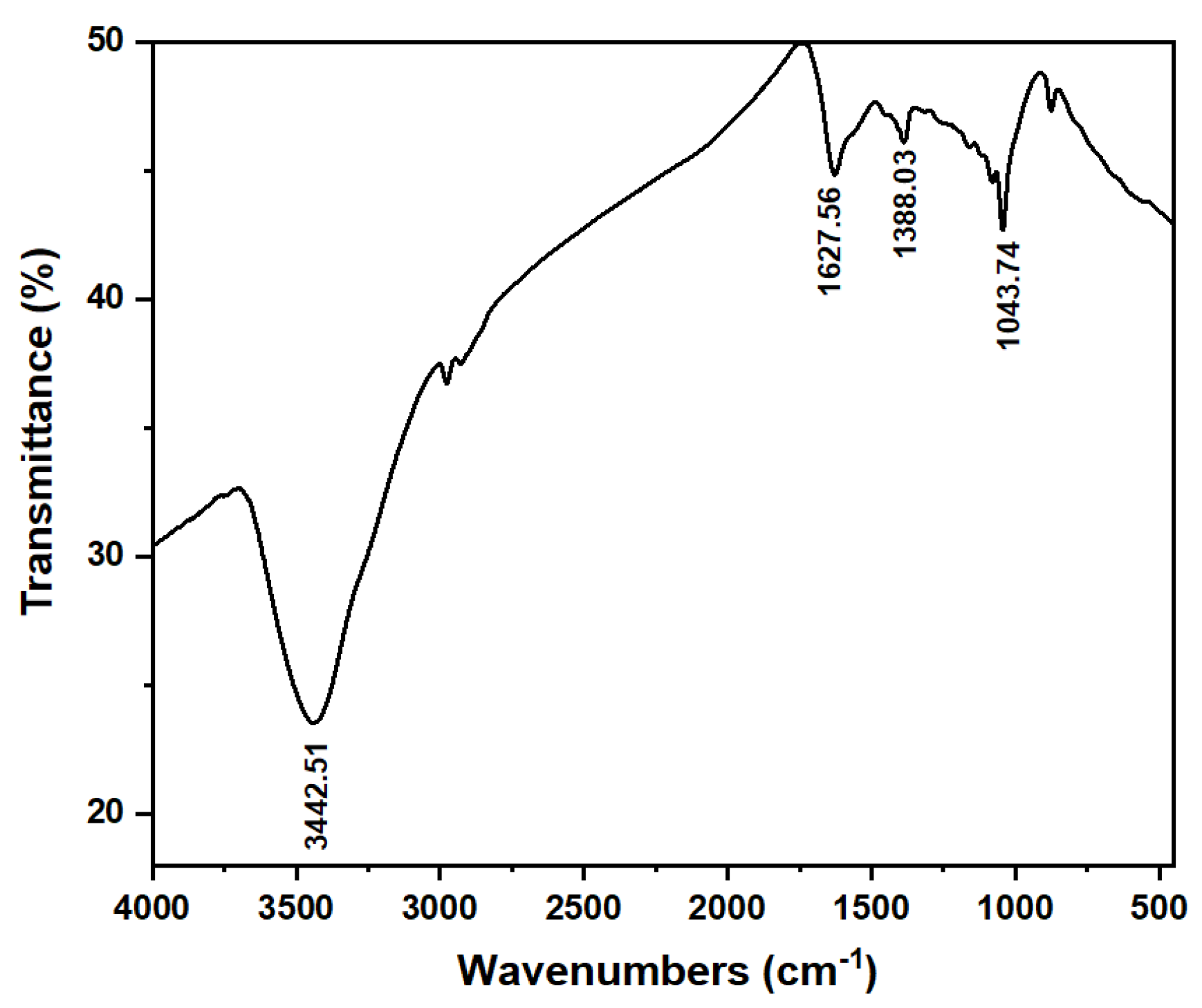
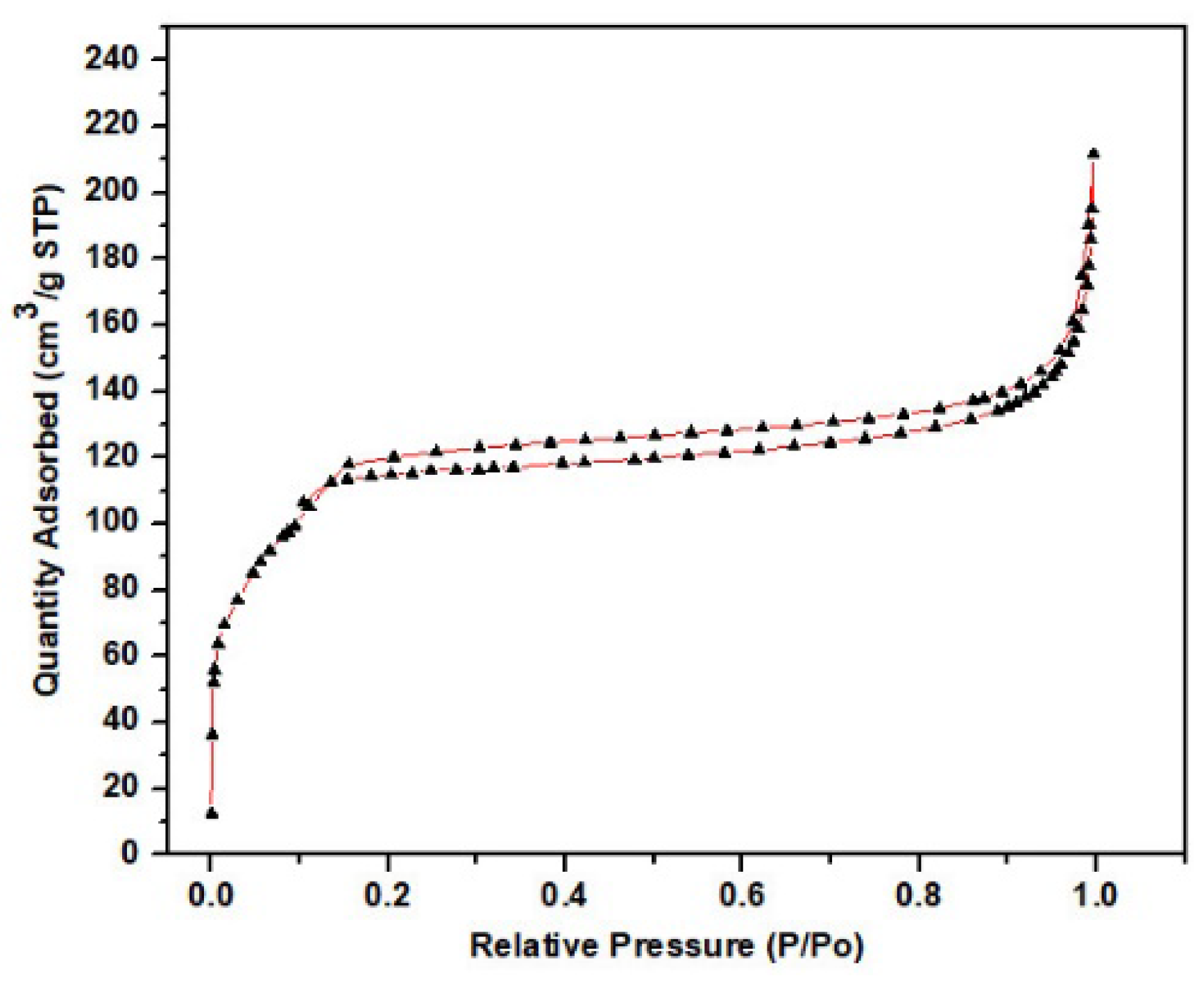
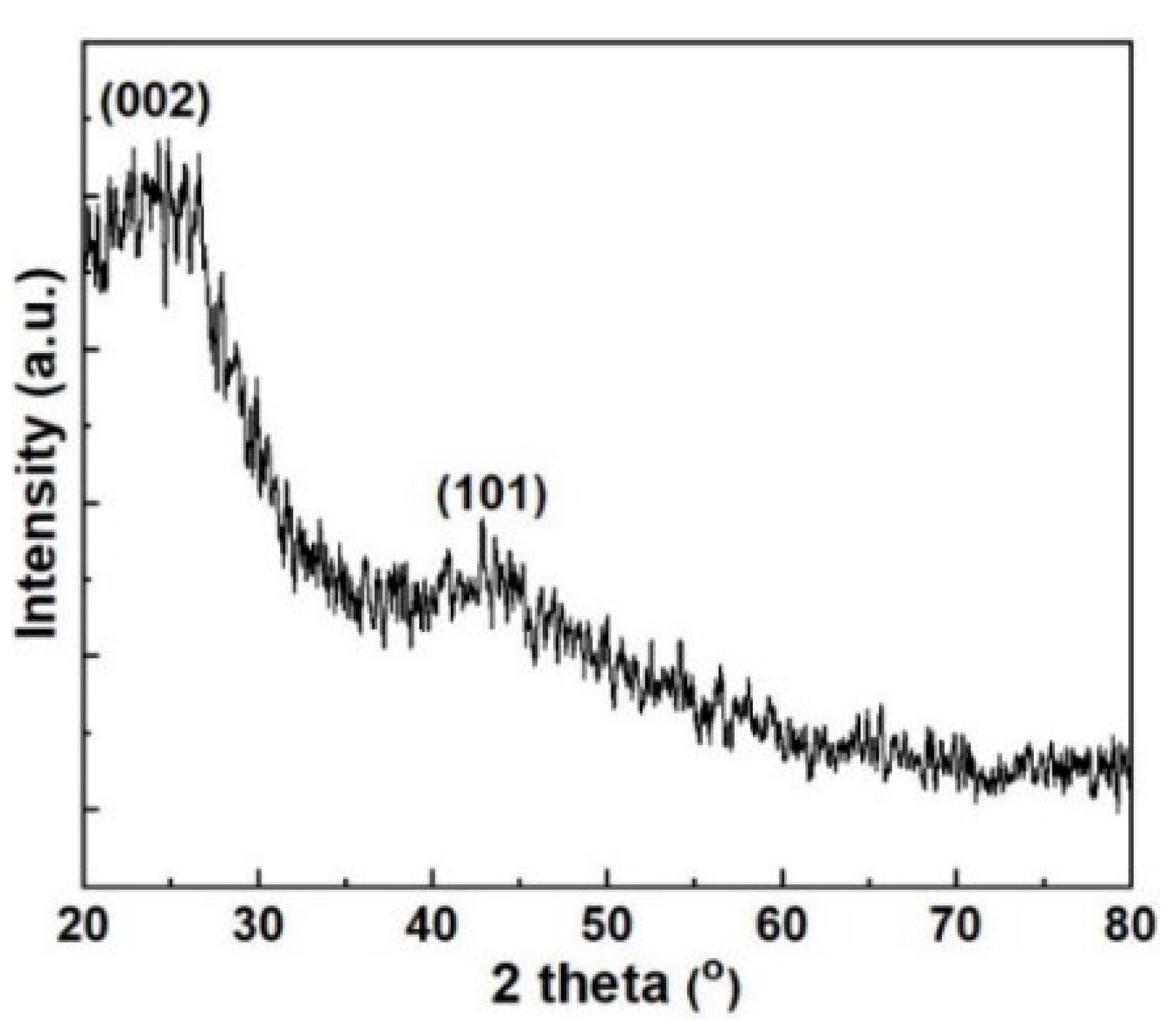
3.1. Investigations on the Physical Propertie, and Surface Characteristics of the MP and ACMP
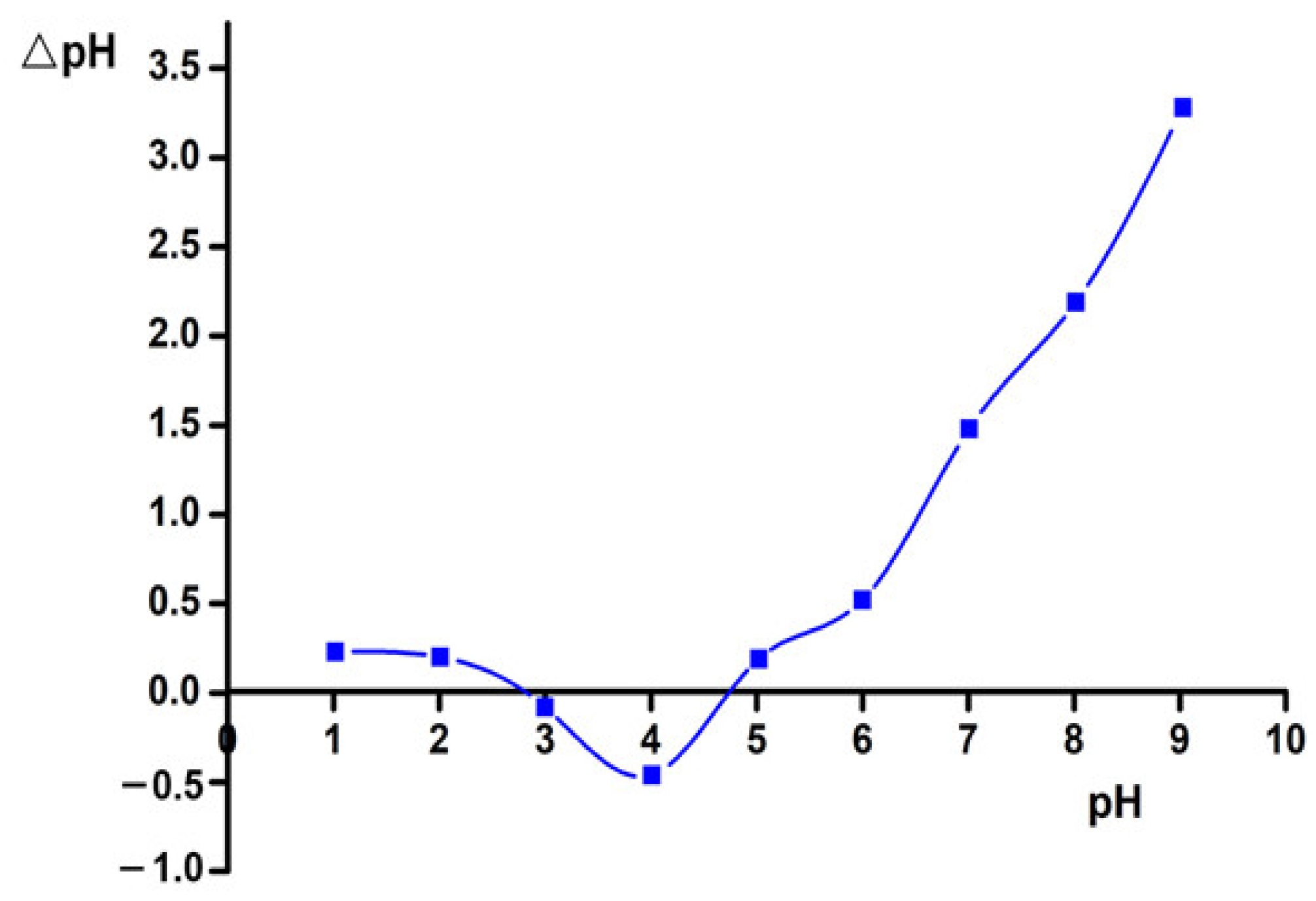
3.2. Isoelectric Point of the ACMP
3.3. Physical Parameters and Iodine Index of the ACMP
3.4. Investigations of the Influencing Effects on the CIP Adsorption Capability of the ACMP
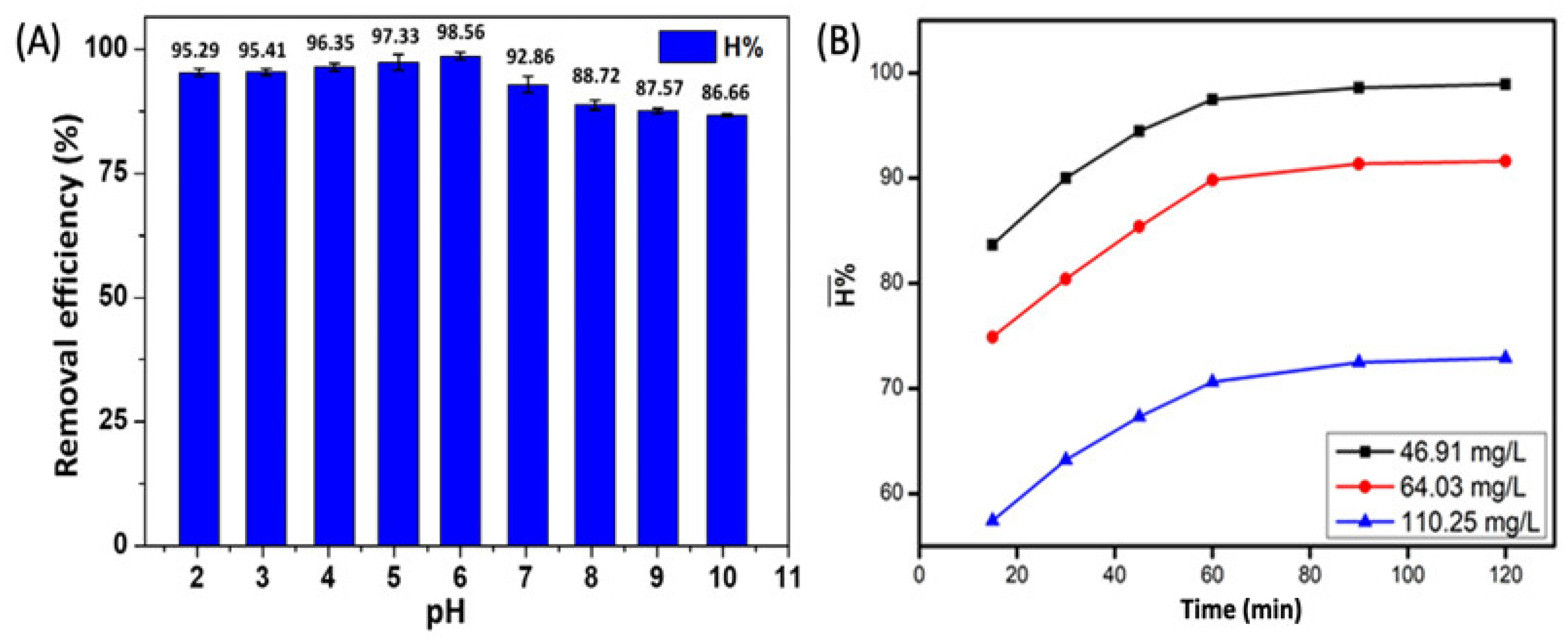
3.4.1. Effect of pH
3.4.2. Effect of Reaction Time on Adsorption Equilibrium
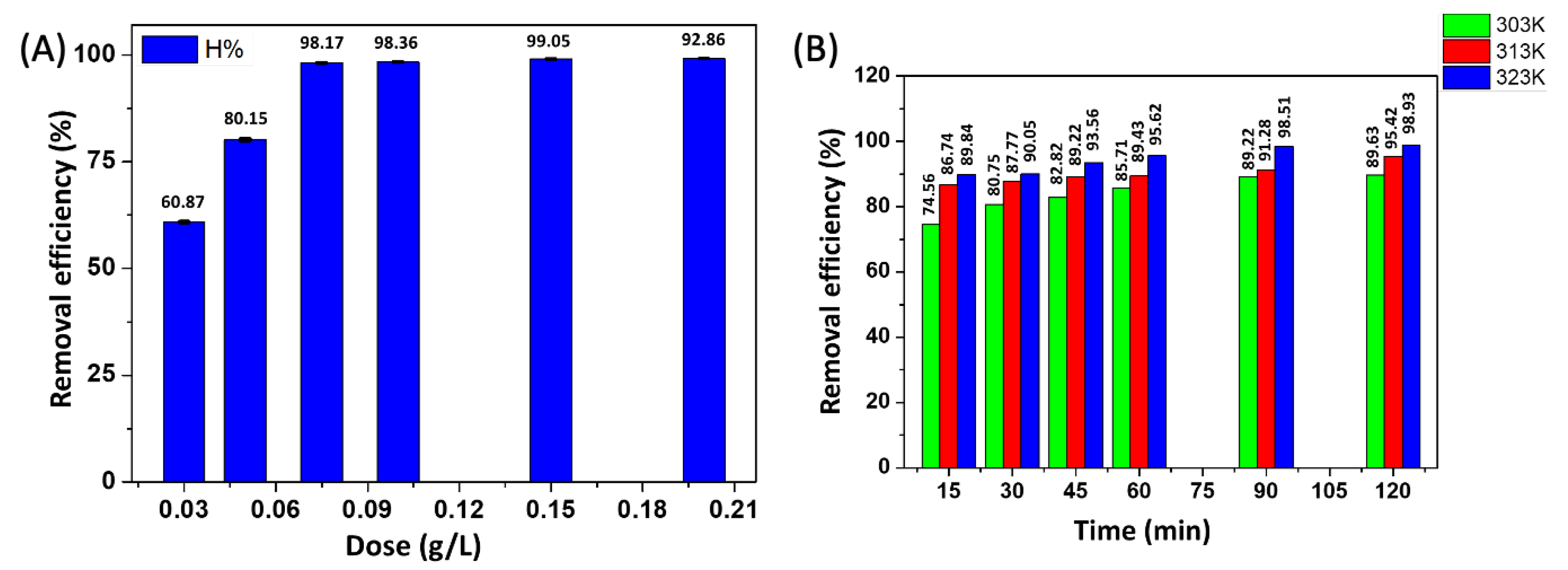
3.4.3. Effect of the ACMP Dosages on the CIP Adsorption Efficiency
3.4.4. Effect of Temperatures
3.5. Isotherm Adsorption Model
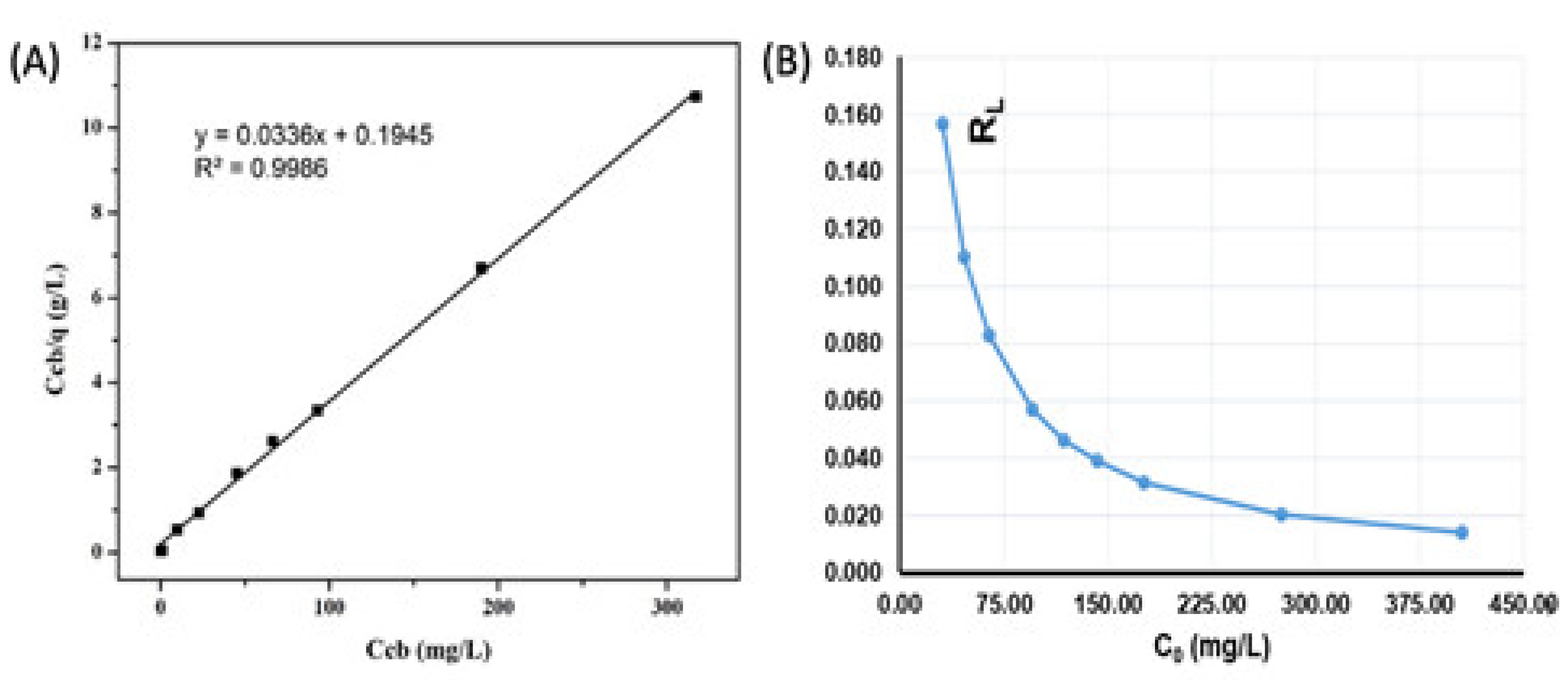
3.5.1. Langmuir Isotherm Adsorption Model
3.5.2. Freundlich Isotherm Adsorption Model
3.5.3. Tempkin Isotherm Adsorption Model
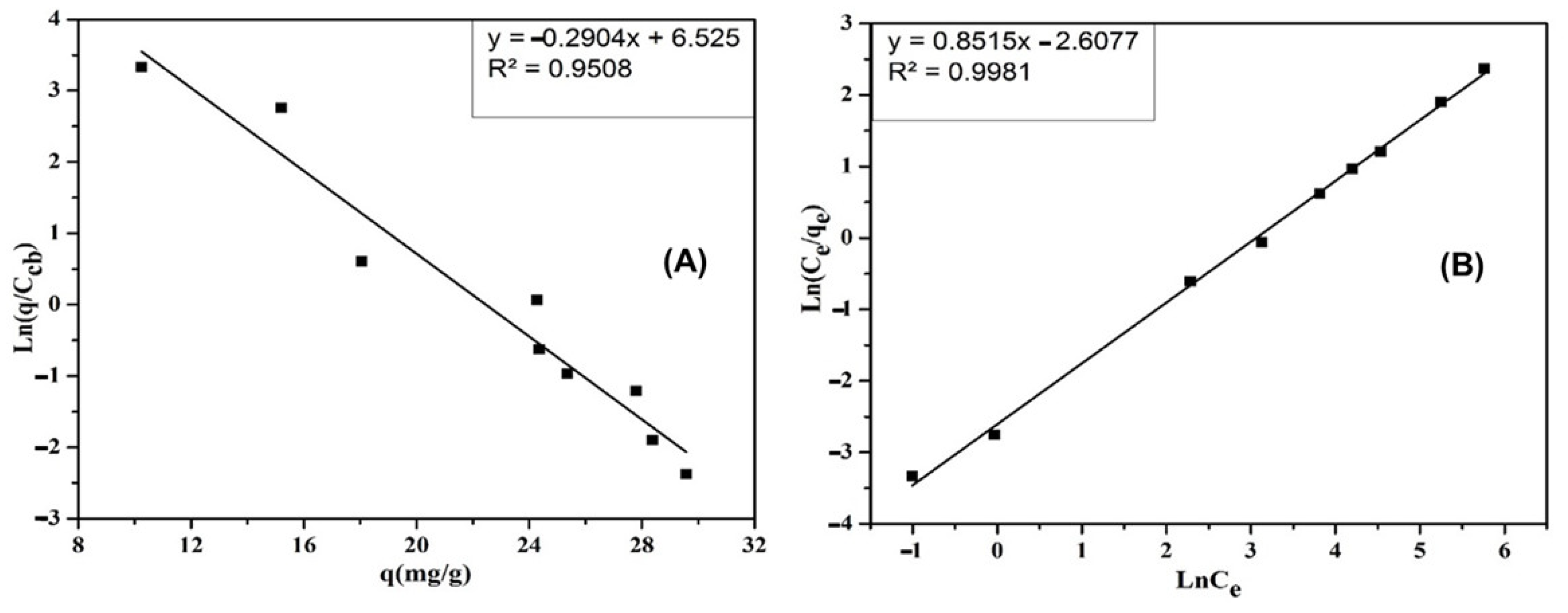
3.5.4. Elovich Isotherm Adsorption Model
3.5.5. Redlich–Peterson Isotherm Adsorption Model
3.6. CIP Adsorption Kinetics of the ACMP
3.7. Activation Energy
3.8. CIP Adsorption Thermodynamics of the ACMP
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Daughton, C.G.; Ternes, T.A. Pharmaceuticals and Personal Care Products in the Environment: Agents of Subtle Change? Environ. Health Perspect. 1999, 107, 32. [Google Scholar] [CrossRef] [PubMed]
- De Witte, B.; Dewulf, J.; Demeestere, K.; Van Langenhove, H. Ozonation and Advanced Oxidation by the Peroxone Process of Ciprofloxacin in Water. J. Hazard. Mater. 2009, 161, 701–708. [Google Scholar] [CrossRef]
- Ghosh, G.C.; Okuda, T.; Yamashita, N.; Tanaka, H. Occurrence and Elimination of Antibiotics at Four Sewage Treatment Plants in Japan and Their Effects on Bacterial Ammonia Oxidation. Water Sci. Technol. 2009, 59, 779–786. [Google Scholar] [CrossRef]
- Yilmaz, M.; Al-Musawi, T.J.; Saloot, M.k.; Khatibi, A.D.; Baniasadi, M.; Balarak, D. Synthesis of Activated Carbon from Lemna Minor Plant and Magnetized with Iron (III) Oxide Magnetic Nanoparticles and Its Application in Removal of Ciprofloxacin. Biomass Conv. Bioref. 2022. [Google Scholar] [CrossRef]
- Lin, C.-C.; Lee, C.-Y. Adsorption of Ciprofloxacin in Water Using Fe3O4 Nanoparticles Formed at Low Temperature and High Reactant Concentrations in a Rotating Packed Bed with Co-Precipitation. Mater. Chem. Phys. 2020, 240, 122049. [Google Scholar] [CrossRef]
- Gulen, B.; Demircivi, P. Adsorption Properties of Flouroquinolone Type Antibiotic Ciprofloxacin into 2:1 Dioctahedral Clay Structure: Box-Behnken Experimental Design. J. Mol. Struct. 2020, 1206, 127659. [Google Scholar] [CrossRef]
- Igwegbe, C.A.; Oba, S.N.; Aniagor, C.O.; Adeniyi, A.G.; Ighalo, J.O. Adsorption of Ciprofloxacin from Water: A Comprehensive Review. J. Ind. Eng. Chem. 2021, 93, 57–77. [Google Scholar] [CrossRef]
- Avcı, A.; İnci, İ.; Baylan, N. Adsorption of Ciprofloxacin Hydrochloride on Multiwall Carbon Nanotube. J. Mol. Struct. 2020, 1206, 127711. [Google Scholar] [CrossRef]
- Wang, M.; Li, G.; Huang, L.; Xue, J.; Liu, Q.; Bao, N.; Huang, J. Study of Ciprofloxacin Adsorption and Regeneration of Activated Carbon Prepared from Enteromorpha Prolifera Impregnated with H3PO4 and Sodium Benzenesulfonate. Ecotoxicol. Environ. Saf. 2017, 139, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Wang, Z.; Guo, J.; Li, Y.; Zhang, H.; Zhu, J.; Xie, X. Enhanced Adsorption of Ciprofloxacin by KOH Modified Biochar Derived from Potato Stems and Leaves. Water Sci. Technol. 2018, 77, 1127–1136. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Lu, S.; Liu, Y.; Meng, W.; Zheng, B. Adsorption of Sulfamethoxazole (SMZ) and Ciprofloxacin (CIP) by Humic Acid (HA): Characteristics and Mechanism. RSC Adv. 2017, 7, 50449–50458. [Google Scholar] [CrossRef]
- Wang, L.; Chen, G.; Ling, C.; Zhang, J.; Szerlag, K. Adsorption of Ciprofloxacin on to Bamboo Charcoal: Effects of PH, Salinity, Cations, and Phosphate. Environ. Prog. Sustain. Energy 2017, 36, 1108–1115. [Google Scholar] [CrossRef]
- Yin, D.; Xu, Z.; Shi, J.; Shen, L.; He, Z. Adsorption Characteristics of Ciprofloxacin on the Schorl: Kinetics, Thermodynamics, Effect of Metal Ion and Mechanisms. J. Water Reuse Desalination 2018, 8, 350–359. [Google Scholar] [CrossRef]
- Li, X.; Chen, S.; Fan, X.; Quan, X.; Tan, F.; Zhang, Y.; Gao, J. Adsorption of Ciprofloxacin, Bisphenol and 2-Chlorophenol on Electrospun Carbon Nanofibers: In Comparison with Powder Activated Carbon. J. Colloid Interface Sci. 2015, 447, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Y.; Yu, F.; Ma, J.; Chen, J. Adsorption of Ciprofloxacin onto Graphene–Soy Protein Biocomposites. New J. Chem. 2015, 39, 3333–3336. [Google Scholar] [CrossRef]
- Carabineiro, S.A.C.; Thavorn-Amornsri, T.; Pereira, M.F.R.; Figueiredo, J.L. Adsorption of Ciprofloxacin on Surface-Modified Carbon Materials. Water Res. 2011, 45, 4583–4591. [Google Scholar] [CrossRef] [PubMed]
- González Labrada, K.; Alcorta Cuello, D.R.; Saborit Sánchez, I.; García Batle, M.; Manero, M.-H.; Barthe, L.; Jáuregui-Haza, U.J. Optimization of Ciprofloxacin Degradation in Wastewater by Homogeneous Sono-Fenton Process at High Frequency. J. Environ. Sci. Health Part A 2018, 53, 1139–1148. [Google Scholar] [CrossRef]
- Basaleh, A.S.; Shawky, A.; Zaki, Z.I. Visible Light-Driven Photodegradation of Ciprofloxacin over Sol-Gel Prepared Bi2O3-Modified La-Doped NaTaO3 Nanostructures. Ceram. Int. 2021, 47, 19205–19212. [Google Scholar] [CrossRef]
- Mukherjee, I.; Cilamkoti, V.; Dutta, R.K. Sunlight-Driven Photocatalytic Degradation of Ciprofloxacin by Carbon Dots Embedded in ZnO Nanostructures. ACS Appl. Nano Mater. 2021, 4, 7686–7697. [Google Scholar] [CrossRef]
- Das, S.; Ghosh, S.; Misra, A.; Tamhankar, A.; Mishra, A.; Lundborg, C.; Tripathy, S. Sunlight Assisted Photocatalytic Degradation of Ciprofloxacin in Water Using Fe Doped ZnO Nanoparticles for Potential Public Health Applications. Int. J. Environ. Res. Public Health 2018, 15, 2440. [Google Scholar] [CrossRef] [Green Version]
- Ulyankina, A.; Molodtsova, T.; Gorshenkov, M.; Leontyev, I.; Zhigunov, D.; Konstantinova, E.; Lastovina, T.; Tolasz, J.; Henych, J.; Licciardello, N.; et al. Photocatalytic degradation of ciprofloxacin in water at nano-ZnO prepared by pulse alternating current electrochemical synthesis. J. Water Process Eng. 2021, 40, 101809. [Google Scholar] [CrossRef]
- Do, T.H.; Ha, X.L.; Duong, T.T.A.; Nguyen, P.C.; Hoang, N.B.; Tran, T.K.N. Optimization, Kinetics, Thermodynamic and Arrhenius Model of the Removal of Ciprofloxacin by Internal Electrolysis with Fe-Cu and Fe-C Materials. Processes 2021, 9, 2110. [Google Scholar] [CrossRef]
- Jiang, J.-Q.; Zhou, Z.; Pahl, O. Preliminary Study of Ciprofloxacin (CIP29) Removal by Potassium Ferrate(VI). Sep. Purif. Technol. 2012, 88, 95–98. [Google Scholar] [CrossRef]
- Li, L.; Liu, J.; Zeng, J.; Li, J.; Liu, Y.; Sun, X.; Xu, L.; Li, L. Complete Degradation and Detoxification of Ciprofloxacin by a Micro-/Nanostructured Biogenic Mn Oxide Composite from a Highly Active Mn2+-Oxidizing Pseudomonas Strain. Nanomaterials 2021, 11, 1660. [Google Scholar] [CrossRef] [PubMed]
- Sayğılı, H.; Güzel, F.; Önal, Y. Conversion of Grape Industrial Processing Waste to Activated Carbon Sorbent and Its Performance in Cationic and Anionic Dyes Adsorption. J. Clean. Prod. 2015, 93, 84–93. [Google Scholar] [CrossRef]
- Iradukunda, Y.; Wang, G.; Li, X.; Shi, G.; Hu, Y.; Luo, F.; Yi, K.; Albashir, A.I.M.; Niu, X.; Wu, Z. High Performance of Activated Carbons Prepared from Mangosteen (Garcinia Mangostana) Peels Using the Hydrothermal Process. J. Energy Storage 2021, 39, 102577. [Google Scholar] [CrossRef]
- Kongsune, P.; Rattanapan, S.; Chanajaree, R. The Removal of Pb2+ from Aqueous Solution Using Mangosteen Peel Activated Carbon: Isotherm, Kinetic, Thermodynamic and Binding Energy Calculation. Groundw. Sustain. Dev. 2021, 12, 100524. [Google Scholar] [CrossRef]
- Ahmad, M.A.; Alrozi, R. Optimization of Preparation Conditions for Mangosteen Peel-Based Activated Carbons for the Removal of Remazol Brilliant Blue R Using Response Surface Methodology. Chem. Eng. J. 2010, 165, 883–890. [Google Scholar] [CrossRef]
- Chen, Y.; Huang, B.; Huang, M.; Cai, B. On the Preparation and Characterization of Activated Carbon from Mangosteen Shell. J. Taiwan Inst. Chem. Eng. 2011, 42, 837–842. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Factors Affecting the Carbon Yield and Adsorption Capability of the Mangosteen Peel Activated Carbon Prepared by Microwave Assisted K2CO3 Activation. Chem. Eng. J. 2012, 180, 66–74. [Google Scholar] [CrossRef]
- Pangsupa, W.; Hunsom, M. Preparation of Mangosteen Shell-Derived Activated Carbon Via KOH Activation for Adsorptive Refining of Crude Biodiesel. J. Am. Oil Chem. Soc. 2016, 93, 1697–1708. [Google Scholar] [CrossRef]
- Nasrullah, A.; Saad, B.; Bhat, A.H.; Khan, A.S.; Danish, M.; Isa, M.H.; Naeem, A. Mangosteen Peel Waste as a Sustainable Precursor for High Surface Area Mesoporous Activated Carbon: Characterization and Application for Methylene Blue Removal. J. Clean. Prod. 2019, 211, 1190–1200. [Google Scholar] [CrossRef]
- Xue, M.; Chen, C.; Tan, Y.; Ren, Z.; Li, B.; Zhang, C. Mangosteen Peel-Derived Porous Carbon: Synthesis and Its Application in the Sulfur Cathode for Lithium Sulfur Battery. J. Mater. Sci. 2018, 53, 11062–11077. [Google Scholar] [CrossRef]
- Giraldo, L.; Moreno-Piraján, J.C. CO2 Adsorption on Activated Carbon Prepared from Mangosteen Peel: Study by Adsorption Calorimetry. J. Therm. Anal. Calorim. 2018, 133, 337–354. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, L.; Liu, Y.; Feng, R.; Zou, T.; Zhang, Y.; Kang, Y.; Zhou, P. Efficient Removal of Methylene Blue Using the Mesoporous Activated Carbon Obtained from Mangosteen Peel Wastes: Kinetic, Equilibrium, and Thermodynamic Studies. Microporous Mesoporous Mater. 2021, 315, 110904. [Google Scholar] [CrossRef]
- Do, T.H.; Nguyen, V.T.; Dung, N.Q.; Chu, M.N.; Van Kiet, D.; Ngan, T.T.K.; Van Tan, L. Study on Methylene Blue Adsorption of Activated Carbon Made from Moringa Oleifera Leaf. Mater. Today Proc. 2021, 38, 3405–3413. [Google Scholar] [CrossRef]
- Kam, S.-K.; Lee, M.-G. Response Surface Modeling for the Adsorption of Dye Eosin Y by Activated Carbon Prepared from Waste Citrus Peel. Appl. Chem. Eng. 2018, 29, 270–277. [Google Scholar] [CrossRef]
- Balarak, D.; Bazrafshan, E.; Mahdavi, Y.; Lalhmunsiama, L.; Lee, S.-M. Kinetic, Isotherms and Thermodynamic Studies in the Removal of 2-Chlorophenol from Aqueous Solution Using Modified Rice Straw. DWT 2017, 63, 203–211. [Google Scholar] [CrossRef]
- Ayawei, N.; Ebelegi, A.N.; Wankasi, D. Modelling and Interpretation of Adsorption Isotherms. J. Chem. 2017, 2017, 3039817. [Google Scholar] [CrossRef]
- Abramian, L.; El-Rassy, H. Adsorption Kinetics and Thermodynamics of Azo-Dye Orange II onto Highly Porous Titania Aerogel. Chem. Eng. J. 2009, 150, 403–410. [Google Scholar] [CrossRef]


| Experiment/Parameters | pH | Time (min) | Material Mass (g) | CIP Initial Concentration (mg/L) | Temperature (K) |
|---|---|---|---|---|---|
| Effect of pH | 3 to 10 | 60 | 0.075 | 50 | 298 |
| Effect of time | 6 | 15–120 | 0.075 | 50 | 298 |
| Effect of mass of initial materials | 6 | 60 | 0.03–0.2 | 50 | 298 |
| Effect of CIP initial concentration | 6 | 60 | 0.075 | 50–400 | 298 |
| Effect of temperature | 6 | 60 | 0.075 | 100 | 303, 313, and 323 |
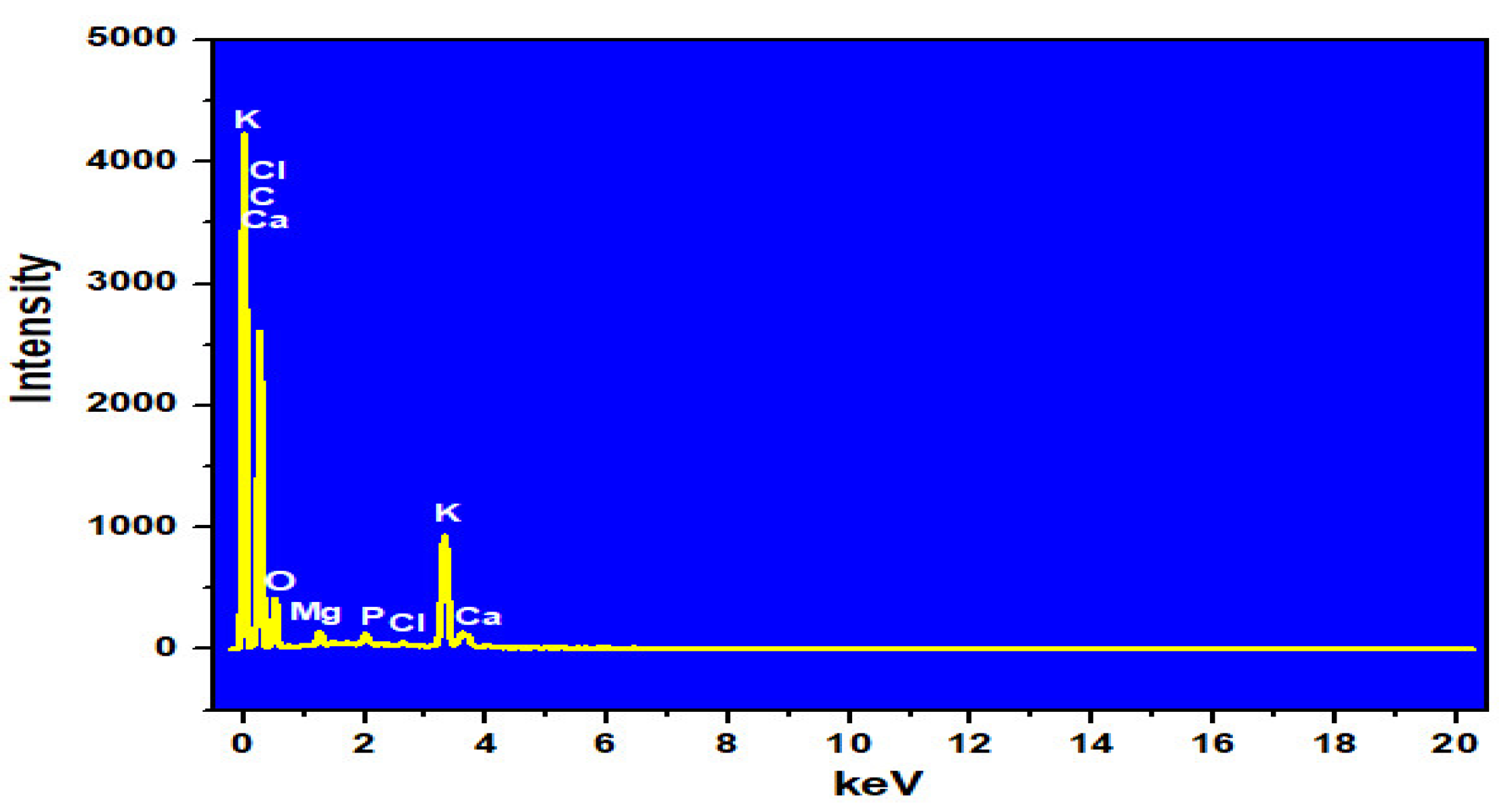
| Element | % Weight | % Atomic |
|---|---|---|
| C | 68.81 | 76.86 |
| O | 24.73 | 20.74 |
| Mg | 0.70 | 0.39 |
| P | 0.38 | 0.16 |
| Cl | 0.13 | 0.05 |
| K | 4.76 | 1.63 |
| Ca | 0.50 | 0.17 |
| Total | 100.00 | 100.00 |
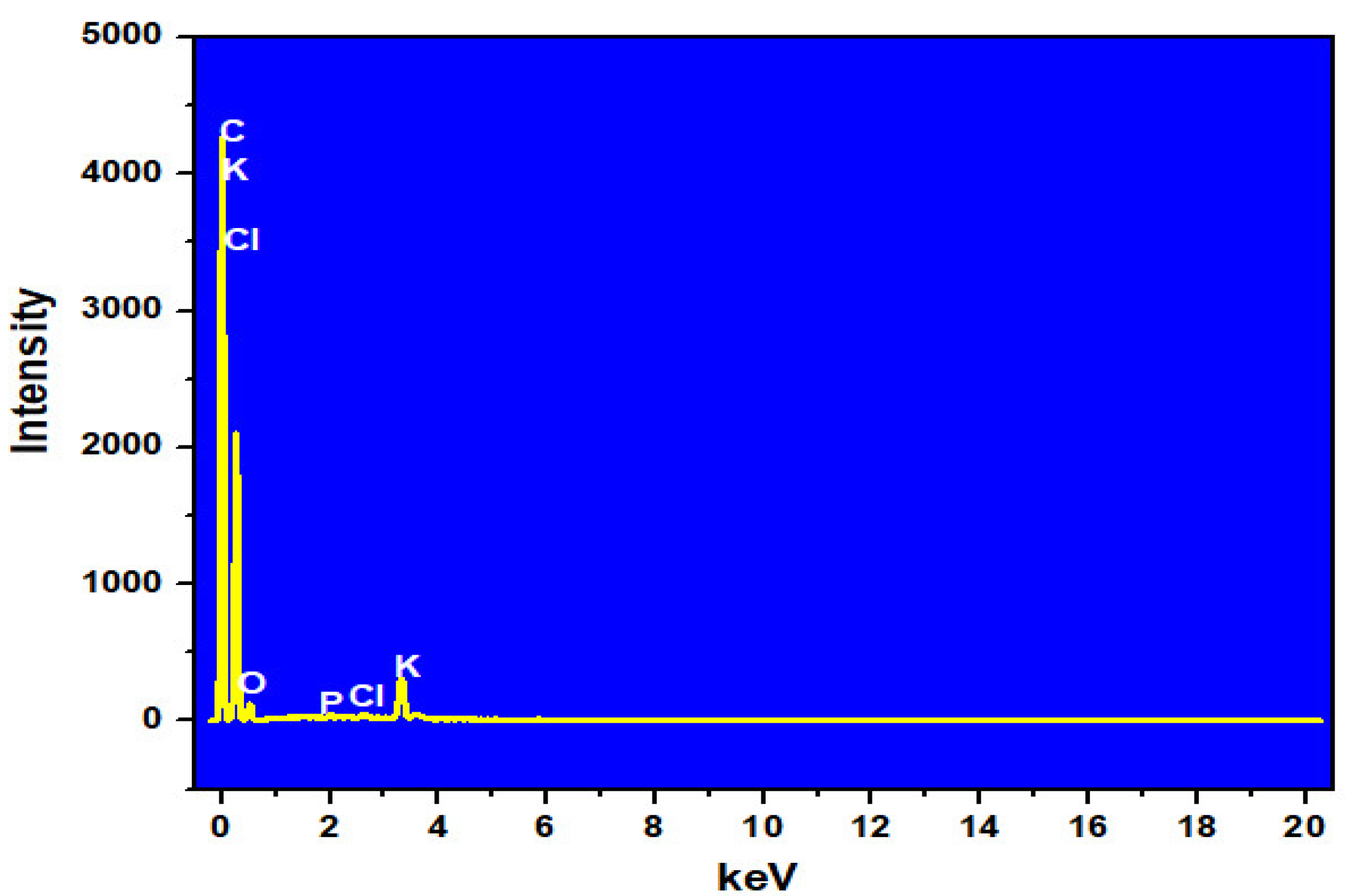
| Element | % Weight | % Atomic |
|---|---|---|
| C | 81.57 | 86.74 |
| O | 15.31 | 12.22 |
| P | 0.20 | 0.08 |
| Cl | 0.22 | 0.08 |
| K | 2.70 | 0.88 |
| Total | 100.00 | 100.00 |
| Ash content (%) | 55.56 ± 1 |
| Moisture content (%) | 4.54 ± 0.03 |
| Density (g/cm3) | 0.85 ± 0.05 |
| Iodine index | 825 ± 0.05 |
| No. | Adsorbents | Adsorption Capacity (mg/g) | Reference |
|---|---|---|---|
| 1 | Activated carbon from Lemna minor plant | 25.00 | [4] |
| 2 | Nano Fe3O4 | 24.00 | [5] |
| 3 | Montmorillonite clay | 128.00 | [6] |
| 4 | Potato stems and leaves | 23.36 | [10] |
| 5 | Humic acid | 13.64 | [11] |
| 6 | Bamboo charcoal | 36.02 | [12] |
| 7 | Porous graphene–soy protein | 160.00 | [15] |
| 8 | ACMP | 29.76 | This work |
| Isotherm Models | Parameters | |
|---|---|---|
| Langmuir | b (L/mg) | 0.173 |
| qmax (mg/g) | 29.76 | |
| R2 | 0.9986 | |
| Freundlich | KF (mg/g)·(mg/L)1/n | 13.57 |
| N | 6.73 | |
| R2 | 0.9421 | |
| Tempkin | KT | 138.49 |
| bT (kJ·mol) | 0.88 | |
| R2 | 0.9653 | |
| Elovich | qmax (mg/g) | 3.44 |
| Ke | 198.25 | |
| R2 | 0.9508 | |
| Redlich–Peterson | β | 0.8515 |
| A (mg·L−1) | 13.57 | |
| R2 | 0.9982 | |
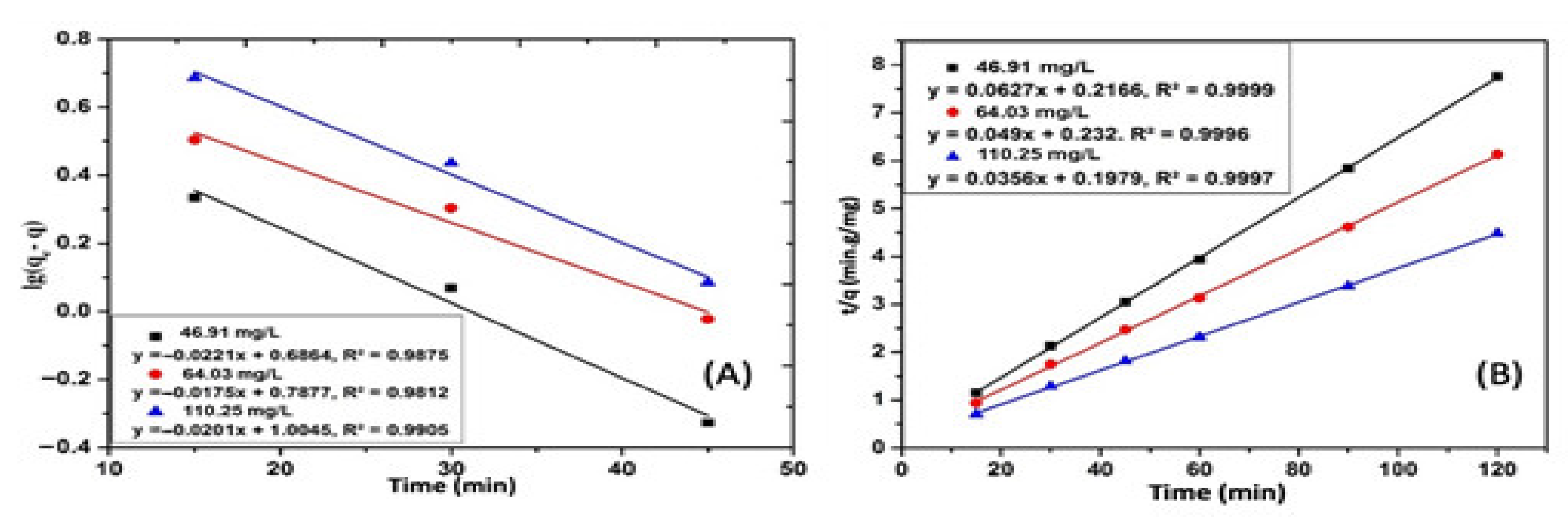
| CIP Concentration (mg/L) | qt,exp Experiment (mg/g) | qt,cal Calculation (mg/g) | Constant k1 (min−1) | R2 |
|---|---|---|---|---|
| 46.91 | 15.24 | 4.86 | 0.0509 | 0.9875 |
| 64.03 | 19.17 | 6.13 | 0.0403 | 0.9812 |
| 110.25 | 25.96 | 10.10 | 0.0463 | 0.9905 |
| CIP Concentration (mg/L) | qt,exp Experiment (mg/g) | qt,cal Calculation (mg/g) | Constant k2 (min−1 L·mg−1) | R2 | %∆q |
|---|---|---|---|---|---|
| 46.91 | 15.24 | 15.95 | 0.0122 | 0.9999 | 1.04 |
| 64.03 | 19.17 | 20.41 | 0.0103 | 0.9996 | 3.02 |
| 110.25 | 25.96 | 28.09 | 0.0094 | 0.9997 | 3.73 |
| Concentration CIP (mg/L) | Second-Order Kinetics Equation | Constant k2 (g/mg min) | h | E (kJ/moL) |
|---|---|---|---|---|
| 46.91 | t/qt = 0.0627·t + 0.2166 | 0.0182 | 4.62 | 13.72 |
| 64.03 | t/qt = 0.049·t + 0.232 | 0.0103 | 4.31 | 14.96 |
| 110.25 | t/qt = 0.0356·t + 0.1979 | 0.0064 | 5.05 | 16.53 |
| T(K) | ∆G0 (kJ/mol) | ∆H0 (kJ/mol) | ∆S0(kJ/mol·K) |
|---|---|---|---|
| 303 | −0.845 | 66.957 | 0.223 |
| 313 | −2.696 | ||
| 323 | −5.329 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, Q.T.; Do, T.H.; Ha, X.L.; Nguyen, H.P.; Nguyen, A.T.; Ngo, T.C.Q.; Chau, H.D. Study of the Ciprofloxacin Adsorption of Activated Carbon Prepared from Mangosteen Peel. Appl. Sci. 2022, 12, 8770. https://doi.org/10.3390/app12178770
Tran QT, Do TH, Ha XL, Nguyen HP, Nguyen AT, Ngo TCQ, Chau HD. Study of the Ciprofloxacin Adsorption of Activated Carbon Prepared from Mangosteen Peel. Applied Sciences. 2022; 12(17):8770. https://doi.org/10.3390/app12178770
Chicago/Turabian StyleTran, Quoc Toan, Tra Huong Do, Xuan Linh Ha, Huyen Phuong Nguyen, Anh Tien Nguyen, Thi Cam Quyen Ngo, and Hung Dung Chau. 2022. "Study of the Ciprofloxacin Adsorption of Activated Carbon Prepared from Mangosteen Peel" Applied Sciences 12, no. 17: 8770. https://doi.org/10.3390/app12178770
APA StyleTran, Q. T., Do, T. H., Ha, X. L., Nguyen, H. P., Nguyen, A. T., Ngo, T. C. Q., & Chau, H. D. (2022). Study of the Ciprofloxacin Adsorption of Activated Carbon Prepared from Mangosteen Peel. Applied Sciences, 12(17), 8770. https://doi.org/10.3390/app12178770







