Exploring Serum NMR-Based Metabolomic Fingerprint of Colorectal Cancer Patients: Effects of Surgery and Possible Associations with Cancer Relapse
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Cohort
2.2. Samples Collection
2.3. NMR Analysis
2.3.1. Acquisition of NMR Data
2.3.2. Spectral Processing
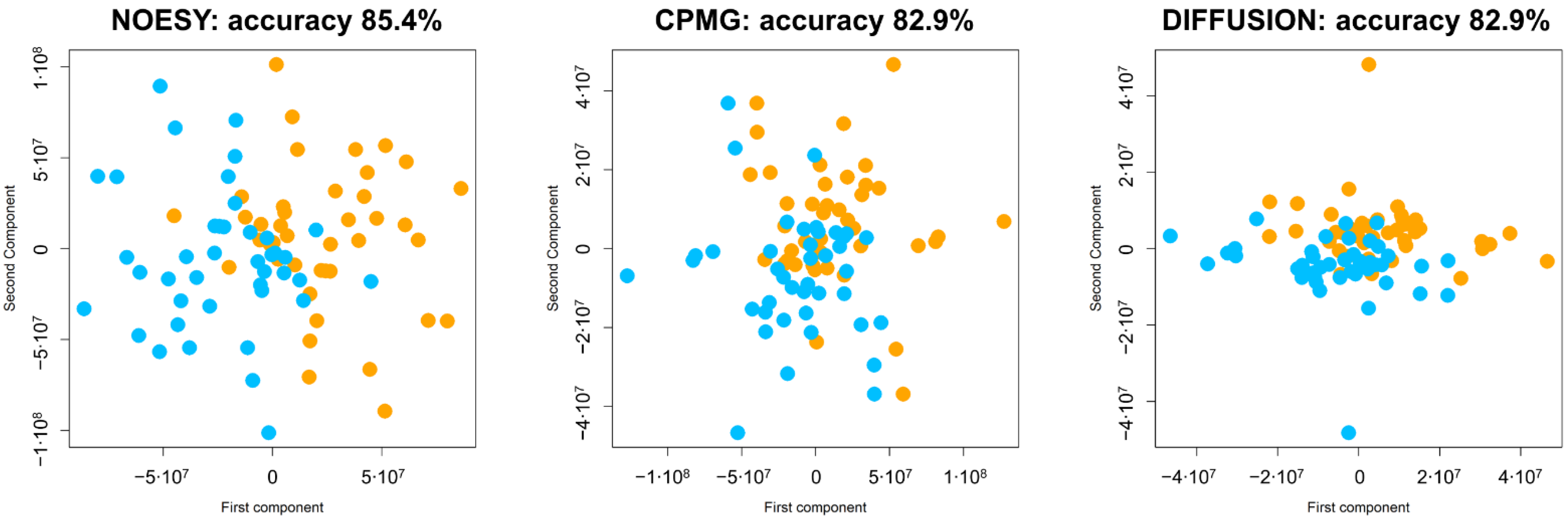
2.4. Statistical Analysis
3. Results
3.1. Characteristics of Enrolled Patients
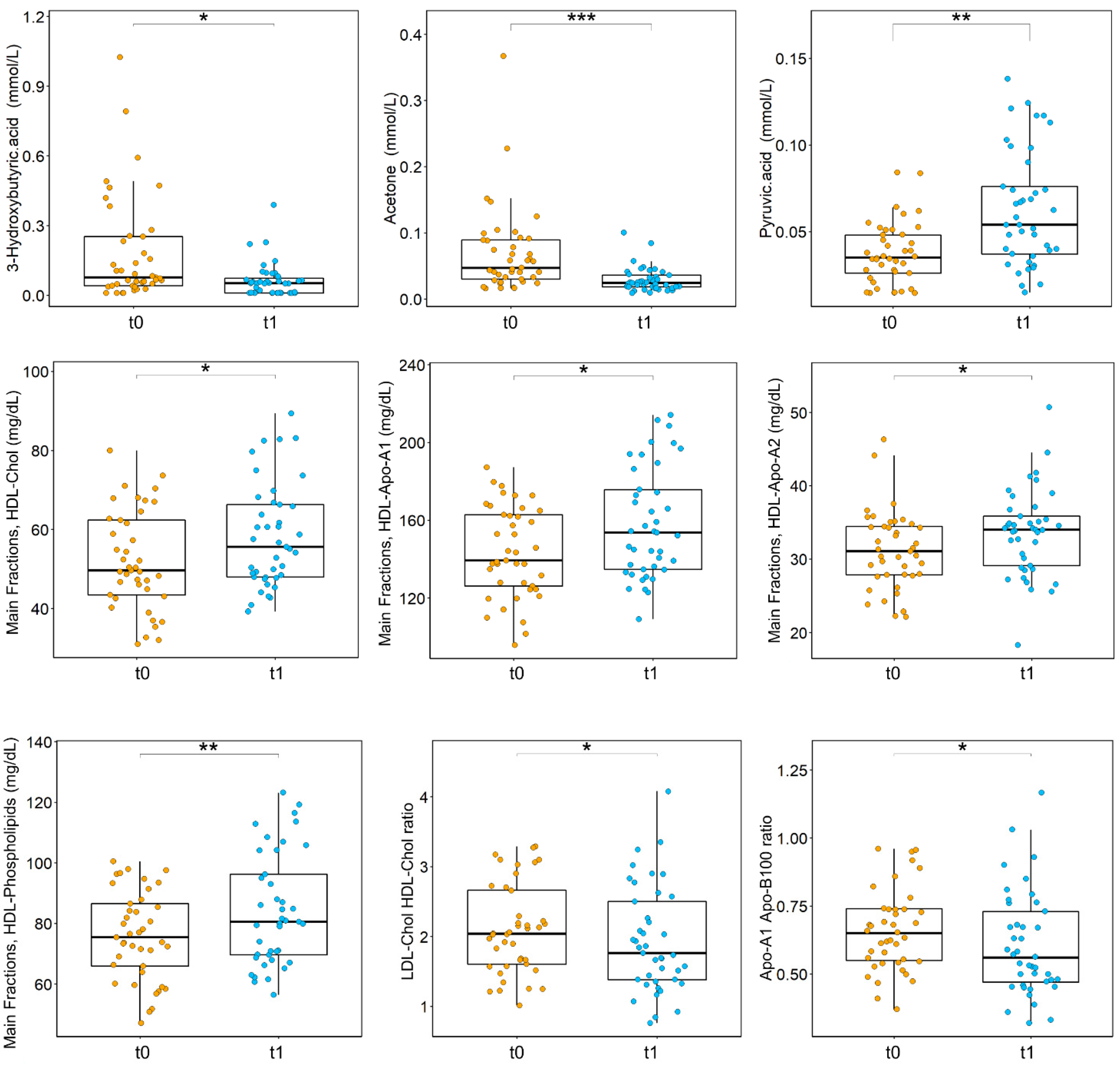
3.2. Effects of Surgery on the Metabolome of CRC Patients
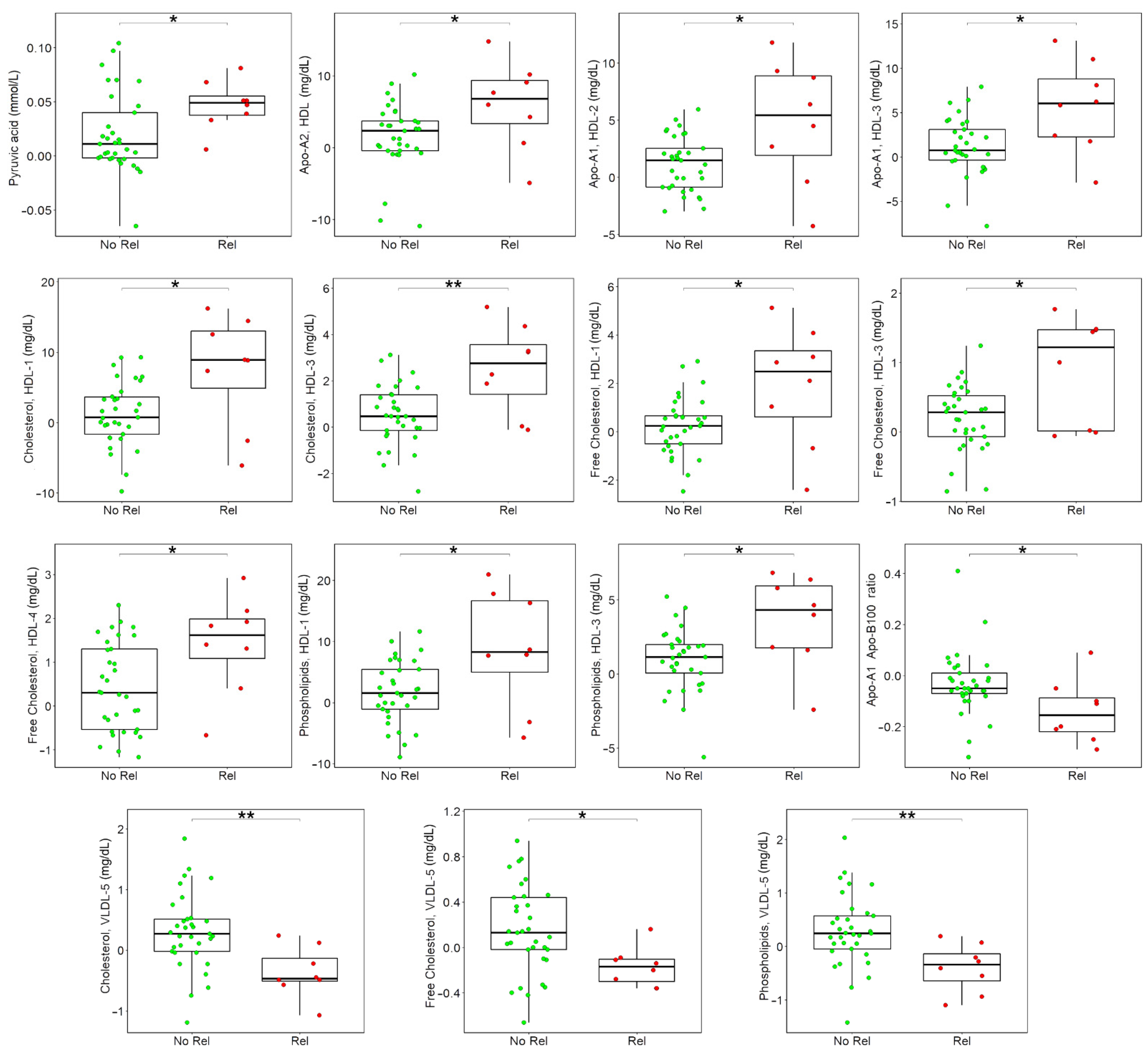
3.3. Associations between Metabolome Variations after Surgery and Cancer Relapse
3.4. Associations between Metabolites and Clinical Variables
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- NCCN Guidelines for Colon Cancer 2021. Available online: https://www.nccn.org/guidelines/guidelines-detail (accessed on 5 November 2021).
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- AIOM: Linee Guida Tumori del Colon 2020. Available online: https://www.aiom.it/linee-guida-aiom-2020-tumori-del-colon/ (accessed on 5 November 2021).
- Reinert, T.; Schøler, L.V.; Thomsen, R.; Tobiasen, H.; Vang, S.; Nordentoft, I.; Lamy, P.; Kannerup, A.-S.; Mortensen, F.V.; Stribolt, K.; et al. Analysis of Circulating Tumour DNA to Monitor Disease Burden Following Colorectal Cancer Surgery. Gut 2016, 65, 625–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guraya, S.Y. Pattern, Stage, and Time of Recurrent Colorectal Cancer After Curative Surgery. Clin. Colorectal Cancer 2019, 18, e223–e228. [Google Scholar] [CrossRef]
- Hall, M.J.; Morris, A.M.; Sun, W. Precision Medicine Versus Population Medicine in Colon Cancer: From Prospects of Prevention, Adjuvant Chemotherapy, and Surveillance. Am. Soc. Clin. Oncol. Educ. Book 2018, 38, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Dienstmann, R.; Mason, M.J.; Sinicrope, F.A.; Phipps, A.I.; Tejpar, S.; Nesbakken, A.; Danielsen, S.A.; Sveen, A.; Buchanan, D.D.; Clendenning, M.; et al. Prediction of Overall Survival in Stage II and III Colon Cancer beyond TNM System: A Retrospective, Pooled Biomarker Study. Ann. Oncol. 2017, 28, 1023–1031. [Google Scholar] [CrossRef]
- SEER Cancer Statistics Review, 1975–2017. Available online: https://seer.cancer.gov/csr/1975_2017/index.html (accessed on 30 March 2021).
- Renfro, L.A.; Grothey, A.; Xue, Y.; Saltz, L.B.; André, T.; Twelves, C.; Labianca, R.; Allegra, C.J.; Alberts, S.R.; Loprinzi, C.L.; et al. ACCENT-Based Web Calculators to Predict Recurrence and Overall Survival in Stage III Colon Cancer. J. Natl. Cancer Inst. 2014, 106, dju333. [Google Scholar] [CrossRef] [Green Version]
- Benson, A.B.; Schrag, D.; Somerfield, M.R.; Cohen, A.M.; Figueredo, A.T.; Flynn, P.J.; Krzyzanowska, M.K.; Maroun, J.; McAllister, P.; Van Cutsem, E.; et al. American Society of Clinical Oncology Recommendations on Adjuvant Chemotherapy for Stage II Colon Cancer. J. Clin. Oncol. 2004, 22, 3408–3419. [Google Scholar] [CrossRef]
- Kumar, A.; Kennecke, H.F.; Renouf, D.J.; Lim, H.J.; Gill, S.; Woods, R.; Speers, C.; Cheung, W.Y. Adjuvant Chemotherapy Use and Outcomes of Patients with High-Risk versus Low-Risk Stage II Colon Cancer. Cancer 2015, 121, 527–534. [Google Scholar] [CrossRef]
- Di Donato, S.; Vignoli, A.; Biagioni, C.; Malorni, L.; Mori, E.; Tenori, L.; Calamai, V.; Parnofiello, A.; Di Pierro, G.; Migliaccio, I.; et al. A Serum Metabolomics Classifier Derived from Elderly Patients with Metastatic Colorectal Cancer Predicts Relapse in the Adjuvant Setting. Cancers 2021, 13, 2762. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.K.; Lindon, J.C. Systems Biology: Metabonomics. Nature 2008, 455, 1054–1056. [Google Scholar] [CrossRef]
- Vignoli, A.; Risi, E.; McCartney, A.; Migliaccio, I.; Moretti, E.; Malorni, L.; Luchinat, C.; Biganzoli, L.; Tenori, L. Precision Oncology via NMR-Based Metabolomics: A Review on Breast Cancer. Int. J. Mol. Sci. 2021, 22, 4687. [Google Scholar] [CrossRef]
- Vignoli, A.; Ghini, V.; Meoni, G.; Licari, C.; Takis, P.G.; Tenori, L.; Turano, P.; Luchinat, C. High-Throughput Metabolomics by 1D NMR. Angew. Chem. Int. Ed. 2019, 58, 968–994. [Google Scholar] [CrossRef] [PubMed]
- Wishart, D.S. Emerging Applications of Metabolomics in Drug Discovery and Precision Medicine. Nat. Rev. Drug Discov. 2016, 15, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Vignoli, A.; Tenori, L.; Giusti, B.; Valente, S.; Carrabba, N.; Baizi, D.; Barchielli, A.; Marchionni, N.; Gensini, G.F.; Marcucci, R.; et al. Differential Network Analysis Reveals Metabolic Determinants Associated with Mortality in Acute Myocardial Infarction Patients and Suggests Potential Mechanisms Underlying Different Clinical Scores Used To Predict Death. J. Proteome Res. 2020, 19, 949–961. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhu, B.; Zeng, Y.; Shen, H.; Zhang, J.; Wang, X. Clinical Lipidomics in Understanding of Lung Cancer: Opportunity and Challenge. Cancer Lett. 2020, 470, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Pietzner, M.; Stewart, I.D.; Raffler, J.; Khaw, K.-T.; Michelotti, G.A.; Kastenmüller, G.; Wareham, N.J.; Langenberg, C. Plasma Metabolites to Profile Pathways in Noncommunicable Disease Multimorbidity. Nat. Med. 2021, 27, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Vignoli, A.; Paciotti, S.; Tenori, L.; Eusebi, P.; Biscetti, L.; Chiasserini, D.; Scheltens, P.; Turano, P.; Teunissen, C.; Luchinat, C.; et al. Fingerprinting Alzheimer’s Disease by 1H Nuclear Magnetic Resonance Spectroscopy of Cerebrospinal Fluid. J. Proteome Res. 2020, 19, 1696–1705. [Google Scholar] [CrossRef] [PubMed]
- Vignoli, A.; Tenori, L.; Giusti, B.; Takis, P.G.; Valente, S.; Carrabba, N.; Balzi, D.; Barchielli, A.; Marchionni, N.; Gensini, G.F.; et al. NMR-Based Metabolomics Identifies Patients at High Risk of Death within Two Years after Acute Myocardial Infarction in the AMI-Florence II Cohort. BMC Med. 2019, 17, 3. [Google Scholar] [CrossRef] [Green Version]
- Auclin, E.; Zaanan, A.; Vernerey, D.; Douard, R.; Gallois, C.; Laurent-Puig, P.; Bonnetain, F.; Taieb, J. Subgroups and Prognostication in Stage III Colon Cancer: Future Perspectives for Adjuvant Therapy. Ann. Oncol. 2017, 28, 958–968. [Google Scholar] [CrossRef]
- Copija, A.; Waniczek, D.; Witkoś, A.; Walkiewicz, K.; Nowakowska-Zajdel, E. Clinical Significance and Prognostic Relevance of Microsatellite Instability in Sporadic Colorectal Cancer Patients. Int. J. Mol. Sci. 2017, 18, 107. [Google Scholar] [CrossRef] [PubMed]
- Nannini, G.; Meoni, G.; Amedei, A.; Tenori, L. Metabolomics Profile in Gastrointestinal Cancers: Update and Future Perspectives. World J. Gastroenterol. 2020, 26, 2514–2532. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zhang, P.; Wang, F.; Liu, W.; Yang, J.; Qin, H. An Integrated Proteomics and Metabolomics Approach for Defining Oncofetal Biomarkers in the Colorectal Cancer. Ann. Surg. 2012, 255, 720–730. [Google Scholar] [CrossRef]
- Nishiumi, S.; Kobayashi, T.; Ikeda, A.; Yoshie, T.; Kibi, M.; Izumi, Y.; Okuno, T.; Hayashi, N.; Kawano, S.; Takenawa, T.; et al. A Novel Serum Metabolomics-Based Diagnostic Approach for Colorectal Cancer. PLoS ONE 2012, 7, e40459. [Google Scholar] [CrossRef]
- Qiu, Y.; Cai, G.; Zhou, B.; Li, D.; Zhao, A.; Xie, G.; Li, H.; Cai, S.; Xie, D.; Huang, C.; et al. A Distinct Metabolic Signature of Human Colorectal Cancer with Prognostic Potential. Clin. Cancer Res. 2014, 20, 2136–2146. [Google Scholar] [CrossRef] [Green Version]
- Farshidfar, F.; Weljie, A.M.; Kopciuk, K.; Buie, W.D.; Maclean, A.; Dixon, E.; Sutherland, F.R.; Molckovsky, A.; Vogel, H.J.; Bathe, O.F. Serum Metabolomic Profile as a Means to Distinguish Stage of Colorectal Cancer. Genome Med. 2012, 4, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farshidfar, F.; Weljie, A.M.; Kopciuk, K.A.; Hilsden, R.; McGregor, S.E.; Buie, W.D.; MacLean, A.; Vogel, H.J.; Bathe, O.F. A Validated Metabolomic Signature for Colorectal Cancer: Exploration of the Clinical Value of Metabolomics. Br. J. Cancer 2016, 115, 848–857. [Google Scholar] [CrossRef] [Green Version]
- Bertini, I.; Cacciatore, S.; Jensen, B.V.; Schou, J.V.; Johansen, J.S.; Kruhøffer, M.; Luchinat, C.; Nielsen, D.L.; Turano, P. Metabolomic NMR Fingerprinting to Identify and Predict Survival of Patients with Metastatic Colorectal Cancer. Cancer Res. 2012, 72, 356–364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ISO/DIS 23118 Molecular In Vitro Diagnostic Examinations—Specifications for Pre-Examination Processes in Metabolomics in Urine, Venous Blood Serum and Plasma. Available online: https://www.iso.org/obp/ui/#iso:std:iso:23118:ed-1:v1:en (accessed on 4 June 2021).
- Bruzzone, C.; Bizkarguenaga, M.; Gil-Redondo, R.; Diercks, T.; Arana, E.; García de Vicuña, A.; Seco, M.; Bosch, A.; Palazón, A.; San Juan, I.; et al. SARS-CoV-2 Infection Dysregulates the Metabolomic and Lipidomic Profiles of Serum. iScience 2020, 23, 101645. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- van Velzen, E.J.J.; Westerhuis, J.A.; van Duynhoven, J.P.M.; van Dorsten, F.A.; Hoefsloot, H.C.J.; Jacobs, D.M.; Smit, S.; Draijer, R.; Kroner, C.I.; Smilde, A.K. Multilevel Data Analysis of a Crossover Designed Human Nutritional Intervention Study. J. Proteome Res. 2008, 7, 4483–4491. [Google Scholar] [CrossRef]
- Westerhuis, J.A.; van Velzen, E.J.; Hoefsloot, H.C.; Smilde, A.K. Multivariate Paired Data Analysis: Multilevel PLSDA versus OPLSDA. Metabolomics 2010, 6, 119–128. [Google Scholar] [CrossRef] [Green Version]
- Cortes, C.; Vapnik, V. Support-Vector Networks. J. Mach. Learn. Res. 1995, 20, 273–297. [Google Scholar] [CrossRef]
- Jiménez, B.; Holmes, E.; Heude, C.; Tolson, R.F.; Harvey, N.; Lodge, S.L.; Chetwynd, A.J.; Cannet, C.; Fang, F.; Pearce, J.T.M.; et al. Quantitative Lipoprotein Subclass and Low Molecular Weight Metabolite Analysis in Human Serum and Plasma by 1H NMR Spectroscopy in a Multilaboratory Trial. Anal. Chem. 2018, 90, 11962–11971. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, X.-W.; Liu, D.-B.; Han, C.-Z.; Du, L.-L.; Jing, J.-X.; Wang, Y. Lipid Levels in Serum and Cancerous Tissues of Colorectal Cancer Patients. World J. Gastroenterol. 2014, 20, 8646–8652. [Google Scholar] [CrossRef] [PubMed]
- Mayengbam, S.S.; Singh, A.; Pillai, A.D.; Bhat, M.K. Influence of Cholesterol on Cancer Progression and Therapy. Transl. Oncol. 2021, 14, 101043. [Google Scholar] [CrossRef] [PubMed]

| Whole Sample (N = 41) | Stratified by Progression Status | Stratified by Chemotherapy Treatment | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Not Relapsed (N = 33) | Relapsed (N = 8) | p-Value | Capecitabine (N = 9) | XELOX (N = 10) | No CT (N = 22) | p-Value | |||
| Age at study entry | Median (min; max) | 73 (51;92) | 71 (51;92) | 78 (68;86) | 0.032 | 77 (68;86) | 65 (51;72) | 78 (54;92) | 0.001 |
| Sex | F | 21 (51%) | 18 (55%) | 3 (38%) | 0.454 | 2 (22%) | 7 (70%) | 12 (55%) | 0.109 |
| M | 20 (49%) | 15 (45%) | 5 (62%) | 7 (78%) | 3 (30%) | 10 (45%) | |||
| ECOG PS | PS 0 | 29 (71%) | 23 (70%) | 6 (75%) | 1 | 7 (78%) | 10 (100%) | 12 (55%) | 0.102 |
| PS 1 | 8 (20%) | 7 (21%) | 1 (12%) | 1 (11%) | 0 (0%) | 7 (32%) | |||
| PS 2 | 4 (10%) | 3 (9%) | 1 (12%) | 1 (11%) | 0 (0%) | 3 (14%) | |||
| pT | pT1 | 6 (15%) | 6 (18%) | 0 (0%) | 0.086 | 0 (0%) | 1 (10%) | 5 (23%) | 0.477 |
| pT2 | 8 (20%) | 8 (24%) | 0 (0%) | 1 (11%) | 1 (10%) | 6 (27%) | |||
| pT3 | 23 (56%) | 17 (52%) | 6 (75%) | 7 (78%) | 7 (70%) | 9 (41%) | |||
| pT4 | 4 (10%) | 2 (6%) | 2 (25%) | 1 (11%) | 1 (10%) | 2 (9%) | |||
| N | N0 | 24 (59%) | 23 (70%) | 1 (12%) | 0.005 | 3 (33%) | 3 (30%) | 18 (82%) | 0.005 |
| N+ | 17 (41%) | 10 (30%) | 7 (88%) | 6 (67%) | 7 (70%) | 4 (18%) | |||
| Stage risk | Stage I | 11 (27%) | 11 (33%) | 0 (0%) | 0.035 | 0 (0%) | 0 (0%) | 11 (50%) | 0.002 |
| Stage II Low risk | 2 (5%) | 2 (6%) | 0 (0%) | 0 (0%) | 0 (0%) | 2 (9%) | |||
| Stage II High risk | 11 (27%) | 10 (30%) | 1 (12%) | 3 (33%) | 3 (30%) | 5 (23%) | |||
| Stage III | 17 (41%) | 10 (30%) | 7 (88%) | 6 (67%) | 7 (70%) | 4 (18%) | |||
| Grading | G1 | 2 (5%) | 1 (3%) | 1 (12%) | 0.168 | 1 (11%) | 1 (10%) | 0 (0%) | 0.205 |
| G2 | 19 (48%) | 17 (53%) | 2 (25%) | 2 (22%) | 4 (40%) | 13 (62%) | |||
| G3 | 17 (42%) | 13 (41%) | 4 (50%) | 5 (56%) | 5 (50%) | 7 (33%) | |||
| G4 | 2 (5%) | 1 (3%) | 1 (12%) | 1 (11%) | 0 (0%) | 1 (5%) | |||
| NA | 1 | 1 | 0 | 0 | 0 | 1 | |||
| Localization | Left-sided | 13 (32%) | 12 (36%) | 1 (12%) | 0.398 | 3 (33%) | 6 (60%) | 4 (18%) | 0.07 |
| Right-sided | 28 (68%) | 21 (64%) | 7 (88%) | 6 (67%) | 4 (40%) | 18 (82%) | |||
| Comorbidities | No com. | 13 (32%) | 8 (24%) | 5 (62%) | 0.111 | 4 (44%) | 3 (30%) | 6 (27%) | 0.519 |
| No vascular com. | 8 (20%) | 8 (24%) | 0 (0%) | 0 (0%) | 3 (30%) | 5 (23%) | |||
| Vascular com. | 20 (49%) | 17 (52%) | 3 (38%) | 5 (56%) | 4 (40%) | 11 (50%) | |||
| MSI | Instable | 1 (11%) | 1 (14%) | 0 (0%) | 1 | 0 (0%) | 0 (0%) | 1 (33%) | 0.278 |
| MSI | 1 (11%) | 1 (14%) | 0 (0%) | 0 (0%) | 0 (0%) | 1 (33%) | |||
| Stable | 7 (78%) | 5 (71%) | 2 (100%) | 2 (100%) | 4 (100%) | 1 (33%) | |||
| NA | 32 | 26 | 6 | 7 | 6 | 19 | |||
| CDX2 | Positive | 1 (100%) | 0 (0%) | 1 (100%) | - | 1 (100%) | 0 (0%) | 0 (0%) | - |
| NA | 40 | 33 | 7 | 8 | 10 | 22 | |||
| KRAS | Mutated | 5 (29%) | 1 (11%) | 4 (50%) | 0.131 | 3 (50%) | 0 (0%) | 2 (67%) | 0.042 |
| WT | 12 (71%) | 8 (89%) | 4 (50%) | 3 (50%) | 8 (100%) | 1 (33%) | |||
| NA | 24 | 24 | 0 | 3 | 2 | 19 | |||
| NRAS | WT | 13 (100%) | 8 (100%) | 5 (100%) | - | 4 (100%) | 8 (100%) | 1 (100%) | - |
| NA | 28 | 25 | 3 | 5 | 2 | 21 | |||
| BRAF | Mutated | 4 (24%) | 3 (33%) | 1 (12%) | 0.576 | 1 (17%) | 3 (38%) | 0 (0%) | 0.461 |
| WT | 13 (76%) | 6 (67%) | 7 (88%) | 5 (83%) | 5 (62%) | 3 (100%) | |||
| NA | 24 | 24 | 0 | 3 | 2 | 19 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vignoli, A.; Mori, E.; Di Donato, S.; Malorni, L.; Biagioni, C.; Benelli, M.; Calamai, V.; Cantafio, S.; Parnofiello, A.; Baraghini, M.; et al. Exploring Serum NMR-Based Metabolomic Fingerprint of Colorectal Cancer Patients: Effects of Surgery and Possible Associations with Cancer Relapse. Appl. Sci. 2021, 11, 11120. https://doi.org/10.3390/app112311120
Vignoli A, Mori E, Di Donato S, Malorni L, Biagioni C, Benelli M, Calamai V, Cantafio S, Parnofiello A, Baraghini M, et al. Exploring Serum NMR-Based Metabolomic Fingerprint of Colorectal Cancer Patients: Effects of Surgery and Possible Associations with Cancer Relapse. Applied Sciences. 2021; 11(23):11120. https://doi.org/10.3390/app112311120
Chicago/Turabian StyleVignoli, Alessia, Elena Mori, Samantha Di Donato, Luca Malorni, Chiara Biagioni, Matteo Benelli, Vanessa Calamai, Stefano Cantafio, Annamaria Parnofiello, Maddalena Baraghini, and et al. 2021. "Exploring Serum NMR-Based Metabolomic Fingerprint of Colorectal Cancer Patients: Effects of Surgery and Possible Associations with Cancer Relapse" Applied Sciences 11, no. 23: 11120. https://doi.org/10.3390/app112311120
APA StyleVignoli, A., Mori, E., Di Donato, S., Malorni, L., Biagioni, C., Benelli, M., Calamai, V., Cantafio, S., Parnofiello, A., Baraghini, M., Garzi, A., Monte, F. D., Romagnoli, D., Migliaccio, I., Luchinat, C., Tenori, L., & Biganzoli, L. (2021). Exploring Serum NMR-Based Metabolomic Fingerprint of Colorectal Cancer Patients: Effects of Surgery and Possible Associations with Cancer Relapse. Applied Sciences, 11(23), 11120. https://doi.org/10.3390/app112311120











