Techno-Economic Evaluation of Hand Sanitiser Production Using Oil Palm Empty Fruit Bunch-Based Bioethanol by Simultaneous Saccharification and Fermentation (SSF) Process
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Kinetic Evaluation of Fermentation
2.1.1. Preparation of Materials
2.1.2. OPEFB Pretreatment and Analysis
2.1.3. Simultaneous Saccharification and Fermentation
2.1.4. Analysis of Fermentation Products
2.1.5. Reaction Constants Determination
- The kinetic equation for yeast cell formation:
- The kinetic equation for product formation (bioethanol):
- The kinetic equation for the residual substrate:
2.2. Hand Sanitiser Plant Design
3. Results and Discussion
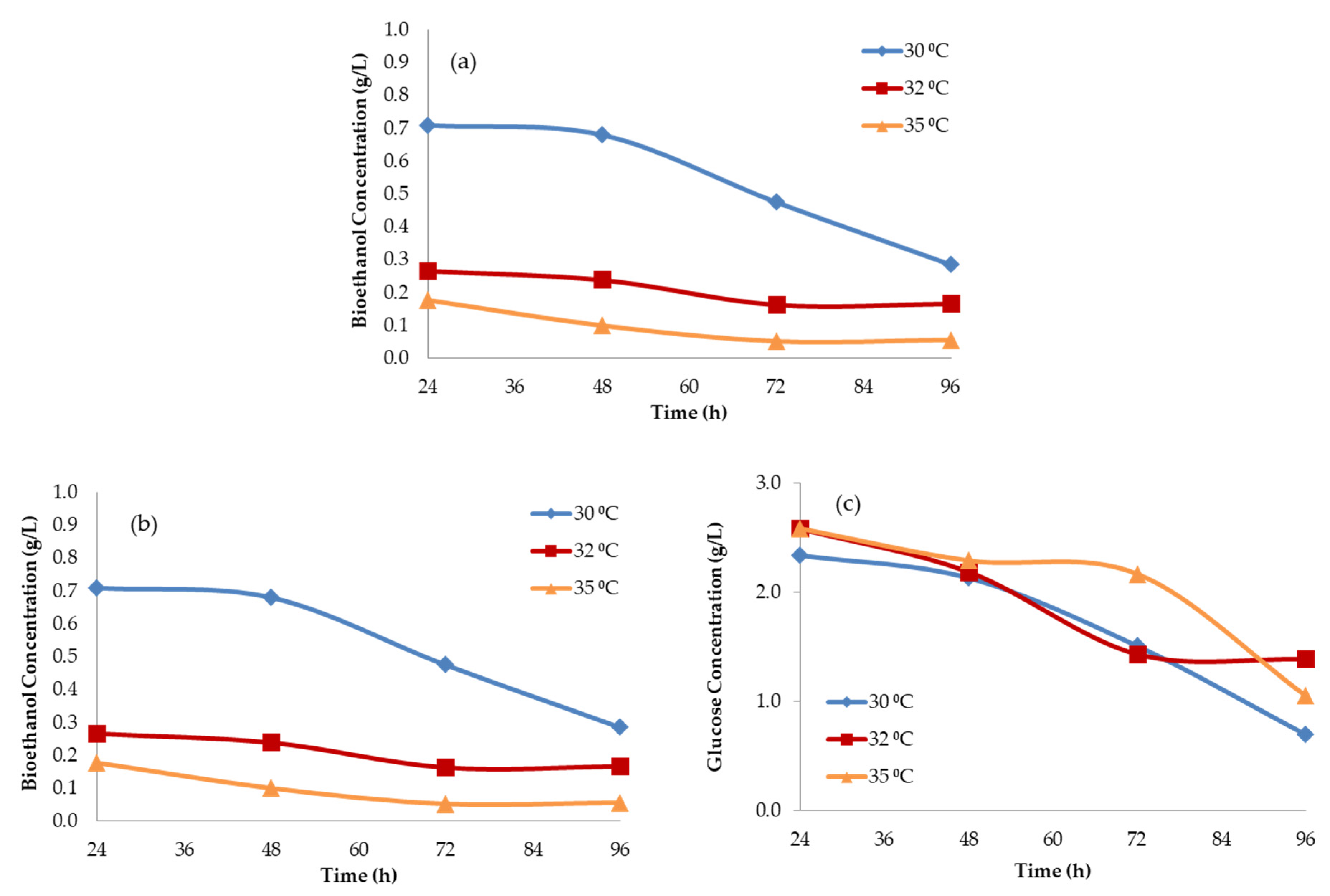
3.1. Kinetic Parameters of Fermentation
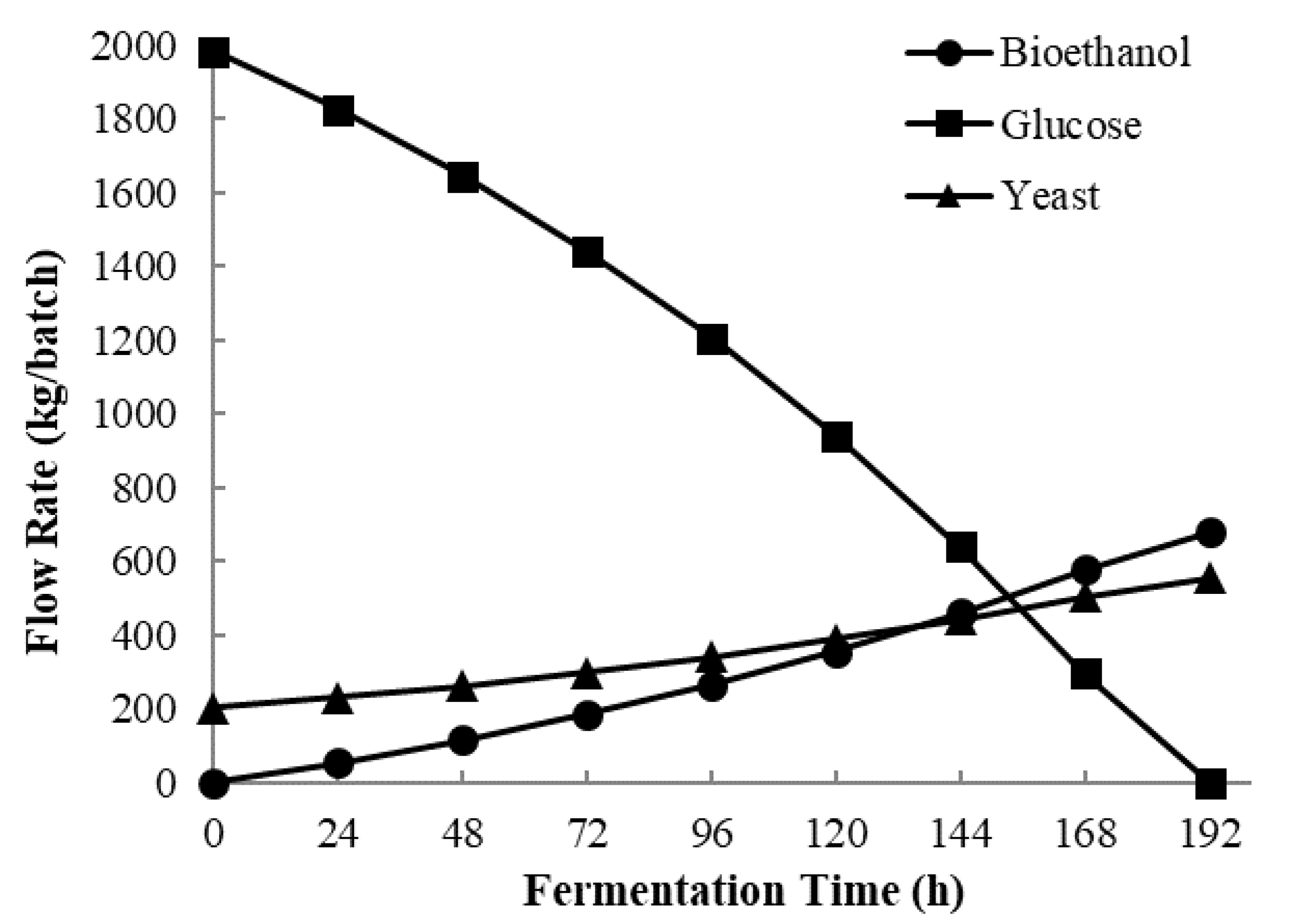
3.2. Process Simulation
3.3. Cost and Economic Parameters
3.4. N/A: Not Available
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization (WHO). (27 April 2020). WHO Timeline-COVID-19. Available online: https://www.who.int/news-room/detail/27-04-2020-who-timeline---covid-19 (accessed on 27 May 2020).
- Kampf, G.; Todt, D.; Pfaender, S.; Steinmann, E. Persistence of Coronaviruses on Inanimate Surfaces and Their Inactivation with Biocidal Agents. J. Hosp. Infect. 2020, 104, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Rai, H.; Knighton, S.; Zabarsky, T.F.; Donskey, C.J. Comparison of Ethanol Hand Sanitizer Versus Moist Towelette Packets for Mealtime Patient Hand Hygiene. Am. J. Infect. Control. 2017, 45, 1033–1034. [Google Scholar] [CrossRef] [PubMed]
- Foddai, A.C.G.; Grant, I.R.; Dean, M. Efficacy of Instant Hand Sanitizers against Foodborne Pathogens Compared with Hand Washing with Soap and Water in Food Preparation Settings: A Systematic Review. J. Food Prot. 2016, 79, 1040–1054. [Google Scholar] [CrossRef] [PubMed]
- Jing, J.L.J.; Yi, T.P.; Bose, R.J.C.; McCarthy, J.R.; Tharmalingam, N.; Madheswaran, T. Hand Sanitizers: A Review on Formulation Aspects, Adverse Effects, and Regulations. Int. J. Environ. Res. Public Health 2020, 17, 3326. [Google Scholar] [CrossRef] [PubMed]
- Golin, A.P.; Choi, D.; Ghahary, A. Hand Santizers: A Review of Ingredients, Mechanisms of Action, Modes of Delivery, and Efficacy against Coronaviruses. Am. J. Infect. Control. 2020, in press. [Google Scholar] [CrossRef]
- WHO. (April 2010). Guide to Local Production: WHO-Recommended Handrub Formulations. Available online: https://www.who.int/gpsc/5may/Guide_to_Local_Production.pdf?ua=1 (accessed on 26 May 2020).
- Rastogi, M.; Shrivastava, S. Recent Advances in Second Generation Bioethanol Production: An Insight to Pretreatment, Saccharification and Fermentation Processes. Renew. Sustain. Energy Rev. 2017, 80, 330–340. [Google Scholar] [CrossRef]
- Rosales-Calderon, O.; Arantes, V. A Review on Commercial-Scale High-Value Products That Can be Produced alongside Cellulosic Ethanol. Biotechnol. Biofuels 2019, 12, 240. [Google Scholar] [CrossRef]
- Su, T.; Zhao, D.; Khodadadi, M.; Len, C. Lignocellulosic Biomass for Bioethanol: Recent Advances, Technology Trends, and Barriers to Industrial Development. Curr. Opin. Green Sustain. Chem. 2020, 24, 56–60. [Google Scholar] [CrossRef]
- Samsuri, M.; Gozan, M.; Mardias, R.; Baiquni, M.; Hermansyah, H.; Wijanarko, A.; Prasetya, B.; Nasikin, M. Pemanfaatan Sellulosa Bagas untuk Produksi Ethanol Melalui Sakarifikasi dan Fermentasi Serentak dengan Enzim Xylanase. Makara 2007, 11, 17–24. [Google Scholar] [CrossRef]
- Arlofa, N.; Gozan, M.; Pradita, T.; Jufri, M. Optimization of Bioethanol Production from Durian Skin by Encapsulation of Saccharomyces cerevisiae. Asian J. Chem. 2019, 31, 1027–1033. [Google Scholar] [CrossRef]
- Darmawan, M.A.; Hermawan, Y.A.; Samsuri, M.; Gozan, M. Conversion of Paper Waste to Bioethanol using Selected Enzyme Combination (Cellulase and Cellobiase) Through Simultaneous Saccharification and Fermentation. In Proceedings of the 11th Regional Conference on Chemical Engineering (RCChE 2018), Yogyakarta, Indonesia, 7–8 November 2019; Teguh, A., Imam, P., Rochmadi, N., Rofiqoh Eviana, P., Eds.; American Institute of Physics Inc.: College Park, MD, USA, 2019. [Google Scholar]
- Sahlan, M.; Hermansyah, H.; Wijanarko, A.; Gozan, M.; Lischer, K.; Ahmudi, A.; Pujianto, P. Ethanol Production by Encapsulated Rhyzopus oryzae from Oil Palm Empty Fruit Bunch. Evergreen 2020, 7, 92–96. [Google Scholar] [CrossRef]
- Koppram, R.; Nielsen, F.; Albers, E. Simultaneous Saccharification and Co-Fermentation for Bioethanol Production using Corncobs at Lab, PDU and Demo Scales. Biotechnol. Biofuels 2013, 6. [Google Scholar] [CrossRef] [PubMed]
- Khuong, L.D.; Kondo, R.; De Leon, R.; Anh, T.K.; Shimizu, K.; Kamei, I. Bioethanol Production from Alkaline-Pretreated Sugarcane Bagasse by Consolidated Bioprocessing using Phlebia sp. MG-60. Int. Biodeterior. Biodegrad. 2014, 88, 62–68. [Google Scholar] [CrossRef]
- Lennartsson, P.R.; Erlandsson, P.; Taherzadeh, M.J. Integration of the First and Second Generation Bioethanol Processes and the Importance of By-Products. Bioresour. Technol. 2014, 165, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Larroche, C.; Dussap, C.G. Comprehensive Assessment of 2G Bioethanol Production. Bioresour. Technol. 2020, 313, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Shinoj, S.; Visvanathan, R.; Panigrahi, S.; Kochubabu, M. Oil Palm Fiber (OPF) and Its Composites: A Review. Ind. Crops Prod. 2011, 33, 7–22. [Google Scholar] [CrossRef]
- Purwandari, F.A.; Sanjaya, A.P.; Millati, R.; Cahyanto, M.N.; Horváth, I.S.; Niklasson, C.; Taherzadeh, M.J. Pretreatment of Oil Palm Empty Fruit Bunch (OPEFB) by N-methylmorpholine-N-oxide (NMMO) for Biogas Production: Structural Changes and Digestion Improvement. Bioresour. Technol. 2013, 128, 461–466. [Google Scholar] [CrossRef]
- Medina, J.D.C.; Woiciechowski, A.; Filho, A.Z.; Nigam, P.S.; Ramos, L.P.; Soccol, C.R. Steam Explosion Pretreatment of Oil Palm Empty Fruit Bunches (EFB) using Autocatalytic Hydrolysis: A Biorefinery Approach. Bioresour. Technol. in press. [CrossRef]
- Panjaitan, J.R.H.; Gozan, M. Formic Acid Production from Palm Oil Empty Fruit Bunches. Int. J. Appl. Eng. Res. 2017, 12, 4382–4390. [Google Scholar]
- Harahap, A.F.P.; Rahman, A.A.; Sadrina, I.N.; Gozan, M. Production of Formic Acid from Oil Palm Empty Fruit Bunch via Dilute Acid Hydrolysis by Response Surface Methodology. In Proceedings of the Broad Exposure to Science and Technology 2019 (BEST2019), Bali, Indonesia, 7–8 August 2019; IOP Publishing Ltd.: Bristol, UK, 2019. [Google Scholar]
- Gozan, M.; Panjaitan, J.R.H.; Tristantini, D.; Alamsyah, R.; Yoo, Y.J. Evaluation of Separate and Simultaneous Kinetic Parameters for Levulinic Acid and Furfural Production from Pretreated Palm Oil Empty Fruit Bunches. Int. J. Chem. Eng. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- McInnes, A. A Comparison of Leading Palm Oil Certification Standards; Forest Peoples Programme: Moreton-in-Marsh, UK, 2017; pp. 5–37. [Google Scholar]
- Bernhard, T. Definition and Scope of Systems Engineering. In Systems Engineering: Principles and Practice of Computer-Based Systems Engineering; John Wiley & Sons Ltd.: Chichester, West Sussex, UK, 1993; pp. 1–23. [Google Scholar]
- Dimian, A.; Bildea, C.; Kiss, A. Integrated Design and Simulation of Chemical Processes, 2nd ed.; Elsevier Science: Amsterdam, The Netherland, 2014; pp. 35–71. [Google Scholar]
- Feibel, B.J. Investment Performance Measurement, 1st ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2003; pp. 215–298. [Google Scholar]
- Khor, C.; Elkamel, A.; Anderson, W. Incorporating the Systems Approach in Future Undergraduate Chemical Engineering Education Curriculum: Illustration via Computer-Aided Process Simulation Laboratory Exercises. World Rev. Sci. Technol. Sustain. Dev. 2008, 5, 402–413. [Google Scholar] [CrossRef]
- Seider, W.D.; Seader, J.D.; Lewin, D.R.; Widagdo, S. Product and Process Design Principles: Synthesis, Analysis, and Evaluation, 3rd ed.; John Wiley & Sons, Inc.: New York, NY, USA, 2004; pp. 110–147. [Google Scholar]
- Zhang, K.; Pei, Z.; Wang, D. Organic Solvent Pretreatment of Lignocellulosic Biomass for Biofuels and Biochemicals: A Review. Bioresour. Technol. 2016, 199, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Zulkiple, N.; Maskat, M.Y.; Hassan, O. Pretreatment of Oil Palm Empty Fruit Fiber (OPEFB) with Aqueous Ammonia for High Production of Sugar. Procedia Chem. 2016, 18, 155–161. [Google Scholar] [CrossRef]
- Badan Standardisasi Nasional (BSN). Standar Nasional Indonesia (Indonesian National Standardization) SNI 0444: 2009 Pulp—Cara Uji Kadar Selulosa Alfa, Beta dan Gamma; BSN: Jakarta, Indonesia, 2009. [Google Scholar]
- Badan Standardisasi Nasional (BSN). Standar Nasional Indonesia (Indonesian National Standardization) SNI 14-1304-1989 Cara Uji Kadar Pentosan Pulp Kayu; BSN: Jakarta, Indonesia, 1989. [Google Scholar]
- Badan Standardisasi Nasional (BSN). Standar Nasional Indonesia (Indonesian National Standardization) SNI 0492:2008 Pulp dan Kayu—Cara Uji Kadar Lignin-Metode Klason; BSN: Jakarta, Indonesia, 2008. [Google Scholar]
- Badan Standardisasi Nasional (BSN). Standar Nasional Indonesia (Indonesian National Standardization) SNI 0442: 2009 Kertas, Karton, dan Pulp-Cara Uji Kadar Abu pada 525 °C; BSN: Jakarta, Indonesia, 2009. [Google Scholar]
- Fogler, H.S. Elements of Chemical Reaction Engineering, 4th ed.; Prentice Hall: Laflin, PA, USA, 2006; pp. 91–98. [Google Scholar]
- Fengel, D.; Wegener, G. Wood: Chemistry, Ultrastructure, Reactions, 2nd ed.; Walter de Gruyter: Berlin, Germany, 1989; pp. 162–197. [Google Scholar]
- Amenaghawon, N.A.; Okieimen, C.O.; Ogbeide, S.E. Kinetic Modelling of Ethanol Inhibition during Alcohol Fermentation of Corn Stover using Saccharomyces cerevisiae. Int. J. Eng. Res. Appl. 2012, 2, 798–803. [Google Scholar]
- Dodic, J.M.; Vucurovic, D.G.; Dodic, S.N.; Grahovac, J.A.; Popov, S.D.; Nedeljkovic, N.M. Kinetic Modelling of Batch Ethanol Production from Sugar Beet Raw Juice. Appl. Energy 2012, 99, 192–197. [Google Scholar] [CrossRef]
- Phisalaphong, M.; Srirattana, N.; Tanthapanichakoon, W. Mathematical Modeling to Investigate Temperature Effect on Kinetic Parameters of Ethanol Fermentation. Biochem. Eng. J. 2006, 28, 36–43. [Google Scholar] [CrossRef]
- Peters, M.S.; Timmerhaus, K.D. Plant Design and Economics for Chemical Engineers, 4th ed.; McGraw-Hill: New York, NY, USA, 1991; pp. 120–252. [Google Scholar]
- Konda, N.M.; Shi, J.; Singh, S.; Blanch, H.W.; Simmons, B.A.; Marcuschamer, D.K. Understanding Cost Drivers and Economic Potential of Two Variants of Ionic Liquid Pretreatment for Cellulosic Biofuel Production. Biotechnol. Biofuels 2014, 7, 1–11. [Google Scholar] [CrossRef]
- Damodaran, A. (January 2020). Margins by Sector (US). Available online: http://pages.stern.nyu.edu/~adamodar/New_Home_Page/datafile/margin.html (accessed on 10 July 2020).




| Reaction | Kinetics Parameters Input |
|---|---|
| Glucose → Yeast cell | |
| Input: | |
| α = 1 | |
| μ max = μ max | |
| (S1 − Term) = 1 | |
| (S2 − Term) = | |
| β = − kd | |
| (B − Term) = Cc | |
| Glucose → Ethanol | |
| Input: | |
| α = (ethanol mass/cell mass) | |
| μ max = μ max | |
| (S1 − Term) = 1 | |
| (S2 − Term) = | |
| β = 0 | |
| (B − Term) = Cc | |
| Glucose → Glucose(residue) | |
| Input: | |
| α = | |
| μ max = μ max | |
| (S1 − Term) = 1 | |
| (S2 − Term) = | |
| β = − m | |
| (B − Term) = Cc |
| Parameters | Value |
|---|---|
| Year of analysis | 2020 |
| Construction period | 30 months |
| Start-up period | 4 months |
| Project lifetime | 15 years |
| Interest rate | 7.0% |
| OPEFB capacity | 2000 kg/batch |
| Labour wage | $0.92/h |
| Parameters | Composition (%) |
|---|---|
| Ash | 1.51 |
| Lignin | 19.39 |
| Cellulose | 63.97 |
| Hemicellulose | 10.58 |
| Kinetic Parameters | Temperature (°C) | ||
|---|---|---|---|
| 30 | 32 | 35 | |
| µmax (h−1) | 0.009 | 0.013 | 0.018 |
| ks (g/dm3) | 0.004 | 0.010 | 0.025 |
| kd (h−1) | 0.009 | 0.009 | 0.213 |
| Total Plant Direct Cost (TPDC) | |
|---|---|
| 1. Equipment purchase cost | 101,000 |
| 2. Installation | 33,000 |
| 3. Process piping | 35,000 |
| 4. Instrumentation | 41,000 |
| 5. Insulation | 3000 |
| 6. Electrical | 10,000 |
| 7. Buildings | 46,000 |
| 8. Yard improvement | 15,000 |
| 9. Auxiliary facilities | 41,000 |
| TPDC | 325,000 |
| Total Plant Indirect Cost (TPIC) | |
| 10. Engineering | 81,000 |
| 11. Construction | 114,000 |
| TPIC | 195,000 |
| Total Plant Cost (TPC = TPDC + TPIC) | |
| TPC | 520,000 |
| Contractor’s Fee and Contingency (CFC) | |
| 12. Contractor’s Fee | 26,000 |
| 13. Contingency | 52,000 |
| CFC | 78,000 |
| Direct Fixed Capital Cost (DFC = TPC + CFC) | |
| DFC | 598,000 |
| Material/Utility | Unit Cost ($) | Annual Amount | Reference Unit | Annual Cost ($) |
|---|---|---|---|---|
| Air | 0.00 | 5,914,956 | kg | 0 |
| Cellulase | 2.50 | 4058 | kg | 10,146 |
| Citric Acid | 0.63 | 2866 | kg | 1791 |
| Glycerol 98% | 0.60 | 471 | kg | 283 |
| H2O2 3% | 0.40 | 1356 | kg | 542 |
| OPEFB | 0.01 | 80,000 | kg | 488 |
| Sodium Citrate | 0.76 | 5431 | kg | 4127 |
| Sodium Hydroxide | 0.43 | 80,000 | kg | 34,000 |
| Water | 0.01 | 4600 | MT | 46 |
| Yeast | 1.95 | 8158 | kg | 15,907 |
| Standard Power | 0.10 | 588,067 | kW-h | 58,807 |
| Low Pressure Steam | 3.34 | 1983 | MT | 6624 |
| High Pressure Steam | 5.78 | 1119 | MT | 6468 |
| Cooling Water | 0.05 | 186,277 | MT | 9314 |
| Chilled Water | 0.19 | 78,573 | MT | 14,536 |
| Product Price (USD/L) | IRR (%) | PBP (Year) | Gross Margin (%) | ROI (%) | NPV (USD) |
|---|---|---|---|---|---|
| 2 | N/A | N/A | −295 | −26 | −1,824,000 |
| 4 | N/A | N/A | −97 | −14 | −1,278,000 |
| 6 | N/A | N/A | −32 | −3 | −732,000 |
| 8 | 0.4 | 10.9 | 1 | 9 | −224,000 |
| 10 | 9.8 | 6.1 | 21 | 16 | 108,000 |
| 12 | 17.0 | 4.3 | 34 | 24 | 440,000 |
| 14 | 23.2 | 3.3 | 44 | 31 | 772,000 |
| 16 | 28.7 | 2.6 | 51 | 38 | 1,104,000 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harahap, A.F.P.; Panjaitan, J.R.H.; Curie, C.A.; Ramadhan, M.Y.A.; Srinophakun, P.; Gozan, M. Techno-Economic Evaluation of Hand Sanitiser Production Using Oil Palm Empty Fruit Bunch-Based Bioethanol by Simultaneous Saccharification and Fermentation (SSF) Process. Appl. Sci. 2020, 10, 5987. https://doi.org/10.3390/app10175987
Harahap AFP, Panjaitan JRH, Curie CA, Ramadhan MYA, Srinophakun P, Gozan M. Techno-Economic Evaluation of Hand Sanitiser Production Using Oil Palm Empty Fruit Bunch-Based Bioethanol by Simultaneous Saccharification and Fermentation (SSF) Process. Applied Sciences. 2020; 10(17):5987. https://doi.org/10.3390/app10175987
Chicago/Turabian StyleHarahap, Andre Fahriz Perdana, Jabosar Ronggur Hamonangan Panjaitan, Catia Angli Curie, Muhammad Yusuf Arya Ramadhan, Penjit Srinophakun, and Misri Gozan. 2020. "Techno-Economic Evaluation of Hand Sanitiser Production Using Oil Palm Empty Fruit Bunch-Based Bioethanol by Simultaneous Saccharification and Fermentation (SSF) Process" Applied Sciences 10, no. 17: 5987. https://doi.org/10.3390/app10175987
APA StyleHarahap, A. F. P., Panjaitan, J. R. H., Curie, C. A., Ramadhan, M. Y. A., Srinophakun, P., & Gozan, M. (2020). Techno-Economic Evaluation of Hand Sanitiser Production Using Oil Palm Empty Fruit Bunch-Based Bioethanol by Simultaneous Saccharification and Fermentation (SSF) Process. Applied Sciences, 10(17), 5987. https://doi.org/10.3390/app10175987





