Observation of Potential Contaminants in Processed Biomass Using Fourier Transform Infrared Spectroscopy
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Isolation of Cellulose, Hemicellulose, and Lignin
2.3. Fourier Transform Infrared (FTIR) Analysis
3. Results and Discussion
3.1. Major Components of Biomass
3.2. Commonly Used Pretreatment and Preprocessing Solvents
3.3. Acids and Alkalis
3.4. Ionic Liquids
3.5. Enzymes
3.6. Biomass-Derived Chemicals
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pu, Y.; Cao, S.; Ragauskas, A.J. Application of quantitative 31P NMR in biomass lignin and biofuel precursors characterization. Energy Environ. Sci. 2011, 4, 3154–3166. [Google Scholar] [CrossRef]
- Yoo, C.G.; Pu, Y.; Li, M.; Ragauskas, A.J. Elucidating Structural Characteristics of Biomass using Solution-State 2D NMR with a Mixture of Deuterated Dimethylsulfoxide and Hexamethylphosphoramide. ChemSusChem 2016, 9, 1090–1095. [Google Scholar] [CrossRef] [PubMed]
- Yoo, C.G.; Yang, Y.; Pu, Y.; Meng, X.; Muchero, W.; Yee, K.L.; Thompson, O.A.; Rodriguez, M.; Bali, G.; Engle, N.L. Insights of biomass recalcitrance in natural Populus trichocarpa variants for biomass conversion. Green Chem. 2017, 19, 5467–5478. [Google Scholar] [CrossRef]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of structural carbohydrates and lignin in biomass. Lab. Anal. Proced. 2010, 1617, 1–16. [Google Scholar]
- Jung, S.; Foston, M.; Kalluri, U.C.; Tuskan, G.A.; Ragauskas, A.J. 3D chemical image using TOF-SIMS revealing the biopolymer component spatial and lateral distributions in biomass. Angew. Chem. 2012, 124, 12171–12174. [Google Scholar] [CrossRef]
- Tolbert, A.K.; Yoo, C.G.; Ragauskas, A.J. Understanding the Changes to Biomass Surface Characteristics after Ammonia and Organosolv Pretreatments by Using Time-of-Flight Secondary-Ion Mass Spectrometry (TOF-SIMS). ChemPlusChem 2017, 82, 686–690. [Google Scholar] [CrossRef]
- Sannigrahi, P.; Kim, D.H.; Jung, S.; Ragauskas, A. Pseudo-lignin and pretreatment chemistry. Energy Environ. Sci. 2011, 4, 1306–1310. [Google Scholar] [CrossRef]
- Figueira, M.; Volesky, B.; Mathieu, H. Instrumental analysis study of iron species biosorption by Sargassum biomass. Environ. Sci. Technol. 1999, 33, 1840–1846. [Google Scholar] [CrossRef]
- Kok, M.V.; Özgür, E. Thermal analysis and kinetics of biomass samples. Fuel Process. Technol. 2013, 106, 739–743. [Google Scholar] [CrossRef]
- Pu, Y.; Meng, X.; Yoo, C.G.; Li, M.; Ragauskas, A.J. Analytical methods for biomass characterization during pretreatment and bioconversion. In Valorization of Lignocellulosic Biomass in a Biorefinery: From Logistics to Environmental and Performance Impact; Kumar, R., Ed.; Nova Science Publishers: New York, NY, USA, 2016; pp. 37–78. [Google Scholar]
- Acquah, G.E.; Via, B.K.; Fasina, O.O.; Eckhardt, L.G. Rapid quantitative analysis of forest biomass using fourier transform infrared spectroscopy and partial least squares regression. J. Anal. Methods Chem. 2016, 2016, 1839598. [Google Scholar] [CrossRef]
- Perkins, W. Fourier transform infrared spectroscopy. Part II. Advantages of FT-IR. J. Chem. Educ. 1987, 64, A269. [Google Scholar] [CrossRef]
- Di Fidio, N.; Raspolli Galletti, A.M.; Fulignati, S.; Licursi, D.; Liuzzi, F.; De Bari, I.; Antonetti, C. Multi-Step Exploitation of Raw Arundo donax L. for the Selective Synthesis of Second-Generation Sugars by Chemical and Biological Route. Catalysts 2020, 10, 79. [Google Scholar] [CrossRef]
- Licursi, D.; Antonetti, C.; Fulignati, S.; Corsini, A.; Boschi, N.; Galletti, A.M.R. Smart valorization of waste biomass: Exhausted lemon peels, coffee silverskins and paper wastes for the production of levulinic acid. Chem. Eng. Trans. 2018, 65, 637. [Google Scholar]
- Lara-Serrano, M.; Morales-delaRosa, S.; Campos-Martín, J.M.; Fierro, J.L.G. Fractionation of Lignocellulosic Biomass by Selective Precipitation from Ionic Liquid Dissolution. Appl. Sci. 2019, 9, 1862. [Google Scholar] [CrossRef]
- Shen, X.J.; Wen, J.L.; Mei, Q.Q.; Chen, X.; Sun, D.; Yuan, T.Q.; Sun, R.C. Facile fractionation of lignocelluloses by biomass-derived deep eutectic solvent (DES) pretreatment for cellulose enzymatic hydrolysis and lignin valorization. Green Chem. 2019, 21, 275–283. [Google Scholar] [CrossRef]
- Agbor, V.B.; Cicek, N.; Sparling, R.; Berlin, A.; Levin, D.B. Biomass pretreatment: Fundamentals toward application. Biotechnol. Adv. 2011, 29, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Yoo, C.G.; Pu, Y.; Ragauskas, A.J. Ionic liquids: Promising green solvents for lignocellulosic biomass utilization. Curr. Opin. Green Sustain. Chem. 2017, 5, 5–11. [Google Scholar] [CrossRef]
- Nguyen, T.Y.; Cai, C.M.; Osman, O.; Kumar, R.; Wyman, C.E. CELF pretreatment of corn stover boosts ethanol titers and yields from high solids SSF with low enzyme loadings. Green Chem. 2016, 18, 1581–1589. [Google Scholar] [CrossRef]
- Yang, B.; Dai, Z.; Ding, S.-Y.; Wyman, C.E. Enzymatic hydrolysis of cellulosic biomass. Biofuels 2011, 2, 421–449. [Google Scholar] [CrossRef]
- Rasmussen, H.; Sørensen, H.R.; Meyer, A.S. Formation of degradation compounds from lignocellulosic biomass in the biorefinery: Sugar reaction mechanisms. Carbohydr. Res. 2014, 385, 45–57. [Google Scholar] [CrossRef]
- Li, M.; Pu, Y.; Yoo, C.G.; Gjersing, E.; Decker, S.R.; Doeppke, C.; Shollenberger, T.; Tschaplinski, T.J.; Engle, N.L.; Sykes, R.W. Study of traits and recalcitrance reduction of field-grown COMT down-regulated switchgrass. Biotechnol. Biofuels 2017, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Faix, O. Fourier transform infrared spectroscopy. In Methods in Lignin Chemistry; Lin, S.Y., Dence, C.W., Eds.; Springer: Berlin/Heidelberg, Germany, 1992; pp. 83–109. [Google Scholar]
- Sim, S.F.; Mohamed, M.; Lu, N.A.L.M.I.; Sarman, N.S.P.; Samsudin, S.N.S. Computer-assisted analysis of fourier transform infrared (FTIR) spectra for characterization of various treated and untreated agriculture biomass. BioResources 2012, 7, 5367–5380. [Google Scholar] [CrossRef]
- Pandey, K. A study of chemical structure of soft and hardwood and wood polymers by FTIR spectroscopy. J. Appl. Polym. Sci. 1999, 71, 1969–1975. [Google Scholar] [CrossRef]
- Ciolacu, D.; Ciolacu, F.; Popa, V.I. Amorphous cellulose—Structure and characterization. Cell. Chem. Technol. 2011, 45, 13. [Google Scholar]
- Yang, H.; Yan, R.; Chen, H.; Lee, D.H.; Zheng, C. Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 2007, 86, 1781–1788. [Google Scholar] [CrossRef]
- Le, D.M.; Nielsen, A.D.; Sørensen, H.R.; Meyer, A.S. Characterisation of Authentic Lignin Biorefinery Samples by Fourier Transform Infrared Spectroscopy and Determination of the Chemical Formula for Lignin. Bioenergy Res. 2017, 10, 1025–1035. [Google Scholar] [CrossRef]
- Liu, C.F.; Xu, F.; Sun, J.X.; Ren, J.L.; Curling, S.; Sun, R.C.; Fowler, P.; Baird, M.S. Physicochemical characterization of cellulose from perennial ryegrass leaves (Lolium perenne). Carbohydr. Res. 2006, 341, 2677–2687. [Google Scholar] [CrossRef]
- Xu, F.; Sun, J.-X.; Sun, R.; Fowler, P.; Baird, M.S. Comparative study of organosolv lignins from wheat straw. Ind. Crop. Prod. 2006, 23, 180–193. [Google Scholar] [CrossRef]
- Sills, D.L.; Gossett, J.M. Using FTIR to predict saccharification from enzymatic hydrolysis of alkali-pretreated biomasses. Biotechnol. Bioeng. 2012, 109, 353–362. [Google Scholar] [CrossRef]
- Mojet, B.L.; Ebbesen, S.D.; Lefferts, L. Light at the interface: The potential of attenuated total reflection infrared spectroscopy for understanding heterogeneous catalysis in water. Chem. Soc. Rev. 2010, 39, 4643–4655. [Google Scholar] [CrossRef]
- Zhang, K.; Pei, Z.; Wang, D. Organic solvent pretreatment of lignocellulosic biomass for biofuels and biochemicals: A review. Bioresour. Technol. 2016, 199, 21–33. [Google Scholar] [CrossRef] [PubMed]
- The Virtual Planetary Laboratory Molecular Database. Available online: http://vpl.astro.washington.edu/spectra/allmoleculeslist.htm (accessed on 8 May 2020).
- Plyler, E.K. Infrared Spectra of Methanol, Ethanol, and n-Propanol. J. Res. Natl. Bur. Stand. 1952, 48, 281–286. [Google Scholar] [CrossRef]
- Conklin, A., Jr.; Goldcamp, M.J.; Barrett, J. Determination of ethanol in gasoline by FT-IR spectroscopy. J. Chem. Educ. 2014, 91, 889–891. [Google Scholar] [CrossRef]
- Hu, S.; Li, Y. Two-step sequential liquefaction of lignocellulosic biomass by crude glycerol for the production of polyols and polyurethane foams. Bioresour. Technol. 2014, 161, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Elfadly, A.; Zeid, I.; Yehia, F.; Abouelela, M.; Rabie, A. Production of aromatic hydrocarbons from catalytic pyrolysis of lignin over acid-activated bentonite clay. Fuel Process. Technol. 2017, 163, 1–7. [Google Scholar] [CrossRef]
- Dwivedi, A.; Baboo, V.; Bajpai, A. Fukui Function Analysis and Optical, Electronic, and Vibrational Properties of Tetrahydrofuran and Its Derivatives: A Complete Quantum Chemical Study. J. Theor. Chem. 2015, 2015, 345234. [Google Scholar] [CrossRef]
- Borowski, P.; Gac, W.; Pulay, P.; Woliński, K. The vibrational spectrum of 1, 4-dioxane in aqueous solution–theory and experiment. New J. Chem. 2016, 40, 7663–7670. [Google Scholar] [CrossRef]
- IR Spectroscopy Tutorial. Available online: https://orgchemboulder.com/Spectroscopy/irtutor/tutorial.shtml (accessed on 8 May 2020).
- Wen Yee, T.; Tin Sin, L.; Rahman, W.; Samad, A. Properties and interactions of poly (vinyl alcohol)-sago pith waste biocomposites. J. Compos. Mater. 2011, 45, 2199–2209. [Google Scholar] [CrossRef]
- Danish, M.; Mumtaz, M.W.; Fakhar, M.; Rashid, U. Response surface methodology based optimized purification of the residual glycerol from biodiesel production process. Chiang Mai J. Sci. 2015, 43, 1570–1582. [Google Scholar]
- Swoboda, A.; Kunze, G. Infrared study of pyridine adsorbed on montmorillonite surfaces. Clay Clay Miner. 1964, 13, 277. [Google Scholar] [CrossRef]
- Testa, A.C. Molecular Vibrations of Pyridine. Available online: http://facpub.stjohns.edu/~testaa/anim27vib.html (accessed on 6 May 2020).
- Segneanu, A.E.; Gozescu, I.; Dabici, A.; Sfirloaga, P.; Szabadai, Z. Organic compounds FT-IR spectroscopy. In Macro To Nano Spectroscopy; Uddin, J., Ed.; InTech: Rijeka, Croatia, 2012; pp. 145–164. [Google Scholar]
- Eisazadeh, A.; Kassim, K.A.; Nur, H. Physicochemical characteristics of phosphoric acid stabilized bentonite. Electron. J. Geotech. Eng. 2010, 15, 327–335. [Google Scholar]
- Arai, Y.; Sparks, D.L. ATR–FTIR spectroscopic investigation on phosphate adsorption mechanisms at the ferrihydrite–water interface. J. Colloid Interface Sci. 2001, 241, 317–326. [Google Scholar] [CrossRef]
- Valentin, R.; Horga, R.; Bonelli, B.; Garrone, E.; Renzo, F.D.; Quignard, F. FTIR spectroscopy of NH3 on acidic and ionotropic alginate aerogels. Biomacromolecules 2006, 7, 877–882. [Google Scholar] [CrossRef]
- Dharaskar, S.A.; Varma, M.N.; Shende, D.Z.; Yoo, C.K.; Wasewar, K.L. Synthesis, characterization and application of 1-butyl-3 methylimidazolium chloride as green material for extractive desulfurization of liquid fuel. Sci. World J. 2013, 2013, 395274. [Google Scholar] [CrossRef] [PubMed]
- Seethalakshmi, K.; Jasmine Vasantha Rani, E.; Padmavathy, R. Study of vibrational spectra and solvation number of non-aqueous solutions of 1-benzyl-3-dimethylimidazolium chloride through ultrasonic technique. Int. J. Recent Sci. Res. 2015, 6, 2347–2349. [Google Scholar]
- Yue, D.; Jia, Y.; Yao, Y.; Sun, J.; Jing, Y. Structure and electrochemical behavior of ionic liquid analogue based on choline chloride and urea. Electrochim. Acta 2012, 65, 30–36. [Google Scholar] [CrossRef]
- Du, C.; Zhao, B.; Chen, X.-B.; Birbilis, N.; Yang, H. Effect of water presence on choline chloride-2urea ionic liquid and coating platings from the hydrated ionic liquid. Sci. Rep. 2016, 6, 29225. [Google Scholar] [CrossRef]
- Dega-Szafran, Z.; Dutkiewicz, G.; Kosturkiewicz, Z.; Szafran, M. Crystal structure and spectroscopic properties of the complex of trigonelline hydrate with p-hydroxybenzoic acid. J. Mol. Struct. 2011, 985, 219–226. [Google Scholar] [CrossRef]
- Sun, R.; Tomkinson, J.; Bolton, J. Separation and characterization of lignins from the black liquor of oil palm trunk fiber pulping. Sep. Sci. Technol. 1999, 34, 3045–3058. [Google Scholar] [CrossRef]
- Shareef, B.A.; Waheed, I.F.; Jalaot, K.K. Preparation and Analytical Properties of 4-Hydroxybenzaldehyde, Biuret and Formaldehyde Terpolymer Resin. Orient. J. Chem. 2014, 29, 1391–1397. [Google Scholar] [CrossRef][Green Version]
- Kaur, J.; Katopo, L.; Hung, A.; Ashton, J.; Kasapis, S. Combined spectroscopic, molecular docking and quantum mechanics study of β-casein and p-coumaric acid interactions following thermal treatment. Food Chem. 2018, 252, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Moosavinejad, S.M.; Madhoushi, M.; Vakili, M.; Rasouli, D. Evaluation of degradation in chemical compounds of wood in historical buildings using FT-IR and FT-Raman vibrational spectroscopy. Maderas-Cienc. Tecnol. 2019, 21. [Google Scholar] [CrossRef]
- Asare, S. Synthesis, Characterization and Molecular Dynamic Simulations of Aqueous Choline Chloride Deep Eutectic Solvents. Ph.D. Thesis, South Dakota State University, Brookings, SD, USA, 2018. [Google Scholar]
- Bohara, R.A.; Thorat, N.D.; Pawar, S.H. Immobilization of cellulase on functionalized cobalt ferrite nanoparticles. Korean J. Chem. Eng. 2016, 33, 216–222. [Google Scholar] [CrossRef]
- Zhang, D.; Hegab, H.E.; Lvov, Y.; Snow, L.D.; Palmer, J. Immobilization of cellulase on a silica gel substrate modified using a 3-APTES self-assembled monolayer. SpringerPlus 2016, 5, 48. [Google Scholar] [CrossRef] [PubMed]
- Swarnalatha, V.; Ester, R.A.; Dhamodharan, R. Immobilization of α-amylase on gum acacia stabilized magnetite nanoparticles, an easily recoverable and reusable support. J. Mol. Catal. B-Enzym. 2013, 96, 6–13. [Google Scholar] [CrossRef]
- Bai, H.; Wang, H.; Sun, J.; Irfan, M.; Han, M.; Huang, Y.; Han, X.; Yang, Q. Production, purification and characterization of novel beta glucosidase from newly isolated Penicillium simplicissimum H-11 in submerged fermentation. EXCLI J. 2013, 12, 528. [Google Scholar]
- Tsilomelekis, G.; Josephson, T.R.; Nikolakis, V.; Caratzoulas, S. Origin of 5-hydroxymethylfurfural stability in water/dimethyl sulfoxide mixtures. ChemSusChem 2014, 7, 117–126. [Google Scholar] [CrossRef]
- Garba, N.A.; Muduru, I.; Sokoto, M.A.; Dangoggo, S.M. Production of liquid hydrocarbons from millet husk via catalytic hydrodeoxygenation in NIO/AL2O3 catalysts. In WIT Transactions on Ecology and the Environment; Syngellakis, S., Magaril, E., Eds.; WIT PRESS: Billerica, MA, USA, 2018; pp. 125–130. [Google Scholar]
- Allen, G.; Bernstein, H.J. Internal rotation: VIII. The infrared and raman spectra of furfural. Can. J. Chem. 1955, 33, 1055–1061. [Google Scholar] [CrossRef]
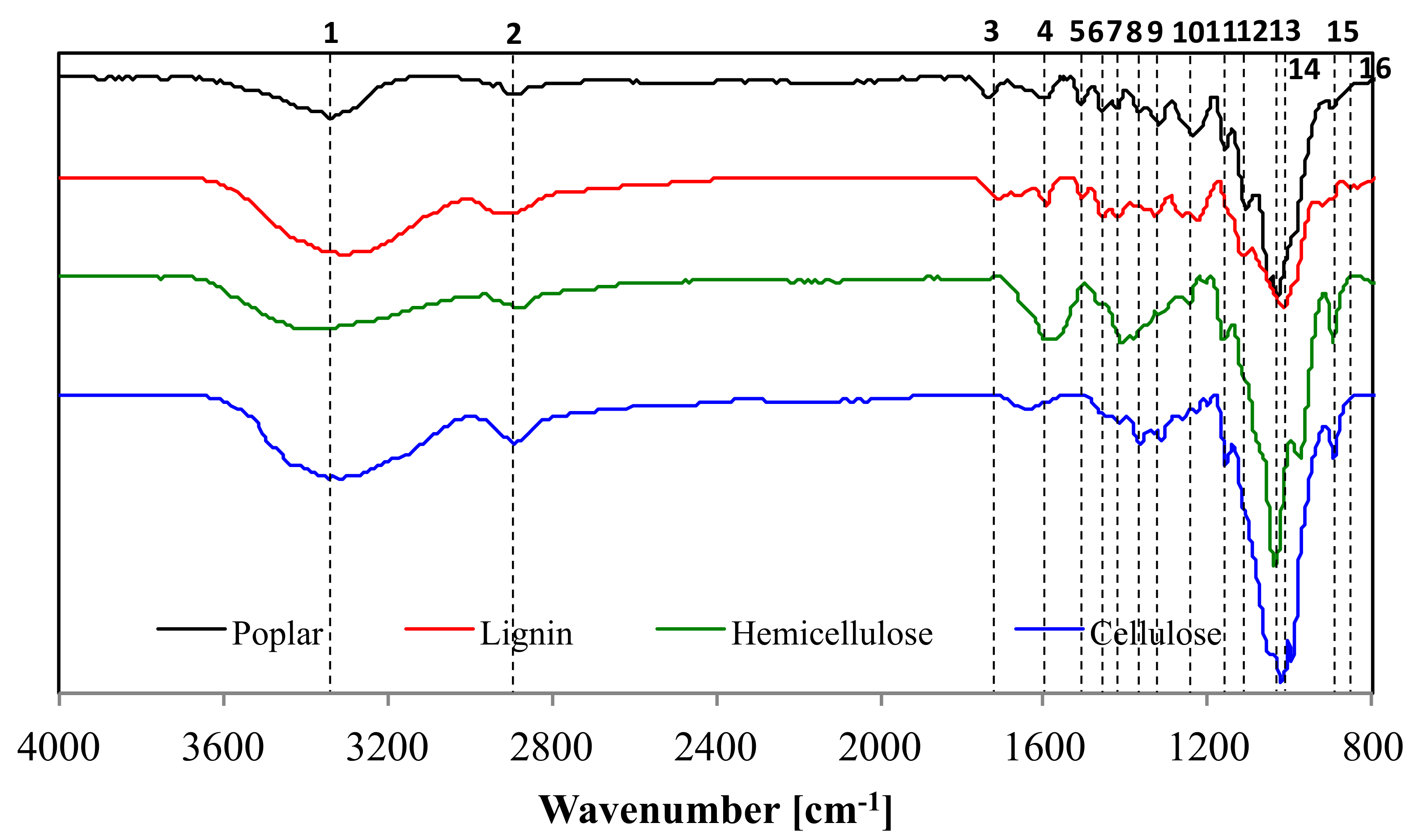
| Wavenumber [cm−1] | Assignment | Components | |
|---|---|---|---|
| 1 | 3367 | O-H stretching | Cellulose, Hemicellulose, Lignin |
| 2 | 2914 | C-H stretching | Cellulose, Hemicellulose, Lignin |
| 3 | 1745 | C=O stretching | Hemicellulose, Lignin |
| 4 | 1618 | Aromatic skeletal vibration, C=O stretching, adsorbed O-H | Hemicellulose, Lignin |
| 5 | 1508 | C=C-C aromatic ring stretching and vibration | Lignin |
| 6 | 1457 | C-H deformation (in methyl and methylene) | Lignin |
| 7 | 1424 | Symmetric CH2 bending vibration, symmetric stretching band of carboxyl group, C-H deformation | Cellulose, Hemicellulose, Lignin |
| 8 | 1370 | C-H bending, C-H stretching in CH3 | Cellulose, Hemicellulose, Lignin |
| 9 | 1317 | CH2 wagging, C-O stretching of C5 substituted aromatic units | Cellulose, Hemicellulose, Lignin |
| 10 | 1235 | C-O stretching of guaiacyl unit | Lignin |
| 11 | 1160 | C-O-C stretching | Cellulose, Hemicellulose |
| 12 | 1108 | Aromatic C-H in plane deformation | Lignin |
| 13 | 1053 | C-OH stretching vibration, C-O deformation | Cellulose, Hemicellulose, Lignin |
| 14 | 1032 | C-O stretching, aromatic C-H in plane deformation | Cellulose, Lignin |
| 15 | 896 | C-O-C stretching | Cellulose, hemicellulose |
| 16 | 846 | Aromatic C-H out of plane bending | Lignin |
| Contaminants | Wavenumber [cm−1] | Assignments |
|---|---|---|
| Water | 3354 | O-H stretching |
| 1653 | O-H-O scissors bending | |
| Acetone | 3005 | CH3 stretching |
| 2908 | CH3 stretching | |
| 1713 | C=O stretching | |
| 1431 | CH3 deforming | |
| 1364 | CH3 deforming | |
| 1222 | C-C stretching | |
| Ethanol | 3350 | O-H stretching |
| 2980 | C-H stretching | |
| 1056 | C-O stretching | |
| Methanol | 3352 | O-H stretching |
| 2952 | C-H asymmetric stretching | |
| 2879 | C-H symmetric stretching | |
| 1465 | C-H asymmetric bending | |
| 1450 | C-H symmetric bending | |
| 1336 | O-H bending | |
| 1068 | CH3 rocking | |
| 1026 | C-O stretching | |
| Tetrahydrofuran | 2977 | C-H stretching |
| 2875 | C-H stretching | |
| 1063 | Ring deformation | |
| 912 | CH2 twisting | |
| Toluene | 3069 | C-H stretching |
| 1497 | C-C stretching | |
| 728 | C-H out of plane bending | |
| Dioxane | 2960 | C-H stretching |
| 2890 | C-H stretching | |
| 1457 | Symmetric CH2 deformation | |
| 1322 | CH2 wagging | |
| 1255 | CH2 twisting | |
| 1119 | C-O-C symmetric stretching | |
| 1057 | Ring trigonal deformation | |
| 889 | C-C stretching | |
| 872 | C-O-C stretching | |
| Glycerol | 3341 | O-H bending |
| 2948 | C-H stretching | |
| 2897 | C-H stretching | |
| 1333 | C-H deformation | |
| 1239 | C-H deformation | |
| 1034 | C-O stretching | |
| 923 | O-H bending | |
| Chloroform | 1220 | C-H bending |
| 755 | CCl3 stretching | |
| Pyridine | 3036 | C-H stretching |
| 1583 | C-C bonding | |
| 1485 | C-N stretching | |
| 1438 | C-H in plane wagging | |
| 1203 | Symmetric C-H wagging | |
| 1069 | C-H wagging | |
| 1032 | C-C in plane wagging | |
| 750/693 | C-H out of plane bending |
| Contaminants | Wavenumber [cm−1] | Assignments |
|---|---|---|
| Sulfuric acid | 3370 | O-H stretching |
| 1660 | O-H-O scissors bending | |
| 1362 | S=O stretching | |
| 750 | S-O stretching | |
| Hydrochloric acid | 3370 | O-H stretching |
| 2905 | H-Cl stretching | |
| 1660 | O-H-O scissors bending | |
| Phosphoric acid | 3370 | O-H stretching |
| 2905 | P-OH bond | |
| 1660 | O-H-O scissors bending | |
| 1161 | P=O stretching | |
| 1031 | P=O stretching | |
| Acetic acid | 3351 | O-H stretching |
| 2916 | Symmetric CH3 stretching | |
| 1706 | C=O stretching | |
| 1427 | CH3 deformation | |
| 1234 | O-H bending | |
| 1031 | CH3 rocking | |
| Ammonium hydroxide | 3350 | N-H stretching & O-H stretching |
| 2914 | N-H stretching | |
| 1660 | O-H-O scissors bending |
| Contaminants | Wavenumber [cm−1] | Assignments |
|---|---|---|
| 1-Butyl-3-methylimidazolium chloride | 3341 | Quaternary amine salt formation |
| 1658 | C=C stretching | |
| 1604 | C=N stretching | |
| 1-Benzyl-3-methylimidazolium chloride | 2961 | C-H stretching |
| 1574 | C-C stretching ring vibration | |
| 633 | C-N/C-Cl in-plane bending | |
| ChCl-Urea | 3435 | NH2 asymmetric stretching |
| 3340 | NH2 symmetric stretching | |
| 1669 | NH2 bending vibration | |
| 1597 | OH bending vibration | |
| 1474 | CH3 rocking | |
| 1152 | C-N stretching | |
| 1062 | CH2 rocking | |
| 961 | Asymmetric stretching of COO | |
| 790 | C=O bonding | |
| ChCl–PHA | 3180 | O-H stretching |
| 1681 | C=O stretching | |
| 1581 | Asymmetric stretching of COO | |
| 1282 | C-O stretching vibration | |
| 1082 | C-O stretching | |
| 953 | C-N stretching | |
| 861 | CH2 rocking vibrations | |
| 838 | Aromatic C-H out-of-plane bending | |
| 786 | C–C stretching | |
| ChCl–PB | 3122 | The stretching vibration of the phenolic O-H |
| 1667 | The stretching vibration of carbonyl group | |
| 1272 | Methylene | |
| 1030 | C-H binding | |
| ChCl-PCA | 3126 | Bending vibration of –NH2 |
| 1675 | C=O stretch of carboxylic acid | |
| 1606 | C=C stretching | |
| 1160 | C-OH stretching | |
| 848 | C-H stretching | |
| 771 | Stretching of the -OH group | |
| Cellulase | 3353 | N-H stretching & O-H stretching |
| 2942 | C-H stretching (asymmetric) | |
| 2900 | C-H stretching (symmetric) | |
| 1642 | NH2 scissoring & C=N vibration | |
| 1334 | C-N stretching | |
| 1036 | C-N stretching | |
| β-glucosidase | 3351 | N-H stretching |
| 1646 | N-H bonding & C=O stretching | |
| 1432 | N-H stretching | |
| 620 | N-H out of plane bending | |
| HMF | 3364 | O-H stretching |
| 1661 | Carbonyl stretching | |
| 1561 | C=C stretching (furan ring) | |
| Furfural | 3134 | C-H stretching |
| 2859 | C-H vibration of aldehyde group | |
| 1671 | C=O in conjugated carbonyl group | |
| 1465 | C=C stretching of furan ring | |
| 1276/1021 | C-O stretching vibration | |
| 928/884/755 | C-H bending out of plane peaks |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhuang, J.; Li, M.; Pu, Y.; Ragauskas, A.J.; Yoo, C.G. Observation of Potential Contaminants in Processed Biomass Using Fourier Transform Infrared Spectroscopy. Appl. Sci. 2020, 10, 4345. https://doi.org/10.3390/app10124345
Zhuang J, Li M, Pu Y, Ragauskas AJ, Yoo CG. Observation of Potential Contaminants in Processed Biomass Using Fourier Transform Infrared Spectroscopy. Applied Sciences. 2020; 10(12):4345. https://doi.org/10.3390/app10124345
Chicago/Turabian StyleZhuang, Jingshun, Mi Li, Yunqiao Pu, Arthur Jonas Ragauskas, and Chang Geun Yoo. 2020. "Observation of Potential Contaminants in Processed Biomass Using Fourier Transform Infrared Spectroscopy" Applied Sciences 10, no. 12: 4345. https://doi.org/10.3390/app10124345
APA StyleZhuang, J., Li, M., Pu, Y., Ragauskas, A. J., & Yoo, C. G. (2020). Observation of Potential Contaminants in Processed Biomass Using Fourier Transform Infrared Spectroscopy. Applied Sciences, 10(12), 4345. https://doi.org/10.3390/app10124345








