Abstract
In alpine environments, open habitats alternate with wood to create a habitat mosaic that shapes insect community composition and diversity. Dung beetles are an important group of insects specialized in feeding on vertebrate dung whose availability also depends on habitat type. Although the habitat preferences of dung beetles have been extensively studied, few studies have addressed the influence of habitat structure and temperature on dung beetle communities in alpine environments. We sampled dung beetles in pastures, at the edges between pasture and wood, in inner and outer woods of two alpine areas at different altitudes (two sites per area). We found that pastures had higher mean temperatures compared to other habitat types and hosted the highest number of species. However, the interaction between habitat and altitude significantly affected species richness and abundance, suggesting that habitat type and temperature modulate the response of dung beetles in the study area. Edges hosted intermediate communities between pastures and woodlands and were populated by both pasture and woodland specialists. Our results suggest that maintaining pastures is crucial to preserving dung beetle communities.
1. Introduction
Understanding the effects of habitat transformation and climate change on species distribution is important to predict future changes in biodiversity and ecosystem functioning [1]. Insects are experiencing a global decline in richness and abundance due to unprecedented rates of habitat loss, degradation, and fragmentation [2,3,4]. Agricultural intensification and urbanization, by replacing traditional low-intensity agriculture and natural areas, have led to a dramatic reduction in both local and regional diversity [3]. A severe effect of land use changes on insects and the ecosystem services they provide has been reported in disparate insect orders such as Coleoptera [5], Diptera [6], and Lepidoptera [7]. Furthermore, land use intensification is a major driver of biotic homogenization and can favor generalist insects at the expense of specialists that feed on a limited range of food sources [8]. In this context, knowledge about the effect of habitat transformation on biodiversity is essential to plan effective conservation strategies.
Dung beetles (Coleoptera: Scarabaeoidea) are an important group of insects specialized in feeding on small particles of fresh dung rich in dead and alive microbial biomass or on other particulate debris from the vertebrate’s gut [9], especially ungulates [10]. Based on their nesting behavior, they can be classified as dwellers, tunnelers, and rollers [11]. Dwellers live and lay eggs inside the dung, tunnelers dig tunnels beneath the dung pad where they build brood balls for larvae nutrition, and rollers shape dung into round balls and bury them far away from the dung pad. Through their feeding and nesting activity, dung beetles are involved in multiple processes that can also support pivotal ecosystem services. For example, they contribute to the mineralization of organic matter [12], nutrient transfer (e.g., carbon, nitrogen, phosphorus, potassium) into the soil [13,14], secondary seed dispersal [15,16], herbage growth [17], and parasite control and suppression [18].
The availability of dung, which mostly depends on the presence and abundance of large-bodied vertebrates, is a key factor influencing dung beetle community structure [19,20]. However, in a study on altitudinal transects in Mt. Kilimanjaro, Gebert et al. [21] reported no correlation between defecation rate and species richness, suggesting that factors other than dung availability can affect dung beetle communities. Habitat type has a significant impact on dung beetle community composition and diversity because it influences both the habitat structure [22] and the presence and abundance of large-bodied vertebrates whose dung is used by dung beetles for feeding [19]. The effect of habitat type on dung beetles is context-dependent and is affected by multiple environmental variables in both the local and biogeographic context [23]. Open habitats such as pastures can have reduced richness and diversity, as shown by Korasaki et al. [24] and Costa et al. [25] in tropical forests and Giménez Gómez et al. [26] in Atlantic forests. However, Nichols et al. [27], based on an extensive literature review, pointed out that the decline in dung beetle diversity and abundance of species typical of forests can be complemented by an increase in the abundance and richness of species characteristic of more open habitats. Within the same habitat, multiple factors can shape its structure and thus influence the taxonomic diversity and composition of dung beetles. For example, minor variations in the density of canopy cover and leaf litter as well as soil characteristics can affect the nesting activity of some dung beetle species [23,28,29]. Besides the effect on taxonomic diversity and composition, habitat type can also influence the functional composition of dung beetle communities. For example, land use can select species based on their nesting strategy. A higher abundance of tunnelers in pastures compared to dwellers and rollers has been observed [30], although other studies have found the absence of endocoprids from open habitats [31].
The effect of habitat on dung beetle communities is often modulated by the spatial position and the distance from the habitat edge. At first, habitat edges present different physical and chemical properties (e.g., soil moisture, light conditions, temperature) compared to the habitats from which they originate [32]. Moreover, high abundances of a habitat specialist species in its preferred habitat could promote the persistence of this species in unfavorable habitats through dispersal processes [33]. For example, Martínez-Falcón et al. [34] showed that dung beetle species responded differently to the edge effect, while community-level measures (e.g., species richness, diversity) masked the effect observed at the species level. The effect of distance from the habitat edge is less clear, with some studies reporting a negligible effect of distance on species turnover [35] and others showing gradual changes in beta-diversity [36]. Lastly, climatic conditions particular to each habitat type may be also responsible for its use. Temperature, for instance, has been acknowledged to be a driver of dung beetle diversity in several ecosystems.
Here, we aimed at understanding the effect of habitat type and temperature on dung beetle diversity in Alpine areas of Northern Italy, which host a huge diversity of dung beetles, especially in open habitats [37,38]. In this context, we aimed at understanding the effect of (i) of habitat type (pasture, edges, outer wood and inner wood), (ii) spatial position within the wood, and (iii) temperature on dung beetle diversity in two study areas located at different altitudes. We hypothesize a higher richness at the edge between pasture and wood due to a positive edge effect. We also hypothesize a gradual decrease in richness from pasture to inner forest areas due to the general higher richness of dung beetles in the open habitats of the study area.
2. Materials and Methods
2.1. Dung Beetle Collection
Dung beetles were collected from the Stura valley (Piedmont region, northwestern Italy) following a hierarchical study design (Figure 1). This valley has an extraordinarily long history of extensive grazing by cattle, horses, and sheep; it hosts the indigenous Sambucana sheep breed, which is a hardy animal capable of living even on uncomfortable and steep rocky pastures. The wild ungulate community includes deer, roe deer, and wild boars.
Two areas with different altitudes, the first (1) being at nearly 900 a.s.l. (44°18′18.0″ N 7°14′16.0″ E) and the second (2) at nearly 1500 m a.s.l. (44°15′56″ N 7°03′21″ E), were selected. Secondly, two sites located at a horizontal distance of 300 m apart from each other (hereafter denoted as 1_1, site 1 in area 1; 1_2, site 2 in area 1, etc.) were selected from each area. Lastly, for each site, four habitat types (pasture, edge, outer wood, and inner wood) were identified. Outer and inner wood were 50 m and 400 m from the edge, defined as the ecotonal zone between pasture and wood. The area 1 wood was mainly composed of Castanea sativa Mill., 1768, with the sporadic presence of Fagus sylvatica L., Picea abies (L.) H. Karst., 1881, and Betula pendula Roth, 1788, while Corylus avellana L., 1753, Acer pseudoplatanus L., 1753, and Fraxinus excelsior Boiss, 1841 characterized the edge. The area 2 wood was characterized by Abies alba Mill. and Fagus sylvatica L., 1753, with low abundances of Larix decidua Mill., 1768 and P. abies. In area 1, the tree height ranged from 20 to 30 m and the density was nearly 120 trees ha−1. In area 2, the tree height ranged from 30 to 40 m, and the density was nearly 100 trees ha−1. In both areas, the ground cover in wooded habitats was mainly composed of leaf litter, with sporadic mosses and ferns in area 2.
Temperature was recorded for each habitat at four-hour intervals from 00:00 to 20:00 within each habitat using Hygro Button data loggers (Proges Plus, Lille, France). These small buttons (Ø × H: 16 × 6 mm) were attached to a wooden stake driven into the ground standing between the three traps to measure the air temperature just above the surface of the soil.
Dung beetles were collected using three baited pit-fall traps for each habitat according to Palestrini et al. [39]. Each trap consisted of a 1.5 L clear plastic bottle 9 cm in diameter, which was cut at the top one-third. The top one-third was then inserted into the lower part of the bottle to act as a funnel. The traps were baited with 200 g of fresh cow dung, suspended in gauze on a tripod made with three sticks 50 cm in length, and placed over the trap close to the entrance of the funnel. Dung beetles were collected using this procedure biweekly from 9 July 2022 to 20 October 2022, for a total of 11 sampling dates, to cover the adult flight period in the study area. The position of the baited traps was the same throughout the sampling period. Organisms were identified using a stereomicroscope according to Dellacasa and Dellacasa [40].
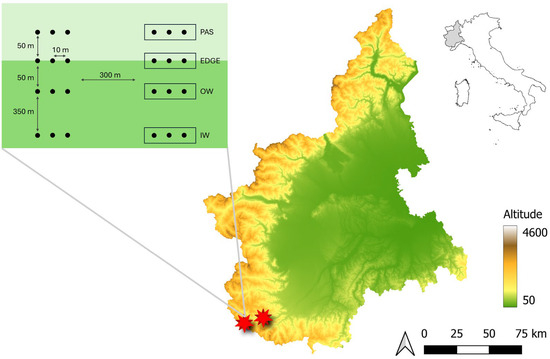
Figure 1.
Study area, located in northwestern Italy (light gray). Sampling areas were located at nearly 900 and 1500 a.s.l., respectively. For each area, two sites were selected 300 m distant from each other (IW = inner wood, OW = outer wood, PAS = pasture). Maps were created with QGIS [41].
2.2. Data Analysis
Temperature changes among different habitats at the four sites were investigated with generalized additive mixed models (GAMM) using the gamm() function of the mgcv package version 1.9-1 [42] for the R statistical software version 4.4.1 [43]. The habitat structure nested within sites was used as both a fixed effect and a smooth factor in interaction with the Julian day calculated from the first day of temperature recording (19 June 2022). Cyclic cubic regression splines were used as the smoothing basis. A corARMA structure with p = 3 conditional on the habitat structure nested within sites was used to deal with temporal autocorrelation. Pairwise comparisons of the temperature trend among habitats within sites and among sites within habitats were performed using the difference_smooth() function of the gratia package version 1.9-1 [44].
The numbers of shared and unshared species among all the sites, together and separately for each site, were represented as Venn diagrams using the ggVennDiagram package version 1.5.2 [45]. The effects of habitat and area on species richness, abundance, and nesting strategy were tested with a linear mixed-effect model using the pitfall trap ID as a random effect. Abundance was log-transformed to meet the assumptions of linear effect models, and the binomial family was used to model nesting strategies, measured as percentages. Models were fitted with the lmer() function of the lmerTest package version 3.1-3 [46], which builds upon the lme4 package [47]. Functional analyses of nesting strategy were performed starting from the community-weighted mean (CWM). For each species, data on nesting strategies (e.g., tunnelers, dwellers) were obtained from [48]. CWM was then calculated from the species × abundance matrix and the species × traits matrix using the function dbFD() of the FD package version 1.0-12.3 [49]. A post hoc test was performed to test pairwise difference among habitats within areas with the package emmeans version 1.10.3 [50].
We searched for indicator species of the investigated habitats with generalized linear effect models for each area separately. Habitat and collection date were considered as fixed effects, while the trap ID was considered as the random effect. The negative binomial distribution was used instead of the Poisson distribution as the error distribution family when overdispersion was significantly different from 1. A post hoc test was performed to test pairwise differences among habitats. Models were fitted for species with more than 10 individuals and recovered in more than 5% of the samples to avoid problems with model convergence.
Beta diversity patterns in dung beetle composition were explored with non-metric multidimensional scaling (nMDS). nMDS was performed on two axes using the vegan package version 2.6-6.1 [51]. Bray–Curtis was used as a dissimilarity measure and stress served as a measure of the model’s goodness of fit. Convex hulls were fitted for each habitat and site separately with the hypervolume package version 3.1.4 [52]. The pairwise overlap among convex hulls of different habitats was used as an estimate of the overall community similarity.
3. Results
3.1. Temperature Trends in the Investigated Habitats
The mean temperature during the study period was 17.6 ± 4.53 °C (mean ± sd): 17.6 ± 5.12 °C, 12.9 ± 4.15 °C, and 14.1 ± 5.14 °C at study sites 1_1, 1_2, 2_1, and 2_2, respectively. With respect to the habitat, the temperatures were 14.8 ± 3.6 °C, 14.5 ± 3.99 °C, 14.5 ± 4.54 °C, and 18.9 ± 6.33 °C for internal wood, external wood, edge, and pasture. Therefore, at all sites, pastures generally showed higher temperatures and variability than the other habitats, while the edge, outer, and inner wood showed no differences among each other (Supplementary Materials Figure S1). Temporal trends were similar among habitats and sites (Figure 2).
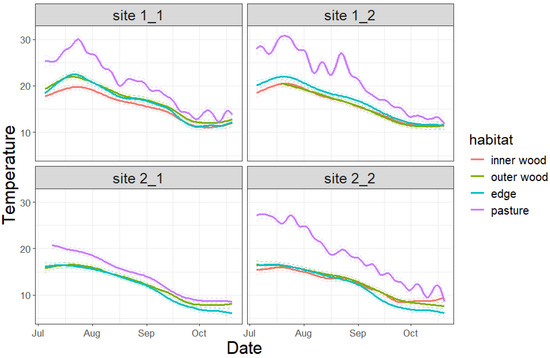
Figure 2.
Temperature trends of each habitat at the four investigated sites. Dashed lines represent confidence intervals.
3.2. Species Shared among Habitats for the Overall Dataset and within Each Site
A total of 27,089 individuals were collected and 37 species were identified. The most abundant species was Onthophagus fracticornis (Preyssler, 1790) with 10,063 individuals, followed by Volinus sticticus (Panzer, 1798) with 6559 and Anoplotrupes stercorosus (Scriba, 1791) with 3094. Overall, 17 species were found in all the investigated habitats and 7 were only found in pastures, namely Euoniticellus fulvus (Goeze, 1777), Onthophagus furcatus (Fabricius, 1781), Trypocopris vernalis fauveli (Bedel, 1911), Nialus varians (Duftschmid, 1805), Otophorus haemorrhoidalis (Linnaeus, 1758), Nimbus contaminatus (Herbst, 1783), and Onthophagus medius (Kugelann, 1792). However, species found in the inner wood, outer wood, and edge were also present in other habitats (Figure 3). Of the species exclusive to pasture habitats, O. furcatus (n = 2), N. varians (n = 2), N. contaminatus (n = 7), and O. medius (n = 5) had few individuals, while E. fulvus (n = 50), T. vernalis fauveli (n = 17), and O. haemorrhoidalis (n = 22) had slightly more individuals.
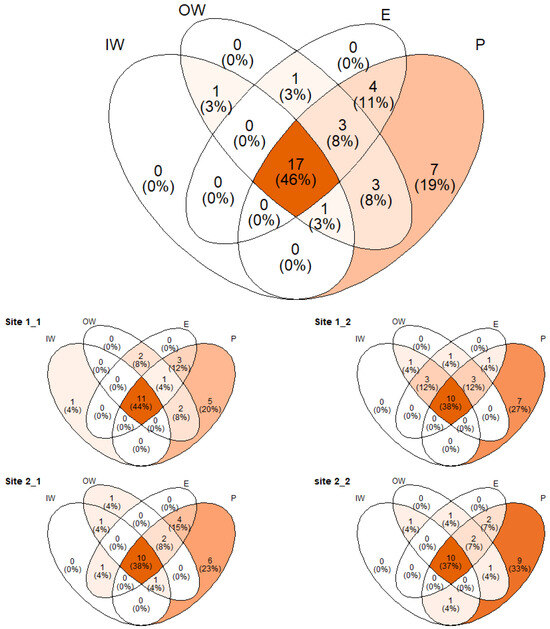
Figure 3.
Venn diagram representing the species shared and unshared within the 4 investigated habitats (IW = internal wood, EW = external wood, M = edge, P = pasture). Overall (upper panel) and site-level results are reported.
Similar patterns were found at the site level, with pastures showing the highest number of exclusive species compared to the other habitats, although some discrepancies due to rare species appeared (e.g., site 1_1 had an exclusive species in the inner wood, Figure 3). Species abundances for each site and habitat are presented in Supplementary Materials Table S1.
3.3. Species Habitat Preferences
The results of the GLMM showed differences in the habitat preferences of different species (Figure 4). In area 1, Acrossus depressus (Kugelann, 1792), Limarus zenkeri (Germar, 1813), and V. sticticus preferred forested habitats (IW, OW and E), while Aphodius fimetarius (Linnaeus, 1758), Onthophagus coenobita (Herbst, 1783), and, to a lesser extent, Geotrupes spiniger (Marsham, 1802) preferred pastures. Some species showed increasing abundance from wood to pasture (e.g., O. fracticornis and Onthophagus taurus (Preyssler, 1790)) and from pasture to wood (e.g., A. stercorosus, Parammoecius corvinus (Erichson, 1848)).
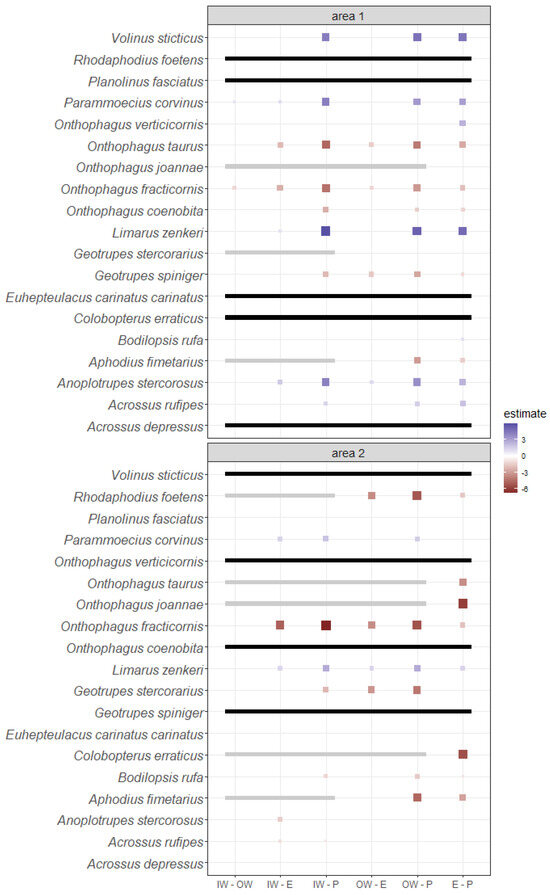
Figure 4.
Pairwise comparisons among the estimates of the habitat effect calculated for each species with mixed-effect models. Size and color intensity are proportional to the magnitude of the difference between the abundances of the ith species of the habitat under comparison (blue indicates that the first habitat has a greater effect than the second, while red indicates the opposite). The lack of an estimate means that the difference between the effect of two habitats includes 0 and should be interpreted as not significant. Black bars mean that the species was not present in the area, while grey bars mean that those habitats were not tested because of problems of convergence in mixed models (usually because the abundance was 0).
In area 2, P. corvinus preferred inner and outer woods, while A. fimetarius, Bodilopsis rufa (Moll, 1782), Onthophagus joannae (Goljan, 1953), O. taurus, and Rhodaphodius foetens (Fabricius, 1787) preferred edges and/or pastures. Some species showed increasing abundance from wood to pastures (O. fracticornis and, to a lesser extent, Geotrupes stercorarius (Linnaeus, 1758)) and from pastures to wood (L. zenkeri).
Species generally showed the same or similar habitat preferences between the two investigated areas, except for A. depressus and A. stercorosus, which changed their habitat preferences between the two investigated areas. Species exclusive to only one habitat (see Section 3.2) were not tested because of complete separation problems in GLMMs.
3.4. Patterns of Diversity Variation among Habitats and Sites
The number of species was significantly related to habitat (F3454.1 = 21.6; p-value ≤ 0.0001) and the interaction between habitat and area (F3388.0 = 28.0; p-value ≤ 0.0001). The post hoc test showed that, on average, edges had slightly higher richness values than inner wood (t35.7 = −2.824, p-value = 0.0370) and pastures (t36.3 = −2.985, p-value = 0.0249) at area 1, whereas pastures had higher richness than inner wood (t44.3 = −10.20, p-value ≤ 0.0001), outer wood (t39.0 = −9.613, p-value ≤ 0.0001), and edge (t39.3 = −7.483, p-value ≤ 0.0001) than inner wood (t42.8 = −2.968, p-value = 0.0243) at area 2 (Supplementary Materials Figure S1). Species richness was significantly higher in area 1 than in area 2 in inner wood (t23.7 = −2.518, p-value = 0.0190), outer wood (t20.0 = −2.385, p-value = 0.0271), and edge (t21.7 = −2.313, p-value = 0.0308), while the opposite was found in pastures (t21.9 = −8.156, p-value ≤ 0.0001).
Like species richness, abundance was significantly related to habitat (F3452.1 = 14.9; p-value ≤ 0.0001) and the interaction between habitat and area (F3452.2 = 8.13; p-value ≤ 0.0001). The post hoc test showed that, on average, pastures had a higher abundance than inner wood (t44.3 = −7.48, p-value ≤ 0.0001), outer wood (t39.0 = −6.20, p-value ≤ 0.0001), and edge (t39.3 = −4.00, p-value = 0.0015) than inner wood (t42.8 = −3.631, p-value = 0.0040) at area 2. No differences were found between the abundances of the two areas.
The percentage of dwellers was related to habitat (F3452.3= 7.64; p-value ≤ 0.0001) and the interaction between habitat and area (F3453.3 = 30.6; p-value ≤ 0.0001) (Supplementary Materials Figure S1). The post hoc test showed than pastures had a lower percentage than inner wood (t35.6 = 8.20, p-value ≤ 0.0001), outer wood (t34.6 = 8.03, p-value ≤ 0.0001), and edge (t36.5 = 8.12, p-value ≤ 0.0001) at site 1, whereas a higher percentage was found in pasture compared to edge (t39.3 = −4.30, p-value = 0.0060) at site 2.
The percentage of tunnelers was related to habitat (F3452.3= 76.5; p-value ≤ 0.0001) and the interaction between habitat and area (F3452.4 = 7.90; p-value ≤ 0.0001) (Supplementary Materials Figure S1). At area 1, pasture showed a higher percentage than inner wood (t35.6 = −7.96, p-value ≤ 0.0001), outer wood (t34.6 = −8.63, p-value ≤ 0.0001), and edge (t36.6 = −6.47, p-value ≤ 0.0001). The same was true at area 2, where percentages were higher in pastures than in than inner wood (t44.3 = −11.5, p-value ≤ 0.0001), outer wood (t39.0 = −8.36, p-value ≤ 0.0001), and edge (t39.3 = −3.61, p-value = 0.045). However, at area 2, edge had a higher percentage than inner wood (t42.8 = −8.08, p-value ≤ 0.0001) and outer wood (t37.6 = −4.80, p-value = 0.0001) and a higher percentage outer wood than inner wood (t42.5 = 3.43, p-value = 0.0071).
Plots of temporal trends of species richness, abundance, and nesting strategies are presented in the Supplementary Materials Figure S1.
3.5. Effects of Habitat and Time on Community Composition
The stress values of the four nMDS were 15.3, 14.6, 17.8, and 13.8 for site 1_1, site 1_2, site 2_1, and site 2_2, respectively. A stress value lower than 20.0 means that the ordination is good and can be interpreted. Convex hulls fitted on nMDS ordinations generally showed a clear separation between pasture and wood communities (Figure 5). This separation was evident for sites 1_1, 1_2, and 2_2 (Figure 5a,b,d), but not for site 2_1 (Figure 5c). These results suggest that edge communities showed intermediate characteristics between the communities of pasture and wood. Visual results were confirmed by the analysis of the pairwise overlap among habitats, which generally showed low overlap between pastures and wooded habitats (Table 1).
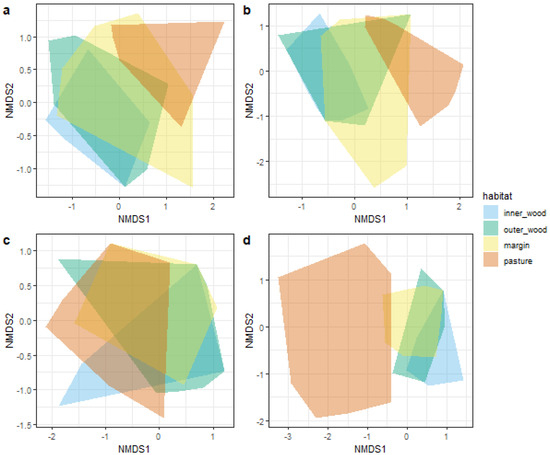
Figure 5.
Non-metric multidimensional scaling ordinations for (a) site 1_1, (b) site 1_2, (c) site 2_1, and (d) site 2_2. Convex hulls are reported for each habitat type.

Table 1.
Overlap between pairwise combinations of convex hulls for all the sites individually (iw = inner wood, ow = outer wood, pas = pasture). Values represent the percentage of the overlap area over the overall volume of two convex hulls.
4. Discussion
Understanding the effect of habitat type and temperature on insect communities is crucial in order to promote effective conservation strategies. In this work, we showed how habitat type affect dung beetle communities, with the two investigated areas having different diversity patterns. Focusing on habitat type, we showed that pastures host communities with more tunnelers, while the other habitats are inhabited by more dwellers. Lastly, we demonstrated that most of the investigated species tend to have similar habitat preferences independently of altitude. However, this was not true for a few species, pointing to the importance of local environmental characteristics when studying the habitat preferences of dung beetles. We also highlighted the possible effect of temperature on the composition of local communities.
4.1. Diversity Patterns within and between Habitats
Our results showed that pastures had the most diverse fauna among habitat types, both overall and in the two investigated areas. Between areas, pastures differed in their species richness, while the other habitats did not. The increased species richness in area 2 was likely due to the presence of some Aphodiinae species, most of which are adapted to cold environments, probably because of their biogeographic origin [53]. A greater abundance of Aphodiinae with increasing altitude has been found in other alpine environments [54] and in other areas, such as in the neotropics [55], probably because of cold adaptation. However, differences in the number of species between pastures of the two study areas could be due to other factors. For example, grazing intensity can decrease the abundance of large species and tunnelers [56,57], thus inflating diversity estimates. Similarly, the use of anthelmintics [58] and the size of pastures compared to the other habitats [59] can affect the number of species in pastures.
Among habitats, we found a higher richness, but not abundance, in the edge and outer wood habitats at lower altitudes, while at higher altitudes, we found higher richness and abundance in pastures compared to the other habitats. The presence of an edge effect in one of the investigated areas but not in the other one corroborates the context dependency of this process. For example, an edge effect on dung beetle richness has been detected in some areas [60], but not in others [34,61], in different environmental contexts. In our study, differences in species richness between pastures in different areas affected the role of edges and outer wood in hosting habitat specialists of both closed and open areas. As stated before, this result may be due to an interaction between the increased richness in Aphodiinae with altitude and different management practices in pastures.
Dwellers’ percentage in the community increased from pasture to wood in both areas, while tunnelers’ percentage showed the opposite trend. Tunnelers are often dominant in open habitats because of a competitive advantage over the other groups [30]. In particular, the most abundant tunnelers in pastures of both study areas were Onthophaginae, some of which (e.g., O. fracticornis, O. taurus) can tolerate high temperatures such as those in the study areas because of their heat tolerance and limited water loss [62]. Heat tolerance is important in the studied sites because of the pastures having higher temperatures than the other habitats. Moreover, tunnellers can be less affected than other groups to problems related to dung desiccation by burying feeding and reproductive ovoids into the soils. On the contrary, high rates of dung desiccation can severely affect reproductive success in dwellers [63].
4.2. Habitat Preferences and the Effect of Temperature
The habitat preferences of dung beetles differed among species and among the investigated areas. Some species showed strong habitat preferences for open habitats and edges and were not found in closed habitats (e.g., O. joannae, O. taurus). Other species showed preferences for closed environments, although some individuals were found in both edges and pastures (e.g., L. zenkeri, V. sticticus). Among the species shared by both areas, some of them showed consistent preferences (e.g., L. zenkeri, O. fracticornis), while others changed their preferences (e.g., A. rufipes, A. stercorosus). The habitat selection of most species likely reflected their physiological performances related to temperature. For instance, E. fulvus was exclusive to pastures because of its high survival rate under high temperatures [62,64]. Analogously, O. fracticornis and O. taurus were mainly found in pastures because of their good upper thermal tolerance and very high desiccation resistance [64]. Lastly, A. stercorosus probably preferred wooded and/or partially wooded habitats to pastures (especially in area 1) because of its low tolerance to heat [65].
Although our sampling design does not allow for disentangling the effect of altitude and habitat types on habitat preferences, we can speculate about the causes of the changed habitat preferences between the two studied areas. Changes in habitat preferences along altitudinal gradients are often due to temperature changes. For example, some dung beetle species prefer closed habitats at lower altitudes and open habitats at higher altitudes because closed habitats at higher altitudes are too cold [66,67]. Similarly to the results of Menéndez and Gutiérrez (2004) [66], we found that A. rufipes and A. stercorosus shifted habitat preferences from closed habitats to open habitats with increasing altitude. Vegetation structure, on the other hand, can affect species distribution directly by changing microclimatic conditions [68] and affecting the dispersion of dung odors [69], and indirectly by affecting the distribution of wild fauna species [70]. This is probably a minor effect in our study area given the low tree density and the absence of undercover vegetation. Lastly, the effects of altitude and vegetation structure probably cannot be fully disentangled because vegetational changes naturally occur along altitudinal gradients.
Changes in dung beetle habitat preferences influenced the community structure in both the studied areas. We found that edges showed a high overlap percentage with outer wood, meaning that the community composition in these two habitats is more similar than the similarity in community composition between edges and the other habitat. This is especially true for edges and pasture, which, except for one site, showed a small overlap. These results are probably due to wood specialists not colonizing nearby habitats or colonizing them with low abundances (edges, outer wood), while pasture generalists gradually decrease in abundance going from edges to inner wood.
In three out of four sites, the ordering produced by nMDS highlighted an obvious difference (i.e., a limited overlap of convex hulls in ordination plot) between the pastures and the wooded sites. This mirrored the trend of temperatures, which were significantly higher and more variable in the pastures compared to the other habitats. It is also noteworthy to underline how in one site (2_1), the community composition of the four habitats appeared to be overlapping just because it reflected a certain uniformity of temperatures in that site. These results, taken together, indicate that local microclimatic conditions, including temperature, may shape the composition of dung beetle communities by driving habitat selection of species.
4.3. Caveats
Multiple interactive factors can shape dung beetle communities. Feeding resources may be one of these factors. In this respect, a caveat of the present study is the use of only cow dung as bait for the sampling traps. This may have limited the number of species collected and hidden some patterns of habitat preferences and community composition. Future studies, therefore, will have to rely on traps with different types of dung as bait [71].
Another possible caveat of this study is that we have considered the temperature of the air only and not the microclimate of the studied sites. Dung beetles spend most of their life in dung and soil, where the microclimatic conditions are very different. Dwellers found in large dung pats defecated by cattle, for example, might encounter humidity limitations because they move in the dung when it is fresh, with high water content. In future studies, therefore, the analyses of microclimatic factors should also include dung and soil.
4.4. Management Implications
Our study reinforces the idea that pastures are essential to preserve dung beetle diversity in alpine areas [38]. This is especially important in traditional managed pastures that show a high potential to preserve local dung beetle diversity [72]. In our study, as the mean richness of dung beetles varied between the two study areas, we highlight the need to consider the interaction between local grazing pressures and altitude to plan effective management strategies. Moreover, the high abundance of tunnelers in pastures is key for ecosystem services. Through their digging activities, they in fact contribute to aerating soil and to promoting the fertility of pasture areas [18].
We contributed evidence to support the context dependency of the edge effect as refugia for wood and pasture dung beetles. Because of the high flight ability due to the unpredictability of foraging resources, dung beetles could be less dependent on local habitat characteristics than other groups (e.g., carabids) while being more susceptible to large-scale habitat configuration. Because multiple interactive factors can shape community composition and dynamics, more studies are needed to understand the relative importance of local and regional processes in alpine ecosystems.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/environments11080178/s1, All figures should be included in the SM part following the information: Figure S1: Pairwise differences of temperature in the pasture habitat among the four investigated sites. Mean (black continuous line) and confidence intervals (grey dashed lines) are shown; Figure S2. Pairwise differences of temperature in the edge habitat among the four investigated sites. Mean (black continuous line) and confidence intervals (grey dashed lines) are shown; Figure S3. Pairwise differences of temperature in the outer wood habitat among the four investi-gated sites. Mean (black continuous line) and confidence intervals (grey dashed lines) are shown; Figure S4. Pairwise differences of temperature in the inner wood habitat among the four investi-gated sites. Mean (black continuous line) and confidence intervals (grey dashed lines) are shown. The data logger of site 21 was not working; Figure S5. Pairwise differences of temperature in different habitats of site 1_1. Mean (black con-tinuous line) and confidence intervals (grey dashed lines) are shown; Figure S6. Pairwise differences of temperature in different habitats of site 1_2. Mean (black con-tinuous line) and confidence intervals (grey dashed lines) are shown; Figure S7. Pairwise differences of temperature in different habitats of site 2_1. Mean (black con-tinuous line) and confidence intervals (grey dashed lines) are shown. The data logger of the inner wood habitat was not working; Figure S8. Pairwise differences of temperature in different habitats of site 2_2. Mean (black con-tinuous line) and confidence intervals (grey dashed lines) are shown; Figure S9. Pairwise comparison of species number between habitats within areas; Figure S10. Pairwise comparison of species number between areas within habitat; Figure S11. Pairwise comparison of abundance between habitats within areas; Figure S12. Pairwise comparison of dwellers percentage between habitats within areas; Figure S13. Pairwise comparison of dwellers percentage between areas within habitat; Figure S14. Pairwise comparison of tunnelers percentage between habitats within areas; Figure S15. Pairwise comparison of tunnellers percentage between areas within habitat; Figure S16. Temporal richness of species richness in the four study sites; Figure S17. Temporal richness of abundance in the four study sites; Figure S18. Temporal richness of dwellers’ percentage in the four study sites; Figure S19. Temporal richness of tunnellers’ percentage in the four study sites; Table S1: Species abundances for each site and habitat.
Author Contributions
Conceptualization, A.R. (Antonio Rolando) and C.P.; research coordination, C.P.; methodology, A.R. (Angela Roggero) and M.C.; formal analysis, A.L.; writing—original draft preparation, A.L. and A.R. (Antonio Rolando), writing—review and editing, all authors. All authors have read and agreed to the published version of the manuscript.
Funding
The work was supported by the project CN_00000033 funded under the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.4—Call for tender No. 3138 of 16 December 2021, rectified by Decree n. 3175 of 18 December 2021 of the Italian Ministry of University and Research, funded by the European Union–NextGenerationEU.
Data Availability Statement
Data are available from the authors upon reasonable request due to the reasons. (e.g., privacy, legal or ethical reasons).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Tittensor, D.P.; Walpole, M.; Hill, S.L.L.; Boyce, D.G.; Britten, G.L.; Burgess, N.D.; Butchart, S.H.M.; Leadley, P.W.; Regan, E.C.; Alkemade, R.; et al. A Mid-Term Analysis of Progress toward International Biodiversity Targets. Science 2014, 346, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Forister, M.L.; Pelton, E.M.; Black, S.H. Declines in Insect Abundance and Diversity: We Know Enough to Act Now. Conserv. Sci. Pract. 2019, 1, e80. [Google Scholar] [CrossRef]
- Habel, J.C.; Samways, M.J.; Schmitt, T. Mitigating the Precipitous Decline of Terrestrial European Insects: Requirements for a New Strategy. Biodivers. Conserv. 2019, 28, 1343–1360. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Ssymank, A.; Sorg, M.; de Kroon, H.; Jongejans, E. Insect Biomass Decline Scaled to Species Diversity: General Patterns Derived from a Hoverfly Community. Proc. Natl. Acad. Sci. USA 2021, 118, e2002554117. [Google Scholar] [CrossRef] [PubMed]
- Rusch, A.; Bommarco, R.; Chiverton, P.; Öberg, S.; Wallin, H.; Wiktelius, S.; Ekbom, B. Response of Ground Beetle (Coleoptera, Carabidae) Communities to Changes in Agricultural Policies in Sweden over Two Decades. Agric. Ecosyst. Environ. 2013, 176, 63–69. [Google Scholar] [CrossRef]
- Jovičić, S.; Burgio, G.; Diti, I.; Krašić, D.; Markov, Z.; Radenković, S.; Vujić, A. Influence of Landscape Structure and Land Use on Merodon and Cheilosia (Diptera: Syrphidae): Contrasting Responses of Two Genera. J. Insect Conserv. 2017, 21, 53–64. [Google Scholar] [CrossRef]
- Fox, R. The Decline of Moths in Great Britain: A Review of Possible Causes. Insect Conserv. Divers. 2013, 6, 5–19. [Google Scholar] [CrossRef]
- Gossner, M.M.; Lewinsohn, T.M.; Kahl, T.; Grassein, F.; Boch, S.; Prati, D.; Birkhofer, K.; Renner, S.C.; Sikorski, J.; Wubet, T.; et al. Land-Use Intensification Causes Multitrophic Homogenization of Grassland Communities. Nature 2016, 540, 266–269. [Google Scholar] [CrossRef]
- Holter, P. Herbivore Dung as Food for Dung Beetles: Elementary Coprology for Entomologists. Ecol. Entomol. 2016, 41, 367–377. [Google Scholar] [CrossRef]
- Buse, J.; Hoenselaar, G.; Langenbach, F.; Langenbach, F.; Schleicher, P.; Twietmeyer, S.; Popa, F.; Heurich, M. Dung beetle richness is positively affected by the density of wild ungulate populations in forests. Biodivers. Conserv. 2021, 30, 3115–3131. [Google Scholar] [CrossRef]
- Halffter, G.; Edmonds, W. The Nesting Behavior of Dung Beetles (Scarabaeinae); Publication 10; Instituto de Ecologia: Mexico City, Mexico, 1982; 176p. [Google Scholar]
- Kaleri, A.R.; Ma, J.; Abro, S.A.; Faqir, Y.; Nabi, F.; Hakeem, A.; Ahmed, A.; Ahmed, S.; Jakhar, A.M.; Shah, S.M.; et al. Dung Beetle Improves Soil Bacterial Diversity and Enzyme Activity and Enhances Growth and Antioxidant Content of Chinese Cabbage (Brassica rapa ssp. pekinensis). J. Soil Sci. Plant Nutr. 2021, 21, 3387–3401. [Google Scholar] [CrossRef]
- Stanbrook, R.; Harris, E.; Jones, M.; Wheater, C.P. The Effect of Dung Beetle Size on Soil Nutrient Mobilization in an Afrotropical Forest. Insects 2021, 12, 141. [Google Scholar] [CrossRef]
- Yamada, D.; Imura, O.; Shi, K.; Shibuya, T. Effect of Tunneler Dung Beetles on Cattle Dung Decomposition, Soil Nutrients and Herbage Growth. Grassl. Sci. 2007, 53, 121–129. [Google Scholar] [CrossRef]
- Milotić, T.; Baltzinger, C.; Eichberg, C.; Eycott, A.E.; Heurich, M.; Müller, J.; Noriega, J.A.; Menendez, R.; Stadler, J.; Ádám, R.; et al. Functionally Richer Communities Improve Ecosystem Functioning: Dung Removal and Secondary Seed Dispersal by Dung Beetles in the Western Palaearctic. J. Biogeogr. 2019, 46, 70–82. [Google Scholar] [CrossRef]
- Andresen, E. Dung Beetles in a Central Amazonian Rainforest and Their Ecological Role as Secondary Seed Dispersers. Ecol. Entomol. 2002, 27, 257–270. [Google Scholar] [CrossRef]
- Nervo, B.; Caprio, E.; Celi, L.; Lonati, M.; Lombardi, G.; Falsone, G.; Iussig, G.; Palestrini, C.; Said-Pullicino, D.; Rolando, A. Ecological Functions Provided by Dung Beetles Are Interlinked across Space and Time: Evidence from 15N Isotope Tracing. Ecology 2017, 98, 433–446. [Google Scholar] [CrossRef]
- Nichols, E.; Gómez, A. Dung Beetles and Fecal Helminth Transmission: Patterns, Mechanisms and Questions. Parasitology 2014, 141, 614–623. [Google Scholar] [CrossRef]
- Andresen, E.; Laurance, S.G.W. Possible Indirect Effects of Mammal Hunting on Dung Beetle Assemblages in Panama. Biotropica 2007, 39, 141–146. [Google Scholar] [CrossRef]
- Barbero, E.; Palestrini, C.; Rolando, A. Dung Beetle Conservation: Effects of Habitat and Resource Selection (Coleoptera: Scarabaeoidea). J. Insect Conserv. 1999, 3, 75–84. [Google Scholar] [CrossRef]
- Gebert, F.; Steffan-Dewenter, I.; Moretto, P.; Peters, M.K. Climate Rather than Dung Resources Predict Dung Beetle Abundance and Diversity along Elevational and Land Use Gradients on Mt. Kilimanjaro. J. Biogeogr. 2020, 47, 371–381. [Google Scholar] [CrossRef]
- Hanski, I.; Cambefort, Y. Dung Beetle Ecology; Princeton University Press: Princeton, NJ, USA, 1991; ISBN 978-0-691-08739-9. [Google Scholar]
- Nichols, E.; Uriarte, M.; Bunker, D.E.; Favila, M.E.; Slade, E.M.; Vulinec, K.; Larsen, T.; Vaz-de-Mello, F.Z.; Louzada, J.; Naeem, S.; et al. Trait-Dependent Response of Dung Beetle Populations to Tropical Forest Conversion at Local and Regional Scales. Ecology 2013, 94, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Korasaki, V.; Braga, R.F.; Zanetti, R.; Moreira, F.M.S.; Vaz-de-Mello, F.Z.; Louzada, J. Conservation Value of Alternative Land-Use Systems for Dung Beetles in Amazon: Valuing Traditional Farming Practices. Biodivers. Conserv. 2013, 22, 1485–1499. [Google Scholar] [CrossRef]
- Costa, F.C.; Pessoa, K.K.T.; Liberal, C.N.; Filgueiras, B.K.C.; Salomão, R.P.; Iannuzzi, L. What Is the Importance of Open Habitat in a Predominantly Closed Forest Area to the Dung Beetle (Coleoptera, Scarabaeinae) Assemblage? Rev. Bras. Entomol. 2013, 57, 329–334. [Google Scholar] [CrossRef]
- Giménez Gómez, V.C.; Verdú, J.R.; Gómez-Cifuentes, A.; Vaz-de-Mello, F.Z.; Zurita, G.A. Influence of Land Use on the Trophic Niche Overlap of Dung Beetles in the Semideciduous Atlantic Forest of Argentina. Insect Conserv. Divers. 2018, 11, 554–564. [Google Scholar] [CrossRef]
- Nichols, E.; Spector, S.; Louzada, J.; Larsen, T.; Amezquita, S.; Favila, M.E. Ecological Functions and Ecosystem Services Provided by Scarabaeinae Dung Beetles. Biol. Conserv. 2008, 141, 1461–1474. [Google Scholar] [CrossRef]
- Silva, P.G.d.; Hernández, M.I.M. Spatial Variation of Dung Beetle Assemblages Associated with Forest Structure in Remnants of Southern Brazilian Atlantic Forest. Rev. Bras. Entomol. 2016, 60, 73–81. [Google Scholar] [CrossRef]
- Chamberlain, D.; Tocco, C.; Longoni, A.; Mammola, S.; Palestrini, C.; Rolando, A. Nesting Strategies Affect Altitudinal Distribution and Habitat Use in Alpine Dung Beetle Communities. Ecol. Entomol. 2015, 40, 372–380. [Google Scholar] [CrossRef]
- Martello, F.; Andriolli, F.; de Souza, T.B.; Dodonov, P.; Ribeiro, M.C. Edge and Land Use Effects on Dung Beetles (Coleoptera: Scarabaeidae: Scarabaeinae) in Brazilian Cerrado Vegetation. J. Insect Conserv. 2016, 20, 957–970. [Google Scholar] [CrossRef]
- Gómez-Cifuentes, A.; Munevar, A.; Gimenez, V.C.; Gatti, M.G.; Zurita, G.A. Influence of Land Use on the Taxonomic and Functional Diversity of Dung Beetles (Coleoptera: Scarabaeinae) in the Southern Atlantic Forest of Argentina. J. Insect Conserv. 2017, 21, 147–156. [Google Scholar] [CrossRef]
- Laurance, W.F.; Lovejoy, T.E.; Vasconcelos, H.L.; Bruna, E.M.; Didham, R.K.; Stouffer, P.C.; Gascon, C.; Bierregaard, R.O.; Laurance, S.G.; Sampaio, E. Ecosystem Decay of Amazonian Forest Fragments: A 22-Year Investigation. Conserv. Biol. 2002, 16, 605–618. [Google Scholar] [CrossRef]
- Shmida, A.; Wilson, M.V. Biological Determinants of Species Diversity. J. Biogeogr. 1985, 12, 1–20. [Google Scholar] [CrossRef]
- Martínez-Falcón, A.P.; Zurita, G.A.; Ortega-Martínez, I.J.; Moreno, C.E. Populations and Assemblages Living on the Edge: Dung Beetles Responses to Forests-Pasture Ecotones. PeerJ 2018, 6, e6148. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.J.; Pelissari, T.D.; Krinski, D.; Canale, G.; Vaz-de-Mello, F.Z. Abrupt Species Loss of the Amazonian Dung Beetle in Pastures Adjacent to Species-Rich Forests. J. Insect Conserv. 2017, 21, 487–494. [Google Scholar] [CrossRef]
- Marsh, C.J.; Feitosa, R.M.; Louzada, J.; Ewers, R.M. Is β-Diversity of Amazonian Ant and Dung Beetles Communities Elevated at Rainforest Edges? J. Biogeogr. 2018, 45, 1966–1979. [Google Scholar] [CrossRef]
- Tocco, C.; Negro, M.; Rolando, A.; Palestrini, C. Does Natural Reforestation Represent a Potential Threat to Dung Beetle Diversity in the Alps? J. Insect Conserv. 2013, 17, 207–217. [Google Scholar] [CrossRef]
- Tocco, C.; Probo, M.; Lonati, M.; Lombardi, G.; Negro, M.; Nervo, B.; Rolando, A.; Palestrini, C. Pastoral Practices to Reverse Shrub Encroachment of Sub-Alpine Grasslands: Dung Beetles (Coleoptera, Scarabaeoidea) Respond More Quickly Than Vegetation. PLoS ONE 2013, 8, e83344. [Google Scholar] [CrossRef] [PubMed]
- Palestrini, C.; Roggero, A.; Gorret, R.; Tocco, C.; Negro, M.; Barbero, E. Scarabaeoidea coprofagi della Val Veni e della Val Ferret (Valle d’Aosta, Italia). Rev. Valdôtaine Hist. Nat. 2007, 61–62, 241–253. [Google Scholar]
- Dellacasa, G.; Dellacasa, M. Coleoptera. Aphodiidae-Aphodiinae; Edagricole Calderini: Bologna, Italy, 2010; ISBN 978-88-506-5203-7. [Google Scholar]
- QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project. 2024. Available online: http://qgis.osgeo.org (accessed on 21 July 2024).
- Wood, S.N. Fast Stable Restricted Maximum Likelihood and Marginal Likelihood Estimation of Semiparametric Generalized Linear Models. J. R. Stat. Soc. Ser. B Stat. Methodol. 2011, 73, 3–36. [Google Scholar] [CrossRef]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2024. [Google Scholar]
- Simpson, G.L.; Singmann, H. Gratia: Graceful ’ggplot’-Based Graphics and Other Functions for GAMs Fitted Using “mgcv”. R Package Version 0.9.2. 2024. Available online: https://CRAN.R-project.org/package=gratia (accessed on 21 July 2024).
- Gao, C.; Dusa, A. ggVennDiagram: A ‘ggplot2’ Implement of Venn Diagram. R Package Version 1.5.2. 2024. Available online: https://CRAN.R-project.org/package=ggVennDiagram (accessed on 21 July 2024).
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef]
- Bates, D.; Machler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using Lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Doube, B.M. A Functional Classification for Analysis of the Structure of Dung Beetle Assemblages. Ecol. Entomol. 1990, 15, 371–383. [Google Scholar] [CrossRef]
- Laliberté, E.; Legendre, P. A Distance-Based Framework for Measuring Functional Diversity from Multiple Traits. Ecology 2010, 91, 299–305. [Google Scholar] [CrossRef]
- Lenth, R.; Buerkner, P.; Herve, M.; Love, J.; Riebl, H.; Singmann, H. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. R Package Version 1.10.3. 2024. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 21 July 2024).
- Oksanen, J.; Simpson, G.; Blanchet, F.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.; Solymos, P.; Stevens, M.; Szoecs, E.; et al. vegan: Community Ecology Package. R Package Version 2.6–6.1. 2024. Available online: https://CRAN.R-project.org/package=vegan (accessed on 21 July 2024).
- Blonder, B.; Morrow, C.B.; Brown, S.; Butruille, G.; Chen, D.; Laini, A.; Harris, D.J. Hypervolume: High Dimensional Geometry, Set Operations, Projection, and Inference Using Kernel Density Estimation, Support Vector Machines, and Convex Hulls. R package Version 3.1.4. 2024. Available online: https://CRAN.R-project.org/package=hypervolume (accessed on 21 July 2024).
- Cabrero-Sañudo, F.J.; Lobo, J.M. Biogeography of Aphodiinae Dung Beetles Based on the Regional Composition and Distribution Patterns of Genera. J. Biogeogr. 2009, 36, 1474–1492. [Google Scholar] [CrossRef]
- Negro, M.; Palestrini, C.; Giraudo, M.T.; Rolando, A. The Effect of Local Environmental Heterogeneity on Species Diversity of Alpine Dung Beetles (Coleoptera: Scarabaeidae). Eur. J. Entomol. 2013, 108, 91–98. [Google Scholar] [CrossRef]
- Noriega, J.A.; Realpe, E. Altitudinal Turnover of Species in a Neotropical Peripheral Mountain System: A Case Study with Dung Beetles (Coleoptera: Aphodiinae and Scarabaeinae). Environ. Entomol. 2018, 47, 1376–1387. [Google Scholar] [CrossRef]
- Negro, M.; Rolando, A.; Palestrini, C. The Impact of Overgrazing on Dung Beetle Diversity in the Italian Maritime Alps. Environ. Entomol. 2011, 40, 1081–1092. [Google Scholar] [CrossRef] [PubMed]
- Perrin, W.; Moretti, M.; Vergnes, A.; Borcard, D.; Jay-Robert, P. Response of Dung Beetle Assemblages to Grazing Intensity in Two Distinct Bioclimatic Contexts. Agric. Ecosyst. Environ. 2020, 289, 106740. [Google Scholar] [CrossRef]
- Verdú, J.R.; Lobo, J.M.; Sánchez-Piñero, F.; Gallego, B.; Numa, C.; Lumaret, J.-P.; Cortez, V.; Ortiz, A.J.; Tonelli, M.; García-Teba, J.P.; et al. Ivermectin Residues Disrupt Dung Beetle Diversity, Soil Properties and Ecosystem Functioning: An Interdisciplinary Field Study. Sci. Total Environ. 2018, 618, 219–228. [Google Scholar] [CrossRef]
- Buse, J.; Šlachta, M.; Sladecek, F.X.J.; Pung, M.; Wagner, T.; Entling, M.H. Relative Importance of Pasture Size and Grazing Continuity for the Long-Term Conservation of European Dung Beetles. Biol. Conserv. 2015, 187, 112–119. [Google Scholar] [CrossRef]
- Durães, R.; Martins, W.P.; Vaz-de-Mellos, F.Z. Dung Beetle (Coleoptera: Scarabaeidae) Assemblages across a Natural Forest-Cerrado Ecotone in Minas Gerais, Brazil. Neotrop. Entomol. 2005, 34, 721–731. [Google Scholar] [CrossRef][Green Version]
- Villada-Bedoya, S.; Cultid-Medina, C.A.; Escobar, F.; Guevara, R.; Zurita, G. Edge Effects on Dung Beetle Assemblages in an Andean Mosaic of Forest and Coffee Plantations. Biotropica 2017, 49, 195–205. [Google Scholar] [CrossRef]
- Nervo, B.; Roggero, A.; Isaia, M.; Chamberlain, D.; Rolando, A.; Palestrini, C. Integrating Thermal Tolerance, Water Balance and Morphology: An Experimental Study on Dung Beetles. J. Therm. Biol. 2021, 101, 103093. [Google Scholar] [CrossRef]
- deCastro-Arrazola, I.; Andrew, N.R.; Berg, M.P.; Curtsdotter, A.; Lumaret, J.-P.; Menéndez, R.; Moretti, M.; Nervo, B.; Nichols, E.S.; Sánchez-Piñero, F.; et al. A Trait-Based Framework for Dung Beetle Functional Ecology. J. Anim. Ecol. 2023, 92, 44–65. [Google Scholar] [CrossRef]
- Nervo, B.; Roggero, A.; Chamberlain, D.; Caprio, E.; Rolando, A.; Palestrini, C. Physiological, Morphological and Ecological Traits Drive Desiccation Resistance in North Temperate Dung Beetles. BMC Zool. 2021, 6, 26. [Google Scholar] [CrossRef]
- Nervo, B.; Laini, A.; Roggero, A.; Palestrini, C.; Rolando, A. Spatio-Temporal Modelling Suggests That Some Dung Beetle Species (Coleoptera: Geotrupidae) May Respond to Global Warming by Boosting Dung Removal. Sci. Total Environ. 2024, 908, 168127. [Google Scholar] [CrossRef]
- Menéndez, R.; Gutiérrez, D. Shifts in Habitat Associations of Dung Beetles in Northern Spain: Climate Change Implications. Écoscience 2004, 11, 329–337. [Google Scholar] [CrossRef]
- Menéndez, R.; González-Megías, A.; Jay-Robert, P.; Marquéz-Ferrando, R. Climate Change and Elevational Range Shifts: Evidence from Dung Beetles in Two European Mountain Ranges. Glob. Ecol. Biogeogr. 2014, 23, 646–657. [Google Scholar] [CrossRef]
- Numa, C.; Verdú, J.R.; Sánchez, A.; Galante, E. Effect of Landscape Structure on the Spatial Distribution of Mediterranean Dung Beetle Diversity. Divers. Distrib. 2009, 15, 489–501. [Google Scholar] [CrossRef]
- Romero-Alcaraz, E.; Ávila, J.M. Landscape Heterogeneity in Relation to Variations in Epigaeic Beetle Diversity of a Mediterranean Ecosystem. Implications for Conservation. Biodivers. Conserv. 2000, 9, 985–1005. [Google Scholar] [CrossRef]
- Boan, J.J.; McLaren, B.E.; Malcolm, J.R. Influence of Post-Harvest Silviculture on Understory Vegetation: Implications for Forage in a Multi-Ungulate System. For. Ecol. Manag. 2011, 262, 1704–1712. [Google Scholar] [CrossRef]
- Englmeier, J.; von Hoermann, C.; Rieker, D.; Benbow, M.E.; Benjamin, C.; Fricke, U.; Ganuza, C.; Haensel, M.; Lackner, T.; Mitesser, O.; et al. Dung-Visiting Beetle Diversity Is Mainly Affected by Land Use, While Community Specialization Is Driven by Climate. Ecol. Evol. 2022, 12, e9386. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, M.; Verdú, J.R.; Zunino, M. Effects of the Progressive Abandonment of Grazing on Dung Beetle Biodiversity: Body Size Matters. Biodivers. Conserv. 2018, 27, 189–204. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).