Evaluation of Black Soldier Fly (Hermetia illucens) Larvae and Pre-Pupae Raised on Household Organic Waste, as Potential Ingredients for Poultry Feed
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Production of Black Soldier Flies
2.2. Diets
2.3. Animals and Experimental Design
2.4. Villus Height and Crypt Depth
2.5. Egg Quality
2.6. Calcium, Inorganic Phosphorus and Magnesium Concentration in the Plasma
2.7. SCFA in the Cecal Content
2.8. PCR Amplicon Sequencing of 16S rRNA Genes
2.9. Taxonomic Analysis Based on 16S rDNA
2.10. Statistical Analysis
3. Results
3.1. BSF Larvae and Pre-Pupae Composition
3.2. Body Weight, Liver Weight, and Egg-Laying Rate
3.3. Villus Height, Crypt Depth and Goblet Cell Density
3.4. Egg Quality
3.5. Concentrations of Calcium, Inorganic Phosphorus and Magnesium in Plasma
3.6. SCFA in the Cecal Content
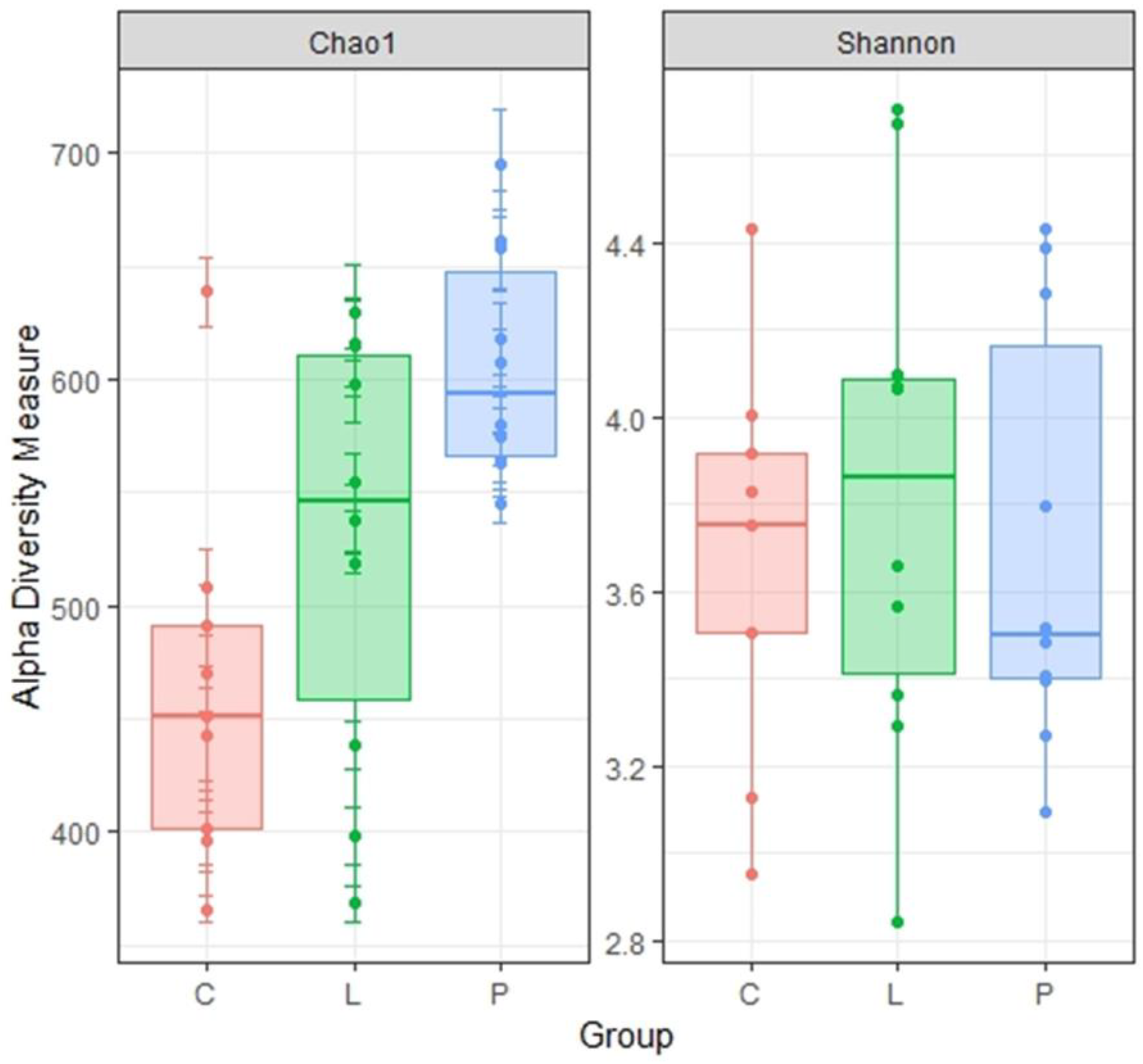
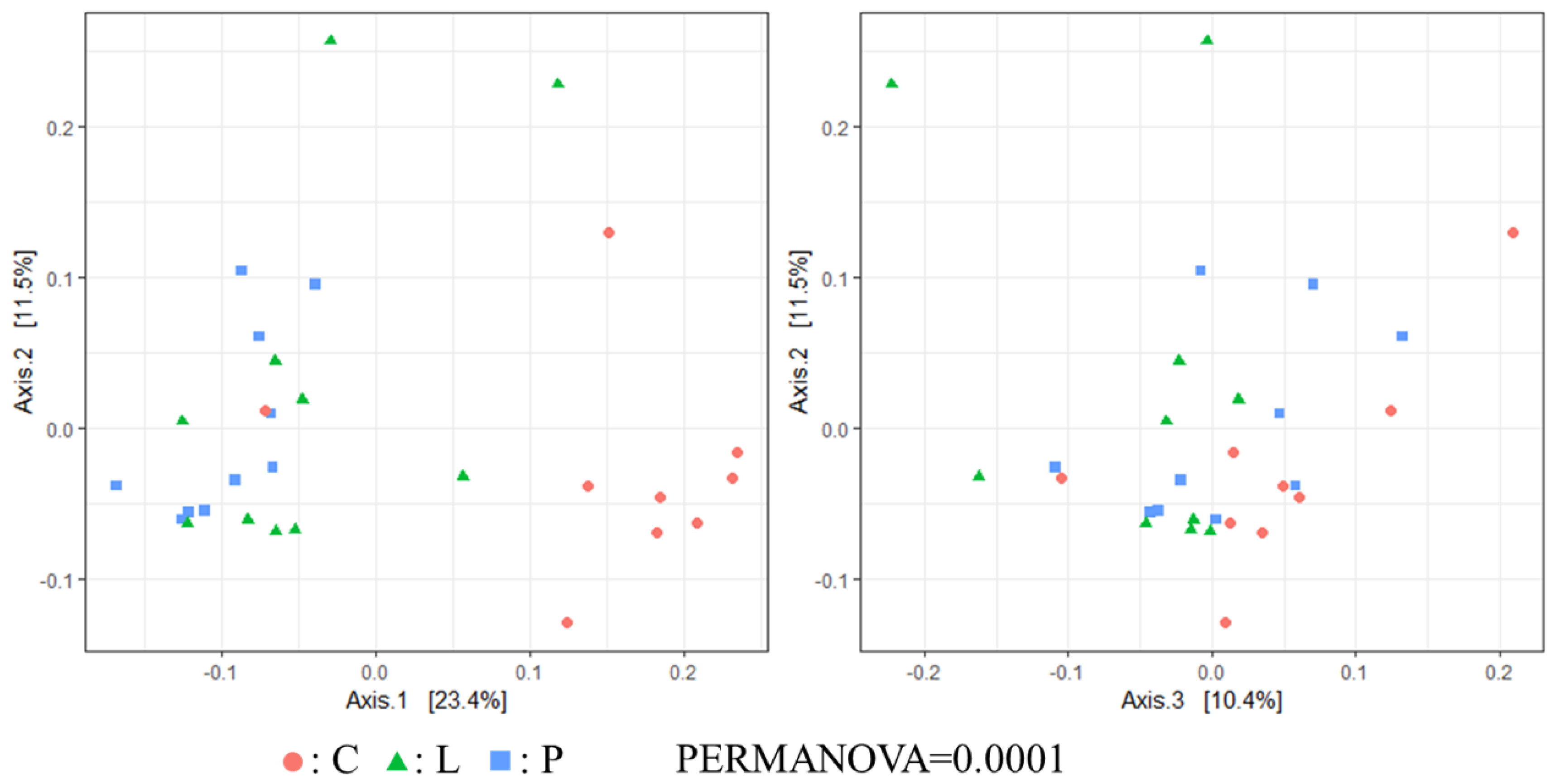
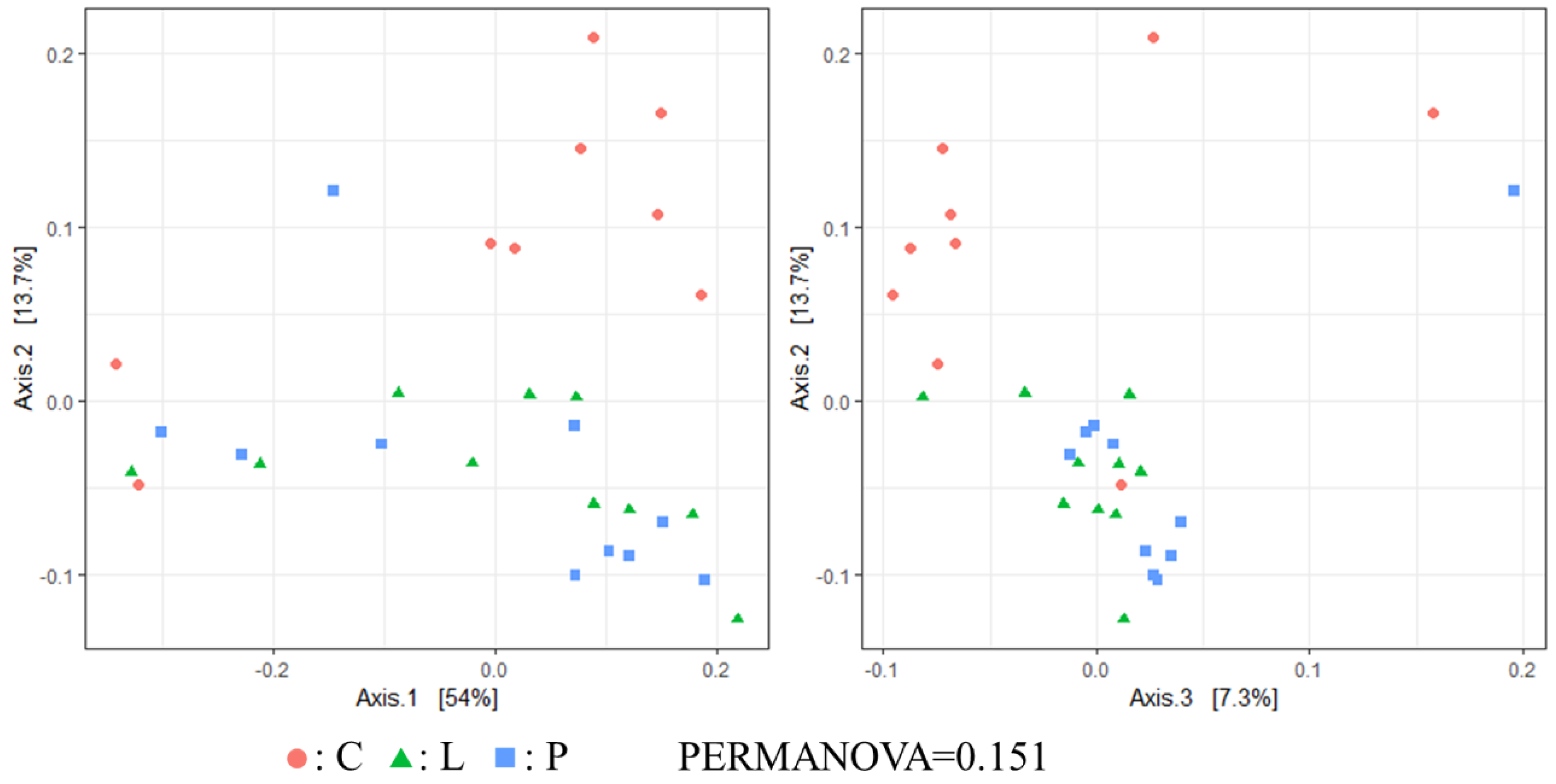
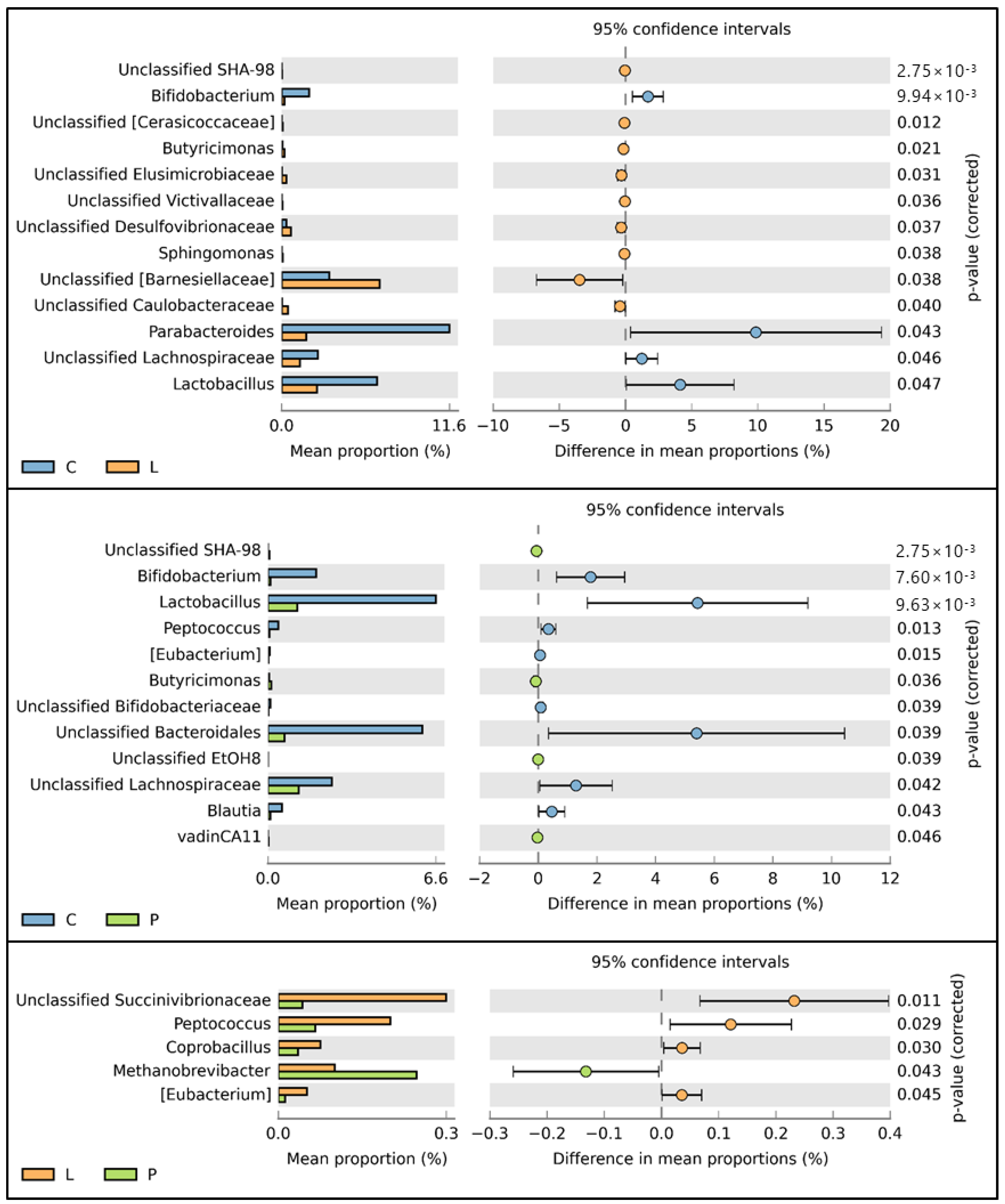
3.7. Cecal Microbiota
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Marco, M.; Martínez, S.; Hernandez, F.; Madrid, J.; Gai, F.; Rotolo, L.; Belforti, M.; Bergero, D.; Katz, H.; Dabbou, S.; et al. Nutritional value of two insect larval meals (Tenebrio molitor and Hermetia illucens) for broiler chickens: Apparent nutrient digestibility, apparent ileal amino acid digestibility and apparent metabolizable energy. Anim. Feed Sci. Technol. 2015, 209, 211–218. [Google Scholar] [CrossRef]
- Maurer, V.; Holinger, M.; Amsler, Z.; Früh, B.; Wohlfahrt, J.; Stamer, A.; Leiber, F. Replacement of soybean cake by Hermetia illucens meal in diets for layers. J. Insects Food Feed 2016, 2, 83–90. [Google Scholar] [CrossRef]
- Schiavone, A.; De Marco, M.; Martínez, S.; Dabbou, S.; Renna, M.; Madrid, J.; Hernandez, F.; Rotolo, L.; Costa, P.; Gai, F.; et al. Nutritional value of a partially defatted and a highly defatted black soldier fly larvae (Hermetia illucens L.) meal for broiler chickens: Apparent nutrient digestibility, apparent metabolizable energy and apparent ileal amino acid digestibility. J. Anim. Sci. Biotechnol. 2017, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cutrignelli, M.I.; Messina, M.; Tulli, F.; Randazzi, B.; Olivotto, I.; Gasco, L.; Loponte, R.; Bovera, F. Evaluation of an insect meal of the Black Soldier Fly (Hermetia illucens) as soybean substitute: Intestinal morphometry, enzymatic and microbial activity in laying hens. Res. Vet. Sci. 2018, 117, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Kupferschmidt, K. Buzz food. Science 2015, 350, 267–269. [Google Scholar] [CrossRef]
- Wang, Y.-S.; Shelomi, M. Review of black soldier fly (Hermetia illucens) as animal feed and human food. Foods 2017, 6, 91. [Google Scholar] [CrossRef]
- Moula, N.; Scippo, M.L.; Douny, C.; Degand, G.; Dawans, E.; Cabaraux, J.F.; Hornick, J.L.; Medigo, R.C.; Leroy, P.; Francis, F.; et al. Performances of local poultry breed fed black soldier fly larvae reared on horse manure. Anim. Nutr. 2018, 4, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Schiavone, A.; Cullere, M.; De Marco, M.; Meneguz, M.; Biasato, I.; Bergagna, S.; Dezzutto, D.; Gai, F.; Dabbou, S.; Gasco, L.; et al. Partial or total replacement of soybean oil by black soldier fly larvae (Hermetia illucens L.) fat in broiler diets: Effect on growth performances, feed-choice, blood traits, carcass characteristics and meat quality. Ital. J. Anim. Sci. 2017, 16, 93–100. [Google Scholar] [CrossRef]
- Marono, S.; Loponte, R.; Lombardi, P.; Vassalotti, G.; Pero, M.E.; Russo, F.; Gasco, L.; Parisi, G.; Piccolo, G.; Nizza, S.; et al. Productive performance and blood profiles of laying hens fed Hermetia illucens larvae meal as total replacement of soybean meal from 24 to 45 weeks of age. Poult. Sci. 2017, 96, 1783–1790. [Google Scholar] [CrossRef] [PubMed]
- Park, S.O.; Park, K.H.; Park, B.S.; Nam, S.H.; Choi, C.Y. Effect of dietary black soldier fly (Hermetia illucens (Diptera: Stratmyidae) pupa on egg production in laying hens. Korean J. Nat. Conserv. 2013, 7, 136–141. [Google Scholar] [CrossRef]
- Mwaniki, Z.; Neijat, M.; Kiarie, E. Egg production and quality responses of adding up to 7.5% defatted black soldier fly larvae meal in a corn–soybean meal diet fed to Shaver White Leghorns from wk 19 to 27 of age. Poult. Sci. 2018, 97, 2829–2835. [Google Scholar] [CrossRef] [PubMed]
- Al-Qazzaz, M.F.A.; Ismail, D.; Akit, H.; Idris, L.H. Effect of using insect larvae meal as a complete protein source on quality and productivity characteristics of laying hens. Rev. Bras. Zootec. 2016, 45, 518–523. [Google Scholar] [CrossRef]
- Ruhnke, I.; Normant, C.; Campbell, D.L.M.; Iqbal, Z.; Lee, C.; Hinch, G.N.; Roberts, J. Impact of on-range choice feeding with black soldier fly larvae (Hermetia illucens) on flock performance, egg quality, and range use of free-range laying hens. Anim. Nutr. 2018, 4, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Waśko, A.; Bulak, P.; Polak-Berecka, M.; Nowak, K.; Polakowski, C.; Bieganowski, A. The first report of the physicochemical structure of chitin isolated from Hermetia illucens. Int. J. Biol. Macromol. 2016, 92, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, L.; Coretti, L.; Dipineto, L.; Bovera, F.; Menna, F.; Chiariotti, L.; Nizza, A.; Lembo, F.; Fioretti, A. Insect-based diet, a promising nutritional source, modulates gut microbiota composition and SCFAs production in laying hens. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Caligiani, A.; Marseglia, A.; Leni, G.; Baldassarre, S.; Maistrello, L.; Dossena, A.; Sforza, S. Composition of black soldier fly prepupae and systematic approaches for extraction and fractionation of proteins, lipids and chitin. Food Res. Int. 2018, 105, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.; Ichiki, R.T.; Shimoda, M.; Morioka, S. Small-scale rearing of the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae), in the laboratory: Low-cost and year-round rearing. Appl. Entomol. Zool. 2016, 51, 161–166. [Google Scholar] [CrossRef]
- Çevikkalp, S.A.; Löker, G.B.; Yaman, M.; Amoutzopoulos, B. A simplified HPLC method for determination of tryptophan in some cereals and legumes. Food Chem. 2016, 193, 26–29. [Google Scholar] [CrossRef]
- AOAC (Association of Official Analytical Chemists). Official Methods of Analysis of the Association of Official Analytical Chemists, 17th ed.; Horwitz, W., Ed.; AOAC International: Gaithersburg, MA, USA, 2002; ISBN 0-93558-67-6. [Google Scholar]
- National Agriculture and Food Research Organization. Japanese Feeding Standard for Poultry; Japanese Livestock Industry Association: Tsukuba, Japan, 2011; ISBN 4901311581.
- Marono, S.; Piccolo, G.; Loponte, R.; Di Meo, C.; Attia, Y.A.; Nizza, A.; Bovera, F. In vitro crude protein digestibility of tenebrio molitor and hermetia illucens insect meals and its correlation with chemical composition traits. Ital. J. Anim. Sci. 2015, 14, 338–343. [Google Scholar] [CrossRef]
- Xiao, L.; Xiao, M.; Jin, X.; Kawasaki, K.; Ohta, N.; Sakaguchi, E. Transfer of blood urea nitrogen to cecal microbial nitrogen is increased by mannitol feeding in growing rabbits fed timothy hay diet. Animal 2012, 6, 1757–1763. [Google Scholar] [CrossRef]
- Klindworth, A.; Peplies, J.; Pruesse, E.; Schweer, T.; Glöckner, F.O.; Quast, C.; Horn, M. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucl. Acids Res. 2012, 41, e1. [Google Scholar] [CrossRef]
- Knight, R.; Clemente, J.C.; Haas, B.J.; Edgar, R.C.; Quince, C. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar]
- Navas-Molina, J.A.; Peralta-Sánchez, J.M.; González, A.; McMurdie, P.J.; Vázquez-Baeza, Y.; Xu, Z.; Ursell, L.K.; Lauber, C.; Zhou, H.; Song, S.J.; et al. Advancing our Understanding of the Human Microbiome Using QIIME, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2013; Volume 531, ISBN 9780124078635. [Google Scholar]
- McMurdie, P.J.; Holmes, S. Phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef]
- Parks, D.H.; Tyson, G.W.; Hugenholtz, P.; Beiko, R.G. STAMP: Statistical analysis of taxonomic and functional profiles. Bioinformatics 2014, 30, 3123–3124. [Google Scholar] [CrossRef]
- Tortora, G.J.; Derrickson, B. Introduction to the Human Body, 10th ed.; John Wiley & Sons, Inc.: New York, NY, USA, 2015; ISBN 1118583183. [Google Scholar]
- Baurhoo, B.; Phillip, L.; Ruiz-Feria, C.A. Effects of purified lignin and mannan oligosaccharides on intestinal integrity and microbial populations in the ceca and litter of broiler chickens. Poult. Sci. 2007, 86, 1070–1078. [Google Scholar] [CrossRef]
- An, S.H.; Kim, D.W.; An, B.K. Effects of dietary calcium levels on productive performance, eggshell quality and overall calcium status in aged laying hens. Asian Aust. J. Anim. Sci. 2016, 29, 1477–1482. [Google Scholar] [CrossRef]
- Nys, Y. Nutritional factors affecting eggshell quality. Czech J. Anim. Sci. 1999, 44, 135–143. [Google Scholar]
- Lutz, T.; Scharrer, E. Effect of short-chain fatty acids on calcium absorption by the rat colon. Exp. Physiol. 1991, 76, 615–618. [Google Scholar] [CrossRef]
- Bedford, A.; Gong, J. Implications of butyrate and its derivatives for gut health and animal production. Anim. Nutr. 2018, 4, 151–159. [Google Scholar] [CrossRef]
- Mountzouris, K.C.; Tsirtsikos, P.; Kalamara, E.; Nitsch, S.; Schatzmayr, G.; Fegeros, K. Evaluation of the efficacy of a probiotic containing Lactobacillus, Bifidobacterium, Enterococcus, and Pediococcus strains in promoting broiler performance and modulating cecal microflora composition and metabolic activities. Poult. Sci. 2014, 86, 309–317. [Google Scholar] [CrossRef]
- Santini, C.; Baffoni, L.; Gaggia, F.; Granata, M.; Gasbarri, R.; Di Gioia, D.; Biavati, B. Characterization of probiotic strains: An application as feed additives in poultry against Campylobacter jejuni. Int. J. Food Microbiol. 2010, 141, S98–S108. [Google Scholar] [CrossRef]
- Forte, C.; Acuti, G.; Manuali, E.; Casagrande-Proietti, P.; Pavone, S.; Trabalza-Marinucci, M.; Moscati, L.; Onofri, A.; Lorenzetti, C. Effects of two different probiotics on microflora, morphology, and morphometry of gut in organic laying hens. Poult. Sci. 2016, 95, 2528–2535. [Google Scholar] [CrossRef]
- De Cesare, A.; Sirri, F.; Manfreda, G.; Moniaci, P.; Giardini, A.; Zampiga, M.; Meluzzi, A. Effect of dietary supplementation with Lactobacillus acidophilus D2/CSL (CECT 4529) on caecum microbioma and productive performance in broiler chickens. PLoS ONE 2017, 12, 1–21. [Google Scholar] [CrossRef]
- Haghighi, H.R.; Gong, J.; Gyles, C.L.; Hayes, M.A.; Zhou, H.; Sanei, B.; Chambers, J.R.; Sharif, S. Probiotics stimulate production of natural antibodies in chickens. Clin. Vaccine Immunol. 2006, 13, 975–980. [Google Scholar] [CrossRef]
- Teo, A.Y.; Tan, H.M. Evaluation of the performance and intestinal gut microflora of broilers fed on corn-soy diets supplemented with bacillus subtilis PB6 (CloSTAT). J. Appl. Poult. Res. 2007, 16, 296–303. [Google Scholar] [CrossRef]
- Kabir, S.M.L.; Rahman, M.M.; Rahman, M.B.; Rahman, M.M.; Ahmed, S.U. The dynamics of probiotics on growth performance and immune response in broilers. Int. J. Poult. Sci. 2004, 3, 361–364. [Google Scholar]
- Lang, J.M.; Pan, C.; Cantor, R.M.; Tang, W.H.W.; Garcia-Garcia, J.C.; Kurtz, I.; Hazen, S.L.; Bergeron, N.; Krauss, R.M.; Lusis, A.J. Impact of individual traits, saturated fat, and protein source on the gut microbiome. mBio 2018, 9, 1–14. [Google Scholar] [CrossRef]
- Zeitz, J.O.; Fennhoff, J.; Kluge, H.; Stangl, G.I.; Eder, K. Effects of dietary fats rich in lauric and myristic acid on performance, intestinal morphology, gut microbes, and meat quality in broilers. Poult. Sci. 2015, 94, 2404–2413. [Google Scholar] [CrossRef]
| Ingredients | Concentration (w/w %) |
|---|---|
| Cabbage | 17 |
| Potato | 16 |
| Carrot | 17 |
| Apple pomace | 5 |
| Grapefruit pomace | 4 |
| Orange pomace | 4 |
| Banana peel | 5 |
| Ground pork | 8 |
| Eggshell | 2 |
| Horse mackerel | 10 |
| Rice | 3 |
| Bread | 3 |
| Wheat noodle | 3 |
| Chinese noodle | 3 |
| Total | 100 |
| Ingredients (%) | C | L | P |
|---|---|---|---|
| Maize grain | 68.0 | 68.0 | 68.0 |
| Soybean meal | 19.0 | 10.0 | 10.0 |
| Larvae | 0.0 | 10.0 | 0.0 |
| Pupa | 0.0 | 0.0 | 10.0 |
| Soybean oil | 1.0 | 0.0 | 0.0 |
| Tricalcium phosphate | 1.0 | 1.0 | 1.0 |
| Calcium carbonate | 9.0 | 9.0 | 9.0 |
| Salt | 1.0 | 1.0 | 1.0 |
| Vitamin and mineral mix | 1.0 | 1.0 | 1.0 |
| Chemical composition (% as feed) and energy content (Kcal/kg) | |||
| Dry matter a | 89.6 | 90.5 | 90.6 |
| Crude protein a | 14.4 | 14.6 | 16.5 |
| Crude fiber a | 2.3 | 2.8 | 3.0 |
| ADFom a | 4.3 | 5.1 | 4.8 |
| NDFom a | 17.6 | 20.8 | 20.8 |
| Ether extract a | 3.5 | 3.7 | 5.1 |
| Ash a | 12.7 | 14.7 | 14.8 |
| Ca a | 3.6 | 3.5 | 3.3 |
| P a | 0.5 | 0.5 | 0.5 |
| Mg a | 0.2 | 0.2 | 0.2 |
| Chitin b | - | 0.39 | 0.67 |
| Gross energy b (kcal/kg) | 3159.4 | 3103.0 | 3169.3 |
| Amino Acid | Larvae | Pre-Pupae |
|---|---|---|
| Arginine | 1.94 | 2.20 |
| Alanine | 2.45 | 2.28 |
| Aspartic acid | 3.43 | 3.74 |
| Cysteine | 0.28 | 0.28 |
| Glutamic acid | 3.99 | 4.30 |
| Glycine | 1.90 | 2.11 |
| Histidine | 1.32 | 1.30 |
| Isoleucine | 1.57 | 1.71 |
| Leucine | 2.59 | 2.81 |
| Lysine | 2.22 | 2.51 |
| Methionine | 0.58 | 0.74 |
| Phenylalanine | 1.51 | 1.69 |
| Proline | 2.16 | 2.14 |
| Serine | 1.62 | 1.70 |
| Threonine | 1.42 | 1.55 |
| Tryptophan | 0.53 | 0.66 |
| Tyrosine | 2.30 | 2.63 |
| Valine | 2.25 | 2.38 |
| Total | 34.06 | 36.73 |
| Fatty Acid | Larvae | Pre-Pupae |
|---|---|---|
| C10:0 | 0.7 | 0.8 |
| C12:0 | 14.1 | 16.1 |
| C14:0 | 1.9 | 1.8 |
| C16:0 | 5.3 | 4.2 |
| C16:1 | 1.1 | 1.0 |
| C18:0 | 0.9 | 0.6 |
| C18:1n-7 | 0.2 | 0.1 |
| C18:1n-9 | 7.3 | 5.6 |
| C18:2n-6 | 2.7 | 2.5 |
| C18:3n-3 | 0.2 | 0.2 |
| C20:0 | 0.0 | 0.0 |
| C20:1 | 0.0 | 0.0 |
| C20:5n-3 | 0.3 | 0.3 |
| C22:0 | 0.0 | 0.0 |
| Saturated fatty acids (SFA) | 22.9 | 23.5 |
| Monounsaturated fatty acids (MUFA) | 8.6 | 6.7 |
| Polyunsaturated fatty acids (PUFA) | 3.2 | 3.0 |
| Item | C | L | P |
|---|---|---|---|
| Feed intake (g/day) | 79.8 ± 0.6 | 77.5 ± 1.3 | 76.2 ± 4.9 |
| Egg-laying rate (%) | 70.3 ± 5.5 | 70.6 ± 6.3 | 70.7 ± 3.1 |
| Egg weight c (g) | 48.0 ± 0.3 a | 49.0 ± 0.7 a | 51.1 ± 0.3 b |
| Feed requirement rate | 2.5 ± 0.1 | 2.3 ± 0.2 | 2.2 ± 0.1 |
| Body weight c (g/bird) | 1230.7 ± 166.6 | 1234.6 ± 124.4 | 1328.7 ± 98.1 |
| Liver weight c (% of BW) | 1.8 ± 0.1 | 1.8 ± 0.1 | 1.8 ± 0.1 |
| Item | C | L | P |
|---|---|---|---|
| Villus height | |||
| Duodenum | 1028.9 ± 51.8 | 993.2 ± 63.0 | 893.8 ± 48.4 |
| Jejunum | 604.6 ± 30.7 | 590.1 ± 23.4 | 561.2 ± 31.6 |
| Ileum | 318.4 ± 17.5 | 351.1 ± 26.0 | 321.7 ± 17.2 |
| Crypt depth | |||
| Duodenum | 103.9 ± 3.2 | 107.0 ± 8.8 | 98.1 ± 3.1 |
| Jejunum | 63.3 ± 4.1 | 62.1 ± 2.6 | 63.5 ± 2.4 |
| Ileum | 59.9 ± 3.0 | 66.1 ± 2.3 | 58.4 ± 4.4 |
| Villus height/Crypt depth | |||
| Duodenum | 10.1 ± 0.2 | 10.2 ± 0.8 | 9.5 ± 0.5 |
| Jejunum | 9.9 ± 0.5 | 9.9 ± 0.4 | 9.2 ± 0.5 |
| Ileum | 5.5 ± 0.2 | 5.5 ± 0.3 | 6.1 ± 0.4 |
| Item | C | L | P |
|---|---|---|---|
| Egg weight (g) | 45.66 ± 1.15 a | 45.61 ± 0.88 a | 49.91 ± 0.60 b |
| Egg yolk weight (g) | 12.02 ± 0.31 | 12.03 ± 0.25 | 12.56 ± 0.24 |
| Albumin weight (g) | 26.79 ± 0.81 a | 27.10 ± 0.63 a | 30.04 ± 0.46 b |
| Egg shell weight (g) | 6.86 ± 0.30 | 6.49 ± 0.29 | 7.32 ± 0.23 |
| Egg shell thickness (mm) | 0.38 ± 0.02 ab | 0.37 ± 0.02 a | 0.43 ± 0.01 b |
| Egg shell strength (kgf/cm2) | 3.69 ± 0.33 | 3.64 ± 0.30 | 4.44 ± 0.17 |
| Egg yolk color score | 5.21 ± 0.30 a | 6.28 ± 0.22 b | 5.92 ± 0.36 ab |
| Albumin height (mm) | 7.16 ± 0.32 a | 7.64 ± 0.14 ab | 8.08 ± 0.18 b |
| Haugh unit | 88.59 ± 1.94 | 91.70 ± 0.69 | 92.66 ± 0.93 |
| Mineral | C | L | P |
|---|---|---|---|
| Calcium | 23.1 ± 1.7 ab | 17.6 ± 1.6 a | 24.6 ± 2.3 b |
| Inorganic phosphorus | 2.1 ± 0.1 | 1.7 ± 0.1 | 2.3 ± 0.1 |
| Magnesium | 2.7 ± 0.1 | 1.7 ± 0.1 | 2.7 ± 0.4 |
| SCFA (µg/L) | C | L | P |
|---|---|---|---|
| Acetic acid | 87.4 ± 17.3 | 110.2 ± 13.0 | 95.4 ± 11.0 |
| Propionic acid | 41.7 ± 7.7 | 64.9 ± 9.7 | 53.1 ± 5.9 |
| n-butyric acid | 204.9 ± 83.5 | 197.5 ± 45.7 | 276.6 ± 138.7 |
| Total | 334.0 ± 102.5 | 372.5 ± 61.2 | 425.1 ± 142.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kawasaki, K.; Hashimoto, Y.; Hori, A.; Kawasaki, T.; Hirayasu, H.; Iwase, S.-i.; Hashizume, A.; Ido, A.; Miura, C.; Miura, T.; et al. Evaluation of Black Soldier Fly (Hermetia illucens) Larvae and Pre-Pupae Raised on Household Organic Waste, as Potential Ingredients for Poultry Feed. Animals 2019, 9, 98. https://doi.org/10.3390/ani9030098
Kawasaki K, Hashimoto Y, Hori A, Kawasaki T, Hirayasu H, Iwase S-i, Hashizume A, Ido A, Miura C, Miura T, et al. Evaluation of Black Soldier Fly (Hermetia illucens) Larvae and Pre-Pupae Raised on Household Organic Waste, as Potential Ingredients for Poultry Feed. Animals. 2019; 9(3):98. https://doi.org/10.3390/ani9030098
Chicago/Turabian StyleKawasaki, Kiyonori, Yuka Hashimoto, Akihiro Hori, Toshiya Kawasaki, Hirofumi Hirayasu, Shun-ichiro Iwase, Atsushi Hashizume, Atsushi Ido, Chiemi Miura, Takeshi Miura, and et al. 2019. "Evaluation of Black Soldier Fly (Hermetia illucens) Larvae and Pre-Pupae Raised on Household Organic Waste, as Potential Ingredients for Poultry Feed" Animals 9, no. 3: 98. https://doi.org/10.3390/ani9030098
APA StyleKawasaki, K., Hashimoto, Y., Hori, A., Kawasaki, T., Hirayasu, H., Iwase, S.-i., Hashizume, A., Ido, A., Miura, C., Miura, T., Nakamura, S., Seyama, T., Matsumoto, Y., Kasai, K., & Fujitani, Y. (2019). Evaluation of Black Soldier Fly (Hermetia illucens) Larvae and Pre-Pupae Raised on Household Organic Waste, as Potential Ingredients for Poultry Feed. Animals, 9(3), 98. https://doi.org/10.3390/ani9030098






