Data Collection for the Fourth Multicentre Confidential Enquiry into Perioperative Equine Fatalities (CEPEF4) Study: New Technology and Preliminary Results
Abstract
:Simple Summary
Abstract
1. Introduction
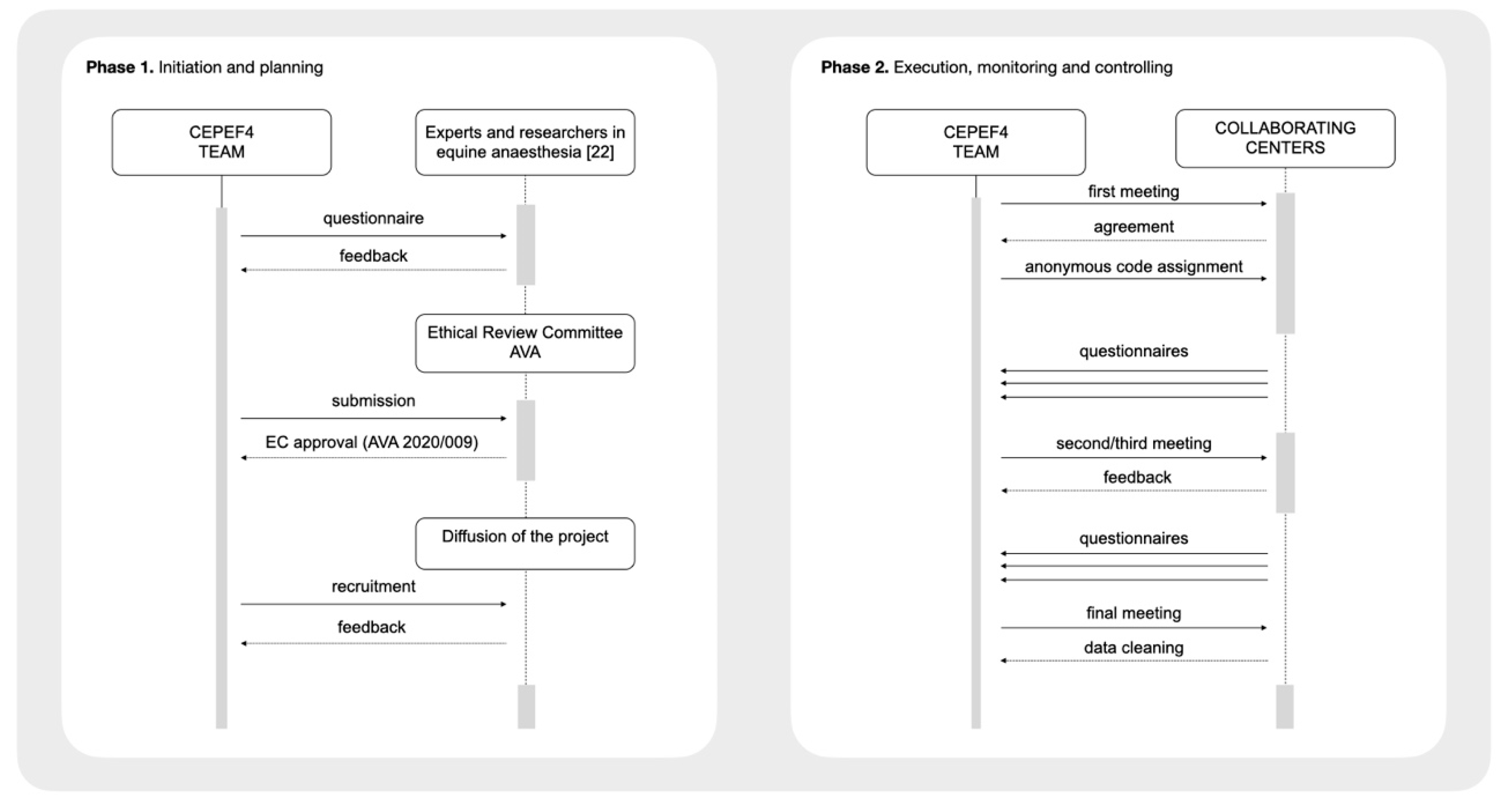
2. Materials and Methods
- (1)
- Preparation of the questionnaire
- (2)
- Definition of perioperative equine fatalities
- (3)
- Ethics statement
- (4)
- Recruitment of collaborating centres
- (5)
- The Ambassador figure and the Agreement Form
- (6)
- Anonymity and confidentiality of each patient, owner and centre
- (7)
- The communication process (recruitment and follow-up meetings)
- (8)
- Data collection and storage
- (9)
- Data cleaning and statistics
- (10)
- Final meeting with each ambassador to double-check data and outcomes
- (11)
- Reporting the results
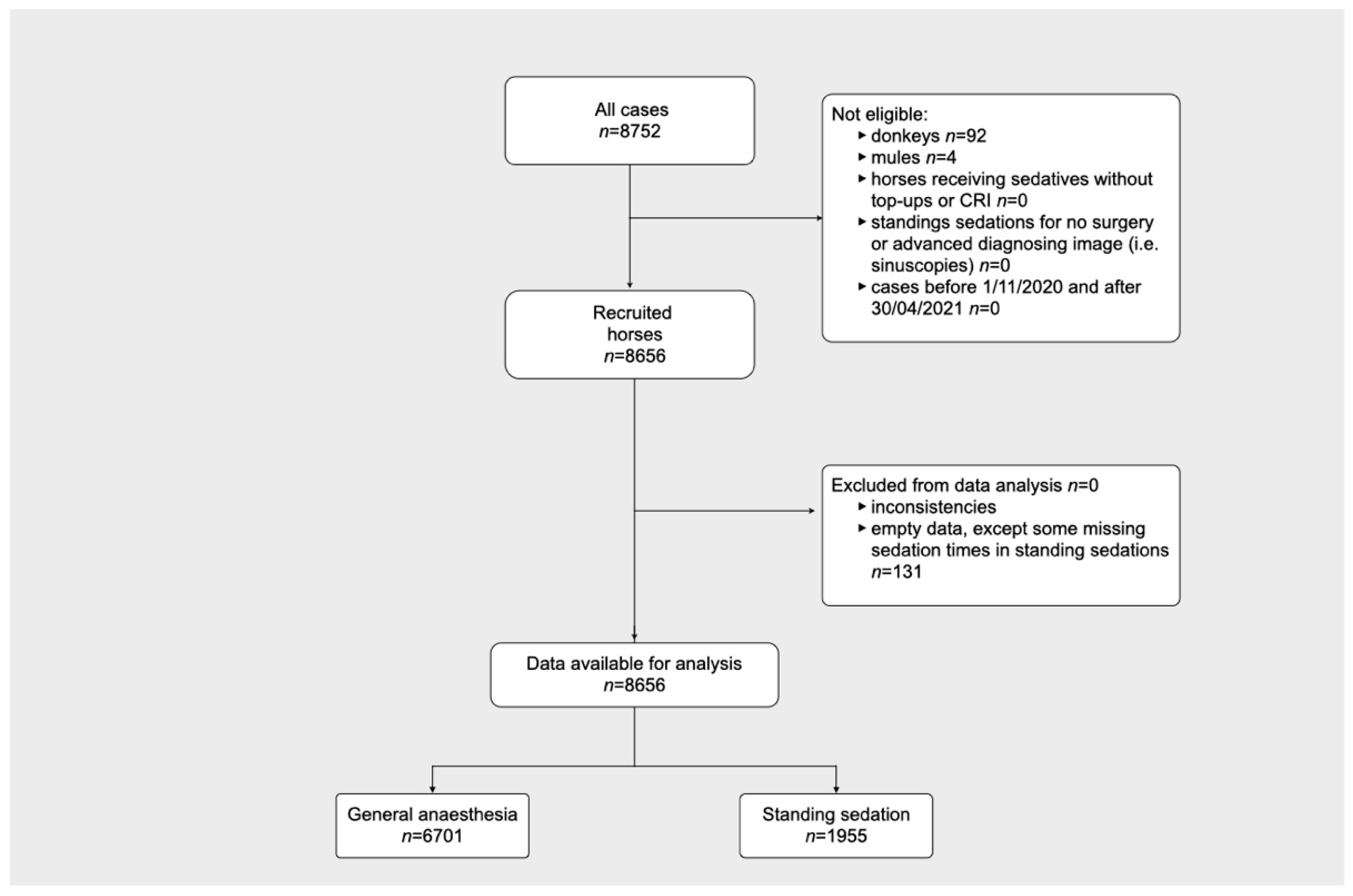
3. Results
3.1. Perioperative Equine Fatalitiess
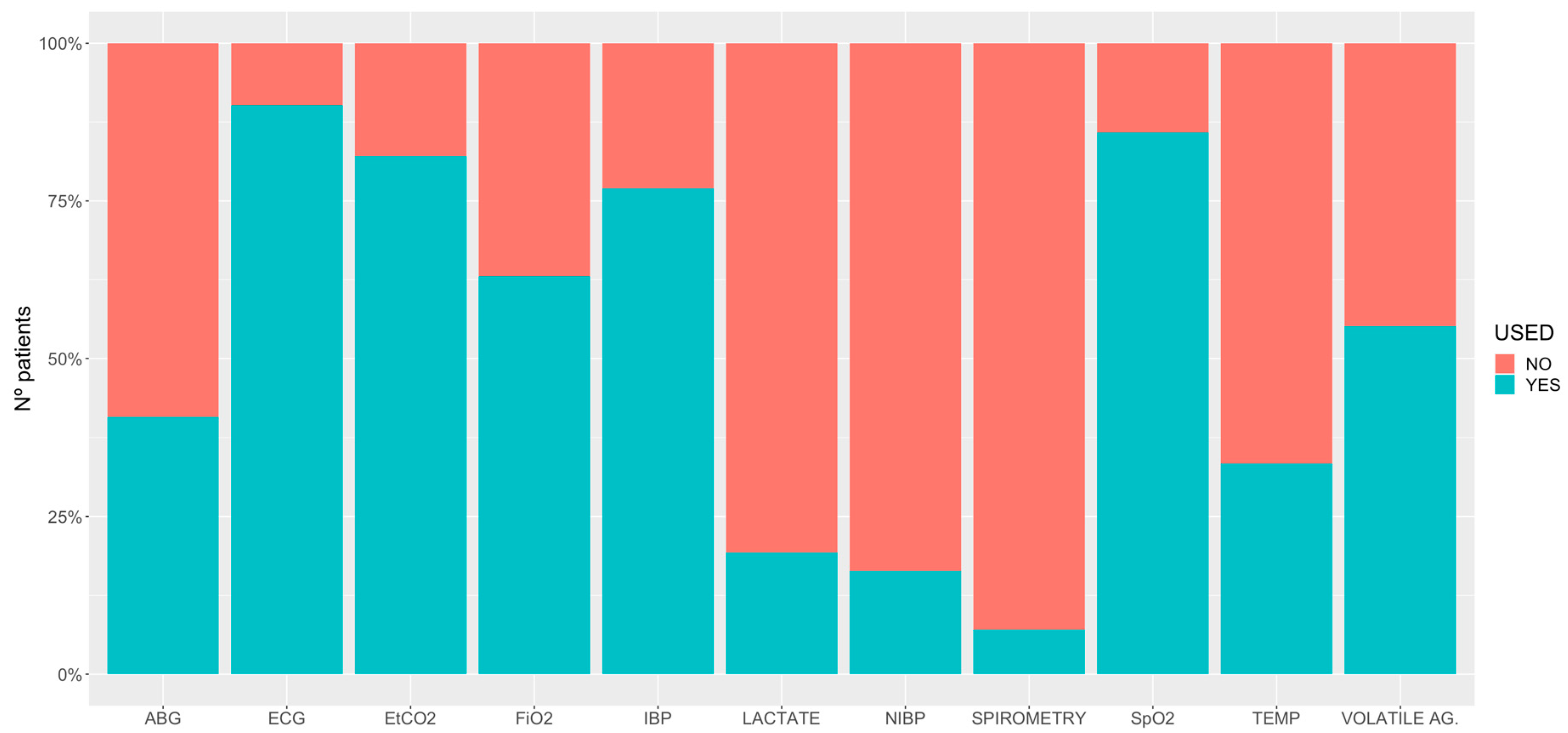
3.2. General Anaesthesia
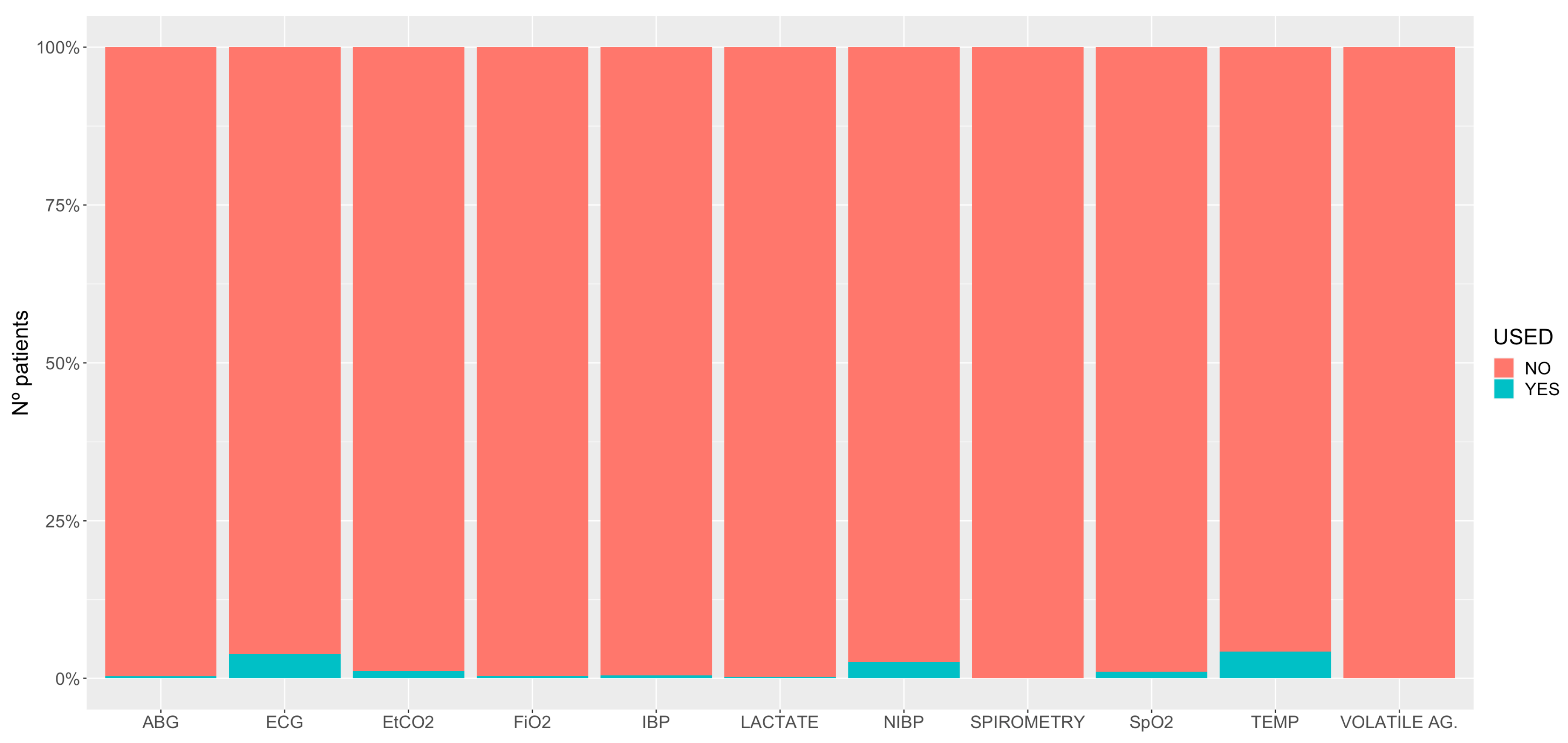
3.3. Standing Sedation
3.4. Results of Survey for Feedback in Final Meeting
4. Discussion
- (1)
- The proposed method for data collection with interactive data handling and cleaning
- (2)
- Outcome of horses under general anaesthesia and standing sedation
- (3)
- Preliminary results for general anaesthetics in horses
- (4)
- Preliminary results of standing sedations in horses
- (5)
- Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
- −
- Argentina: Centro de Cirugía Equina Santa Fe, Santa Fe (Natalia Rossetti); Hospital Escuela de Grandes Animales FCV UNNE, Corrientes (Eduardo Darío Saravia)
- −
- Australia: Goulburn Valley Equine Hospital, Congupna, Victoria (Valentina Di Sevo); Scone Equine Hospital, Scone, New South Wales (Maria Helena Bellei)
- −
- Austria: Veterinärmedizinische Universität Wien, Wien (Natalí Verdier)
- −
- Belgium: Dierenkliniek de Bosdreef, Moerbeke-Waas, Oost-Vlaanderen (Katherine Bafort and Hans Wilderjans); Equitom Equine Clinic, Meldert, Limbürg (Jamien Deman and Tom Marien); Université de Liège, Liège, Wallonie (Julien Dupont and Charlotte Sandersen); Universiteit Gent, Merelbeke, Oost-Vlaanderen (Stijn Schauvliege and Frank Gasthuys)
- −
- Brasil: Universidade Federal de Minas Gerais, Belo Horizonte, MG (Suzane Lilian Beier); Universidade Federal do Paraná, Curitiba, PR (Juan Carlos Duque Moreno); Universidade de São Paulo, Pirassununga, SP (Adriano Carregaro); Universidade de São Paulo, São Paulo, SP (Denise Tabacchi Fantoni)
- −
- Canada: Université de Montréal, Saint Hyacinthe, Québec (Mathilde Ducroq, Javier Benito de la Víbora and Paulo Steagall); Western College of Veterinary Medicine, University of Saskatchewam, Saskatchewam (Barbara Ambros)
- −
- Chile: HCV Universidad Nacional Andrés Bello, Santiago de Chile, Región Metropolitana (Karina Aros Villarroel)
- −
- Colombia: Gustavo Villa Díaz (ambulatory clinician), Medellín, Antioquia; Universidad CES, Medellín, Antioquia (Gustavo Villa Díaz); Universidad Lasallista, Caldas, Antioquia (Gustavo Villa Díaz); Universidad Tecnológica de Pereira, Pereira, Risaralda (David Latorre Galeano)
- −
- France: Centre Hospitalier Vétérinaire Équin de Livet, Calvados (Valerie Picanvet); Clinique de Grosbois, Boissy Saint Leger, Val de Marne (Valerie Deniau); Clinique Équine Acy Romance, Acy Romance, Grand Est (Justine Dalla Valle and Anneleen Michielsen); Clinique Équine de la Boisrie, Chaylloue, Orne (Peggy Moreau); Clinique Équine de Meslay, Pays de Loire (Bienvenida Román Durá and Tamara de Beauregard); Clinique Vétérinaire Équine de Meheudin, Écouche, Orne (Elsa Autard and Florence Polle)
- −
- Germany: TIHO Hannover, Hannover, Niedersachsen (Lara Twele, Álvaro Gutiérrez Bautista and Sabine Kästner)
- −
- Italy: Università di Bologna, Bologna (Carlotta Lambertini and Noemi Romagnoli); Università di Perugia, Perugia (Sara Nannarone)
- −
- Japan: Shadai Horse Clinic, Hokkaido (Tsukasa Suzuki)
- −
- Mexico: Central Quirúrgica para Caballos, Naucalpan, Estado de México (Jorge Eduardo Mendoza Flores); EQCOVET, Zapopan, Jalisco (Edgardo Hernández Díaz); Jorge Eduardo Mendoza Flores (ambulatory clinician), Ciudad de México, Estado de México
- −
- Republic of Ireland: Fethard Equine Hospital, Clonmel, Tipperary (Erica Sogus); Somerton Equine Hospital, Friarstown, Kildare (Monica McNichol and Valerie Hyde); University College Dublin, Dublin, Dublin (Vilhelmiina Huuskonen)
- −
- Spain: Hospital de Referencia La Equina, Manilva, Málaga (Joana Maia Ramos, Maya Caparrós Buscarons and Miguel Valdés Vázquez); Hospital Veteriario Costa de La Luz, Conil de la Frontera, Cádiz (Ana del Ojo Morales); Universidad Alfonso X El Sabio, Villanueva de la Cañada, Madrid (Rocío Cañellas Blanco and Verónica Salazar Nussio); Universidad Autónoma de Barcelona, Barcelona (Lucas Troya Portillo and Cristina Costa Farré); Universidad Cardenal Herrera CEU, Valencia (Eva Zoe Hernández Magaña, Jaime Viscasillas Monteagudo and José Ignacio Redondo García); Universidad de Córdoba, Córdoba (Esther Caravaca Paredes and María del Mar Granados Machuca); Universidad Complutense de Madrid, Madrid (Marta Villalba Díez, Isabel Santiago Llorente and Ignacio Álvarez Gómez de Segura); Universidad de Murcia, Murcia (Eliseo Belda Mellado and Francisco Ginés Laredo Álvarez)
- −
- Switzerland: Bern Universität, Bern (Ilaria Petruccione and Olivier Levionnois); Pferdeklinik Dalchenhof, Brittnau, Aargau (Orquidea Kiesse); Pferdeklinik Niederlenz, Niederlenz, Lenzburg (Theo Tschanz); Vetsuisse Faculty, University of Zürich, Zürich (Simone Katja Ringer and Regula Bettschart-Wolfensberger)
- −
- The Netherlands: Dierenhospital Visdonk, Roosendaal (Els van den Berg); Universiteit Utrecht, Utrecht (Janny de Grauw and Thijs van Loon)
- −
- United Kingdom: Ashbrook Equine Hospital, Allostock, Cheshire (Julia Shrubb); Bell Equine Veterinary Clinic, Mereworth, Kent (Heloïse Lesca and Karen Coumbe); Bourton Vale Equine Clinic, Cheltenham, Gloucestershire (Lydia Coles); Hird & Partners, Shelf, West Yorkshire (John Hird); Liphook Equine Hospital, Liphook, Hampshire (Rachel Tucker); Pool House Equine Clinic, Fradley, Staffordshire (Federica Cantatore, Cristina Bertone and Sophie Malorey); Rainbow Equine Hospital, Malton, North Yorkshire (Kate Loomes); Rossdales Ltd., Newmarket, Suffolk (Luis Louro and Catriona McKenzie); Royal Veterinary College, Hatfield, Hertfordshire (Kata Veres-Nyeki); The Donkey Sanctuary, Sidmouth, Devon (Abigail Sefton, Dom Doyle and Rebekah Sullivan); The Universisty of Edinburgh, Easter Bush Campus, Midlothian (Chiara Pressanto, Tom Beeston and Miguel Gozalo-Marcilla); University of Glasgow, Bearsden, Glasgow (Josephine Kropf and Andrew Bell); Wright and Morten, Somerford, Cheshire (Nicola Harries and Rachel Helm)
- −
- United States of America: Colorado State University, Fort Collins, CO (Rachel Hector and Marlis Rezende); University of Missouri, Columbia, MO (Kelley Varner); University of Pennsilvania—New Bolton Centre, Kennett Square, PA (Klaus Hopster)
- −
- Uruguay: Facultad de Veterinaria—UDELAR, Montevideo (Sabrina Castro Peri, Gastón Pérez and Camila Nan); María Noel Acevedo (ambulatory clinician), Montevideo; Servicio de Veterinaria y Remonta, Canelones (Sabrina Castro Peri and María Noel Acevedo)
- −
- Belgium (Stijn Schauvliege)
- −
- France (Latifa Khenissi and Stijn Schauvliege)
- −
- Japan (Paulo Steagall)
- −
- México (José Antonio Ibancovichi)
- −
- South America (Adriano Carregaro and Natalia Rossetti)
- −
- The Netherlands (Janny de Grauw)
References
- Ahern, N.R. Using the Internet to conduct research. Nurse Res. 2005, 13, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Cantrell, M.A.; Lupinacci, P. Methodological issues in online data collection. J. Adv. Nurs. 2007, 60, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Pace, W.D.; Staton, E.W. Electronic data collection options for practice-based research networks. Ann. Fam. Med. 2005, 3 (Suppl. S1), S21–S29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pittard, W.S.; Li, S. The essential toolbox of data science: Python, R, Git, and Docker. Methods Mol. Biol. 2020, 2104, 265–311. [Google Scholar] [CrossRef] [PubMed]
- Van den Broeck, J.; Cunningham, S.A.; Eeckels, R.; Herbst, K. Data cleaning: Detecting, diagnosing, and editing data abnormalities. PLoS Med. 2005, 2, e267. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, B. Equine anaesthesia: An assessment of techniques used in clinical practice. Equine Vet. J. 1969, 1, 261–275. [Google Scholar] [CrossRef]
- Tevik, A. The role of anesthesia in surgical mortality in horses. Nord. Vet. Med. 1983, 35, 175–179. [Google Scholar]
- Young, S.S.; Taylor, P.M. Factors influencing the outcome of equine anaesthesia: A review of 1,314 cases. Equine Vet. J. 1993, 25, 147–151. [Google Scholar] [CrossRef]
- Mee, A.M.; Cripps, P.J.; Jones, R.S. A retrospective study of mortality associated with general anaesthesia in horses: Elective procedures. Vet. Rec. 1998, 142, 275–276. [Google Scholar] [CrossRef]
- Mee, A.M.; Cripps, P.J.; Jones, R.S. A retrospective study of mortality associated with general anaesthesia in horses: Emergency procedures. Vet. Rec. 1998, 142, 307–309. [Google Scholar] [CrossRef]
- Bidwell, L.A.; Bramlage, L.R.; Rood, W.A. Equine perioperative fatalities associated with general anaesthesia at a private practice--a retrospective case series. Vet. Anaesth. Analg. 2007, 34, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Jago, R.C.; Corletto, F.; Wright, I.M. Peri-anaesthetic complications in an equine referral hospital: Risk factors for post anaesthetic colic. Equine Vet. J. 2015, 47, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Dugdale, A.H.; Obhrai, J.; Cripps, P.J. Twenty years later: A single-centre, repeat retrospective analysis of equine perioperative mortality and investigation of recovery quality. Vet. Anaesth. Analg. 2016, 43, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Laurenza, C.; Ansart, L.; Portier, K. Risk factors of anesthesia-related mortality and morbidity in one equine hospital: A retrospective study on 1,161 cases undergoing elective or emergency surgeries. Front. Vet. Sci. 2020, 6, 514. [Google Scholar] [CrossRef] [PubMed]
- Proudman, C.J.; Dugdale, A.H.; Senior, J.M.; Edwards, G.B.; Smith, J.E.; Leuwer, M.L.; French, N.P. Pre-operative and anaesthesia-related risk factors for mortality in equine colic cases. Vet. J. 2006, 171, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Johnston, G.M.; Taylor, P.M.; Holmes, M.A.; Wood, J.L. Confidential enquiry of perioperative equine fatalities (CEPEF-1): Preliminary results. Equine Vet. J. 1995, 27, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Senior, J.M.; Pinchbeck, G.L.; Allister, R.; Dugdale, A.H.; Clark, L.; Clutton, R.E.; Coumbe, K.; Dyson, S.; Clegg, P.D. Reported morbidities following 861 anaesthetics given at four equine hospitals. Vet. Rec. 2007, 160, 407–408. [Google Scholar] [CrossRef] [Green Version]
- Johnston, G.M.; Eastment, J.K.; Wood, J.; Taylor, P.M. The confidential enquiry into perioperative equine fatalities (CEPEF): Mortality results of Phases 1 and 2. Vet. Anaesth. Analg. 2002, 29, 159–170. [Google Scholar] [CrossRef]
- Dugdale, A.H.; Taylor, P.M. Equine anaesthesia-associated mortality: Where are we now? Vet. Anaesth. Analg. 2016, 43, 242–255. [Google Scholar] [CrossRef] [Green Version]
- Gent, T.C.; Bettschart-Wolfensberger, R. Peri-anaesthetic mortality in horses—the need for CEPEF-4. Vet. Anaesth. Analg. 2013, 40, e1–e2. [Google Scholar] [CrossRef]
- Redondo, J.I. Anaesthetic-related deaths in dogs in Spain: COMPLRED 2016–2019. In Proceedings of the Autumn Meeting of the Association of Veterinary Anaesthetists 2019, Ghent, Belgium, 11–13 September 2019; p. 166. Available online: https://ava.eu.com/wp-content/uploads/2019/09/Proceedings-Ghent-2019-main-congress.pdf (accessed on 6 June 2021).
- Gozalo-Marcilla, M.; Redondo, J.I.; Johnston, M.; Taylor, P.; Bettschart-Wolfensberger, R. CEPEF4: Update and plan. Vet. Anaesth. Analg. 2020, 47, 724–725. [Google Scholar] [CrossRef]
- Gozalo-Marcilla, M.; Redondo, J.I.; Johnston, M.; Taylor, P.M.; Bettschart-Wolfensberger, R. CEPEF4 going live. In Proceedings of the Spring Meeting of the Association of Veterinary Anaesthetists 2020, Dublin, Ireland, 11–13 March 2020; p. 90. Available online: http://avadublin2020.ie/wp-content/uploads/2020/03/FINAL-AVA-Proceedings.pdf (accessed on 6 June 2021).
- Gozalo-Marcilla, M.; Redondo, J.I.; Johnston, M.; Taylor, P.; Bettschart-Wolfensberger, R. A new equine anaesthetic mortality study two decades after CEPEF2: CEPEF4 is going live! Equine Vet. J. 2020, 52, 891–892. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 6 June 2021).
- Rich, B. Table 1: Tables of Descriptive Statistics in HTML. 2021, R Package Version 1.4.1. Available online: https://CRAN.R-project.org/package=table1 (accessed on 6 June 2021).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- South, A. Rnaturalearth: World Map Data from Natural Earth. R Package Version 0.1.0. 2017. Available online: https://CRAN.R-project.org/package=rnaturalearth (accessed on 6 June 2021).
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Int. J. Surg. 2014, 12, 1495–1499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brodbelt, D.C.; Blissitt, K.J.; Hammond, R.A.; Neath, P.J.; Young, L.E.; Pfeiffer, D.U.; Wood, J.L. The risk of death: The confidential enquiry into perioperative small animal fatalities. Vet. Anaesth. Analg. 2008, 35, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.W.; Paton, B.S. Combined use of detomidine with opiates in the horse. Equine Vet. J. 1988, 20, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Lopes, C.; Luna, S.P.; Rosa, A.C.; Quarterone, C.; Crosignani, N.; Taylor, P.M.; Pantoja, J.C.; Puoli, J.N. Antinociceptive effects of methadone combined with detomidine or acepromazine in horses. Equine Vet. J. 2016, 48, 613–618. [Google Scholar] [CrossRef]
- Gozalo-Marcilla, M.; Luna, S.P.; Crosignani, N.; Filho, J.N.P.; Possebon, F.S.; Pelligand, L.; Taylor, P.M. Sedative and antinociceptive effects of different combinations of detomidine and methadone in standing horses. Vet. Anaesth. Analg. 2017, 44, 1116–1127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, L.W. Equine anaesthesia: Discovery and rediscovery. Equine Vet. J. 1983, 15, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.M. The stress response to anaesthesia in ponies: Barbiturate anaesthesia. Equine Vet. J. 1990, 22, 307–312. [Google Scholar] [CrossRef]
- Posner, L.P.; Kasten, J.I.; Kata, C. Propofol with ketamine following sedation with xylazine for routine induction of general anaesthesia in horses. Vet. Rec. 2013, 173, 550. [Google Scholar] [CrossRef]
- Johnston, G.M.; Eastment, J.K.; Taylor, P.M.; Wood, J.L. Is isoflurane safer than halothane in equine anaesthesia? Results from a prospective multicentre randomised controlled trial. Equine Vet. J. 2004, 36, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Gozalo-Marcilla, M.; Gasthuys, F.; Schauvliege, S. Partial intravenous anaesthesia in the horse: A review of intravenous agents used to supplement equine inhalation anaesthesia. Part 1: Lidocaine and ketamine. Vet. Anaesth. Analg. 2014, 41, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Gozalo-Marcilla, M.; Gasthuys, F.; Schauvliege, S. Partial intravenous anaesthesia in the horse: A review of intravenous agents used to supplement equine inhalation anaesthesia. Part 2: Opioids and alpha-2 adrenoceptor agonists. Vet. Anaesth. Analg. 2015, 42, 1–16. [Google Scholar] [CrossRef] [PubMed]
- White, K. Total and partial intravenous anaesthesia of horses. Practice 2015, 37, 189–197. [Google Scholar] [CrossRef] [Green Version]
- Devisscher, L.; Schauvliege, S.; Dewulf, J.; Gasthuys, F. Romifidine as a constant rate infusion in isoflurane anaesthetized horses: A clinical study. Vet. Anaesth. Analg. 2010, 37, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Sacks, M.; Ringer, S.K.; Bischofberger, A.S.; Berchtold, S.M.; Bettschart-Wolfensberger, R. Clinical comparison of dexmedetomidine and medetomidine for isoflurane balanced anaesthesia in horses. Vet. Anaesth. Analg. 2017, 44, 1128–1138. [Google Scholar] [CrossRef]
- Valverde, A.; Gunkelt, C.; Doherty, T.J.; Giguère, S.; Pollak, A.S. Effect of a constant rate infusion of lidocaine on the quality of recovery from sevoflurane or isoflurane general anaesthesia in horses. Equine Vet. J. 2005, 37, 559–564. [Google Scholar] [CrossRef]
- Larenza, M.P.; Peterbauer, C.; Landoni, M.F.; Levionnois, O.L.; Schatzmann, U.; Spadavecchia, C.; Thormann, W. Stereoselective pharmacokinetics of ketamine and norketamine after constant rate infusion of a subanesthetic dose of racemic ketamine or S-ketamine in Shetland ponies. Am. J. Vet. Res. 2009, 70, 831–839. [Google Scholar] [CrossRef]
- Chesnel, M.A.; Clutton, R.E. A comparison of two morphine doses on the quality of recovery from general anaesthesia in horses. Res. Vet. Sci. 2013, 95, 1195–1200. [Google Scholar] [CrossRef]
- Gozalo-Marcilla, M.; Steblaj, B.; Schauvliege, S.; Duchateau, L.; Gasthuys, F. Comparison of the influence of two different constant-rate infusions (dexmedetomidine versus morphine) on anaesthetic requirements, cardiopulmonary function and recovery quality in isoflurane anaesthetized horses. Res. Vet. Sci. 2013, 95, 1186–1194. [Google Scholar] [CrossRef]
- Santos, M.; Fuente, M.; Garcia-Iturralde, R.; Herran, R.; Lopez-Sanroman, J.; Tendillo, F.J. Effects of alpha-2 adrenoceptor agonists during recovery from isoflurane anaesthesia in horses. Equine Vet. J. 2003, 35, 170–175. [Google Scholar] [CrossRef]
- Woodhouse, K.J.; Brosnan, R.J.; Nguyen, K.Q.; Moniz, G.W.; Galuppo, L.D. Effects of postanesthetic sedation with romifidine or xylazine on quality of recovery from isoflurane anesthesia in horses. J. Am. Vet. Med. Assoc. 2013, 242, 533–539. [Google Scholar] [CrossRef]
- Guedes, A.G.P.; Tearney, C.C.; Cenani, A.; Aristizabal, F.; Nieto, J. Comparison between the effects of postanesthetic xylazine and dexmedetomidine on characteristics of recovery from sevoflurane anesthesia in horses. Vet. Anaesth. Analg. 2017, 44, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Wohlfender, F.D.; Doherr, M.G.; Driessen, B.; Hartnack, S.; Johnston, G.M.; Bettschart-Wolfensberger, R. International online survey to assess current practice in equine anaesthesia. Equine Vet. J. 2015, 47, 65–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arndt, S.; Hopster, K.; Sill, V.; Rohn, K.; Kastner, S.B.R. Comparison between head-tail-rope assisted and unassisted recoveries in healthy horses undergoing general anesthesia for elective surgeries. Vet. Surg. 2020, 49, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Ruegg, M.; Bettschart-Wolfensberger, R.; Hartnack, S.; Junge, H.K.; Theiss, F.; Ringer, S.K. Comparison of non-assisted versus head and tail rope-assisted recovery after emergency abdominal surgery in horses. Pferdeheilkunde 2016, 32, 469–478. [Google Scholar] [CrossRef] [Green Version]
- del Barrio, M.C.N.; David, F.; Hughes, J.M.L.; Clifford, D.; Wilderjans, H.; Bennett, R. A retrospective report (2003–2013) of the complications associated with the use of a one-man (head and tail) rope recovery system in horses following general anaesthesia. Ir. Vet. J. 2018, 71, 6. [Google Scholar] [CrossRef] [Green Version]

| Country | University Hospitals | Referral Centres | Ambulatory Clinicians | Total Centres | Cases 1 | % Cases |
|---|---|---|---|---|---|---|
| Belgium | 2 | 2 | 0 | 4 | 1906 | 21.78 |
| United Kingdom | 3 | 10 | 0 | 13 | 1670 | 19.08 |
| Republic of Ireland | 1 | 2 | 0 | 3 | 1068 | 12.20 |
| France | 0 | 6 | 0 | 6 | 1025 | 11.71 |
| Australia | 0 | 2 | 0 | 2 | 898 | 10.26 |
| Switzerland | 2 | 2 | 0 | 4 | 444 | 5.07 |
| Spain | 6 | 2 | 0 | 8 | 399 | 4.56 |
| United States of America | 3 | 0 | 0 | 3 | 327 | 3.74 |
| Germany | 1 | 0 | 0 | 1 | 162 | 1.85 |
| The Netherlands | 1 | 1 | 0 | 2 | 150 | 1.71 |
| Canada | 2 | 0 | 0 | 2 | 142 | 1.62 |
| Italy | 2 | 0 | 0 | 2 | 120 | 1.37 |
| Austria | 1 | 0 | 0 | 1 | 97 | 1.11 |
| Japan | 0 | 1 | 0 | 1 | 82 | 0.94 |
| Argentina | 1 | 1 | 0 | 2 | 55 | 0.63 |
| Chile | 1 | 0 | 0 | 1 | 48 | 0.55 |
| Uruguay | 1 | 1 | 1 | 3 | 46 | 0.53 |
| Brazil | 4 | 0 | 0 | 4 | 44 | 0.50 |
| Colombia | 3 | 0 | 1 | 4 | 35 | 0.40 |
| Mexico | 0 | 2 | 1 | 3 | 34 | 0.39 |
| Total | 34 | 32 | 3 | 69 | 8752 | 100 |
| Variable | Categories | General Anaesthesia (n = 6701) | Standing Sedations (n = 1955) | Overall (n = 8656) |
|---|---|---|---|---|
| Sex | Female | 2699 | 817 | 3516 |
| Male | 4002 | 1138 | 5140 | |
| Age | Neonate (≤1 month) | 229 | 30 | 259 |
| Foal (1–12 months) | 1407 | 27 | 1434 | |
| Young (1–5 years) | 2314 | 328 | 2642 | |
| Adult (5–14 years) | 2029 | 1131 | 3160 | |
| Geriatric (>14 years) | 722 | 439 | 1161 | |
| BCS | Normal | 5766 | 1585 | 7351 |
| Thin | 415 | 129 | 544 | |
| Fat | 520 | 241 | 761 | |
| ASA | I | 3007 | 1200 | 4207 |
| II | 2354 | 666 | 3020 | |
| III | 546 | 79 | 625 | |
| IV | 470 | 9 | 479 | |
| V | 324 | 1 | 325 | |
| Reason 1 | Abdominal | 218 | 61 | 279 |
| Colic | 917 | 6 | 923 | |
| Diagnostic | 479 | 959 | 1438 | |
| ENT | 377 | 138 | 515 | |
| Fracture | 166 | 28 | 192 | |
| Orthopaedics | 3346 | 138 | 3484 | |
| Urogenital | 820 | 103 | 923 | |
| Miscellaneous | 898 | 573 | 1471 | |
| Colic/noncolic | Noncolic surgery | 5784 | 1949 | 7733 |
| Colic surgery | 917 | 6 | 923 | |
| Type of anaesthesia | Standing sedation | 0 | 1955 | 1955 |
| Inhalatory | 2282 | 0 | 2282 | |
| PIVA | 3718 | 0 | 3718 | |
| TIVA | 701 | 0 | 701 | |
| Duration * | <1 h | 2317 | 594 | 2911 |
| 1–2 h | 2815 | 744 | 3559 | |
| 2–3 h | 1123 | 333 | 1456 | |
| >3 h | 446 | 153 | 599 | |
| Use of locoregional techniques | No | 5602 | 1114 | 6716 |
| Yes | 1099 | 841 | 1940 | |
| Use of mechanical ventilation | No | 1870 | 1955 | 3825 |
| Yes | 4831 | 0 | 4831 | |
| Timetable | Normal | 5866 | 1918 | 7784 |
| Out of hours | 835 | 37 | 872 | |
| Scheduling | Scheduled | 5120 | 1817 | 6937 |
| Non scheduled | 380 | 80 | 460 | |
| Urgent | 1201 | 58 | 1259 |
| Alive | Dead | % Deaths | PTS | % PTS | Total | |
|---|---|---|---|---|---|---|
| General anaesthesia | ||||||
| Noncolic surgery | 5673 | 35 | 0.6% | 76 | 1.3% | 5784 |
| Abdominal | 198 | 3 | 1.4% | 17 | 7.8% | 218 |
| Diagnostic | 440 | 3 | 0.6% | 36 | 7.5% | 479 |
| ENT | 373 | 4 | 1.1% | 0 | 0.0% | 377 |
| Fracture | 154 | 7 | 4.2% | 4 | 2.4% | 166 |
| Miscellaneous | 882 | 5 | 0.6% | 11 | 1.2% | 898 |
| Orthopaedics | 3306 | 13 | 0.4% | 27 | 0.8% | 3346 |
| Urogenital | 803 | 7 | 0.9% | 10 | 1.2% | 820 |
| Colic surgery | 633 | 31 | 3.4% | 253 | 27.6% | 917 |
| Overall | 6306 | 66 | 1.0% | 329 | 4.9% | 6701 |
| Standing sedation | ||||||
| Noncolic surgery | 1934 | 4 | 0.2% | 11 | 0.6% | 1949 |
| Abdominal | 59 | 1 | 1.6% | 1 | 1.6% | 61 |
| Diagnostic | 950 | 0 | 0.0% | 9 | 0.9% | 959 |
| ENT | 137 | 0 | 0.0% | 1 | 0.7% | 138 |
| Fracture | 27 | 1 | 3.6% | 0 | 0.0% | 28 |
| Miscellaneous | 570 | 1 | 0.2% | 2 | 0.4% | 573 |
| Orthopaedics | 138 | 0 | 0.0% | 0 | 0.0% | 138 |
| Urogenital | 100 | 1 | 1.0% | 2 | 1.9% | 103 |
| Colic surgery | 4 | 0 | 0.0% | 2 | 33.3% | 6 |
| Overall | 1938 | 4 | 0.2% | 13 | 0.7% | 1955 |
| Pre-Existing Condition and Brief Noncolic Death Description | n | Phase | Protocol |
|---|---|---|---|
| General anaesthesia | |||
| ASA I and II | 18 | ||
| Fracture in recovery (2 ASA I, 3 ASA II) | 5 | Recovery | 2 PIVA, 2 INH, 1 TIVA |
| Post-operative colic (5 ASA II) | 5 | Days 3, 4, 5, 5 and 5 | 3 PIVA, 2 INH |
| Post-operative myelomalacia (2 ASA II) | 2 | Days 2 and 4 | 2 PIVA |
| Cardiac arrest (ASA I) | 1 | Maintenance | INH |
| Upper airway obstruction in recovery (ASA II) | 1 | Recovery | PIVA |
| Fracture C2 (ASA II) | 1 | Recovery | PIVA |
| Sudden collapse after standing (ASA I) | 1 | Recovery | TIVA |
| Presumed spinal cord malacia (ASA II) | 1 | Recovery | PIVA |
| Small colon prolapse (ASA II) | 1 | Day 1 | PIVA |
| ASA III, IV and V | 17 | ||
| Re-fractures in recovery (3 ASA IV) | 3 | Recovery | 3 PIVA |
| Recumbent on arrival. Unable to stand after anaesthesia (2 ASA III) | 2 | Recovery | 1 PIVA, 1 TIVA |
| Intraoperative bleeding without response to treatment (2 ASA III) | 2 | Maintenance | 2 PIVA |
| Catastrophic recovery (<1 year with sling) (ASA III) | 1 | Recovery | PIVA |
| Post-operative colic (1 ASA III) | 1 | Day 3 | INH |
| Re-fracture olecranon in the stable (ASA III) | 1 | Day 3 | PIVA |
| Unknown, could not stand, no fracture diagnosed in X-rays (ASA III) | 1 | Recovery | INH |
| Severe chronic sinusitis. Sudden death. Necropsy: communication between sinus fistula and cranial cavity (ASA IV) | 1 | Recovery | TIVA |
| Foal with sepsis, diarrhoea (ASA IV) | 1 | Day 5 | TIVA |
| Bladder rupture on caesarean section (ASA V)Fracture rib repair, hemothorax and hemopericardium (ASA V) | 1 | Maintenance | PIVA |
| 1 | Maintenance | INH | |
| Fracture/abductor tear after dystocia (ASA V)Intraoperative bleeding dystocia (ASA V) | 1 | Day 2 | PIVA |
| 1 | Day 1 | INH | |
| Standing sedation | 4 | ||
| Post-operative colitis (ASA I, ASA II) | 2 | Days 1 and 6 | CRI |
| Post-operative colic (ASA III) | 1 | Day 2 | CRI |
| Re-fracture in stable (ASA III) | 1 | Day 1 | CRI |
| PREM | IND | MAIN | REC | 1D | 2D | 3D | 4D | 5D | 6D | 7D | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Colic | Deaths | 1 | 3 | 7 | 10 | 6 | 1 | 1 | 1 | 0 | 0 | 1 |
| PTS | 0 | 0 | 186 | 12 | 14 | 15 | 9 | 5 | 9 | 0 | 5 | |
| Noncolic | Deaths | 0 | 0 | 5 | 18 | 4 | 3 | 3 | 1 | 4 | 1 | 0 |
| PTS | 0 | 0 | 38 | 3 | 15 | 12 | 8 | 4 | 3 | 1 | 3 | |
| Total | Deaths | 1 | 3 | 12 | 28 | 10 | 4 | 4 | 2 | 4 | 1 | 1 |
| PTS | 0 | 0 | 224 | 15 | 29 | 27 | 17 | 9 | 12 | 1 | 8 |
| Drugs | PREM | IND | MAIN—(Bolus If Not Inhalant) | MAIN-CRI | POST |
|---|---|---|---|---|---|
| Acepromazine | 2776 | 0 | 42 | 0 | 162 |
| Xylazine | 3231 | 0 | 327 | 724 | 2303 |
| Detomidine | 1569 | 0 | 44 | 274 | 340 |
| Romifidine | 1874 | 0 | 89 | 1132 | 1699 |
| Medetomidine | 236 | 0 | 81 | 471 | 287 |
| Dexmedetomidine | 5 | 0 | 65 | 296 | 52 |
| Midazolam | 87 | 2795 | 40 | 125 | 2 |
| Diazepam | 112 | 3293 | 17 | 0 | 2 |
| Morphine | 1270 | 0 | 239 | 7 | 222 |
| Methadone | 498 | 0 | 66 | 2 | 26 |
| Pethidine | 0 | 0 | 0 | 0 | 0 |
| Fentanyl | 6 | 0 | 4 | 3 | 0 |
| Buprenorphine | 2 | 0 | 1 | 0 | 5 |
| Butorphanol | 3320 | 0 | 261 | 238 | 94 |
| Phenylbutazone | 2647 | 0 | 64 | 0 | 657 |
| Flunixin | 2223 | 0 | 73 | 0 | 673 |
| Meloxicam | 413 | 0 | 5 | 0 | 143 |
| Ketoprofen | 93 | 0 | 1 | 0 | 6 |
| Propofol | 0 | 316 | 40 | 8 | 21 |
| Alfaxalone | 0 | 0 | 1 | 0 | 0 |
| Ketamine | 0 | 6633 | 1158 | 1090 | 37 |
| Thiopental | 0 | 95 | 234 | 0 | 23 |
| TLT-ZLZ | 0 | 41 | 1 | 0 | 0 |
| GGE | 0 | 84 | 44 | 442 | 0 |
| Halothane | 0 | 0 | 0 | 0 | 0 |
| Isoflurane | 0 | 1 | 5250 | 0 | 0 |
| Sevoflurane | 0 | 1 | 547 | 0 | 0 |
| Desflurane | 0 | 1 | 203 | 0 | 0 |
| Lidocaine | 17 | 0 | 0 | 1255 | 86 |
| Dobutamine | 0 | 0 | 0 | 4281 | 20 |
| Phenylephrine | 0 | 0 | 12 | 277 | 960 |
| Drugs and Combinations | n | % |
|---|---|---|
| Alpha2 + Partial/Agonists-Antagonists Opioids | 2214 | 33.0% |
| ACP + Alpha2 + Partial/Agonists-Antagonists Opioids | 1026 | 15.3% |
| ACP + Alpha2 + Pure Opioids | 1017 | 15.2% |
| Alpha2 alone | 872 | 13.0% |
| ACP + Alpha2 | 721 | 10.8% |
| Alpha2 + Pure Opioids | 704 | 10.5% |
| Partial/Agonist-Antagonist Opioid alone | 62 | 0.9% |
| Pure Opioid alone | 31 | 0.5% |
| Benzodiazepine alone | 27 | 0.4% |
| ACP + Alpha2 + Pure Opioids + Partial/Agonists-Antagonists Opioids | 11 | 0.2% |
| Alpha2 + Pure Opioids + Partial/Agonists-Antagonists Opioids | 8 | 0.1% |
| ACP + Partial/Agonists-Antagonists Opioids | 1 | 0.0% |
| None | 7 | 0.1% |
| Induction Drugs | n | % |
|---|---|---|
| Ketamine + Benzodiazepine | 5919 | 88.3% |
| Ketamine + Propofol | 295 | 4.4% |
| Ketamine alone | 247 | 3.7% |
| Ketamine + Thiopental | 91 | 1.4% |
| Ketamine + GGE | 80 | 1.2% |
| Tiletamine + Zolazepam | 41 | 0.6% |
| Propofol | 21 | 0.3% |
| Thiopental + GGE | 3 | 0.1% |
| Ketamine + Inhalatory | 1 | 0.0% |
| Inhalatory + GGE | 1 | 0.0% |
| Thiopental alone | 1 | 0.0% |
| Inhalatory | 1 | 0.0% |
| Maintenance Drugs | n | % |
|---|---|---|
| Isoflurane | 5250 | 78.3% |
| Sevoflurane | 547 | 8.2% |
| Triple drip alone | 277 | 4.1% |
| Desflurane | 203 | 3.0% |
| Ketamine alone | 87 | 1.3% |
| Thiopental alone | 4 | 0.1% |
| Ketamine + Thiopental | 3 | 0.0% |
| None | 330 | 5.0% |
| CRI Drugs | n | % |
|---|---|---|
| Alpha2 alone | 1780 | 47.9% |
| Lidocaine alone | 1041 | 28.0% |
| Alpha2 + Ketamine | 384 | 10.3% |
| Alpha2 + Ketamine + Butorphanol | 147 | 4.0% |
| Alpha2 + Lidocaine | 78 | 2.1% |
| Ketamine alone | 70 | 1.9% |
| Alpha2 + Butorphanol | 68 | 1.8% |
| Lidocaine + Ketamine | 65 | 1.7% |
| Alpha2 + Lidocaine + Ketamine | 47 | 1.3% |
| Alpha2 + Lidocaine + Butorphanol | 18 | 0.5% |
| Other combinations | 20 | 0.5% |
| Drugs and Combinations | n | % |
|---|---|---|
| Alpha2 alone | 4220 | 63.0% |
| Alpha2 + Pure Opioids | 197 | 2.9% |
| ACP + Alpha2 | 91 | 1.4% |
| Alpha2 + Partial/Agonists-Antagonists Opioids | 65 | 1.0% |
| ACP alone | 38 | 0.6% |
| Pure Opioid alone | 37 | 0.6% |
| ACP + Alpha2 + Partial/Agonists-Antagonists Opioids | 18 | 0.3% |
| Partial/Agonist-Antagonist Opioid alone | 11 | 0.2% |
| ACP + Alpha2 + Pure Opioids | 10 | 0.1% |
| ACP + Partial/Agonists-Antagonists Opioids | 3 | 0.0% |
| Benzodiazepine alone | 2 | 0.0% |
| ACP + Alpha2 + Pure Opioids + Partial/Agonists-Antagonists Opioids | 1 | 0.0% |
| ACP + Pure Opioids | 1 | 0.0% |
| Pure Opioids + Partial/Agonists-Antagonists Opioids | 1 | 0.0% |
| None | 2006 | 29.9% |
| Induction | INH (n = 2282) | PIVA (n = 3718) | TIVA (n = 701) | Overall (n = 6701) |
|---|---|---|---|---|
| Personnel assisted | 1498 (65.6%) | 2010 (54.1%) | 570 (81.3%) | 4078 (60.8%) |
| Gate | 353 (15.5%) | 1207 (32.5%) | 86 (12.3%) | 1646 (24.6%) |
| Free | 407 (17.8%) | 479 (12.9%) | 44 (6.3%) | 930 (13.9%) |
| Table | 18 (0.8%) | 9 (0.2%) | 0 (0%) | 27 (0.4%) |
| Sling | 6 (0.3%) | 13 (0.3%) | 1 (0.1%) | 20 (0.3%) |
| INH (n = 2225) | PIVA (n = 3543) | TIVA (n = 693) | Overall * (n = 6461) | |
|---|---|---|---|---|
| Free | 1049 (47.2%) | 1770 (50.0%) | 462 (66.7%) | 3281 (50.8%) |
| Ropes | 942 (42.3%) | 1598 (45.1%) | 116 (16.7%) | 2656 (41.1%) |
| Manual | 234 (10.5%) | 175 (4.9%) | 115 (16.6%) | 524 (8.1%) |
| Drugs | PREM | MAIN—(Bolus) | MAIN-CRI | POST |
|---|---|---|---|---|
| Acepromazine | 918 | 15 | 0 | 1 |
| Xylazine | 61 | 17 | 29 | 1 |
| Detomidine | 1574 | 1178 | 525 | 1 |
| Romifidine | 405 | 302 | 60 | 0 |
| Medetomidine | 11 | 8 | 10 | 0 |
| Dexmedetomidine | 4 | 7 | 4 | 0 |
| Midazolam | 15 | 61 | 34 | 0 |
| Diazepam | 3 | 10 | 1 | 0 |
| Morphine | 532 | 54 | 76 | 7 |
| Methadone | 223 | 20 | 4 | 2 |
| Pethidine | 2 | 0 | 0 | 0 |
| Fentanyl | 2 | 0 | 0 | 0 |
| Buprenorphine | 3 | 0 | 0 | 0 |
| Butorphanol | 922 | 437 | 94 | 0 |
| Phenylbutazone | 253 | 5 | 0 | 115 |
| Flunixin | 513 | 3 | 0 | 137 |
| Meloxicam | 39 | 2 | 0 | 14 |
| Ketamine | 0 | 8 | 24 | 0 |
| Phenylephrine | 0 | 1 | 3 | 14 |
| Drugs and Combinations | n | % |
|---|---|---|
| Alpha2 + Partial/Agonists-Antagonists Opioids | 588 | 30.1% |
| ACP + Alpha2 + Pure Opioids | 393 | 20.1% |
| Alpha2 + Pure Opioids | 338 | 17.3% |
| ACP + Alpha2 + Partial/Agonists-Antagonists Opioids | 312 | 16.0% |
| ACP + Alpha2 | 203 | 10.4% |
| Alpha2 alone | 93 | 4.8% |
| Alpha2 + Pure Opioids + Partial/Agonists-Antagonists Opioids | 14 | 0.7% |
| ACP + Alpha2 + Pure Opioids + Partial/Agonists-Antagonists Opioids | 10 | 0.5% |
| Pure Opioid alone | 3 | 0.1% |
| Partial/Agonist-Antagonist Opioid alone | 1 | 0.0% |
| CRI Drugs | n | % |
|---|---|---|
| Alpha2 alone | 448 | 22.9% |
| Alpha2 + Butorphanol | 88 | 4.5% |
| Alpha2 + Morphine | 66 | 3.4% |
| Alpha2 + Ketamine | 10 | 0.5% |
| Alpha2 + Ketamine + Morphine | 8 | 0.4% |
| Alpha2 + Ketamine + Butorphanol | 4 | 0.2% |
| Alpha2 + Methadone | 3 | 0.2% |
| Other combinations | 6 | 0.3% |
| No CRI (only top-ups) | 1322 | 67.6% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gozalo-Marcilla, M.; Bettschart-Wolfensberger, R.; Johnston, M.; Taylor, P.M.; Redondo, J.I. Data Collection for the Fourth Multicentre Confidential Enquiry into Perioperative Equine Fatalities (CEPEF4) Study: New Technology and Preliminary Results. Animals 2021, 11, 2549. https://doi.org/10.3390/ani11092549
Gozalo-Marcilla M, Bettschart-Wolfensberger R, Johnston M, Taylor PM, Redondo JI. Data Collection for the Fourth Multicentre Confidential Enquiry into Perioperative Equine Fatalities (CEPEF4) Study: New Technology and Preliminary Results. Animals. 2021; 11(9):2549. https://doi.org/10.3390/ani11092549
Chicago/Turabian StyleGozalo-Marcilla, Miguel, Regula Bettschart-Wolfensberger, Mark Johnston, Polly M. Taylor, and Jose I. Redondo. 2021. "Data Collection for the Fourth Multicentre Confidential Enquiry into Perioperative Equine Fatalities (CEPEF4) Study: New Technology and Preliminary Results" Animals 11, no. 9: 2549. https://doi.org/10.3390/ani11092549
APA StyleGozalo-Marcilla, M., Bettschart-Wolfensberger, R., Johnston, M., Taylor, P. M., & Redondo, J. I. (2021). Data Collection for the Fourth Multicentre Confidential Enquiry into Perioperative Equine Fatalities (CEPEF4) Study: New Technology and Preliminary Results. Animals, 11(9), 2549. https://doi.org/10.3390/ani11092549








