Zoonotic Parasites in Playgrounds in Southern Spain: A One Health Approach
Abstract
1. Introduction
2. Materials and Methods
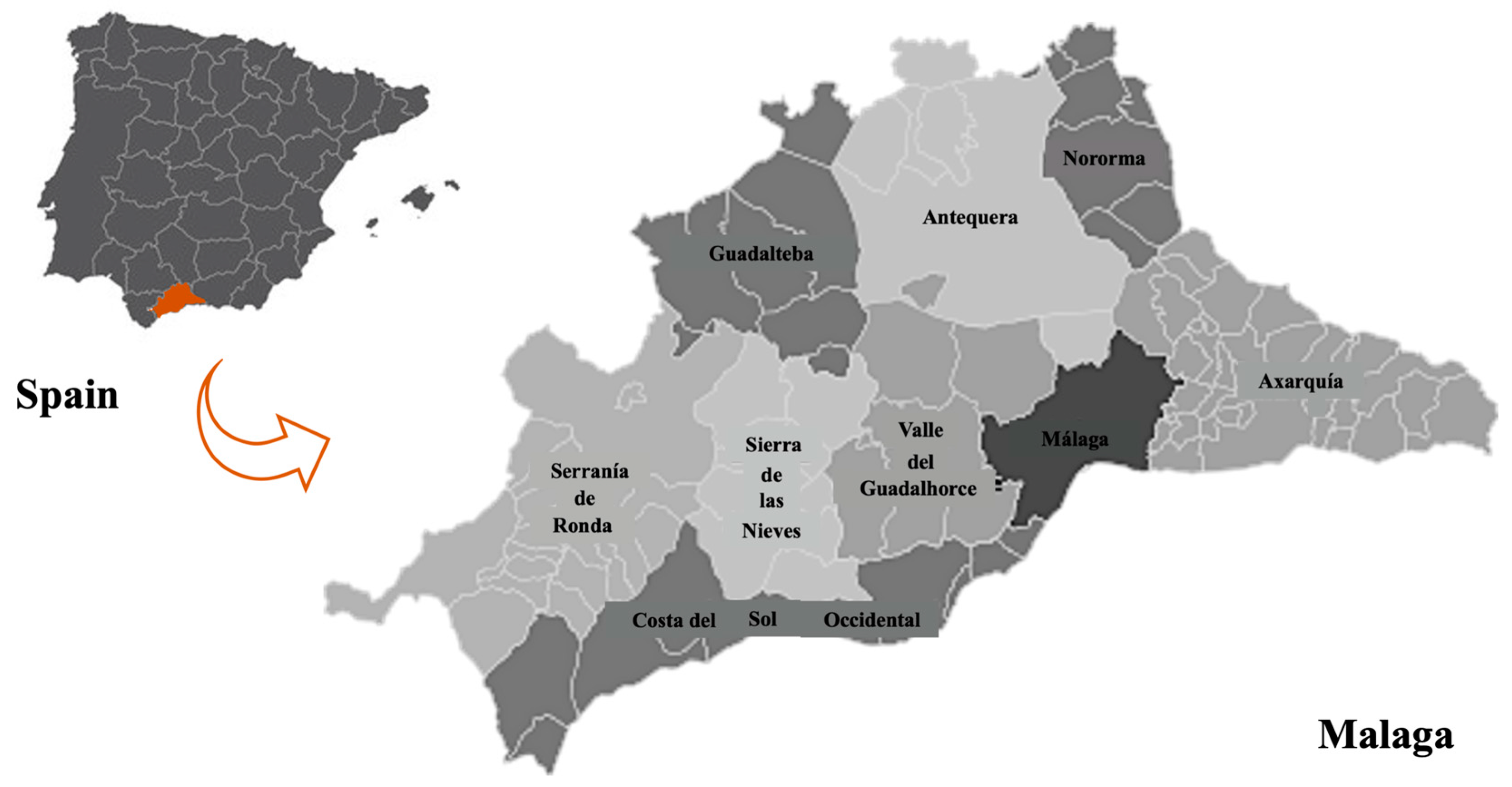
2.1. Study Playgrounds
2.2. Sampling
2.3. Laboratory Diagnosis
2.4. Statistical Testing
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fletcher, S.M.; Stark, D.; Harkness, J.; Ellis, J. Enteric Protozoa in the Developed World: A Public Health Perspective. Clin. Microbiol. Rev. 2012, 25, 420–449. [Google Scholar] [CrossRef] [PubMed]
- De Mello, C.C.S.; Nizoli, L.Q.; Ferraz, A.; Chagas, B.C.; Azario, W.J.D.; Villela, M.M. Helminth Eggs with Zoonotic Potential in the Vicinity of Public Schools in Southern Brazil. Rev. Bras. Parasitol. Vet. 2020, 29, e016419. [Google Scholar] [CrossRef]
- Candela, E.; Goizueta, C.; Periago, M.V.; Muñoz-Antoli, C. Prevalence of Intestinal Parasites and Molecular Characterization of Giardia Intestinalis, Blastocystis Spp. and Entamoeba Histolytica in the Village of Fortín Mbororé (Puerto Iguazú, Misiones, Argentina). Parasites Vectors 2021, 14, 510. [Google Scholar] [CrossRef]
- Ola-Fadunsin, S.D.; Abdulrauf, A.B.; Abdullah, D.A.; Ganiyu, I.A.; Hussain, K.; Sanda, I.M.; Rabiu, M.; Akanbi, O.B. Epidemiological Studies of Gastrointestinal Parasites Infecting Dogs in Kwara Central, North Central, Nigeria. Comp. Immunol. Microbiol. Infect. Dis. 2023, 93, 101943. [Google Scholar] [CrossRef]
- Ziam, H.; Kelanemer, R.; Belala, R.; Medrouh, B.; Khater, H.F.; Djerbal, M.; Kernif, T. Prevalence and Risk Factors Associated with Gastrointestinal Parasites of Pet Dogs in North-Central Algeria. Comp. Immunol. Microbiol. Infect. Dis. 2022, 86, 101817. [Google Scholar] [CrossRef]
- FEDIAC Annual Report 2022-2. 2023. Available online: https://europeanpetfood.org/wp-content/uploads/2023/02/Annual-Report-2022-2.pdf (accessed on 7 January 2023).
- Santos de Mello, C.C.; Nizoli, L.Q.; Ferraz, A.; Chagas, B.C.; Azario, W.J.D.; da Motta, S.P.; Villela, M.M. Soil Contamination by Ancylostoma spp. and Toxocara spp. Eggs in Elementary School Playgrounds in the Extreme South of Brazil. Rev. Bras. Parasitol. Vet. 2022, 31, e019121. [Google Scholar] [CrossRef]
- Mohammed, A.S.; Abd Alla, A.; Galander, A.; Elfaki, T.; Ibrahim Hashim, A.; Altayb, H.N. Frequency of Parasitic Infections in Arachishypogaea L. (Groundnuts), Citrulluslanatus Seeds (Watermelon Seeds) and Ziziphusspina-Christi (Nabag) Sold by Street Vendors in Khartoum State, Sudan: A Cross-Sectional Study. F1000Res 2021, 10, 586. [Google Scholar] [CrossRef]
- Zottler, E.M.; Bieri, M.; Basso, W.; Schnyder, M. Intestinal Parasites and Lungworms in Stray, Shelter and Privately Owned Cats of Switzerland. Parasitol. Int. 2019, 69, 75–81. [Google Scholar] [CrossRef]
- De Souza, C.T.V.; Dorr, A.P.; Silva, V.L.D.B.; Silva, F.D.L.; da Silva, E.B.; Ramos, D.G.D.S.; Pacheco, R.D.C.; Sousa, V.R.F. Occurrence of Gastrointestinal Parasites in Dogs from Cuiabá, Mato Grosso. Rev. Bras. Parasitol. Vet. 2023, 32, e012422. [Google Scholar] [CrossRef]
- Ebani, V.V.; Guardone, L.; Bertelloni, F.; Perrucci, S.; Poli, A.; Mancianti, F. Survey on the Presence of Bacterial and Parasitic Zoonotic Agents in the Feces of Wild Birds. Vet. Sci. 2021, 8, 171. [Google Scholar] [CrossRef]
- Vargas-Nava, A.I.; Nava, V.; Castro, N.; Campo, D.; Verdugo, I.E.; José, J.; Loera, P.; Barraza Tizoc, C.L.; Maribel, S.; Camacho, G. Prevalence and Viability of Toxocara spp. Eggs in Soil of Public Parks in Northwestern Mexico. Iran. J. Parasitol. 2020, 15, 196. [Google Scholar] [CrossRef]
- Raičević, J.G.; Pavlović, I.N.; Coghill, T.A.G. Canine Intestinal Parasites as a Potential Source of Soil Contamination in the Public Areas of Kruševac, Serbia. J. Infect. Dev. Ctries. 2021, 15, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Eisen, A.K.A.; Demoliner, M.; de Oliveira, K.G.; Troian, E.A.; Mallmann, L.; Filippi, M.; de Almeida, P.R.; Spilki, F.R. Soil Contamination of a Public Park by Human and Canine, as well as Hookworms and Toxocara spp. Eggs. Rev. Inst. Med. Trop. Sao Paulo 2019, 61, e60. [Google Scholar] [CrossRef]
- Galán-Puchades, M.T.; Trelis, M.; Sáez-Durán, S.; Cifre, S.; Gosálvez, C.; Sanxis-Furió, J.; Pascual, J.; Bueno-Marí, R.; Franco, S.; Peracho, V.; et al. One Health Approach to Zoonotic Parasites: Molecular Detection of Intestinal Protozoans in an Urban Population of Norway Rats, Rattus Norvegicus, in Barcelona, Spain. Pathogens 2021, 10, 311. [Google Scholar] [CrossRef]
- Ashiri, A.; Rafiei, A.; Beiromvand, M.; Khanzadeh, A.; Alghasi, A. Screening of Strongyloides Stercoralis Infection in High-Risk Patients in Khuzestan Province, Southwestern Iran. Parasites Vectors 2021, 14, 37. [Google Scholar] [CrossRef] [PubMed]
- Ngwese, M.M.; Manouana, G.P.; Alvyn, P.; Moure, N.; Ramharter, M.; Esen, M.; Adégnika, A.A. Tropical Medicine and Infectious Disease Diagnostic Techniques of Soil-Transmitted Helminths: Impact on Control Measures. Trop. Med. Infect. Dis. 2020, 5, 93. [Google Scholar] [CrossRef]
- Ash, L.; Oriel, T. Atlas de Parasitología Humana; Editorial Médica Panamericana: Buenos Aires, Argentina, 2010. [Google Scholar]
- Mikaeili, F.; Mathis, A.; Deplazes, P.; Mirhendi, H.; Barazesh, A.; Ebrahimi, S.; Kia, E.B. Differentiation of Toxocara canis and Toxocara cati Based on PCR-RFLP Analyses of RDNA-ITS and Mitochondrial Cox1 and Nad1 Regions. Acta Parasitol. 2017, 62, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Cociancic, P.; Deferrari, G.; Zonta, M.L.; Navone, G.T. Intestinal Parasites in Canine Feces Contaminating Urban and Recreational Areas in Ushuaia (Argentina). Vet. Parasitol. Reg. Stud. Rep. 2020, 21, 100424. [Google Scholar] [CrossRef]
- Köchle, B.R.; Garijo-Toledo, M.M.; Llobat, L.; Sansano-Maestre, J. Prevalence of Toxocara Eggs in Public Parks in the City of Valencia (Eastern Spain). Vet. Sci. 2022, 9, 232. [Google Scholar] [CrossRef]
- Montoya, A.; García, M.; Gálvez, R.; Checa, R.; Marino, V.; Sarquis, J.; Barrera, J.P.; Rupérez, C.; Caballero, L.; Chicharro, C.; et al. Implications of Zoonotic and Vector-Borne Parasites to Free-Roaming Cats in Central Spain. Vet. Parasitol. 2018, 251, 125–130. [Google Scholar] [CrossRef]
- Ahmed, F.; Cappai, M.G.; Morrone, S.; Cavallo, L.; Berlinguer, F.; Dessì, G.; Tamponi, C.; Scala, A.; Varcasia, A. Raw Meat Based Diet (RMBD) for Household Pets as Potential Door Opener to Parasitic Load of Domestic and Urban Environment. Revival of Understated Zoonotic Hazards? A Review. One Health 2021, 13, 100327. [Google Scholar] [CrossRef]
- Sanchez-Thevenet, P.; Carmena, D.; Adell-Aledón, M.; Dacal, E.; Arias, E.; Saugar, J.M.; Rodríguez, E.; Dea-Ayuela, M.A. High Prevalence and Diversity of Zoonotic and Other Intestinal Parasites in Dogs from Eastern Spain. Vector-Borne Zoonotic Dis. 2019, 19, 915–922. [Google Scholar] [CrossRef]
- Morelli, S.; Colombo, M.; Traversa, D.; Iorio, R.; Paoletti, B.; Bartolini, R.; Barlaam, A.; di Cesare, A. Zoonotic Intestinal Helminthes Diagnosed in a 6-Year Period (2015–2020) in Privately Owned Dogs of Sub-Urban and Urban Areas of Italy. Vet. Parasitol. Reg. Stud. Rep. 2022, 29, 100689. [Google Scholar] [CrossRef]
- Patterson, J. Toxocarosis in Humans: How Much of a Problem Is It in the UK? Drug Ther. Bull. 2023, 61, 7–11. [Google Scholar] [CrossRef]
- Chen, J.; Liu, Q.; Liu, G.H.; Zheng, W.b.; Hong, S.J.; Sugiyama, H.; Zhu, X.Q.; Elsheikha, H.M. Toxocariasis: A Silent Threat with a Progressive Public Health Impact. Infect. Dis. Poverty 2018, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rostami, A.; Riahi, S.M.; Holland, C.V.; Taghipour, A.; Khalili-Fomeshi, M.; Fakhri, Y.; Omrani, V.F.; Hotez, P.J.; Gasser, R.B. Seroprevalence Estimates for Toxocariasis in People Worldwide: A Systematic Review and Meta-Analysis. PLoS Negl. Trop. Dis. 2019, 13, e0007809. [Google Scholar] [CrossRef]
- Gonzalez-Quintela, A.; Gude, F.; Campos, J.; Garea, M.T.; Romero, P.A.; Rey, J.; Meijide, L.M.; Fernandez-Merino, M.C.; Vidal, C. Toxocara Infection Seroprevalence and its Relationship with Atopic Features in a General Adult Population. Int. Arch. Allergy Immunol. 2006, 139, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Rehbein, S.; Knaus, M.; Mallouk, Y.; Breiltgens, T.; Brianti, E.; Capári, B.; Dantas-Torres, F.; Gau, M.; Joachim, A.; Kaulfuß, K.H.; et al. Efficacy against Nematode Infections and Safety of Afoxolaner plus Milbemycin Oxime Chewable Tablets in Domestic Dogs under Field Conditions in Europe. Parasitol. Res. 2017, 116, 259–269. [Google Scholar] [CrossRef]
- Mahdavi, F.; Sadrebazzaz, A.; Chahardehi, A.M.; Badali, R.; Omidian, M.; Hassanipour, S.; Asghari, A. Global Epidemiology of Giardia Duodenalis Infection in Cancer Patients: A Systematic Review and Meta-Analysis. Int. Health 2022, 14, 5–17. [Google Scholar] [CrossRef] [PubMed]
- De Lucio, A.; Bailo, B.; Aguilera, M.; Cardona, G.A.; Fernández-Crespo, J.C.; Carmena, D. No Molecular Epidemiological Evidence Supporting Household Transmission of Zoonotic Giardia Duodenalis and Cryptosporidium spp. from Pet Dogs and Cats in the Province of Álava, Northern Spain. Acta Trop. 2017, 170, 48–56. [Google Scholar] [CrossRef]
- Papini, R.; Marangi, M.; Mancianti, F.; Giangaspero, A. Occurrence and Cyst Burden of Giardia Duodenalis in Dog Faecal Deposits from Urban Green Areas: Implications for Environmental Contamination and Related Risks. Prev. Vet. Med. 2009, 92, 158–162. [Google Scholar] [CrossRef]
- Lecová, L.; Hammerbauerová, I.; Tůmová, P.; Nohýnková, E. Companion Animals as a Potential Source of Giardia Intestinalis Infection in Humans in the Czech Republic—A Pilot Study. Vet. Parasitol. Reg. Stud. Rep. 2020, 21, 100431. [Google Scholar] [CrossRef] [PubMed]
- Yılmaz, A.; Uslu, H. Examination of Giardia Intestinalis with Direct Microscopy and Direct Fluorescent Antibody in Patients with Diarrhea. Turk. Parazitol. Derg. 2020, 44, 187–190. [Google Scholar] [CrossRef]
- Munoz, J.; Mayer, D.C.G. Toxoplasma Gondii and Giardia Duodenalis Infections in Domestic Dogs in New York City Public Parks. Vet. J. 2016, 211, 97–99. [Google Scholar] [CrossRef]
- Šmigová, J.; Papajová, I.; Šoltys, J.; Pipiková, J.; Šmiga, Ľ.; Šnábel, V.; Takáčová, J.; Takáč, L. The Occurence of Endoparasites in Slovakian Household Dogs and Cats. Vet. Res. Commun. 2021, 45, 243–249. [Google Scholar] [CrossRef]
- Cociancic, P.; Zonta, M.L.; Navone, G.T. A Cross-Sectional Study of Intestinal Parasitoses in Dogs and Children of the Periurban Area of La Plata (Buenos Aires, Argentina): Zoonotic Importance and Implications in Public Health. Zoonoses Public Health 2018, 65, e44–e53. [Google Scholar] [CrossRef]
- Martínez-Moreno, F.J.; Hernández, S.; López-Cobos, E.; Becerra, C.; Acosta, I.; Martínez-Moreno, A. Estimation of Canine Intestinal Parasites in Córdoba (Spain) and Their Risk to Public Health. Vet. Parasitol. 2007, 143, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Duncan, K.T.; Koons, N.R.; Litherland, M.A.; Little, S.E.; Nagamori, Y. Prevalence of Intestinal Parasites in Fecal Samples and Estimation of Parasite Contamination from Dog Parks in Central Oklahoma. Vet. Parasitol. Reg. Stud. Rep. 2020, 19, 100362. [Google Scholar] [CrossRef]
- Villamizar, X.; Higuera, A.; Herrera, G.; Vasquez-A, L.R.; Buitron, L.; Muñoz, L.M.; Gonzalez-C, F.E.; Lopez, M.C.; Giraldo, J.C.; Ramírez, J.D. Molecular and Descriptive Epidemiology of Intestinal Protozoan Parasites of Children and Their Pets in Cauca, Colombia: A Cross-Sectional Study. BMC Infect. Dis. 2019, 19, 190. [Google Scholar] [CrossRef] [PubMed]
| Phylum | Order | Family | Genus | Species | n | Total (%) |
|---|---|---|---|---|---|---|
| Nematoda | Strongylida | Ancylostomatidae | Ancylostoma | A. caninum | 4 | 3.0 ± 1.6 b |
| Ascaridida | Ascarididae | Toxocara | Toxocara spp. | 21 | 17.0 ± 3.5 a | |
| Rhabditida | Ancylostomatidae | Uncinaria | U. stenocephala | 11 | 9.0 ± 2.6 ab | |
| Sarcomastigophora | Diplomonadida | Hexamitidae | Giardia | G. duodenalis | 20 | 17.0 ± 3.4 a |
| Platyhelminthes | Cyclophyllidea | Dilepidiidae | Dipylidium | D. caninum | 4 | 3.0 ± 1.6 b |
| Parasites | n | Total (%) | ||
|---|---|---|---|---|
| Toxocara canis | Giardia duodenalis | 5 | 33.0 | |
| Toxocara canis | Uncinaria stenocephala | 3 | 20.0 | |
| Giardia duodenalis | Uncinaria stenocephala | 2 | 13.0 | |
| Toxocara canis | Ancylostoma caninum | 1 | 7.0 | |
| Uncinaria stenocephala | Dipylidium caninum | 1 | 7.0 | |
| Uncinaria stenocephala | Ancylostoma caninum | 1 | 7.0 | |
| Giardia duodenalis | Dipylidium caninum | Uncinaria stenocephala | 1 | 7.0 |
| Toxocara canis | Toxocara catis | Dipylidium caninum | 1 | 7.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lorenzo-Rebenaque, L.; López-Fernández, S.; Marco-Jiménez, F.; Montoro-Dasi, L.; Marin, C.; Vega, S.; Martínez-Manzanares, E.; Fariñas, F. Zoonotic Parasites in Playgrounds in Southern Spain: A One Health Approach. Microorganisms 2023, 11, 721. https://doi.org/10.3390/microorganisms11030721
Lorenzo-Rebenaque L, López-Fernández S, Marco-Jiménez F, Montoro-Dasi L, Marin C, Vega S, Martínez-Manzanares E, Fariñas F. Zoonotic Parasites in Playgrounds in Southern Spain: A One Health Approach. Microorganisms. 2023; 11(3):721. https://doi.org/10.3390/microorganisms11030721
Chicago/Turabian StyleLorenzo-Rebenaque, Laura, Sandra López-Fernández, Francisco Marco-Jiménez, Laura Montoro-Dasi, Clara Marin, Santiago Vega, Eduardo Martínez-Manzanares, and Fernando Fariñas. 2023. "Zoonotic Parasites in Playgrounds in Southern Spain: A One Health Approach" Microorganisms 11, no. 3: 721. https://doi.org/10.3390/microorganisms11030721
APA StyleLorenzo-Rebenaque, L., López-Fernández, S., Marco-Jiménez, F., Montoro-Dasi, L., Marin, C., Vega, S., Martínez-Manzanares, E., & Fariñas, F. (2023). Zoonotic Parasites in Playgrounds in Southern Spain: A One Health Approach. Microorganisms, 11(3), 721. https://doi.org/10.3390/microorganisms11030721






