Respiratory Tract Infections and Laboratory Diagnostic Methods: A Review with A Focus on Syndromic Panel-Based Assays
Abstract
1. Introduction
2. The Epidemiology of RTIs
3. The Human Respiratory Tract and the Classification of RTIs
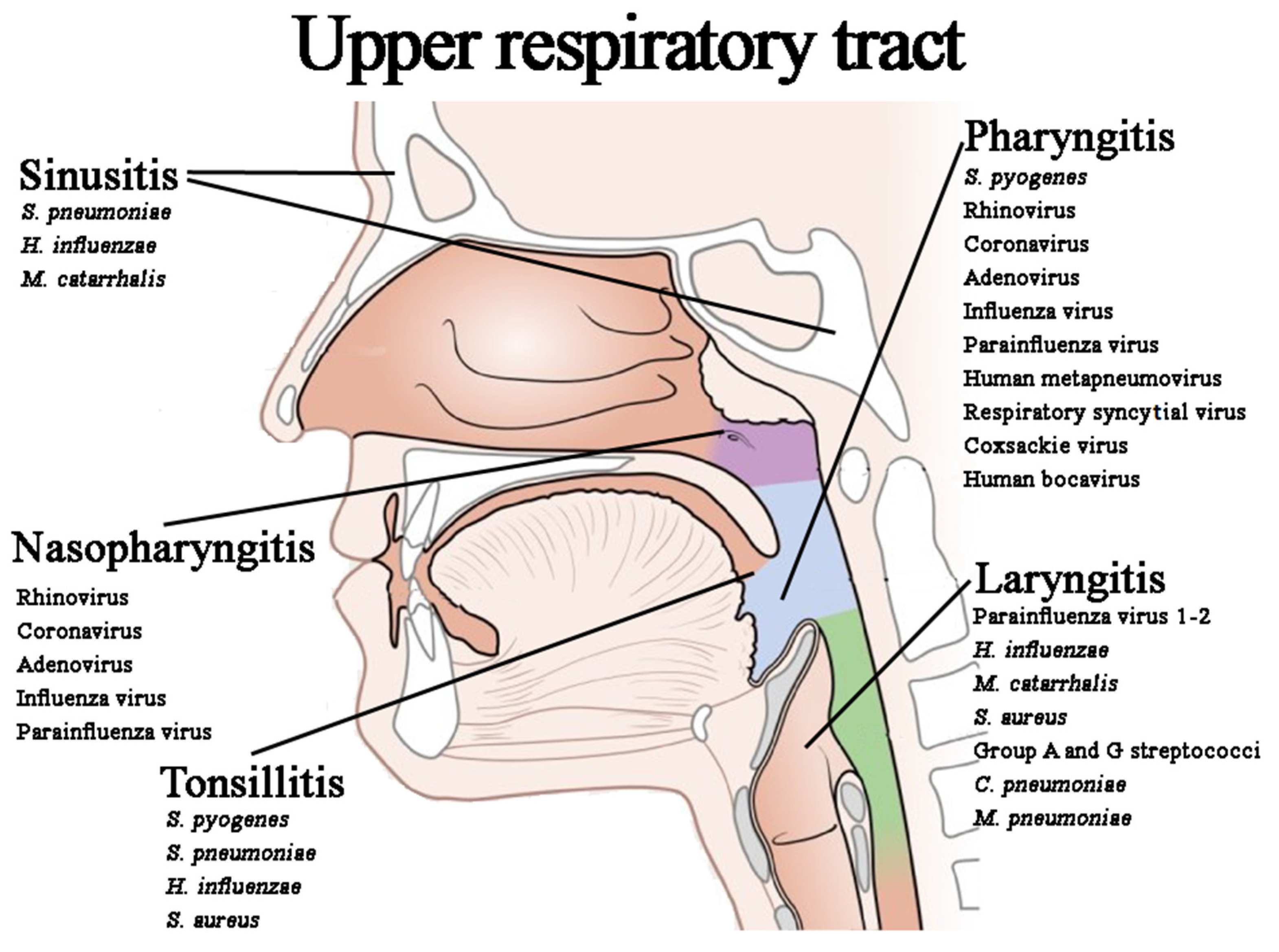
3.1. Upper Respiratory Tract Infections (URIs)
3.1.1. Acute Nasopharyngitis (AN)
3.1.2. Acute Sinusitis (AS)
3.1.3. Acute Pharyngitis (AP)
3.1.4. Acute Tonsillitis (AT)
3.1.5. Acute Laryngitis (AL) and Laryngotracheobronchitis (LTB)
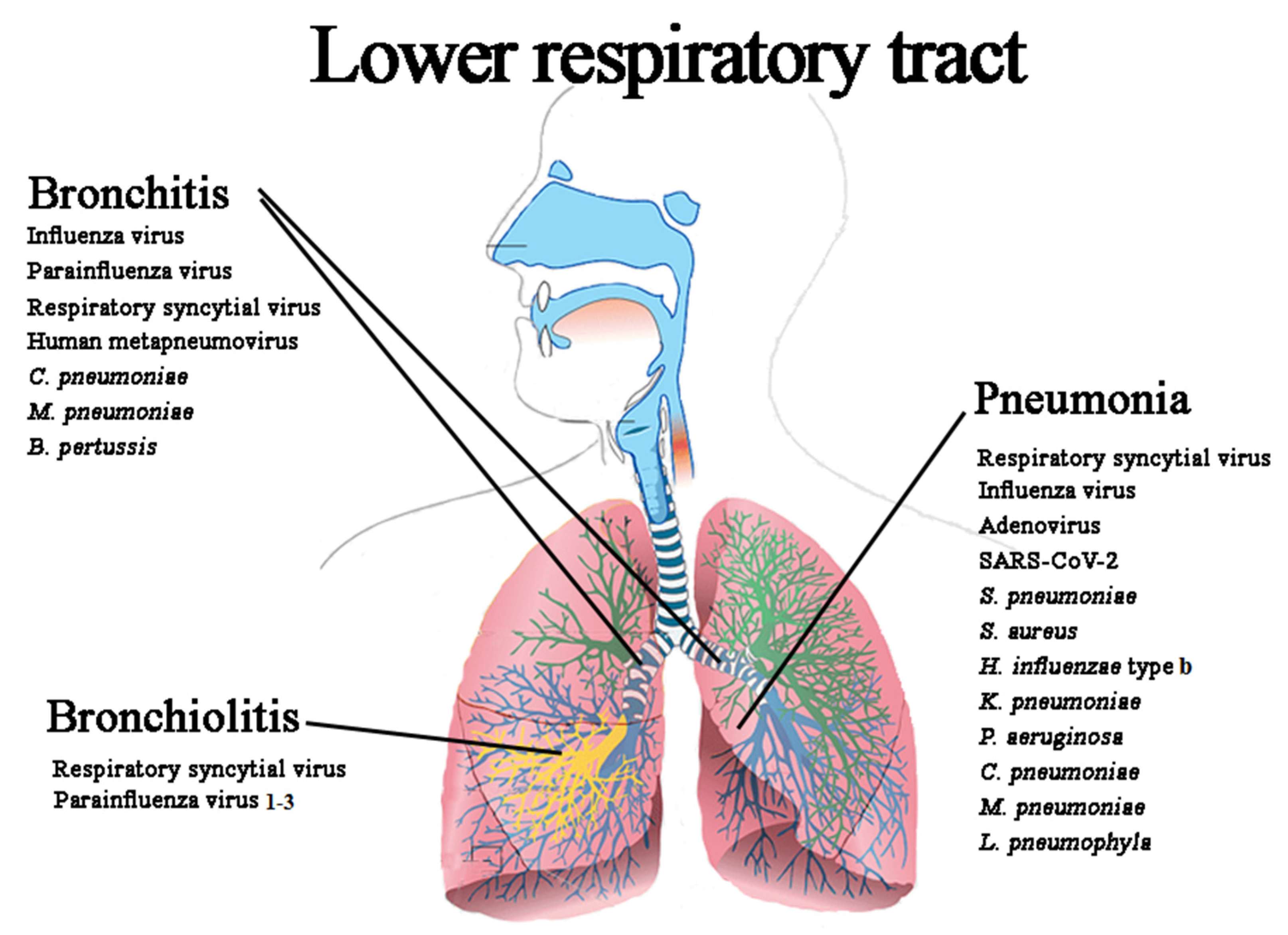
3.2. Lower Respiratory Tract Infections (LRIs)
3.2.1. Acute Bronchitis (AB)
3.2.2. Acute Bronchiolitis (ABR)
3.2.3. Pneumonia
4. Laboratory Diagnosis
4.1. Specimen Collection
4.2. Microscopy
4.3. Culture
4.4. Antigen Detection Assays
4.5. Serology
4.6. Nucleic Acid Amplification Tests
5. Multiplex Panel Assays
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO Severe Acute Respiratory Infections Treatment Centre. Practical Manual to Set Up and Manage a SARI Treatment Centre and a SARI Screening Facility in Health Care Facilities; World Health Organization: Geneva, Switzerland, 2020; p. 120. [Google Scholar]
- WHO. Infection Prevention and Control of Epidemic- and Pandemic-Prone Acute Respiratory Infections in Health Care: WHO Guidelines; World Health Organization: Geneva, Switzerland, 2014; p. 157. [Google Scholar]
- Mirsaeidi, M.; Motahari, H.; Taghizadeh Khamesi, M.; Sharifi, A.; Campos, M.; Schraufnagel, D.E. Climate Change and Respiratory Infections. Ann. Am. Thorac. Soc. 2016, 13, 1223–1230. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Guo, F. Loss of Life Expectancy Due to Respiratory Infectious Diseases: Findings from the Global Burden of Disease Study in 195 Countries and Territories 1990–2017. J. Popul. Res. 2022, 39, 1–43. [Google Scholar] [CrossRef] [PubMed]
- Yanagihara, K. The Role of Molecular Diagnosis in Acute Respiratory Tract Infection. Respir. Investig. 2019, 57, 511. [Google Scholar] [CrossRef] [PubMed]
- Kuti, E.L.; Patel, A.A.; Coleman, C.I. Impact of Inappropriate Antibiotic Therapy on Mortality in Patients with Ventilator-Associated Pneumonia and Blood Stream Infection: A Meta-Analysis. J. Crit. Care 2008, 23, 91–100. [Google Scholar] [CrossRef]
- Webber, D.M.; Wallace, M.A.; Burnham, C.A.; Anderson, N.W. Evaluation of the BioFire FilmArray Pneumonia Panel for Detection of Viral and Bacterial Pathogens in Lower Respiratory Tract Specimens in the Setting of a Tertiary Care Academic Medical Center. J. Clin. Microbiol. 2020, 58, e00343-20. [Google Scholar] [CrossRef]
- Rytter, H.; Jamet, A.; Coureuil, M.; Charbit, A.; Ramond, E. Which Current and Novel Diagnostic Avenues for Bacterial Respiratory Diseases? Front. Microbiol. 2020, 11, 616971. [Google Scholar] [CrossRef]
- Leland, D.S.; Ginocchio, C.C. Role of Cell Culture for Virus Detection in the Age of Technology. Clin. Microbiol. Rev. 2007, 20, 49–78. [Google Scholar] [CrossRef]
- Couturier, M.R.; Bard, J.D. Direct-from-Specimen Pathogen Identification: Evolution of Syndromic Panels. Clin. Lab. Med. 2019, 39, 433–451. [Google Scholar] [CrossRef]
- WHO The Global Health Observatory. Global Health Estimates: Leading Causes of Death. Cause-Specific Mortality, 2000–2019. Available online: https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates/ghe-leading-causes-of-death (accessed on 29 July 2022).
- WHO The Global Health Observatory. Global Health Estimates: Leading Causes of DALYs. Disease Burden, 2000–2019. Available online: https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates/global-health-estimates-leading-causes-of-dalys (accessed on 29 July 2022).
- The World Bank World Bank Country and Lending Groups. Country Classification. Available online: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups (accessed on 29 July 2022).
- Simoes, E.A.F.; Cherian, T.; Chow, J.; Shahid-Salles, S.A.; Laxminarayan, R.; John, T.J. Acute Respiratory Infections in Children; Jamison, D.T., Breman, J.G., Measham, A.R., Alleyne, G., Claeson, M., Evans, D.B., Jha, P., Mills, A., Musgrove, P., Eds.; The International Bank for Reconstruction and Development/The World Bank: Washington, DC, USA, 2006; ISBN 0-8213-6179-1. [Google Scholar]
- WHO International Statistical Classification of Diseases and Related Health Problems (ICD). Available online: https://www.who.int/standards/classifications/classification-of-diseases (accessed on 29 July 2022).
- Grief, S.N. Upper Respiratory Infections. Prim. Care 2013, 40, 757–770. [Google Scholar] [CrossRef]
- Miller, J.M.; Binnicker, M.J.; Campbell, S.; Carroll, K.C.; Chapin, K.C.; Gilligan, P.H.; Gonzalez, M.D.; Jerris, R.C.; Kehl, S.C.; Patel, R.; et al. A Guide to Utilization of the Microbiology Laboratory for Diagnosis of Infectious Diseases: 2018 Update by the Infectious Diseases Society of America and the American Society for Microbiology. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2018, 67, 813–816. [Google Scholar] [CrossRef]
- Flores, A.R.; Caserta, M.T. Pharyngitis. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2015; pp. 753–759.e2. [Google Scholar]
- Arnold, J.C.; Nizet, V. Pharyngitis. In Principles and Practice of Pediatric Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2018; pp. 202–208.e2. [Google Scholar]
- Mustafa, Z.; Ghaffari, M. Diagnostic Methods, Clinical Guidelines, and Antibiotic Treatment for Group A Streptococcal Pharyngitis: A Narrative Review. Front. Cell. Infect. Microbiol. 2020, 10, 563627. [Google Scholar] [CrossRef]
- Wolford, R.W.; Goyal, A.; Belgam Syed, S.Y.; Schaefer, T.J. Pharyngitis; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Alcaide, M.L.; Bisno, A.L. Pharyngitis and Epiglottitis. Infect. Dis. Clin. N. Am. 2007, 21, 449–469. [Google Scholar] [CrossRef]
- Weber, R. Pharyngitis. Prim. Care 2014, 41, 91–98. [Google Scholar] [CrossRef]
- Anderson, J.; Paterek, E. Tonsillitis; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Bartlett, A.; Bola, S.; Williams, R. Acute Tonsillitis and Its Complications: An Overview. J. R. Nav. Med. Serv. 2015, 101, 69–73. [Google Scholar] [CrossRef]
- Tristram, D. Laryngitis, Tracheitis, Epiglottitis, and Bronchiolitis: Sore Throat, Change in Voice, Feverora Wheezing Infant in Respiratory Distress. In Introduction to Clinical Infectious Diseases: A Problem-Based Approach; Springer: Cham, Switzerland, 2018; pp. 75–85. [Google Scholar]
- Caserta, M.T. Acute Laryngitis. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2015; pp. 760–761.e1. [Google Scholar]
- Blot, M.; Bonniaud-Blot, P.; Favrolt, N.; Bonniaud, P.; Chavanet, P.; Piroth, L. Update on Childhood and Adult Infectious Tracheitis. Med. Mal. Infect. 2017, 47, 443–452. [Google Scholar] [CrossRef]
- Bower, J.; McBride, J.T. Croup in Children (Acute Laryngotracheobronchitis). In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2015; pp. 762–766.e1. [Google Scholar]
- Woodfork, K. Bronchitis. In xPharm: The Comprehensive Pharmacology Reference; Elsevier: Amsterdam, The Netherlands, 2007; pp. 1–13. [Google Scholar]
- Walsh, E.E. Acute Bronchitis. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2015; pp. 806–809.e1. [Google Scholar]
- de Conto, F.; Conversano, F.; Medici, M.C.; Ferraglia, F.; Pinardi, F.; Arcangeletti, M.C.; Chezzi, C.; Calderaro, A. Epidemiology of Human Respiratory Viruses in Children with Acute Respiratory Tract Infection in a 3-Year Hospital-Based Survey in Northern Italy. Diagn. Microbiol. Infect. Dis. 2019, 94, 260–267. [Google Scholar] [CrossRef]
- Rossi, G.A.; Medici, M.C.; Merolla, R. Incidence of Respiratory Syncytial Virus Positivity in Young Italian Children Referred to the Emergency Departments for Lower Respiratory Tract Infection over Two Consecutive Epidemic Seasons. Infection 2005, 33, 18–24. [Google Scholar] [CrossRef]
- Segal, L.N.; Weiden, M.D.; Horowitz, H.W. Acute Exacerbations of Chronic Obstructive Pulmonary Disease. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2015; pp. 810–817.e3. [Google Scholar]
- Medici, M.C.; Arcangeletti, M.C.; Merolla, R.; Chezzi, C. Incidence of Respiratory Syncytial Virus Infection in Infants and Young Children Referred to the Emergency Departments for Lower Respiratory Tract Diseases in Italy. Acta Biomed. 2004, 75, 26–33. [Google Scholar]
- WHO Pneumonia. Available online: https://www.who.int/news-room/fact-sheets/detail/pneumonia (accessed on 29 July 2022).
- Fauci, A.S. Harrison’s Principles of Internal Medicine; McGraw-Hill Education: New York, NY, USA, 2015; ISBN 0071802134. [Google Scholar]
- Lanks, C.W.; Musani, A.I.; Hsia, D.W. Community-Acquired Pneumonia and Hospital-Acquired Pneumonia. Med. Clin. N. Am. 2019, 103, 487–501. [Google Scholar] [CrossRef]
- Calderaro, A.; Buttrini, M.; Montecchini, S.; Piccolo, G.; Martinelli, M.; Dell’Anna, M.L.; di Maio, A.; Arcangeletti, M.C.; Maccari, C.; de Conto, F.; et al. Detection of SARS-CoV-2 and Other Infectious Agents in Lower Respiratory Tract Samples Belonging to Patients Admitted to Intensive Care Units of a Tertiary-Care Hospital, Located in an Epidemic Area, during the Italian Lockdown. Microorganisms 2021, 9, 185. [Google Scholar] [CrossRef]
- Weinberger, S.E.; Cockrill, B.A.; Mandel, J. Principles of Pulmonary Medicine E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2017; ISBN 0323523722. [Google Scholar]
- Albano, D.; Bertagna, F.; Alongi, P.; Baldari, S.; Baldoncini, A.; Bartolomei, M.; Boccaletto, F.; Boero, M.; Borsatti, E.; Bruno, A.; et al. Prevalence of Interstitial Pneumonia Suggestive of COVID-19 at (18)F-FDG PET/CT in Oncological Asymptomatic Patients in a High Prevalence Country during Pandemic Period: A National Multi-Centric Retrospective Study. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2871–2882. [Google Scholar] [CrossRef]
- Das, S.; Dunbar, S.; Tang, Y.-W. Laboratory Diagnosis of Respiratory Tract Infections in Children-the State of the Art. Front. Microbiol. 2018, 9, 2478. [Google Scholar] [CrossRef]
- Murdoch, D.R.; Werno, A.M.; Jennings, L.C. Microbiological Diagnosis of Respiratory Illness: Recent Advances. In Kendig’s Disorders of the Respiratory Tract in Children; Elsevier: Amsterdam, The Netherlands, 2019; pp. 396–405.e3. [Google Scholar]
- Campbell, S.; Forbes, B.A. The Clinical Microbiology Laboratory in the Diagnosis of Lower Respiratory Tract Infections. J. Clin. Microbiol. 2011, 49, S30–S33. [Google Scholar] [CrossRef]
- Ramanan, P.; Bryson, A.L.; Binnicker, M.J.; Pritt, B.S.; Patel, R. Syndromic Panel-Based Testing in Clinical Microbiology. Clin. Microbiol. Rev. 2018, 31, e00024-17. [Google Scholar] [CrossRef]
- Leber, A.L. Clinical Microbiology Procedures Handbook; John Wiley & Sons: New York, NY, USA, 2020; ISBN 1555818811. [Google Scholar]
- Centers for Disease Control and Prevention. Specimen Collection Guidelines; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2020. [Google Scholar]
- Charalambous, B.M.; Batt, S.L.; Peek, A.C.; Mwerinde, H.; Sam, N.; Gillespie, S.H. Quantitative Validation of Media for Transportation and Storage of Streptococcus Pneumoniae. J. Clin. Microbiol. 2003, 41, 5551–5556. [Google Scholar] [CrossRef]
- Poulsen, C.S.; Kaas, R.S.; Aarestrup, F.M.; Pamp, S.J. Standard Sample Storage Conditions Have an Impact on Inferred Microbiome Composition and Antimicrobial Resistance Patterns. Microbiol. Spectr. 2021, 9, e0138721. [Google Scholar] [CrossRef]
- Skevaki, C.L.; Papadopoulos, N.G.; Tsakris, A.; Johnston, S.L. Microbiologic Diagnosis of Respiratory Illness: Practical Applications. In Kendig & Chernick’s Disorders of the Respiratory Tract in Children; Elsevier: Amsterdam, The Netherlands, 2012; pp. 399–423. [Google Scholar]
- Cooley, L.; Dendle, C.; Wolf, J.; Teh, B.W.; Chen, S.C.; Boutlis, C.; Thursky, K.A. Consensus Guidelines for Diagnosis, Prophylaxis and Management of P Neumocystis Jirovecii Pneumonia in Patients with Haematological and Solid Malignancies, 2014. Intern. Med. J. 2014, 44, 1350–1363. [Google Scholar] [CrossRef]
- Falsey, A.R.; Walsh, E.E. Novel Coronavirus and Severe Acute Respiratory Syndrome. Lancet 2003, 361, 1312–1313. [Google Scholar] [CrossRef]
- Ksiazek, T.G.; Erdman, D.; Goldsmith, C.S.; Zaki, S.R.; Peret, T.; Emery, S.; Tong, S.; Urbani, C.; Comer, J.A.; Lim, W.; et al. A Novel Coronavirus Associated with Severe Acute Respiratory Syndrome. N. Engl. J. Med. 2003, 348, 1953–1966. [Google Scholar] [CrossRef]
- Goldsmith, C.S.; Miller, S.E. Modern Uses of Electron Microscopy for Detection of Viruses. Clin. Microbiol. Rev. 2009, 22, 552–563. [Google Scholar] [CrossRef]
- Zhang, Y.; Hung, T.; Song, J.; He, J. Electron Microscopy: Essentials for Viral Structure, Morphogenesis and Rapid Diagnosis. Sci. China Life Sci. 2013, 56, 421–430. [Google Scholar] [CrossRef] [PubMed]
- LaSala, P.R.; Bufton, K.K.; Ismail, N.; Smith, M.B. Prospective Comparison of R-Mix Shell Vial System with Direct Antigen Tests and Conventional Cell Culture for Respiratory Virus Detection. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2007, 38, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Ginocchio, C.C. Detection of Respiratory Viruses Using Non-Molecular Based Methods. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2007, 40 (Suppl. S1), S11–S14. [Google Scholar] [CrossRef]
- Hematian, A.; Sadeghifard, N.; Mohebi, R.; Taherikalani, M.; Nasrolahi, A.; Amraei, M.; Ghafourian, S. Traditional and Modern Cell Culture in Virus Diagnosis. Osong Public Health Res. Perspect. 2016, 7, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Slinger, R.; Milk, R.; Gaboury, I.; Diaz-Mitoma, F. Evaluation of the QuickLab RSV Test, a New Rapid Lateral-Flow Immunoassay for Detection of Respiratory Syncytial Virus Antigen. J. Clin. Microbiol. 2004, 42, 3731–3733. [Google Scholar] [CrossRef]
- Chartrand, C.; Tremblay, N.; Renaud, C.; Papenburg, J. Diagnostic Accuracy of Rapid Antigen Detection Tests for Respiratory Syncytial Virus Infection: Systematic Review and Meta-Analysis. J. Clin. Microbiol. 2015, 53, 3738–3749. [Google Scholar] [CrossRef]
- WHO. Antigen-Detection in the Diagnosis of SARS-CoV-2 Infection; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- Loens, K.; van Heirstraeten, L.; Malhotra-Kumar, S.; Goossens, H.; Ieven, M. Optimal Sampling Sites and Methods for Detection of Pathogens Possibly Causing Community-Acquired Lower Respiratory Tract Infections. J. Clin. Microbiol. 2009, 47, 21–31. [Google Scholar] [CrossRef]
- Chkhaidze, I.; Manjavidze, N.; Nemsadze, K. Serodiagnosis of Acute Respiratory Infections in Children in Georgia. Indian J. Pediatr. 2006, 73, 569–572. [Google Scholar] [CrossRef]
- Kuypers, J.; Wright, N.; Ferrenberg, J.; Huang, M.-L.; Cent, A.; Corey, L.; Morrow, R. Comparison of Real-Time PCR Assays with Fluorescent-Antibody Assays for Diagnosis of Respiratory Virus Infections in Children. J. Clin. Microbiol. 2006, 44, 2382–2388. [Google Scholar] [CrossRef]
- File, T.M.J.; Tan, J.S.; Plouffe, J.F. The Role of Atypical Pathogens: Mycoplasma Pneumoniae, Chlamydia Pneumoniae, and Legionella Pneumophila in Respiratory Infection. Infect. Dis. Clin. N. Am. 1998, 12, 569–592. [Google Scholar] [CrossRef]
- Saikku, P. Diagnosis of Chlamydia Pneumoniae. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 1998, 4 (Suppl. S4), S7–S13. [Google Scholar] [CrossRef]
- Graham, F.F.; Hales, S.; White, P.S.; Baker, M.G. Review Global Seroprevalence of Legionellosis—A Systematic Review and Meta-Analysis. Sci. Rep. 2020, 10, 7337. [Google Scholar] [CrossRef]
- WHO. Laboratory Manual for the Diagnosis of Wooping Cough Caused by Bordetella Pertussis/Bordetella Parapertussis; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Loeffelholz, M.; Chonmaitree, T. Advances in Diagnosis of Respiratory Virus Infections. Int. J. Microbiol. 2010, 2010, 126049. [Google Scholar] [CrossRef]
- WHO. Clinical Care for Severe Acute Respiratory Infection; WHO: Geneva, Switzerland, 2022. [Google Scholar]
- Messacar, K.; Parker, S.K.; Todd, J.K.; Dominguez, S.R. Implementation of Rapid Molecular Infectious Disease Diagnostics: The Role of Diagnostic and Antimicrobial Stewardship. J. Clin. Microbiol. 2017, 55, 715–723. [Google Scholar] [CrossRef]
- Dien Bard, J.; McElvania, E. Panels and Syndromic Testing in Clinical Microbiology. Clin. Lab. Med. 2020, 40, 393–420. [Google Scholar] [CrossRef]
- Hanson, K.E.; Couturier, M.R. Multiplexed Molecular Diagnostics for Respiratory, Gastrointestinal, and Central Nervous System Infections. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2016, 63, 1361–1367. [Google Scholar] [CrossRef]
- Zanella, M.-C.; Meylan, P.; Kaiser, L. Syndromic Panels or “Panel Syndrome”? A Perspective through the Lens of Respiratory Tract Infections. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2020, 26, 665–668. [Google Scholar] [CrossRef]
- Fox, A.S.; Rao, S.N. Syndromic Testing for the Diagnosis of Infectious Diseases: The Right Test If Used for the Right Patient. J. Antimicrob. Chemother. 2021, 76, iii2–iii3. [Google Scholar] [CrossRef]
- Cassidy, H.; van Genne, M.; Lizarazo-Forero, E.; Niesters, H.G.M.; Gard, L. Evaluation of the QIAstat-Dx RP2.0 and the BioFire FilmArray RP2.1 for the Rapid Detection of Respiratory Pathogens Including SARS-CoV-2. Front. Microbiol. 2022, 13, 854209. [Google Scholar] [CrossRef]
- Lebourgeois, S.; Storto, A.; Gout, B.; le Hingrat, Q.; Ardila Tjader, G.; del Carmen Cerdan, M.; English, A.; Pareja, J.; Love, J.; Houhou-Fidouh, N.; et al. Performance Evaluation of the QIAstat-Dx® Respiratory SARS-CoV-2 Panel. Int. J. Infect. Dis. 2021, 107, 179–181. [Google Scholar] [CrossRef]
- Yu, C.Y.; Chan, K.G.; Yean, C.Y.; Ang, G.Y. Nucleic Acid-Based Diagnostic Tests for the Detection SARS-CoV-2: An Update. Diagnostics 2021, 11, 53. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, H.; van Genne, M.; Lizarazo-Forero, E.; Gard, L.; Niesters, H.G.M. A Discussion of Syndromic Molecular Testing for Clinical Care. J. Antimicrob. Chemother. 2021, 76, iii58–iii66. [Google Scholar] [CrossRef] [PubMed]
- Popowitch, E.B.; O’Neill, S.S.; Miller, M.B. Comparison of the Biofire FilmArray RP, Genmark ESensor RVP, Luminex XTAG RVPv1, and Luminex XTAG RVP Fast Multiplex Assays for Detection of Respiratory Viruses. J. Clin. Microbiol. 2013, 51, 1528–1533. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Ruan, S.-Y.; Pan, S.-C.; Lee, T.-F.; Chien, J.-Y.; Hsueh, P.-R. Performance of a Multiplex PCR Pneumonia Panel for the Identification of Respiratory Pathogens and the Main Determinants of Resistance from the Lower Respiratory Tract Specimens of Adult Patients in Intensive Care Units. J. Microbiol. Immunol. Infect. 2019, 52, 920–928. [Google Scholar] [CrossRef]
- Murphy, C.N.; Fowler, R.; Balada-Llasat, J.M.; Carroll, A.; Stone, H.; Akerele, O.; Buchan, B.; Windham, S.; Hopp, A.; Ronen, S.; et al. Multicenter Evaluation of the BioFire FilmArray Pneumonia/Pneumonia Plus Panel for Detection and Quantification of Agents of Lower Respiratory Tract Infection. J. Clin. Microbiol. 2020, 58, e00128-20. [Google Scholar] [CrossRef]
- Buchan, B.W.; Windham, S.; Balada-Llasat, J.-M.; Leber, A.; Harrington, A.; Relich, R.; Murphy, C.; Dien Bard, J.; Naccache, S.; Ronen, S.; et al. Practical Comparison of the BioFire FilmArray Pneumonia Panel to Routine Diagnostic Methods and Potential Impact on Antimicrobial Stewardship in Adult Hospitalized Patients with Lower Respiratory Tract Infections. J. Clin. Microbiol. 2020, 58, e00135-20. [Google Scholar] [CrossRef]
- Tazi, S.; Kabbaj, H.; Zirar, J.; Zouaki, A.; el Amin, G.; el Himeur, O.; Seffar, M. Comparative Performance Evaluation of FilmArray BioFire RP2.1 and MAScIR 2.0 Assays for SARS-CoV-2 Detection. Adv. Virol. 2022, 2022, 4510900. [Google Scholar] [CrossRef]
- Dumkow, L.E.; Worden, L.J.; Rao, S.N. Syndromic Diagnostic Testing: A New Way to Approach Patient Care in the Treatment of Infectious Diseases. J. Antimicrob. Chemother. 2021, 76, iii4–iii11. [Google Scholar] [CrossRef]
- Rappo, U.; Schuetz, A.N.; Jenkins, S.G.; Calfee, D.P.; Walsh, T.J.; Wells, M.T.; Hollenberg, J.P.; Glesby, M.J. Impact of Early Detection of Respiratory Viruses by Multiplex PCR Assay on Clinical Outcomes in Adult Patients. J. Clin. Microbiol. 2016, 54, 2096–2103. [Google Scholar] [CrossRef]
- Rogers, B.B.; Shankar, P.; Jerris, R.C.; Kotzbauer, D.; Anderson, E.J.; Watson, J.R.; O’Brien, L.A.; Uwindatwa, F.; McNamara, K.; Bost, J.E. Impact of a Rapid Respiratory Panel Test on Patient Outcomes. Arch. Pathol. Lab. Med. 2015, 139, 636–641. [Google Scholar] [CrossRef]
- Srinivas, P.; Rivard, K.R.; Pallotta, A.M.; Athans, V.; Martinez, K.; Loutzenheiser, S.; Lam, S.W.; Procop, G.W.; Richter, S.S.; Neuner, E.A. Implementation of a Stewardship Initiative on Respiratory Viral PCR-based Antibiotic Deescalation. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2019, 39, 709–717. [Google Scholar] [CrossRef]
- Brendish, N.J.; Mills, S.; Ewings, S.; Clark, T.W. Impact of Point-of-Care Testing for Respiratory Viruses on Antibiotic Use in Adults with Exacerbation of Airways Disease. J. Infect. 2019, 79, 357–362. [Google Scholar] [CrossRef]
| Bacteria | Viruses | Fungi |
|---|---|---|
| Streptococcus pneumoniae Staphylococcus aureus Haemophylus influenzae type b Klebsiella pneumoniae Pseudomonas aeruginosa Chlamydophila pneumoniae Mycoplasma pneumoniae Legionella pneumophyla Chlamydia trachomatis Chlamydophila psittaci Coxiella burnetii | Influenza virus A Parainfluenza virus 1, 2, 3 Respiratory syncytial virus Adenovirus 1–7, 14, 21 SARS-CoV-1, 2 * MERS ** Epstein-Barr virus Coxsackie A virus Cytomegalovirus | Histoplasma capsulatum Coccidioides immitis Blastomyces brasiliensis Aspergillus spp. Candida spp. Cryptococcus neoformans Pneumocystis jirovecii |
| Assay | Company | Target | Time-to-Result | Type of Amplification | Reference |
|---|---|---|---|---|---|
| QIAstat-Dx Respiratory SARS-CoV-2 Panel | QIAGEN GmbH | 22 Pathogens (FLU A, FLU A subtype H1N1/2009, H1, and H3; FLU B; CoV 229E, HKU1, NL63, OC43; SARS-CoV-2; PIV 1, 2, 3, and 4; RSV A/B; h-MPV A/B; ADV; h-BocaV; RV/Enterovirus; Mycoplasma pneumoniae; Legionella pneumophila; Bordetella pertussis) | About 1 h | Multiplex real-time RT-PCR | [76,77] |
| BioCode Respiratory Pathogen Panel (RPP) | Applied BioCode, Inc. | 17 Pathogens (ADV; CoV 229E, OC43, HKU1, NL63; h-MPV A/B; FLU A, FLU A subtype H1N1/2009, H1, and H3; FLU B; PIV 1, 2, 3, and 4; RSV A/B; RV/Enterovirus; Bordetella pertussis; Chlamydia pneumoniae; Mycoplasma pneumoniae) | About 5 h | RT-PCR | [78] |
| ePlex Respiratory Pathogen Panel 2 | GenMarkDiagnostics, Inc. | 18 Pathogens (ADV; CoV 229E, OC43, HKU1, NL63; SARS-CoV-2; h-MPV A/B; FLU A, FLU A subtype H1N1/2009, H1, and H3; FLU B, PIV 1, 2, 3, and 4; RSV A, B; RV/Enterovirus; Chlamydia pneumoniae; Mycoplasma pneumoniae) | About 2 h | RT-PCR | [10,79] |
| eSensor Respiratory Viral Panel (RVP) | Clinical MicroSensors, Inc. | 14 Pathogens (FLU A, FLU A subtype H1N1/2009, H1, and H3; FLU B; RSV A, B; PIV 1, 2, and 3; h-MPV; RV; ADV species B/E; ADV species C) | About 8 h | Multiplex microarray, competitive DNA hybridization | [80] |
| FilmArray Pneumonia plus Panel | BioFireDiagnostics, LLC | 27 Pathogens and 7 resistant genes (Semi-quantitative detection: Acinetobacter calcoaceticus-baumannii complex; Enterobacter cloacae; Escherichia coli; Haemophilus influenzae; Klebsiella aerogenes; Klebsiella oxytoca; Klebsiella pneumoniae group; Moraxella catarrhalis; Proteus spp.; Pseudomonas aeruginosa; Serratia marcescens; Staphylococcus aureus; Streptococcus agalactiae; Streptococcus pneumoniae; Streptococcus pyogenes Qualitative detection: Legionella pneumophila; Mycoplasma pneumoniae; Chlamydia pneumoniae; FLU A; FLU B; ADV; CoV; PIV; RSV; RV/Enterovirus; h-MPV; Middle East Respiratory Syndrome Coronavirus Resistance: ESBL: CTX-M; Carbapenemases: KPC, NDM, Oxa48-like, VIM, IMP; Methicilin Resistance: mecA/mecC and MREJ) | About 1 h | Nested multiplex RT-PCR | [7,81,82,83] |
| FilmArray Respiratory Panel (RP) | BioFireDiagnostics, LLC | 20 Pathogens (ADV; CoV 229E, HKU1, OC43, NL63; h-MPV; RV/Enterovirus; FLU A, FLU A subtype H1N1/2009, H1, and H3; FLU B; PIV 1, 2, 3, and 4; RSV; Bordetella pertussis; Chlamydophila pneumoniae; Mycoplasma pneumoniae) | About 1 h | Nested multiplex RT-PCR | [80] |
| NxTAG Respiratory Pathogen Panel | LuminexMolecularDiagnostics, Inc. | 22 Pathogens (FLU A, FLU A subtype H1N1/2009, H1, and H3; FLU B; RSV A, B; CoV 229E, OC43, NL63, HKU1; PIV 1, 2, 3, and 4; h-MPV; ADV; h-BocaV; RV/Enterovirus; Chlamydophila pneumoniae; Mycoplasma pneumoniae; Legionella pneumophyla) | About 4 h | RT-PCR | [10,79,80] |
| xTAG Respiratory Viral Panel Fast (RVP FAST) | LuminexMolecularDiagnostics, Inc. | 18 Pathogens (FLU A, FLU A subtype H1 and H3; FLU B; RSV A, B; CoV 229E, OC43, NL63, HKU1; PIV 1, 2, 3, and 4; h-MPV; ADV; h-BocaV; RV/Enterovirus) | About 4 h | RT-PCR | [10] |
| Verigene Respiratory Pathogens Flex NucleicAcid Test (RP Flex) | Nanosphere, Inc. | 16 Pathogens (ADV; h-MPV; FLU A, FLU A subtype H1 and H3; FLU B; PIV 1, 2, 3, and 4; RV; RSV A, B; Bordetella pertussis; Bordetella parapertussis/bronchiseptica; Bordetella holmesii) | About 2 h | RT-PCR and microarray hybridization | [10,79] |
| FilmArray Respiratory Panel 2.1 (RP 2.1) | BioFireDiagnostics, LLC | 19 Pathogens (ADV; CoV 229E, HKU1, OC43, NL63; SARS-CoV-2; h-MPV; RV/Enterovirus; FLU A, FLU A subtype H1N1/2009, H1, and H3; FLU B; PIV; RSV; Bordetella pertussis; Bordetella parapertussis; Chlamydophila pneumoniae; Mycoplasma pneumoniae) | About 1 h | Nested multiplex RT-PCR | [76,84] |
| FilmArray Respiratory Panel 2.1 plus (RP2PLUS) | BioFireDiagnostics, LLC | 21 Pathogens (ADV; CoV 229E, HKU1, OC43, NL63; Middle East Respiratory Syndrome Coronavirus; SARS-CoV-2; h-MPV; RV/Enterovirus; FLU A, FLU A subtype H1N1/2009, H1, and H3; FLU B; PIV 1, 2, 3, and 4; RSV; Bordetella pertussis; Bordetella parapertussis; Chlamydophila pneumoniae; Mycoplasma pneumoniae) | About 45 min | Nested multiplex RT-PCR | [84] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calderaro, A.; Buttrini, M.; Farina, B.; Montecchini, S.; De Conto, F.; Chezzi, C. Respiratory Tract Infections and Laboratory Diagnostic Methods: A Review with A Focus on Syndromic Panel-Based Assays. Microorganisms 2022, 10, 1856. https://doi.org/10.3390/microorganisms10091856
Calderaro A, Buttrini M, Farina B, Montecchini S, De Conto F, Chezzi C. Respiratory Tract Infections and Laboratory Diagnostic Methods: A Review with A Focus on Syndromic Panel-Based Assays. Microorganisms. 2022; 10(9):1856. https://doi.org/10.3390/microorganisms10091856
Chicago/Turabian StyleCalderaro, Adriana, Mirko Buttrini, Benedetta Farina, Sara Montecchini, Flora De Conto, and Carlo Chezzi. 2022. "Respiratory Tract Infections and Laboratory Diagnostic Methods: A Review with A Focus on Syndromic Panel-Based Assays" Microorganisms 10, no. 9: 1856. https://doi.org/10.3390/microorganisms10091856
APA StyleCalderaro, A., Buttrini, M., Farina, B., Montecchini, S., De Conto, F., & Chezzi, C. (2022). Respiratory Tract Infections and Laboratory Diagnostic Methods: A Review with A Focus on Syndromic Panel-Based Assays. Microorganisms, 10(9), 1856. https://doi.org/10.3390/microorganisms10091856









