Role of Probiotics and Diet in the Management of Neurological Diseases and Mood States: A Review
Abstract
1. Introduction
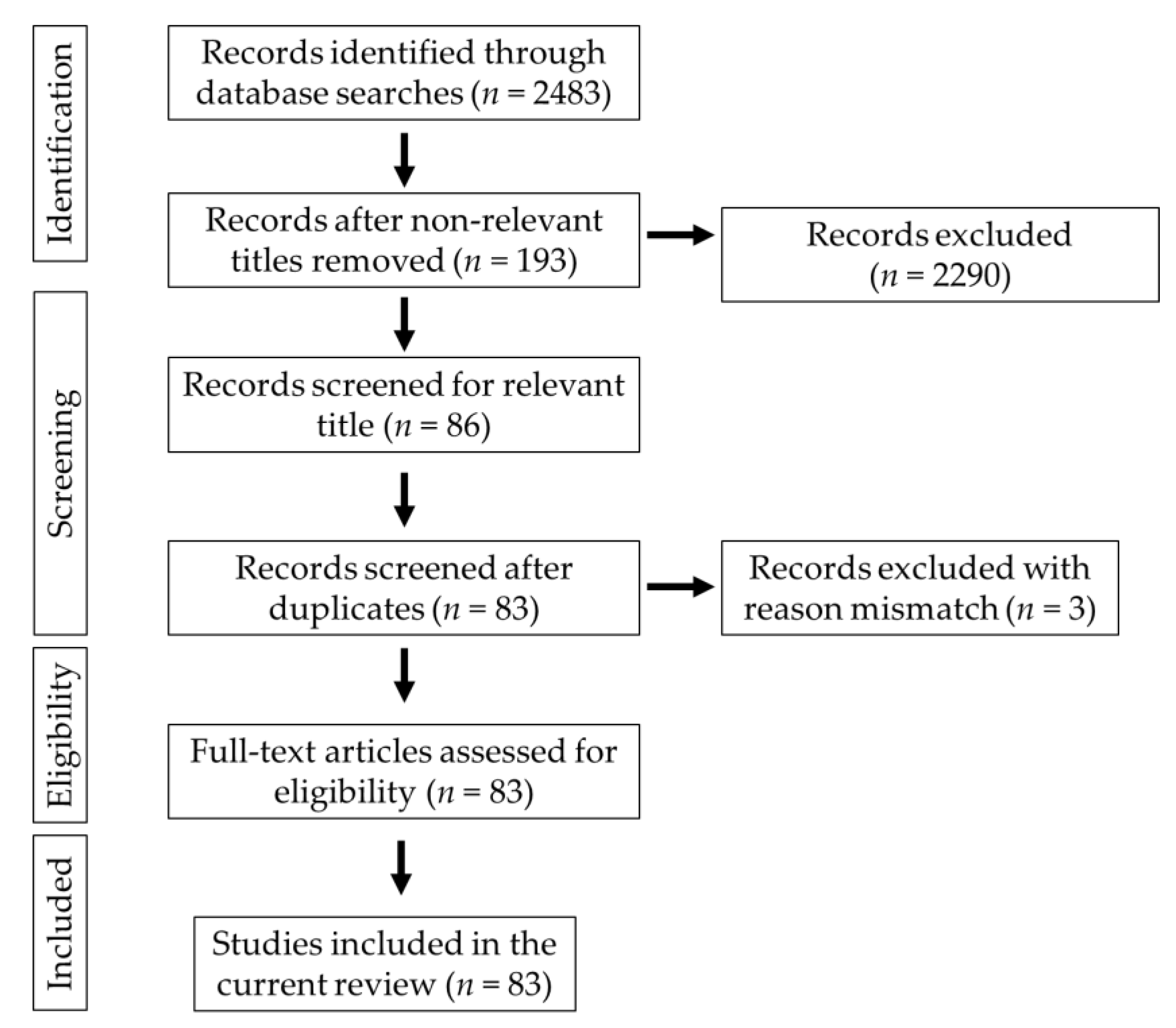
2. Methodology
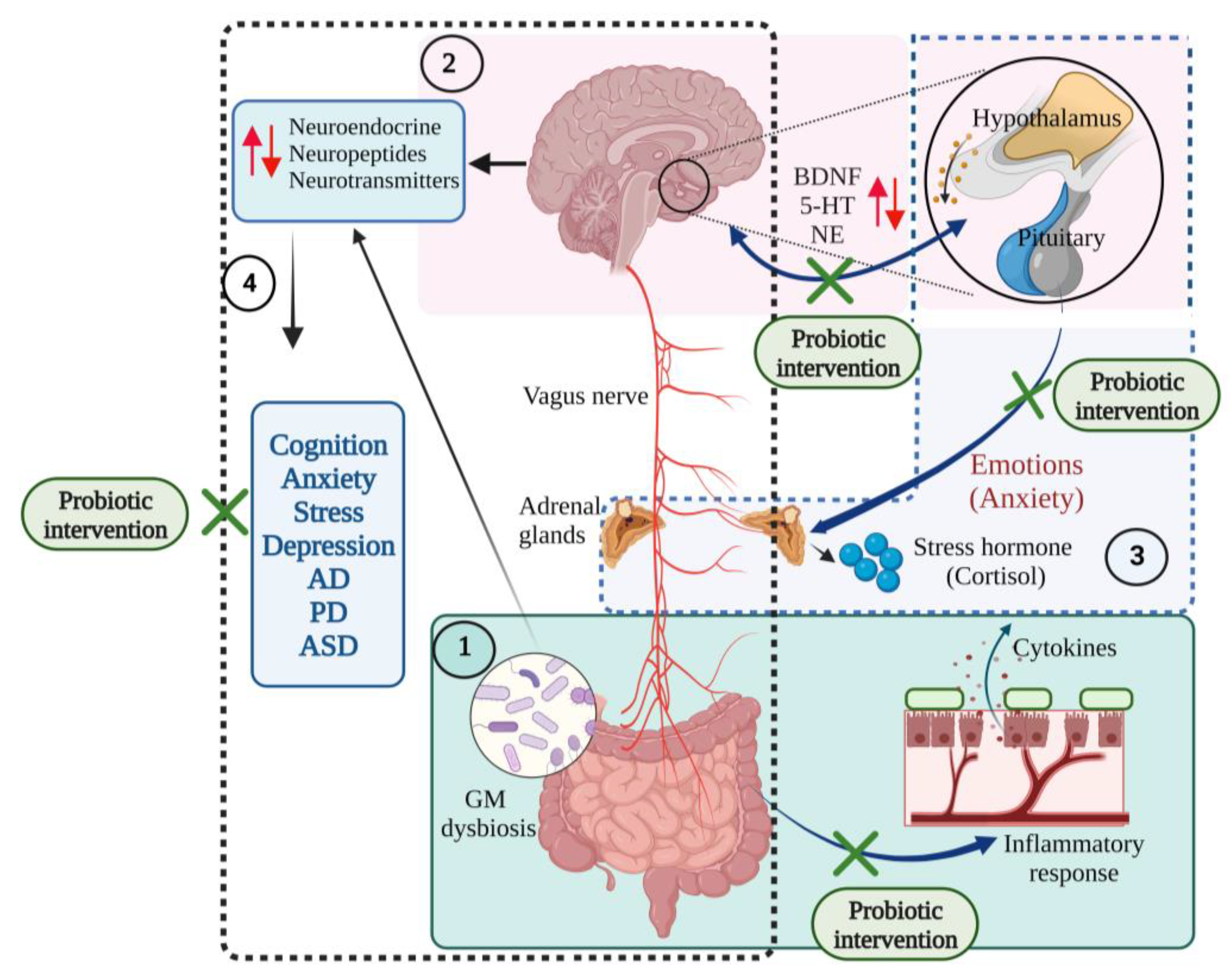
3. Probiotics and Brain Health
4. Role of Psychobiotics in Neurological Diseases, Mood States and Neurological Injuries
4.1. Autism Spectrum Disorder (ASD)
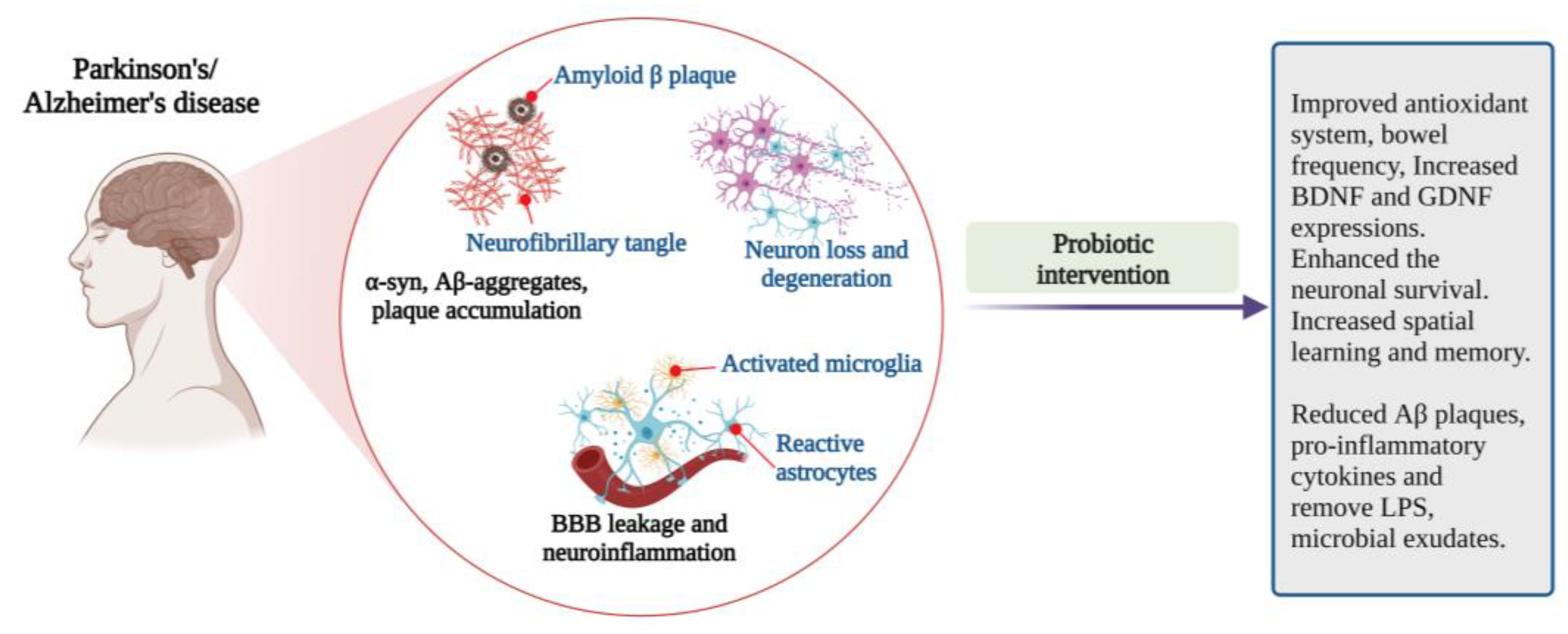
4.2. Parkinson’s Disease (PD)
4.3. Alzheimer’s Disease
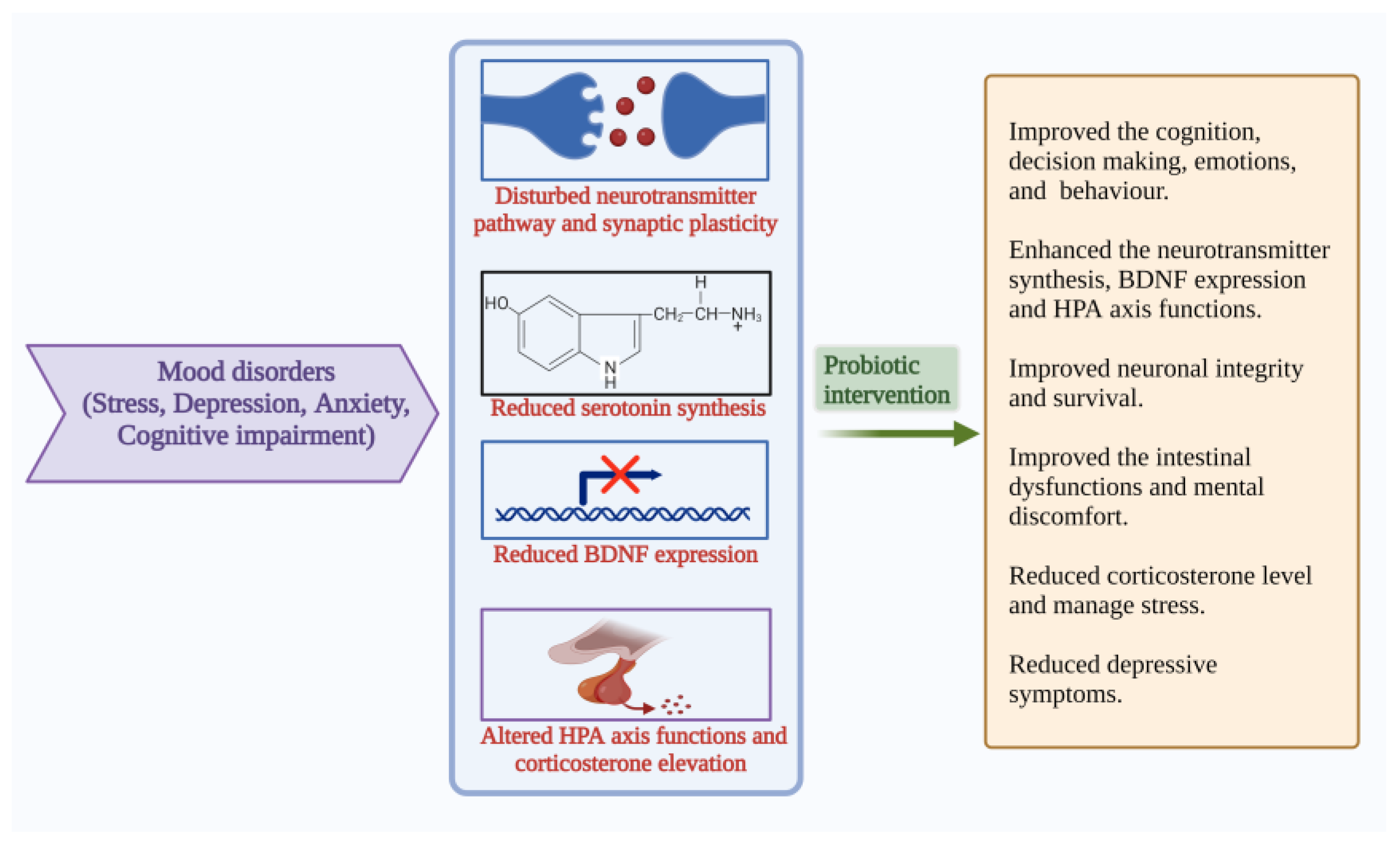
4.4. Mood States: Anxiety, Stress, Depression
4.5. Cognitive Impairment
4.6. CNS Injuries and Probiotics
5. Diet and Neuroprotection
6. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Holzapfel, W.H.; Schillinger, U. Introduction to pre- and probiotics. Food Res. Int. 2002, 35, 109–116. [Google Scholar] [CrossRef]
- Nazir, Y.; Hussain, S.A.; Abdul Hamid, A.; Song, Y. Probiotics and their potential preventive and therapeutic role for cancer, high serum cholesterol, and allergic and HIV diseases. BioMed. Res. Int. 2018, 2018, 3428437. [Google Scholar] [CrossRef] [PubMed]
- de Souza, M.; Barbalho, S.; Goulart, R.; de Carvalho, A. The current and future role of drugs and probiotics in the management of inflammatory bowel disease. JSBM 2015, 3, 76–85. [Google Scholar] [CrossRef]
- Alagiakrishnan, K.; Halverson, T. Microbial therapeutics in neurocognitive and psychiatric disorders. J. Clin. Med. Res. 2021, 13, 439–459. [Google Scholar] [CrossRef] [PubMed]
- Bear, T.L.K.; Dalziel, J.E.; Coad, J.; Roy, N.C.; Butts, C.A.; Gopal, P.K. The role of the gut microbiota in dietary interventions for depression and anxiety. Adv. Nutr. 2020, 11, 890–907. [Google Scholar] [CrossRef]
- Sarkar, A.; Lehto, S.M.; Harty, S.; Dinan, T.G.; Cryan, J.F.; Burnet, P.W.J. Psychobiotics and the manipulation of bacteria-gut-brain signals. Trends Neurosci. 2016, 39, 763–781. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Dinan, T.G.; Stanton, C.; Cryan, J.F. Psychobiotics: A novel class of psychotropic. Biol. Psychiatry 2013, 74, 720–726. [Google Scholar] [CrossRef]
- Cheng, L.H.; Liu, Y.W.; Wu, C.C.; Wang, S.; Tsai, Y.C. Psychobiotics in mental health, neurodegenerative and neurodevelopmental disorders. J. Food Drug Anal. 2019, 27, 632–648. [Google Scholar] [CrossRef]
- Sharma, R.; Gupta, D.; Mehrotra, R.; Mago, P. Psychobiotics: The next-generation probiotics for the brain. Curr. Microbiol. 2021, 78, 449–463. [Google Scholar] [CrossRef]
- Messaoudi, M.; Violle, N.; Bisson, J.F.; Desor, D.; Javelot, H.; Rougeot, C. Beneficial psychological effects of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in healthy human volunteers. Gut Microbes 2011, 2, 256–261. [Google Scholar] [CrossRef]
- Mohammadi, A.A.; Jazayeri, S.; Khosravi-Darani, K.; Solati, Z.; Mohammadpour, N.; Asemi, Z.; Adab, Z.; Djalali, M.; Tehrani-Doost, M.; Hosseini, M.; et al. The effects of probiotics on mental health and hypothalamic–pituitary–adrenal axis: A randomized, double-blind, placebo-controlled trial in petrochemical workers. Nutr. Neurosci. 2016, 19, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Logan, A.C.; Katzman, M. Major depressive disorder: Probiotics may be an adjuvant therapy. Med. Hypotheses 2005, 64, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef]
- Ait-Belgnaoui, A.; Durand, H.; Cartier, C.; Chaumaz, G.; Eutamene, H.; Ferrier, L.; Houdeau, E.; Fioramonti, J.; Bueno, L.; Theodorou, V. Prevention of gut leakiness by a probiotic treatment leads to attenuated HPA response to an acute psychological stress in rats. Psychoneuroendocrinology 2012, 37, 1885–1895. [Google Scholar] [CrossRef]
- Janik, R.; Thomason, L.A.M.; Stanisz, A.M.; Forsythe, P.; Bienenstock, J.; Stanisz, G.J. Magnetic resonance spectroscopy reveals oral Lactobacillus promotion of increases in brain GABA, N-acetyl aspartate and glutamate. Neuroimage 2016, 125, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Allen, A.P.; Hutch, W.; Borre, Y.E.; Kennedy, P.J.; Temko, A.; Boylan, G.; Murphy, E.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Bifidobacterium longum 1714 as a translational psychobiotic: Modulation of stress, electrophysiology and neurocognition in healthy volunteers. Transl. Psychiatry. 2016, 6, e939. [Google Scholar] [CrossRef] [PubMed]
- Panther, E.J.; Dodd, W.; Clark, A.; Lucke-Wold, B. Gasterointestinal microbiome and neurological injury. Biomedicines 2022, 10, 500. [Google Scholar] [CrossRef] [PubMed]
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The gut-brain axis: Interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015, 28, 203–209. [Google Scholar]
- Li, X.J.; You, X.Y.; Wang, C.Y.; Li, X.L.; Sheng, Y.Y.; Zhuang, P.W.; Zhang, Y.J. Bidirectional Brain-gut-microbiota Axis in increased intestinal permeability induced by central nervous system injury. CNS Neurosci. Ther. 2020, 26, 783–790. [Google Scholar] [CrossRef]
- Agirman, G.; Yu, K.B.; Hsiao, E.Y. Signaling inflammation across the gut-brain axis. Science 2021, 374, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Willman, J.; Willman, M.; Reddy, R.; Fusco, A.; Sriram, S.; Mehkri, Y.; Charles, J.; Goeckeritz, J.; Lucke-Wold, B. Gut microbiome and neurosurgery: Implications for treatment. Clin. Transl. Discov. 2022, 2, e139. [Google Scholar] [CrossRef] [PubMed]
- Sasselli, V.; Pachnis, V.; Burns, A.J. The enteric nervous system. Dev. Biol. 2012, 366, 64–73. [Google Scholar] [CrossRef]
- Thangaleela, S.; Sivamaruthi, B.S.; Kesika, P.; Bharathi, M.; Chaiyasut, C. Nasal microbiota, olfactory health, neurological disorders, and aging-A review. Microorganisms 2022, 10, 1405. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, J.R.; Ravel, J. The vocabulary of microbiome research: A proposal. Microbiome 2015, 3, 31. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A.; Tillisch, K.; Gupta, A. Gut/brain axis and the microbiota. J. Clin. Investig. 2015, 125, 926–938. [Google Scholar] [CrossRef]
- Bik, E.M.; Ugalde, J.A.; Cousins, J.; Goddard, A.D.; Richman, J.; Apte, Z.S. Microbial biotransformations in the human distal gut. Br. J. Pharmacol. 2018, 175, 4404–4414. [Google Scholar] [CrossRef]
- Kang, S.S.; Jeraldo, P.R.; Kurti, A.; Miller, M.E.; Cook, M.D.; Whitlock, K.; Goldenfeld, N.; Woods, J.A.; White, B.A.; Chia, N.; et al. Diet and exercise orthogonally alter the gut microbiome and reveal independent associations with anxiety and cognition. Mol. Neurodegener. 2014, 9, 36. [Google Scholar] [CrossRef]
- Lyte, M. Probiotics function mechanistically as delivery vehicles for neuroactive compounds: Microbial endocrinology in the design and use of probiotics. BioEssays 2011, 33, 574–581. [Google Scholar] [CrossRef]
- Rijkers, G.T.; Bengmark, S.; Enck, P.; Haller, D.; Herz, U.; Kalliomaki, M.; Kudo, S.; Lenoir-Wijnkoop, I.; Mercenier, A.; Myllyluoma, E.; et al. Guidance for substantiating the evidence for beneficial effects of probiotics: Current status and recommendations for future research. J. Nutr. 2010, 140, 671S–676S. [Google Scholar] [CrossRef]
- Azzalini, D.; Rebollo, I.; Tallon-Baudry, C. Visceral signals shape brain dynamics and cognition. Trends. Cogn. Sci. 2019, 23, 488–509. [Google Scholar] [CrossRef] [PubMed]
- Losifescu, D.V. The relation between mood, cognition and psychosocial functioning in psychiatric disorders. European Neuropsychopharmacol. 2012, 22 (Suppl. 3), S499–S504. [Google Scholar] [CrossRef] [PubMed]
- Battle, D.E. Diagnostic and Statistical Manual of Mental Disorders (DSM). CoDAS 2013, 25, 191–192. [Google Scholar]
- Lord, C.; Brugha, T.S.; Charman, T.; Cusack, J.; Dumas, G.; Frazier, T.; Jones, E.; Jones, R.M.; Pickles, A.; State, M.W.; et al. Autism spectrum disorder. Nat. Rev. Dis. Primers. 2020, 6, 5. [Google Scholar] [CrossRef] [PubMed]
- Campion, D.; Ponzo, P.; Alessandria, C.; Saracco, G.M.; Balzola, F. The role of microbiota in autism spectrum disorders. Minerva Gastroenterol. Dietol. 2018, 64, 333–350. [Google Scholar] [CrossRef]
- Williams, B.L.; Hornig, M.; Buie, T.; Bauman, M.L.; Cho Paik, M.; Wick, I.; Bennett, A.; Jabado, O.; Hirschberg, D.L.; Lipkin, W.I. Impaired carbohydrate digestion and transport and mucosal dysbiosis in the intestines of children with autism and gasterointestinal disturbances. PLoS ONE 2011, 6, e24585. [Google Scholar] [CrossRef]
- MacFabe, D.F. Enteric short-chain fatty acids: Microbial messengers of metabolism, mitochondria, and mind: Implications in autism spectrum disorders. Microb. Ecol. Health Dis. 2015, 26, 28177. [Google Scholar] [CrossRef] [PubMed]
- Sivamaruthi, B.S.; Suganthy, N.; Kesika, P.; Chaiyasut, C. The role of microbiome, dietary supplements, and probiotics in autism spectrum disorder. Int. J. Environ. Res. Public Health 2020, 17, 2647. [Google Scholar] [CrossRef]
- Critchfield, J.W.; Van Hemert, S.; Ash, M.; Mulder, L.; Ashwood, P. The potential role of probiotics in the management of childhood autism spectrum disorders. Gastroenterol. Res. Pract. 2011, 2011, 161358. [Google Scholar] [CrossRef]
- Liu, X.; Cao, S.; Zhang, X. Modulation of Gut Microbiota-Brain Axis by Probiotics, Prebiotics, and Diet. J. Agric. Food. Chem. 2015, 63, 7885–7895. [Google Scholar] [CrossRef]
- Do, R.; Roberts, E.; Sichel, L.S.; Sichel, J. Improvements in gastrointestinal symptoms among children with autism spectrum disorder receiving the Delpro® probiotic and immunomodulator formulation. J. Probiotics Health 2013, 1, 1–6. [Google Scholar]
- Santocchi, E.; Guiducci, L.; Prosperi, M.; Calderoni, S.; Gaggini, M.; Apicella, F.; Tancredi, R.; Billeci, L.; Mastromarino, P.; Grossi, E.; et al. Effects of probiotic supplementation on gastrointestinal, sensory and core symptoms in autism spectrum disorders: A randomized controlled trial. Front. Psychiatry 2020, 11, 550593. [Google Scholar] [CrossRef] [PubMed]
- Grossi, E.; Melli, S.; Dunca, D.; Terruzzi, V. Unexpected improvement in core autism spectrum disorder symptoms after long-term treatment with probiotics. SAGE Open Med. Case Rep. 2016, 4, 2050313X16666231. [Google Scholar] [CrossRef]
- Hsiao, E.Y.; McBride, S.W.; Hsien, S.; Sharon, G.; Hyde, E.R.; McCue, T.; Codelli, J.A.; Chow, J.; Reisman, S.E.; Petrosino, J.F.; et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell 2013, 155, 1451–1463. [Google Scholar] [CrossRef]
- Donaldson, Z.R.; Young, L.J. Oxytocin, vasopressin, and the neurogenetics of sociality. N. Y. Sci. J. 2008, 322, 900–904. [Google Scholar] [CrossRef] [PubMed]
- Sachdev, P.S.; Blacker, D.; Blazer, D.G.; Ganguli, M.; Jeste, D.V.; Paulsen, J.S.; Petersen, R.C. Classifying neurocognitive disorders: The DSM-5 approach. Nat. Rev. Neurol. 2014, 10, 634–642. [Google Scholar] [CrossRef]
- Thangaleela, S.; Sivamaruthi, B.S.; Kesika, P.; Bharathi, M.; Chaiyasut, C. Role of the gut-brain axis, gut microbial composition, diet, and probiotic intervention in Parkinson’s disease. Microorganisms 2022, 10, 1544. [Google Scholar] [CrossRef]
- Petrov, V.A.; Saltykova, I.V.; Zhukova, I.A.; Alifrova, V.M.; Zhukova, N.G.; Dorofeeva, Y.B.; Tyakht, A.V.; Kovarsky, B.A.; Alekseev, D.G.; Kostryukova, E.S.; et al. Analysis of gut microbiota in patients with Parkinson’s disease. Bull. Exp. Biol. Med. 2017, 162, 734–737. [Google Scholar] [CrossRef]
- Clapp, M.; Aurora, N.; Herrera, L.; Bhatia, M.; Wilen, E.; Wakefield, S. Gut microbiota’s effect on mental health: The gut-brain axis. Clin. Pract. 2017, 7, 987. [Google Scholar] [CrossRef]
- Georgescu, D.; Ancusa, O.E.; Georgescu, L.A.; Ionita, I.; Reisz, D. Nonmotor gastrointestinal disorders in older patients with Parkinson’s disease: Is there hope? Clin. Interv. Aging 2016, 11, 1601–1608. [Google Scholar] [CrossRef]
- Cassani, E.; Privitera, G.; Pezzoli, G.; Pusani, C.; Madio, C.; Iorio, L.; Barichella, M. Use of probiotics for the treatment of constipation in Parkinson’s disease patients. Minerva Gastroenterol. Dietol. 2011, 57, 117–121. [Google Scholar] [PubMed]
- Barichella, M.; Pacchetti, C.; Bolliri, C.; Cassani, E.; Iorio, L.; Pusani, C.; Pinelli, G.; Privitera, G.; Cesari, I.; Faierman, S.A.; et al. Probiotics and prebiotic fiber for constipation associated with Parkinson disease: An RCT. Neurology 2016, 87, 1274–1280. [Google Scholar] [CrossRef] [PubMed]
- Magistrelli, L.; Amoruso, A.; Mogna, L.; Graziano, T.; Cantello, R.; Pane, M.; Comi, C. Probiotics may have beneficial effects in Parkinson’s disease: In vitro Evidence. Front. Immunol. 2019, 10, 969. [Google Scholar] [CrossRef] [PubMed]
- Borzabadi, S.; Oryan, S.; Eidi, A.; Aghadavod, E.; Daneshvar Kakhaki, R.; Tamtaji, O.R.; Taghizadeh, M.; Asemi, Z. The effects of probiotic supplementation on gene expression related to inflammation, insulin and lipid in patients with Parkinson’s disease: A randomized, double-blind, placebo-controlled trial. Arch. Iran Med. 2018, 21, 289–295. [Google Scholar]
- Castelli, V.; d’Angelo, M.; Lombardi, F.; Alfonsetti, M.; Antonosante, A.; Catanesi, M.; Benedetti, E.; Palumbo, P.; Cifone, M.G.; Giordano, A.; et al. Effects of the probiotic formulation SLAB51 in in vitro and in vivo Parkinson’s disease models. Aging 2020, 912, 4641–4659. [Google Scholar] [CrossRef]
- Srivastav, S.; Neupane, S.; Bhurtel, S.; Katila, N.; Maharjan, S.; Choi, H. Probiotics mixture increases butyrate, and subsequently rescues the nigral dopaminergic neurons from MPTP and rotenone-induced neurotoxicity. J. Nutr. Biochem. 2019, 69, 73–86. [Google Scholar] [CrossRef]
- Goya, M.E.; Xue, F.; Sampedro-Torres-Quevedo, C.; Arnaouteli, S.; Riquelme-Dominguez, L.; Romanowski, A.; Brydon, J.; Ball, K.L.; Stanley-Wall, N.R.; Doitsidou, M. Probiotic Bacillus subtilis protects against α-synuclein aggregation in C. elegans. Cell Rep. 2020, 30, 367–380. [Google Scholar] [CrossRef]
- Sun, H.; Zhao, F.; Liu, Y.; Ma, T.; Jin, H.; Quan, K.; Leng, B.; Zhao, J.; Yuan, X.; Li, Z.; et al. Probiotics synergized with conventional regimen in managing Parkinson’s disease. npj. Park. Dis. 2022, 8, 62. [Google Scholar]
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R.; Kawas, C.H., Jr.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R.; et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers. Dement. 2011, 7, 263–269. [Google Scholar] [CrossRef]
- Bonfili, L.; Cecarini, V.; Cuccioloni, M.; Angeletti, M.; Berardi, S.; Scarpona, S.; Rossi, G.; Eleuteri, A.M. SLAB51 Probiotic formulation activates SIRT1 pathway promoting antioxidant and neuroprotective effects in an AD mouse model. Mol. Neurobiol. 2018, 55, 7987–8000. [Google Scholar] [CrossRef]
- Bloom, G.S. Amyloid-beta and tau: The trigger and bullet in Alzheimer disease pathogenesis. JAMA Neurol. 2014, 71, 505–508. [Google Scholar] [CrossRef]
- Kesika, P.; Suganthy, N.; Sivamaruthi, B.S.; Chaiyasut, C. Role of gut-brain axis, gut microbial composition, and probiotic intervention in Alzheimer’s disease. Life Sci. 2021, 264, 118627. [Google Scholar] [CrossRef]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef]
- Wu, S.C.; Cao, Z.S.; Chang, K.M.; Juang, J.L. Intestinal microbial dysbiosis aggravates the progression of Alzheimer’s disease in Drosophila. Nat. Commun. 2017, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Akbari, E.; Asemi, Z.; Daneshvar Kakhaki, R.; Bahmani, F.; Kouchaki, E.; Tamtaji, O.R.; Hamidi, G.A.; Salami, M. Effect of probiotic supplementation on cognitive function and metabolic status in Alzheimer’s disease: A randomized, double-blind and controlled trial. Front. Aging Neurosci. 2016, 8, 256. [Google Scholar] [CrossRef] [PubMed]
- Tamtaji, O.R.; Heidari-Soureshjani, R.; Mirhosseini, N.; Kouchaki, E.; Bahmani, F.; Aghadavod, E.; Tajabadi-Ebrahimi, M.; Asemi, Z. Probiotic and selenium co-supplementation, and the effects on clinical, metabolic and genetic status in Alzheimer’s disease: A randomized, double-blind, controlled trial. Clin. Nutri. 2019, 38, 2569–2575. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Y.; Wang, Y.; Xu, H.; Mei, X.; Yu, D.; Wang, Y.; Li, W. Antioxidant properties of probiotic bacteria. Nutrients 2017, 9, 521. [Google Scholar] [CrossRef] [PubMed]
- Duntas, L.H. Selenium and inflammation: Underlying anti-inflammatory mechanisms. Horm. Metab. Res. 2009, 41, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Rezaeiasl, Z.; Salami, M.; Sepehri, G. The effects of Probiotic Lactobacillus and Bifidobacterium Strains on memory and learning behavior, long-term potentiation (LTP), and some biochemical parameters in β-amyloid-induced rat’s model of Alzheimer’s disease. Prev. Nutr. Food Sci. 2019, 24, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Mehrabadi, S.; Sadr, S.S. Assessment of probiotics mixture on memory function, inflammation markers, and oxidative stress in an Alzheimer’s disease model of rats. Iran Biomed. J. 2020, 24, 220–228. [Google Scholar] [CrossRef]
- Desbonnet, L.; Garrett, L.; Clarke, G.; Bienenstock, J.; Dinan, T.G. The probiotic Bifidobacteria infantis: An assessment of potential antidepressant properties in the rat. J. Psychiatr. Res. 2008, 43, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Xu, J.; Yang, B.; Chen, K.; Kong, Y.; Fang, N.; Gong, T.; Wang, F.; Ling, Z.; Liu, J. Effect of Clostridium butyricum against microglia-mediated neuroinflammation in Alzheimer’s disease via regulating gut microbiota and metabolites butyrate. Mol. Nutr. Food Res. 2020, 64, e1900636. [Google Scholar] [CrossRef] [PubMed]
- Athari Nik Azm, S.; Djazayeri, A.; Safa, M.; Azami, K.; Ahmadvand, B.; Sabbaghziarani, F.; Sharifzadeh, M.; Vafa, M. Lactobacilli and bifidobacteria ameliorate memory and learning deficits and oxidative stress in β-amyloid (1-42) injected rats. Appl. Physiol. Nutr. Metab. 2018, 43, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Sugahara, H.; Shimada, K.; Mitsuyama, E.; Kuhara, T.; Yasuoka, A.; Kondo, T.; Abe, K.; Xiao, J.Z. Therapeutic potential of Bifidobacterium breve strain A1 for preventing cognitive impairment in Alzheimer’s disease. Sci. Rep. 2017, 7, 13510. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. What Are Anxiety Disorders? Available online: https://psychiatry.org/patients-families/anxiety-disorders/what-are-anxiety-disorders. (accessed on 7 November 2022).
- Tolentino, J.C.; Schmidt, S.L. DSM-5 criteria and Depression Severity: Implications for Clinical Practice. Front Psychiatry 2018, 9, 450. [Google Scholar] [CrossRef]
- Neufeld, K.M.; Kang, N.; Bienenstock, J.; Foster, J.A. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol. Motil. 2011, 23, 255–264. [Google Scholar] [CrossRef]
- Sivamaruthi, B.S.; Prasanth, M.I.; Kesika, P.; Chaiyasut, C. Probiotics in human mental health and diseases—A minireview. Trop. J. Pharm. Res. 2021, 18, 889–895. [Google Scholar] [CrossRef]
- Feng, Q.; Chen, W.D.; Wang, Y.D. Gut microbiota: An integral moderator in health and disease. Front. Microbiol. 2018, 9, 151. [Google Scholar] [CrossRef] [PubMed]
- Powley, T.L.; Wang, X.Y.; Fox, E.A.; Phillips, R.J.; Liu, L.W.; Huizinga, J.D. Ultrastructural evidence for communication between intramuscular vagal mechanoreceptors and interstitial cells of Cajal in the rat fundus. Neurogastroenterol. Motil. 2008, 20, 69–79. [Google Scholar] [CrossRef]
- Breit, S.; Kupferberg, A.; Rogler, G.; Hasler, G. Vagus nerve as modulator of the brain-gut axis in psychiatric and inflammatory disorders. Front. Psychiatry. 2018, 9, 44. [Google Scholar] [CrossRef]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Delgado, P.L.; Charney, D.S.; Price, L.H.; Aghajanian, G.K.; Landis, H.; Heninger, G.R. Serotonin function and the mechanism of antidepressant action: Reversal of antidepressant-induced remission by rapid depletion of plasma tryptophan. JAMA Psychiatry 1990, 47, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Sudo, N.; Chida, Y.; Aiba, Y.; Sonoda, J.; Oyama, N.; Yu, X.N.; Kubo, C.; Koga, Y. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J. Physiol. 2004, 558, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Kalynchuk, L.E.; Pinel, J.P.; Meaney, M.J. Serotonin receptor binding and mRNA expression in the hippocampus of fearful amygdala-kindled rats. Neurosci. Lett. 2006, 396, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Lesch, K.P.; Zeng, Y.; Reif, A.; Gutknecht, L. Anxiety-related traits in mice with modified genes of the serotonergic pathway. Eur. J. Pharmacol. 2003, 480, 185–204. [Google Scholar] [CrossRef]
- Kash, T.L.; Baucum, A.J., 2nd; Conrad, K.L.; Colbran, R.J.; Winder, D.G. Alcohol exposure alters NMDAR function in the bed nucleus of the stria terminalis. Neuropsychopharmacology 2009, 34, 2420–2429. [Google Scholar] [CrossRef]
- Li, Q.; Holmes, A.; Ma, L.; Van de Kar, L.D.; Garcia, F.; Murphy, D.L. Medial hypothalamic 5-hydroxytryptamine (5-HT) 1A receptors regulate neuroendocrine responses to stress and exploratory locomotor activity: Application of recombinant adenovirus containing 5-HT1A sequences. J. Neurosci. 2004, 24, 10868–10877. [Google Scholar] [CrossRef]
- Desbonnet, L.; Garrett, L.; Clarke, G.; Kiely, B.; Cryan, J.F.; Dinan, T.G. Effects of the probiotic Bifidobacterium infantis in the maternal separation model of depression. Neuroscience 2010, 170, 1179–1188. [Google Scholar] [CrossRef]
- Savignac, H.M.; Kiely, B.; Dinan, T.G.; Cryan, J.F. Bifidobacteria exert strain-specific effects on stress-related behavior and physiology in BALB/c mice. Neurogastroenterol. Motil. 2014, 26, 1615–1627. [Google Scholar] [CrossRef]
- Logan, A.C.; Venket Rao, A.; Irani, D. Chronic fatigue syndrome: Lactic acid bacteria may be of therapeutic value. Med. Hypotheses 2003, 60, 915–923. [Google Scholar] [CrossRef]
- Marcos, A.; Wärnberg, J.; Nova, E.; Gómez, S.; Alvarez, A.; Alvarez, R.; Mateos, J.A.; Cobo, J.M. The effect of milk fermented by yogurt cultures plus Lactobacillus casei DN-114001 on the immune response of subjects under academic examination stress. Eur. J. Nutr. 2004, 43, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Bergami, M.; Rimondini, R.; Santi, S.; Blum, R.; Gotz, M.; Canossa, M. Deletion of TrkB in adult progenitors alters newborn neuron integration into hippocampal circuits and increases anxiety-like behavior. Proc. Natl. Acad. Sci. USA 2008, 105, 15570–15575. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.P.; Unger, T.J.; Byrnes, J.; Rios, M. Examination of behavioral deficits triggered by targeting BDNF in fetal or postnatal brains of mice. Neuroscience 2006, 142, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Flowers, S.A.; Ellingrod, V.L. The microbiome in mental health: Potential contribution of gut microbiota in disease and pharmacotherapy management. Pharmacotherapy 2015, 35, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.R.; Ediger, J.P.; Graff, L.A.; Greenfeld, J.M.; Clara, I.; Lix, L.; Rawsthorne, P.; Miller, N.; Rogala, L.; McPhail, C.M.; et al. The Manitoba IBD cohort study: A population-based study of the prevalence of lifetime and 12- month anxiety and mood disorders. Am. J. Gastroenterol. 2008, 103, 1989–1997. [Google Scholar] [CrossRef]
- Ma, T.; Jin, H.; Kwok, L.Y.; Sun, Z.; Liong, M.T.; Zhang, H. Probiotic consumption relieved human stress and anxiety symptoms possibly via modulating the neuroactive potential of the gut microbiota. Neurobiol. Stress 2021, 14, 100294. [Google Scholar] [CrossRef]
- Pinto-Sanchez, M.I.; Hall, G.B.; Ghajar, K.; Nardelli, A.; Bolino, C.; Lau, J.T.; Martin, F.P.; Cominetti, O.; Welsh, C.; Rieder, A.; et al. Probiotic Bifidobacterium longum NCC3001 reduces depression scores and alters brain activity: A pilot study in patients with irritable bowel syndrome. Gastroenterology 2017, 153, 448–459.e8. [Google Scholar] [CrossRef]
- Akkasheh, G.; Kashani-Poor, Z.; Tajabadi-Ebrahimi, M.; Jafari, P.; Akbari, H.; Taghizadeh, M.; Memarzadeh, M.R.; Asemi, Z.; Esmaillzadeh, A. Clinical and metabolic response to probiotic administration in patients with major depressive disorder: A randomized, double-blind, placebo-controlled trial. Nutrition 2016, 32, 315–320. [Google Scholar] [CrossRef]
- Kazemi, A.; Noorbala, A.A.; Azam, K.; Eskandari, M.H.; Djafarian, K. Effect of probiotic and prebiotic vs placebo on psychological outcomes in patients with major depressive disorder: A randomized clinical trial. Clin. Nutr. 2019, 38, 522–528. [Google Scholar] [CrossRef]
- Steenbergen, L.; Sellaro, R.; van Hemert, S.; Bosch, J.A.; Colzato, L.S. A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav. Immun. 2015, 48, 258–264. [Google Scholar] [CrossRef]
- Tillisch, K.; Labus, J.; Kilpatrick, L.; Jiang, Z.; Stains, J.; Ebrat, B.; Guyonnet, D.; Legrain-Raspaud, S.; Trotin, B.; Naliboff, B.; et al. Consumption of fermented milk product with probiotic modulates brain activity. Gastroenterology 2013, 144, 1394–1401. [Google Scholar] [CrossRef]
- Goh, K.K.; Liu, Y.W.; Kuo, P.H.; Chung, Y.E.; Lu, M.L.; Chen, C.H. Effect of probiotics on depressive symptoms: A meta-analysis of human studies. Psychiatry Res. 2019, 282, 112568. [Google Scholar] [CrossRef] [PubMed]
- Gottfredson, L.S.; Deary, I.J. Intelligence predicts health and longevity, but why? Curr. Dir. Psychol. Sci. 2004, 13, 1–4. [Google Scholar] [CrossRef]
- Gale, C.R.; Batty, G.D.; Tynelius, P.; Deary, I.J.; Rasmussen, F. Intelligence in early adulthood and subsequent hospitalization for mental disorders. Epidemiology 2010, 21, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Hugo, J.; Ganguli, M. Dementia and cognitive impairment: Epidemiology, diagnosis, and treatment. Clin. Geriatr. Med. 2014, 30, 421–442. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Li, B.; Yang, W.; Ge, T.; Cui, R. Brain-immune interaction mechanisms: Implications for cognitive dysfunction in psychiatric disorders. Cell Prolif. 2022, 55, e13295. [Google Scholar] [CrossRef] [PubMed]
- Esteban-Cornejo, I.; Tejero-Gonzalez, C.M.; Sallis, J.F.; Veiga, O.L. Physical activity and cognition in adolescents: A systematic review. J. Sci. Med. Sport. 2015, 18, 534–539. [Google Scholar] [CrossRef]
- Keeley, T.J.H.; Fox, K.R. The impact of physical activity and fitness on academic achievement and cognitive performance in children. Int Rev Sport Exerc Psychol. 2009, 2, 198–214. [Google Scholar] [CrossRef]
- Bioque, M.; González-Rodríguez, A.; Garcia-Rizo, C.; Cobo, J.; Monreal, J.A.; Usall, J.; Soria, V.; PNECAT Group; Labad, J. Targeting the microbiome-gut-brain axis for improving cognition in schizophrenia and major mood disorders: A narrative review. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 105, 110130. [Google Scholar] [CrossRef]
- Van Hemert, S.; Ormel, G. Influence of the multispecies probiotic Ecologic® BARRIER on parameters of intestinal barrier function. Food Nutr. Sci. 2014, 5, 1739–1745. [Google Scholar]
- Papalini, S.; Michels, F.; Kohn, N.; Wegman, J.; van Hemert, S.; Roelofs, K.; Arias-Vasquez, A.; Aarts, E. Stress matters: Randomized controlled trial on the effect of probiotics on neurocognition. Neurobiol. Stress 2018, 10, 100141. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Kinoshita, T.; Matsumoto, A.; Yoshino, K.; Saito, I.; Xiao, J.Z. Bifidobacterium breve A1 supplementation improved cognitive decline in older adults with mild cognitive impairment: An open-label, single-arm study. J. Prev. Alzheimer’s Dis. 2019, 6, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Kuhara, T.; Oki, M.; Xiao, J.Z. Effects of Bifidobacterium breve A1 on the cognitive function of older adults with memory complaints: A randomised, double-blind, placebo-controlled trial. Benef. Microbes 2019, 10, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.W.; Liu, W.H.; Wu, C.C.; Juan, Y.C.; Wu, Y.C.; Tsai, H.P.; Wang, S.; Tsai, Y.C. Psychotropic effects of Lactobacillus plantarum PS128 in early life-stressed and naïve adult mice. Brain Res. 2016, 1631, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Buffington, S.A.; Di Prisco, G.V.; Auchtung, T.A.; Ajami, N.J.; Petrosino, J.F.; Costa-Mattioli, M. Microbial reconstitution reverses maternal diet-induced social and synaptic deficits in offspring. Cell 2016, 165, 1762–1775. [Google Scholar] [CrossRef]
- Wang, X.; Yang, J.; Zhang, H.; Yu, J.; Yao, Z. Oral probiotic administration during pregnancy prevents autism-related behaviors in offspring induced by maternal immune activation via anti-inflammation in mice. Autism Res. 2019, 12, 576–588. [Google Scholar] [CrossRef]
- El-Ansary, A.; Bacha, A.B.; Bjørklund, G.; Al-Orf, N.; Bhat, R.S.; Moubayed, N.; Abed, K. Probiotic treatment reduces the autistic-like excitation/inhibition imbalance in juvenile hamsters induced by orally administered propionic acid and clindamycin. Metab. Brain Dis. 2018, 33, 1155–1164. [Google Scholar] [CrossRef]
- Hwang, Y.H.; Park, S.; Paik, J.W.; Chae, S.W.; Kim, D.H.; Jeong, D.G.; Ha, E.; Kim, M.; Hong, G.; Park, S.H.; et al. Efficacy and safety of Lactobacillus Plantarum C29-fermented soybean (DW2009) in individuals with mild cognitive impairment: A 12-week, multi-center, randomized, double-blind, placebo-controlled clinical trial. Nutrients 2019, 11, 305. [Google Scholar] [CrossRef]
- Bermúdez-Humarán, L.G.; Salinas, E.; Ortiz, G.G.; Ramirez-Jirano, L.J.; Morales, J.A.; Bitzer-Quintero, O.K. From probiotics to psychobiotics: Live beneficial bacteria which act on the brain-gut axis. Nutrients 2019, 11, 890. [Google Scholar] [CrossRef]
- Musa, N.H.; Mani, V.; Lim, S.M.; Vidyadaran, S.; Abdul Majeed, A.B.; Ramasamy, K. Lactobacilli-fermented cow’s milk attenuated lipopolysaccharide-induced neuroinflammation and memory impairment in vitro and in vivo. J. Dairy. Res. 2017, 84, 488–495. [Google Scholar] [CrossRef]
- Parracho, H.M.R.T.; Gibson, G.R.; Knott, F.; Bosscher, D.; Kleerebezem, M.; McCartney, A.L. A double-blind, placebo-controlled, crossover-designed probiotic feeding study in children diagnosed with autistic spectrum disorders. Int. J. Probiotics Prebiotics 2010, 5, 69–74. [Google Scholar]
- Tomova, A.; Husarova, V.; Lakatosova, S.; Bakos, J.; Vlkova, B.; Babinska, K.; Ostatnikova, D. Gastrointestinal microbiota in children with autism in Slovakia. Physiol. Behav. 2015, 138, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.W.; Liong, M.T.; Chung, Y.E.; Huang, H.Y.; Peng, W.S.; Cheng, Y.F.; Lin, Y.S.; Wu, Y.Y.; Tsai, Y.C. Effects of Lactobacillus plantarum PS128 on children with autism spectrum disorder in Taiwan: A randomized, double-blind, placebo-controlled trial. Nutrients 2019, 11, 820. [Google Scholar] [CrossRef]
- Ibrahim, A.; Ali, R.; Manaf, M.; Ahmad, N.; Tajurruddin, F.W.; Qin, W.Z.; Desa, S.; Ibrahim, N.M. Multi-strain probiotics (Hexbio) containing MCP BCMC strains improved constipation and gut motility in Parkinson’s disease: A randomised controlled trial. PLoS ONE 2020, 15, e0244680. [Google Scholar] [CrossRef] [PubMed]
- Nimgampalle, M.; Kuna, Y. Anti-Alzheimer Properties of Probiotic, Lactobacillus plantarum MTCC 1325 in Alzheimer’s Disease induced Albino Rats. J. Clin. Diagn. Res. 2017, 11, KC01–KC05. [Google Scholar] [CrossRef]
- Diop, L.; Guillou, S.; Durand, H. Probiotic food supplement reduces stress-induced gastrointestinal symptoms in volunteers: A double-blind, placebo-controlled, randomized trial. Nutr. Res. 2008, 28, 1–5. [Google Scholar] [CrossRef]
- Gruenwald, J.; Graubaum, H.J.; Harde, A. Effect of a probiotic multivitamin compound on stress and exhaustion. Adv. Ther. 2002, 19, 141–150. [Google Scholar] [CrossRef]
- Talbott, S.M.; Stephens, B.J.; Talbott, J.A.; Oddou, M.P. Effect of coordinated probiotic/prebiotic/phytobiotic supplementation on microbiome balance and psychological mood state in healthy stressed adults. FASEB J. 2019, 32, 533.85. [Google Scholar] [CrossRef]
- Chahwan, B.; Kwan, S.; Isik, A.; van Hemert, S.; Burke, C.; Roberts, L. Gut feelings: A randomised, triple-blind, placebo-controlled trial of probiotics for depressive symptoms. J. Affect. Disord. 2019, 253, 317–326. [Google Scholar] [CrossRef]
- Cornelius, C.; Crupi, R.; Calabrese, V.; Graziano, A.; Pennisi, G.; Radak, Z.; Calabrese, E.J.; Cuzzocrea, S. Traumatic brain injury: Oxidative stress and neuroprotection. Antioxid. Redox Signal. 2013, 19, 836–853. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Z.; Peng, J.; garner, S.Y.; Yin, S.; Jiang, Y. Gut microbiota-brain interaction: An emerging immunotherapy for traumatic brain injury. Exp. Neurol. 2021, 18, 113585. [Google Scholar] [CrossRef] [PubMed]
- Pavelescu, D.; Mirea, L.; Grintescu, I. Could selected probiotics have beneficial effects on clinical outcome of severe traumatic brain injury patients? Crit. Care. 2014, 18, P472. [Google Scholar] [CrossRef]
- Cavaleri, F.; Bashar, E. Potential synergies of β-hydroxybutyrate and butyrate on the modulation of metabolism, inflammation, cognition and general health. J. Nutr. Metab. 2018, 2018, 7195760. [Google Scholar] [CrossRef]
- Sun, J.; Wang, F.; Ling, Z.; Yu, X.; Chen, W.; Li, H.; Jin, J.; Pang, M.; Zhang, H.; Yu, J.; et al. Clostridium butyricum attenuates cerebral ischemia/reperfusion injury in diabetic mice via modulation of gut microbiota. Brain Res. 2016, 1042, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Sivamaruthi, B.S.; Kesika, P.; Chaiyasut, C. Impact of fermented foods on human cognitive function-A review of outcome of clinical trials. Sci. Pharm. 2018, 86, 22. [Google Scholar] [CrossRef]
- Horrocks, L.A.; Yeo, Y.K. Health benefits of docosahexaenoic acid (DHA). Pharmacol. Res. 1999, 40, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Hills, R.D., Jr.; Pontefract, B.A.; Mishcon, H.R.; Black, C.A.; Sutton, S.C.; Theberge, C.R. Gut Microbiome: Profound Implications for Diet and Disease. Nutrients 2019, 11, 1613. [Google Scholar] [CrossRef]
- Wikoff, W.R.; Anfora, A.T.; Liu, J.; Schultz, P.G.; Lesley, S.A.; Peters, E.C.; Siuzdak, G. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc. Natl. Acad. Sci. USA 2009, 106, 3698–3703. [Google Scholar] [CrossRef]
- Myers, S.P.; Hawrelak, J. The causes of intestinal dysbiosis: A review. Altern. Med. Rev. 2004, 9, 180–197. [Google Scholar]
- Lynch, S.V.; Pedersen, O. The Human Intestinal Microbiome in Health and Disease. N. Engl. J. Med. 2016, 375, 2369–2379. [Google Scholar] [CrossRef]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Pinilla, F. Brain foods: The effects of nutrients on brain function. Nat. Rev. Neurosci. 2008, 9, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P. Dietary factors, hormesis and health. Ageing Res. Rev. 2007, 7, 43–48. [Google Scholar] [CrossRef]
- Greenwood, C.E.; Winocur, G. High-fat diets, insulin resistance and declining cognitive function. Neurobiol. Aging 2005, 26 (Suppl. 1), 42–45. [Google Scholar] [CrossRef]
- Molteni, R.; Barnard, J.R.; Ying, Z.; Roberts, C.K.; Gomez-Pinilla, F. A high-fat, refined sugar diet reduces hippocampal brain-derived neurotrophic factor, neuronal plasticity, and learning. Neuroscience 2002, 112, 803–814. [Google Scholar] [CrossRef]
- Moriguchi, T.; Greiner, R.S.; Salem, N., Jr. Behavioral deficits associated with dietary induction of decreased brain docosahexaenoic acid concentration. J. Neurochem. 2000, 75, 2563–2573. [Google Scholar] [CrossRef] [PubMed]
- Horrobin, D.F. Schizophrenia: The illness that made us human. Med. Hypotheses 1998, 50, 269–288. [Google Scholar] [CrossRef]
- Freeman, M.P.; Hibbeln, J.R.; Wisner, K.L.; Davis, J.M.; Mischoulon, D.; Peet, M.; Keck, P.E., Jr.; Marangell, L.B.; Richardson, A.J.; Lake, J.; et al. Omega-3 fatty acids: Evidence basis for treatment and future research in psychiatry. J. Clin. Psychiatry. 2006, 67, 1954–1967. [Google Scholar] [CrossRef]
- Lin, P.Y.; Chiu, C.C.; Huang, S.Y.; Su, K.P. A Meta-analytic review of polyunsaturated fatty acid compositions in dementia. J. Clin. Psychiatry 2012, 73, 1245–1254. [Google Scholar] [CrossRef]
- McCann, J.C.; Ames, B.N. Is docosahexaenoic acid, an n-3 long-chain polyunsaturated fatty acid, required for development of normal brain function? An overview of evidence from cognitive and behavioral tests in humans and animals. Am. J. Clin. Nutr. 2005, 82, 281–295. [Google Scholar] [CrossRef]
- Wu, A.; Ying, Z.; Gomez-Pinilla, F. Omega-3 fatty acids supplementation restores mechanisms that maintain brain homeostasis in traumatic brain injury. J. Neurotrauma 2007, 24, 1587–1595. [Google Scholar] [CrossRef] [PubMed]
- Theodore, L.E.; Kellow, N.J.; McNeil, E.A.; Close, E.O.; Coad, E.G.; Cardoso, B.R. Nut consumption for cognitive performance: A systematic review. Adv. Nutr. 2021, 12, 777–792. [Google Scholar] [CrossRef]
- Zhu, R.Z.; Chen, M.Q.; Zhang, Z.W.; Wu, T.Y.; Zhao, W.H. Dietary fatty acids and risk for Alzheimer’s disease, dementia, and mild cognitive impairment: A prospective cohort meta-analysis. Nutrition 2021, 90, 111355. [Google Scholar] [CrossRef] [PubMed]
- Sydenham, E.; Dangour, A.D.; Lim, W.S. Omega 3 fatty acid for the prevention of cognitive decline and dementia. Cochrane Database Syst. Rev. 2012, 6, CD005379. [Google Scholar]
- Martinez-Lapiscina, E.H.; Clavero, P.; Toledo, E.; San Julian, B.; Sanchez-Tainta, A.; Corella, D.; Lamuela-Raventos, R.M.; Martinez, J.A.; Martinez-Gonzalez, M.A. Virgin olive oil supplementation and long-term cognition: The PREDIMED-NAVARRA randomized, trial. J. Nutr. Health Aging 2013, 17, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Schaffer, S.; Asseburg, H.; Kuntz, S.; Muller, W.E.; Eckert, G.P. Effects of polyphenols on brain ageing and Alzheimer’s disease: Focus on mitochondria. Mol. Neurobiol. 2012, 46, 161–178. [Google Scholar] [CrossRef] [PubMed]
- Pu, F.; Mishima, K.; Irie, K.; Motohashi, K.; Tanaka, Y.; Orito, K.; Egawa, T.; Kitamura, Y.; Egashira, N.; Iwasaki, K.; et al. Neuroprotective effects of quercetin and rutin on spatial memory impairment in an 8-arm radial maze task and neuronal death induced by repeated cerebral ischemia in rats. J. Pharmacol. Sci. 2007, 104, 329–334. [Google Scholar] [CrossRef] [PubMed]
- van Praag, H.; Lucero, M.J.; Yeo, G.W.; Stecker, K.; Heivand, N.; Zhao, C.; Yip, E.; Afanador, M.; Schroeter, H.; Hammerstone, J.; et al. Plant-derived flavanol (−) epicatechin enhances angiogenesis and retention of spatial memory in mice. J. Neurosci. 2007, 27, 5869–5878. [Google Scholar] [CrossRef]
- Sawaengsri, H.; Wang, J.; Reginaldo, C.; Steluti, J.; Wu, D.; Meydani, S.N.; Selhub, J.; Paul, L. High folic acid intake reduces natural killer cell cytotoxicity in aged mice. J. Nutr. Biochem. 2016, 30, 102–107. [Google Scholar] [CrossRef]
- Politis, A.; Olgiati, P.; Malitas, P.; Albani, D.; Signorini, A.; Polito, L.; De Mauro, S.; Zisaki, A.; Piperi, C.; Stamouli, E.; et al. Vitamin B12 levels in Alzheimer’s disease: Association with clinical features and cytokine production. J. Alzheimer’s Dis. 2010, 19, 481–488. [Google Scholar] [CrossRef]
- Navarro, A.; Gómez, C.; Sánchez-Pino, M.J.; González, H.; Bández, M.J.; Boveris, A.D.; Boveris, A. Vitamin E at high doses improves survival, neurological performance, and brain mitochondrial function in aging male mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 289, R1392–R1399. [Google Scholar] [CrossRef]
- Cordero, K.; Coronel, G.G.; Serrano-Illán, M.; Cruz-Bracero, J.; Figueroa, J.D.; De León, M. Effects of dietary vitamin e supplementation in bladder function and spasticity during spinal cord injury. Brain Sci. 2018, 8, 38. [Google Scholar] [CrossRef]
- Schirinzi, T.; Martella, G.; Imbriani, P.; Di Lazzaro, G.; Franco, D.; Colona, V.L.; Alwardat, M.; Sinibaldi Salimei, P.; Mercuri, N.B.; Pierantozzi, M.; et al. Dietary vitamin e as a protective factor for Parkinson’s Disease: Clinical and experimental evidence. Front. Neurol. 2019, 10, 148. [Google Scholar] [CrossRef] [PubMed]
- Malar, D.S.; Prasanth, M.I.; Brimson, J.M.; Sharika, R.; Sivamaruthi, B.S.; Chaiyasut, C.; Tencomnao, T. Neuroprotective properties of green tea (Camellia sinensis) in Parkinson’s disease: A review. Molecules 2020, 25, 3926. [Google Scholar] [CrossRef]
- Wang, J.; Ho, L.; Zhao, W.; Ono, K.; Rosensweig, C.; Chen, L.; Humala, N.; Teplow, D.B.; Pasinetti, G.M. Grape-derived polyphenolics prevent Abeta oligomerization and attenuate cognitive deterioration in a mouse model of Alzheimer’s disease. J. Neurosci. 2008, 28, 6388–6392. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Ho, L.; Faith, J.; Ono, K.; Janle, E.M.; Lachcik, P.J.; Cooper, B.R.; Jannasch, A.H.; D’Arcy, B.R.; Williams, B.A.; et al. Role of intestinal microbiota in the generation of polyphenol-derived phenolic acid mediated attenuation of Alzheimer’s disease β-amyloid oligomerization. Mol. Nutr. Food Res. 2015, 59, 1025–1040. [Google Scholar] [CrossRef]
- Liu, J. The effects and mechanisms of mitochondrial nutrient α-lipoic acid on improving age-associated mitochondrial and cognitive dysfunction: An overview. Neurochem. Res. 2008, 33, 194–203. [Google Scholar] [CrossRef]
- Holmquist, L.; Stuchbury, G.; Berbaum, K.; Muscat, S.; Young, S.; Hager, K.; Engel, J.; Münch, G. Lipoic acid as a novel treatment for Alzheimer’s disease and related dementias. Pharmacol. Ther. 2007, 113, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, E.; Miller, M.; Yaxley, A.; Isenring, E. Malnutrition in the elderly: A narrative review. Maturitas 2013, 76, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Corish, C.A.; Bardon, L.A. Malnutrition in older adults: Screening and determinants. Proc. Nutr. Soc. 2019, 78, 372–379. [Google Scholar] [CrossRef]
- Feng, L.; Chu, Z.; Quan, X.; Zhang, Y.; Yuan, W.; Yao, Y.; Zhao, Y.; Fu, S. Malnutrition is positively associated with cognitive decline in centenarians and oldest-old adults: A cross-sectional study. E Clin. Med. 2022, 47, 101336. [Google Scholar] [CrossRef] [PubMed]


| Probiotic Strain | Study Model | Age | No. of Subjects | Dose and Duration | Results | Ref. |
|---|---|---|---|---|---|---|
| Cognition | ||||||
| Probiotic sticks (B. longum 1714) | Healthy volunteers | 25.5 mean years | 22 | The probiotic stick contained maltodextrin, magnesium stearate and 1 × 109 CFU of probiotics per stick; 1 stick per day for 4 weeks | Improved cognitive performance. Improved visuospatial memory performance and anti-stress, precognitive effects | [17] |
| Ecologic® Barrier (B. bifidum W23, B. lactis W52, L. acidophilus W37, L. brevis W63, L. casei W56, L. salivarius W24, L. lactis W16, and L. lactis W58). | Healthy female volunteers | 18 to 40 years | 61 | 5 × 109 CFU per day for 4 weeks. | Improved cognitive response and working memory performance | [112] |
| B. breve A1 | Patients with MCI | - | 27 | 2.0 × 1010 CFU per day for 24 weeks | Improved cognitive function | [113] |
| B. breve A1 | Older adults with memory complaints | 50 to 80 years | 121 | 2.0 × 1010 CFU per day for 12 weeks | Improved cognitive function | [114] |
| B. bifidum, B. infantis, L. helveticus, fructooligosaccharides and maltodextrin. | C57BL/6J mice | 21 days * | - | Bifidobacterium (1.9 × 108 CFU/g); Lactobacillus (6.4 × 109 CFU/g) were administered with drinking water to female mice from embryonic day 0.5 to postnatal day 21. | Decreased the incidence of ASD Prevented a series of MIA-induced ASD-relevant deficits. Reduced impairments in social behaviour, repetitive behaviour, stereotyped behaviour, depression, and anxiety-like behaviour. Prevented the decrease in PV+ neurons and GABA levels | [117] |
| ProtexinR restore (L. casei, L. rhamnosus, S. thermophilus, B. breve, L. acidophilus, B. infantis, L. bulgaricus) | Young male golden Syrian hamsters | - | 50 | 2 × 108 CFU/Kg body weight dissolved in PBS; once a day for 27 days | Ameliorated the glutamate excitotoxicity | [118] |
| L. plantarum C29 mediated fermented soybean (DW2009) | Physically healthy men and women with MCI | 55 to 85 years | 100 | 1.25 × 1010 CFU/g per day for 12 weeks | Enhanced cognitive function. Increased BDNF levels Attenuated the memory impairment. | [119] |
| L. plantarum (LAB1, LAB11, LAB12), L. fermentum (LAB9, LAB10), and L. casei (LABPC) | Male ICR mice and BV2 microglia cell lines | 2 months | 30 | L. fermentum (109 CFU) and L. casei (109 CFU) per day for 28 days | Attenuated the LPS-induced memory deficit. Increased the antioxidants SOD, GSH, and GPx. Reduced the neuroinflammation by decreasing MDA, AChE, and pro-inflammatory cytokines | [121] |
| Autism spectrum disorder | ||||||
| Delpro® (L. acidophilus, L. casei, L. delbrueckii, B. longum and B. bifidum) with lyophilized peptidoglycan, muramyl peptides and DNA motifs derived from L. rhamnosus V strain | Children with ASD and frequent GI distress | 3 to 16 years | 33 | 2 billion CFU of each strain/ thrice a day for 21 days | Improved GI symptoms and autism treatment evaluation domains such as speech, communication, sociability, sensory awareness, physical behaviour, and health | [41] |
| DSF Vivomixx® (S. thermophilus, B. breve, B. longum, B. infantis, L. acidophilus, L. plantarum, L. paracasei, L. delbrueckii subsp. bulgaricus) | ASD children | 18 to 72 months | 85 | 450 billion/ packet. Two packets/day in the first month; 1 packet/day in the next 5 months | Showed positive effects on core autism symptoms. Significantly improved GI symptoms, multisensory processing, and adaptive functioning. | [42] |
| VSL#3 (L. delbrueckii subsp. Bulgaricus, L. acidophilus, B. breve, B. longum, B. infantis, L. paracasei, L. plantarum, S. thermophiles) | ASD children | 12 years | - | 9 × 1010 CFU/g of B. breve, B. longum, B. infantis; 8 × 1010 CFU/g of L. acidophilus, L. plantarum, L. paracasei, L. bulgaricus, L. delbrueckii subsp.; 20 × 1010 CFU/g of S. thermophilus and S. salivaricus/ day for 4 weeks | Reduced the severity of abdominal symptoms. Autistic core symptoms improved. The score of the social affect domain improved | [43] |
| Lactobacillus plantarum WCSF1 | ASD children | 4 to 16 years | - | 4.5 × 1010 CFU per day for 12 weeks | Increased the abundance of Enterococci and Lactobacilli. Decreased the abundance of Clostridium cluster XIVa. Improved stool consistency and behavioural scores | [122] |
| A mixture of 3 Lactobacillus strains, 2 Bifidobacterium strains, and one Streptococcus strain (60: 25: 15 ratio) | ASD children and their non-autistic siblings and other healthy children as control | ASD (2 to 9 years) Siblings (5 to 7 years) Control (2 to 11 years) | ASD (n = 10); Siblings (n = 9); Control (n = 10) | 4 months | Probiotic supplementation normalized Bacteroidetes/Firmicutes ratio. Decreased the abundance of Desulfovibrio spp. and decreased TNFα level in feces | [123] |
| L. plantarum PS128 | ASD boys | 7 to 15 years | 80 | 3 × 1010 CFU per day for 4 weeks | Ameliorated defiance behaviours. Improved the Swanson, Nolan, Pelham-IV-Taiwan version (SNAP-IV) scores | [124] |
| Parkinson’s disease | ||||||
| L. acidophilus, B. infantis | PD patients with GI-NMS | 76.05 ± 2.09 years | 120 | 3 months | Improved abdominal pain and bloating compared to the drug trimebutine. | [50] |
| L. casei Shirota | PD patients with chronic constipation | - | 40 | 65 mL of fermented milk containing 6.5 × 109 CFU/ day for 6 weeks. | Improved stool consistency and bowel habits | [51] |
| Fermented milk with prebiotic fibre (L. rhamnosus GG, L. acidophilus, L. plantarum, L. paracasei, L. delbrueckii subsp. Bulgaricus, Bifidobacterium, prebiotic fibre) | PD patients with ROME III criteria constipation | Test: 71.8 ± 7.7 years; placebo: 69.5 ± 10.3 years | 120 | 125 mL fermented milk containing 250 × 109 CFU/day for 4 weeks | Improved frequency of bowel movements | [52] |
| L. salivarius LS01, L. plantarum LP01, L. acidophilus LA02, L. rhamnosus LR06, B. breve BR03, B. animalis subsp. lactis BS01 | PBMCs (isolated from PD patients) | 70 ± 8 years | - | 1 × 106 cells/plate of PBMCs treated with probiotic strains in a 1:1 ratio for 24 h | Decreased the pro-inflammatory cytokines (TNF-α, IL-17A, and IL-6) and oxidative stress levels. Reduced the growth of pathogens such as E. coli, Klebsiella pneumoniae | [53] |
| L. acidophilus, B. bifidum, L. reuteri, L. fermentum | PD patients | 50 to 80 years | 50 | 2 × 109 CFU per strain; 8 × 109 CFU per capsule; one capsule per day for 12 weeks. | Improved expression of IL-1, IL-8, TNF-α, TGF-β, and PPAR-γ. No changes were observed in inflammation and oxidative stress marker | [54] |
| S. thermophilus DSM 32245, B. lactis DSM 32246, B. lactis DSM 32247, L. acidophilus DSM 32241, L. helviticus DSM 32242, L. paracasei DSN 32243, L. plantarum DSM 32244, L. brevis DSM 27961 | Male C57BL/6 mice, SH-SY5Y cell line. | 9 weeks | 30 | One sachet containing 200 billion bacteria dissolved in 10 mL of drinking water. Mice received 270 µL using oral gavage daily for 2 weeks | SLAB51® was able to counteract the detrimental effect of 6-OHDA. Improved anti-inflammatory and antioxidant activities. Protected dopaminergic neurons and improved behavioural impairments. | [55] |
| The commercial probiotic mixture contained L. rhamnosus GG, B. animalis lactis, L. acidophilus | MPTP-induced male C57BL/6 mice PD model | 7 weeks old | - | 2 × 106 CFU per day for 30 days | Induced the butyrate synthesis and increased the BDNF and GDNF levels Enhanced the survival and proliferation of dopaminergic neurons. Reduced the expression of monoamine oxidase B expression and increased the expression of neurotrophic factors. | [56] |
| Hexbio® (L. acidophilus, L. casei, L. lactis, B. infantis, B. longum) ProbioM8 (Bifidobacterium animalis subsp. lactis) | PD patients with ROME III criteria constipation PD patients | 50 to 80 years 69.41 ± 6.05 years | 48 Probiotic (n = 50) Probiotic + conventional drug (n = 50) | 107 mg of each strain (30 × 109 CFU), 2% FOS and lactose/ twice daily for 8 weeks 2G Probio M8 contains 3 × 1010 CFU/day; maltodextrin as an excipient, once daily for 3 months. | Improved bowel opening frequency and whole gut transit time Reduced anxiety and depression. Regulate gut lipid metabolism, SCFAs, and neurotransmitters. Increased dopaminergic synthesis | [125] [58] |
| Alzheimer’s disease | ||||||
| L. acidophilus, L. casei, L. fermentum, B. bifidum, | AD patients | 60 to 95 years | 60 | 200 mL probiotic milk containing 2 × 109 CFU/g of each strain per day for 12 weeks. | Improved MMSE score. Decreased MDA and serum hs-CRP | [65] |
| L. acidophilus, B. bifidum, B. longum with selenium | AD patients | 55 to 100 years | 79 | 200 µg selenium and probiotic containing 2 × 109 CFU of each strain per day for 12 weeks | Improved cognitive function. Reduced serum triglycerides, LDL, total-/HDL cholesterol, hs-CRP | [66] |
| Iranian Prodigest Capsule composed of L. acidophilus, B. bifidum and B. longum | Aβ injected Male Sprague-Dawley rats. | - | 40 | 15 × 109 in 1 mL drinking water for 4 weeks and given via ICV for 4 weeks | Improved learning. Increased paired-pulse facilitation ratios. Reduced serum triglycerides and very LDL | [69] |
| L. reuteri, L. rhamnosus, B. infantis | Aβ injected male Wistar rats. | - | 50 | 1 × 1010 CFU per day for 10 weeks | Significantly improved spatial memory Reduced Aβ plaques in AD rats. Reduced inflammatory markers IL-1β and TNF-α, and oxidative stress in AD rats. | [70] |
| Clostridium butyricum WZMC1016 | APPswe/PS1dE9 Tg mice | 2 months | 20 | 1 × 109 CFU per day for 4 weeks | Attenuated microglia-mediated neuroinflammation | [72] |
| L. acidophilus (1688FL431-16LA02), L. fermentum (ME3), B. lactis (1195SL609-16BS01), B. longum (1152SL593-16BL03) | Aβ injected rats | - | 60 | 1 × 1010 CFU each strain per day in drinking water (30 mL) every morning. | Improved memory deficit and inhibited AD pathology by modifying microbiota | [73] |
| B. breve A1 | Aβ injected mice | 10 weeks | 1× 109 CFU | Improved cognitive function and ameliorated behaviour deficits. Suppressed the immune reactive and inflammation genes in the hippocampus. Significantly enhanced acetate level | [74] | |
| L. plantarum MTCC1325 | D-Galactose-induced AD albino rats | - | 48 | D-Galactose and probiotics 12 × 108 CFU/mL; 10 mL/kg body weight per day for 60 days | Ameliorated cognitive deficits and restored Ach Produced anti-Alzheimer properties against D-Galactose-induced AD rats. Restored spatial memory impairment | [126] |
| Depression, anxiety and stress | ||||||
| B. longum 1714, and B. breve 1205 | Innately anxious BALB/c mice | 7 weeks | - | Probiotics reconstituted in PBS; 1 × 109 CFU per day for 6 weeks | Reduced anxiety, stress and depression-related behaviours and weight loss in the anxious mouse Reduced stress-induced hyperthermia | [90] |
| Fermented milk containing Actimel® (L. delbrueckii subsp. bulgaricus, S. salivarius subsp. thermophilus, L. casei DN114001) | Healthy students under examination stress | 18 to 23 years | 155 | 200 mL fermented milk containing 1 × 107/mL CFU L. delbrueckii subsp. bulgaricus, 1 × 108 CFU/mL S. salivarius subsp. thermophilus, and L. casei DN114001 per day for 6 weeks | Significantly increased circulating lymphocytes and CD 56 immune cells | [92] |
| L. plantarum P-8 | Stressed adults | - | Probiotic (n = 43) Placebo (n = 36) | 2 × 1010 CFU per day for 12 weeks | Improved the production of neurotransmitters and neuroactive metabolites. Modulates the gut microbes. Reduced stress and anxiolytic effects | [97] |
| B. longum NCC3001 | IBS patients with mild to moderate anxiety and depression scores | - | 44 | Probiotic sachet containing 1.0 × 1010 CFU/g and maltodextrin dissolved in 100–200 mL lactose-free milk per day for 6 weeks | Decreased HAD scores and depression levels significantly. Reduced responses to negative stimuli | [98] |
| Probiotic capsule containing L. acidophilus, L. casei and B. longum | Patients with MDD | 20 to 55 years | 40 | 2 × 109 CFU per day for 8 weeks | Decreased Beck’s depression scores significantly Significantly raised plasma total glutathione levels | [99] |
| L. acidophilus Rosell-52, B. longum Rosell-175 | Healthy human volunteers with symptoms of stress | 18 to 60 years | 75 | 3 × 109 CFU each strain per day for 3 weeks | No significant changes were observed in psychological symptoms and sleep problems. Reduced stress-induced GI symptoms such as abdominal pain, nausea, and vomiting | [127] |
| A multivitamin capsule containing probiotics L. acidophilus, B. bifidum, B. longum. | Adult men and women suffered from stress and exhaustion. | - | 42 | 1 g capsule containing multivitamins and 10 million strains per day for 6 months | Improved immune health and GI health | [128] |
| Synbiotics supplement contained L. helveticus R0052, B. longum R0175, L. rhamnosus R0011, galactooligosaccharide and other nutrients. | Healthy stressed individuals | - | 32 | For 4 weeks | Increased beneficial bacteria load Lactobacillus and Bifidobacterium. Increased the psychological indices significantly for both positive and negative mood states | [129] |
| Ecologic ® Barrier containing B. bifidum W23, B. lactis W51, W52, L. acidophilus W37, L. brevis W63, L. casei W56, L. salivarius W24, L. lactis W19 and L. lactis W58. | Participants with mild to severe depression | 18 and above years | 71 | 1 × 1010 CFU per day for 8 weeks | Improved cognitive reactivity. No significant differences were found between groups in the BDI, DASS and BAI scores | [130] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thangaleela, S.; Sivamaruthi, B.S.; Kesika, P.; Chaiyasut, C. Role of Probiotics and Diet in the Management of Neurological Diseases and Mood States: A Review. Microorganisms 2022, 10, 2268. https://doi.org/10.3390/microorganisms10112268
Thangaleela S, Sivamaruthi BS, Kesika P, Chaiyasut C. Role of Probiotics and Diet in the Management of Neurological Diseases and Mood States: A Review. Microorganisms. 2022; 10(11):2268. https://doi.org/10.3390/microorganisms10112268
Chicago/Turabian StyleThangaleela, Subramanian, Bhagavathi Sundaram Sivamaruthi, Periyanaina Kesika, and Chaiyavat Chaiyasut. 2022. "Role of Probiotics and Diet in the Management of Neurological Diseases and Mood States: A Review" Microorganisms 10, no. 11: 2268. https://doi.org/10.3390/microorganisms10112268
APA StyleThangaleela, S., Sivamaruthi, B. S., Kesika, P., & Chaiyasut, C. (2022). Role of Probiotics and Diet in the Management of Neurological Diseases and Mood States: A Review. Microorganisms, 10(11), 2268. https://doi.org/10.3390/microorganisms10112268








