Relationship Between Mean Faecal Gastrointestinal Nematode Egg Excretion in Horses and Its Variability: Implications for Control
Abstract
1. Introduction
2. Materials and Methods
2.1. Characteristics of Sampling Sites
2.2. Parasitological Methods
2.3. Statistical Methods
3. Results
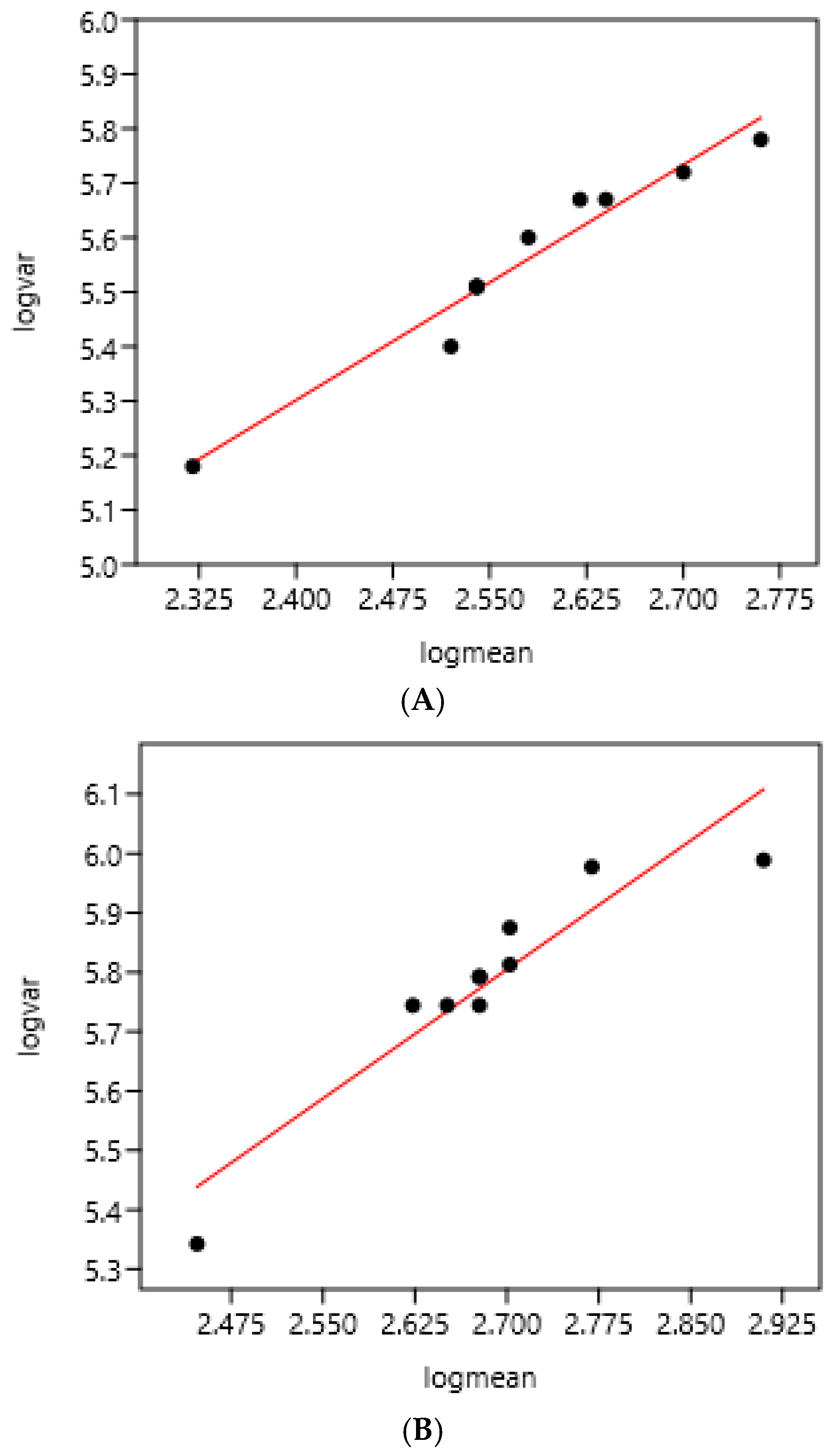
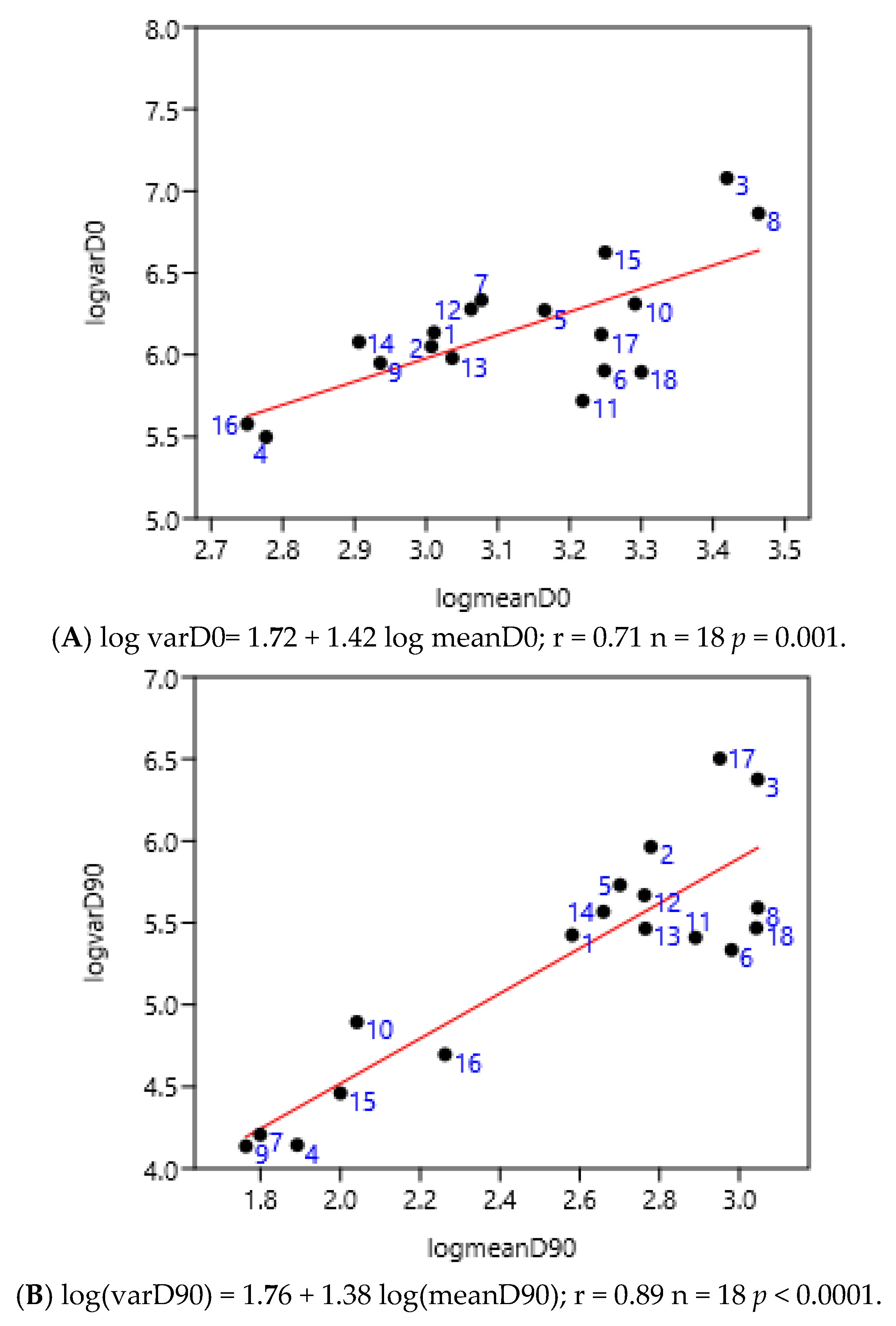
3.1. Relationship Between Average FEC and Its Variance: Factors of Variation
3.1.1. Sampling Day on a Farm
3.1.2. Age of Horses
3.1.3. Category of Horses
3.1.4. Regions of Three Countries
3.1.5. Previous Anthelmintic Treatment
3.2. Composite Faecal Egg Count to Establish Average FEC
3.2.1. Composite from Faeces Collected from Horses
3.2.2. Composite from Faeces Collected from the Ground
3.3. Relation Between Mean FEC and the Percentage of Horses with Different Indicators of FEC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Poulin, R. Are there general laws in parasite ecology? Parasitology 2007, 134, 763–776. [Google Scholar] [CrossRef]
- Gaba, S.; Ginot, V.; Cabaret, J. Modelling macroparasite aggregation using a nematode-sheep system: The Weibull distribution as an alternative to the negative binomial distribution? Parasitology 2005, 131, 393–401. [Google Scholar] [CrossRef]
- Relf, V.E.; Morgan, E.R.; Hodgkinson, J.E.; Matthews, J.B. Helminth egg excretion with regard to age, gender and management practices on UK Thoroughbred studs. Parasitology 2013, 140, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.M.; Nielsen, M.K. An evidence-based approach to equine parasite control: It ain’t the 60s anymore. Equine Vet. Educ. 2010, 22, 306–316. [Google Scholar] [CrossRef]
- Morrill, A.; Poulin, R.; Forbes, M.R. Interrelationships a, d properties of parasite aggregation measures: A user’s guide. Int. J. Parasitol. 2023, 53, 763–776. [Google Scholar] [CrossRef]
- Barger, I.A. The statistical distribution of trichostrongylid nematodes in grazing lambs. Int. J. Parasitol. 1985, 15, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Boag, B.; Hackett, C.A.; Topham, P.B. The use of Taylor’s Power Law to describe the aggregated distribution of gastro-intestinal nematodes of sheep. Int. J. Parasitol. 1992, 22, 267–270. [Google Scholar] [CrossRef]
- Taylor, L.R. Aggregation, variance, and the mean. Nature 1961, 189, 732–735. [Google Scholar] [CrossRef]
- Smith, H.F. An empirical law describing heterogeneity in the yields of agricultural crops. J. Agric. Sci. 1938, 28, 1–23. [Google Scholar] [CrossRef]
- Taylor, R.A. Taylor’s Power Law: Order and Pattern in Nature; Academic Press: London, UK, 2019; 640p. [Google Scholar]
- Morgan, E.R.; Segonds-Pichon, A.; Ferté, H.; Duncan, P.; Cabaret, J. Anthelmintic Treatment and the Stability of Parasite Distribution in Ruminants. Animals 2023, 13, 1882. [Google Scholar] [CrossRef]
- Murphy, D.; Love, S. The pathogenic effects of experimental cyathostome infections in ponies. Vet. Parasitol. 1997, 70, 99–110. [Google Scholar] [CrossRef]
- Kilani, M.; Chermette, R.; Guillot, J.; Polack, B.; Duncan, J.L.; Cabaret, J. Gastrointestinal nematodes. In Infectious and Parasitic Diseases of Livestock. 2. Bacterial Diseases, Fungal Diseases, Parasitic Diseases; Tec & Toc Lavoisier: Paris, France, 2010; pp. 1481–16003. [Google Scholar]
- Kaplan, R.M. Anthelmintic resistance in nematodes of horses. Vet. Res. 2002, 33, 491–507. [Google Scholar] [CrossRef]
- Silva, P.A.; Cernea, M.; Madeira de Carvalho, L. Anthelmintic Resistance in Equine Nematodes-A Review on the Current Situation, with Emphasis in Europe. Bull. Univ. Agric. Sci. Vet. 2019, 76. [Google Scholar] [CrossRef] [PubMed]
- Uriarte, J.; Cabaret, J.; Tanco, J.A. The distribution and abundance of parasitic infections in sheep grazing on irrigated or on non-irrigated pastures in North-Eastern Spain. Ann. De Rech. Vétérinaires 1985, 16, 321–325. [Google Scholar]
- Duncan, J.L.; Love, S. Preliminary observations on an alternative strategy for the control of horse strongyles. Equine Vet. J. 1991, 23, 226–228. [Google Scholar] [CrossRef]
- Gomez, H.H.; Georgi, J.R. Equine helminth infections: Control by selective chemotherapy. Equine Vet. J. 1991, 23, 198–200. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.K. Sustainable equine parasite control: Perspectives and research needs. Vet. Parasitol. 2012, 185, 32–44. [Google Scholar] [CrossRef]
- Pfister, K.; van Doorn, D. New perspectives in equine intestinal parasitic disease: Insights in monitoring helminth infections. Vet. Clin. North America. Equine Pract. 2018, 34, 141–153. [Google Scholar] [CrossRef]
- Matthee, S.; McGeoch, M.A. Helminths in horses: Use of selective treatment for the control of strongyles. J. S. Afr. Vet. Assoc. 2004, 75, 129–136. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Becher, A.M.; Mahling, M.; Nielsen, M.K.; Pfister, K. Selective anthelmintic therapy of horses in the Federal states of Bavaria (Germany) and Salzburg (Austria): An investigation into strongyle egg shedding consistency. Vet. Parasitol. 2010, 171, 116–122. [Google Scholar] [CrossRef]
- Lüthin, S.; Zollinger, A.; Basso, W.; Bisig, M.; Caspari, N.; Eng, V.; Frey, C.F.; Grimm, F.; Igel, P.; Lüthi, S.; et al. Strongyle faecal egg counts in Swiss horses: A retrospective analysis after the introduction of a selective treatment strategy. Vet. Parasitol. 2023, 323, 110027. [Google Scholar] [CrossRef] [PubMed]
- Jürgenschellert, L.; Krücken, J.; Bousquet, E.; Bartz, J.; Heyer, N.; Nielsen, M.K.; von Samson-Himmelstjerna, G. Occurrence of strongylid nematode parasites on horse farms in Berlin and Brandenburg, Germany, with high seroprevalence of Strongylus vulgaris infection. Front. Vet. Sci. 2022, 9, 892920. [Google Scholar] [CrossRef]
- Sallé, G.; Cortet, J.; Koch, C.; Reigner, F.; Cabaret, J. Economic assessment of FEC-based targeted selective drenching in horses. Vet. Parasitol. 2015, 214, 159–166. [Google Scholar] [CrossRef]
- Vineer, H.R.; Vande Velde, F.; Bull, K.; Claerebout, E.; Morgan, E.R. Attitudes towards worm egg counts and targeted selective treatment against equine cyathostomins. Prev. Vet. Med. 2017, 144, 66–74. [Google Scholar] [CrossRef]
- Kornaś, S.; Cabaret, J.; Skalska, M.; Nowosad, B. Horse infection with intestinal helminths in relation to age, sex, access to grass and farm system. Vet. Parasitol. 2010, 174, 285–291. [Google Scholar] [CrossRef]
- Pagnon, R. Résistance aux Anthelminthiques des Strongles Chez les Equidés: Enquête Dans un Centre Equestre du Sud de la France. Ph.D. Thesis, Université de Toulouse, Toulouse, France, 2005; 121p. [Google Scholar]
- Kaplan, R.K.; Denwood, M.J.; Nielsen, M.K.; Thamsborg, S.M.; Torgerson, P.R.; Gilleard, J.S.; Dobson, R.J.; Vercruysse, J.; Levecke, B. World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) guideline for diagnosing anthelmintic resistance using the faecal egg count. reduction test in ruminants, horses and swine. Vet. Parasitol. 2023, 318, 109936. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, S.A.; Korsholm, H. A method for culture and recovery of gastrointestinal strongyle larvae. Nordisk Vet. Med. 1983, 35, 429–430. [Google Scholar]
- Amer, M.M.; Desouky, A.Y.; Helmy, N.M.; Abdou, A.M.; Sorour, S.S. Identifying 3rd larval stages of common strongylid and non-strongylid nematodes (class: Nematoda) infecting Egyptian equines based on morphometric analysis. BMC Vet. Res. 2022, 18, 432. [Google Scholar] [CrossRef] [PubMed]
- Eisler, Z.; Bartos, I.; Kertész, J. Fluctuation scaling in complex systems: Taylor’s law and beyond. Adv. Phys. 2008, 57, 89–142. [Google Scholar] [CrossRef]
- Johnson, P.T.J.; Wilber, M.Q. Biological and statistical processes jointly drive population aggregation: Using host–parasite interactions to understand Taylor’s power law. Proc. R. Soc. B 2017, 284, 20171388. [Google Scholar] [CrossRef]
- McVinish, R.; Lester, R.J.G. Measuring aggregation in parasite populations. J. R. Soc. Interface 2020, 17, 20190886. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, R.C.; Vineer, H.R.; Grainger-Wood, R.; Morgan, E.R. Heterogeneity in helminth infections: Factors influencing aggregation in a simple host–parasite system. Parasitology 2020, 147, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Galvani, A.P. Immunity, antigenic heterogeneity, and aggregation of helminth parasites. J. Parasitol. 2003, 89, 232–241. [Google Scholar] [CrossRef]
- Boag, B.; Lello, J.; Fenton, A.; Tompkins, D.M.; Hudson, P.J. Patterns of parasite aggregation in the wild European rabbit (Oryctolagus cuniculus). Int. J. Parasitol. 2001, 31, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Morgan, E.R.; Cavill, L.; Curry, G.E.; Wood, R.M.; Mitchell, E.S.E. Effects of aggregation and sample size on composite faecal egg counts in sheep. Vet. Parasitol. 2005, 131, 79–87. [Google Scholar] [CrossRef]
- Eysker, M.; Bakker, J.; Van Den Berg, M.; Van Doorn, D.C.K.; Ploeger, H.W. The use of age-clustered pooled faecal samples for monitoring worm control in horses. Vet. Parasitol. 2008, 151, 249–255. [Google Scholar] [CrossRef]
- Nielsen, M.K.; Baptiste, K.E.; Tolliver, S.C.; Collins, S.S.; Lyons, E.T. Analysis of multiyear studies in horses in Kentucky to ascertain whether counts of eggs and larvae per gram of feces are reliable indicators of numbers of strongyles and ascarids present. Vet. Parasitol. 2010, 174, 77–84. [Google Scholar] [CrossRef]
- Martínez-Ortiz de Montellano, C.; González-Reyes, L.; Toledo-Alvarado, H.O. Epidemiological patterns of cyathostominae burden in Warmblood horses in Mexico. WAAVP 2025. In Proceedings of the 30th Conference of the World Association for the Advancements of Veterinary Parasitology, Curitiba, Brazil, 17–21 August 2025; pp. 61–62. [Google Scholar]
- Matthews, J.B. Anthelmintic resistance in equine nematodes. Int. J. Parasitol. Drugs Drug Resist. 2014, 4, 310–315. [Google Scholar] [CrossRef]
- Nielsen, M.K.; Mittel, L.; Grice, A.; Erskine, M.; Graves, E.; Vaala, W.; Tully, R.C.; French, D.D. AAEP Parasite Control Guidelines. 2013. Available online: https://static1.squarespace.com/static/624e2ee7d1ae6057e493a94f/t/62533421a0850d236f1acd08/1649620001387/AAEP_ParasiteControl_Detailed_2013.pdf (accessed on 15 November 2025).
- Rendle, D.; Austin, C.; Bowen, M.; Cameron, I.; Furtado, T.; Hodgkinson, J.; McGorum, B.; Matthews, J. Equine de-worming: A consensus on current best practice. UK-Vet Equine 2019, 3, 1–14. [Google Scholar] [CrossRef]
- Wilkes, E.J.A.; Woodgate, R.G.; Raidal, S.L.; Hughes, K.J. The application of faecal egg count results and statistical inference for clinical decision making in foals. Vet. Parasitol. 2019, 270, 7–12. [Google Scholar] [CrossRef]
- Denwood, M.J.; Love, S.; Innocent, G.T.; Matthews, L.; McKendrick, I.J.; Hillary, N.; Reid, S.W.J. Quantifying the sources of variability in equine faecal egg counts: Implications for improving the utility of the method. Vet. Parasitol. 2012, 188, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Lester, H.E.; Morgan, E.R.; Hodgkinson, J.E.; Matthews, J.B. Analysis of strongyle egg shedding consistency in horses and factors that affect it. J. Equine Vet. Sci. 2018, 60, 113–119. [Google Scholar] [CrossRef]
- Kuzmina, T.A.; Lyons, E.T.; Tolliver, S.C.; Dzeverin, I.I.; Kharchenko, V.A. Fecundity of various species of strongylids (Nematoda: Strongylidae)—Parasites of domestic horses. Parasitol. Res. 2012, 111, 2265–2271. [Google Scholar] [CrossRef] [PubMed]
- Fleurance, G.; Martin-Rosset, W.; Dumont, B.; Duncan, P.; Farrugia, A.; Lecomte, T. Environmental impact of horses. In Equine nutrition; Martin-Rosset, W., Ed.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2013; pp. 481–504. [Google Scholar]








| Country | Climate on Yearly Basis Rainfall in mm (Temperature) | Horse Breed | Type of Horse (Number) | Number of Farms/Duration in Years | Anthelmintic Treatments | Anthelmintic Efficacy (%) | Percentage of Cyathostomins |
|---|---|---|---|---|---|---|---|
| Poland (South-East Part) | 680 (8.7 °C) | Pure-Blood Arabian, Thoroughbred | Racing (12,450 altogether) | 74 (10) | Pyrantel, Fenbendazole Ivermectin Moxidectin | 51 * 23 67 100 | 99 |
| France 1 | 689 (11.8 °C) | Welsh Pony | Leisure (100 × 10 sampling events) | 1 (3) | Ivermectin, Moxidectin Pyrantel | 90 100 90 | 98 |
| France 2 | 579 (14.8 °C) | Diverse | Riding club (35 × 4 sampling events) | 1 (0.5) | Ivermectin, Mebendazole | 96 76 | Nd ** |
| France 3 | 1237 (10.8 °C) | Selle Français, Anglo-Arab | Racing (160 × 6 sampling events) | 1 (2) | Ivermectin, Pyrantel Fenbendazole | 96 97 55 | 95 |
| Mexico 1 | 1240 (25.0 °C) | American Quarter | Racing (42 × 4 sampling events) | 1 (0.25) | Ivermectin, Moxidectin, Febantel | 99 90 97 | 64 |
| Mexico 2 | 490 (24.0 °C) | American Quarter | Racing (54 × 4 sampling events) | 1 (0.25) | Ivermectin, Moxidectin, Febantel | 99 98 90 | 50 |
| Mexico 3 | 426 (22.5 °C) | American Quarter | Racing (71 × 4 sampling events) | 1 (0.25) | Ivermectin, Moxidectin, Febantel and Metrifonate | 100 100 93 | 30 |
| Mexico 4 | 747 (26.5 °C) | Raza Española | Racing (99 × 4 sampling events) | 1 (0.25) | Ivermectin, Moxidectin, Febantel and Metrifonate | 100 100 100 | 65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cabaret, J.; Guerrero Molina, C.; Martínez-Ortiz-de Montellano, C.; Alcala Canto, Y. Relationship Between Mean Faecal Gastrointestinal Nematode Egg Excretion in Horses and Its Variability: Implications for Control. Pathogens 2026, 15, 156. https://doi.org/10.3390/pathogens15020156
Cabaret J, Guerrero Molina C, Martínez-Ortiz-de Montellano C, Alcala Canto Y. Relationship Between Mean Faecal Gastrointestinal Nematode Egg Excretion in Horses and Its Variability: Implications for Control. Pathogens. 2026; 15(2):156. https://doi.org/10.3390/pathogens15020156
Chicago/Turabian StyleCabaret, Jacques, Cristina Guerrero Molina, Cintli Martínez-Ortiz-de Montellano, and Yazmin Alcala Canto. 2026. "Relationship Between Mean Faecal Gastrointestinal Nematode Egg Excretion in Horses and Its Variability: Implications for Control" Pathogens 15, no. 2: 156. https://doi.org/10.3390/pathogens15020156
APA StyleCabaret, J., Guerrero Molina, C., Martínez-Ortiz-de Montellano, C., & Alcala Canto, Y. (2026). Relationship Between Mean Faecal Gastrointestinal Nematode Egg Excretion in Horses and Its Variability: Implications for Control. Pathogens, 15(2), 156. https://doi.org/10.3390/pathogens15020156








