Granulomatous Secondary Syphilis: A Case Report with a Brief Overview of the Diagnostic Role of Immunohistochemistry
Abstract
:1. Introduction
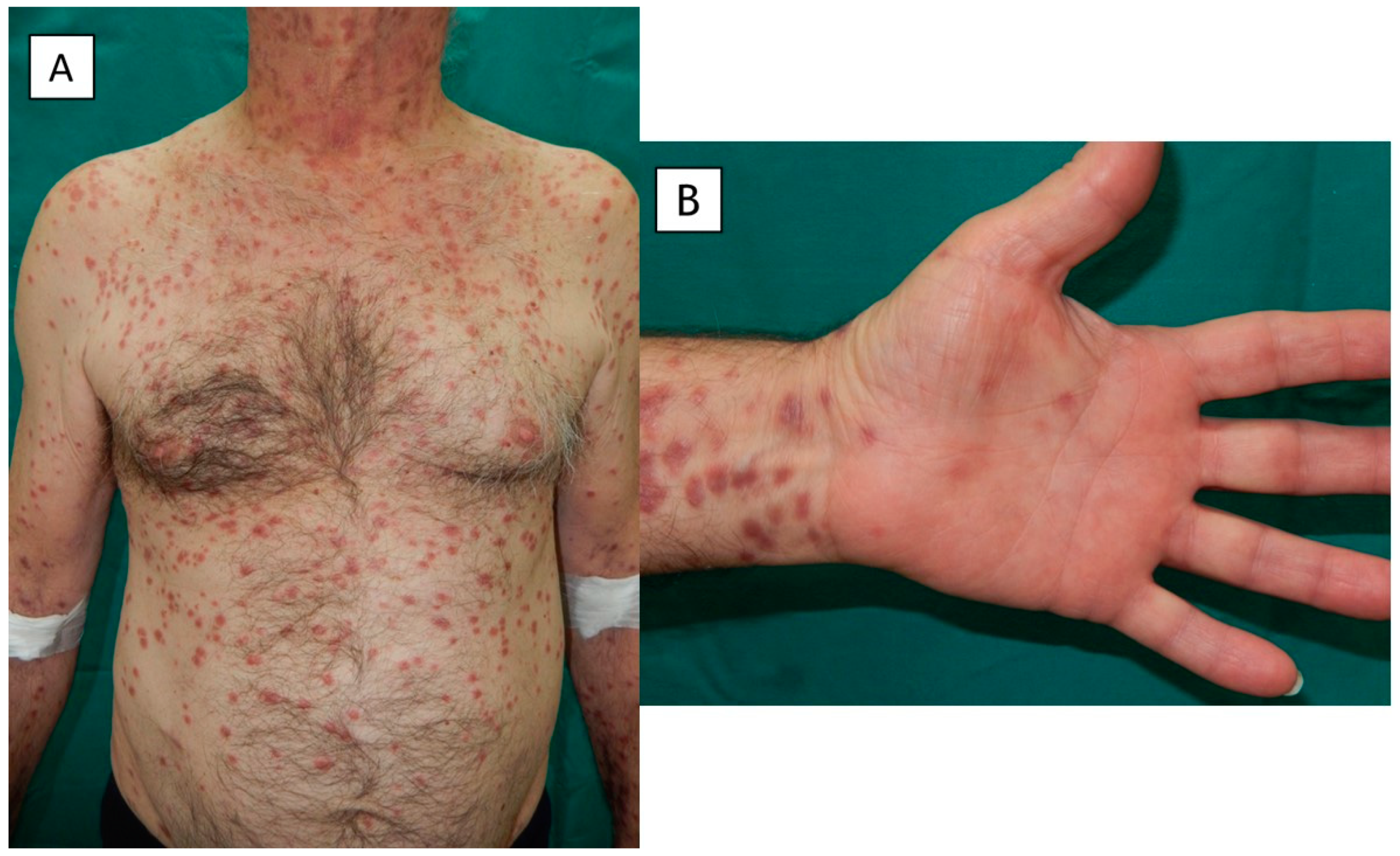
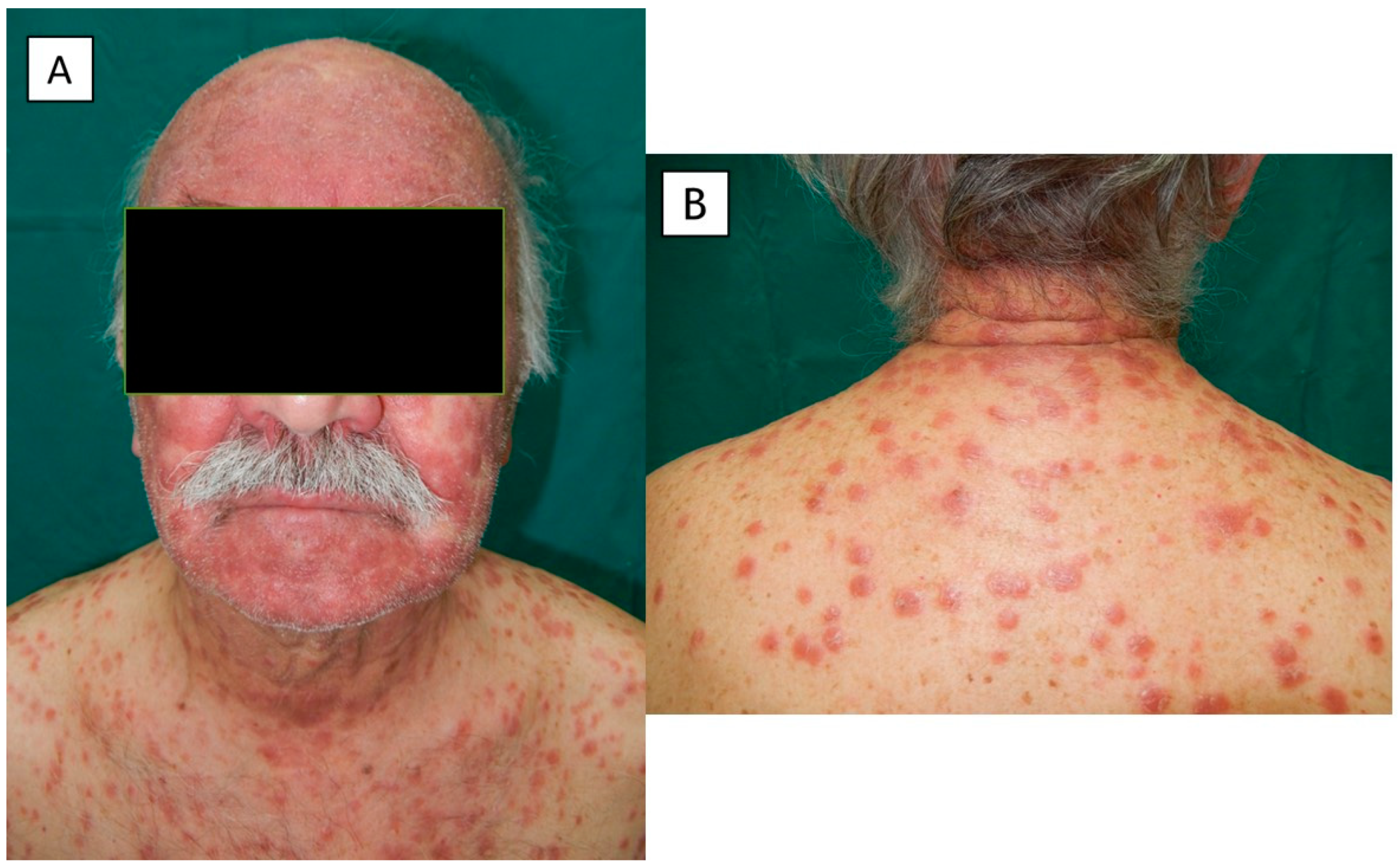
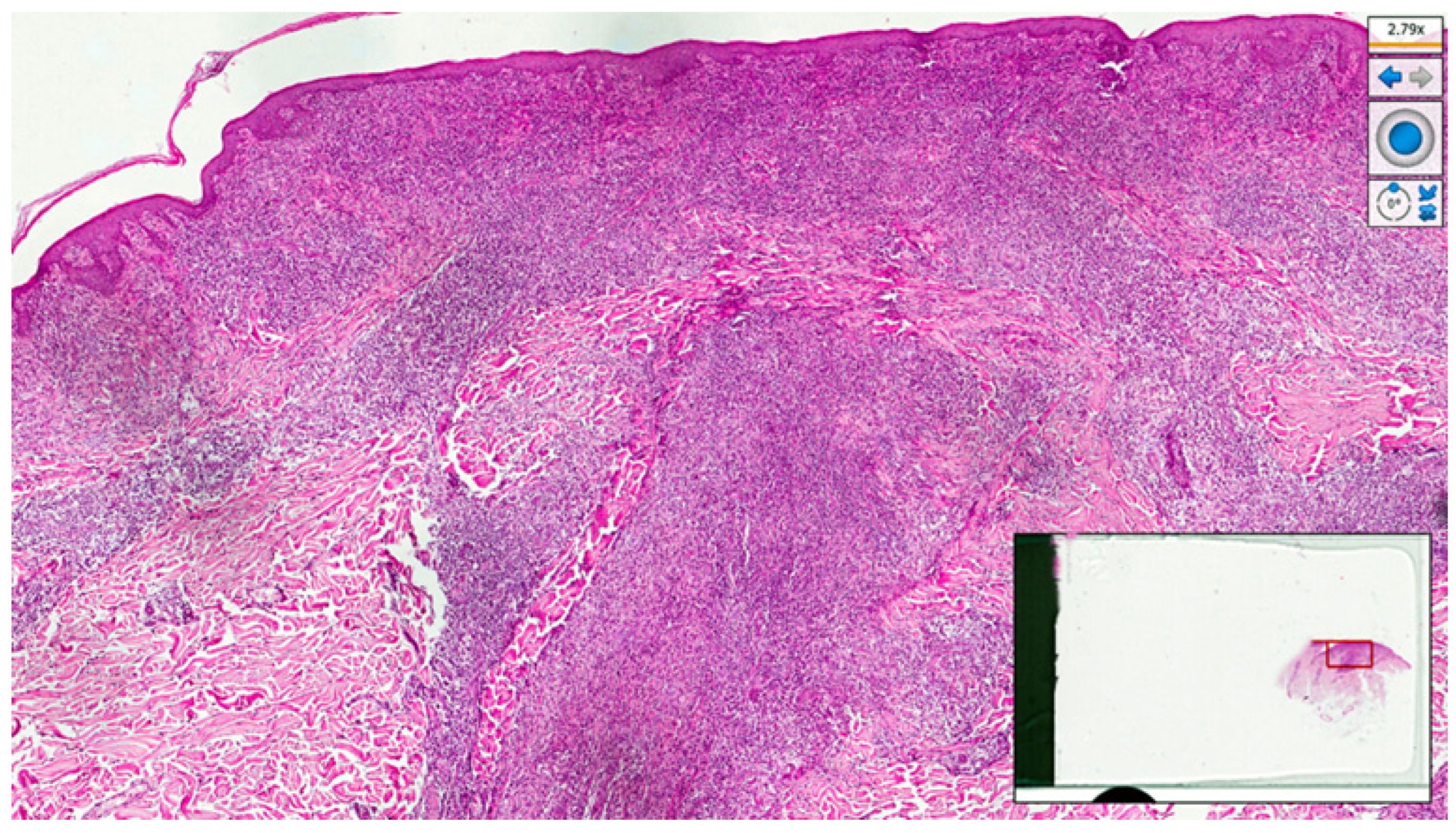
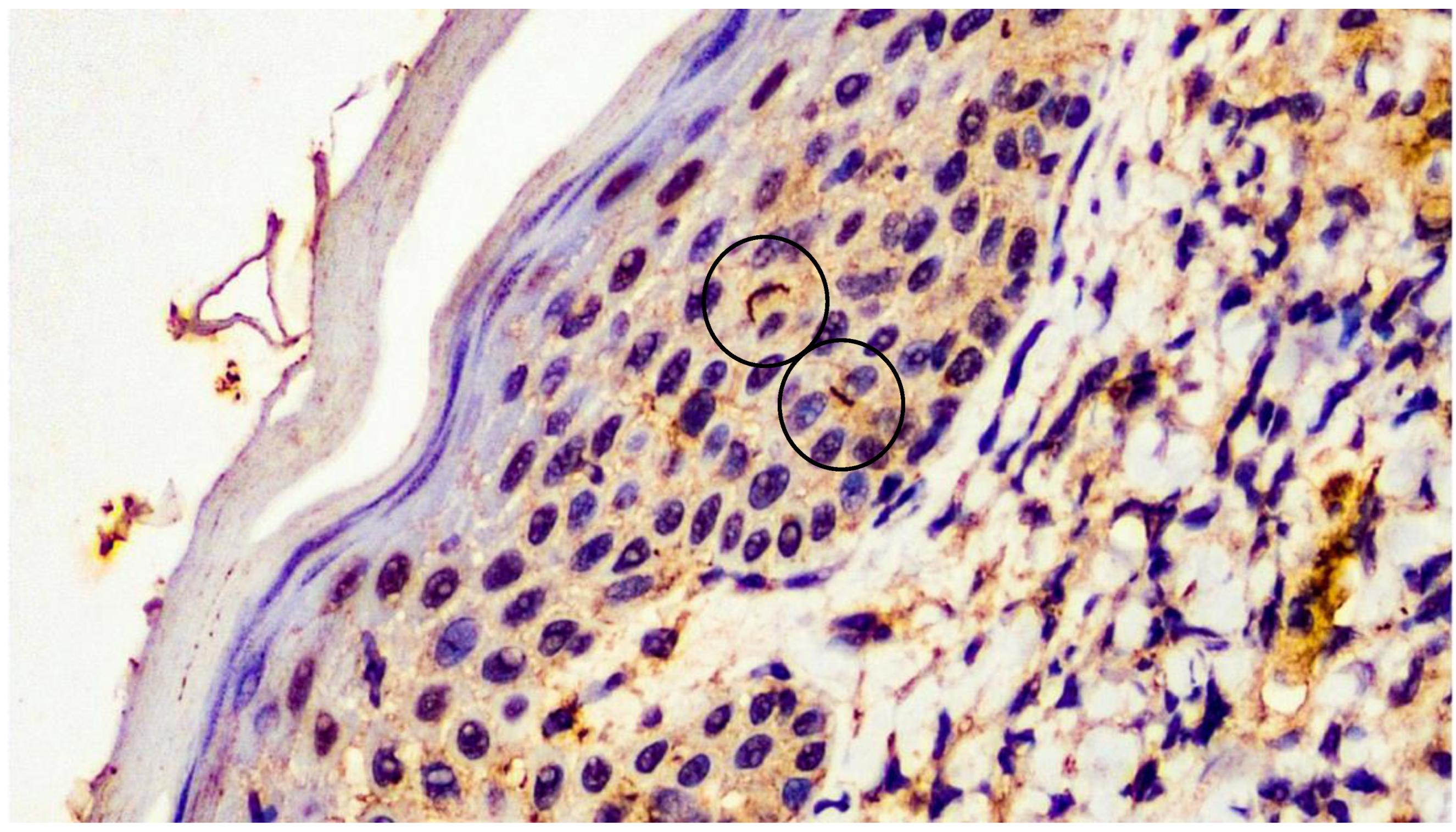
2. Case Presentation
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ghanem, K.G.; Ram, S.; Rice, P.A. The Modern Epidemic of Syphilis. N. Engl. J. Med. 2020, 382, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Rysgaard, C.; Alexander, E.; Swick, B.L. Nodular secondary syphilis with associated granulomatous inflammation: Case report and literature review. J. Cutan. Pathol. 2014, 41, 370–379. [Google Scholar] [CrossRef]
- Magdaleno-Tapial, J.; Valenzuela-Oñate, C.; Ortiz-Salvador, J.M.; Hernández-Bel, P.; Alegre-De Miquel, V. Treponema Pallidum Epidermotropism in Nodular Secondary Syphilis. Indian J. Dermatol. 2018, 63, 509–511. [Google Scholar] [PubMed]
- Li, F.; Wang, T.; Wang, L. Secondary syphilis primarily presenting with multiple nodules on the scalp: Case report and published work review. J. Dermatol. 2017, 44, 1401–1403. [Google Scholar] [CrossRef]
- Sezer, E.; Luza, B.; Calonje, E. Secondary syphilis with an interstitial granuloma annulare-like histopathologic pattern. J. Cutan. Pathol. 2011, 38, 439–442. [Google Scholar] [CrossRef]
- Jin, A.; McLarney, B.D.; Hsu, S.; Lee, J.B. Granulomatous secondary syphilis: Another diagnostic pitfall for the dermatopathologist. J. Cutan. Pathol. 2023, 50, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Theel, E.S.; Katz, S.S.; Pillay, A. Molecular and Direct Detection Tests for Treponema pallidum Subspecies pallidum: A Review of the Literature, 1964–2017. Clin. Infect. Dis. 2020, 71, S4–S12. [Google Scholar] [CrossRef]
- Buffet, M.; Grange, P.A.; Gerhardt, P.; Carlotti, A.; Calvez, V.; Bianchi, A.; Dupin, N. Diagnosing Treponema pallidum in secondary syphilis by PCR and immunohistochemistry. J. Investig. Dermatol. 2007, 127, 2345–2350. [Google Scholar] [CrossRef]
- Magri, F.; Donà, M.G.; Panetta, C.; Pontone, M.; Pimpinelli, F.; Cameli, N.; Cristaudo, A.; Zaccarelli, M.; Latini, A. Unusual clinical manifestation and challenging serological interpretation of syphilis: Insights from a case report. BMC Infect. Dis. 2021, 21, 521. [Google Scholar] [CrossRef] [PubMed]
- Martín-Ezquerra, G.; Fernandez-Casado, A.; Barco, D.; Jucglà, A.; Juanpere-Rodero, N.; Manresa, J.M.; de Almeida, L.M.; Rodríguez-Peralto, J.L.; Kutzner, H.; Cerroni, L.; et al. Treponema pallidum distribution patterns in mucocutaneous lesions of primary and secondary syphilis: An immunohistochemical and ultrastructural study. Hum. Pathol. 2009, 40, 624–630. [Google Scholar] [CrossRef]
- Hoang, M.P.; High, W.A.; Molberg, K.H. Secondary syphilis: A histologic and immunohistochemical evaluation. J. Cutan. Pathol. 2004, 31, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Rosa, M.F.A.P.; Quintella, L.P.; Ferreira, L.C.; Cuzzi, T. Immunohistochemical detection of Treponema pallidum in skin samples with clinical and histopathological correlations and Warthin-Starry staining critical analysis. An. Bras. Dermatol. 2023, 98, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Flores, A. Immunostaining for Treponema pallidum: Caution in its evaluation. Am. J. Dermatopathol. 2010, 32, 523–525. [Google Scholar] [CrossRef] [PubMed]
- Pettit, C.; McMurray, S.; Randall, M.B.; Jones, A.; Fisher, K. Highlighting a Potential Pitfall: Positive Treponema pallidum Immunohistochemical Stain in a Patient Without Syphilis. Am. J. Dermatopathol. 2019, 41, 924–926. [Google Scholar] [CrossRef]
- Glatz, M.; Achermann, Y.; Kerl, K.; Bosshard, P.P.; Cozzio, A. Nodular secondary syphilis in a woman. BMJ Case. Rep. 2013, 2013, bcr-2013-009130. [Google Scholar] [CrossRef]
- Hinojosa, J.A.; Maxim, E.; Garza-Chapa, J.I.; Tovar-Garza, A.; Susa, J. Diffuse Papular Rash in a Young Woman: Challenge. Am. J. Dermatopathol. 2019, 41, 82–83. [Google Scholar] [CrossRef]
- Hinojosa, J.A.; Maxim, E.; Garza-Chapa, J.I.; Tovar-Garza, A.; Susa, J. Diffuse Papular Rash in a Young Woman: Answer. Am. J. Dermatopathol. 2019, 41, 615–616. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Yang, T.H.; Chang, Y.S.; Chang, I.J. Granulomatous secondary syphilis with a facial annular sarcoidosis-like clinical and histopathological pattern. Australas. J. Dermatol. 2020, 61, e364–e365. [Google Scholar] [CrossRef]
- Phan, K.; Gorji, M.; Smith, S.D.; Harvey, R. Granulomatous secondary syphilis: Uncommon and atypical presentation. Int. J. Dermatol. 2020, 59, 270–272. [Google Scholar] [CrossRef]
- Henebeng, E.; Karasko, E.; Robinson-Bostom, L.; DiMarco, C. Secondary Syphilis With Elastophagocytosis: An Unusual Histologic Finding. Am. J. Dermatopathol. 2021, 43, 225–227. [Google Scholar] [CrossRef]
- Fernández-Camporro, A.; Calzada-González, J.M.; Arcos-González, P.; Rodríguez-Díaz, E. Granulomatous Secondary Syphilis Suggested by Histology: A Case Report. Actas. Dermosifiliogr. 2022, 113, S26–S28. [Google Scholar] [CrossRef] [PubMed]
- Yousefian, F.; Wang, H.; Yadlapati, S.; Cohen, J.A.; Browning, J.C. Granulomatous Secondary Syphilis: An Uncommon Presentation of the Great Imitator. Am. J. Med. 2023, 136, 25–26. [Google Scholar] [CrossRef]
- Saal, R.C.; Borda, L.J.; Pariser, R.J. Grouped follicular secondary syphilis: Case report and review of literature. Australas. J. Dermatol. 2023, 64, e168–e170. [Google Scholar] [CrossRef] [PubMed]
- Flamm, A.; Alcocer, V.M.; Kazlouskaya, V.; Kwon, E.J.; Elston, D. Histopathologic features distinguishing secondary syphilis from its mimickers. J. Am. Acad. Dermatol. 2020, 82, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.K.; Li, J. Histologic Features of Secondary Syphilis. Dermatology 2020, 236, 145–150. [Google Scholar] [CrossRef]



| First Author [Reference] | Sex | Age (yrs) | Cutaneous Lesions (Duration, Type and Distribution) | Other Reported Clinical Information | Histopathology | IHC Results | Special Stains * |
|---|---|---|---|---|---|---|---|
| Sezer [5] | Male | 47 | 3 months Non-pruritic macules and papules mostly on the trunk and extremities A few lesions on a palm. | - | Minimal acanthosis, focal basal vacuolar changes; rare dyskeratotic keratinocytes In the superficial and mid-to-deep dermis, moderate perivascular and interstitial infiltrate with numerous plasma cells, lymphocytes and a few neutrophils; histiocytes with occasional multinucleated giant cells (interstitial granuloma annulare-like pattern) | Positive (numerous spirochetes, mostly within endothelial cells and less frequently within macrophages or the basal epidermis) | - |
| Glatz [15] | Female | 21 | 2 months Painless red patches and scaling, ulcerated, nodules on the face, neck and upper parts of the trunk and arms | No involvement of palms and soles No mucosal lesions No lymphadenopathy Headache No fever, malaise or weight loss | Hyperkeratosis, acanthosis and spongiosis of the epidermis with focal erosion; interface dermatitis Noncaseating granulomas in the whole dermis with multinucleated giant cells, interstitial and perivascular lymphohistiocytic dermal infiltrate, eosinophils and plasma cells | Positive (spirochetes in the dermis) | PAS, Brown-Brenn-Gram, ZN |
| Rysgaard [2] | Male | 62 | 10 days Numerous nonpruritic red-brown, focally orange, papules and nodules on the trunk and upper extremities | No involvement of palms and soles No mucosal lesions | Marked perivascular and periadnexal lymphohistiocytic infiltrate in the dermis; non-palisading epithelioid granulomas with numerous plasma cells and multinucleated giant cells with elastophagocytosis | Positive (occasional spirochetes within granulomatous foci) | Gram, GMS, AFB, WS |
| Magdaleno-Tapial [3] | Male | 55 | 30 days Multiple erythematous nodules and annular plaques on the face, neck, arms and upper parts of the trunk | No involvement of palms and soles No mucosal lesions No lymphadenopathy No systemic symptoms Alopecia in the occipital region | Psoriasiform epidermal hyperplasia Granulomatous infiltrate in the papillary and reticular dermis with epithelioid histiocytes and numerous multinucleated giant cells (tuberculoid granuloma), surrounded by a dense lymphoplasmacytic infiltrate; capillaries with edematous walls and prominent endothelial cells | Positive (especially in the epidermis and the epidermal ridges) | - |
| Hinojosa [16,17] | Female | 25 | 8 weeks Hundreds of indurated erythematous itchy and painful papules on the face, neck, trunk and extremities (papulo-squamous on the trunk); some nodular or umbilicated lesions. A few brown palmoplantar macules |
Dyspnea, malaise, myalgia (recent episode of fever, chills and sore throat) Genital lesions | Mild acanthosis, parakeratosis; neutrophils in the cornified layer Superficial and deep lymphohistiocytic infiltrate with periadnexal accentuation, epithelioid granulomas containing multinucleated giant cells and neutrophilic abscesses with a few plasma cells and eosinophils | Positive (multiple spirochetes in the epidermis, dermis and within granulomatous infiltrate) | Gram, PAS, Fite |
| Lee [18] | Male | 29 | 2–3 weeks Asymptomatic, erythematous annular plaques only on the face | No involvement of palms and soles No mucosal or genital lesions No systemic symptoms No lymphadenopathy | Upper dermal perivascular and interstitial inflammatory infiltrates composed of lymphocytes and scattered plasma cells with a small naked granuloma | Positive | PAS; Alcian blue |
| Phan [19] | Female | 30 | 4 weeks Erythematous papules with inward facing scale, primarily around the buttocks, trunk, and abdomen | No systemic symptoms Mild alterations of liver function tests Abdominal lymphadenopathy | Irregular acanthosis, mild spongiosis Granulomatous inflammation in the superficial dermis with epithelioid histiocytes forming granulomata, mostly around hair follicles and eccrine ducts; focal necrosis with several neutrophils within most granulomata; perivascular lymphohistiocytic infiltrate with occasional plasma cells and neutrophils in the superficial dermis | Positive (scattered elongated organisms) | PAS, GMS, ZN, Wade-Fite, PUTT |
| Henebeng [20] | Male | 65 | 5 months Violaceous macules on the arms, legs, palms, and soles |
At the onset, fever, rigors, and cervical lymphadenopathy No headache, weight changes, myalgia, numbness, or vision problems |
Vacuolar interface alteration, focal exocytosis of lymphocytes Mild superficial and mild dermal perivascular lymphoplasmacytic inflammation; granuloma with interstitial giant cells and elastophagocytosis | Positive (rare intraepidermal spirochetes) | - |
| Fernández Camporro [21] | Female | 51 | 15 days Mildly pruritic papules predominantly on the abdomen and top of the limbs | No involvement of palms and soles No fever or other systemic symptoms No genital or oral lesions No lymphadenopathy | Normal epidermis Predominantly perivascular nodular inflammatory infiltrate in the upper dermis comprising histiocytic cells, several multinucleated giant cells, epithelioid cells, lymphocytes, and abundant plasma cells, without caseous degeneration; endothelial edema and swelling | Positive (abundant intracellular and extracellular helical structures) | - |
| Jin [6] | Female | 44 | 1 month Pruritic scattered erythematous, edematous papules and nodules on the extremities, trunk, and face | No involvement of palms and soles | Dense nodular lymphohistiocytic dermal infiltrate with periadnexal involvement; tuberculoid granulomas with numerous plasma cells | Negative | AFB, GMS, WS |
| Yousefian [22] | Male | 20 | 6 weeks Multiple erythematous nodules on the forehead, nose, and upper limbs | Severe headache, photophobia, fatigue Cervical lymphadenopathy | Dense dermal granulomatous infiltrate with lymphocytes and numerous plasma cells | Positive (numerous spirochetes) | - |
| Saal [23] | Male | 25 | 3 months Mildly pruritic edematous, grouped, and follicular papules mostly on the trunk and upper extremities A small, hyperpigmented patch on the heel of the left foot | No systemic symptoms Alopecic patches on the scalp | Mild lichenoid infiltrate and multiple non-caseating granulomas in the dermis; prominent infiltrate of lymphocytes and plasma cells | Positive (numerous spirochetes in the epidermis and dermis) | PAS, AFB |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ambrogio, F.; Cazzato, G.; Foti, C.; Grandolfo, M.; Mennuni, G.B.; Vena, G.A.; Cassano, N.; Lettini, T.; Castronovi, C.; Ingordo, V.; et al. Granulomatous Secondary Syphilis: A Case Report with a Brief Overview of the Diagnostic Role of Immunohistochemistry. Pathogens 2023, 12, 1054. https://doi.org/10.3390/pathogens12081054
Ambrogio F, Cazzato G, Foti C, Grandolfo M, Mennuni GB, Vena GA, Cassano N, Lettini T, Castronovi C, Ingordo V, et al. Granulomatous Secondary Syphilis: A Case Report with a Brief Overview of the Diagnostic Role of Immunohistochemistry. Pathogens. 2023; 12(8):1054. https://doi.org/10.3390/pathogens12081054
Chicago/Turabian StyleAmbrogio, Francesca, Gerardo Cazzato, Caterina Foti, Mauro Grandolfo, Gisella Biagina Mennuni, Gino Antonio Vena, Nicoletta Cassano, Teresa Lettini, Cosimo Castronovi, Vito Ingordo, and et al. 2023. "Granulomatous Secondary Syphilis: A Case Report with a Brief Overview of the Diagnostic Role of Immunohistochemistry" Pathogens 12, no. 8: 1054. https://doi.org/10.3390/pathogens12081054
APA StyleAmbrogio, F., Cazzato, G., Foti, C., Grandolfo, M., Mennuni, G. B., Vena, G. A., Cassano, N., Lettini, T., Castronovi, C., Ingordo, V., Romita, P., & Filotico, R. (2023). Granulomatous Secondary Syphilis: A Case Report with a Brief Overview of the Diagnostic Role of Immunohistochemistry. Pathogens, 12(8), 1054. https://doi.org/10.3390/pathogens12081054








