Distribution of Epstein–Barr Virus LMP1 Variants in Patients with Infectious Mononucleosis and Association with Selected Biochemical and Hematological Parameters
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Study Design
2.2. PCR and Population-Based Sequencing
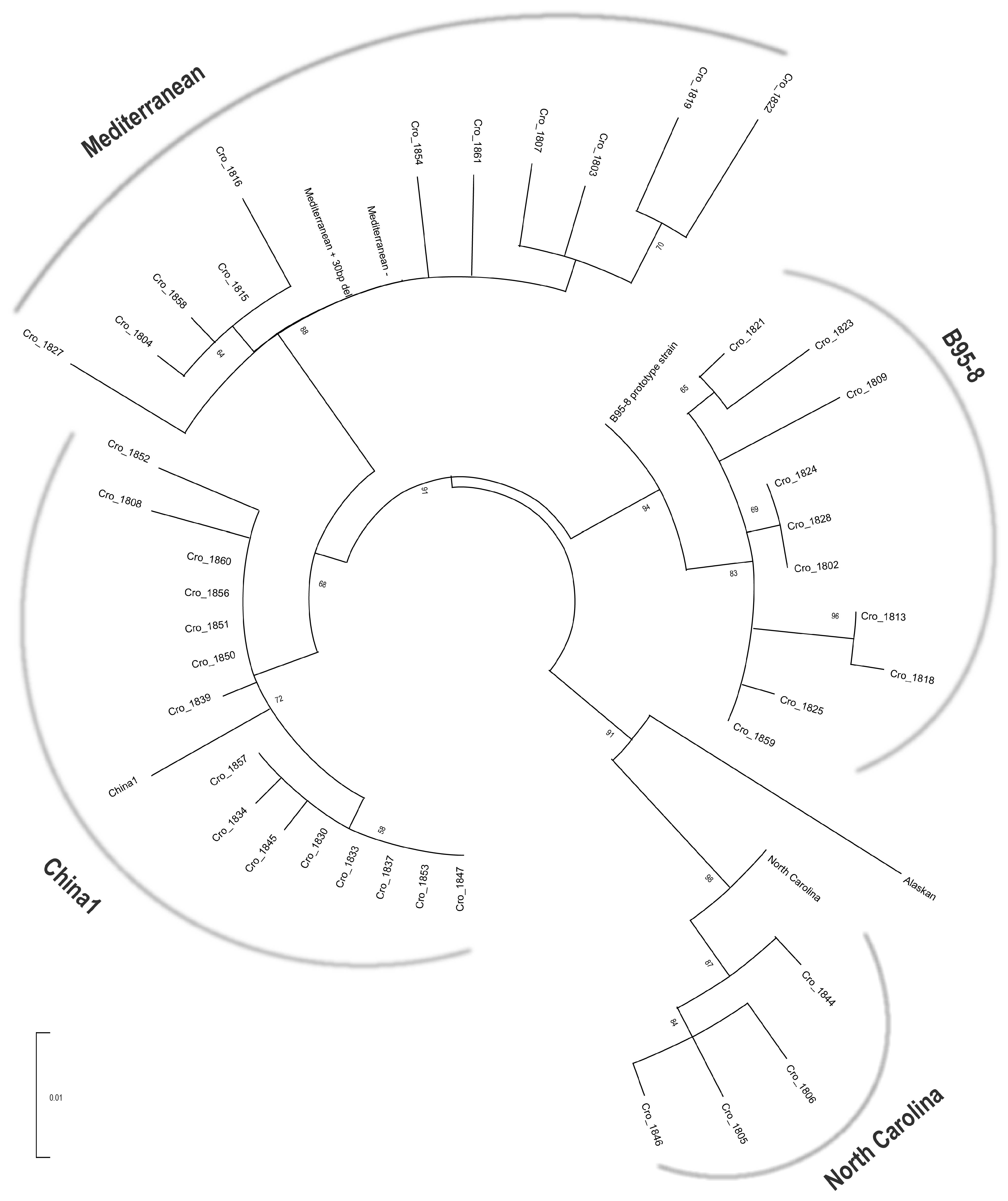
2.3. Phylogenetic Analysis LMP1 Sequences
2.4. Statistical Analysis
3. Results
3.1. Distribution and Characterization of EBV LMP1 Variants in Patients with IM
3.2. Detection of Amino Acid Substitutions in LMP1 Gene in Croatian IM Isolates
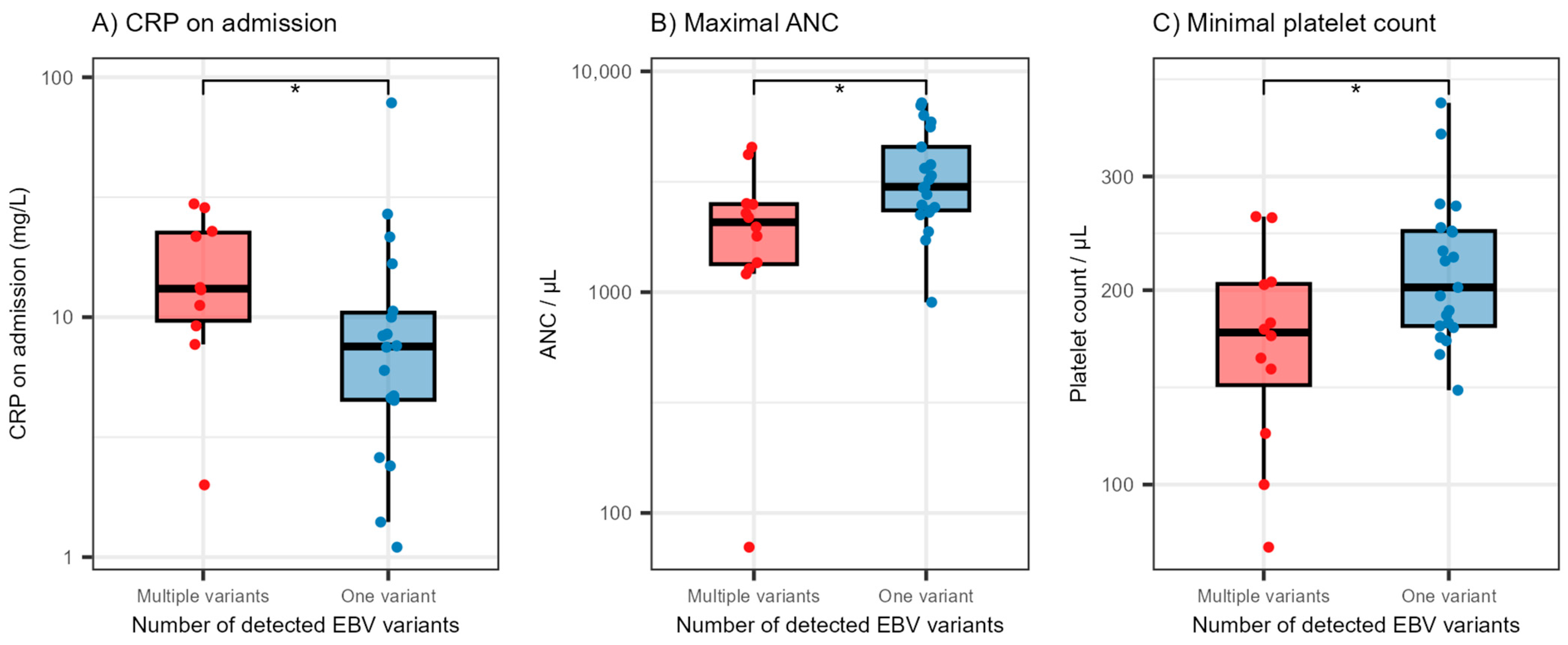
3.3. Association between EBV LMP1 Variants and Selected Laboratory Findings in IM
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- International Committee on Taxonomy of Viruses ICTV. Current ICTV Taxonomy Release: Taxon Details: Epstein-Barr Virus. 2022. Available online: https://ictv.global/taxonomy/taxondetails?taxnode_id=19780268 (accessed on 10 May 2023).
- Cao, Y.; Xie, L.; Shi, F.; Tang, M.; Li, Y.; Hu, J.; Bode, A.M. Targeting the signaling in Epstein–Barr virus-associated diseases: NCMechanism, regulation, and clinical study. Signal Transduct. Target. Ther. 2021, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- de Paor, M.; Boland, F.; Cai, X.; Smith, S.; Ebell, M.H.; Mac Donncha, E.; Fahey, T. Derivation and validation of clinical prediction rules for diagnosis of infectious mononucleosis: A prospective cohort study. BMJ Open 2023, 13, e068877. [Google Scholar] [CrossRef] [PubMed]
- Dunmire, S.K.; Hogquist, K.A.; Balfour, H.H. Infectious Mononucleosis. Curr. Top. Microbiol. Immunol. 2015, 390, 211–240. [Google Scholar]
- Shannon-Lowe, C.; Rickinson, A. The Global Landscape of EBV-Associated Tumors. Front. Oncol. 2019, 9, 713. [Google Scholar] [CrossRef]
- Bjornevik, K.; Cortese, M.; Healy, B.C.; Kuhle, J.; Mina, M.J.; Leng, Y.; Elledge, S.J.; Niebuhr, D.W.; Scher, A.I.; Munger, K.L.; et al. Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis. Science 2022, 375, 296–301. [Google Scholar] [CrossRef]
- Bjornevik, K.; Münz, C.; Cohen, J.I.; Ascherio, A. Epstein-Barr virus as a leading cause of multiple sclerosis: Mechanisms and implications. Nat. Rev. Neurol. 2023, 19, 160–171. [Google Scholar] [CrossRef]
- Rozman, M.; Korać, P.; Jambrosic, K.; Židovec Lepej, S. Progress in Prophylactic and Therapeutic EBV Vaccine Development Based on Molecular Characteristics of EBV Target Antigens. Pathogens 2022, 11, 864. [Google Scholar] [CrossRef] [PubMed]
- Palser, A.L.; Grayson, N.E.; White, R.E.; Corton, C.; Correia, S.; Ba Abdullah, M.M.; Watson, S.J.; Cotton, M.; Arrand, J.R.; Murray, P.G.; et al. Genome diversity of Epstein-Barr virus from multiple tumor types and normal infection. J. Virol. 2015, 89, 5222–5237. [Google Scholar] [CrossRef] [PubMed]
- Neves, M.; Marinho-Dias, J.; Ribeiro, J.; Sousa, H. Epstein-Barr virus strains and variations: Geographic or disease-specific variants? J. Med. Virol. 2017, 89, 373–387. [Google Scholar] [CrossRef]
- Wang, L.W.; Jiang, S.; Gewurz, B.E. Epstein-Barr Virus LMP1-Mediated Oncogenicity. J. Virol. 2017, 19, e01718-16. [Google Scholar] [CrossRef]
- Hulse, M.; Johnson, S.M.; Boyle, S.; Caruso, L.B.; Tempera, I. Epstein-Barr Virus-Encoded Latent Membrane Protein 1 and B-Cell Growth Transformation Induce Lipogenesis through Fatty Acid Synthase. J. Virol. 2021, 95, e01857-20. [Google Scholar] [CrossRef]
- Edwards, R.H.; Seillier-Moiseiwitsch, F.; Raab-Traub, N. Signature amino acid changes in latent membrane protein 1 distinguish Epstein-Barr virus strains. Virology 1999, 261, 79–95. [Google Scholar] [CrossRef]
- Gantuz, M.; Lorenzetti, M.A.; Chabay, P.A.; Preciado, M.V. A novel recombinant variant of latent membrane protein 1 from Epstein Barr virus in Argentina denotes phylogeographical association. PLoS ONE 2017, 12, e0174221. [Google Scholar] [CrossRef]
- Kaymaz, Y.; Oduor, C.I.; Aydemir, O.; Luftig, M.A.; Otieno, J.A.; Ong’echa, J.M.; Bailey, J.A.; Moormann, A.M. Epstein-Barr Virus Genomes Reveal Population Structure and Type 1 Association with Endemic Burkitt Lymphoma. J. Virol. 2020, 94, e02007–e02019. [Google Scholar] [CrossRef]
- van Baarle, D.M.; Hovenkamp, E.; Dukers, N.H.; Renwick, N.; Kersten, M.J.; Goudsmit, J.; Coutinho, R.A.; Miedema, F.; van Oers, M.H. High prevalence of Epstein-Barr virus type 2 among homosexual men is caused by sexual transmission. J. Infect. Dis. 2000, 181, 2045–2049. [Google Scholar] [CrossRef] [PubMed]
- Crawford, D.H.; Macsween, K.F.; Higgins, C.D.; Thomas, R.; McAulay, K.; Williams, H.; Harrison, N.; Reid, S.; Conacher, M.; Douglas, J.; et al. A cohort study among university students: Identification of risk factors for Epstein-Barr virus seroconversion and infectious mononucleosis. Clin. Infect. Dis. 2006, 43, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Higgins, C.D.; Swerdlow, A.J.; Macsween, K.F.; Harrison, N.; Williams, H.; McAulay, K.; Thomas, R.; Reid, S.; Conacher, M.; Britton, K.; et al. A study of risk factors for acquisition of Epstein-Barr virus and its subtypes. J. Infect. Dis. 2007, 195, 474–482. [Google Scholar] [CrossRef]
- Correia, S.; Palser, A.; Elgueta Karstegl, C.; Middeldorp, J.M.; Ramayanti, O.; Cohen, J.I.; Hildesheim, A.; Fellner, M.D.; Wiels, J.; White, R.E.; et al. Natural Variation of Epstein-Barr Virus Genes, Proteins, and Primary MicroRNA. J. Virol. 2017, 91, e00375-17. [Google Scholar] [CrossRef]
- Wang, Y.J.; Ba, Y.; Chen, Q.Y.; Han, Y.Q. Clinical Significance of Peripheral Blood EBV-DNA Determination and Genotyping in Lymphoma Patients. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2021, 29, 1802–1806. [Google Scholar] [PubMed]
- Palma, I.; Sánchez, A.E.; Jiménez-Hernández, E.; Alvarez-Rodríguez, F.; Nava-Frias, M.; Valencia-Mayoral, P.; Salinas-Lara, C.; Velazquez-Guadarrama, N.; Portilla-Aguilar, J.; Pena, R.Y.; et al. Detection of Epstein-Barr virus and genotyping based on EBNA2 protein in Mexican patients with hodgkin lymphoma: A comparative study in children and adults. Clin. Lymphoma Myeloma Leuk. 2013, 13, 266–272. [Google Scholar] [CrossRef]
- Salahuddin, S.; Khan, J.; Azhar, J.B.; Whitehurst, C.; Qadri, I.; Shackelford, J.; Pagano, J.S.; Muhammad, D.; Richards, K.L. Prevalence of Epstein–Barr Virus Genotypes in Pakistani Lymphoma Patients. Asian Pac. J. Cancer Prev. 2018, 19, 3153–3159. [Google Scholar]
- Kafita, D.; Kaile, T.; Malyangu, E.; Tembo, R.; Zulu, E.; Chisanga, C.; Kalonda, A.; Samutela, M.; Polepole, P.; Kwenda, G. Evidence of EBV infection in lymphomas diagnosed in Lusaka, Zambia. Pan Afr. Med. J. 2018, 29, 181. [Google Scholar] [CrossRef]
- Tabibzadeh, A.; Karbalaie Niya, M.H.; Esghaei, M.; Bokharaei-Salim, F.; Ataei-Pirkooh, A.; Kiani, S.J.; Monavari, S.H.R. Molecular Epidemiology of Epstein-Barr virus (EBV) in Patients with Hematologic Malignancies. Asian Pac. J. Cancer Prev. 2020, 21, 693–698. [Google Scholar] [CrossRef]
- Knecht, H.; Bachmann, E.; Brousset, P.; Sandvej, K.; Nadal, D.; Bachmann, F.; Odermatt, B.F.; Delsol, G.; Pallesen, G. Deletions within the LMP1 oncogene of Epstein-Barr virus are clustered in Hodgkin’s disease and identical to those observed in nasopharyngeal carcinoma. Blood 1993, 82, 2937–2942. [Google Scholar] [CrossRef] [PubMed]
- Sandvej, K.; Peh, S.; Andresen, B.; Pallesen, G. Identification of potential hot spots in the carboxy-terminal part of the Epstein-Barr virus (EBV) BNLF-1 gene in both malignant and benign EBV-associated diseases: High frequency of a 30-bp deletion in Malaysian and Danish peripheral T-cell lymphomas. Blood 1994, 84, 4053–4060. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.S.; Su, I.J.; Chung, P.J.; Shu, C.H.; Ng, C.K.; Wu, S.J.; Liu, S.T. Detection of an Epstein-Barr-virus variant in T-cell-lymphoma tissues identical to the distinct strain observed in nasopharyngeal carcinoma in the Taiwanese population. Int. J. Cancer 1995, 62, 673–677. [Google Scholar] [CrossRef]
- Khanim, F.; Yao, Q.Y.; Niedobitek, G.; Sihota, S.; Rickinson, A.B.; Young, L.S. Analysis of Epstein-Barr virus gene polymorphisms in normal donors and in virus-associated tumors from different geographic locations. Blood 1996, 88, 3491–3501. [Google Scholar] [CrossRef]
- Dirnhofer, S.; Angeles-Angeles, A.; Ortiz-Hidalgo, C.; Reyes, E.; Gredler, E.; Krugmann, J.; Fend, F.; Quintanilla-Martinez, L. High prevalence of a 30-base pair deletion in the Epstein-Barr virus (EBV) latent membrane protein 1 gene and of strain type B EBV in Mexican classical Hodgkin’s disease and reactive lymphoid tissue. Hum. Pathol. 1999, 30, 781–787. [Google Scholar] [CrossRef]
- Alves, P.; Larrate, M.; Garcia-Costa, A.; Rohan, P.; Gama, B.E.; Abdelhay, E.; Delatorre, E.; Hassan, R. Spatial Dispersal of Epstein-Barr Virus in South America Reveals an African American Variant in Brazilian Lymphomas. Viruses 2022, 12, 762. [Google Scholar] [CrossRef]
- Montes-Mojarro, I.A.; Chen, B.J.; Ramirez-Ibarguen, A.F.; Quezada-Fiallos, C.M.; Perez-Baez, W.B.; Duenas, D.; Casavilca Zambrano, S.; Ortiz-Mayor, M.; Rojas-Bilbao, E.; Garcia-Rivello, H.; et al. Mutational profile and EBV strains of extranodal NK/T-cell lymphoma, nasal type in Latin America. Mod. Pathol. 2020, 33, 781–791. [Google Scholar] [CrossRef]
- Begić, V.; Korać, P.; Gašparov, S.; Rozman, M.; Simicic, P.; Zidovec-Lepej, S. Molecular Characterisation of Epstein-Barr Virus in Classical Hodgkin Lymphoma. Int. J. Mol. Sci. 2022, 23, 15635. [Google Scholar] [CrossRef] [PubMed]
- Banko, A.; Lazarevic, I.; Cupic, M.; Stevanovic, G.; Boricic, I.; Jovanovic, T. Carboxy-terminal sequence variation of LMP1 gene in Epstein-Barr-virus-associated mononucleosis and tumors from Serbian patients. J. Med. Virol. 2012, 84, 632–642. [Google Scholar] [CrossRef] [PubMed]
- Banko, A.; Lazarevic, I.; Stevanovic, G.; Cirkovic, A.; Karalic, D.; Cupic, M.; Banko, B.; Milovanovic, J.; Jovanovic, T. Analysis of the Variability of Epstein-Barr Virus Genes in Infectious Mononucleosis: Investigation of the Potential Correlation with Biochemical Parameters of Hepatic Involvement. Med. Biochem. 2016, 35, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Mendes, T.M.; Oliveira, L.C.; Yamamoto, L.; Del Negro, G.M.; Okay, T.S. Epstein-Barr virus nuclear antigen-2 detection and typing in immunocompromised children correlated with lymphoproliferative disorder biopsy findings. Braz. J. Infect. Dis. 2008, 12, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Li, D.J.; Bei, J.X.; Mai, S.J.; Xu, J.F.; Chen, L.Z.; Zhang, R.H.; Yu, X.J.; Hong, M.H.; Zeng, Y.X.; Kang, T. The dominance of China 1 in the spectrum of Epstein-Barr virus strains from Cantonese patients with nasopharyngeal carcinoma. J. Med. Virol. 2009, 81, 1253–1260. [Google Scholar] [CrossRef]
- Guindon, S.; Gascuel, O. A simple, fast and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 2003, 52, 696–704. [Google Scholar] [CrossRef]
- Tavaré, S. Some probabilistic and statistical problems in the analysis of DNA sequences. Lect. Math. Life Sci. 1986, 17, 57–86. [Google Scholar]
- Walling, D.M.; Shebib, N.; Weaver, S.C.; Nichols, C.M.; Flaitz, C.M.; Webster-Cyriaque, J. The molecular epidemiology and evolution of Epstein-Barr virus: Sequence variation and genetic recombination in the latent membrane protein-1 gene. J. Infect. Dis. 1999, 179, 763–774. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; Scientific Research; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: https://www.R-project.org/ (accessed on 15 May 2022).
- Coleman, C.B.; Lang, J.; Sweet, L.A.; Smith, N.A.; Freed, B.M.; Pan, Z.; Haverkos, B.; Pelanda, R.; Rochford, R. Epstein-Barr Virus Type 2 Infects T Cells and Induces B Cell Lymphomagenesis in Humanized Mice. J. Virol. 2018, 92, e00813-18. [Google Scholar] [CrossRef]
- Simbiri, K.O.; Smith, N.A.; Otieno, R.; Wohlford, E.E.; Daud, I.I.; Odada, S.P.; Middleton, F.; Rochford, R. Epstein-Barr virus genetic variation in lymphoblastoid cell lines derived from Kenyan pediatric population. PLoS ONE 2015, 10, e0125420. [Google Scholar] [CrossRef]
- Coleman, C.B.; Wohlford, E.M.; Smith, N.A.; King, C.A.; Ritchie, J.A.; Baresel, P.C.; Kimura, H.; Rochford, R. Epstein-Barr virus type 2 latently infects T cells, inducing an atypical activation characterized by expression of lymphotactic cytokines. J. Virol. 2015, 89, 2301–2312. [Google Scholar] [CrossRef] [PubMed]
- Coleman, C.B.; Daud, I.I.; Ogolla, S.O.; Ritchie, J.A.; Smith, N.A.; Sumba, P.O.; Dent, A.E.; Rochford, R. Epstein-Barr Virus Type 2 Infects T Cells in Healthy Kenyan Children. J. Infect. Dis. 2017, 216, 670–677. [Google Scholar] [CrossRef] [PubMed]
- Sproston, N.R.; Ashworth, J.J. Role of C-Reactive Protein at Sites of Inflammation and Infection. Front. Immunol. 2018, 9, 754. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.; Asprusten, T.T.; Godang, K.; Leegaard, T.M.; Osnes, L.T.; Skovlund, E.; Tjade, T.; Øie, M.G.; Wyller, V.B.B. Predictors of chronic fatigue in adolescents six months after acute Epstein-Barr virus infection: A prospective cohort study. Brain Behav. Immun. 2019, 75, 94–100. [Google Scholar] [CrossRef]
- Kristiansen, M.S.; Stabursvik, J.; O’Leary, E.C.; Pedersen, M.; Asprusten, T.T.; Leegaard, T.; Osnes, L.T.; Tjade, T.; Skovlund, E.; Godang, K.; et al. Clinical symptoms and markers of disease mechanisms in adolescent chronic fatigue following Epstein-Barr virus infection: An exploratory cross-sectional study. Brain Behav. Immun. 2019, 80, 551–563. [Google Scholar] [CrossRef]
- Páez-Guillán, E.M.; Campos-Franco, J.; Alende, R.; Gonzalez-Quintela, A. Hematological Abnormalities Beyond Lymphocytosis During Infectious Mononucleosis: Epstein-Barr Virus-Induced Thrombocytopenia. Mediterr. J. Hematol. Infect. Dis. 2023, 15, e2023023. [Google Scholar]
- Hammond, W.P.; Harlan, J.M.; Steinberg, S.E. Severe neutropenia in infectious mononucleosis. West. J. Med. 1979, 131, 92–97. [Google Scholar]
- Zhang, Y.; Huang, C.; Zhang, H.; Duan, Z.; Liu, Q.; Li, J.; Zong, Q.; Wei, Y.; Liu, F.; Duan, W.; et al. Characteristics of immunological events in Epstein-Barr virus infection in children with infectious mononucleosis. Front. Pediatr. 2023, 11, 1060053. [Google Scholar] [CrossRef]
- Rzymski, P.; Szuster-Ciesielska, A.; Dzieciątkowski, T.; Gwenzi, W.; Fal, A. mRNA vaccines: The future of prevention of viral infections? J. Med. Virol. 2023, 95, e28572. [Google Scholar] [CrossRef]
- Guo, M.; Duan, X.; Peng, X.; Jin, Z.; Huang, H.; Xiao, W.; Zheng, Q.; Deng, Y.; Fan, N.; Chen, K.; et al. A lipid-based LMP2-mRNA vaccine to treat nasopharyngeal carcinoma. Nano Res. 2023, 16, 5357–5367. [Google Scholar] [CrossRef]
| Gene | Primers |
|---|---|
| EBNA-2 (PCR) | F: 5′-AGGGATGCCTGGACACAAGA-3′ R: 5′-TGGTGCTGCTGGTG GTGGCAAT-3′ |
| EBNA-2 (nested PCR, EBV type 1) | F: 5′-TCTTGATAGGGATCCGCTAGGATA-3′ R: 5′-ACCGTGGTTCTGGACTATCTGGATC-3′ |
| EBNA-2 (nested PCR, EBV type 2) | F: 5’-CATGGTAGCCTTAGGACATA-3’ R: 5’-AGACTTAGTTGATGCCCTAG-3’ |
| LMP1 | F: 5’-GCT AAG GCA TTC CCA GTA AA-3’ R:5’-GAT GAA CAC CAC CAC GAT G-3’ |
| EBV LMP1 Variant | Number of 33 Base Pair Tandem Repeats | Deletions in the LMP1 Gene | ||
|---|---|---|---|---|
| 3–4.5 | 5–7 | |||
| B95-8 Wild-Type | 10 | - | No deletion | 10 |
| China1 | 2 | 13 | 30 base pair deletions | 15 |
| Mediterranean | 5 | 6 | No deletion | 9 |
| 30 base pair deletion | 1 | |||
| 69 base pair deletion | 1 | |||
| North Carolina | 3 | 1 | No deletion | 4 |
| Total | 20 | 20 | ||
| aa Position | 212 | 212 | 213 | 214 | 214 | 214 | 218 | 221 | 224 | 229 | 231 2 | 240 | 248 | 250 | 250 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B95-8 Prototype 1 | G | G | H | E | E | E | N | E | H | S | A | Q | G | D | D |
| aa Change | S | H | Q | K | Q | G | T | D | Q | T | S | R | A | N | E |
| Number of Cases in Croatian IM Isolates | |||||||||||||||
| B95-8 | 7 | 1 | 1 | 1 | 1 | ||||||||||
| China1 | 8 | 1 | 2 | 1 | 1 | 1 | |||||||||
| Mediterranean | 7 | 1 | 1 | 1 | 1 | 11 | 1 | 1 | 1 | ||||||
| North Carolina | 3 | 4 | |||||||||||||
| aa Position | 252 | 255 | 262 | 266 | 267 2 | 270 | 282 | 293 | 306 | 309 | 313 | 314 | 315 2 | 317 | 317 |
| B95-8 Prototype 1 | G | D | N | D | P | T | D | D | L | S | S | A | G | D | D |
| aa change | A | G | S | G | R | A | G | G | Q | N | P | G | E | E | N |
| Number of Cases in Croatian IM Isolates | |||||||||||||||
| B95-8 | 1 | 1 | 1 | ||||||||||||
| China1 | 15 | ||||||||||||||
| Mediterranean | 4 | 1 | 2 | 5 | 9 | 11 | 1 | 1 | 1 | 1 | |||||
| North Carolina | 1 | 4 | 4 | 4 | |||||||||||
| aa Position | 319 | 321 2 | 322 | 322 | 322 | 322 | 325 | 328 | 329 | 330 | 331 | 333 | 333 | 334 | 335 |
| B95-8 Prototype 1 | G | P | Q | Q | Q | Q | E | E | N | K | G | D | D | Q | G |
| aa Change | D | T | E | T | N | D | K | Q | I | T | Q | A | G | R | D |
| Number of Cases in Croatian IM Isolates | |||||||||||||||
| B95-8 | 1 | 1 | 10 | ||||||||||||
| China1 | 15 | 1 | 15 | ||||||||||||
| Mediterranean | 2 | 9 | 1 | 1 | 1 | 2 | 11 | ||||||||
| North Carolina | 4 | 4 | 1 | ||||||||||||
| aa Position | 335 | 338 | 338 | 349 | 352 | 352 | 352 | 356 | 356 | 356 | 357 | 358 | 359 | 361 | 361 |
| B95-8 Prototype 1 | G | L | L | D | H | H | H | D | D | D | P | H | L | T | T |
| aa Change | S | S | P | A | S | R | N | H | A | N | S | P | V | M | R |
| Number of Cases in Croatian IM Isolates | |||||||||||||||
| B95-8 | 3 | 3 | 1 | 2 | 2 | 1 | 2 | ||||||||
| China1 | 13 | 1 | 1 | ||||||||||||
| Mediterranean | 1 | 11 | 4 | 1 | 8 | ||||||||||
| North Carolina | 4 | 4 | 4 | ||||||||||||
| Selected Parameters | Infectious Mononucleosis Patients (Number of Patients, Percentage) |
|---|---|
| Number of patients | 33 |
| Gender distribution (number of males, percentage) | 15 males (45.5%) |
| Age (mean, range) | 11.9 years (0.89–17.83) |
| Hospitalized patients (number, percentage) | 7 (21.2%) |
| Days since symptom onset at clinical presentation (median, interquartile range) | 5 (3–7) |
| Clinical symptoms (number, percentage) | |
| Pharyngitis | 30 (90.1%) |
| Lymphadenitis | 25 (75.8%) |
| Respiratory symptoms | 21 (63.6%) |
| Hepatosplenomegaly | 12 (36.4%) |
| Upper eyelid edema | 6 (18.2%) |
| Periglandural edema | 6 (18.2%) |
| Rash | 3 (9.1%) |
| EBV LMP1 variant distribution | |
| Wild-type | 14 (42.4%) |
| Wild-type/China1 co-infection | 8 (24.2%) |
| North Carolina | 4 (12.1%) |
| Wild-type/North Carolina co-infection | 4 (12.1%) |
| Mediterranean | 3 (9.1%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zidovec-Lepej, S.; Batovic, M.; Rozman, M.; Bodulić, K.; Prtorić, L.; Šokota, A.; Nikcevic, A.; Simicic, P.; Tešović, G. Distribution of Epstein–Barr Virus LMP1 Variants in Patients with Infectious Mononucleosis and Association with Selected Biochemical and Hematological Parameters. Pathogens 2023, 12, 915. https://doi.org/10.3390/pathogens12070915
Zidovec-Lepej S, Batovic M, Rozman M, Bodulić K, Prtorić L, Šokota A, Nikcevic A, Simicic P, Tešović G. Distribution of Epstein–Barr Virus LMP1 Variants in Patients with Infectious Mononucleosis and Association with Selected Biochemical and Hematological Parameters. Pathogens. 2023; 12(7):915. https://doi.org/10.3390/pathogens12070915
Chicago/Turabian StyleZidovec-Lepej, Snjezana, Margarita Batovic, Marija Rozman, Kristian Bodulić, Laura Prtorić, Ante Šokota, Andrea Nikcevic, Petra Simicic, and Goran Tešović. 2023. "Distribution of Epstein–Barr Virus LMP1 Variants in Patients with Infectious Mononucleosis and Association with Selected Biochemical and Hematological Parameters" Pathogens 12, no. 7: 915. https://doi.org/10.3390/pathogens12070915
APA StyleZidovec-Lepej, S., Batovic, M., Rozman, M., Bodulić, K., Prtorić, L., Šokota, A., Nikcevic, A., Simicic, P., & Tešović, G. (2023). Distribution of Epstein–Barr Virus LMP1 Variants in Patients with Infectious Mononucleosis and Association with Selected Biochemical and Hematological Parameters. Pathogens, 12(7), 915. https://doi.org/10.3390/pathogens12070915





