A Randomized, Double-Blind, Placebo-Controlled, Multicenter Study to Evaluate the Safety and Efficacy of ThymoQuinone Formula (TQF) for Treating Outpatient SARS-CoV-2
Abstract
1. Introduction
2. Results
Clinical Study Results
3. Discussion
4. Materials and Methods
4.1. Clinical Study Design
4.2. Participants
4.3. Randomization and Masking
4.4. Procedures
4.5. Outcomes
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Salem, E.M.; Yar, T.; Bamosa, A.O.; Al-Quorain, A.; Yasawy, M.I.; Alsulaiman, R.M.; Randhawa, M.A. Comparative study of Nigella Sativa and triple therapy in eradication of Helicobacter Pylori in patients with non-ulcer dyspepsia. Saudi J. Gastroenterol. 2010, 16, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.S.; Riffat, S. Field trial of Saussurea lappa roots against nematodes and Nigella sativa seeds against cestodes in children. J. Pak. Med. Assoc. 1991, 41, 185–187. [Google Scholar] [PubMed]
- Fard, F.A.; Zahrani, S.T.; Bagheban, A.A.; Mojab, F. Therapeutic Effects of Nigella Sativa Linn (Black Cumin) on Candida albicans Vaginitis. Arch. Clin. Infect. Dis. 2015, 10, e22991. [Google Scholar] [CrossRef]
- Barakat, E.M.; El Wakeel, L.M.; Hagag, R.S. Effects of Nigella sativa on outcome of hepatitis C in Egypt. World J. Gastroenterol. 2013, 19, 2529–2536. [Google Scholar] [CrossRef] [PubMed]
- Onifade, A.A.; Jewell, A.P.; Adedeji, W.A. Nigella sativa concoction induced sustained seroreversion in HIV patient. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 332–335. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Onifade, A.A.; Jewell, A.P.; Okesin, A.B. Seronegative conversion of an hiv positive subject treated with Nigella sativa and honey. Afr. J. Infect. Dis. 2015, 9, 47–50. [Google Scholar] [CrossRef]
- Rahman, M.T. Potential benefits of combination of Nigella sativa and Zn supplements to treat COVID-19. J. Herb. Med. 2020, 23, 100382. [Google Scholar] [CrossRef]
- Koshak, A.E.; Koshak, E.A.; Mobeireek, A.F.; Badawi, M.A.; Wali, S.O.; Malibary, H.M.; Atwah, A.F.; Alhamdan, M.M.; Almalki, R.A.; Madani, T.A. Nigella sativa for the treatment of COVID-19: An open-label randomized controlled clinical trial. Complement. Ther. Med. 2021, 61, 102769. [Google Scholar] [CrossRef]
- Tenforde, M.W.; Kim, S.S.; Lindsell, C.J.; Rose, E.B.; Shapiro, N.I.; Files, D.C.; Gibbs, K.W.; Erickson, H.L.; Steingrub, J.S.; Smithline, H.A.; et al. Symptom Duration and Risk Factors for Delayed Return to Usual Health Among Outpatients with COVID-19 in a Multistate Health Care Systems Network—United States, March–June 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 993–998. [Google Scholar] [CrossRef]
- Richard, S.A.; Epsi, N.; Pollett, S.; Lindholm, D.A.; Malloy, A.M.W.; Maves, R.; Utz, G.C.; Lalani, T.; Smith, A.G.; Mody, R.M.; et al. Performance of the inFLUenza Patient-Reported Outcome Plus (FLU-PRO Plus©) instrument in patients with COVID-19. Open Forum Infect. Dis. 2021, 8, ofab517. [Google Scholar] [CrossRef]
- Java, A.; Apicelli, A.J.; Liszewski, M.K.; Coler-Reilly, A.; Atkinson, J.P.; Kim, A.H.; Kulkarni, H.S. The complement system in COVID-19: Friend and foe? JCI Insight 2020, 5, e140711. [Google Scholar] [CrossRef] [PubMed]
- Lowery, S.A.; Sariol, A.; Perlman, S. Innate immune and inflammatory responses to SARS-CoV-2: Implications for COVID-19. Cell Host Microbe 2021, 29, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, L.; Mescia, F.; Turner, L.; Hanson, A.L.; Kotagiri, P.; Dunmore, B.J.; Ruffieux, H.; De Sa, A.; Huhn, O.; Morgan, M.D.; et al. Longitudinal analysis reveals that delayed bystander CD8+ T cell activation and early immune pathology distinguish severe COVID-19 from mild disease. Immunity 2021, 54, 1257–1275.e1258. [Google Scholar] [CrossRef] [PubMed]
- Kreutmair, S.; Unger, S.; Núñez, N.G.; Ingelfinger, F.; Alberti, C.; De Feo, D.; Krishnarajah, S.; Kauffmann, M.; Friebel, E.; Babaei, S.; et al. Distinct immunological signatures discriminate severe COVID-19 from non-SARS-CoV-2-driven critical pneumonia. Immunity 2021, 54, 1578–1593.e1575. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Martin, J.C. Pathological inflammation in patients with COVID-19: A key role for monocytes and macrophages. Nat. Rev. Immunol. 2020, 20, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Kaseb, A.O.; Mohamed, Y.I.; Malek, A.E.; Raad, I.I.; Altameemi, L.; Li, D.; Kaseb, O.A.; Kaseb, S.A.; Selim, A.; Ma, Q. The Impact of Angiotensin-Converting Enzyme 2 (ACE2) Expression on the Incidence and Severity of COVID-19 Infection. Pathogens 2021, 10, 379. [Google Scholar] [CrossRef]
- Ciaglia, E.; Vecchione, C.; Puca, A.A. COVID-19 Infection and Circulating ACE2 Levels: Protective Role in Women and Children. Front. Pediatr. 2020, 8, 206. [Google Scholar] [CrossRef]
- Li, M.-Y.; Li, L.; Zhang, Y.; Wang, X.-S. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect. Dis. Poverty 2020, 9, 45. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; Place, S.; Van Laethem, Y.; Cabaraux, P.; Mat, Q.; Huet, K.; Plzak, J.; Horoi, M.; Hans, S.; et al. Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate coronavirus disease 2019. J. Intern. Med. 2020, 288, 335–344. [Google Scholar] [CrossRef]
- Wu, J.; Li, W.; Shi, X.; Chen, Z.; Jiang, B.; Liu, J.; Wang, D.; Liu, C.; Meng, Y.; Cui, L.; et al. Early antiviral treatment contributes to alleviate the severity and improve the prognosis of patients with novel coronavirus disease (COVID-19). J. Intern. Med. 2020, 288, 128–138. [Google Scholar] [CrossRef]
- Yu, F.; Yan, L.; Wang, N.; Yang, S.; Wang, L.; Tang, Y.; Gao, G.; Wang, S.; Ma, C.; Xie, R.; et al. Quantitative Detection and Viral Load Analysis of SARS-CoV-2 in Infected Patients. Clin. Infect. Dis. 2020, 71, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Bi, Q.; Wu, Y.; Mei, S.; Ye, C.; Zou, X.; Zhang, Z.; Liu, X.; Wei, L.; Truelove, S.A.; Zhang, T.; et al. Epidemiology and transmission of COVID-19 in 391 cases and 1286 of their close contacts in Shenzhen, China: A retrospective cohort study. Lancet Infect. Dis. 2020, 20, 911–919. [Google Scholar] [CrossRef]
- Tolossa, T.; Wakuma, B.; Gebre, D.S.; Atomssa, E.M.; Getachew, M.; Fetensa, G.; Ayala, D.; Turi, E. Time to recovery from COVID-19 and its predictors among patients admitted to treatment center of Wollega University Referral Hospital (WURH), Western Ethiopia: Survival analysis of retrospective cohort study. PLoS ONE 2021, 16, e0252389. [Google Scholar] [CrossRef] [PubMed]
- SeyedAlinaghi, S.; Abbasian, L.; Solduzian, M.; Yazdi, N.A.; Jafari, F.; Adibimehr, A.; Farahani, A.; Khaneshan, A.S.; Alavijeh, P.E.; Jahani, Z.; et al. Predictors of the prolonged recovery period in COVID-19 patients: A cross-sectional study. Eur. J. Med. Res. 2021, 26, 41. [Google Scholar] [CrossRef] [PubMed]
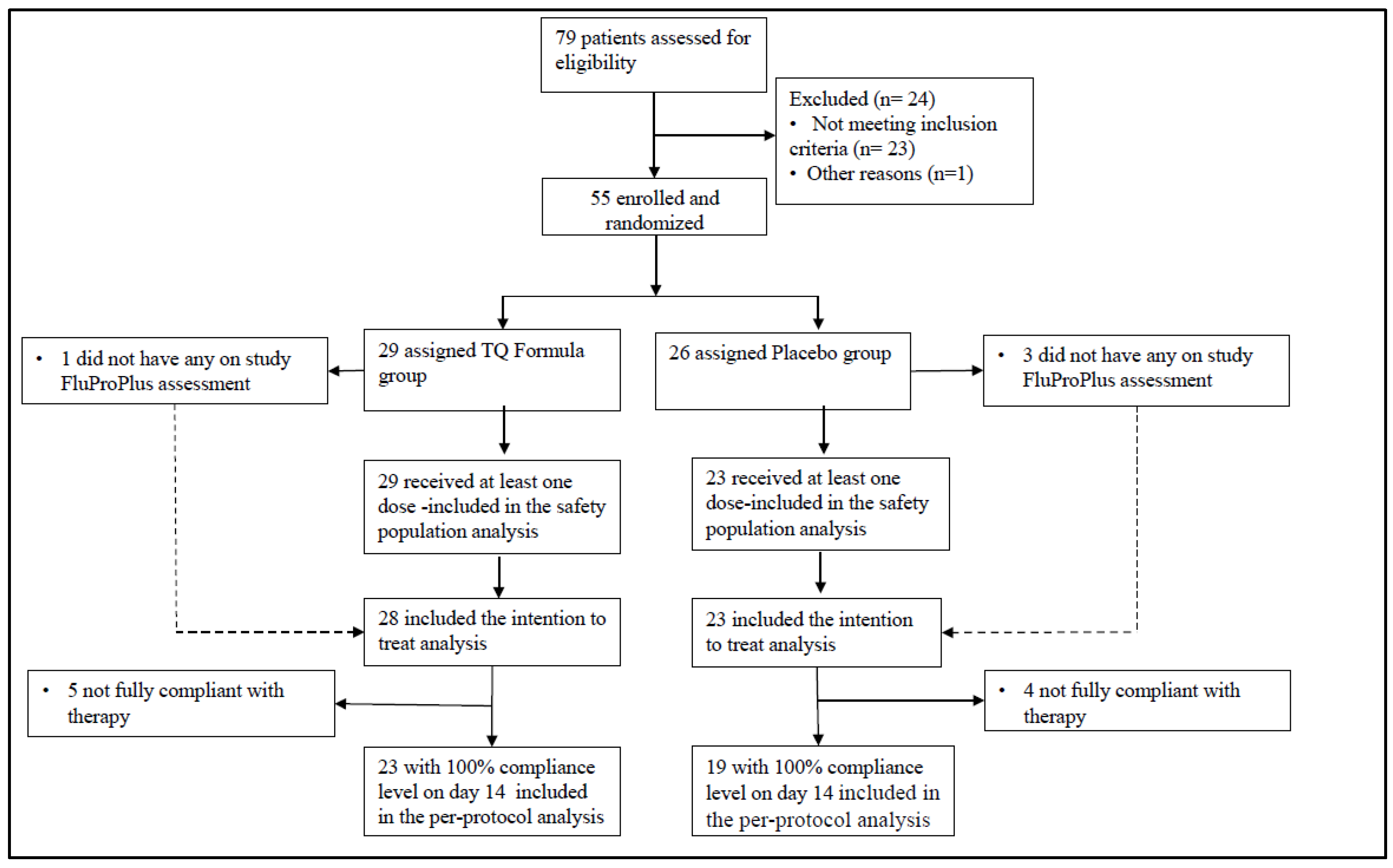
| All | TQ Formula | Placebo | ||
|---|---|---|---|---|
| N (%) | N (%) | N (%) | ||
| Sex | ||||
| Female | 31 (56.36) | 16 (55.17) | 15 (57.69) | |
| Male | 24 (43.64) | 13 (44.83) | 11 (42.31) | |
| Race/Ethnicity | ||||
| Asian | Non-Hispanic/Latino | 1 (1.82) | 1 (3.45) | |
| Black or African American | Hispanic/Latino | 3 (5.45) | 1 (3.45) | 2 (7.69) |
| Non-Hispanic/Latino | 4 (7.27) | 1 (3.45) | 3 (11.54) | |
| White | Hispanic/Latino | 20 (36.36) | 11 (37.93) | 9 (34.62) |
| Non-Hispanic/Latino | 5 (9.09) | 2 (6.9) | 3 (11.54) | |
| Others | Hispanic/Latino | 22 (40.00) | 13 (44.83) | 9 (34.62) |
| Age | ||||
| Years (Mean ± SD) | 45.69 ± 17.35 | 45.48 ± 19.29 | 45.92 ± 15.27 | |
| ≤55 years | 39 (70.91) | 21 (72.41) | 18 (69.23) | |
| >55 years | 16 (29.09) | 8 (27.5) | 8 (30.76) | |
| Common Comorbidities | ||||
| Diabetes Mellitus 2 | 10 (18.18) | 5 (17.24) | 5 (19.23) | |
| Hypertension | 22 (40.00) | 11 (37.93) | 11 (42.31) | |
| BMI | ||||
| Underweight | 1 (1.82) | 1 (3.45) | ||
| Normal weight | 9 (16.36) | 2 (6.90) | 7 (26.92) | |
| Overweight | 24 (43.64) | 14 (48.28) | 10 (38.46) | |
| Obese | 21 (38.18) | 12 (41.38) | 9 (34.62) | |
| SARS-CoV-2 Vaccination * | 9 (16.36) | 7 (24.13) | 2 (7.69) | |
| N | Min | Q1 | Median | Q3 | Max | Mean | SD | p-Value * | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Viral Load Day 0 | Placebo | 23 | 59 | 1683 | 25,000 | 25,000 | 25,000 | 15,418 | 11,465 | 0.69 |
| TQ Formula | 25 | 87 | 9575 | 25,000 | 25,000 | 25,000 | 17,819 | 9991 | ||
| Viral Load Day 7 | Placebo | 18 | 0 | 0 | 12,282 | 25,000 | 25,000 | 12,479 | 12,539 | 0.81 |
| TQ Formula | 23 | 0 | 0 | 216 | 25,000 | 25,000 | 10,674 | 12,407 | ||
| Viral Load Day 14 | Placebo | 19 | 0 | 0 | 0 | 25,000 | 25,000 | 9056 | 11,907 | 0.18 |
| TQ Formula | 21 | 0 | 0 | 0 | 0 | 25,000 | 4274 | 8513 | ||
| Objectives/Purpose | Endpoints/Outcome Measures | Justification and Results for Endpoints |
|---|---|---|
| Primary | ||
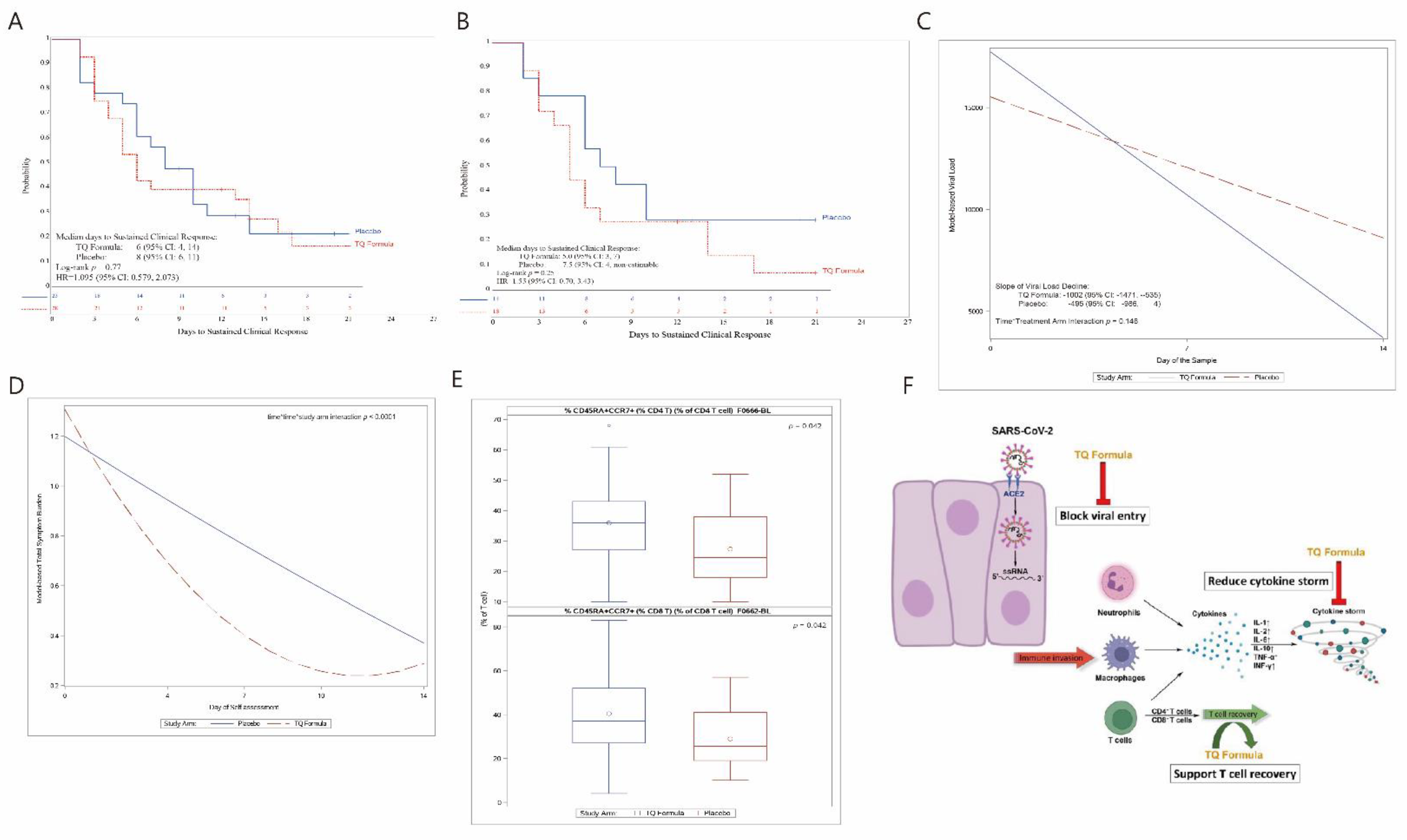
| To evaluate if treatment with 3 g TQ Formula (500 mg per capsule, 3 capsules BID) given orally on outpatient basis can significantly reduce median time-to-sustained-clinical-response compared to placebo in participants with COVID-19 infection treated in the outpatient setting. | Measurement of the difference in median time-to-sustained-clinical-response in participants taking 3 g TQ Formula (500 mg per capsule, 3 capsules BID) versus participants taking placebo. Sustained clinical response is defined as a reduction of scores to ≤2 on all symptoms of the Modified FLU-PRO Plus. | A reduction in time-to-sustained-clinical-response is a direct measure of treatment effectiveness. Results: median time-to-SCR of 6 days in TQ Formula arm (95% CI: 4.14) versus 8 days in placebo arm (95% CI: 6.11). (p = 0.77). |
| To evaluate the safety and tolerability of TQ Formula (500 mg oral capsule, 3 capsules BID) when given to participants with COVID-19 infection. | Number of overall adverse events, related adverse reactions, and hospitalizations reported in participants taking 3 g TQ Formula (500 mg per capsule, 3 capsules BID) versus participants taking placebo. All AEs/SAEs will be captured throughout the study as per schedule of assessments. | Comparing the number of AEs and SAEs and any relationship to IP is important for determining the safety profile of TQ Formula. Results: TQF arm had no SAEs and had 50% less AE incidence compared to placebo while this difference was not significant (p = 0.16). |
| Secondary | ||
| To compare the viral load profile over time (from baseline through to day 14) between treatment with 3 g TQ Formula (500 mg per capsule, 3 capsules BID) given orally on outpatient basis and placebo in participants with COVID-19 infection. | Measurement of change in quantitative viral load from baseline, day 7, and day 14 using RT-PCR in participants taking 3 g TQ Formula (500 mg per capsule, 3 capsules BID) versus participants taking placebo with COVID-19 infection. | Faster decline in viral load is hypothesized to lead to faster recovery from the illness and less infectivity. Results: trend of a faster decline in viral load with TQ formula treatment (p = 0.146). |
| To compare the percentage of RT-PCR negative/undetectable (i.e., viral clearance) on day 7 and day 14 in participants taking 3 g TQ Formula (500 mg per capsule, 3 capsules BID) versus participants taking placebo. | Percentage of negative/undetectable RT-PCR (i.e., viral clearance) on day 7 and day 14 in participants taking 3 g TQ Formula (500 mg per capsule, 3 capsules BID) versus participants taking placebo. | Treatment with 3 g TQ Formula (500 mg per capsule, 3 capsules BID) given orally on outpatient basis is hypothesized to result in higher percentages of viral clearance by RT-PCR Faster decline in viral load is hypothesized to lead to faster recovery from the illness and less infectivity. Results: Lower percentage of RT-PCR positive cases in TQ Formula arm on day 14 (24% vs. 42%, p = 0.22). |
| To compare the duration and severity of symptoms (measured by Modified FLU-PRO Plus) overtime from day 1 through day 14 in total Modified FLU-PRO Plus symptom severity score overall and in sub-domain scores (namely, Nose, Throat, Eyes, Chest/Respiratory, Gastrointestinal, Body/Systemic, Taste/Smell), between treatment with 3 g TQ Formula (500 mg per capsule, 3 capsules BID) given orally on outpatient basis and placebo in participants with COVID-19 infection. | Measurement of severity of and change in COVID-19 symptoms per total score as well as sub-scores (Nose, Throat, Eyes, Chest/Respiratory, Gastrointestinal, Body/Systemic, Taste/Smell) measured through Modified FLU-PRO Plus from day 1 through day 14 in participants with COVID-19 infection treated either with 3 g TQ Formula (500 mg per capsule, 3 capsules BID) or placebo. | FLU-PRO is a validated measure that has been used on multiple virus studies. The Modified FLU-PRO Plus version has additional questions (Taste/Smell) that are COVID-19 specific. The Modified FLU-PRO Plus has been shortened to reduce number of symptoms. Results: oral TQ Formula treated arm led to significantly faster decline in overall symptom burden from day 1 through day 14 as compared to placebo treated arm (p < 0.0001) as well as in throat (p = 0.0003), body/systemic (p = 0.0011), Chest/Respiratory (p = 0.024), and Smell/Taste (p = 0.032) subdomains. |
| To investigate if there exists an association between viral load and symptom severity by study arm and if such associations change overtime. | Correlation Coefficient of quantitative viral load and symptom severity at baseline, at day 7, and day 14 in participants taking 3 g TQ Formula (500 mg per capsule, 3 capsules BID) versus participants taking placebo | Decreases in viral load are hypothesized to be correlated with better clinical outcomes. Results: raw baseline Viral Load measure is negatively associated with total symptom burden (Spearman’s rank correlation = −0.30, p = 0.037). This negative rank-association does not remain significant on day 7 (p = 0.48) while show significance on day 14 (Spearman’s rank correlation = −0.46, p = 0.0031). |
| Tertiary/Exploratory | ||
| To evaluate the basic pharmacokinetics of TQ Formula’s main active ingredient (thymoquinone) at same time points (Days 1, 7, and 14) in participants with COVID-19 infection. | Measurement of thymoquinone and metabolites’ concentration in the plasma on day 1, day 7 and 14 using HPLC in patients treated with TQ Formula. | Thymoquinone is the main active ingredient of TQ Formula and the pharmacokinetics of thymoquinone and its’ metabolites in the plasma of treated patients are used to correlate with effectiveness of treatment. Results: analysis ongoing. |
| To explore the effect of TQ Formula on inflammatory cytokines, coagulation factors and effector immune cells at same time points (Days 1, 7, and 14) in participants with COVID-19 infection. | Measurement of the inflammatory cytokine production, coagulation factors and the various effector immune cell subsets in the Peripheral Blood Mononuclear Cells (PBMC) of these patients on day 1, day 7 and day 14 using FACS. | The inflammatory cytokines and immunological markers are of importance because of their correlation with disease severity in COVID-19. Results: Statistically significant increases in CD4 and CD8 T-cell Percentages on day 14 (p = 0.042 for both). |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bencheqroun, H.; Ahmed, Y.; Kocak, M.; Villa, E.; Barrera, C.; Mohiuddin, M.; Fortunet, R.; Iyoha, E.; Bates, D.; Okpalor, C.; et al. A Randomized, Double-Blind, Placebo-Controlled, Multicenter Study to Evaluate the Safety and Efficacy of ThymoQuinone Formula (TQF) for Treating Outpatient SARS-CoV-2. Pathogens 2022, 11, 551. https://doi.org/10.3390/pathogens11050551
Bencheqroun H, Ahmed Y, Kocak M, Villa E, Barrera C, Mohiuddin M, Fortunet R, Iyoha E, Bates D, Okpalor C, et al. A Randomized, Double-Blind, Placebo-Controlled, Multicenter Study to Evaluate the Safety and Efficacy of ThymoQuinone Formula (TQF) for Treating Outpatient SARS-CoV-2. Pathogens. 2022; 11(5):551. https://doi.org/10.3390/pathogens11050551
Chicago/Turabian StyleBencheqroun, Hassan, Yasir Ahmed, Mehmet Kocak, Enrique Villa, Cesar Barrera, Mariya Mohiuddin, Raul Fortunet, Emmanuel Iyoha, Deborah Bates, Chinedu Okpalor, and et al. 2022. "A Randomized, Double-Blind, Placebo-Controlled, Multicenter Study to Evaluate the Safety and Efficacy of ThymoQuinone Formula (TQF) for Treating Outpatient SARS-CoV-2" Pathogens 11, no. 5: 551. https://doi.org/10.3390/pathogens11050551
APA StyleBencheqroun, H., Ahmed, Y., Kocak, M., Villa, E., Barrera, C., Mohiuddin, M., Fortunet, R., Iyoha, E., Bates, D., Okpalor, C., Agbosasa, O., Mohammed, K., Pondell, S., Mohamed, A., Mohamed, Y. I., Gok Yavuz, B., Kaseb, M. O., Kasseb, O. O., Gocio, M. Y., ... Kaseb, A. O. (2022). A Randomized, Double-Blind, Placebo-Controlled, Multicenter Study to Evaluate the Safety and Efficacy of ThymoQuinone Formula (TQF) for Treating Outpatient SARS-CoV-2. Pathogens, 11(5), 551. https://doi.org/10.3390/pathogens11050551






