Phenotypic Selection of Dairy Cattle Infected with Bovine Leukemia Virus Demonstrates Immunogenetic Resilience through NGS-Based Genotyping of BoLA MHC Class II Genes
Abstract
:1. Introduction
2. Results
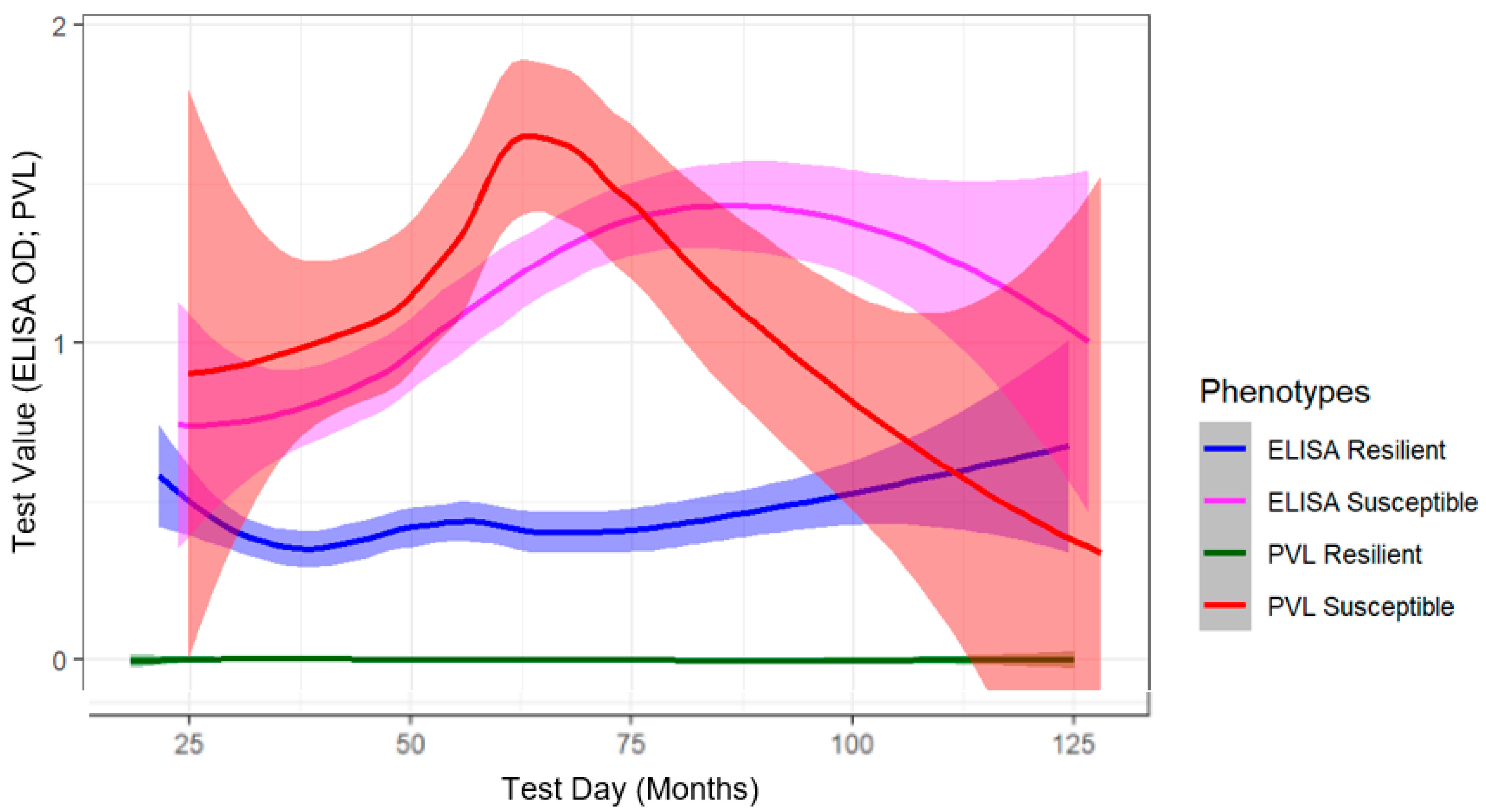
2.1. Phenotype Determination Using Longitudinal Diagnostic Testing of BLV-Infected Cows
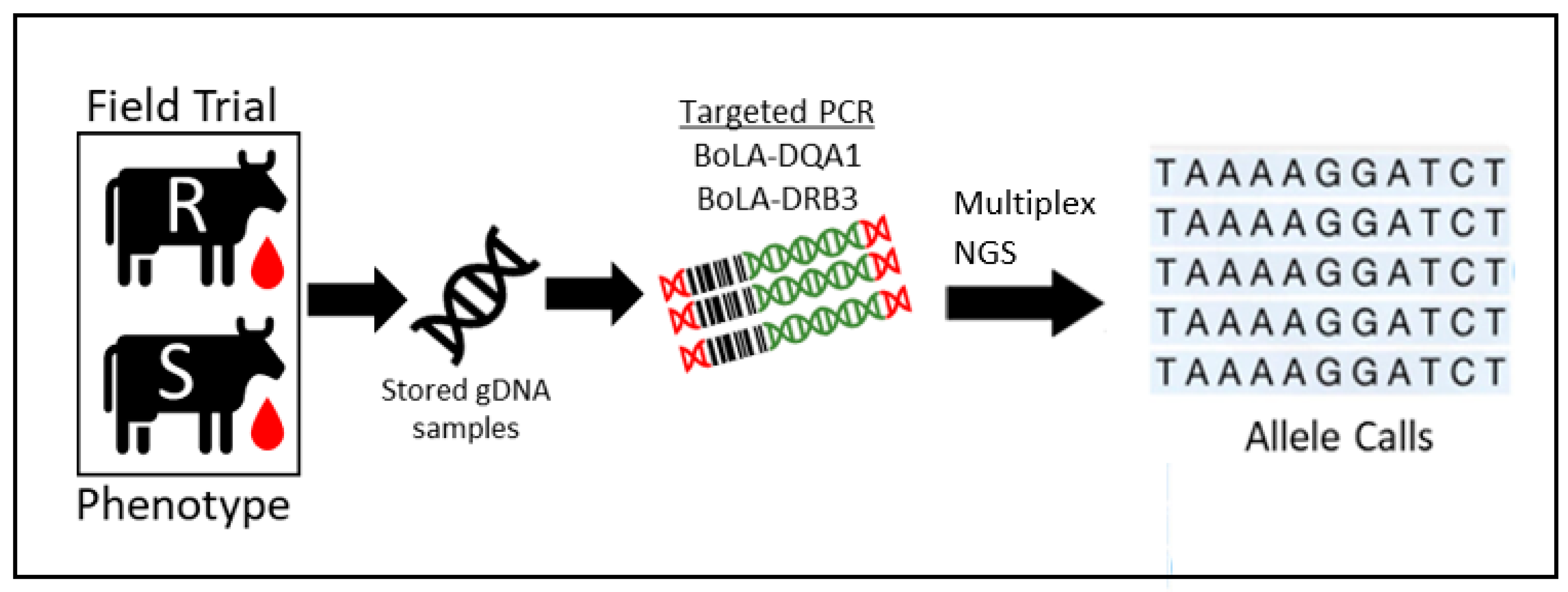
2.2. Development of Multiplex Sequencing Approach
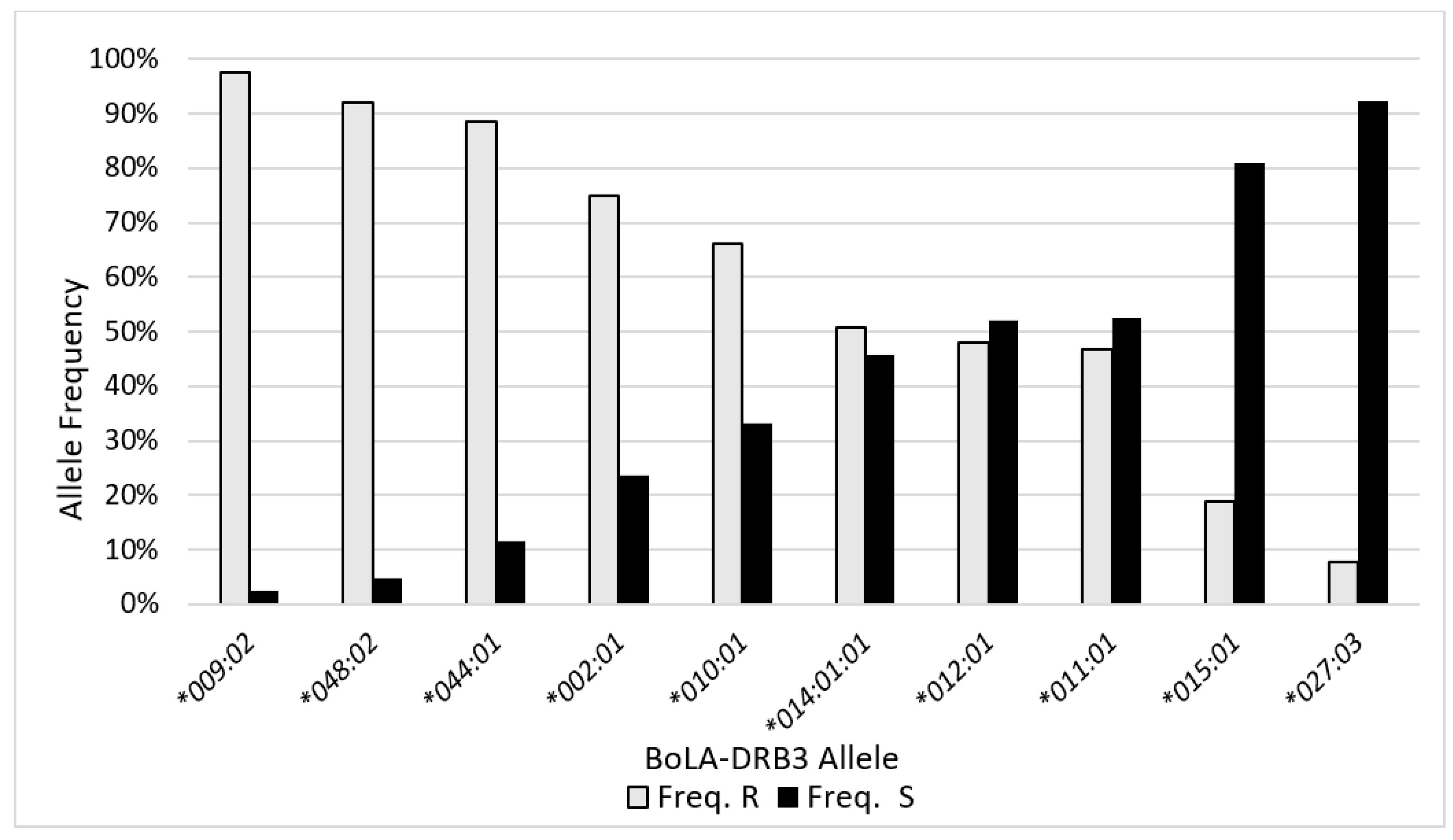
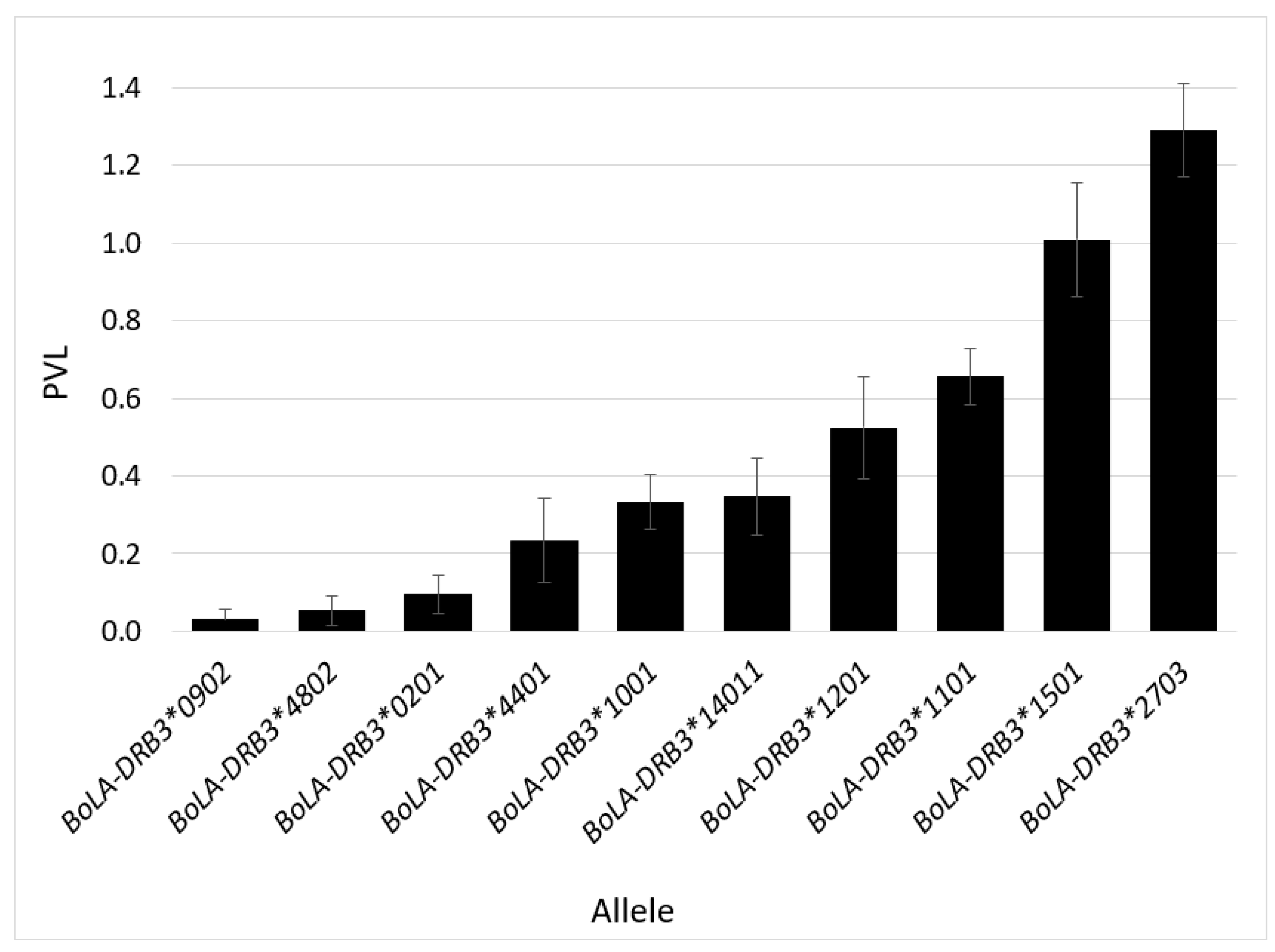
2.3. BLV Phenotype Associates with BoLA-DRB3 Genotype
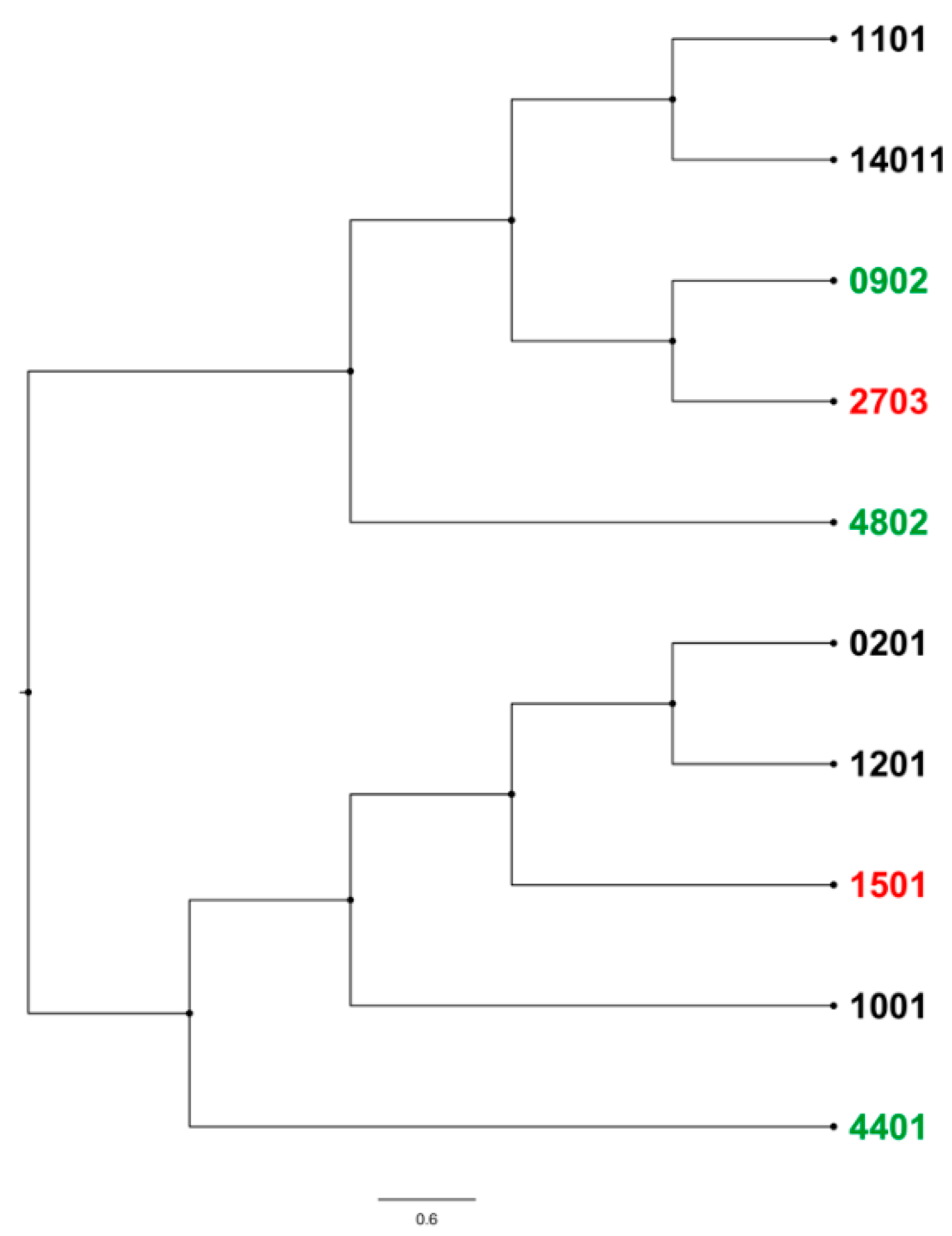
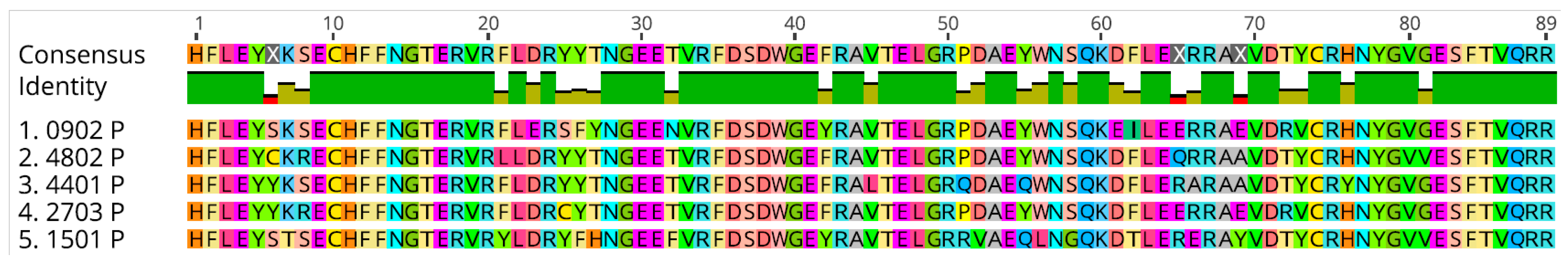
2.4. BoLA-DRB3 Alleles Found to Be Associated with Differential BLV Phenotypes Are Genetically Related
3. Discussion
3.1. Disease Resilience, Tolerance, and Resistance
3.2. Longitudinal Diagnostic Outcomes for Phenotype Determination
3.3. Lack of Diversity in BoLA-MHC Class II Genes May Lead to Increased Disease Susceptibility
3.4. BLV Phenotyping Prior to BoLA Genotyping Agrees with Current Literature
3.5. BLV Phenotype Is Likely Affected by Various Genetic Factors
4. Materials and Methods
4.1. Sample Collection, Diagnosis, and Phenotype Determination
4.2. Detection of Anti-BLV gp51 Antibody via Enzyme-Linked Immunosorbent Assay (ELISA)
4.3. DNA Extraction and BLV Proviral Load Determination
4.4. Next Generation Sequencing (NGS) of the BoLA-DRB3 Gene
4.5. Bioinformatics
4.6. Sequencing Controls
4.7. Phylogenetics and Amino Acid Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- LaDronka, R.M.; Ainsworth, S.; Wilkins, M.J.; Norby, B.; Byrem, T.M.; Bartlett, P.C. Prevalence of Bovine Leukemia Virus Antibodies in US Dairy Cattle. Vet. Med. Int. 2018, 2018, 5831278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrer, J.F.; Marshak, R.R.; Abt, D.A.; Kenyon, S.J. Persistent lymphocytosis in cattle: Its cause, nature and relation to lymphosarcoma. Ann. Rech. Vét. 1978, 9, 851–857. [Google Scholar]
- Johnson, R.; Gibson, C.D.; Kaneene, J.B. Bovine leukemia virus: A herd-based control strategy. Prev. Vet. Med. 1985, 3, 339–349. [Google Scholar] [CrossRef]
- Juliarena, M.A.; Barrios, C.N.; Ceriani, M.C.; Esteban, E.N. Hot topic: Bovine leukemia virus (BLV)-infected cows with low proviral load are not a source of infection for BLV-free cattle. J. Dairy Sci. 2016, 99, 4586–4589. [Google Scholar] [CrossRef] [Green Version]
- Furtado, M.D.S.B.S.; Andrade, R.G.; Romanelli, L.C.F.; Ribeiro, M.A.; Ribas, J.G.; Torres, E.B.; Barbosa-Stancioli, E.F.; Proietti, A.B.D.F.C.; Martins, M.L. Monitoring the HTLV-1 proviral load in the peripheral blood of asymptomatic carriers and patients with HTLV-associated myelopathy/tropical spastic paraparesis from a Brazilian cohort: ROC curve analysis to establish the threshold for risk disease. J. Med Virol. 2012, 84, 664–671. [Google Scholar] [CrossRef]
- Ellis, S.A.; Hammond, J.A. The Functional Significance of Cattle Major Histocompatibility Complex Class I Genetic Diversity. Annu. Rev. Anim. Biosci. 2014, 2, 285–306. [Google Scholar] [CrossRef] [PubMed]
- Amills, M.; Ramiya, V.; Norimine, J.; Lewin, H.A. The major hystocompatibility complex of ruminants. Rev. Sci. Tech. l’OIE 1998, 17, 108–120. [Google Scholar] [CrossRef] [Green Version]
- Behl, J.D.; Verma, N.K.; Tyagi, N.; Mishra, P.; Behl, R.; Joshi, B.K. The Major Histocompatibility Complex in Bovines: A Review. ISRN Vet. Sci. 2012, 2012, 872710. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muazzam, A.G.; Mansoor, A.; Ali, L.; Siddiqi, S.; Hameed, A.; Ajmal, M.; Mazhar, K. Association of HLA-DRB1 and -DQB1alleles and haplotypes with rheumatoid arthritis in a Pakistani population. Arthritis Res. Ther. 2013, 15, R95. [Google Scholar] [CrossRef] [Green Version]
- Rao, V.; Raha, O.; Chowdhury, S.; Dasgupta, S.; Raychaudhuri, P.; Sarkar, B.; Raju, P. Approaches in type 1 diabetes research: A status report. Int. J. Diabetes Dev. Ctries. 2009, 29, 85–101. [Google Scholar] [CrossRef] [Green Version]
- Taylor, G.M.; Gokhale, D.A.; Crowther, D.; Woll, P.J.; Harris, M.; Ryder, D.; Ayres, M.; Radford, J.A. Further investigation of the role of HLA-DPB1 in adult Hodgkin’s disease (HD) suggests an influence on susceptibility to different HD subtypes. Br. J. Cancer 1999, 80, 1405–1411. [Google Scholar] [CrossRef] [Green Version]
- Scherf, B.D.; Pilling, D. The Second Report on the State of World’s Animal Genetic Resources for Food and Agriculture. Available online: http://www.fao.org/3/a-i4787e.pdf (accessed on 21 October 2021).
- Abt, D.A.; Marshak, R.R.; Kulp, H.W.; Pollock, R.J. Studies on the Relationship between Lymphocytosis and Bovine Leukosis1. In Comparative Leukemia Research 1969; Dutcher, R.M., Ed.; Karger: Basel, Switzerland, 1970; Volume 36, pp. 527–536. [Google Scholar] [CrossRef]
- Lewin, H.A.; Bernoco, D. Evidence for BoLA-linked resistance and susceptibility to subclinical progression of bovine leukaemia virus infection. Anim. Genet. 1986, 17, 197–207. [Google Scholar] [CrossRef]
- Xu, A.; van Eijk, M.J.; Park, C.; Lewin, H.A. Polymorphism in BoLA-DRB3 exon 2 correlates with resistance to persistent lympho-cytosis caused by bovine leukemia virus. J. Immunol. 1993, 151, 6977–6985. Available online: http://www.jimmunol.org/content/151/12/6977.abstract (accessed on 19 July 2021).
- Maccari, G.; Robinson, J.; Ballingall, K.; Guethlein, L.A.; Grimholt, U.; Kaufman, J.; Ho, C.-S.; De Groot, N.G.; Flicek, P.; Bontrop, R.; et al. IPD-MHC 2.0: An improved inter-species database for the study of the major histocompatibility complex. Nucleic Acids Res. 2017, 45, D860–D864. [Google Scholar] [CrossRef]
- Burke, M.G.; Stone, R.T.; Muggli-Cockett, N.E. Nucleotide sequence and Northern analysis of a bovine major histocompatibility class II DRß-like cDNA. Anim. Genet. 2009, 22, 343–352. [Google Scholar] [CrossRef]
- Miyasaka, T.; Takeshima, S.N.; Jimba, M.; Matsumoto, Y.; Kobayashi, N.; Matsuhashi, T.; Sentsui, H.; Aida, Y. Identification of bovine leukocyte antigen class II haplotypes associated with variations in bovine leukemia virus proviral load in Japanese Black cattle. Tissue Antigens 2013, 81, 72–82. [Google Scholar] [CrossRef]
- Carignano, H.A.; Beribe, M.J.; Caffaro, M.E.; Amadio, A.F.; Nani, J.P.; Gutierrez, G.; Alvarez, I.; Trono, K.; Miretti, M.; Poli, M.A. BOLA-DRB3gene polymorphisms influence bovine leukaemia virus infection levels in Holstein and Holstein × Jersey crossbreed dairy cattle. Anim. Genet. 2017, 48, 420–430. [Google Scholar] [CrossRef]
- Esteban, E.N.; Poli, M.A.; Ceriani, C.; Gutierrez, C.; Dolcini, G.L.; Pérez, S.E.; Lützelschwab, C.M.; Feldman, L.; Juliarena, M.A. Bovine Leukemia Virus (BLV), Proposed Control and Eradication Programs by Marker Assisted Breeding of Genetically Resistant Cattle. In Animal Genetics; Rechi, L.J., Ed.; Nova Science Publishers: Hauppaige, NY, USA, 2009. [Google Scholar]
- Hayashi, T.; Mekata, H.; Sekiguchi, S.; Kirino, Y.; Mitoma, S.; Honkawa, K.; Horii, Y.; Norimine, J. Cattle with the BoLA class II DRB3*0902 allele have significantly lower bovine leukemia proviral loads. J. Vet. Med. Sci. 2017, 79, 1552–1555. [Google Scholar] [CrossRef] [Green Version]
- Juliarena, M.A.; Poli, M.; Sala, L.; Ceriani, C.; Gutierrez, S.; Dolcini, G.; Rodrguez, E.M.; Mario, B.; Rodrguez-Dubra, C.; Esteban, E.N.; et al. Association of BLV infection profiles with alleles of theBoLA-DRB3.2gene. Anim. Genet. 2008, 39, 432–438. [Google Scholar] [CrossRef]
- Udina, I.G.; Karamysheva, E.E.; Turkova, S.O.; Orlova, A.R.; Sulimova, G.E. Genetic mechanisms of resistance and susceptibility to leukemia in Ayrshire and black pied cattle breeds determined by allelic distribution of gene Bola-DRB3. Russ. J. Genet. 2003, 39, 306–317. [Google Scholar] [CrossRef]
- Takeshima, S.-N.; Ohno, A.; Aida, Y. Bovine leukemia virus proviral load is more strongly associated with bovine major histocompatibility complex class II DRB3 polymorphism than with DQA1 polymorphism in Holstein cow in Japan. Retrovirology 2019, 16, 14. [Google Scholar] [CrossRef]
- Taxis, T.M.; DeJong, T.N.; Swenson, C.L.; Sporer, K.R.; Droscha, C.; Niles, D.; Bartlett, P.C. Reducing bovine leukemia virus prevalence on a large midwestern dairy farm by using lymphocyte counts, ELISA antibody testing, and proviral load. Bov. Pract. 2020, 54, 136–144. [Google Scholar]
- Ruggiero, V.J.; Bartlett, P.C. Control of Bovine Leukemia Virus in Three US Dairy Herds by Culling ELISA-Positive Cows. Vet. Med. Int. 2019, 2019, 3202184. [Google Scholar] [CrossRef] [Green Version]
- Hutchinson, H.C.; Norby, B.; Droscha, C.J.; Sordillo, L.M.; Coussens, P.M.; Bartlett, P.C. Bovine leukemia virus detection and dynamics following experimental inoculation. Res. Vet. Sci. 2020, 133, 269–275. [Google Scholar] [CrossRef]
- Lupón, J.; Gavidia-Bovadilla, G.; Ferrer, E.; de Antonio, M.; Perera-Lluna, A.; López-Ayerbe, J.; Domingo, M.; Núñez, J.; Zamora, E.; Moliner, P.; et al. Dynamic Trajectories of Left Ventricular Ejection Fraction in Heart Failure. J. Am. Coll. Cardiol. 2018, 72, 591–601. [Google Scholar] [CrossRef]
- Eijk, M.J.T.; Stewart-Haynes, J.A.; Lewin, H.A. Extensive polymorphism of the BOLA-DRB3 gene distinguished by PCR-RFLP. Anim Genet. 1992, 23, 483–496. [Google Scholar] [CrossRef]
- Lo, C.-W.; Borjigin, L.; Saito, S.; Fukunaga, K.; Saitou, E.; Okazaki, K.; Mizutani, T.; Wada, S.; Takeshima, S.-N.; Aida, Y. BoLA-DRB3 Polymorphism is Associated with Differential Susceptibility to Bovine Leukemia Virus-Induced Lymphoma and Proviral Load. Viruses 2020, 12, 352. [Google Scholar] [CrossRef] [Green Version]
- Berghof, T.V.L.; Poppe, M.; Mulder, H. Opportunities to Improve Resilience in Animal Breeding Programs. Front. Genet. 2019, 9, 692. [Google Scholar] [CrossRef] [Green Version]
- Bishop, S.C.; Woolliams, J.A. Genomics and disease resistance studies in livestock. Livest. Sci. 2014, 166, 190–198. [Google Scholar] [CrossRef] [Green Version]
- Råberg, L.; Sim, D.; Read, A.F. Disentangling Genetic Variation for Resistance and Tolerance to Infectious Diseases in Animals. Science 2007, 318, 812–814. [Google Scholar] [CrossRef] [Green Version]
- Matzaraki, V.; Kumar, V.; Wijmenga, C.; Zhernakova, A. The MHC locus and genetic susceptibility to autoimmune and infectious diseases. Genome Biol. 2017, 18, 76. [Google Scholar] [CrossRef]
- Fellay, J.; Shianna, K.V.; Ge, D.; Colombo, S.; Ledergerber, B.; Weale, M.; Zhang, K.; Gumbs, C.; Castagna, A.; Cossarizza, A.; et al. A Whole-Genome Association Study of Major Determinants for Host Control of HIV-1. Science 2007, 317, 944–947. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Sonstegard, T.S.; Cole, J.B.; Van Tassell, C.P.; Wiggans, G.R.; Crooker, B.A.; Tan, C.; Prakapenka, D.; Liu, G.E.; Da, Y. Genome changes due to artificial selection in U.S. Holstein cattle. BMC Genom. 2019, 20, 128. [Google Scholar] [CrossRef]
- Giovambattista, G.; Moe, K.K.; Polat, M.; Borjigin, L.; Hein, S.T.; Moe, H.H.; Takeshima, S.-N.; Aida, Y. Characterization of bovine MHC DRB3 diversity in global cattle breeds, with a focus on cattle in Myanmar. BMC Genet. 2020, 21, 95. [Google Scholar] [CrossRef]
- Takeshima, S.-n.; Ikegami, M.; Morita, M.; Nakai, Y.; Aida, Y. Identification of new cattle BoLA-DRB3 alleles by sequence-based typing. Immunogenetics 2001, 53, 74–81. [Google Scholar] [CrossRef]
- Gutiérrez, S.E.; Esteban, E.N.; Lützelschwab, C.M.; Juliarena, M.A. Major Histocompatibility Complex-Associated Resistance to Infectious Diseases: The Case of Bovine Leukemia Virus Infection. In Trends and Advances in Veterinary Genetics; Abubakar, M., Ed.; InTech: Rijeka, Croatia, 2017. [Google Scholar] [CrossRef] [Green Version]
- Gao, X.; Nelson, G.W.; Karacki, P.; Martin, M.P.; Phair, J.; Kaslow, R.; Goedert, J.J.; Buchbinder, S.; Hoots, K.; Vlahov, D.; et al. Effect of a Single Amino Acid Change in MHC Class I Molecules on the Rate of Progression to AIDS. N. Engl. J. Med. 2001, 344, 1668–1675. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, T.; Mukoyama, H.; Furuta, H.; Kondo, Y.; Takeshima, S.; Aida, Y.; Kosugiyama, M.; Tomogane, H. Association of the amino acid motifs ofBoLA-DRB3alleles with mastitis pathogens in Japanese Holstein cows. Anim. Sci. J. 2009, 80, 510–519. [Google Scholar] [CrossRef]
- Lopalco, L. CCR5: From Natural Resistance to a New Anti-HIV Strategy. Viruses 2010, 2, 574–600. [Google Scholar] [CrossRef] [Green Version]
- Bai, L.; Sato, H.; Kubo, Y.; Wada, S.; Aida, Y. CAT1/SLC7A1 acts as a cellular receptor for bovine leukemia virus infection. FASEB J. 2019, 33, 14516–14527. [Google Scholar] [CrossRef] [Green Version]
- Deykin, A.V.; Kubekina, M.V.; Silaeva, Y.Y.; Krivonogova, A.S.; Isaeva, A.G. Using CRISPR/Cas9 for generation the cd209 knockout is a way to get cattle breeds resistant to the Bovine leukemia virus (BLV). E3S Web Conf. 2020, 176, 01007. [Google Scholar] [CrossRef]
- Klimov, E.; Shevtsova, A.; Kovalchuk, S. The Search for a Receptor for Cell Infection by Bovine Leukemia Virus: Data Mining and Signaling Pathways Analysis. Annu. Res. Rev. Biol. 2018, 28, 1–4. [Google Scholar] [CrossRef]
- Kashima, S.; Rodrigues, E.S.; Azevedo, F.; da Cruz Castelli, E.; Mendes-Junior, C.T.; Yoshioka, F.K.N.; da Silva, I.T.; Takayanagui, O.M.; Covas, D.T. DC-SIGN (CD209) gene promoter polymorphisms in a Brazilian population and their association with human T-cell lymphotropic virus type 1 infection. J. Gen. Virol. 2009, 90, 927–934. [Google Scholar] [CrossRef]
- Li, H. Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. arXiv 2013, arXiv:1303.3997. [Google Scholar]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R.; 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [Green Version]
- Garrison, E.; Marth, G. Haplotype-based variant detection from short-read sequencing. arXiv 2012, arXiv:1207.3907. [Google Scholar]
- Edgar, R.C. MUSCLE: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinform. 2004, 5, 113. [Google Scholar] [CrossRef] [Green Version]
| Timepoints | |||||
|---|---|---|---|---|---|
| 2016 | 2017 | 2018 | 2019 | Total | |
| # Farms | 6 | 6 | 9 | 9 | 9 |
| # ELISA | 80 | 866 | 6633 | 6854 | 14,433 |
| # PCR | 65 | 30 | 1655 | 2335 | 4085 |
| # Genotyped Animals | 95 | 95 | 384 | 574 | |
| Primer Name | Fluidigm CS Sequence | TruSeq Index | Gene-Specific Primer Sequence |
|---|---|---|---|
| BoLA-DRB3.1 F | ACACTGACGACATGGTTCTACA | TCGTGGAGCG | ATCCTCTCTCTGCACGAGATTTCC |
| BoLA-DRB3.4 F | ACACTGACGACATGGTTCTACA | TGCCTGGTGG | ATCCTCTCTCTGCACGAGATTTCC |
| BoLA-DRB3.12 F | ACACTGACGACATGGTTCTACA | GTGTGGCGCT | ATCCTCTCTCTGCACGAGATTTCC |
| BoLA-DRB3.20 F | ACACTGACGACATGGTTCTACA | CGCACATGGC | ATCCTCTCTCTGCACGAGATTTCC |
| BoLA-DRB3 R | TACGGTAGCAGAGACTTGGTCTTCGCCGCTGCACAGTGAAACTCTC | ||
| BoLA-DRB3 Allele | Total No. | Total Freq. | Freq. R | Freq. S |
|---|---|---|---|---|
| *009:02 | 199 | 20.2% | 97.5% | 2.5% |
| *011:01 | 171 | 17.4% | 46.8% | 52.6% |
| *010:01 | 124 | 12.6% | 66.1% | 33.1% |
| *002:01 | 72 | 7.3% | 75.0% | 23.6% |
| *048:02 | 62 | 6.3% | 91.9% | 4.8% |
| *014:01:01 | 57 | 5.8% | 50.9% | 45.6% |
| *044:01 | 52 | 5.3% | 88.5% | 11.5% |
| *012:01 | 50 | 5.1% | 48.0% | 52.0% |
| *027:03 | 39 | 4.0% | 7.7% | 92.3% |
| *015:01 | 37 | 3.8% | 18.9% | 81.1% |
| *016:01 | 35 | 3.6% | 85.7% | 11.4% |
| *007:01 | 33 | 3.4% | 78.8% | 18.2% |
| *006:01 | 27 | 2.7% | 92.6% | 7.4% |
| *001:01 | 9 | 0.9% | 33.3% | 66.7% |
| *037:01 | 6 | 0.6% | 83.3% | 16.7% |
| *018:01 | 6 | 0.6% | 50.0% | 50.0% |
| *008:01 | 4 | 0.4% | 50.0% | 50.0% |
| *061:01 | 1 | 0.1% | 0.0% | 100.0% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lohr, C.E.; Sporer, K.R.B.; Brigham, K.A.; Pavliscak, L.A.; Mason, M.M.; Borgman, A.; Ruggiero, V.J.; Taxis, T.M.; Bartlett, P.C.; Droscha, C.J. Phenotypic Selection of Dairy Cattle Infected with Bovine Leukemia Virus Demonstrates Immunogenetic Resilience through NGS-Based Genotyping of BoLA MHC Class II Genes. Pathogens 2022, 11, 104. https://doi.org/10.3390/pathogens11010104
Lohr CE, Sporer KRB, Brigham KA, Pavliscak LA, Mason MM, Borgman A, Ruggiero VJ, Taxis TM, Bartlett PC, Droscha CJ. Phenotypic Selection of Dairy Cattle Infected with Bovine Leukemia Virus Demonstrates Immunogenetic Resilience through NGS-Based Genotyping of BoLA MHC Class II Genes. Pathogens. 2022; 11(1):104. https://doi.org/10.3390/pathogens11010104
Chicago/Turabian StyleLohr, Chaelynne E., Kelly R. B. Sporer, Kelsey A. Brigham, Laura A. Pavliscak, Matelyn M. Mason, Andrew Borgman, Vickie J. Ruggiero, Tasia M. Taxis, Paul C. Bartlett, and Casey J. Droscha. 2022. "Phenotypic Selection of Dairy Cattle Infected with Bovine Leukemia Virus Demonstrates Immunogenetic Resilience through NGS-Based Genotyping of BoLA MHC Class II Genes" Pathogens 11, no. 1: 104. https://doi.org/10.3390/pathogens11010104
APA StyleLohr, C. E., Sporer, K. R. B., Brigham, K. A., Pavliscak, L. A., Mason, M. M., Borgman, A., Ruggiero, V. J., Taxis, T. M., Bartlett, P. C., & Droscha, C. J. (2022). Phenotypic Selection of Dairy Cattle Infected with Bovine Leukemia Virus Demonstrates Immunogenetic Resilience through NGS-Based Genotyping of BoLA MHC Class II Genes. Pathogens, 11(1), 104. https://doi.org/10.3390/pathogens11010104








