Aspergillus Section Fumigati Pneumonia and Oxalate Nephrosis in a Foal
Abstract
:1. Introduction
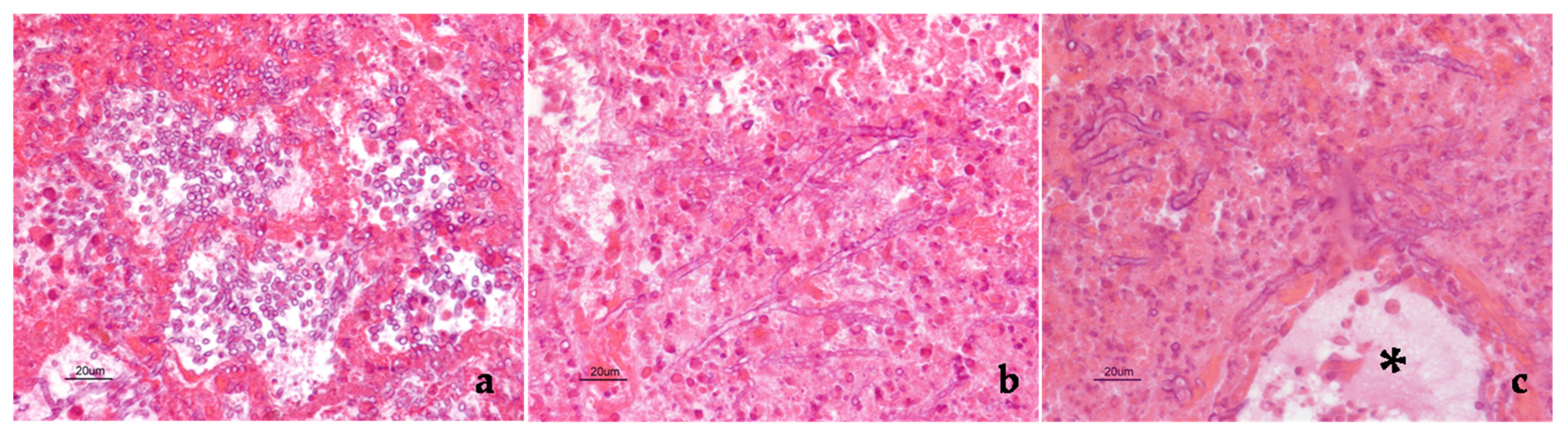
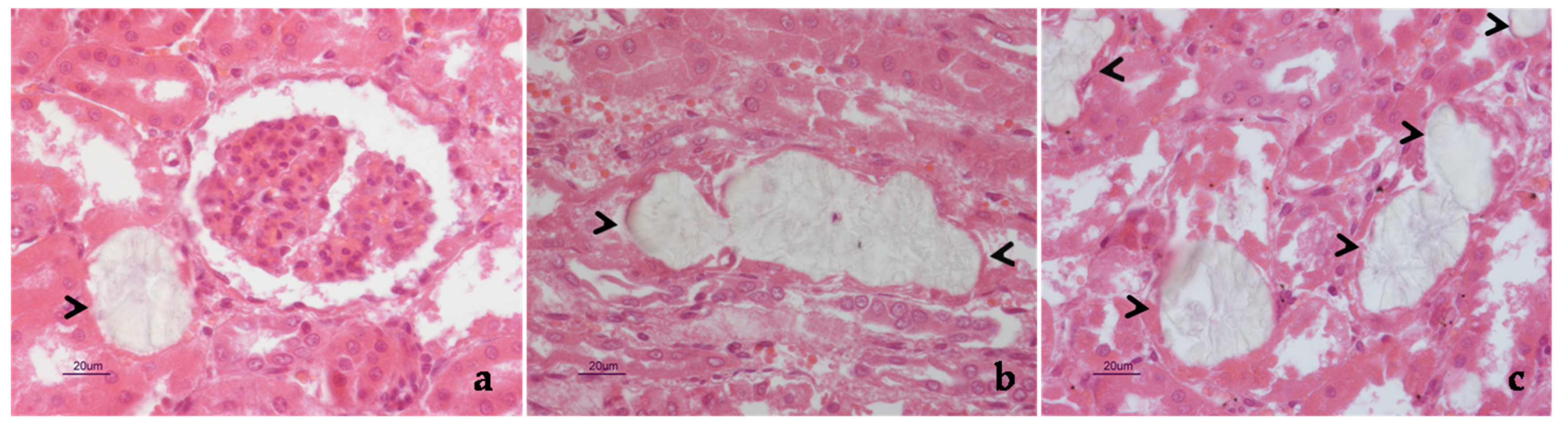
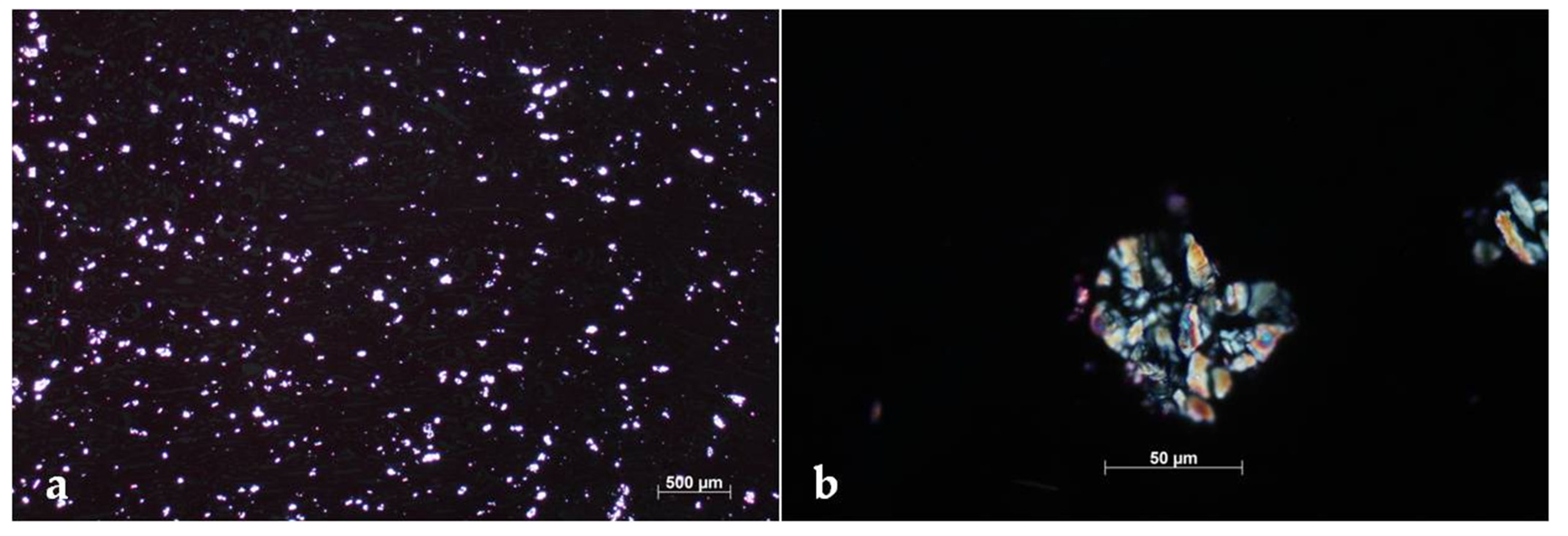
2. Case Description
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Seyedmousavi, S.; Guillot, J.; Arné, P.; de Hoog, G.S.; Mouton, J.W.; Melchers, W.J.; Verweij, P.E. Aspergillus and aspergilloses in wild and domestic animals: A global health concern with parallels to human disease. Med. Mycol. 2015, 53, 765–797. [Google Scholar] [CrossRef] [PubMed]
- Cafarchia, C.; Figueredo, L.A.; Otranto, D. Fungal diseases of horses. Vet. Microbiol. 2013, 167, 215–234. [Google Scholar] [CrossRef] [PubMed]
- Slocombe, R.F.; Slauson, D.O. Invasive pulmonary aspergillosis of horses: An association with acute enteritis. Vet. Pathol. 1988, 25, 277–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshioka, I.; Kobayashi, K.; Kirimura, K. Overexpression of the gene encoding alternative oxidase for enhanced glucose consumption in oxalic acid producing Aspergillus niger expressing oxaloacetate hydrolase gene. J. Biosci. Bioeng. 2020, 129, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Barnes, W.G.; Schaetzel, W.P. Pulmonary aspergillosis and the importance of oxalate crystal recognition in cytology specimens. Arch. Pathol. Lab. Med. 1986, 110, 1176–1179. [Google Scholar] [PubMed]
- Payne, C.L.; Dark, M.J.; Conway, J.A.; Farina, L.L. A retrospective study of the prevalence of calcium oxalate crystals in veterinary Aspergillus cases. J. Vet. Diagn. Investig. 2017, 29, 51–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandhu, G.S.; Kline, B.C.; Stockman, L.; Roberts, G.D. Molecular probes for diagnosis of fungal infections. J. Clin. Microbiol. 1995, 33, 2913–2919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sweeney, C.R.; Habecker, P.L. Pulmonary aspergillosis in horses: 29 cases (1974–1997). J. Am. Vet. Med. Assoc. 1999, 214, 808–811. [Google Scholar]
- Headley, S.A.; de Carvalho, P.H.; Cunha Filho, L.F.; Yamamura, A.A.; Okano, W. Equine pulmonary aspergillosis with encephalitic, myocardial, and renal dissemination. Mycopathologia 2014, 177, 129–135. [Google Scholar] [CrossRef]
- Carrasco, L.; Mendez, A.; Jensen, H.E. Chronic bronchopulmonary aspergillosis in a horse with Cushing’s syndrome. Mycoses 1996, 39, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Buechner-Maxwell, V.; Zhang, C.; Robertson, J.; Jain, N.C.; Antczak, D.F.; Feldman, B.F.; Murray, M.J. Intravascular leukostasis and systemic aspergillosis in a horse with subleukemic acute myelomonocytic leukemia. J. Vet. Intern. Med. 1994, 8, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, L.; Tarradas, M.C.; Gómez-Villamandos, J.C.; Luque, I.; Arenas, A.; Méndez, A. Equine pulmonary mycosis due to Aspergillus niger and Rhizopus stolonifer. J. Comp. Pathol. 1997, 117, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Hattel, A.L.; Drake, T.R.; Anderholm, B.J.; McAllister, E.S. Pulmonary aspergillosis associated with acute enteritis in a horse. J. Am. Vet. Med. Assoc. 1991, 199, 589–590. [Google Scholar] [PubMed]
- Pace, L.W.; Wirth, N.R.; Foss, R.R.; Fales, W.H. Endocarditis and pulmonary aspergillosis in a horse. J. Vet. Diagn. Investig. 1994, 6, 504–506. [Google Scholar] [CrossRef]
- Johnson, P.J.; Moore, L.A.; Mrad, D.R.; Turk, J.R.; Wilson, D.A. Sudden death of two horses associated with pulmonary aspergillosis. Vet. Rec. 1999, 145, 16–20. [Google Scholar] [CrossRef]
- Long, J.R.; Mitchell, L. Pulmonary aspergillosis in a mare. Can. Vet. J. 1971, 12, 16–18. [Google Scholar] [PubMed]
- Thirion-Delalande, C.; Guillot, J.; Jensen, H.E.; Crespeau, F.L.; Bernex, F. Disseminated acute concomitant aspergillosis and mucormycosis in a pony. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2005, 52, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Blue, J.; Perdrizet, J.; Brown, E. Pulmonary aspergillosis in a horse with myelomonocytic leukemia. J. Am. Vet. Med. Assoc. 1987, 190, 1562–1564. [Google Scholar]
- Dutton, M.V.; Evans, C.S. Oxalate production by fungi: Its role in pathogenicity and ecology in the soil environment. Can. J. Microbiol 1996, 42, 881–895. [Google Scholar] [CrossRef]
- Maxie, M.G.; Newman, S.J. Urinary system. In Jubb, Kennedy & Palmer’s Pathology of Domestic Animals, 5th ed.; Maxie, M.G., Ed.; Saunders Elsevier: Philadelphia, PA, USA, 2007; Volume 2, pp. 523–654. [Google Scholar]
- Maeno, T.; Sasaki, M.; Shibue, Y.; Mimura, K.; Oka, H. Calcium oxalate in the sputum may aid in the diagnosis of pulmonary aspergillosis: A report of two cases. Med. Mycol. Case Rep. 2015, 8, 32–36. [Google Scholar] [CrossRef]
- Severo, L.C.; Londero, A.T.; Geyer, G.R.; Picon, P.D. Oxalosis associated with an Aspergillus niger fungus ball. Report of a case. Mycopathologia 1981, 73, 29–31. [Google Scholar] [CrossRef] [PubMed]
- Metzger, J.B.; Garagusi, V.F.; Kerwin, D.M. Pulmonary oxalosis caused by Aspergillus niger. Am. Rev. Respir. Dis. 1984, 129, 501–502. [Google Scholar] [CrossRef] [PubMed]
- Ghio, A.J.; Peterseim, D.S.; Roggli, V.L.; Piantadosi, C.A. Pulmonary oxalate deposition associated with Aspergillus niger infection. An oxidant hypothesis of toxicity. Am. Rev. Respir Dis. 1992, 145, 1499–1502. [Google Scholar] [CrossRef]
- Roehrl, M.H.; Croft, W.J.; Liao, Q.; Wang, J.Y.; Kradin, R.L. Hemorrhagic pulmonary oxalosis secondary to a noninvasive Aspergillus niger fungus ball. Virchows Arch. 2007, 451, 1067–1073. [Google Scholar] [CrossRef]
- Cho, C.; Asuncion, A.; Tatum, A.H. False-positive antineutrophil cytoplasmic antibody in aspergillosis with oxalosis. Arch. Pathol. Lab. Med. 1995, 119, 558–561. [Google Scholar] [PubMed]
- Vaideeswar, P.; Sakhdeo, U.M. Pulmonary aspergilloma with renal oxalosis: Fatal effect at a distance. Mycoses 2009, 52, 272–275. [Google Scholar] [CrossRef] [PubMed]
- Kuwabara, H.; Shibayama, Y. Pulmonary aspergilloma with prominent oxalate deposition. Indian J. Pathol. Microbiol. 2012, 55, 589–590. [Google Scholar] [CrossRef] [PubMed]
- Trujillo, H.; Fernández-Ruiz, M.; Gutiérrez, E.; Sevillano, Á.; Caravaca-Fontán, F.; Morales, E.; López-Medrano, F.; Aguado, J.M.; Praga, M.; Andrés, A. Invasive pulmonary aspergillosis associated with COVID-19 in a kidney transplant recipient. Transpl. Infect. Dis. 2021, 23, e13501. [Google Scholar] [CrossRef] [PubMed]
- Jae-Hoon, K.; Kyung-Il, K.; Won-Il, K.; Hyun-Joo, S.; Sang-Kyung, L.; Young-Hwa, J. Pulmonary Aspergillosis and Renal Oxalosis in an Elk. Korean J. Vet. Res. 2002, 42, 383–387. [Google Scholar]
- Wyand, D.S.; Langheinrich, K.; Helmboldt, C.F. Aspergillosis and renal oxalosis in a white-tailed deer. Case history. J. Wildl. Dis. 1971, 7, 52–56. [Google Scholar] [CrossRef] [Green Version]
| Number of EPA Cases | Predisposing Factors | Aspergillus Species | Sites of Infection (Other Than Lung) | Sites of Oxalates Precipitation | Reference |
|---|---|---|---|---|---|
| 19 | Enterocolitis (n = 14), intensive corticosteroid treatment (n = 2), urolithiasis (n = 1), myelogenous leukemia (n = 1), hemangiosarcoma (n = 1) | Aspergillus sp. (n = 17), Aspergillus fumigatus (n = 2) | Kidneys (n = 2), brain (n = 1) | Not reported | [3] |
| 11 | Not reported | Aspergillus sp. (n = 11), Aspergillus fumigatus (n = 2), Aspergillus glaucus (n = 1), Aspergillus flavus (n = 1) | Colon (n = 1), kidney (n = 1), heart (n = 1), skeletal muscle (n = 1), guttural pouches (n = 1) | Lungs (n = 1) and kidneys (n = 2) | [6] |
| 29 | Loss of integrity of the gastrointestinal tract (n = 25) | Aspergillus sp. | Not specified organs (n = 12) | Not reported | [8] |
| 2 | None | Aspergillus fumigatus | Kidneys, myocardium, brain | Not reported | [9] |
| 1 | Cushing’s syndrome | Aspergillus sp. | None | Not reported | [10] |
| 1 | Leukemia | Aspergillus sp. | Gut | Not reported | [11] |
| 1 | Exposure to high spore count | Aspergillus niger | None | Not reported | [12] |
| 1 | Ehrlichia risticii infection, acute enteritis | None | Not reported | [13] | |
| 1 | Volvulus of the colon and peritonitis | Aspergillus sp. | Heart | Not reported | [14] |
| 2 | Sarcocystis neurona infection (n = 1), small intestine obstruction (n = 1) | Aspergillus sp. | None | Not reported | [15] |
| 1 | Unknown | Aspergillus fumigatus | None | Not reported | [16] |
| 1 | Enterocolitis | Aspergillus fumigatus | Colon | Not reported | [17] |
| 1 | Myelomonocytic leukemia | Aspergillus sp. | None | Not reported | [18] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hattab, J.; Vulcano, A.; D’Arezzo, S.; Verni, F.; Tiscar, P.G.; Lanteri, G.; Gjurcevic, E.; Tosi, U.; Marruchella, G. Aspergillus Section Fumigati Pneumonia and Oxalate Nephrosis in a Foal. Pathogens 2021, 10, 1087. https://doi.org/10.3390/pathogens10091087
Hattab J, Vulcano A, D’Arezzo S, Verni F, Tiscar PG, Lanteri G, Gjurcevic E, Tosi U, Marruchella G. Aspergillus Section Fumigati Pneumonia and Oxalate Nephrosis in a Foal. Pathogens. 2021; 10(9):1087. https://doi.org/10.3390/pathogens10091087
Chicago/Turabian StyleHattab, Jasmine, Antonella Vulcano, Silvia D’Arezzo, Fabiana Verni, Pietro Giorgio Tiscar, Giovanni Lanteri, Emil Gjurcevic, Umberto Tosi, and Giuseppe Marruchella. 2021. "Aspergillus Section Fumigati Pneumonia and Oxalate Nephrosis in a Foal" Pathogens 10, no. 9: 1087. https://doi.org/10.3390/pathogens10091087
APA StyleHattab, J., Vulcano, A., D’Arezzo, S., Verni, F., Tiscar, P. G., Lanteri, G., Gjurcevic, E., Tosi, U., & Marruchella, G. (2021). Aspergillus Section Fumigati Pneumonia and Oxalate Nephrosis in a Foal. Pathogens, 10(9), 1087. https://doi.org/10.3390/pathogens10091087







