Molecular Detection of Selected Pathogens with Zoonotic Potential in Deer Keds (Lipoptena fortisetosa)
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
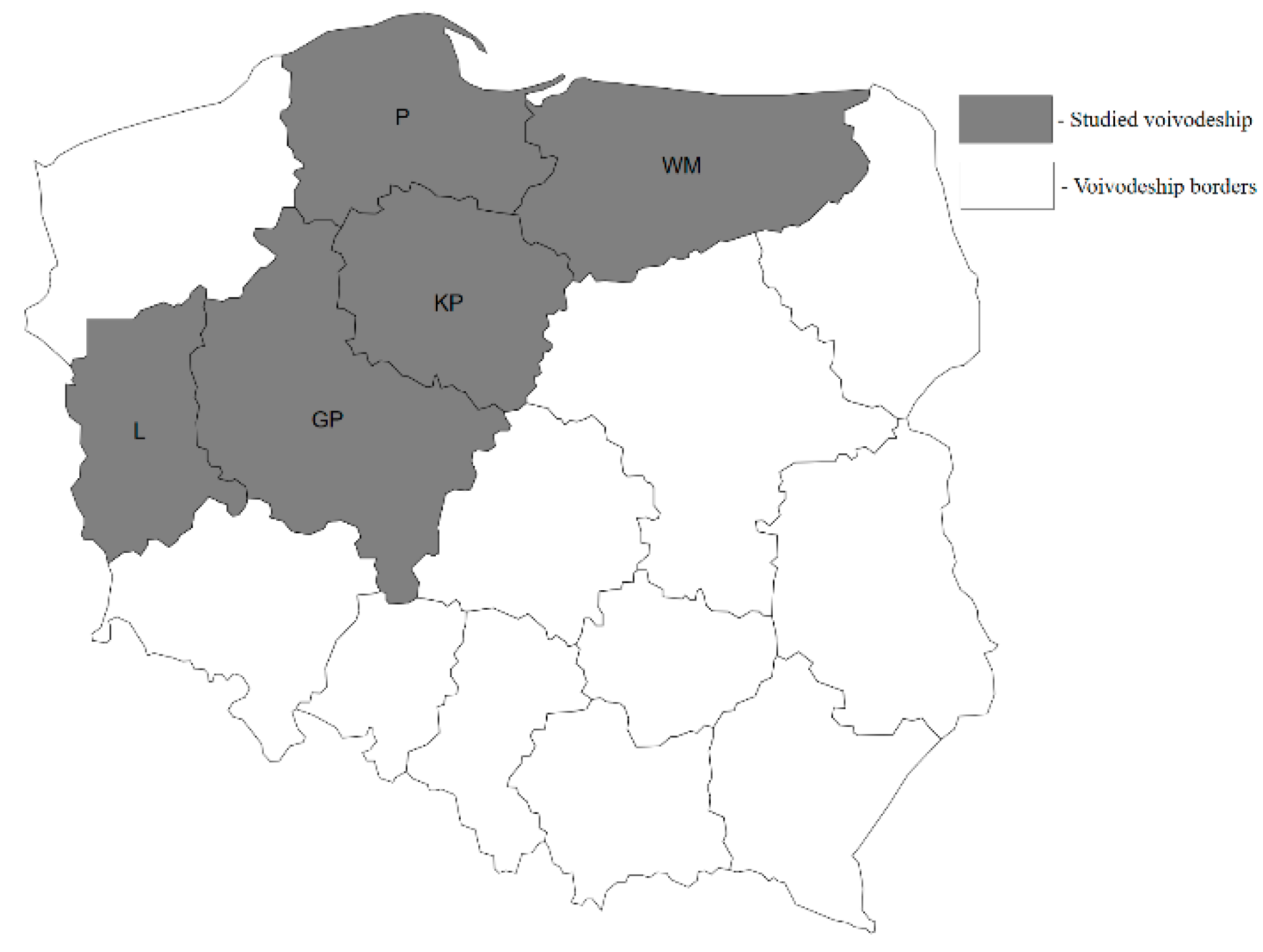
4.1. Sample Collection
4.2. Species Identification
4.3. DNA Extraction
4.4. Selection of Pathogens for Analysis
4.5. Polymerase Chain Reaction
4.6. Sequencing
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bueno-Marí, R.; Jiménez-Peydró, R. Anopheles plumbeus Stephens, 1828: A neglected malaria vector in Europe. Malar. Rep. 2011, 1, e2. [Google Scholar] [CrossRef]
- Baldacchino, F.; Desquesnes, M.; Mihok, S.; Foil, L.D.; Duvallet, G.; Jittapalapong, S. Tabanids: Neglected subjects of research, but important vectors of disease agents! Infect. Genet. Evol. 2014, 28, 596–615. [Google Scholar] [CrossRef]
- Salvetti, M.; Bianchi, A.; Marangi, M.; Barlaam, A.; Giacomelli, S.; Bertoletti, I.; Roy, L.; Giangaspero, A. Deer keds on wild ungulates in northern Italy, with a taxonomic key for the identification of Lipoptena spp. of Europe. Med. Vet. Entomol. 2019, 34, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Bezerra-Santos, M.A.; Otranto, D. Keds, the enigmatic flies and their role as vectors of pathogens. Acta Trop. 2020, 209, 105521. [Google Scholar] [CrossRef] [PubMed]
- Kowal, J.; Nosal, P.; Kornaś, S.; Wajdzik, M.; Matysek, M.; Basiaga, M. Biodiversity and importance of hippoboscids infection in cervids. Med. Weter. 2016, 72, 745–749. [Google Scholar]
- Maa, T.C. A synopsis of the Lipopteninae. J. Med. Entomol. 1965, 2, 233–248. [Google Scholar] [CrossRef] [PubMed]
- Borowiec, L.; Zatwarnicki, T. Lipoptena fortisetosa Maa, 1965 (Diptera, Hippoboscidae), nowy gatunek dla fauny Polski. Prz. Zool. 1989, 33, 579–582. [Google Scholar]
- Kowal, J.; Nosal, P.; Rościszewska, M.; Matysek, M. New records of Lipoptena fortisetosa Maa, 1965 (Diptera:Hippoboscidae) in Poland. Dipteron 2009, 25, 27–29. [Google Scholar]
- Cydzik, K.; Kadulski, S. Parasitic Insects of the Red Deer (Cervus elaphus L.) in Northeastern Poland. Stawonogi. Inwazje i Ich Ograniczanie; Akapit: Lublin, Poland, 2009; pp. 113–115. [Google Scholar]
- Jędrysik, D.; Kadulski, S. Parasitic Arthropods of Roe Deer Capreolus capreolus (L.) of the Region of Pojezierze Południowobałtyckie (The Southern Baltic Lake District). Arthropods. The Medical and Economic Importance; Akapit: Lublin, Poland, 2012; pp. 95–103. [Google Scholar]
- Matysek, M.; Kowal, J. Dwa nowe gatunki muchówek. Tatry 2014, 48, 64–65. [Google Scholar]
- Kadulski, S. Lipoptena Nitzsch, 1818 in Poland. Wiadomości Parazytologiczne 1998, 44, 462. [Google Scholar]
- Sokół, R.; Gałęcki, R. Prevalence of keds on city dogs in central Poland. Med. Vet. Entomol. 2017, 31, 114–116. [Google Scholar] [CrossRef] [PubMed]
- Gałęcki, R.; Jaroszewski, J.; Xuan, X.; Bakuła, T. Temporal-Microclimatic Factors Affect the Phenology of Lipoptena fortisetosa in Central European Forests. Animals 2020, 10, 2012. [Google Scholar] [CrossRef] [PubMed]
- Hermosilla, C.; Pantchev, N.; Bachmann, R.; Bauer, C. Lipoptena cervi (deer ked) in two naturally infested dogs. Vet. Rec. 2006, 159, 286. [Google Scholar] [CrossRef] [PubMed]
- Kadulski, S. Ectoparasites of Cervidae in north-east Poland. Acta Parasitol. 1996, 41, 204–210. [Google Scholar]
- Szczurek, B.; Kadulski, S. Ectoparasites on fallow deer, Dama dama (L.) in Pomerania, Poland. Acta Parasitol. 2004, 49, 80–86. [Google Scholar]
- Izdebska, J.N. European bison arthropod parasites from closed Polish breeding facilities. Acta Parasitol. 2001, 46, 135–137. [Google Scholar]
- Metelitsa, A.K.; Veselkin, G.A. Parasitism of the louse fly Lipoptena fortisetosa on cattle. Parazitologiia 1989, 23, 276–277. [Google Scholar]
- Dehio, C.; Sauder, U.; Hiestand, R. Isolation of Bartonella schoenbuchensis from Lipoptena cervi, a blood-sucking arthropod causing deer ked dermatitis. J. Clin. Microbiol. 2004, 42, 5320–5323. [Google Scholar] [CrossRef]
- Ivanov, V.I. Anthropophilia of the deer blood sucker Lipoptena cervi L. (Diptera, Hippoboscidae). Med. Parazitol. 1975, 44, 491–495. [Google Scholar]
- Buczek, W.; Buczek, A.M.; Bartosik, K.; Buczek, A. Comparison of skin lesions caused by Ixodes ricinus ticks and Lipoptena cervi deer keds infesting humans in the natural environment. Int. J. Environ. Res. Public Health 2020, 17, 3316. [Google Scholar] [CrossRef]
- Maślanko, W.; Bartosik, K.; Raszewska-Famielec, M.; Szwaj, E.; Asman, M. Exposure of Humans to Attacks by Deer Keds and Consequences of Their Bites—A Case Report with Environmental Background. Insects 2020, 11, 859. [Google Scholar] [CrossRef]
- Vikøren, T.; Lillehaug, A.; Handeland, K. Helseovervakingsprogrammet for Hjortevilt (HOP), Årsrapport for 2006 og 2007; National Veterinary Inst. Report Series; National Veterinary Institute: Oslo, Norway, 2008. [Google Scholar]
- Madslien, K.; Ytrehus, B.; Vikøren, T.; Malmsten, J.; Isaksen, K.; Hygen, H.O.; Solberg, E.J. Hair-loss epizootic in moose (Alces alces) associated with massive deer ked (Lipoptena cervi) infestation. J. Wildl. Dis. 2011, 47, 893–906. [Google Scholar] [CrossRef]
- Rantanen, T.; Reunala, T.; Vuojolahti, P.; Hackman, W. Persistent pruritic papules from deer ked bites. Acta Derm. Venereol. 1982, 62, 307–311. [Google Scholar]
- Laukkanen, A.; Ruoppi, P.; Mäkinen-Kiljunen, S. Deer ked-induced occupational allergic rhinoconjunctivitis. Ann. Allergy Asthma Immunol. 2005, 94, 604–608. [Google Scholar] [CrossRef]
- Decastello, A.; Farkas, R. Anaphylactic reaction following forest fly (Hippobosca equina) bite: A human case. Clin. Exp. Med J. 2010, 4, 193–198. [Google Scholar] [CrossRef]
- Benelli, G.; Duggan, M.F. Management of arthropod vector data–Social and ecological dynamics facing the One Health perspective. Acta Trop. 2018, 182, 80–91. [Google Scholar] [CrossRef]
- Mencke, N. Future challenges for parasitology: Vector control and ‘One health’in Europe: The veterinary medicinal view on CVBDs such as tick borreliosis, rickettsiosis and canine leishmaniosis. Vet. Parasitol. 2013, 195, 256–271. [Google Scholar] [CrossRef]
- Baker, J.R. A review of the role played by the Hippoboscidae (Diptera) as vectors of endoparasites. J. Parasitol. 1967, 53, 412–418. [Google Scholar] [CrossRef]
- Regier, Y.; Komma, K.; Weigel, M.; Pulliainen, A.T.; Göttig, S.; Hain, T.; Kempf, V.A. Microbiome analysis reveals the presence of Bartonella spp. and Acinetobacter spp. in deer keds (Lipoptena cervi). Front. Microbiol. 2018, 9, 3100. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, K.T.; Kwon, O.D.; Ock, Y.; Kim, T.; Choi, D.; Kwak, D. Novel detection of Coxiella spp.; Theileria luwenshuni, and T. ovis endosymbionts in deer keds (Lipoptena fortisetosa). PLoS ONE 2016, 11, e0156727. [Google Scholar]
- Böse, R.; Petersen, K. Lipoptena cervi (Diptera), a potential vector of Megatrypanum trypanosomes of deer (Cervidae). Parasitol. Res. 1991, 77, 723–725. [Google Scholar] [CrossRef]
- Werszko, J.; Steiner-Bogdaszewska, Ż.; Jeżewski, W.; Szewczyk, T.; Kuryło, G.; Wołkowycki, M.; Wróblewski, P.; Karbowiak, G. Molecular detection of Trypanosoma spp. in Lipoptena cervi and Lipoptena fortisetosa (Diptera: Hippoboscidae) and their potential role in the transmission of pathogens. Parasitology 2020, 147, 1–7. [Google Scholar] [CrossRef]
- Hornok, S.; de la Fuente, J.; Biró, N.; Fernández de Mera, I.G.; Meli, M.L.; Elek, V.; Gönczi, E.; Meili, T.; Tánczos, B.; Farkas, R.; et al. First molecular evidence of Anaplasma ovis and Rickettsia spp. in keds (Diptera: Hippoboscidae) of sheep and wild ruminants. Vector Borne Zoonotic Dis. 2011, 11, 1319–1321. [Google Scholar] [CrossRef]
- Víchová, B.; Majláthová, V.; Nováková, M.; Majláth, I.; Čurlík, J.; Bona, M.; Komjáti-Nagyová, M.; Pet’ko, B. PCR detection of re-emerging tick-borne pathogen, Anaplasma phagocytophilum, in deer ked (Lipoptena cervi) a blood-sucking ectoparasite of cervids. Biologia 2011, 66, 1082. [Google Scholar] [CrossRef]
- Doby, J.M.; Bigaignon, G.; Degeilh, B.; Guiguen, C. Ectoparasites of large wild mammals (deer and wild boars) and Lyme borreliosis. Search for Borrelia burgdorferi in more than 1400 ticks, lice, Pupipara Diptera and fleas. Rev. Med. Vet-Toulouse 1994, 145, 743–748. [Google Scholar]
- Hulinsky, V.; Smetana, K. Molecular and microscopical evidence of Ehrlichia spp. and Borrelia burgdorferi sensu lato in patients, animals and ticks in the Czech Republic. New Microbiol. 2002, 25, 437–448. [Google Scholar]
- De Bruin, A.; Van Leeuwen, A.D.; Jahfari, S.; Takken, W.; Földvári, M.; Dremmel, L.; Sprong, H.; Földvári, G. Vertical transmission of Bartonella schoenbuchensis in Lipoptena cervi. Parasites Vectors 2015, 8, 176. [Google Scholar] [CrossRef]
- Korhonen, E.M.; Vera, C.P.; Pulliainen, A.T.; Sironen, T.; Aaltonen, K.; Kortet, R.; Härkönen, L.; Härkönen, S.; Paakkonen, T.; Nieminen, P.; et al. Molecular detection of Bartonella spp. in deer ked pupae, adult keds and moose blood in Finland. Epidemiol. Infect. 2015, 143, 578–585. [Google Scholar] [CrossRef]
- Songailaitė, L.; Klepeckienė, K.; Paulauskas, A.; Radzijevskaja, J. Molecular detection of Bartonella spp. in deer keds Lipoptena cervi and Lipoptena fortisetosa in Lithuania. In Proceedings of the Smart Bio: ICSB 2nd International Conference, Kaunas, Lithuania, 3–5 May 2018; Abstracts book. Vytautas Magnus University: Kaunas, Lithuania, 2018. [Google Scholar]
- Szewczyk, T.; Werszko, J.; Steiner-Bogdaszewska, Ż.; Jeżewski, W.; Laskowski, Z.; Karbowiak, G. Molecular detection of Bartonella spp. in deer ked (Lipoptena cervi) in Poland. Parasites Vectors 2017, 10, 487. [Google Scholar] [CrossRef]
- Andreani, A.; Sacchetti, P.; Belcari, A. Comparative morphology of the deer ked Lipoptena fortisetosa first recorded from Italy. Med. Vet. Entomol. 2019, 33, 140–153. [Google Scholar] [CrossRef]
- Kurina, O.; Kirik, H.; Õunap, H.; Õunap, E. The northernmost record of a blood-sucking ectoparasite, Lipoptena fortisetosa Maa (Diptera: Hippoboscidae), in Estonia. Biodivers. Data J. 2019, 7, e47857. [Google Scholar] [CrossRef]
- Paakkonen, T. Ecophysiology of the Deer Ked (Lipoptena cervi) and Its Hosts. Ph.D. Thesis, University of Eastern Finland, Joensuu, Finland, 2012. Available online: https://core.ac.uk/download/pdf/15169155.pdf (accessed on 23 April 2020).
- Härkönen, S.; Laine, M.; Vornanen, M.; Reunala, T. Deer ked (Lipoptena cervi) dermatitis in humans–an increasing nuisance in Finland. Alces 2009, 45, 73–79. [Google Scholar]
- Kortet, R.; Härkönen, L.; Hokkanen, P.; Härkönen, S.; Kaitala, A.; Kaunisto, S.; Laaksonen, S.; Kekäläinen, J.; Ylönen, H. Experiments on the ectoparasitic deer ked that often attacks humans; preferences for body parts, colour and temperature. Bull. Entomol. Res. 2010, 100, 279. [Google Scholar] [CrossRef]
- Halkman, W. Immigration of Lipoptera cervi (Diptera, Hippoboscidae) in Finland, with notes on its biology and medical significance. Not. Entomol. 1983, 63, 53–59. [Google Scholar]
- Davis, J.W. Deer ked infestation on white-tailed deer in East Texas. J Wildl. Manag. 1973, 37, 183–186. [Google Scholar] [CrossRef]
- Samuel, W.; Trainer, D. Lipoptena mazamae Rondani, 1878 (Diptera: Hippoboscidae) on white-tailed deer in southern Texas. J. Med. Entomol. 1972, 9, 104–106. [Google Scholar] [CrossRef] [PubMed]
- Frischknecht, F. The skin as interface in the transmission of arthropod-borne pathogens. Cell. Microbiol. 2007, 9, 1630–1640. [Google Scholar] [CrossRef] [PubMed]
- Jaenson, T.G.; Tälleklint, L. Incompetence of roe deer as reservoirs of the Lyme borreliosis spirochete. J. Med. Entomol. 1992, 29, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Haarløv, N. Life cycle and distribution pattern of Lipoptena cervi (L.) (Dipt.; Hippobosc.) on Danish deer. Oikos 1964, 15, 93–129. [Google Scholar] [CrossRef]
- Barns, S.M.; Grow, C.C.; Okinaka, R.T.; Keim, P.; Kuske, C.R. Detection of diverse new Francisella-like bacteria in environmental samples. Appl. Environ. Microbiol. 2005, 71, 5494–5500. [Google Scholar] [CrossRef]
- Shabbir, M.Z.; Akram, S.; ul Hassan, Z.; Hanif, K.; Rabbani, M.; Muhammad, J.; Chaudhary, M.H.; Abbas, T.; Ghori, M.T.; Rashid, H.; et al. Evidence of Coxiella burnetii in Punjab province, Pakistan. Acta Trop. 2016, 163, 61–69. [Google Scholar] [CrossRef]
- Watanabe, Y.; Fujihara, M.; Obara, H.; Matsubara, K.; Yamauchi, K.; Harasawa, R. Novel hemoplasma species detected in free-ranging sika deer (Cervus nippon). J. Vet. Med. Sci. 2010, 72, 1527–1530. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.S. Causation and disease: The Henle-Koch postulates revisited. Yale J. Biol. Med. 1976, 49, 175. [Google Scholar]
- Gradmann, C. A spirit of scientific rigour: Koch’s postulates in twentieth-century medicine. Microbes Infect. 2014, 16, 885–892. [Google Scholar] [CrossRef] [PubMed]
- Byrd, A.L.; Segre, J.A. Adapting Koch’s postulates. Science 2016, 351, 224–226. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.S.; Estrada-Peña, A.; Zintl, A. Vectors of babesiosis. Annu. Rev. Entomol. 2019, 64, 149–165. [Google Scholar] [CrossRef] [PubMed]
- Buss, M.; Case, L.; Kearney, B.; Coleman, C.; Henning, J.D. Detection of Lyme disease and anaplasmosis pathogens via PCR in Pennsylvania deer ked. J. Vector Ecol. 2016, 41, 292–294. [Google Scholar] [CrossRef]
- Hornok, S.; Takács, N.; Szekeres, S.; Szőke, K.; Kontschán, J.; Horváth, G.; Sugár, L. DNA of Theileria orientalis, T. equi and T. capreoli in stable flies (Stomoxys calcitrans). Parasites Vectors 2020, 13, 1–6. [Google Scholar] [CrossRef]
- Yanovski, A.D.; Ogston, C.W. Sex differences in size of the blood meal in the bed bug Cimex hemipterus (Hemiptera: Cimicidae). J. Med. Entomol. 1982, 19, 45–47. [Google Scholar] [CrossRef]
- Lehane, M. The Biology of Blood-Sucking in Insects; Cambridge University Press: Cambridge, UK, 2005; pp. 202–258. [Google Scholar]
- Mustonen, A.M.; Käkelä, R.; Paakkonen, T.; Nieminen, P. Life stage-related differences in fatty acid composition of an obligate ectoparasite, the deer ked (Lipoptena cervi)—Influence of blood meals and gender. CBP Part A Mol. Integr. Physiol. 2015, 179, 62–70. [Google Scholar] [CrossRef]
- Kauffmann, M.; Rehbein, S.; Hamel, D.; Lutz, W.; Heddergott, M.; Pfister, K.; Silaghi, C. Anaplasma phagocytophilum and Babesia spp. in roe deer (Capreolus capreolus), fallow deer (Dama dama) and mouflon (Ovis musimon) in Germany. Mol. Cell. Probes 2017, 31, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Sawczuk, M.; Maciejewska, A.; Skotarczak, B. Identification and molecular characterization of Theileria sp. infecting red deer (Cervus elaphus) in northwestern Poland. Eur. J. Wildl. Res. 2008, 54, 225–230. [Google Scholar] [CrossRef]
- Bruno, P.; Bruno, G.; Claudine, P.E. Detection of spirochaetes of Borrelia burgdorferi complexe in the skin of cervids by PCR and culture. Eur. J. Epidemiol. 2000, 16, 869–873. [Google Scholar] [CrossRef]
- Sacristán, C.; das Neves, C.G.; Suhel, F.; Sacristán, I.; Tengs, T.; Hamnes, I.S.; Madslien, K. Bartonella spp. detection in ticks, Culicoides biting midges and wild cervids from Norway. Transbound. Emerg. Dis. 2020, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Dehio, C.; Lanz, C.; Pohl, R.; Behrens, P.; Bermond, D.; Piémont, Y.; Pelz, K.; Sander, A. Bartonella schoenbuchii sp. nov.; isolated from the blood of wild roe deer. Int. J. Syst. Evol. Microbiol. 2001, 51, 1557–1565. [Google Scholar] [CrossRef] [PubMed]
- Astobiza, I.; Barral, M.; Ruiz-Fons, F.; Barandika, J.F.; Gerrikagoitia, X.; Hurtado, A.; García-Pérez, A.L. Molecular investigation of the occurrence of Coxiella burnetii in wildlife and ticks in an endemic area. Vet. Microbiol. 2011, 147, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Oaaland, T.; Christiansen, E.; Jonsson, B.; Kapperud, G.; Wiger, R. A survey of tularemia in wild mammals from Fennoscandia. J. Wildl. Dis. 1977, 13, 393–399. [Google Scholar] [CrossRef]
- Bourque, M.; Higgins, R. Serologic studies on brucellosis, leptospirosis and tularemia in moose (Alces alces) in Quebec. J. Wildl. Dis. 1984, 20, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, A.A.; Hansen, D.E.; Starkey, E.E.; McLean, R.G. Serologic survey of wild cervids for potential disease agents in selected national parks in the United States. Prev. Vet. Med. 1995, 21, 313–322. [Google Scholar] [CrossRef]
- Grazziotin, A.L.; Santos, A.P.; Guimaraes, A.M.S.; Mohamed, A.; Cubas, Z.S.; de Oliveira, M.J.; dos Santos, L.C.; de Moraes, W.; da Costa Vieira, R.F.; Donatti, L.; et al. Mycoplasma ovis in captive cervids: Prevalence, molecular characterization and phylogeny. Vet. Microbiol. 2011, 152, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Grazziotin, A.L.; Duarte, J.M.B.; Szabó, M.P.J.; Santos, A.P.; Guimarães, A.M.S.; Mohamed, A.; da Costa Vieira, R.F.; de Barros Filho, I.R.; Biondo, A.W.; Messick, J.B. Prevalence and molecular characterization of Mycoplasma ovis in selected free-ranging Brazilian deer populations. J. Wildl. Dis. 2011, 47, 1005–1011. [Google Scholar] [CrossRef][Green Version]
- Stefanidesova, K.; Kocianova, E.; Boldis, V.; Kostanova, Z.; Kanka, P.; Nemethova, D.; Spitalska, E. Evidence of Anaplasma phagocytophilum and Rickettsia helvetica infection in free-ranging ungulates in central Slovakia. Eur. J. Wildl. Res. 2008, 54, 519–524. [Google Scholar] [CrossRef]
- Inokuma, H.; Seino, N.; Suzuki, M.; Kaji, K.; Takahashi, H.; Igota, H.; Inoue, S. Detection of Rickettsia helvetica DNA from peripheral blood of sika deer (Cervus nippon yesoensis) in Japan. J. Wildl. Dis. 2008, 44, 164–167. [Google Scholar] [CrossRef]
- Elfving, K.; Malmsten, J.; Dalin, A.M.; Nilsson, K. Serologic and molecular prevalence of Rickettsia helvetica and Anaplasma phagocytophilum in wild cervids and domestic mammals in the central parts of Sweden. Vector Borne Zoonotic Dis. 2015, 15, 529–534. [Google Scholar] [CrossRef]
- Ochirkhuu, N.; Konnai, S.; Odbileg, R.; Murata, S.; Ohashi, K. Molecular epidemiological survey and genetic characterization of Anaplasma species in Mongolian livestock. Vector Borne Zoonotic Dis. 2017, 17, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Bown, K.J.; Lambin, X.; Ogden, N.H.; Petrovec, M.; Shaw, S.E.; Woldehiwet, Z.; Birtles, R.J. High-resolution genetic fingerprinting of European strains of Anaplasma phagocytophilum by use of multilocus variable-number tandem-repeat analysis. J. Clin. Microbiol. 2007, 45, 1771–1776. [Google Scholar] [CrossRef]
- Hilpertshauser, H.; Deplazes, P.; Schnyder, M.; Gern, L.; Mathis, A. Babesia spp. identified by PCR in ticks collected from domestic and wild ruminants in southern Switzerland. Appl. Environ. Microbiol. 2006, 72, 6503–6507. [Google Scholar] [CrossRef] [PubMed]
- Jensen, W.A.; Fall, M.Z.; Rooney, J.; Kordick, D.L.; Breitschwerdt, E.B. Rapid Identification and Differentiation of Bartonella Species Using a Single-Step PCR Assay. J. Clin. Microbiol. 2000, 38, 1717–1722. [Google Scholar] [CrossRef]
- Barbour, A.G.; Maupin, G.O.; Teltow, G.J.; Carter, C.J.; Piesman, J. Identification of an uncultivable Borrelia species in the hard tick Amblyomma americanum: Possible agent of a Lyme disease-like illness. J. Infect. Dis. 1996, 173, 403–409. [Google Scholar] [CrossRef]
- Zheng, W.; Liu, M.; Moumouni, P.F.A.; Liu, X.; Efstratiou, A.; Liu, Z.; Liu, Y.; Tao, H.; Guo, H.; Wang, G.; et al. First molecular detection of tick-borne pathogens in dogs from Jiangxi, China. J. Vet. Sci. 2017, 79, 248–254. [Google Scholar] [CrossRef]
- Long, G.W.; Oprandy, J.J.; Narayanan, R.B.; Fortier, A.H.; Porter, K.R.; Nacy, C.A. Detection of Francisella tularensis in blood by polymerase chain reaction. J. Clin. Microbiol. 1993, 31, 152–154. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Ruttayaporn, N.; Saechan, V.; Jirapattharasate, C.; Vudriko, P.; Moumouni, P.F.A.; Cao, S.; Inpankaew, T.; Ybañez, A.P.; Suzuki, H.; et al. Molecular survey of canine vector-borne diseases in stray dogs in Thailand. Parasitol. Int. 2016, 65, 357–361. [Google Scholar] [CrossRef]
- Labruna, M.B.; Whitworth, T.; Horta, M.C.; Bouyer, D.H.; McBride, J.W.; Pinter, A.; Popov, V.; Gennari, S.M.; Walker, D.H. Rickettsia species infecting Amblyomma cooperi ticks from an area in the state of Sao Paulo, Brazil, where Brazilian spotted fever is endemic. J. Clin. Microbiol. 2004, 42, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, Z.; Liu, Z.; Liu, J.; Yang, J.; Li, Q.; Li, Y.; Cen, S.; Guan, G.; Ren, Q.; et al. Molecular identification of Theileria parasites of northwestern Chinese Cervidae. Parasites Vectors 2014, 7, 1–7. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Molecular Evolutionary Genetics Analysis. Available online: https://www.megasoftware.net (accessed on 3 September 2019).
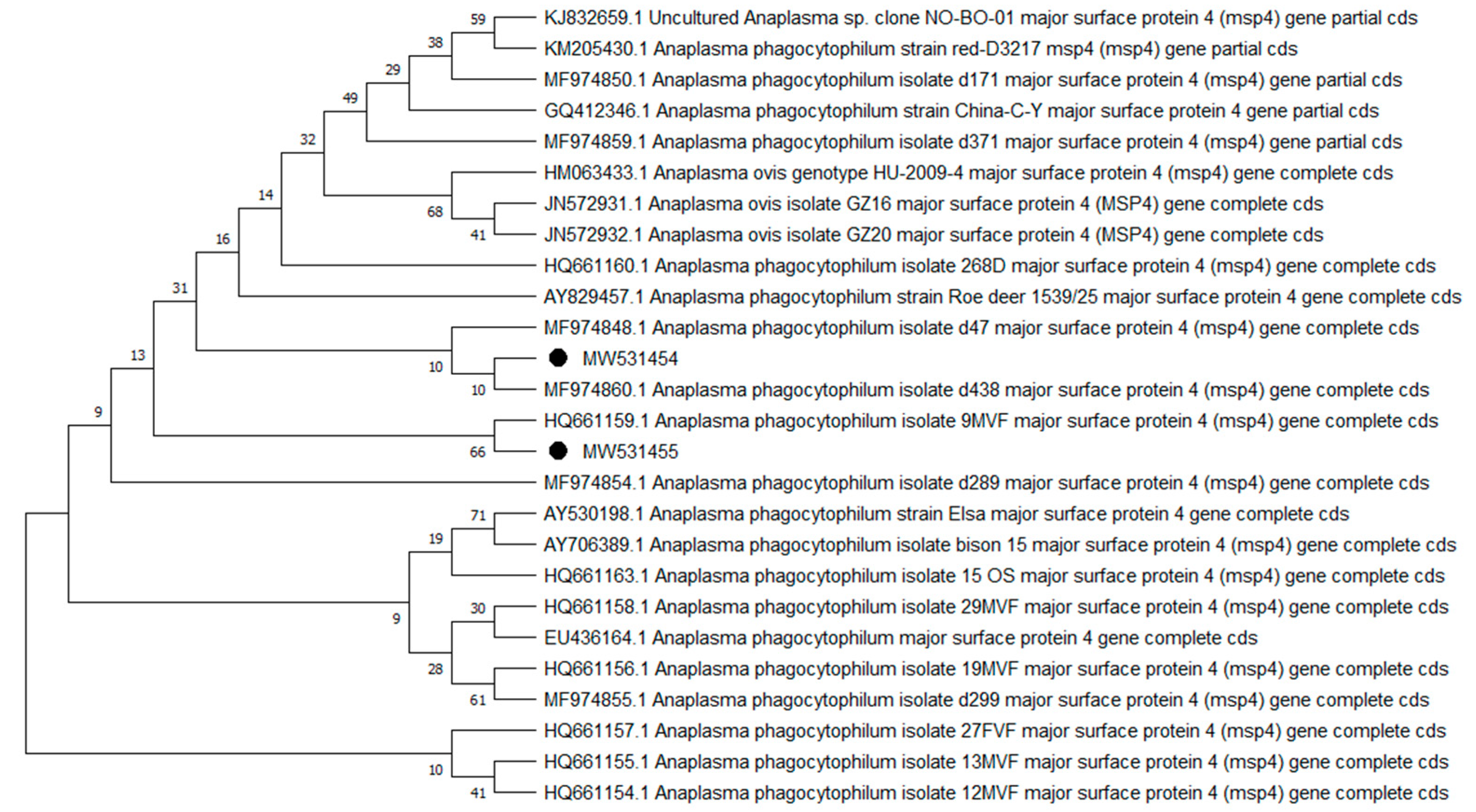

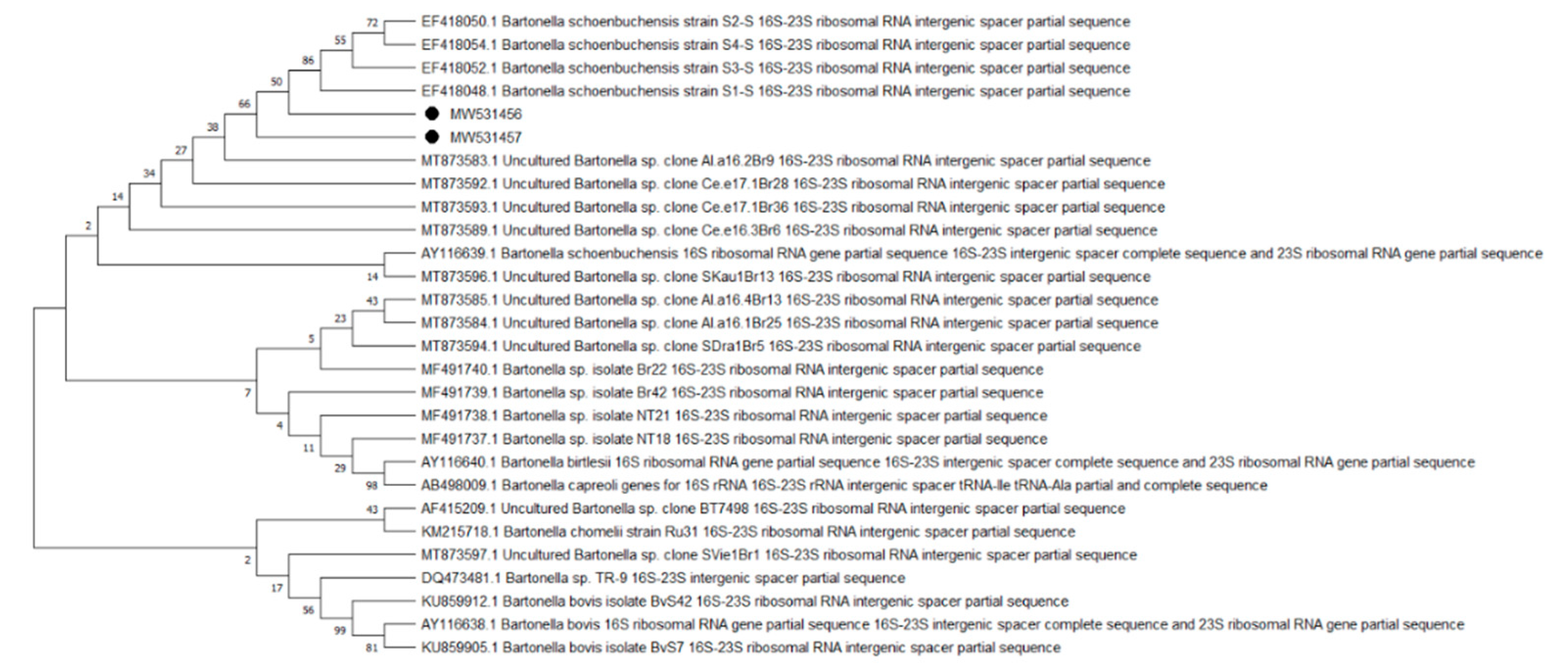





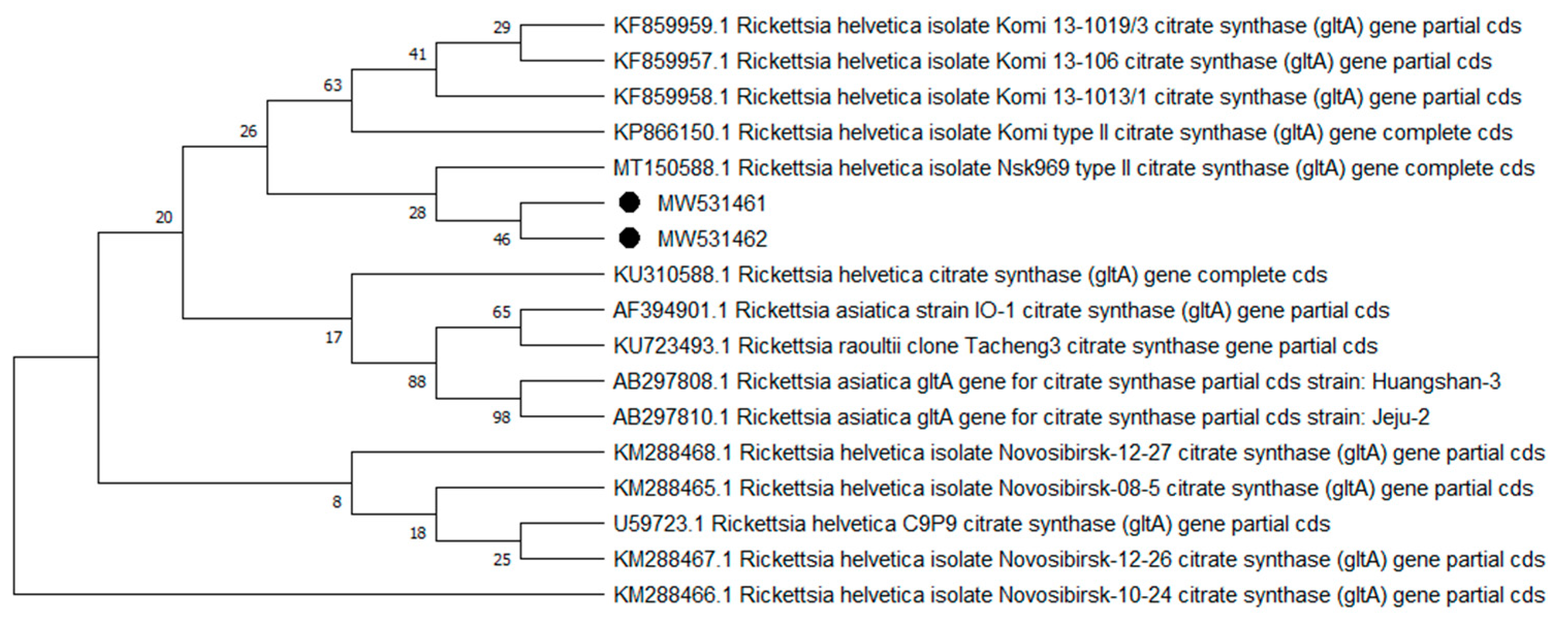
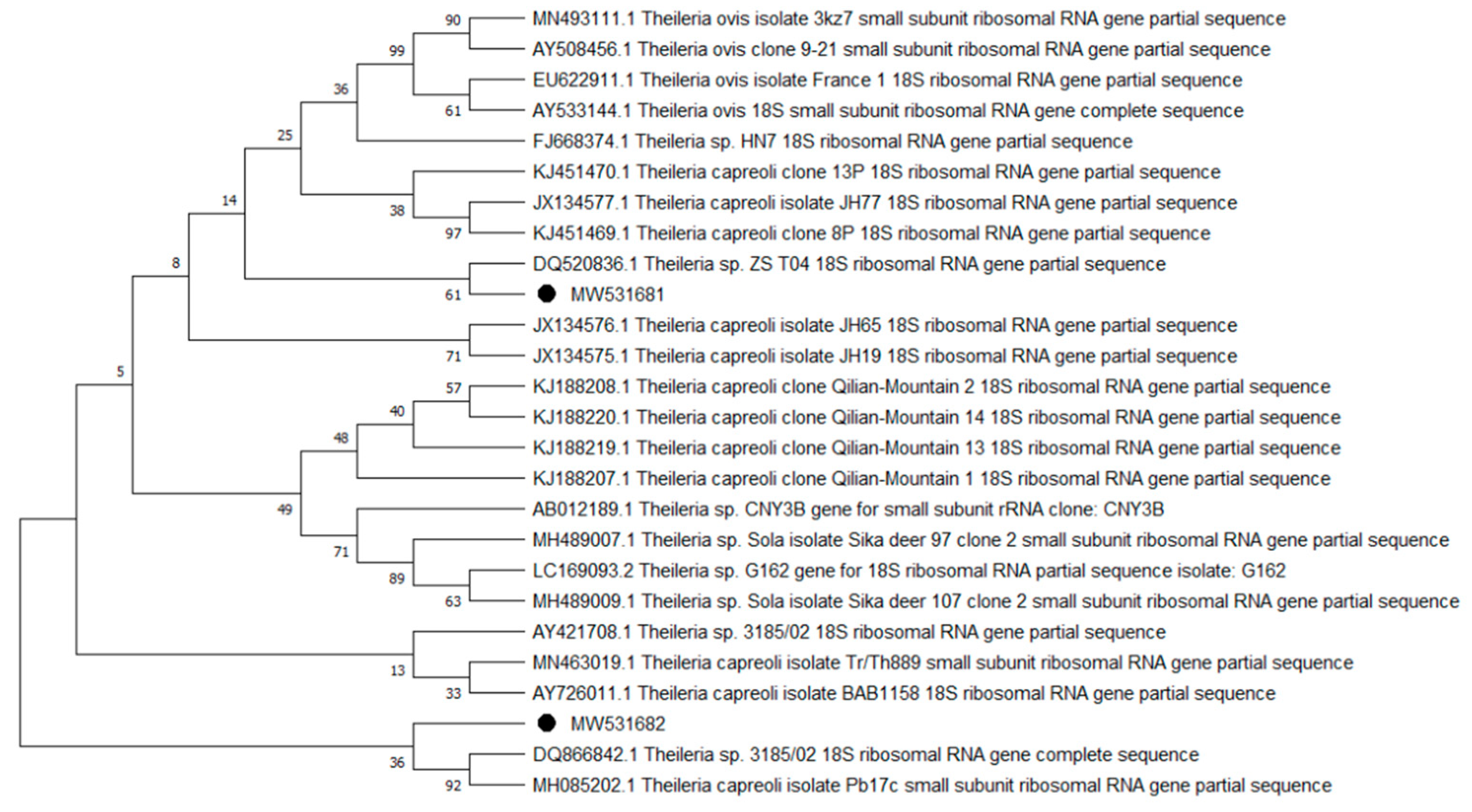
| Species | Group A | Group E | Total n-500 | ||
|---|---|---|---|---|---|
| n-250 | n-250 | ||||
| M | F | M | F | ||
| n-99 | n-151 | n-115 | n-135 | ||
| Anaplasma ovis | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Anaplasma phagocytophilum | 6 (6.0%) | 14 (9.3%) | 0 (0%) | 0 (0%) | 20 (4%) |
| Babesia spp. | 21 (21.21%) | 30 (19.86%) | 0 (0%) | 0 (0%) | 51 (10.2%) |
| Bartonella spp. | 64 (64.65%) | 94 (62.25%) | 37 (32.17) | 52 (38.52%) | 247 (49.4%) |
| Borrelia spp. | 3 (3.03%) | 9 (6.6%) | 0 (0%) | 0 (0%) | 12 (24.30%) |
| Coxiella spp. | 3 (3.03%) | 9 (6.6%) | 1 (2.87%) | 2 (1.48%) | 15 (3%) |
| Francisella spp. | 2 (2.02%) | 5 (3.31%) | 0 (0%) | 2 (1.48%) | 9 (1.8%) |
| Mycoplasma spp. | 22 (22.22%) | 52 (34.44%) | 9 (7.82%) | 8 (5.93%) | 91 (18.2%) |
| Rickettsia spp. | 30 (30.30%) | 46 (30.46%) | 14 (12.17%) | 16 (3.2%) | 106 (21.2%) |
| Theileria spp. | 22 (22.22%) | 27 (17.88%) | 0 (0%) | 0 (0%) | 49 (9.8%) |
| Parasite Species (Dependent Variables) | p-Value (for the Model) | Independent Variables | SE | W | p-Value * | OR | |
|---|---|---|---|---|---|---|---|
| Total number | <0.001 | sex | M | 0.32 | 4.06 | 0.044 | 0.584 |
| F | 1.476 | ||||||
| group | A | 0.22 | 92.32 | <0.001 | 8.04 | ||
| E | 0.12 | ||||||
| Anaplasma phagocytophilum | sex | M | 0.56 | 2.85 | 0.03 | 1.24 | |
| F | 0.88 | ||||||
| Babesia spp. | site | WM | 0.54 | 4.33 | 0.037 | 3.09 | |
| L | 0.55 | 3.67 | 0.045 | 2.85 | |||
| Bartonella spp. | group | A | 0.20 | 26.04 | <0.001 | 2.803 | |
| E | 0.36 | ||||||
| Coxiella spp. | group | A | 0.582 | 4.445 | 0.035 | 3.410 | |
| E | 0.293 | ||||||
| Mycoplasma spp. | group | A | 0.283 | 35.53 | <0.001 | 5.40 | |
| E | 0.19 | ||||||
| Rickettsia spp. | group | A | 0.226 | 16.559 | <0.001 | 2.51 | |
| E | 0.40 | ||||||
| Theileria spp. | site | L | 0.653 | 6.483 | 0.011 | 0.19 | |
| Species | Anaplasma phagocytophilum | Babesia spp. | Bartonella spp. | Borrelia spp. | Coxiella spp. | Francisella tularensis | Mycoplasma spp. | Rickettsia spp. | Theileria spp. |
|---|---|---|---|---|---|---|---|---|---|
| Anaplasma phagocytophilum | - | 0.033 b | 0.013 b | 0.21 *,a | 0.20 *,a | 0.057 b | 0.062 b | 0.035 b | 0.040 b |
| Babesia spp. | 0.079 a | - | 0.024 b | 0.026 b | 0.029 b | 0.11 b | 0.041 b | 0.011 b | 0.050 b |
| Bartonella spp. | 0.026 a | 0.094 *,a | - | 0.005 b | 0.016 b | 0.037 b | 0.029 b | 0.009 b | 0.04 b |
| Borrelia spp. | 0.21 *,a | 0.077 a | 0.032 a | - | 0.032 b | 0.043 b | 0.1 b | 0.027 b | 0.078 b |
| Coxiella spp. | 0.16 *,a | 0.010 a | 0.037 a | 0.043 a | - | 0.045 b | 0.073 b | 0.080 b | 0.025 b |
| Francisella spp. | 0.035 a | 0.12 *,a | 0.024 a | 0.025 a | 0.30 a | - | 0.063 b | 0.059 b | 0.041 b |
| Mycoplasma spp. | 0.01 a | 0.13 *,a | 0.028 a | 0.128 *,a | 0.032 a | 0.13 a | - | 0.028 b | 0.055 b |
| Rickettsia spp. | 0.01 a | 0.069 a | 0.077 a | 0.0080 a | 0.11 *,a | 0.039 a | 0.050 a | - | 0.046 b |
| Theileria spp. | 0.08 *,a | 0.16 *,a | 0.11 a | 0.12 *,a | 0.012 a | 0.008 a | 0.052 a | 0.090 a | - |
| Species | Target Gene | Name of Primer | Primer Sequence (5′–3′) | Expected Size (bp) | PCR Cycle Conditions | Reference |
|---|---|---|---|---|---|---|
| Anaplasma ovis | MSP4 | MSP43R | CCG GAT CCT TAG CTG AAC AGG AAT CTT GC | 347 | 94 °C/5 min; 40 cycles: 94 °C/30 s, 60 °C/30 s, 72 °C/1 min; 72 °C/7 min | Ochirkhuu et al. [81] |
| MSP45F | GGG AGC TCC TAT GAA TTA CAG AGA ATT GTT TAC | |||||
| Anaplasma phagocytophilum | MSP4 | Aphamsp4F | ATGAATTACAGAGAATTGCTTGTAGG | 849 | 94 °C/5 min; 40 cycles: 94 °C/10 s, 58 °C/10 s, 72 °C/50 s; 72 °C/5 min | Bown et al. [82] |
| Aphamsp4R | TTAATTGAAAGCAAATCTTGCTCCTATG | |||||
| Bovine * Babesia spp. | 18S rRNA | BabsppF1 | GTTTCTGMCCCATCAGCTTGAC | 440 | 45 cycles: 94 °C/30 s, 61 °C/30 s, 72 °C/45 s; 72 °C/10 min | Hilpertshauser et al. [83] |
| BabsppR | CAAGACAAAAGTCTGCTTGAAAC | |||||
| Bartonella spp. | 16–23S gene | Bartonella spp. F | (C/T)CTTCGTTTCTCTTTCTTCA | 154-260 | 95 °C/2min; 45 cycles: 95 °C/1 min, 60 °C/1 min, 72 °C/30 s; 72 °C/5 min | Jensen et al. [84] |
| Bartonella spp. R | AACCAACTGAGCTACAAGCC | |||||
| Borrelia spp. | flagellin | Borrelia spp. flaF | ACATATTCAGATGCAGACAGAGGT | 350 | 95 °C/5 min; 40 cycles: 95 °C/1 min, 55 °C/1 min, 72 °C/30 s; 72 °C/5 min | Barbour et al. [85] |
| Borrelia spp. flaR | AACAGCTGAAGAGCTTGGAATG | |||||
| Coxiella burnetii | 16S rRNA | Coxiella 16SrRNA F | ATTGAAGAGTTTGATTCTGG | 1457 | 95 °C/3 m; (2 cycles: 95 °C/30 s, 58 °C/30 s, 72 °C/2 min; 10 cycles: 95 °C/30 s, 58–50 °C/30 s (–2 every 2 cycles), 72 °C/2 min; 30 cycles: 95 °C/30 s, 48 °C/30 s, 72 °C/2 min); 72 °C/5 min | Zheng et al. [86] |
| Coxiella 16SrRNA R | CGGCTTCCCGAAGGTTAG | |||||
| Francisella tularensis | TUL4 | Ff393 | ATGGCGAGTGATACTGCTTG | 248 | 98 °C/30 s; 35 cycles: 98 °C/10 s, 53.4 °C/ 30 s, 72 °C/1 min; 72 °C/10 min | Long et al. [87] |
| Ff642 | GCATCATCAGAGCCACCTAA | |||||
| Mycoplasma spp. | 16S rRNA | HBT-F | ATA CGG CCC ATA TTC CTA CG | 595 | 94 °C/10 min; 40 cycles: 95 °C/30 s, 60 °C/30 s, 72 °C/30 s; 72 °C/10 min | Liu et al. [88] |
| HBT-R | TGC TCC ACC ACT TGT TCA | |||||
| Rickettsia spp. | gltA | Rickettsia gltA F | GCAAGTATCGGTGAGGATGTAAT | 401 | 95 °C/3 min; 40 cycles: 95 °C/15 s, 48 °C/30 s, 72 °C/30 s; 72 °C/7 min | Labruna et al. [89] |
| Rickettsia gltA R | GCTTCCTTAAAATTCAATAAATCAGGAT | |||||
| Theileria spp. | 18S rRNA | 989 | AGT TTC TGA CCT ATC AG | 1098 | 94 °C/5 min; 35 cycles: 94 °C/30 s, 53 °C/30 s, 72 °C/1 min; 72 °C/7 min | Li et al. [90] |
| 990 | TTG CCT TAA ACT TCC TTG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gałęcki, R.; Jaroszewski, J.; Bakuła, T.; Galon, E.M.; Xuan, X. Molecular Detection of Selected Pathogens with Zoonotic Potential in Deer Keds (Lipoptena fortisetosa). Pathogens 2021, 10, 324. https://doi.org/10.3390/pathogens10030324
Gałęcki R, Jaroszewski J, Bakuła T, Galon EM, Xuan X. Molecular Detection of Selected Pathogens with Zoonotic Potential in Deer Keds (Lipoptena fortisetosa). Pathogens. 2021; 10(3):324. https://doi.org/10.3390/pathogens10030324
Chicago/Turabian StyleGałęcki, Remigiusz, Jerzy Jaroszewski, Tadeusz Bakuła, Eloiza M. Galon, and Xuenan Xuan. 2021. "Molecular Detection of Selected Pathogens with Zoonotic Potential in Deer Keds (Lipoptena fortisetosa)" Pathogens 10, no. 3: 324. https://doi.org/10.3390/pathogens10030324
APA StyleGałęcki, R., Jaroszewski, J., Bakuła, T., Galon, E. M., & Xuan, X. (2021). Molecular Detection of Selected Pathogens with Zoonotic Potential in Deer Keds (Lipoptena fortisetosa). Pathogens, 10(3), 324. https://doi.org/10.3390/pathogens10030324









