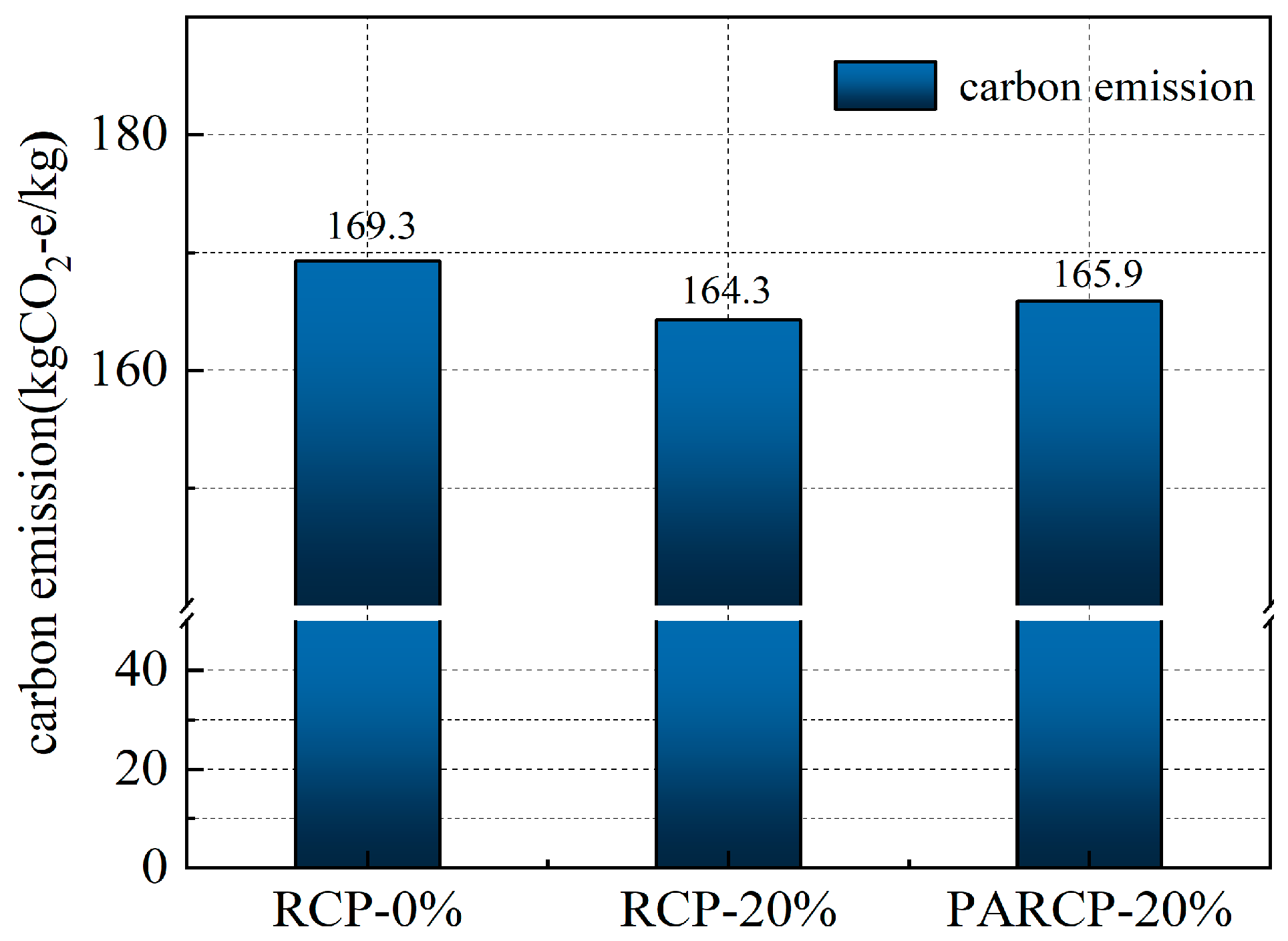Abstract
Recycled concrete powder (RCP) utilization as an auxiliary cementitious material absorbs construction waste and promotes low-carbon transition in construction by replacing high-carbon materials. This study optimized RCP’s particle size and amorphous SiO2 content through physical activation, systematically investigating its effects on alkali-activated cementitious materials (AACMs). The results demonstrated that 20% activated RCP enhanced compressive strength by 9% (34.2 MPa), only 12.7% lower than that of the reference samples. Hydration analysis revealed activated RCP delayed exothermic peaks but increased total heat via active particles. Life-cycle assessment showed substituting 20% ground granulated blast-furnace slag (GGBS)/fly ash (FA) with RCP reduced carbon emissions from 169.3 to 165.9 kg CO2-e/ton (−2.1%). Although activation slightly raised emissions to 166.6 kg CO2-e/ton, RCP’s carbon contribution remained at 9% versus GGBS’s 83% dominance. Crucially, the activation’s 0.7 kg CO2-e/ton increase was offset by 4.7 kg CO2-e/ton reductions from material substitution and waste recycling benefits, confirming its net carbon-neutral potential.
1. Introduction
Ordinary Portland cement (OPC) and its derived concrete are widely used in various construction projects around the world due to their superior strength and excellent plasticity, and have become the most common building materials [1,2]. With the continuous development of the global economy and the expansion of the scale of transportation and civil facilities, the demand for OPC is increasing. According to relevant research, the production of one ton of OPC produces one ton of CO2, and the carbon emissions caused by the construction sector will reach nearly 10 billion tons, accounting for 37% of the total global CO2 emissions [3,4]. Therefore, it is urgent to find a green cementitious material with low energy consumption, low pollution and high performance. On the other hand, with the continuous advancement of modern social processes around the world, the emissions of building solid waste generated during construction, renovation and demolition are increasing year by year. According to statistics, the annual emissions of global construction solid waste have exceeded ten billion tons [5,6]. These large amounts of construction solid waste are randomly stacked or simply landfilled, which not only leads to the waste and scarcity of land resources but also poses serious environmental pollution [7,8,9]. Therefore, many researchers are exploring how to realize the recycling of construction solid waste to increase economic, social and environmental benefits.
At present, the valorization of construction solid waste is in the use of recycled aggregate to prepare recycled concrete. By crushing it, recycled coarse aggregate (5–31.5 mm) and recycled fine aggregate (0.15–5 mm) can be obtained. In this process, a certain amount of active RCP (<0.75 mm) will be produced. These three materials can replace natural aggregate to prepare mortar or concrete. All these recycled products can partially or fully replace natural aggregates for the preparation of mortar and concrete. In particular, a study [10] focusing on recycled concrete from the precast concrete industry verified that high-quality multi-recycled coarse aggregates can achieve equivalent or even superior compressive strength to ordinary concrete at a 100% replacement rate of natural coarse aggregate, which further validates the engineering application feasibility of such recycled products.
The main composition of the recycled micropowder is similar to that of FA, showing a potential pozzolanic effect and good microaggregate filling function [11]. Its main components are unhydrated cement slurry and some inert materials. Xiao et al. [12] used waste concrete powder as an auxiliary cementitious material to prepare foamed concrete. With the increase in dosage, the thermal conductivity of concrete gradually decreased. When the content reached 30%, the 28 d compressive strength of foam concrete was reduced by 59.9%. W et al. [13] showed that the inactive particles of RCP filled the gap between hydration products, which was beneficial to the densification of microstructure. Moreover, due to its low particle size distribution, RCP can be utilized as an ultra-fine aggregate for the preparation of cementitious materials. The addition of a small amount of RCP helps to improve the particle size distribution and increase the strength. Qin et al. [3] used RCP to prepare recycled concrete and found that when the fineness of RCP is less than the fineness of cement, the use of RCP can improve the durability of concrete. However, due to its low activity, it is difficult to apply in practical engineering. Given this background, scholars across the globe have performed systematic investigations focusing on the activation treatment of RCP to improve its reactivity. The current main activated methods include chemical activation, thermal activation or physical grinding to improve its activity [14,15,16,17]. Wu et al. [18] showed that thermal activation could promote the formation of new active ingredients in RCP, with the highest activity index of 90%. Some researchers have used pretreatment methods to improve RCP activity and replace cement-based cementing materials to prepare alkali-activated mortar. The results show that both calcination and carbonization can improve the properties of RCP, and RCP exhibits better mechanical properties and lower temporary pore size at 600 °C. Increasing the carbonization time of RCP increases its internal calcite content. With the increase in substitution rate, the mechanical properties of alkali-activated mortar decrease gradually. Liu et al. [19] used MgO-Na2CO3 to activate RCP. Through the combined activated effect between the two, a large amount of gel material was generated in an alkaline environment to fill the pores of the structure, thus forming a dense microstructure. Chen et al. [20] demonstrated that chemical activation enabled the mortar activity index to reach 68.3% when RCP was incorporated at a replacement rate of 30%. In contrast, the activity index of the thermal activation system at 700 °C reached 77.4%. Although thermal activation technology and chemical activation significantly increase the activity index of RCP, the calcination cost is high, the energy consumption is high, and carbon emissions are increased, making it difficult to apply in practical engineering.
Compared with thermal activation and chemical activation, physical grinding of RCP can make the powder finer and more regular, which is beneficial in improving the early activity of the powder. Yang et al. [21] showed that the particle size distribution of RCP can be optimized by physical activation. It released internal unhydrated cement particles and encapsulated active substances to increase its specific surface area. At the same time, due to the action of mechanical force, SiO2 in RCP was transformed into an amorphous state, and the amorphous state of SiO2 increased the activity of RCP. Gao et al. [22] showed that when RCP was added at a proportion of 30%, an activity index of 77.79% could be achieved with 1 h of physical activation treatment, which was beneficial to the formation of ettringite (AFt) and CaCO3 phase. A study by Wang physically activated RCP by selecting grinding times of 15, 30, 45, 60, and 75 min. The research indicated that beyond 60 min, the content of fine particles within 10–20 μm increased significantly within the RCP, with pronounced agglomeration phenomena [23]. However, existing research still has significant limitations and has yet to establish a systematic technical framework or scientific understanding. First, it primarily focuses on the macroscopic effects of physical activation on RCP activity, lacks in-depth analysis of its regulation of the microscopic hydration mechanism of RCP in alkaline activation systems, and fails to clarify the core mechanism behind performance enhancement. Second, it has not systematically quantified the carbon emission reduction benefits of physical activation nor established relevant correlation relationships, making it difficult to support low-carbon applications. According to the life cycle assessment, the carbon emissions per ton of construction waste generated by each treatment far exceed the carbon emissions generated by physical activation due to the complexity of the process, whether it is calcination or carbonization [24,25,26].
Under the global initiative of advancing low-carbon development and emission reduction, the problems of high energy consumption and pollution in the cement production process have become increasingly prominent—cement production alone accounts for approximately 8% of global carbon emissions. Meanwhile, the insufficient recycling rate of industrial solid waste (only 30–50%) has formed a dual environmental challenge that urgently needs to be addressed. Alkali-activated cementitious materials (AACMs) are cementitious materials generated by the alkaline activation reaction of potential cementitious components and alkaline activators; they have the advantages of simple preparation, low energy consumption, excellent corrosion resistance and high-temperature resistance. Compared with traditional Portland cement, AACMs emit less than 40% of the CO2, along with additional merits, such as low water demand, low hydration heat, high strength and good durability. Due to these low-carbon characteristics and strong capacity for absorbing solid waste, AACMs have become the focus of current research, as they can react with industrial solid wastes (e.g., GGBS and FA) via alkaline activators, realizing the dual-path coupling of “solid waste recycling + carbon emission reduction.”
Previous studies on recycled concrete powder (RCP) have mainly focused on chemical and thermal activation methods. Both of these widely used activation approaches have notable limitations: thermal activation requires high-temperature calcination to enhance the pozzolanic activity of RCP, which inevitably leads to high energy consumption and additional carbon emissions; and chemical activation generally relies on strong alkaline activators, which not only increases material costs, but also brings potential risks of alkali-aggregate reaction and poor volume stability of the hardened products. In contrast, physical activation is more feasible and advantageous for engineering applications, avoiding the drawbacks of thermal and chemical activation. Furthermore, in OPC systems, the physical activation of RCP mainly enhances material performance through the micro-aggregate filling effect and a weak pozzolanic reaction, with only limited improvement in reaction activity. In AACM systems, physical activation optimizes the particle size of RCP and increases its amorphous SiO2 content, allowing it to react sufficiently with alkali activators to produce more C–S–H or C–A–S–H gels, thereby achieving a much stronger enhancement in reactivity. Meanwhile, AACMs themselves are a low-carbon binder system, and their combination with physically activated RCP realizes the coupling effect of solid waste utilization and carbon emission reduction, which is not attainable in OPC systems. This study innovatively constructed a technical framework of “solid waste regeneration–alkali-activated synergistic carbon reduction.” Specifically, RCP was physically activated by controlling the grinding time, and the changes in its particle size distribution were analyzed; the activity index of RCP was evaluated in accordance with the standard JG/T 573-2020 [27] “Recycled micropowder for concrete and mortar.” Subsequently, RCP treated by different methods was used to prepare cementitious materials under alkali-activated conditions. Moreover, the hydration mechanism and the characteristics of resultant products of RCP in alkali-activated systems were analyzed via XRD, SEM and hydration heat analysis. This study not only improved the replacement rate of RCP in AACMs to 20% through physical activation but also systematically quantified its carbon emission reduction benefits, reducing CO2 emissions to 165.9 kg CO2-e/ton and achieving a carbon sink effect of 𢄬3.4 kg CO2-e/ton via solid waste recycling. Compared to traditional thermal activation and chemical activation methods, it offers greater engineering applicability and low-carbon environmental advantages. This study ultimately provides sufficient scientific support for the optimized application of recycled micropowder in alkali-activated systems and forms a collaborative paradigm that integrates high-value utilization of construction solid waste with precise carbon emission regulation, which is of great significance for boosting the recycling and resource utilization of construction solid waste.
2. Materials and Methods
2.1. Materials
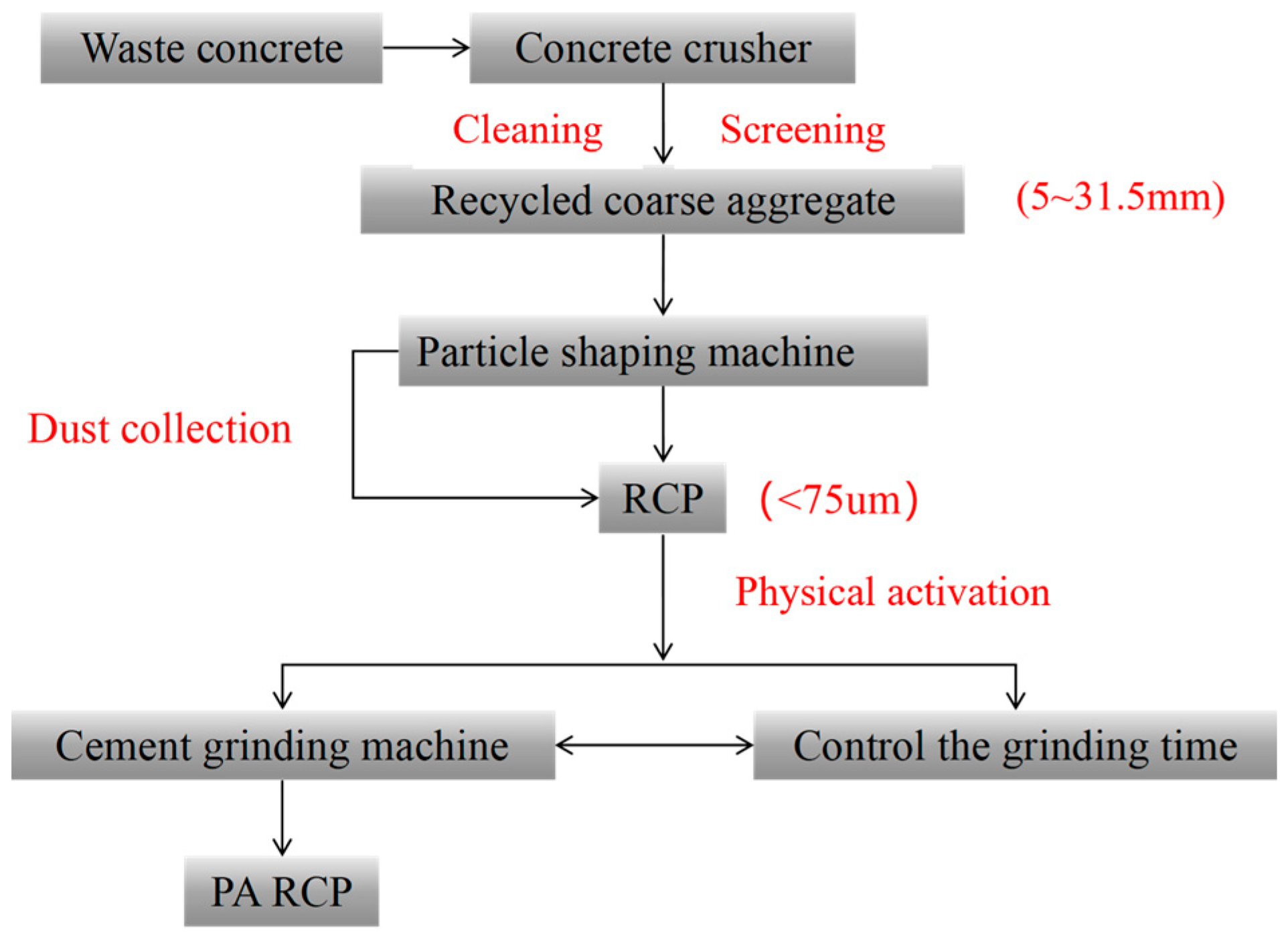
The cementitious materials employed in this study were RCP, GGBS, and FA. All raw materials were supplied by Shandong Qingdao Green Sail Recycled Building Materials Co., Ltd., a formal local supplier with a stable and sufficient material supply. The RCP was produced from construction and demolition waste (mainly waste concrete) via a sequential process, including crushing, cleaning, particle shaping, and dry screening, followed by dust collection to obtain fine powder with a particle size of less than 0.75 mm.
Table 1 provides the chemical composition of the alkali-activated precursor (GGBS, FA, and RCP), obtained via X-ray fluorescence spectroscopy. The main component of GGBS is CaO, accounting for 51.49% of its main components. FA mainly comprises SiO2 and Al2O3, which account for 72.88% of the total composition. The composition of RCP is similar to that of FA, with the mass fraction of SiO2 reaching 59.63%, followed by Al2O3.

Table 1.
Chemical composition of cementitious materials (%).
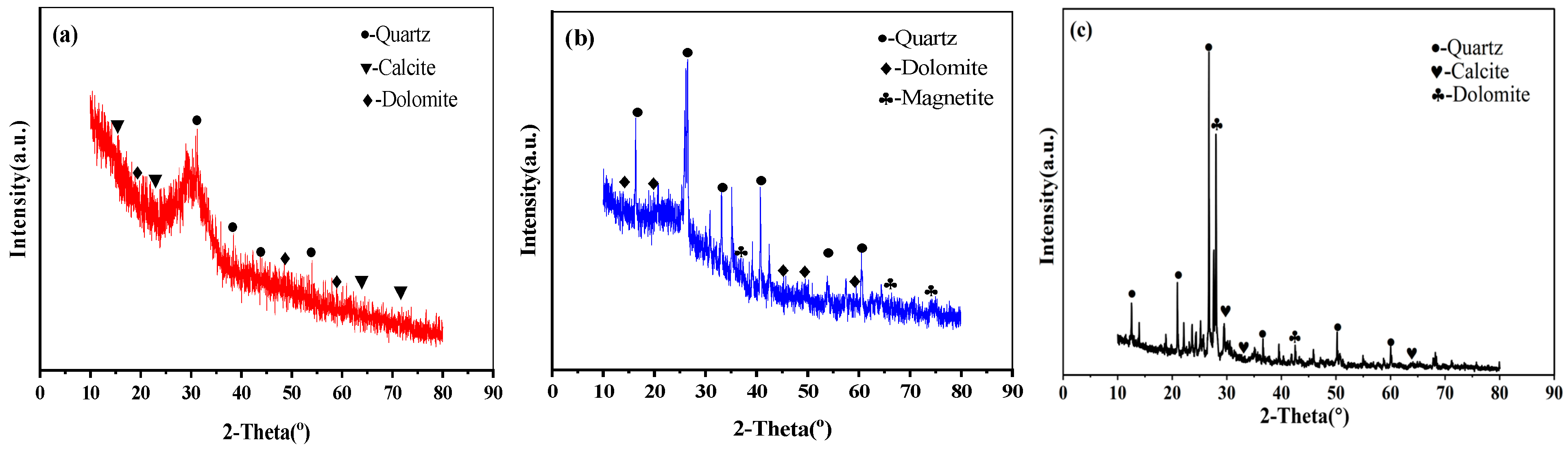
Figure 1 shows the XRD analysis patterns of GGBS, FA and RCP. The XRD pattern of RCP shows that its main crystal phase is SiO2 and that it contains a large amount of albite and some CaCO3. The presence of CaCO3 in RCP can be attributed to the existence of a certain amount of Ca(OH)2 in the mortar attached to the surface of the recycled aggregate or in the hardened cement paste of waste concrete, which reacts with CO2 in the air.
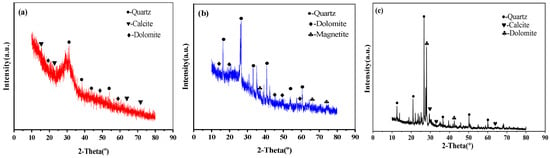
Figure 1.
XRD of GGBS, FA, and RCP: (a) GGBS; (b) FA; and (c) RCP.
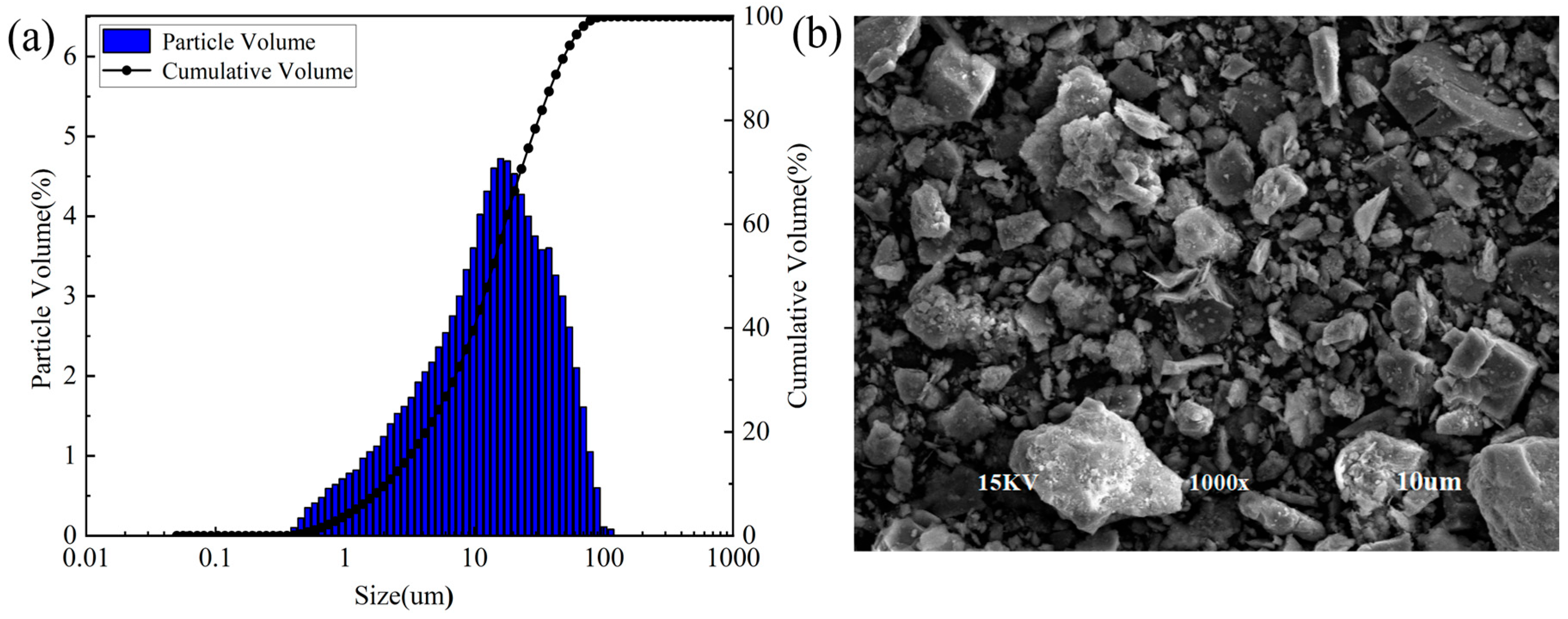
Figure 2 shows the particle size distribution, particle size accumulation and SEM images spectra of RCP. The results of the particle size analysis suggest that the size of RCP particles is distributed within 0.1–100 μm, with a peak particle size of 15.99 μm. The volume fraction of particles less than 10 μm is determined as 39.51%, and particles with a size below 14.14 μm constitute 50% of the total sample. The SEM image of RCP indicates that RCP particles have irregular shapes, with the edges and corners of the particles clearly observed. The surface of some particles is rough and uneven, and there is a large number of pores.
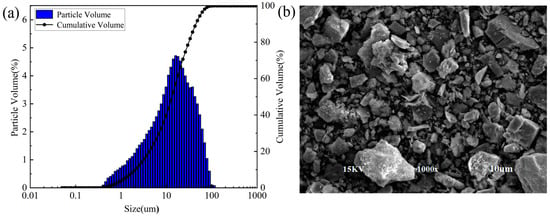
Figure 2.
Particle size distribution and SEM image of RCP: (a) particle size distribution and (b) SEM image of RCP.
The alkaline activator for the experiment was prepared from an aqueous sodium silicate solution with a modulus of 3.3 and a Na2O concentration of 8.53%, solid sodium hydroxide with a purity of 99%, and tap water, yielding an alkali solution with a modulus of 1 and a concentration of 3.5%. The employed sand was stored for 24 h before the test, with the second type of recycled sand used in the test. The properties of the recycled sand are shown in Table 2.

Table 2.
Performance indicators of recycled sand.
2.2. Preparation of AACMs
Pretreatment of RCP: The cement test mill manufactured by the Tianjin Gangyuan Experimental Instrument Factory was used to mechanically grind RCP; the grinding time was 15, 30, and 45 min. Before the test begins, remove the RCP and dry it to a constant weight to prevent increased moisture content from affecting the grinding test results. In each experiment, 2 kg of RCP was ground. The preparation process is shown in Figure 3. The resulting RCP samples were labeled RCP-M, where M is the grinding time in minutes. For example, RCP-15 represents the RCP ground for 15 min.
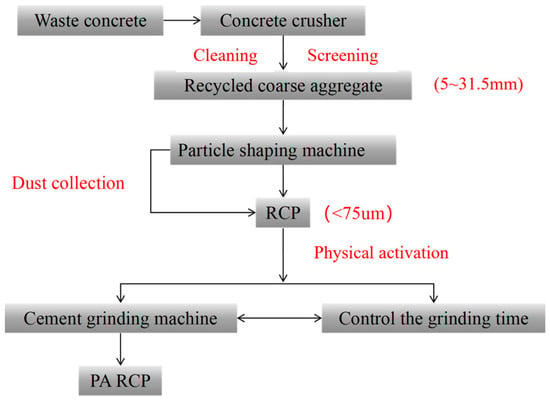
Figure 3.
Preparation and activation of RCP.
Kaptan et al. [28] reported that for use as an auxiliary cementitious material, RCP must exhibit high reactivity and small particle size. These properties facilitate hydration by promoting nucleation, generating new hydration products, and enhancing the density of the material through the filling effect.
Preparation of the AACMs: First, the GGBS, FA, reclaimed sand, and RCP ground for different durations were added to a stirring pot in predetermined proportions. The mixture was homogenized using a JJ-5 cement mixer for 30 s, followed by the addition of the alkaline activator (the above aqueous solution of Na2SiO3 and NaOH) and mixing for 1 min. The prepared slurry was poured into a mold (40 mm × 40 mm × 160 mm) and shaken on the vibration table for 25 s to remove bubbles. Finally, the surface of the slurry was coated with a polyethylene film to prevent water loss. Among them, the preparation of the PAAACMs (AACMs incorporating activated RCP) was selected as the group of RCP with the highest activity index.
2.3. Material Test Methods
The RCP activity index was evaluated according to the JG/T 573-2020 [27] “Recycled micropowder for concrete and mortar” standard: the mortar samples with the 30% replacement rate were prepared using the unactivated and activated RCP. In addition, the pozzolanic activity of RCP ground for different durations was determined, according to the activity index evaluation method in the standard (the 28 d compressive strength of the tested mortar specimens was contrasted against that of the control mortar).
The fluidity of the prepared mortar was tested according to the GB/T 2419-2005 [29] “cement mortar fluidity test standard.” and the flexural and compressive strengths of the prepared mortar samples were tested according to the JGJ/T 70-2009 [30] “building mortar basic performance test standar.”
The hydration heat of the AACMs was tested according to the GB/T 12959-2008 [31] “Determination method of cement hydration heat.” standard. Three groups of samples (without RCP, with unactivated RCP, and with activated RCP; RCP content = 20% in all cases) were used to prepare paste samples (removal of reclaimed sand) and placed in insulation bottles. A TAM Air eight-channel microcalorimeter (USA) was used to measure the heat flow of the samples for 72 h.
Phase compositions of the prepared samples were identified by X-ray diffraction on a Rigaku Miniflex 600 system Rigaku Corporation, Tokyo, Japan, with data collected at 10°/min within the 2θ range of 10° to 80°. A ZEISS GeminiSEM 300 microscope Carl Zeiss AG, Oberkochen, Germany was employed to characterize the microstructural features of the samples.2.4 Mixing ratio.
AACMs: Two groups of mortar specimens with different cementitious systems were prepared, group A (RCP not activated) and group B (RCP activated). RCP replaced the GGBS and FA at substitution rates of 0%, 10%, 20%, and 30%. The ratio of residual GGBS to FA by mass was 7:3. The sol ratio of the two groups was 0.5, the mortar ratio was 1:2, with the mix ratio shown in Table 3.

Table 3.
The mixing ratios of the AACMs (g).
3. Results
3.1. The Properties and Structure of the Preprocessed RCP
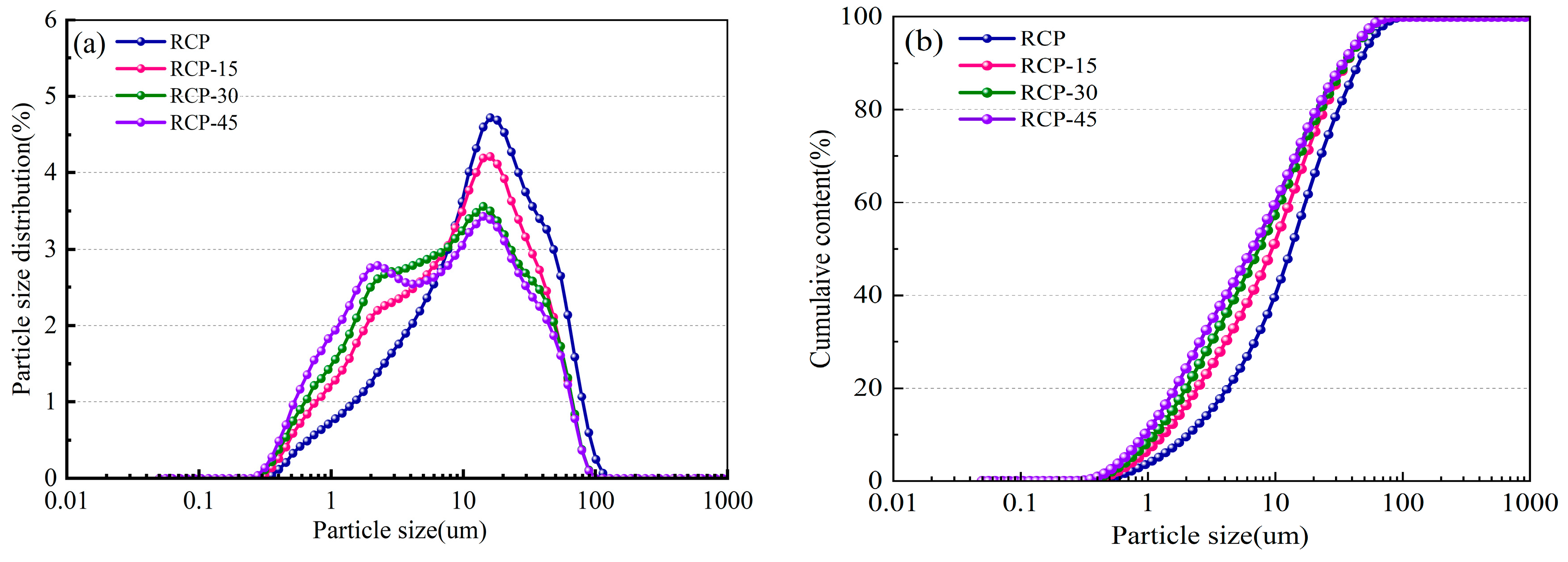
Figure 4 shows the particle size distribution and the particle size cumulative distribution curves for RCP-0, RCP-15, RCP-30, and RCP-45. With increasing grinding time, the overall particle size distribution shifts to smaller particle sizes (i.e., to the left) and the proportion of the particles with peak size gradually decreases. In particular, after grinding for 30 min, the median particle size is 8.37 μm, and the proportion of particles smaller than 10 μm is 57.23%.
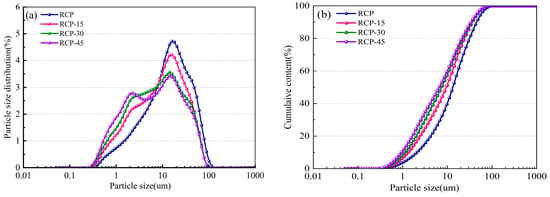
Figure 4.
Particle size distribution and particle size accumulation of RCP ground for different durations: (a) particle size distribution and (b) particle size accumulation.
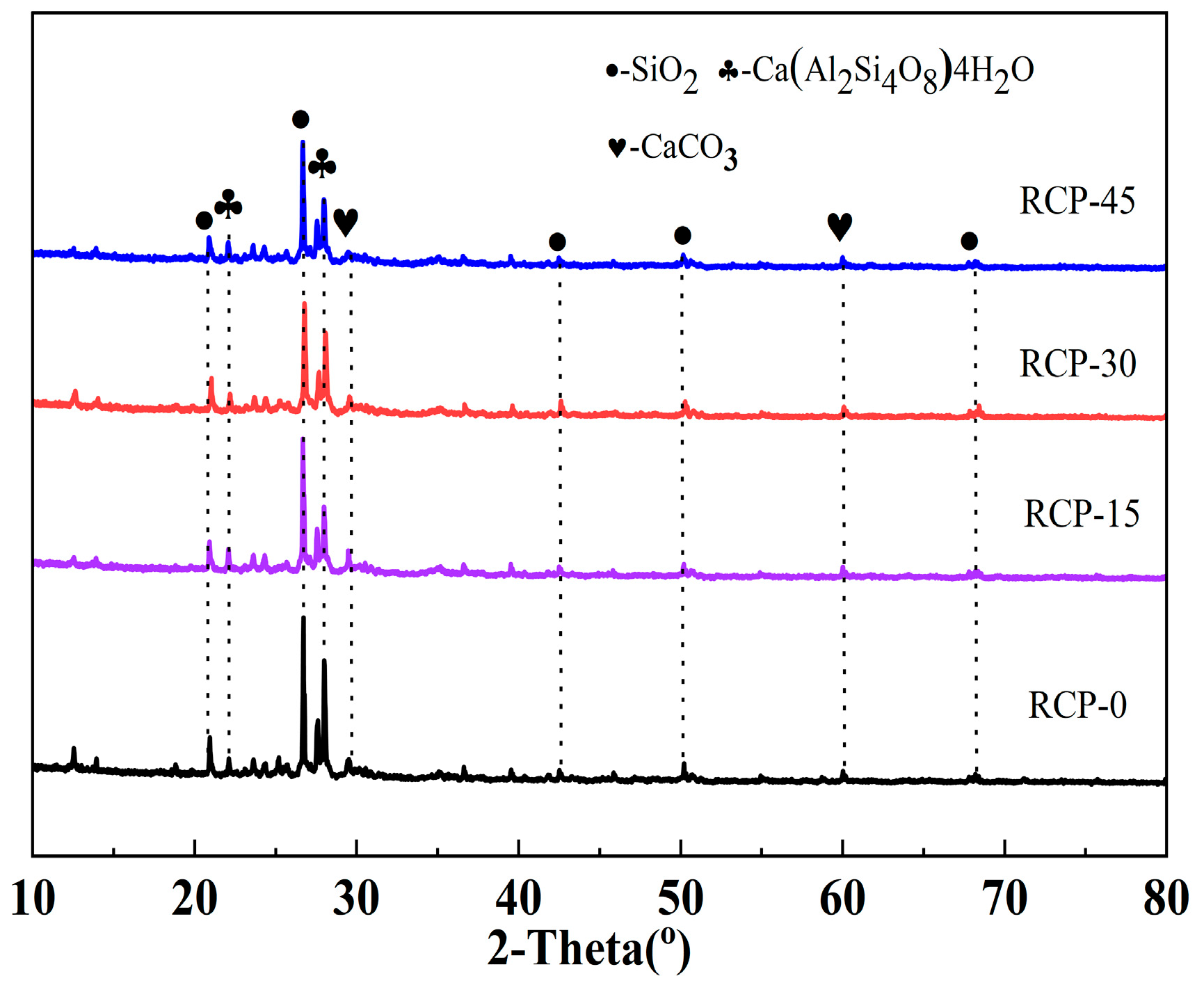
Figure 5 shows the XRD patterns of RCP under four experimental conditions (RCP-0, RCP-15, RCP-30, and RCP-45). After mechanical grinding, some of the RCP diffraction peaks decrease in intensity, whereas some increase in intensity. For example, the crystallization peaks of SiO2 located at approximately 21° and 27° decrease in intensity after grinding. This is attributed to the distortion of the regular tetrahedral structure of SiO2 caused by mechanical grinding, which transforms crystalline SiO2 into amorphous SiO2 [32] and thereby considerably increases the activity of the AACMs. Furthermore, the partial crystallization peak of CaCO3 in RCP-30 has a higher intensity than that of CaCO3 in RCP-0. Such a behavior can be explained by the fact that Ca(OH)2 contained in RCP reacts with CO2 at high temperatures and consequently forms CaCO3, as reported in reference [33].
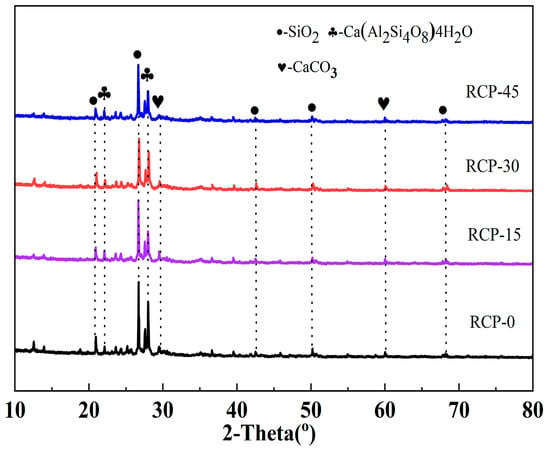
Figure 5.
XRD of RCP ground for different durations.
Figure 6 shows the SEM images for RCP-0 and RCP-30. After grinding, RCP particles become finer, the surface edges and corners become passivated, and the particle shape becomes more regular. The incorporation of such particles into mortar improves the particle gradation in the cementitious material, decreasing the number of internal pores in the mortar and increasing its compactness [34].

Figure 6.
SEM images of (a) RCP-0 and (b) RCP-30.
3.2. Activity Index of RCP After Activation
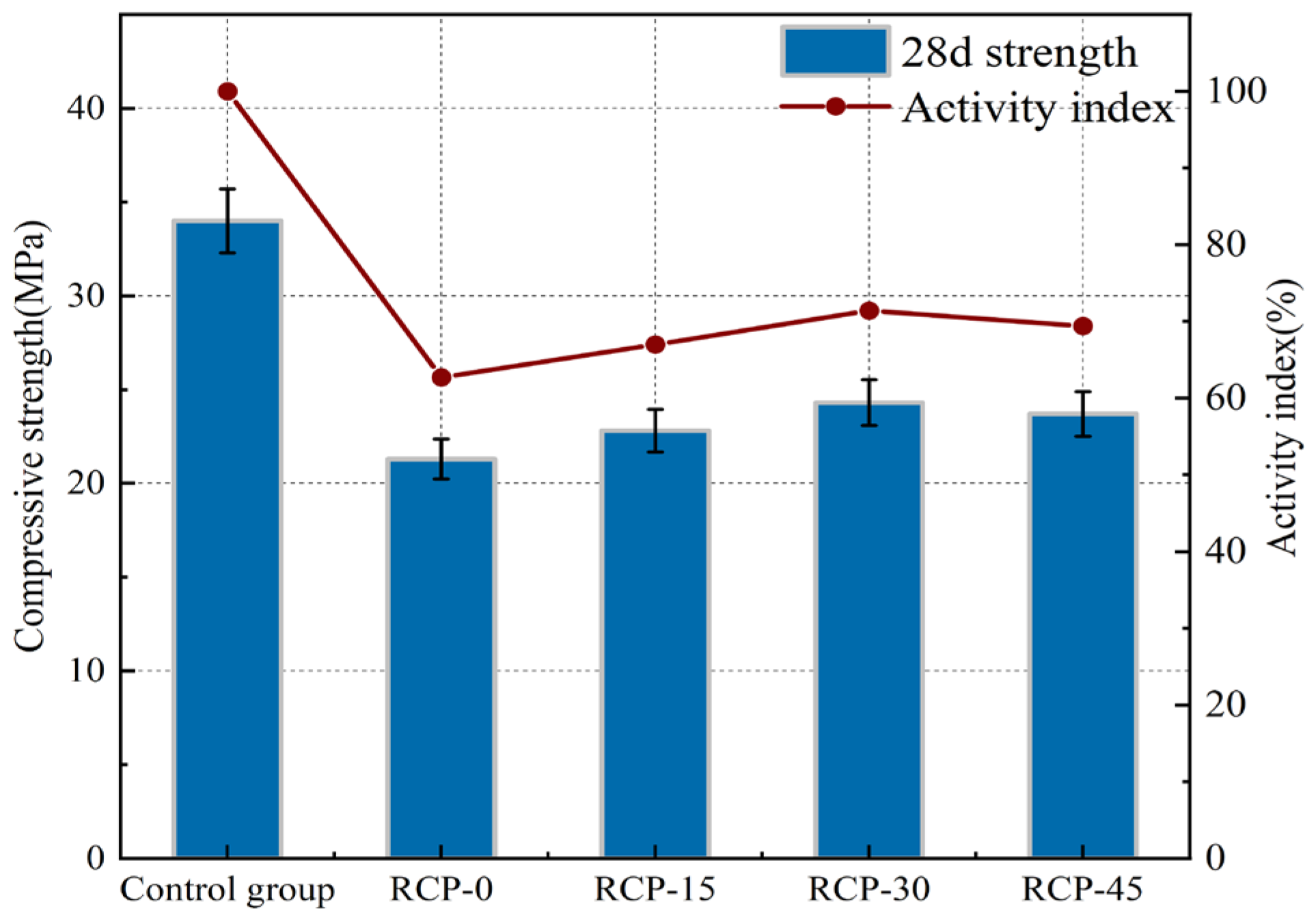
Figure 7 shows the 28-day compressive strength and activity index of RCP surfaces at different grinding times. Specifically, the activity indices for RCP-0, RCP-15, RCP-30, and RCP-45 were 62.7%, 67%, 71.4%, and 69.8%, respectively. This is attributed to physical grinding causing distortion of the SiO2 tetrahedral structure within RCP, transforming it into an amorphous form. The increased amorphous SiO2 enhances the reactivity of RCP. The activity index of RCP-30 was 13.8% higher than that of RCP-0. As the grinding time increased, the specific surface area of RCP expanded, leading to higher water absorption and water demand ratios. This change reduced the availability of free water, thereby inhibiting hydration reactions. Additionally, agglomeration phenomena [35] caused some RCP particles to remain unground, resulting in a slight overall increase in the activity index.
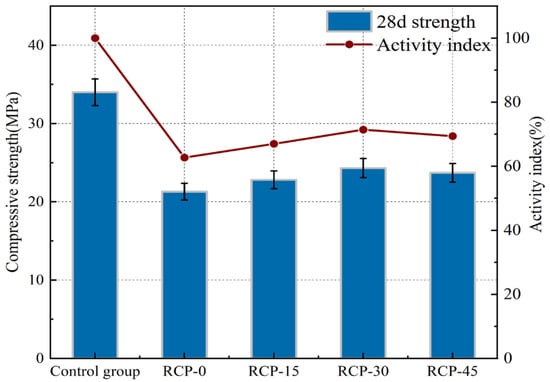
Figure 7.
Activity index of RCP ground for different durations.
3.3. Fluidity of the AACMs
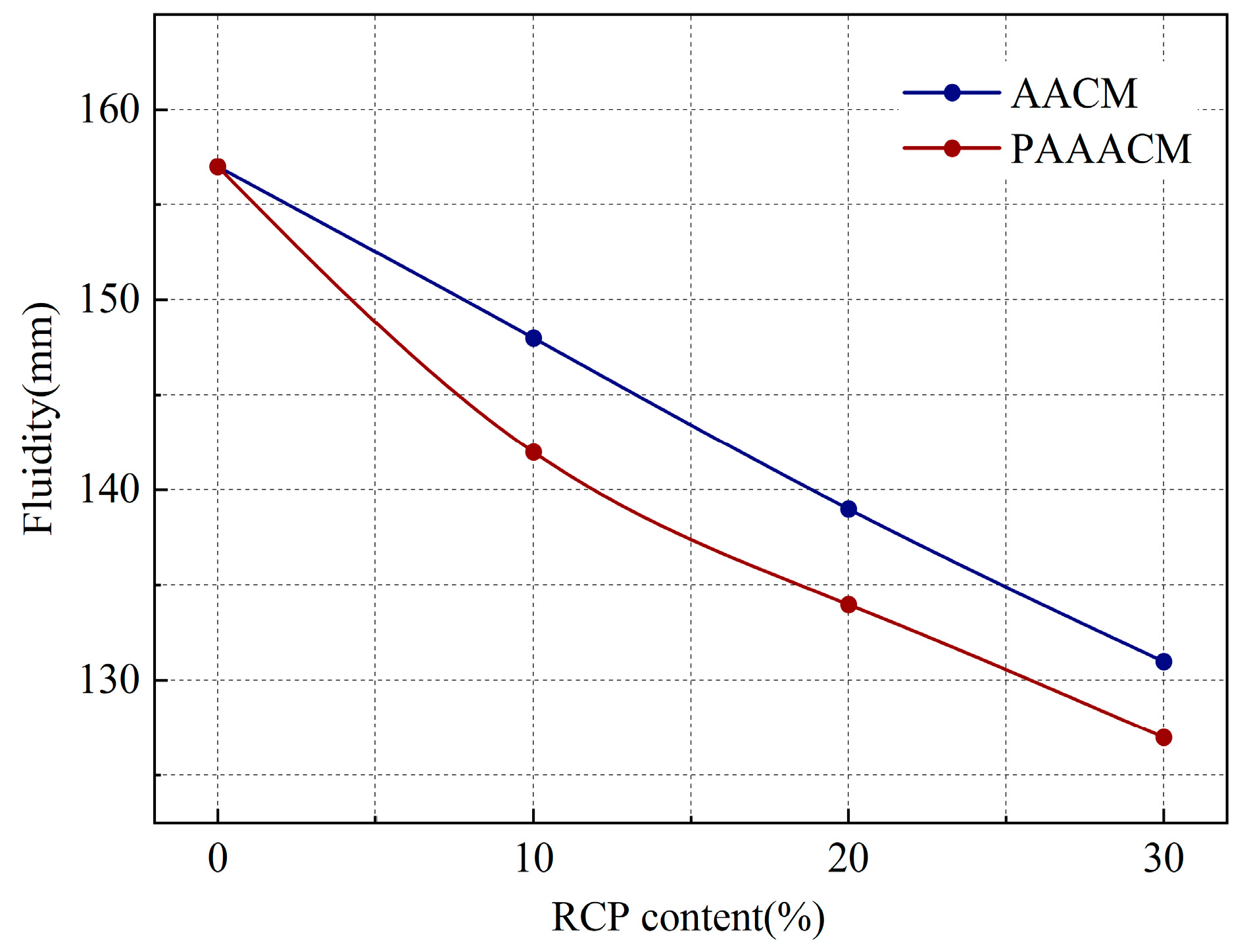
As shown in Figure 8, with increasing RCP content, the fluidity of the mortar decreases, with the overall reduction rate reaching 6% to 17% in the group with the AACMs and 10% to 19% in the group with the PAAACMs. This is attributed to the rough surface of RCP: more edges and corners led to greater water absorption and thereby a decrease in free water. In the case of excessive RCP dosage, a flocculation structure that is difficult to destroy is formed in the alkali-activated GGBS and FA system [13], reducing fluidity. After mechanical grinding, RCP particles are smaller, more regular in shape, and their edges and corners are passivated. These particles act as an ultrafine aggregate in the cementitious system [15], improving the particle gradation. At 20% content, the fluidity of the PAAACM group is 4% lower than that of the RCP group. This is attributed to the ball and microaggregate effects [18,33] of the passivated fine particles, which increase the fluidity of the mortar. However, this effect is weaker than the increase in water absorption caused by the increase in the specific surface area, resulting in a decrease in fluidity.
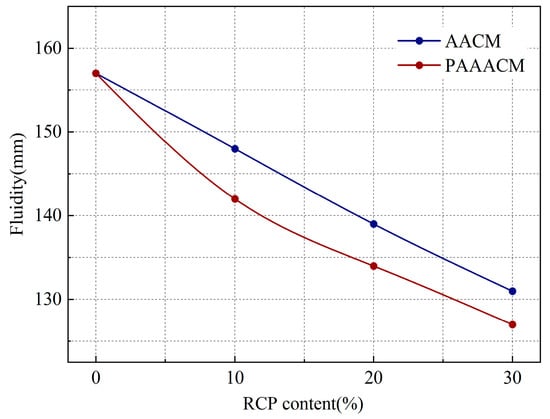
Figure 8.
Fluidity of mortar with different RCP contents.
3.4. The Mechanical Properties of the AACMs
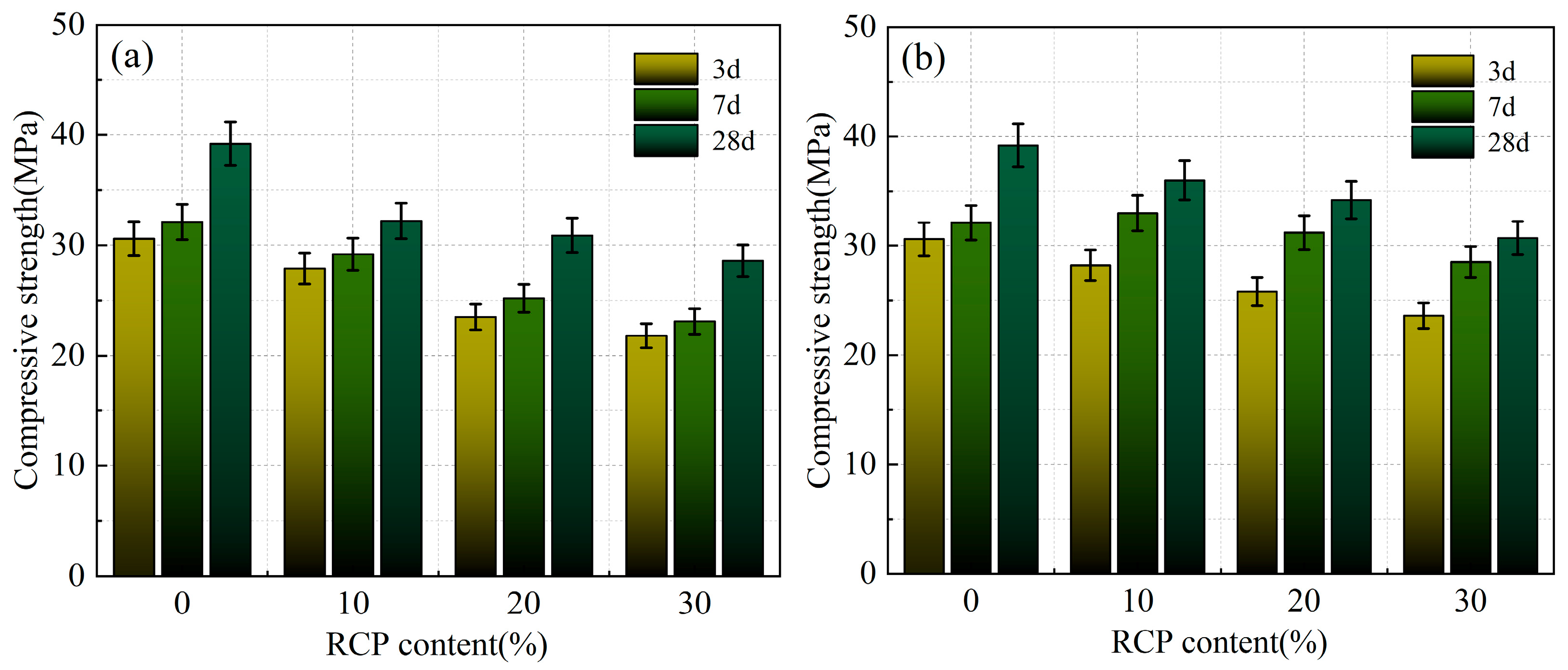
Figure 9a and Figure 10a present the compressive and flexural strengths of the AACMs at various curing ages with varying RCP contents and activity levels. As the RCP content increases, both strengths of the alkali-activated mortar show a continuous decreasing trend. A compressive strength of 39.2 MPa was obtained for the mortar with an RCP-0 content of 0%. As the RCP content increases to 10%, 20%, and 30%, the 28-day compressive strength of the AACM mortar shows reductions of 15%, 22%, and 32% relative to the control group, while the corresponding values for the PAAACM group decrease by 7%, 15%, and 22%. As a result, the inclusion of RCP is unfavorable to the overall performance of alkali-activated cementitious systems. This is attributed to the low activity of RCP itself. Thus, the increase in RCP content hinders the reaction between the powder and the alkali solution. [14,22] Moreover, a greater water demand leads to insufficient available free water and a decreased yield of hydration products, which in turn gives rise to the decline in mortar strength.
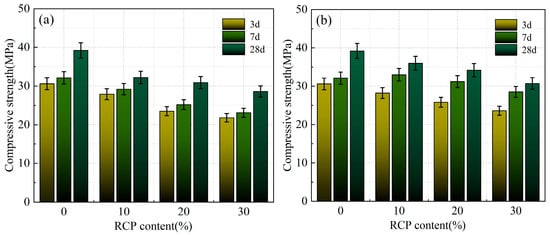
Figure 9.
The compressive strength of the AACMs and PAAACMs under different dosages of RCP: (a) the AACMs and (b) the PAAACMs.
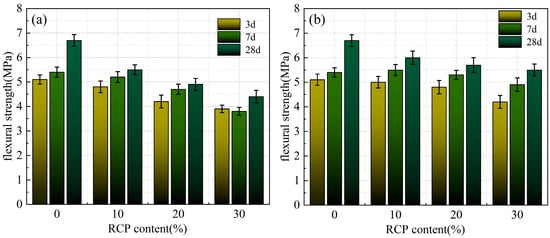
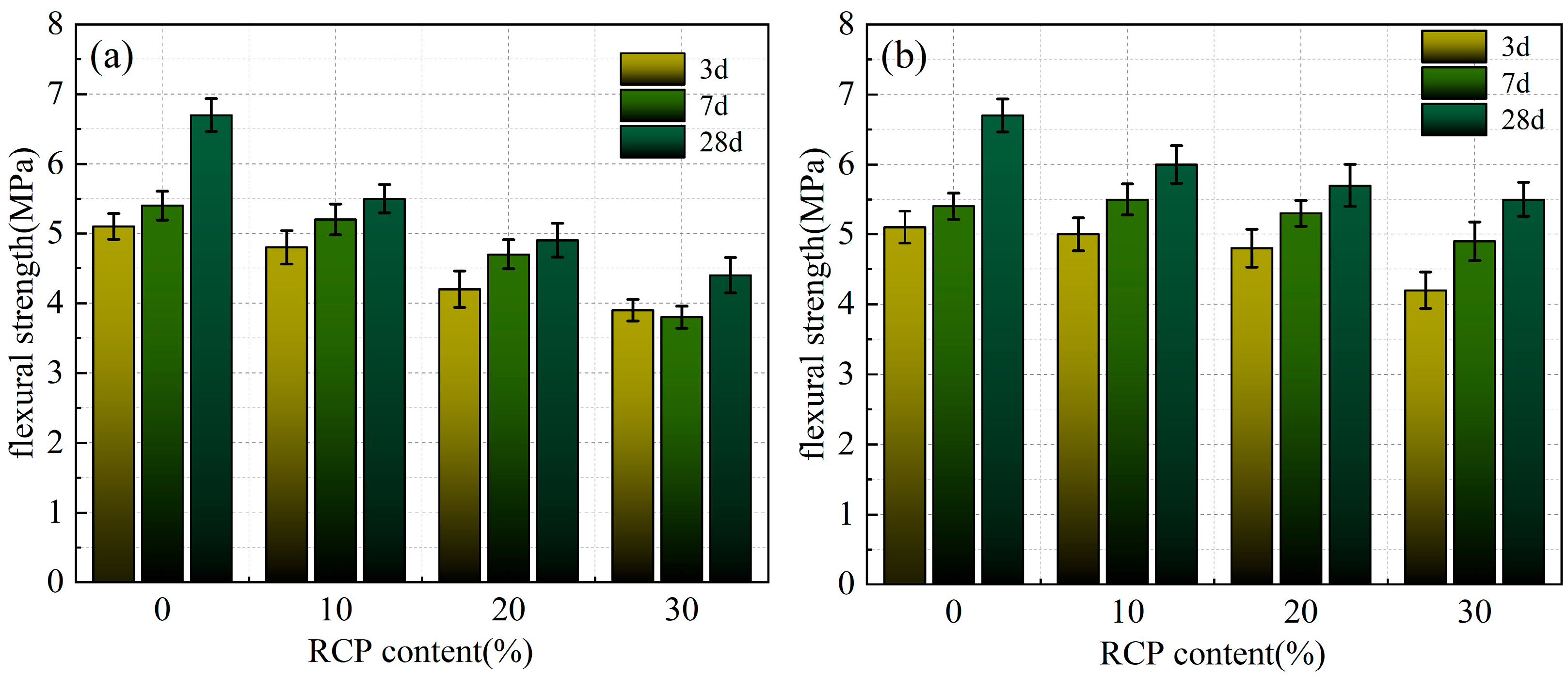
Figure 10.
The flexural strength of the AACMs and PAAACMs under different dosages of RCP: (a) the AACMs and (b) the PAAACMs.
The statistical parameters, including the mean, standard deviation (SD) and coefficient of variation (CV), tabulated in Table 4 and Table 5 further confirm the excellent repeatability and reliability of the experimental data. The CV values for all compressive and flexural strength results range from 3.51% to 6.67%, demonstrating the high precision of the test measurements and low dispersion of the mechanical property data.

Table 4.
CV of compressive and flexural strength of AACMs with various RCP dosages.

Table 5.
CV of compressive and flexural strength of PAAACMs with various RCP dosages.
According to Figure 9b, at RCP contents of 10%, 20%, and 30%, the compressive strength of the PAAACMs is 7%, 9%, and 15% higher than that of the AACMs. Furthermore, the compressive strength of the PAAACMs rapidly increases in the early stage, indicating that the physically activated RCP positively affects the early hydration of the AACM system; however, the excessive content of RCP negatively impacts the compressive strength. This is attributed to the small size and uniformity of RCP particles after physical activation, which is beneficial for the particle gradation in cementitious materials. Concurrently, an elevated content of active silica accelerated the reaction between the powder and alkaline-activator solution, contributing to the formation of the calcium aluminum silicate hydrate (C–A–S–H) and calcium silicate hydrate (C–S–H) gels [17]. However, with increasing RCP content, the impact of the filling effect on the hydration reaction and activation is not sufficient to compensate for the reduction in the amounts of active material and free water. The elevated water demand ratio reduces the generation of hydration products, which in turn gives rise to the degradation of the mortar’s compressive strength.
3.5. Microstructure
3.5.1. XRD of the Hydration Products
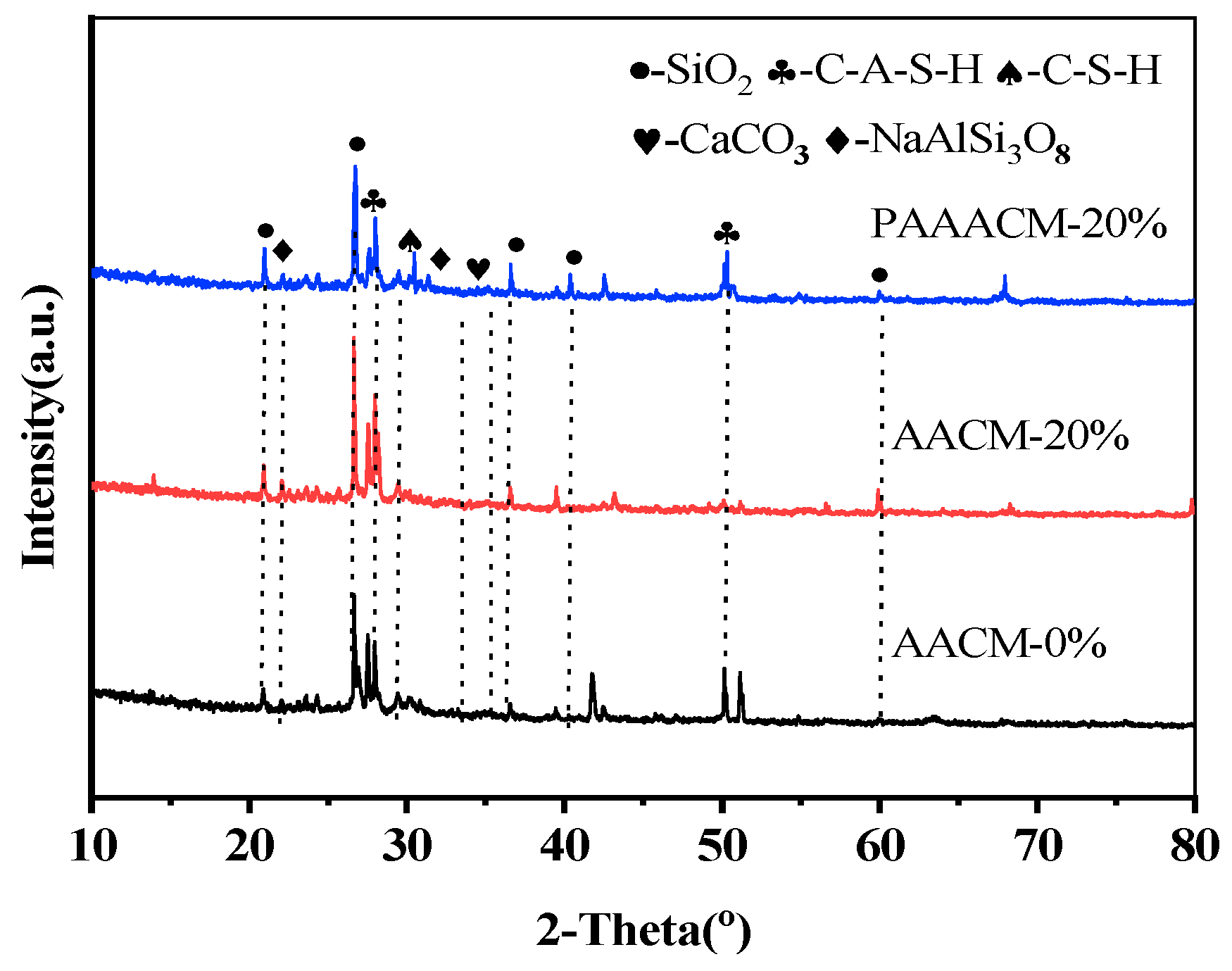
Figure 11 shows the XRD patterns of the alkali-activated mortar with different RCP dosages and different activities. The hydration products in the three groups of samples include SiO2, albite, CaCO3, C–S–H and C–A–S–H gels [36]. The XRD patterns of AACM-0 and AACM-20% show no notable differences in the type and number of main peaks. With increasing RCP content, the SiO2 and C–A–S–H gel contents of AACM-20% also increase. After grinding, the activity of SiO2 in RCP is higher, so the SiO2 diffraction peak in the mortar with activated RCP is weaker and narrower, and the peaks of C–S–H, C–A–S–H, and other gels have higher intensity than those in the mortar with unactivated RCP. This suggests that under alkaline conditions, only a small fraction of reactive particles participate in hydration to form gel phases, while most inert particles remain unreacted [18]. The activated RCP particles are more likely to dissolve in an alkaline environment, undergo the hydration reaction, and generate gels. At an RCP incorporation rate above 20%, the overabundance of RCP particles damages the gel framework, resulting in an obvious deterioration in compressive strength.
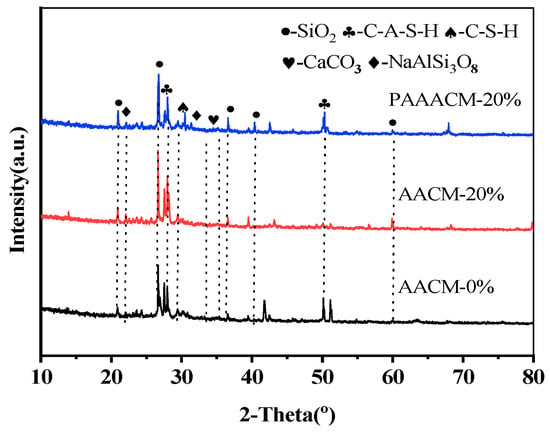
Figure 11.
XRD plots of AACM-0%, AACM-20%, and PAAACM-20%.
3.5.2. SEM Analysis
Figure 12 shows the effect of the content and activity of RCP on the structure of the AACMs. With the incorporation of RCP, the microstructure becomes more uneven, and the number of surface cracks and damaged pores increases. In the group with PAAACM-20%, the amount of hydration products is higher, and the structure of the resulting AACMs is denser than in the group with AACM-20%. No RCP particles are observed on the surface. Based on previous studies [9,18], this is attributed to the encapsulation of RCP by the already formed gel material, forming a dense microstructure; at the same time, RCP particles not involved in the reaction become new nucleation sites, thus promoting the formation of novel reaction products over the surface of RCP grains.

Figure 12.
SEM images of the AACMs at 28 d: (a,b) the AACM-0% group; (c,d) the AACM-20% group; and (e,f) the PAAACM-30% group.
3.6. Hydration Heat Analysis
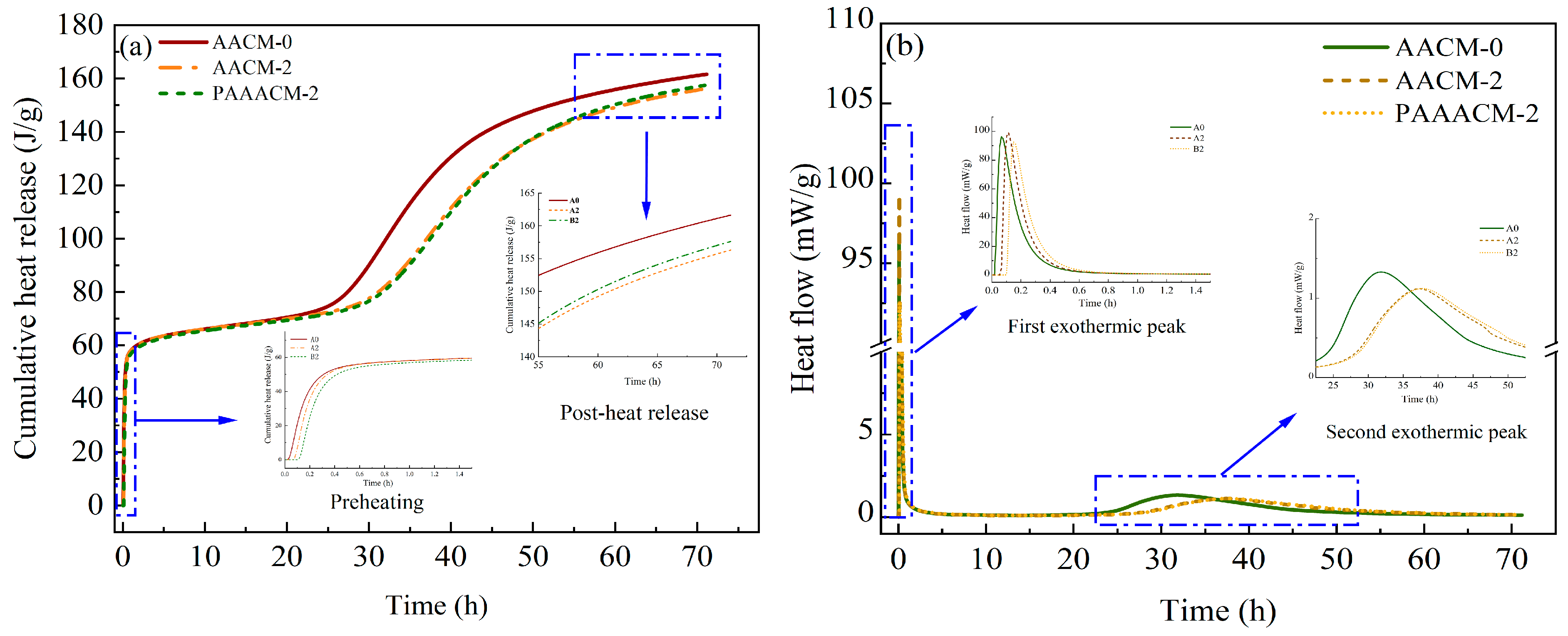
Figure 13a shows that a rise in both the dosage and activity of the RCP results in a decreased heat release rate and lower total heat release for the alkali-activated cementitious system. At an RCP content of 20%, the total heat release is 6–7 J/g lower than that in the system without RCP (group AACM-0%). Compared with the system with unactivated RCP (group AACM-20%), the total heat release in the system with activated RCP is higher by 1–2 J/g. This is attributed to the inactive material in RCP hindering the hydration reaction, resulting in a decrease in the heat release rate; after physical activation, the active particle content in RCP increases, promoting the partial hydration reaction and thereby increasing the total heat release [37]. The reduction in total hydration heat indicates insufficient formation of the hydration products, C–S–H and C–A–S–H gels, leading to a looser microstructure and consequently decreased compressive and flexural strengths of the mortar. This pattern aligns perfectly with the continuous strength decline observed in Figure 9 and Figure 10 as the RCP dosage increases. Conversely, physical activation enhances RCP activity, increasing hydration heat release and hydration product formation. This results in a denser microstructure, leading to significantly higher strength in the PAAACM group compared to the AACM group. Additionally, the rational control of early heat release rate mitigates drying shrinkage, indirectly ensuring the stability of strength development.
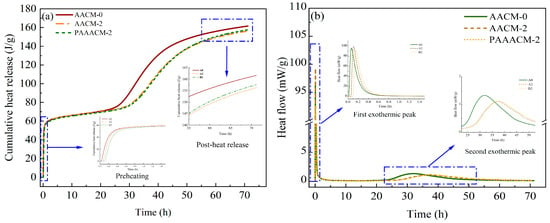
Figure 13.
The hydration test results of the AACMs under different dosages of RCP and different activation times: (a) the total heat release graph and (b) the total heat release rate graph.
The hydration heat release of cement-based materials and AACMs is a complex process with multiple reaction mechanisms and is mainly divided into five stages [38,39,40,41]: the initial period, induction period, acceleration period, deceleration period, and recession period. As shown in Figure 13b, two exothermic peaks are observed for the alkali-activated cementitious system, and their occurrence time varies among systems. The appearance of these two exothermic peaks can be attributed to the production of C–S–H and C–A–S–H gels during the course of hydration [40,41]. In the initial period, alkaline ions (OH−) released through the breaking of covalent bonds in the mineral powder glass by SO42− provide numerous nucleation sites for the formation of early hydration products [42]. According to the heat flow curves for AACM-0 and AACM-20, the incorporation of RCP delays the initial period of the hydration reaction. This is attributed to the presence of large amounts of inert material and calcite in RCP, which suppresses the hydration process in GGBS and FA, as well as the limited reactive materials contained in RCP within the alkaline environment. In the rapid reaction stage, the hydration reaction is intense, and large amounts of C–S–H and C–A–S–H gels are formed [43,44]. The generated hydration products are densified and wrapped around the material particles, reducing the area of the material participating in the hydration reaction, resulting in a decrease in the reaction rate and the transition to the induction period at 0.2–0.3 h. It can be seen from the figure that in the first exothermic peak, the reaction rate of the activated RCP is actually slower than that of the unactivated one, which indicates that it is helpful to a certain extent in delaying the drying shrinkage of concrete. Figure 13b shows that the second exothermic peak of group A0 is between 22.5 and 45 h, while the second exothermic peak for groups A2 and B2 is observed at 27.5–50 h, indicating a delay in the second exothermic peak by 26.5%. At this point, the hydration reaction enters the acceleration period. In this stage, a large amount of hydration products is generated, and the total heat release increases. For group PAAACM-20%, the heat release rate gradually increases after reaching the second heat release peak, exceeding that of group AACM-20%. It is worth noting that the time for it to enter the induction period is delayed by 25%. This is because, during the initial period, the activity of PARCP was high enough to generate sufficient gel-like substances that completely covered its surface, preventing it from entering the next reaction stage. The overall heat release decreases with the incorporation of RCP because of the reduction in the amount of the formed hydration products, resulting in a decrease in the strength of the mortar [11,45]. Simultaneously, the accumulation of inert materials and insufficient free water resulting from excessive RCP further exacerbates microstructural porosity, leading to a greater reduction in strength (e.g., at a 30% RCP dosage, the strength reduction reached 32% in the AACM group and 22% in the PAAACM group).
At the end of the acceleration period, the concentration of basic ions decreases, and the reaction enters the deceleration period. Relevant studies [46,47,48] have shown that the formation of a C–S–H gel with a low calcium–silicon ratio is preferred in an alkaline environment, resulting in a decrease in the alkali concentration. As the alkaline ions in the alkali solution are gradually consumed and the thickness of hydration products on the surface of the active particles increases, the hydration reaction gradually stabilizes and enters the recession period. The heat liberation over 72 h and the formation of hydration products in the PAAACM-20% group are both higher than the corresponding values in the AACM-20% group, which agrees well with the mortar’s compressive strength results.
Hydration heat monitoring results demonstrate that the introduction of RCP exerts an inhibitory effect on the hydration between solid powders and alkaline solutions. This contributes to a more porous microstructure, postponed induction and rapid reaction intervals, and a weakened hydration rate peak in the acceleration period. Furthermore, physical activation increases the total heat release from RCP particles in the alkaline environment, which serves to mitigate the drying shrinkage of AACMs and elevate the total content of hydration products produced.
3.7. Carbon Emission Analysis of AACMs
Based on the previous experimental results, this study selected three typical mix ratios to construct the AACM carbon footprint evaluation system: group I (base group, 0% content), group II (20% unactivated RCP content), and group III (20% physically activated RCP content). According to the calculation standard of GB/T 51366-2019 [49] “Standard for Building Carbon Emission Calculation.” the calculation program and system boundary of the AACM carbon emission are preliminarily determined. Through the product relationship between the carbon emission factor (EFi) of each component material and the unit volume dosage (Mi) of each component, the total carbon dioxide emissions of mortars with different mix ratios were systematically quantified. Liao et al. [50] also pointed out that carbon emissions are determined by multiplying the material consumption (M) by the carbon emission coefficient (Cef). The specific formula is shown in Equation (1):
Mi is expressed as the unit volume consumption of each component material, EFi is expressed as the carbon emission factor of each component material and preparation process, Tj is expressed as the equipment running time, and Wj is expressed as the equipment power.
The carbon emission factor data used in this study are derived from the international authoritative literature database and references [50], including standardized data sources such as Ecoinvent and the China Life Cycle Database (CLCD). Table 6 systematically summarizes the carbon emission factors of each production stage and their corresponding data sources, in which the key parameters are localized to ensure the regional applicability of the calculation results. Using the data in Table 3 and Table 6, we calculated the total carbon emissions of the AACMs.

Table 6.
Carbon emission factors of each material.
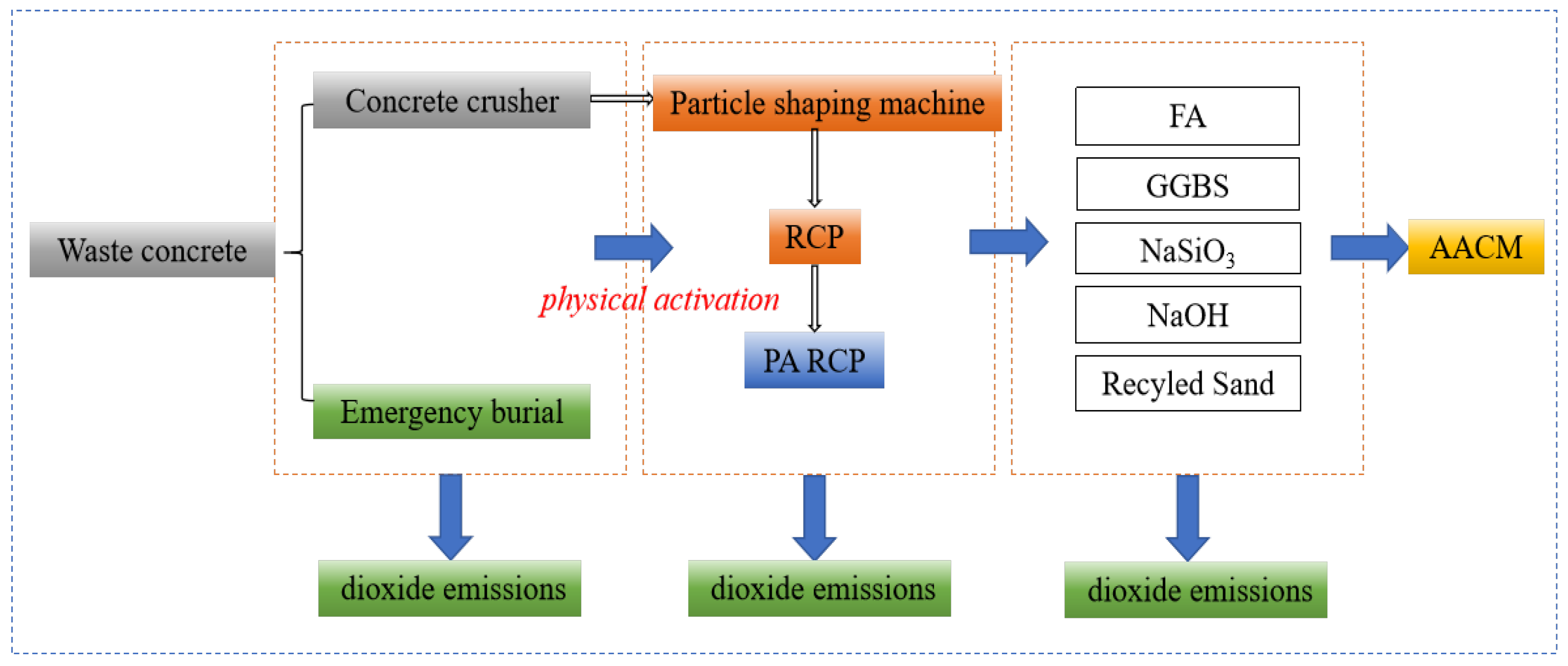
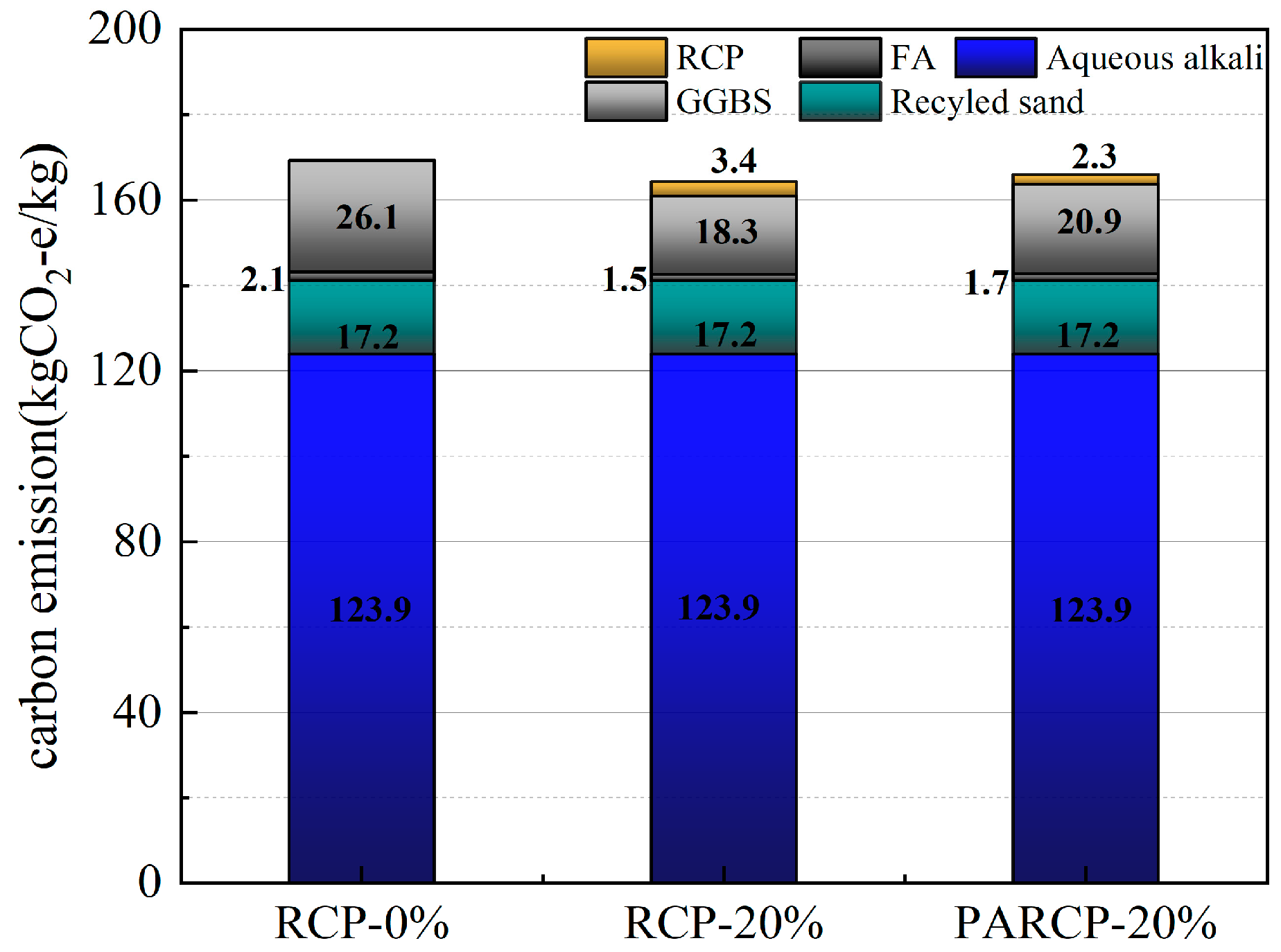
Based on the life cycle assessment (LCA) method, Figure 14 illustrates the carbon footprint distribution of RCP from construction solid waste crushing and sorting, physical activation treatment, to waste disposal and preparation of AACM. According to Figure 15 and the data in Figure 16, when the RCP content is 0%, the carbon emission of the AACMs reaches 169.3 kgCO2-e/ton. When the content reaches 20%, the carbon emission of the AACMs reaches 165.9 kgCO2-e/ton. After RCP simultaneously replaces GGBS and FA, it is 2.1% lower than that of the control group. At the same time, the emission of activated RCP reaches 166.6 kgCO2-e/ton. However, compared with the carbon emission structure dominated by GGBS in the whole cementitious material (83.9%), it only accounts for 9.2%. Although the physical activation process of RCP produces certain carbon emissions, its replacement of high-carbon-footprint materials (such as GGBS) in the alkali-activated cementitious system can reduce certain carbon emission intensity. Combined with the carbon offset effect of building solid waste recycling [51,52], the application of RCP significantly promotes the construction and environmental sustainability of a low-carbon building materials system.
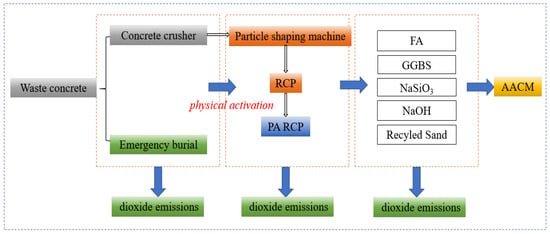
Figure 14.
Preparation and activation of RCP and carbon footprint of AACMs.
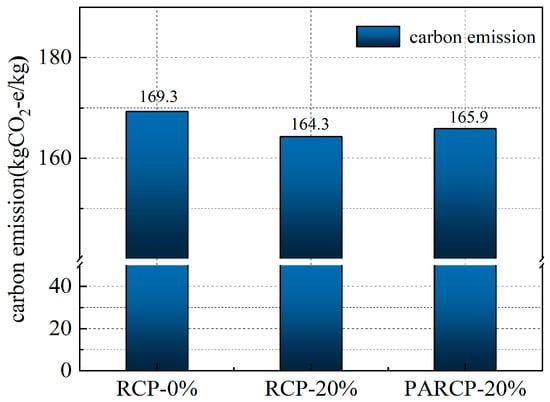
Figure 15.
Total carbon emissions.
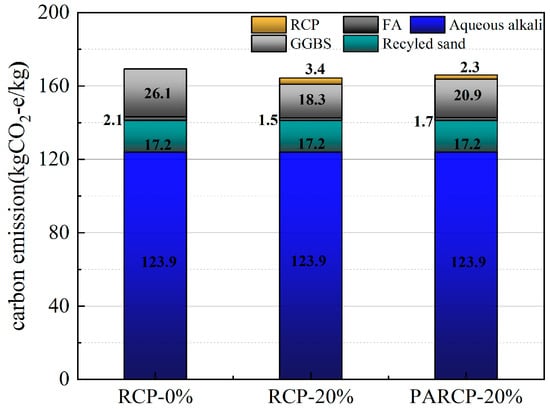
Figure 16.
Carbon emissions of mortar components.
4. Discussion
According to the study in this paper, it can be seen that RCP, after physical activation, has higher activity, which helps to promote the hydration reaction of alkali-activated specimens. Nonetheless, the incorporation of excessive RCP still degrades both the workability and mechanical properties of the specimens, while also causing a delay in the hydration exothermic peak. Given the current research scope, the durability and long-term aging performance of the prepared mortars were not tested in this work. For future research, systematic investigations should be carried out on the drying shrinkage, durability (including impermeability, frost resistance, and carbonation resistance), and long-term aging characteristics of mortar or concrete prepared with physically activated RCP, so as to complete the performance evaluation system of this kind of low-carbon cementitious material.
5. Conclusions
- When 20% physically activated RCP is incorporated as a replacement, the AACM system demonstrates a 2.1% reduction in life cycle carbon emissions, validating the substantial contribution of construction waste recycling to carbon neutrality in the construction materials industry, while maintaining an engineering-applicable compressive strength of 34.2 MPa (9% higher than unactivated systems).
- Physical activation establishes a dual carbon-reduction mechanism at the microscopic scale by optimizing RCP particle size distribution and enhancing amorphous SiO2 content: the 27% improvement in utilization rate of active components reduces raw material consumption, while the strengthened pozzolanic reaction decreases cement demand by 15%.
- Although RCP incorporation delays the first exothermic peak in alkali-activated reactions by 1.2 h, the activated treatment significantly increases total heat release by 17.8% (reaching 214 J/g) through enhanced active site density. This accelerated reaction kinetics improves the 3-day compressive strength by 12%, effectively compensating for early-stage performance losses.
- At a 20% replacement of slag and FA with RCP, the system achieves a 2.1% carbon emission reduction (from 169.3 to 165.9 kg CO2-e/ton). While activation slightly increases emissions to 166.6 kg CO2-e/ton, RCP’s own carbon contribution remains limited to 9% (versus 83% from GGBS). The incremental emission (0.7 kg CO2-e/ton) is fully offset by substitution-induced reductions (4.7 kg CO2-e/ton) and carbon offsets from waste recycling, yielding a net emission reduction of 6.3 kg CO2-e/ton.
Author Contributions
Conceptualization, Y.G., Z.G., Z.Z. and Q.L.; methodology, Y.G., Z.G., Z.Z. and Q.L.; software, J.Y. and C.S.; validation, Y.G., Z.G., Z.Z., L.W. and M.C.; formal analysis, Q.L. and M.C.; investigation, Z.G., J.Y., Q.L. and Z.Z.; resources, Q.L. and Z.Z.; data curation, Y.G., Z.G., Z.Z. and Q.L.; writing—original draft preparation, Z.G. and Z.Z.; writing—review and editing, Y.G., Z.G., Z.Z., M.C., L.W. and Q.L.; visualization, J.Y. and C.S.; supervision, J.Y. and C.S.; project administration, Y.G. and Q.L.; funding acquisition, Y.G. and Q.L. All authors have read and agreed to the published version of the manuscript.
Funding
The National Natural Science Foundation of China [grant numbers 52478262]; the Qingdao Natural Science Foundation Youth Project [grant numbers 23–2–1–105–zyyd–jch]; the Shandong Province City level School Enterprise Cooperation Project [grant numbers 2425282, 2425244, 2424360, 2423272]; and Graduate Innovation Program of Qingdao Agricultural University [grant numbers QNYCX25022].
Data Availability Statement
Every dataset generated or employed throughout the present work can be found in the published paper. Supplementary information will be provided by the corresponding author when reasonably requested.
Conflicts of Interest
Author Changhai Shao was employed by the company Shandong Guanrunjia Environmental Protection New Materials Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Nwankwo, O.C.; Bamigboye, O.G.; Davies, E.I.; Michaels, A.T. High volume Portland cement replacement: A review. Constr. Build. Mater. 2020, 260, 120445. [Google Scholar] [CrossRef]
- Holt, P.S.; Berge, D.N. Life-cycle assessment of using liquid hazardous waste as an alternative energy source during Portland cement manufacturing: A United States case study. J. Clean. Prod. 2018, 195, 1057–1068. [Google Scholar] [CrossRef]
- Qin, L.; Gao, X.; Li, Q. Upcycling carbon dioxide to improve mechanical strength of Portland cement. J. Clean. Prod. 2018, 196, 726–738. [Google Scholar] [CrossRef]
- Nie, S.; Zhou, J.; Yang, F.; Lan, M.; Li, J.; Zhang, Z.; Chen, Z.; Xu, M.; Li, H.; Sanjayan, J.G. Analysis of theoretical carbon dioxide emissions from cement production: Methodology and application. J. Clean. Prod. 2022, 334, 130270. [Google Scholar] [CrossRef]
- Kuoribo, E.; Shokry, H.; Mahmoud, H. Attaining material circularity in recycled construction waste to produce sustainable concrete blocks for residential building applications. J. Build. Eng. 2024, 96, 110503. [Google Scholar] [CrossRef]
- Alsaif, S.A.; Alqarni, S.A. Characteristics of concrete incorporating construction and demolition waste aggregate and post-consumer tires rubber and steel fibers. Constr. Build. Mater. 2024, 435, 136820. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, X.; Kong, L. Evaluation of carbon and economic benefits of producing recycled aggregates from construction and demolition waste. J. Clean. Prod. 2023, 425, 138946. [Google Scholar] [CrossRef]
- Yu, B.; Wang, J.; Li, R.; Wang, Q.; Wu, H.; Xu, X.; Yuan, H.; Zuo, J. Unveiling the NIMBY effect in construction and demolition waste landfilling: Factors, paths, and solutions. J. Environ. Manag. 2024, 366, 121925. [Google Scholar] [CrossRef]
- Luo, H.; Aguiar, J.; Wan, X.; Wang, Y.; Cunha, S.; Jia, Z. Application of Aggregates from Construction and Demolition Wastes in Concrete: Review. Sustainability 2024, 16, 4277. [Google Scholar] [CrossRef]
- Salesa, Á.; Pérez-Benedicto, J.A.; Colorado-Aranguren, D.; Lopez-Julian, P.L.; Esteban, L.M.; Sanz-Balduz, L.J.; Saez-Hostaled, J.L.; Ramis, J.; Olivares, D. Physico-mechanical properties of multi-recycled concrete from the precast concrete industry. J. Clean. Prod. 2017, 141, 248–255. [Google Scholar] [CrossRef]
- Nacer-Eddine, M.H.; François, B.; Walid, D.; Harifidy, R.; Nordine, L.; Karima, A. Hydration, mechanical and transfer properties of blended cement pastes and mortars prepared with recycled powder or limestone filler. J. Build. Eng. 2023, 78, 107541. [Google Scholar] [CrossRef]
- Xiao, J.; Hao, L.; Cao, W.; Ye, T. Influence of recycled powder derived from waste concrete on mechanical and thermal properties of foam concrete. J. Build. Eng. 2022, 61, 105203. [Google Scholar] [CrossRef]
- Wan, X.; Li, H.; Che, X.; Xu, P.; Li, C.; Yu, Q. A Study on the Application of RCP in an Alkali-Activated Cementitious System. Processes 2023, 11, 203. [Google Scholar] [CrossRef]
- Feng, Y.; Li, J.; Zhang, B.; Fu, H.; Chen, W.; Xue, Z.; Lu, Z.; Yang, J.; Xie, J. Concrete improvement incorporating recycled powder and aggregates treated via a combination of calcination and carbonation: The impact behaviors. J. Clean. Prod. 2023, 418, 138069. [Google Scholar] [CrossRef]
- Ma, Z.; Hu, R.; Yao, P.; Wang, C. Utilizing heat-mechanical synergistic treatment for separating concrete waste into high-quality recycled aggregate, active recycled powder and new concrete. J. Build. Eng. 2023, 68, 106161. [Google Scholar] [CrossRef]
- Deng, Y.; Wu, L. Effect of recycled concrete fine powder after calcination on the properties of autoclaved aerated concrete. Case Stud. Constr. Mater. 2024, 20, e02961. [Google Scholar] [CrossRef]
- Tang, Y.; Xiao, J.; Wang, D.; Zhang, M. Effect of carbonation treatment on fracture behavior of low-carbon mortar with recycled sand and recycled powder. Cem. Concr. Compos. 2023, 142, 105178. [Google Scholar] [CrossRef]
- Wu, H.; Xu, J.; Yang, D.; Ma, Z. Utilizing thermal activation treatment to improve the properties of waste cementitious powder and its newmade cementitious materials. J. Clean. Prod. 2021, 322, 129074. [Google Scholar] [CrossRef]
- Liu, J.; Tian, Y.; Ge, X.; Liu, B.; Liu, K.; Song, G. Experimental Study on MgO-Na2CO3 Combined Activated Recycled Fine-Powder-Slag Cementitious System and Modification. Buildings 2024, 14, 592. [Google Scholar] [CrossRef]
- Chen, L.; Wei, M.; Lei, N.; Li, H. Effect of chemical–thermal activation on the properties of recycled fine powder cementitious materials. Case Stud. Constr. Mater. 2024, 20, e02956. [Google Scholar] [CrossRef]
- Yang, Y.; Kang, Z.; Zhan, B.; Gao, P.; Yu, Q.; Wang, J.; Zhao, W.; Zhang, Y.; Bi, W.; Yang, C.; et al. Short Review on the Application of Recycled Powder in Cement-Based Materials: Preparation, Performance and Activity Activated. Buildings 2022, 12, 1568. [Google Scholar] [CrossRef]
- Gao, S.; Zhang, H. Multi-scale study on strength development, hydration behavior and phase evolution of recycled powder multi-component cementitious materials. Constr. Build. Mater. 2024, 431, 136451. [Google Scholar] [CrossRef]
- Wang, C.; Zhan, B.; Yu, Q.; Gao, P.; Guo, B.; Zhu, L.; Qin, H.; Xu, Y. Investigation on combined mechanical-chemical activation and its synergistic benefits for recycled concrete powder. J. Build. Eng. 2026, 117, 114870. [Google Scholar] [CrossRef]
- Li, X.; Ma, W.; Li, S.; Hou, S.; Chen, Y.; Wen, X.; Dan, J.; Huang, J.; Lv, Y. Study on the properties and carbon footprint of low heat cement clinker prepared by RCP and calcium carbide slag. Constr. Build. Mater. 2024, 456, 139337. [Google Scholar] [CrossRef]
- Zhang, B.; Feng, Y.; Xie, J.; Chen, W.; Xue, Z.; Zhao, G.; Li, Y.; Li, J.; Yang, J. Compressive behaviour and microstructures of concrete incorporating pretreated recycled powder/aggregates: The coupling effects of calcination and carbonization. J. Build. Eng. 2023, 68, 106158. [Google Scholar] [CrossRef]
- Li, J.H.; Wenting, M. Evaluating Carbon Emissions of Construction and Demolition Waste in Building Energy Retrofit Projects. Energy 2023, 281, 128201. [Google Scholar] [CrossRef]
- JG/T 573-2020; Recycled Fine Powder Used in Concrete and Mortar. China Quality Standard Publishing Media Co., Ltd.: Beijing, China, 2020.
- Kaptan, K.; Cunha, S.; Aguiar, J. A review of the utilization of recycled powder from concrete waste as a cement partial replacement in cement-based materials: Fundamental properties and activation methods. Appl. Sci. 2024, 14, 9775. [Google Scholar] [CrossRef]
- GB/T 2419-2005; Test Method for Fluidity of Cement Mortar. China Standards Press: Beijing, China, 2005.
- JGJ/T 70-2009; Standard for Test Method of Basic Properties of Construction Mortar. China Architecture & Building Press: Beijing, China, 2009.
- GB/T 12959-2008; Test Methods for Heat of Hydration of Cement. Standards Press of China: Beijing, China, 2008.
- Yuan, C.; Wang, D.; Setiawan, H.; Wei, Y. Effect and mechanism of different activated modes on the activities of the recycled brick micropowder. Sci. Eng. Compos. Mater. 2021, 28, 676–688. [Google Scholar] [CrossRef]
- Li, S.; Gao, J.; Li, Q.; Zhao, X. Investigation of using recycled powder from the preparation of recycled aggregate as a supplementary cementitious material. Constr. Build. Mater. 2021, 267, 120976. [Google Scholar] [CrossRef]
- Wu, H.; Gao, J.; Liu, C.; Guo, Z.; Luo, X. Reusing waste clay brick powder for low-carbon cement concrete and alkali-activated concrete: A critical review. J. Clean. Prod. 2024, 449, 141755. [Google Scholar] [CrossRef]
- Zhang, B.; Feng, Y.; Xie, J.; Dai, J.; Chen, W.; Xue, Z.; Li, L.; Li, Y.; Li, J. Effects of pretreated recycled powder substitution on mechanical properties and microstructures of alkali-activated cement. Constr. Build. Mater. 2023, 406, 133360. [Google Scholar] [CrossRef]
- Li, J.; Deng, X.; Lu, Z.; Li, X.; Hou, L.; Jiang, J.; Yang, F.; Zhang, J.; He, K. Recycled concrete fines as a supplementary cementitious material: Mechanical performances, hydration, and microstructures in cementitious systems. Case Stud. Constr. Mater. 2024, 21, e03575. [Google Scholar] [CrossRef]
- Lou, Y.; Huang, M.; Kang, S.; Hu, M.; Wu, W.; Chen, S. Study on basic performance and drying shrinkage of binary solid waste geopolymer prepared with recycled powders and slag. Case Stud. Constr. Mater. 2024, 20, e03195. [Google Scholar] [CrossRef]
- Wang, W.; Wang, B. Study on the influence of nano-silica substitution of reactive Si on the properties and hydration process of alkali-activated cementitious materials paste. Constr. Build. Mater. 2024, 443, 137759. [Google Scholar] [CrossRef]
- Chen, X.; Li, Y.; Zhu, Z.; Ma, L. Evaluation of waste concrete recycled powder (WCRP) on the preparation of low-exothermic cement. J. Build. Eng. 2022, 53, 104511. [Google Scholar] [CrossRef]
- Yu, S.; He, J.; Sang, G.; Yang, S.; Liu, G. Study on hydration process of alkali-activated slag cement activated by weakly alkaline components. Constr. Build. Mater. 2024, 413, 134716. [Google Scholar] [CrossRef]
- Liu, S.; Wang, Y.; Wu, A.; Shi, D.; Yang, S.; Ruan, Z.; Song, X.; Zhang, M. Early mechanical strength, hydration mechanism and leaching behavior of alkali-activated slag/FA paste filling materials. J. Build. Eng. 2024, 84, 108481. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, W.; Zhang, Y.; Gu, X. Research on hydration characteristics of OSR-GGBFS-FA alkali-activated materials. Constr. Build. Mater. 2024, 411, 134321. [Google Scholar] [CrossRef]
- Guo, L.; Liu, J.; Chen, D.; An, S. Mechanical properties and microstructure evolution of alkali-activated GGBS-FA-steel slag ternary cements. Constr. Build. Mater. 2024, 444, 137727. [Google Scholar] [CrossRef]
- Xu, R.; Kong, F.; Yang, R.; Wang, H.; Hong, T. Influences of silicate modulus and alkali content on macroscopic properties and microstructure of alkali-activated blast furnace slag-copper slag. Constr. Build. Mater. 2024, 442, 137622. [Google Scholar] [CrossRef]
- Deng, M.; Xie, X.; Zhuo, J.; He, Y.; Wang, K. Experimental Study on the Strength and Hydration Products of Cement Mortar with Hybrid Recycled Powders Based on Industrial-Construction Residue Cement Stabilization of Crushed Aggregate. Materials 2023, 16, 4233. [Google Scholar] [CrossRef] [PubMed]
- Ba, H.; Li, J.; Ni, W.; Li, Y.; Ju, Y.; Zhao, B.; Wen, G.; Hitch, M. Effect of calcium to silicon ratio on the microstructure of hydrated calcium silicate gels prepared under medium alkalinity. Constr. Build. Mater. 2023, 379, 131240. [Google Scholar] [CrossRef]
- L’Hôpital, E.; Lothenbach, B.; Kulik, D.; Scrivener, K. Influence of calcium to silica ratio on aluminium uptake in calcium silicate hydrate. Cem. Concr. Res. 2016, 85, 111–121. [Google Scholar] [CrossRef]
- Yu, J.; Wang, K.; Yang, P.; Li, M.; Dong, B.; Jin, Z.; Hong, S.; Ma, H. Simulation of calcium carbonate nucleation processes in confined C-S-H nanopores with different calcium-silicon ratios. Constr. Build. Mater. 2024, 438, 137157. [Google Scholar] [CrossRef]
- GB/T 51366-2019; Standard for Building Carbon Emission Calculation. China Architecture & Building Press: Beijing, China, 2019.
- Liao, G.; Wang, D.; Wang, W.; He, Y. Microstructure, strength development mechanism, and CO2 emission analyses of alkali-activated FA-slag mortars. J. Clean. Prod. 2024, 442, 141116. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, C.; He, B.; Yi, S.; Tang, L. Recycling fine powder collected from construction and demolition wastes as partial alternatives to cement: A comprehensive analysis on effects, mechanism, cost and CO2 emission. J. Build. Eng. 2023, 71, 106507. [Google Scholar] [CrossRef]
- Yi, Y.; Liu, J.; Lavagnolo, C.M.; Manzardo, A. Evaluating the carbon emission reduction in construction and demolition waste management in China. Energy Build. 2024, 324, 114932. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.