Abstract
To address the problem of excessive sulfur in high-sulfur magnetite concentrates when used directly, this study systematically investigated the desulfurization behavior and mechanism during oxidative roasting. Green pellets were prepared by mixing high-sulfur iron concentrate fines with 1% bentonite, followed by roasting experiments in air at 800–1200 °C. Thermogravimetric analysis (TG), real-time flue gas analysis (DOAS), X-ray diffraction (XRD), and scanning electron microscopy–energy dispersive spectroscopy (SEM–EDS) were employed to characterize the process and products. The results show that sulfur release is mainly concentrated in two stages: intensive oxidative decomposition of FeS/FeS2 in the range of 480–580 °C and release of reacted sulfur originally encapsulated within the pellets in the range of 940–1080 °C. It was found that alkali metal oxides CaO and MgO in the feed can fix sulfur at a high temperature. They react with released SO2 and iron oxides to form Ca/Mg sulfate–iron oxide composite phases, such as (Ca0.75Mg0.25)SO4·0.38Fe2O3 and (Ca0.91Mg0.09)SO4·3.66Fe2O3·1.47MgO, which slow the SO2 emission rate. A desulfurization ratio above 99% can be achieved when roasting at 1100 °C and above. This study clarifies the sulfur migration mechanism during the roasting of high-sulfur iron concentrate pellets, providing a theoretical basis for optimizing the roasting process to achieve efficient desulfurization and recovery of iron resources.
1. Introduction
Clean and efficient utilization of iron ore resources is the cornerstone of sustainable development of the iron and steel industry [1]. With the increasing depletion of high-grade iron ore resources, the exploitation of complex and difficult-to-process resources such as high-sulfur iron ores has been growing [2]. The sulfur content in such ores is typically far above ironmaking requirements. Sulfur is one of the most detrimental impurity elements in steels: it not only deteriorates the mechanical properties and causes “hot shortness”, but the SO2 generated by sulfur oxidation during roasting is also a major atmospheric pollutant [3,4]. Therefore, achieving clean and efficient utilization of high-sulfur iron ores and controlling sulfur migration from the source is of great theoretical and practical significance.
At present, desulfurization of high-sulfur iron ores mainly relies on two technical routes: pre-desulfurization by beneficiation and pyrometallurgical desulfurization by roasting. Flotation is a widely used beneficiation method for sulfur removal, by optimizing reagent schemes and process parameters, separation between sulfides and iron oxide minerals can be realized [5,6,7]. However, for ores with complex dissemination, especially those in which pyrrhotite has magnetic properties close to magnetite, the sulfur content in the iron concentrate can still remain high after beneficiation, and subsequent pyrometallurgical processing is required for deep desulfurization.
Because the oxidative roasting of pellets provides longer material residence time and controllable operating conditions, it is an ideal process for treating high-sulfur iron ores and achieving synergy between consolidation and desulfurization [8,9]. The principle of oxidative desulfurization is to convert metal sulfides into oxides and SO2 gas under high-temperature oxidizing atmospheres. Considerable studies have been carried out on the oxidation kinetics and thermodynamics of single sulfide minerals such as pyrite and pyrrhotite [10,11,12,13,14]. However, high-sulfur iron concentrates constitute a complex mineral system. Besides major iron minerals and a small amount of sulfides, they often contain gangue components such as CaO, MgO, and SiO2. During roasting, these gangue components are not inert; at high temperatures they interact with the desulfurization product SO2, thereby altering sulfur migration pathways and final occurrence states [15,16]. For example, alkali metal oxides (CaO and MgO) have strong chemical adsorption and fixation capability for SO2 and can form stable sulfates [17]. Ma et al. systematically showed that during pellet oxidation, FeS2 transforms through sulfate intermediates and the released SO2 is readily captured by alkaline components to form stable sulfates (CaSO4 and MgSO4) [18]. Existing studies mostly focus on overall “desulfurization degree” and sulfur release kinetics [19,20]. Systematic understanding is still insufficient regarding how sulfur competes and distributes between release and fixation by alkaline gangue components in a complex iron ore system, particularly during pellet roasting as well as the composition, structure, and stability of the resulting solid sulfur-fixation products.
Accordingly, this study uses high-sulfur iron concentrate pellets as the research object. Through oxidative roasting experiments, we aim to clarify the stage-wise sulfur release behavior during oxidative roasting of high-sulfur iron concentrate pellets and to reveal the sulfur-fixation mechanism of gangue components, especially CaO and MgO within specific temperature ranges and the microstructural characteristics of the composite products formed, thereby identifying optimized conditions for deep desulfurization. This work provides theoretical guidance and data support for precise control and efficient desulfurization in pellet roasting of high-sulfur iron ores.
2. Materials and Methods
2.1. Raw Materials
The chemical composition of the high-sulfur iron concentrate fines is shown in Table 1. After magnetic separation, the T.Fe and S contents of the concentrate are 63% and 3.46%, respectively. The sulfur content is far above the requirement for iron concentrates, and the material belongs to a high-sulfur magnetite concentrate. Iron mainly exists as iron oxides (Fe3O4 and Fe2O3), and sulfur exists as iron sulfides, namely pyrite (FeS2) and pyrrhotite (FeS). The concentrate also contains gangue and other impurities, non-target mineral phases associated with the valuable minerals. The main gangue components are MgO and SiO2, with additional CaO and Al2O3.

Table 1.
Chemical composition of high-sulfur iron concentrate fines (%).
2.2. Experimental Procedure
The experimental work mainly consisted of preparation of high-sulfur iron concentrate pellets and roasting tests.
Pellet preparation: After drying, the high-sulfur iron concentrate fines were mixed with 1% sodium bentonite. Then, 6% water was added and mixed again, followed by aging for 45 min. Green pellets were prepared using a Φ500 mm disk pelletizer. The disk inclination angle was fixed at 45°, the rotation speed was controlled at 30 r/min, and the pelletizing time was controlled at 16 min. The obtained green pellets were screened using a round-hole sieve, and pellets with a diameter of 10–13 mm were selected for subsequent roasting tests.
Roasting tests: The roasting setup mainly included a thermogravimetric furnace (HeFei Kejing Materials Technology Co., Ltd., Hefei, China) and a TY-6400 ultraviolet (DOAS) gas analyzer (Wuhan Tianyu Intelligent Control Technology Co., Ltd., Wuhan, China); the technical specifications of the TY-6400 UV (DOAS) gas analyzer are listed in Table 2. The green pellets were dried at 105 °C for 2 h. To avoid SO2 emissions exceeding the analyzer measurement range, approximately 5 g of pellets with a diameter of 10–13 mm were weighed, loaded into a corundum crucible, and placed in the furnace chamber. Before each test, the gas supply device and the gas analyzer were turned on, and air was continuously supplied at 500 mL/min. After 20 min, the furnace was heated from room temperature to the set temperature at a heating rate of 10 °C/min and held for 20 min, followed by cooling to room temperature at 10 °C/min. The SO2 generated during heating and roasting was analyzed in real time by the gas analyzer.

Table 2.
Technical specifications of the TY-6400 UV (DOAS) gas analyzer.
3. Results and Discussion
3.1. Mineralogical Phases of the Raw Material
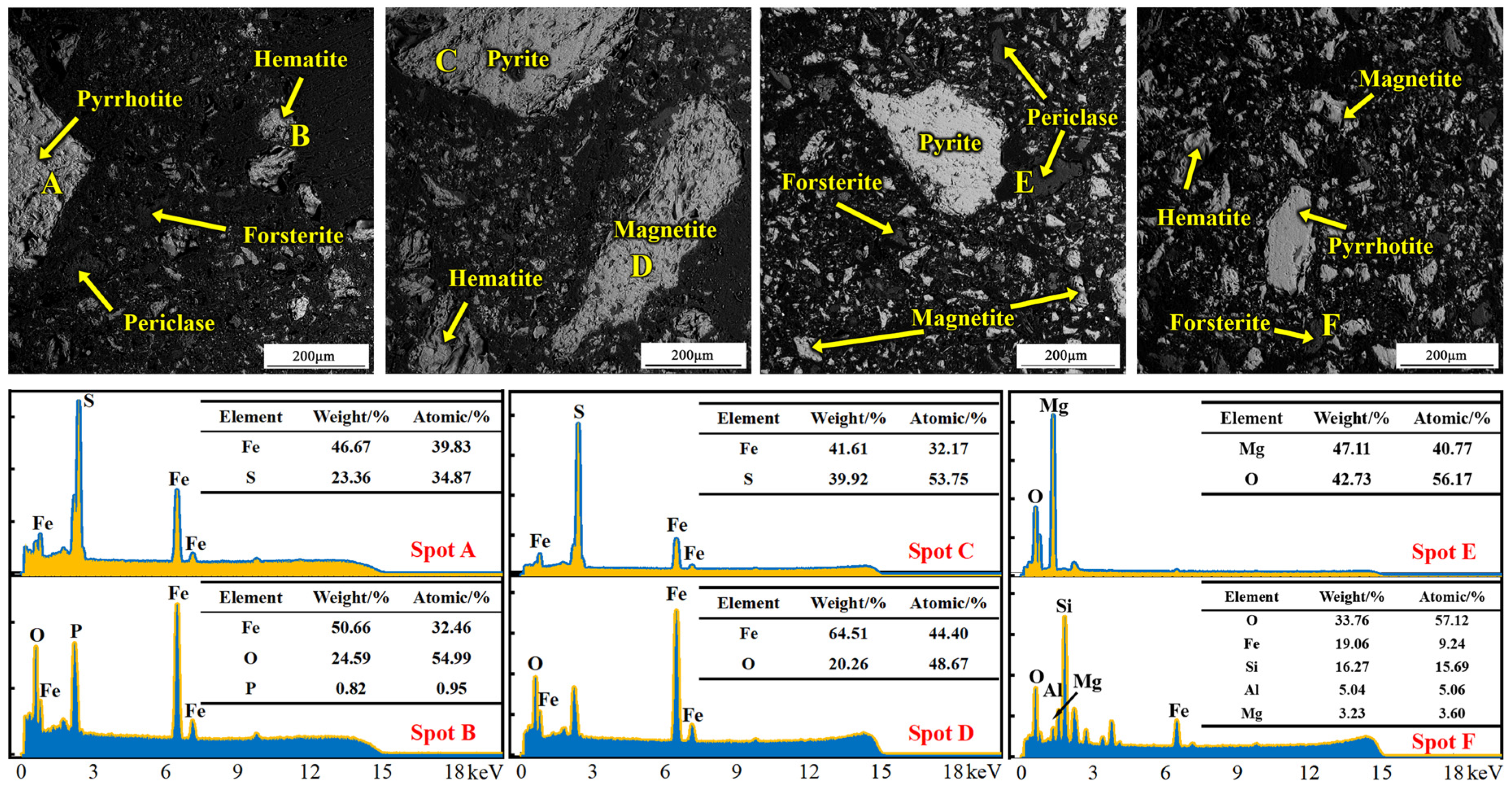
SEM analysis results of the magnetically separated high-sulfur iron concentrate fines are shown in Figure 1. The phases at points A–F correspond to pyrrhotite, hematite, pyrite, magnetite, periclase, and forsterite, respectively, together with the corresponding point-scan EDS spectra. The SEM micrographs indicate that most phases in the concentrate exist as liberated particles, while a small portion occurs as encapsulated inclusions.
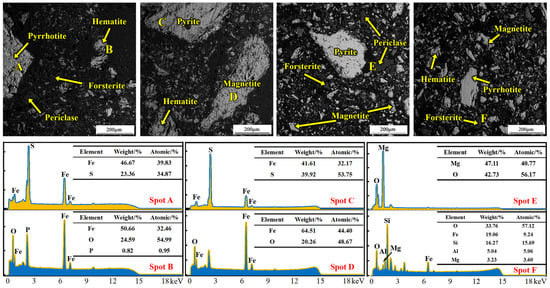
Figure 1.
SEM–EDS microstructural analysis of the high-sulfur iron concentrate.
3.2. Sulfur Migration Behavior During Roasting
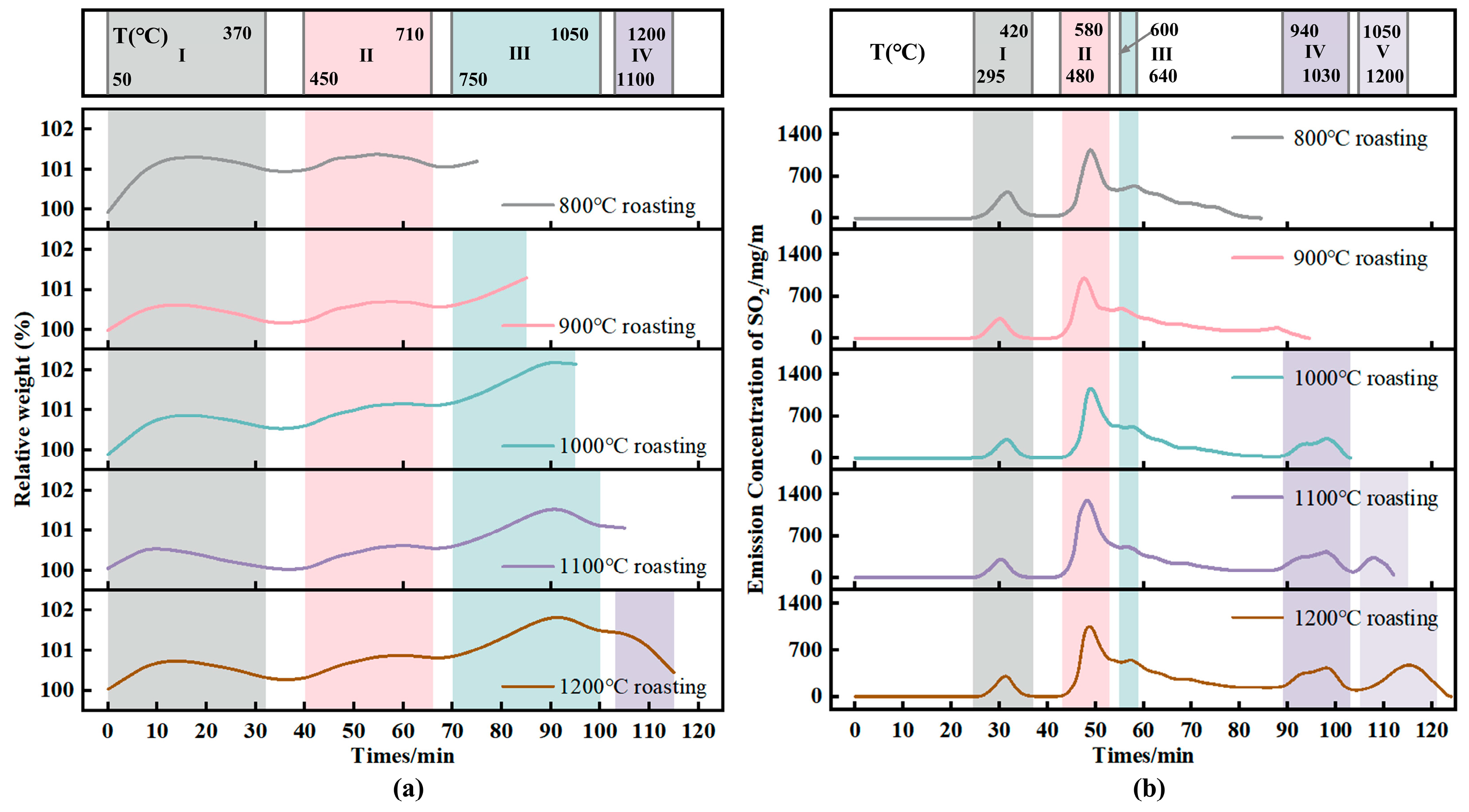
The pellet mass change during the entire roasting process at 800 °C, 900 °C, 1000 °C, 1100 °C and 1200 °C is shown in Figure 2a. Temperature intervals I–IV correspond to the temperature ranges of each peak segment during roasting. Under different roasting temperatures, the pellet mass-change trends are basically consistent. In temperature interval I (50–370 °C), the pellet mass first increases and then decreases. In interval II (450–710 °C), the pellet mass also first increases and then decreases, reaching a peak at approximately 580 °C. In this interval, oxidation and sulfation of FeS and FeS2 cause mass gain, while the formed FeSO4 and Fe2(SO4)3 decompose above 580 °C, resulting in mass loss. When the sulfate formation rate becomes lower than the sulfate decomposition rate, a peak appears in the mass curve. In interval III (750–1050 °C), the pellet mass first increases and then decreases, with a peak at approximately 950 °C. In this interval, oxidation of Fe3O4 to Fe2O3 and sulfur-fixation reactions by alkali metal oxides components contribute to mass gain; with further temperature increase, partial decomposition of alkali metal oxides sulfates leads to mass loss.
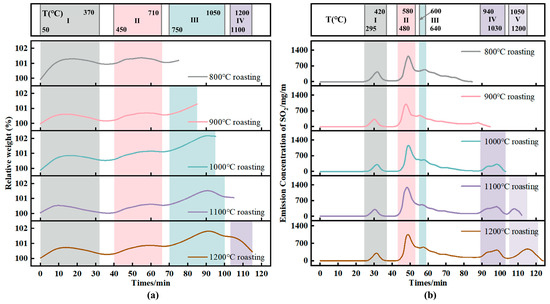
Figure 2.
Pellet mass change and SO2 emission concentration curves during roasting at different set temperatures. (a) Pellet mass change; (b) SO2 emission concentration curves.
To ensure consistency among SO2 emission concentration curves, the measured SO2 concentration was divided by the initial sample mass to obtain the SO2 emission concentration per gram of pellets. The SO2 emission curves for green pellets roasted at 900 °C, 1000 °C, 1100 °C, and 1200 °C are shown in Figure 2b. Temperature intervals I–V correspond to the peak segments in the roasting process. For different roasting temperatures, the peak temperature ranges in intervals I–III are identical; because the holding temperatures differ, interval IV appears at the same temperature range when the set roasting temperatures are 1000, 1100, and 1200 °C.
As the temperature increases, SO2 generation begins at approximately 310 °C. In interval I (295–420 °C), the SO2 concentration reaches a peak at approximately 365 °C; correspondingly, the pellet mass shows a trough after approximately 365 °C in Figure 2a. With further temperature increase, the highest SO2 peak of the whole process appears in interval II (480–580 °C), with a peak temperature at approximately 540 °C. In this temperature range, oxidation and oxidative sulfation of FeS and FeS2 produce a large amount of SO2, and the peak SO2 concentration is about 1050–1200 mg/m3. In interval III (600–640 °C), a peak occurs at approximately 635 °C. With a further temperature increase, interval IV appears at 940–1080 °C, with a peak at approximately 985 °C. In this range, low-melting-point salts melt above 900 °C, exposing sulfur-bearing phases encapsulated inside the pellets to the oxidizing atmosphere and thus generating SO2 via oxidation. As the temperature continues to rise, sulfur-fixation reactions by alkali metal oxides components occur, so the generated SO2 cannot be released in time, leading to a decline of the curve. Interval V appears in the range of 1050–1200 °C, with a peak at approximately 1150 °C; this interval is mainly associated with the decomposition of alkali metal oxides sulfates.
3.3. Phase Evolution of the Roasted Products
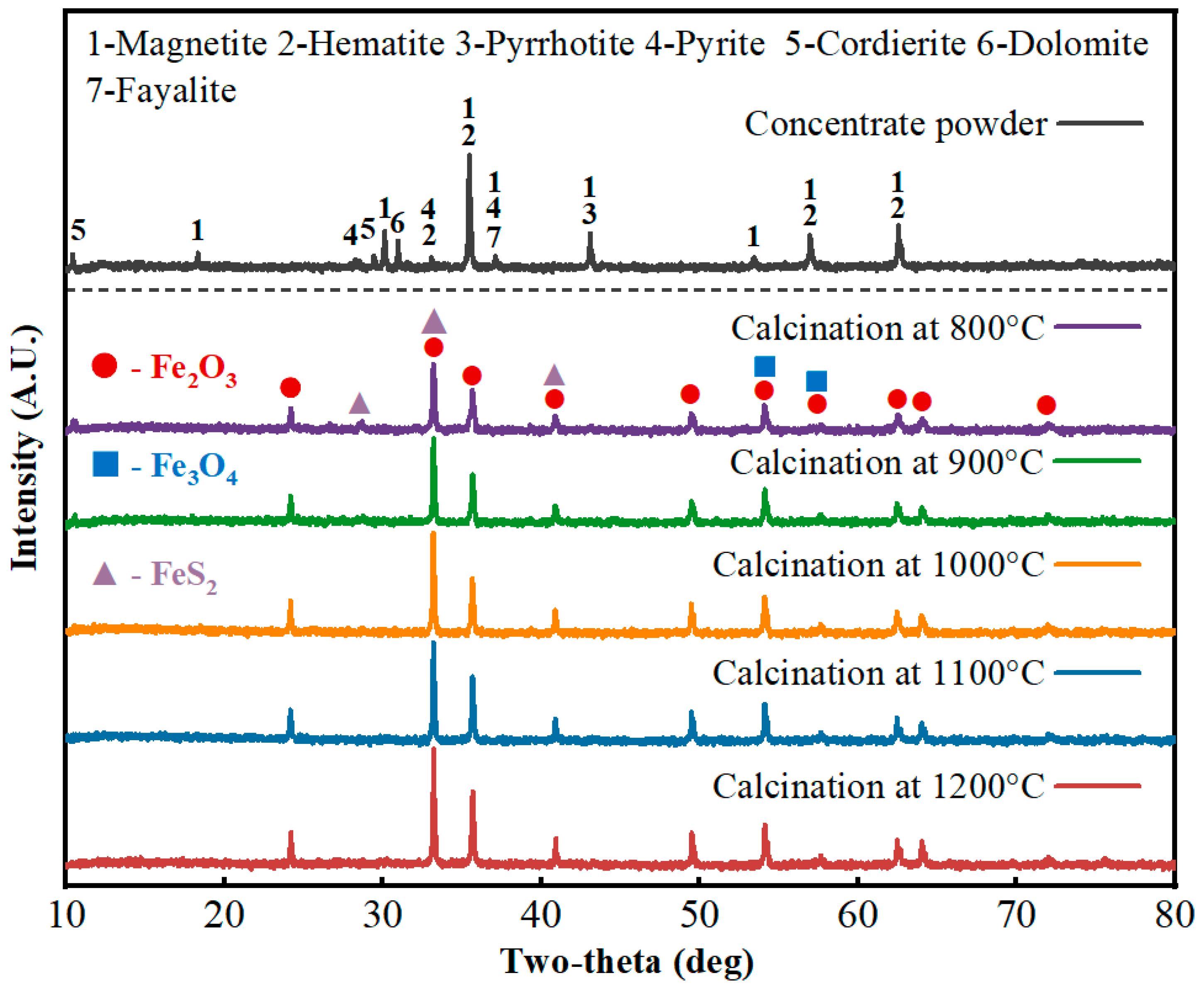
XRD was used to analyze phase compositions of the high-sulfur iron concentrate fines and the interior of pellets after roasting at different temperatures; results are shown in Figure 3. The main iron-bearing phases in the concentrate are magnetite and hematite; the sulfur-bearing phases are mainly pyrite and pyrrhotite; and gangue minerals include fayalite, dolomite, cordierite, etc. In roasted pellets, the main iron-bearing phases are ferric oxide phases and magnetite phases, similar to those before roasting, while sulfur-bearing phases include unreacted residual pyrite. With increasing roasting temperature, diffraction peak intensities increase, mainly because ferric oxide phases are present in most peaks and their content increases as reactions proceed. In Figure 4, the pyrite peak positions overlap with some ferric oxide peaks. During roasting, pyrite gradually oxidizes to form ferric oxide; nevertheless, the intensity of the overlapping peaks still increases with temperature. This is mainly because the diffraction intensity of pyrite planes is weaker than that of ferric oxide planes, and the changes caused by decreasing pyrite content are relatively minor. Therefore, the diffraction peaks at positions shared by pyrite and ferric oxide still increase in intensity as the roasting temperature rises.
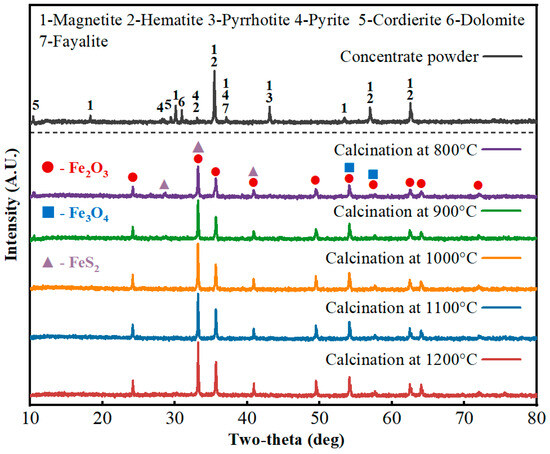
Figure 3.
XRD patterns of the high-sulfur iron concentrate and the pellet interior before and after roasting at different temperatures.
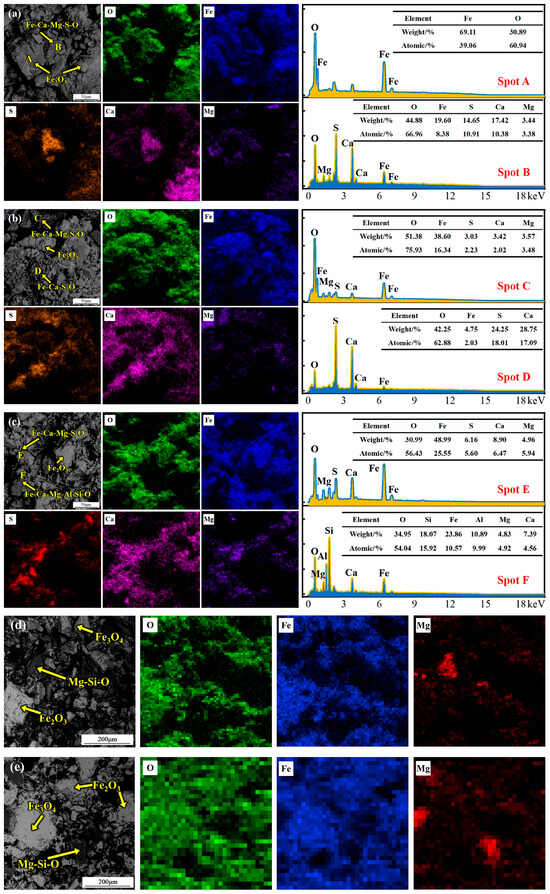
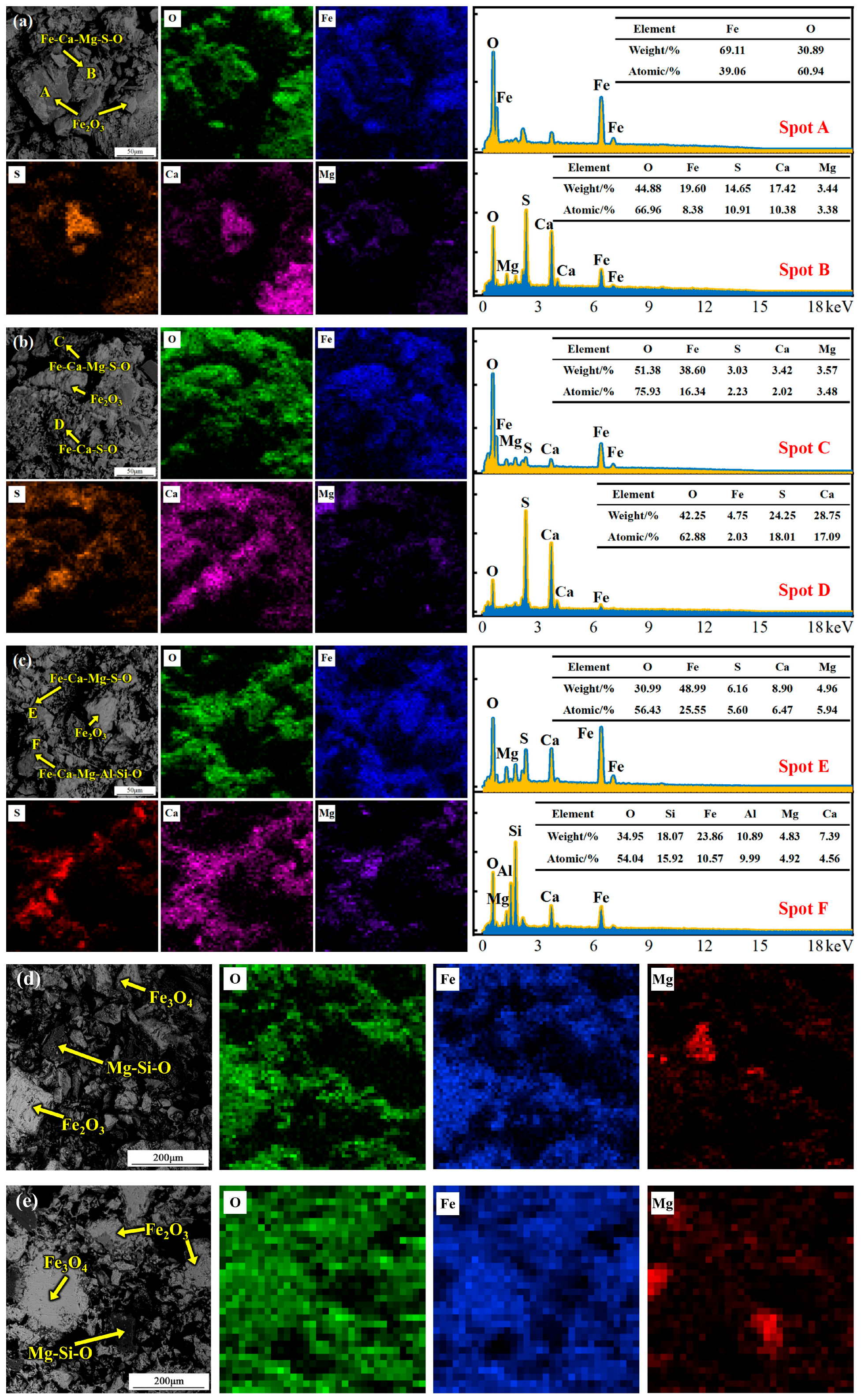
Figure 4.
SEM micrographs and point-analysis results of the pellet interior after roasting at 800, 900, 1000, 1100 and 1200 °C. (a) 800 °C; (b) 900 °C; (c) 1000 °C; (d) 1100 °C; (e) 1200 °C.
Based on the above analysis of sulfur migration behavior, the microstructure and phase assemblages inside pellets roasted at different temperatures were systematically characterized. SEM micrographs and point-analysis results of the pellet interior after roasting at 800 °C are shown in Figure 4a. Sulfur mainly exists as calcium–iron sulfate composite oxides. Point analysis indicates that local Ca-rich regions formed a calcium–magnesium sulfate–iron oxide composite phase with a composition of (Ca0.75Mg0.25)SO4·0.38Fe2O3 via sulfur fixation. Formation of this phase indicates that MgO also participates in sulfur-fixation reactions and forms a composite structure with iron oxides; an independent iron oxide phase also exists as shown at point A. When the temperature increases to 900 °C, SEM micrographs and point-analysis results are shown in Figure 4b. Sulfur-bearing phases become more complex. Point C corresponds to a calcium–magnesium sulfate–iron oxide–magnesium oxide composite phase. Based on EDS results, its chemical formula is (Ca0.91Mg0.09)SO4·3.66Fe2O3·1.47MgO. Formation of this phase is mainly attributed to partial decomposition of magnesium sulfate at a higher roasting temperature. In addition, a calcium sulfate–iron oxide composite phase is also found, as shown at point D, with a chemical formula of CaSO4·0.06Fe2O3. At 1000 °C, sulfur-bearing phases are still dominated by calcium–iron sulfate composite oxides (Figure 4c). With a further increase in the roasting temperature, CaSO4 begins to decompose. The product at point E has a chemical formula of (Ca0.52Mg0.48)SO4·2.28Fe2O3·0.63CaO·0.58MgO based on EDS mapping results.
When the roasting temperature is further increased to 1100 °C and 1200 °C, SEM micrographs of the pellet interior are shown in Figure 4d,e. Because sulfur content is low, sulfur-bearing phases are difficult to identify. The system mainly transforms into iron oxides, Fe3O4, and gangue phases. This series of phase evolutions—especially the various Ca/Mg–Fe sulfate composite phases formed in the intermediate temperature range (800–1000 °C)—demonstrates the sulfur-fixation effect of alkali metal oxides in the gangue.
Sulfur migration during pellet roasting follows a staged release–fixation–re-release pathway. The primary SO2 peak at 480–580 °C originates from rapid oxidation of FeS and FeS2. At 800–1000 °C, part of the SO2 is temporarily immobilized by CaO and MgO, leading to the formation of Ca/Mg sulfate–iron oxide composite phases as indicated by SEM–EDS. With further temperature increase above 1050 °C, these composite sulfates gradually decompose, and sulfur becomes difficult to detect because of its very low level, which agrees with the high desulfurization degree above 99% achieved at 1100 °C and higher. In contrast to prior studies that mainly emphasize the overall desulfurization degree or single-sulfide oxidation kinetics, this work directly correlates gas-phase SO2 evolution with the formation and breakdown of composite sulfate phases, providing a clearer basis for optimizing temperature windows.
3.4. Desulfurization Performance
The pellet mass before and after oxidative roasting and the sulfur content in pulverized roasted pellet samples were measured; results are shown in Table 3. The roasted sample mass decreases compared with the original pellets. The mass loss is mainly due to moisture, volatiles, and SO2 generated during roasting. The mass-loss ratio refers to the ratio of the mass loss of the pellets after roasting to the initial mass of the pellets. The mass-loss ratio increases with the roasting temperature, indicating that reactions of sulfur-bearing phases become more complete at higher temperatures. The sulfur content decreases with increasing the roasting temperature. The initial sulfur content in the pellets is 3.46%. After roasting at 1100 °C and 1200 °C, the desulfurization ratio exceeds 99%. Moreover, the increase in the desulfurization ratio becomes gradually smaller as the roasting temperature increases; thus, the roasting temperature can be set according to practical production requirements.

Table 3.
Changes in element content and desulfurization ratio in pellets after oxidative roasting.
4. Discussion
Oxidative roasting offers a simpler route for deep desulfurization of high-sulfur iron concentrate pellets. Sulfur is removed during induration under controllable residence time and temperature, which is particularly advantageous when sulfides are finely disseminated and difficult to separate. By coupling SO2 release with in situ fixation by CaO and MgO, roasting forms sulfate–iron oxide composite phases that allow phase-based prediction of sulfur migration, thereby facilitating optimization of the roasting temperature profile for deep desulfurization. Future work should validate these findings under industrial pellet induration conditions using plant-like thermal profiles, bed flux, and controlled gas compositions to improve scalability and energy efficiency.
5. Conclusions
Oxidative roasting desulfurization of high-sulfur iron concentrate pellets shows clear stage-wise sulfur release behavior. Under air atmosphere and a heating rate of 10 °C/min, SO2 is mainly released in two temperature stages: the 480–580 °C range corresponds to intense oxidative decomposition of FeS and FeS2 and produces the maximum SO2 emission peak of the whole process; SO2 emission in the 940–1080 °C range is related to exposure and further oxidation of sulfur-bearing phases encapsulated inside the pellets at high temperature.
In the range of 800–1000 °C, the generated SO2 can react with CaO and MgO to form Ca/Mg sulfate–iron oxide composite phases such as (Ca0.75Mg0.25)SO4·0.38Fe2O3 and (Ca0.91Mg0.09)SO4·3.66Fe2O3·1.47MgO. Formation of these sulfur-fixation products transfers part of sulfur from the gas phase to the solid phase, thereby slowing SO2 discharge and leading to a “release–fixation–re-release” process at high temperatures.
The sulfur content of the high-sulfur iron concentrate is 3.46% and decreases continuously with increasing roasting temperature. When the roasting temperature reaches 1100 °C and above, the desulfurization ratio can stably exceed 99%; however, further increasing the temperature to 1200 °C yields only a small improvement. This suggests that, while ensuring deep desulfurization, the roasting temperature range can be optimized by considering energy consumption and production requirements.
Author Contributions
Conceptualization, Z.J., B.Y. and M.J.; Methodology, Z.J. and B.Y.; Investigation, Z.J., B.Y. and G.L.; Resources, M.J., B.Z. and G.Y.; Data collection and analysis, Z.J., B.Y. and Y.X.; Formal analysis, Z.J., G.L. and Y.X.; Writing—original draft preparation, B.Y. and B.Z.; Writing—review and editing, Z.J., G.Y., C.L., G.L. and Y.X.; Supervision, G.Y.; Project administration, B.Z. and C.L.; Funding acquisition, B.Z. and Z.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (52374412); Liao Ning Revitalization Talents Program (XLYC2403111).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding authors.
Conflicts of Interest
Authors Zhongshuai Jia, Guoying Yan, and Guoli Luo were employed by the company Baogang Group Mining Research Institute. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Cai, H.; Zhang, J.; Liu, Z.; Wang, Y.; Li, Z.; Jiang, H. Study on the Effects of Mg–Si Gangue Compositions within Magnetite on Iron Oxide Crystallization during Pellet Roasting. Steel Res. Int. 2025, 96, 395–405. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Y.; Zhang, T.; Xu, J. Summary of sulfur hazards in high-sulfur bauxite and desulfurization methods. Sci. Total Environ. 2024, 948, 174631. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.W.; Xu, M.; Zhang, J.L.; Kong, L.T.; Wan, T.J.; Huang, W.D. Behaviour of sulphur and control of sulphur in CHARP process. J. Iron Steel Res. 2007, 19, 10–15. [Google Scholar]
- Yang, J.; Okumura, K.; Kuwabara, M.; Sano, M. Behavior of magnesium in the desulfurization process of molten iron with magnesium vapor produced in-situ by aluminothermic reduction of magnesium oxide. ISIJ Int. 2002, 42, 685–693. [Google Scholar] [CrossRef]
- Amin, S.A.S.; Shayanfar, S. Desulfurization of iron ore concentrate using a combination of magnetic separation and reverse flotation. J. Chem. Technol. Metall. 2021, 56, 1002–1110. [Google Scholar]
- Lin, S.; Liu, R.; Li, W.; Sun, W.; Hu, Y. Clean desulfurization of sulfur–rich tungsten concentrates by reverse flotation. J. Clean. Prod. 2020, 244, 118876. [Google Scholar] [CrossRef]
- Nakhaei, F.; Irannajad, M. Sulphur removal of iron ore tailings by flotation. J. Dispers. Sci. Technol. 2017, 38, 1755–1763. [Google Scholar] [CrossRef]
- Yur’ev, B.P.; Dudko, V.A. Study of desulphurization processes at heat treatmet of iron ore pellets. Ferr. Metall. Bull. Sci. Tech. Econ. Inf. 2018, 1, 21–28. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, J.; Chen, F.; Wang, S.; Guo, Y.; Chen, S.; Heng, H.; Xu, F.; Wen, Y.; Yang, L. Vanadium Extracted Pellets Desulfurization Consolidation Behavior and Roasted Pellets Reduction Characteristic: Performance and Mechanism. J. Environ. Chem. Eng. 2025, 13, 117814. [Google Scholar] [CrossRef]
- Chen, X.; Zhao, N.; Li, Z.; Wang, Z. Study on the Migration and Release of Sulfur during the Oxidizing Roasting of High-Sulfur Iron Ore. Minerals 2024, 14, 276. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Q.; Liu, X.; Xu, B.; Yang, Y.; Jiang, T. A thermodynamic analysis on the roasting of pyrite. Minerals 2019, 9, 220. [Google Scholar] [CrossRef]
- Urtnasan, E.; Kumar, A.; Wang, J.P. Correlation between Thermodynamic Studies and Experimental Process for Roasting Cobalt-Bearing Pyrite. Metals 2024, 14, 777. [Google Scholar] [CrossRef]
- Tong, S.; Ai, L.; Hong, L.; Zhou, M.; Chen, J. Desulfurization Behavior of Magnetite Pellets Reduced by Isothermal Pure Hydrogen. Steel Res. Int. 2025, 96, 2400698. [Google Scholar] [CrossRef]
- Ning, X.Y.; Xue, Q.G.; Wang, G.; Zeng, F.-B.; She, X.-F.; Wang, J.-S. Desulphurisation mechanism of direct reduction and melting in carbon bearing pellets. Ironmak. Steelmak. 2014, 41, 583–590. [Google Scholar] [CrossRef]
- Chen, X.; Ren, Y.; Gao, W.; Zhao, N. Study of the mechanism of high-efficient in-situ SO2 fixation during oxidative roasting of high-sulfur iron ores and DFT calculation. Fuel Process. Technol. 2025, 267, 108171. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, J.; Liu, Z.; Liu, X.-L.; Xu, C.-Y.; Li, Y. Effects of magnesium olivine on the mineral structure and compressive strength of pellets. Ironmak. Steelmak. 2020, 47, 100–105. [Google Scholar] [CrossRef]
- Abzalov, V.M.; Sudai, A.V.; Yur’Ev, B.P. Desulfurization in roasting iron-ore pellets. Steel Transl. 2008, 38, 1003–1007. [Google Scholar] [CrossRef]
- Ma, K.; Yi, J.; Su, Z.; Bai, X.; Fu, G.; Jiang, T.; Zhang, Y. New insights on desulfurization mechanism of sulfur-containing phases during iron pellet oxidation: Constraints and mitigation strategies. J. Environ. Chem. Eng. 2025, 13, 116414. [Google Scholar] [CrossRef]
- Shi, T.; Guo, H.; Liu, Y.; Wang, L.; Lei, Y.; Hao, H.; Liu, F. DFT combined with XANES to investigate the sulfur fixation mechanisms of H2S on different CaO surfaces. Fuel 2022, 327, 125204. [Google Scholar] [CrossRef]
- Zhang, C.; Yan, A.; Guo, H.; Lei, Y.; Hao, H.; Liu, F. DFT combined with XPS to investigate sulfur removal and sulfur-fixation mechanisms of copper oxide during coal pyrolysis and semi-coke combustion. Fuel 2022, 317, 123482. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.