Abstract
This study developed a composite binder cold briquetting-direct reduction process for zinc removal and resource recovery from zinc-containing dust. Through systematic briquetting and reduction experiments, the optimal briquette parameters were identified, and the mechanisms of zinc migration and removal during reduction were discussed. The results showed that under optimized reduction conditions at 1275 °C for 25 min and with 4% carbon content in the briquettes, the process achieved a zinc removal rate of 98.25% and an iron metallization rate of 90.54%, indicating high Zn removal performance under the tested conditions. Notably, compared with briquettes prepared with conventional organic binders (OB1), the composite binder (CB1) briquettes exhibited higher compressive strength while maintaining comparable Zn removal and metallization performance. The CB1 offers both economic advantages and improved mechanical strength, being successfully applied in industrial lines. Moreover, this process offers an industrially applicable route for the efficient treatment and resource utilization of zinc-bearing dust in the steel industry.
1. Introduction
With the continuous expansion of the global steel industry, the generation of solid wastes from metallurgical processes has increased significantly, among which zinc-bearing dust is one of the most typical by-products. According to data from the World Steel Association, global crude steel production reached 1.883 billion tons in 2024. During this production, approximately 80 million tons of zinc-bearing dust were generated, accounting for about 3–4% of the total crude steel output [1]. This type of dust primarily originates from the dedusting systems of sintering machines, blast furnaces, converters and electric arc furnaces [2,3]. Its chemical composition is highly complex, typically containing valuable elements such as Fe (15–55%), C (1–10%), Zn (0.5–30%) and Pb (0.2–3%), as well as trace amounts of hazardous impurities including Cd, Cr and As [4,5,6]. Due to the coexistence of multiple heavy metals, zinc-bearing dust has been classified as a hazardous solid waste by the U. S. Environmental Protection Agency (USEPA) [7].
The disposal and utilization of zinc-bearing dust have long posed serious challenge to the iron and steel industry due to its intricate composition and hazardous nature. Existing treatment technologies are generally categorized into hydrometallurgical and pyrometallurgical routes [8,9,10]. The hydrometallurgical process, typically involving acid (such as; under high pressure and under an atmospheric pressure), alkaline, or ammonia leaching, offers low energy consumption and simple equipment requirements. However, the presence of stable spinel phases such as ZnFe2O4 significantly limits the leaching efficiency of zinc, making it difficult to achieve complete recovery under ambient conditions [11,12]. Moreover, this approach generates large volumes of wastewater containing residual heavy metals, posing additional environmental concerns [13]. In contrast, pyrometallurgical processes—including rotary kiln, rotary hearth furnace (RHF), and electric arc furnace technologies—rely on high-temperature reduction to volatilize zinc and lead while converting iron oxides into metallic iron [14,15,16]. Among them, the RHF process has gained wide industrial adoption due to its operational flexibility and high zinc removal efficiency, typically exceeding 90% [17]. Nevertheless, conventional carbothermal reduction depends heavily on fossil carbon sources such as coke or coal, resulting in high energy consumption, excessive CO2 emissions, and elevated operational costs [18,19]. Additionally, the reduced products often exhibit poor compressive strength and non-uniform metallization, which limit their direct use as burden materials in blast furnaces and usually were recycled by sintering, leading to a waste of energy [20]. To mitigate these drawbacks, emerging approaches such as integrated pyro–hydrometallurgical, microwave-assisted, and hydrogen-based reduction processes have been proposed to enhance zinc removal and reduce carbon emissions [2,21,22]. Although these methods have shown promising results, issues such as high equipment investment, process complexity, and insufficient mechanical stability of reduced products still hinder large-scale industrial application.
In response to these limitations, cold briquetting technology has recently gained increasing attention as a low-energy and environmentally friendly agglomeration method for treating zinc-bearing dust [23,24,25]. This process involves the compaction of fine dust particles into briquettes at room temperature with binders, followed by high-temperature reduction to achieve zinc volatilization and iron metallization. The performance of the briquettes—particularly their compressive strength, reducibility, and thermal stability—is highly dependent on the binder compositions [26]. Conventional organic or inorganic binders, such as starch, bentonite, and sodium silicate, often fail to retain adequate bonding strength under high-temperature reduction conditions, leading to friability and dust generation during reduction [27].
This study develops a cold briquetting–high-temperature reduction route for treating zinc-bearing dust, enabling zinc removal and iron metallization, and the reduced briquettes can be potentially used for BF or BOF. The effects of briquetting pressure, carbon content, and reduction temperature on compressive strength, Zn removal, and metallization were systematically examined, and briquettes prepared with different binder systems were comparatively evaluated. By positioning against representative zinc-removal routes, the main contribution of this work is to improve briquette integrity using a composite binder; under the optimized conditions (1275 °C, 25 min, and 4% carbon), high Zn removal (98.25%) and metallization (90.54%) were achieved, while the compressive strength was markedly enhanced compared with the conventional organic binder. Overall, the composite binder improves structural compactness and thermal stability, supporting the practical applicability of this route for efficient resource recovery of zinc-bearing dust.
2. Experimental
2.1. Raw Materials
The six types of zinc-bearing dust samples used in this study were collected from one Chinese steel mill, which included blast furnace dust (D1 and D2), electric arc furnace dust (D3), converter dust (D4), converter sludge (D5), and cold-rolled mill sludge (D6). The chemical compositions of these raw materials are listed in Table 1. As shown in the table, D1 (62.86% Fe), D5 (59.46% Fe), and D4 (56.38% Fe) had significantly higher iron contents compared to D6 (25.08% Fe) and D2 (34.76% Fe). Among these materials, D2 contained the highest carbon content (26.37%), while the others showed considerably lower levels ranging from 1.75% C to 6.04% C. The presence of high-carbon constituents helped create a partially reducing atmosphere during subsequent briquetting and reduction processes. The zinc contents in D3, D6, D4, and D5 were measured to be 7.89%, 6.43%, 4.42%, and 3.12%, respectively. Due to their elevated zinc content, these samples require pre-treatment to remove zinc.

Table 1.
Chemical compositions of zinc-bearing dusts/wt%.
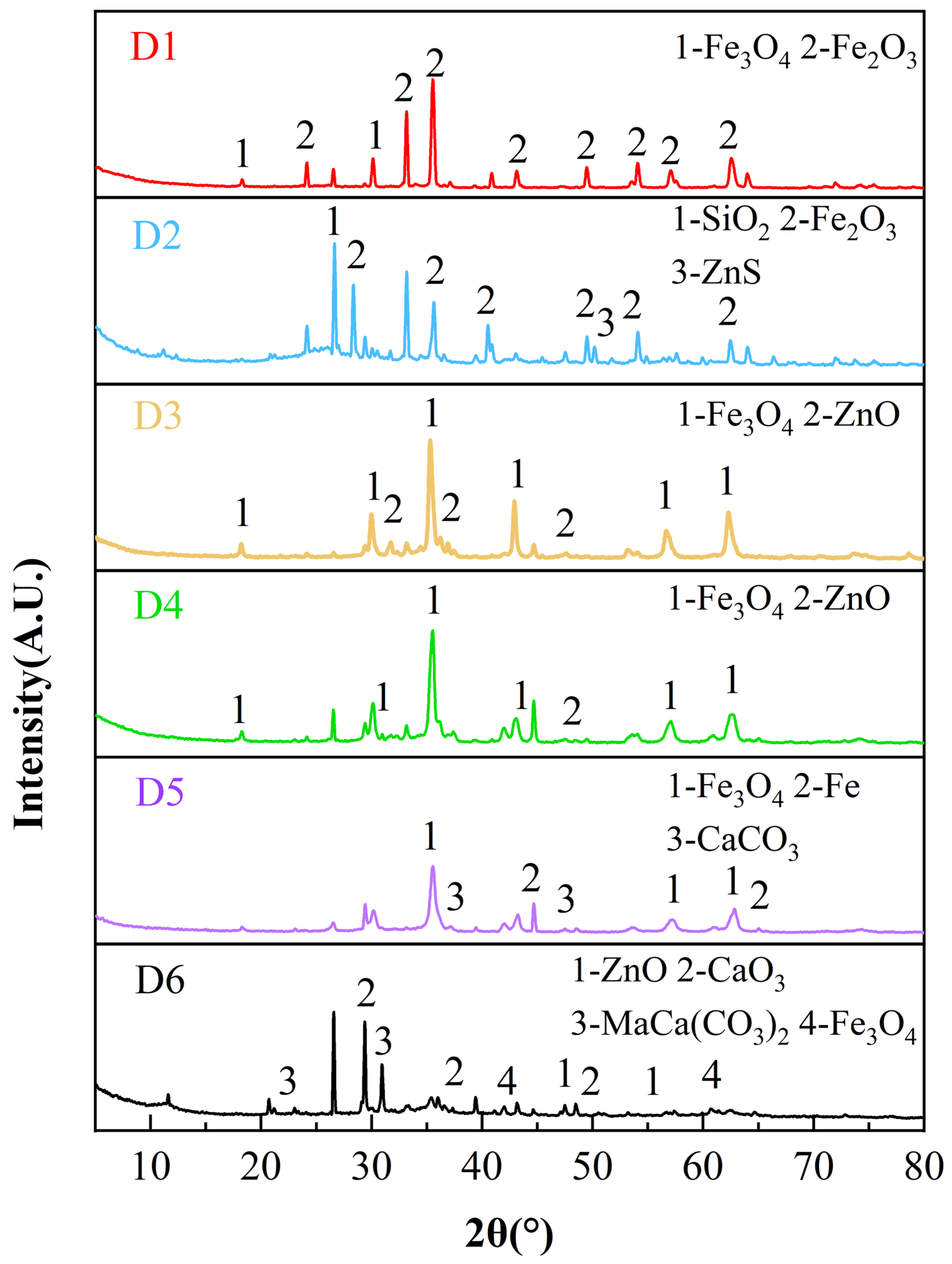
X-ray diffraction (XRD) was performed to identify the major mineral phases of the representative zinc-bearing dusts. The XRD patterns of the six zinc-bearing dust samples (D1–D6) are presented in Figure 1. The XRD patterns indicate that iron mainly exists as Fe3O4 and Fe2O3, while Zn is primarily present as ZnO (D3, D4 and D6) and ZnS (D2). Pb-bearing crystalline phases were not clearly detected by XRD, likely due to the relatively low Pb content and/or peak overlap; thus Pb is reported as total Pb by XRF.
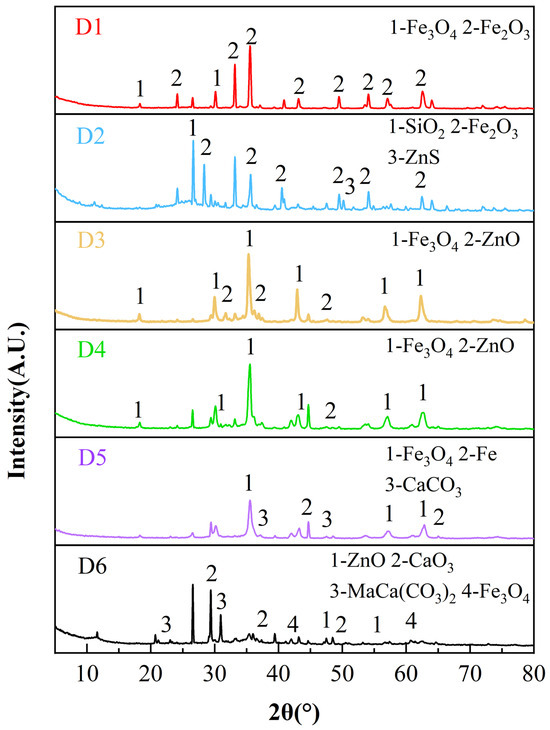
Figure 1.
XRD patterns of six zinc-bearing dusts.
The particle size distributions and specific surface area (SSA) of the six Zinc-bearing dust samples of D1–D5 are presented in Table 2. Most samples (D1–D5) exhibited fine particles dominated by the −0.025 mm fraction (46.66–93.23%), whereas D6 was characterized by a coarser size distribution with 39.35% in the +0.150 mm fraction and only 13.63% in the −0.025 mm fraction (Table 2). All the samples were too fine and have to be agglomerated before direct reduction for zinc removal.

Table 2.
Size distributions and specific surface areas of zinc-bearing dusts.
The reducing agent used for the direct reduction and zinc removal of zinc-bearing dust briquettes was obtained from coke dry quenching (CDQ) dust collected from a coking plant. Its chemical composition and particle size distribution are shown in Table 3 and Table 4, respectively. The CDQ dust had a fixed carbon content of 83.89% and a volatile matter content of 1.72%, with over 85% of the particles passing a 200-mesh sieve. The fine particle size facilitated the uniform dispersion of the reducing agent within the mixture, thereby increasing the specific surface areas available for reactions. The incorporation of this reducing agent during the briquetting process enhanced the reduction efficiency and consequently improved the zinc removal rate.

Table 3.
Chemical compositions of CDQ dust/wt%.

Table 4.
Size distributions of CDQ dust/%.
This study used two binders for comparison: one conventional organic binder (OB1) currently used in production line and a novel composite binder (CB1) developed by the authors. The chemical composition and ash content of the two binders are presented in Table 5 and Table 6, respectively. OB1 contains over 90% carbon (C), hydrogen (H) and oxygen (O), with its binding properties primarily originating from organic components. As shown in Table 5, the total C–H–O content decreases from 90.87 wt% (OB1) to 69.82 wt% (CB1), indicating a reduced organic fraction in CB1. Meanwhile, Table 6 shows that the ash of CB1 is dominated by SiO2 (49.43 wt%) together with CaO and Al2O3, reflecting an enhanced inorganic (ash-forming oxide) fraction compared with OB1. The ash content of CB1 is composed of high-melting-point compounds—including CaO·SiO2 and 2CaO·SiO2—formed by the reaction between SiO2 and CaO [28]. This composition effectively suppresses the melting tendency of zinc-bearing dust briquettes during processing, thereby enhancing the mechanical strength of the briquette products.

Table 5.
Chemical compositions of the binders/wt%.

Table 6.
Chemical compositions of the binder ash/wt%.
2.2. Experimental Methods
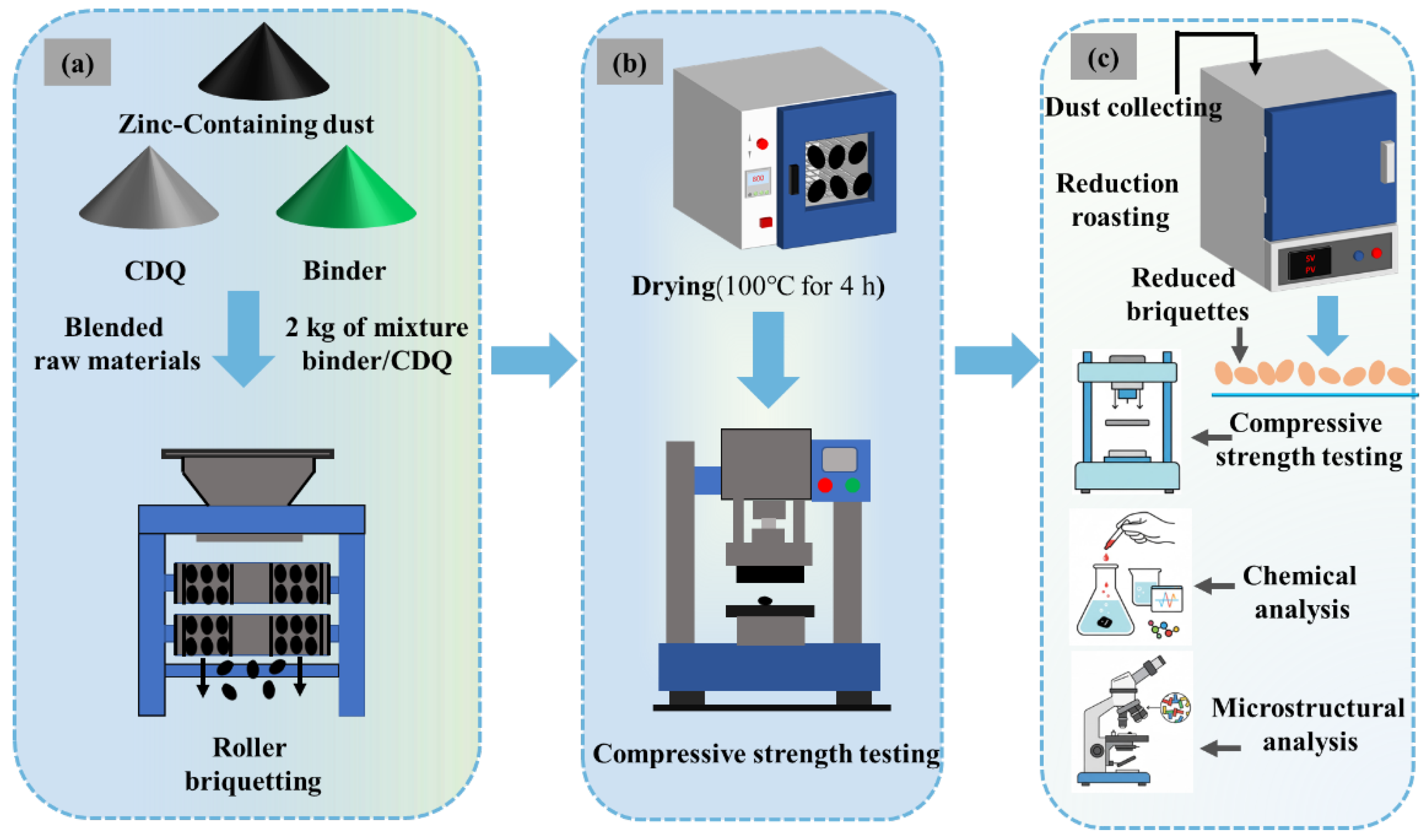
The experimental procedure is schematically illustrated in Figure 2. The raw material formulation was first determined based on the relative generation rate of zinc-bearing dusts from different section in the steel mill, and a mixture was prepared with the following composition: D1 (17.58%), D2 (10.99%), D3 (10.99%), D4 (18.32%), D5 (36.63%), and D6 (5.49%). The chemical composition of the mixture is shown in Table 7. The content of Na2O and Zn is high, and they need to be removed as much as possible during direct reduction. Then, predetermined amounts of binder and CDQ were added to the mixture of dusts. For each batch, 2 kg of the blended material was accurately weighed and thoroughly homogenized before being fed into a double-roll briquetting machine (YQ250 × 50, Hengchang Metallurgical Building Materials Equipment Co., Ltd., Gongyi, China) for agglomeration. Under optimized briquetting parameters (briquetting pressure 20 MPa, binder dosage 4%, moisture content 14%), the resulting green briquettes were dried in a forced-air oven at a constant temperature of 100 °C for 4 h. Finally, the compressive strength of the dried briquettes was measured using a universal testing machine (WDW-50, Fangchen Instrument & Equipment Co., Ltd., Jinan, China), and those meeting the specified standards were selected for subsequent reduction experiments.
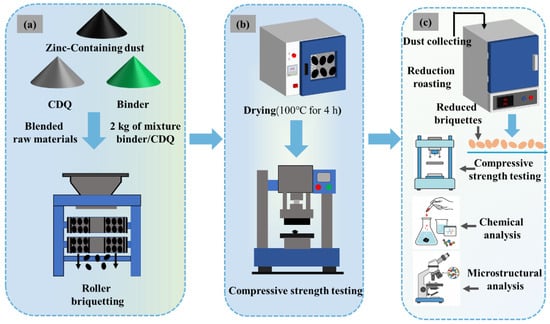
Figure 2.
Schematic diagram of the experimental system. (a) Briquetting experiment; (b) Compression test; (c) Reduction experiment.

Table 7.
Chemical composition of the mixed zinc-bearing dusts/wt%.
Twenty dried briquettes were randomly selected and placed in cylindrical crucibles (80 mm diameter × 80 mm height). The loaded crucibles were then transferred into a muffle furnace (KJ-M1700, Kejia Electric Furnace Co., Ltd., Zhengzhou, China) for high-temperature reduction experiments. No external reducing gas was supplied during the holding stage; the reducing atmosphere was mainly generated by internal carbon in the briquettes. The furnace chamber was operated in air; therefore, N2 purging during cooling was applied to minimize oxidation. Upon completion of the reduction process, high-purity nitrogen was immediately introduced to cool the system and effectively prevent sample oxidation. After cooling, two reduced briquette samples were randomly selected, crushed, and prepared for subsequent chemical composition analysis and phase identification.
The iron metallization rate (ηFe) and the zinc removal rate (φZn) were determined by the following formulas:
where ηFe represents the iron metallization rate (%), MFe and TFe are the contents of metallic iron and total iron content, respectively.
where φZn represents the zinc removal rate (%), m0 and m1 are the masses of the briquettes before and after reduction (g), and Zn0 and Zn1 are the zinc contents of the briquettes before and after reduction (%).
ηFe = (MFe/TFe) × 100
φZn = [1 − (m1 × Zn1)/(m0 × Zn0)] × 100
All experiments were conducted in triplicate (n = 3). Each data point represents the mean of three independent replicates, and error bars denote the standard deviation (SD).
The elemental composition of the samples was determined using X-ray fluorescence (XRF) to quantify both major and minor elements. The microstructure was characterized using a Leica DMLP optical microscope (Wetzlar, Germany) and a scanning electron microscope equipped with an energy-dispersive spectrometer (SEM-EDS, JEOL JSM-6490LV, Tokyo, Japan) to analyze the morphology and elemental distribution of the samples.
3. Results and Discussion
3.1. Optimization of Briquetting Parameters
3.1.1. Effect of Briquetting Pressure
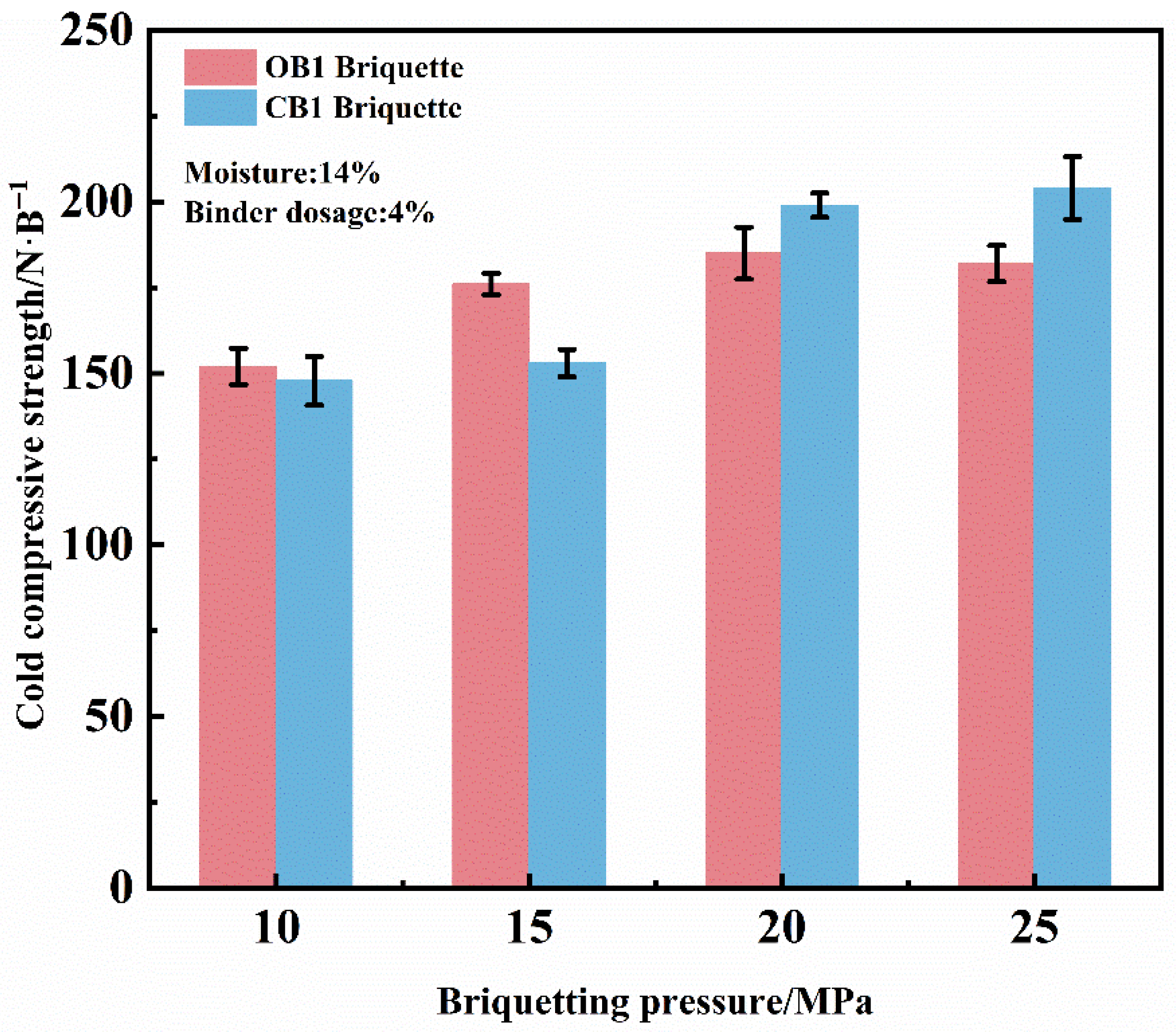
The effect of briquetting pressure on the cold compressive strength (CCS) of dry briquettes was investigated under the conditions of 4% binder, and 14% moisture content, as shown in Figure 3. The results indicate that as the briquetting pressure increased from 10 MPa to 20 MPa, the compressive strength of dry OB1 briquettes increased from 152.4 N/briquette to 186.0 N/briquette, while the compressive strength of dry CB1 briquettes increased from 147.8 N/briquette to 198.2 N/briquette. At a briquetting pressure of 20 MPa, the compressive strengths of both types of briquettes met the production requirements. Although further increase in pressure enhanced the compressive strength, it resulted in substantially higher production costs with a diminishing rate of strength improvement. Moreover, excessively high pressure produced an overly dense microstructure that promoted crack formation and complicated demolding. Conversely, at briquetting pressures below 20 MPa, the briquettes exhibited low compressive strength due to insufficient densification, and was accompanied by an elevated degradation index and return fines ratio. Critically, such high densification impedes the diffusion of reducing gases in subsequent reduction and zinc removal processes, potentially lowering the metallization rate of the final product [20]. Therefore, a briquetting pressure of 20 MPa is recommended for zinc-bearing dust briquettes preparation.
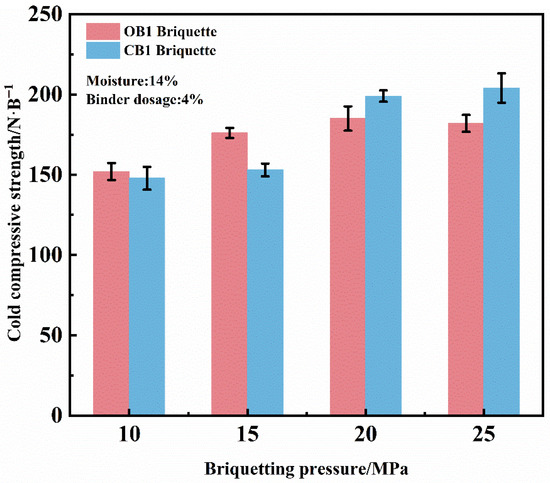
Figure 3.
Effect of briquetting pressure on the CCS of dry briquettes of zinc-bearing dust.
3.1.2. Effect of Binder Dosage
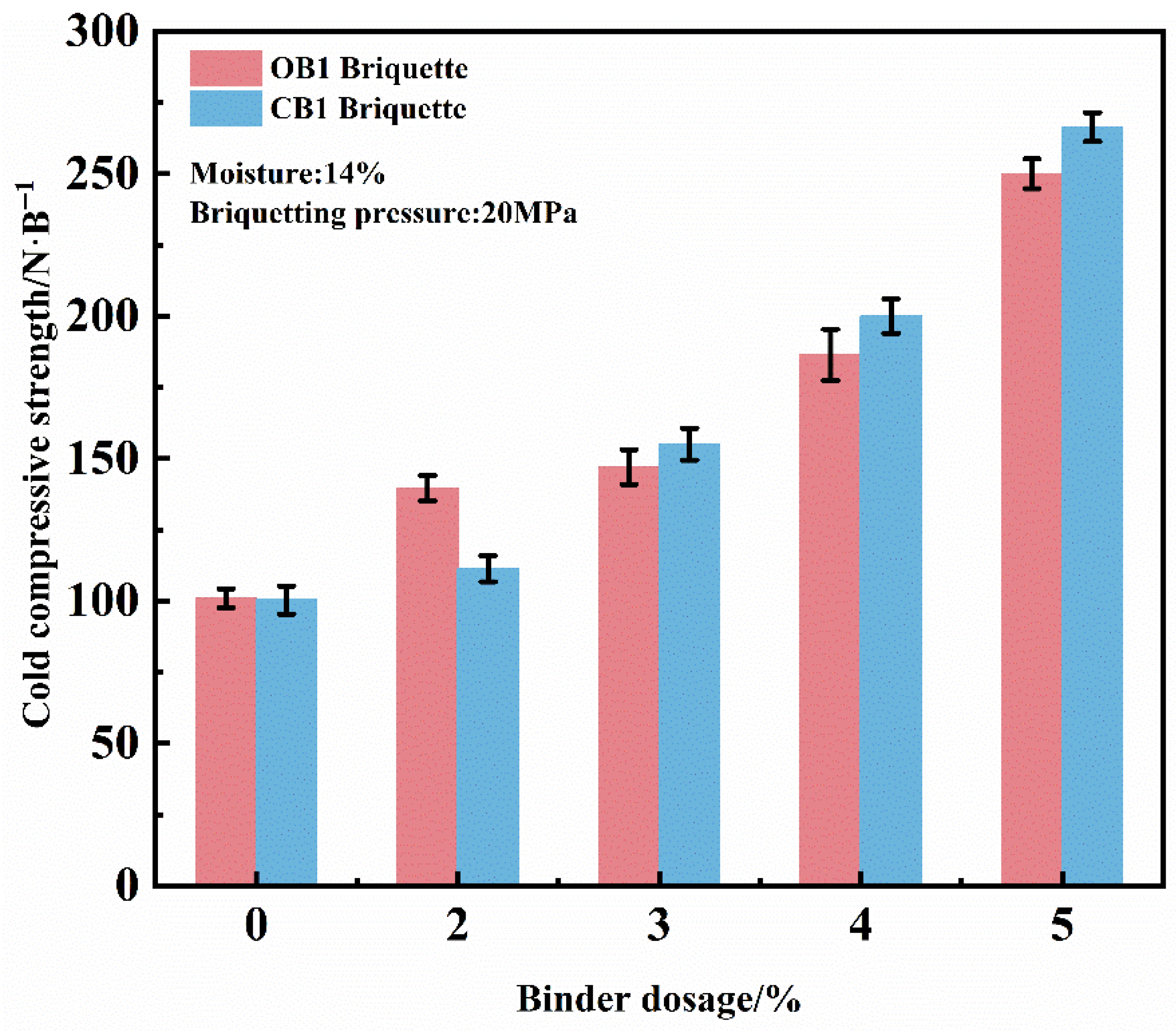
Figure 4 illustrates the influence of binder dosage on the compressive strength of dry briquettes under constant conditions of 20 MPa briquetting pressure and 14% moisture. The compressive strength of both briquette types exhibits a significant increasing trend with higher binder content. In the absence of binder, the briquettes possess a compressive strength of approximately 100 N/briquette, primarily maintained through the mechanical interlocking of particles. As the binder content was raised to 2–3.3%, a marked enhancement in compressive strength was observed. A further increase to 4–5% resulted in compressive strengths of 200–270 N/briquette, representing a more pronounced improvement. At the initial stage, the compressive strength of briquettes prepared with the conventional organic binder (OB1) is slightly higher than that of those with the novel composite binder (CB1). However, once the binder content exceeds 3.3%, the compressive strength of CB1 briquettes increases rapidly and remains consistently higher than that of OB1 briquettes thereafter, demonstrating the superior reinforcing performance of the composite binder at higher dosages. Considering both production efficiency and mechanical performance, a binder content of 4% is recommended as optimal.
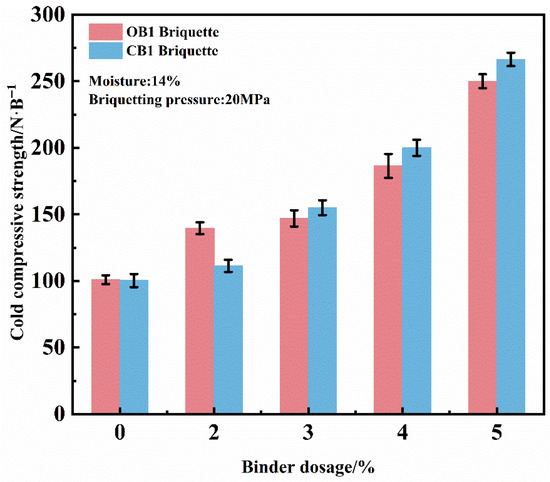
Figure 4.
Effect of binder dosage on the CCS of dry briquettes of zinc-bearing dust.
3.1.3. Effect of Mixture Moisture
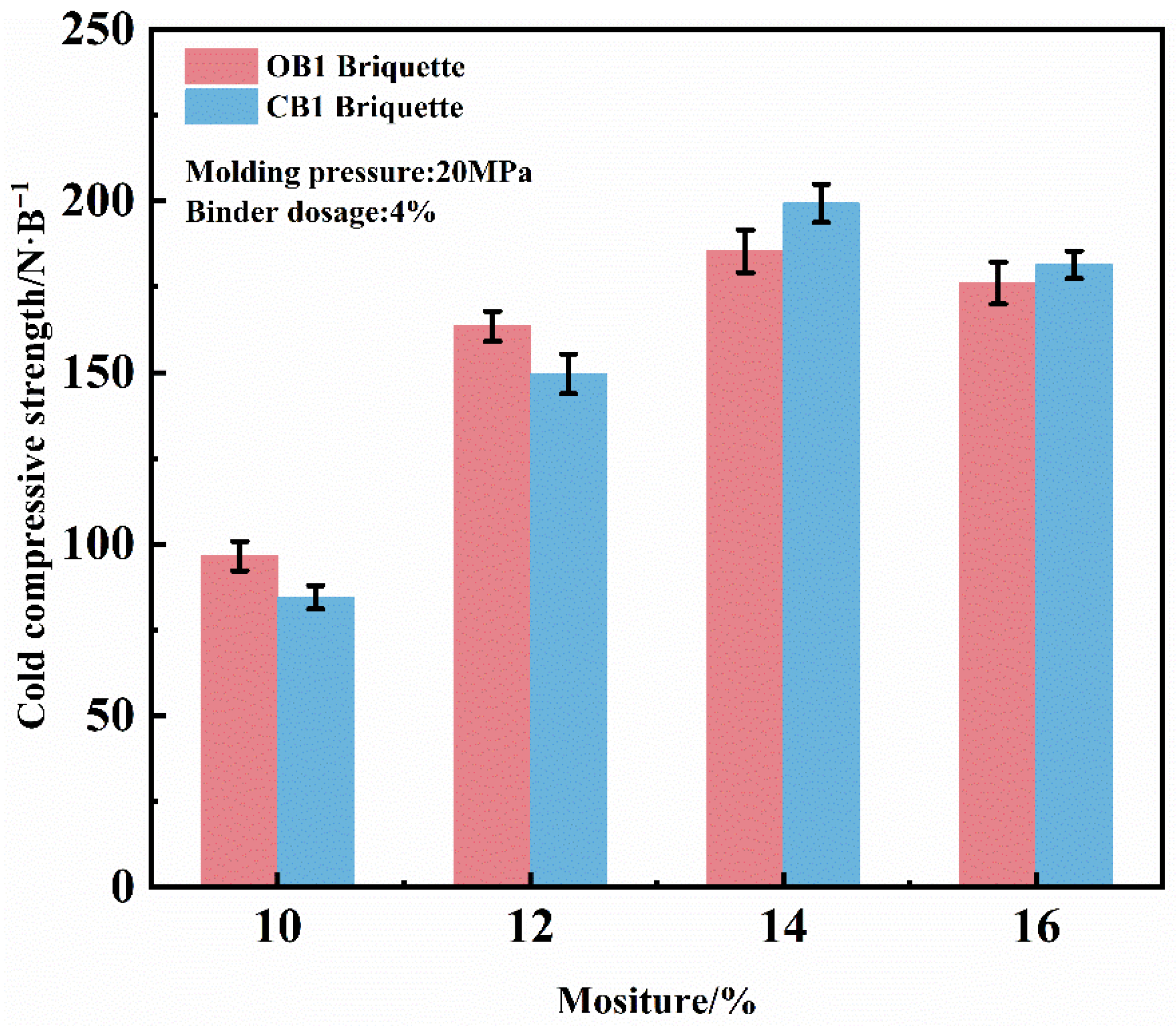
The influence of moisture on the briquette performance was evaluated under fixed parameters of 4% binder and 20 MPa molding pressure, as illustrated in Figure 5. The results reveal that the compressive strength initially increases gradually with moisture content up to a critical point. Beyond 14% moisture content, however, the strength of both briquette types exhibits a clear declining trend. Accordingly, an optimal molding moisture content of 14% is suggested. Under this condition, the compressive strengths of dry OB1 and CB1 briquettes reached 184 N/briquette and 199 N/briquette, respectively, demonstrating the superior strengthening capability of the novel composite binder.
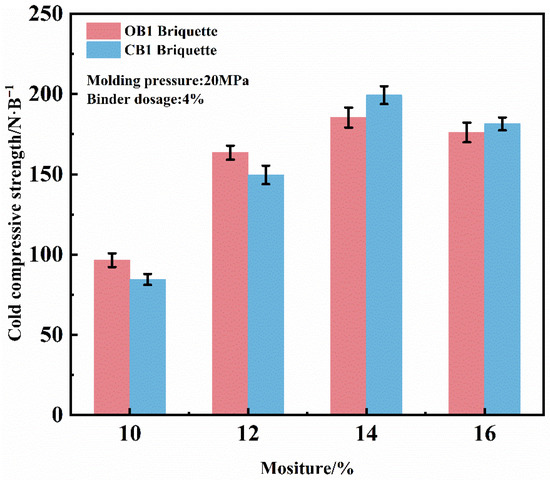
Figure 5.
Effect of mixture moisture on the CCS of briquettes zinc-bearing dust.
The moisture-dependent strengthening behavior is attributed to enhanced capillary forces. As moisture content increases, the diameters of the capillaries between mineral particles decrease and they become rapidly saturated, thereby strengthening the interparticle bonding forces. However, when the moisture content becomes excessively high, the resulting free water disrupts these capillary forces, while the incompressibility of water simultaneously prevents tight particle consolidation [29,30]. Furthermore, overly high moisture content can lead to roll sticking, which complicates the demolding process and adversely affects production efficiency.
3.2. Optimization of Direct Reduction Parameters
3.2.1. Effect of Reduction Temperature
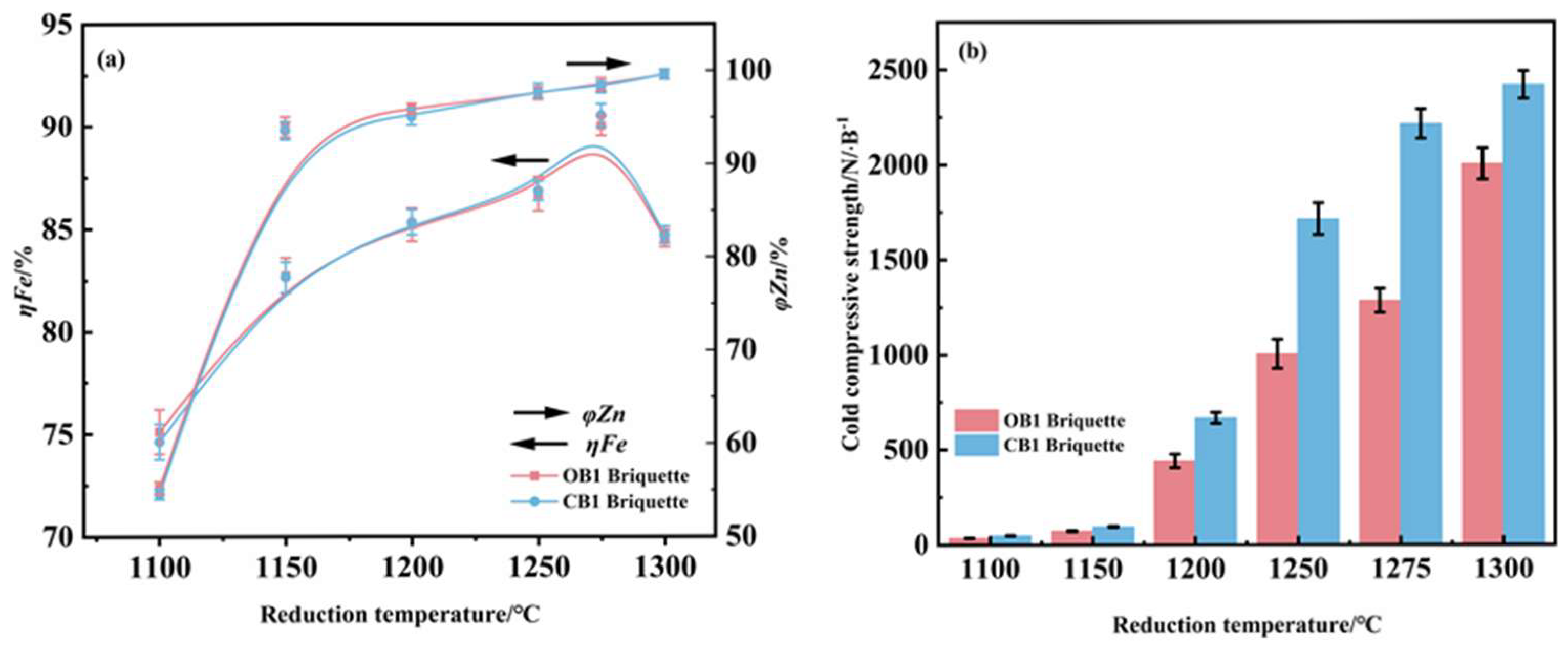
The effect of reduction temperature on the metallurgical behavior of zinc-bearing dust briquettes was examined under constant process parameters of reducing for 25 min and at 4% carbon content, and the results were presented in Figure 6. The iron metallization rate showed a non-monotonic dependence on temperature, peaking at 1275 °C before declining at higher temperatures. This decline is mechanistically explained by accelerated carbon consumption at 1300 °C, which transformed the furnace atmosphere from reducing to oxidizing conditions and promoted re-oxidation of metallic iron [6]. In contrast, both compressive strength and zinc removal efficiency increased progressively with temperature. Under the optimized condition of 1275 °C, OB1 and CB1 briquettes attained iron metallization rates of 90.04% and 90.54%, zinc removal rates of 98.52% and 98.25%, and compressive strengths of 1285 N/briquette and 2214 N/briquette, respectively. The findings indicate that while the two briquettes provide similar reduction performance, CB1 briquettes exhibit markedly more enhanced mechanical integrity than that of OB1.
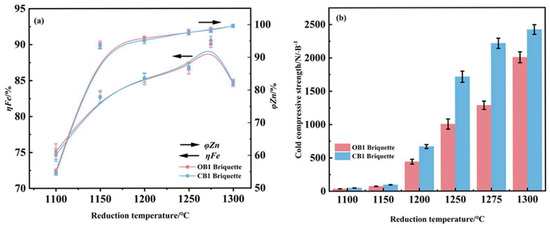
Figure 6.
Influence of reduction temperature on the metallization degree and zinc removal rate (a) and compressive strength (b) of reduced briquettes. (reduction time 25 min; carbon content 4%).
3.2.2. Effect of Reduction Time
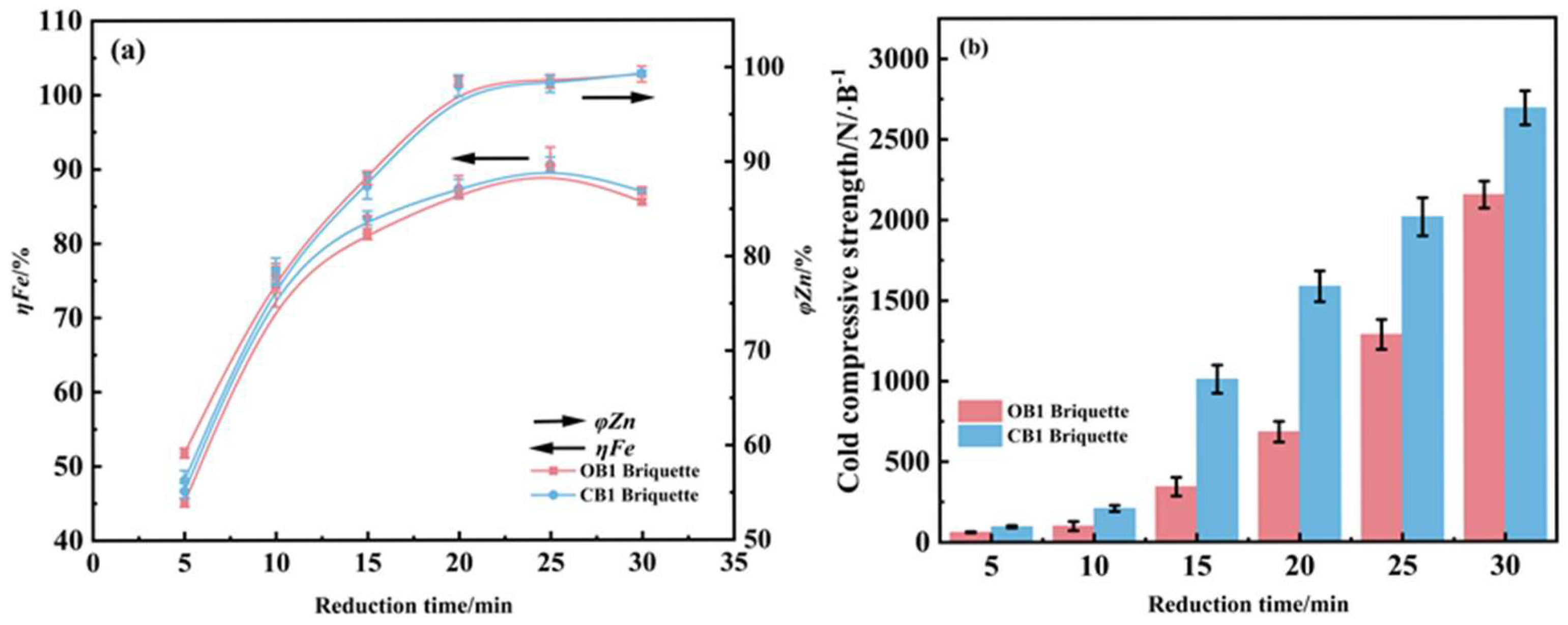
Under fixed conditions of reduction at 1275 °C and 4% carbon content, this work systematically investigated the influence of reduction time on the iron metallization rate and zinc removal efficiency of two types of briquettes, with experimental results shown in Figure 7. The analysis revealed that reduction time plays a critical role in modulating the reduction process. As the reduction duration increased, the iron metallization rate initially rose and then declined, constrained by the limited carbon content within the briquettes. In comparison, the zinc removal rate increased gradually before stabilizing, whereas the compressive strength showed continuous enhancement as the reduction time increased from 5 to 25 min. Based on these observed trends, an optimal reduction time of 25 min was recommended for the system. Although the reduction performance of the two briquettes is very close to each other, the CCS of the reduced briquettes of CB1 is much higher than that of OB1.
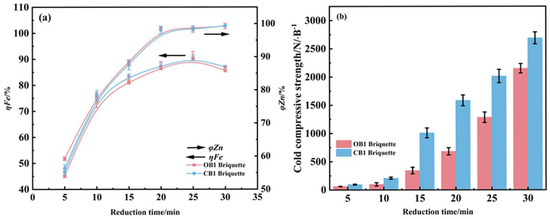
Figure 7.
Influence of reduction time on the metallization degree and zinc removal rate (a) and compressive strength (b) of reduced briquettes (reduction temperature 1275 °C; carbon content 4%).
3.2.3. Effect of Carbon Content
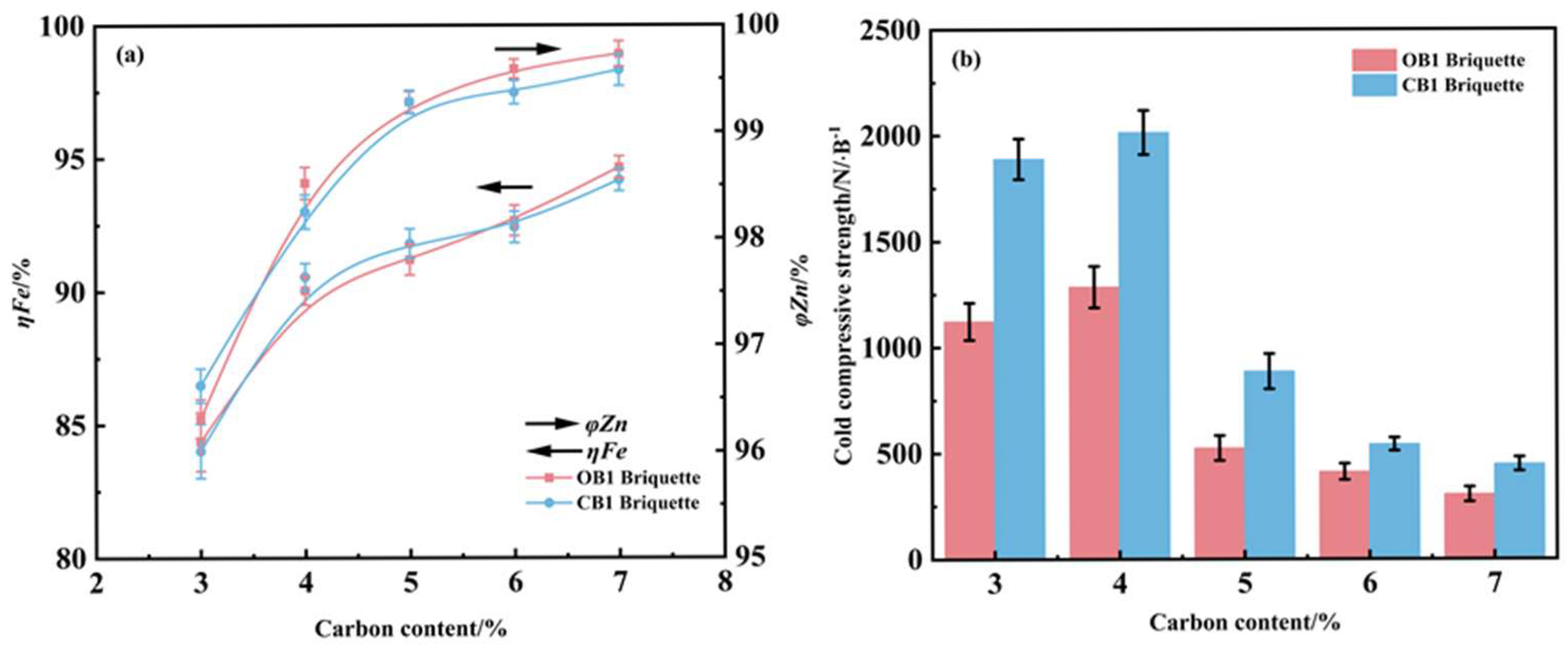
When reducing conditions at 1275 °C for 25 min, the effect of carbon content on the iron metallization rate and zinc removal efficiency of OB1 and CB1 briquettes was investigated, and the corresponding results are given in Figure 8. As the carbon content increased, both types of briquettes exhibited a concurrent enhancement in iron metallization rate and zinc removal efficiency. At a carbon content of 4%, the iron metallization rates of OB1 and CB1 briquettes both exceeded 90%, while their zinc removal efficiencies reached over 95%. Further increasing the carbon content beyond 4% resulted in stabilization of the iron metallization rate and a noticeably reduced improvement in zinc removal efficiency. When the carbon content was raised from 3% to 4%, the compressive strength of both briquettes increased significantly; however, a further increase in carbon content led to a decline in compressive strength. This decline can be attributed to increased porosity caused by excessive gas generation and residual carbon accumulation during the reduction process, which consequently weakened the mechanical integrity of the metallized briquettes [31]. Based on these findings, a carbon content of 4% is recommended as the optimal value for this reduction process.
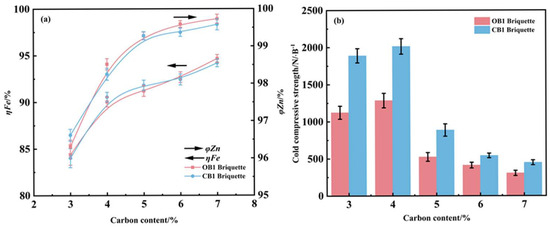
Figure 8.
Influence of carbon content on the metallization degree and zinc removal rate (a) and compressive strength (b) of reduced briquettes (reduction temperature 1275 °C; reduction time 25 min).
3.3. Mechanism of Zinc Removal During the Reduction of Briquettes
Macroscopic images of the directly reduced OB1 and CB1 briquettes are shown in Figure 9. The CB1 briquettes exhibit a higher porosity and a notable metallic luster due to the complete volatilization of zinc from the surface. Consequently, both the metallization degree and the zinc removal rate are significantly enhanced, resulting in a higher compressive strength. In contrast, the OB1 briquettes have a dull surface color, evident cracks, and a looser structure, which leads to their lower compressive strength.

Figure 9.
Macroscopic Images of OB1 briquettes (a) and CB1 briquettes (b) after direct reduction.
The OB1 briquettes prepared with the organic binder exhibited severe microstructural degradation after reduction at 1275 °C for 25 min with a carbon addition of 4 wt%. The internal structure was relatively loose, and the metallic iron grains showed poor aggregation (Figure 10), which accounted for their inferior compressive strength. Micro-area EDS analyses (Table 8) identified the chemical compositions of the residual gangue and partially reduced iron oxides. Sodium (Na) was detected in all analyzed gangue regions (Areas 2–6), which was attributed to the chemistry of high content of Na2O in the organic binder. In the partially reduced iron oxide region (Area 5), high Fe and O contents confirmed the presence of a (Fe, Mg)O solid solution close to the FeO stage, containing approximately 1.43 at.% Na. Sodium, mainly existing as Na2O, entered the iron oxide lattice during reduction. As the reaction proceeded to the FeO stage, a large amount of alkali metal dissolved into the lattice, catalyzing the localized growth of metallic iron crystals and generating interfacial stresses [32]. When the accumulated stress reached a critical level, microcracks propagated, increasing the briquette pulverization rate.

Figure 10.
(a,b) SEM micrograph of reduced OB1 briquettes prepared under the reduction conditions (c) Microstructure of region (a); (d) Microstructure of region (b). (T = 1275 °C, t = 25 min, carbon content = 4%).

Table 8.
The EDS results responding to Figure 10.
Furthermore, Na promoted the formation of low-melting phases within the gangue matrix. Si- and Al-rich regions (Areas 4 and 6, identified as CaxSiOy phases) contained 1.85 at.% and 2.17 at.% Na, respectively. At 1275 °C, molten Na2O reacted with Si and Al to form sodium aluminosilicates [33]. Due to restricted crystallization kinetics, these products predominantly existed as ultrafine or amorphous phases, and such non-equilibrium crystallization behavior further contributed to structural loosening and deterioration of the overall mechanical integrity of the briquettes. Usually high content of Na2O in the briquettes will lead to the degradation of briquettes, in the meantime, the high content of Na2O in the reduced briquettes will do damages to BF when used an BF burden. The metal iron particles are dispersed in the reduced briquettes, and less interlinkage being formed.
These observations explain why OB1 briquettes exhibit poorer performance during reduction, namely more severe cracking/loosening and lower post-reduction compressive strength than CB1 briquettes. Therefore, binders with lower Na2O are preferred for zinc-bearing dust briquetting.
As shown in Figure 11 the briquettes prepared with the composite binder (CB1) exhibited a highly compact internal structure and substantially improved mechanical strength after reduction at 1275 °C for 25 min with 4 wt% carbon addition. As shown in Table 9, EDS analysis revealed that the dominant gangue phases, including Mg-rich ferrite (Area 2), calcium aluminosilicate (Areas 3 and 6), and calcium silicate (Areas 4 and 5), had completely transformed into high-melting stable compounds such as MgFe2O4, Ca2Al2SiO7, and (Ca, Fe)2[SiO4]. The suppression of low-melting sodium aluminosilicate formation effectively prevented the structural degradation observed in OB1 briquettes.

Figure 11.
(a,b) SEM micrograph of reduced CB1 briquettes prepared under the reduction conditions (c) Microstructure of region (a); (d) Microstructure of region (b). (T = 1275 °C, t = 25 min, carbon content = 4%).

Table 9.
The EDS results responding to Figure 11.
In this system, CaO served dual functions: promoting the decomposition of Fe-bearing silicates to metallic iron, thereby reducing the residual FeO content, and reacted with SiO2 to generate high-melting calcium silicates (CaSiO3, Ca2SiO4), forming a robust, refractory gangue framework [28]. Moreover, the homogeneous dispersion of inorganic components within the composite binder decreased porosity and improved particle packing, facilitating the migration and coalescence of reduced iron into a dense, continuous metallic network intimately bonded to the stable gangue matrix. This compact microstructure and enhanced interfacial cohesion were responsible for the remarkable improvement in mechanical strength and the reduced degradation tendency of the CB1 briquettes. The microstructure of the reduced briquettes of OB1 is much better than of OB1, closer interlinkage of metal iron being formed in the former briquettes.
It can also be found that the reduced briquettes containing CB1 have much lower content of Na2O, which means that high Na2O removal rate was achieved from the reduction of CB1 briquettes, and the reduced briquettes of CB1 have better quality than of OB1 briquettes.
4. Conclusions
An innovative composite binder was developed and applied to zinc-bearing dust briquetting and a dry briquette-direct reduction process was investigated; additionally, a low-carbon and efficient route for recycling was developed. The main conclusions are as follows.
- (1)
- The zinc-bearing dusts investigated in this study are characterized by a high zinc content with an uneven distribution, accompanied by elevated levels of iron and carbon. This complex chemical composition and mineralogical structure hinder their direct utilization through conventional treatment methods.
- (2)
- Under the optimized parameters of the composite binder cold-briquetting-direct reduction process, reduced briquettes with an iron metallization degree of 90.54%, a zinc removal rate of 98.25%, and a compressive strength of 2214 N/briquette were obtained. These briquettes exhibit excellent mechanical and metallurgical properties, making them suitable for use as feedstock in electric arc furnaces or BF.
- (3)
- The composite binder enhances the mechanical strength of the briquettes by improving their microstructural stability. Its mechanism lies in suppressing the formation of low-melting sodium aluminosilicates and promoting the generation of high-melting structural phases such as MgFe2O4 and Ca2Al2SiO7, thereby producing a denser and more robust metallic iron network. In addition, alkali-related degradation was alleviated for CB1 briquettes after reduction. Quantitative Na2O measurements will be conducted in future work.
Author Contributions
G.L.: Investigation, Data curation, Writing—original draft preparation. D.Z.: Methodology, Writing—review & editing. M.H.: Investigation, Data curation. J.P. and C.Y.: Supervision, Validation. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the financial support from the National Key Research and Development Program of China grant number (2023YFC3903900, 2023YFC3903904).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
Author Mingzhou Hou was employed by the company Zhejiang Huayou Cobalt Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- World Steel in Figures 2024. Available online: https://worldsteel.org/data/world-steel-in-figures/world-steel-in-figures-2024/ (accessed on 18 November 2025).
- Zhang, W.; Dong, T.; Zhu, D.; Jiang, Y.; Pan, J.; Guo, Z.; Li, S.; Ma, W. Improving Zinc Reduction and Removal from Pellets of Zinc-Bearing Dusts via Vacuum Microwave-Assisted Carbothermal Reduction Process. Process Saf. Environ. Prot. 2024, 192, 896–906. [Google Scholar] [CrossRef]
- Zhu, D.; Wang, D.; Pan, J.; Tian, H.; Xue, Y. A Study on the Zinc Removal Kinetics and Mechanism of Zinc-Bearing Dust Pellets in Direct Reduction. Powder Technol. 2021, 380, 273–281. [Google Scholar] [CrossRef]
- Xue, Y.; Hao, X.; Liu, X.; Zhang, N. Recovery of Zinc and Iron from Steel Mill Dust—An Overview of Available Technologies. Materials 2022, 15, 4127. [Google Scholar] [CrossRef]
- Zhu, R.; Huang, R.; Xu, A.; Li, B.; Zang, Y.; Deng, X.; Yang, J.; Li, M.; Long, X. Research Status and Development of Extraction Process of Zinc-Bearing Dust from Ironmaking and Steelmaking—A Critical Review. J. Iron Steel Res. Int. 2023, 30, 1303–1323. [Google Scholar] [CrossRef]
- Gao, X.; Chai, Y.; Wang, Y.; Luo, G.; An, S.; Peng, J. Process and Mechanism of Preparing Metallized Blast Furnace Burden from Metallurgical Dust and Sludge. Sci. Rep. 2024, 14, 9760. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency Organization. Hazardous Waste. Available online: https://www.epa.gov/hw (accessed on 18 November 2025).
- Wang, D.; Zhu, D.; Pan, J.; Guo, Z.; Tian, H.; Xue, Y. A High-Efficiency Separation Process of Fe and Zn from Zinc-Bearing Dust by Direct Reduction. J. Iron Steel Res. Int. 2022, 29, 1559–1572. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Cui, K.; Fu, T.; Gao, J.; Hussain, S.; AlGarni, T.S. Pyrometallurgical Recovery of Zinc and Valuable Metals from Electric Arc Furnace Dust—A Review. J. Clean. Prod. 2021, 298, 126788. [Google Scholar] [CrossRef]
- Jha, M.K.; Kumar, V.; Singh, R.J. Review of Hydrometallurgical Recovery of Zinc from Industrial Wastes. Resour. Conserv. Recycl. 2001, 33, 1–22. [Google Scholar] [CrossRef]
- Xie, Z.; Jiang, T.; Chen, F.; Guo, Y.; Wang, S.; Yang, L. Phase Transformation and Zinc Extraction from Zinc Ferrite by Calcium Roasting and Ammonia Leaching Process. Crystals 2022, 12, 641. [Google Scholar] [CrossRef]
- Binnemans, K.; Jones, P.T.; Manjón Fernández, Á.; Masaguer Torres, V. Hydrometallurgical Processes for the Recovery of Metals from Steel Industry By-Products: A Critical Review. J. Sustain. Metall. 2020, 6, 505–540. [Google Scholar] [CrossRef]
- Remeteiová, D.; Ružičková, S.; Mičková, V.; Heželová, M.; Pikna, Ľ. Treatment of Strong Alkaline Wastewater from Neutral Leaching of EAF Dust by Precipitation and Ion Exchange. Appl. Water Sci. 2023, 13, 138. [Google Scholar] [CrossRef]
- Xie, Z.; Li, G.; Guo, Y.; Wang, S.; Chen, F.; Yang, L.; Fu, G.; Jiang, T. Mineral Phase Reconstruction and Separation Behavior of Zinc and Iron from Zinc-Containing Dust. Materials 2023, 16, 3481. [Google Scholar] [CrossRef]
- Kim, J.; Cho, M.-K.; Jung, M.; Kim, J.; Yoon, Y.-S. Rotary Hearth Furnace for Steel Solid Waste Recycling: Mathematical Modeling and Surrogate-Based Optimization Using Industrial-Scale Yearly Operational Data. Chem. Eng. J. 2023, 464, 142619. [Google Scholar] [CrossRef]
- Zhao, G.; Li, R.; Xing, X.; Ju, J.; Li, X.; Zu, J. Removal Behavior of Zn and Alkalis from Blast Furnace Dust in Pre-Reduction Sinter Process. Green Process. Synth. 2023, 12, 20230045. [Google Scholar] [CrossRef]
- Wu, Y.; Jiang, Z.; Zhang, X.; Wang, P.; She, X. Numerical Simulation of the Direct Reduction of Pellets in a Rotary Hearth Furnace for Zinc-Containing Metallurgical Dust Treatment. Int. J. Miner. Metall. Mater. 2013, 20, 636–644. [Google Scholar] [CrossRef]
- Khaidarov, T.B.; Khanna, R.; Khaidarov, B.B.; Li, K.; Suvorov, D.S.; Metlenkin, D.A.; Burmistrov, I.N.; Gorokhovsky, A.V.; Volokhov, S.V.; Kuznetsov, D.V. An Innovative Approach Toward Enhancing the Environmental and Economic Sustainability of Resource Recovery from Hazardous Zn-Bearing Dusts from Electric Arc Furnace Steelmaking. Sustainability 2025, 17, 2773. [Google Scholar] [CrossRef]
- Leuchtenmueller, M.; Legerer, C.; Brandner, U.; Antrekowitsch, J. Carbothermic Reduction of Zinc Containing Industrial Wastes: A Kinetic Model. Metall. Mater. Trans. B 2021, 52, 548–557. [Google Scholar] [CrossRef]
- Somerville, M. The Strength and Density of Green and Reduced Briquettes Made with Iron Ore and Charcoal. J. Sustain. Metall. 2016, 2, 228–238. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, S.; Chen, B.; Xin, C.; Ding, W.; Zhang, H.; Bao, S. Clean Treatment of Zinc-Bearing Dust: Zinc-Iron Separation and Nano Zinc Oxide Preparation Based on Low-Temperature Roasting and Water Leaching Process. Sep. Purif. Technol. 2025, 373, 133527. [Google Scholar] [CrossRef]
- Guo, Z.; Xu, X.; Li, S.; Zhu, D.; Pan, J.; Yang, C. Hydrogen Reduction Process for Zinc-Bearing Dust Treatment: Reduction Kinetic Mechanism and Microstructure Transformations in a Novel and Environmentally Friendly Metallurgical Technique. J. Environ. Chem. Eng. 2023, 11, 110836. [Google Scholar] [CrossRef]
- Barisetty, S.; Kalshetty, S.; Ramakrishna, S.; Vishwanath, S.C.; Balachandran, G. Cold Briquetting of DRI Fines for Use in Steel Making Process. Trans. Indian Inst. Met. 2020, 73, 449–455. [Google Scholar] [CrossRef]
- Wu, S.; Chang, F.; Zhang, J.; Lu, H.; Kou, M. Cold Strength and High Temperature Behaviors of Self-Reducing Briquette Containing Electric Arc Furnace Dust and Anthracite. ISIJ Int. 2017, advpub. [Google Scholar] [CrossRef]
- Paknahad, P.; Askari, M.; Shahahmadi, S.A. Cold-Briquetted Iron and Carbon (CBIC), Investigation of Steelmaking Behavior. J. Mater. Res. Technol. 2020, 9, 6655–6664. [Google Scholar] [CrossRef]
- Lemos, L.R.; da Rocha, S.H.F.S.; de Castro, L.F.A. Reduction Disintegration Mechanism of Cold Briquettes from Blast Furnace Dust and Sludge. J. Mater. Res. Technol. 2015, 4, 278–282. [Google Scholar] [CrossRef]
- Han, H.; Duan, D.; Yuan, P. Binders and Bonding Mechanism for RHF Briquette Made from Blast Furnace Dust. ISIJ Int. 2014, 54, 1781–1789. [Google Scholar] [CrossRef]
- Nekhoroshev, E.; Shevchenko, M.; Cheng, S.; Shishin, D.; Jak, E. Thermodynamic Re-Optimization of the CaO–SiO2 System Integrated with Experimental Phase Equilibria Studies. Ceram. Int. 2024, 50, 33686–33702. [Google Scholar] [CrossRef]
- Hamid, M.F.; Idroas, M.Y.; Ishak, M.Z.; Alauddin, Z.A.Z.; Miskam, M.A.; Abdullah, M.K. An Experimental Study of Briquetting Process of Torrefied Rubber Seed Kernel and Palm Oil Shell. BioMed Res. Int. 2016, 2016, 1679734. [Google Scholar] [CrossRef]
- Silviana, S.; Hadiyanto, H.; Widayat, W.; Dalanta, F.; Sa’adah, A.N. Modification of Charcoal Briquette Using Binding Agents Improving the Product Quality: A Review. AIP Conf. Proc. 2023, 2683, 030068. [Google Scholar] [CrossRef]
- Ning, X.; Ren, Z.; Zhang, N.; Wang, G.; Zhang, X.; Wu, J.; Liu, J.; Karasev, A.; Wang, C. Reduction Study of Carbon-Bearing Briquettes in the System of Multiple Reductants. Materials 2025, 18, 4408. [Google Scholar] [CrossRef]
- Jiang, X.; Lu, F.; Ju, J.; Guo, W.; Zu, J. The Influence of Alkali Metals on the Sintering Mineralization Process of Iron Ore. Materials 2025, 18, 227. [Google Scholar] [CrossRef]
- Sharma, T.; Gupta, R.C.; Prakash, B. Effect of Gangue Content on the Swelling Behaviour of Iron Ore Pellets. Miner. Eng. 1990, 3, 509–516. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.