Recovering Lithium from the Cathode Active Material in Lithium-Ion Batteries via Thermal Decomposition
Abstract
1. Introduction
2. Materials and Methods
2.1. Lithium Leaching
2.1.1. Cathode Model Sample
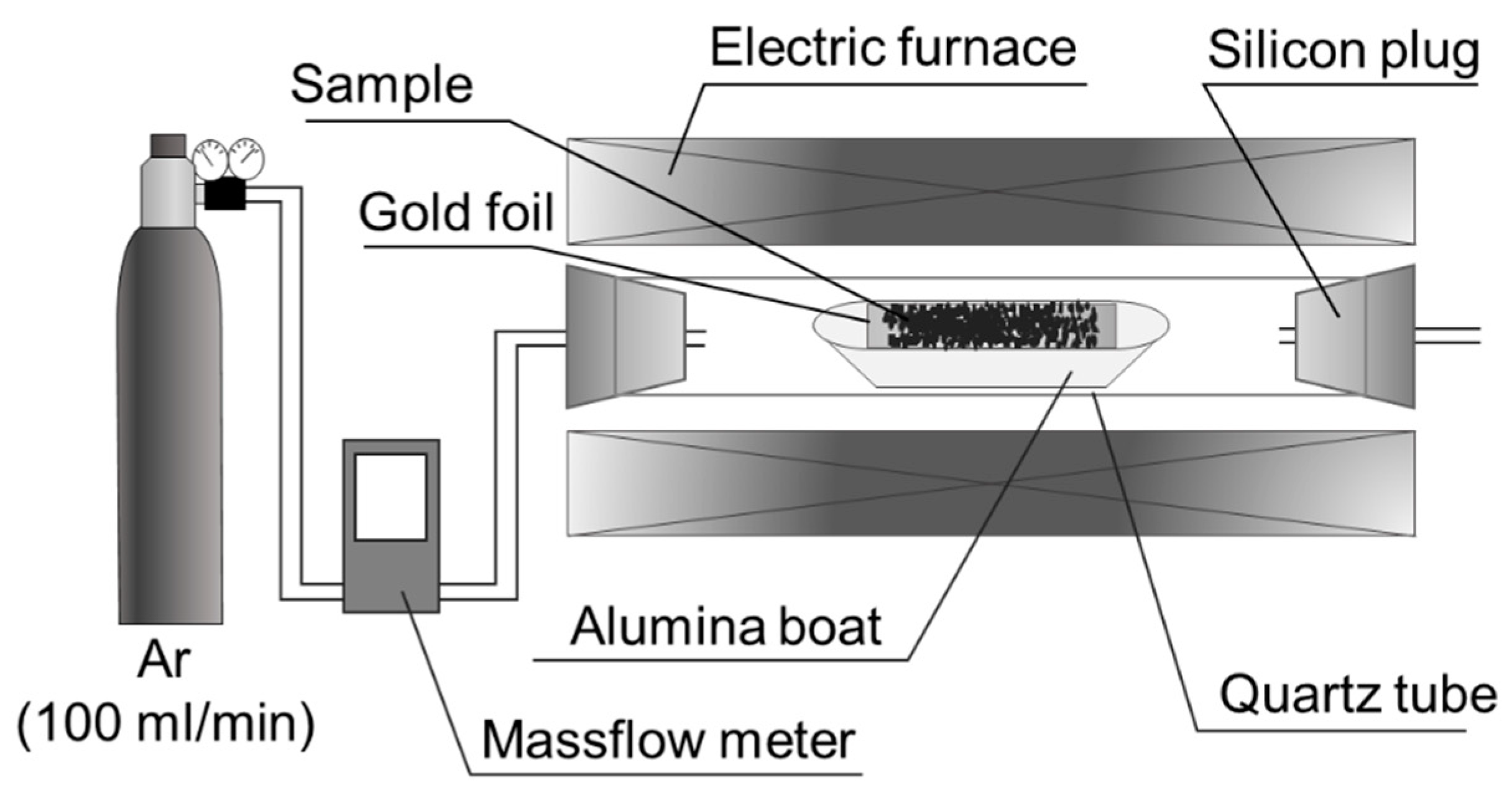
2.1.2. Calcination Test
2.1.3. Lithium Leaching Test
2.2. Lithium Carbonate Purification
2.2.1. Dissolution Test
2.2.2. Carbonation Test
2.3. Characterization
2.3.1. Thermogravimetric (TG) and Differential Thermal Analysis (DTA)
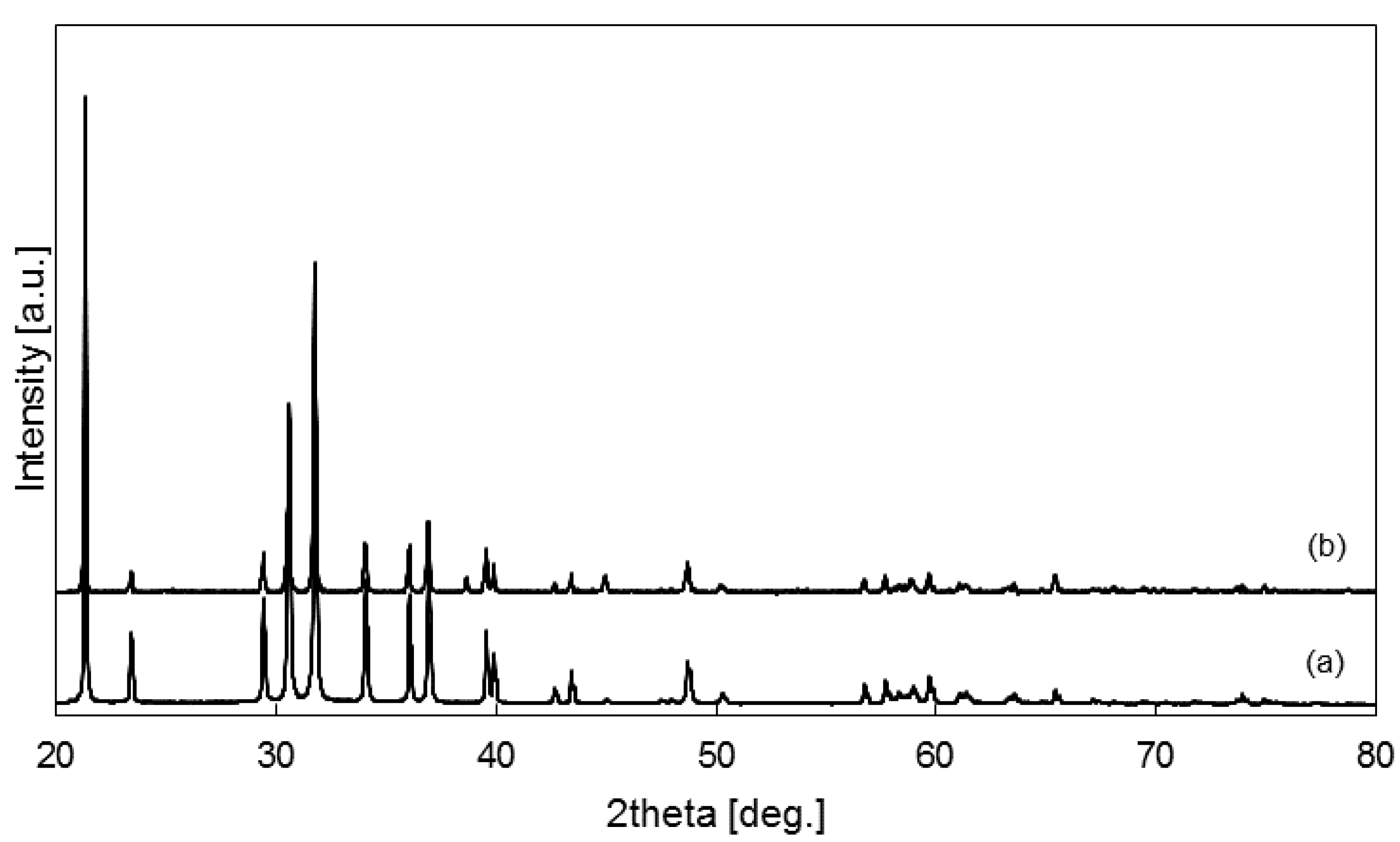
2.3.2. Crystalline Phase Identification
2.3.3. Lithium and Fluorine Quantification
2.3.4. Lithium Balance
2.3.5. Carbon Analysis of Precipitate
2.3.6. Calculation of Lithium Carbonate Concentration in the Precipitate
- The weight of F in the precipitate was calculated by subtracting the weight of F in the filtrate from the weight of added fluoride.
- The weight of F in the precipitate was converted to the weight of LiF.
- The Li2CO3 concentration in the precipitate was obtained by subtracting the weight of the LiF from the total weight of the precipitate.
3. Results and Discussion
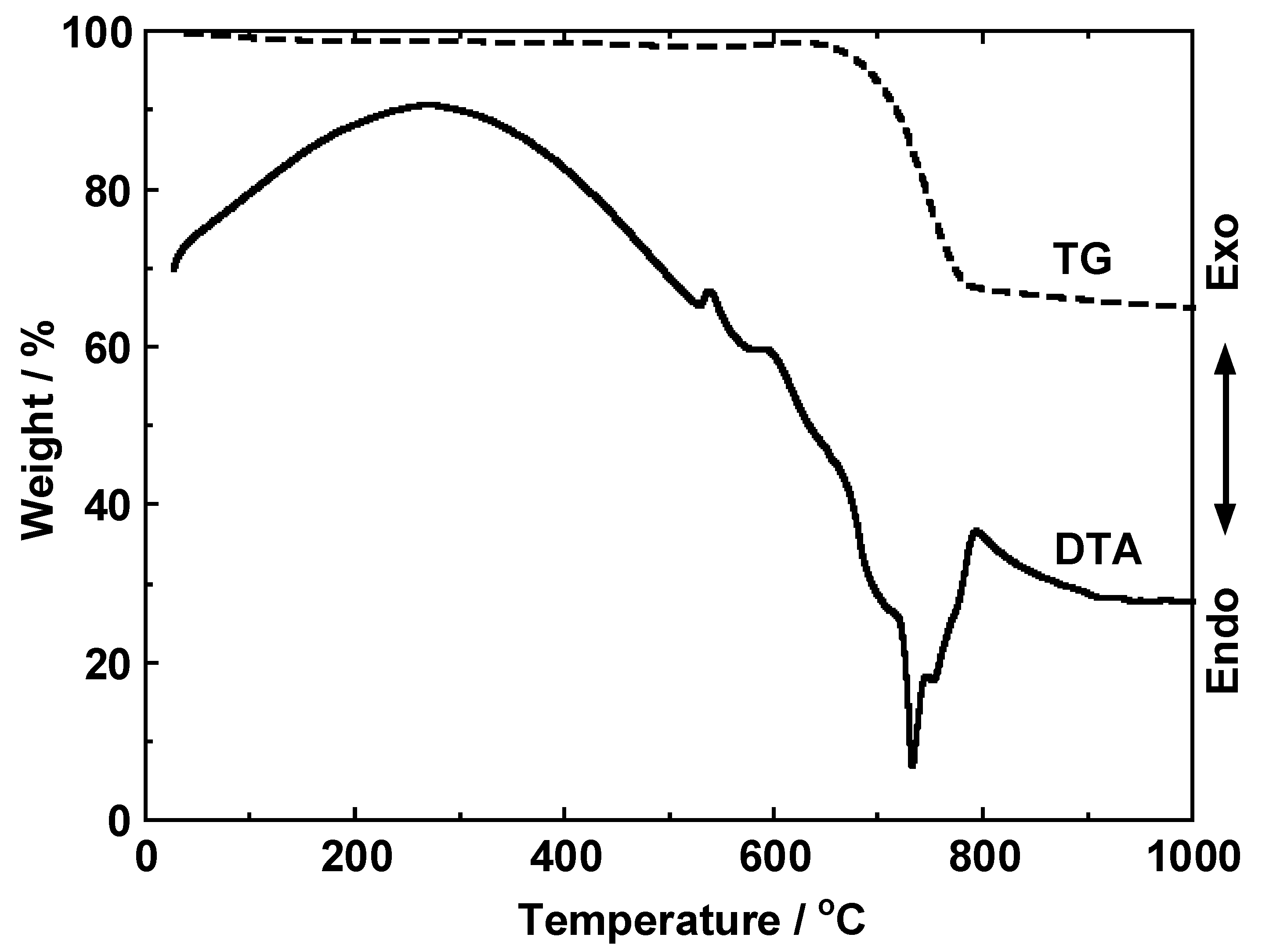
3.1. Thermal Behavior of Cathode Model Sample
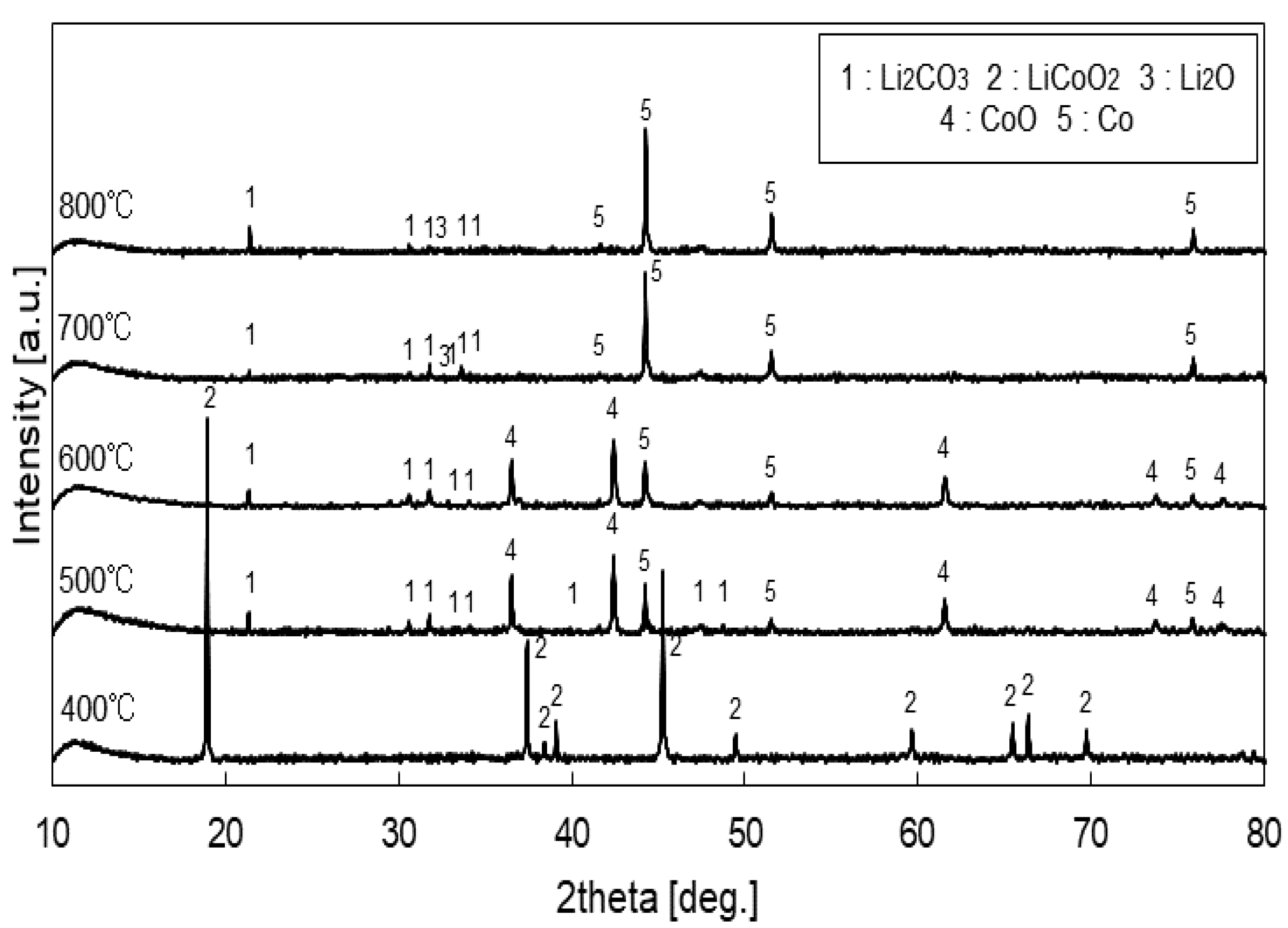
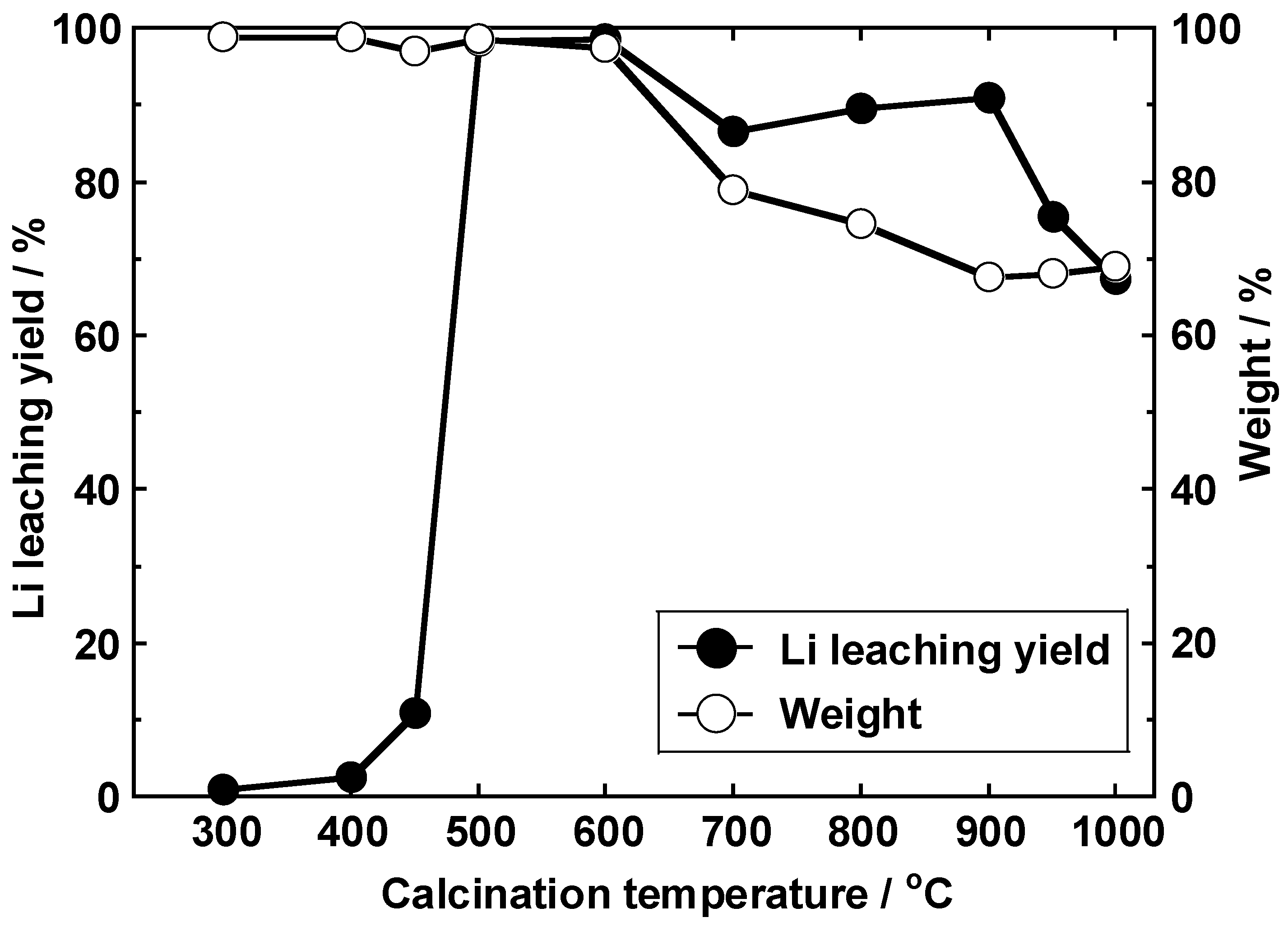
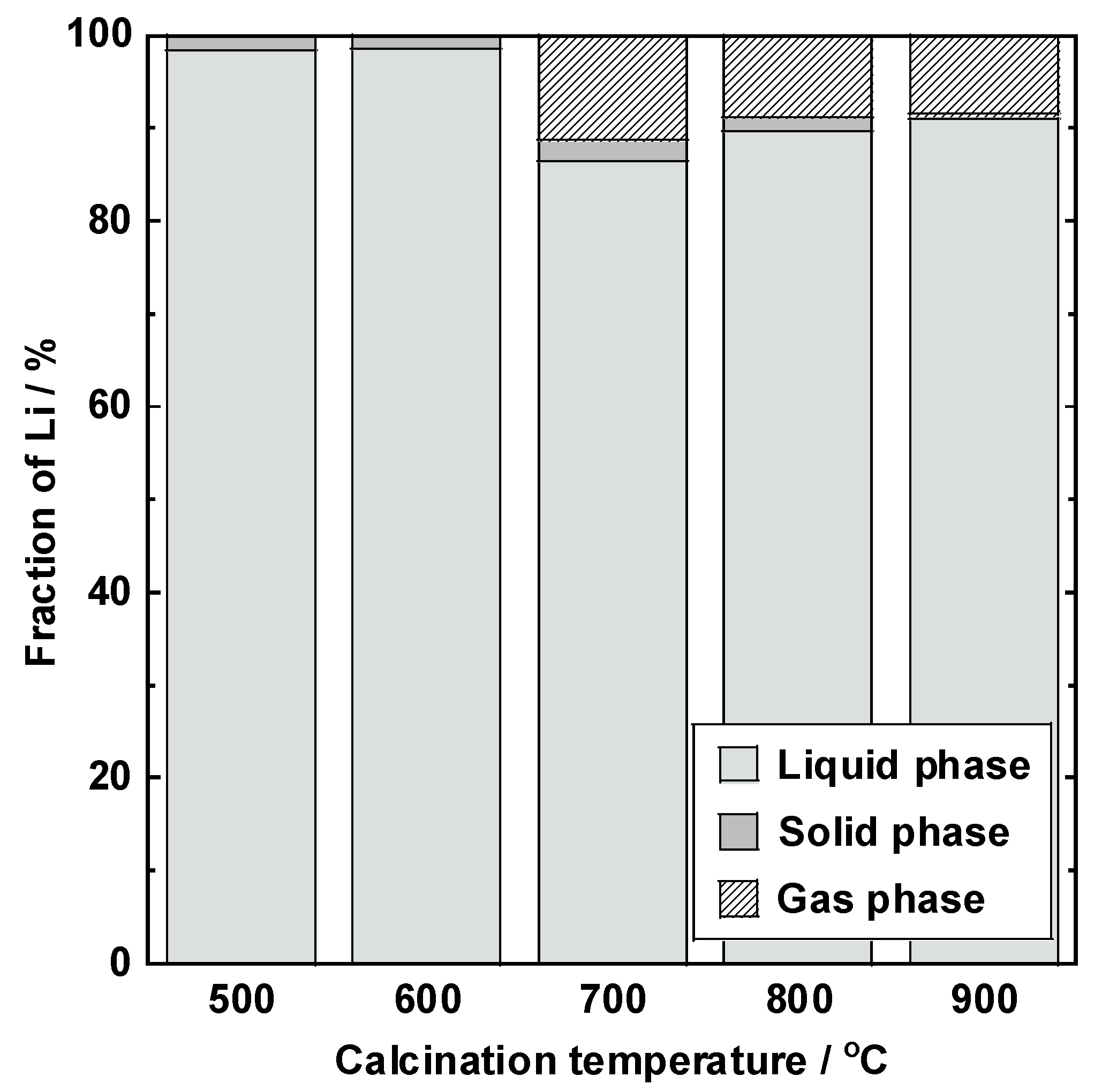
3.2. Lithium Leaching Yield
3.3. Lithium Balance
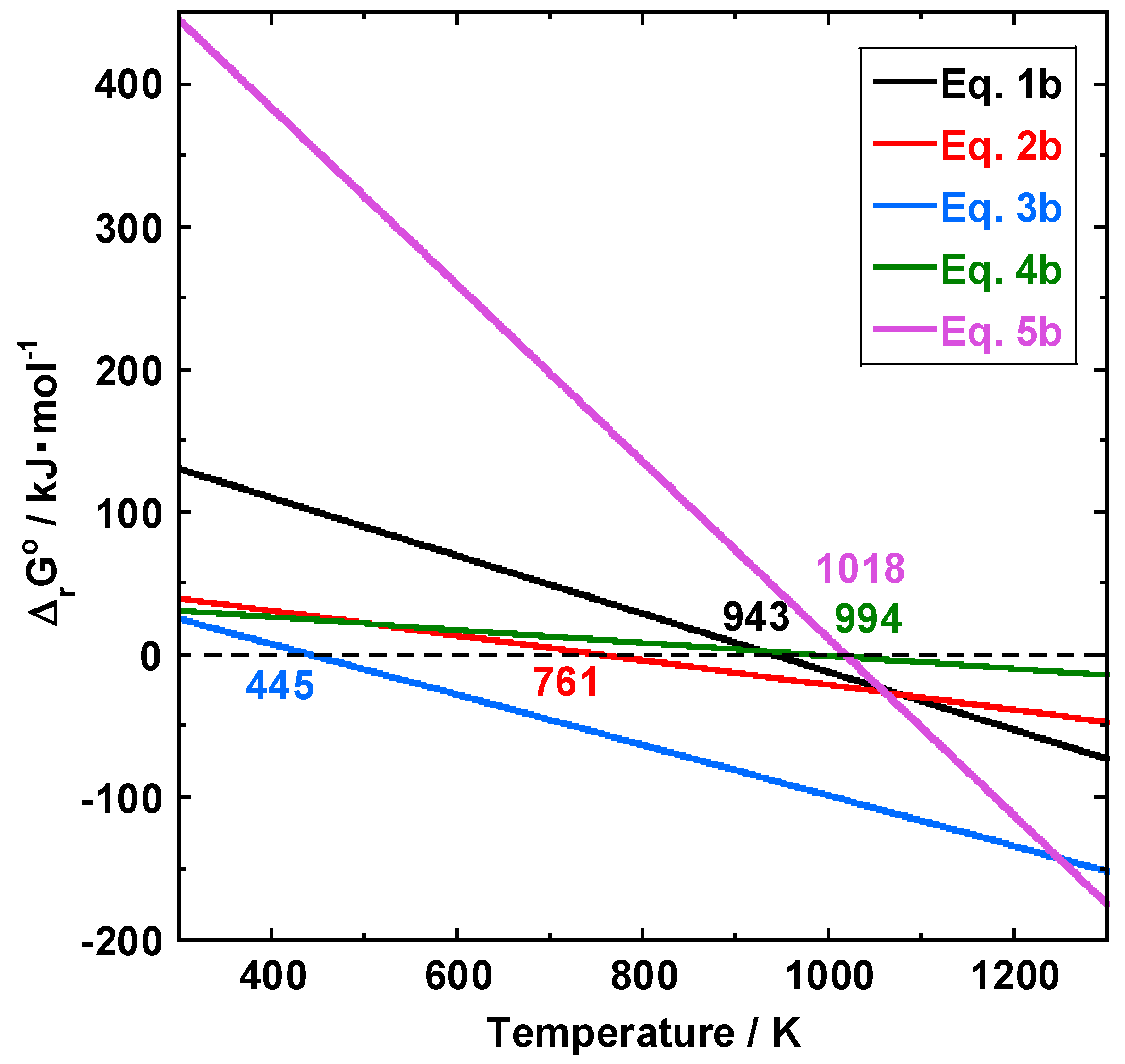
3.4. Estimated Reaction Mechanisms
3.4.1. At 520 to 550 °C
3.4.2. At 650 to 800 °C
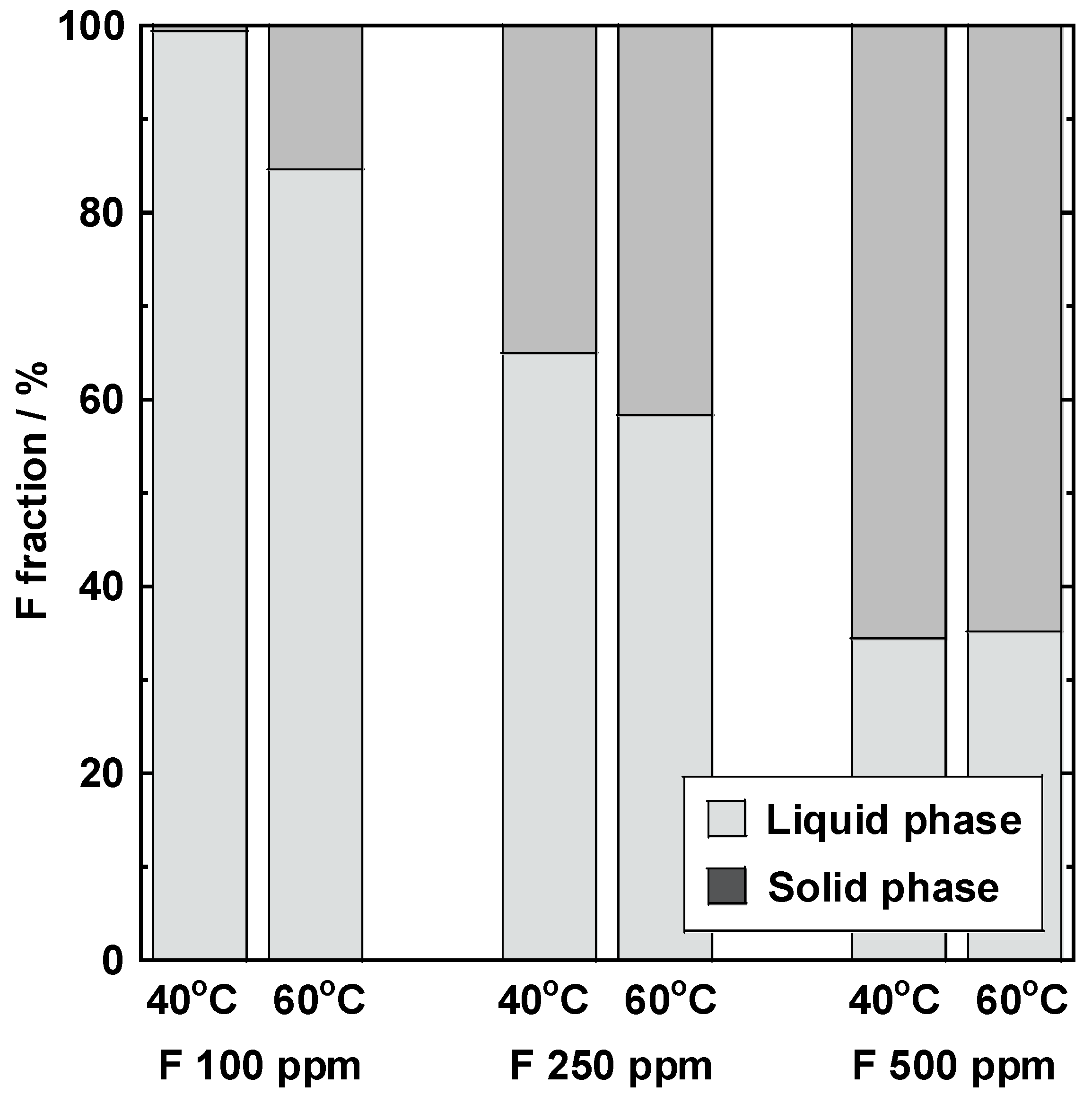
3.5. Lithium Carbonate Recovery
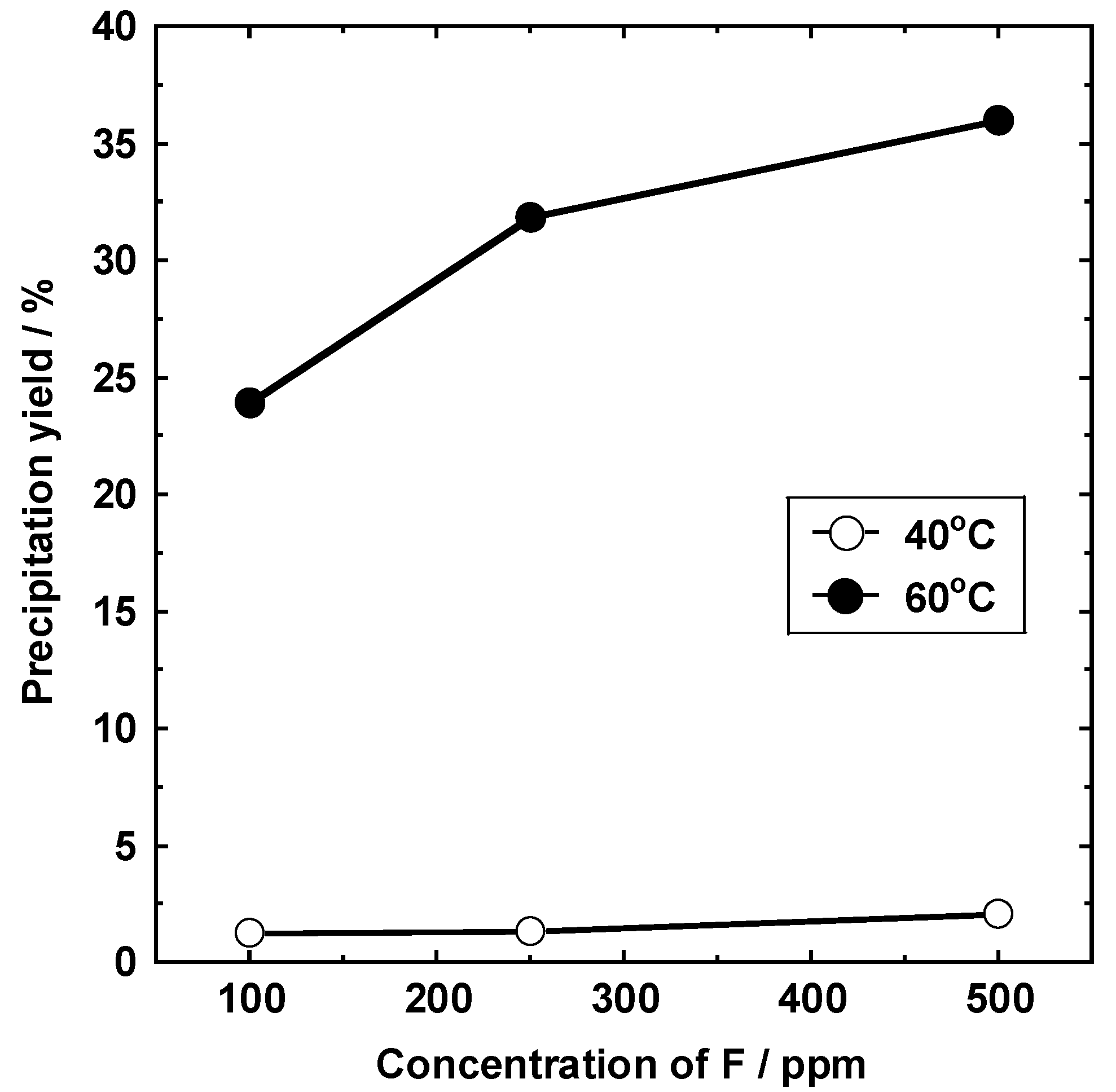
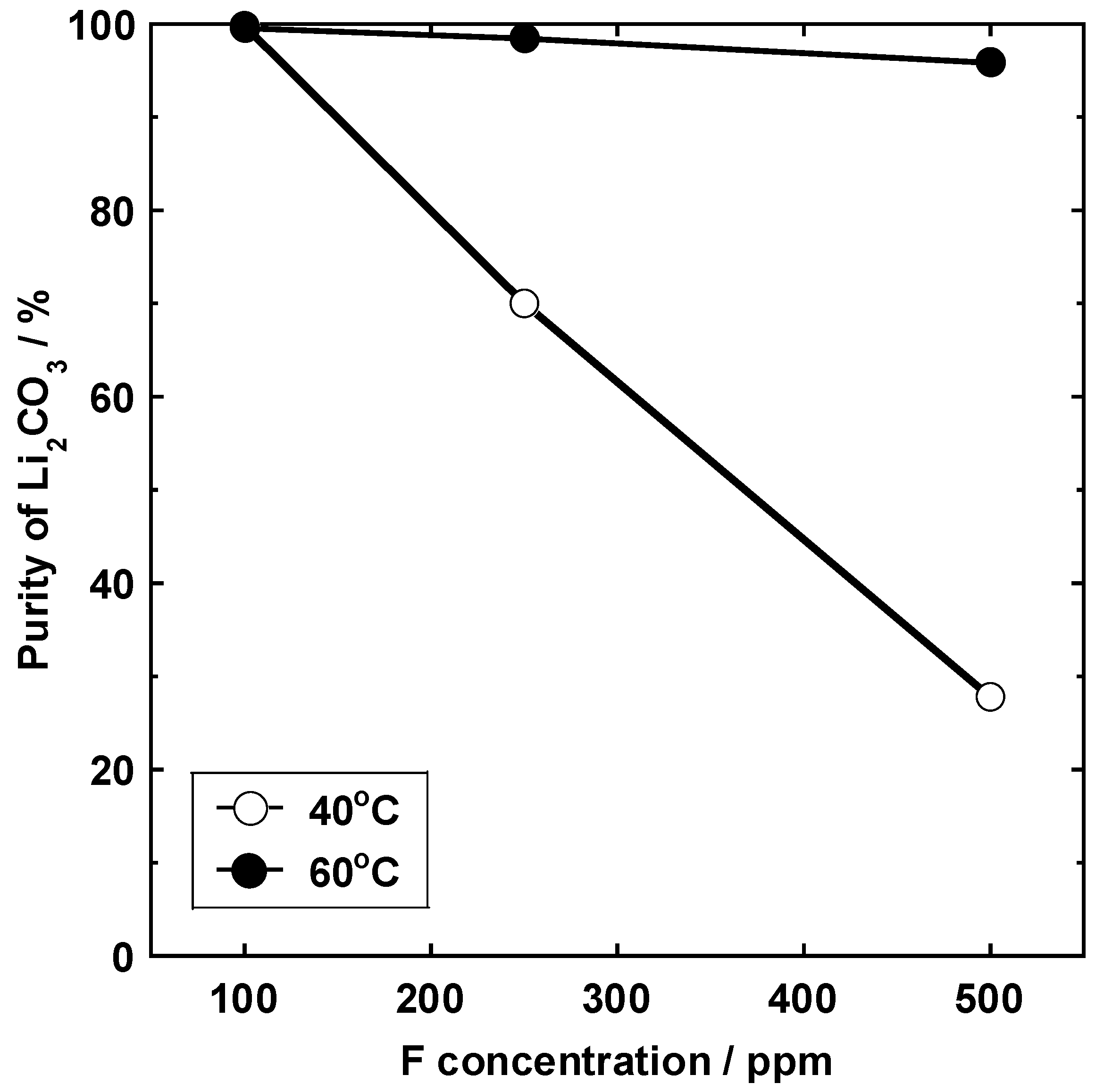
3.6. Relationship between the Concentration of the Fluorine Solution and the Lithium Carbonate Concentration
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Primary Battery Sales Statistics by Volume. Available online: http://www.baj.or.jp/statistics/14.html (accessed on 2 November 2019). (In Japanese).
- Li, J.; Wang, G.; Xu, Z. Environmentally friendly oxygen-free roasting/wet magnetic separation technology for in situ recycling cobalt, lithium carbonate and graphite from spent LiCoO2/graphite lithium batteries. J. Hazard. Mater. 2016, 302, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Sit, K.; Li, P.K.C.; Ip, C.W.; Li, C.W.; Wan, L.; Lam, Y.F.; Lai, P.Y.; Fan, J.; Magnuson, D. Studies of the energy and power of current commercial prismatic and cylindrical Li-ion cells. J. Power Sources 2004, 125, 124–134. [Google Scholar] [CrossRef]
- Natarajan, S.; Boricha, A.B.; Bajaj, H.C. Recovery of value-added products from cathode and anode material of spent lithium-ion batteries. Waste Manag. 2018, 77, 455–465. [Google Scholar] [CrossRef] [PubMed]
- Swain, B. Recovery and recycling of lithium: A review. Sep. Purif. Technol. 2017, 172, 388–403. [Google Scholar] [CrossRef]
- Heelan, J.; Gratz, E.; Zheng, Z.; Wang, Q.; Chen, M.; Apelian, D.; Wang, Y. Current and prospective Li-ion battery recycling and recovery processes. JOM 2016, 68, 2632–2638. [Google Scholar] [CrossRef]
- Bertuol, D.A.; Machado, C.M.; Silva, M.L.; Calgaro, C.O.; Dotto, G.L.; Tanabe, E.H. Recovery of cobalt from spent lithium-ion batteries using supercritical carbon dioxide extraction. Waste Manag. 2016, 51, 245–251. [Google Scholar] [CrossRef]
- Biswal, B.K.; Jadhav, U.U.; Madhaiyan, M.; Ji, L.; Yang, E.-H.; Cao, B. Biological leaching and chemical precipitation methods for recovery of Co and Li from spent lithium-ion batteries. ACS Sustain. Chem. Eng. 2018, 6, 12343–12352. [Google Scholar] [CrossRef]
- da Cunha, J.M.; Klein, L.; Bassaco, M.M.; Tanabe, E.H.; Bertuol, D.A.; Dotto, G.L. Cobalt recovery from leached solutions of lithium-ion batteries using waste materials as adsorbents. Can. J. Chem. Eng. 2015, 93, 2198–2204. [Google Scholar] [CrossRef]
- Seo, Y.; Morimoto, S. Domestic cobalt flow and mid-term demand forecast. J. MMIJ 2016, 132, 66–70. (In Japanese) [Google Scholar] [CrossRef]
- Nansai, K.; Nakajima, K.; Kagawa, S.; Kondo, Y.; Shigetomi, Y.; Suh, S. Global mining risk footprint of critical metals necessary for low-carbon technologies: The case of neodymium, cobalt, and platinum in Japan. Environ. Sci. Technol. 2015, 49, 2022–2031. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, C.; He, W.; Li, G.; Huang, J. A review on management of spent lithium ion batteries and strategy for resource recycling of all components from them. Waste Manag. Res. 2018, 36, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gaustad, G.; Babbitt, C.W. Targeting high value metals in lithium-ion battery recycling via shredding and size-based separation. Waste Manag. 2016, 51, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Jha, M.K.; Kumari, A.; Jha, A.K.; Kumar, V.; Hait, J.; Pandey, B.D. Recovery of lithium and cobalt from waste lithium ion batteries of mobile phone. Waste Manag. 2013, 33, 1890–1897. [Google Scholar] [CrossRef] [PubMed]
- Dorella, G.; Mansur, M.B. A study of the separation of cobalt from spent Li-ion battery residues. J. Power Sources 2007, 170, 210–215. [Google Scholar] [CrossRef]
- Meng, K.; Cao, Y.; Zhang, B.; Ou, X.; Li, D.-M.; Zhang, J.-F.; Ji, X. Comparison of the ammoniacal leaching behavior of layered LiNixCoyMn1-x-yO2 (x = 1/3, 0.5, 0.8) cathode materials. ACS Sustain. Chem. Eng. 2019, 7, 7750–7759. [Google Scholar] [CrossRef]
- Meshram, P.; Pandey, B.D.; Mankhand, T.R. Hydrometallurgical processing of spent lithium ion batteries (LIBs) in the presence of a reducing agent with emphasis on kinetics of leaching. Chem. Eng. J. 2015, 281, 418–427. [Google Scholar] [CrossRef]
- Guo, Y.; Li, F.; Zhu, H.; Li, G.; Huang, J.; He, W. Leaching lithium from the anode electrode materials of spent lithium-ion batteries by hydrochloric acid (HCl). Waste Manag. 2016, 51, 227–233. [Google Scholar] [CrossRef]
- Song, D.; Wang, X.; Zhou, E.; Hou, P.; Guo, F.; Zhang, L. Recovery and heat treatment of the Li(Ni1/3Co1/3Mn1/3)O2 cathode scrap material for lithium ion battery. J. Power Sources 2013, 232, 348–352. [Google Scholar] [CrossRef]
- Song, D.; Wang, X.; Nie, H.; Shi, H.; Wang, D.; Guo, F.; Shi, X.; Zhang, L. Heat treatment of LiCoO2 recovered from cathode scraps with solvent method. J. Power Sources 2014, 249, 137–141. [Google Scholar] [CrossRef]
- Xiao, J.; Li, J.; Xu, Z. Novel approach for in situ recovery of lithium carbonate from spent lithium ion batteries using vacuum metallurgy. Environ. Sci. Technol. 2017, 51, 11960–11966. [Google Scholar] [CrossRef]
- Myoung, J.; Jung, Y.; Lee, J.; Tak, Y. Cobalt oxide preparation from waste LiCoO2 by electrochemical–hydrothermal method. J. Power Sources 2002, 112, 639–642. [Google Scholar] [CrossRef]
- Iizuka, A.; Yamashita, Y.; Nagasawa, H.; Yamasaki, A.; Yanagisawa, Y. Separation of lithium and cobalt from waste lithium-ion batteries via bipolar membrane electrodialysis coupled with chelation. Sep. Purif. Technol. 2013, 113, 33–41. [Google Scholar] [CrossRef]
- Kim, S.; Joo, H.; Moon, T.; Kim, S.-H.; Yoon, J. Rapid and selective lithium recovery from desalination brine using an electrochemical system. Environ. Sci. Process. Impacts 2019, 21, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Naseri, T.; Seyyed, N.B.-H.; Mousavi, M. Bacterial leaching as a green approach for typical metals recovery from end-of-life coin cells batteries. J. Clean. Prod. 2019, 220, 483–492. [Google Scholar] [CrossRef]
- Xin, B.; Zhang, D.; Zhang, X.; Xia, Y.; Wu, F.; Chen, S.; Li, L. Bioleaching mechanism of Co and Li from spent lithium-ion battery by the mixed culture of acidophilic sulfur-oxidizing and iron-oxidizing bacteria. Biores. Technol. 2009, 100, 6163–6169. [Google Scholar] [CrossRef]
- Zeng, G.; Deng, X.; Luo, S.; Luo, X.; Zou, J. A copper-catalyzed bioleaching process for enhancement of cobalt dissolution from spent lithium-ion batteries. J. Hazard. Mater. 2012, 199–200, 164–169. [Google Scholar] [CrossRef]
- Zhang, T.; He, Y.; Wang, F.; Ge, L.; Zhu, X.; Li, H. Chemical and process mineralogical characterizations of spent lithium-ion batteries: An approach by multi-analytical techniques. Waste Manag. 2014, 34, 1051–1058. [Google Scholar] [CrossRef]
- Wang, M.-M.; Zhang, C.-C.; Zhang, F.-S. Recycling of spent lithium-ion battery with polyvinyl chloride by mechanochemical process. Waste Manag. 2017, 67, 232–239. [Google Scholar] [CrossRef]
- Larsson, F.; Andersson, P.; Blomqvist, P.; Mellander, B.-E. Toxic fluoride gas emissions from lithium-ion battery fires. Sci. Rep. 2017, 7, 10018. [Google Scholar] [CrossRef]
- Mineral Resources Material Flow 2018. Available online: http://mric.jogmec.go.jp/wp-content/uploads/2019/03/material_flow2018_Li.pdf (accessed on 2 November 2019). (In Japanese)
- Martin, G.; Rentsch, L.; Höck, M.; Bertau, M. Lithium market research—global supply, future demand and price development. Energy Storage Mater. 2017, 6, 171–179. [Google Scholar] [CrossRef]
- Lithium Statistics Information. Available online: https://www.usgs.gov/centers/nmic/lithium-statistics-and-information (accessed on 2 November 2019).
- Liu, P.; Xiao, L.; Tang, Y.; Chen, Y.; Ye, L.; Zhu, Y. Study on the reduction roasting of spent LiNixCoyMnzO2 lithium-ion battery cathode materials. J. Therm. Anal. Calorim. 2019, 136, 1323–1332. [Google Scholar] [CrossRef]
- Wang, M.; Tan, Q.; Liu, L.; Li, J. Efficient separation of aluminum foil and cathode materials from spent lithium-ion batteries using a low-temperature molten salt. ACS Sustain. Chem. Eng. 2019, 7, 8287–8294. [Google Scholar] [CrossRef]
- Zhang, G.; Du, Z.; He, Y.; Wang, H.; Xie, W.; Zhang, T. A sustainable process for the recovery of anode and cathode materials derived from spent lithium-ion batteries. Sustainability 2019, 11, 2363. [Google Scholar] [CrossRef]
- Gachot, G.; Ribiére, P.; Mathiron, D.; Grugeon, S.; Armand, M.; Leriche, J.-B.; Pilard, S.; Laruelle, S. Gas chromatography/mass spectrometry as a suitable tool for the Li-ion battery electrolyte degradation mechanisms study. Anal. Chem. 2011, 83, 478–485. [Google Scholar] [CrossRef]
- Zulfiqar, S.; Zulfiqar, M.; Rizvi, M.; Munir, A.; McNeill, I.C. Study of the thermal degradation of polychlorotrifluoroethylene, poly(vinylidenefluoride) and copolymers of chlorotrifluoroethylene and vinylidene fluoride. Polym. Degrad. Stab. 1994, 43, 423–430. [Google Scholar] [CrossRef]
- Inoue, R.; Ueda, S.; Wakuta, K.; Sasaki, K.; Ariyama, T. Thermodynamic consideration on the absorption properties of carbon dioxide to basic oxide. ISIJ Int. 2010, 50, 1532–1538. [Google Scholar] [CrossRef]
- CRC Handbook of Chemistry and Physics, 100th ed.; CRC Press: Boca Raton, FL, USA, 2019; pp. 5–181.

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuzuhara, S.; Ota, M.; Tsugita, F.; Kasuya, R. Recovering Lithium from the Cathode Active Material in Lithium-Ion Batteries via Thermal Decomposition. Metals 2020, 10, 433. https://doi.org/10.3390/met10040433
Kuzuhara S, Ota M, Tsugita F, Kasuya R. Recovering Lithium from the Cathode Active Material in Lithium-Ion Batteries via Thermal Decomposition. Metals. 2020; 10(4):433. https://doi.org/10.3390/met10040433
Chicago/Turabian StyleKuzuhara, Shunsuke, Mina Ota, Fuka Tsugita, and Ryo Kasuya. 2020. "Recovering Lithium from the Cathode Active Material in Lithium-Ion Batteries via Thermal Decomposition" Metals 10, no. 4: 433. https://doi.org/10.3390/met10040433
APA StyleKuzuhara, S., Ota, M., Tsugita, F., & Kasuya, R. (2020). Recovering Lithium from the Cathode Active Material in Lithium-Ion Batteries via Thermal Decomposition. Metals, 10(4), 433. https://doi.org/10.3390/met10040433



